This site displays a prototype of a “Web 2.0” version of the daily Federal Register. It is not an official legal edition of the Federal Register, and does not replace the official print version or the official electronic version on GPO’s govinfo.gov.
The documents posted on this site are XML renditions of published Federal Register documents. Each document posted on the site includes a link to the corresponding official PDF file on govinfo.gov. This prototype edition of the daily Federal Register on FederalRegister.gov will remain an unofficial informational resource until the Administrative Committee of the Federal Register (ACFR) issues a regulation granting it official legal status. For complete information about, and access to, our official publications and services, go to About the Federal Register on NARA's archives.gov.
The OFR/GPO partnership is committed to presenting accurate and reliable regulatory information on FederalRegister.gov with the objective of establishing the XML-based Federal Register as an ACFR-sanctioned publication in the future. While every effort has been made to ensure that the material on FederalRegister.gov is accurately displayed, consistent with the official SGML-based PDF version on govinfo.gov, those relying on it for legal research should verify their results against an official edition of the Federal Register. Until the ACFR grants it official status, the XML rendition of the daily Federal Register on FederalRegister.gov does not provide legal notice to the public or judicial notice to the courts.
AGENCY:
Centers for Medicare & Medicaid Services (CMS), Department of Health and Human Services (HHS).
ACTION:
Final rule with comment period.
SUMMARY:
This final rule with comment period revises the Medicare Hospital Outpatient Prospective Payment System (OPPS) and the Medicare Ambulatory Surgical Center (ASC) payment system for calendar year 2026 based on our continuing experience with these systems. We also describe the changes to the amounts and factors used to determine the payment rates for Medicare services paid under the OPPS and those paid under the ASC payment systems. In addition, this final rule with comment period announces the closure of a teaching hospital and the opportunity to apply for available slots, and updates and refines the requirements for the Hospital Outpatient Quality Reporting Program, Rural Emergency Hospital Quality Reporting Program, Ambulatory Surgical Center Quality Reporting Program, Overall Hospital Quality Star Rating, and hospitals to make public their standard charge information and enforcement of hospital price transparency, as well as summarizes comments received in response to a request for information on measure concepts regarding Well-Being and Nutrition for consideration in future years for the OQR, REHQR, and ASCQR programs.
DATES:
Effective Date: The provisions of this rule are effective January 1, 2026.
Comment period: To be assured consideration, comments must be received at one of the addresses provided below, by January 20, 2026.
Deadline for hospitals to submit applications for Available Resident Slots: Application submissions for Round 26 are due no later than February 19, 2026 (see section XXII.C. of this final rule with comment period for further details on the application process).
ADDRESSES:
In commenting, please refer to file code CMS-1834-FC.
Comments, including mass comment submissions, must be submitted in one of the following three ways (please choose only one of the ways listed):
1. Electronically. You may submit electronic comments on this regulation to http://www.regulations.gov. Follow the “Submit a comment” instructions.
2. By regular mail. You may mail written comments to the following address ONLY: Centers for Medicare & Medicaid Services, Department of Health and Human Services, Attention: CMS-1834-FC, P.O. Box 8010, Baltimore, MD 21244-8010.
Please allow sufficient time for mailed comments to be received before the close of the comment period.
3. By express or overnight mail. You may send written comments to the following address ONLY: Centers for Medicare & Medicaid Services, Department of Health and Human Services, Attention: CMS-1834-FC, Mail Stop C4-26-05, 7500 Security Boulevard, Baltimore, MD 21244-1850.
For information on viewing public comments, see the beginning of the SUPPLEMENTARY INFORMATION section.
FOR FURTHER INFORMATION CONTACT:
Regulation coordination questions, contact Gina Aughenbaugh via email at 410-786-7756 or OutpatientPPS@cms.hhs.gov.
Add-on Payment for Radiopharmaceutical Technetium-99m (Tc-99m) Derived from Domestically Produced Molybdenum-99, contact Au'Sha Washington via email at ausha.washington@cms.hhs.gov or Leone Kisler at leone.kisler@cms.hhs.gov.
Adjusting Payment under the OPPS for Services Predominantly Performed in the ASC or Physician Office Settings Request for Information, contact Elise Barringer via email at Elise.Barringer@cms.hhs.gov.
Advisory Panel on Hospital Outpatient Payment (HOP Panel), contact the HOP Panel mailbox at APCPanel@cms.hhs.gov.
Ambulatory Surgical Center Covered Procedures List (ASC CPL), contact Abigail Cesnik via email at Abigail.Cesnik@cms.hhs.gov.
Ambulatory Surgical Center Quality Reporting (ASCQR) Program measures, contact Marsha Hertzberg via email at Marsha.Hertzberg@cms.hhs.gov.
Ambulatory Surgical Center Quality Reporting (ASCQR) Program policies, contact Anita Bhatia via email at Anita.Bhatia@cms.hhs.gov.
All-Inclusive Rate (AIR) Add-On Payment for High-Cost Drugs Provided by Indian Health Service (IHS) and Tribal Facilities, contact Nate Vercauteren via email at Nathan.Vercauteren@cms.hhs.gov.
Blood and Blood Products, contact Gil Ngan via email at Gil.Ngan@cms.hhs.gov.
Cancer Hospital Payments, contact Scott Talaga via email at Scott.Talaga@cms.hhs.gov.
CMS Web Posting of the OPPS and ASC Payment Files, contact Gil Ngan via email at Gil.Ngan@cms.hhs.gov.
Composite APCs (Multiple Imaging and Mental Health) and Comprehensive APCs (C-APCs), contact Elise Barringer via email at Elise.Barringer@cms.hhs.gov.
Device-Intensive Status and No Cost/Full Credit and Partial Credit Devices, contact Scott Talaga via email at Scott.Talaga@cms.hhs.gov.
Graduate Medical Education (GME) Accreditation, contact DAC@cms.hhs.gov.
Hospital Outpatient Quality Reporting (OQR) Program policies, contact Kimberly Go via email at Kimberly.Go@cms.hhs.gov.
Hospital Outpatient Quality Reporting (OQR) Program measures, contact Kristina Rabarison via email at Kristina.Rabarison@cms.hhs.gov.
Hospital Outpatient Visits (Emergency Department Visits and Critical Care Visits), contact Elise Barringer via email at Elise.Barringer@cms.hhs.gov.
Hospital Price Transparency, contact Sarah Wheat via email at PriceTransparencyHospitalCharges@cms.hhs.gov.
Inpatient Only (IPO) Procedures List, contact Abigail Cesnik via email at Abigail.Cesnik@cms.hhs.gov.
Market-Based Data Collection and Market-Based MS-DRG Relative Weight Methodology Issues, contact DAC@cms.hhs.gov.
Medical Review of Certain Inpatient Hospital Admissions under Medicare Part A for CY 2026 and Subsequent Years (2-Midnight Rule), contact Nate Vercauteren via email at Nathan.Vercauteren@cms.hhs.gov.
Medicare OPPS Drug Acquisition Cost Survey, contact Cory Duke via email at Cory.Duke@cms.hhs.gov or Gil Ngan at Gil.Ngan@cms.hhs.gov or Nate Vercauteren at Nathan.Vercauteren@cms.hhs.gov.
Method to Control Unnecessary Increases in the Volume of Outpatient Services, contact Elise Barringer via email at Elise.Barringer@cms.hhs.gov.
New Technology Intraocular Lenses (NTIOLs), contact Scott Talaga via email at Scott.Talaga@cms.hhs.gov.
Non-Opioid Policy or Implementation of Section 4135 of the Consolidated Appropriations Act (CAA), 2023, contact Cory Duke via email at Cory.Duke@cms.hhs.gov or Nicole Marcos via email at Nicole.Marcos@cms.hhs.gov.
OPPS Brachytherapy, contact Cory Duke via email at Cory.Duke@cms.hhs.gov and Scott Talaga via email at Scott.Talaga@cms.hhs.gov.
OPPS Data (APC Weights, Conversion Factor, Copayments, Cost-to-Charge Ratios (CCRs), Data Claims, Geometric Mean Calculation, Outlier Payments, and Wage Index), contact Erick Chuang via email at Erick.Chuang@cms.hhs.gov or Scott Talaga via email at Scott.Talaga@cms.hhs.gov. ( printed page 53449)
OPPS Drugs, Radiopharmaceuticals, Biologicals, and Biosimilar Products, contact Gil Ngan via email at Gil.Ngan@cms.hhs.gov, Cory Duke via email at Cory.Duke@cms.hhs.gov, or Nate Vercauteren via email at Nathan.Vercauteren@cms.hhs.gov.
OPPS New Technology Procedures/Services, contact the New Technology APC mailbox at NewTechAPCapplications@cms.hhs.gov.
OPPS Packaged Items/Services, contact Cory Duke via email at Cory.Duke@cms.hhs.gov.
OPPS Pass-Through Devices, contact the Device Pass-Through mailbox at DevicePTapplications@cms.hhs.gov.
OPPS Status Indicators (SI) and Comment Indicators (CI), contact Marina Kushnirova via email at Marina.Kushnirova@cms.hhs.gov or Tonya Gierke at Tonya.Gierke@cms.hhs.gov.
Overall Hospital Quality Star Rating policies, contact Tyson Nakashima Sr. via email Tyson.Nakashima@cms.hhs.gov.
Partial Hospitalization Program (PHP), Intensive Outpatient (IOP), and Community Mental Health Center (CMHC) Issues, contact the PHP Payment Policy Mailbox at PHPPaymentPolicy@cms.hhs.gov.
Remote Services, contact Elise Barringer via email at Elise.Barringer@cms.hhs.gov or Nate Vercauteren via email at Nathan.Vercauteren@cms.hhs.gov.
Rural Emergency Hospital Quality Reporting (REHQR) Program policies, contact Anita Bhatia via email at Anita.Bhatia@cms.hhs.gov.
Rural Emergency Hospital Quality Reporting (REHQR) Program measures, contact Melissa Hager via email at Melissa.Hager@cms.hhs.gov.
Skin Substitute Products, contact Susan Janeczko via email at Susan.Janeczko@cms.hhs.gov, Cory Duke via email at Cory.Duke@cms.hhs.gov, or Nicole Marcos via email at Nicole.Marcos@cms.hhs.gov.
Software as a Service, contact Nicole Marcos via email at Nicole.Marcos@cms.hhs.gov.
Virtual Direct Supervision of Outpatient Therapeutic and Diagnostic Services in Hospitals and CAHs, contact Nate Vercauteren via email at Nathan.Vercauteren@cms.hhs.gov.
All Other Issues Related to Hospital Outpatient Payments Not Previously Identified, contact the OPPS mailbox at OutpatientPPS@cms.hhs.gov.
All Other Issues Related to the Ambulatory Surgical Center Payments Not Previously Identified, contact the ASC mailbox at ASCPPS@cms.hhs.gov.
SUPPLEMENTARY INFORMATION:
Inspection of Public Comments: All comments received before the close of the comment period are available for viewing by the public, including any personally identifiable or confidential business information that is included in a comment. We post all comments received before the close of the comment period on the following website as soon as possible after they have been received: https://www.regulations.gov. Follow the search instructions on that website to view public comments. CMS will not post on Regulations.gov public comments that make threats to individuals or institutions or suggest that the individual will take actions to harm the individual. CMS continues to encourage individuals not to submit duplicative comments. We will post acceptable comments from multiple unique commenters even if the content is identical or nearly identical to other comments.
Addenda Available Only Through the Internet on the CMS Website
In the past, a majority of the addenda referred to in our OPPS/ASC proposed and final rules were published in the Federal Register as part of the annual rulemakings. However, beginning with the calendar year (CY) 2012 OPPS/ASC proposed rule, the addenda no longer appear in the Federal Register as part of the annual OPPS/ASC proposed and final rules to decrease administrative burden and reduce costs associated with publishing lengthy tables. Instead, these addenda are published and available only on the CMS website. The addenda relating to the OPPS are available at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices. The addenda relating to the ASC payment system are available at https://www.cms.gov/medicare/payment/prospective-payment-systems/ambulatory-surgical-center-asc/asc-regulations-and-notices.
Current Procedural Terminology (CPT) Copyright Notice
Throughout this final rule with comment period, we use CPT codes and descriptions to refer to a variety of services. We note that CPT codes and descriptions are copyright 2025 American Medical Association (AMA). All Rights Reserved. CPT is a registered trademark of the AMA. Applicable Federal Acquisition Regulations and Defense Federal Acquisition Regulations apply.
I. Summary and Background
A. Executive Summary of This Document
1. Purpose
In this final rule with comment period, we are updating the payment policies and payment rates for services furnished to Medicare beneficiaries in hospital outpatient departments (HOPDs) and ambulatory surgical centers (ASCs), beginning January 1, 2026. Section 1833(t) of the Social Security Act (the Act) requires us to annually review and update the payment rates for services payable under the Hospital Outpatient Prospective Payment System (OPPS). Specifically, section 1833(t)(9)(A) of the Act requires the Secretary of the Department of Health and Human Services (the Secretary) to review certain components of the OPPS not less often than annually, and to revise the groups, the relative payment weights, and the wage and other adjustments to take into account changes in medical practice, changes in technology, and the addition of new services, new cost data, and other relevant information and factors. In addition, under section 1833(i)(D)(v) of the Act, we annually review and update the ASC payment rates. This final rule with comment period also includes additional policy changes made in accordance with our experience with the OPPS and the ASC payment system and recent changes in our statutory authority. We describe these and various other statutory authorities in the relevant sections of this final rule with comment period. In addition, this final rule with comment period announces the closure of a teaching hospital and the opportunity to apply for available slots, and updates the requirements for the Hospital Outpatient Quality Reporting (OQR) Program, the Rural Emergency Hospital Quality Reporting (REHQR) Program, the Ambulatory Surgical Center Quality Reporting (ASCQR) Program, and Overall Hospital Quality Star Rating. Finally, we are updating and refining the requirements for hospitals to make public their standard charges and CMS enforcement of hospital price transparency (HPT) regulations.
2. Summary of the Major Provisions
- OPPS Update: For CY 2026, we are increasing the payment rates under the OPPS by an outpatient department (OPD) fee schedule increase factor of 2.6 percent. This increase factor is based on the final inpatient hospital market basket percentage increase of 3.3 percent for inpatient services paid under the hospital inpatient prospective payment system (IPPS), reduced by a final productivity adjustment of 0.7 percentage point. Based on this update, we estimate that total payments to OPPS providers (including beneficiary cost sharing and estimated changes in enrollment, utilization, and case mix) for calendar year (CY) 2026 will be approximately $101.0 billion, an increase of approximately $8.0 billion compared to estimated CY 2025 OPPS payments. ( printed page 53450)
We are continuing to implement the statutory 2.0 percentage point reduction in payments for hospitals that fail to meet the hospital outpatient quality reporting requirements by applying a reporting factor of 0.9805 to the OPPS payments and copayments for all applicable services. We note that under the final 340B remedy offset, payments for services at hospitals subject to the 340B remedy offset will be reduced by 0.5 percentage points.
- ASC Payment Update: For CYs 2019 through 2023, we adopted a policy to update the ASC payment system using the hospital market basket update. In light of the impact of the COVID-19 public health emergency (PHE) on healthcare utilization, we extended our policy to update the ASC payment system using the hospital market basket update an additional 2 years—through CYs 2024 and 2025. In this final rule with comment period, we are extending our utilization of the hospital market basket update as the update factor for the ASC payment system for 1 additional year (through CY 2026). Using the hospital market basket update, for CY 2026, we are increasing payment rates under the ASC payment system by 2.6 percent for ASCs that meet the quality reporting requirements under the ASCQR Program. This increase is based on a final hospital market basket percentage increase of 3.3 percent reduced by a final productivity adjustment of 0.7 percentage point. Based on this final update, we estimate that total payments to ASCs (including beneficiary cost sharing and estimated changes in enrollment, utilization, and case-mix) for CY 2026 will be approximately $9.2 billion, an increase of approximately $450 million compared to estimated CY 2025 Medicare payments.
- Device Pass-Through Payment Applications: For CY 2026, we received eight complete applications for device pass-through payments. We sought public comment on seven applications and make final determinations on these applications in this final rule with comment period.
- Changes to the List of ASC Covered Surgical Procedures and Ancillary Services Lists: For CY 2026, we are expanding the ASC covered procedures list (CPL) by revising the criteria under § 416.166 to modify the general standard criteria and to eliminate five of the general exclusion criteria, moving them into a new section as nonbinding physician considerations for patient safety. We also are adding 276 procedures to the ASC CPL based on these criteria changes and adding an additional 271 codes to the ASC CPL that we are finalizing for removal from the IPO list for CY 2026.
- Changes to the Inpatient Only (IPO) List: For CY 2026, we are phasing out the IPO list over 3 years, beginning with the removal of 285 mostly musculoskeletal services for CY 2026.
- Add-on Payment for Radiopharmaceutical Technetium-99m (Tc-99m) Derived from Domestically Produced Molybdenum-99 (Mo-99): In the CY 2025 OPPS/ASC final rule with comment period, we finalized that for CY 2026 the add-on payment for radiopharmaceuticals produced without the use of Tc-99m derived from non-Highly Enriched Uranium sources would be replaced with an add-on payment for radiopharmaceuticals that use Tc-99m derived from domestically produced Mo-99. For CY 2026, we are finalizing a $10 per dose amount for this add-on payment, and that at least 50 percent of the Mo-99 used in the Tc-99m generator that produces a dose of Tc-99m must be domestically produced for the dose to qualify for the add-on payment. We are also codifying our definition for domestically produced Mo-99, and to establish new HCPCS C-code C9176 (Tc-99m from domestically produced non-HEU Mo-99, [minimum 50 percent], full cost recovery add-on, per study dose).
- Cross-Program Updates for the Hospital Outpatient Quality Reporting (OQR), Rural Emergency Hospital Quality Reporting (REHQR), and Ambulatory Surgical Center Quality Reporting (ASCQR) Programs: We are finalizing the removal of: (1) the COVID-19 Vaccination Coverage Among Healthcare Personnel (HCP) measure from the Hospital OQR and ASCQR Program measure sets beginning with the CY 2024 reporting period/CY 2026 payment determination; (2) the Hospital Commitment to Health Equity (HCHE) measure from the Hospital OQR and REHQR Program measure sets, and the Facility Commitment to Health Equity (FCHE) measure from the ASCQR Program measure set beginning with the CY 2025 reporting period/CY 2027 payment or program determination; and (3) the Screening for Social Drivers of Health (SDOH) measure and the Screen Positive Rate for SDOH measure from the Hospital OQR, REHQR, and ASCQR Program measure sets beginning with the CY 2025 reporting period. Additionally, we received comments regarding measure concepts related to well-being and nutrition for future consideration in the Hospital OQR, REHQR, and ASCQR Programs. We are finalizing our proposal to update and codify the Extraordinary Circumstance Exception (ECE) policy to clarify that CMS has the discretion to grant an extension in response to an ECE request for the Hospital OQR, REHQR, and ASCQR Programs.
- Hospital Outpatient Quality Reporting (OQR) Program: In addition to the cross-program measure and policy updates, we are finalizing: (1) adoption of the Emergency Care Access & Timeliness eCQM with 1 year of voluntary reporting for the CY 2027 reporting period followed by mandatory reporting for the CY 2028 reporting period/CY 2030 payment determination and subsequent years; (2) removal of the Median Time from Emergency Department (ED) Arrival to ED Departure for Discharged ED Patients and the Left Without Being Seen measures beginning with the CY 2028 reporting period/2030 payment determination; and (3) modification of the Excessive Radiation Dose or Inadequate Image Quality for Diagnostic Computed Tomography (CT) in Adults (Hospital Level—Outpatient) measure (Excessive Radiation eCQM) from mandatory reporting beginning with the CY 2027 reporting period to continue voluntary reporting in the CY 2027 reporting period and subsequent years.
- Rural Emergency Hospital Quality Reporting (REHQR) Program: In addition to the cross-program measure and policy updates, we are finalizing the: (1) adoption of the Emergency Care Access & Timeliness eCQM beginning with the CY 2027 reporting period/CY 2029 program determination; and (2) related eCQM data submission and reporting requirements, including that REHs will be provided the option of reporting either the Emergency Care Access and Timeliness eCQM or the Median Time from Emergency Department (ED) Arrival to ED Departure for Discharged ED Patients measure beginning with the CY 2027 reporting period/CY 2029 program determination.
- Ambulatory Surgical Center Quality Reporting (ASCQR) Program: We are not finalizing the adoption of the Patient Understanding of Key Information Related to Recovery After a Facility-Based Outpatient Procedure or Surgery, Patient Reported Outcome-Based Performance Measure (Information Transfer PRO-PM) at this time.
- Overall Hospital Quality Star Rating Modification to Emphasize the Safety of Care Measure Group: We proposed to update the methodology that will be used to calculate the Overall Hospital Quality Star Rating through implementation of a 2-stage methodologic update. We are finalizing our proposed updates to the methodology to emphasize the ( printed page 53451) importance of the Safety of Care measure group, particularly to address the issue of hospitals receiving a high Star Rating despite performing in the lowest quartile of the Safety of Care measure group. The first-stage methodology update is a narrow but focused transitional step that limits hospitals to a maximum of four out of five stars (based on at least three Safety of Care measure scores) if they performed in the lowest quartile of the Safety of Care measure group in the 2026 Overall Hospital Quality Star Rating. The second stage of the methodology update replaces the first stage update and reduces the Star Rating of any hospital in the lowest quartile of Safety of Care (based on at least three Safety of Care measure scores) by one star, to a minimum 1-star rating for the 2027 Overall Hospital Quality Star Rating and later years. These changes will prioritize safety for both patients and healthcare workers and reflect CMS' fundamental commitment to ensuring high-quality, safe care as a central component of health system performance.
- Partial Hospitalization and Intensive Outpatient Programs: We finalizing changes to our methodology used to calculate the CY 2026 Community Mental Health Center (CMHC) Partial Hospitalization Program (PHP), and Intensive Outpatient Program (IOP) costs based on 40 percent of the corresponding proposed hospital-based PHP and IOP costs. This change will resolve a cost inversion in CMHC cost data that resulted in higher geometric mean costs for 3-service days than for 4-service days. It also stabilizes rates for CMHCs by basing them on data from a much larger set of providers while preserving the adjustment for the structural differences between CMHC and hospital costs.
- Notice of Intent to Conduct a Medicare OPPS Drugs Acquisition Cost Survey: Section 1833(t)(14)(D)(ii) of the Act requires the Secretary to periodically conduct surveys of hospital acquisition costs for each specified covered outpatient drug for use in setting the payment rates for such drugs. Additionally, on April 18, 2025, President Trump signed Executive Order (E.O.) 14273, “Lowering Drug Prices by Once Again Putting Americans First”. Section 5 of the E.O., “Appropriately Accounting for Acquisition Costs of Drugs in Medicare”, which directs the Secretary of HHS to publish in the Federal Register a plan to conduct a survey under section 1833(t)(14)(D)(ii) of the Act so he can determine the hospital acquisition cost for covered outpatient drugs at hospital outpatient departments. Accordingly, we will be conducting a survey, with the survey submission window opening by early CY 2026, of the acquisition costs for each separately payable drug acquired by all hospitals paid under the OPPS. We intend for the survey to be completed in time for the survey results to be used to inform policymaking beginning with the CY 2027 OPPS/ASC proposed rule.
- Two-Midnight Rule Medical Review Activities Exemptions: For CY 2026, we are continuing our existing policy exempting procedures that are removed from the IPO list under the OPPS from certain medical review activities related to the two-midnight policy. Under this policy, procedures removed from the IPO list are exempted from site-of-service claim denials, Medicare review contractor referrals to the Recovery Audit Contractor (RAC) for persistent noncompliance with the 2-midnight rule, and RAC reviews for “patient status” (that is, site-of-service) until claims data demonstrates that the procedures are more commonly billed in the outpatient setting than the inpatient setting. We are also revising 42 CFR 412.3(d)(2) for clarity.
- Virtual Direct Supervision of Pulmonary Rehabilitation (PR), Coronary Rehabilitation (CR), Intensive Coronary Rehabilitation and Diagnostic Services. For CY 2026, we are revising § 410.27(a)(1)(iv)(B)( 1) and § 410.28(e)(2)(iii) to make the availability of the direct supervision of CR, ICR, PR services and diagnostic services via audio-video real-time communications technology (excluding audio-only) permanent, except for diagnostic services that have a global period indicator of 010 or 090.
- Prospective Adjustment to Payments for Non-Drug Items and Services to Offset the Increased Payments for Non-Drug Items and Services Made in CY 2018 Through CY 2022 as a Result of the 340B Payment Policy. For CY 2026, we are applying the previously finalized reduction to the OPPS conversion factor under § 419.32(b)(1)(iv)(B)(12) used to determine the payment amounts for non-drug items and services for hospitals for whom this adjustment applies of 0.5 percent. The Remedy for the 340B-Acquired Drug Payment Policy for Calendar Years 2018-2022 (88 FR 77150) codified a 0.5 percent reduction in the OPPS conversion factor applicable to non-drug items and services, excluding hospitals that enrolled in Medicare after January 1, 2018.
- Payment for Skin Substitute Products under the OPPS. For CY 2026, we are finalizing a policy for CMS to separately pay for the provision of certain groups of skin substitute products as supplies when they are used during a covered application procedure paid under the PFS in the non-facility setting or under the OPPS. We are finalizing a policy to group skin substitutes that are not drugs or biologicals using three FDA regulatory categories (PMAs, 510(k)s, and 361 HCT/Ps) to set payment rates. To effectuate this categorization into a payment policy under the OPPS, we are creating three new APCs for HCPCS codes that describe skin substitute products organized by clinical and resource similarity. These three APCs will divide skin substitutes by their FDA regulatory pathway. Specifically, we are creating: APC 6000 (PMA Skin Substitute Products); APC 6001 (510(k) Skin Substitute Products); and APC 6002 (361 HCT/P Skin Substitute Products). This will result in an initial payment rate of $127.14 for each of the new APCs. We are implementing this policy in both the non-facility, ambulatory surgical center setting, and outpatient hospital settings.
Method to Control Unnecessary Increases in the Volume of Outpatient Services Furnished in Excepted Off-Campus Provider-Based Departments (PBDs): For CY 2026, we are finalizing a policy to use our authority under section 1833(t)(2)(F) of the Act to apply the Physician Fee Schedule equivalent rate for any HPCPCs codes assigned to the drug administration services APCs, when provided at an off-campus PBD excepted from section 1833(t)(21) of the Act. We are finalizing a policy to exempt rural Sole Community Hospitals from this method to control the unnecessary volume of drug administration services.
Final Market-Based MS-DRG Relative Weight Data Collection and Change in Methodology for Calculating MS-DRG Relative Weights Under the Inpatient Prospective Payment System: As discussed in section XX. of this final rule with comment period, in order to reduce the Medicare program's reliance on the hospital chargemaster, and to support the development of a market-based approach to payment under the Medicare FFS system, we are finalizing a policy that hospitals will be required to report certain market-based payment rate information on their Medicare cost report for cost reporting periods ending on or after January 1, 2026, to be used in a finalized change to the methodology for calculating the IPPS MS-DRG relative weights to reflect ( printed page 53452) relative market-based pricing. Specifically, we are finalizing a requirement for facilities to report market-based rate information on the Medicare cost report; the hospital will be required to report the median of the payer-specific negotiated charges by MS-DRG that the hospital has disclosed for all of its MAOs on the most recent version of the machine-readable file (MRF) that the hospital is required to disclose under the hospital price transparency regulations at 45 CFR part 180. We also are finalizing a change to the methodology for calculating the IPPS MS-DRG relative weights to incorporate this market-based rate information, beginning in FY 2029. This finalized MS-DRG relative weight methodology will utilize the finalized median payer-specific negotiated charge information, collected on the cost report, for calculating the MS-DRG relative weights.
Graduate Medical Education (GME) Accreditation: In order to ensure that accreditation for approved medical residency programs is in compliance with applicable laws prohibiting race-based and other unlawful discrimination and to improve the accreditation process, we are finalizing that accrediting organizations may not use accreditation criteria that promote or encourage discrimination on the basis of race, color, national origin, sex, age, disability, or religion, including the use of those characteristics or intentional proxies for those characteristics as a selection criterion for employment, program participation, resource allocation, or similar activities, opportunities, or benefits. The effective date of this policy will be January 1, 2026.
Final Updates to Requirements for Hospitals to Make Public a List of Their Standard Charges: We are finalizing our proposals with modifications to the HPT regulations to enhance clarity and standardization in hospital disclosure of standard charges. Specifically, we are finalizing with modification revisions to 45 CFR 180.20 to add definitions for “tenth (10th) percentile allowed amount”, “median allowed amount”, and “ninetieth (90th) percentile allowed amount”, which are values a hospital will encode when a payer-specific negotiated charge is based on a percentage or algorithm, to more accurately reflect the distribution of actual amounts that a hospital has received for an item or service. In tandem with that, we are finalizing revisions to § 180.50 to remove the requirement for hospitals to disclose the estimated allowed amount, and, instead, require hospitals to disclose the 10th percentile, median, and 90th percentile allowed amounts, as well as the count of allowed amounts, in MRFs when payer-specific negotiated charges are based on percentages or algorithms. We are also finalizing with modification our proposal to require that hospitals use electronic data interchange (EDI) 835 electronic remittance advice (ERA) transaction data or an alternative, equivalent source of remittance data to calculate and encode the allowed amounts. We are finalizing our proposals to require that hospitals comply with specific instructions regarding the methodology that must be used to calculate such allowed amounts (including a lookback period), with some modifications. Additionally, we are finalizing, with modifications, our proposals to require hospitals to encode the attestation statement and the name of the hospital chief executive officer, president, or senior official designated to oversee the encoding of true, accurate, and complete data in the MRF. To advance the comparability of HPT data with other healthcare data, we are finalizing our proposal to require that hospitals encode their organizational, or Type 2, National Provider Identifier(s) (NPIs) in the MRFs. These policies are effective as of January 1, 2026, but we will delay enforcement of the requirements until April 1, 2026.
Finally, to encourage faster resolution and payment of CMPs, and in exchange for a hospital's admission of having violated HPT requirements, we are finalizing with clarifying edits our proposal to update § 180.90 to reduce the amount of a CMP by 35 percent, under certain conditions, when a hospital waives its right to an ALJ hearing, beginning January 1, 2026. These changes aim to improve transparency in hospital pricing, facilitate efficient enforcement of the HPT requirements, and empower consumers with actionable pricing information.
3. Summary of Costs and Benefits
In section XXVI. of this final rule with comment period, we set forth a detailed analysis of the regulatory and Federalism impacts that the final changes will have on affected entities and beneficiaries. Key estimated impacts are described below.
a. Impacts of all OPPS Changes
Table 167 in section XXVI.C. of this final rule with comment period displays the distributional impact of all the OPPS changes on various groups of hospitals and CMHCs for CY 2026 compared to all estimated OPPS payments in CY 2025. We estimate that the final policies in this final rule with comment period will result in a 2.4 percent increase in OPPS payments to providers for services. We estimate that total OPPS payments for CY 2026, including beneficiary cost-sharing, to the approximately 3,600 facilities paid under the OPPS (including general acute care hospitals, children's hospitals, cancer hospitals, and CMHCs) will increase by approximately $1.77 billion compared to CY 2025 payments due to the OPD update, excluding changes in enrollment, utilization, and case-mix. However, for providers subject to the 340B remedy offset, the 340B remedy offset is estimated to reduce payments by $275 million in CY 2026.
We estimated the isolated impact of our OPPS policies on CMHCs because CMHCs have historically only been paid for partial hospitalization services under the OPPS. Beginning CY 2024, they are also paid for IOP services under the OPPS. Based on our policy to calculate CMHC PHP and IOP costs based on 40 percent of the corresponding proposed hospital-based PHP and IOP costs, we estimate a 2.2 percent increase in CY 2026 payments to CMHCs relative to their CY 2025 payments.
b. Impacts of the Updated Wage Indexes
We estimate that our update of the wage indexes based on the fiscal year (FY) 2026 IPPS final rule wage indexes will result in a 0.1 percent increase for urban hospitals under the OPPS and a 0.2 percent increase for rural hospitals. These wage indexes include continued implementation of the Office of Management and Budget (OMB) labor market area delineations based on 2020 Decennial Census data, with updates, as discussed in section II.C. of this final rule with comment period.
c. Impacts of the Rural Adjustment and the Cancer Hospital Payment Adjustment
For CY 2026, we are continuing to provide additional payments to cancer hospitals so that a cancer hospital's payment-to-cost ratio (PCR) after the additional payments is equal to the weighted average PCR for the other OPPS hospitals using the most recently submitted or settled cost report data. Section 16002(b) of the 21st Century Cures Act requires that this weighted average PCR be reduced by 1.0 percentage point. In light of the COVID-19 PHE impact on claims and cost data used to calculate the target PCR, we maintained the CY 2021 target PCR of 0.89 through CYs 2022 and 2023. However, in CY 2024, we finalized a policy to reduce the target PCR by 1.0 percentage point each calendar year ( printed page 53453) until the target PCR equals the PCR of non-cancer hospitals using the most recently submitted or settled cost report data. For CY 2025, we finalized a target PCR of 0.87. For CY 2026, we are finalizing a target PCR of 0.87, the same PCR of non-cancer hospitals using the most recently submitted or settled cost report data, to determine the CY 2026 cancer hospital payment adjustment to be paid at cost report settlement. That is, the payment adjustments would be the additional payments needed to result in a PCR equal to 0.87 for each cancer hospital.
d. Impacts of the OPD Fee Schedule Increase Factor
For the CY 2026 OPPS/ASC, we are establishing an OPD fee schedule increase factor of 2.6 percent and applying that increase factor to the conversion factor for CY 2025. As a result of the OPD fee schedule increase factor and other budget neutrality adjustments, we estimate that urban hospitals will experience an increase in payments of approximately 2.8 percent and that rural hospitals will experience an increase in payments of 2.4 percent. Classifying hospitals by teaching status, we estimate non-teaching hospitals will experience an increase in payments of 2.7 percent, minor teaching hospitals will experience an increase in payments of 2.9 percent, and major teaching hospitals will experience an increase in payments of 2.6 percent. We also classified hospitals by the type of ownership. We estimate that hospitals with voluntary ownership will experience an increase of 2.8 percent in payments, while hospitals with government ownership will experience an increase of 2.3 percent in payments. We estimate that hospitals with proprietary ownership will experience an increase of 3.4 percent in payments.
e. Impacts of the ASC Payment Update
For impact purposes, the surgical procedures on the ASC covered surgical procedure list are aggregated into surgical specialty groups using CPT and HCPCS code range definitions. The percentage change in estimated total payments by specialty groups under the final CY 2026 payment rates, compared to estimated CY 2025 payment rates, ranges between an increase of 2 percent and an increase of 12 percent.
f. Impacts of the Market-Based MS-DRG Relative Weight Data Collection and Change in Methodology for Calculating MS-DRG Relative Weights Under the Inpatient Prospective Payment System
In section XX. of this final rule, we are finalizing a methodology for estimating the MS-DRG relative weights beginning in FY 2029 based on the median payer-specific negotiated charge information we are finalizing to collect on the cost report. We note that the estimated total annual burden hours for this data collection are as follows: 3,038 hospitals times 20 hours per hospital equals 60,760 annual burden hours and $4,857,458.20. We refer readers to section XXIII.E. of this final rule with comment period for further analysis of this assessment.
g. Impacts of Hospital Price Transparency
We finalizing a policy to require hospitals to report four new data elements when the payer-specific negotiated charge is based on a percentage or algorithm—the median allowed amount (which would replace the estimated allowed amount data element), the 10th percentile allowed amount, the 90th percentile allowed amount, and the count of allowed amounts. We are also finalizing new attestation language that hospitals must include in the machine-readable file (MRF) and requiring hospitals to encode the name of the chief executive officer, president or senior official designated to oversee the encoding of true, accurate and complete data in the MRF. Additionally, we are finalizing our proposal to require hospitals to add their National Provider Identifiers (NPIs) to the MRF. The policy will advance the comparability of standard charge information across hospitals and of the hospital price transparency (HPT) data with other healthcare data, including health plan transparency data from the Transparency in Coverage (TiC) MRFs. These new policies include a one-time burden of $1,461.80 per hospital, and a total national cost of $10,840,708.80 ($1,461.80 × 7,416 hospitals). As discussed in detail in sections XIX. and XXIII. of this final rule with comment period, we believe that the benefits to the public (and to hospitals themselves) outweigh the burden imposed on hospitals.
B. Legislative and Regulatory Authority for the Hospital OPPS
When Title XVIII of the Act was enacted, Medicare payment for hospital outpatient services was based on hospital-specific costs. In an effort to ensure that Medicare and its beneficiaries pay appropriately for services and to encourage more efficient delivery of care, the Congress mandated replacement of the reasonable cost-based payment methodology with a prospective payment system (PPS). The Balanced Budget Act of 1997 (BBA) (Pub. L. 105-33) added section 1833(t) to the Act, authorizing implementation of a PPS for hospital outpatient services. The OPPS was first implemented for services furnished on or after August 1, 2000. Implementing regulations for the OPPS are located at 42 CFR parts 410 and 419.
The Medicare, Medicaid, and SCHIP Balanced Budget Refinement Act of 1999 (BBRA) (Pub. L. 106-113) made major changes in the hospital OPPS. The following Acts made additional changes to the OPPS: the Medicare, Medicaid, and SCHIP Benefits Improvement and Protection Act of 2000 (BIPA) (Pub. L. 106-554); the Medicare Prescription Drug, Improvement, and Modernization Act of 2003 (MMA) (Pub. L. 108-173); the Deficit Reduction Act of 2005 (DRA) (Pub. L. 109-171), enacted on February 8, 2006; the Medicare Improvements and Extension Act under Division B of Title I of the Tax Relief and Health Care Act of 2006 (MIEA-TRHCA) (Pub. L. 109-432), enacted on December 20, 2006; the Medicare, Medicaid, and SCHIP Extension Act of 2007 (MMSEA) (Pub. L. 110-173), enacted on December 29, 2007; the Medicare Improvements for Patients and Providers Act of 2008 (MIPPA) (Pub. L. 110-275), enacted on July 15, 2008; the Patient Protection and Affordable Care Act (Pub. L. 111-148), enacted on March 23, 2010, as amended by the Health Care and Education Reconciliation Act of 2010 (HCERA, Pub. L. 111-152), enacted on March 30, 2010 (these two public laws are collectively known as the Affordable Care Act); the Medicare and Medicaid Extenders Act of 2010 (MMEA, Pub. L. 111-309); the Temporary Payroll Tax Cut Continuation Act of 2011 (TPTCCA, Pub. L. 112-78), enacted on December 23, 2011; the Middle Class Tax Relief and Job Creation Act of 2012 (MCTRJCA, Pub. L. 112-96), enacted on February 22, 2012; the American Taxpayer Relief Act of 2012 (Pub. L. 112-240), enacted January 2, 2013; the Pathway for SGR Reform Act of 2013 (Pub. L. 113-67) enacted on December 26, 2013; the Protecting Access to Medicare Act of 2014 (PAMA, Pub. L. 113-93), enacted on March 27, 2014; the Medicare Access and CHIP Reauthorization Act (MACRA) of 2015 (Pub. L. 114-10), enacted April 16, 2015; the Bipartisan Budget Act of 2015 (Pub. L. 114-74), enacted November 2, 2015; the Consolidated Appropriations Act, 2016 (Pub. L. 114-113), enacted on December 18, 2015, the 21st Century Cures Act (Pub. L. 114-255), enacted on December 13, 2016; the Consolidated Appropriations Act, 2018 (Pub. L. 115- ( printed page 53454) 141), enacted on March 23, 2018; the Substance Use Disorder- Prevention that Promotes Opioid Recovery and Treatment for Patients and Communities Act (Pub. L. 115-271), enacted on October 24, 2018; the Further Consolidated Appropriations Act, 2020 (Pub. L. 116-94), enacted on December 20, 2019; the Coronavirus Aid, Relief, and Economic Security Act (Pub. L. 116-136), enacted on March 27, 2020; the Consolidated Appropriations Act, 2021 (Pub. L. 116-260), enacted on December 27, 2020; the Inflation Reduction Act, 2022 (Pub. L. 117-169), enacted on August 16, 2022; and the Consolidated Appropriations Act (CAA), 2023 (Pub. L. 117-238), enacted December 29, 2022.
Under the OPPS, we generally pay for hospital Part B services on a rate-per-service basis that varies according to the APC group to which the service is assigned. We use the Healthcare Common Procedure Coding System (HCPCS) (which includes certain Current Procedural Terminology (CPT) codes) to identify and group the services within each APC. The OPPS includes payment for most hospital outpatient services, except those identified in section I.C of this final rule. Section 1833(t)(1)(B) of the Act provides for payment under the OPPS for hospital outpatient services designated by the Secretary (which includes partial hospitalization services furnished by CMHCs), and certain inpatient hospital services that are paid under Medicare Part B.
The OPPS rate is an unadjusted national payment amount that includes the Medicare payment and the beneficiary copayment. This rate is divided into a labor-related amount and a nonlabor-related amount. The labor-related amount is adjusted for area wage differences using the hospital inpatient wage index value for the locality in which the hospital or CMHC is located.
All services and items within an APC group are comparable clinically and with respect to resource use, as required by section 1833(t)(2)(B) of the Act. In accordance with section 1833(t)(2)(B) of the Act, subject to certain exceptions, items and services within an APC group cannot be considered comparable with respect to the use of resources if the highest median cost (or mean cost, if elected by the Secretary) for an item or service in the APC group is more than 2 times greater than the lowest median cost (or mean cost, if elected by the Secretary) for an item or service within the same APC group (referred to as the “2 times rule”). In implementing this provision, we generally use the cost of the item or service assigned to an APC group.
For new technology items and services, special payments under the OPPS may be made in one of two ways. section 1833(t)(6) of the Act provides for temporary additional payments, which we refer to as “transitional pass-through payments”, for at least 2 but not more than 3 years for certain drugs, biological agents, brachytherapy devices used for the treatment of cancer, and categories of other medical devices. For new technology services that are not eligible for transitional pass-through payments, and for which we lack sufficient clinical information and cost data to appropriately assign them to a clinical APC group, we have established special APC groups based on costs, which we refer to as New Technology APCs. These New Technology APCs are designated by cost bands which allow us to provide appropriate and consistent payment for designated new procedures that are not yet reflected in our claims data. Similar to pass-through payments, an assignment to a New Technology APC is temporary; that is, we retain a service within a New Technology APC until we acquire sufficient data to assign it to a clinically appropriate APC group.
C. Excluded OPPS Services and Hospitals
Section 1833(t)(1)(B)(i) of the Act authorizes the Secretary to designate the hospital outpatient services that are paid under the OPPS. While most hospital outpatient services are payable under the OPPS, section 1833(t)(1)(B)(iv) of the Act excludes payment for ambulance, physical and occupational therapy, and speech-language pathology services, for which payment is made under a fee schedule. It also excludes screening mammography, diagnostic mammography, and effective January 1, 2011, an annual wellness visit providing personalized prevention plan services. The Secretary exercises the authority granted under the statute to also exclude from the OPPS certain services that are paid under fee schedules or other payment systems. Such excluded services include, for example, the professional services of physicians and nonphysician practitioners paid under the Medicare Physician Fee Schedule (MPFS); certain laboratory services paid under the Clinical Laboratory Fee Schedule (CLFS); services for beneficiaries with end-stage renal disease (ESRD) that are paid under the ESRD prospective payment system; and services and procedures that require an inpatient stay that are paid under the hospital IPPS. In addition, section 1833(t)(1)(B)(v) of the Act does not include applicable items and services (as defined in subparagraph (A) of paragraph (21)) that are furnished on or after January 1, 2017, by an off-campus outpatient department of a provider (as defined in subparagraph (B) of paragraph (21)). We set forth the services that are excluded from payment under the OPPS in regulations at 42 CFR 419.22.
Under § 419.20(b) of the regulations, we specify the types of hospitals that are excluded from payment under the OPPS. These excluded hospitals are:
- Critical access hospitals (CAHs);
- Hospitals located in Maryland and paid under Maryland's All-Payer or Total Cost of Care Model;
- Hospitals located outside of the 50 States, the District of Columbia, and Puerto Rico;
- Indian Health Service (IHS) hospitals; and
- Rural emergency hospitals (REHs).
D. Prior Rulemaking
On April 7, 2000, we published in the Federal Register a final rule with comment period (65 FR 18434) to implement a prospective payment system for hospital outpatient services. The hospital OPPS was first implemented for services furnished on or after August 1, 2000. Section 1833(t)(9)(A) of the Act requires the Secretary to review certain components of the OPPS, not less often than annually, and to revise the groups, the relative payment weights, and the wage and other adjustments to take into account changes in medical practices, changes in technology, the addition of new services, new cost data, and other relevant information and factors.
Since initially implementing the OPPS, we have published final rules in the Federal Register annually to implement statutory requirements and changes arising from our continuing experience with this system. These rules can be viewed on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices.
E. Advisory Panel on Hospital Outpatient Payment (the HOP Panel or the Panel)
1. Authority of the Panel
Section 1833(t)(9)(A) of the Act, as amended by section 201(h) of Public Law 106-113, and redesignated by section 202(a)(2) of Public Law 106-113, requires that we consult with an expert outside advisory panel composed of an appropriate selection of representatives of providers to annually review (and ( printed page 53455) advise the Secretary concerning) the clinical integrity of the payment groups and their weights under the OPPS. In CY 2000, based on section 1833(t)(9)(A) of the Act, the Secretary established the Advisory Panel on Ambulatory Payment Classification Groups (APC Panel) to fulfill this requirement. In CY 2011, based on section 222 of the Public Health Service Act (the PHS Act), which gives discretionary authority to the Secretary to convene advisory councils and committees, the Secretary expanded the panel's scope to include the supervision of hospital outpatient therapeutic services in addition to the APC groups and weights. To reflect this new role of the panel, the Secretary changed the panel's name to the Advisory Panel on Hospital Outpatient Payment (the HOP Panel). The HOP Panel is not restricted to using data compiled by CMS, and in conducting its review, it may use data collected or developed by organizations outside the Department.
2. Establishment of the Panel
On November 21, 2000, the Secretary signed the initial charter establishing the Panel, and, at that time, named the APC Panel. This expert panel is composed of appropriate representatives of providers (currently employed full-time, not as consultants, in their respective areas of expertise) who review clinical data and advise CMS about the clinical integrity of the APC groups and their payment weights. Since CY 2012, the Panel also is charged with advising the Secretary on the appropriate level of supervision for individual hospital outpatient therapeutic services. The Panel is technical in nature, and it is governed by the provisions of the Federal Advisory Committee Act (FACA). The current charter specifies, among other requirements, that the Panel—
- May advise on the clinical integrity of Ambulatory Payment Classification (APC) groups and their associated weights;
- May advise on the appropriate supervision level for hospital outpatient services;
- May advise on OPPS APC rates for ASC covered surgical procedures;
- Continues to be technical in nature;
- Is governed by the provisions of the FACA;
- Has a Designated Federal Official (DFO); and
- Is chaired by a Federal Official designated by the Secretary.
The Panel's charter was amended on November 15, 2011, renaming the Panel and expanding the Panel's authority to include supervision of hospital outpatient therapeutic services and to add critical access hospital (CAH) representation to its membership. The Panel's charter was also amended on November 6, 2014 (80 FR 23009), and the number of members was revised from up to 19 to up to 15 members. The Panel's current charter was approved on November 21, 2024, for a 2-year period.
The current Panel membership and other information pertaining to the Panel, including its charter, Federal Register notices, membership, meeting dates, agenda topics, and meeting reports, can be viewed on the CMS website at https://www.cms.gov/Regulations-and-Guidance/Guidance/FACA/AdvisoryPanelonAmbulatoryPaymentClassificationGroups.html.
3. Panel Meetings and Organizational Structure
The Panel has held many meetings, with the last meeting taking place on August 25, 2025. The recommendations of the Panel for the most recent meeting are available on the CMS website at https://www.cms.gov/medicare/regulations-guidance/advisory-committees/hospital-outpatient-payment. Prior to each meeting, we publish a notice in the Federal Register to announce the meeting, new members, and any other changes of which the public should be aware. Beginning in CY 2017, we have transitioned to one meeting per year (81 FR 31941). In CY 2022, we published a Federal Register notice requesting nominations to fill vacancies on the Panel (87 FR 68499). We are currently accepting nominations at: https://mearis.cms.gov.
In addition, the Panel has established an administrative structure that, in part, currently includes the use of two subcommittee workgroups to provide preparatory meeting and subject support to the larger panel. The two current subcommittees include the following:
- APC Groups and Status Indicator Assignments Subcommittee, which advises and provides recommendations to the Panel on the appropriate status indicators to be assigned to HCPCS codes, including but not limited to whether a HCPCS code or a category of codes should be packaged or separately paid, as well as the appropriate APC assignment of HCPCS codes regarding services for which separate payment is made; and
- Data Subcommittee, which is responsible for studying the data issues confronting the Panel and for recommending options for resolving them.
Each of these workgroup subcommittees was established by a majority vote from the full Panel during a scheduled Panel meeting, and the Panel recommended at the August 25, 2025, meeting that these subcommittees continue. We accepted this recommendation.
For discussions of earlier Panel meetings and recommendations, we refer readers to previously published OPPS/ASC proposed and final rules, the CMS website mentioned earlier in this section, and the FACA database at https://facadatabase.gov.
F. Public Comments Received on the CY 2026 OPPS/ASC Proposed
We received approximately 3,039 timely pieces of correspondence on the CY 2026 OPPS/ASC proposed rule that appeared in the Federal Register on July 17, 2025 (90 FR 33476). We received comments from elected officials, providers and suppliers, practitioners, and advocacy groups. We provide summaries of the public comments, and our responses are set forth in the various sections of this final rule with comment period under the appropriate headings. We note that we received some public comments that were outside the scope of the CY 2026 OPPS/ASC proposed rule. Out-of-scope-public comments are not addressed in this CY 2026 OPPS/ASC final rule with comment period.
G. Public Comments Received on the CY 2025 OPPS/ASC Final Rule With Comment Period
We received approximately 29 timely pieces of correspondence on the CY 2025 OPPS/ASC final rule with comment period that appeared in the Federal Register on November 27, 2024 (89 FR 93912).
II. Updates Affecting OPPS Payments
A. Recalibration of APC Relative Payment Weights
1. Database Construction
a. Database Source and Methodology
Section 1833(t)(9)(A) of the Act requires that the Secretary review not less often than annually and revise the relative payment weights for Ambulatory Payment Classifications (APCs). In the April 7, 2000 OPPS final rule with comment period (65 FR 18482), we explained in detail how we calculated the relative payment weights that were implemented on August 1, 2000, for each APC group.
For the CY 2026 OPPS, we proposed to recalibrate the APC relative payment weights for services furnished on or after January 1, 2026, and before January 1, 2027 (CY 2026), using the same basic methodology that we described in the CY 2025 OPPS/ASC final rule with comment period (89 FR 93921 through ( printed page 53456) 93922), using CY 2024 claims data. That is, we proposed to recalibrate the relative payment weights for each APC based on claims and cost report data for hospital outpatient department (HOPD) services to construct a database for calculating APC group weights.
For the purpose of recalibrating the proposed APC relative payment weights for CY 2026, we began with approximately 143 million final action claims (claims for which all disputes and adjustments have been resolved and payment has been made) for HOPD services furnished on or after January 1, 2024, and before January 1, 2025, before applying our exclusionary criteria and other methodological adjustments. After the application of those data processing changes, we used approximately 76 million final action claims to develop the proposed CY 2026 OPPS payment weights. For exact numbers of claims used and additional details on the claims accounting process, we refer readers to the claims accounting narrative under supporting documentation for the CY 2026 OPPS/ASC proposed rule on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient.
Addendum N to the CY 2026 OPPS/ASC proposed rule (which is available via the internet on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices) includes the proposed list of bypass codes for CY 2026. The proposed list of bypass codes contains codes that are reported on claims for services in CY 2024 and, therefore, includes codes that were in effect in CY 2024 and used for billing. We proposed to retain these deleted bypass codes on the proposed CY 2026 bypass list because these codes existed in CY 2024 and were covered HOPD services in that period, and CY 2024 claims data were used to calculate proposed CY 2026 payment rates. Keeping these deleted bypass codes on the bypass list potentially allows us to create more “pseudo” single procedure claims for ratesetting purposes. “Overlap bypass codes” that are members of the proposed multiple imaging composite APCs are identified by asterisks (*) in the third column of Addendum N to the CY 2026 OPPS/ASC proposed rule. HCPCS codes that we proposed to add for CY 2026 are identified by asterisks (*) in the fourth column of Addendum N.
We did not receive any public comments on our general proposal to recalibrate the relative payment weights for each APC based on claims and cost report data for HOPD services or on our proposed bypass code process. We are finalizing as proposed the “pseudo” single claims process and the CY 2026 proposed list of bypass codes, finalized in Addendum N to this final rule with comment period (which is available via the internet on the CMS website). For this final rule with comment period, for the purpose of recalibrating the final APC relative payment weights for CY 2026, we used approximately 81 million final action claims (claims for which all disputes and adjustments have been resolved and payment has been made) for HOPD services furnished on or after January 1, 2024, and before January 1, 2025. For the exact numbers of claims used and additional details on the claims accounting process, we refer readers to the claims accounting narrative under supporting documentation for this final rule with comment period on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient.
b. Calculation and Use of Cost-to-Charge Ratios (CCRs)
For CY 2026, we proposed to continue to use the hospital-specific overall ancillary and departmental cost-to-charge ratios (CCRs) to convert charges to estimated costs through application of a revenue code-to-cost center crosswalk. To calculate the APC costs on which the proposed CY 2026 APC payment rates are based, we calculated hospital-specific departmental CCRs for each hospital for which we had CY 2024 claims data by comparing these claims data to the most recently available hospital cost reports, which, in most cases, are from CY 2023. For the proposed CY 2026 OPPS payment rates, we used the set of claims processed during CY 2024. We applied the hospital-specific CCR to the hospital's charges at the most detailed level possible, based on a revenue code-to-cost center crosswalk that contains a hierarchy of CCRs used to estimate costs from charges for each revenue code. To ensure the completeness of the revenue code-to-cost center crosswalk, we reviewed changes to the list of revenue codes for CY 2024 (the year of claims data we used to calculate the proposed CY 2026 OPPS payment rates) and updates to the National Uniform Billing Committee (NUBC) 2024 Data specifications Manual. That crosswalk is available for review and continuous comment on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient.
In accordance with our longstanding policy, similar to our finalized policy for CY 2025 OPPS ratesetting, we proposed to calculate CCRs for the standard cost centers—cost centers with a predefined label—and nonstandard cost centers—cost centers defined by a hospital—accepted by the electronic cost report database. In general, the most detailed level at which we calculate CCRs is the hospital-specific departmental level.
While we generally view the use of additional cost data as improving our OPPS ratesetting process, we have historically not included cost report lines for certain nonstandard cost centers in the OPPS ratesetting database construction when hospitals have reported these nonstandard cost centers on cost report lines that do not correspond to the cost center number. We believe it is important to further investigate the accuracy of these cost report data before including such data in the ratesetting process. Further, we believe it is appropriate to gather additional information from the public as well before including the data in OPPS ratesetting. For CY 2026, we proposed not to include the nonstandard cost centers reported in this way in the OPPS ratesetting database construction.
We did not receive any public comments on the general CCR process and therefore, we are finalizing our proposal for CY 2026 to continue to use the hospital-specific overall ancillary and departmental CCRs to convert charges to estimated costs through application of a revenue code-to-cost center crosswalk and we are also finalizing the proposed methodology.
2. Final Data Development and Calculation of Costs Used for Ratesetting
In this section of this final rule with comment period, we discuss the use of claims to calculate the OPPS payment rates for CY 2026. The Hospital OPPS page on the CMS website on which this final rule is posted ( https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient) provides an accounting of claims used in the development of the final payment rates. That accounting provides additional detail regarding the number of claims derived at each stage of the process. In addition, later in this section we discuss the file of claims that comprises the data set that is available upon payment of an administrative fee under a CMS data use agreement. The CMS website https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient, includes information about obtaining the “OPPS Limited Data Set,” which now includes the additional variables ( printed page 53457) previously available only in the OPPS Identifiable Data Set, including International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM) diagnosis codes and revenue code payment amounts. This file is derived from the CY 2024 claims that are used to calculate the final payment rates for the CY 2026 OPPS/ASC final rule with comment period.
Previously, the OPPS established the scaled relative weights on which payments are based using APC median costs, a process described in the CY 2012 OPPS/ASC final rule with comment period (76 FR 74188). However, as discussed in more detail in section II.A.2.f. of the CY 2013 OPPS/ASC final rule with comment period (77 FR 68259 through 68271), we finalized the use of geometric mean costs to calculate the relative weights on which the CY 2013 OPPS payment rates were based. While this policy changed the cost metric on which the relative payments are based, the data process in general remained the same under the methodologies that we used to obtain appropriate claims data and accurate cost information in determining estimated service cost.
We used the methodology described in sections II.A.2.a. through II.A.2.c. of this final rule with comment period to calculate the costs we used to establish the final relative payment weights used in calculating the OPPS payment rates for CY 2026 shown in Addenda A and B to this final rule with comment period (which are available via the internet on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices). We refer readers to section II.A.4. of this final rule with comment period for a discussion of the conversion of APC costs to scaled payment weights.
We note that under the OPPS, CY 2019 was the first year in which the claims data used for setting payment rates (CY 2017 data) contained lines with the modifier “PN,” which indicates nonexcepted items and services furnished and billed by off-campus provider-based departments (PBDs) of hospitals. Because nonexcepted items and services are not paid under the OPPS, in the CY 2019 OPPS/ASC final rule with comment period (83 FR 58832), we finalized a policy to remove those claim lines reported with modifier “PN” from the claims data used in ratesetting for the CY 2019 OPPS and subsequent years. For the CY 2026 OPPS, we proposed to continue to remove claim lines with modifier “PN” from the ratesetting process.
We did not receive any public comments on our proposal to continue to remove claim lines reported with modifier “PN” from the ratesetting process and are finalizing as proposed.
For details of the claims accounting process used in this CY 2026 OPPS/ASC final rule with comment period, we refer readers to the claims accounting narrative under supporting documentation for this final rule with comment period on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient.
a. Calculation of Single Procedure APC Criteria-Based Costs
(1) Blood and Blood Products
Since the implementation of the OPPS in August 2000, we have made separate payments for blood and blood products through APCs rather than packaging payment for them into payments for the procedures with which they are administered. Hospital payments for the costs of blood and blood products, as well as for the costs of collecting, processing, and storing blood and blood products, are made through the OPPS payments for specific blood product APCs.
We proposed to continue to establish payment rates for blood and blood products using our blood-specific CCR methodology (88 FR 49562), which utilizes actual or simulated CCRs from the most recently available hospital cost reports to convert hospital charges for blood and blood products to costs. This methodology has been our standard ratesetting methodology for blood and blood products since CY 2005. It was developed in response to data analysis indicating that there was a significant difference in CCRs for those hospitals with and without blood-specific cost centers and past public comments indicating that the former OPPS policy of defaulting to the overall hospital CCR for hospitals not reporting a blood-specific cost center often resulted in an underestimation of the true hospital costs for blood and blood products. To address the differences in CCRs and to better reflect hospitals' costs, our methodology simulates blood CCRs for each hospital that does not report a blood cost center by calculating the ratio of the blood-specific CCRs to hospitals' overall CCRs for those hospitals that do report costs and charges for blood cost centers and applies this mean ratio to the overall CCRs of hospitals not reporting costs and charges for blood cost centers on their cost reports. We proposed to calculate the costs upon which the proposed payment rates for blood and blood products are based using the actual blood-specific CCR for hospitals that reported costs and charges for a blood cost center and a hospital-specific, simulated, blood-specific CCR for hospitals that did not report costs and charges for a blood cost center.
We stated in the CY 2026 OPPS/ASC proposed rule (90 FR 33487) that we continue to believe that the hospital-specific, simulated, blood-specific CCR methodology takes into account the unique charging and cost accounting structure of each hospital, as it better responds to the absence of a blood-specific CCR for a hospital than alternative methodologies, such as defaulting to the overall hospital CCR or applying an average blood-specific CCR across hospitals. This methodology also yields more accurate estimated costs for these products and results in payment rates for blood and blood products that appropriately reflect the relative estimated costs of these products for hospitals without blood cost centers and for these blood products in general.
For a more detailed discussion of payments for blood and blood products through APCs, we refer readers to:
- The CY 2005 OPPS proposed rule (69 FR 50524 and 50525) for a more comprehensive discussion of the blood-specific CCR methodology;
- The CY 2008 OPPS/ASC final rule with comment period (72 FR 66807 through 66810) for a detailed history of the OPPS payment for blood and blood products; and
- The CY 2015 OPPS/ASC final rule with comment period (79 FR 66795 and 66796) for additional discussion of our policy not to make separate payments for blood and blood products when they appear on the same claims as services assigned to a C-APC.
We did not receive public comments on this provision, and therefore, we are finalizing without modification our proposal to calculate the costs upon which the payment rates for blood and blood products are based using the actual blood-specific CCR for hospitals that reported costs and charges for a blood cost center and a hospital specific, simulated, blood-specific CCR for hospitals that did not report costs and charges for a blood cost center. We are also finalizing without modification our proposal to continue to establish payment rates for blood and blood products using our blood-specific CCR methodology, which utilizes actual or simulated CCRs from the most recently available hospital cost reports to convert hospital charges for blood and blood products to costs. Please refer to Addendum B to this final rule with comment period (which is available via ( printed page 53458) the internet on the CMS website) for the final CY 2026 payment rates for blood and blood products.
(2) Brachytherapy Sources
Section 1833(t)(2)(H) of the Act mandates the creation of additional groups of covered OPD services that classify devices of brachytherapy—cancer treatment through solid source radioactive implants—consisting of a seed or seeds (or radioactive source) (“brachytherapy sources”) separately from other services or groups of services. The statute provides certain criteria for the additional groups. For the history of OPPS payment for brachytherapy sources, we refer readers to prior OPPS final rules, such as the CY 2012 OPPS/ASC final rule with comment period (77 FR 68240 and 68241). As we have stated in prior OPPS updates, we believe that adopting the general OPPS prospective payment methodology for brachytherapy sources is appropriate for several reasons (77 FR 68240). The general OPPS methodology uses costs based on claims data to set the relative payment weights for hospital outpatient services. This payment methodology results in more consistent, predictable, and equitable payment amounts per source across hospitals by averaging the extremely high and low values, in contrast to payment based on hospitals' charges adjusted to costs. We believe that the OPPS methodology, as opposed to payment based on hospitals' charges adjusted to cost, also would provide hospitals with incentives for efficiency in the provision of brachytherapy services to Medicare beneficiaries. Moreover, this approach is consistent with our payment methodology for most items and services paid under the OPPS. We refer readers to the CY 2016 OPPS/ASC final rule with comment period (80 FR 70323 through 70325) for further discussion of the history of OPPS payment for brachytherapy sources.
For CY 2026, except where otherwise indicated, we proposed to continue our policy and use the costs derived from CY 2024 claims data to set the proposed CY 2026 payment rates for brachytherapy sources because we proposed to use CY 2024 data to set the proposed payment rates for most other items and services that would be paid under the CY 2026 OPPS. With the exception of the proposed payment rate for brachytherapy source C2645 (Brachytherapy planar source, palladium-103, per square millimeter) and the proposed payment rates for low-volume brachytherapy APCs discussed in section III.D. of the CY 2026 OPPS/ASC proposed rule, we proposed to base the payment rates for brachytherapy sources on the geometric mean unit costs for each source, consistent with the methodology that we proposed for other items and services paid under the OPPS, as discussed in section II.A.2. of the CY 2026 OPPS/ASC proposed rule. We also proposed for CY 2026 and subsequent years to continue the other payment policies for brachytherapy sources that we finalized and first implemented in the CY 2010 OPPS/ASC final rule with comment period (74 FR 60537). For CY 2026 and subsequent years, we proposed to pay for the stranded and nonstranded not otherwise specified (NOS) codes, HCPCS codes C2698 (Brachytherapy source, stranded, not otherwise specified, per source) and C2699 (Brachytherapy source, non-stranded, not otherwise specified, per source), at a rate equal to the lowest stranded or nonstranded prospective payment rate for such sources, respectively, on a per-source basis (as opposed to, for example, per mCi), which is based on the policy we established in the CY 2008 OPPS/ASC final rule with comment period (72 FR 66785). For CY 2026 and subsequent years, we also proposed to continue the policy we implemented in the CY 2010 OPPS/ASC final rule with comment period (74 FR 60537) regarding payment for new brachytherapy sources for which we have no claims data, for the same reasons we discussed in the CY 2008 OPPS/ASC final rule with comment period (72 FR 66786; which was delayed until January 1, 2010, by section 142 of Pub. L. 110-275). Specifically, this policy is intended to enable us to assign new HCPCS codes for new brachytherapy sources to their own APCs, with prospective payment rates set based on our consideration of external data and other relevant information regarding the expected costs of the sources to hospitals. The proposed CY 2026 payment rates for brachytherapy sources are included in Addendum B to the OPPS/ASC proposed rule (which is available via the internet on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices) and identified with status indicator “U (Brachytherapy Sources, Paid under OPPS; separate APC payment).”
For CY 2018, we assigned status indicator “U” to HCPCS code C2645 (Brachytherapy planar source, palladium-103, per square millimeter) in the absence of claims data and established a payment rate using external data (invoice price) at $4.69 per mm2 for the brachytherapy source's APC—APC 2648 (Brachytx planar, p-103) (82 FR 49233 through 49244). For CY 2019, in the absence of sufficient claims data, we continued to establish a payment rate for C2645 at $4.69 per mm2 for APC 2648 (Brachytx planar, p-103) (83 FR 58834 through 58836). Our CY 2018 claims data available for the CY 2020 OPPS/ASC final rule with comment period (84 FR 61142) included two claims with a geometric mean cost for HCPCS code C2645 of $1.02 per mm2 . In response to comments from interested parties, we agreed that, given the limited claims data available and a new outpatient indication for C2645, a payment rate for HCPCS code C2645 based on the geometric mean cost of $1.02 per mm2 may not adequately reflect the cost of HCPCS code C2645. In the CY 2020 OPPS/ASC final rule with comment period, we finalized our policy to use our equitable adjustment authority under section 1833(t)(2)(E) of the Act, which states that the Secretary shall establish, in a budget neutral manner, other adjustments as determined to be necessary to ensure equitable payments, to maintain the CY 2019 payment rate of $4.69 per mm2 for HCPCS code C2645 for CY 2020. Similarly, in the absence of sufficient claims data to establish an APC payment rate, in the CY 2021, CY 2022, CY 2023, CY 2024, and CY 2025 OPPS/ASC final rules with comment period (85 FR 85879 through 85880, 86 FR 63469, 87 FR 71760-71761, 88 FR 81553, and 89 FR 93925), we finalized our policy to use our equitable adjustment authority under section 1833(t)(2)(E) of the Act to maintain the CY 2019 payment rate of $4.69 per mm2 for HCPCS code C2645 for CYs 2021 through 2025.
There were no CY 2024 claims available that reported HCPCS code C2645 for the CY 2026 OPPS/ASC proposed rule. Therefore, in the absence of claims data, we proposed to continue to use our equitable adjustment authority under section 1833(t)(2)(E) of the Act to maintain the CY 2025 payment rate of $4.69 per mm2 for HCPCS code C2645, which we proposed to be assigned to APC 2648 (Brachytx planar, p-103), for CY 2026.
Additionally, for CY 2022 and subsequent calendar years, we adopted a Universal Low Volume APC policy for clinical and brachytherapy APCs. As discussed in further detail in section X.C. of the CY 2022 OPPS/ASC final rule with comment period (86 FR 63743 through 63747), we adopted this policy to mitigate wide variation in payment rates that occur from year to year for APCs with low utilization. Such volatility in payment rates from year to ( printed page 53459) year can result in even lower utilization and potential barriers to access. Brachytherapy APCs that have fewer than 100 single claims used for ratesetting purposes are designated as Low Volume APCs unless an alternative payment rate is applied, such as the use of our equitable adjustment authority under section 1833(t)(2)(E) of the Act in the case of APC 2648 (Brachytx planar, p-103), for which HCPCS code C2645 (Brachytherapy planar source, palladium-103, per square millimeter) is the only code assigned as discussed previously in this section.
For CY 2026, we proposed to designate six brachytherapy APCs as Low Volume APCs as these APCs met our criteria to be designated as Low Volume APCs.
We did not receive public comments on this provision, and therefore, we are finalizing as proposed. Except for brachytherapy APCs designated as Low Volume APCs and APC 2648, we will continue our policy and use the costs derived from CY 2024 claims data to set the final CY 2026 payment rates for brachytherapy sources. We will continue to pay for the stranded and nonstranded not otherwise specified (NOS) codes, HCPCS codes C2698 (Brachytherapy source, stranded, not otherwise specified, per source) and C2699 (Brachytherapy source, non-stranded, not otherwise specified, per source), at a rate equal to the lowest stranded or nonstranded prospective payment rate for such sources, respectively, on a per-source basis. Further, we will use our equitable adjustment authority under section 1833(t)(2)(E) of the Act to maintain the CY 2025 payment rate of $4.69 per mm2 for HCPCS code C2645, which we are assigning to APC 2648 (Brachytx planar, p1-103), for CY 2026. We refer readers to section III.D. of this final rule with comment period for information on the brachytherapy APCs we are finalizing to designate as Low Volume APCs.
The final CY 2026 payment rates for brachytherapy sources are included in Addendum B to this final rule with comment period (which is available via the internet on the CMS website) and are identified with status indicator “U.” We continue to invite interested parties to submit recommendations for new codes to describe new brachytherapy sources. Such recommendations should be directed via email to outpatientpps@cms.hhs.gov.
b. Comprehensive APCs (C-APCs) for CY 2026
(1) Background
In the CY 2014 OPPS/ASC final rule with comment period (78 FR 74861 through 74910), we finalized a comprehensive payment policy that packages payment for adjunctive and secondary items, services, and procedures into the most costly primary procedure under the OPPS at the claim level. The policy was finalized in CY 2014, but the effective date was delayed until January 1, 2015, to allow additional time for further analysis, opportunity for public comment, and systems preparation. The comprehensive APC (C-APC) policy was implemented effective January 1, 2015, with modifications and clarifications in response to public comments received regarding specific provisions of the C-APC policy (79 FR 66798 through 66810).
A C-APC is defined as a classification for the provision of a primary service and all adjunctive services provided to support the delivery of the primary service. We established C-APCs as a category broadly for OPPS payment and implemented 25 C-APCs beginning in CY 2015 (79 FR 66809 and 66810). We have gradually added new C-APCs since the policy was implemented beginning in CY 2015, with the number of C-APCs now totaling 72 (80 FR 70332; 81 FR 79584 and 79585; 83 FR 58844 through 58846; 84 FR 61158 through 61166; 85 FR 85885; 86 FR 63474; 87 FR 71769; 88 FR 81562; and 89 FR 93926).
Under our C-APC policy, we designate a service described by a HCPCS code assigned to a C-APC as the primary service when the service is identified by OPPS status indicator “J1”. When such a primary service is reported on a hospital outpatient claim, taking into consideration the few exceptions that are discussed below, we make payment for all other items and services reported on the hospital outpatient claim as being integral, ancillary, supportive, dependent, and adjunctive to the primary service (hereinafter collectively referred to as “adjunctive services”) and representing components of a complete comprehensive service (78 FR 74865 and 79 FR 66799). Payments for adjunctive services are packaged into the payments for the primary services. This results in a single prospective payment for each of the primary, comprehensive services based on the costs of all reported services at the claim level. One example of a primary service would be a partial mastectomy, and an example of a secondary service packaged into that primary service would be a radiation therapy procedure.
Services excluded from the C-APC policy under the OPPS include services that are not covered OPD services, services that cannot, by statute, be paid for under the OPPS, and services that are required by statute to be separately paid. This includes certain mammography and ambulance services that are not covered OPD services in accordance with section 1833(t)(1)(B)(iv) of the Act; brachytherapy seeds, which also are required by statute to receive separate payment under section 1833(t)(2)(H) of the Act; pass-through payment drugs and devices, which also require separate payment under section 1833(t)(6) of the Act; self-administered drugs (SADs) that are not otherwise packaged as supplies because they are not covered under Medicare Part B under section 1861(s)(2)(B) of the Act; and certain preventive services (78 FR 74865 and 79 FR 66800 and 66801). A list of services excluded from the C-APC policy is included in Addendum J to this final rule with comment period (which is available via the internet on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices). If a service does not appear on this list of excluded services, payment for it will be packaged into the payment for the primary C-APC service when it appears on an outpatient claim with a primary C-APC service.
The C-APC policy payment methodology set forth in the CY 2014 OPPS/ASC final rule with comment period and modified and implemented beginning in CY 2015 is summarized as follows (78 FR 74887 and 79 FR 66800):
Basic Methodology. As stated in the CY 2015 OPPS/ASC final rule with comment period, we define the C-APC payment policy as including all covered OPD services on a hospital outpatient claim reporting a primary service that is assigned to status indicator “J1,” [1] excluding services that are not covered OPD services or that cannot by statute be paid for under the OPPS. Services and procedures described by HCPCS codes assigned to status indicator “J1” are assigned to C-APCs based on our usual APC assignment methodology by evaluating the geometric mean costs of the primary service claims to establish resource similarity and the clinical ( printed page 53460) characteristics of each procedure to establish clinical similarity within each APC.
In the CY 2016 OPPS/ASC final rule with comment period, we expanded the C-APC payment methodology to qualifying extended assessment and management encounters through the “Comprehensive Observation Services” C-APC (C-APC 8011). Services within this APC are assigned status indicator “J2.” [2] Specifically, we make a payment through C-APC 8011 for a claim that:
- Does not contain a procedure described by a HCPCS code to which we have assigned status indicator “T;” [3]
- Contains 8 or more units of services described by HCPCS code G0378 (Hospital observation services, per hour);
- Contains services provided on the same date of service or 1 day before the date of service for HCPCS code G0378 that are described by one of the following codes: HCPCS code G0379 (Direct admission of patient for hospital observation care) on the same date of service as HCPCS code G0378; CPT code 99281 (Emergency department visit for the evaluation and management of a patient (Level 1)); CPT code 99282 (Emergency department visit for the evaluation and management of a patient (Level 2)); CPT code 99283 (Emergency department visit for the evaluation and management of a patient (Level 3)); CPT code 99284 (Emergency department visit for the evaluation and management of a patient (Level 4)); CPT code 99285 (Emergency department visit for the evaluation and management of a patient (Level 5)) or HCPCS code G0380 (Type B emergency department visit (Level 1)); HCPCS code G0381 (Type B emergency department visit (Level 2)); HCPCS code G0382 (Type B emergency department visit (Level 3)); HCPCS code G0383 (Type B emergency department visit (Level 4)); HCPCS code G0384 (Type B emergency department visit (Level 5)); CPT code 99291 (Critical care, evaluation and management of the critically ill or critically injured patient; first 30-74 minutes); or HCPCS code G0463 (Hospital outpatient clinic visit for assessment and management of a patient); and
- Does not contain services described by a HCPCS code to which we have assigned status indicator “J1.”
The assignment of status indicator “J2” to a specific set of services performed in combination with each other allows for all other OPPS payable services and items reported on the claim (excluding services that are not covered OPD services or that cannot by statute be paid for under the OPPS) to be deemed adjunctive services representing components of a comprehensive service and resulting in a single prospective payment for the comprehensive service based on the costs of all reported services on the claim (80 FR 70333 through 70336).
Services included under the C-APC payment packaging policy, that is, services that are typically adjunctive to the primary service and provided during the delivery of the comprehensive service, include diagnostic procedures, laboratory tests, and other diagnostic tests and treatments that assist in the delivery of the primary procedure; visits and evaluations performed in association with the procedure; uncoded services and supplies used during the service; durable medical equipment as well as prosthetic and orthotic items and supplies when provided as part of the outpatient service; and any other components reported by HCPCS codes that represent services that are provided during the complete comprehensive service (78 FR 74865 and 79 FR 66800).
In addition, payment for hospital outpatient department services that are similar to therapy services, such as speech language pathology, and delivered either by therapists or nontherapists is included as part of the payment for the packaged complete comprehensive service. These services that are provided during the perioperative period are adjunctive services and are deemed not to be therapy services as described in section 1834(k) of the Act, regardless of whether the services are delivered by therapists or other nontherapist health care workers. We have previously noted that therapy services are those provided by therapists under a plan of care in accordance with section 1835(a)(2)(C) and section 1835(a)(2)(D) of the Act and are paid for under section 1834(k) of the Act, subject to annual therapy caps as applicable (78 FR 74867 and 79 FR 66800). However, certain other services similar to therapy services are considered and paid for as hospital outpatient department services. Payment for these nontherapy outpatient department services that are reported with therapy codes and provided with a comprehensive service is included in the payment for the packaged complete comprehensive service. We note that these services, even though they are reported with therapy codes, are hospital outpatient department services and not therapy services. We refer readers to the July 2016 OPPS Change Request 9658 (Transmittal 3523)[4] for further instructions on reporting these services in the context of a C-APC service.
Items included in the packaged payment provided in conjunction with the primary service also include all drugs, biologicals, and radiopharmaceuticals, regardless of cost, except those drugs with pass-through payment status and SADs, unless they function as packaged supplies (78 FR 74868, 74869, and 74909 and 79 FR 66800). We refer readers to Section 50.2M, Chapter 15 of the Medicare Benefit Policy Manual for a description of our policy on SADs treated as hospital outpatient supplies, including lists of SADs that function as supplies and those that do not function as supplies.[5]
We define each hospital outpatient claim reporting a single unit of a single primary service assigned to status indicator “J1” as a single “J1” unit procedure claim (78 FR 74871 and 79 FR 66801). Line-item charges for services included on the C-APC claim are converted to line-item costs, which are then summed to develop the estimated APC costs. These claims are then assigned one unit of the service with status indicator “J1” and later used to develop the geometric mean costs for the C-APC relative payment weights. (We note that we use the term “comprehensive” to describe the geometric mean cost of a claim reporting “J1” service(s) or the geometric mean cost of a C-APC, inclusive of all the items and services included in the C-APC service payment bundle.) Charges for services that would otherwise be separately payable are added to the charges for the primary service. This process differs from our traditional cost accounting methodology only in that all such services on the claim are packaged (except certain services as described above). We apply our standard data trims, which exclude claims with extremely high primary units or extreme costs.
The comprehensive geometric mean costs are used to establish resource similarity and, along with clinical similarity, dictate the assignment of the primary services to the C-APCs. We ( printed page 53461) establish a ranking of each primary service (single unit only) to be assigned to status indicator “J1” according to its comprehensive geometric mean costs. For the minority of claims reporting more than one primary service assigned to status indicator “J1” or units thereof, we identify one “J1” service as the primary service for the claim based on our cost-based ranking of primary services. We then assign these multiple “J1” procedure claims to the C-APC to which the service designated as the primary service is assigned. If the reported “J1” services on a claim map to different C-APCs, we designate the “J1” service assigned to the C-APC with the highest comprehensive geometric mean cost as the primary service for that claim. If the reported multiple “J1” services on a claim map to the same C-APC, we designate the most costly service (at the HCPCS code level) as the primary service for that claim. This process results in initial assignments of claims for the primary services assigned to status indicator “J1” to the most appropriate C-APCs based on both single and multiple procedure claims reporting these services and clinical and resource homogeneity.
Complexity Adjustments. We use complexity adjustments to provide increased payment for certain comprehensive services. We apply a complexity adjustment by promoting qualifying paired “J1” service code combinations or paired code combinations of “J1” services and certain add-on codes (as described further below) from the originating C-APC (the C-APC to which the designated primary service is first assigned) to the next higher paying C-APC in the same clinical family of C-APCs. We apply this type of complexity adjustment when the paired code combination represents a complex, costly form or version of the primary service according to the following criteria:
- Frequency of 25 or more claims reporting the code combination (frequency threshold); and
- Violation of the 2 times rule, as stated in section 1833(t)(2) of the Act and section III.B.2. of this final rule with comment period, in the originating C-APC (cost threshold).
These criteria identify paired code combinations that occur commonly and exhibit materially greater resource requirements than the primary service. The CY 2017 OPPS/ASC final rule with comment period (81 FR 79582) included a revision to the complexity adjustment eligibility criteria. Specifically, we finalized a policy to discontinue the requirement that a code combination (that qualifies for a complexity adjustment by satisfying the frequency and cost criteria thresholds described above) also not create a 2 times rule violation in the higher level or receiving APC.
After designating a single primary service for a claim, we evaluate that service in combination with each of the other procedure codes reported on the claim assigned to status indicator “J1” (or certain add-on codes) to determine if there are paired code combinations that meet the complexity adjustment criteria. For a new HCPCS code, we determine initial C-APC assignment and qualification for a complexity adjustment using the best available information, crosswalking the new HCPCS code to a predecessor code(s) when appropriate.
Once we have determined that a particular code combination of “J1” services (or combinations of “J1” services reported in conjunction with certain add-on codes) represents a complex version of the primary service because it is sufficiently costly, frequent, and a subset of the primary comprehensive service overall according to the criteria described above, we promote the claim including the complex version of the primary service as described by the code combination to the next higher cost C-APC within the clinical family, unless the primary service is already assigned to the highest cost APC within the C-APC clinical family or assigned to the only C-APC in a clinical family. We do not create new APCs with a comprehensive geometric mean cost that is higher than the highest geometric mean cost (or only) C-APC in a clinical family just to accommodate potential complexity adjustments. Therefore, the highest payment for any claim including a code combination for services assigned to a C-APC would be the highest paying C-APC in the clinical family (79 FR 66802).
We package payment for all add-on codes into the payment for the C-APC. However, certain primary service add-on combinations may qualify for a complexity adjustment. As noted in the CY 2016 OPPS/ASC final rule with comment period (80 FR 70331), all add-on codes that can be appropriately reported in combination with a base code that describes a primary “J1” service are evaluated for a complexity adjustment.
To determine which combinations of primary service codes reported in conjunction with an add-on code may qualify for a complexity adjustment for CY 2026, we apply the frequency and cost criteria thresholds discussed above, testing claims reporting one unit of a single primary service assigned to status indicator “J1” and any number of units of a single add-on code for the primary “J1” service. If the frequency and cost criteria thresholds for a complexity adjustment are met and reassignment to the next higher cost APC in the clinical family is appropriate (based on meeting the criteria outlined above), we make a complexity adjustment for the code combination; that is, we reassign the primary service code reported in conjunction with the add-on code to the next higher cost C-APC within the same clinical family of C-APCs. As previously stated, we package payment for add-on codes into the C-APC payment rate. If any add-on code reported in conjunction with the “J1” primary service code does not qualify for a complexity adjustment, payment for the add-on service continues to be packaged into the payment for the primary service and is not reassigned to the next higher cost C-APC. We list the final complexity adjustments for “J1” and add-on code combinations for CY 2026, along with all the other final complexity adjustments, in Addendum J to this final rule with comment period (which is available via the internet on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices).
Addendum J to this final rule with comment period includes the cost statistics for each code combination that would qualify for a complexity adjustment (including primary code and add-on code combinations). Addendum J to this final rule with comment period also contains summary cost statistics for each of the paired code combinations that describe a complex code combination that would qualify for a complexity adjustment and be reassigned to the next higher cost C-APC within the clinical family. The combined statistics for all final reassigned complex code combinations are represented by an alphanumeric code with the first four digits of the designated primary service followed by a letter. For example, the final geometric mean cost listed in Addendum J for the code combination described by complexity adjustment assignment 3320R, which is assigned to C-APC 5224 (Level 4 Pacemaker and Similar Procedures), includes all paired code combinations that will be reassigned to C-APC 5224 when CPT code 33208 is the primary code. Providing the information contained in Addendum J to this final rule with comment period allows interested parties the ( printed page 53462) opportunity to better assess the impact associated with the assignment of claims with each of the paired code combinations eligible for a complexity adjustment.
We received public comments on these proposals. The following is a summary of the comments received and our responses.
Comment: We received support from commenters for a variety of existing and proposed complexity adjustments.
Response: We thank the commenters for their support.
Comment: Multiple commenters requested that CMS apply a complexity adjustment to additional code combinations. The specific C-APC complexity adjustment code combinations requested by the commenters for CY 2026 are listed in Table 1.
( printed page 53463) ( printed page 53464) ( printed page 53465) ( printed page 53466)Response: We reviewed each of the requested code combinations suggested by commenters, listed in Table 1, against our complexity adjustment criteria. The following code combinations met our cost and frequency criteria, qualifying for a complexity adjustment for CY 2026:
- Primary HCPCS code 93454 with secondary HCPCS code 0523T.
- Primary HCPCS code 93460 with secondary HCPCS code 0523T.
- Primary HCPCS code 28740 with secondary HCPCS code 20902.
- Primary HCPCS 28750 code with secondary HCPCS code 20900.
- Primary HCPCS code 28750 with secondary HCPCS code 28308.
- Primary HCPCS 22513 code with secondary HCPCS code 22515.
- Primary HCPCS 22514 code with secondary HCPCS code 22515.
- Primary HCPCS 43255 code with secondary HCPCS code 43245.
- Primary HCPCS 37187 code with secondary HCPCS code 37248.
All the remaining code combinations listed failed to meet our cost or frequency criteria and do not qualify for complexity adjustments for CY 2026. Addendum J to this final rule with comment period includes the cost statistics for each code combination that was evaluated for a complexity adjustment.
Comment: Several commenters brought to our attention that some qualifying complexity adjustments pairings were promoted up two APC levels. Commenters requested that we clarify that code pairings that qualify for a complexity adjustment are only promoted to an APC one level higher. Other commenters flagged code pairings that qualified for complexity adjustments but were not mapped to the next highest APC in their clinical family. Other commenters found that some code pairings in Addendum J which qualified for complexity adjustments in the “Complexity Adjustment Evaluation” tab were not listed in the “Complexity Adjustments” tab.
Response: We thank the commenters for bringing this to our attention. It has been our longstanding policy to promote coding pairings that qualify for complexity adjustments to the next highest APC in their clinical family. In Addendum J to this final rule with comment period, all code pairings that qualify for complexity adjustments are mapped to the next highest APC in their clinical family. All code pairings that qualify for complexity adjustments can be found in both “Complexity Adjustment Evaluation” tab and the “Complexity Adjustments” tab of Addendum J.
Comment: We received requests to evaluate HCPCS code pairings for complexity adjustments that were not any combination of “J1” or add-on codes. Commenters requested that CMS evaluate codes with status indicators “S”, which indicates a code is paid separately and is not subject to multiple procedure discounting. Commenters specifically requested that CMS consider G0390 (trauma activation with critical care) and G0257 (emergency/unscheduled dialysis) because the commenters believe that complexity adjustments do not currently recognize the cost of trauma cases.
Response: As stated in the CY 2015 OPPS/ASC final rule with comment period (79 FR 66770 through 67034), under our C-APC policy, we designate a service described by a HCPCS code assigned to a C-APC as the primary service when the service is identified by OPPS status indicator “J1.” We use complexity adjustments to provide increased payment for certain ( printed page 53467) comprehensive services. We apply a complexity adjustment by promoting qualifying paired “J1” service code combinations or paired code combinations of “J1” services and certain add-on codes from the originating C-APC (the C-APC to which the designated primary service is first assigned) to the next higher paying C-APC in the same clinical family of C-APCs. If a code pairing is not “J1” plus “J1” or “J1” plus an add-on code, it would not be evaluated for a complexity adjustment. In the CY 2026 OPPS/ASC proposed rule, we did solicit comments on revising our complexity adjustment methodology, which is summarized below in section II.B.2. of this final rule with comment period. Additionally, the assigned status indicators for HCPCS codes are open for public comment through our annual rulemaking process if commenters feel that any code may need to be reevaluated.
Comment: Commenters requested that CMS provide additional information so that other interested parties are able to replicate Addendum J in its entirety. Commenters specifically requested additional clarity around the treatment of add-on codes.
Response: We refer commenters to the claims accounting narrative under supporting documentation for this CY 2026 OPPS/ASC final rule with comment period on the CMS website. The claims accounting narrative provides a detailed overview of how we processed the CY 2024 claims data to produce the proposed prospective CY 2026 OPPS payment rates.
After consideration of public comments, we are finalizing the C-APC complexity adjustment policy for CY 2026 as proposed. We are also finalizing the proposed complexity adjustments, with the addition of nine new code combinations suggested by commenters that meet our complexity adjustment criteria. We have made additional updates to the claims accounting narrative, specifically the section on Comprehensive APCs, in order to provide additional clarity on the claims accounting process used for determining complexity adjustments. We have also updated Addendum J to provide additional transparency on this issue.
(2) Comment Solicitation on C-APC Complexity Adjustment Criteria
In response to a variety of requests from interested parties, as well as public comments in past rulemaking, related to our C-APC complexity adjustment criteria, in the CY 2026 OPPS/ASC proposed rule (90 FR 33491) we included a comment solicitation on C-APC adjustment criteria. Interested parties and commenters have requested that CMS modify the established C-APC complexity adjustment eligibility criteria of 25 or more claims reporting the code combination (frequency threshold) and a violation of the 2 times rule in the originating C-APC (cost threshold) to allow additional code combinations to qualify for complexity adjustments. Interested parties and commenters have also requested expanding the qualifying code combinations for complexity adjustments to allow clusters of procedures, consisting of a “J1” code pair and multiple other associated add-on codes, to be used in combination with that “J1” code pair to qualify. These interested parties and commenters have noted these expanded combinations may allow for a more accurate reflection of medical practice when multiple procedures are performed together or there are certain complex procedures that include numerous add-on codes.
For CY 2026, we solicited comments on potential refinements to our C-APC complexity adjustment criteria. Under this solicitation, we sought comment on expanding code combinations that qualify for complexity adjustments, including any specifications related to determining specific combination types and how they represent a complex, costly subset of the primary service. We sought comment on how CMS could identify service pairings or clusters of services for complexity adjustments that are clinically appropriate but are currently not evaluated for complexity adjustments. Additionally, if we were to expand our complexity adjustment criteria to allow for clusters of codes, we sought comment on what the appropriate cost and frequency thresholds could be used to identify which code clusters truly reflect complex and resource-intensive code combinations that are commonly performed in the hospital outpatient department setting.
We sought comment on which services are clinically integral to the provision of “J1” services that would qualify for a complexity adjustment under an expanded evaluation framework. Specifically, we sought comment on what criteria we could add, reflecting clinical practice, that would determine the costly additional components that are often associated with other high-cost packaged items and services. Finally, we sought comment on how we might address the unintended consequences of granular coding on the mechanics of the complexity adjustment criteria and if highly specific coding truly reflects clinical practice in hospital outpatient departments.
We received public comments on this comment solicitation. The following is a summary of the comments we received and our responses.
Comment: We received a number of comments on C-APC complexity adjustment criteria. Commenters shared their ideas on how to refine the complexity adjustment criteria and methodology, on the mechanics of how complexity adjustments are evaluated, and how the data is presented to the public.
Many commenters expressed their need for additional information so that interested parties would be able to accurately replicate Addendum J. To that end, commenters requested that CMS provide sufficient detail in the CY 2026 OPPS final rule with comment period Claims Accounting Narrative such that Addendum J could be fully replicated in its entirety. Other commenters suggested that CMS detail the step-by-step claims accounting process used to count claims for the purpose of evaluating complexity adjustment eligibility.
Some commenters requested that CMS include the full list of add-on codes eligible for evaluation for the complexity adjustment, along with the method CMS used to determine whether or not a code was eligible for complexity adjustment evaluation. Commenters also requested that CMS provide additional information and greater transparency on the methodology used to evaluate the complexity adjustment frequency criteria for “J1” and add-on codes. Specifically, commenters suggested that CMS should evaluate the total costs of “J1 + N” code combinations in the same manner as single J1 procedures and “J1 + J1” code combinations. Commenters indicated that this would be consistent with how CMS evaluates the cost of single frequency “J1” procedures and “J1 + J1” procedure code combinations.
Many comments suggested CMS could modify the methodology used when determining the cost threshold for a code combination to qualify for a complexity adjustment. Commenters recommended that CMS use the “lower of” methodology to determine the eligibility cost threshold:
- Current methodology using the two times rule, OR
- The lowest GMC of significant procedures in the APC to which the code combination would be eligible for complexity adjustment.
Commenters contended that the current methodology may be appropriate for lower cost APCs where the differences between the APC levels ( printed page 53468) and procedures are less significant. However, for higher-cost APCs, they say that using the two-times rule becomes problematic, and in some instances, the threshold is higher than the cost of any single procedure in the higher paying APC. By adopting the recommended methodology, commenters explained that more code combinations would be eligible for complexity adjustments. Commenters went on to say that the purpose of complexity adjustments is to ensure appropriate payment under the C-APC methodology, and therefore it is critical that CMS employ a methodology that reflects a more appropriate eligibility cost threshold consistent with the single “J1” procedures included in each APC.
Nearly all commenters on this issue agreed that CMS should expand its review of procedure combinations to include clusters of “J1” primary service and add-on codes, rather than only code pairs. Commenters asserted that this would better reflect medical practice when multiple procedures are performed together. Some commenters even suggested that CMS consider procedure combinations that include clusters of “J1” and add-on codes, and certain select HCPCS device codes. Commenters had specific suggestions on how using code clusters could work. One commenter suggested using clusters but maintaining cost and frequency thresholds, further suggesting using eligibility for Transitional Pass-Through payment as one criterion by which to identify instances where a code cluster would be appropriate for an expanded complexity adjustment. Other commenters suggested that CMS could limit the evaluation of code clusters to those nominated by the public on an annual basis.
Multiple commenters requested that CMS revise the complexity adjustment policy by allowing promotion of qualifying code combinations, even when the primary code is already assigned to the highest level of APCs within a clinical family (for example, creating a new APC level to accommodate these higher cost cases). Other commenters asserted that in order to maintain stability and predictability of payments associated with complex procedures, CMS should allow established qualifying codes to maintain the complexity adjusted payment for three calendar years before they are required to go through the eligibility review. Further, commenters said that APC reassignment for codes that qualify for 3 consecutive years should be made permanent.
In response to our request for comments on whether highly specific coding truly reflects clinical practice in hospital outpatient departments, commenters asserted that CMS' broad C-APC packaging policy, including the current eligibility criteria for complexity adjustments, has discouraged complete and accurate hospital reporting of packaged costs. Commenters explained that since hospitals receive the same C-APC payment for furnishing multiple packaged services, there is no incentive to report costs that do not drive reimbursement. Commenters asserted that this underreporting of packaged costs, coupled with CMS' claims edits for device-intensive procedures, leads to underpayment for APCs that rely heavily on packaged items, especially those with expensive routine supplies.
We also received a variety of other comments on ways to expand the scope of the complexity adjustment methodology, including establishing a provisional complexity adjustment process for code combinations involving newly removed IPO list procedures, reviewing bilateral procedure claims with high-cost implantable supplies, evaluating non-J1 procedure codes such as status indicators “S” and “T” for significant cost variation, and waiving the Administrative Procedures Act requirements for public comment to adopt suggested changes in the CY 2026 OPPS/ASC final rule for January 1, 2026.
Response: We sincerely thank commenters for their interest and engagement on this important issue. Given the wide array of information presented through this public comment process, we will take the technical recommendations, alternate methodological approaches, and other detailed feedback provided into consideration for future notice and comment rulemaking. We welcome ongoing dialogue and engagement from interested parties regarding suggestions for potential future C-APC complexity adjustment criteria revisions.
(3) Exclusion of Procedures Assigned to New Technology APCs From the C-APC Policy
Services that are assigned to New Technology APCs are typically new procedures that do not have sufficient claims history to establish an accurate payment for them. Beginning in CY 2002, we retain services within New Technology APC groups until we gather sufficient claims data to enable us to assign the service to an appropriate clinical APC. This policy allows us to move a service from a New Technology APC in less than 2 years if sufficient data are available. It also allows us to retain a service in a New Technology APC for more than 2 years if sufficient data upon which to base a decision for reassignment have not been collected (82 FR 59277).
The C-APC payment policy packages payment for adjunctive and secondary items, services, and procedures into the most costly primary procedure under the OPPS at the claim level. Prior to CY 2019, when a procedure assigned to a New Technology APC was included on the claim with a primary procedure, identified by OPPS status indicator “J1,” payment for the new technology service was typically packaged into the payment for the primary procedure. Because the new technology service was not separately paid in this scenario, the overall number of single claims available to determine an appropriate clinical APC for the new service was reduced. This was contrary to the objective of the New Technology APC payment policy, which is to gather sufficient claims data to enable us to assign the service to an appropriate clinical APC.
To address this issue and ensure that there are sufficient claims data for services assigned to New Technology APCs, in the CY 2019 OPPS/ASC final rule with comment period (83 FR 58847), we finalized excluding payment for any procedure that is assigned to a New Technology APC (APCs 1491 through 1599 and APCs 1901 through 1908) from being packaged when included on a claim with a “J1” service assigned to a C-APC. In the CY 2020 OPPS/ASC final rule with comment period, we finalized that beginning in CY 2020, payment for services assigned to a New Technology APC would be excluded from being packaged into the payment for comprehensive observation services assigned status indicator “J2” when they are included on a claim with a “J2” service (84 FR 61167).
(4) Exclusion of Drugs and Biologicals Described by HCPCS Code C9399 (Unclassified Drugs or Biologicals) From the C-APC Policy
Section 1833(t)(15) of the Act, as added by section 621(a)(1) of the Medicare Prescription Drug, Improvement, and Modernization Act of 2003 (Pub. L. 108-173), provides for payment under the OPPS for new drugs and biologicals until HCPCS codes are assigned. Under this provision, we are required to make payment for a covered outpatient drug or biological that is furnished as part of covered outpatient department services but for which a HCPCS code has not yet been assigned in an amount equal to 95 percent of average wholesale price (AWP) for the drug or biological. ( printed page 53469)
In the CY 2005 OPPS/ASC final rule with comment period (69 FR 65805), we implemented section 1833(t)(15) of the Act by instructing hospitals to bill for a drug or biological that is newly approved by the Food and Drug Administration (FDA) and that does not yet have a HCPCS code by reporting the National Drug Code (NDC) for the product along with the newly created HCPCS code C9399 (Unclassified drugs or biologicals). We explained that when HCPCS code C9399 appears on a claim, the Outpatient Code Editor (OCE) suspends the claim for manual pricing by the Medicare Administrative Contractor (MAC). The MAC prices the claim at 95 percent of the drug or biological's AWP, using Red Book or an equivalent recognized compendium, and processes the claim for payment. We emphasized that this approach enables hospitals to bill and receive payment for a new drug or biological concurrent with its approval by the FDA. The hospital does not have to wait for the next quarterly release or for approval of a product specific HCPCS code to receive payment for a newly approved drug or biological or to resubmit claims for adjustment. We instructed that hospitals would discontinue billing HCPCS code C9399 and the NDC upon implementation of a product specific HCPCS code, status indicator, and appropriate payment amount with the next quarterly update. We also note that HCPCS code C9399 is paid in a similar manner in the ASC setting, as 42 CFR 416.171(b) outlines that certain drugs and biologicals for which separate payment is allowed under the OPPS are considered covered ancillary services for which the OPPS payment rate, which is 95 percent of AWP for HCPCS code C9399, applies.
Since the implementation of the C-APC policy in 2015, payment for drugs and biologicals described by HCPCS code C9399 had been included in the C-APC payment when these products appear on a claim with a primary C-APC service. Packaging payment for these drugs and biologicals that appear on a hospital outpatient claim with a primary C-APC service is consistent with our C-APC packaging policy under which we make payment for all items and services, including all non-pass-through drugs, reported on the hospital outpatient claim as being integral, ancillary, supportive, dependent, and adjunctive to the primary service and representing components of a complete comprehensive service, with certain limited exceptions (78 FR 74869). It was our position that the total payment for the C-APC with which payment for a drug or biological described by HCPCS code C9399 is packaged includes payment for the drug or biological at 95 percent of its AWP.
However, we determined that in certain instances, drugs and biologicals described by HCPCS code C9399 are not being paid at 95 percent of their AWPs when payment for them is packaged with payment for a primary C-APC service. In order to ensure payment for new drugs and biologicals described by HCPCS code C9399 at 95 percent of their AWP, for CY 2023 and subsequent years, we finalized our proposal to exclude any drug or biological described by HCPCS code C9399 from packaging when the drug or biological is included on a claim with a “J1” service, which is the status indicator assigned to a C-APC, and a claim with a “J2” service, which is the status indicator assigned to comprehensive observation services. See Addendum J for the CY 2026 C-APC payment policy exclusions.
In the CY 2023 OPPS/ASC final rule with comment period, we finalized the proposal in section XI., “CY 2023 OPPS Payment Status and Comment Indicators”, to add a new definition to status indicator “A” to include unclassified drugs and biologicals that are reportable with HCPCS code C9399 (87 FR 72051). The current definition, as finalized in the CY 2023 OPPS/ASC final rule with comment period, can be found in Addendum D1, would ensure the MAC prices claims for drugs or biologicals billed with HCPCS code C9399 at 95 percent of the drug or biological's AWP and pays separately for the drug or biological under the OPPS when it appears on the same claim as a primary C-APC service.
(5) Exclusion of Cell and Gene Therapies From the C-APC Policy
As previously discussed in this section, and in the CY 2014 OPPS/ASC final rule with comment period (78 FR 74865), the C-APC policy packages payment for items and services that are typically integral, ancillary, supportive, dependent, or adjunctive to the primary service and provided during the delivery of the comprehensive service, including diagnostic procedures, laboratory tests and other diagnostic tests and treatments that assist in the delivery of the primary procedure. In the CY 2014 OPPS/ASC final rule with comment period (78 FR 74861), we finalized defining a comprehensive APC as a classification for the provision of a primary service and all adjunctive services provided to support the delivery of the primary service. Because a comprehensive APC treats all individually reported codes as representing components of the comprehensive service, we make a single prospective payment based on the cost of all individually reported codes that represent the provision of a primary service and all adjunctive services provided to support that delivery of the primary service.
As discussed in the CY 2025 OPPS/ASC proposed rule (89 FR 59201 through 59204), we generally treat all items and services reported on a C-APC claim as integral, ancillary, supportive, dependent, and adjunctive to the primary service and representing components of a comprehensive service. Historically, items packaged for payment provided in conjunction with the primary C-APC service also include all drugs, biologicals, and radiopharmaceuticals, regardless of cost, except those drugs with pass-through payment status and those drugs that are usually self-administered (SADs), unless they function as supplies (78 FR 74868 through 74869 and 74909).
However, we recognized in the CY 2025 OPPS/ASC proposed rule (89 FR 59201 through 59204) that there are rare instances in which cell and gene therapies appear on the same claim as a primary C-APC service and therefore, have their payment packaged with payment for the primary C-APC service. As stated in the CY 2025 OPPS/ASC final rule with comment period (89 FR 93932 through 93938), given the unique nature of these therapies, we do not believe they function as integral, ancillary, supportive, dependent, or adjunctive to any of the current primary C-APC services. Additionally, we stated that when these products are administered, they are the primary treatment being administered to a patient and thus, are not integral, ancillary, supportive, dependent, or adjunctive to any primary C-APC services.
Therefore, we finalized a policy for CY 2025 and subsequent years (89 FR 93932 through 93938), to not package payment for cell and gene therapies into C-APCs, when those cell and gene therapies are not functioning as integral, ancillary, supportive, dependent, or adjunctive to the primary C-APC service. For new cell and gene therapy products that are not integral, ancillary, supportive, dependent, or adjunctive to any C-APC primary service, we will continue to add their product specific HCPCS codes, when created, to the C-APC exclusion list. The proposed list of qualifying products can be found in Table 2.
( printed page 53470)We list all final C-APC exclusion categories for CY 2026 in Addendum J to this final rule with comment period (which is available via the internet on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices). Comments on our proposed exclusions are below.
Comment: Commenters generally supported and thanked CMS for proposing to continue the exclusion of Cell and Gene Therapies from C-APC packaging in order to support innovation and patient access.
Response: We thank commenters for their support.
Comment: A few commenters had suggestions on potential modifications and expansions for this policy. For example, one commenter suggested excluding drugs that are the primary therapy and exceed a cost threshold that aligns with the drug's cost relative to the total C-APC claim cost from comprehensive packaging. Bladder cancer drugs were one example suggested for exclusion as C-APC packaging of their product would be cost prohibitive.
Response: We thank commenters for their feedback, analysis, and recommendations on potential future approaches for structuring C-APC payment. We are not expanding our C-APC exclusion policy at this time to include additional classes of drugs, but we will take this information into consideration for future rulemaking.
Comment: A few commenters asked for CMS to add HCPCS code Q2056 (Ciltacabtagene autoleucel, up to 100 million autologous b-cell maturation antigen (bcma) directed car-positive t cells, including leukapheresis and dose preparation procedures, per therapeutic dose) to this list of Cell and Gene Therapies excluded from C-APC packaging for CY 2026. Commenters noted that this product's pass-through status expired June 30, 2025, and that it was previously indicated as a cell and gene therapy that would be excluded from C-APC packaging. Several commenters asked CMS to be vigilant with adding new products as they are approved and to introduce a formal process for the public to alert CMS that there is a new cell and gene therapy HCPCS code that should be excluded from payment.
Response: We thank commenters for recommending the addition of HCPCS code Q2056 to the cell and gene therapy C-APC exclusion list. This HCPCS code has been added to the table of cell and gene therapies excluded from C-APC packaging for CY 2026. We want to clarify for commenters, that although HCPCS code Q2056 was omitted from the CY 2026 OPPS/ASC proposed rule table, the code was excluded from C-APC packaging effective July 1, 2025, ( printed page 53471) after its drug pass-through status expired. Per our finalized policy in the CY 2025 OPPS/ASC final rule with comment period (89 FR 93932 through 93938), for new cell and gene therapy products that are not integral, ancillary, supportive, dependent, or adjunctive to any C-APC primary service, we will continue to add their product specific HCPCS codes, when created, to the C-APC exclusion list. We review products that are updated through the quarterly process to determine if there are qualifying cell and gene therapies that should be excluded from C-APC packaging. We welcome readers to contact us if they have a suggestion of a new qualifying cell and gene therapy that should be excluded from C-APC packaging.
We note that we did not make a proposal to alter the substance of the overall policy excluding cell and gene therapies from the C-APC packaging; consistent with public comments received, we are continuing this policy for CY 2026. In response to comments, the finalized list of qualifying products can be found in Table 3 consistent with our finalized policy in the CY 2025 OPPS/ASC final rule with comment period (89 FR 93932 through 93938).
(6) Exclusion of Non-Opioid Products for Pain Relief Under Section 4135 of the Consolidated Appropriations Act, 2023 From the C-APC Policy
The Consolidated Appropriations Act (CAA), 2023 (Pub. L. 117-328), was signed into law on December 29, 2022. Section 4135(a) and (b) of the CAA, 2023, titled “Access to Non-Opioid Treatments for Pain Relief,” amended section 1833(t)(16) and section 1833(i) of the Social Security Act, respectively, to provide for temporary additional payments for non-opioid treatments for pain relief (as that term is defined in section 1833(t)(16)(G)(i) of the Act). In particular, section 1833(t)(16)(G) provides that with respect to a non-opioid treatment for pain relief furnished on or after January 1, 2025, ( printed page 53472) and before January 1, 2028, the Secretary shall not package payment for the non-opioid treatment for pain relief into payment for a covered OPD service (or group of services) and shall make an additional payment for the non-opioid treatment for pain relief as specified in clause (ii) of that section. Clauses (ii) and (iii) of section 1833(t)(16)(G) of the Act provide for the amount of additional payment and set a limitation on that amount. As stated earlier in this section, our current policy is to exclude from the packaged C-APC payment those items and services that are required by statute to be separately paid.
Accordingly, in the CY 2025 OPPS/ASC final rule with comment period, we finalized a policy to exclude the non-opioid treatments for pain relief identified as satisfying the required criteria for payment under section 4135 of the CAA, 2023 from the C-APC policy to ensure payment is not packaged into any C-APC and that separate payment is made in accordance with the statute (89 FR 93938 through 93939).
(7) C-APCs for CY 2026
For CY 2026 and subsequent years, we proposed to continue to apply the C-APC payment policy methodology. We refer readers to the CY 2017 OPPS/ASC final rule with comment period (81 FR 79583) for a discussion of the C-APC payment policy methodology and revisions.
Each year, in accordance with section 1833(t)(9)(A) of the Act, we review and revise the services within each APC group and the APC assignments under the OPPS. As a result of our annual review of the services and the APC assignments under the OPPS, we did not propose to convert any standard APCs to C-APCs in CY 2026; thus, we proposed that the number of C-APCs for CY 2026 would be the same as the number for CY 2025, which is 72 C-APCs.
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: A few commenters requested that CMS reconsider our packaging policies for C-APC 8011 (Comprehensive Observation Services). They requested that CMS remove the rule that the presence of a SI “T” [6] procedure on a claim excludes payment of C-APC 8011 and instead package the payment of the SI “T” procedure into C-APC 8011, as is already done with SI “Q” [7] procedures. Commenters stated that this requirement violates the basic tenet of the packaging concept in that when observation services are ordered and furnished, the observation services become the primary service provided to such patients and the SI “T” procedure is provided ancillary to that primary service. Commenters cited scenarios in which hospitals provide significant, resource-intensive services to a patient but are paid significantly less than if a SI “T” procedure was not done.
Response: We thank the commenters for bringing this to our attention. In the CY 2016 OPPS/ASC final rule with comment period (80 FR 70334 through70336), in response to commenters' concerns regarding packaging payment for potentially high-cost surgical procedures into the payment for an observation C-APC, we finalized a policy that claims reporting procedures assigned status indicator “T” do not qualify for payment through C-APC 8011, regardless of whether the procedure assigned status indicator “T” was furnished before or after observation services (described by HCPCS code G0378) were provided. In the CY 2017 OPPS/ASC final rule with comment period (81 FR 79562), we stated that services that would otherwise qualify for C-APC 8011 are not considered to be observation services when they are associated with a surgical procedure (assigned to status indicator “T”). Instead, they are considered to be perioperative recovery, which is always packaged in with the surgical procedure (81 FR 79583). We will continue to review the impacts of this issue and may revisit it in future rulemaking.
Comment: Some commenters expressed concerns with the C-APC methodology for surgical insertion codes for brachytherapy treatment, stating that these concerns impact beneficiary access to brachytherapy in the HOPD setting. These commenters stated that the C-APC methodology lacks the appropriate charge capture mechanisms to accurately reflect the services associated with the C-APC, that there are significant variations in the clinical practice and billing patterns in the hospital claims data used for ratesetting, and that the C-APC rates do not accurately or fully reflect the services and costs associated with the primary procedure. Commenters urged the agency to explore alternatives, including that CMS discontinue the C-APC policy for all brachytherapy insertion codes. Alternatively, one commenter suggested that CMS could continue to pay for “J1” brachytherapy insertion codes under the C-APC payment methodology but exclude and make separate payment for designated preparation and planning services in addition to the C-APC payment. Another commenter called for education on whether services, like brachytherapy, that are assigned to a “J1” indicators and delivered over multiple patient encounters may be reported per encounter.
Response: We appreciate the comments on the C-APC methodology. However, we believe that the current C-APC methodology is appropriately applied to surgical insertion for Brachytherapy treatment and is accurately capturing costs, particularly as the brachytherapy sources used for these procedures are excluded from C-APC packaging and are separately payable. We will evaluate if provider education may be appropriate in this circumstance. We will continue to examine these concerns and will determine if any modifications to this policy are warranted in future rulemaking.
After consideration of the public comments we received, we are finalizing the C-APCs as proposed. Table 4 lists the final C-APCs for CY 2026. All C-APCs are displayed in Addendum J to this CY 2026 OPPS/ASC final rule with comment period (which is available via the internet on the CMS website). Addendum J to this final rule with comment period also contains all the data related to the C-APC payment policy methodology, including the list of complexity adjustments and other information for CY 2026.
( printed page 53473) ( printed page 53474)c. Calculation of Composite APC Criteria-Based Costs
As discussed in the CY 2008 OPPS/ASC final rule with comment period (72 FR 66613), we believe it is important that the OPPS enhance incentives for hospitals to provide necessary, high-quality care as efficiently as possible. For CY 2008, we developed composite APCs to provide a single payment for groups of services that are typically performed together during a single clinical encounter and that result in the provision of a complete service. Combining payment for multiple, independent services into a single OPPS payment in this way enables hospitals ( printed page 53475) to manage their resources with maximum flexibility by monitoring and adjusting the volume and efficiency of services themselves. An additional advantage to the composite APC model is that we can use data from correctly coded multiple procedure claims to calculate payment rates for the specified combinations of services, rather than relying upon single procedure claims which may be low in volume and/or incorrectly coded. Under the OPPS, we currently have composite policies for mental health services and multiple imaging services. We refer readers to the CY 2008 OPPS/ASC final rule with comment period (72 FR 66611 through 66614 and 66650 through 66652) for a full discussion of the development of the composite APC methodology, and the CY 2012 OPPS/ASC final rule with comment period (76 FR 74163) and the CY 2018 OPPS/ASC final rule with comment period (82 FR 59241, 59242, and 59246 through 52950) for further background.
(1) Mental Health Services Composite APC
For CY 2026, we proposed to continue our longstanding policy of limiting the aggregate payment for specified less resource-intensive mental health services furnished on the same date to the payment for a day of partial hospitalization services provided by a hospital, which we consider to be the most resource-intensive of all outpatient mental health services (88 FR 49572). We refer readers to the April 7, 2000, OPPS final rule with comment period (65 FR 18452 through 18455) for the initial discussion of this longstanding policy and the CY 2012 OPPS/ASC final rule with comment period (76 FR 74168) for further background.
In the CY 2018 OPPS/ASC proposed rule and final rule with comment period (82 FR 33580 and 33581 and 82 FR 59246 and 59247), we proposed and finalized the policy for CY 2018 and subsequent years that, when the aggregate payment for specified mental health services provided by one hospital to a single beneficiary on a single date of service, based on the payment rates associated with the APCs for the individual services, exceeds the maximum per diem payment rate for partial hospitalization services provided by a hospital, those specified mental health services will be paid through composite APC 8010 (Mental Health Services Composite). In addition, we set the payment rate for composite APC 8010 for CY 2018 at the same payment rate for APC 5863, which was the maximum partial hospitalization per diem payment rate for a hospital, and finalized a policy that the hospital would continue to be paid the payment rate for composite APC 8010. This policy applied in CYs 2018 through 2023.
In the CY 2024 OPPS/ASC proposed rule, we stated that APC 5863 was no longer the maximum partial hospitalization per diem payment rate for a hospital due to the creation of APC 5864, which is four or more hospital-based PHP services per day (88 FR 49572). We solicited comment on whether APC 5864 would be appropriate to use as the daily mental health cap, as we have historically set the daily mental health cap for composite APC 8010 at the maximum partial hospitalization per diem payment rate for a hospital (88 FR 49572). Based on public comments received and our longstanding policy, in the CY 2024 OPPS/ASC final rule, we finalized APC 5864, four hospital-based PHP services per day, as the daily mental health cap (88 FR 81566).
In the CY 2026 OPPS/ASC proposed rule, we stated that we continue to believe that the costs associated with administering a partial hospitalization program represent the most resource intensive of all outpatient mental health services. For CY 2026 and subsequent years, we proposed to continue this policy that when the aggregate payment for specified mental health services provided by one hospital to a single beneficiary on a single date of service, based on the payment rates associated with the APCs for the individual services, exceeds the per diem payment rate for four partial hospitalization services provided in a day by a hospital (the payment amount for APC 5864), those specified mental health services would be paid through composite APC 8010. In addition, we proposed to continue to set the payment rate for composite APC 8010 at the same payment rate that we proposed for APC 5864, which is a partial hospitalization per diem payment rate for four partial hospitalization services furnished in a day by a hospital.
Under the proposed policy, the Integrated OCE (I/OCE) would continue to determine whether to pay for these specified mental health services individually, or to make a single payment at the same payment rate established for APC 5864 for all the specified mental health services furnished by the hospital on that single date of service by paying for the services through composite APC 5863.
We did not receive public comments on this provision, and therefore, we are finalizing our proposal regarding APC 8010 without modification. When the aggregate payment for specified mental health services provided by one hospital to a single beneficiary on a single date of service, based on the payment rates associated with the APCs for the individual services, exceeds the maximum per diem payment rate for four partial hospitalization services provided in a day by a hospital (the payment amount for APC 5864), those specified mental health services would be paid through composite APC 8010 for CY 2026. In addition, we are finalizing setting the payment rate for composite APC 8010 for CY 2026 at the same payment rate that we set for APC 5864, which is the maximum partial hospitalization per diem payment rate for a hospital.
(2) Multiple Imaging Composite APCs (APCs 8004, 8005, 8006, 8007, and 8008)
Effective January 1, 2009, we provide a single payment each time a hospital submits a claim for more than one imaging procedure within an imaging family on the same date of service, to reflect and promote the efficiencies hospitals can achieve when performing multiple imaging procedures during a single session (73 FR 41448 through 41450). We utilize three imaging families based on imaging modality for purposes of this methodology: (1) ultrasound; (2) computed tomography (CT) and computed tomographic angiography (CTA); and (3) magnetic resonance imaging (MRI) and magnetic resonance angiography (MRA). The HCPCS codes subject to the multiple imaging composite policy and their respective families are listed in Table 5.
While there are three imaging families, there are five multiple imaging composite APCs due to the statutory requirement under section 1833(t)(2)(G) of the Act that we differentiate payment for OPPS imaging services provided with and without contrast. While the ultrasound procedures included under the policy do not involve contrast, both CT/CTA and MRI/MRA scans can be provided either with or without contrast. The five multiple imaging composite APCs established in CY 2009 are:
- APC 8004 (Ultrasound Composite);
- APC 8005 (CT and CTA without Contrast Composite);
- APC 8006 (CT and CTA with Contrast Composite);
- APC 8007 (MRI and MRA without Contrast Composite); and
- APC 8008 (MRI and MRA with Contrast Composite).
We define the single imaging session for the “with contrast” composite APCs as having at least one or more imaging ( printed page 53476) procedures from the same family performed with contrast on the same date of service. For example, if the hospital performs an MRI without contrast during the same session as at least one other MRI with contrast, the hospital will receive payment based on the payment rate for APC 8008, the “with contrast” composite APC.
We make a single payment for those imaging procedures that qualify for payment based on the composite APC payment rate, which includes any packaged services furnished on the same date of service. The standard (noncomposite) APC assignments continue to apply for single imaging procedures and multiple imaging procedures performed across families. For a full discussion of the development of the multiple imaging composite APC methodology, we refer readers to the CY 2009 OPPS/ASC final rule with comment period (73 FR 68559 through 68569).
For CY 2026, we proposed to continue to pay for all multiple imaging procedures within an imaging family performed on the same date of service using the multiple imaging composite APC payment methodology. In the CY 2026 OPPS/ASC proposed rule, we stated that we continue to believe that this policy would reflect and promote the efficiencies hospitals can achieve when performing multiple imaging procedures during a single session.
For CY 2026, except where otherwise indicated, we proposed to use the costs derived from CY 2024 claims data to set the proposed CY 2026 payment rates. Therefore, for CY 2026, the proposed payment rates for the five multiple imaging composite APCs (APCs 8004, 8005, 8006, 8007, and 8008) were based on proposed geometric mean costs calculated from CY 2024 claims available for the CY 2026 OPPS/ASC proposed rule that qualify for composite payment under the current policy (that is, those claims reporting more than one procedure within the same family on a single date of service). To calculate the proposed geometric mean costs, we used the same methodology that we used to calculate the geometric mean costs for these composite APCs since CY 2014, as described in the CY 2014 OPPS/ASC final rule with comment period (78 FR 74918). The imaging HCPCS codes referred to as “overlap bypass codes” that we removed from the bypass list for purposes of calculating the proposed multiple imaging composite APC geometric mean costs, in accordance with our established methodology as stated in the CY 2014 OPPS/ASC final rule with comment period (78 FR 74918), are identified by asterisks in Addendum N to this final rule with comment period (which is available via the internet on the CMS website https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices) and are discussed in more detail in section II.A.1.a. of this final rule with comment period.
We did not receive any public comments on this policy. We are finalizing without modification our proposal to continue the use of multiple imaging composite APCs to pay for the provision of more than one imaging procedure from the same imaging family on the same date. Table 5 lists the final HCPCS codes that would be subject to the multiple imaging composite APC policy and their respective families and approximate composite APC final geometric mean costs for CY 2026.
( printed page 53477) ( printed page 53478) ( printed page 53479) ( printed page 53480) ( printed page 53481)3. Changes to Packaged Items and Services
a. Background and Rationale for Packaging in the OPPS
Like other prospective payment systems, the OPPS relies on the concept of averaging to establish a payment rate for services. The payment may be more or less than the estimated cost of providing a specific service or a bundle of specific services for a particular beneficiary. The OPPS packages payments for multiple interrelated items and services into a single payment to create incentives for hospitals to furnish services most efficiently and to manage their resources with maximum flexibility. Our packaging policies support our strategic goal of using larger payment bundles in the OPPS to maximize hospitals' incentives to provide care in the most efficient manner. For example, where there are a variety of devices, drugs, items, and supplies that could be used to furnish a service, some of which are more costly than others, packaging encourages hospitals to use the most cost-efficient item that meets the patient's needs, rather than to routinely use a more expensive item, which may occur if separate payment is provided for the item.
Packaging also encourages hospitals to effectively negotiate with manufacturers and suppliers to reduce the purchase price of items and services or to explore alternative group purchasing arrangements, thereby encouraging the most economical health care delivery. Similarly, packaging encourages hospitals to establish protocols that ensure that necessary services are furnished, while scrutinizing the services ordered by practitioners to maximize the efficient use of hospital resources. Packaging payments into larger payment bundles promotes the predictability and accuracy of payment for services over time. Finally, packaging may reduce the importance of refining service-specific payments because packaged payments include costs associated with higher cost cases requiring many ancillary items and services and lower cost cases requiring fewer ancillary items and services. Packaging encourages efficiency and is an essential component of a prospective payment system; therefore, packaging payments for items and services that are typically integral, ancillary, supportive, dependent, or adjunctive to a primary service has been a fundamental part of the OPPS since its implementation in August 2000. As we continue to develop larger payment groups that more broadly reflect services provided in an encounter or episode of care, we have expanded the OPPS packaging policies. Most, but not necessarily all, categories of items and services currently packaged in the OPPS are listed in 42 CFR 419.2(b). Our overarching goal is to make payments for all services under the OPPS more consistent with those of a prospective payment system and less like those of a per-service fee schedule, which pays separately for each coded item. As a part of this effort, we have continued to examine the payment for items and services provided under the OPPS to determine which OPPS services can be packaged to further achieve the objective of advancing the OPPS toward a more prospective payment system.
b. Final CY 2026 Policy on Packaged Items and Services
For CY 2026, we examined the items and services currently provided under the OPPS, reviewing categories of integral, ancillary, supportive, dependent, or adjunctive items and services for which we believe payment would be appropriately packaged into payment for the primary service that they support. Specifically, we examined the HCPCS code definitions (including CPT code descriptors) and hospital outpatient department billing patterns to determine whether there were categories of codes for which packaging would be appropriate according to existing OPPS packaging policies or a logical expansion of those existing OPPS packaging policies.
For CY 2026, we did not propose any changes to the overall packaging policy discussed. We proposed to continue to conditionally package the costs of selected newly identified ancillary services into payment for a primary service where we believe that the packaged item or service is integral, ancillary, supportive, dependent, or adjunctive to the provision of care that was reported by the primary service HCPCS code (90 FR 33503).
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: Several commenters expressed broad support for unpackaging payments. One commenter believed that CMS packaging policies may encourage efficiencies and help lower costs, but they believed that packaging policies could penalize hospitals that provide complex care to sicker patients. Similarly, commenters believed that unpackaging payment could support patient access and innovations, including to certain drugs, biologicals, and services. Specifically, one commenter suggested that CMS consider unpackaging their product, a contrast agent, and believed CMS's reasoning in the CY 2026 OPPS/ASC proposed rule for unpackaging and paying separately for diagnostic radiopharmaceuticals applies equally or even more to their product, since the cost of their product is over 500 times greater than the amount reported for the policy packaged drugs offset associated with the Level II Urology APC. The commenter believed lack of separate payment was a barrier to beneficiary access and recommended CMS pay for products like theirs when the product costs exceeded a certain threshold.
Response: We thank the commenters for their perspectives on packaging within the OPPS, including specific examples of cost exceeding offset amounts. We continue to believe that our packaging policies are a fundamental principle that distinguishes a prospective payment system from a fee schedule. In general, packaging the costs of supportive items and services into the payment for the primary procedure or service with which they are associated encourages hospital efficiencies and enables hospitals to manage their resources with maximum flexibility. We will take the information commenters provided into consideration as appropriate for possible future rulemaking.
Comment: Several commenters recommended CMS reassess its policy packaging principles regarding laboratory testing, with a particular emphasis on screening tests and antimicrobial stewardship, including those tests used in the emergency department setting. These commenters explained the public health threat of antibiotic-resistant infections, including the patient and financial impacts. Specifically, these commenters discussed that current APC assignments do not reflect substantial investments in the reagents, instruments, and analytic software that are required for these tests. Therefore, they requested CMS exclude these products from packaging through a narrowly defined exception, similar to preventative services.
Response: We thank the commenters for their feedback on these issues, including the importance of antimicrobial stewardship. We note that these costs are generally accounted for through packaging under our policies outlined in 42 CFR 419.2(b). As previously discussed in this section, in general, packaging the costs of supportive items and services into the payment for the primary procedure or service with which they are associated encourages hospital efficiencies and ( printed page 53482) enables hospitals to manage their resources with maximum flexibility. Our overarching goal is to make payments for services under the OPPS more consistent with those of a prospective payment system and less like those of a per-service fee schedule, which pays separately for each coded item. At this time, we do not believe that unpackaging the tests as suggested by commenters helps us to achieve this goal. However, we will take these comments into consideration for any future modifications to our broader packaging policies.
Additionally, we received specific recommendations regarding C-APC packaging of Cell and Gene Therapies and associated products, which are addressed in section II.b.4. of this final rule with comment period, and the packaging of non-opioid treatments for pain relief, which are addressed in section XIII.F. of this final rule with comment period. Commenters also made recommendations on our packaging policies in the context of our diagnostic radiopharmaceutical proposal, which is discussed in the next section.
After consideration of public comments, we are finalizing our proposal to continue to conditionally package the costs of selected newly-identified ancillary services into payment for a primary service where we believe that the packaged item or service is integral, ancillary, supportive, dependent, or adjunctive to the provision of care that was reported by the primary service HCPCS code, as proposed for CY 2026.
c. Payment for Diagnostic Radiopharmaceuticals
(1) Background on OPPS Packaging Policy for Diagnostic Radiopharmaceuticals
Under the OPPS, we package several categories of nonpass-through drugs, biologicals, and radiopharmaceuticals, regardless of the cost of the products. Because the products are packaged according to the policies in § 419.2(b), we refer to them as “policy-packaged” drugs, biologicals, and radiopharmaceuticals. In particular, under § 419.2(b)(15), payment for drugs, biologicals, and, prior to CY 2025, all radiopharmaceuticals that function as supplies when used in a diagnostic test or procedure are packaged with the payment for the related procedure or service. Packaging costs into a single aggregate payment for a service, encounter, or episode of care is a fundamental principle that distinguishes a prospective payment system from a fee schedule. In general, packaging the costs of supportive items and services into the payment for the primary procedure or service with which they are associated encourages hospital efficiencies and enables hospitals to manage their resources with maximum flexibility.
In the CY 2008 OPPS/ASC final rule with comment period, we finalized the packaging status of diagnostic radiopharmaceuticals as part of our overall enhanced packaging approach for the CY 2008 OPPS and subsequent years (72 FR 66635 through 66641). Importantly, we noted that we believe diagnostic radiopharmaceuticals are always intended to be used with a diagnostic nuclear medicine procedure and function as supplies when used in a diagnostic test or procedure, making it appropriate to package the payment for the diagnostic radiopharmaceutical into the payment for the related nuclear medicine procedure. Higher cost diagnostic radiopharmaceuticals were one specific type of product that, prior to CY 2025, was policy packaged under the category described by § 419.2(b)(15). Since we implemented this policy in CY 2008, interested parties raised concerns regarding policy packaging of diagnostic radiopharmaceuticals.
In the CY 2025 OPPS/ASC proposed rule (89 FR 59213 through 59222), we stated that we continue to believe diagnostic radiopharmaceuticals are always intended to be used with a diagnostic nuclear medicine procedure and function as supplies when used in a diagnostic test or procedure, generally making it appropriate to package payment for them with payment for the related nuclear medicine procedure. However, we stated there are certain situations in which the packaged payment amount attributed to the diagnostic radiopharmaceutical used in an imaging procedure assigned to a nuclear medicine APC may not adequately account for the cost of a diagnostic radiopharmaceutical that has a significantly higher cost, but lower utilization relative to the other diagnostic radiopharmaceuticals that may be used with the procedure.
In the CY 2025 OPPS/ASC final rule with comment period (89 FR 93948 through 93963) we finalized a policy to pay separately for any diagnostic radiopharmaceutical with a per day cost greater than $630 for CY 2025. We proposed to use the same methodology that was finalized in the CY 2025 OPPS/ASC final rule with comment period in order to calculate the per day costs for diagnostic radiopharmaceuticals for CY 2026 and future years (89 FR 93953 through 93955). We noted that any diagnostic radiopharmaceutical with a per day cost at or below that threshold will continue to be policy packaged under our longstanding policy at § 419.2(b)(15). Additionally, we finalized the policy that starting in CY 2026 and for subsequent years, we will update the threshold amount of $630 by a forecast of the Producer Price Index (PPI) for Pharmaceuticals for Human Use, Prescription (Bureau of Labor Statistics (BLS) series code WPUSI07003) from IHS Global, Inc (IGI) (89 FR 93955).
In the CY 2025 OPPS/ASC final rule with comment period, we also finalized a policy to pay for nonpass-through, separately payable diagnostic radiopharmaceuticals with per day costs above the designated threshold based on our authority under section 1833(t)(14)(A)(iii)(II) of the Act. As we found that the ASP data we had was not usable for the purpose of paying for diagnostic radiopharmaceuticals, we finalized a policy to pay for qualifying nonpass-through diagnostic radiopharmaceuticals with claims data based on mean unit cost data derived from hospital claims. Additionally, we finalized corresponding modifications to the regulation text at § 419.2(b)(15) and § 419.41 to codify our finalized payment policy for diagnostic radiopharmaceuticals and our existing policy for therapeutic radiopharmaceuticals. For additional information regarding the policy finalized for CY 2025, reference 89 FR 93948 through 93963.
(2) Diagnostic Radiopharmaceutical Packaging Threshold
For CY 2026, we proposed to continue the policy finalized in CY 2025 (90 FR 33504). Specifically, we proposed to continue to calculate the per day cost of diagnostic radiopharmaceuticals based on the methodology described in section V.B.1.b. of the CY 2026 OPPS/ASC proposed rule, which relies on the methodology finalized in the CY 2006 OPPS final rule with comment period (70 FR 68636 through 68638).
As finalized in the CY 2025 OPPS/ASC final rule with comment period (89 FR 93955), starting in the OPPS/ASC rulemaking for CY 2026 and for subsequent years, we stated we would update the proposed threshold amount of $630 by a forecast of the PPI for Pharmaceuticals for Human Use, Prescription (BLS series code WPUSI07003) from IHS Global, Inc (IGI) by using most recently available four-quarter moving average PPI levels to trend from the third quarter of the year 2 years prior to the applicable calendar year to the third quarter of the year prior ( printed page 53483) to the applicable calendar year (for example, from the third quarter of 2024 to the third quarter of 2025 for CY 2026). We proposed a technical refinement to this policy. We proposed to use the most recently available four-quarter moving average PPI levels to trend the CY 2025 final threshold forward from the third quarter of the CY 2025 to the third quarter of the payment year (CY 2026) and round the resulting dollar amount to the nearest $5 increment. We believed using the most recently available four-quarter moving average PPI levels more appropriately updates the packaging threshold from CY 2025 for payment in CY 2026. For CY 2027 and subsequent updates, we proposed to trend the CY 2025 threshold of $630 forward using the four-quarter moving average PPI levels for Pharmaceuticals for Human Use, Prescription for CY 2025 (third quarter) forward using the PPI for Pharmaceuticals for Human Use, Prescription for the applicable payment year (third quarter) (90 FR 3362324). This is the same as the update factor used for the OPPS drug packaging threshold, where we originally used the four-quarter moving average PPI levels for Pharmaceutical Preparations, Prescription (BLS series code WPUSI07003, formerly BLS series code 32541DRX) to trend the $50 threshold forward from the third quarter of CY 2005 (when the Pub. L. 108-173 mandated threshold became effective) to the third quarter of the applicable payment year (71 FR 68085 and 68086).
Therefore, for CY 2026, we proposed to update the CY 2025 $630 threshold amount by the four-quarter moving average PPI levels for Pharmaceuticals for Human Use, Prescription to trend the $630 threshold forward. Specifically, we proposed to use the most recently available forecast of the four-quarter moving average PPI levels for Pharmaceutical for Human Use, Prescription from the third quarter of 2025 to the third-quarter of 2026, and to round the resulting dollar amount to the nearest $5 increment. Based on this methodology, we trended the $630 threshold forward and rounded the resulting dollar amount ($654.23) to the nearest $5 increment, which yields a proposed figure of $655 per day for CY 2026. Consistent with our methodology and practices listed in section V.B.1.b. of the CY 2026 OPPS/ASC proposed rule, we also proposed that if more recent data are subsequently available (for example, a more recent estimate of the PPI for Pharmaceuticals for Human Use, Prescription), we would use such data, if appropriate, to determine the CY 2026 diagnostic radiopharmaceutical packaging threshold in the final rule.
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: Most commenters were supportive of our proposal to maintain a per day cost threshold in order to determine which diagnostic radiopharmaceuticals should be paid separately under this policy. In general, commenters believed this threshold would help distinguish between older, lower-cost diagnostic radiopharmaceuticals and newer, higher-cost precision diagnostic radiopharmaceuticals. Similarly, most commenters were supportive of the proposed update methodology and the corresponding updated per day cost threshold of $655.
Response: We thank commenters for their support.
Comment: Some commenters offered feedback on the threshold CMS proposed for CY 2026. Specifically, a few commenters requested that CMS maintain the CY 2025 per day cost threshold of $630 and not update the cost threshold according to the proposed methodology for CY 2026. These commenters believed the policy was too new to warrant an increase in the payment threshold, and commenters noted that these products had unique cost structures, utilization patterns, and roles in patient care. Some requested CMS delay any changes in the threshold until the policy has been in place for at least 2 years. Some commenters were not convinced that the proposed update factor is appropriate or representative of diagnostic radiopharmaceuticals, but generally did not provide an alternative update methodology. Some commenters believed that incorporating radiopharmaceutical-specific cost data would be more appropriate. A commenter also recommended CMS ensure no unintended consequences of this policy occur, such as manufacturers purposefully pricing their products just above the payment threshold.
Response: We thank commenters for their feedback. We will monitor the effects of this policy and will consider proposing modifications in future rulemaking if appropriate. We do not believe it is appropriate to maintain the same threshold that was finalized in CY 2025. We continue to believe it is appropriate to subject the diagnostic radiopharmaceutical packaging threshold to the same update factor that is used for the OPPS drug packaging threshold as supported by the majority of commenters. Updating the threshold by the PPI for Pharmaceuticals for Human Use (Prescription) is consistent with our longstanding policy to update the OPPS drug packaging threshold annually. This PPI update factor provides aggregate changes in the selling prices of pharmaceuticals, which makes it an appropriate factor with which to update the diagnostic radiopharmaceutical packaging threshold to ensure that as diagnostic radiopharmaceuticals' costs change over time, the threshold continues to identify products with costs that significantly exceed the otherwise applicable APC payment amounts as determined in this final rule with comment period and that therefore should be eligible for separate payment. We appreciate the recommendation to consider an update factor more specific to diagnostic radiopharmaceuticals, which we will consider for future rulemaking.
Comment: A commenter requested that CMS consider unpackaging all radiopharmaceuticals regardless of their cost to ensure proper payment and avoid perverse incentives. They believed that this action would lead to an overall reduction in industry costs by eliminating the incentive for manufacturers to price products above the threshold.
Response: We thank the commenter for their concern regarding the avoidance of perverse financial incentives. The threshold amount was originally designed to ensure payment only for those products with costs that significantly exceed their packaged payment. We continue to believe a threshold is an appropriate method to ensure targeted payment as it continues the packaging of most diagnostic radiopharmaceuticals. As previously mentioned in this section, packaging is a fundamental principle that distinguishes a prospective payment system from a fee schedule. In general, packaging the costs of supportive items and services into the payment for the primary procedure or service with which they are associated encourages hospital efficiencies and enables hospitals to manage their resources with maximum flexibility. However, we will continue to monitor this policy for any unintended consequences.
After consideration of public comments, we are finalizing our policy as proposed. We are finalizing our proposal to update the CY 2025 $630 threshold amount by the four-quarter moving average PPI levels for Pharmaceuticals for Human Use, Prescription to trend the threshold forward. Specifically, we are using the most recently available forecast of the four-quarter moving average PPI levels for Pharmaceutical for Human Use, ( printed page 53484) Prescription from the third quarter of 2025 to the third quarter of 2026, and to round the resulting dollar amount to the nearest $5 increment.
We also proposed, and are now finalizing, a policy that if more recent data were to subsequently become available (for example, a more recent estimate of the PPI for Pharmaceuticals for Human Use, Prescription), we would use such data, if appropriate, to determine the CY 2026 diagnostic radiopharmaceutical packaging threshold in the final rule. Based on this methodology, using the most recent data available for this final rule with comment period, we trended the $630 threshold forward and rounded the resulting dollar amount ($656.65) to the nearest $5 increment, which yields a final diagnostic radiopharmaceutical packaging threshold figure of $655 per day for CY 2026.
(3) Amount of Separate Payment for Diagnostic Radiopharmaceuticals Exceeding the Threshold
As discussed in the CY 2025 OPPS/ASC final rule with comment period (89 FR 93955 through 93959), once we determine that the per day cost of a nonpass-through diagnostic radiopharmaceutical exceeds the cost threshold, proposed to be $655 per day for CY 2026, we will then assign that radiopharmaceutical to an APC, making it a specified covered outpatient drug (SCOD) per section 1833(t)(14)(B) of the Act. We proposed to continue our current policy for CY 2026, and proposed to pay for those nonpass-through, separately payable diagnostic radiopharmaceuticals based on our authority under section 1833(t)(14)(A)(iii)(II) of the Act. While, under this authority, we would ordinarily use the ASP methodology under section 1847A of the Act, we continued to find that the ASP data we had was not usable for payment purposes. We continued to believe that arithmetic mean unit cost (MUC) would be an appropriate proxy for the average price for a diagnostic radiopharmaceutical for a given year, as it is calculated based on the average costs for a particular year and is directly reflective of the actual cost data that hospitals submit to CMS. Therefore, we proposed to continue our current policy and proposed for CY 2026 to pay for qualifying diagnostic radiopharmaceuticals with per day costs above the diagnostic radiopharmaceutical packaging threshold based on their arithmetic MUC, which would be derived from calendar year 2024 claims data.
Although we proposed to base payment for qualifying radiopharmaceuticals on their arithmetic MUC for CY 2026, we continued to encourage manufacturers to submit ASP information for diagnostic radiopharmaceuticals, if possible. While we proposed to continue to use MUC to pay for separately payable diagnostic radiopharmaceuticals in CY 2026, we noted that manufacturers can begin, or continue, to report ASP data for potential future use in paying for diagnostic radiopharmaceuticals. For CY 2026, ASP reporting is voluntary for diagnostic radiopharmaceuticals paid under the OPPS. We encouraged interested parties to submit comments regarding potential issues that may arise that prevent appropriate ASP reporting for diagnostic radiopharmaceuticals. We referred readers to the CY 2025 OPPS/ASC final rule with comment period as it discusses some of the known concerns regarding ASP reporting for diagnostic radiopharmaceuticals (89 FR 93948 through 93963). We reiterated our stance from the CY 2025 OPPS/ASC final rule with comment period, that if we were to use average sales price as the basis of calculating a payment, we believed there must be more consistent, validated, and universal reporting in order for ASP to be a viable payment methodology (89 FR 93961).
We also reiterated, as we stated in the CY 2025 OPPS/ASC final rule with comment period (89 FR 93957), that there could be potential value in the use of ASP data for payment purposes for diagnostic radiopharmaceuticals when reported correctly and by all manufacturers who manufacture a product that is described by a given HCPCS code. We continue to believe that the use of ASP information for OPPS payment could provide an opportunity to improve payment accuracy for separately payable diagnostic radiopharmaceuticals by applying an established methodology that has already been successfully implemented under the OPPS for other separately payable drugs and biologicals, as well as for therapeutic radiopharmaceuticals.
To facilitate potential future payment for diagnostic radiopharmaceuticals based on ASP, we sought comment from interested parties on how CMS can ensure more consistent, validated, and universal reporting in order for ASP to be a viable payment methodology utilized in future rulemaking. For example, we sought comment on how CMS may update its past guidance, Submission of OPPS ASP Data for Nonpass-Through Separately Payable Therapeutic Radiopharmaceuticals and Radiopharmaceuticals with Pass-Through Status,[8] to reflect current clinical practices and to reflect ASP reporting for diagnostic radiopharmaceuticals.
Additionally, as discussed in section V.B.5. of the CY 2026 OPPS/ASC proposed rule (Proposed Payment for Nonpass-Through Drugs, Biologicals, and Radiopharmaceuticals with HCPCS Codes but Without OPPS Hospital Claims Data), we proposed to set the payment rate for new diagnostic radiopharmaceuticals that exceed the diagnostic radiopharmaceutical packaging threshold and with HCPCS codes, but which do not have pass-through status and are without claims data, at ASP plus 6 percent (90 FR 33624). If ASP data for these diagnostic radiopharmaceuticals were not available, we proposed to pay WAC plus 3 percent during the product's initial sales period, consistent with our policy described in section V.B.2. of the CY 2026 OPPS/ASC proposed rule. If the WAC also is unavailable, we proposed to make payment for new diagnostic radiopharmaceuticals at 95 percent of the products' most recent AWP. Following the initial sales period, a payment rate of WAC plus 6 percent would apply, if ASP data for these diagnostic radiopharmaceuticals remain unavailable. We believed the volume of products in this category would typically be very low; however, in these rare situations, we believed it would continue to be appropriate to use ASP plus six percent, WAC plus 3 or 6 percent, or 95 percent of AWP until a MUC is available. As we stated in the CY 2025 OPPS/ASC final rule with comment period, it is appropriate to use this payment hierarchy until a MUC is available. There is typically only one manufacturer for a diagnostic radiopharmaceutical that is new and described by a HCPCS code, but without claims data, so CMS does not have to ensure all manufacturers are reporting ASP for that particular HCPCS code prior to establishing a separate payment amount based on ASP. Additionally, although reporting of ASP is not a condition of CMS approving a HCPCS application, CMS has the opportunity to actively engage with the manufacturer, or sponsor of a HCPCS application, during the HCPCS application process. This allows for ongoing dialogue and education regarding the unique ASP reporting requirements that may be ( printed page 53485) associated with a particular product, including how to ensure the reported ASP aligns with the dose descriptor for the newly assigned HCPCS code (89 FR 93958). We believed the hierarchy previously specified is appropriate to determine the payment for a diagnostic radiopharmaceutical that is new and described by a HCPCS code, but without claims data, as it is consistent with the typical hierarchy associated with payment for drugs and biologicals paid under the OPPS as discussed in section V.A. and V.B. of the CY 2026 OPPS/ASC proposed rule.
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: Commenters were generally in favor of separate payment for diagnostic radiopharmaceuticals. Specifically, for diagnostic radiopharmaceuticals that have claims data, commenters appreciated that CMS is paying for these products separately, but recommended that CMS eventually transition away from an MUC-based payment methodology, which commenters generally viewed as an interim approach.
Commenters believe MUC is problematic because hospitals may inconsistently report diagnostic radiopharmaceutical units and costs, which ultimately impact the MUC calculation. Commenters provided suggestions to CMS on how to assess hospital claims data for appropriate submissions. Commenters also stated that many factors contribute to the inaccuracy of MUCs, including charge compression, varied revenue code use, cost-to-charge ratios, outdated data, and inconsistent cost center use by hospitals. These commenters provided data detailing that MUC is consistently lower for their products than ASP. Commenters highlighted how MUC may disadvantage their products. For example, one commenter stated that their product is under a competitive disadvantage while being paid at MUC compared to newer products that may be receiving separate ASP-based payment through pass-through payment status.
For these reasons, commenters suggested CMS transition to an average sales price (ASP) based payment, including for purposes of completing the per day cost calculations, for these diagnostic radiopharmaceuticals at the earliest point possible. Commenters stated that ASP is more reflective of the true cost of diagnostic radiopharmaceuticals compared to MUC. Commenters expressed their views on the benefits of ASP, including that ASP is updated quarterly, is more consistent, transparent, and aligned with payment for many other Part B drugs. Commenters stated that a portion of manufacturers are already reporting ASP, and many have recent experience reporting ASP for OPPS drug pass-through status. Some commenters suggested that diagnostic radiopharmaceuticals that currently report ASP, or have been paid based on ASP while on pass-through status, continue to receive ASP-based payment post pass-through status expiration. These commenters believed that an MUC-based payment methodology should only be used in instances where ASP is not reported. Other commenters recommended CMS consider WAC and AWP based payment methodologies.
Some commenters stated that if MUC is going to be used as the basis of payment for separately paid diagnostic radiopharmaceuticals, CMS should consider additional instruction to hospitals on reporting, or consider a low volume MUC policy or minimum claim volume to avoid fluctuations in payment due to low claims volume.
Broadly, commenters requested CMS provide additional clarity regarding why CMS proposed to continue to pay diagnostic radiopharmaceuticals at their arithmetic mean unit cost rather than ASP.
Response: We thank commenters for their support of our proposal to continue to pay diagnostic radiopharmaceuticals separately. We thank commenters for expressing their interest in use of ASP data and appropriate ASP reporting. As we have previously stated, and as commenters have noted, we recognize the complexities associated with reporting ASP for radiopharmaceuticals. We reiterate that we agree that there could be value in the use of ASP for determining separately paid diagnostic radiopharmaceutical payment amounts in the future. However, for CMS to use an ASP-based methodology to set payment rates for separately paid diagnostic radiopharmaceuticals, we believe there must be more consistent, validated, and universal reporting of ASP data for diagnostic radiopharmaceuticals. While commenters have communicated that they are currently, or will, report ASP, we still do not have universal reporting of ASP data to CMS for diagnostic radiopharmaceuticals. While the number of products reporting ASP has slightly increased, we continue to have concerns regarding the accuracy of the reported data. For several diagnostic radiopharmaceuticals that have reported their ASP, their reported ASP exceeds the calculated arithmetic MUCs by several thousand percent. Some of these discrepancies between reported ASP and MUC are so significant, that if we were to accept the ASPs as submitted and base payment for these diagnostic radiopharmaceuticals off the reported ASP, the result would be more than one diagnostic radiopharmaceutical accounting for billions of dollars in projected payment in CY 2026 and these diagnostic radiopharmaceuticals would be estimated to be among the top 10 highest paid HCPCS codes in all of the OPPS. Based on claims data, these extremely high ASP data appear to be erroneously reported. Based on this, coupled with the lack of universal reporting, among other factors, we continue to believe that an ASP-based methodology is not appropriate for setting CY 2026 payment for separately paid diagnostic radiopharmaceuticals.
We appreciate the insight from commenters regarding their concerns with CMS continuing to use arithmetic MUC as the payment methodology for diagnostic radiopharmaceuticals. We are taking these comments into consideration for future rulemaking.
We note that we rely on providers to accurately report the use of HCPCS codes in accordance with code descriptors and CPT and CMS instructions, to report services accurately on claims, and to report charges and costs accurately for the services on their Medicare hospital cost reports.
Regarding the suggestion to develop a low-volume MUC methodology when we calculate payment rates for diagnostic radiopharmaceuticals, we will take this suggestion under advisement for future notice and comment rulemaking. We did not propose to subject low volume diagnostic radiopharmaceutical APCs to the broader OPPS low volume policy, and we note that the low volume APC policy does not apply to APCs to which single drugs, biologicals, or radiopharmaceuticals are assigned, even if there is a low volume of claims for these items. We understand the commenter's concerns; however, we do not believe it would be appropriate to implement this policy modification without further engagement from interested parties.
Finally, in response to comments suggesting we adopt WAC or AWP based payment methodologies, we continue to believe that neither WAC nor AWP is an appropriate proxy to provide OPPS payment for average radiopharmaceutical acquisition costs and associated handling costs when manufacturers are not required to ( printed page 53486) submit ASP data. This is because payment based on WAC or AWP for separately payable drugs and biologicals is usually temporary for a calendar quarter until a manufacturer is able to submit the required ASP data in accordance with the quarterly ASP submission timeframes for reporting under section 1847A of the Act. WAC and AWP reported to compendia may not be reflective of a patient ready dose. The absence of an ASP reporting requirement and inappropriate or no reporting of ASP could result in payment for a separately payable diagnostic radiopharmaceutical based on WAC or AWP indefinitely, a result which we believe would be inappropriate, as these pricing metrics do not capture all of the pricing discounts that may be reflected in the ASP.
Comment: A few commenters question the legal validity of CMS using MUC as the basis of payment, and some of those commenters believe that radiopharmaceutical manufacturers are required to report ASP by statute. These commenters state that they believe CMS has the authority to require reporting and pay based on ASP.
Response: We proposed to pay for nonpass-through, separately payable diagnostic radiopharmaceuticals based on our authority under section 1833(t)(14)(A)(iii)(II) of the Act, and we continue to find this appropriate, as we find that the ASP data we have are not usable for the purpose of paying for diagnostic radiopharmaceuticals. Instead, we are paying based on mean unit cost data derived from hospital claims. We believe that paying for diagnostic radiopharmaceuticals using mean unit cost would appropriately pay for the average price of nonpass-through separately payable diagnostic radiopharmaceuticals for the applicable year. We believe MUC is an appropriate proxy for the average price for a diagnostic radiopharmaceutical for a given year, as it is calculated based on the average costs for a particular year and is directly reflective of the actual cost data that hospitals submit to CMS. We will continue to explore our authority regarding requiring ASP reporting for diagnostic radiopharmaceuticals under the OPPS.
Comment: Commenters broadly requested that CMS work collaboratively with manufacturers to identify and implement best practices for average sales price (ASP) data submission for radiopharmaceutical therapies. Some commenters stated that they look forward to working with CMS to identify and implement best practices for ASP data submission, and believed that clear guidance and streamlined reporting processes will help ensure that ASP data is complete and accurate, which supports a sustainable payment environment. A few commenters provided detailed recommendations and requests for how CMS may update our ASP reporting guidelines in the future for diagnostic radiopharmaceuticals.
Response: We thank commenters for their helpful feedback regarding future guidance on diagnostic radiopharmaceutical ASP reporting. We will take these comments under advisement for potential updated sub-regulatory guidance CMS intends to explore. We encourage interested parties to continue to engage with CMS on this issue.
After consideration of public comments, we are finalizing our proposal without modification. In summary, for CY 2026 we are paying separately for any diagnostic radiopharmaceutical with a per-day cost greater than $655 using the methodology described. Any diagnostic radiopharmaceutical with a per-day cost at or below that threshold would continue to be policy packaged under our longstanding policy codified at § 419.2(b)(15). We are finalizing our proposal to pay for those nonpass-through, separately payable diagnostic radiopharmaceuticals based on our authority under section 1833(t)(14)(A)(iii)(II) of the Act, and to pay for qualifying nonpass-through diagnostic radiopharmaceuticals with claims data based on mean unit cost data derived from hospital claims. As discussed in section V.B.5. of this final rule with comment period, we proposed and are finalizing a policy to set the payment rate for new diagnostic radiopharmaceuticals with HCPCS codes that exceed the diagnostic radiopharmaceutical packaging threshold, but which do not have pass-through status and are without claims data, at ASP plus 6 percent. If ASP data for these diagnostic radiopharmaceuticals were not available, we finalized a policy to pay WAC plus 3 percent during the product's initial sales period, consistent with our policy described in section V.B.2. of this CY 2026 OPPS/ASC final rule with comment period. If the WAC also is unavailable, we finalized a policy to make payment for new diagnostic radiopharmaceuticals at 95 percent of the products' most recent AWP. Following the initial sales period, a payment rate of WAC plus 6 percent would apply, if ASP data for these diagnostic radiopharmaceuticals remain unavailable.
(4) Qualifying Diagnostic Radiopharmaceuticals Above the Diagnostic Radiopharmaceutical Packaging Threshold
The HCPCS codes that describe diagnostic radiopharmaceuticals with per day costs that exceed the proposed diagnostic radiopharmaceutical packaging threshold were proposed to be assigned to a status indicator of “K”, indicating separate payment to be paid based on that HCPCS code's arithmetic MUC. A proposed APC and a proposed payment rate would be assigned as shown in Addendum B to the CY 2026 OPPS/ASC proposed rule. HCPCS codes that describe diagnostic radiopharmaceuticals with per day costs that are at or below the proposed diagnostic radiopharmaceutical packaging threshold were proposed to continue to be assigned to a status indicator of “N”, indicating packaged payment.
The proposed list of diagnostic radiopharmaceuticals that we calculated as having per day costs that exceeded $655 and their proposed status indicators can be found in Table 6 (originally set forth in Table 4 of the CY 2026 OPPS/ASC proposed rule at 90 FR 33506).
( printed page 53487)Proposed definitions of status indicators can be found in Addendum D1 to the CY 2026 OPPS/ASC proposed rule. Addenda to the CY 2026 OPPS/ASC proposed rule can be found on the CMS OPPS web page.
Based on the policy finalized in this section to pay separately at arithmetic MUC for any diagnostic radiopharmaceutical with a HCPCS code and claims data, and with a per-day cost greater than $655 using the methodology previously described, the HCPCS codes that describe diagnostic radiopharmaceuticals with per day costs that exceed the finalized diagnostic radiopharmaceutical packaging threshold are assigned to a status indicator of “K”, indicating separate payment to be paid based on that HCPCS code's arithmetic MUC. A final APC and payment rate are assigned as shown in Addendum B to this final rule with comment period. HCPCS codes that describe diagnostic radiopharmaceuticals with per day costs that are at or below the proposed diagnostic radiopharmaceutical packaging threshold are assigned to a status indicator of “N”, indicating packaged payment.
The finalized list of diagnostic radiopharmaceuticals that we calculated as having per day costs that exceed $655 and their proposed status indicators can be found in Table 7.
( printed page 53488)Finalized definitions of status indicators can be found in Addendum D1 to this final rule with comment period. Addenda to this rule can be found on the CMS OPPS web page.
4. Implementation of Section 4135 of the Consolidated Appropriations Act (CAA), 2023
The Consolidated Appropriations Act (CAA), 2023 (Pub. L. 117-328), was signed into law on December 29, 2022. Section 4135(a) and (b) of the CAA, 2023, titled Access to Non-Opioid Treatments for Pain Relief, amended sections 1833(t)(16) and 1833(i) of the Act, respectively, to provide for temporary additional payments for non-opioid treatments for pain relief (as that term is defined in section 1833(t)(16)(G)(i) of the Act). In particular, section 1833(t)(16)(G) of the Act provides that with respect to a non-opioid treatment for pain relief furnished on or after January 1, 2025, and before January 1, 2028, the Secretary shall not package payment for the non-opioid treatment for pain relief into payment for a covered OPD service (or group of services) and shall make an additional payment for the non-opioid treatment for pain relief as specified in clause (ii) of that section. Clauses (ii) and (iii) of section 1833(t)(16)(G) of the Act provide for the amount of additional payment and set a limitation on that amount, respectively.
The additional payments required under section 1833(t)(16)(G) of the Act began on January 1, 2025, based on the policy finalized in the CY 2025 OPPS/ ( printed page 53489) ASC final rule with comment period (89 FR 94343 through 94361). In section XIII.F. of the CY 2026 OPPS/ASC proposed rule (90 FR 33742 through 33749), we proposed to continue the policy finalized in the CY 2025 OPPS/ASC final rule with comment period for CY 2026. We also proposed non-opioid treatments for pain relief that would qualify under this policy for CY 2026 and sought public comment on those product evaluations.
We refer readers to section XIII.F. of this final rule with comment period for a summary of comments received on this proposal, as well as the finalized policy and qualifying products for CY 2026.
5. Calculation of OPPS Scaled Payment Weights
We established a policy in the CY 2013 OPPS/ASC final rule with comment period (77 FR 68283) of using geometric mean-based APC costs to calculate relative payment weights under the OPPS. In the CY 2025 OPPS/ASC final rule with comment period (89 FR 93964 through 93965), we applied this policy and calculated the relative payment weights for each APC for CY 2025 that were shown in Addenda A and B of the CY 2025 OPPS/ASC final rule with comment period (which were made available via the internet on the CMS website) using the APC costs discussed in sections II.A.1. and II.A.2. of the CY 2025 OPPS/ASC final rule with comment period (89 FR 93921 through 93947). For CY 2026, as we did for CY 2025, we proposed to continue to apply the policy established in CY 2013 and calculate relative payment weights for each APC for CY 2026 using geometric mean-based APC costs.
For CY 2012 and CY 2013, outpatient clinic visits were assigned to one of five levels of clinic visit APCs, with APC 0606 representing a mid-level clinic visit. In the CY 2014 OPPS/ASC final rule with comment period (78 FR 75036 through 75043), we finalized a policy that created alphanumeric HCPCS code G0463 (Hospital outpatient clinic visit for assessment and management of a patient), representing all clinic visits under the OPPS. HCPCS code G0463 was assigned to APC 0634 (Hospital Clinic Visits). We also finalized a policy to use CY 2012 claims data to develop the CY 2014 OPPS payment rates for HCPCS code G0463 based on the total geometric mean cost of the levels one through five CPT Evaluation or Assessment and Management (E/M) codes for clinic visits previously recognized under the OPPS (CPT codes 99201 through 99205 and 99211 through 99215). In addition, we finalized a policy to no longer recognize a distinction between new and established patient clinic visits.
For CY 2016, we deleted APC 0634 and reassigned the outpatient clinic visit HCPCS code G0463 to APC 5012 (Level 2 Examinations and Related Services) (80 FR 70372). For CY 2026, as we did for CY 2025, we proposed to continue to standardize all the relative payment weights to APC 5012. We believe that standardizing relative payment weights to the geometric mean of the APC to which HCPCS code G0463 is assigned maintains consistency in calculating unscaled weights that represent the cost of some of the most frequently provided OPPS services. For CY 2026, as we did for CY 2025, we proposed to assign APC 5012 a relative payment weight of 1.00 and to divide the geometric mean cost of each APC by the geometric mean cost for APC 5012 to derive the unscaled relative payment weight for each APC. The choice of the APC on which to standardize the relative payment weights does not affect payments made under the OPPS because we scale the weights for budget neutrality.
Section 1833(t)(9)(B) of the Act requires that APC reclassification and recalibration changes, wage index changes, and other adjustments be made in a budget neutral manner. Budget neutrality ensures that the estimated aggregate weight under the OPPS for CY 2026 is neither greater than nor less than the estimated aggregate weight that would have been calculated without the changes. To comply with this requirement concerning the APC changes, we proposed to compare the estimated aggregate weight using the CY 2025 scaled relative payment weights to the estimated aggregate weight using the proposed CY 2026 unscaled relative payment weights.
For CY 2025, we multiplied the CY 2025 scaled APC relative payment weight applicable to a service paid under the OPPS by the volume of that service from CY 2024 claims to calculate the total relative payment weight for each service. We then added together the total relative payment weight for each of these services to calculate an estimated aggregate weight for the year. For CY 2026, we proposed to apply the same process using the estimated CY 2026 unscaled relative payment weights rather than scaled relative payment weights. We proposed to calculate the weight scalar by dividing the CY 2025 estimated aggregate weight by the unscaled CY 2026 estimated aggregate weight.
For a detailed discussion of the weight scalar calculation, we refer readers to the OPPS claims accounting document available on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices. Click on the link labeled “Hospital Outpatient Prospective Payment—Notice of Final Rulemaking” for 2026, which can be found under the heading “Hospital Outpatient Regulations and Notices” and open the claims accounting document link, which is labeled “2026 Final Rule OPPS Claims Accounting (PDF).”
We proposed to compare the estimated unscaled relative payment weights in CY 2026 to the estimated total relative payment weights in CY 2025 using CY 2024 claims data, holding all other components of the payment system constant to isolate changes in total weight. Based on this comparison, we proposed to adjust the calculated CY 2026 unscaled relative payment weights for purposes of budget neutrality. We proposed to adjust the estimated CY 2026 unscaled relative payment weights by multiplying them by a proposed weight scalar of 1.4624 to ensure that the proposed CY 2026 relative payment weights are scaled to be budget neutral. The proposed CY 2026 relative payment weights listed in Addenda A and B to the CY 2026 OPPS/ASC proposed rule (which are available via the internet on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices) are scaled and incorporate the recalibration adjustments discussed in sections II.A.1. and II.A.2. of the CY 2026 OPPS/ASC proposed rule.
Section 1833(t)(14) of the Act provides the methodology for payment rates for certain specified covered outpatient drugs (SCODs). Section 1833(t)(14)(H) of the Act provides that additional expenditures resulting from this paragraph shall not be taken into account in establishing the conversion factor, weighting, and other adjustment factors for 2004 and 2005 under paragraph (9) but shall be taken into account for subsequent years. Therefore, the cost of those SCODs (as discussed in section V.B.2. of this final rule with comment period) is included in the budget neutrality calculations for the CY 2026 OPPS.
We did not receive any public comments on the proposed weight scalar calculation, and we are finalizing our proposal to use the calculation process described in the CY 2026 OPPS/ASC proposed rule, without modification, for CY 2026. For CY 2026, as we did for CY 2025, we will continue to apply the policy established in CY ( printed page 53490) 2013 and calculate relative payment weights for each APC for CY 2026 using geometric mean-based APC costs. For CY 2026, as we did for CY 2025, we will assign APC 5012 a relative payment weight of 1.00; and we will divide the geometric mean cost of each APC by the geometric mean cost for APC 5012 to derive the unscaled relative payment weight for each APC. To comply with this requirement concerning the APC changes, we will compare the estimated aggregate weight using the CY 2025 scaled relative payment weights to the estimated aggregate weight using the CY 2026 unscaled relative payment weights.
Using updated final rule claims data, we are updating the estimated CY 2026 unscaled relative payment weights by multiplying them by a weight scalar of 1.4879 to ensure that the final CY 2026 relative payment weights are scaled to be budget neutral. The final CY 2026 relative payments weights listed in Addenda A and B of this final rule with comment period (available via the internet on the CMS website) were scaled and incorporate the recalibration adjustments discussed in sections II.A.1 and II.A.2. of this final rule with comment period.
B. Final Conversion Factor Update
1. OPD Fee Schedule Increase Factor
Section 1833(t)(3)(C)(ii) of the Act requires the Secretary to update the conversion factor used to determine the payment rates under the OPPS on an annual basis by applying the OPD fee schedule increase factor. For purposes of section 1833(t)(3)(C)(iv) of the Act, subject to sections 1833(t)(17) and 1833(t)(3)(F) of the Act, the OPD fee schedule increase factor is equal to the hospital inpatient market basket percentage increase applicable to hospital discharges of the Act (or an amount that is computed and applied with respect to covered OPD services). In the FY 2026 IPPS/Long Term Care Hospital (LTCH) proposed rule (90 FR 18266), consistent with current law, based on IHS Global, Inc.'s (IGI's) fourth quarter 2024 forecast, the proposed FY 2026 IPPS market basket percentage increase was 3.2 percent. We noted that under our regular process for the CY 2026 OPPS/ASC final rule with comment period, we would use the market basket update for the FY 2026 IPPS/LTCH PPS final rule. If that forecast is different than the IPPS market basket percentage increase used for the CY 2026 OPPS/ASC proposed rule, the CY 2026 OPPS/ASC final rule with comment period OPD fee schedule increase factor would reflect that updated forecast of the market basket percentage increase.
For CY 2026, we proposed to use the estimate of the hospital inpatient market basket percentage increase of 3.2 percent as one component to calculate the OPD fee schedule increase factor.
Comment: One commenter supported the CY 2026 update factor.
Response: We thank the supporter for the support.
Comment: Several commenters expressed concerns that the proposed CY 2026 market basket update of 2.4 percent, through which the majority of hospitals would only receive a 0.06 percent net increase, does not adequately keep up with the increased costs of delivering care across all settings.
One commenter suggested that CMS expand the data set to ensure the use of accurate, timely data that reflect real labor costs. Specifically, they requested CMS apply its exceptions and adjustments authority to make a one-time retrospective adjustment of 10 to15 percent to the market basket to account for the update hospitals should have received in 2022 when accounting for inflation. The commenter suggested CMS establish a threshold such that if the payment differential between what was provided and actual costs is greater than 1.5 percentage points, there is a retroactive adjustment to payments above the threshold. The commenter also suggested CMS recalibrate the market basket more frequently, at least once every 3 years to ensure the market basket reflects the appropriate mix of services.
Another commenter suggested that the proposed increase is insufficient if the 340B repayment acceleration and volume control policy for medication administration services are finalized. The commenter also stated that CMS should reconsider the data source for workforce costs because the healthcare industry has shifted since the pandemic. The commenter stated that workers have demanded higher salaries to keep up with economic changes and the ECI survey of hospital employment that CMS employs does not account for contracted or contingent workers.
Another commenter expressed concern that the update is insufficient and unsustainable for rural hospitals. The commenter stated 196 hospitals have closed or ceased to provide inpatient services since 2010, and there are 432 rural hospitals vulnerable to closure. The commenter stated the Consumer Price Index (CPI) was 4.2 percent as of June 2025 and that the Medicare reimbursement continues to fall behind the actual cost of providing care to beneficiaries. The commenter stated CMS' projections for updating payment rates are lower than actual inflation due to the use of historical data. The commenter stated CMS must explore how it can accurately pay rural hospitals by accounting for inflation and historical underpayment.
Commenters stated that since the COVID-19 PHE, IGI has shown a consistent trend of under-forecasting the market basket growth and expressed concern this may indicate a more systematic issue with IGI's forecasting. Several commenters, including many associations, urged CMS to use its special exceptions and adjustments authority under section 1886(d)(5)(I)(i) of the Act to implement a retrospective one-time adjustment for CY 2026 to account for the underestimation of the market basket updates over the last several years. Commenters recommended that CMS implement various one-time adjustments of 4.3 percent to account for underpayments in 1 or more years between CY 2021 and CY 2025 as well as for forecasted underpayments for CY 2026. The commenters stated the underestimation is, in large part, because the market basket is a time-lagged estimate that cannot fully account for unexpected changes that occur, such as historic inflation and increased labor and supply costs.
Response: We acknowledge commenters' concerns on the proposed CY 2026 market basket of 2.4 percent, including the challenging financial landscape in which rural hospitals operate. According to section 1833(t)(3)(C)(iv) of the Act, the OPD fee schedule increase factor for a year must be equal to the IPPS market basket percentage increase factor applicable under section 1886(b)(3)(B)(iii) of the Act to hospital discharges in the fiscal year ending in such year. Therefore, we are unable to adopt a final OPD fee schedule increase factor different than the IPPS market basket percentage increase factor finalized in the FY 2026 IPPS/LTCH PPS final rule. We refer commenters to the FY 2026 IPPS/LTCH PPS final rule for responses regarding the market basket issues commenters raised (90 FR 36900 to 36903).
2. Productivity Adjustment
Section 1833(t)(3)(F)(i) of the Act requires that, for 2012 and subsequent years, the OPD fee schedule increase factor under subparagraph (C)(iv) be reduced by the productivity adjustment described in section 1886(b)(3)(B)(xi)(II) of the Act. Section 1886(b)(3)(B)(xi)(II) of the Act defines the productivity adjustment as equal to the 10-year ( printed page 53491) moving average of changes in annual economy-wide, private nonfarm business multifactor productivity (MFP) (as projected by the Secretary for the 10-year period ending with the applicable fiscal year, year, cost reporting period, or other annual period) (the “productivity adjustment”). In the FY 2012 IPPS/LTCH PPS final rule (76 FR 51689 through 51692), we finalized our methodology for calculating and applying the productivity adjustment. The U.S. Department of Labor's Bureau of Labor Statistics (BLS) publishes the official measures of private nonfarm business productivity for the U.S. economy. We note that previously the productivity measure referenced in section 1886(b)(3)(B)(xi)(II) of the Act was published by BLS as private nonfarm business multifactor productivity. Beginning with the November 18, 2021, release of productivity data, BLS replaced the term multifactor productivity (MFP) with total factor productivity (TFP). BLS noted that this is a change in terminology only and will not affect the data or methodology. As a result of the BLS name change, the productivity measure referenced in section 1886(b)(3)(B)(xi)(II) of the Act is now published by BLS as private nonfarm business total factor productivity. However, as mentioned, the data and methods are unchanged. Please see www.bls.gov for the BLS historical published TFP data. A complete description of IGI's TFP projection methodology is available on the CMS website at https://www.cms.gov/data-research/statistics-trends-and-reports/medicare-program-rates-statistics/market-basket-research-and-information. In addition, we note that beginning with the FY 2022 IPPS/LTCH PPS final rule, we refer to this adjustment as the productivity adjustment rather than the MFP adjustment to more closely track the statutory language in section 1886(b)(3)(B)(xi)(II) of the Act. We note that the adjustment continues to rely on the same underlying data and methodology. In the FY 2026 IPPS/LTCH PPS proposed rule (90 FR 18266), the proposed productivity adjustment for FY 2026 was 0.8 percentage point.
Therefore, we proposed that the productivity adjustment for the CY 2026 OPPS/ASC proposed rule would be 0.8 percentage point. We also proposed that if more recent data subsequently become available after the publication of the CY 2026 OPPS/ASC proposed rule (for example, a more recent estimate of the market basket percentage increase and/or the productivity adjustment), we would use such data, if appropriate, to determine the CY 2026 hospital inpatient market basket update and the productivity adjustment for the final rule with comment period, which are components in calculating the OPD fee schedule increase factor under sections 1833(t)(3)(C)(iv) and 1833(t)(3)(F) of the Act.
We note that section 1833(t)(3)(F) of the Act provides that application of this subparagraph may result in the OPD fee schedule increase factor under section 1833(t)(3)(C)(iv) of the Act being less than 0.0 percent for a year and may result in OPPS payment rates being less than rates for the preceding year. As described in further detail below, we proposed for CY 2026 an OPD fee schedule increase factor of 2.4 percent for the CY 2026 OPPS/ASC proposed rule (which is the proposed estimate of the hospital inpatient market basket percentage increase of 3.2 percent, less the proposed 0.8 percentage point productivity adjustment).
Comment: Commenters requested that CMS eliminate or waive the productivity adjustment for CY 2026 and going forward using CMS' special exceptions and adjustments authority under section 1886(b)(3)(B)(xi) of the Act, which would justify special circumstances. The commenters stated the COVID-19 pandemic years distorted productivity measurements and the application of the full productivity adjustment penalizes hospitals for costs beyond their control. The commenters requested the waiver due to strong concerns about the productivity adjustment cuts given the uncertainty which hospitals and healthcare systems are currently operating.
Commenters expressed concerns that the productivity adjustment, which reflects the private nonfarm business total factor productivity, are not achieved by hospitals and the health care field. Commenters stated the TFP, or productivity adjustment does not account for non-profit or government business which account for 60 percent of hospitals and health systems. The commenter requested we provide transparency into the data on the 10-year moving average periods in calculating the productivity adjustment. The commenter is concerned that the productivity adjustment was artificially and inappropriately increased.
Response: While we understand the commenters' concerns, section 1833(t)(3)(F)(i) of the Act requires that after determining the OPD fee schedule increase factor under subparagraph (C)(iv), the Secretary shall reduce such increase factor by the productivity adjustment described in section 1886(b)(3)(B)(xi) of the Act. As required by statute, the FY 2026 productivity adjustment is derived based on the 10-year moving average growth in economy-wide productivity for the period ending FY 2026.
We thank the commenters for their comments. After consideration of the comments received and consistent with our proposal, we are finalizing an OPD fee schedule increase factor with modification of 2.4 percent for CY 2026, which consists of the IPPS market basket increase factor of 3.2 percent less a 0.8 percentage point productivity adjustment.
3. Other Conversion Factor Adjustments
To set the OPPS conversion factor for 2026, we proposed to increase the CY 2025 conversion factor of $89.169 by 2.4 percent. In accordance with section 1833(t)(9)(B) of the Act, we proposed to further adjust the conversion factor for CY 2026 to ensure that any revisions made to the wage index and rural adjustment are made on a budget neutral basis. We proposed to apply an overall budget neutrality factor of 1.0116 for wage index changes by comparing proposed total estimated payments from our simulation model using the proposed FY 2026 IPPS wage indexes to those payments using the CY 2025 OPPS wage indexes. We further proposed to calculate an additional budget neutrality factor of 0.9955 to account for our proposed policy to cap wage index reductions for hospitals at 5 percent on an annual basis and the CY 2026 proposed transitional exception for low wage index hospitals.
For CY 2026, we proposed to maintain the current rural adjustment policy, as discussed in section II.E. of this final rule with comment period. Therefore, the proposed budget neutrality factor for the rural adjustment was 1.0000.
We proposed to calculate a CY 2026 budget neutrality adjustment factor for the cancer hospital payment adjustment. We previously finalized transitioning from the target PCR of 0.89 for CYs 2020 through 2023 (which included the 1.0 percentage point reduction as required by section 16002(b) of the 21st Century Cures Act) and incrementally reducing the target PCR by an additional 1.0 percentage point for each calendar year, beginning with CY 2024, until the target PCR equals the PCR of non-cancer hospitals calculated using the most recent data minus 1.0 percentage point as required by section 16002(b) of the 21st Century Cures Act. Based on the most recent data available for this final rule with comment period, the target PCR now equals the PCR of non-cancer ( printed page 53492) hospitals. We proposed a CY 2026 target PCR equal to 0.87 for the cancer hospital payment adjustment, which includes the 1.0 percentage point reduction as required by section 16002(b) of the 21st Century Cures Act. We note that this proposed target PCR is the same as the final target PCR established in the CY 2025 OPPS (89 FR 93979). Therefore, we proposed to apply a budget neutrality adjustment factor of 1.0000 to the conversion factor for the cancer hospital payment adjustment.
For the CY 2026 OPPS/ASC proposed rule, we estimated that proposed pass-through spending for drugs, biologicals, and devices for CY 2026 will equal approximately $587 million, which represents 0.59 percent of total projected CY 2026 OPPS spending. Therefore, we stated that the proposed conversion factor would be adjusted by the difference between the 0.37 percent estimate of pass-through spending for CY 2025 and the 0.59 percent estimate of proposed pass-through spending for CY 2026, resulting in a proposed decrease to the conversion factor for CY 2026 of 0.22 percentage point.
We proposed that estimated payments for outliers would be 1.0 percent of total OPPS payments for CY 2026. We estimate for the CY 2026 OPPS/ASC proposed rule that outlier payments would be approximately 0.92 percent of total OPPS payments in CY 2025; the 1.00 percent for proposed outlier payments in CY 2026 would constitute a 0.08 percentage point increase in payment in CY 2026 relative to CY 2025.
For CY 2026, we proposed to use a conversion factor of $91.747 in the calculation of the national unadjusted payment rates for those items and services for which payment rates are calculated using geometric mean costs; that is, the proposed OPD fee schedule increase factor of 1.024 (2.4 percent for CY 2026), the required proposed wage index budget neutrality adjustment of approximately 1.0116, the proposed 5 percent annual cap for individual hospital wage index reductions adjustment and the proposed transitional exception of approximately 0.9955, the proposed cancer hospital payment adjustment of 1.0000, and the proposed adjustment factor of 0.9978 (a decrease of 0.22 percentage point) for the difference in pass-through spending, and a 0.08 percentage point increase in projected OPPS spending for the projected increase in outlier payments, which resulted in a proposed conversion factor for CY 2026 of $91.747.
For CY 2026, we also proposed that hospitals that fail to meet the reporting requirements of the Hospital OQR Program would continue to be subject to a further reduction of 2.0 percentage points to the OPD fee schedule increase factor. For hospitals that fail to meet the requirements of the Hospital OQR Program, we proposed to make all other adjustments discussed above and apply an adjustment factor of 0.9805 to the proposed CY 2026 conversion factor of $91.747. We proposed that the hospitals that fail to meet the requirements of the Hospital OQR Program will use a reduced OPD fee schedule update factor of 0.4 percent (that is, the proposed OPD fee schedule increase factor of 2.4 percent further reduced by 2.0 percentage points).
For CY 2026, we proposed to reduce payments for non-drug items and services for hospitals for whom the annual reduction to payment amounts under § 419.32(b)(1)(iv)(B)(12) applies with a 2 percentage point reduction to the OPD fee schedule increase factor, explained in more detail in section V.B.7. of this final rule with comment period. This would result in a proposed reduced conversion factor for CY 2026 of approximately $89.958 for this group of hospitals. The calculations we performed to determine the CY 2026 proposed conversion factor are shown in Table 8.
Comment: Commenters expressed concerns with the proposed 340B accelerated offset and its reduction on the CY 2026 proposed rate increase.
Response: We acknowledge commenters' concerns on the proposed 340B accelerated offset. For a discussion of the CY 2026 340B remedy offset, we refer readers to section V.B.7 of this final rule with comment period.
For this CY 2026 OPPS/ASC final rule with comment period, based on more recent data available, the OPD fee schedule increase factor for the CY 2026 OPPS is 2.6 percent (which reflects the 3.3 percent final estimate of the hospital inpatient market basket percentage increase with a—0.7 percentage point productivity adjustment). For CY 2026, we are using a conversion factor of $91.415 in the calculation of the national unadjusted payment rates for those items and services for which payment rates are calculated using geometric mean costs; that is, the OPD fee schedule increase factor of 2.6 percent for CY 2026, the required wage index budget neutrality adjustment of 0.9990, the 5 percent annual cap for individual hospital wage index reductions of 0.9995, the cancer hospital payment adjustment of 1.0000, and the adjustment of 0.07 (or 0.37 less 0.30) percentage point of projected OPPS spending for the difference in pass-through spending that results in a conversion factor for CY 2026 of $91.415. We are also finalizing a reduced conversion factor of $89.632 in the calculation of payments for hospitals that fail to meet the Hospital OQR Program requirements (a difference of −1.783 in the conversion factor relative to hospitals that met the requirements).
( printed page 53493) ( printed page 53494)C. Wage Index Changes
Section 1833(t)(2)(D) of the Act requires the Secretary to determine a wage adjustment factor to adjust the portion of payment and coinsurance attributable to labor-related costs for relative differences in labor and labor-related costs across geographic regions in a budget neutral manner (codified at 42 CFR 419.43(a)). This portion of the OPPS payment rate is called the OPPS labor-related share. The budget neutrality calculation methodology is discussed in section II.A.5. of this final rule with comment period.
The OPPS labor-related share is 60 percent of the national OPPS payment. This labor-related share is based on a regression analysis that determined that, for all hospitals, approximately 60 percent of the costs of services paid under the OPPS were attributable to wage costs. We confirmed that this labor-related share for outpatient services is appropriate during our regression analysis for the payment adjustment for rural hospitals in the CY 2006 OPPS/ASC final rule with comment period (70 FR 68553). We proposed to continue this policy for the CY 2026 OPPS/ASC final rule with comment period. We refer readers to section II.C. of this final rule with comment period for a description and an example of how the wage index for a particular hospital is used to determine payment for the hospital.
We did not receive any public comments on our proposed labor-related share, and we are finalizing our proposal without modification.
As discussed in the claims accounting narrative included with the supporting documentation for this final rule with comment period (which is available via the internet on the CMS website ( https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices)), for estimating APC costs, we would standardize 60 percent of estimated claims costs for geographic area wage variation using the same FY 2026 pre-reclassified wage index that we use under the IPPS to standardize costs. This standardization process removes the effects of differences in area wage levels from the determination of a national unadjusted OPPS payment rate and copayment amount.
Under §§ 419.41(c)(1) and 419.43(c) (published in the OPPS April 7, 2000, final rule with comment period (65 FR 18495 and 18545)), the OPPS adopted the final fiscal year IPPS post-reclassified wage index as the calendar year wage index for adjusting the OPPS standard payment amounts for labor market differences. Therefore, the wage index that applies to a particular acute care, short-stay hospital under the IPPS also applies to that hospital under the OPPS. As initially explained in the September 8, 1998, OPPS/ASC proposed rule (63 FR 47576), we believe that using the IPPS wage index as the source of an adjustment factor for the OPPS is reasonable and logical, given the inseparable, subordinate status of the HOPD within the hospital overall. In accordance with section 1886(d)(3)(E) of the Act, the IPPS wage index is updated annually.
The Affordable Care Act contained several provisions affecting the wage index. These provisions were discussed in the CY 2012 OPPS/ASC final rule with comment period (76 FR 74191). Section 10324 of the Affordable Care Act added section 1886(d)(3)(E)(iii)(II) to the Act, which defines a frontier State and amended section 1833(t) of the Act to add paragraph (19), which requires a frontier State wage index floor of 1.00 in certain cases, and states that the frontier State floor shall not be applied in a budget neutral manner. We codified these requirements at § 419.43(c)(2) and (3) of our regulations. For CY 2026, we proposed to implement this provision in the same manner as we have since CY 2011. Under this policy, the frontier State hospitals would receive a wage index of 1.00 if the otherwise applicable wage index (including reclassification, the rural floor, and rural floor budget neutrality) is less than 1.00. Because the HOPD receives a wage index based on the geographic location of the specific inpatient hospital with which it is associated, the frontier State wage index adjustment applicable for the inpatient hospital also would apply for any associated HOPD. We refer readers to the FY 2011 through FY 2025 IPPS/LTCH PPS final rules for discussions regarding this provision, including our methodology for identifying which areas meet the definition of “frontier States” as provided for in section 1886(d)(3)(E)(iii)(II) of the Act: for FY 2011, 75 FR 50160 through 50161; for FY 2012, 76 FR 51793, 51795, and 51825; for FY 2013, 77 FR 53369 and 53370; for FY 2014, 78 FR 50590 and 50591; for FY 2015, 79 FR 49971; for FY 2016, 80 FR 49498; for FY 2017, 81 FR 56922; for FY 2018, 82 FR 38142; for FY 2019, 83 FR 41380; for FY 2020, 84 FR 42312; for FY 2021, 85 FR 58765; for FY 2022, 86 FR 45178; FY 2023, 87 FR 49006; FY 2024, 88 FR 58977; and for FY 2025, 89 FR 69300.
In addition to the changes required by the Affordable Care Act, we note that the proposed FY 2026 IPPS wage indexes continue to reflect a number of adjustments implemented in past years, including, but not limited to, reclassification of hospitals to different geographic areas, the rural floor provisions, the imputed floor wage index adjustment in all-urban States, an adjustment for occupational mix, an adjustment to the wage index based on commuting patterns of employees (the out-migration adjustment), and the permanent 5 percent cap on any decrease to a hospital's wage index from its wage index in a prior FY. Beginning with FY 2024, we include hospitals with § 412.103 reclassification along with geographically rural hospitals in all rural wage index calculations, and to exclude “dual reclass” hospitals (hospitals with simultaneous § 412.103 and Medicare Geographic Classification Review Board (MGCRB) reclassifications) implicated by the hold harmless provision at section ( printed page 53495) 1886(d)(8)(C)(ii) of the Act (88 FR 58971 through 58973). We refer readers to the FY 2026 IPPS/LTCH PPS proposed rule (90 FR 18217 through 18236) for a detailed discussion of all proposed changes to the FY 2026 IPPS wage indexes.
Comment: MedPAC expressed support for CMS' annual process to update the OPPS wage index with newer wage data and Office of Management and Budget delineations. However, MedPAC also expressed its concerns with flaws in the wage index system. MedPAC emphasized that, in order to improve the accuracy and fairness of Medicare's wage index systems for IPPS and OPPS hospitals and other providers, Medicare needs wage indexes that are less manipulable, that more accurately and precisely reflect geographic differences in market-wide labor costs, and that limit how much wage index values can differ among providers that are competing for the same pool of labor. To address these concerns, MedPAC stated that in its June 2023 report to Congress, it recommended that the Congress repeal the existing Medicare wage index statutes, including current exceptions, and require the Secretary to phase in new wage index systems for hospitals and other types of providers that (1) use all-employer, occupation-level wage data with different occupation weights for the wage index of each provider type; (2) reflect local area level differences in wages between and within metropolitan statistical areas and statewide rural areas; and (3) smooth wage index differences across adjacent local areas.
Response: We appreciate MedPAC's concerns, but as MedPAC acknowledges in its comment, the Congress would need to change current law to implement MedPAC's suggestions.
Comment: One commenter expressed support for CMS' policy to treat urban hospitals re-designated as rural under § 412.103 the same as geographically rural hospitals for the wage index calculation. The commenter opined that treating urban hospitals re-designated as rural under § 412.103 the same as geographically rural hospitals for the calculation of the rural wage index and rural floor is an appropriate and fair implementation of the statute.
Response: We thank the commenter for their support. We note that in the FY 2023 IPPS/LTCH PPS final rule (87 FR 49018 through 49021), we finalized a permanent approach to smooth year-to-year decreases in hospitals' wage indexes. Specifically, for FY 2023 and subsequent years, we apply a 5 percent cap on any decrease to a hospital's wage index from its wage index in the prior FY, regardless of the circumstances causing the decline. That is, a hospital's wage index for FY 2026 would not be less than 95 percent of its final wage index for FY 2025. Except for newly opened hospitals, we apply the cap for a fiscal year using the final wage index applicable to the hospital on the last day of the prior fiscal year. A newly opened hospital would be paid the wage index for the area in which it is geographically located for its first full or partial fiscal year (subject to any reclassification), and it would not receive a cap for that first year, because it would not have been assigned a wage index in the prior year (in accordance with 42 CFR 419.41(c)(1) and 419.43(c), as noted previously).
Comment: Several commenters, including MedPAC, supported the policy to cap wage index decreases. MedPAC urged CMS to apply a cap to wage index increases as well. Several commenters urged CMS to exclude the wage index cap policy from budget neutrality.
Response: We thank the commenters for their support. We appreciate MedPAC's suggestion that the cap on wage index changes should also be applied to increases in the wage index. However, as we stated in the FY 2023 IPPS/LTCH PPS final rule (87 FR 49021), one purpose of the wage index policy is to help mitigate the significant negative impacts of certain wage index changes. That is, we cap decreases because we believe that a hospital would be able to more effectively budget and plan when there is predictability about its expected minimum level of OPPS payments in the upcoming fiscal year. We do not have a policy to limit wage index increases because we do not believe such a policy is needed to enable hospitals to more effectively budget and plan their operations. Therefore, we believe it is appropriate for hospitals that experience an increase in their wage index value to receive that wage index value. With respect to excluding the wage index cap policy from budget neutrality, under the OPPS, section 1833(t)(2)(D) of the Act requires the Secretary to determine a wage adjustment factor to adjust the portion of payment and coinsurance attributable to labor-related costs for relative differences in labor and labor-related costs across geographic regions in a budget neutral manner. This statutory requirement is inconsistent with the commenters' request to exclude the wage index cap policy from budget neutrality.
Consistent with the FY 2026 IPPS/LTCH PPS proposed rule (90 FR 18233), we proposed to discontinue for CY 2026 and subsequent years the low wage index hospital policy under the OPPS. Under the low wage index hospital policy that we adopted for the OPPS (84 FR 61186 through 61188), we increased the wage index for hospitals with a wage index value below the 25th percentile wage index value for a calendar year by half the difference between the otherwise applicable final wage index value for a year for that hospital and the 25th percentile wage index value for that year across all hospitals. We removed the low wage index hospital policy from the IPPS wage index calculation for FY 2025 after considering the Court of Appeals for the D.C. Circuit's decision in Bridgeport Hosp. v. Becerra, 108 F.4th 882 (D.C. Cir. 2024). On July 23, 2024, the court held in Bridgeport Hosp. v. Becerra that the Secretary lacked authority under section 1886(d)(3)(E) of the Act or under the “adjustments” language of section 1886(d)(5)(I)(i) of the Act to adopt the low wage index hospital policy for FY 2020 for the IPPS, and that the policy for FY 2020 and related budget neutrality adjustment in the IPPS must be vacated. After considering the court's decision, in the interim final action with comment period (IFC) titled “Medicare Program; Changes to the Fiscal Year 2025 Hospital Inpatient Prospective Payment System (IPPS) Rates Due to Court Decision” (referred to herein as the FY 2025 IFC) (89 FR 80405 through 80421), we recalculated the FY 2025 IPPS hospital wage index to remove the low wage index hospital policy for FY 2025 and also removed the low wage index budget neutrality factor from the FY 2025 standardized amounts.
In the FY 2026 IPPS/LTCH PPS proposed rule, after considering the D.C. Circuit's decision in Bridgeport Hosp. v. Becerra, we proposed to discontinue the low wage index hospital policy for FY 2026 and subsequent fiscal years. We refer the reader to the FY 2025 IFC (89 FR 80405 through 80421) and FY 2026 IPPS/LTCH PPS proposed rule (90 FR 18233 through 18236) for a detailed discussion regarding the removal of the low wage index hospital policy from the IPPS for FYs 2025 and 2026.
As discussed previously, from the establishment of the OPPS in 2000 through 2024, we adopted the IPPS wage index on a calendar year basis in the OPPS. From FY 2020 to FY 2024, the IPPS wage index included the low wage index hospital policy and we correspondingly adopted the low wage index hospital policy under the OPPS for CY 2020 to CY 2024. However, when the Bridgeport decision was issued in July 2024, the OPPS did not remove the ( printed page 53496) low wage index hospital policy from the calculation of the CY 2025 wage index. As discussed in the CY 2025 OPPS/ASC final rule with comment period, this decision to continue the low wage index hospital policy under the OPPS for CY 2025 (and thus to diverge from the IPPS wage index for FY 2025) was due principally to the unique circumstances presented by the timing of the court decision and subsequent IFC and the statutory authority that CMS relied upon to implement the low wage index hospital policy under the OPPS was different than the statutory authority relied upon for the policy under the IPPS. We took this approach for the CY 2025 OPPS given the unusual circumstances wherein an appellate court ruled that CMS lacked authority under the IPPS statute for a policy under the FY 2020 IPPS wage index that the OPPS/ASC proposed rule had already proposed to include in the OPPS wage index. Under these circumstances, we concluded that continuing the low wage index hospital policy for CY 2025 would avoid unexpected and arguably unfair payment consequences for hospitals that were not plaintiffs in Bridgeport. Additionally, we believed that the same reasons underlying adoption of the IFC policies for the FY 2025 IPPS wage index weighed against incorporating those policies for purposes of the CY 2025 OPPS wage index. Specifically, we noted in the IFC that the intention of the policies implemented therein was to “promote certainty regarding . . . payments” and “provide for payment stability and promote predictability,” in light of the court's decision in Bridgeport (89 FR 80408) and we determined that those interests would be better served by finalizing the OPPS wage index methodology as proposed, including the low wage index hospital policy. Based on these considerations, we continued the low wage index hospital policy under the OPPS for CY 2025 as proposed but indicated that we would explore options for realigning the IPPS and OPPS wage index values through future rulemaking. We refer readers to the CY 2025 OPPS/ASC final rule with comment period for a detailed discussion regarding our retention of the low wage index hospital policy under the OPPS for CY 2025 (89 FR 93975 through 93976).
Given the proposal to discontinue the low wage index hospital policy under the IPPS in the FY 2026 IPPS/LTCH PPS proposed rule and the absence of the timing issues which compelled us to continue the low wage index hospital policy under the OPPS for CY 2025, we stated in the CY 2026 OPPS/ASC proposed rule that we think it is now appropriate to return to our longstanding policy of using the IPPS wage index as the source of an adjustment factor for the OPPS. Consequently, to effectuate full realignment of the IPPS and OPPS wage index values in CY 2026, we proposed to eliminate the low wage index hospital policy under the OPPS and use the IPPS wage index in CY 2026 and subsequent years.
To effectuate full realignment of the IPPS and OPPS wage index values in CY 2026, we proposed that the 5 percent cap that will apply to the CY 2026 OPPS wage index will be based off the IPPS wage index for FY 2025 rather than the OPPS wage index for CY 2025. We noted that because the CY 2025 OPPS wage index was different than the FY 2025 IPPS wage index (due to the continuation of the low wage index hospital policy under the OPPS), using the FY 2026 IPPS wage index for the CY 2026 OPPS wage index would result in decreases greater than 5 percent to some hospitals' wage indexes under the OPPS. Therefore, under our proposal the 5 percent cap on wage index decreases in the CY 2026 OPPS would apply in a similar manner to years prior to the CY 2025 OPPS, in which IPPS hospitals would receive the same wage index with the cap on wage index decreases as they would under the FY IPPS, and non-IPPS hospitals and CMHCs would receive a similar corresponding wage index with the cap on wage index decreases policy under the broader wage index adoption.
Comment: Most commenters supported our proposal to effectuate full realignment of the IPPS and OPPS wage index values in CY 2026 by eliminating the low wage index hospital policy under the OPPS and using the IPPS wage index in CY 2026 and subsequent years.
Response: We thank commenters for their support.
Comment: One commenter expressed concern about our proposal to eliminate the low wage index hospital policy, stating that eliminating it without a permanent, equitable alternative will disproportionately harm hospitals that serve vulnerable and underserved populations. Another commenter urged CMS to reconsider its approach to the area wage index following the removal of the low wage index policy and to take corrective action to address its inequities, particularly for rural States.
Response: We understand the commenters' concerns that the rationale for implementing the low wage index hospital policy remains. However, we believe it is important to return the IPPS and OPPS wage index values to their historical and longstanding alignment. As initially explained in the September 8, 1998, OPPS/ASC proposed rule (63 FR 47576), we believe that using the IPPS wage index as the source of an adjustment factor for the OPPS is reasonable and logical, given the inseparable, subordinate status of the HOPD within the hospital overall.
After consideration of the public comments we received, we are finalizing our proposal without modification to discontinue the low wage index hospital policy under the OPPS and use the FY 2026 IPPS post-reclassified wage index for urban and rural areas as finalized in the FY 2026 IPPS final rule as the wage index for the OPPS. We are also finalizing our proposal without modification that the 5 percent cap that will apply to the CY 2026 OPPS wage index and will be based off the IPPS wage index for FY 2025 rather than the OPPS wage index for CY 2025.
We note that in the FY 2026 IPPS proposed rule (90 FR 18233 through 18235) we proposed, using our authority under section 1886(d)(5)(I)(i) of the Act, to adopt a narrow transitional exception to the calculation of FY 2026 IPPS payments for low wage index hospitals significantly impacted by the discontinuation of the low wage index hospital policy. As indicated in that rule, we proposed this temporary payment exception “to mitigate short-term instability and payment fluctuations that can negatively impact hospitals consistent with principles of certainty and predictability under prospective payment systems.” To address these same concerns under the OPPS, we correspondingly proposed a transitional payment exception for CY 2026 under the OPPS using our equitable adjustment authority under section 1833(t)(2)(E) of the Act. This authority allows the Secretary to establish, in a budget neutral manner, adjustments as determined to be necessary to ensure equitable payments.
The transitional exception policy we proposed would apply to hospitals that benefited from the CY 2024 low wage index hospital policy. For those hospitals, we proposed to compare the hospital's proposed CY 2026 wage index to the hospital's CY 2024 wage index. If the hospital is significantly impacted by the discontinuation of the low wage index hospital policy, meaning the hospital's proposed CY 2026 wage index is decreasing by more than 9.75 percent from the hospital's CY 2024 wage index, then the transitional payment exception for CY 2026 for that ( printed page 53497) hospital would be equal to the additional CY 2026 amount the hospital would be paid under the OPPS if its CY 2026 wage index were equal to 90.25 percent of its CY 2024 wage index. This proposed transitional payment exception would be applied after the application of the 5-percent cap described at 42 CFR 412.64(h)(7). We proposed to make this policy budget neutral under the OPPS through the second wage index budget neutrality adjustment applied to the OPPS conversion factor (which currently includes the 5 percent hold harmless cap policy).
Comment: Multiple commenters supported CMS' proposal to establish a transitional payment exception for CY 2026. Commenters also requested that the proposed transitional payment exception be applied in a non-budget neutral manner.
Response: We appreciate the commenters' support of our policy to establish a transitional payment exception for CY 2026. For the OPPS, section 1833(t)(2)(D) of the Act requires the Secretary to determine a wage adjustment factor to adjust the portion of payment and coinsurance attributable to labor-related costs for relative differences in labor and labor-related costs across geographic regions in a budget neutral manner. Commenters' request to exclude the proposed transitional payment exception from budget neutrality is inconsistent with the budget neutral requirement under 1833(t)(2)(D) of the Act.
Comment: One commenter stated that as proposed, the transitional payment exception would cause disproportionate harm to rural, safety-net, and OPPS hospitals that cannot be reclassified. To avoid this outcome, the commenter recommended that CMS: (1) anchor the 5 percent cap to the CY 2025 OPPS wage-index baseline, not the FY 2025 IPPS baseline; (2) broaden the transitional exception to >5 percent decline, not >9.75 percent, and set the floor at 95 percent of CY 2024, not 90.25 percent; and (3) retain an OPPS low-wage policy or equivalent hold-harmless policies until alignment can occur without producing greater than 5 percent decreases from baseline.
Response: We appreciate the alternative methods suggested by the commenter to help mitigate the effect on hospitals of eliminating the low wage index hospitals policy. We note, however, that we had described in the CY 2025 OPPS/ASC final rule with comment period our intention to realign the wage index and by adopting into the OPPS transitional payment exception the same percentages and methodologies used by the IPPS transitional payment exception, we can fully align the wage index across both systems in CY 2026 rather than having discrepancies and inconsistencies that continue into future years based on ongoing transition policies.
After consideration of the public comments we received, we are finalizing our proposal without modification to adopt a transitional payment exception for CY 2026 under the OPPS using our equitable adjustment authority under section 1833(t)(2)(E) of the Act.
Core Based Statistical Areas (CBSAs) are made up of one or more constituent counties. Each CBSA and constituent county has its own unique identifying codes. The FY 2018 IPPS/LTCH PPS final rule (82 FR 38130) discussed the two different lists of codes to identify counties: Social Security Administration (SSA) codes and Federal Information Processing Standard (FIPS) codes. Historically, CMS listed and used SSA and FIPS county codes to identify and crosswalk counties to CBSA codes for purposes of the IPPS and OPPS wage indexes. However, the SSA county codes are no longer being maintained and updated, although the FIPS codes continue to be maintained by the U.S. Census Bureau. The Census Bureau's most current statistical area information is derived from ongoing census data received since 2010; the most recent data are from 2015. The Census Bureau maintains a complete list of changes to counties or county equivalent entities on the website at https://www.census.gov/programs-surveys/geography/technical-documentation/county-changes.html. In the FY 2018 IPPS/LTCH PPS final rule (82 FR 38130), for purposes of crosswalking counties to CBSAs for the IPPS wage index, we finalized our proposal to discontinue the use of the SSA county codes and begin using only the FIPS county codes. Similarly, for the purposes of crosswalking counties to CBSAs for the OPPS wage index, in the CY 2018 OPPS/ASC final rule with comment period (82 FR 59260), we finalized our proposal to discontinue the use of SSA county codes and begin using only the FIPS county codes. For CY 2026, under the OPPS, we are continuing to use only the FIPS county codes for purposes of crosswalking counties to CBSAs.
We proposed to use the FY 2026 IPPS post-reclassified wage index for urban and rural areas as the wage index for the OPPS to determine the wage adjustments for both the OPPS payment rate and the copayment rate for CY 2026. Therefore, any policies and adjustments that are finalized for the FY 2026 IPPS post-reclassified wage index would be reflected in the final CY 2026 OPPS wage index beginning on January 1, 2026, if appropriate. We refer readers to the FY 2026 IPPS/LTCH PPS proposed rule (90 FR 18217 through 18236) and the proposed FY 2026 hospital wage index files posted on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/fy-2026-ipps-proposed-rule-home-page. Regarding budget neutrality for the CY 2026 OPPS wage index, we refer readers to section II.C. of this final rule with comment period. We continue to believe that using the IPPS post-reclassified wage index as the source of an adjustment factor for the OPPS is reasonable and logical, given the inseparable, subordinate status of the HOPD within the hospital overall.
Hospitals that are paid under the OPPS, but not under the IPPS, do not have an assigned hospital wage index under the IPPS. Therefore, for non-IPPS hospitals paid under the OPPS, it is our longstanding policy to assign the wage index that would be applicable if the hospital was paid under the IPPS, based on its geographic location and any applicable wage index policies and adjustments. We proposed to continue this policy for CY 2026. We refer readers to the FY 2026 IPPS/LTCH PPS proposed rule (90 FR 18217 through 18236) for a detailed discussion of the proposed changes to the FY 2026 IPPS wage indexes.
It has been our longstanding policy to allow non-IPPS hospitals paid under the OPPS to qualify for the out-migration adjustment if they are located in a “section 505 out-migration county” (that is, a county identified under section 505 of the Medicare Prescription Drug, Improvement, and Modernization Act of 2003 (MMA)) (Pub. L. 108-173). Applying this adjustment is consistent with our policy of adopting IPPS wage index policies for hospitals paid under the OPPS. We note that, because non-IPPS hospitals cannot reclassify, they are eligible for the out-migration wage index adjustment if they are located in a section 505 out-migration county. This is the same out-migration adjustment policy that would apply if the hospital were paid under the IPPS. For CY 2026, we proposed to continue our policy of allowing non-IPPS hospitals paid under the OPPS to qualify for the outmigration adjustment if they are located in a section 505 out-migration county (section 505 of the MMA) (88 FR 49585 and 49586). Furthermore, we proposed that the wage index that would apply for CY 2026 to non-IPPS hospitals paid ( printed page 53498) under the OPPS would continue to include the rural floor adjustment and any policies and adjustments applied to the IPPS wage index. In addition, we proposed that the wage index that would apply to non-IPPS hospitals paid under the OPPS would include the 5 percent cap on wage index decreases and the previously described proposed transitional payment exception for hospitals significantly impacted by the discontinuation of the low wage index hospital policy. We did not receive any comments on these proposals and are finalizing as proposed.
For CMHCs, for CY 2026, we proposed to continue to calculate the wage index by using the post-reclassification IPPS wage index based on the CBSA where the CMHC is located. Furthermore, we proposed that the wage index that would apply to a CMHC for CY 2026 would continue to include the rural floor adjustment and any policies and adjustments applied to the IPPS wage index. In addition, the wage index that would apply to CMHCs would include the 5 percent cap on wage index decreases. Also, we proposed that the wage index that would apply to CMHCs would not include the outmigration adjustment because that adjustment only applies to hospitals.
We did not receive any public comments on these proposals, and we are finalizing our proposals regarding CMHC wage index calculations without modification.
Table 4A associated with the FY 2026 IPPS/LTCH PPS final rule (available via the internet on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/fy-2026-ipps-final-rule-home-page) identifies counties that would be eligible for the out-migration adjustment. Table 2 associated with the FY 2026 IPPS/LTCH PPS final rule (available for download via the website noted previously) identifies IPPS hospitals that would receive the out-migration adjustment for FY 2026. We are including the outmigration adjustment information from Table 2 associated with the FY 2026 IPPS/LTCH PPS final rule as Addendum L to this final rule with comment period, with the addition of non-IPPS hospitals that would receive the section 505 outmigration adjustment under this final rule with comment period. Addendum L is available via the internet on the CMS website. We refer readers to the CMS website for the OPPS at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices. At this link, readers will find a link to the final FY 2026 IPPS wage index tables and Addendum L.
D. Final Statewide Average Default Cost-to-Charge Ratios (CCRs)
In addition to using CCRs to estimate costs from charges on claims for ratesetting, we use overall hospital-specific CCRs calculated from the hospital's most recent cost report (OMB control number: 0938-0050 for Form CMS-2552-10) to determine outlier payments, payments for pass-through devices, and monthly interim transitional corridor payments under the OPPS during the PPS year. For certain hospitals, under the regulations at 42 CFR 419.43(d)(5)(iii), we use the statewide average default CCRs to determine the payments mentioned earlier if it is not possible to determine an accurate CCR for a hospital in certain circumstances. This includes hospitals that are new, hospitals that have not accepted assignment of an existing hospital's provider agreement, and hospitals that have not yet submitted a cost report. We also use the statewide average default CCRs to determine payments for hospitals whose CCR falls outside the predetermined ceiling threshold for a valid CCR or for hospitals in which the most recent cost report reflects an all-inclusive rate status (Medicare Claims Processing Manual (Pub. L. 100-04), Chapter 4, Section 10.11).
We discussed our policy for using default CCRs, including setting the ceiling threshold for a valid CCR, in the CY 2009 OPPS/ASC final rule with comment period (73 FR 68594 through 68599) in the context of our adoption of an outlier reconciliation policy for cost reports beginning on or after January 1, 2009. For details on our process for calculating the statewide average CCRs, we refer readers to the Claims Accounting Narrative for this final rule with comment period, which is posted on the CMS website. We proposed to calculate the default ratios for CY 2026 using the most recent cost report data.
We did not receive any public comments on our proposal, and we are finalizing our proposal without modification to calculate the default ratios for CY 2026 using the most recent cost report data, which are from a June 2025 HCRIS cost report extract.
We no longer publish a table in the Federal Register containing the statewide average CCRs in the annual OPPS/ASC proposed rule and final rule with comment period. These CCRs and the upper limit CCR value at which we would apply statewide CCRs will be available for download with each CY OPPS/ASC proposed rule and final rule with comment period on the CMS website. We refer readers to our website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices; click on the link on the left of the page titled “Annual Policy Files” and then select the relevant year to download the statewide CCRs and upper limits in the “Downloads” section of the web page.
E. Adjustment for Rural Sole Community Hospitals (SCHs) and Essential Access Community Hospitals (EACHs) Under Section 1833(t)(13)(B) of the Act for CY 2026
In the CY 2006 OPPS final rule with comment period (70 FR 68556), we finalized a payment increase for rural sole community hospitals (SCHs) of 7.1 percent for all services and procedures paid under the OPPS, excluding separately payable drugs and biologicals, brachytherapy sources, items paid at charges reduced to costs, and devices paid under the pass-through payment policy, in accordance with section 1833(t)(13)(B) of the Act, as added by section 411 of the Medicare Prescription Drug, Improvement, and Modernization Act of 2003 (MMA) (Pub. L. 108-173). Section 1833(t)(13) of the Act provides the Secretary the authority to make an adjustment to OPPS payments for rural hospitals, effective January 1, 2006, if justified by a study of the difference in costs by APC between hospitals in rural areas and hospitals in urban areas. Our analysis showed a difference in costs for rural SCHs. Therefore, for the CY 2006 OPPS, we finalized a payment adjustment for rural SCHs of 7.1 percent for all services and procedures paid under the OPPS, excluding separately payable drugs and biologicals, brachytherapy sources, items paid at charges reduced to costs, and devices paid under the pass-through payment policy, in accordance with section 1833(t)(13)(B) of the Act.
In the CY 2007 OPPS/ASC final rule with comment period (71 FR 68010 and 68227), for purposes of receiving this rural adjustment, we revised our regulations at § 419.43(g) to clarify that essential access community hospitals (EACHs) are also eligible to receive the rural SCH adjustment, assuming these entities otherwise meet the rural adjustment criteria. Currently, two hospitals are classified as EACHs, and as of CY 1998, under section 4201(c) of the Balanced Budget Act of 1997 (BBA) (Pub. L. 105-33), a hospital can no longer become newly classified as an EACH.
This adjustment for rural SCHs is budget neutral and applied before calculating outlier payments and ( printed page 53499) copayments. We stated in the CY 2006 OPPS final rule with comment period (70 FR 68560) that we would not reestablish the adjustment amount on an annual basis, but we may review the adjustment in the future and, if appropriate, would revise the adjustment. We provided the same 7.1 percent adjustment to rural SCHs, including EACHs, again in CYs 2008 through 2025 (89 FR 93977).
For CY 2026, we proposed to continue the current policy of a 7.1 percent payment adjustment for rural SCHs, including EACHs, for all services and procedures paid under the OPPS, excluding separately payable drugs and biologicals, brachytherapy sources, items paid at charges reduced to costs, and devices paid under the pass-through payment policy, applied in a budget neutral manner (90 FR 33514).
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: Multiple commenters supported our proposal to continue to apply a 7.1 percent payment adjustment for rural SCHs, including EACHs, in CY 2026.
Response: We thank the commenters for their support of our proposal.
Comment: Several commenters stated that the 7.1 percent payment adjustment should be extended to urban SCHs and Medicare dependent hospitals (MDHs). The commenters stated MDHs are rural hospitals and suggested CMS perform another study to look at the costs that MDHs incur, and opined that CMS has the authority to extend the adjustment to MDHs and urban SCHs without legislation.
Response: We note that our authority for an adjustment under section 1833(t)(13)(B) of the Act only applies to rural hospitals, so we would not have authority to extend this adjustment to a hospital classified as urban. We are not performing another study under 1833(t)(13)(A) of the Act for this rule, but we will consider if this is appropriate for future rulemaking.
Comment: One commenter stated the supplemental adjustments for rural SCHs and EACHs are essential for their sustainability but stated recipients of these payments need greater accountability for improving beneficiary access. The commenter suggested that CMS publish annual data on outpatient service availability and closures or reductions of service, to ensure that these supplemental payments are tied to tangible results for patients.
Response: We thank the commenter for their input and will consider the suggestion for future rulemaking.
After consideration of these public comments, we are finalizing our proposal, without modification, to continue our current policy of utilizing a budget neutral 7.1 percent payment adjustment for rural SCHs, including EACHs, for all services and procedures paid under the OPPS, excluding separately payable drugs and biologicals, brachytherapy sources, devices paid under the pass-through payment policy, and items paid at charges reduced to costs.
F. Payment Adjustment for Certain Cancer Hospitals for CY 2026
1. Background
Since the inception of the OPPS, which was authorized by the BBA, Medicare has paid the 11 hospitals that meet the criteria for cancer hospitals identified in section 1886(d)(1)(B)(v) of the Act under the OPPS for covered outpatient department services. These cancer hospitals are exempted from payment under the IPPS. With the Medicare, Medicaid and SCHIP Balanced Budget Refinement Act of 1999 (Pub. L. 106-113), the Congress added section 1833(t)(7) of the Act, “Transitional Adjustment to Limit Decline in Payment,” which requires the Secretary to determine OPPS payments to cancer and children's hospitals based on their pre-BBA payment amount (these hospitals are often referred to under this policy as “held harmless” and their payments are often referred to as “hold harmless” payments).
As required under section 1833(t)(7)(D)(ii) of the Act, a cancer hospital receives the full amount of the difference between payments for covered outpatient department services under the OPPS and a “pre-BBA amount.” That is, cancer hospitals are permanently held harmless to their “pre-BBA amount,” and they receive transitional outpatient payments (TOPs) or hold harmless payments to ensure that they do not receive a payment that is lower in amount under the OPPS than the payment amount they would have received before implementation of the OPPS, as set forth in section 1833(t)(7)(F) of the Act. The “pre-BBA amount” is the product of the hospital's reasonable costs for covered outpatient department services occurring in the current year and the base payment-to-cost ratio (PCR) for the hospital defined in section 1833(t)(7)(F)(ii) of the Act. The “pre-BBA amount” and the determination of the base PCR are defined at § 419.70(f). TOPs are calculated on Worksheet E, Part B, of the Hospital Cost Report or the Hospital Health Care Complex Cost Report (Form CMS-2552-96 or Form CMS-2552-10 (OMB No: 0938-0050), respectively), as applicable each year. Section 1833(t)(7)(I) of the Act exempts TOPs from budget neutrality calculations.
Section 3138 of the Affordable Care Act (Pub. L. 111-148) amended section 1833(t) of the Act by adding a new paragraph (18), which instructs the Secretary to conduct a study to determine if, under the OPPS, outpatient costs incurred by cancer hospitals described in section 1886(d)(1)(B)(v) of the Act with respect to APC groups exceed outpatient costs incurred by other hospitals furnishing services under section 1833(t) of the Act, as determined appropriate by the Secretary. Section 1833(t)(18)(A) of the Act requires the Secretary to take into consideration the cost of drugs and biologicals incurred by cancer hospitals and other hospitals. Section 1833(t)(18)(B) of the Act provides that, if the Secretary determines that cancer hospitals' costs are higher than those of other hospitals, the Secretary shall provide an appropriate adjustment under section 1833(t)(2)(E) of the Act to reflect these higher costs. In 2011, after conducting the study required by section 1833(t)(18)(A) of the Act, we determined that outpatient costs incurred by the 11 specified cancer hospitals were greater than the costs incurred by other OPPS hospitals. For a complete discussion regarding the cancer hospital cost study, we refer readers to the CY 2012 OPPS/ASC final rule with comment period (76 FR 74200 and 74201).
Based on these findings, we finalized a policy to provide a payment adjustment to the 11 specified cancer hospitals that reflects their higher outpatient costs, as discussed in the CY 2012 OPPS/ASC final rule with comment period (76 FR 74202 through 74206). Specifically, we adopted a policy to provide additional payments to the cancer hospitals so that each cancer hospital's final PCR for services provided in a given calendar year is equal to the weighted average PCR (which we refer to as the “target PCR”) for other hospitals paid under the OPPS. The target PCR is set in advance of the calendar year and is calculated using the most recently submitted or settled cost report data that are available at the time of final rulemaking for the calendar year. The amount of the payment adjustment is made on an aggregate basis at cost report settlement. We note that the changes made by section 1833(t)(18) of the Act do not affect the existing statutory provisions that provide for TOPs for cancer hospitals. ( printed page 53500) The TOPs are assessed, as usual, after all payments, including the cancer hospital payment adjustment, have been made for a cost reporting period. Table 9 displays the target PCR for purposes of the cancer hospital adjustment for CY 2012 through CY 2025.
2. Policy for CY 2026
Section 16002(b) of the 21st Century Cures Act (Pub. L. 114-255) amended section 1833(t)(18) of the Act by adding subparagraph (C), which requires that in applying § 419.43(i) (that is, the payment adjustment for certain cancer hospitals) for services furnished on or after January 1, 2018, the Secretary shall use a target PCR that is 1.0 percentage point less than the target PCR that would otherwise apply. Section 16002(b) of the 21st Century Cures Act also provides that, in addition to the percentage reduction, the Secretary may consider making an additional percentage point reduction to the target PCR that takes into account payment rates for applicable items and services described under section 1833(t)(21)(C) of the Act for hospitals that are not cancer hospitals described under section 1886(d)(1)(B)(v) of the Act. Further, in making any budget neutrality adjustment under section 1833(t) of the Act, section 16002(b) of the 21st Century Cures Act provides that the Secretary shall not take into account the reduced expenditures that result from application of section 1833(t)(18)(C) of the Act.
We proposed to provide additional payments to the 11 specified cancer hospitals so that each cancer hospital's proposed PCR is equal to the weighted average PCR (or “target PCR”) for the other OPPS hospitals, generally using the most recent submitted or settled cost report data that are available, reduced by 1.0 percentage point, to comply with section 16002(b) of the 21st Century Cures Act. As discussed further below, we did not propose an additional reduction beyond the 1.0 percentage point reduction required by section 16002(b) of the 21st Century Cures Act for CY 2026.
To calculate the proposed CY 2026 target PCR, we proposed to use the same extract of cost report data from HCRIS used to estimate costs for the CY 2026 OPPS which, in most cases, would be the most recently available hospital cost reports. Using these cost report data, we included data from Worksheet E, Part B, for each hospital, using data from each hospital's most recent cost report, whether as submitted or settled.
We then limited the dataset to the hospitals with CY 2024 claims data that we used to model the impact of the proposed CY 2026 APC relative payment weights (3,388 hospitals) because we believe it is appropriate to use the same set of hospitals that are being used to calibrate the modeled CY 2026 OPPS. The cost report data for the hospitals in this dataset were from cost report periods with fiscal year ends ranging from 2022 to 2024; however, the cost reporting periods were predominantly from fiscal years ending in 2023 and 2024. We then removed the cost report data of the 49 hospitals located in Puerto Rico from our dataset because we did not believe their cost structure reflected the costs of most hospitals paid under the OPPS, and, therefore, their inclusion may bias the calculation of hospital-weighted statistics. We also removed the cost report data of 12 hospitals because these hospitals had cost report data that were not complete (missing aggregate OPPS payments, missing aggregate cost data, or missing both), so that all cost reports in the study would have both the payment and cost data necessary to calculate a PCR for each hospital, leading to a proposed analytic file of 3,327 hospitals with cost report data.
Using this smaller dataset of cost report data, we estimated that, on average, the OPPS payments to other hospitals furnishing services under the OPPS were approximately 88 percent of reasonable cost (weighted average PCR of 0.88). Therefore, after applying the 1.0 percentage point reduction, as required by section 16002(b) of the 21st Century Cures Act, using our standard process the payment amount associated with the cancer hospital payment adjustment to be determined at cost report settlement would be the additional payment needed to result in a proposed target PCR equal to 0.87 for each cancer hospital.
In the CY 2024 OPPS/ASC final rule with comment period (88 FR 81586 through 81589), we explained that we believe we should begin to take into consideration the PCR of non-cancer hospitals based on the most recently available data for calculating the target PCR. We noted that we do not know if the changes in the data that have yielded lower PCRs for non-cancer hospitals are likely to continue in future years or if, when data from after the PHE ( printed page 53501) is available, we will see the target PCR increase toward its historical norm. Therefore, in the CY 2024 OPPS/ASC final rule with comment period, we finalized our proposal to transition from the target PCR of 0.89 we finalized for CYs 2020 through 2024 (which included the 1.0 percentage point reduction as required by section 16002(b) of the 21st Century Cures Act) and incrementally reduce the target PCR by an additional 1.0 percentage point for each calendar year, beginning with CY 2024, until the target PCR equals the PCR of non-cancer hospitals calculated using the most recent data minus 1.0 percentage point as required by section 16002(b) of the 21st Century Cures Act. Therefore, utilizing this methodology, we finalized in the CY 2025 OPPS/ASC final rule with comment period (89 FR 93977 through 93980) our policy to reduce the CY 2024 target PCR of 0.88 by 1 percentage point and finalized a cancer hospital target PCR of 0.87 for CY 2025.
Since the target PCR based on the OPPS payments to other hospitals furnishing services under the OPPS would be 0.87 after applying the 1.0 percentage point reduction, as required by the section 16002(b) of the 21st Century Cures Act, and would equal the CY 2025 target PCR, it is no longer necessary to continue our transition policy of gradually reducing the pre-COVID-19 PHE target PCR by 1.0 percentage point in lieu of our target PCR calculation. For CY 2026 and subsequent years, we proposed to calculate the target PCR based on our longstanding target PCR calculation methodology described in the CY 2026 OPPS/ASC proposed rule, and then apply the 1.0 percentage point reduction as required by section 16002(b) of the 21st Century Cures Act.
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: Some commenters supported the cancer hospital payment adjustment while one commenter recommended the agency conduct a review to determine if the cancer hospital payment adjustment was still appropriate, including reviewing the accuracy of cost report information.
Response: As previously discussed, section 1833(t)(7)(D)(ii) of the Act requires that the cancer hospitals that meet the statutory criteria receive the full amount of the difference between payments for covered outpatient department services under the OPPS and a “pre-BBA amount.” Additionally, 1833(t)(18)(B) of the Act requires that if the Secretary determines that cancer hospitals' costs are higher than those of other hospitals, to provide an appropriate adjustment under section 1833(t)(2)(E) to reflect these higher costs. The statute does not provide flexibility to discontinue such cancer hospital payment adjustments. Further, we note that Medicare Administrative Contractors routinely perform audits on hospital cost reports to ensure accuracy and completeness.
Comment: One commenter supported the Agency's discontinuation of the gradual reduction policy but recommended that we reinstate such a policy in the future to avoid dramatic and unexpected negative reductions in the target PCR.
Response: We appreciate the commenter's support and will take their comment regarding reinstating the gradual reduction policy into consideration for future rulemaking.
After consideration of public comments we received, we are finalizing without modification our proposal to calculate the target PCR based on our longstanding target PCR calculation methodology described in this final rule with comment and then apply the 1.0 percentage point reduction as required by section 16002(b) of the 21st Century Cures Act. For this final rule with comment period, we are using the most recent cost report data through June 30, 2025 to update the adjustment. We limited the dataset to hospitals with CY 2024 claims data that we used to model the impact of the CY 2026 APC relative payment weights (3,395 hospitals) because it is appropriate to use the same set of hospitals that we are using to calibrate the modeled CY 2026 OPPS. The cost report data for the hospitals in the dataset were from cost reporting periods with fiscal years ends ranging from 2019 to 2024. We then removed the cost report data of the 48 hospitals located in Puerto Rico from our dataset because we do not believe that their cost structure reflects the costs of most hospitals paid under the OPPS and, therefore, their inclusion may bias the calculation of hospital-weighted statistics. We also removed the cost report data of 6 hospitals because these hospitals had cost report data that were not complete (missing aggregate OPPS payments, missing aggregate cost data, or missing both), so that all cost reports in the study would have both the payment and cost data necessary to calculate a PCR for each hospital, leading to an analytic file of 3,341 hospitals with cost report data.
Using this smaller dataset of updated cost report data, we estimate that, on average, the OPPS payments to other hospitals furnishing services under the OPPS were approximately 88 percent of reasonable cost (weighted average PCR of 0.88). Therefore, after applying the 1.0 percentage point reduction, as required by section 16002(b) of the 21st Century Cures Act, we are finalizing a target PCR of 0.87. Table 10 shows the estimated percentage increase in OPPS payments to each cancer hospital for CY 2026, due to the cancer hospital payment adjustment policy. The actual, final amount of the CY 2026 cancer hospital payment adjustment for each cancer hospital will be determined at cost report settlement and will depend on each hospital's CY 2026 payments and costs from the settled CY 2026 cost report. We note that the requirements contained in section 1833(t)(18) of the Act do not affect the existing statutory provisions that provide for TOPs for cancer hospitals. The TOPs will be assessed, as usual, after all payments, including the cancer hospital payment adjustment, have been made for a cost reporting period.
( printed page 53502)G. Hospital Outpatient Outlier Payments
1. Background
The OPPS provides outlier payments to hospitals to help mitigate the financial risk associated with high-cost and complex procedures, where a very costly service could present a hospital with significant financial loss. As explained in the CY 2015 OPPS/ASC final rule with comment period (79 FR 66832 through 66834), we set our projected target for aggregate outlier payments at 1.0 percent of the estimated aggregate total payments under the OPPS for the prospective year. Outlier payments are provided on a service-by-service basis when the cost of a service exceeds the APC payment amount multiplier threshold (the APC payment amount multiplied by a certain amount) as well as the APC payment amount plus a fixed-dollar amount threshold (the APC payment plus a certain dollar amount). In CY 2025, the outlier threshold was met when the hospital's cost of furnishing a service exceeded 1.75 times the APC payment amount (the multiplier threshold) and exceeded the APC payment amount plus $7,175 (the fixed-dollar amount threshold) (89 FR 93980 through 93982). If the hospital's cost of furnishing a service exceeds both the multiplier threshold and the fixed-dollar threshold, the outlier payment is calculated as 50 percent of the amount by which the hospital's cost of furnishing the service exceeds 1.75 times the APC payment amount. Beginning with CY 2009 payments, outlier payments are subject to a reconciliation process similar to the IPPS outlier reconciliation process for cost reports, as discussed in the CY 2009 OPPS/ASC final rule with comment period (73 FR 68594 through 68599).
It has been our policy to report the actual amount of outlier payments as a percent of total spending in the claims being used to model the OPPS. Our estimate of total outlier payments as a percent of total CY 2024 OPPS payments, using CY 2024 claims available for this final rule with comment period, is approximately 0.82 percent. Therefore, for CY 2024, we estimate that we did not meet the outlier target by 0.18 percent of total aggregated OPPS payments.
For the CY 2026 OPPS/ASC proposed rule, using CY 2024 claims data and CY 2025 payment rates, we estimated that the aggregate outlier payments for CY 2025 would be approximately 0.92 percent of the total CY 2025 OPPS payments (90 FR 33517). We provided estimated CY 2026 outlier payments for hospitals and CMHCs with claims included in the claims data that we used to model impacts in the Hospital-Specific Impacts—Provider-Specific Data file on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient.
2. Outlier Calculation for CY 2026
For CY 2026, we proposed to continue our policy of estimating outlier payments to be 1.0 percent of the estimated aggregate total payments under the OPPS. We proposed that a portion of that 1.0 percent, an amount equal to less than 0.01 percent of outlier payments (or 0.0001 percent of total OPPS payments), would be allocated to CMHCs for partial hospitalization program (PHP) and intensive outpatient program (IOP) outlier payments. This is the amount of estimated outlier payments that would result from the proposed CMHC outlier threshold as a proportion of total estimated OPPS outlier payments. We proposed to continue our outlier policy that if a CMHC's cost for PHP and IOP services exceeds 3.40 times the APC payment rate, the outlier payment would be calculated as 50 percent of the amount ( printed page 53503) by which the cost exceeds 3.40 times the proposed APC payment rate.
For further discussion of CMHC outlier payments, we refer readers to section VIII.C. of this final rule with comment period.
To ensure that the estimated CY 2026 aggregate outlier payments would equal 1.0 percent of estimated aggregate total payments under the OPPS, we proposed that the hospital outlier threshold be set so that outlier payments would be triggered when a hospital's cost of furnishing a service exceeds 1.75 times the APC payment amount and exceeds the APC payment amount plus the fixed-dollar threshold.
We calculated the proposed fixed-dollar threshold using the standard methodology most recently used for CY 2025 (89 FR 93980 through 93982). For purposes of estimating outlier payments for CY 2026, we used the hospital-specific overall ancillary CCRs available in the April 2025 update to the Outpatient Provider-Specific File (OPSF). The OPSF contains provider-specific data, such as the most current CCRs, which are maintained by the MACs and used by the OPPS Pricer to pay claims. The claims that we generally use to model each OPPS update lag by 2 years.
To estimate the CY 2026 proposed hospital outlier payments (90 FR 33841), we inflated the charges on the CY 2024 claims using the same proposed charge inflation factor of 1.1118 that we used to estimate the IPPS fixed-loss cost threshold for the FY 2026 IPPS/LTCH PPS proposed rule (90 FR 18434 through 18436). We used an inflation factor of 1.05440 to estimate CY 2025 charges from the CY 2024 charges reported on CY 2024 claims before applying CY 2025 CCRs to estimate the percent of outliers paid in CY 2025. The proposed methodology for determining these charge inflation factors is discussed in the FY 2026 IPPS/LTCH PPS proposed rule (90 FR 18434). As we stated in the CY 2005 OPPS final rule with comment period (69 FR 65844 through 65846), we believed that the use of the same charge inflation factors is appropriate for the OPPS because, with the exception of the inpatient routine service cost centers, hospitals use the same ancillary and cost centers to capture costs and charges for inpatient and outpatient services.
As noted in the CY 2007 OPPS/ASC final rule with comment period (71 FR 68011), we were concerned that we could systematically overestimate the OPPS hospital outlier threshold if we did not apply a CCR inflation adjustment factor. Therefore, we proposed to apply the same CCR adjustment factor that we proposed to apply for the FY 2026 IPPS outlier calculation to the CCRs used to simulate the proposed CY 2026 OPPS outlier payments to determine the fixed-dollar threshold. Specifically, for CY 2026, we proposed to apply an adjustment factor of 0.970113 to the CCRs that were in the April 2025 OPSF to trend them forward from CY 2025 to CY 2026. The methodology for calculating the proposed CCR adjustment factor is discussed in the FY 2026 IPPS/LTCH PPS proposed rule (90 FR 18434 through 18435).
To model hospital outlier payments for the CY 2026 OPPS/ASC proposed rule, we applied the overall CCRs from the April 2025 OPSF after adjustment (using the proposed CCR inflation adjustment factor of 0.970113 to approximate CY 2026 CCRs) to charges on CY 2024 claims that were adjusted (using the proposed charge inflation factor of 1.1118 to approximate CY 2026 charges). We simulated aggregated CY 2024 hospital outlier payments using these costs for several different fixed-dollar thresholds, holding the 1.75 multiplier threshold constant and assuming that outlier payments would continue to be made at 50 percent of the amount by which the cost of furnishing the service would exceed 1.75 times the APC payment amount, until the total outlier payments equaled 1.0 percent of aggregated estimated total CY 2026 OPPS payments. We estimated that a proposed fixed-dollar threshold of $6,450 combined with the proposed multiplier threshold of 1.75 times the APC payment rate, would allocate 1.0 percent of aggregated total OPPS payments to outlier payments for CY 2026. For CMHCs, we proposed that, if a CMHC's cost for partial hospitalization or intensive outpatient services exceeds 3.40 times the APC payment rate, the outlier payment would be calculated as 50 percent of the amount by which the cost exceeds 3.40 times the APC payment rate.
Section 1833(t)(17)(A) of the Act, which applies to hospitals, as defined under section 1886(d)(1)(B) of the Act, requires that hospitals that fail to report data required for the quality measures selected by the Secretary, in the form and manner required by the Secretary under section 1833(t)(17)(B) of the Act, incur a 2.0 percentage point reduction to their OPD fee schedule increase factor; that is, the annual payment update factor. The application of a reduced OPD fee schedule increase factor results in reduced national unadjusted payment rates that would apply to certain outpatient items and services furnished by hospitals that are required to report outpatient quality data and that fail to meet the Hospital Outpatient Quality Reporting (OQR) Program requirements. For hospitals that fail to meet the Hospital OQR Program requirements, we proposed to continue the policy that we implemented in CY 2010 that the hospitals' costs would be compared to the reduced payments for purposes of outlier eligibility and payment calculation. For more information on the Hospital OQR Program, we refer readers to section XV. of this final rule with comment period.
We received one public comment in support of our proposal. After consideration of the public comment we received, we are finalizing our proposal, without modification, to continue to our policy of estimating outlier payments to be 1.0 percent of the estimated aggregate total payments under the OPPS and to use our established methodology to set the OPPS outlier fixed-dollar loss threshold for CY 2026.
3. Final Outlier Calculation for CY 2026
Consistent with historical practice, we used updated data for this final rule with comment period for outlier calculations. For CY 2026, we are applying the overall ancillary CCRs from the July 2025 OPSF file after adjustment (using the CCR adjustment factor of 0.956081 to approximate CY 2026 CCRs) to charges on CY 2024 claims that were adjusted using a charge inflation factor of 1.11313 to approximate CY 2026 charges. These are the same CCR adjustment and charge inflation factors that were used to set the IPPS fixed-dollar thresholds for the FY 2026 IPPS/LTCH PPS final rule (90 FR 37227). We simulated aggregated CY 2026 hospital outlier payments using these costs for several different fixed-dollar thresholds, holding the 1.75 multiple-threshold constant and assuming that outlier payments will continue to be made at 50 percent of the amount by which the cost of furnishing the service would exceed 1.75 times the APC payment amount, until the total outlier payment equaled 1.0 percent of aggregated estimated total CY 2026 OPPS payments. We estimate that a final fixed-dollar threshold of $6,225 combined with the multiple threshold of 1.75 times the APC payment rate, will allocate 1.0 percent of aggregated total OPPS payments to outlier payments.
For CMHCs, if a CMHC's cost for partial hospitalization or intensive outpatient services exceeds 3.40 times the APC payment rate, the outlier payment will be calculated as 50 percent of the amount by which the cost ( printed page 53504) exceeds the 3.40 times the APC payment rate.
H. Calculation of an Adjusted Medicare Payment From the National Unadjusted Medicare Payment
The national unadjusted payment rate is the payment rate for most APCs before accounting for the wage index adjustment or any applicable adjustments. The basic methodology for determining prospective payment rates for HOPD services under the OPPS is set forth in existing regulations at 42 CFR part 419, subparts C and D. For this final rule with comment period, the payment rate for most services and procedures for which payment is made under the OPPS is the product of the conversion factor calculated in accordance with section II.B. of this final rule with comment period and the relative payment weight described in section II.A. of this final rule with comment period. The national unadjusted payment rate for most APCs contained in Addendum A to this final rule with comment period (which is available on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient-pps/quarterly-addenda-updates and for most HCPCS codes to which separate payment under the OPPS has been assigned in Addendum B to this final rule with comment period (which is available on the CMS website, see link above) is calculated by multiplying the final CY 2026 scaled weight for the APC by the CY 2026 conversion factor.
We note that section 1833(t)(17) of the Act, which applies to hospitals, as defined under section 1886(d)(1)(B) of the Act, requires that hospitals that fail to submit data required to be submitted on quality measures selected by the Secretary, in the form and manner and at a time specified by the Secretary, incur a reduction of 2.0 percentage points to their OPD fee schedule increase factor, that is, the annual payment update factor. The application of a reduced OPD fee schedule increase factor results in reduced national unadjusted payment rates that apply to certain outpatient items and services provided by hospitals that are required to report outpatient quality data and that fail to meet the Hospital OQR Program requirements. For further discussion of the payment reduction for hospitals that fail to meet the requirements of the Hospital OQR Program, we refer readers to section XIV. of this final rule with comment period.
Below we demonstrate the steps used to determine the APC payments that will be made in a CY under the OPPS to a hospital that fulfills the Hospital OQR Program requirements and to a hospital that fails to meet the Hospital OQR Program requirements for a service that has any of the following status indicator assignments: “J1,” “J2,” “P,” “Q1,” “Q2,” “Q3,” “Q4,” “R,” “S,” “T,” “U,” or “V” (as defined in Addendum D1 to this final rule with comment period, which is available via the internet on the CMS website), in a circumstance in which the multiple procedure discount does not apply, the procedure is not bilateral, and conditionally packaged services (status indicator of “Q1” and “Q2”) qualify for separate payment. We note that, although blood and blood products with status indicator “R” and brachytherapy sources with status indicator “U” are not subject to wage adjustment, they are subject to reduced payments when a hospital fails to meet the Hospital OQR Program requirements.
Individual providers interested in calculating the payment amount that they would receive for a specific service from the national unadjusted payment rates presented in Addenda A and B to this final rule with comment period (which are available via the internet on the CMS website) should follow the formulas presented in the following steps. For purposes of the payment calculations below, we refer to the national unadjusted payment rate for hospitals that meet the requirements of the Hospital OQR Program as the “full” national unadjusted payment rate. We refer to the national unadjusted payment rate for hospitals that fail to meet the requirements of the Hospital OQR Program as the “reduced” national unadjusted payment rate. The reduced national unadjusted payment rate is calculated by multiplying the reporting ratio of 0.9805 times the “full” national unadjusted payment rate. The national unadjusted payment rate used in the calculations below is either the full national unadjusted payment rate or the reduced national unadjusted payment rate, depending on whether the hospital met its Hospital OQR Program requirements to receive the full CY 2025 OPPS fee schedule increase factor.
Step 1. Calculate 60 percent (the labor-related portion) of the national unadjusted payment rate. Since the initial implementation of the OPPS, we have used 60 percent to represent our estimate of that portion of costs attributable, on average, to labor. We refer readers to the April 7, 2000 OPPS final rule with comment period (65 FR 18496 through 18497) for a detailed discussion of how we derived this percentage. During our regression analysis for the payment adjustment for rural hospitals in the CY 2006 OPPS final rule with comment period (70 FR 68553), we confirmed that this labor-related share for hospital outpatient services is appropriate.
The formula below is a mathematical representation of Step 1 and identifies the labor-related portion of a specific payment rate for a specific service.
X is the labor-related portion of the national unadjusted payment rate.
X = .60 * (national unadjusted payment rate).
Step 2. Determine the wage index area in which the hospital is located and identify the wage index level that applies to the specific hospital. The wage index values assigned to each area would reflect the geographic statistical areas (which are based upon OMB standards) to which hospitals are assigned for FY 2026 under the IPPS, reclassifications through the Medicare Geographic Classification Review Board (MGCRB), section 1886(d)(8)(B) “Lugar” hospitals, and reclassifications under section 1886(d)(8)(E) of the Act, as implemented in § 412.103 of the regulations. For CY 2026, we proposed to apply for the CY 2026 OPPS wage index any adjustments for the FY 2026 IPPS post-reclassified wage index, including, but not limited to, the rural floor adjustment and a wage index floor of 1.00 in frontier states, in accordance with section 10324 of the Affordable Care Act of 2010. For further discussion of the wage index we are applying for the CY 2026 OPPS, including the low wage index hospital policy, we refer readers to section II.C. of this final rule with comment period.
Step 3. Adjust the wage index of hospitals located in certain qualifying counties that have a relatively high percentage of hospital employees who reside in the county, but who work in a different county with a higher wage index, in accordance with section 505 of the Medicare Prescription Drug, Improvement, and Modernization Act of 2003 (Pub. L. 108-173). Addendum L to this final rule with comment period (which is available via the internet on the CMS website) contains the qualifying counties and the associated wage index increase developed for the final FY 2026 IPPS wage index, which are listed in Table 3 associated with the FY 2026 IPPS final rule and available via the internet on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps. (Click on the link on the left side of the screen titled “FY 2026 IPPS Final Rule Home Page” and select “FY 2026 Final Rule Tables.”) This step is to be followed only if the ( printed page 53505) hospital is not reclassified or redesignated under section 1886(d)(8) or section 1886(d)(10) of the Act.
Step 4. Multiply the applicable wage index determined under Steps 2 and 3 by the amount determined under Step 1 that represents the labor-related portion of the national unadjusted payment rate.
The formula below is a mathematical representation of Step 4 and adjusts the labor-related portion of the national unadjusted payment rate for the specific service by the wage index.
Xais the labor-related portion of the national unadjusted payment rate (wage adjusted).
Xa = labor-portion of the national unadjusted payment rate * applicable wage index.
Step 5. Calculate 40 percent (the nonlabor-related portion) of the national unadjusted payment rate and add that amount to the resulting product of Step 4. The result is the wage index adjusted payment rate for the relevant wage index area.
The formula below is a mathematical representation of Step 5 and calculates the remaining portion of the national payment rate, the amount not attributable to labor, and the adjusted payment for the specific service.
Yis the nonlabor-related portion of the national unadjusted payment rate.
Y = 0.40 * (national unadjusted payment rate).
Step 6. If a provider is an SCH, as set forth in the regulations at § 412.92, or an EACH, which is considered to be an SCH under section 1886(d)(5)(D)(iii)(III) of the Act, and located in a rural area, as defined in § 412.64(b), or is treated as being located in a rural area under § 412.103, multiply the wage index adjusted payment rate by 1.071 to calculate the total payment.
The formula below is a mathematical representation of Step 6 and applies the rural adjustment for rural SCHs.
Adjusted Medicare Payment (SCH or EACH) = Adjusted Medicare Payment * 1.071.
Step 7. The adjusted payment rate is the sum of the wage adjusted labor-related portion of the national unadjusted payment rate and the nonlabor-related portion of the national unadjusted payment rate.
Xais the labor-related portion of the national unadjusted payment rate (wage adjusted).
Yis the nonlabor-related portion of the national unadjusted payment rate.
Adjusted Medicare Payment = Xa + Y
We are providing examples below of the calculation of both the full and reduced national unadjusted payment rates that would apply to certain outpatient items and services performed by hospitals that meet and that fail to meet the Hospital OQR Program requirements, using the steps outlined previously. For purposes of this example, we are using a provider that is located in Brooklyn, New York that is assigned to CBSA 35614. This provider bills one service that is assigned to APC 5071 (Level 1 Excision/Biopsy/Incision and Drainage). The final CY 2026 full national unadjusted payment rate for APC 5071 is $723.47. The final reduced national adjusted payment rate for APC 5071 for a hospital that fails to meet the Hospital OQR Program requirements is $709.36. This reduced rate is calculated by multiplying the reporting ratio of 0.9805 by the full unadjusted payment rate for APC 5071.
Step 1. The labor-related portion of the final full national unadjusted payment is approximately $434.08 (0.60 * $723.47). The labor-related portion of the final reduced national adjusted payment is approximately $425.62 (0.60 * $709.36).
Step 2 & 3. The FY 2026 wage index for a provider located in CBSA 35614 in New York, which includes the adoption of the final IPPS 2026 wage index policies, is 1.3697.
Step 4. The wage adjusted labor-related portion of the final full national unadjusted payment is approximately $594.56 ($434.08 * 1.3697). The wage adjusted labor-related portion of the final reduced national adjusted payment is approximately $582.97 ($425.62 * 1.3697).
Step 5. The nonlabor-related portion of the final full national unadjusted payment is approximately $289.39 (0.40 * $723.47). The nonlabor-related portion of the final reduced national adjusted payment is approximately $283.74(0.40 * $709.36).
Step 6. For this example of a provider located in Brooklyn, New York, the rural adjustment for rural SCHs does not apply.
Step 7. The sum of the labor-related and nonlabor-related portions of the final full national unadjusted payment is approximately $883.95 ($594.56 + $289.39). The sum of the portions of the final reduced national adjusted payment is approximately $866.71 ($582.97 + $283.74) as shown in Table 11.
We did not receive any public comments on these steps under the methodology that we included in the CY 2026 OPPS/ASC proposed rule to determine the APC payments for CY 2026. Therefore, we are using the steps in the methodology specified above to demonstrate the calculation of the final CY 2026 OPPS payments using the same parameters.
I. Beneficiary Copayments
1. Background
Section 1833(t)(3)(B) of the Act requires the Secretary to set rules for determining the unadjusted copayment amounts to be paid by beneficiaries for covered OPD services. Section 1833(t)(8)(C)(ii) of the Act specifies that the Secretary must reduce the national unadjusted copayment amount for a covered OPD service (or group of such services) furnished in a year in a manner so that the effective copayment rate (determined on a national unadjusted basis) for that service in the year does not exceed a specified percentage. As specified in section 1833(t)(8)(C)(ii)(V) of the Act, the effective copayment rate for a covered OPD service paid under the OPPS in CY 2006, and in CYs thereafter, shall not exceed 40 percent of the APC payment rate.
Section 1833(t)(3)(B)(ii) of the Act provides that, for a covered OPD service (or group of such services) furnished in a year, the national unadjusted copayment amount cannot be less than 20 percent of the OPD fee schedule ( printed page 53506) amount. However, section 1833(t)(8)(C)(i) of the Act limits the amount of beneficiary copayment that may be collected for a procedure (including items such as drugs and biologicals) performed in a year to the amount of the inpatient hospital deductible for that year.
Section 4104 of the Affordable Care Act eliminated the Medicare Part B coinsurance for preventive services furnished on and after January 1, 2011, that meet certain requirements, including flexible sigmoidoscopies and screening colonoscopies, and waived the Part B deductible for screening colonoscopies that become diagnostic during the procedure. For a discussion of the changes made by the Affordable Care Act with regard to copayments for preventive services furnished on and after January 1, 2011, we refer readers to section XII.B. of the CY 2011 OPPS/ASC final rule with comment period (75 FR 72013).
Section 122 of the Consolidated Appropriations Act (CAA), 2021 (Pub. L. 116-260), Waiving Medicare Coinsurance for Certain Colorectal Cancer Screening Tests, amended section 1833(a) of the Act to offer a special coinsurance rule for screening flexible sigmoidoscopies and screening colonoscopies, regardless of the code that is billed for the establishment of a diagnosis as a result of the test, or for the removal of tissue or other matter or other procedure, that is furnished in connection with, as a result of, and in the same clinical encounter as the colorectal cancer screening test. We refer readers to section “X.B. Changes to Beneficiary Coinsurance for Certain Colorectal Cancer Screening Tests”, of the CY 2022 OPPS/ASC final rule with comment period for the full discussion of this policy (86 FR 63740 through 63743). Under the regulation at 42 CFR 410.152(l)(5)(i)(B), the Medicare Part B payment percentage for colorectal cancer screening tests described in the regulation at § 410.37(j) that are furnished in CY 2023 through CY 2026 is 85 percent, with beneficiary coinsurance equal to 15 percent.
On August 16, 2022, the Inflation Reduction Act of 2022 (IRA) (Pub. L. 117-169) was signed into law. Section 11101(a) of the IRA amended section 1847A of the Act by adding a new subsection (i), which requires the payment of rebates into the Supplementary Medical Insurance Trust Fund for Part B rebatable drugs if the payment limit amount exceeds the inflation-adjusted payment amount, which is calculated as set forth in section 1847A(i)(3)(C) of the Act. The provisions of section 11101 of the IRA were initially implemented through program instruction, as permitted under section 1847A(c)(5)(C) of the Act. On February 9, 2023 and December 14, 2023, we issued initial [9] and revised [10] guidance, respectively, implementing the Medicare Part B Inflation Rebate Program, including the computation of inflation-adjusted beneficiary coinsurance under section 1847A(i)(5) of the Act and amounts paid under section 1833(a)(1)(EE) of the Act.[11] For additional information regarding implementation of section 11101 of the IRA, please see the inflation rebates resources page at https://www.cms.gov/inflation-reduction-act-and-medicare/inflation-rebates-medicare.
Section 11101(b) of the IRA amended sections 1833(i) and 1833(t)(8) of the Act by adding a new paragraph (9) and subparagraph (F), respectively. Section 1833(i)(9) of the Act requires under the ASC payment system that, in the case of a Part B rebatable drug for which payment is not packaged into a payment for a service, in lieu of calculation of coinsurance that would otherwise apply under the ASC payment system, the provisions of section 1847A(i)(5) of the Act shall, as determined appropriate by the Secretary, apply for calculation of beneficiary coinsurance in the same manner as the provisions of section 1847A(i)(5) of the Act apply under that section. Similarly, section 1833(t)(8)(F) of the Act requires under the OPPS that in the case of a Part B rebatable drug (except for a drug that has no copayment applied under subparagraph (E) of such section or for which payment is packaged into the payment for a covered OPD service or group of services), in lieu of the calculation of the copayment amount that would otherwise apply under the OPPS, the provisions of section 1847A(i)(5) of the Act shall, as determined appropriate by the Secretary, apply in the same manner as the provisions of section 1847A(i)(5) of the Act apply under that section. Section 1847A(i)(5) of the Act requires that for Part B rebatable drugs, as defined in section 1847A(i)(2)(A) of the Act, furnished on or after April 1, 2023, in quarters in which the payment amount described in section 1847A(i)(3)(A)(ii)(I) of the Act (or, in the case of selected drugs described under section 1192(c) of the Act, the payment amount described in section 1847A(b)(1)(B) of the Act), exceeds the inflation-adjusted payment amount determined in accordance with section 1847A(i)(3)(C) of the Act, the coinsurance will be 20 percent of the inflation-adjusted payment amount for such quarter (hereafter, the inflation-adjusted coinsurance amount). This inflation-adjusted coinsurance amount is applied as a percent, as determined by the Secretary, to the payment amount that would otherwise apply for such calendar quarter in accordance with section 1847A(b)(1)(B) or (C) of the Act, as applicable, including in the case of a selected drug described under section 1192(c) of the Act.
Paragraph (9) of section 1833(i) of the Act and subparagraph (F) of section 1833(t)(8) of the Act, as added by section 11101(b) of the IRA, also provide that in lieu of the amounts of payment otherwise applicable under the ASC payment system and the OPPS, the provisions of paragraph (1)(EE) of subsection (a) of section 1833 of the Act shall apply, as determined appropriate by the Secretary. Section 11101(b) of the IRA amended section 1833(a)(1) of the Act by adding a new subparagraph (EE), which requires that if the payment amount under section 1847A(i)(3)(A)(ii)(I) of the Act or, in the case of a selected drug described under section 1192(c) of the Act, the payment amount described in section 1847A(b)(1)(B) of the Act, for that drug exceeds the inflation-adjusted payment amount for a Part B rebatable drug, the Part B payment amount would, subject to the Part B deductible and sequestration, equal the difference between such payment amount and the inflation-adjusted coinsurance amount. Consistent with the policy adopted in section 40 of the revised Medicare Part B Drug Inflation Rebate Guidance, the calculation to determine the applicable beneficiary coinsurance amount would not be adjusted for sequestration. CMS codified the Medicare payment for Part B rebatable drugs in the CY 2024 PFS final rule by adding new paragraph (m) to § 410.152 (88 FR 79043).
In the CY 2024 OPPS/ASC final rule with comment period (88 FR 81594), we codified the OPPS program payment and cost as required by section 1833(t)(8)(F) of the Act by adding a new paragraph (e) to § 419.41, which cross-references the regulations adopted in the CY 2024 PFS final rule (§§ 410.152(m) and 489.30(b)(6)). We ( printed page 53507) also amended the regulation text to reflect our longstanding policies for calculating the Medicare program payment and cost sharing amounts for separately payable drugs and biologicals by adding a new paragraph (d) to § 419.41. Similarly, we codified the ASC cost sharing amounts for Part B rebatable drugs as required by section 1833(i)(9) of the Act by revising § 416.172(d) to include a cross-reference to 42 CFR 489.30(b)(6), which codified the cost sharing amounts for Part B rebatable drugs with prices increasing at a rate faster than inflation.
In the CY 2025 PFS final rule (89 FR 98228 through 98275), we codified regulations implementing section 11101 of the IRA in newly added 42 CFR part 427, chapter IV, including new provisions at §§ 427.200 and 427.201 to codify the policies regarding the computation of the inflation-adjusted beneficiary coinsurance, defined in § 427.200, for Part B rebatable drugs as required by section 1847A(i)(5) of the Act. As finalized, § 427.201(a) establishes that CMS will use the methodology established in such section to calculate the inflation-adjusted beneficiary coinsurance and associated adjusted Medicare payment percentage and incorporates references to the existing provisions at §§ 410.152(m), 419.41(e), and 489.30(b)(6). Section 427.201(c) provides that any category of products that is excluded from the identification of Part B rebatable drugs at § 427.101(b) is not subject to the inflation-adjusted beneficiary coinsurance. Examples of these excluded products include separately payable radiopharmaceuticals, skin substitute products, and qualifying biosimilar biological products.
Section 427.201(b) sets forth the calculation of the inflation-adjusted beneficiary coinsurance. We will compare the payment amount in paragraph (b)(3) of such section to the inflation-adjusted payment amount for an applicable calendar quarter; if the payment amount exceeds the inflation-adjusted payment amount, the inflation-adjusted beneficiary coinsurance is calculated by multiplying the inflation-adjusted payment amount by 0.20. Section 427.201(b)(3) specifies that CMS will use the published payment amount in quarterly pricing files [12, 13, 14] to determine if a Part B rebatable drug should have an adjusted beneficiary coinsurance. If so, such adjusted beneficiary coinsurance shall be equal to 20 percent of the inflation-adjusted payment amount as described in section 1847A(i)(3)(C) of the Act for a calendar quarter. This approach deviates from the rebate calculation approach set forth in § 427.302, which relies on the specified amount defined at § 427.20 even when the specified amount and the published payment amount in quarterly pricing files differ.
We note that the cost sharing amounts of rebatable drugs paid under the OPPS published in the quarterly Addendum A and B updates reflect the inflation-adjusted coinsurance applied as a percent of the payment amount that would otherwise apply in accordance with section 1847A(b)(1)(B) or (C) of the Act, as determined by the Secretary pursuant to 1847A(i)(5) of the Act using the methodology in § 427.201. As we explained in the CY 2025 PFS final rule (89 FR 98237), this policy is intended to hold beneficiaries harmless in situations where the payment amount is calculated differently from the specified amount, and we believe this approach is consistent with the statutory language and appropriately reflects the differences in the statutory text of section 1847A(i)(5) of the Act, which sets forth the payment amount that is used to determine whether coinsurance should be adjusted, and section 1847A(i)(3)(A) of the Act, which sets forth the “specified amount” used to determine rebate amounts. We refer readers to the full discussion at 89 FR 98237 and 98238 for additional details.
2. OPPS Copayment Policy
For CY 2026, we proposed to determine copayment amounts for new and revised APCs using the same methodology that we implemented beginning in CY 2004. We refer readers to the November 7, 2003 OPPS final rule with comment period for a discussion of that methodology (68 FR 63458). In addition, we proposed to use the same standard rounding principles that we have historically used in instances where the application of our standard copayment methodology would result in a copayment amount that is less than 20 percent and cannot be rounded, under standard rounding principles, to 20 percent. We refer readers to the CY 2008 OPPS/ASC final rule with comment period (72 FR 66687) in which we discuss our rationale for applying these rounding principles. The final national unadjusted copayment amounts for services payable under the OPPS that would be effective January 1, 2026, are included in Addenda A and B to this final rule with comment period (which are available via the internet on the CMS website).
As discussed in section XIV.E. of this final rule with comment period, for CY 2026, the Medicare beneficiary's minimum unadjusted copayment and national unadjusted copayment for a service to which a reduced national unadjusted payment rate applies will equal the product of the reporting ratio and the national unadjusted copayment, or the product of the reporting ratio and the minimum unadjusted copayment, respectively, for the service.
We note that OPPS copayments may increase or decrease each year based on changes in the calculated APC payment rates, due to updated cost report and claims data, and any changes to the OPPS cost modeling process. However, as described in the CY 2004 OPPS final rule with comment period, the development of the copayment methodology generally moves beneficiary copayments closer to 20 percent of OPPS APC payments (68 FR 63458 through 63459).
In the CY 2004 OPPS final rule with comment period (68 FR 63459), we adopted a new methodology to calculate unadjusted copayment amounts in situations including reorganizing APCs, and we finalized the following rules to determine copayment amounts in CY 2004 and subsequent years.
- When an APC group consists solely of HCPCS codes that were not paid under the OPPS the prior year because they were packaged or excluded or are new codes, the unadjusted copayment amount would be 20 percent of the APC payment rate.
- If a new APC that did not exist during the prior year is created and consists of HCPCS codes previously assigned to other APCs, the copayment amount is calculated as the product of the APC payment rate and the lowest coinsurance percentage of the codes comprising the new APC.
- If no codes are added to or removed from an APC and, after recalibration of its relative payment weight, the new payment rate is equal to orgreater than the prior year's rate, the copayment amount remains constant (unless the resulting coinsurance percentage is less than 20 percent).
- If no codes are added to or removed from an APC and, after recalibration of its relative payment weight, the new payment rate isless than the prior year's rate, the copayment amount is calculated as the product of the new payment rate and the prior year's coinsurance percentage.
- If HCPCS codes are added to or deleted from an APC and, after ( printed page 53508) recalibrating its relative payment weight, holding its unadjusted copayment amount constant results in a decrease in the coinsurance percentage for the reconfigured APC, the copayment amount would not change (unless retaining the copayment amount would result in a coinsurance rate less than 20 percent).
- If HCPCS codes are added to an APC and, after recalibrating its relative payment weight, holding its unadjusted copayment amount constant results in an increase in the coinsurance percentage for the reconfigured APC, the copayment amount would be calculated as the product of the payment rate of the reconfigured APC and the lowest coinsurance percentage of the codes being added to the reconfigured APC.
We noted in the CY 2004 OPPS final rule with comment period that we would seek to lower the copayment percentage for a service in an APC from the prior year if the copayment percentage was greater than 20 percent. We noted that this principle was consistent with section 1833(t)(8)(C)(ii) of the Act, which accelerates the reduction in the national unadjusted coinsurance rate so that beneficiary liability will eventually equal 20 percent of the OPPS payment rate for all OPPS services to which a copayment applies, and with section 1833(t)(3)(B) of the Act, which achieves a 20 percent copayment percentage when fully phased in and gives the Secretary the authority to set rules for determining copayment amounts for new services. We further noted that the use of this methodology would, in general, reduce the beneficiary coinsurance rate and copayment amount for APCs for which the payment rate changes as the result of the reconfiguration of APCs and/or recalibration of relative payment weights (68 FR 63459).
We did not receive any public comments on our proposal and we are finalizing our proposal to determine copayment amounts for new and revised APCs using the same methodology that we implemented beginning in CY 2004. The finalized national unadjusted copayment amounts for services payable under the OPPS that will be effective January 1, 2026, are included in Addenda A and B to the CY 2026 OPPS/ASC final rule (which are available on the CMS website).
3. Calculation of an Adjusted Copayment Amount for an APC Group
Individuals interested in calculating the national copayment liability for a Medicare beneficiary for a given service provided by a hospital that met or failed to meet its Hospital OQR Program requirements should follow the formulas presented in the following steps.
Step 1. Calculate the beneficiary payment percentage for the APC by dividing the APC's national unadjusted copayment by its proposed payment rate. For example, using APC 5071, $144.69 is 20 percent of the full national unadjusted payment rate of $723.47. For APCs with only a minimum unadjusted copayment in Addenda A and B to this final rule with comment period (which are available via the internet on the CMS website), the beneficiary payment percentage is 20 percent.
The formula below is a mathematical representation of Step 1 and calculates the national copayment as a percentage of national payment for a given service.
B is the beneficiary payment percentage.
B = National unadjusted copayment for APC/national unadjusted payment rate for APC.
Step 2. Calculate the appropriate wage-adjusted payment rate for the APC for the provider in question, as indicated in Steps 2 through 4 under section II.H. of this final rule with comment period. Calculate the rural adjustment for eligible providers, as indicated in Step 6 under section II.H. of this final rule with comment period.
Step 3. Multiply the percentage calculated in Step 1 by the payment rate calculated in Step 2. The result is the wage-adjusted copayment amount for the APC.
The formula below is a mathematical representation of Step 3 and applies the beneficiary payment percentage to the adjusted payment rate for a service calculated under section II.H. of this final rule with comment period, with and without the rural adjustment, to calculate the adjusted beneficiary copayment for a given service.
Wage-adjusted copayment amount for the APC = Adjusted Medicare Payment * B.
Wage-adjusted copayment amount for the APC (SCH or EACH) = (Adjusted Medicare Payment * 1.071) * B.
Step 4. For a hospital that failed to meet its Hospital OQR Program requirements, multiply the copayment calculated in Step 3 by the reporting ratio of 0.9805.
The unadjusted copayments for services payable under the OPPS that would be effective January 1, 2026, are shown in Addenda A and B to this final rule with comment period (which are available via the CMS website). We note that the final national unadjusted payment rates and copayment rates shown in Addenda A and B to this final rule with comment period reflect the CY 2026 OPD fee schedule increase factor discussed in section II.B. of this final rule with comment period.
In addition, as noted earlier, section 1833(t)(8)(C)(i) of the Act limits the amount of beneficiary copayment that may be collected for a procedure performed in a year to the amount of the inpatient hospital deductible for that year.
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: Several commenters requested CMS reconsider the coinsurance policy for diagnostic radiopharmaceuticals on pass-through status, that are above the radiopharmaceutical packaging threshold. The commenters disagreed that these radiopharmaceuticals are no longer considered policy-packaged under § 419.2(b)(15) and instead, treated like separately payable drugs assigned to an APC, and subject to a coinsurance. The commenters believe the coinsurance on diagnostic radiopharmaceuticals on pass-through status will be too financially burdensome for beneficiaries and undermines the intent of the statute and the advantages of having pass-through status. The commenters stated if both pass-through and non-pass-through diagnostic radiopharmaceuticals are subject to coinsurance, the commenters believe the distinction between these categories become meaningless. The commenters request CMS to clarify how the current policy and the cost-sharing calculations for diagnostic radiopharmaceuticals meets the intent of the statute.
Response: We disagree with the commenter's interpretation that these diagnostic radiopharmaceuticals would remain policy-packaged under 42 CFR 419.2(b)(15) after we implemented the CY 2025 diagnostic radiopharmaceuticals separate payment policy (89 FR 93948). We consider diagnostic radiopharmaceuticals to be drugs for purposes of pass-through payment (89 FR 94226). We note the copayment for pass-through drugs depends on their OPPS payment status absent pass-through status. For example, if a drug is policy-packaged, the pass-through payment amount is equal to a payment rate calculated using the ASP methodology (89 FR 94226). In accordance with section 1833(t)(8) of the Act, there is no copayment on the pass-through payment amount. Therefore, policy packaged drugs have a zero-dollar copayment amount when granted OPPS drug pass-through status. ( printed page 53509) However, for those drugs that do not fall into the category of policy packaged drugs, those drugs are separately payable drugs in the OPPS. The pass-through amount is the difference between the amount authorized under section 1842(o) of the Act, which is generally ASP plus 6 percent, and the portion of the otherwise applicable OPD fee schedule, which is also generally ASP plus 6 percent, is $0 (89 FR 94225 through 94226). We reiterate that the copayment for pass-through drugs depends on their OPPS payment status absent pass-through status. Therefore, the copay amount, absent pass-through status, under the OPD fee are still subject to the co-insurance established by section 1833(t)(3)(B) of the Act and § 419.41(d) for separately payable drugs. We refer readers to the CY 2026 OPPS/ASC proposed rule for further discussion on pass-through payment for drugs, biologicals, and radiopharmaceuticals (90 FR 33614).
We also note that the co-insurance for a separately payable drug under the OPPS shall not exceed the amount of inpatient hospital deductible for that year.
Comment: One commenter suggested beneficiaries should not be responsible for drug pricing increases and that CMS should limit or eliminate cost-sharing for beneficiaries, citing the unintended consequences and negative effects on access to health care and health outcomes (such as reduced use of medically necessary services), and increased use of emergency rooms.
Response: We thank the commenter for the input. We note that the beneficiary copayment is established by section 1833(t)(3)(B) of the Act and § 419.41(d). Section 1833(t)(8)(F) of the Act provides for an adjustment to the beneficiary coinsurance for Part B drugs and biologicals that are not packaged into payment for an OPD service with prices that have increased faster than the rate of inflation beginning April 1, 2023. In the CY 2024 OPPS/ASC final rule with comment period and the CY 2024 PFS final rule, we codified this inflation-adjusted coinsurance amount at §§ 419.41(e), 410.152(m), and 489.30(b)(6), respectively. For these drugs and biologicals, the beneficiary coinsurance is 20 percent of the inflation-adjusted payment amount, which is less than what the beneficiary would pay in coinsurance otherwise. Therefore, beneficiaries are insulated from coinsurance amounts calculated based on drug prices that outpace inflation. More information about the beneficiary coinsurance adjustment and the Medicare Part B Inflation Rebate Program is available at https://www.cms.gov/inflation-reduction-act-and-medicare/inflation-rebates-medicare.
III. OPPS Ambulatory Payment Classification (APC) Group Policies
A. OPPS Treatment of New and Revised HCPCS Codes
Payments for OPPS procedures, services, and items are generally based on medical billing codes, specifically, Healthcare Common Procedure Coding System (HCPCS) codes, that are reported on hospital outpatient department (HOPD) claims. HCPCS codes are used to report surgical procedures, medical services, items, and supplies under the hospital OPPS. The HCPCS is divided into two principal subsystems, referred to as Level I and Level II of the HCPCS. Level I is comprised of CPT (Current Procedural Terminology) codes, a numeric and alphanumeric coding system that is established and maintained by the American Medical Association (AMA), and consists of Category I, II, III, MAAA, and PLA CPT codes. Level II, which is established and maintained by CMS, is a standardized coding system that is used primarily to identify products, supplies, and services not included in the CPT codes. Together, Level I and II HCPCS codes are used to report procedures, services, items, and supplies under the OPPS payment system. Specifically, we recognize the following codes on OPPS claims:
- Category I CPT codes, which describe surgical procedures, diagnostic and therapeutic services, and vaccine codes;
- Category III CPT codes, which describe new and emerging technologies, services, and procedures;
- MAAA CPT codes, which describe laboratory multianalyte assays with algorithmic analyses (MAA);
- PLA CPT codes, which describe proprietary laboratory analyses (PLA) services; and
- Level II HCPCS codes (also known as alpha-numeric codes), which are used primarily to identify drugs, devices, supplies, temporary procedures, and services not described by CPT codes.
The codes are updated and changed throughout the year. CPT and Level II HCPCS code changes that affect the OPPS are published through the annual rulemaking cycle and through the OPPS quarterly update Change Requests (CRs). Generally, these code changes are effective January 1, April 1, July 1, or October 1. CPT code changes are released by the AMA (via their website) while Level II HCPCS code changes are released to the public via the CMS HCPCS website. CMS recognizes the release of new CPT and Level II HCPCS codes outside of the formal rulemaking process via OPPS quarterly update CRs. Based on our review, we assign the new codes to interim status indicators (SIs) and APCs. These interim assignments are finalized in the OPPS/ASC final rules. This quarterly process offers hospitals access to codes that more accurately describe the items or services furnished and provides payment for these items or services in a timelier manner than if we waited for the annual rulemaking process. We solicit public comments on the new CPT and Level II HCPCS codes, status indicators, and APC assignments through our annual rulemaking process.
We note that, under the OPPS, the APC assignment determines the payment rate for an item, procedure, or service. The items, procedures, or services not exclusively paid separately under the hospital OPPS are assigned to appropriate status indicators. Certain payment status indicators provide separate payment while other payment status indicators do not. In section XI. “CY 2026 Payment Status and Comment Indicators” of this final rule with comment period, we discuss the various status indicators and comment indicators used under the OPPS. We also provide a complete list of the status indicators and their definitions in Addendum D1 to this final rule with comment period.
1. April 2025 HCPCS Codes Proposed Rule Comment Solicitation
For the April 2025 update, 104 new HCPCS codes were established and made effective on April 1, 2025. Through the April 2025 OPPS quarterly update CR (Transmittal 13135, Change Request 13993, dated March 20, 2025), we recognized several new HCPCS codes for payment under the OPPS. We solicited public comments on the proposed APC and status indicator assignments for the codes listed in Table 9 (New HCPCS Codes Effective April 1, 2025) of the CY 2026 OPPS/ASC proposed rule (90 FR 33525 through 33528), which are also displayed in Table 12.
We received some public comments on the proposed OPPS APC and SI assignments for the new Level II HCPCS codes that were effective on April 1, 2025. The comments and our responses are addressed in the applicable sections of this final rule with comment period, which include, but are not limited to sections III.C. (New Technology APCs); III.E. (OPPS APC-Specific Policies); and IV. (OPPS Payment for Devices). For ( printed page 53510) those April 2025 codes for which we received no comments, we are finalizing the proposed APC and status indicator assignments as proposed. In addition, in prior years we included the final OPPS status indicators and APC assignments in the coding preamble tables, however, because the same information can be found in Addendum B, we no longer include them in Table 12. Therefore, readers are advised to refer to the OPPS Addendum B for the final OPPS status indicators, APC assignments, and payment rates for all codes reportable under the hospital OPPS. These new codes that were effective April 1, 2025, were assigned to comment indicator “NP” in Addendum B to the CY 2026 OPPS/APC proposed rule to indicate that the codes are assigned to an interim APC assignment and comments would be accepted on their interim APC assignments. The complete list of status indicators and definitions used under the OPPS can be found in Addendum D1 to this final rule with comment period, while the complete list of comment indicators and definitions can be found in Addendum D2 to this final rule with comment period. We note that OPPS Addendum B (OPPS payment file by HCPCS code), Addendum D1 (OPPS Status Indicators), and Addendum D2 (OPPS Comment Indicators) are available via the internet on the CMS website.
( printed page 53511) ( printed page 53512) ( printed page 53513) ( printed page 53514)2. July 2025 HCPCS Codes Proposed Rule Comment Solicitation
For the July 2025 update, 110 new codes were established and made effective July 1, 2025. Through the July 2025 OPPS quarterly update CR (Transmittal 13258, Change Request 14091, dated June 23, 2025) we recognized several new codes for payment and assigned them to appropriate interim OPPS status indicators and APCs. We solicited public comments on the proposed APC and status indicator assignments for the codes listed in Table 10 (New HCPCS Codes Effective July 1, 2025) of the CY 2026 OPPS/ASC proposed rule (90 FR 33529 through 33533), which are also listed in Table 13.
We received some public comments on the proposed OPPS APC and SI assignments for the new Level II HCPCS codes implemented on July 1, 2025. The comments and our responses are addressed in pertinent sections of this final rule with comment period, which include, but are not limited to sections III.C (New Technology APCs); III.E (OPPS APC-Specific Policies); and IV (OPPS Payment for Devices). For those July 1, 2025, codes for which we received no comments, we are finalizing the proposed APC and status indicator assignments. Additionally, we note that in prior years we included the final OPPS status indicators and APC assignments in the coding preamble tables, however, because the same information can be found in Addendum B, we no longer include them in Table 13. Therefore, readers are advised to refer to the OPPS Addendum B for the final OPPS status indicators, APC assignments, and payment rates for all codes reportable under the OPPS. These new codes that were effective July 1, 2025, were assigned to comment indicator “NP” in Addendum B to the CY 2026 OPPS/ASC proposed rule to indicate that the codes are assigned to an interim APC assignment and comments would be accepted on their interim APC assignments. The complete list of status indicators and definitions used under the OPPS can be found in Addendum D1 to this final rule with comment period, while the complete list of comment indicators and definitions can be found in Addendum D2 to this final rule with comment period. We note that OPPS Addendum B (OPPS payment file by HCPCS code), Addendum D1 (OPPS Status Indicators), and Addendum D2 (OPPS Comment Indicators) are available via the internet on the CMS website.
( printed page 53515) ( printed page 53516) ( printed page 53517) ( printed page 53518) ( printed page 53519) ( printed page 53520)3. October 2025 HCPCS Codes Final Rule Comment Solicitation
For the October 2025 update, 100 codes were established and made effective October 1, 2025. Through the October 2025 OPPS quarterly update CR (Transmittal 13425, Change Request 14223, dated September 22, 2025), we recognized several new codes for separate payment and assigned them to appropriate interim OPPS status indicators and APCs. For CY 2026, consistent with our established policy, we proposed in the CY 2026 OPPS/ASC proposed rule (90 FR 33533) that the HCPCS codes that would be effective October 1, 2025, would be flagged with comment indicator “N1” in Addendum B to the CY 2026 OPPS/ASC final rule with comment period to indicate that we have assigned the codes to interim OPPS status indicators for CY 2026. Table 14 lists the codes that were effective October 1, 2025. We note that several of the temporary C-codes have been replaced with permanent J-codes effective January 1, 2026. We are inviting public comments in this final rule with comment period on the interim payment indicators, which will be finalized in the CY 2027 OPPS/ASC final rule with comment period. We note the proposed APC assignments and status indicators for these same codes will be subject to comment in the CY 2027 OPPS/ASC proposed rule with comment period and will be finalized in the CY 2027 OPPS/ASC final rule with comment period.
( printed page 53521) ( printed page 53522) ( printed page 53523) ( printed page 53524) ( printed page 53525)4. January 2026 HCPCS Codes
a. New Level II HCPCS Codes Final Rule Comment Solicitation
Consistent with past practice, we are soliciting comments on the new Level II HCPCS codes that will be effective January 1, 2026, in the CY 2026 OPPS/ASC final rule with comment period, thereby allowing us to finalize the status indicators and APC assignments for the codes in the CY 2027 OPPS/ASC final rule with comment period. Unlike the CPT codes that are effective January 1 and are included in the OPPS/ASC proposed rules, and except for the proposed new C-codes and G-codes listed in Addendum O of the CY 2026 OPPS/ASC proposed rule, most Level II HCPCS codes are not released until sometime around November to be effective January 1. Because these codes are not available until November, we were unable to include them in the OPPS/ASC proposed rules. Consequently, for CY 2026, we proposed to include the new Level II HCPCS codes that will be effective January 1, 2026 (that would be incorporated in the January 2026 OPPS quarterly update CR), in Addendum B to the CY 2026 OPPS/ASC final rule with comment period. Specifically, for CY 2026, we proposed to continue our established policy of assigning comment indicator “N1” in Addendum B to this final rule with comment period to the new HCPCS codes that will be effective January 1, 2026, to indicate that we are assigning them an interim status indicator, which is subject to public comment. We are inviting public comments in this final rule with comment period on the status indicators and APC assignments, which would then be finalized in the CY 2027 OPPS/ASC final rule with comment period. Similar to the codes effective October 1, 2025, the proposed APC assignments and status indicators for these new Level II HCPCS codes that will be effective January 1, 2026, will also be subject to comment in the CY 2027 OPPS/ASC proposed rule, and will be finalized in the CY 2027 OPPS/ASC final rule with comment period.
b. New CPT Codes Proposed Rule Comment Solicitation
In the CY 2015 OPPS/ASC final rule with comment period (79 FR 66841 through 66844), we finalized a revised process of assigning APC and status indicators for new and revised Category I and III CPT codes that would be effective January 1. Specifically, for the new/revised CPT codes that we receive in a timely manner from the AMA's CPT Editorial Panel, we finalized our proposal to include the codes that would be effective January 1 in the OPPS/ASC proposed rules, along with proposed APC and status indicator assignments for them, and to finalize the APC and status indicator assignments in the OPPS/ASC final rules beginning with the CY 2016 OPPS update. For those new/revised CPT codes that were received too late for inclusion in the OPPS/ASC proposed rule, we finalized our proposal to establish and use HCPCS G-codes that mirror the predecessor CPT codes and retain the current APC and status indicator assignments for a year until we can propose APC and status indicator assignments in the following year's rulemaking cycle. We note that even if we find that we need to create HCPCS G-codes in place of certain CPT codes for the PFS proposed rule, we do not anticipate that these HCPCS G-codes will always be necessary for OPPS purposes. We will make every effort to include proposed APC and status indicator assignments for all new and revised CPT codes that the AMA makes publicly available in time for us to include them in the proposed rule, and to avoid resorting to use of HCPCS G-codes and the resulting delay in utilization of the most current CPT codes. Also, we finalized our proposal to make interim APC and status indicator assignments for CPT codes that are not available in time for the proposed rule and that describe wholly new services (such as new technologies or new surgical procedures), to solicit public comments in the final rule, and ( printed page 53526) to finalize the specific APC and status indicator assignments for those codes in the following year's rule.
For the CY 2026 OPPS update, we received the CPT codes that will be effective January 1, 2026, from the AMA in time to be included in the CY 2026 OPPS/ASC proposed rule. The new, revised, and deleted CPT codes can be found in Addendum B to the proposed rule (which is available via the internet on the CMS website). We note that the new and revised CPT codes are assigned to comment indicator “NP” in Addendum B to the proposed rule to indicate that the code is new for the next calendar year or the code is an existing code with substantial revision to its code descriptor in the next calendar year as compared to the current calendar year with a proposed APC assignment, and that comments would be accepted on the proposed APC assignment and status indicator.
Further, we noted that the CPT code descriptors that appeared in Addendum B were short descriptors and did not accurately describe the complete procedure, service, or item described by the CPT code. Therefore, we included the 5-digit placeholder codes and the long descriptors for the new and revised CY 2026 CPT codes in Addendum O to the CY 2026 OPPS/ASC proposed rule (which is available via the internet on the CMS website) so that the public could adequately comment on the proposed APCs and SI assignments. The 5-digit placeholder codes were included in Addendum O to the CY 2026 OPPS/ASC proposed rule, specifically under the column labeled “CY 2026 OPPS/ASC Proposed Rule 5-Digit AMA/CMS Placeholder Code.” We noted that the final CPT code numbers would be included in this CY 2026 OPPS/ASC final rule with comment period. We also noted that not every code listed in Addendum O is subject to public comment. For the new and revised Category I and III CPT codes, we requested public comments on only those codes that are assigned comment indicator “NP”.
In summary, in the CY 2026 OPPS/ASC proposed rule, we solicited public comments on the proposed CY 2026 status indicators and APC assignments for the new and revised CPT codes that would be effective January 1, 2026. The CPT codes listed in Addendum B to the CY 2026 OPPS/ASC proposed rule appear with short descriptors only. We listed them again, with long descriptors, in Addendum O to the CY 2026 OPPS/ASC proposed rule. We also proposed to finalize the status indicator and APC assignments for these codes (with their final CPT code numbers) in the CY 2026 OPPS/ASC final rule with comment period. The proposed status indicator and APC assignments for these codes were included in Addendum B to the CY 2026 OPPS/ASC proposed rule (which is available via the internet on the CMS website). We received comments on several of the new CPT codes that were assigned to comment indicator “NP” in Addendum B to the CY 2026 OPPS/ASC proposed rule. We have responded to those public comments in sections III.C., III.E., and IV. of this final rule with comment period.
The final SIs, APC assignments, and payment rates for the new CPT codes that are effective January 1, 2026, can be found in Addendum B to this final rule with comment period. In addition, the SI definitions can be found in Addendum D1 to this final rule with comment period. Addenda B and D1 are available via the internet on the CMS website.
Finally, Table 15, which is a reprint of Table 11 from the CY 2026 OPPS/ASC proposed rule (90 FR 33535), shows the comment timeframe for new and revised HCPCS codes. The table provides information on our current process for updating codes through our OPPS quarterly update CRs, seeking public comments, and finalizing the treatment of these codes under the OPPS.
( printed page 53527)B. OPPS Changes—Variations Within APCs
1. Background
Section 1833(t)(2)(A) of the Act requires the Secretary to develop a classification system for covered hospital outpatient department services. In addition, section 1833(t)(2)(B) of the Act provides that the Secretary may establish groups of covered OPD services within this classification system, so that services classified within each group are comparable clinically and with respect to the use of resources. In accordance with these provisions, we developed a grouping classification system, referred to as Ambulatory Payment Classifications (APCs), as set forth in regulations at 42 CFR 419.31. We use Level I (also known as CPT codes) and Level II HCPCS codes (also known as alphanumeric codes) to identify and group the services within each APC. The APCs are organized such that each group is homogeneous both clinically and in terms of resource use. Using this classification system, we have established distinct groups of similar services. We also have developed separate APC groups for certain medical devices, drugs, biologicals, therapeutic radiopharmaceuticals, and brachytherapy devices that are not packaged into the payment for the procedure.
We have packaged into the payment for each procedure or service within an APC group, the costs associated with those items and services that are typically ancillary and supportive to a primary diagnostic or therapeutic modality and, in those cases, are an integral part of the primary service they support. Therefore, we do not make separate payment for these packaged items or services. In general, packaged items and services include, but are not limited to, the items and services listed in regulations at 42 CFR 419.2(b). A further discussion of packaged services is included in section II.A.3. of this final rule with comment period.
Under the OPPS, we generally pay for covered hospital outpatient services on a rate-per-service basis, where the service may be reported with one or more HCPCS codes. Payment varies according to the APC group to which the independent service or combination of services is assigned. For CY 2026, we proposed that each APC relative payment weight represents the hospital cost of the services included in that APC, relative to the hospital cost of the services included in APC 5012 (Clinic Visits and Related Services). The APC relative payment weights are scaled to APC 5012 because it is the hospital clinic visit APC and clinic visits are among the most frequently furnished services in the hospital outpatient setting.
2. Application of the 2 Times Rule
Section 1833(t)(9)(A) of the Act requires the Secretary to review, not less often than annually, and revise the APC groups, the relative payment weights, and the wage and other adjustments described in section 1833(t)(2) of the Act to consider changes in medical practice, changes in technology, the addition of new services, new cost data, and other relevant information and factors. Section 1833(t)(9)(A) of the Act also requires the Secretary to consult with an expert outside advisory panel composed of an appropriate selection of representatives of providers to review (and advise the Secretary concerning) the clinical integrity of the APC groups and the relative payment weights. We note that the Advisory Panel on Hospital Outpatient Payment (also known as the HOP Panel or the Panel) recommendations for specific services for the CY 2026 OPPS update will be discussed in the relevant specific sections throughout this final rule with comment period.
In addition, section 1833(t)(2) of the Act provides that, subject to certain exceptions, the items and services within an APC group cannot be considered comparable regarding the use of resources if the highest cost for an item or service in the group is more than 2 times greater than the lowest cost for an item or service within the same group (referred to as the “2 times rule”). The statute authorizes the Secretary to make exceptions to the 2 times rule in unusual cases, such as for low-volume items and services (but the Secretary may not make such an exception in the case of a drug or biological that has been designated as an orphan drug under section 526 of the Federal Food, Drug, and Cosmetic Act). In determining the APCs with a 2 times rule violation, we consider only those HCPCS codes that are significant based on the number of claims. We note that, for purposes of identifying significant procedure codes for examination under the 2 times rule, we consider procedure codes that have more than 1,000 single major claims or procedure codes that both have more than 99 single major claims and contribute at least 2 percent of the single major claims used to establish the APC cost to be significant (75 FR 71832). This longstanding definition of when a procedure code is significant for purposes of the 2 times rule was selected because we believe that a subset of 1,000 or fewer claims is negligible within the set of approximately 100 million single procedure or single session claims we use for establishing costs. Similarly, a procedure code for which there are fewer than 99 single claims and that comprises less than 2 percent of the single major claims within an APC will have a negligible impact on the APC cost (75 FR 71832). In the CY 2026 OPPS/ASC proposed rule, we proposed to make exceptions to this limit on the variation of costs within each APC group in unusual cases, such as for certain low-volume items and services.
For the CY 2026 OPPS update, we identified the APCs with violations of the 2 times rule, and we proposed changes to the procedure codes assigned to these APCs (with the exception of those APCs for which we proposed a 2 times rule exception) in Addendum B to the CY 2026 OPPS/ASC proposed rule. We note that Addendum B does not appear in the printed version of the Federal Register as part of this final rule with comment period. Rather, it is published and made available via the internet on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices.
To eliminate a violation of the 2 times rule and improve clinical and resource homogeneity in the APCs for which we did not propose a 2 times rule exception, we proposed to reassign these procedure codes to new APCs that contain services that are similar with regard to both their clinical and resource characteristics. In many cases, the proposed procedure code reassignments and associated APC reconfigurations for CY 2026 included in the CY 2026 OPPS/ASC proposed rule are related to changes in costs of services that were observed in the CY 2024 claims data available for CY 2026 ratesetting. Addendum B to the CY 2026 OPPS/ASC proposed rule identifies with a comment indicator “CH” those procedure codes for which we proposed a change to the APC assignment or status indicator, or both, that were initially assigned in the July 1, 2025, OPPS Addendum B Update, which is available via the internet on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/addendum-a-b-update.
3. APC Exceptions to the 2 Times Rule
While considering the APC changes that we proposed for CY 2026, we reviewed all of the APCs for which we identified 2 times rule violations to determine whether any of the APCs ( printed page 53528) would qualify for an exception. We used the following criteria to evaluate whether to propose exceptions to the 2 times rule for affected APCs:
- Resource homogeneity;
- Clinical homogeneity;
- Hospital outpatient setting utilization;
- Frequency of service (volume); and
- Opportunity for upcoding and code fragments.
For a detailed discussion of these criteria, we refer readers to the April 7, 2000 final rule (65 FR 18457 through 18458).
Based on the CY 2024 claims data available for the CY 2026 OPPS/ASC proposed rule, we found 26 APCs with violations of the 2 times rule. We applied the criteria as described above to identify the APCs for which we proposed to make exceptions under the 2 times rule for CY 2026 and found that all of the 26 APCs we identified meet the criteria for an exception to the 2 times rule based on the CY 2024 claims data available for the CY 2026 OPPS/ASC proposed rule. We note that, on an annual basis, based on our analysis of the latest claims data, we identify violations to the 2 times rule and propose changes when appropriate. Those APCs that violate the 2 times rule are identified and appear in Table 16. In addition, we did not include in that determination those APCs where a 2 times rule violation was not a relevant concept, such as APC 5401 (Dialysis), which only has two HCPCS codes assigned to it that have similar geometric mean costs and do not create a 2 times rule violation. Therefore, we have only identified those APCs, including those with criteria-based costs, such as device-dependent CPT/HCPCS codes, with violations of the 2 times rule, where a 2 times rule violation is a relevant concept.
Table 12 of the CY 2026 OPPS/ASC proposed rule (90 FR 33537) listed the APCs for which we proposed to make an exception under the 2 times rule for CY 2026 based on the criteria cited above and claims data submitted between January 1, 2024, and December 31, 2024, and CCRs, if available. The proposed geometric mean costs for covered hospital outpatient services for these and all other APCs that were used in the development of the CY 2026 OPPS/ASC proposed rule can be found via the internet on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices.
Based on the updated final rule CY 2024 claims data used for this final rule with comment period, we found a total of 27APCs with violations of the 2 times rule. Of these 27 total APCs, 24 were identified in the proposed rule and three are newly identified in this final rule with comment period. The following two APCs appeared in Table 12 of the CY 2026 OPPS/ASC proposed rule (90 FR 33537) as violating the 2 times rule. However, after conducting data analysis for this final rule with comment period, we found that the APCs no longer violate the 2 times rule:
- APC 5613 (Level 3 Therapeutic Radiation Treatment Preparation).
- APC 5811 (Manipulation Therapy).
In addition, the following three APCs are newly identified with 2 times rule violations using updated data for this final rule with comment period:
- APC 5024 (Level 4 Type A ED Visits).
- APC 5052 (Level 2 Skin Procedures).
- APC 5722 (Level 2 Diagnostic Tests and Related Services).
We received comments on the APCs located in Table 12 of the CY 2026 OPPS/ASC proposed rule (90 FR 33537), along with comments on APC assignments for specific HCPCS codes. These comments and our responses can be found in section III.E. of this final rule with comment period.
Based on our analysis of the CY 2024 costs from hospital claims and cost report data available for this final rule with comment period, we are finalizing our proposals with some modifications. Specifically, we are finalizing our proposal to except the 24 proposed APCs that continue to have 2 times violations in this final rule with comment period data from the 2 times rule for CY 2024 claims data and also except three additional APCs that did not violate the 2 times rule in the CY 2026 OPPS/ASC proposed rule data, but do violate the 2 times rule in this final rule with comment period data, for a total of 27APCs for which we identified 2 times rule violations but that qualify for exceptions.
In summary, Table 16 lists the 27 APCs that we are excepting from the 2 times rule for CY 2026 based on the criteria described earlier and a review of updated claims data for dates of service between January 1, 2024, and December 31, 2024, that were processed on or before June 30, 2025, and updated CCRs, if available. We note that, for cases in which a recommendation by the HOP Panel appears to result in or allow a violation of the 2 times rule, we generally accept the HOP Panel's recommendation because those recommendations are based on explicit consideration of resource use, clinical homogeneity, site of service, and the quality of the claims data used to determine the APC payment rates. The geometric mean costs for hospital outpatient services for these and all other APCs that were used in the development of this final rule with comment period can be found via the internet on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices.
( printed page 53529)C. New Technology APCs
1. Background
In the CY 2002 OPPS final rule (66 FR 59903), we finalized changes to the time period in which a service can be eligible for payment under a New Technology APC. Beginning in CY 2002, we retain services within New Technology APC groups until we gather sufficient claims data to enable us to assign the service to an appropriate clinical APC. This policy allows us to move a service from a New Technology APC in less than 2 years if sufficient data are available. It also allows us to retain a service in a New Technology APC for more than 2 years if sufficient data upon which to base a decision for reassignment have not been collected.
We also adopted in the CY 2002 OPPS final rule the following criteria for assigning a complete or comprehensive service to a New Technology APC: (1) the service must be truly new, meaning it cannot be appropriately reported by an existing HCPCS code assigned to a clinical APC and does not appropriately fit within an existing clinical APC; (2) the service is not eligible for transitional pass-through payment (however, a truly new, comprehensive service could qualify for assignment to a new technology APC even if it involves a device or drug that could, on its own, qualify for pass-through payment); and (3) the service falls within the scope of Medicare benefits under section 1832(a) of the Act and is reasonable and necessary in accordance with section 1862(a)(1)(A) of the Act (66 FR 59898 through 59903). For additional information about our New Technology APC policy, we refer readers to https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/HospitalOutpatientPPS/passthroughpayment on the CMS website and then follow the instructions to access the MEARISTM system for OPPS New Technology APC applications.[15]
In the CY 2004 OPPS final rule with comment period (68 FR 63416), we restructured the New Technology APCs to make the cost intervals more consistent across payment levels and refined the cost bands for these APCs to retain two parallel sets of New Technology APCs: one set with a status indicator of “S” (Significant Procedures, Not Discounted when Multiple. Paid under OPPS; separate APC payment) and the other set with a status indicator of “T” (Significant Procedure, Multiple Reduction Applies. Paid under OPPS; separate APC payment). These current New Technology APC configurations allow us to price new technology services more appropriately and consistently.
For CY 2025, there were 52 New Technology APC levels, ranging from the lowest cost band assigned to APC 1491 (New Technology—Level 1A ($0-$10)) to the highest cost band assigned to APC 1908 (New Technology—Level 52 ($145,001-$160,000)). We note that the cost bands for the New Technology APCs, specifically, APCs 1491 through 1599 and 1901 through 1908, vary with increments ranging from $10 to $14,999. These cost bands identify the APCs to which new technology procedures and services with estimated service costs that fall within those cost bands are ( printed page 53530) assigned under the OPPS. Payment for each APC is made at the mid-point of the APC's assigned cost band. For example, payment for APC 1507 (New Technology—Level 7 ($501-$600)) is made at $550.50.
Under the OPPS, one of our goals is to make payments that are appropriate for the services that are necessary for the treatment of Medicare beneficiaries. The OPPS, like other Medicare payment systems, is budget neutral and increases are limited to the annual hospital market basket increase reduced by the productivity adjustment. We believe that our payment rates reflect the costs that are associated with providing care to Medicare beneficiaries and are adequate to ensure access to services (80 FR 70374). For many emerging technologies, there is a transitional period during which utilization may be low, often because providers are first learning about the technologies and their clinical utility. Quite often, parties request that Medicare make higher payments under the New Technology APCs for new procedures in that transitional phase. These requests, and their accompanying estimates for expected total patient utilization, often reflect very low rates of patient use of expensive equipment, resulting in high per-use costs for which requesters believe Medicare should make full payment. Medicare does not, and we believe should not, assume responsibility for more than its share of the costs of procedures based on projected utilization for Medicare beneficiaries and does not set its payment rates based on initial projections of low utilization for services that require expensive capital equipment. For the OPPS, we rely on hospitals to make informed business decisions regarding the acquisition of high-cost capital equipment, taking into consideration their knowledge about their entire patient base (Medicare beneficiaries included) and an understanding of Medicare's and other payers' payment policies. We refer readers to the CY 2013 OPPS/ASC final rule with comment period (77 FR 68314) for further discussion regarding this payment policy.
Some services assigned to New Technology APCs have low annual volume, which we consider to be fewer than 100 claims in the year of claims data used for ratesetting (86 FR 63528). Where utilization of services assigned to a New Technology APC is low, it can lead to wide variation in payment rates from year to year, resulting in even lower utilization and potential barriers to access of new technologies, which ultimately limits our ability to assign the service to the appropriate clinical APC. To mitigate these issues, we finalized a policy in the CY 2019 OPPS/ASC final rule with comment period to utilize our equitable adjustment authority at section 1833(t)(2)(E) of the Act to adjust how we determine the costs for low-volume services assigned to New Technology APCs (83 FR 58892 through 58893). Specifically, in the CY 2019 OPPS/ASC final rule with comment period (83 FR 58893), we established that, in each of our annual rulemakings, we would calculate and present the result of each statistical methodology (arithmetic mean, geometric mean, and median) based on up to 4 years of claims data and solicit public comment on which methodology should be used to establish the payment rate for the low-volume new technology service. In the CY 2022 OPPS/ASC final rule (86 FR 63529), we replaced the New Technology APC low volume policy with the universal low volume APC policy. Unlike the New Technology APC low volume policy, the universal low volume APC policy applies to clinical APCs and brachytherapy APCs, in addition to procedures assigned to New Technology APCs, and uses the highest of the geometric mean, arithmetic mean, or median based on up to 4 years of claims data to set the payment rate for the APC. We refer readers to the CY 2022 OPPS/ASC final rule with comment period (86 FR 63529) for further discussion regarding this policy.
Despite the universal low volume APC policy, we continued to see payment instability for services with very low claims volume of fewer than 10 claims in the 4-year lookback period used under the universal low volume APC policy. For CY 2025, we finalized a policy to exempt services assigned to New Technology APCs with fewer than 10 claims over the 4-year lookback period used for the universal low volume policy. Instead of assigning these services to a different New Technology APC based on the very few claims available, we maintained the New Technology APC assignment for each service from the prior year, CY 2024. We refer readers to the CY 2025 OPPS/ASC final rule with comment period for a discussion on the policy (89 FR 94016 through 94018).
Finally, we note that, in a budget-neutral system, payments may not fully cover hospitals' costs in a particular circumstance, including those for the purchase and maintenance of capital equipment. We rely on hospitals to make their decisions regarding the acquisition of high-cost equipment with the understanding that the Medicare program must be careful to establish its initial payment rates, including those made through New Technology APCs, for new services that lack hospital claims data based on realistic utilization projections for all such services delivered in cost-efficient hospital outpatient settings. As the OPPS acquires claims data regarding hospital costs associated with new procedures, we regularly examine the claims data and any available new information regarding the clinical aspects of new procedures to confirm that our OPPS payments remain appropriate for procedures as they transition into mainstream medical practice (77 FR 68314). For CY 2026, we included the proposed payment rates for New Technology APCs 1491 to 1599 and 1901 through 1908 in Addendum A to the CY 2026 OPPS/ASC proposed rule (which is available on the CMS website at https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/HospitalOutpatientPPS/Hospital-Outpatient-Regulations-and-Notices.
2. Continue To Exempt Services With Under 10 Claims in the 4-Year Lookback Period From APC Reassignment Based on the Universal Low Volume Policy
We continue to be concerned about payment stability for services assigned to New Technology APCs, specifically services with fewer than 10 claims in the 4-year lookback period used under the universal low volume APC policy. We also continue to believe that determining initial cost estimates for these services may be particularly challenging, given the lack of cost information for new and innovative technologies, and that we generally utilize claims data from hospitals as soon as these data become available.
We proposed to continue our policy to exempt services assigned to New Technology APCs with fewer than 10 claims over the 4-year lookback period from the universal low volume policy. Instead of assigning these services to a different clinical or New Technology APC based on the very few claims available, we proposed to continue maintaining the New Technology APC assignment for each service from the prior year. For example, for CY 2026, services assigned to New Technology APCs with fewer than 10 claims in the previous 4 years would maintain their New Technology APC assignment from CY 2025. We proposed to continue this policy in future years, until, or unless, an alternative policy is finalized. We maintain that it is appropriate to apply this policy to services assigned to New Technology APCs because these services represent new technologies for which it ( printed page 53531) may be more challenging to determine an appropriate cost than for other, more established services. We continue to believe 10 claims is an appropriate ceiling for exempting services from reassignment based on the universal low volume APC policy because we believe that at 10 claims a rough standard distribution begins to appear. We also continue to believe that services with so few claims over the 4-year lookback period would be especially vulnerable to large changes in payment rates year-to-year as a result of one or two new claims being available or one or two claims from what was previously the fourth year of the lookback period no longer being included in that period.
Consistent with our overall policy regarding use of updated claims data in the final rule, we proposed to perform a similar analysis for the final rule using updated claims data, including determining whether specific HCPCS codes continue to meet the criteria for our universal low volume APC policy or would be subject to our proposed policy to continue exempting services with fewer than 10 claims in the 4-year lookback period from the universal low volume APC policy and maintain the New Technology APC assignment from the previous year. We would update the APC placement as needed in the final rule.
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: Commenters generally supported our low volume APC policies. Comments were received regarding specific services assigned to New Technology APCs with low claims volume that supported maintaining the APC assignment as a result of our proposal to continue to exempt services assigned to New Technology APCs with fewer than 10 claims over the 4-year lookback period from the universal low volume policy.
Response: We thank the commenters for their support.
After consideration of public comments, we are finalizing our proposal to continue to exempt services assigned to New Technology APCs with fewer than 10 claims over the 4-year lookback period from the universal low volume policy.
3. Procedures Assigned to New Technology APC Groups for CY 2026
As we described in the CY 2002 OPPS final rule (66 FR 59902), we generally retain a procedure in the New Technology APC to which it is initially assigned until we have obtained sufficient claims data to justify reassignment of the procedure to a clinically appropriate APC. In addition, in cases where we find that our initial New Technology APC assignment was based on inaccurate or inadequate information (although it was the best information available at the time), where we obtain new information that was not available at the time of our initial New Technology APC assignment, or where the New Technology APCs are restructured, we may, based on more recent resource utilization information (including claims data) or the availability of refined New Technology APC cost bands, reassign the procedure or service to a different New Technology APC that more appropriately reflects its cost (66 FR 59903).
Consistent with our current policy, for CY 2026, we proposed to retain services within New Technology APC groups until we obtain sufficient claims data to justify reassignment of the service to an appropriate clinical APC. The flexibility associated with this policy allows us to reassign a service from a New Technology APC in less than 2 years if we have obtained sufficient claims data. It also allows us to retain a service in a New Technology APC for more than 2 years if we have not obtained sufficient claims data upon which to base a reassignment decision (66 FR 59902).
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: A commenter stated that the New Technology APC pathway is vital for bringing innovative services to patients before sufficient claims data exist for permanent APC assignment and suggested that we incorporate the perspectives of patients and caregivers into our review to ensure payment decisions reflect unmet needs and treatment burdens.
Response: We thank the commenter for their comment. We note that we accept a variety of information as part of the New Technology APC application process.
After consideration of public comments, we are finalizing our proposal to retain services within New Technology APC groups until we obtain sufficient claims data to justify reassignment of the service to an appropriate clinical APC.
a. Administration of Subretinal Therapies Requiring Vitrectomy (APC 1563)
Effective January 1, 2021, CMS established HCPCS code C9770 (Vitrectomy, mechanical, pars plana approach, with subretinal injection of pharmacologic/biologic agent) and assigned it to a New Technology APC based on the geometric mean cost of CPT code 67036 (Vitrectomy, mechanical, pars plana approach) due to similar resource utilization. For CY 2021, HCPCS code C9770 was assigned to APC 1561 (New Technology—Level 24 ($3001-$3500)). This code may be used to describe the administration of HCPCS code J3398 (Injection, voretigene neparvovec-rzyl, 1 billion vector genomes). This procedure was previously discussed in depth in the CY 2021 OPPS/ASC final rule with comment period (85 FR 85939 through 85940). For CY 2022, we maintained the APC assignment of APC 1561 (New Technology—Level 24 ($3001-$3500)) for HCPCS code C9770 (86 FR 63531 through 63532).
HCPCS code J3398 (Injection, voretigene neparvovec-rzyl, 1 billion vector genomes) is for a gene therapy product indicated for a rare mutation-associated retinal dystrophy. Voretigene neparvovec-rzyl (Luxturna®) was approved by FDA in December of 2017 and is an adeno-associated virus vector-based gene therapy indicated for the treatment of patients with confirmed biallelic RPE65 mutation-associated retinal dystrophy.[16] This therapy is administered through a subretinal injection, which interested parties describe as an extremely delicate and sensitive surgical procedure. The FDA-approved package insert describes one of the steps for administering Luxturna as, “after completing a vitrectomy, identify the intended site of administration. The subretinal injection can be introduced via pars plana.”
Interested parties, including the manufacturer of Luxturna®, recommended CPT code 67036 (Vitrectomy, mechanical, pars plana approach) for the administration of the gene therapy.[17] However, the manufacturer previously contended the administration was not accurately described by any existing codes as CPT code 67036 (Vitrectomy, mechanical, pars plana approach) does not account for the administration itself. CMS recognized the need to accurately describe the unique procedure that is required to administer the therapy described by HCPCS code J3398. Therefore, in the CY 2021 OPPS/ASC ( printed page 53532) final rule with comment period, we established a new HCPCS code, C9770 (Vitrectomy, mechanical, pars plana approach, with subretinal injection of pharmacologic/biologic agent) to describe this process. For CY 2021, we assigned HCPCS code C9770 to APC 1561 (New Technology—Level 24 ($3001-$3500)) using the geometric mean cost of CPT code 67036. For CY 2022, we continued to assign HCPCS code C9770 to APC 1561 (New Technology—Level 24 ($3001-$3500)) using the geometric mean cost of CPT code 67036.
CY 2023 was the first year that claims data were available for HCPCS code C9770; therefore, we proposed and finalized a policy to base the payment rate of HCPCS code C9770 on claims data for that code rather than on the geometric mean cost of CPT code 67036. Given the low number of claims for this procedure, we designated HCPCS code C9770 as a low volume procedure under our universal low volume APC policy and used the greater of the geometric mean, arithmetic mean, or median cost calculated based on the available claims data to calculate an appropriate payment rate for purposes of assigning HCPCS code C9770 to a New Technology APC.
Based on the claims data available for the CY 2023 OPPS/ASC final rule with comment period, we found the median was the statistical methodology that estimated the highest cost for the service. The payment rate calculated using this methodology fell within the cost band for APC 1562 (New Technology—Level 25 ($3501-$4000)). Therefore, we finalized our proposal to assign HCPCS code C9770 to APC 1562 for CY 2023.
For CY 2024, we proposed and finalized that we would delete HCPCS code C9770 effective December 31, 2023 and recognize CPT code 0810T (Subretinal injection of a pharmacologic agent, including vitrectomy and 1 or more retinotomies) starting January 1, 2024 (88 FR 81617 through 81619). We determined the payment rate for CPT code 0810T using the claims data for HCPCS code C9770 and designated CPT code 0810T as a low volume procedure under our universal low volume APC policy and used the greater of the geometric mean, arithmetic mean, or median cost calculated based on the available claims data for HCPCS code C9770 to calculate an appropriate payment rate for purposes of assigning CPT code 0810T to a New Technology APC. For CY 2024, we finalized assignment of CPT code 0810T to APC 1563 (New Technology—Level 26 ($4001-$4500)) (88 FR 81617 through 81619). For 2025, claims data for CPT code 0810T was not yet available. Therefore, we continued to use claims data for HCPCS code C9770 to determine the appropriate APC for CPT code 0810T and finalized to continue to assign CPT code 0810T to APC 1563 for CY 2025.
CY 2026 is the first year that we have claims data available for CPT code 0810T, and there are 6 claims available. Since the procedure described by CPT code 0810T was billed using HCPCS code C9770 prior to January 1, 2024, we proposed to use the available combined 42 claims for both codes during this time period to allow for a more accurate picture of the costs associated with this procedure. For CY 2026, we proposed to designate CPT code 0810T as a low volume procedure under our universal low volume APC policy, given that there were only 42 combined claims available. This is below the threshold of 100 claims for a service within a year required to designate a service as a low volume service and apply our universal low volume APC policy. Therefore, we proposed to use the greater of the geometric mean, arithmetic mean, or median cost calculated based on the available claims data from a 4-year lookback period to calculate an appropriate payment rate for purposes of assigning CPT code 0810T to a New Technology APC.
Using all available claims for CPT code 0810T and HCPCS code C9770 from the 4-year lookback period, based on 42 claims, we determined the geometric mean cost to be approximately $4,040, the arithmetic mean cost to be $4,327, and the median cost to be $3,999. Because the arithmetic mean is the statistical methodology that estimated the highest cost for the service, we proposed to use this cost to determine the New Technology APC placement. The arithmetic mean of $4,327 falls within the cost band for APC 1563 (New Technology—Level 26 ($4001-$4500)). Therefore, we proposed to continue to assign CPT code 0810T to APC 1563 for CY 2026. Additionally, we proposed to perform a similar analysis using updated claims data, including determining if CPT code 0810T continues to meet the criteria for our universal low volume APC policy, in the CY 2026 OPPS/ASC final rule with comment period and update the APC assignment as needed.
We did not receive any public comments on our proposal to continue to assign HCPCS code 0810T to APC 1563 for CY 2026.
One additional claim for CY 2024 has been processed since the CY 2026 OPPS/ASC proposed rule. Our analysis of the updated claims data found that the greater of the geometric mean, arithmetic mean, or median cost calculated for HCPCS codes C9770 and 0810T is approximately $4,239. This continues to fall into the cost band of New Technology APC 1563. Therefore, we are finalizing our proposal without modification to continue to assign CPT code 0810T to APC 1563 (New Technology—Level 26 ($4001-$4500)).
Refer to Table 17 for the final OPPS New Technology APC and status indicator assignment for CPT codes 0810T for CY 2026. The final CY 2026 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website. In addition, we refer readers to Addendum D1 to this final rule with comment period for the status indicator meanings for all codes reported under the OPPS. Addendum D1 can also be found via the internet on the CMS website.
( printed page 53533)b. BgRT (APC 1521 and 1525)
Biology Guided Radiation Therapy (BgRT) uses positron-emitting radiopharmaceuticals to control delivery of radiation therapy to treat primary and metastatic lung or bone tumors. During radiation treatment delivery, the same system applies these firing filters to the real-time positron emission tomography (PET) data collected by the radiation treatment delivery machine. Effective January 1, 2024, CMS created HCPCS codes C9794 (Therapeutic radiology simulation-aided field setting; complex, including acquisition of PET and CT imaging data required for radiopharmaceutical-directed radiation therapy treatment planning ( i.e., modeling) and C9795 (Stereotactic body radiation therapy, treatment delivery, per fraction to 1 or more lesions, including image guidance and real-time positron emissions-based delivery adjustments to 1 or more lesions, entire course not to exceed 5 fractions) to describe the modeling and treatment delivery portions of the BgRT service. We assigned HCPCS code C9794 to APC 1521 (New Technology—Level 21 ($1901-$2000)) and HCPCS code C9795 to APC 1525 (New Technology—Level 25 ($3501-$4000)) for CY 2024.
For CY 2025, we continued to assign HCPCS code C9794 to APC 1521 (New Technology—Level 21 ($1901-$2000)) with a payment rate of $1,950.50 and HCPCS code C9795 to APC 1525 (New Technology—Level 25 ($3501-$4000)) with a payment rate of $3,750.50 because we did not have any claims data for the service.
Effective January 1, 2025, HCPCS codes C9794 and C9795 were replaced by HCPCS codes G0562 and G0563, respectively. For CY 2026, the proposed OPPS payment rates are based on available CY 2024 claims data. There are no CY 2024 claims for HCPCS codes G0562 and G0563 since they were not effective until CY 2025. However, as HCPCS codes C9794 and C9795 were still in use until December 31, 2024, we proposed to determine the payment rate for HCPCS codes G0562 and G0563 using the available claims data for HCPCS codes C9794 and C9795, respectively. For CY 2026, we proposed to designate HCPCS codes G0562 and G0563 as low volume procedures under our universal low volume APC policy, given that there are only 16 claims for C9794 and 28 claims for C9795 during the claims period. For HCPCS code G0562, using all available claims for C9794, we determined, for the CY 2026 OPPS/ASC proposed rule, the arithmetic mean cost to be $1,241, the median cost to be $1,203, and the geometric mean cost to be $1,121. Because the arithmetic mean cost is the statistical methodology that estimated the highest cost for the service, we proposed to use this cost to determine the New Technology APC placement. The arithmetic mean cost of $1,241 falls within the cost band for APC 1514 (New Technology—Level 14 ($1201-$1300)). Therefore, we proposed to assign HCPCS code G0562 to APC 1514 (New Technology—Level 14 ($1201-$1300) with a payment rate of $1,250.50 for CY 2026. For HCPCS code G0563, using all available claims for C9795, we determined the arithmetic mean cost to be $3,606; the median cost to be $2,915, and the geometric mean cost to be $3,348. The arithmetic mean cost is the statistical methodology that estimated the highest cost for the service; therefore, we proposed to use this cost to determine the New Technology APC placement. The arithmetic mean cost of $3,606 falls within the cost band for APC 1525 (New Technology—Level 25 ($3501-$4000)). Therefore, we proposed to assign HCPCS code G0563 to APC 1525 (New Technology—Level 25 ($3501-$4000) with a payment rate of $3750.50 for CY 2026.
Additionally, we proposed to perform a similar analysis using updated claims data, including determining if HCPCS codes G0562 and G0563 continue to meet the criteria for our universal low volume APC policy, in the CY 2026 OPPS/ASC final rule with comment period and update the APC assignments as needed.
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: Commenters supported CMS' proposal to assign HCPCS code G0563 to APC 1525 (New Technology—Level 25 ($3501-$4000) with a payment rate of $3750.50 for CY 2026.
Response: We thank the commenters for their support.
Comment: Several commenters did not support the proposal to assign HCPCS code G0562 to APC 1514 (New Technology—Level 14 ($1201-$1300) with a payment rate of $1,250.50 for CY 2026. Commenters explained that the resulting decrease in payment would not cover the costs to provide the service, especially because the modeling service described by HCPCS code G0562 happens on a different day than the treatment, but on the same high-cost device as the treatment. Commenters emphasized the few single frequency claims available and urged CMS to allot hospitals more time to understand how costs for HCPCS code G0562 should be reported versus a diagnostic CT or PET scan that involves different equipment, workflows, and time. Commenters requested that we maintain the APC assignment for HCPCS code G0562 for CY 2026.
Response: We thank the commenters for their input. We agree with commenters who expressed concern that the proposed payment rate was based on an extremely limited number of claims and may not accurately reflect the true resource costs to hospitals associated with furnishing this service. Additionally, we are concerned that, if we were to finalize as proposed, the payment rate for this service would decrease 36 percent based on only 16 single frequency claims and only one year of claims data. As we have stated in prior rules, when only a limited number of claims are available for a given service, it is possible that those claims may not be representative of the full range of hospital costs. We ( printed page 53534) anticipate that, as hospitals gain additional experience furnishing the service and as more claims data becomes available in future years, the claims data will more accurately reflect the typical resource costs of the service.
We note that since the CY 2026 OPPS/ASC proposed rule published, we have one additional claim for HCPCS code C9795 to use for HCPCS code G0563 ratesetting, and the revised statistical methodologies are: the geometric mean cost is $3,277, the arithmetic mean is $3,449, and the median is $3,228. The highest of these is the arithmetic mean, which falls outside of the proposed APC assignment of APC 1525 (New Technology—Level 25 ($3501-$4000)). After consideration of public comments and the revised statistical methodologies, we are not finalizing our proposals for HCPCS codes G0562 and G0563. For CY 2026, we are finalizing the assignment of HCPCS code G0562 to APC 1521 and status indicator “S” and HCPCS code G0563 to APC 1524 (New Technology—Level 24 ($3001-$3500)) and status indicator “S”. Refer to Table 18 for the final OPPS New Technology APC and status indicator assignment for HCPCS codes G0562 and G0563 for CY 2026. The final CY 2026 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website. In addition, we refer readers to Addendum D1 to this final rule with comment period for the status indicator meanings for all codes reported under the OPPS. Addendum D1 can also be found via the internet on the CMS website.
c. Blinded Procedure for NYHA Class III/IV Heart Failure (APC 1590)
A randomized, double-blinded, controlled IDE study was conducted for the V-Wave interatrial shunt. The V-Wave interatrial shunt is for patients with severe symptomatic heart failure and is designed to regulate left atrial pressure in the heart. All participants who passed initial screening for the study receive a right heart catheterization procedure described by CPT code 93451 (Right heart catheterization including measurement(s) of oxygen saturation and cardiac output, when performed). Participants assigned to the experimental group also receive the V-Wave interatrial shunt procedure while participants assigned to the control group only receive right heart catheterization. The developer of V-Wave was concerned that the current coding of these services by Medicare would reveal to the study participants whether they had received the interatrial shunt because an additional procedure code, CPT code 93799 (Unlisted cardiovascular service or procedure), would be included on the claims for participants receiving the interatrial shunt. Therefore, for CY 2020, we created a temporary HCPCS code to describe the V-Wave interatrial shunt procedure for both the experimental group and the control group in the study. Specifically, we established HCPCS code C9758 (Blinded procedure for NYHA class III/IV heart failure; transcatheter implantation of interatrial shunt or placebo control, including right heart catheterization, trans-esophageal echocardiography (TEE)/intracardiac echocardiography (ICE), and all imaging with or without guidance (for example, ultrasound, fluoroscopy), performed in an approved investigational device exemption (IDE) study) to describe the service, and we assigned the service to APC 1589 (New Technology—Level 38 ($10,001-$15,000)) with a payment rate of $12,500.50.
In the CY 2021 OPPS/ASC final rule with comment period (85 FR 85946), we stated that we believe similar resources and device costs are involved with the V-Wave interatrial shunt procedure and the Corvia Medical interatrial shunt procedure (HCPCS code C9760), except that payment for HCPCS codes C9758 and C9760 differs based on how often the interatrial shunt is implanted when each code is billed. An interatrial shunt is implanted one-half of the time HCPCS code C9758 is billed, whereas an interatrial shunt is implanted every time HCPCS code C9760 is billed. Accordingly, for CY 2021, we reassigned HCPCS code C9758 to APC 1590 (New Technology—Level 39 ($15,001-$20,000)), which reflects the cost of furnishing the interatrial shunt one-half of the time the procedure is performed. Since CY 2021, HCPCS code C9758 has continued to be assigned to APC 1590. ( printed page 53535)
For CY 2026, the developer of the V-Wave interatrial shunt informed us that the IDE study had concluded and HCPCS code C9758 was no longer being utilized. Therefore, we proposed to delete HCPCS code C9758 for CY 2026.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter requested that CMS not delete HCPCS code C9758, as Corvia Medical is conducting an ongoing clinical study that utilizes this code.
Response: Since HCPCS code C9758 is currently being utilized, we will continue to keep this code active for CY 2026. Therefore, we are not finalizing our proposal to delete HCPCS code C9758 for CY 2026.
Our updated claims data for the 4-year lookback period for the universal low volume APC policy shows only 8 claims for HCPCS code C9758. Because we are finalizing our proposal to maintain current New Technology APC assignments for CY 2026 for New Technology APC services with fewer than 10 claims in the 4-year lookback period, we are continuing to assign HCPCS code C9758 to APC 1590 for CY 2026. Refer to Table 19 for the final OPPS New Technology APC and status indicator assignment for HCPCS code C9758 for CY 2026. The final CY 2026 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website. In addition, we refer readers to Addendum D1 to this final rule with comment period for the status indicator meanings for all codes reported under the OPPS. Addendum D1 can also be found via the internet on the CMS website.
d. Bronchoscopy With Transbronchial Ablation of Lesion(s) by Microwave Energy (APC 1562)
Effective January 1, 2019, CMS established HCPCS code C9751 (Bronchoscopy, rigid or flexible, transbronchial ablation of lesion(s) by microwave energy, including fluoroscopic guidance, when performed, with computed tomography acquisition(s) and 3-D rendering, computer-assisted, image-guided navigation, and endobronchial ultrasound (EBUS) guided transtracheal and/or transbronchial sampling ( e.g., aspiration[s]/biopsy[ies]) and all mediastinal and/or hilar lymph node stations or structures and therapeutic intervention(s)). This microwave ablation procedure utilizes a flexible catheter to access the lung tumor via a working channel and may be used as an alternative procedure to a percutaneous microwave approach. Based on our review of the New Technology APC application for this service and the service's clinical similarity to existing services paid under the OPPS, we estimated the likely cost of the procedure would be between $8,001 and $8,500. We assigned the procedure to APC 1571 (New Technology—Level 34 ($8001-$8500)) for CY 2019.
In claims data available from CY 2019 for the CY 2021 OPPS/ASC final rule with comment period, there were four claims reported for bronchoscopy with transbronchial ablation of lesions by microwave energy. Given the low volume of claims for the service, we proposed for CY 2021 to apply the universal low volume APC policy we adopted in CY 2019, under which we utilize our equitable adjustment authority under section 1833(t)(2)(E) of the Act to calculate the geometric mean, arithmetic mean, and median costs to determine an appropriate payment rate for purposes of assigning bronchoscopy with transbronchial ablation of lesions by microwave energy to a New Technology APC. Based on this analysis using claims from CY 2019, we assigned HCPCS code C9751 to APC 1562 (New Technology—Level 25 ($3501-$4000)) with a $3750.50 payment rate for CY 2021.
There have been no separately payable claims reported for HCPCS code C9751 since 2019. Therefore, we have continued to use claims from CY 2019 to determine the payment rate for this service in CY 2023, CY 2024, and CY 2025 OPPS/ASC final rules with comment period. Based on the information available, we continue to assign HCPCS code C9751 to APC 1562 (New Technology—Level 25 ($3501-$4000)), with a payment rate of $3,750.50.
For CY 2026, we were informed that the Neuwave Flex program is no longer available for commercial use, and that HCPCS code C9751 is no longer being utilized. Therefore, we proposed to delete HCPCS code C9751 for CY 2026.
We did not receive public comments on our proposal to delete HCPCS code C9751 for CY 2026. Additionally, our updated claims data remain unchanged. Therefore, we are finalizing as proposed.
Refer to Table 20 for the final OPPS New Technology APC and status indicator assignment for HCPCS code C9751 for CY 2026. In addition, we refer readers to Addendum D1 to this final rule with comment period for the status indicator meanings for all codes reported under the OPPS. Addendum D1 can also be found via the internet on the CMS website.
( printed page 53536)e. Cardiac Positron Emission Tomography (PET)/Computed Tomography (CT) Studies (APCs 1519 and 1522)
For CY 2026, the OPPS payment rates for the service described by CPT codes 78431, 78432, and 78433 were proposed to be based on available CY 2024 claims data. CPT code 78431 had over 30,000 single frequency claims in CY 2024. The geometric mean cost for CPT code 78431 is approximately $2,200. The geometric mean falls within APC 1522 (New Technology—Level 22 ($2001-$2500)) with a payment rate of $2,250.50, which is the current APC assignment for this service. Therefore, we proposed, for CY 2026, to continue to assign CPT code 78431 to APC 1522 (New Technology—Level 22 ($2001-$2500)) with a payment rate of $2,250.50.
There were only 31 single frequency claims in CY 2024 for CPT code 78432. As this is below the threshold of 100 claims for a service within a year, we proposed to apply our universal low volume New Technology APC policy and use the highest of the geometric mean cost, arithmetic mean cost, or median cost based on up to 4 years of claims data to assign CPT code 78432 to the appropriate New Technology APC. Using available claims data from CY 2021, CY 2022, and CY 2023, our analysis found the geometric mean cost of the service is approximately $1,591, the arithmetic mean cost of the service is approximately $1,737, and the median cost of the service is approximately $1,364. The arithmetic mean is the statistical methodology that estimates the highest cost for the service. The arithmetic mean cost of $1,737, is an amount that is below the cost band for APC 1520 (New Technology—Level 20 ($1801-$1900)), where the procedure is currently assigned. Therefore, we proposed, for CY 2026, to assign CPT code 78432 to APC 1519 (New Technology—Level 19 ($1701-$1800)) with a payment rate of $1,750.50.
There were over 1,400 single frequency claims for CPT code 78433 in CY 2024. The geometric mean for CPT code 78433 is approximately $2,037, which is an amount that is above the current New Technology APC cost band APC 1521 (New Technology—Level 21 ($1901-$2000)) to which it is assigned. Therefore, for CY 2026, we proposed to reassign CPT code 78433 to APC 1522 (New Technology—Level 22 ($2001-$2500)) with a payment rate of $2,250.50.
We note that, over the past several years, the claims volumes for CPT codes 78431 and 78433 have increased significantly while the geometric mean costs of the codes have remained relatively stable. However, CPT code 78432, which is closely related to CPT codes 78431 and 78433, continues to have low claims frequency and fluctuating geometric mean costs. Due to our concerns regarding CPT code 78432 and the lack of an appropriate clinical APC for CPT codes 78431 and 78433 at this time based on resource cost similarity, we proposed to continue to assign CPT codes 78431 through 78433 to New Technology APCs for CY 2026.
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: Commenters supported the proposed APC assignments for CPT codes 78431 and 78433 for CY 2026 based on our analysis of the available claims data.
Response: We thank the commenters for their support.
Comment: Some commenters did not support the proposed APC assignment for CPT code 78432 to APC 1519 (New Technology—Level 19 ($1701-$1800)) with a payment rate of $1,750.50. A commenter explained that CPT code 78432 consumes more resources than CPT code 78431. The commenter stated that with similar, but enhanced, clinical staff and radiotracer workflows to CPT code 78431, it is not appropriate for CPT code 78432 to be assigned to an APC with payments lower than CPT code 78431.
Response: As we have stated in previous rulemaking, New Technology APCs are cost bands rather than clinical groupings. Unlike when we assign a service to a clinical APC and consider resource and clinical similarities to other services in a clinical APC, we assign services to New Technology APCs based on cost. While we appreciate the commenter's information regarding the clinical differences between CPT codes 78431 and 78432, we adjust New Technology APC assignments based on the claims data available rather than clinical characteristics of a service.
We note that additional claims for CPT codes 78431 through 78433 have been processed since the CY 2026 OPPS/ASC proposed rule. Based on updated claims data, CPT code 78431 has an updated geometric mean cost of approximately $2,182. Because the geometric mean cost of CPT code 78431 is still within the range for APC 1522, the proposed APC assignment for CPT code 78431 for CY 2026, we are finalizing the proposed APC assignment of CPT code 78431 without modification.
There were three additional single frequency claims for CY 2024 processed for CPT code 78432 since the CY 2026 OPPS/ASC proposed rule, bringing the total number of single frequency claims to 34 for CPT code 78432 for CY 2024. Based on the updated claims data for CPT code 78432, the geometric mean cost is approximately $1,428; the ( printed page 53537) arithmetic mean cost is approximately $1,517; and the median cost is approximately $1,274. Of these, the highest statistical methodology is the arithmetic mean cost of $1,517. Since the updated arithmetic mean cost for CPT code 78432 is outside of the cost band for APC 1519 (New Technology—Level 19 ($1701-$1800), we are not finalizing our proposal to assign CPT code 78432 to APC 1519 for CY 2026. Based on the updated statistical methodologies, we are assigning CPT code 78432 to APC 1517 for CY 2026 (New Technology—Level 17 ($1501-$1600)) with a payment rate of $1,550.50.
Based on updated claims data, CPT code 78433 has an updated geometric mean cost of approximately $2004. Because the geometric mean cost of CPT code 78433 is still within the range for APC 1522, the proposed APC assignment for CPT code 78433 for CY 2026, we are finalizing the proposed APC assignment of CPT code 78433 without modification.
Refer to Table 21 for the final OPPS New Technology APC and status indicator assignments for CPT codes 7843, 78432, and 78433 for CY 2026. The final CY 2026 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website. In addition, we refer readers to Addendum D1 to this final rule with comment period for the status indicator meanings for all codes reported under the OPPS. Addendum D1 can also be found via the internet on the CMS website.
f. CardiAMP (APC 1590)
The CardiAMP cell therapy IDE studies are two randomized, double-blinded, controlled IDE studies: the CardiAMP Cell Therapy Chronic Myocardial Ischemia Trial [18] and the CardiAMP Cell Therapy Heart Failure ( printed page 53538) Trial.[19] The two trials are designed to investigate the safety and efficacy of autologous bone marrow mononuclear cell treatment for the following: (1) patients with medically refractory and symptomatic ischemic cardiomyopathy; and (2) patients with refractory angina pectoris and chronic myocardial ischemia. On April 1, 2022, we established HCPCS code C9782 to describe the CardiAMP cell therapy IDE studies and assigned HCPCS code C9782 to APC 1574 (New Technology—Level 37 ($9,501-$10,000)) with the status indicator “T.” We subsequently revised the descriptor for HCPCS code C9782 to: (Blinded procedure for New York Heart Association (NYHA) Class II or III heart failure, or Canadian Cardiovascular Society (CCS) Class III or IV chronic refractory angina; transcatheter intramyocardial transplantation of autologous bone marrow cells ( e.g., mononuclear) or placebo control, autologous bone marrow harvesting and preparation for transplantation, left heart catheterization including ventriculography, all laboratory services, and all imaging with or without guidance ( e.g., transthoracic echocardiography, ultrasound, fluoroscopy), all device(s), performed in an approved Investigational Device Exemption (IDE) study) to clarify the inclusion of the Helix trans endocardial injection catheter device in the descriptor. Additionally, we determined that APC 1590 (New Technology—Level 39 ($15,001-$20,000)) most accurately accounted for the resources associated with furnishing the procedure described by HCPCS code C9782.
For CY 2025, the OPPS payment rates were based on available CY 2023 claims data. We identified three single frequency paid claims for C9782 for ratesetting for CY 2025. Because we finalized our proposal to maintain current New Technology APC assignments for CY 2025 for New Technology APC services with fewer than 10 claims in the 4-year lookback period, we continued to assign HCPCS code C9782 to APC 1590 with a payment rate of $17,500.50 for CY 2025.
For CY 2026, there were no new claims reported for HCPCS code C9782. Therefore, there are still only three single frequency claims available for HCPCS code C9782 in the 4-year lookback period. Given our proposal to maintain current New Technology APC assignments for CY 2026 for New Technology APC services with fewer than 10 claims in the 4-year lookback period applicable for the universal low-volume APC policy moving forward, we proposed to continue to assign HCPCS code C9782 to APC 1590 (New Technology—Level 39 ($15,001-$20,000)) with a payment rate of $17,500.50.
We did not receive public comments on this provision, and our updated claims data did not show any additional claims for HCPCS Code C9782. Therefore, we are finalizing our proposal to continue to assign HCPCS code C9782 to New Technology APC 1590 with a status indication of “T” for CY 2026. Refer to Table 22 for the final OPPS New Technology APC and status indicator assignment for HCPCS code C9782. The final CY 2026 payment rates can be found in Addendum B to this final rule with comment via the internet on the CMS website.
g. Atherosclerosis Imaging-Quantitative Computer Tomography (AI-QCT) (APC 1511)
Atherosclerosis Imaging-Quantitative Computer Tomography (AI-QCT) is a Software as a Service (SaaS) that assesses the extent of coronary artery disease severity. This procedure is performed to quantify the extent of coronary plaque and stenosis in patients who have undergone coronary computed tomography analysis (CCTA). The AMA CPT Editorial Panel established the following four codes associated with this service, effective January 1, 2021:
0623T: Automated quantification and characterization of coronary atherosclerotic plaque to assess severity of coronary disease, using data from coronary computed tomographic angiography; data preparation and transmission, computerized analysis of data, with review of computerized analysis output to reconcile discordant data, interpretation and report.
0624T: Automated quantification and characterization of coronary atherosclerotic plaque to assess severity of coronary disease, using data from coronary computed tomographic angiography; data preparation and transmission.
0625T: Automated quantification and characterization of coronary atherosclerotic plaque to assess severity of coronary disease, using data from coronary computed tomographic angiography; computerized analysis of ( printed page 53539) data from coronary computed tomographic angiography.
0626T: Automated quantification and characterization of coronary atherosclerotic plaque to assess severity of coronary disease, using data from coronary computed tomographic angiography; review of computerized analysis output to reconcile discordant data, interpretation and report.
Of these four CPT codes, only CPT code 0625T was determined to be separately payable in the OPPS and was assigned to status indicator = “S” (Procedure or Service, Not Discounted When Multiple) starting October 1, 2022. We assigned CPT code 0625T to a separately payable status indicator based on the technology and its potential utilization in the HOPD setting, our evaluation of the service, as well as input from our medical advisors. The procedure was assigned to APC 1511 (New Technology—Level 11 ($900-$1000)) with a payment rate of $950.50.
For CY 2024, the OPPS payment rates were based on available CY 2022 claims data. There were 37 claims for CPT code 0625T during this time period. As this was below the threshold of 100 claims for a service within a year, we explained that we could propose to designate CPT code 0625T as a low volume service under our universal low volume New Technology APC policy and use the highest of the geometric mean cost, arithmetic mean cost, or median cost based on up to 4 years of claims data to assign code 0625T to the appropriate New Technology APC. We found the geometric mean cost for the service to be approximately $3.70, the arithmetic mean cost to be approximately $4.10, and the median cost to be approximately $3.50. Under our universal low volume New Technology APC policy, we would use the greatest of the statistical methodologies, the arithmetic mean, to assign CPT code 0625T to New Technology 1491 (New Technology Level 1A—(0-$10)) with a payment rate of $5.00. However, we acknowledged that, because CPT code 0625T was only made separately payable as part of the OPPS in October 2022, and, therefore, the CY 2022 claims available only reflected two months of data, we were concerned that we did not have sufficient claims data to justify reassignment to another New Technology APC (66 FR 69902). Therefore, consistent with our current policy to retain services within New Technology APC groups until we obtain sufficient claims data to justify reassignment (66 FR 59902), for CY 2024, we finalized our proposal to maintain CPT code 0625T's assignment to APC 1511 (New Technology—Level 11 ($901-$1000) with a payment rate of $950.50 rather than applying the universal low volume APC policy. For 2025, there were only 3 available claims for 0625T. We continued to have concerns that we did not have sufficient claims data to justify reassignment to another New Technology APC based on the CY 2023 geometric mean cost of $180. Therefore, we used our authority under section 1833(t)(2)(E) for CY 2025 to continue to assign CPT code 0625T to APC 1511 (New Technology—Level 11 ($901-$1000) with a payment rate of $950.50.
Effective January 1, 2026, the AMA CPT Editorial Panel is creating a new Category I CPT code for AI-QCT, which is currently described by CPT code 75577 (placeholder code 75XX6) (Quantification and characterization of coronary atherosclerotic plaque to assess severity of coronary disease, derived from augmentative software analysis of the data set from a coronary computed tomographic angiography, with interpretation and report by a physician or other qualified healthcare professional). CPT codes 0623T-0626T are being deleted and replaced with CPT code 75577 (placeholder code 75XX6). Since CPT placeholder code 75XX6 will not be effective until January 1, 2026, we will not have claims data available for ratesetting for this code until the CY 2028 rulemaking cycle. However, as CPT code 0625T will still be in use until December 31, 2025, we proposed to determine the payment rate for CPT placeholder code 75XX6 using the available CY 2024 claims data for CPT code 0625T.
For the CY 2026 OPPS/ASC proposed rule, there were 22 separately payable claims in the CY 2024 data reported for CPT code 0625T with a geometric mean cost of approximately $496. Given that there were fewer than 100 claims, CPT code 0625T would fall under our universal low volume New Technology APC policy where we would use the highest of the geometric mean cost, arithmetic mean cost, or median cost based on up to 4 years of claims data to assign CPT code 0625T to the appropriate New Technology APC. Using a 4-year lookback of claims data, we determined the geometric mean cost to be $13.21, the arithmetic mean cost to be $243, and the median cost to be $3.51. However, this lookback includes the claims from CY 2021 and CY 2022 that indicate that the cost of the procedure is less than $5, which would not appear to cover the basic costs of this procedure including computing time, generating a report, and having medical personnel interpret the report. The claims were also significantly lower than the expected cost of this procedure based on evidence submitted by the manufacturer when this technology was initially evaluated for placement in a New Technology APC. For CY 2024, the geometric mean cost of around $496 based on 22 claims may better reflect the cost of the procedure described by CPT code 0625T, but there are not enough claims to be confident about the result. Due to these issues, we are not confident that the results of the 4-year lookback period accurately reflect the actual costs of CPT code 0625T. Additionally, we recognize that software-based technologies are unique and rapidly evolving and that a significant fluctuation in payment may hinder patient access to these new services. We issued a comment solicitation in section III.F. of the CY 2026 OPPS/ASC proposed rule to collect information on alternative and consistent payment methods that seek to reflect the underlying value of SaaS under the OPPS to consider in future rulemaking. We hope to identify whether specific adjustments to our payment policies for SaaS are needed to more accurately and appropriately pay for these products and services across settings of care. Therefore, we proposed to use our authority under section 1833(t)(2)(E) to assign CPT code 75577 (placeholder code 75XX6) to APC 1511 (New Technology—Level 11 ($901-$1000) with a payment rate of $950.50 for CY 2026, which based on the information currently available to us, best reflects the cost of the service as described by the New Technology APC application.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: Several commenters supported the assignment of CPT code 75577 (placeholder code 75XX6) to APC 1511 (New Technology—Level 11 ($901-$1000) for CY 2025.
Response: We note that since the CY 2026 OPPS/ASC proposed rule was issued, nine additional claims for CPT code 0625T have been processed, with the updated geometric mean cost decreasing to approximately $375. Although we have nine additional claims, we are not certain that we have enough claims data to be confident in the calculated geometric mean cost for CPT code 0625T. Therefore, we are finalizing our proposal, without modification, to assign CPT code 75577 to APC 1511 (New Technology—Level 11 ($901-$1000).
Comment: Multiple commenters requested that CMS proactively ensures ( printed page 53540) that Medicare Administrative Contractors (MACs) do not issue an edit that restricts certain revenue codes for CPT code 75577 (placeholder code 75XX6), as had previously been issued for CPT code 75580.
Response: We are able to confirm that there are no MAC edits in place for CPT code 75577. Facilities may bill CPT 75577 with any appropriate revenue code. As a reminder, it is longstanding CMS policy that hospital outpatient facilities are responsible for reporting the appropriate cost centers and revenue codes on claims. As stated in section 20.5 in Chapter 4 (Part B Hospital) of the Medicare Claims Processing Manual, CMS “does not instruct hospitals on the assignment of HCPCS codes to revenue codes for services provided under OPPS since hospitals' assignment of cost vary. Where explicit instructions are not provided, HOPDs should report their charges under the revenue code that will result in the charges being assigned to the same cost center to which the cost of those services are assigned in the cost report.”
After consideration of the public comments we received, we are finalizing our proposal without modification. Refer to Table 23 for the final OPPS New Technology APC and status indicator assignment for HCPCS codes 0625T and 75577 for CY 2026. The final CY 2026 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website. In addition, we refer readers to Addendum D1 to this final rule with comment period for the status indicator meanings for all codes reported under the OPPS. Addendum D1 can also be found via the internet on the CMS website.
h. Corvia Medical Interatrial Shunt Procedure (APC 1592)
On July 1, 2020, we established HCPCS code C9760 (Non-randomized, non-blinded procedure for nyha class ii, iii, iv heart failure; transcatheter implantation of interatrial shunt or placebo control, including right and left heart catheterization, transeptal puncture, trans-esophageal echocardiography (tee)/intracardiac echocardiography (ice), and all imaging with or without guidance (for example, ultrasound, fluoroscopy), performed in an approved investigational device exemption (ide) study) to facilitate payment for the implantation of the Corvia Medical interatrial shunt.
As we stated in the CY 2021 OPPS final rule with comment period (85 FR 85947), we believe that similar resources and device costs are involved with the Corvia Medical interatrial shunt procedure and the V-Wave interatrial shunt procedure. Unlike the V-Wave interatrial shunt, which is implanted half the time the associated interatrial shunt procedure described by HCPCS code C9758 is billed, the Corvia Medical interatrial shunt is implanted every time the associated interatrial shunt procedure (HCPCS code C9760) is billed. Therefore, for CY 2021, we assigned HCPCS code C9760 to APC 1592 (New Technology—Level 41 ($25,001-$30,000)) with a payment rate of $27,500.50. We also modified the code descriptor for HCPCS code C9760 to remove the phrase “or placebo control,” from the descriptor.
For CY 2025, the OPPS payment rates were based on available CY 2023 claims data. There were two claims for HCPCS code C9760 in CY 2023. We continued to assign HCPCS code C9760 to APC 1592 (New Technology—Level 41 ($25,001- $30,000)) based on our CY 2025 policy to maintain current New Technology APC assignments for CY 2025 for New Technology APC services with fewer than 10 claims in the 4-year lookback period applicable for the universal low-volume APC policy.
For CY 2026, the OPPS payment rates were proposed to be based on available CY 2024 claims data. There were no claims for HCPSC code C9760 in CY 2024. Therefore, for CY 2026, given our proposal to maintain current New Technology APC assignments for CY 2026 for New Technology APC services with fewer than 10 claims in the 4-year lookback period applicable for the universal low-volume APC policy moving forward, we proposed to continue to assign HCPCS code C9760 ( printed page 53541) to APC 1592 (New Technology—Level 41 ($25,001- $30,000)) with a payment rate of $27,500.50.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter supported CMS' proposal and stated that it would preserve access.
Response: We thank the commenter for their support.
We note that there were no additional claims for HCPCS code C9760 in our updated claims data. Therefore, for CY 2026, we are finalizing our proposal without modification. Specifically for CY 2026, we are assigning HCPCS code C9760 to APC 1592 (New Technology—Level 41 ($25,001-$30,000)) with a payment rate of $27,500.50.
Refer to Table 24 for the final OPPS New Technology APC and status indicator assignments for HCPCS code C9760 for CY 2026. The CY 2026 payment rates can be found in Addendum B to this final rule via the internet on the CMS website. In addition, we refer readers to Addendum D1 to this final rule with comment period for the status indicator meanings for all codes reported under the OPPS. Addendum D1 can also be found via the internet on the CMS website.
i. DARI Motion Procedure (APC 1505)
Effective January 1, 2022, CPT code 0693T (Comprehensive full body computer-based markerless 3D kinematic and kinetic motion analysis and report) is associated with the DARI Motion Procedure, a service that provides human motion analysis to aid clinicians in pre- and post-operative surgical intervention and in making other treatment decisions, including selecting the best course of physical therapy and rehabilitation. The technology consists of eight cameras that surround a patient, which send live video to a computer workstation that analyzes the video to create a 3D reconstruction of the patient without the need for special clothing, markers, or devices attached to the patient's clothing or skin.
Since CPT code 0693T became effective January 1, 2022, we have had no claims for the DARI Motion Procedure and, therefore, have maintained its initial APC assignment to APC 1505 (New Technology—Level 5 ($301-$400)) with a payment of $350.50.
For CY 2026, the OPPS payment rates were proposed based on available CY 2024 claims data. Because we did not have any available claims data, we proposed to continue to assign CPT code 0693T to APC 1505 (New Technology—Level 5 ($301-400)), with a payment rate of $350.50, for CY 2026.
CMS did not receive any public comments on our proposal, and there continue to be no claims for this service. Therefore, for CY 2026, we are finalizing our proposal without modification to continue to assign CPT Code 0693T to New Technology APC 1505 (New Technology—Level 5 ($301-$400)) with a status indicator of “S” for CY 2026, found in Table 25. The CY 2026 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website.
( printed page 53542)j. Instillation of Anti-Neoplastic Pharmacologic/Biologic Agent Into Renal Pelvis (APC 1551)
Effective October 1, 2023, CMS established HCPCS code C9789 (Instillation of anti-neoplastic pharmacologic/biologic agent into renal pelvis, any method, including all imaging guidance, including volumetric measurement if performed) and assigned it to APC 1559 (New Technology—Level 22 ($2001-$2500)), with a payment rate of $2,250.50 based on our review of the clinical and resource characteristics of this service.
This code may be used to describe the unique procedure associated with the administration of the drug described by HCPCS code J9281 (Mitomycin pyelocalyceal instillation, 1 mg) or similar products. HCPCS code J9281 may be used to describe the product, JELMYTO® (mitomycin for pyelocalyceal solution). The FDA approved JELYMTO® in 2020, and the FDA approved indication and usage for JELMYTO® is as an alkylating drug indicated for the treatment of adult patients with low-grade Upper Tract Urothelial Cancer (LG-UTUS).[20]
For CY 2025, the OPPS payment rates were based on available CY 2023 claims data. Because we created HCPCS code C9789 effective October 1, 2023, we had limited claims data from CY 2023 available for CY 2025 rulemaking. Specifically, we only had 6 claims available for ratesetting, so we maintained the New Technology APC assignment of APC 1559 (New Technology—Level 22 ($2001-$2500)) with a payment of $2,250.50 for CY 2025, based on our CY 2025 policy to maintain the New Technology APC assignment for New Technology APC services with fewer than 10 claims in the 4-year lookback period applicable for the universal low-volume APC policy.
For CY 2026, the OPPS payment rates were proposed based on available CY 2024 claims data. HCPCS code C9789 had 109 single frequency claims in CY 2024, which exceeds the 100 claims threshold generally used for the universal low volume APC policy. The geometric mean cost for HCPCS code C9789 is approximately $1,401. Therefore, for CY 2026, we proposed to assign HCPCS code C9789 to APC 1553 (New Technology—Level 16 ($1401-$1500)) with a payment rate of $1,450.50.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter supported HCPCS code C7989 remaining in a New Technology APC but requested that HCPCS C7989 remain assigned to New Technology APC 1559 (New Technology—Level 22 ($2001-$2500)). The commenter stated they do not believe providers are consistently reporting HCPCS code C9789 when JELMYTO® is administered and are using alternative CPT codes for the procedure. As a result, the commenter believes the 2024 claims do not adequately reflect the costs for administering JELMYTO®. The commenter expressed concerns about beneficiaries having future access to the instillation procedure for JELMYTO® if the procedure was no longer assigned to a New Technology APC and requested that CMS issue a MLN Matters® or similar guidance to provide information on how to code and bill the instillation procedure with the drug JELMYTO®.
Response: Providing coding guidance is out of scope for the OPPS/ASC final rule with comment period. We note that if hospitals have questions about appropriate coding that they cannot resolve on their own, the initial first step would be to review the HCPCS code descriptors or consult the appropriate Medicare Administrative Contractor (MAC) for their jurisdiction. We note that HCPCS code J9281 is assigned to a status indicator of `K' (Nonpass-Through Drugs and Nonimplantable Biologicals, Including Therapeutic Radiopharmaceuticals; Paid under OPPS; separate APC payment.) and procedures and services assigned to a New Technology APC are excluded from the C-APC packaging policy. (See the Medicare Claims Processing Manual, Chapter 4, Section 10.2.3 for a list of exclusions to the comprehensive APC packaging policy.) Therefore, providers may receive separate payment for both the drug and installation procedure when providing this service.
In response to maintaining the CY 2025 New Technology APC assignment, we note that HCPCS code C9789 has over 100 claims, and therefore, the New Technology APC assignment is based on the geometric mean cost for that code. We note that the geometric mean cost and claims data for HCPCS code C9789 has changed since the CY 2026 OPPS/ASC proposed rule. Based on the updated claims data for this final rule with comment period, the geometric mean cost for HCPCS code C9789 is $1,211 based on 222 single frequency claims. We believe that 222 single frequency claims is adequate for ratesetting for this service. As we do every year, we will reevaluate the APC assignments for these codes in the next rulemaking cycle. We remind hospitals that we review, on an annual basis, the APC assignments for all items and services paid under the OPPS.
In summary, after consideration of the public comment we received, we are finalizing with modification a New Technology APC assignment for HCPCS code C9789 to APC 1551 (New Technology—Level 14 ($1201-$1300)) with a payment rate of $1,250.50.
Refer to Table 26 for the final OPPS New Technology APC and status indicator assignments for CPT code C9789 for CY 2026. The final CY 2026 payment rates for this code can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 to this final rule with comment period for the SI definitions for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
( printed page 53543)k. LimFlow TADV Procedure CPT Code 0620T (APC 1580)
The LimFlow TADV procedure which is described by CPT code 0620T (Endovascular venous arterialization, tibial or peroneal vein, with transcatheter placement of intravascular stent graft(s) and closure by any method, including percutaneous or open vascular access, ultrasound guidance for vascular access when performed, all catheterization(s) and intraprocedural roadmapping and imaging guidance necessary to complete the intervention, all associated radiological supervision and interpretation, when performed) is an endovascular procedure that is used to treat patients with chronic limb-threatening ischemia. According to the developer, these patients are no longer eligible for conventional endovascular or open bypass surgery to treat their artery blockage, and without this procedure, they are likely to face limb amputation.
CPT code 0620T was established in January 2021 and was assigned to APC 5194 (Level 4 Endovascular Procedures) with a payment rate of approximately $17,400, which is the highest-paying APC for endovascular procedures. While we proposed to continue to assign CPT code 0620T to APC 5194 for CY 2024, we finalized a reassignment from a clinical APC to a New Technology APC with a higher payment rate based on comments received expressing concern that the low payment rate of the procedure would discourage providers from performing the procedure and deny access to the procedure. For CY 2024, the procedure was assigned to APC 1578 (New Technology—Level 41 ($25,001-$30,000)). For CY 2025 ratesetting, there were 11 single frequency claims for CPT code 0620T in the CY 2023 claims data. As this is below the threshold of 100 claims for a service within a year, we applied our universal low volume APC policy and used the highest of the geometric mean cost, arithmetic mean cost, or median cost based on up to 4 years of claims data to assign the service to the appropriate New Technology APC. Based on our review of the available claims and the application of the universal low volume APC policy, we assigned HCPCS code 0620T to APC 1579 (New Technology—Level 42 ($30,001-$40,000)) with a payment rate of $35,000.50 based on the median cost of approximately $36,400.
For CY 2026, the OPPS payment rates were proposed to be based on available CY 2024 claims data. There were 19 single frequency claims for 0620T in the CY 2024 claims data. As this is below the threshold of 100 claims for a service within a year, we proposed to again apply our universal low volume APC policy and use the highest of the geometric mean cost, arithmetic mean cost, or median cost based on up to 4 years of claims data to assign the service to the appropriate New Technology APC. Based on our review of the available claims, we have determined that the arithmetic mean is approximately $39,000; the median is approximately $38,000; and the geometric mean cost is approximately $35,000. Of these, the arithmetic mean is the statistical methodology that estimated the highest cost for the service. The payment rate calculated using this methodology falls within the cost band for APC 1579 (New Technology—Level 42 ($30,001-$40,000)) with a payment rate of $35,000.50. Therefore, for CY 2026, we proposed to designate this service as a low volume service under our universal low volume APC policy and to continue to assign HCPCS code 0620T to APC 1579 (New Technology—Level 42 ($30,001-$40,000)) with a payment rate of $35,000.50.
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: A commenter supported the proposal to assign CPT code 0620T to APC 1579 (New Technology—Level 42 ($30,001-$40,000)) with a payment rate of $35,000.50 for CY 2026 based on the application of the universal low volume APC policy. The commenter stated that the assignment accurately reflects the resources used in the procedure.
Response: Based on the public comments received, we are finalizing our proposal to apply our universal low volume APC policy and use the highest of the geometric mean cost, arithmetic mean cost, or median cost based on up to 4 years of claims data to assign the service to the appropriate New Technology APC. Three additional claims for CY 2024 have been processed since the CY 2026 OPPS/ASC proposed rule. Our analysis of the updated claims data found that the greater of the geometric mean, arithmetic mean, or median cost calculated for CPT code 0620T is $43,748.64 based on the arithmetic mean. This value falls within APC 1580 (New Technology—Level 43 ($40,001-$50,000)) with a payment rate of $45,000.50. Therefore, for CY 2026, we are assigning CPT code 0620T to APC 1580 based on the application of the universal low volume APC policy. Refer to Table 27 for the final OPPS New Technology APC and status indicator assignments for CPT code 0620T for CY 2026. The final CY 2026 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website.
( printed page 53544)l. Liver Histotripsy Service (APC 1579)
CPT code 0686T (Histotripsy ( i.e., non-thermal ablation via acoustic energy delivery) of malignant hepatocellular tissue, including image guidance) was first effective July 1, 2021, and describes the histotripsy service associated with the use of the HistoSonics system. Histotripsy is a non-invasive, non-thermal, mechanical process that uses a focused beam of sonic energy to destroy cancerous liver tumors and is currently in a non-randomized, prospective clinical trial to evaluate the efficacy and safety of the device for the treatment of primary or metastatic tumors located in the liver.[21] When HCPCS code 0686T was first effective, the histotripsy procedure was designated as a Category A IDE clinical study (NCT04573881). Since devices in Category A IDE studies are excluded from Medicare payment, payment for CPT code 0686T only reflected the cost of the service that is performed (absent the cost of the device) each time it is reported on a claim. On March 2, 2023, the histotripsy IDE clinical study was re-designated as a Category B (Non-experimental/Investigational) IDE study. Due to this new designation, payment for CPT code 0686T in CY 2024 reflected payment for both the service that was performed and the device used each time it was reported on a claim. For CY 2024, we assigned CPT code 0686T to APC 1576 (New Technology—Level 39 ($15,001-$20,000)) with a payment rate of $17,500.50. For CY 2025, we continued to assign CPT code 0686T to APC 1576 (New Technology—Level 39 ($15,001-$20,000) due to our CY 2025 policy to maintain current New Technology APC assignments for CY 2025 for New Technology APC services with fewer than 10 claims in the 4-year lookback period applicable for the universal low volume APC policy, and based on the fact that there were only 3 claims for CPT code 0686T in the prior 4-year period.
For CY 2026, the OPPS payment rates were proposed to be based on available CY 2024 claims data. For the CY 2026 OPPS/ASC proposed rule, we identified 94 claims for CPT code 0686T within this period. As this is below the threshold of 100 claims for a service within a year, we propose to apply our universal low volume APC policy and use the highest of the geometric mean cost, arithmetic mean cost, or median cost based on up to 4 years of claims data to assign CPT code 0686T to the appropriate New Technology APC. We identified $32,307.41 as the arithmetic mean, $20,577.77 as the median, and $21,264.91 as the geometric mean. The arithmetic mean was the statistical methodology that estimated the highest cost for CPT code 0686T. For CY 2026, we proposed to reassign CPT code 0686T to APC 1579 (New Technology—Level 42 ($30,001-$40,000)) with a payment rate of $35,000.50.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter supported the proposal to reassign CPT code 0686T to APC 1579 (New Technology—Level 42 ($30,001-$40,000)) for CY 2026 based on the 94 claims data available.
Response: We thank the commenter for their input.
Six additional claims for CY 2024 have been processed since the CY 2026 OPPS/ASC proposed rule, bringing the total number of claims to 100. Since the total number of CY 2024 single frequency claims for CY code 0686T surpasses the 99 claim threshold for the universal low volume APC policy, we would use the geometric mean cost of the CY 2024 claims data to set the payment rate for CY 2026 under our standard ratesetting methodology, rather than the highest of the three statistical methodologies over a 4-year lookback period. Based on the updated claims data available for this final rule with comment period, the geometric mean cost for HCPCS code 0686T is around $16,008. Due to the updated claims data available for this final rule with comment period, we are finalizing a New Technology APC assignment for HCPCS code 0686T to APC 1576 (New Technology—Level 39 ($15,001-$20,000)) with a payment rate of around $17,500.50. This is the same APC to which the service is currently assigned in CY 2025.
Refer to Table 28 for the final OPPS New Technology APC and status indicator assignments for CPT code 0686T for CY 2026. We refer readers to Addendum B to this final rule with comment period for the final CY 2026 OPPS payment rate for this code. ( printed page 53545) Addendum B is available via the internet on the CMS website.
m. LiverMultiScan Service (APC 1511)
CPT codes 0648T (Quantitative magnetic resonance for analysis of tissue composition ( e.g., fat, iron, water content), including multiparametric data acquisition, data preparation and transmission, interpretation and report, obtained without diagnostic mri examination of the same anatomy ( e.g., organ, gland, tissue, target structure) during the same session; single organ) and 0649T (Quantitative magnetic resonance for analysis of tissue composition ( e.g., fat, iron, water content), including multiparametric data acquisition, data preparation and transmission, interpretation and report, obtained with diagnostic mri examination of the same anatomy ( e.g., organ, gland, tissue, target structure); single organ (list separately in addition to code for primary procedure)) became effective July 1, 2021 and are associated with the LiverMultiScan service.
LiverMultiScan is a Software as a medical Service (SaaS) that is intended to aid the diagnosis and management of chronic liver disease, the most prevalent of which is Non-Alcoholic Fatty Liver Disease (NAFLD). It provides standardized, quantitative imaging biomarkers for the characterization and assessment of inflammation, hepatocyte ballooning, and fibrosis, as well as steatosis, and iron accumulation. LiverMultiScan receives MR images acquired from patients' providers and analyzes the images using their proprietary Artificial Intelligence (AI) algorithms. It then sends the providers a quantitative metric report of the patient's liver fibrosis and inflammation. In accordance with our SaaS add-on codes policy (87 FR 72032 to 72033), SaaS CPT add-on codes are assigned to the same APCs and status indicators as their standalone codes. Thus, CPT code 0649T, the add-on code for LiverMultiScan, is assigned to the identical APC and status indicator as CPT code 0648T, the standalone code for the same service.
For CY 2024 and CY 2025, we used our equitable adjustment authority under section 1833(t)(2)(E) to continue to assign CPT codes 0648T and 0649T to APC 1511 (New Technology—Level 11 ($901-$1,000) with a payment rate of $950.50.
For CY 2026, the OPPS payment rates were proposed based on available CY 2024 claims data. We identified 107 single frequency claims for CPT code 0648T and 104 single frequency claims CPT code 0649T for CY 2024. The geometric mean cost for CPT code 0648T was $253.68 and the geometric mean cost for CPT code 0649T was $162.96. Based on the geometric mean cost for CPT code 0648T, we would have assigned CPT codes 0648T and 0649T to APC 1504 (New Technology—Level 4 ($201-$300)) with a payment rate of $250.50. However, assigning these SaaS technologies based on the geometric costs would have decreased the payment rate by around 75 percent. We recognized that software-based technologies, like those described by CPT codes 0648T and 0649T, continue to evolve and that the limited claims data may not have truly represented the cost of this service. We issued a comment solicitation in section III.F. of the CY 2026 OPPS/ASC proposed rule to collect information on alternative and consistent payment methods that seek to reflect the underlying value of SaaS technologies under the OPPS to consider in future rulemaking. We hoped to identify whether specific adjustments to our payment policies for SaaS technologies are needed to more accurately and appropriately pay for these products and services across settings of care. Therefore, we proposed to use our authority under section 1833(t)(2)(E) of Act for CY 2026 to continue to assign CPT codes 0648T and 0649T to APC 1511 (New Technology—Level 11 ($901-$1000)) with a payment rate of $950.50, which we believed best reflected the cost of the service, based on information provided by the applicant.
We note that since the CY 2026 OPPS/ASC proposed rule was published, CPT code 0648T has an updated geometric mean cost of around $269 based on 114 single frequency claims, and CPT code 0649T has an updated geometric mean cost of around $158 based on 111 single frequency claims.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A number of comments were received in support of maintaining the APC assignments to New Technology APC 1511. Commenters stated that maintaining the current payment rate for LiverMultiScan will allow continued access to this valuable non-invasive imaging tool service. Many commenters noted the clinical relevance of these procedures in obtaining information on a patient's liver health and developing appropriate treatment plans.
A commenter supported the proposed New Technology APC assignment but also provided possible explanations for the payment variability, including the inappropriate use of CPT codes and distorted data due to inappropriate cost to charge ratios. The commenter noted the need for reliable claims data for ratesetting.
Response: We appreciate the commenters' input and support for the proposed APC assignment. We hope to glean valuable information from the SaaS comment solicitation that will help us understand the potential factors that affect payment consistency. We hope by having this additional information, we can put forth a policy in future rulemaking that provides a ( printed page 53546) more stable payment method for SaaS technologies.
After consideration of the public comment we received, we are finalizing our proposal without modification. We will use our equitable adjustment authority under section 1833(t)(2)(E) of the Act to continue to assign CPT codes 0648T and 0649T to New Technology APC 1511 (New Technology—Level 11 ($901-$1,000) with a payment rate of $950.50 for CY 2026.
Refer to Table 29 for the OPPS New Technology APC and status indicator assignments for CPT codes 0648T and 0649T for CY 2026. The final CY 2026 payment rates can be found in Addendum B to this final rule via the internet on the CMS website. In addition, we refer readers to Addendum D1 to this final rule with comment period for the status indicator meanings for all codes reported under the OPPS. Addendum D1 can also be found via the internet on the CMS website.
n. Optellum Lung Cancer Prediction (LCP) (APC 1508)
CPT codes 0721T (Quantitative computed tomography (CT) tissue characterization, including interpretation and report, obtained without concurrent CT examination of any structure contained in previously acquired diagnostic imaging) and 0722T (Quantitative computed tomography (CT) tissue characterization, including interpretation and report, obtained with concurrent CT examination of any structure contained in the concurrently acquired diagnostic imaging dataset (list separately in addition to code for primary procedure)) became effective July 1, 2022, and are associated with the Optellum LCP technology. The Optellum LCP applies an algorithm to a patient's CT scan to produce a raw risk score for a patient's pulmonary nodule. The physician uses the risk score to quantify the risk of lung cancer and to determine what the next management step should be for the patient (for example, CT surveillance versus invasive procedure). In accordance with our SaaS add-on codes policy (87 FR 72032 to 72033), SaaS CPT add-on codes are assigned to the same APCs and status indicators as their standalone codes. Thus, CPT code 0722T, the add-on code for the Optellum LCP service, is assigned to the identical APC and status indicator as CPT code 0721T, the standalone code for the same service. For CY 2024, we assigned CPT codes 0721T and 0722T to APC New Technology 1508 (New Technology—Level 8 ($601-$700)).
For CY 2025, we continued to assign CPT codes 0721T and 0722T to APC 1508 (New Technology—Level 8 ($601-$700)) with a payment rate of $650.50 based on our CY 2025 policy to maintain New Technology APC assignments for CY 2025 for New Technology APC services with fewer than 10 claims in the 4-year lookback period applicable for the universal low-volume APC policy.
For CY 2026, OPPS payment rates were proposed based on available CY 2024 claims data. There were 496 combined claims for CPT codes 0721T and 0722T for CY 2024: 7 claims for CPT code 0721T and 489 claims for 0722T. The geometric mean cost of CPT code 0721T is $30.24 and the geometric mean cost for CPT code 0722T is $60.47. Based on the geometric mean cost for CPT code 0722T, which has a significantly greater number of claims than 0721T, we would assign CPT codes 0721T and 0722T to APC 1502 (New Technology—Level 2 ($51-$100) with a payment rate of $75.50. However, assigning these SaaS technologies based on the geometric costs would decrease the payment rate by close to 90 percent in 1 year. We recognize that software-based technologies, like those described by CPT codes 0721T and 0722T, continue to evolve and that the limited claims data that we have may not truly represent the cost of this service. We issued a comment solicitation in section III.F. of the CY 2026 OPPS/ASC proposed rule to collect information on alternative and consistent payment methods that seek to reflect the underlying value of SaaS under the OPPS to consider in future rulemaking. We hope to identify whether specific adjustments to our payment policies for SaaS technologies are needed to more accurately and appropriately pay for these products and services across settings of care.
While we recognize that there are certain unknowns regarding the cost of technologies like the Optellum LCP service, we believe it would be unlikely for the cost to be 90 percent less than the initial estimated costs based on our ( printed page 53547) review of the information provided in the New Technology APC application. Therefore, we proposed to use our authority under section 1833(t)(2)(E) for CY 2026 to continue to assign CPT codes 0721T and 0722T to APC 1508 (New Technology—Level 8 ($601-$700)) with a payment rate of $650.50 based on the information provided to us by the manufacturer in their application, which we believed may better reflect the cost of the service at the time of the CY 2026 OPPS/ASC proposed rule than the available claims data.
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: Commenters were supportive of the proposal to use our equitable adjustment authority to continue to assign CPT codes 0721T and 0722T to APC 1508. Commenters believe the proposed APC assignment aligned with the cost of the service. Commenters expressed concern that the claims data do not accurately reflect the true cost to hospitals and imaging providers of delivering the service. A commenter stated that they believe the inaccurate claims data was the result of hospitals reporting inappropriate revenue codes for the service and expressed support for the creation of a new cost center with revenue codes for AI-based services.
Response: While CMS does not provide billing advice to hospitals, we encourage manufacturers and distributors to provide outreach to hospitals regarding billing practices that are most appropriate for their individual technologies. We will consider the commenter's suggestion to create a new cost center for AI-based services as we explore how to appropriately pay for software as a service in future rulemaking. We note that we solicited comments on payment policies for “software as a service” in the CY 2026 OPPS/ASC proposed rule and refer readers to section III.F. of this final rule with comment period for a summary of the comments received.
After consideration of the public comments, we are finalizing our proposal without modification. Specifically, for CY 2026, we are finalizing our proposal to assign CPT codes 0721T and 0722T to APC 1508 (New Technology—Level 8 ($601-$700)) with a payment rate of $650.50.
Refer to Table 30 for the proposed and final OPPS New Technology APC and status indicator assignments for HCPCS codes 0721T and 0722T for CY 2026. The final CY 2026 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website.
o. Quantitative Magnetic Resonance (QMR) for Analysis of Tissue Composition (APC 1511)
Effective January 1, 2022, CPT codes 0697T (Quantitative magnetic resonance for analysis of tissue composition ( e.g., fat, iron, water content), including multiparametric data acquisition, data preparation and transmission, interpretation and report, obtained without diagnostic mri examination of the same anatomy ( e.g., organ, gland, tissue, target structure) during the same session; multiple organs) and 0698T (Quantitative magnetic resonance for analysis of tissue composition ( e.g., fat, iron, water content), including multiparametric data acquisition, data preparation and transmission, interpretation and report, obtained with diagnostic mri examination of the same anatomy ( e.g., organ, gland, tissue, target structure); multiple organs (list separately in addition to code for primary procedure)) are associated with the CoverScan Software as a medical Service (SaaS). This service is a medical image management and processing software package that analyzes MR data and provides quantified metrics of multiple organs such as the heart, lungs, liver, spleen, pancreas, and kidney. For CY 2024, we assigned CPT codes 0697T and 0698T to APC 1511 (New Technology—Level 11 ($900-$1,000)).
For CY 2025, there were fewer than 100 claims for ratesetting and because we recognized that the number of claims used to apply our universal low volume policy (using the highest of the geometric mean cost, arithmetic mean cost, or median cost based on up to 4 ( printed page 53548) years of claims data) may not have represented the cost of this SaaS, we used our equitable adjustment authority under section 1833(t)(2)(E) to continue to assign CPT codes 0697T and 0698T to APC 1511 (New Technology—Level 11 ($900-$1,000)) with a payment of $950.50. In accordance with our SaaS add-on codes policy (87 FR 72032 to 72033), SaaS CPT add-on codes are assigned to the same APCs and status indicators as their standalone codes. Thus, CPT code 0698T, the add-on code for CoverScan was assigned to the identical APC and status indicator as CPT code 0697T, the standalone code for the same service.
For CY 2026, the proposed OPPS payment rates were based on available CY 2024 claims data. We identified 55 single frequency claims for CPT code 0698T and no claims for CPT code 0697T in CY 2024. Because the SaaS standalone and add-on services are identical, we believe it is important for purposes of ratesetting to use the data that is available, whether it is associated with the standalone code or the add-on code. As the 55 single frequency claims are below the threshold of 100 claims for a service within a year, we would have proposed applying our universal low volume APC policy and would have used the highest of the geometric mean cost, arithmetic mean cost, or median cost based on up to 4 years of claims data to assign CPT codes 0697T and 0698T to the appropriate New Technology APC. Our analysis of the combined data, zero claims for CPT code 0697T and 137 claims for CPT code 0698T, yielded a geometric mean cost of approximately $422, an arithmetic mean cost of approximately $600, and a median cost of approximately $777. The median cost is the statistical methodology that estimated the highest cost for CPT codes 0697T and 0698T. Based on the median cost, we would have proposed to assign CPT codes 0697T and 0698T to APC 1509 (New Technology—Level 9 ($701-$800)) with a payment of $750.50.
As in CY 2025, for the CY 2026 OPPS/ASC proposed rule, we recognized that the few claims available for CPT codes 0697T and 0698T may not have truly represented the cost of this SaaS. We recognized that software-based technologies, like those described by CPT codes 0697T and 0698T, are unique and rapidly evolving and that a significant fluctuation in payment may hinder patient access to these new services. We issued a comment solicitation in section III.F of the CY 2026 OPPS/ASC proposed rule to collect information on alternative and consistent payment methods that seek to reflect the underlying value of SaaS under the OPPS to consider in future rulemaking. We hoped to identify whether specific adjustments to our payment policies for SaaS are needed to more accurately and appropriately pay for these products and services across settings of care.
Because we have continued to have the same concerns about payment variability and the possible effects the payment may have on patient access to SaaS, we proposed to use our authority under section 1833(t)(2)(E) for CY 2026 to continue to assign CPT codes 0697T and 0698T to APC 1511 (New Technology—Level 11 ($900-$1,000)) with a payment of $950.50 which we believe best reflects the cost of the service at this time.
Comment: A commenter provided possible explanations for the payment variability, including the inappropriate use of CPT codes and distorted data due to inappropriate cost to charge ratios and stated the need for reliable claims data for ratesetting. While concerns were expressed as to the payment variability, the commenter supported the proposed APC assignment to APC 1511 (New Technology—Level 11 ($900-$1,000)) with a payment of $950.50. The commenter indicated that this APC assignment provides adequate payment for this service which enables beneficiaries to have continued access to these technologies.
Response: We appreciate the commenter's input and support for the proposed APC assignment. We hope to glean valuable information from the SaaS comment solicitation that will help us understand the potential factors that affect payment consistency. We hope by having this additional information, we can put forth a policy in future rulemaking that provides a more stable payment method for SaaS technologies.
After consideration of the public comment we received, we are finalizing our proposal without modification. We will use our equitable adjustment authority under section 1833(t)(2)(E) to continue to assign CPT codes 0697T and 0698T to New Technology APC 1511 (New Technology—Level 11 ($901-$1,000) with a payment rate of $950.50 for CY 2026. Refer to Table 31 for the OPPS New Technology APC and status indicator assignments for CPT codes 0697T and 0698T for CY 2026. The final CY 2026 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website. In addition, we refer readers to Addendum D1 to this final rule with comment period for the status indicator meanings for all codes reported under the OPPS. Addendum D1 can also be found via the internet on the CMS website.
( printed page 53549)p. Quantitative Magnetic Resonance Cholangiopancreatography (QMRCP) (APC 1511)
Effective July 1, 2022, CPT codes 0723T (Quantitative magnetic resonance cholangiopancreatography (QMRCP) including data preparation and transmission, interpretation and report, obtained without diagnostic magnetic resonance imaging (MRI) examination of the same anatomy ( e.g., organ, gland, tissue, target structure) during the same session) and 0724T (Quantitative magnetic resonance cholangiopancreatography (QMRCP), including data preparation and transmission, interpretation and report, obtained with diagnostic magnetic resonance imaging (MRI) examination of the same anatomy ( e.g., organ, gland, tissue, target structure) (list separately in addition to code for primary procedure)) are associated with the QMRCP Software as a medical Service (SaaS). The service performs quantitative assessment of the biliary tree and gallbladder. It uses a proprietary algorithm that produces a three-dimensional reconstruction of the biliary tree and pancreatic duct and also provides precise quantitative information of biliary tree volume and duct metrics. In accordance with our SaaS add-on codes policy (87 FR 72032 to 72033), SaaS CPT add-on codes are assigned to the same APCs and status indicators as their standalone codes. Consistent with our SaaS add-on codes policy, CPT code 0724T, the add-on code for QMRCP is assigned to the identical APC and status indicator as CPT code 0723T, the standalone code for the same service. For CY 2024, we assigned CPT codes 0723T and 0724T to APC 1511 (New Technology—Level 11 ($900-$1,000)). For CY 2025, we continued to assign CPT codes 0723T and 0724T to APC 1511 (New Technology—Level 11 ($900-$1,000)) based on there being fewer than 10 claims in the 4-year lookback period and the exception from the universal low-volume APC policy.
For CY 2026, the OPPS payment rates were proposed to be based on available CY 2024 claims data. There were only four new claims for HCPCS code 0724T and no claims for CPT code 0723T. Given our proposal to maintain current New Technology APC assignments for CY 2026 for New Technology APC services with fewer than 10 claims in the 4-year lookback period due to an exception from the universal low-volume APC policy, we proposed, for CY 2026, to continue to assign CPT codes 0723T and 0724T to APC 1511 (New Technology—Level 11 ($901-$1000)), with a payment rate of $950.50.
Comment: A commenter supported the proposals to continue to assign 0723T and 0724T to APC 1511.
Response: We thank the commenter for their support.
Our updated claims data for the 4-year lookback period for the universal low volume APC policy shows no claims for HCPCS code 0723T and four single claims for 0724T. Because we are finalizing our proposal to maintain current New Technology APC assignments for CY 2026 for New Technology APC services with fewer than 10 claims in the 4-year lookback period, we are continuing to assign HCPCS code 0723T and 0724T to APC 1511.
Refer to Table 32 for the final OPPS New Technology APC and status indicator assignments for CPT codes 0723T and 0724T for CY 2026. The final CY 2026 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website.
( printed page 53550)q. Supervised Visits for Esketamine Self-Administration (APCs 1512 and 1518)
On March 5, 2019, FDA approved Spravato[TM] (esketamine) nasal spray, used in conjunction with an oral antidepressant.[22] for treatment of depression in adults who have tried other antidepressant medicines but have not benefited from them (treatment-resistant depression (TRD)). This is the first FDA approval of esketamine for any use.
Esketamine is a noncompetitive N-methyl D-aspartate (NMDA) receptor antagonist. It is a nasal spray supplied as an aqueous solution of esketamine hydrochloride in a vial with a nasal spray device. Each device delivers two sprays containing a total of 28 mg of esketamine. Patients would require either two (2) devices (for a 56 mg dose) or three (3) devices (for an 84 mg dose) per treatment.
Because of the risk of serious adverse outcomes resulting from sedation and dissociation and respiratory depression caused by esketamine nasal spray administration, and the potential for abuse and misuse of the product, it is only available through a restricted distribution system under a Risk Evaluation and Mitigation Strategy (REMS). A REMS is a drug safety program that the FDA can require for certain medications with serious safety concerns to help ensure the benefits of the medication outweigh its risks. The SpravatoTM REMS program requires, among other requirements, that the esketamine nasal spray be dispensed and administered to enrolled patients in health care settings that are certified in the REMS. See www.fda.gov for more information regarding the SpravatoTM REMS program requirements.
A treatment session of esketamine consists of instructed nasal self-administration by the patient followed by a period of at least 2 hours post-administration observation of the patient under direct supervision of a health care professional in the certified health care setting. Refer to the CY 2020 PFS final rule and interim final rule for more information about supervised visits for esketamine nasal spray self-administration (84 FR 63102 through 63105); see also the Spravato REMS document and Spravato labeling available on the FDA website.[23]
To facilitate prompt beneficiary access to the new, potentially life-saving treatment for TRD using esketamine, we created two new HCPCS G codes, G2082 and G2083, effective January 1, 2020. HCPCS code G2082 is for an outpatient visit for the evaluation and management of an established patient who requires the supervision of a physician or other qualified health care professional and provision of up to 56 mg of esketamine through nasal self-administration and includes two hours of post-administration observation. HCPCS code G2083 describes a similar service to HCPCS code G2082 but involves the administration of more than 56 mg of esketamine.
For CY 2025, HCPCS code G2082 was assigned to APC 1513 (New Technology—Level 13 ($1101-$1200)) with a payment rate of $1,150.50 and HCPCS code G2083 was assigned to APC 1516 (New Technology—Level 16 ($1401-$1,500)) with a payment rate of $1,450.50.
For CY 2026, the OPPS payment rates were proposed based on available CY 2024 claims data as the available single frequency claims exceed the 100 claims threshold generally used for our universal low volume policy. Therefore, for CY 2026, we proposed to assign HCPCS codes G2082 and G2083 to New Technology APCs based on each of the codes' geometric mean costs. Specifically, we proposed to assign HCPCS code G2082 to APC 1512 (New Technology—Level 12 ($1001-$1100)) with a payment rate of $1,050.50 based on its geometric mean cost of $1,019, which was calculated using the available 558 single frequency claims from CY 2024 claims data. We also proposed to assign HCPCS code G2083 to APC 1517 (New Technology—Level 17 ($1501-$1600)) with a payment rate ( printed page 53551) of $1,550.50 based on its geometric mean cost of $1,549, which was calculated using the available 4,138 single frequency claims from CY 2024 claims data. As we continue to gather adequate claims data on these codes, we invited public comment on the appropriate clinical APC assignments for HCPCS codes G2082 and G2083.
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: Commenters supported the proposed APC assignment. They encouraged CMS to continue to provide adequate payment and not to undervalue this needed treatment, as that would cause access issues for beneficiaries in need of this service. Some commenters requested that we maintain the APC assignment for HCPCS code G2082 in APC 1513 (New Technology—Level 13 ($1101-$1200)) with a payment of $1,150.50, stating that adequate payment is needed to preserve access while additional cost data is collected. Another commenter suggested that we create a new clinical APC family with two levels that are specific to this service. The commenter stated that the creation of the new APC would ensure clinical and resource homogeneity and provide an opportunity in the future for similar services to be placed in the same APC.
Response: As readers are aware, we have been contemplating potential clinical APC assignments for the past number of rulemaking cycles but are not convinced as to what clinical APC would be appropriate in terms of clinical and resource homogeneity. We appreciate the public's suggestion of creating a new APC for this service.
We note the geometric mean costs for both HCPCS codes G2082 and G2083 have changed since the CY 2026 OPPS/ASC proposed rule. Based on the updated claims data available for this final rule, the geometric mean cost for HCPCS code G2082 is around $1,015 and the geometric mean cost for HCPCS code G2083 is around $1,612. Based on updated claims data available for this final rule with comment period, we are finalizing a New Technology APC assignment for HCPCS code G2083 to APC 1518 (New Technology—Level 18 ($1601-$1700)) with a payment of $1,650.50.
Finally, we note that because we have gathered additional claims data and seen increases in claims volume, we will continue to consider potential clinical APC placements for HCPCS codes G2082 and G2083 through future rulemaking.
Refer to Table 33 for the CY 2026 proposed and final APC and status indicator assignments for HCPCS codes G2082 and G2083. The CY 2026 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website.
r. Surfacer® Inside-Out® Access Catheter System (APC 1534)
HCPCS code C9780 (Insertion of central venous catheter through central venous occlusion via inferior and superior approaches ( e.g., inside-out technique), including imaging guidance) describes the procedure associated with the use of the Surfacer® Inside-Out® Access Catheter System that is designed to address central venous occlusion. HCPCS code C9780 was established on October 1, 2021, and since its establishment the code has been assigned to APC 1534 (New Technology—Level 34 ($8001-$8500)).
For the CY 2026 OPPS/ASC proposed rule, there were only three new claims for HCPCS code C9780. Therefore, there are only seven single frequency claims available for HCPCS code C9780 in the 2 years of data since the code has been available. Given our proposal to maintain current New Technology APC ( printed page 53552) assignments for CY 2026 for New Technology APC services with fewer than 10 claims in the 4-year lookback period applicable for the universal low-volume APC policy, we proposed for CY 2026 to continue to assign HCPCS code C9780 to APC 1534 (New Technology—Level 34 ($8001-$8500)) with a payment rate of $8,250.50.
We did not receive any public comments on our proposal to continue to assign CPT code C9870 to APC 1534 (New Technology—Level 34 ($8001-$8500)). We note that there were no additional claims in our updated claims data. Therefore, given our policy to maintain current New Technology APC assignments for CY 2026 for New Technology APC services with fewer than 10 claims in the 4-year lookback period applicable for the universal low volume APC policy, we are finalizing as proposed to continue to assign CPT code C9870 to APC 1534. Refer to Table 34 for the final OPPS New Technology APC and status indicator assignment for HCPCS code C9780. The final CY 2026 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website.
s. Transcatheter Atrial Shunt System (TASS) (APC 1537)
The Transcatheter Atrial Shunt System (TASS) is a nitinol self-expanding cardiovascular implant consisting of four arms including two left atrial (LA) arms and two coronary sinus (CS) arms placed between the left atrium and coronary sinus to create a 7mm flow diameter channel for blood to flow from the high pressure region of the left atrium to the lower pressure region of the right atrium via the coronary sinus.
TASS was designated as a Category A IDE clinical study (NCT03523416) on July 31, 2019. Effective October 1, 2023 CMS created HCPCS code C9792 (Blinded or nonblinded procedure for symptomatic New York Heart Association (NYHA) Class II, III, IVa heart failure; transcatheter implantation of left atrial to coronary sinus shunt using jugular vein access, including all imaging necessary to intra procedurally map the coronary sinus for optimal shunt placement ( e.g., TEE or ICE ultrasound, fluoroscopy), performed under general anesthesia in an approved investigational device exemption (IDE) study) to describe the TASS service and assigned it to APC 1537 (New Technology—Level 37 ($9501-$10000)) with a payment rate of $9750.50. Since devices in Category A IDE studies are not covered by Medicare during the study, the payment for HCPCS code C9792 reflects only the cost of the service that is performed each time it is reported on a claim.
For CY 2025, there were no claims available, so we maintained the APC assignment for HCPCS code C9792 to APC 1537 (New Technology—Level 37 ($9501-$10000)).
For CY 2026, the proposed OPPS payment rates are based on available CY 2024 claims data. We do not have any claims data for HCPCS code C9792. Therefore, for CY 2026, we proposed to continue to assign HCPCS code C9792 to APC 1537 (New Technology—Level 37 ($9501-$10000)) with a payment rate of $9,750.50.
We did not receive public comments on this provision, and therefore, we are finalizing as proposed. HCPCS Code C9792 will remain assigned to APC 1537 (New Technology—Level 37 ($9,501-$10,000)) with a payment rate of $9,750.50.
Refer to Table 35 for the final OPPS New Technology APC and status indicator assignment for HCPCS code C9792. The final CY 2026 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website.
( printed page 53553)t. Magnetic Resonance Imaging With Inhaled Hyperpolarized Xenon-129 Contrast Agent (APC 1551)
HCPCS code C9791 (Magnetic resonance imaging with inhaled hyperpolarized xenon-129 contrast agent, chest, including preparation and administration of agent) was established on October 1, 2023. For CY 2023, we assigned HCPCS code C9791 to APC 1551 (New Technology—Level 14 ($1201-$1300)). Due to the effective date of the service of October 1, 2023, there were no claims available for HCPCS code C9791 for rate setting in CY 2024. Therefore, in CY 2024, we continued to assign HCPCS code C9791 to APC 1551(New Technology—Level 14 ($1201-$1300)). There were no claims available for HCPCS code C9791 when we were setting rates for CY 2025, so we continued to assign HCPCS code C9791 to APC 1551 (New Technology—Level 14 ($1201-$1300)).
For CY 2026, the proposed OPPS payment rates were based on the available CY 2024 data. There were only four new claims for HCPCS code C9791. Given our proposal to maintain current New Technology APC assignments for CY 2026 for New Technology APC services with fewer than 10 claims in the 4-year lookback period applicable for the universal low-volume APC policy, we proposed for CY 2026 to continue to assign HCPCS code C9791 to APC 1551—New Technology—Level 14 ($1201-$1300)), with a payment rate of $1,250.50.
We did not receive public comments on our proposal to continue to assign HCPCS code C9791 to APC 1551.
Our updated claims data for the 4-year lookback period for the universal low volume APC policy shows only five claims for HCPCS code C9791. Because we are finalizing our proposal to maintain current New Technology APC assignments for CY 2026 for New Technology APC services with fewer than 10 claims in the 4-year lookback period, we are finalizing our proposal without modification to continue to assign HCPCS code C9791 to APC 1551.
Refer to Table 36 for the final OPPS New Technology APC and status indicator assignment for HCPCS code C9791 for CY 2026. The final CY 2026 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website. In addition, we refer readers to Addendum D1 to this final rule with comment period for the status indicator meanings for all codes reported under the OPPS. Addendum D1 can also be found via the internet on the CMS website.
u. SAINT Neuromodulation System (APCs 1511 and 1525)
The SAINT Neuromodulation System is a non-invasive repetitive transcranial magnetic stimulation (rTMS) system that identifies an individualized target and delivers navigationally directed repetitive magnetic pulses to that individualized target located within the left dorsolateral prefrontal cortex to treat major depressive disorder (MDD). The patient first receives structural MRI and functional MRI scans that are analyzed by the provider to identify and localize the personalized stimulation target in the patient's dorsolateral prefrontal cortex. Once the areas targeted for treatment are identified, the patient ( printed page 53554) receives non-invasive magnetic stimulation in the targeted area. The patient has 10 treatment sessions per day with each treatment session lasting 10 minutes followed by 50 minutes of rest before another treatment session occurs. The treatment is administered over five days for a total of 50 sessions of non-invasive magnetic stimulation therapy. There are four CPT codes listed in Table 37 that describe the MRI scans that are used to target the treatment and describe the administration of the non-invasive magnetic stimulation therapy.
For CY 2025, the OPPS payment rates were proposed based on available CY 2023 claims data. However, CPT codes 0889T, 0890T, 0891T, and 0892T did not become effective until July 1, 2024, which means there were no claims data for the procedures described these CPT codes. We assigned our proposed rates for these services based on our evaluation of the resources needed to perform these services.
For CY 2026, the OPPS payment rates were proposed based on available CY 2024 claims data. There were only five claims for CPT code 0889T and three claims for CPT code 0892T within this period. Given our proposal to maintain current New Technology APC assignments for CY 2026 for New Technology APC services with fewer than 10 claims in the 4-year lookback period applicable for the universal low-volume APC policy, we proposed to continue to assign CPT code 0889T to APC 1511 (New Technology—Level 11 ($901-$1000)) with a payment of $950.50 and CPT code 0892T to APC 1525 (New Technology—Level 25 ($3501-$4000)) with a payment of $3750.50.
There were 12 single frequency claims for CPT 0890T and 39 single frequency claims for CPT 0891T. As this is above the threshold of 10 claims and below the threshold of 100 claims for a service within a year, we proposed to apply our universal low volume New Technology APC policy and use the highest of the geometric mean cost, arithmetic mean cost, or median cost based on up to 4 years of claims data to assign CPT codes 0890T and 0891T to the appropriate New Technology APCs.
Using available claims data from CY 2024, our analysis found the geometric mean cost of CPT 0890T was approximately $1,646, the median cost was approximately $1,009, and the arithmetic mean cost was approximately $1,950. The arithmetic mean was the statistical methodology that estimates the highest cost for the service. Therefore, we proposed, for CY 2026, to assign CPT code 0890T to APC 1521 (New Technology—Level 21 ($1901-$2000)) with a payment rate of $1,950.50.
For CPT 0891T, using the available claims data from CY 2024, our analysis found the geometric mean cost was approximately $1,692, the median cost was approximately $1,009, and the arithmetic mean cost was approximately $2,010. The arithmetic mean was the statistical methodology that estimated the highest cost for the service. Therefore, we proposed, for CY 2026, to assign CPT code 0891T to APC 1522 (New Technology—Level 22 ($2001-$2500)) with a payment rate of $2,250.50.
Since the CY 2026 OPPS/ASC proposed rule was published, we note that CPT code 0889T now has 11 single frequency claims; CPT code 0890T has an updated geometric mean cost of $1,687; and CPT code 0891T now has 41 single frequency claims and a geometric mean cost of around $1,690. There were no changes to the claims information for CPT code 0892T.
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: Many commenters requested that CMS maintain the current New Technology APC assignments as the current assignments more appropriately reflect the resources required to provide this highly resource intensive therapy. Commenters noted that if CMS finalized the proposed rates, it would result in a 40-48 percent decrease in payment for two of the SAINT codes. They indicated that not only would the payment reduction affect current access to these services for patients who are suffering from major depressive disorder and treatment-resistant depression, but it would also impede providers from implementing SAINT. As a result, this would further widen the disparities in care, especially in rural and underserved communities where treatment options may be limited.
Commenters stated that the limited claims data (less than 1 year) does not accurately reflect the costs of providing SAINT and that CMS should not use the small dataset that is available for determining the rates for CY 2026. A commenter noted that one of the early providers of SAINT confirmed with ( printed page 53555) them that their reported costs were made in error and were highly inaccurate. The commenter stated that the charges and revenue code assignments for CPT codes 0890T and 0891T dramatically under-reported costs for SAINT, potentially by 80 percent of actual costs for providing this service.
Response: We agree that the proposed rates based on a partial year of claims do not accurately reflect the costs for implementing, providing, and maintaining this service.
After consideration of the public comments we received, we are not finalizing our proposal for CY 2026. For CY 2026, we are using our equitable adjustment authority under section 1833(t)(2)(E) of the Act to maintain the current APC assignments for CPT codes 0889T, 0890T, 0891T, and 0892T. Refer to Table 38 for the proposed and final OPPS New Technology APC and status indicator assignments for CPT codes 0889T, 0890T, 0891T, and 0892T. The final CY 2026 payment rates for these codes can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 to this final rule with comment period for the SI definitions for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
v. Implantable Glucose Monitoring System (APC 1563)
Effective January 1, 2017, the AMA CPT Editorial Panel established CPT codes 0446T (Creation of subcutaneous pocket with insertion of implantable interstitial glucose sensor, including system activation and patient training) and 0448T (Removal of implantable interstitial glucose sensor with creation of subcutaneous pocket at different anatomic site and insertion of new implantable sensor, including system activation) to describe an implantable glucose sensor for patients with diabetes. These codes were used to describe sensors with a 90-day or 180-day battery life. Although these CPT codes were effective January 1, 2017, the implantable interstitial glucose sensor did not receive FDA approval for marketing until June 6, 2019. For CY 2021, we assigned CPT codes 0446T and 0448T to APC 5054 (Level 4 Skin Procedures) and a status indicator of ( printed page 53556) “T” (Procedure or Service, Multiple Procedure Reduction Applies; Paid under OPPS; separate APC payment.) and have maintained these APC assignments since then.
In the CY 2025 OPPS/ASC final rule with comment period, we created the following two HCPCS G codes effective January 1, 2025, to describe the implantable interstitial glucose sensor with a 365-day battery life.
- G0546 (Creation of subcutaneous pocket with insertion of 365 day implantable interstitial glucose sensor, including system activation and patient training); and
- G0565 (Removal of implantable interstitial glucose sensor with creation of subcutaneous pocket at different anatomic site and insertion of new 365 day implantable sensor, including system activation).
We assigned HCPCS codes G0564 and G0565 to APC 1561 (New Technology—Level 24 ($3001-$3500)) with a payment rate of $3,250.50.
For the April 1, 2025, quarterly update, we deleted HCPCS codes G0564 and G0565 and assigned 0446T and 0448T to APC 1561 (New Technology—Level 24 ($3001-$3500)) with a payment rate of $3,250.50 to describe the new implantable interstitial glucose sensor with a 365-day battery life. The 365-day glucose sensor replaced previous versions of the implantable interstitial glucose sensor with shorter battery lives. Therefore, the 365-day sensor is the only sensor on the market and can only be described by CPT codes 0446T and 0448T.
For CY 2026, the proposed OPPS payment rates were based on available CY 2024 claims data. As CPT codes 0446T and 0448T were assigned to New Technology APCs to describe this new sensor for the April 2025 quarterly update and the G codes describing this service were only effective for one quarter, we do not have any claims data for the service. Therefore, for CY 2026, we proposed to continue to assign CPT codes 0446T and 0448T to APC 1561 (New Technology—Level 24 ($3001-$3500)) with a payment rate of $3,250.50.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter requested that CMS reassign CPT codes 0446T and 0448T to APC 1530 (New Technology—Level 30 ($6001-$6500)). The commenter cited the increased cost of the implanted 365-day glucose sensor as the reason for their request and the value of the longer sensor duration, including fewer insertion and removal procedures and better adherence to therapy. They explained that extensive research and development, along with high manufacturing costs, have contributed to the cost of the implantable continuous glucose monitoring system (iCGM system) which they state is $6,800. The commenter also requested that the OPPS payment align with the PFS payment (approximately $5,800) to provide consistent payment regardless of setting.
Response: We appreciate the public comment and understand the implicated value that a longer life sensor brings to Medicare beneficiaries. We agree that there would be inherently increased costs to hospitals for the new technology of a 365-day system, but we do not agree that the costs to hospitals would be almost double the costs of the 180-day system.
In summary, after consideration of the public comment we received, we are finalizing our proposal with modification based on our statutory authority set out at section 1833(t)(2)(E) of the Act, to assign CPT code 0446T and 0448T to APC 1563 (New Technology—Level 26 ($4001-$4500)) with a payment of $4,250.50. We remind hospitals that we review, on an annual basis, the APC assignments for all items and services paid under the OPPS. Refer to Table 39 for code descriptor, APC assignment and status indicator assignments for CPT codes 0446T and 0448T for CY 2026. The final CY 2026 payment rates for these codes can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 to this final rule with comment period for the SI definitions for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
w. Skin Cell Suspension Autograft (SCSA) Procedures (CPT Code 15013 and HCPCS Code C8002) (APC 1567)
Effective January 1, 2025, both CPT code 15013 (Preparation of skin cell suspension autograft, requiring enzymatic processing, manual mechanical disaggregation of skin cells, and filtration; first 25 sq cm or less of harvested skin) and HCPCS code C8002 (Preparation of skin cell suspension autograft, automated, including all enzymatic processing and device components (do not report with manual suspension preparation)) describe the preparation step of a skin cell suspension autograft (SCSA) procedure to treat acute thermal burn injuries. Both codes describe the preparation step of a three-step SCSA procedure: harvesting, preparation, and ( printed page 53557) application. The difference between the codes is that CPT code 15013 describes the manual preparation of the SCSA, and HCPCS code C8002 describes the automated preparation of the SCSA. Due to the similarities between the procedures, in the CY 2025 OPPS/ASC final rule with comment period, we assigned both CPT code 15013 and HCPCS code C8002 to APC 1567 (New Technology—Level 30 ($6,001-$6,500)) with a payment rate of $6,250.50 and status indicator “T”. In the CY 2025 OPPS/ASC final rule with comment period, we noted that we believed the sum of the payment rates for the three-step process should approximate $10,000. However, because of the effect of the multiple procedure reduction, the total payment for the skin cell suspension autograft furnished using the RECELL System would have been approximately $8,000, contrary to the intended target of $10,000 as stated in the CY 2025 OPPS/ASC final rule with comment period. To correct this error, in the CY 2025 OPS/ASC Correction Notice, we assigned both CPT code 15013 and HCPCS code C8002 to APC 1532 (New Technology—Level 32 ($7,001-$7,500)) with a payment rate of $7,250.50 and status indicator “S” (Procedure or service, not discounted when multiple, paid under OPPS; separate APC payment).
For CY 2026, the OPPS payment rates are proposed to be based on available CY 2024 claims data. Since CPT code 15013 and HCPCS code C8002 were not effective until January 1, 2025, we did not have any claims for either code for CY 2024. Therefore, for CY 2026, we proposed to continue to assign CPT code 15013 and HCPCS code C8002 to APC 1532 (New Technology—Level 32 ($7,001-$7,500)) with a payment rate of $7,250.50.
We did not receive public comments on this provision, and therefore, we are finalizing as proposed.
Refer to Table 40 for the proposed and final OPPS New Technology APC and status indicator assignments for CPT code 15013 and HCPCS code C8002 for CY 2026. The final CY 2026 payment rates can be found in Addendum B to this final rule via the internet on the CMS website.
x. Renal Histotripsy Service (APC 1576)
HCPCS code C9790 (Histotripsy (that is, non-thermal ablation via acoustic energy delivery) of malignant renal tissue, including image guidance) was created October 1, 2023, and was used to describe the Medicare approved Category B IDE (investigational device exemption) clinical study involving the renal histotripsy procedure associated with the use of the HistoSonics Edison System. CPT code 0888T (Histotripsy ( i.e., non-thermal ablation via acoustic energy delivery) of malignant renal tissue, including image guidance) replaced HCPCS code C9790 effective July 1, 2024.
Renal histotripsy is a non-invasive, non-thermal, mechanical process that uses a focused beam of sonic energy to destroy solid renal tumors and is currently in a prospective, multi-center, single-arm pivotal trial designed to evaluate the effectiveness and safety of the device for the destruction of kidney tissue by treating primary solid renal tumors.[24] Because the renal histotripsy clinical study is designated as a Category B (non-experimental/investigational) IDE study, the Medicare payment for CPT code 0888T reflects payment for both the service that is performed, and the device used each time it is reported on a claim. For CY 2025 we assigned CPT code 0888T to APC 1576 (New Technology—Level 39 ($15,001-$20,000)) with a payment rate of $17,500.50 based on the previous APC and status indicator assignments for HCPCS code C9790.
For CY 2026, the proposed OPPS payment rates were based on available CY 2024 claims data. We identified one single frequency claim for HCPCS code C9790 and six single frequency claims for CPT code 0888T. Since the CY 2026 OPPS/ASC proposed rule has been published, we have eight single frequency claims for CPT code 0888T. Given our proposal to maintain current New Technology APC assignments for CY 2026 for New Technology services with fewer than 10 claims in the 4-year lookback period applicable for the universal low-volume APC policy, we proposed to continue to assign CPT code 0888T to APC 1576 (New Technology—Level 39 ($15,001-$20,000)) with a payment rate of $17,500.50.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter supported the proposed New Technology APC assignment.
Response: We thank the commenter for their support. ( printed page 53558)
In summary, we are finalizing our proposal without modification. We will continue to assign CPT code 0888T to APC 1576 with a status indicator of `S' for CY 2026.
The New Technology APC and status indicator assignment for CPT code 0888T is shown in Table 41. The final CY 2026 payment rates for this CPT code can be found in Addendum B to this final rule via the internet on the CMS website. In addition, we refer readers to Addendum D1 to this final rule with comment period for the status indicator meanings reported under OPPS. Addendum D1 can also be found via the internet on the CMS website.
D. Universal Low Volume APC Policy for Clinical and Brachytherapy APCs
In the CY 2022 OPPS/ASC final rule with comment period (86 FR 63743 through 63747), we adopted a policy to designate clinical and brachytherapy APCs as low volume APCs if they have fewer than 100 single claims that can be used for ratesetting purposes in the claims year used for ratesetting for the prospective year. For the CY 2026 OPPS/ASC proposed rule, CY 2024 claims were generally the claims used for ratesetting; and clinical and brachytherapy APCs with fewer than 100 single claims from CY 2024 that can be used for ratesetting would be low volume APCs subject to our universal low volume APC policy. As we stated in the CY 2022 OPPS/ASC final rule with comment period, we adopted this policy to reduce the volatility in the payment rate for those APCs with fewer than 100 single claims. Where a clinical or brachytherapy APC has fewer than 100 single claims that can be used for ratesetting, under our low volume APC payment adjustment policy, we determine the APC cost as the greatest of the geometric mean cost, arithmetic mean cost, or median cost based on up to 4 years of claims data. We excluded APC 5853 (Partial Hospitalization for CMHCs) and APC 5863 (Partial Hospitalization for Hospital-based PHPs) from our universal low volume APC policy given the different nature of policies that affect the partial hospitalization program. We also excluded APC 2698 (Brachytx, stranded, nos) and APC 2699 (Brachytx, non-stranded, nos) as our current methodology for determining payment rates for non-specified brachytherapy sources is appropriate.
Based on claims data available for the CY 2026 OPPS/ASC proposed rule, we proposed to designate six brachytherapy APCs and five clinical APCs as low volume APCs under the OPPS (90 FR 33561 through 33562). The six brachytherapy APCs and five clinical APCs meet our criteria of having fewer than 100 single claims in the claims' year used for ratesetting (CY 2024 for the CY 2026 OPPS/ASC proposed rule). Ten of the 11 APCs were designated as low volume APCs in CY 2025. Based on data for the CY 2026 OPPS/ASC proposed rule, APC 2645 (Brachytx, non-stranded, gold-198) had 103 single claims and no longer met our criteria to be designated as a low volume APC; however, APC 2643 (Brachytx, non-stranded, c-131) had only 88 single claims and met our criteria to be designated as a low volume APC.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: Commenters supported our proposal to continue our low volume APC policy.
Response: We thank commenters for their support.
After consideration of public comments, based on CY 2024 claims data available for this final rule with comment period, we are finalizing our proposal to designate six brachytherapy APCs and five clinical APCs as low volume APCs under the OPPS. Table 42 includes the CY 2024 claims available for ratesetting for each of the APCs we are designating as low volume APCs for CY 2026. The final cost statistics for our CY 2026 low volume APCs, such as the median, arithmetic mean, and geometric mean cost are available for download with this final rule with comment period on the CMS website. We refer readers to our website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices; click on the relevant regulation to download the low volume APC cost statistics under the comprehensive (OPPS) ratesetting methodology in the downloads section of the web page.
( printed page 53559)E. APC-Specific Policies
1. APC Structure
a. Diagnostic Tests and Related Services (APCs 5721 Through 5724)
The Diagnostic Tests and Related Services APC series was created as part of the APC restructuring and consolidation in the CY 2016 OPPS (80 FR 70384 through 70386). Since its initial establishment, we have maintained a four-level APC structure for the series. In the CY 2026 OPPS, as part of our standard process of reviewing the OPPS structure based on updated claims data, we proposed to make changes to the APC series, and included those changes in the associated cost statistics files and addenda made available with each proposed and final rule via the internet on the CMS website.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: Several commenters requested that CMS does not finalize the proposed changes to the Diagnostic Tests and Related Services APC series. They requested that CMS refrain from shifting services in the APC family until there was sufficient opportunity for meaningful public comment on such changes. Commenters noted the impact on geometric mean costs for each of the APCs. Other commenters requested that CMS delay the changes and conduct further analysis so that the public can evaluate them. A commenter requested that CMS rerun the cost modeling for the APCs excluding low volume or anomalous facility reports and consider volatility guards, public analysis regarding observed changes, and maintain payment levels that preserve beneficiary access until stable cost data can be established.
Commenters noted individual impacts on codes related to the changes in the Diagnostic Tests and Related Services APCs. A commenter noted the impact on CPT code 95924 (Testing of autonomic nervous system function; combined parasympathetic and sympathetic adrenergic function testing with at least 5 minutes of passive tilt), which had an expected decrease in payment of 29 percent. Another commenter noted CPT code 93017 (Cardiovascular stress test using maximal or submaximal treadmill or bicycle exercise, continuous electrocardiographic monitoring, and/or pharmacological stress; tracing only, without interpretation and report) which had an expected decrease of about 29 percent in its payment rate, which the commenter wanted maintained at $311.40. A few commenters recommended that CPT code 90870 (Electroconvulsive therapy (includes necessary monitoring)) be maintained in its current assignment or moved to a clinically coherent APC since the code represents a therapeutic procedure and not a diagnostic test.
Response: As discussed earlier in this rule, section 1833(t) of the Act requires CMS to annually review and update the payment rates for services payable under the OPPS. Section 1833(t)(2)(B) of the Act provides that the Secretary may establish groups of covered OPD services within this classification system, so that services classified within each group are comparable clinically and with respect to the use of resources. In addition, section 1833(t)(2) of the Act provides that, subject to certain exceptions, the items and services within an APC group cannot be considered comparable regarding the use of resources if the highest cost for an item or service in the group is more than 2 times greater than the lowest cost for an item or service within the same group (referred to as the “2 times rule”).
As part of that review and update process we made changes to the APC assignments within the APC series such that the cost and clinical APC groupings would be more reflective of the codes assigned to them. We note that the changes in geometric mean costs for these APCs are associated with the APC recalibrations so that the services with clinical cost patterns that are more similar to each other are assigned to the same APC. In general, we do not believe that the alternative cost modeling the commenter requested is appropriate for ratesetting for these APCs. In the broader OPPS ratesetting process we generally aim to use as much as data is available that is appropriate for ratesetting, and typically only remove claims data from that process through the systematic trims that are described in the claims accounting narrative document made available on the CMS website.
We believe that sufficient opportunity for public notice and comment was provided, as the proposed changes to the APC assignments and the APC levels are reflected through the cost statistics and two times files we make available with each proposed and final rule. In addition, any changes associated with individual codes are noted in the ( printed page 53560) Addendum B through the Change Indicator column.
CPT codes 95924 and 93017 were both proposed to be placed in APC 5722 (Level 2 Diagnostic Tests and Related Services) in the CY 2026 OPPS/ASC proposed rule, which has a cost significant range from approximately $165 to $298. (The addenda for CY 2026 OPPS/ASC proposed rule can be found on the CMS website located at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices/cms-1834-p.) In the final rule claims data available for OPPS ratesetting CPT code 95924 has a geometric mean cost of $281.10, while CPT code 93017 has a geometric mean cost of $271.62. While we recognize that APC geometric mean costs can fluctuate based on a variety of factors, including updated claims and cost report data update, APC level recalibration, and others, the geometric mean costs of both codes suggests that they are appropriately placed in the Level 2 APC based on their estimated resource costs. We note that we generally do not set APC payment rates at predetermined rates for clinical APCs.
In the claims available for OPPS ratesetting for this final rule, CPT code 90870 has an estimated geometric mean cost of $713.14 based on 52,518 single claims. The geometric mean cost of APC 5724 (Level 4 Diagnostic Tests and Related Services) is $884.07. We believe that the proposed placement of CPT code 90870 in APC 5724 remains appropriate based on its geometric mean cost relative to that of the APC and the similarity of its resource costs relative to other procedures assigned to the APC and clinical similarity to some of the related services assigned to the APC series more broadly. As a result, we are finalizing the assignment of CPT code 90870 to APC 5724.
We note that we will continue to monitor the APC series as more claims data are available for the procedures assigned to this series and continue to be interested in suggestions regarding changes to those assignments for future years.
After consideration of the public comments we received, we are finalizing the proposed APC recalibration changes for the Diagnostics Tests and Related Procedures APC series. In addition, we are finalizing the assignments of CPT codes 95924 and 93017 to APC 5722, and CPT code 90870 to APC 5724.
b. Nerve Procedures (APCs 5431 Through 5432)
The current APC structure of the Nerve Procedures series was developed during the broader CY 2016 OPPS reorganization and consolidation of APCs. Since that time, it has maintained that same two-level APC structure (89 FR 70379 through 70380).
Comment: A few commenters noted that there was a significant decrease in the estimated geometric mean cost of the Level 2 Nerve Procedures APC due to the impact of a new eligible complexity adjustment code combination. They requested that CMS either map the complexity adjustment 6471R back into the Level 1 Nerve Procedures APC (APC 5431) or alternatively develop a level 3 APC that could accommodate some of the higher cost procedures in the current Level 2, such that there was less of an impact on some portion of the procedures.
Commenters had requested that these codes be included in that level 3 APC:
61215—Insertion of subcutaneous reservoir, pump or continuous infusion system for connection to ventricular catheter
63741—Creation of shunt, lumbar, subarachnoid-peritoneal, -pleural, or other; percutaneous, not requiring laminectomy
64864—Suture of facial nerve; extracranial
64885—Nerve graft (includes obtaining graft), head or neck; up to 4 cm in length
64886—Nerve graft (includes obtaining graft), head or neck; more than 4 cm length
64890—Nerve graft (includes obtaining graft), single strand, hand or foot; up to 4 cm length
64891—Nerve graft (includes obtaining graft), single strand, hand or foot; more than 4 cm length
64892—Nerve graft (includes obtaining graft), single strand, arm or leg; up to 4 cm length
64895—Nerve graft (includes obtaining graft), multiple strands (cable), hand or foot; up to 4 cm length
64896—Nerve graft (includes obtaining graft), multiple strands (cable), hand or foot; more than 4 cm length
64897—Nerve graft (includes obtaining graft), multiple strands (cable), arm or leg; up to 4 cm length
64898—Nerve graft (includes obtaining graft), multiple strands (cable), arm or leg; more than 4 cm length
64907—Nerve pedicle transfer; second stage
64912—Nerve repair; with nerve allograft, each nerve, first strand (cable)
A commenter also requested special consideration for CPT code 64912, and that the CPT code be included in the Level 3 Nerve Procedures APC.
Response: While we note that APC geometric mean cost changes are expected under the current structure of the C-APCs and the complexity adjustments, we agree that a Level 3 Nerve Procedures APC is appropriate, in particular to resolve what would otherwise be a significant “two times rule” violation in the Level 2 APC. We are including the above requested codes in the Level 3 APC. We believe that there are several additional codes for which it would appropriate from a clinical and resource cost similarity perspective to include in the Level 3 APC. We are finalizing the inclusion of the following additional codes into the Level 3 APC:
61720—Creation of lesion by stereotactic method, including burr hole(s) and localizing and recording techniques, single or multiple stages; globus pallidus or thalamus
62230—Replacement or revision of cerebrospinal fluid shunt, obstructed valve, or distal catheter in shunt system
62350—Implantation, revision or repositioning of tunneled intrathecal or epidural catheter, for long-term medication administration via an external pump or implantable reservoir/infusion pump; without laminectomy
64840—Suture of posterior tibial nerve
64856—Suture of major peripheral nerve, arm or leg, except sciatic; including transposition
64905—Nerve pedicle transfer; first stage
64910—Nerve repair; with synthetic conduit or vein allograft (e.g., nerve tube), each nerve
64911—Nerve repair; with autogenous vein graft (includes harvest of vein graft), each nerve
0442T—Ablation, percutaneous, cryoablation, includes imaging guidance; nerve plexus or other truncal nerve (e.g., brachial plexus, pudendal nerve)
Under the current C-APC methodology, complexity adjustments are mapped to the next higher cost APC within the clinical family. Within this structure, we currently do not have a mechanism for removing eligibility complexity adjustments by assigning them to alternative APCs including the originating primary APC. Therefore, we do not currently believe it is appropriate to manually assign the complexity adjustment represented by 6471 to APC 5431. Despite this, we understand that there are potential opportunities to ( printed page 53561) refine the C-APC and complexity adjustment methodology to address cost modeling concerns. We continue to remain interested in suggestions regarding the C-APC and complexity adjustment methodology, as noted in section II.A.2.b. of this final rule with comment period.
After consideration of the comments, we are finalizing a 3-Level APC structure for the Nerve Procedures APC series.
c. Endovascular Procedures (APCs 5191 Through 5194)
The Endovascular Procedures APC series was initially mapped and assigned as a 3 level series in the CY 2016 OPPS. In the CY 2017 OPPS, an additional APC level was created. Since that time the Endovascular Procedures APC series has been maintained as a 4-level APC series.
Comment: A commenter requested that APC 5200 (Implantation Wireless PA Pressure Monitor) be converted into a Level 5 Endovascular Procedures APC, and that the following codes be included into that APC:
- C9774—Revascularization, endovascular, open or percutaneous, tibial/peroneal artery(ies); with intravascular lithotripsy and atherectomy, includes angioplasty within the same vessel(s), when performed.
- C9775—Revascularization, endovascular, open or percutaneous, tibial/peroneal artery(ies); with intravascular lithotripsy and transluminal stent placement(s), and atherectomy, includes angioplasty within the same vessel(s), when performed.
- C9767—Revascularization, endovascular, open or percutaneous, lower extremity artery(ies), except tibial/peroneal; with intravascular lithotripsy and transluminal stent. placement(s), and atherectomy, includes angioplasty within the same vessel(s), when performed.
- C9797—Vascular embolization or occlusion procedure with use of a pressure-generating catheter (e.g., one-way valve, intermittently occluding), inclusive of all radiological supervision and interpretation, intraprocedural roadmapping, and imaging guidance necessary to complete the intervention; for tumors, organ ischemia, or infarction.
They believe that doing so would be appropriate based on the similarity of the codes' geometric mean costs. That commenter also stated that with the proposal to eliminate the inpatient only list over three years beginning in 2026, creating a Level 5 Endovascular Procedures APC could allow for appropriate clinical APC placement for procedures being removed from the list.
Response: We note that the APC geometric mean cost of APC 5194 (Level 4 Endovascular Procedures) is $18,872.40 while that of APC 5200 (Implantation Wireless PA Pressure Monitor) is $29,529.47. We recognize that the general approximate geometric mean costs for inclusion in the requested Level 5 APC would range from about $24,500 to $27,900. However, that level 5 APC payment weight would still continue to be primarily driven by the estimated costs of the currently assigned HCPCS code 33289 ( Transcatheter implantation of wireless pulmonary artery pressure sensor for long-term hemodynamic monitoring, including deployment and calibration of the sensor, right heart catheterization, selective pulmonary catheterization, radiological supervision and interpretation, and pulmonary artery angiography, when performed), which has a geometric mean cost of $29,529.47.
We recognize the commenter concerns around procedure codes potentially being removed from the IPO list requiring appropriate clinical APC placements. For the CY 2026 OPPS, we do not believe that the procedures being removed from the IPO list would require the level 5 Endovascular Procedures APC that the commenter requested.
At this time, we do not believe that it is appropriate to convert the APC 5200 (Implantation Wireless PA Pressure Monitor) into a level 5 Endovascular Procedures APC. However, we will continue to monitor the available claims and cost data for the APC series and in the context of codes being removed for the IPO list.
After consideration of the comments, we are finalizing the 4-level APC structure of the Endovascular Procedures APC series as proposed.
d. Laparoscopy and Related Services (APCs 5361 Through 5362)
As part of the CY 2016 OPPS APC restructuring process, the four level APC series for Laparoscopy and Related Services was consolidated into a 2-level APC series (80 FR 70379) through. Since that time, we have maintained that 2-level APC structure based on the clinical and resource homogeneity of the services assigned to those APCs.
Comment: A commenter requested that CMS create a Level 3 Laparoscopy and Related Services APCs that would include the highest cost and complexity services in the current Level 2 APC. The commenter believes that this would restore the previously existing structure of the Laparoscopy and Related Services APCs while allowing potential placements for services being removed from the IPO list. The commenter believes that it would be appropriate to place services with geometric mean costs of $12,000 or more into that level 3 APC.
Response: While we monitor the structure of the OPPS and the various APCs, we note that some of the principles guiding the CY 2016 OPPS APC restructuring and consolidation included improved resource and clinical homogeneity, as well as reduced resource overlap in APCs within a clinical family (80 FR 70379).
The Level 2 Laparoscopy and Related Services APC has an estimated geometric mean cost of $10,943.41, with a range of geometric mean costs for two times rule purposes ranging from $8,909.96 for CPT code 58552 (Laparoscopy, surgical, with vaginal hysterectomy, for uterus 250 g or less; with removal of tube(s) and/or ovary(s)) to $13,085.27 for CPT code 55866 (Laparoscopy, surgical, with vaginal hysterectomy, for uterus 250 g or less; with removal of tube(s) and/or ovary(s)). While some of the services in the cost range indicated by the commenter have significant claims volume, we do not believe that there is a current need for an additional APC level given that the estimated geometric mean cost of the requested Level 3 APC would be significantly distinct from a cost perspective.
We note that we are not removing any services from the IPO list in CY 2026 that would require assignments to the Laparoscopic and Related Services APC series. However, we will continue to monitor the claims data as they become available and the need for additional APC levels in the future.
After consideration of the public comments, we are finalizing the 2-level APC structure for the Laparoscopy and Related Services APC series in this final rule with comment period as proposed.
2. ActiGraft System, HCPCS Code G0465 (APC 5054)
Effective April 2021, HCPCS code G0465 (Autologous platelet rich plasma (PRP) or other blood-derived product for diabetic chronic wounds/ulcers, using an FDA-cleared device for this indication, (includes as applicable administration, dressings, phlebotomy, centrifugation or mixing, and all other preparatory procedures, per treatment) describes autologous blood derived products for chronic non-healing wounds. For CY 2026, we proposed to assign HCPCS code G0465 to APC 5054 ( printed page 53562) (Level 4 Skin Procedures) and status indicator “T.”
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter requested that CMS change the status indicator from “T,” which allows for multiple procedure discounting, to a status indicator that pays the full payment rate each time the service is billed on a claim. The commenter explained that CMS recognized that HCPCS code G0465 could be billed twice on the same claim by revising the Medically Unlikely Edit (MUE) for HCPCS code G0465 from “1” to “2” in April 2025. Per the commenter, when more than one treatment is required for a patient with a single large wound or multiple wounds, providers would need to bill HCPCS code G0465 twice on the same claim to reflect the multiple treatments. The commenter believes that applying a multiple procedure discount to HCPCS code G0465 is inappropriate because the cost of the treatment kit would not be paid fully if billed multiple times. The commenter notes that CPT code 43877, which is also proposed to be assigned to APC 5054, is assigned a status indicator other than “T,” specifically “Q2,” to demonstrate that CMS has assigned codes in APC 5054 a status indicator other than “T.” Finally, the commenter requested that CMS revise the place of service (POS) codes for G0465 to allow for the service to be furnished in the nursing home setting.
Response: With regard to the commenter's request to revise the POS codes for HCPCS code G0465, changes to allow for services to be paid in facilities other than the hospital outpatient department or ambulatory surgical center are outside the scope of this rule.
With regard to the request to not finalize the assignment of the proposed status indicator of “T” to HCPCS code G0465, we note that the OPPS uses certain payment principles, including packaging and multiple procedure discounting, in an effort to control costs and promote more efficient care. HCPCS code G0465 describes a service that may be billed more than once on a claim in particular instances when the patient has multiple or large wounds that would require additional product. In reviewing the other services that are assigned to APC 5054, we note there are many other similar skin services (for example, HCPCS code G0460) that are also assigned to status indicator “T” and that are described on a “per treatment” basis, like HCPCS code G0465. To align with the 80 other similar skin procedure codes assigned to APC 5054 and status indicator “T,” we are finalizing our proposal to assign status indicator “T” to HCPCS code G0465 as reflected in Table 43.
The final CY 2026 OPPS payment rate for all the codes payable under the OPPS can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 to this final rule with comment period for the SI meanings for all codes reported under the OPPS. Addendum D1 is available via the internet on the CMS website.
Audiology Related Procedures and Services
3. Audiology-Related Services, CPT Codes 92540, 92579, 92588 (APC 5722)
For CY 2025, we assigned CPT codes 92540 (Basic vestibular evaluation, includes spontaneous nystagmus test with eccentric gaze fixation nystagmus, with recording, positional nystagmus test, minimum of 4 positions, with recording, optokinetic nystagmus test, bidirectional foveal and peripheral stimulation, with recording, and oscillating tracking test, with recording) and 92579 (Visual reinforcement audiometry (vra)) to APC 5721 (Level 1 Diagnostic Tests and Related Services) with a payment rate of $156.46. CPT code 92588 (Distortion product evoked otoacoustic emissions; comprehensive diagnostic evaluation (quantitative analysis of outer hair cell function by cochlear mapping, minimum of 12 frequencies), with interpretation and report) was assigned to APC 5722 (Level 2 Diagnostic Tests and Related Services) with a payment rate of $311.40.
In the CY 2026 OPPS/ASC proposed rule, we proposed to assign all three CPT codes to APC 5722 (Level 2 Diagnostic Tests and Related Services) with a proposed payment rate of $221.14.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter stated that they were concerned with the placement of CPT code 92540 in APC 5721 because the time and resources required to perform CPT 92540 are clinically analogous to the electrophysiological tests in APC 5722. Therefore, the commenter requested a reassignment of CPT 92540 from APC 5721 to APC 5722. The same commenter supported the placement of CPT codes 92579 and 92588 to APC 5722 and urged CMS to finalize the assignment.
Response: As we have already proposed to assign CPT code 92540 to APC 5722, we are finalizing our proposal without modification to assign CPT codes 92540, 92579 and 92588 to APC 5722 for CY 2026.
We refer readers to Addendum B to this final rule with comment period for the payment rates for all codes ( printed page 53563) reportable under the OPPS. Addendum B is available via the internet on the CMS website.
4. Fully Implanted Active Middle Ear Implant (FI-AMEI), CPT Codes 0951T-0955T
Effective July 1, 2025, The AMA CPT Editorial Board created five new Category III CPT codes to report total implantation, revision or replacement with or without mastoidectomy; replacement of sound processor only; and removal, including all implant components of a FI-AMEI. Specifically, the following CPT codes were created:
- 0951T—Totally implantable active middle ear hearing implant; initial placement, including mastoidectomy, placement of and attachment to sound processor;
- 0952T—Totally implantable active middle ear hearing implant; revision or replacement, with mastoidectomy and replacement of sound processor;
- 0953T—Totally implantable active middle ear hearing implant; revision or replacement, without mastoidectomy and replacement of sound processor;
- 0954T—Totally implantable active middle ear hearing implant; replacement of sound processor only, with attachment to existing transducers; and
- 0955T—Totally implantable active middle ear hearing implant; removal, including removal of sound processor and all implant components.
In the CY 2026 OPPS/ASC proposed rule, we proposed to assign CPT codes 0951T-0955T to status indicator “E1” to indicate that these codes are not paid by Medicare when submitted on outpatient claims (any outpatient bill type) because we believe that these codes meet the definition of a hearing aid and therefore are not covered by Medicare.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter stated that FI-AMEI is not a hearing aid but instead is a prosthetic device that meets the definition of an osseointegrated implant, and therefore, should be covered by Medicare.
The commenter stated that an FI-AMEI is a prosthetic device because it produces the perception of sound by replacing the function of the middle ear. More specifically, the commenter stated that an FI-AMEI is an osseointegrated implant.
The commenter requested that CMS change the proposed OPPS status indicator assignment for CPT codes 0951T-0955T from status indicator “E1” to status indicator “S” given the FI-AMEI is a covered prosthetic device as defined by the Medicare Benefit Policy Manual.
The commenter requested CMS assign codes 0951T-0953T to New Technology APC 1577, code 0954T to New Technology APC 1575, and code 0955T to New Technology APC 1534.
Response: We thank the commenter for their input. Section 1862(a)(7) of the Act excludes hearing aids from Medicare coverage. Certain devices that produce perception of sound by replacing the function of the middle ear, cochlea or auditory nerve are excepted and payable by Medicare. Cochlear implants and auditory brainstem implants that replace the function of the cochlea or auditory nerve and provide electrical stimulation to auditory nerve fibers are excepted. Osseointegrated implants that replace the function of the middle ear and provide vibratory mechanical energy through the skull to both cochleae are excepted. Middle ear implants, including fully implanted active middle ear hearing devices, do not function like osseointegrated implants and are not excepted.
Therefore, we are finalizing the assignment of status indicator “E1” for CPT codes 0951T-0955T without modification to indicate that these codes are not paid by Medicare when submitted on outpatient claims (any outpatient bill type).
In addition, we refer readers to Addendum D1 to this final rule with comment period for the SI meanings for all codes reported under the OPPS. Addendum D1 is available via the internet on the CMS website.
Breast and Lymph Procedures
5. Ablation of Breast Tumor Procedures, CPT Codes 0970T and 0971T (APC 5091)
Effective July 1, 2025, the AMA CPT Editorial Panel established the CPT codes 0970T and 0971T. CPT codes 0970T (Ablation, benign breast tumor ( e.g., fibroadenoma), percutaneous, laser, including imaging guidance when performed, each tumor) and 0971T (Ablation, malignant breast tumor(s), percutaneous, laser, including imaging guidance when performed, unilateral) describe procedures for the ablation of breast tumors. For CY 2026, we proposed to maintain both CPT codes in APC 5091 (Level 1 Breast/Lymphatic Surgery and Related Procedures).
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter requested that CMS reassign CPT codes 0970T and 0971T to APC 5093 (Level 3 Breast/Lymphatic Surgery and Related Procedures), which had a proposed payment rate of $6,575.63. The commenter stated that the device associated with these procedures had a cost of around $2,700 and the total costs of the procedure would be more appropriately covered by the payment rate of APC 5093.
Response: After clinical review of these procedures, we continue to believe that these procedures are appropriately assigned to APC 5091. Therefore, we are finalizing our proposal, without modification, to continue to assign CPT codes 0970T and 0971T to APC 5091 (Level 1 Breast/Lymphatic Surgery and Related Procedures). Table 44 shows the finalized status indicator and APC assignment for the procedure codes. We refer readers to Addendum B to this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
( printed page 53564)6. Lymphovenous Bypass, CPT Code 1019T (APC 5092)
The CPT Editorial Panel created Category III CPT code 1019T (placeholder code X476T) (Lymphovenous bypass, including robotic assistance, when performed, per extremity) effective January 1, 2026. For CY 2026, we proposed to assign CPT code 1019T to APC 5091 (Level 1 Breast/Lymphatic Surgery and Related Procedures) with a proposed payment of around $4,049 and a status indicator of `J1' (Hospital Part B Services Paid Through a Comprehensive APC; Paid under OPPS).
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: Several commenters requested higher payment for this procedure providing the following rationale to support their request:
- The procedure is a curative procedure versus managing the condition of lymphedema.
- The procedure requires 2 to 5 hours of operative time.
- The procedure is performed by surgeons who have advanced surgical training in this specialty area.
- The procedure is resource intensive utilizing a highly specialized and expensive equipment (for example, microscope and robot).
A couple of commenters suggested alternative crosswalk codes to determine APC placement such as, CPT code 19357 ( Tissue expander placement in breast reconstruction, including subsequent expansion(s)) and CPT code 35883 ( Revision, femoral anastomosis of synthetic arterial bypass graft in groin, open; with nonautogenous patch graft (e.g., polyester, ePTFE, bovine pericardium). The commenter suggested using the claims information for CPT code 35883 and the cost information associated with that procedure as a basis for determining an appropriate New Technology APC assignment.
Response: After review of the comments, we believe that CPT code 1019T should be reassigned to APC 5092, noting that there are currently other lymph procedures assigned to APC 5092. We do not believe a New Technology APC assignment would be appropriate as the procedure is described by a Category III CPT code and can be assigned to a clinical APC.
In summary, after consideration of the public comments we received, we are finalizing our proposal with modification, to assign CPT code 1019T to APC 5092 as reflected in Table 45. The final CY 2026 payment rates for this code can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 to this final rule with comment period for the SI definitions for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
Cardiac Related Procedures and Services
7. Cardiac CT Services, CPT Codes 75572, 75573, and 75574 (APC 5572)
Cardiac computed tomography services are described by the following Category I CPT codes and have been effective since January 1, 2010:
- 75572—Computed tomography, heart, with contrast material, for evaluation of cardiac structure and morphology (including 3D image postprocessing, assessment of cardiac function, and evaluation of venous structures, if performed);
- 75573—Computed tomography, heart, with contrast material, for evaluation of cardiac structure and morphology in the setting of congenital heart disease (including 3D image postprocessing, assessment of left ventricular [LV] cardiac function, right ventricular [RV] structure and function and evaluation of vascular structures, if performed); and
- 75574— Computed tomographic angiography, heart, coronary arteries and bypass grafts (when present), with contrast material, including 3D image postprocessing (including evaluation of ( printed page 53565) cardiac structure and morphology, assessment of cardiac function, and evaluation of venous structures, if performed).
In prior rulemaking, we have received comments noting that the payment for these codes has declined since 2017. Comments on previous OPPS proposed rules have indicated that the payment amount is insufficient to cover the cost of providing the service and have stated that the payment amount does not consider the hospital resources required to perform these services, including the use of the equipment, medication administration, staff time, and scanner time. We have maintained over the years that an analysis of our claims data for these three codes have shown geometric mean costs consistent with the geometric mean cost for the APC to which they were assigned to since 2015.
We have also received comments in the past urging CMS to allow hospitals the flexibility to submit charges for cardiac CT services with a revenue code other than CT scan (035X) and Radiology Diagnostic (032X) revenue codes, implying that MACs had applied edits to the cardiac CT codes that prevented hospitals from reporting a cardiology (048X) revenue code when appropriate. It is longstanding CMS policy that hospital outpatient facilities are responsible for reporting the appropriate cost centers and revenue codes on claims. As stated in section 20.5 in Chapter 4 (Part B Hospital) of the Medicare Claims Processing Manual, CMS “does not instruct hospitals on the assignment of HCPCS codes to revenue codes for services provided under OPPS since hospitals' assignments of cost vary. Where explicit instructions are not provided, HOPDs should report their charges under the revenue code that will result in the charges being assigned to the same cost center to which the cost of those services are assigned in the cost report.” We have consistently stated that hospital outpatient facilities must determine the most appropriate cost center and revenue code for the cardiac CT codes (87 FR 71849, 88 FR 81664 and 89 FR 94058).
After we issued the CY 2024 OPPS/ASC final rule, interested parties notified us of a specific claims edit that may have limited the revenue codes reported with the cardiac CT codes in prior years' claims data. We removed the outdated revenue code edit in early December 2023 to allow for the cardiac CT codes to be billed with any appropriate revenue code. We informed the public of our findings and the changes that we made in the January 2024 OPPS Update (Transmittal 12421, Change Request 13488), dated December 21, 2023. We believe the edit may have prevented some providers from reporting the cardiology revenue code (048X), which maps to the cardiology cost center (03140), when billing for cardiac CT services. In the past, commenters have indicated that the cardiology cost center has a higher cost-to-charge ratio (CCR) than the imaging cost centers, and they believe the inability to report the cardiology revenue code has resulted in a lower payment rate for cardiac CT services. Since the OPPS ratesetting process utilizes the applicable cost center's CCR to reduce the charges on the claim to estimated cost, utilizing cost centers with lower CCRs results in a lower OPPS payment compared to utilizing cost centers with higher CCRs. With the edit no longer in place, we stated that hospitals may bill for cardiac CT services with whichever revenue code they believe appropriate, including cardiology revenue code 048X.
In CY 2025 rulemaking, we conducted studies to calculate HCPCS geometric mean costs for the cardiac CT codes based on a simulation that assumed that differing numbers of HOPDs (specifically 25 percent, 50 percent, and 75 percent of the total number of HOPDs billing for these services) would have assigned these services to the cardiology revenue code (048X) and cardiology cost center (03410). Based upon the results of the studies, we found that if 50 percent or more of HOPDs had billed these services with the cardiology revenue code (048X) and cardiology cost center (03140), the geometric mean cost for these codes would have increased and would have resulted in a revised APC assignment from APC 5571 (Level 1 Imaging with Contrast) to APC 5572 (Level 2 Imaging with Contrast).
We were persuaded by the public comments submitted that a majority of the providers who bill these codes would have preferred to bill them with the cardiology revenue code but were not able to do so due to the prior revenue code edit and the remaining procedural hurdles that flowed from the prior revenue code edit.
For CY 2025, we used our equitable adjustment authority under section 1833(t)(2)(E) of the Act to utilize an alternative methodology to calculate the payment for the cardiac CT services in CY 2025. We finalized temporary reassignment of the cardiac CT codes (CPT codes 75572, 75573, and 75574) to APC 5572 (Level 2 Imaging with Contrast).
For CY 2026, CPT code 75572 had 33,272 single claims for ratesetting and a geometric mean cost of around $152. CPT code 75573 had 488 single claims for ratesetting and a geometric mean cost of around $216. CPT code 75573 had 94,419 single claims for ratesetting and a geometric mean cost of around $187. Review of the current claims data indicates that there are providers who are utilizing the cardiology revenue codes.
For CY 2026, we proposed to continue assignment of CPT codes 75572, 75573, and 75574 to APC 5572 (Level 2 Imaging with Contrast) with a proposed payment rate of $358.35, noting as we did in last year's final rule that we anticipate that it may take 3 to 4 years to see an impact from changes in billing practices.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comments: Several commenters supported the APC assignment of CPT codes 75572, 75573, and 75574 to APC (Level 2 Imaging with Contrast). Commenters stated that accurate and stable reimbursement is essential in ensuring that Medicare beneficiaries benefit from evidence-based innovations that improve cardiovascular care. Providers believe that the current APC assignment provides adequate reimbursement that appropriately reflects the clinical complexity and resource requirements to provide these services.
Many commenters believe that these services are underutilized, in part due to historical underpayment and billing restrictions that they encountered in the past and expressed concern about the continued challenges with making changes to billing patterns. Commenters requested that CMS provide a specific timeline of when to expect that these codes would be reassigned to APCs, based on their geometric mean costs, if there is not a notable shift in their geometric mean costs over time. Some commenters shared that they have been working with the various departments within their facilities to effectuate the changes in billing practices, but notable changes are slow. Another commenter shared that they are working with major chargemaster software and advice companies to ensure that providers and facilities are aware that may choose the revenue code that they believe is most appropriate for the services provided. This commenter has also reached out to the National Uniform Billing Committee (NUBC) to suggest new revenue codes that better capture mixed modality services. Commenters noted that because the descriptors for these codes contain “CT”, staff are hesitant to use a revenue code that is not an imaging revenue code. ( printed page 53566)
Many commenters requested educational materials and guidance that specifically indicates that it is appropriate to bill the cardiology revenue codes with cardiac CT services. A commenter suggested specific language that they would like to see CMS use.
Response: We agree with commenters that effectuating change in any size health system can be challenging. As stated in the CY 2025 OPPS/ASC final rule, we will continue to monitor the claims data for these services, anticipating that it may take 3 to 4 years before we see changes in the claims data. While we have seen a number of providers utilizing the cardiology revenue codes for these services, we do not believe that it would be beneficial to provide a specific timeline or deadline for when we would anticipate moving these codes based on their geometric mean costs. If we believe that providers continue to need more time to overcome procedural and logistical hurdles with billing the cardiology revenue codes, we do not believe that a `fixed' timeline should be the determining factor of whether we allow that flexibility or not. We will continue to monitor the claims data for changes in billing practices. If we do not see a significant change in the geometric mean costs after several years, we would revert payment for these services to the standard OPPS payment methodology and assign the cardiac CT codes to the appropriate APCs based on their geometric mean costs.
Many commenters requested guidance from CMS (Medicare Learning Network or “MLN”) that explicitly states that it is appropriate to use the cardiology revenue codes when billing for cardiac CT services. We acknowledge that we had stated in last year's final rule that we would provide public education and instruction through MLN and anticipate that we will do so. As a reminder to our readers, we do not provide specific coding guidance. We refer our readers to section 20.5 in Chapter 4 (Part B Hospital) of the Medicare Claims Processing Manual, CMS “does not instruct hospitals on the assignment of HCPCS codes to revenue codes for services provided under OPPS since hospitals' assignments of cost vary. Where explicit instructions are not provided, HOPDs should report their charges under the revenue code that will result in the charges being assigned to the same cost center to which the cost of those services are assigned in the cost report.” We have consistently stated that hospital outpatient facilities must determine the most appropriate cost center and revenue code for the cardiac CT codes (87 FR 71849, 88 FR 81664).
In summary, for CY 2026, we are finalizing our proposal without modification and assigning CT codes 75572, 75573, and 75574 to APC 5572 (Level 2 Imaging with Contrast). See Table 46 for the CY 2026 final OPPS status indicator and APC assignments. The final CY 2026 payment rates for these codes can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 to this final rule with comment period for the SI definitions for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
8. Cardiac Magnetic Resonance (CMR) Imaging, CPT Codes 75557, 75559, 75561, 75563 (APCs 5523, 5524, 5572, and 5573)
For CY 2026, we proposed to continue to assign the following cardiac magnetic resonance imaging (MRI) CPT codes to APC 5523, 5524, 5572, and 5573 respectively:
- CPT code 75557—(Cardiac magnetic resonance imaging for morphology and function without contrast material) to APC 5523 (Level 3 Imaging without Contrast) with a proposed payment of $245.72 based on a geometric mean cost of around $299 and 1,452 single frequency claims used for ratesetting;
- CPT code 75559—(Cardiac magnetic resonance imaging for morphology and function without contrast material; with stress imaging) to APC 5524 (Level 4 Imaging without Contrast) with a proposed payment of $562.07 based on a geometric mean cost of around $479 and 22 single frequency claims used for ratesetting;
- CPT code 75561—(Cardiac magnetic resonance imaging for morphology and function without contrast material(s), followed by contrast material(s) and further sequences) to 5572 (Level 2 Imaging ( printed page 53567) with Contrast) with a proposed payment of $358.35 based on a geometric mean cost of around $459 and 29,162 single frequency claims used for ratesetting; and
- CPT code 75563—(Cardiac magnetic resonance imaging for morphology and function without contrast material(s), followed by contrast material(s) and further sequences; with stress imaging) to APC 5573 (Level 3 Imaging with Contrast) with a proposed payment of $802.38 based on a geometric mean cost of around $819 and 3,202 single frequency claims used for ratesetting.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: Commenters requested that CPT codes 75557, 75559, 75561, 75563 be reassigned to the Nuclear Medicine and Related Services APC family because cardiac MRI procedures are not clinically similar or similar in resource use to the other procedures assigned to the current APCs. The commenter indicated that the cardiac MRI procedures are more resource intensive because of stress protocols, continuous patient monitoring, specialized staff to care for patients undergoing these procedures, longer procedure room time, and post-stress observation time and many of the other procedures that are in these APCs are lower-acuity, non-cardiac tests. The commenter believes that their current APC assignments are directly responsible for the low use of cardiac MRIs in cardiac patients. The commenter stated that reassigning these procedures to APC 5593 (Level 3 Nuclear Medicine and Related Services) would restore appropriate payment for similar advanced cardiac imaging modalities and resolve the distorted payment signals (that is, blended geometric mean costs are suppressed by high-volume, low-acuity studies).
Alternatively, the commenter suggested creating a cardiac imaging sub-APC that would include cardiac MRI services, stress echocardiography, and nuclear cardiology services that include patient monitoring and recovery. In addition, the commenter had several recommendations for CMS to implement in the interim to improve the accuracy of ratesetting:
- Issue sub-regulatory guidance encouraging hospitals to map cardiac MRI charges to cardiac/stress imaging cost centers rather than generic MRI;
- Ensure packaged items are consistently captured in the claims data; and
- Consider a complexity adjustment or modality-neutral cardiac imaging APC framework.
Response: The OPPS payment rates were proposed based on available CY 2024 claims data. Our claims data shows that APC 5593 (Level 3 Nuclear Medicine and Related Services) has a geometric mean cost of around $1,332 which is significantly higher than the geometric mean costs for the cardiac MRI codes ($299 to $819).
We do not believe that cardiac MRI with stress imaging is very similar to myocardial perfusion imaging because both tests are performed under a stress protocol and therefore should be assigned to APC 5593. APC 5593 contains procedures that describe nuclear medicine tests, not MRI services. We have noted in previous rulemaking that we do not believe MRI services should be assigned to the nuclear medicine APCs (81 FR 79630).
In response to issuing sub-regulatory guidance encouraging HOPDs to utilize a certain revenue code or map charges to a certain cost center, we refer readers to the Medicare Claims Processing Manual, Chapter 4 (Part B Hospital), section 20.5, where we state, “Generally, CMS does not instruct hospitals on the assignment of HCPCS codes to revenue codes for services provided under OPPS since hospitals' assignment of cost vary. Where explicit instructions are not provided, providers should report their charges under the revenue code that will result in the charges being assigned to the same cost center to which the cost of those services are assigned in the cost report.”
In summary, after consideration of the public comment, we are finalizing our proposal without modification to maintain the APC assignments for the cardiac MRI codes for CY 2026. The final CY 2026 payment rates for these codes can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 to this final rule with comment period for the SI definitions for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
Refer to Table 47 for code descriptor, APC assignment and status indicator assignment for CPT codes 75557, 75559, 75561, and 75563 for CY 2026.
( printed page 53568)9. Computational Electrocardiogram (ECG) Analysis System (vMap), CPT Code 0897T (APC 5724)
CPT code 0897T (Noninvasive augmentative arrhythmia analysis derived from quantitative computational cardiac arrhythmia simulations, based on selected intervals of interest from 12-lead electrocardiogram and uploaded clinical parameters, including uploading clinical parameters with interpretation and report) utilizes ECG data to identify potential arrhythmia focal points for patients. The vMap provides augmented information which enables physicians to characterize arrhythmia and assists in triage and treatment of abnormal rhythm. CPT code 0897T became effective July 1, 2024, and since its establishment, the code has been assigned to APC 5724 (Level 4 Diagnostic Tests and Related Services) for CY 2024. For CY 2025, we maintained the APC assignment to APC 5724 (Level 4 Diagnostic Tests and Related Services). For CY 2026, we proposed maintaining the current APC assignment noting that there were no single frequency claims for ratesetting under OPPS and 34 multiple frequency claims, meaning the procedure was completed with other primary services.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: The manufacturer requested that CMS reassign CPT code 0897T to a New Technology APC when the procedure is performed on the day of but separate from the ablation procedure itself until there are sufficient claims data available to support an appropriate clinical APC assignment. The commenter believes that the vMap procedure meets the New Technology APC criteria based on the demonstrated costs of the procedure and that there is precedent for SaaS technologies. In addition to vMap, the commenter urged that CMS pay for all SaaS technologies separately from any underlying procedures and assign these services to New Technology APCs until there are sufficient claims data to support an appropriate clinical APC assignment.
Response: We thank the manufacturer for their input. We do not believe that the vMap procedure meets the New Technology APC criteria based on the costs of the service or because there is precedent for SaaS technologies. There are no cost criteria for a New Technology APC placement. We review the information provided to determine if a service is “truly new” meaning there is not a code or combination of codes to describe the complete service. (We refer readers to the final rule in the November 30, 2001, Federal Register (66 FR 59897) for a full discussion of the criteria and information needed for a New Technology APC assignment.) All New Technology APC applications are reviewed to determine if they meet the criteria for a New Technology APC placement. The vMap procedure has a Category III CPT code which we crosswalked to CPT code 75580 (HeartFlow®) (Noninvasive estimate of coronary fractional flow reserve (FFR) derived from augmentative software analysis of the data set from a coronary computed tomography angiography, with interpretation and report by a physician or other qualified health care professional) and assigned to APC 5724 (Level 4 Diagnostic Tests and Related Services).
In summary, after consideration of the public comment we received, we are finalizing our proposal without modification, to assign CPT code 0897T to APC 5724 (Level 4 Diagnostic Tests and Services as reflected in Table 48.
The final CY 2026 payment rates for this code can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 to this final rule with comment period for the SI definitions for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
10. Fractional Flow Reserve Derived From Computed Tomography (FFRct) CPT Code 75580 (APC 5724)
Fractional Flow Reserve Derived from Computed Tomography (FFRct), also known by the trade name HeartFlow®, is a noninvasive diagnostic service that allows physicians to measure coronary artery disease in a patient through the use of coronary CT scans. The HeartFlow® service is indicated for clinically stable symptomatic patients with coronary artery disease, and, in many cases, may avoid the need for an invasive coronary angiogram procedure. HeartFlow® uses a proprietary data analysis process performed at a central facility to develop a three-dimensional image of a patient's coronary arteries, which allows physicians to identify the fractional flow reserve to assess whether patients should undergo further invasive testing (that is, a coronary angiogram).
HeartFlow® is described by CPT code 75580 (Noninvasive estimate of coronary fractional flow reserve (FFR) derived from augmentative software analysis of the data set from a coronary computed tomography angiography, with interpretation and report by a physician or other qualified health care professional) effective January 1, 2024. CPT code 0503T was the predecessor code for HeartFlow®.
HeartFlow® was assigned to APC 5724 (Level 4 Diagnostic Tests and Related Services) for CY 2024 and CY 2025. In last year's rule, we received a comment stating that several of the Medicare Administrative Contractors (MACs) had an edit in place that prohibited the use of the cardiology revenue code (0480) when billing CPT code 75580 as evidenced by claims ( printed page 53569) denials with “Invalid Revenue Code” errors. Based on the information the commenter provided, we were able to identify the outdated edit and removed it. We reminded readers that it is longstanding CMS policy that hospital outpatient facilities are responsible for reporting the appropriate cost centers and revenue codes on claims. We referred readers to Section 20.5 in Chapter 4 (Part B Hospital) of the CMS Medicare Claims Processing Manual, where we state CMS “does not instruct hospitals on the assignment of HCPCS codes to revenue codes for services provided under OPPS since hospitals' assignment of cost vary. Where explicit instructions are not provided, HOPDs should report their charges under the revenue code that will result in the charges being assigned to the same cost center to which the cost of those services are assigned in the cost report.”
We proposed for CY 2026 to continue to assign HeartFlow® (CPT code 75580) to APC 5724 (Level 4 Diagnostic Tests and Related Services).
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comments: Commenters were supportive of the continued APC assignment to APC 5724 (Level 4 Diagnostic Tests and Related Services); however, the commenters universally requested that CMS exclude the “flawed” or “erroneous” data due to the claims edit that was in place that prohibited them for choosing a revenue center that they deemed most appropriate for the service provided. Several commenters also requested guidance when choosing a revenue center code. They note that this would ensure accurate claims reporting and support reliable data for future ratesetting.
Response: As noted previously in this final rule with comment period, we removed the outdated edit as soon as we were aware that there was an edit in place. HOPDs can use whatever revenue code they believe is most appropriate. We generally do not exclude available claims data, based on the assumption that what is being billed to Medicare is in compliance with coding and billing guidance. We acknowledge that there are a number of procedural and logistical hurdles associated with changing billing practices and will continue to monitor the claims data.
Comment: Many commenters expressed concerns about the lower reimbursement rate for APC 5724 and the movement of services within the APC family. We refer the readers to section III.E.1. of this final rule with comment period for a full discussion about the movement of services within the APC family and the payment variability.
In summary, after consideration of the public comments we received, we are finalizing our proposal without modification to continue to assign CPT 75580 to APC 5724 (see Table 49). The final CY 2026 payment rates for this code can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 to this final rule with comment period for the SI definitions for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
11. Chimeric Antigen Receptor (CAR-T) Administration, CPT Codes 38228 (APC 5694)
Chimeric Antigen Receptor T-Cell (CAR T-cell) therapy is a cell-based gene therapy in which T-cells are collected and genetically engineered to express a chimeric antigen receptor that will bind to a certain protein on a patient's cancerous cells. The CAR T-cells are then administered to the patient to attack certain cancerous cells, and the individual is observed for potential serious side effects that would require medical intervention. We refer readers to previous discussions in the OPPS/ASC final rules with comment period for background regarding the specific CAR T-cell products, including the CY 2020 OPPS/ASC final rule with comment period (84 FR 61231 through 61234) and the CY 2019 OPPS/ASC final rule with comment period (83 FR 58904 through 58908). The AMA created four Category III CPT codes that are related to CAR T-cell therapy, effective January 1, 2019. We also finalized that the procedures described by CPT code 0540T would be assigned status indicator “S” (Procedure or Service, Not Discounted when Multiple) and APC 5694 (Level 4 Drug Administration) from CY 2019 through CY 2024 and did not propose to change the APC assignment for CY 2025 when CPT code 0540T was replaced by CPT code 38228.
As listed in Addendum B to the CY 2026 OPPS/ASC proposed rule, we proposed to continue to assign CPT code 38228 to status indicator “S” (Procedure or Service, Not Discounted when Multiple) and APC 5694 (Level 4 Drug Administration).
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: Several commenters recommended the reassignment of CPT code 38228 to APC 5242 (Level 2 Blood Product Exchange and Related Services). A commenter suggested that rationale for the reassignment of CPT code 38228 to APC 5242 is that APC more accurately reflects the higher facility costs associated with the significant nurse monitoring for the outpatient administration of CAR T-cell therapy. The commenter supported this claim as the AMA placed CPT code 38228 in the same section as stem cell transplant codes, which were initially ( printed page 53570) recognized in the inpatient setting under MS-DRGs 016 and 017. Another commenter also stated CPT code 38228 is analogous to CPT code 38241 (Hematopoietic progenitor cell (hpc); autologous transplantation) and that CMS initially assigned CAR-T related codes to autologous stem cell transplant MS-DRGs 016 and 017, which they believe is the appropriate crosswalk for OPPS.
Another commenter disagreed with the current APC 5694 assignment and stated that CPT code 38338 (and CPT codes 67028 and 67516) do not have the facility NA indicator in the Medicare Physician Fee Schedule and the RUC assigned both facility and non-facility RVUs. Thus, the commenter stated that physicians perform and document the services when they perform the services in facilities.
Response: We continue to believe that the procedures described by CPT codes 38225, 38226, and 38227 describe the various steps required to collect and prepare the genetically modified T-cells, and Medicare does not generally pay separately for each step used to manufacture a drug or biological product. We believe CPT code 38228 is appropriately assigned to APC 5694, which shares similar clinical and resource use as other complex cancer drug administrations. We note that the IPPS established MS-DRG 018 for CAR-T and other immunotherapies and CAR-T therapies are no longer assigned to MS-DRGs 016 and 017. We also disagree that CPT code 38228 is similar clinically and in resource to CPT code 38241 because we view CPT code 38228 as the administration of the CAR-T drugs, while CPT code 38241 involves the transplantation of hematopoietic progenitor cells. Therefore, we are not convinced by the commenter's reason to reassign CPT code 38228 to APC 5242. Furthermore, we believe it is inappropriate to use the IPPS MS-DRGs as an analog to the APC assignments in the OPPS because there are significant differences in resource consumption between the HOPD and inpatient setting.
We are also not compelled to reassign CPT code 38228 to APC 5242 because of the lack of the NA indicator in the Medicare Physician Fee Schedule and because the RUC assignment of both facility and non-facility RVUs do not support the reassignment of CPT codes 38338, 67028 and 67516 to APC 5242. We rely on input from a variety of sources for our APC assignments, including, but not limited to, review of the resource costs and clinical similarity of the service to existing procedures; input from CMS medical advisors; and information from interested specialty societies. We evaluated the recommendations, modeled the suggestions, analyzed the cost results of the suggested APC reassignments, and received additional input from our medical advisors. We note that the drug administration codes that the commenter mentioned were assigned to their respective APCs with similar clinical and resource similarity. Furthermore, the commenter only suggested an APC reassignment for CPT code 38228 but did not provide any APC suggestions for CPT codes 67028 and 67516.
After consideration of the public comments we received, we are finalizing our proposed APC assignment and status indicator for CPT code 38228 to APC 5694 without modification. Refer to Table 50 for the final OPPS APC and status indicator assignment for CPT code 38228 for CY 2026. We refer readers to Addendum B to this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
12. CVi® Contrast Delivery System
The CVi® Contrast Delivery System is an automated Contrast Management System with multiuse syringes. Contrast Management Systems are designed to assist physicians in infusing iodinated contrast media administered to patients in common angiograph procedures. Contrast with angiography procedures is often administered through a handheld, manual injection device. Automated contrast management systems use variable rate technology to deliver a specific amount of contrast.
We received public comments on this topic. The following is a summary of the comments we received and our responses.
Comment: A commenter requested that CMS establish a G-code to provide additional payment to recognize the cost of automated contrast management systems with angiography procedures. The commenter stated that providing additional payment through the establishment of a new code would facilitate expanded patient access to automated systems that address important policy goals and address significant unmet needs such as reducing the incidence of Acute Kidney Injury (CA-AKI), reducing costs related to CA-AKI related hospitalization, reducing contrast waste, alleviating contrast media shortages precipitated by supply chain issues and reducing physician radiation exposure. The commenter stated that it is necessary to establish a G-code because existing Medicare payment policies for Percutaneous Coronary Intervention (PCI) and other procedures do not differentiate procedures that involve an automated variable rate contrast injector ( printed page 53571) with multiuse syringe for the administration of iodinated contrast media. They also noted that these procedures are coded and paid identically, regardless of whether the provider has invested in automated contrast management systems that they state provide better health outcomes.
Response: After reviewing the information provided by the commenter and following consultation with our medical officers, we have determined that this equipment is only used in conjunction with another procedure and would be packaged for payment consistent with our policy of packaging items and services that are typically integral, ancillary, supportive, dependent, or adjunctive to a primary service.
Therefore, we are not creating a G-code to describe the use of an automated variable rate contrast injector with multiuse syringe for the administration of iodinated contrast media for CY 2026.
Dental Related Procedures
13. Malignant Tumor/Lesion Removal Dental Procedures, CDT Codes D7440-D7441 (APC 5164)
Effective January 1, 2024, we made over 200 additional dental codes payable under the OPPS when payment and coverage requirements are met, as provided in the relevant PFS payment rules regarding Medicare Part B payment for dental services (88 FR 81540-82185). Of these payable dental codes, we assigned Current Dental Terminology (CDT) codes D7440 ( Excision of malignant tumor-lesion diameter up to 1.25cm) and D7441(Excision of malignant tumor- lesion diameter greater than 1.25 cm) to APC 5164 (Level 4 ENT Procedures) and status indicator “J1.” For CY 2026, we proposed to continue to assign CDT codes D7440 and D7441 to APC 5164 and status indicator “J1.”
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter requested that CMS add CDT codes D7440 and D7441 to the list of covered outpatient services for CY 2026.
Response: We are clarifying that CDT codes D7440 and D7441 are already payable when performed in the outpatient hospital department and coverage requirements are met. Our proposal would maintain the APC and status indicator assignments for CY 2026.
Therefore, we are finalizing our proposal to continue to assign CDT codes D7440 and D7441 to APC 5164 and status indicator “J1” for CY 2026.
Endoscopy Procedures
14. Biliary Endoscopy Procedure, CPT Code 47555 (APC 5341)
CPT code 47555 ( Biliary endoscopy, percutaneous via T-tube or other tract; with dilation of biliary duct stricture(s) without stent) describes the procedure for dilation of bile ducts using an endoscope. Using CY 2024 claims data, CPT code 47555 had geometric mean cost of $8,577.51 in the CY 2026 OPPS/ASC proposed rule. For CY 2026, we believed this procedure was still appropriately assigned to APC 5341 (Level 1 Abdominal/Peritoneal/Biliary and Related Procedures), which had a proposed payment rate of $3,698.49. Therefore, we proposed to continue assigning CPT code to APC 5341.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter requested that CMS reassign CPT code 47555 to APC 5342 (Level 2 Abdominal/Peritoneal/Biliary and Related Procedures), which had a proposed payment rate of $6,667. The commenter stated that the cost of this procedure exceeded the payment rate for APC 5341.
Response: CPT code 47555 has an updated GMC of $8,068.88. After further clinical review, we agree with the commenter that resources and costs associated with CPT code 47555 would be more appropriately reflected by APC 5342. In summary, we are finalizing our proposal with modification to assign CPT code 47555 to APC 5342 (Level 2 Abdominal/Peritoneal/Biliary and Related Procedures) for CY 2026. Table 51 shows the finalized status indicator and APC assignment for the procedure code. We refer readers to Addendum B to this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
15. Endoscopic Procedure—Upper GI Tract, CPT Code 43252 (APC 5302)
CPT code 43252 ( Esophagogastroduodenoscopy, flexible, transoral; with optical endomicroscopy) describes a service that is used to visualize the upper portions of the GI tract from the esophagus to the duodenum. Using CY 2024 claims data, CPT code 43252 had geometric mean cost of $1,739.85 in the CY 2026 OPPS/ASC proposed rule. For CY 2026, we believed this procedure was still appropriately assigned to APC 5302 (Level 2 Upper GI Procedures), which had a proposed payment rate of $1,975.59. Therefore, we proposed to continue assigning CPT code to APC 5302.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A few commenters requested that CMS assign CPT code 43252 to APC 5303 (Level 3 Upper GI Procedures) with a payment rate of around $4,002 for CY 2026. The commenters stated that hospitals have historically underreported the costs for this procedure, therefore skewing the GMC for CPT code 43252.
Response: We have stated regularly over the history of the OPPS, it is the responsibility of providers and other interested parties and not CMS to resolve potential claims and reporting issues for individual CPT codes and medical services payable by Medicare. The updated GMC for the service, which is around $1,737.41, is lower than the payment rate for APC 5302 which is ( printed page 53572) around $1,960. Therefore, we are finalizing our proposal, without modification, to continue to assign CPT code 43252 to APC 5302 (Level 2 Upper GI Procedures). Table 52 shows the finalized status indicator and APC assignment for the procedure codes. We refer readers to Addendum B to this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
16. Endoscopic Retrograde Cholangiopancreatography (ERCP) With Stone Destruction Procedure, CPT Code 43265 (APC 5331)
CPT code 43265 ( Endoscopic retrograde cholangiopancreatography (ERCP); with destruction of calculi, any method (eg., mechanical, electrohydraulic, lithotripsy)) describes the procedure for destruction of stone of bile or pancreatic ducts. Using CY 2024 claims data, CPT code 43265 had geometric mean cost of $9,011.93 in the CY 2026 OPPS/ASC proposed rule. For CY 2026, we believed this procedure was still appropriately assigned to APC 5331 (Complex GI Procedures), which had a proposed payment rate of $6,276.20. Therefore, we proposed to continue assigning CPT code to APC 5331.
Comment: A commenter requested that CMS reassign CPT code 43265 to APC 5362 (Level 2 Laparoscopy and Related Services), which had a proposed payment rate of $10,966.50, due to the similarity to CPT code 47554 (Biliary endoscopy, percutaneous via t-tube or other tract; with removal of calculus/calculi).
Response: We thank the commenter for their input. CPT code 43265 has an updated geometric mean cost of $8,754.94. After clinical review of this procedure, we continue to believe that this procedure is appropriately assigned to the APC 5331. Therefore, we are finalizing our proposal, without modification, to continue to assign CPT codes 43265 to APC 5331 (Complex GI Procedures). Table 53 shows the finalized status indicator and APC assignment for the procedure codes. We refer readers to Addendum B to this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
17. Endoscopic Submucosal Dissection (ESD) Procedure, HCPCS Code C9779 (APC 5303)
We established HCPCS code C9779 ( Endoscopic submucosal dissection (ESD), including endoscopy or colonoscopy, mucosal closure, when performed) effective October 1, 2021, to describe the endoscopic submucosal dissection (ESD) performed during an endoscopy or colonoscopy. HCPCS code C9779 was established based on a New Technology application that was submitted to CMS for New Technology consideration under the OPPS. Based on our assessment, we assigned the code to APC 5313 (Level 3 Lower GI Procedures) because we believed the ESD procedure had similar clinical characteristics and resource costs as the surgical procedures assigned to APC 5313. We announced the assignment to APC 5313 in the October 2021 OPPS quarterly update CR (Transmittal 10997, Change Request 12436, dated September 16, 2021). In CY 2022, we continued to assign the code to APC 5313.
For CY 2023, we assigned HCPCS code C9779 to APC 5303 (Level 3 Upper GI Procedures) after receiving public comments that stated that the ESD procedure's resource requirements and geometric mean cost were more similar to the resource requirements and geometric mean costs of procedures found in APC 5303. Further, commenters noted that the ESD procedure is technically more demanding, requires advanced skills to perform, and is clinically similar to CPT code 43497 (Lower esophageal myotomy, transoral ( i.e., peroral endoscopic myotomy [POEM])), which was assigned to APC 5303. For CY 2026, we proposed to maintain HCPCS code C9779, which had a geometric mean cost of $5,516.81, in APC 5303 (Level 3 Upper GI Procedures). APC 5303 had a proposed CY 2026 payment rate of $4,002.57.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: Commenters requested that CMS reassign HCPCS code C9779 to APC 5331 (Complex GI procedures), which had a proposed payment rate of ( printed page 53573) $6,276.20. The commenters reasoned that HCPCS code C9779 would be more appropriately placed in APC 5331 due to its similarity to CPT code 43479. Additionally, the commenters stated that as HCPCS code C9779 includes both combined upper and lower GI ESD procedures, there is great variability in the costs reported by hospitals which could affect the accuracy of the geometric mean cost.
Response: HCPCS code C9779 has an updated geometric mean cost of $5,182.09. While the geometric mean cost for HCPCS code C9779 is slightly closer to the payment rate for APC 5331, we continue to believe that HCPCS code C9779 is appropriately assigned, based on both clinical and resource similarity, to APC 5303 (Level 3 Upper GI Procedures). Therefore, we are finalizing our proposal, without modification, to continue to assign CPT code C9779 to APC 5303 (Level 3 Upper GI Procedures). Table 54 shows the finalized status indicator and APC assignment for the procedure codes. We refer readers to Addendum B to this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
18. Esophageal Balloon Distention Study, CPT Code 91040 (APC 5723)
Esophageal balloon distension studies are used to diagnose conditions of the esophagus and may be used to determine the source of certain types of pain, such as chest pain. The esophageal balloon study is often performed in conjunction with esophagogastroduodenoscopy procedures. Using CY 2024 claims data, CPT Code 91040 (Esophageal balloon distension study, diagnostic, with provocation when performed) had a proposed geometric mean cost of $2,007.29 in the CY 2026 OPPS/ASC proposed rule. For CY 2026, we proposed to continue to assign CPT code 91040 to APC 5723 (Level 3 Diagnostic Tests and Related Services) with a proposed payment rate of around $382.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: Commenters requested that CMS reassign CPT code 91040 to APC 5724 (Level 4 Diagnostic Test and Related Services). Commenters stated that CPT code 91040 has the highest geometric mean cost of all codes assigned to APC 5723 and had a higher device offset percentage than other procedures in the same APC.
Response: We note that the updated geometric mean cost for CPT code 91040 is $2,001.92, and the geometric mean cost for APC 5723 is $384.16. While this is a significant difference, the CY 2024 claims data shows that only 2.6 percent (approximately 102 claims) of all the claims billed with CPT code 91040 (3,927 total claims frequency) were billed with only CPT code 91040 on the claim. We believe that, in addition to the higher costs of the packaged items, the costs from the other procedures that are performed with CPT code 91040 have driven up the geometric mean cost of CPT code 91040. Based on review of this procedure, other procedures in the same APC family, and the claims data, we believe that the clinical and resource characteristics of CPT code 91040 are sufficiently like other procedures assigned to APC 5723 and CPT code 91040 should continue to be assigned to APC 5723.
Therefore, for CY 2026 we are finalizing our proposal, without modification, to continue to assign CPT code 91040 to APC 5723 (Level 3 Diagnostic Tests and Related Services). Table 55 shows the finalized status indicator and APC assignment for the procedure codes. We refer readers to Addendum B to this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
19. Transnasal EGD, CPT Codes 0652T, 0653T, and 0654T (APCs 5302 and 5303)
CPT codes 0652T ( Esophagogastroduodenoscopy, flexible, transnasal; diagnostic, including collection of specimen(s) by brushing or washing, when performed (separate procedure)), 0653T ( Esophagogastroduodenoscopy, flexible, transnasal; with biopsy, single or multiple), and 06534T ( Esophagogastroduodenoscopy, flexible, transnasal; with insertion of intraluminal tube or catheter) describe the procedures for transnasal esophagogastroduodenoscopy. Using CY 2024 claims data, CPT codes 0652T, 0653T, and 0654T had geometric mean costs of $1,897.72, $1,107.54, $1,064.23, ( printed page 53574) respectively, in the CY 2026 OPPS/ASC proposed rule. For CY 2026, we believed CPT codes 0652T and 0653T were still appropriately assigned to APC 5302 (Level 2 Upper GI Procedures), which had a proposed payment rate of $1,975.59. We also believed that CPT code 0654T was still appropriately assigned to APC 5303 (Level 3 Upper GI Procedures), which had a proposed payment rate of $4,002.57. Therefore, we proposed to continue assigning CPT codes 0652T and 0653T to APC 5302 and CPT code 0654T to APC 5303.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter requested that CMS reassign CPT codes 0652T and 0653T to APC 1562 (New Technology—Level 25 ($3,501-$4,000)) and CPT code 0654T to APC 1563 (New Technology—Level 26 ($4,001-$4,500)). The commenter stated that since these are low volume procedures, reassigning these procedures to New Technology APCs would allow them to receive payment rates appropriate to their resource costs.
Response: We thank the commenter for their input. CPT codes 0652T, 0653T, and 0654T have updated GMCs of $1,744.52, $1,096.83, and $1,111.46 in the final rule data. After further clinical review, we continue to believe that CPT codes 0652T, 0653T, and 0654T are still appropriately assigned to APCs 5302 and 5303. Additionally, all three procedures have GMCs lower than the payment rate of their assigned APCs. Therefore, we are finalizing our proposal, without modification, to continue to assign CPT codes 0652T and 0653T to APC 5302 (Level 2 Upper GI Procedures) and CPT code 0654T to APC 5303 (Level 3 Upper GI Procedures). Table 56 shows the finalized status indicator and APC assignment for the procedure codes. We refer readers to Addendum B to this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
Gastrointestinal Services
20. Gastric Electrophysiology Mapping With Simultaneously Validated Patient System Profiling (GEMS) Service, CPT Code 0868T (APC 5723)
Effective July 1, 2023, based on a New Technology application received by CMS for the GEMS service, CMS established HCPCS code C9787 ( Gastric electrophysiology mapping with simultaneous patient symptom profiling) and assigned it to APC 5723 (Level 3 Diagnostic Tests and Related Services) based on a crosswalk to CPT code 0779T. Effective July 1, 2024, HCPCS code C9787 was deleted and replaced by CPT code 0868T (High-resolution gastric electrophysiology mapping with simultaneous patient symptom profiling, with interpretation and report). CMS assigned CPT code 0868T to APC 5723, the same APC to which its predecessor code, HCPCS code C9787, was assigned. For CY 2026, CMS proposed to continue to assign CPT code 0868T to APC 5723 with a proposed payment rate of $381.96 for CY 2026.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: Several commenters requested that CMS not finalize the proposed APC assignment for CPT code 0868T. A commenter explained that the current payment amount is too low and has impacted the ability for the service to be provided to pediatric patients because Medicare payment rates impact the rates of commercial payors and state Medicaid programs. Some commenters drew distinctions between the CPT codes 0868T and 0779T. For example, a commenter stated that CPT code 0868T is fundamentally different from CPT code 0779T across every domain, including the technology, clinical protocol, staff and facility resources, and diagnostic outputs. Commenters requested that CMS assign CPT code 0868T to APC 5724 (Level 4 Diagnostic Tests and Related Services) or a new technology APC.
Response: We appreciate the commenters' input. First, we note that there is extremely low claims volume currently available for CPT code 0868T to justify a change in APC assignment based on existing claims data at this time. While the geometric mean cost of CPT code 0868T is approximately $3,000, this geometric mean cost is based on only 7 single frequency claims, which means that there is a high degree of variability in the limited claims data available. Additionally, based on our review of the technology and in consultation with our medical officers, we continue to believe that CPT codes 0868T and 0779T are comparable services in that they are both studies measuring gastrointestinal physiological activity. Additionally, there are other similar codes that describe gastrointestinal services assigned to APC 5723, such as CPT code 91020. Therefore, we believe that our proposal to assign CPT code 0868T is appropriate for CY 2026. We will continue to monitor the claims data and adjust the APC placement for CPT code 0868T based on the claims data in future rulemaking.
After consideration of the public comments, we are finalizing our proposal without modification to continue to assign CPT code 0868T to ( printed page 53575) APC 5723. The final CY 2026 payment rate for the code can be found in Addendum B to this final rule with comment period. We also refer readers to Addendum D1 to this final rule with comment period for the SI meanings for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
21. IB—Stim, CPT 64567 (APC 5301)
CPT code 64567 replaces placeholder code 64X11 (Percutaneous electrical nerve field stimulation, cranial nerves, without implantation) and is effective January 1, 2026. The code describes a neuromodulation therapy for the treatment of functional gastrointestinal disorders. CPT code 64567 replaces existing CPT code 0720T (Percutaneous electrical nerve field stimulation, cranial nerves, without implantation). For CY 2026, we proposed to assign CPT code 64567 to APC 5724 (Level 4 Diagnostic Tests and Related Services) and delete predecessor CPT code 0720T.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter requested that CMS reassign CPT code 64567 to APC 1515 (New Technology—Level 15 ($1301-1400)) with a payment rate of $1,350.50 for CY 2026. The commenter believes the procedure requires more resources than other services currently assigned to APC 5724, and that it is not clinically similar to those other procedures since it represents a therapeutic intervention rather than a diagnostic service. They further explained that no appropriate clinical APC exists for this procedure and note that it has very low Medicare utilization rates upon which to base a clinical APC assignment.
Response: After careful review of the service and the comment received as well as discussions with our medical officers, we are finalizing our proposals with modification. First, we are finalizing our proposal to delete CPT code 0720T as CPT code 64567 is replacing the predecessor code. However, we are not finalizing our proposal to assign CPT code 64567 to APC 5724, due to the clinical characteristics of the service. Specifically, we agree with the commenter that continued assignment to APC 5724 would not be appropriate because we believe CPT code 64567 represents a therapeutic service rather than a diagnostic procedure, based on the information available. We note that there were no claims for the service in CY 2024 for us to consider in our analysis. Therefore, due to the service's clinical characteristics, for CY 2026, we are assigning CPT code 64567 to APC 5301 (Level 1 Upper GI Procedures) as reflected in Table 57 with a payment rate of $926.63. We note that we will continue to monitor the claims data and update the payment rate in future rulemaking based on the available claims. The final CY 2026 payment rate for the code can be found in Addendum B to this final rule with comment period. We also refer readers to Addendum D1 to this final rule with comment period for the SI meanings for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
Imaging Services
22. Computed Tomographic Colonography, CPT Code 74263 (APC 5523)
For CY 2024, we assigned CPT code 74263 (Computed tomographic (CT) colonography, screening, including image postprocessing) to status indicator “E1” indicating that the service was not covered and not payable by Medicare under OPPS. For CY 2025, we finalized assigning CPT code 74263 to APC 5523 (Level 3 Imaging without Contrast) using CPT code 74176 (Computed tomography, abdomen and pelvis; without contrast material) as a crosswalk code because of the coverage changes for colorectal cancer screening services. (See 89 FR 94287 through 94290 for a full discussion of the coverage changes for colorectal cancer screening services.)
For the CY 2026 OPPS/ASC proposed rule, we proposed to maintain the APC assignment for CPT code 74263 in APC 5523 (Level 3 Imaging without Contrast) with a proposed payment of around $245 and a status indicator of `S' (Procedure or Service, Not Discounted When Multiple; Paid under OPPS).
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter requested that CMS reassign CPT code 74263 to APC 5524 (Level 4 Imaging without Contrast) with a proposed payment of approximately $562. The commenter noted that CT colonography is a safe, minimally invasive exam that can save lives by detecting early disease. They stated that a lower reimbursement rate is likely to disproportionately affect underserved communities and exacerbate disparities in the early diagnosis of colorectal cancer.
Response: We thank the commenter for the input; however, we disagree with the commenters and believe that CPT code 74263 is appropriately assigned to APC 5523 (Level 3 Imaging without Contrast). Given that any claims data in CY 2024 would be from before the service was payable in the OPPS, it would be difficult to rely on the claims data to determine the APC assignment for this code. We reviewed the claims data and cost information for the crosswalk code and continue to believe that CPT code 74176 is an appropriate crosswalk code for CPT 74263. CPT code 74176 has a geometric mean cost around $164 based on 228,147 single frequency claims used for ratesetting under OPPS. After review and input from our CMS Medical Officers, we believe that CPT code 74263 is ( printed page 53576) appropriately assigned to APC 5523 (Level 3 Imaging without Contrast).
In summary, after consideration of the public comments we received, we are finalizing our proposal without modification, to assign CPT code 74263 to APC 5523 (Level 3 Imaging without Contrast). The final CY 2026 payment rates for this code can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 to this final rule with comment period for the status indicator definitions for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
Refer to Table 58 for code descriptor, APC assignment and status indicator assignment CPT code 74263 for CY 2026.
23. Computed Tomographic Services (Head, Neck, and Cerebral Perfusion), CPT Codes 70471 and 70473 (APCs 5572, 5571)
The CPT Editorial Panel created 3 new Category I CPT codes 70471, 70472, and 70473 effective January 1, 2026 to replace the Category III CPT code 0042T. CPT code 0042T will be deleted effective January 1, 2026. The new final CPT codes (along with their placeholder codes) and the deleted Category III code are listed below with their long descriptors.
- 70471 (70XX1): Computed tomographic angiography (CTA), head and neck, with contrast material(s), including noncontrast images, when performed, and image postprocessing
- 70472 (70XX2): Computed tomographic (CT) cerebral perfusion analysis with contrast material(s), including image postprocessing performed with concurrent CT or CT angiography of the same anatomy (List separately in addition to code for primary procedure)
- 70473 (70XX3): Computed tomographic (CT) cerebral perfusion analysis with contrast material(s), including image postprocessing performed without concurrent CT or CT angiography of the same anatomy
- 0042T: Cerebral perfusion analysis using computed tomography with contrast administration, including post-processing of parametric maps with determination of cerebral blood flow, cerebral blood volume, and mean transit time
For CY 2026, we proposed to assign these codes to the following APCs and status indicators (SIs):
- Assign CPT code 70471 to APC 5572 (Level 2 Imaging with Contrast), SI of `S' (Procedure or Service, Not Discounted When Multiple; Paid under OPPS);
- Assign CPT code 70472 to SI of `N' (Items and Services Packaged into APC Rates; Paid under OPPS; payment is packaged into payment for other services); and
- Assign CPT 70473 to APC 5571 (Level 1 Imaging with Contrast), SI of `S' (Procedure or Service, Not Discounted When Multiple; Paid under OPPS).
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter requested that CMS assign CPT code 70473 to APC 5572 (Level 2 Imaging with Contrast). The commenter indicated that the procedure described by CPT code 70473 is similar clinically and in resource use to the computed tomographic angiography procedure described by CPT code 70471. The commenter noted that the angiography procedure and the perfusion analysis are not always performed on the same date of service.
Response: After review of these new codes and input from our CMS Medical Officers, we believe that CPT code 70473 is more clinically similar to CPT code 70460 (Computed tomography, head or brain; with contrast material(s)),which has a geometric mean cost of around $173 and 2,659 single frequency claims in the CY 2024 claims data used for CY 2026 OPPS ratesetting. CPT code 70460 is currently assigned to APC 5571 (Level 1 Imaging with Contrast) and has a proposed payment of around $179. We remind hospitals that we review, on an annual basis, the APC assignments for all items and services paid under the OPPS.
In summary, after consideration of the public comments we received, we are finalizing our proposal without modification. The final CY 2026 payment rates for these codes can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 to this final rule with comment period for the SI definitions for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
Refer to Table 59 for code descriptors, APC assignments and status indicator assignments for CPT codes 70471, 70472, and 70473 for CY 2026.
( printed page 53577)24. Duplex Scan of Extracranial Arteries, CPT Code 93880 (APC 5523)
For CY 2026, we proposed to continue to assign CPT code 93880 ( Duplex scan of extracranial arteries; complete bilateral study) to APC 5523 (Level 3 Imaging without Contrast) and a status indicator of `S' with a proposed payment of $245.72.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter requested that CMS reassign CPT code 93880 to APC 5524 (Level 4 Imaging without Contrast) with a proposed payment of $562.07. The commenter stated that CPT code 93880 should be reassigned due to its clinical and resource similarity to CPT code 93306 (Echocardiography, transthoracic, real-time with image documentation (2d), includes m-mode recording, when performed, complete, with spectral doppler echocardiography, and with color flow doppler echocardiography), which is assigned to APC 5524.
Response: We thank the commenter for their input; however, we disagree with their recommendation. We review, on an annual basis, the APC assignments for all services and items paid under the OPPS based on our analysis of the latest claims data. For the CY 2026 OPPS update, based on CY 2024 claims data, our analysis for this final rule with comment period supports the continued assignment of CPT code 93880 to APC 5523 (Level 3 Imaging without Contrast) based on its clinical and resource homogeneity to the procedures and services in APC 5523. Specifically, our claims data show a GMC of approximately $230 based on 410,021 single frequency claims for CPT code 93880, which is consistent with the GMC of approximately $246 for APC 5523, rather than the GMC of approximately $563 for APC 5524 (Level 4 Imaging without Contrast). We believe the resource requirements for CPT code 93880 are more similar to procedures found in APC 5523 (Level 3 Imaging without Contrast) rather than APC 5524 (APC Level 4 Imaging without Contrast).
In summary, after consideration of the public comment, we are finalizing our proposal without modification to assign CPT code 93880 to APC 5523 (Level 3 Imaging without Contrast) for CY 2026. The final CY 2026 payment rates for this code can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 to this final rule with comment period for the SI definitions for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
25. Duplex Scan of Hemodialysis Fistula, CPT Code 0876T
The AMA CPT Editorial Panel established CPT code 0876T ( Duplex scan of hemodialysis fistula, computer-aided, limited (volume flow, diameter, and depth, including only body of fistula)) effective July 1, 2024. We assigned CPT code 0876T to status indicator `E1' for July 1, 2024, based on our belief that the implantable marker used with this procedure did not have FDA approval.
For CY 2026, we proposed to continue to assign CPT code 0876T to E1 (Not covered by any Medicare outpatient benefit category; Statutorily excluded by Medicare, Not reasonable and necessary; Not paid by Medicare when submitted on outpatient claims) because we have not been notified by interested parties regarding an updated FDA status.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter requested that CMS assign CPT code 0876T to an APC for payment under the OPPS. The commenter noted that the vast number of end-stage renal disease admissions are related to complications with patients' vascular access and the cost savings of salvaging fistulas versus replacing them.
Response: We are unable to determine if all parts of this service have been approved by the FDA and therefore are maintaining the status indicator assignment of ‘E1’. We look forward to hearing from the manufacturer when FDA approval has been obtained.
In summary, after consideration of the public comment that we received, we are finalizing the status indicator assignment for 0876T without modification. The final CY 2026 payment rates for this code can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 to this final rule with comment period for the SI definitions for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
26. Non-Cardiac Contrast Enhanced Ultrasound (CEUS), CPT Codes 76978 and 76979 (APC 5572)
CPT codes 76978 and 76979 describe non-cardiac contrast enhanced ultrasounds. Their code descriptors are as follows:
- 76978: Ultrasound, targeted dynamic microbubble sonographic ( printed page 53578) contrast characterization (non-cardiac); initial lesion.
- 76979:Ultrasound, targeted dynamic microbubble sonographic contrast characterization (non-cardiac); each additional lesion with separate injection (List separately in addition to code for primary procedure).
For CY 2026, we proposed to assign CPT codes 76978 and 76979 to APC 5571 (Level 1 Imaging with Contrast). CPT code 76978 had a geometric mean cost of around $287 based on 710 single frequency claims and CPT code 76979 was packaged with a primary procedure.
Comment: Several commenters requested reassignment of CPT codes 76978 and 76969 from their current assignment in APC 5571 (Level 1 Imaging with Contrast) to APC 5572 (Level 2 Imaging with Contrast). CPT code 76978 describes ultrasound, targeted dynamic microbubble sonographic contrast characterization (non-cardiac) for the initial lesion, while CPT code 76979 describes each additional lesion with a separate injection and is packaged with the primary procedure code per OPPS policy. The commenters stated that the current APC assignment does not adequately reflect the resource costs associated with these specialized contrast-enhanced ultrasound procedures.
Response: We agree with the commenters' request for APC reassignment. After reviewing the clinical characteristics and resource costs associated with CPT codes 76978, we are reassigning 76978 from APC 5571 (Level 1 Imaging with Contrast) to APC 5572 (Level 2 Imaging with Contrast) for CY 2026 as reflected in Table 60. This reassignment better reflects the complexity and resource intensity of non-cardiac contrast enhanced ultrasound procedures. CPT code 76978 will be assigned status indicator “S” (separately payable) under APC 5572, while CPT code 76979 will maintain status indicator “N” (packaged) and will continue to be packaged with the primary procedure code 76978 under the new APC assignment. This change recognizes the specialized nature of contrast-enhanced ultrasound technology and ensures appropriate payment for these services. The reassignment will be effective beginning January 1, 2026.
27. Irreversible Electroporation Ablation of Tumors (NanoKnife® System), CPT Codes 0600T, 47384, 55877 (APC 5362)
Effective July 1, 2020, the AMA CPT Editorial Panel established CPT code 0600T ( Ablation, irreversible electroporation; 1 or more tumors per organ, including imaging guidance, when performed, percutaneous) which describes a technique in which an electrical field is applied to cells in order to increase the permeability of the cell membranes through the formation of nanoscale defects in the lipid bilayer. The result is creation of nanopores in the cell membrane and disruption of intra-cellular homeostasis, ultimately causing cell death. The procedure received CPT code 0600T from the AMA in July 2020, and we assigned CPT code 0600T to APC 5362 (Level 2 Laparoscopy and Related Services) in the CY 2021 OPPS/ASC final rule with comment period. For the CY 2026 OPPS/ASC proposed rule, CPT code 0600T had a geometric mean cost of around $13,068 and we proposed to continue to assign the procedure to APC 5362, which has a proposed payment rate of around $10,967 and status indicator J1 (Hospital part B services paid through a comprehensive APC). For the CY 2026 OPPS/ASC proposed rule, we proposed CPT code 0600T to be a device intensive procedure with a proposed device offset percentage of 57.02 percent based on claims data for CPT code 0600T.
Effective July 1, 2025, the AMC CPT Editorial Panel established placeholder CPT code 4001X (Ablation, irreversible electroporation, liver, 1 or more tumors, including imaging guidance, percutaneous); and placeholder CPT code 5XX11 (Ablation, irreversible electroporation, prostate, 1 or more tumors, including imaging guidance, percutaneous). CPT code 47384 is the final code for 4001X and CPT code 55877 is the final code for 5XX11. For the CY 2026 OPPS/ASC proposed rule, we proposed to assign CPT code 47384 and CPT code 55877 to APC 5362 and status indicator J1. For the CY 2026 OPPS/ASC proposed rule, we proposed CPT code 47384 and CPT code 55877 to be device intensive procedures with a proposed device offset percentage of 31 percent which is the default for new device intensive procedures that lack claims data, or lack claims data from a predecessor code or a clinically related or similar code.
We note that at the August 25, 2025, HOP Panel Meeting, a presentation was made requesting: (1) reassignment to APC 5377 (Level 7 Urology and Related Services) for CPT code 55877; (2) reassignment to APC 1575 (New Technology—Level 38) for CPT codes 47384 and 0600T; and (3) the use of the claims data for CPT code 0600T to determine the device offset percentage for CPT codes 47384 and 55877. Based ( printed page 53579) on the information presented at the meeting, the Panel recommended that CMS use the claims data for CPT code 0600T to determine the device offset percentage for CPT code 47384 and CPT code 55877. The Panel made no recommendation on the APC assignments for CPT codes 0600T, 47384 and 55877.
We received public comments on this topic. The following is a summary of the comments we received and our responses.
Comment: A few commenters requested reassignment from APC 5362 to APC 5377 (Level 7 Urology and Related Services) for CPT code 55877 and reassignment from APC 5362 to APC 1575 (New Technology—Level 38) for CPT codes 47384 and 0600T. One of the commenters additionally requested device-intensive status for CPT codes 0600T, 47384 and 55877. We refer readers to sections IV.B. and XIII.C. of this final rule with comment period for our response those requests.
The commenters stated that all three procedures need to be reassigned because the CY 2024 claims data for CPT code 0600T shows a geometric mean cost of $13,068, which is higher than the proposed APC 5362 geometric mean cost of $11,137. For which specific APCs the procedures should be assigned to, the commenters stated that it is appropriate to assign CPT code 55877 to APC 5377 (Level 7 Urology and Related Services) based on resource and clinical coherence. The commenters stated that the costs associated with CPT code 0600T are more akin to the geometric mean of APC 5377 and the clinical range of services included in APC 5377 encompass ablation procedures, including CPT 55882, that are used in treating the same patient diagnoses and population. For CPT codes 0600T and 47384, commenters stated that there is no clinically appropriate APC that fits these procedures from both a clinical and resource perspective, so they proposed to reassign them to APC 1575 based on geometric mean cost. Alternatively, the commenter suggested, if CMS believes that all three procedures should be assigned to the same APC, the alternative APC assignment for placeholder code 55877 could be APC 1575.
Response: We thank the commenters for their recommendations. After consideration of the public comments we received and discussion and input from our Medical Officers, we are finalizing our proposal without modification for the APC assignments for CPT codes 0600T, 47384 and 55877. We believe the current claim data for 0600T indicates that APC 5362 is an appropriate assignment for these services. We note that we review the APC assignments for all items and services paid under the OPPS on an annual basis. We will reevaluate the APC assignments for CPT codes 0600T, 47384 and 55877 in the next rulemaking cycle.
Table 61 shows the finalized status indicator and APC assignment for these procedure codes. We refer readers to Addendum B to this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
Laboratory Related Services
Certain clinical diagnostic laboratory tests that are listed on the Clinical Laboratory Fee Schedule (CLFS) are packaged in the OPPS as integral, ancillary, supportive, dependent, or adjunctive to the primary service or services provided in the hospital outpatient setting during the same outpatient stay. Specifically, we conditionally package laboratory tests and only pay separately for laboratory tests when (1) they are the only services provided to a beneficiary on a claim; (2) they are molecular pathology tests; (3) they are advanced diagnostic laboratory tests (ADLTs) that provide an analysis of multiple biomarkers of DNA, RNA, or proteins combined with a unique algorithm to yield a single patient-specific result; or (4) the laboratory tests are considered preventive services. When laboratory tests are not packaged under the OPPS and are listed on the CLFS, they are paid at the CLFS payment rates, outside the OPPS, under Medicare Part B.
28. Diagnostic Biomarker Tests for Alzheimer's Disease, CPT Codes 0551U and 0568U
Recently, the AMA CPT Editorial Panel created two new CPT codes to describe diagnostic biomarker tests for Alzheimer's disease. CPT code 0551U became effective April 1, 2025, and CPT code 0568U became effective October 1, 2025. The CPT codes and their descriptors are as follows:
- 0551U (Tau, phosphorylated, pTau217, by single-molecule array (ultrasensitive digital protein detection), using plasma); and
- 0568U (Neurology (dementia), beta amyloid (Aβ40, Aβ42, Aβ42/40 ratio), tau-protein phosphorylated at residue (eg, pTau217), neurofilament light chain (NfL), and glial fibrillary acidic protein (GFAP), by ultra-high sensitivity molecule array detection, plasma, algorithm reported as positive, intermediate, or negative for Alzheimer pathology).
In the CY 2026 OPPS/ASC proposed rule, we proposed to assign CPT codes ( printed page 53580) 0551U and 0568U status indicator “Q4” under OPPS.
Comment: We received one comment in support of our proposal. A commenter commended CMS's commitment to developing the basis of payment for these new clinical diagnostic laboratory tests with the addition of CPT codes 0551U and 0568U.
Response: We thank the commenter for their support. However, on October 1, 2025, the AMA CPT Editorial Panel deleted CPT code 0551U effective January 1, 2026. Therefore, for CY 2026, we will delete this code from this final rule with comment period and the January 2026 Update. However, we are finalizing our proposal to assign status indicator “Q4” to CPT code 0568U for CY 2026.
We refer readers to Addendum D1 of this final rule with comment period for the SI meanings for all codes reported under the OPPS. Addendum D1 is available via the internet on the CMS website.
29. PreciseBreast Test, CPT Code 0220U
AMA CPT Editorial Panel created CPT code 0220U on October 1, 2020 to describe a clinical diagnostic laboratory test that utilizes an Al-digital risk assessment methodology to measure protein-based biomarkers that are digitized to recapitulate the location, morphology and biological grade of a patient's invasive (ductal) breast cancer.
In the CY 2026 OPPS/ASC proposed rule, we proposed to continue assigning CPT code 0220U to status indicator “Q4” under OPPS.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter requested that CMS update its guidance on the laboratory date of service (DOS) policy for protein-based multianalyte algorithmic tests for cancer indications. Under the DOS regulations at § 414.510(b)(5), a clinical diagnostic laboratory test on a specimen collected during a hospital outpatient encounter can be billed by the performing laboratory if the test involves DNA, RNA or protein biomarkers or is classified as an Advanced Diagnostic Laboratory Test (ADLT). PreciseBreast® test (0220U) 1 is a cancer-related protein-based multi-analyte algorithmic test, however, because it is assigned a Proprietary Laboratory Analyses” (PLA) code by AMA CPT Editorial Panel, it does not meet the exclusion from the DOS policy under CMS's current interpretation. According to the commenter, they cannot bill Medicare for their PreciseBreast® test even though it is performed in their laboratory outside of the hospital. This has significantly limited access to the targeted clinical diagnostic information that the test provides to breast cancer patients.
The commenter requested that CMS update the laboratory DOS policy at § 414.510(b)(5) to clarify that PreciseBreast®, as a cancer-related protein-based MAAA, can be billed by the performing lab with a DOS of the date of test performance. PreciseBreast® should be added to CMS's list of “Laboratory Tests Subject to Exceptions to Laboratory DOS Policy Defined at § 414.510(b)(5))”, reassigned to Status Indicator “A”, and excluded from packaging in the outpatient setting.
Response: We thank the commenter for their input but note that the comment related to the Date of Service (DOS) policy is out of scope for the purposes of this OPPS/ASC final rule with comment period as there was no proposal to modify the DOS regulations in the CY 2026 OPPS/ASC proposed rule.
We also do not believe that PreciseBreast® test qualifies as an exception to the OPPS laboratory packaging policy based on criteria discussed in the Laboratory Related Services section.
Based on that information, we continue to believe that the assignment of status indicator “Q4” to CPT code 0220U is still appropriate and are finalizing without modification, our proposal to assign CPT code 0220U to status indicator “Q4” for CY 2026.
30. Screening DNA/RNA Test for Hepatitis C Virus, HCPCS Code G0567
In the April 2025 quarterly update, effective June 27, 2024, CMS created a new HCPCS code, G0567, to describe a new screening DNA/RNA test for Hepatitis C Virus. We assigned this code to status indicator “A” under OPPS indicating that this code would be paid separately under clinical laboratory fee schedule (CLFS), similar to other preventive screening tests that are excluded from the OPPS packaging policy for the laboratory tests.
In the CY 2026 OPPS/ASC proposed rule, we proposed to continue to assign HCPCS code G0567 to status indicator “A”.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter requested confirmation on the assignment of status indicator “A” where a commenter stated that such an assignment would be fully consistent with CMS precedent for other “screening” G-codes, such as G0472 (Hepatitis c antibody), G0475 (HIV antigen/antibody), G0476 (HPV co-testing), G0499 (Hep B screening)).
Response: We thank the commenter for their feedback and believe that the assignment of status indicator “A” continues to be appropriate for CY 2026. Therefore, we are finalizing our proposal, without modification to assign HCPCS code G0567 to status indicator “A” for CY 2026. We refer readers to OPPS Addendum D1 to this final rule with comment period for the status indicator definitions for all codes reported under the OPPS.
31. Laparoscopic Hernia Repair and Appendectomy, Procedures, CPT Codes 49650, 49651, and 44970 (APC 5342)
CPT codes 49650 ( Laparoscopy, surgical; repair initial inguinal hernia) and 49651 ( Laparoscopy, surgical; repair recurrent inguinal hernia) describe laparoscopic hernia repair procedures. CPT code 44970 ( Laparoscopy, surgical, appendectomy) describes the procedure for laparoscopic appendectomy. Using CY 2024 claims data, CPT codes 49650, 49651, and 44970 had geometric mean costs of $7,050.22, $7,173.25, $6,777.47, respectively, in the CY 2026 OPPS/ASC proposed rule. For CY 2026, we believed these were still appropriately assigned to APC 5361 (Level 1 Laparoscopy and Related Services), which had a proposed payment rate of $6,228.97. Therefore, we proposed to continue assigning CPT codes 49651, 49651, and 44970 to APC 5361.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: The HOP Panel recommended that we reassign CPT codes 49650, 49651, and 44970 to APC 5342 (Level 2 Abdominal/Peritoneal/Biliary and Related Procedures) based on clinical and resource similarity to procedure in that APC. A commenter disagreed with the HOP Panel's recommendation and instead requested that CMS finalize our proposal without modification due to potential disruption to the payment rate for APC 5361 if we were to remove the three procedures.
Response: We thank the commenter for their input. CPT codes 49650, 49651, and 44970 have updated GMCs of $6,902.69, $7,040.05, and $6,638.55 in the final rule data. While these are closer to the payment rate for APC 5342, after clinical review of these procedures, we did not find APC 5342 to be the clinically appropriate APC family for CPT codes 49650, 49651, and 44970. We ( printed page 53581) continue to believe that these procedures belong in the Laparoscopy and Related Procedures family and are appropriately paid based on the payment rate for APC 5361.
After consideration of the public comment we received, we are finalizing our proposal without modification to continue to assign CPT codes 49650, 49651, and 44970 to APC 5361 (Level 1 Laparoscopy and Related Services).
Table 62 shows the finalized status indicator and APC assignment for the procedure codes. We refer readers to Addendum B to this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
32. Medical 3D Printing, CPT Codes 0559T, 0561T (5734)
CPT codes 0559T ( Anatomic model 3D-printed from image data set(s); first individually prepared and processed component of an anatomic structure) and 0561T ( Anatomic guide 3D-printed and designed from image data set(s); first anatomic guide) were established in 2019 to describe medical 3D printing services. For the CY 2026 OPPS/ASC proposed rule, CPT code 0559T had a geometric mean of approximately $183 based on 12 single frequency claims and 16 total frequency claims, and CPT code 0561T had a geometric mean cost of approximately $255 based on 8 single frequency claims and 84 total frequency claims. For CY 2026, we proposed to continue to assign CPT codes 0559T and 0561T to APC 5733 (Level 3 Minor Procedures) and status indicator “Q1.”
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter requested that CMS assign CPT codes 0559T and 0561T to APC 5735 (Level 5 Minor Procedures) because the resources and supplies needed to provide these services are greater than the proposed payment rates. The commenter explained that the low utilization for both codes caused a lack of reliable claims data for ratesetting and urged us to assign the codes to a clinical APC with a higher payment rate to ensure patient access. Per the commenter, the 3D-printed models cost close to $3,000 per patient.
Response: Based on the geometric mean cost of both codes, we agree with the commenter that the proposed payment rate is not appropriate for CPT codes 0559T and 0561T, as the payment rate is significantly lower than the geometric mean cost of the codes. Based on the claims data available, we believe an assignment to APC 5734, a higher payment level within the same clinical APC, would align more closely with the geometric mean costs for both codes.
After consideration of the public comment we received, we are finalizing our proposal with modification to assign CPT codes 0559T and 0561T to APC 5734 and status indicator “Q1” as reflected in Table 63. As we do every year, we will re-evaluate the APC assignments for these codes in the next rulemaking cycle.
Neuro and Nerve Related Procedures
33. Continuous EEG Monitoring, CPT Codes 0956T, 0960T (APC 5117)
Continuous EEG monitoring involves an implantable device. The device is an electroencephalographic (EEG) recording and transmitting device implanted under the scalp. It is a prescription device indicated to acquire, transmit, and store EEGs continuously from patients between 18-75 years of age with drug-resistant epilepsy who are intolerant or not indicated for more conservative monitoring tools. The device is intended to aid a physician's remote assessment and monitoring of the indicated patient's condition.
The medical use of the data acquired by the continuous EEG monitoring system is to be performed under the direction and interpretation of a licensed medical professional. The device does not provide any diagnostic conclusions about the patient's condition.
The AMA CPT Editorial Board created five new Category III CPT codes to describe various procedures associated with a sub-scalp bilateral continuous ( printed page 53582) EEG monitoring system to assist in identifying seizure activity for patients with drug resistant epilepsy (DRE). Specifically, effective July 1, 2025, the three new CPT codes are:
- 0956T—Partial craniectomy, channel creation, and tunneling of electrode for sub-scalp implantation of an electrode array, receiver, and telemetry unit for continuous bilateral electroencephalography monitoring system, including imaging guidance
- 0957T—Revision of sub-scalp implanted electrode array, receiver, and telemetry unit for electrode, when required, including imaging guidance
- 0958T—Removal of sub-scalp implanted electrode array, receiver, and telemetry unit for continuous bilateral electroencephalography monitoring system, including imaging guidance
- 0959T—Removal or replacement of magnet from coil assembly that is connected to continuous bilateral electroencephalography monitoring system, including imaging guidance
- 0960T—Replacement of sub-scalp implanted electrode array, receiver, and telemetry unit with tunneling of electrode for continuous bilateral electroencephalography monitoring system, including imaging guidance
In the CY 2026 OPPS/ASC proposed rule, we proposed to assign CPT codes 0956T and 0960T to status indicator “S” and APC 1577 (New Technology—Level 40 ($20,001-$25,000)) with a proposed payment rate of $22,500.50. CPT code 0957T was assigned to status indicator “J1” and APC 5112 (Level 2 Musculoskeletal Procedures) with a proposed payment rate of $1,659.95. CPT code 0958T was assigned to status indicator “J1”, APC 5113 (Level 3 Musculoskeletal Procedures) with a proposed payment rate of $3,377.20. CPT code 0959T was assigned to status indicator “Q2” APC 5072 (Level 2 Excision/Biopsy/Incision and Drainage) with a proposed payment rate of $1,692.22.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: Several commenters requested that CMS reexamine and revise the reimbursement rates for CPT codes 0956T and 0960T because, according to the commenters, outpatient reimbursement for services associated with sub-scalp EEG monitoring is substantially below the actual resources required to deliver them.
A few commenters urged CMS to move CPT codes 0956T and 0960T to a higher-paying New Technology APC.
A commenter recommended that CMS assign both CPT codes to APC 1579 (New Technology—Level 42 ($30,001-$40,000)). Their analysis indicated that a similar procedure is the implantation of a cochlear implant. In evaluating the CY 2026 OPPS claims data, the procedure costs of implanting a cochlear implant (CPT code 69930) are $8,461. The commenter stated that the cost of the device is $25,000, so total estimated hospital costs of the procedure are $33,461. The commenter requested that CMS reassign CPT codes 0956T and 0960T to APC 1579.
Another commenter estimated that the total procedure costs for 0956T and 0960T exceed $35,000. Device costs are approximately $25,000. Surgical and facility costs for outpatient implantation procedures add approximately $10,000. The commenter stated that with the current proposed reimbursement falling $12,500 short of actual costs, a hospital would be unlikely to offer this service, and patients would not be able to access this service.
Response: Based on clinical similarity and resource homogeneity of the procedures described by CPT codes 0956T and 0960T to existing procedures assigned to the Level 7 Musculoskeletal Procedures APC, and based on input from our medical advisors, we are assigning CPT codes 0956T and 0960 to status indicator “J1” and APC 5117 (Level 7 Musculoskeletal Procedures) for CY 2026 as reflected in Table 64.
The final payment rates for the codes can be found in Addendum B to this final rule with comment period. In addition, we refer to Addendum D1 to this final rule with comment period for the status indicator (SI) meanings for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
34. Transcutaneous Magnetic Peripheral Nerve Stimulation CPT Codes 0766T and 0767T (APC 5722)
Transcutaneous Magnetic Peripheral Nerve Stimulation is intended to stimulate peripheral nerves for relief of chronic intractable pain, post‐traumatic pain, post‐surgical pain and/or for relief of chronic painful diabetic peripheral neuropathy in the lower extremities for patients 18 and older. CPT code 0766T ( Transcutaneous magnetic stimulation by focused low-frequency electromagnetic pulse, peripheral nerve, with identification and marking of the treatment location, including noninvasive electroneurographic localization (nerve conduction ( printed page 53583) localization), when performed; first nerve ) became effective January 1, 2023. For CY 2025, CPT code 0766T was reassigned to APC 5722 (Level 2 Diagnostic Tests and Related Services) with the status indicator “S” (Significant Procedures, Not Discounted when Multiple. Paid under OPPS; separate APC payment). There are no claims available for CPT code 0766T in CY 2024. For CY 2026, we proposed to continue assigning CPT code 0766T to APC 5722.
CPT code 0767T ( Transcutaneous magnetic stimulation by focused low-frequency electromagnetic pulse, peripheral nerve, with identification and marking of the treatment location, including noninvasive electroneurographic localization (nerve conduction localization), when performed; each additional nerve (list separately in addition to code for primary procedure)) became effective January 1, 2023 and assigned status indicator “N” (packaged). CPT code 0767T is an add-on code and we proposed to continue assignment of status indicator “N” for this code for CY 2026.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: Commenters requested CPT code 0766T be reassigned to APC 5724 (Level 4 Diagnostic Tests and Related Services), claiming that CPT code 0766T is not clinically similar and does not use comparable resources to other services in APC 5722. The commenters requested that CMS follows the 2025 HOP Panel recommendations to reassign 0766T to APC 5724 (Level 4 Diagnostic Tests and Related Services) and reassign the 0767T status indicator to “S” (Significant Procedures, Not Discounted when Multiple. Paid under OPPS; separate APC payment).
Response: We disagree that CPT code 0766T is comparable, clinically and in resource costs, to the services assigned to APC 5724. We continue to believe CPT code 0766T is more comparable, clinically and from a resource cost perspective, to CPT code 90867 (Therapeutic repetitive transcranial magnetic stimulation (tms) treatment; initial, including cortical mapping, motor threshold determination, delivery and management), and CPT code 90868 (Therapeutic repetitive transcranial magnetic stimulation (tms) treatment; subsequent delivery and management, per session), which are assigned to APC 5722 (Level 2 Diagnostic Tests and Related Services), because these services share similar magnetic stimulation and nerve localization processes.
We note that CPT code 0767T is an add-on code. Add-on codes are always performed in addition to the primary service or procedure and not reported as a stand-alone code. As specified under regulation 42 CFR 419.2(b)(18), add-on codes are generally packaged under the OPPS, and payment for the codes are bundled with the primary codes. Consequently, CPT code 0767T is not paid separately under the OPPS and its payment is packaged into payment for the primary code.
After consideration of the public comments we received, we are finalizing our proposed APC assignment and status indicators for CPT codes 0766T and 0767T without modification. Refer to Table 65 for the final OPPS APC and status indicator assignment for CPT codes 0766T and 0767T for CY 2026. We refer readers to Addendum B to this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
35. Ultrasound Guided Carpal Tunnel Release Procedure, CPT Code 64728 (APC 5431)
For CY 2026, the AMA CPT Editorial Board created a new Category I CPT code 64728 (Placeholder code 647XX) to describe carpal tunnel release procedures using ultrasound guidance. In the CY 2026 OPPS/ASC proposed rule, we proposed to assign CPT code 647XX to APC 5431 (Level 1 Nerve Procedures) with status indicator “J1” and a proposed payment rate of $1,999.82.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter believes overall hospital resources for this service can be approximated using other similar procedures—for example, endoscopic carpal tunnel release (CPT code 29848 ( Endoscopy, wrist, surgical, with release of transverse ligament)) and ( printed page 53584) open carpal tunnel release (CPT code 64721 ( Neuroplasty and/or transposition; median nerve at carpal tunnel))—and adding the single-use device cost of $1,099, plus the $140 cost of intraprocedure ultrasound to those other costs. Using the geometric mean costs for these services and adding the single-use device and intraprocedure ultrasound costs results in an estimated cost of between $3,048 and $3,454 for the new 647XX procedure.
Based on this information, the commenter suggested that CMS re-assign CPT code 64728 to APC 1524 (New Technology—Level 24 ($3001-$3500) with status indicator “S” and a proposed payment rate of $3,250.50 or alternatively re-assign it to APC 5432 (Level 2 Nerve Procedures) with status indicator “J1” and a proposed payment rate of $6,667.00 or to APC 5113 (Level 3 Musculoskeletal Procedures) with status indictor “J1” with a proposed payment rate of $3,377.20.
Response: Based on the estimated resource costs and clinical similarity of CPT code 64728 to other surgical procedures assigned to APC 5431, we continue to believe that the assignment of CPT code 64728 to APC 5431 is appropriate. Therefore, we are finalizing without modification our proposal to assign CPT code 64728 to APC 5431 for CY 2026.
We refer readers to Addendum B to this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
Neurostimulators
36. Creation of a Level 6 Neurostimulator APC
In prior rulemaking, some interested parties have requested that we create a Level 6 Neurostimulator and Related Procedures APC, due to their concerns around clinical and resource cost similarity in the Level 5 Neurostimulator and Related Procedures APC. We most recently responded to this request in the CY 2025 OPPS/ASC final rule with comment period (89 FR 94064). We noted that we believed that the current 5 level APC structure for the Neurostimulator and Related Procedures series provided for an appropriate distribution of clinical and cost similarity at the different APC levels. As discussed in the CY 2021 OPPS/ASC final rule with comment period, we reiterate that the OPPS is a prospective payment system. We group procedures with similar clinical characteristics and resource costs into APCs and establish a payment rate that reflects the geometric mean of all services in the group even though the cost of any individual service within the APC may be higher or lower than the APC's geometric mean. As a result, in the OPPS, any individual procedure may potentially be paid more or less than the cost of the services because the payment rate is based on the geometric mean of the entire group of services in the APC. However, the impact of these payment differences should be mitigated when distributed across a large number of APCs (85 FR 85968).
While we continued to believe that a five-level structure for the Neurostimulator and Related Procedures APC series remains appropriate, we solicited comment from interested parties on the need for a Level 6 APC, given the clinical and estimated cost characteristics of the services currently assigned to the Level 5 APC and New Technology APC 1580 (New Technology—Level 43 ($40,001-$50,000)).
In summary, for the CY 2026 OPPS, we proposed to maintain the current 5 level structure for the Neurostimulator and Related Procedure APC series. We also solicited comments on potentially creating an additional Level 6 APC in the series.
See Table 66 for proposed CY 2026 SI and APC assignments for specific HCPCS codes in the series and Table 67 for the proposed CY 2026 Neurostimulator and Related Procedures APCs.
( printed page 53585)We received public comments on the proposal to maintain the 5 level APC structure. The following is a summary of the comments we received and our responses.
Comment: Some commenters supported the proposal to continue assigning CPT code 0266T/64XX5 ( Implantation or replacement of carotid sinus baroreflex activation device; total system (includes generator placement, unilateral or bilateral lead placement, intra-operative interrogation, programming, and repositioning, when performed)) to New Technology APC 1580. Commenters also supported maintaining assignment of CPT code 33276 (Insertion of phrenic nerve stimulator system (pulse generator and stimulating lead[s]), including vessel catheterization, all imaging guidance, and pulse generator initial analysis with diagnostic mode activation, when performed) to New Technology APC 1580.
Other commenters noted that CPT code 64568 ( Open implantation of cranial nerve (e.g., vagus nerve) neurostimulator electrode array and pulse generator) which has similar characteristics to 33276 and 0266T/64XX5 should similarly be placed in New Technology APC 1580, based on its geometric mean cost and that continuing to assign CPT code 64568 to the Level 5 Neurostimulator and Related Procedures APC would be an inconsistent treatment of the procedure code, relative to the others. Finally, a commenter suggested that a combined C-code be created to pair 64568 with C1827 ( Generator, neurostimulator (implantable), non-rechargeable, with implantable stimulation lead and external paired stimulation controller).
At the August 2025 HOP Panel meeting, the HOP panel made a recommendation to consider placement of CPT code 64568 to New Technology APC 1580.
Response: We appreciate the commenters' support for the proposed assignments of HCPCS codes 33276 and 0266T/64XX5 to New Technology APC 1580.
We agree that it is appropriate to assign CPT code 64568 to New Technology APC 1580 based on its geometric mean cost of $46,926.67 and apply a similar temporary New Technology APC assignment, as we have for the other neurostimulator procedure codes assigned to that APC. We will continue to monitor the claims data for the code as additional claims become available. Finally, we do not believe the creation of a C-code pairing CPT code 64568 and device code C1827 is necessary or appropriate, given a final placement for the primary procedure code to New Technology APC 1580 in this final rule with comment period.
Comment: Many commenters requested the creation of a Level 6 Neurostimulator and Related Procedures APC, with some requesting that specific codes or codes with similar costs be included in that Level 6 APC. We note that at the August 2025 HOP Panel meeting, the HOP panel made a recommendation to create a Level 6 Neurostimulator APC.
Commenters stated that while they appreciated the assignment of specific codes to New Technology APCs and that those assignments represent a viable short term solution, a Level 6 APC represents a more long term solution to commenter concerns, including those around procedure payment relative to costs. A commenter also noted that some of the factors supporting the creation of the Level 7 Musculoskeletal Procedures APC would also apply to this series, such as the bimodal distribution of cost and potential placement of procedures being removed from the IPO list. A commenter also noted that although a Level 6 Neurostimulator and Related Procedures APC would have relatively low claims volume, that there are already other comparable APCs with low volume in the Intraocular Procedures APC series.
A few commenters supported maintaining the current 5 level structure of the APC series. One of the commenters requested that CMS not make any changes to the APC series structure or any of its assignments until a more comprehensive review of the current APC family could be performed.
Response: We appreciate the thoughtful responses commenters have provided with regard to the Neurostimulator and Related Procedures APC series and our request for comments.
At this time, we believe the 5-level APC structure for the APC series remains appropriate and individual temporary New Technology APC assignments with respect to the high cost procedures in this evolving area resolve some of the concerns around procedure payment relative to cost. We will continue to monitor the claims data for these procedures as more information around their volume and estimated costs become available.
After consideration of public comments, we are assigning CPT code 64568 to New Technology APC 1580 and maintaining the 5-level APC structure for the Neurostimulator and ( printed page 53586) Related Procedures APC series in the CY 2026 OPPS.
See Table 68 for final CY 2026 SI and APC assignments for specific HCPCS codes in the series and Table 69 for the final CY 2026 Neurostimulator and Related Procedures APCs.
( printed page 53587)37. Neurostimulator and Related Procedures (APCs 5461 Through 5465)
In the CY 2021 OPPS/ASC final rule with comment period, we finalized a five-level APC structure for the Neurostimulator and Related Procedures series (85 FR 85968 through 85970). For a detailed discussion of the history of neurostimulators policy, we refer readers to the CY 2015, CY 2020, CY 2021, CY 2023, CY 2024, and CY 2025 OPPS/ASC final rules with comment period (79 FR 66807 through 66808; 84 FR 61162 through 6116, 85 FR 85968 through 85970; 87 FR 71869; 88 FR 81645 through 81658; 89 FR 94062 through 96045).
CPT Codes 61885 and 64590
Effective January 1, 1982, The AMA CPT Editorial Board created Category I CPT code 61885 (Insertion or replacement of cranial neurostimulator pulse generator or receiver, direct or inductive coupling; with connection to a single electrode array) and CPT code 64590 (Insertion or replacement of peripheral, sacral, or gastric neurostimulator pulse generator or receiver, requiring pocket creation and connection between electrode array and pulse generator or receiver).
Based on the estimated resource costs and clinical similarity of HCPCS code 61885 to other procedures assigned to APC 5465 and because the geometric mean cost for that procedure aligned with the geometric mean cost of APC 5465, we proposed to reassign CPT code 61885 from APC 5464 (Level 4 Neurostimulator and Related Procedures) with a proposed payment rate of $20,126.69 for CY 2026 to APC 5465 (Level 5 Neurostimulator and Related Procedures) with a proposed payment rate of $31,751.65 for CY 2026. We also proposed to continue assigning CPT code 64590 to APC 5464 (Level 4 Neurostimulator and Related Procedures).
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: Some commenters supported these proposals and other commenters were concerned that since CMS was proposing to reassign CPT code 61885 from APC 5464 to APC 5465, the OPPS proposed payment rate for APC 5464 will decline significantly, when calculated both with and without the comprehensive APC (C-APC) methodology.
A commenter stated that the decrease in the payment rate for services remaining in APC 5464 will limit access for Medicare beneficiaries to important therapies reported under CPT code 64590 and other codes in that APC.
Other commenters requested that CMS utilize the adjustment authority at section 1833(t)(2)(E) of the Act to ensure equitable payments under the OPPS and calculate the OPPS payment rates for APC 5464 with and without the C-APC methodology using a geometric mean cost that phases in the reassignment of 61855.
During the 4-year phase-in, the payment rate calculation for APC 5464 would include a declining share of the mean cost of 61855 according to the following schedule:
- 2026—75%.
- 2027—50%.
- 2028—25%.
- 2029—0%.
The commenters requested that CMS apply this adjustment only to APC 5464; the calculation of the payment rate for APC 5465 should not be affected by this adjustment.
Response: Under the OPPS, we use the latest claims data to set the annual payment rates. For this final rule with comment period, the OPPS payment rates are based on claims submitted between January 1, 2024, and December 31, 2024, processed through June 30, 2025. Based on our evaluation of the claims data, the geometric mean cost for CPT code 61885 is $31,169.28 based on 3,008 single claims (out of 3,030 total claims), which is much more consistent with the geometric mean cost for APC 5465 (geometric mean cost of $31,767.96) than APC 5464 (geometric mean cost of $19,972.38).
In addition, the geometric mean costs of HCPCS codes that are assigned to APC 5464 are in line with the geometric mean cost for APC 5464. For instance, the geometric mean cost of CPT code 64590 which has over 95 percent of the claims volume in APC 5464, is $20,065.44 based on 11,062 single claims (out of 11,254 total claims) which is comparable with the geometric mean cost of $$19,972.38 for APC 5464.
In summary, after consideration of the public comments, we are finalizing without modification our proposal to assign CPT code 61885 to APC 5465 for CY 2026.
CPT code 61891 ( Responsive neurostimulation (RNS)) is an epilepsy treatment that uses an implanted device to help prevent seizures before they begin, similar to how a pacemaker detects and treats abnormal heart rhythms. With RNS, surgeons implant a small battery-powered device called a ( printed page 53588) neurostimulator in the patient's skull. The neurostimulator is connected to thin wires, which the surgeon places in the area or areas of the brain where the patient's seizures originate. The neurostimulator monitors the brain's electrical activity, and when activity that could lead to a seizure is detected, it delivers a pulse of electrical stimulation that may stop the seizure before it begins. The neurostimulator's battery generally lasts about 11 years. The neurostimulator is surgically replaced on an outpatient basis when the battery is at end of service.
The RNS procedures are described by CPT codes: 61889, 61891 and 61892.
- 61889—Insertion of skull-mounted cranial neurostimulator pulse generator or receiver, including craniectomy or craniotomy, when performed, with direct or inductive coupling, with connection to depth and/or cortical strip electrode array(s).
- 61891—Revision or replacement of skull-mounted cranial neurostimulator pulse generator or receiver with connection to depth and/or cortical strip electrode array(s).
- 61892—Removal of skull-mounted cranial neurostimulator pulse generator or receiver with cranioplasty, when performed.
In the CY 2026 OPPS/ASC proposed rule, we proposed to continue assigning CPT code 61889 to status indicator “C” since this procedure is only performed in the inpatient setting. We proposed to continue assigning CPT code 61891 to status “J1”, APC 5464 (Level 4 Neurostimulator and Related Procedures) with a proposed payment rate of $20,126.69 and we proposed to continue assigning CPT code 61892 to status indicator “J1”, APC 5113 (Level 3 Musculoskeletal Procedures) with a proposed payment rate of $3,377.20.
At the August 25, 2025, HOP Panel Meeting, a presenter advised the Panel to request that CMS reassign CPT code 61891 to APC 5465 (Level 5 Neurostimulator and Related Procedures). Based on the information presented at the meeting, the Panel recommended that CMS reassign CPT code 61891 to APC 5465 for CY 2026.
Comment: Commenters disagreed with CMS' assignment of APC 5464 and requested that CMS reassign CPT code 61891 to APC 5465. The commenters cited the inadequacy of APC 5464 from a cost and payment perspective, the fact that the predecessor CPT code 61886 was assigned to APC 5465, and that revision procedures are extremely rare.
A few commenters stated that while they understood CMS' concern about overpaying for a revision procedure that does not involve replacing the neurostimulator, the likelihood of this occurring is extremely rare. One of the commenters noted that based on internal data collected by NeuroPace, the company that manufactures the RNS System, it is less than one case per year across all payers.
Several commenters also noted that in their own practices, they have never performed a revision to the neurostimulator but have performed a number of replacements.
Another commenter stated that the current APC assignment for CPT code 61891 does not adequately reflect hospital costs, nor does it provide adequate outpatient payment for the service.
A few commenters stated that CY 2024 claims data published with the CY 2026 OPPS/ASC proposed rule demonstrates that the geometric mean cost (GMC) of CPT code 61891 is more appropriately aligned with the GMC of APC 5465. The commenter noted that per the Cost Statistics file, CPT code 61891 has a geometric mean cost (GMC) of $32,487. This is significantly higher than the GMC of current APC 5464 ($20,440) and the proposed payment rate of APC 5464 ($20,127). If finalized, hospitals will incur a loss of over $12,000 per procedure in CY 2026 for this procedure.
Some commenters also pointed out that assigning CPT code 61891 to APC 5465 is consistent with the APC assignment for the predecessor CPT code 61886—Insertion or replacement of cranial neurostimulator pulse generator or receiver, direct or inductive coupling; with connection to two or more electrode arrays. Prior to the implementation of CPT code 61891 in January 2024, the replacement of a skull-mounted cranial neurostimulator was reported with CPT code 61886 and assigned to APC 5465. CPT code 61891 was created to differentiate the services associated with skull-mounted cranial neurostimulators and cranial neurostimulators implanted in the chest (for example, CPT code 61886).
Another commenter acknowledged that the volume of procedures coded with CPT code 61891 remains low. However, a further reduction in payment has the potential to create access issues for the vulnerable patient population of patients with intractable epilepsy in whom these neurostimulator are utilized.
Response: We agree with the commenters and the HOP Panel that CPT code 61891 should be reassigned to APC 5465.
After consideration of the public comments we received, we are assigning CPT code 61891 to APC 5465 for CY 2026. We refer readers to Addendum B to this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
CPT Code 0786T
Effective January 1, 2024, the CPT Editorial Panel separated integrated from non-integrated ( i.e., traditional) sacral neurostimulator procedure by establishing a new CPT code, 0786T to report procedures using integrated sacral neurostimulator devices, while CPT code 64590 was updated to reflect the use of traditional technology:
- 0786T: Insertion or replacement of percutaneous electrode array, sacral, with integrated neurostimulator, including imaging guidance, when performed.
- 64590: Insertion or replacement of peripheral, sacral, or gastric neurostimulator pulse generator or receiver, requiring pocket creation and connection between electrode array and pulse generator or receiver.
In the CY 2026 OPPS/ASC proposed rule, we proposed to continue assigning CPT code 0786T to status indicator “E1” to indicate that it is still pending the FDA approval. However, this service received FDA approval on June 17, 2025.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: At the August 25, 2025 HOP Panel Meeting, the presenter advised the panel to request that CMS assign CPT code 0786T to APC 5464 (Level 4 Neurostimulator and Related Procedures). The HOP Panel agreed with the presenter and made that recommendation.
A commenter stated CPT code 0786T should not map to the same APC (APC 5464) as the traditional peripheral and sacral nerve stimulation devices that require lead insertion and pocket formation for the insertion of an implanted neurostimulator (INS). The traditional approach for a neurostimulator implant is reported with CPT code 64561 ( Percutaneous implantation of neurostimulator electrode array; sacral nerve (transforaminal placement) including image guidance, if performed) or 64555 ( Percutaneous implantation of neurostimulator electrode array; peripheral nerve (excludes sacral nerve)) for a percutaneous lead implant. These codes define the trial lead and permanent lead insertion. CPT code (64590 Insertion or replacement of peripheral, sacral, or gastric ( printed page 53589) neurostimulator pulse. generator or receiver, requiring pocket creation and connection between electrode array and pulse generator or receiver is used to report the insertion of a neurostimulator pulse generator or receiver and requires the creation of a pocket and the connection between the electrode array and the pulse generator ). This does not occur with integrated devices. As a predicate, CPT code 64596 (Insertion or replacement of percutaneous electrode array, peripheral nerve, with integrated neurostimulator, including imaging guidance, when performed; initial electrode array) is used to report the permanent placement of an integrated system that includes the contacts and the receiver on the other end of the lead and maps to APC 5463 (Level 3 Neurostimulator and Related Procedures). Therefore, the commenter recommended assignment of CPT codes 0786T, to either APC 5462 (Level 2 Neurostimulator and Related Procedures) or APC 5463 as either APC is a more accurate assignment given the cost and resources required to perform the procedure with an integrated device.
Another commenter recommended that we assign CPT code 0786T to APC 5462. CMS has assigned CPT code 0587T, describing the percutaneous implantation of an integrated single-device neurostimulation system for bladder dysfunction targeting the posterior tibial nerve, to APC 5462. The commenter believed that assignment of CPT code 0786T to the same APC as CPT code 0587T (APC 5462) would be much more clinically coherent than assignment to APC 5464, because both procedures involve percutaneous implantation of integrated neurostimulators for the treatment of bladder dysfunction with the only difference being the nerve target (sacral vs. posterior tibial nerve).
Another commenter requested that CMS assign CPT code 0786T to APC 5464 with a status indicator of “J1” based on clinical and resource homogeneity. The commenter believes that assignment to APC 5464 creates clinical alignment with other urinary urge incontinence (UUI) procedures, including both integrated and non-integrated systems.
Response: We thank the commenters for their input. We agree with one of the commenters that APC 5463 is the most appropriate assignment based on the cost and resources required to perform the procedure with an integrated device. Therefore, we are reassigning CPT code 0786T from status indicator “E1” to status indicator “J1”, APC 5463 for CY 2026.
We refer readers to Addendum B to this final rule with comment period for the payment rates for all codes reportable under the OPPS. We also refer readers to Addendum D1 to this final rule with comment period for the SI meanings for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
CPT Codes 0817T and 0988T
For CY 2024, the CPT Editorial Panel established four new Category III CPT codes, specifically, CPT codes 0816T, 0817T, 0818T, and 0819T to describe integrated neurostimulation services for bladder dysfunction, effective January 1, 2024.
For CY 2026, we proposed to continue assigning CPT code 0817T— Open insertion or replacement of integrated neurostimulation system for bladder dysfunction including electrode(s) (e.g., array or leadless), and pulse generator or receiver, including analysis, programming, and imaging guidance, when performed, posterior tibial nerve; subfascial, to APC 5464 with status indicator “J1”.
For CY 2026, CPT Editorial Panel created new Category III CPT code 0988T (placeholder code X400T)— Open insertion or replacement of integrated neurostimulation system for bladder dysfunction including electrode(s) (e.g., array or leadless), and pulse generator or receiver, including analysis, programming, and imaging guidance, when performed, posterior tibial nerve; subcutaneous and subfascial, effective January 1, 2026.
In the CY 2026 OPPS/ASC proposed rule, we proposed to assign it to APC 5464 with status indicator “J1”.
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: A commenter recommended assigning CPT codes 0988T and 0817T to either APC 5462 or APC 5463 as either APC is a more accurate assignment given the cost and resources required to perform the procedure with an integrated device.
Another commenter was concerned that the proposed 2026 OPPS payment rate for CPT 0817T is significantly lower than the 2025 rate.
Response: In the CY 2026 OPPS/ASC proposed rule, we inadvertently listed CPT code X400T as receiving FDA-approval even though the Coloplast's implantable tibial nerve stimulator that is described by this code is still pending FDA approval. Therefore, in the Final Rule, we are changing the status indicator for CPT code 0988T to status indicator “E1” Not covered by any Medicare outpatient benefit category; Statutorily excluded by Medicare; Not reasonable and necessary; Not paid by Medicare when submitted on outpatient claims (any outpatient bill type)) for CY 2026 because the device is not yet FDA approved.
We thank the commenters for their feedback related to our proposal for CPT code 0817T and we agree with one of the comments that it is appropriately placed in APC 5464 based on resource cost and clinical homogeneity to other similar codes in that APC. We also believe that its geometric mean cost is in line with the geometric mean cost of APC 5464.
For the CY 2026, based on claims submitted between January 1, 2024, and December 31, 2024, processed through June 30, 2025, our analysis of the latest claims data for this final rule with comment period shows a geometric mean cost of approximately $21,783.06 for CPT code 0817T based on 93 single claims, which is comparable to the geometric mean cost of about $19,972.38 for APC 5464. Based on the data, we continue to believe that assignment to APC 5464 for CPT code 0817T is appropriate.
We refer readers to Addendum D1 to this final rule with comment period for the SI meanings for all codes reported under the OPPS. Addendum D1 is available via the internet on the CMS website.
38. New Technology Applications
a. Digital Mental Health Treatment (DMHT), HCPCS Code G0552 (APC 5012)
HCPCS code G0552 ( Supply of digital mental health treatment device and initial education and onboarding, per course of treatment that augments a behavioral therapy plan). became effective January 1, 2025, and describes digital mental health treatment devices. We proposed to assign HCPCS code G0552 to APC 5012 (Clinic Visits and Related services) and status indicator V (clinic or emergency department visit) for CY 2026.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: Commenters recommended that CMS not finalize the proposal to continue to assign HCPCS code G0552 to APC 5012 for CY 2026, stating that the proposed payment rate of approximately $134 would not cover the costs associated with furnishing the service. A commenter explained that CMS should not use CPT code 98975 ( printed page 53590) ( Remote therapeutic monitoring (e.g., therapy adherence, therapy response, digital therapeutic intervention); initial set-up and patient education on use of equipment) as a crosswalk code to assign HCPCS code G0552 to APC 5012, noting certain differences between the codes. For example, the commenter explained that HCPCS code G0552 treats a condition while a remote monitoring device is designed to monitor a patient's status at home related to treatment. As a result of these differences, the commenter noted that DMHT devices, like those described by HCPCS code G0552, are significantly more costly to design, develop, study, obtain clearance, and commercialize compared with remote monitoring devices.
Response: We thank the commenters for their input. The New Technology APC application for the service described by HCPCS code G0552 is currently under consideration. After careful review and discussion with our CMS medical officers and leadership, we will render a decision through the subregulatory process through which the New Technology APC placement was initially requested.
b. Leadless Pacemaker (WiSE CRT System), CPT Code 0515T (APC 5231)
CPT code 0515T ( Insertion of wireless cardiac stimulator for left ventricular pacing, including device interrogation and programming, and imaging supervision and interpretation, when performed; complete system (includes electrode and generator [transmitter and battery]) became effective January 1, 2019, and describes the insertion of a wireless cardiac stimulator for left ventricular pacing.
CPT code 0515T is currently assigned to APC 5231 (Level 1 ICD and Similar Procedures) and status indicator of J1 (Hospital Part B Services Paid Through a Comprehensive APC; Paid under OPPS).
For CY 2026, we proposed to continue assignment of CPT code 0515T to APC 5231 (Level 1 ICD and Similar Procedures) with a proposed payment of around $22,725. This code had 2 claims for ratesetting and a geometric mean cost (GMC) of $16,837.74.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter requested that CMS reassign CPT code 0515T to New Technology APC 1576—Level 39 ($15,001-$20,000) to ensure that there is adequate payment for the non-device costs as this code is tentatively approved for pass-through payment (device costs).
Response: The New Technology APC application is currently under consideration. After careful review and discussion with our CMS medical officers and leadership, we will render a decision through the sub-regulatory process through which the New Technology APC placement was initially requested. We refer the readers to section IV. of this final rule with comment period for a discussion of applications that have been submitted for pass-through payment.
c. Paired Vagal Nerve Stimulation (Vivistim® System), CPT Code 64568 (APC 5465)
The Vivistim® System is an implanted neurostimulator that is used to stimulate the vagal nerve for upper extremity motor deficits and motor function in chronic ischemic stroke patients with moderate to severe arm impairment. CPT code 64568 ( Open implantation of cranial nerve (e.g., vagus nerve) neurostimulator electrode array and pulse generator) is used to describe the implantation of the Vivistim® System.
CPT code 64568 is assigned to APC 5465 (Level 5 Neurostimulator and Related Procedures), status indicator of `J1' (Hospital Part B Services Paid Through a Comprehensive APC;) and has a geometric mean cost (GMC) of around $49,319 with 151 single frequency claims used for ratesetting under OPPS. We note that the Vivistim® System was granted transitional device pass-through status on January 1, 2023, that is set to expire December 31, 2025.
Comment: Many commenters shared their experience with the Vivistim® System (paired vagus nerve stimulation) noting the effectiveness of the system to restore motor function in patients living with long term disabilities after having a stroke. Many of the commenters expressed concern about the lack of reimbursement once the transitional pass-through payments expire. They encouraged CMS to create a Level 6 Neurostimulator and Related Procedures APC to ensure that there is adequate and sustainable reimbursement for this advanced therapy. Commenters indicate that the current reimbursement for CPT code 64568 will not adequately cover hospital costs, putting access for Medicare beneficiaries at risk.
A commenter provided three options to ensure that there is adequate reimbursement for this advanced neuromodulation. The commenter requested that CMS consider creating a Level 6 Neurostimulator APC that includes CPT code 64568, reassigning 64568 to New Technology APC 1580 (New Technology—Level 43 ($40,001-$50,000)) or creating a C-code through the pending New Technology APC application and assigning the C-code to New Technology APC 1581 (New Technology—Level 44 ($50,001-$60,000)).
Response: We appreciate the input from commenters. As stated earlier in section “36. Creation of a Level 6 Neurostimulator APC”, we are assigning the primary procedure code to New Technology APC 1580 (New Technology—Level 43 ($40,001-$50,000)) in this final rule with comment period. We refer readers to the above noted section for a full discussion of the comments and our responses regarding CPT code 64568.
39. Noncontact Near-Infrared (NIR) Spectroscopy, CPT 0640T (APC 5732)
Effective CY 2024, there are three codes that describe the service related to NIR spectroscopy: CPT codes 0640T ( Noncontact near-infrared spectroscopy (e.g., for measurement of deoxyhemoglobin, oxyhemoglobin, and ratio of tissue oxygenation), other than for screening for peripheral arterial disease, image acquisition, interpretation, and report; first anatomic site), 0859T ( Noncontact near-infrared spectroscopy (e.g., for measurement of deoxyhemoglobin, oxyhemoglobin, and ratio of tissue oxygenation), other than for screening for peripheral arterial disease, image acquisition, interpretation, and report; each additional anatomic site (list separately in addition to code for primary procedure)), and 0860T ( Noncontact near-infrared spectroscopy (e.g., for measurement of deoxyhemoglobin, oxyhemoglobin, and ratio of tissue oxygenation), for screening for peripheral arterial disease, including provocative maneuvers, image acquisition, interpretation, and report, one or both lower extremities). Only CPT code 0640T is currently separately paid under the OPPS. For CY 2026, we proposed to assign CPT code 0640T to APC 5732 (Level 2 Minor Procedures) and status indicator “S” with a payment rate of approximately $39.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter requested that CMS reassign CPT code 0640T to APC 5722 (Level 2 Diagnostic Tests and Related Services), which had a proposed payment rate of approximately $220 for CY 2026, based on a crosswalk to CPT code 0598T (Noncontact real-time fluorescence wound imaging, for bacterial presence, location, and load, per session; first anatomic site ( e.g., lower extremity)). The commentor noted ( printed page 53591) certain similarities between both codes, such as that CPT code 0640T and CPT code 0598T are both Category III CPT codes, diagnostic imaging services, and neither use contrast.
Response: We note that for CY 2026, OPPS payment rates are based on available CY 2024 claims data. Based on our analysis of the claims data for this final rule with comment period, we found a geometric mean cost of approximately $11 for CPT code 0640T based on 347 single frequency claims (out of 1,067 total claims). In contrast, we found a geometric mean cost of approximately $200 for CPT code 0598T based on 1974 single frequency claims (out of 4,063 total claims). Based on the data, the resource cost associated with noncontact real-time fluorescence imaging (CPT code 0598T), is significantly higher compared to noncontact near-infrared (NIR) spectroscopy (CPT code 0640T). We disagree that the resource costs for NIR spectroscopy is similar to noncontact real-time fluorescence imaging based on the claims data available.
After consideration of the public comment, we continue to believe that CPT code 0640T is appropriately assigned to APC 5732. Therefore, for CY 2026, we are finalizing our proposal to assign CPT code 0640T to APC 5732 as reflected in Table 70. The final CY 2026 OPPS payment rate for all the codes payable under the OPPS can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 to this final rule with comment period for the SI meanings for all codes reported under the OPPS. Addendum D1 is available via the internet on the CMS website.
Nuclear Medicine Tests
40. Nuclear Medicine Services: Single-Photon Emission Computed Tomography (SPECT) Studies, CPT 78803 (APC 5592)
CPT code 78803 ( Radiopharmaceutical localization of tumor, inflammatory process or distribution of radiopharmaceutical agent(s) (includes vascular flow and blood pool imaging, when performed); tomographic (spect), single area (e.g., head, neck, chest, pelvis), single day imaging) describes a SPECT scan to find and map a tumor, inflammatory process, or how a radioactive tracer is distributed in a single body area, like the head, chest, or pelvis. For the CY 2026 OPPS/ASC proposed rule, CPT code 78803 had a geometric mean cost of around $585 and we proposed to reassign the procedure from APC 5593 (Level 3 Nuclear Medicine and Related Services) with a proposed payment rate of around $1,323 to APC 5592 (Level 2 Nuclear Medicine and Related Services) with a proposed payment rate of around $559 and status indicator S (Procedure or service not subject to multiple procedure discounting).
Comment: Commenters objected to the reassignment of CPT code 78803 to APC 5592 and requested that CMS not finalize the proposal but rather maintain the current placement in APC 5593. These commenters stated that the significant payment decrease of 57 percent resulting from the reassignment would limit patient access, affect patient care, and restrict hospitals from offering the test. Most commenters referenced the fact that costs were pulled out of 78803 last year when CMS finalized its policy to separately pay for certain radiopharmaceuticals and stated that physicians and hospitals needed time to properly account for resources and inputs associated with 78803 for services that do not use high-cost radiopharmaceuticals. These commenters requested that CMS collect several years of geometric mean data before reassigning CPT code 78803 from its current APC 5593. A commenter stated that CPT code 78803 should remain in APC 5593 because it is used to report SPECT (not planar) imaging, and other SPECT procedures are assigned to APC 5593.
Response: As acknowledged by commenters, last year we finalized a policy to unpackage diagnostic radiopharmaceuticals with per day costs above an annually adjusted threshold and pay separately for them. As a result of this shift from packaged payment to separate payment, the geometric mean cost for CPT 78803 understandably dropped, from around $1,137 for CY 2024 to around $588 for CY 2025. We appreciate commenters' concerns about providing additional time for hospitals to adjust and for additional geometric mean data to accumulate, however given that costs that were previously packaged into CPT code 78803 are now separately paid as a result of the policy we instituted last year, we think it is appropriate to reassign CPT 78803 to an APC that better aligns with 78803's CY 2026 geometric mean cost of around $585.
After consideration of the public comments we received, we are finalizing our policy without modification for CPT code 78803. We note that we review the APC assignments for all items and services paid under the OPPS on an annual basis. We will reevaluate the APC assignment for CPT code 78803 in the next rulemaking cycle.
Table 71 shows the finalized status indicator and APC assignment for this procedure code. We refer readers to Addendum B to this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
( printed page 53592)41. Nuclear Medicine Study (1 Area), CPT Code 78800 (APC 5591)
CPT code 78800 ( Radiopharmaceutical localization of tumor, inflammatory process or distribution of radiopharmaceutical agent(s) (includes vascular flow and blood pool imaging, when performed); planar, single area (e.g., head, neck, chest, pelvis), single day imaging) describes a planar (2D) nuclear medicine scan of a single area on a single day, used to locate tumors, inflammatory processes, or track the distribution of a radioactive tracer. It includes vascular flow and blood pool imaging if they are performed as part of the study. CPT code 78800 is assigned to APC 5591 (Level 1 Nuclear Medicine and Related Services) and status indicator S (Procedure or Service, Not Discounted When Multiple).
Comment: A commenter stated that CPT code 78800 is assigned to APC 5591 in proposed Addendum B but is assigned to APC 5573 in the proposed “Data Addendum B” and “2 Times Rule” files. The commenter believes that the code assignment to APC 5573 in the “Data Addendum B” and “2 Times Rule” files is incorrect, and that the correct APC assignment for CPT code 78800 is APC 5591.
Response: We agree that the correct APC assignment for CPT code 78800 is APC 5591. However, we note that the proposed 2026 “Data Addendum B” and “2 Times Rule” files (which are available via the internet on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices/cms-1834-p) both indicate that the code is assigned to APC 5591.
The final CY 2026 payment rates for this code can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 to this final rule with comment period for the SI definitions for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
Ophthalmology Related Services
42. Administration of Lacrimal Ophthalmic Insert Into Lacrimal Canaliculus, CPT Code 68841 (APC 5503)
HCPCS code J1096 ( Dexamethasone, lacrimal ophthalmic insert, 0.1 mg ), describes the drug Dextenza and is a drug indicated for “the treatment of ocular inflammation and pain following ophthalmic surgery” and for “the treatment of ocular itching associated with allergic conjunctivitis.” [25] Dextenza is administered via a natural opening in the eyelid (called the punctum) and delivers a tapered dose of dexamethasone to the ocular surface for up to 30 days. CPT code 68841 (Insertion of drug-eluting implant, including punctal dilation when performed, into lacrimal canaliculus, each) describes the insertion of the implant to administer Dextenza.
In the CY 2024 OPPS/ASC final rule with comment period (88 FR 81651 through 81653), we stated that based on the claims data available at the time, we continued to believe that the assignment of CPT code 68841 to APC 5503 (Level 3 Extraocular, Repair, and Plastic Eye Procedures) was appropriate based on the geometric mean costs. We also reiterated our reasoning that CPT code 68841 was appropriately assigned to an OPPS status indicator of “Q1” and ASC payment indicator of “N1.” We continued to believe that CPT code 68841 is mostly performed during ophthalmic surgeries, such as cataract surgeries. A status indicator “Q1,” indicating a conditionally packaged procedure, describes a HCPCS code where the payment is packaged when it is provided with a significant procedure but is separately paid when the service appears on the claim without a significant procedure. Because ASC services always include a surgical procedure, HCPCS codes that are conditionally packaged under the OPPS are generally packaged (payment indictor “N1”) under the ASC payment system. Although interested parties stated this is an independent surgical procedure and should not be packaged into the primary ophthalmic procedure in which the drug and drug administration are associated, we did not agree based on observed clinical patterns of how the drug is used. Based on CY 2023 claims data, out of over 7,000 total frequency claims, CPT code 68841 was used independently only about 2 percent of the time, meaning that the other 98 percent of the time CPT code 68841 had its payment packaged into the primary procedure with which it is associated. These data reinforced our belief that Dextenza and CPT code 68841 are not furnished independently of a surgical procedure and should be packaged into the primary ophthalmic procedure with which the drug and drug administration are associated. While we recognized that there are some claims that may only include CPT code 68841 without a primary ophthalmic surgery on the claim, we did not believe that this is a frequent occurrence based on our claims data and clinical use patterns; as previously mentioned, our claims data showed that only 2 percent of claims are performed independently of another primary procedure.
For CY 2025, we continued to assign CPT code 68841 to APC 5503 (Level 3 Extraocular, Repair, and Plastic Eye Procedures). We also maintained the OPPS status indicator “Q1” and an ASC payment indicator of “N1.”
For CY 2026, we proposed to continue to assign CPT code 68841 to APC 5503 (Level 3 Extraocular, Repair, and Plastic Eye Procedures). We also proposed to continue to assign CPT code to OPPS status indicator `Q1' and an ASC payment indicator of `N1'.
We note that CPT code 68841 does not have any single frequency claims ( printed page 53593) out of 2,930 total frequency claims. As stated above, this data once again reinforces our belief that Dextenza and CPT code 68841 are not furnished independently of a surgical procedure and should be packaged into the primary ophthalmic procedure with which the drug and drug administration are associated.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: Commenters requested that CMS revise the status indicator to `J1' (Hospital Part B Services Paid Through a Comprehensive APC; Paid under OPPS) to allow for separate ASC payment. The commenter stated that Dextenza replaces the use of self-administered eye drops, which can be difficult for some patients to administer and adhere to the regimen. The commenter also stated the lack of payment disproportionately and negatively affects the ASC setting where this procedure is done 80 percent of the time.
The commenter did not agree with CMS that the fact that CPT code 68841 was performed as a standalone procedure a small percentage of the time was adequate justification for assigning a `Q1' status indicator. The commenter stated that CMS is treating this procedure (CPT 68841) differently than the other procedures assigned to APC 5503 with a `J1' status indicator and that there are other comparable drug delivery procedures, specifically CPT codes 64415, 66020, 66030, and 0699T that also have J1 status indicators and receive separate payment in the ASC setting.
Response: We have long maintained that Dextenza is a drug that functions as a surgical supply and should be packaged under our packaging policy at § 419.2(b), which lists the types of items and services for which payment is packaged under the OPPS. Specifically, § 419.2(b)(16) includes drugs and biologicals that function as supplies when used in a surgical procedure as packaged costs. Historically, we have stated that we consider all items related to the surgical outcome and provided during the hospital stay in which the surgery is performed, including postsurgical pain management drugs, to be part of the surgery for purposes of our drug and biological surgical supply packaging policy (79 FR 66875). As such, the drug administration procedure, CPT code 68841, is also supporting the main ocular procedure being performed. CPT code 68841 should, therefore, be packaged as an intraoperative service under § 419.2(b)(14). We do not believe the listed HCPCS codes suggested by the commenter are analogous to CPT code 68841:
- 64415—Injection(s), anesthetic agent(s) and/or steroid; brachial plexus, including imaging guidance, when performed; assigned to APC 5443 (Level 3 Nerve Injections), status indicator of `T';
- 66020—Injection, anterior chamber of eye (separate procedure); air or liquid; assigned to APC 5491 (Level 1 Intraocular Procedures), status indicator of `J1';
- 66030—Injection, anterior chamber of eye (separate procedure); medication; assigned to APC 5491 (Level 1 Intraocular Procedures), status indicator of `J1'; and
- 0699T—Injection, posterior chamber of eye, medication; assigned to APC 5491 (Level 1 Intraocular Procedures), status indicator of `J1'.
We disagree with the commenter that lack of payment for the procedure in the ASC setting is a disincentive to use Dextenza. We note the number of claims continue to increase. We also note that HCPCS code J1096, which may be used to describe the drug, Dextenza, is a qualifying product for separate payment in both the OPPS and ASC under our policy to implement section 4135 of the CAA, 2023.
For the reasons discussed, we continue to believe that it is appropriate to assign CPT code 68841 to a status indicator “Q1,” indicating a conditionally packaged procedure, which describes a HCPCS code where the payment is packaged when it is provided with a significant procedure but is separately paid when the service appears on the claim without a significant procedure. Because ASC services always include a surgical procedure, HCPCS codes that are conditionally packaged under the OPPS are generally packaged (payment indictor “N1”) under the ASC payment system.
After consideration of the public comment, we are finalizing our proposal, without modification, to assign CPT code 68841 to APC 5503 with OPPS status indicator “Q1” (STV Packaged Codes) for CY 2026, which typically means there will be a packaged APC payment if this code is billed on the same claims as a HCPCS code assigned to status indictor “S,” “T,” or “V” (Clinic or Emergency Department Visit). In addition, based on the OPPS assignments, we are finalizing an ASC payment indicator of “N1” (Packaged service/item; no separate payment made) for CPT code 68841 for CY 2026.
For the final CY 2026 OPPS payment rates, we refer readers to OPPS Addendum B to this final rule with comment period. In addition, we refer readers to OPPS Addendum D1 to this final rule with comment period for the status indicator definitions for all codes reported under the OPPS. For the final CY 2026 ASC payment rates and payment indicators, we refer readers to Addendum AA and Addendum BB for the ASC payment rates, and Addendum DD1 for the ASC payment indicator and their definitions. The OPPS Addenda B and D1 and ASC Addenda AA, BB, and DD1 are available via the internet on the CMS website.
43. Comprehensive Aqueous Outflow Procedure
The comprehensive aqueous outflow procedure consists of more than 90 degrees of microcatheterization followed by more than 90 degrees of ab interno trabeculotomy used to treat patients with glaucoma.
We received public comments on this topic. The following is a summary of the comments we received and our responses.
Comment: A commenter stated that this service is more complex, utilizes more intraoperative time, and resource utilization. The other commenter stated that the current coding structure does not adequately reflect the differences in procedures currently reported with CPT code 66174 ( Transluminal dilation of aqueous outflow canal (e.g., canaloplasty); without retention of device or stent) and the comprehensive aqueous outflow procedure. The commenter indicated that these differences risk limiting hospitals from adopting the procedure and thus hindering patient access. The commenters requested that CMS create a C code and finalize a New Technology APC assignment that appropriately recognizes this unique procedure.
Response: We thank the commenters for their input. After review of the comment, we do not believe the commenter has provided sufficient explanation to justify the creation of a new C-code at this time.
Orthopedic Related Services
44. First Carpometacarpal Total Joint Arthroplasty, CPT Code 1003T (APC 5115)
The CPT Editorial Panel established CPT code 1003T to describe a total joint arthroplasty procedure involving the thumb effective January 1, 2026. The long descriptor for CPT code 1003T is as follows: Arthroplasty, first carpometacarpal joint, with distal ( printed page 53594) trapezial and proximal first metacarpal prosthetic replacement (e.g., first carpometacarpal total joint). Because the final CY 2026 CPT code numbers were not available when we published the CY 2026 OPPS/ASC proposed rule, the code was listed as placeholder code CPT code X459T in the OPPS Addendum B to the CY 2026 OPPS/ASC proposed rule.
For CY 2026, we proposed to assign CPT code 1003T to APC 5114 (Level 4 Musculoskeletal Procedures) and status indicator `J1' (Hospital Part B Services Paid Through a Comprehensive APC; Paid under OPPS.) with a proposed payment of around $7,533 based on clinical similarity to CPT code 26531 (Arthroplasty, metacarpophalangeal joint; with prosthetic implant, each joint).
At the August 25, 2025, HOP Panel Meeting, a presenter provided information to the Panel regarding new CPT code 1003T. The presenter advised the Panel to request that CMS reassign CPT code 1003T from APC 5114 to APC 5116. The HOP Panel agreed with the presenter and recommended that CMS reassign CPT code 1003T to APC 5116 (Level 6 Musculoskeletal Procedures), with a proposed payment of $18,056.80.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: Commenters noted that while other hand and wrist procedures are assigned to APC 5114, this procedure involves a total joint reconstruction with an implantation of a complex and costly prosthesis. The commenter believes that CPT 1003T is more similar to other arthroplasty procedures in APC 5116 suggesting CPT code 25446 (which describes a total wrist arthroplasty) and CPT code 25442 (which describes a distal ulna arthroplasty) as appropriate crosswalks. Commenters urged CMS to reassign CPT code 1003T to APC 5116 (Level 6 Musculoskeletal Procedures) as the HOP Panel recommended.
Response: We agree with the commenters that CPT code 1003T should not be assigned to APC 5114; however, we disagree that CPT code 1003T should be reassigned to APC 5116. After reviewing the comments and taking into consideration the HOP Panel recommendation, we believe that CPT code 1003T should be reassigned to APC 5115 crosswalking to CPT code 25441 (Arthroplasty with prosthetic replacement; distal radius). As we do every year, we will reevaluate the APC assignments for this code in the next rulemaking cycle. We remind hospitals that we review, on an annual basis, the APC assignments for all items and services paid under the OPPS.
In summary, after consideration of the public comments we received, we are finalizing the APC and status indicator assignment for CPT code 1003T with modification and assigning CPT code 1003T to APC 5115 (Level 5 Musculoskeletal Procedures) with a status indicator of J1. The final CY 2026 payment rates for this code can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 to this final rule with comment period for the SI definitions for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
Refer to Table 72 for code descriptor, APC assignment and status indicator assignment for CPT code 1003T for CY 2026.
45. Fusion of Foot Bones, CPT Code 28740 (APC 5114)
CPT code 28740 ( Arthrodesis, midtarsal or tarsometatarsal, single joint) describes the fusion of foot bones. In the CY 2026 OPPS/ASC proposed rule, we proposed to continue to assign CPT code 28740 to APC 5114 (Level 4 Musculoskeletal Procedures) with a status indicator of `J1' (Hospital Part B Services Paid Through a Comprehensive APC; Paid under OPPS) and proposed payment rate of $7,533.87.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter stated that the assignment of CPT code 28740 to APC 5114 may represent a violation of the 2 times rule, which may impede access to care for beneficiaries. The commenter recommended that CMS consider a volume threshold of 500 single major claims (or the existing greater or equal to 99 claims that constitute greater than or equal to the 2 percent criterion) as the standard for designating an item or service “significant” for purposes of the 2 times rule.
Response: We appreciate the commenter's request. We did not identify a 2-times rule violation for APC 5114. Our updated claims data found CPT code 28740 has fewer than 1,000 claims and does not meet the significance threshold for the 2-times rule evaluation for APC 5114. We believe that the current APC assignment of CPT code 28740 to APC 5114 continues to be appropriate for CY 2026.
However, we will take the commenter's suggestion on the 500-claim significance threshold into consideration for the future rulemaking.
In summary, after consideration of the public comments we received, we are finalizing our proposal without modification, to assign CPT code 28740 to APC 5114 (Level 4 Musculoskeletal Procedures). The final CY 2026 payment rates for this code can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 to this final rule with comment period for the status indicator definitions for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
Refer to Table 73 for code descriptor, APC assignment and status indicator ( printed page 53595) assignment CPT code 28740 for CY 2026.
Oncology Related Services
46. Radiation Oncology Treatment Delivery, CPT Codes 77402, 77407, 77412 (APCs 5621, 5622, and 5623)
At the September 2024 CPT Editorial Panel meeting, the Panel approved the revision of radiation therapy CPT codes 77402, 77407 and 77412 to establish a technique-agnostic family of codes and bundle imaging into the three CPT codes. In addition, Intensity Modulated Radiation Therapy (IMRT) treatment delivery codes 77385 and 77386 and CT guidance code 77014 were deleted and consolidated into this new code structure:
- Revised CPT code 77402 (Radiation treatment delivery; Level 1 (e.g., single electron field, multiple electron fields, or 2D photons), including imaging guidance, when performed) describes the delivery of a low-complexity form of radiation to a cancer or tumor and includes any imaging that is used during the session to ensure the radiation beam is accurately targeting the cancer. For the CY 2026 OPPS/ASC proposed rule, we proposed to continue to assign CPT code 77402 to APC 5621 (Level 1 Radiation Therapy), which has a payment rate of around $108, and status indicator S (Procedure or service not subject to multiple procedure discounting).
- Revised CPT code 77407 (Radiation treatment delivery; Level 2, single isocenter (e.g., 3D or IMRT), photons, including imaging guidance, when performed) describes the delivery of a more complex form of radiation using a single central point of radiation (isocenter) with high-energy photons which may be delivered through 3D conformal radiation therapy (3D-CRT) or Intensity-Modulated Radiation Therapy (IMRT) and includes any imaging that is used during the session. For the CY 2026 OPPS/ASC proposed rule, we proposed to continue to assign CPT code 77407 to APC 5622 (Level 2 Radiation Therapy), which has a payment rate of around $275, and status indicator S.
- Revised CPT code 77412 (Radiation treatment delivery; Level 3, multiple isocenters with photon therapy (e.g., 2D, 3D, or IMRT) OR a single isocenter photon therapy (e.g., 3D or IMRT) with active motion management, OR total skin electrons, OR mixed electron/photon field(s), including imaging guidance, when performed) describes the most complex radiation treatment delivery where multiple points of focus (isocenters) are used with photon therapy (2D, 3D, or IMRT) or a single isocenter is used but with active motion management or specialized techniques like total skin electrons or mixed electron/photon beams are performed and includes any imaging that is used during the session. For the CY 2026 OPPS/ASC proposed rule, we proposed to continue to assign CPT code 77412 to APC 5622 (Level 2 Radiation Therapy), which has a payment rate of around $275, and status indicator S.
A comment letter was submitted to the HOP Panel in advance of the August 25, 2025 HOP Panel Meeting that provided information about CPT codes 77407 and 77412. The letter advised the Panel to request that CMS reassign CPT code 77407 to APC 5623 and CPT code 77412 to APC 5624 for CY 2026. The HOP Panel had no recommendations.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: Many commenters expressed concern that CMS' proposed APC assignments for CPT codes 77407 and 77412 did not adequately reflect the procedures described by the revised codes and would not provide sufficient payment. Many commenters pointed out that these revised codes now included 3D conformal and IMRT treatment delivery that were previously described by deleted CPT codes 77385 and 77386. Several commenters suggested that for the purposes of rate setting, CMS should treat the three revised CPT codes as new codes. A commenter stated that “[a]lthough the CPT code numbers are the same for the remaining three CPT codes in the new treatment delivery family, they represent completely different services. For HOPPS rate setting purposes, CMS should treat 77402, 77407, and 77412 as new codes.”
Several commenters suggested alternative APC assignments, recommending that CPT code 77407 be reassigned from APC 5622 to APC 5623 (Level 3 Radiation Therapy), which has a CY 2026 proposed payment rate of around $600. These commenters reasoned that reassigning CPT code 77407 to APC 5623 would more accurately reflect the higher resource utilization associated with IMRT. On the same basis, roughly half of these commenters suggested that CPT code 77412 be reassigned from APC 5622 to APC 5623 and the other half suggested reassignment to APC 5624 (Level 4 Radiation Therapy) with a CY 2026 proposed payment rate of $716. Proponents of reassignment to APC 5624 stated that the higher APC assignment was warranted as CPT code 77412 is intended to account for the highest complexity therapy and such therapy requires additional time and incremental capital equipment resources to deliver multi-isocenter treatments and active motion management. Finally, a few commenters suggested that CPT code 47702 be reassigned from APC 5621 to APC 5622.
Response: We agree with commenters that the proposed APC assignments and the resulting payment rates for CPT codes 77407 and 77412 could more properly account for the revisions made to those codes and that the geometric mean costs of CPT codes 77385 ($568) and 77386 ($634) should be considered when assigning the appropriate APC for these codes. Accordingly, to establish a geometric mean cost for CPT codes 77407 and 77412 that better reflects the geometric mean costs under CPT codes 77385 and 77386, we have crosswalked the claims volume of CPT codes 77385, 77386, 77407, and 77412 in the following manner: For CPT code 77407, we are imputing all CY 2024 claims in CPT code 77407, the bottom 50 percent (with respect to total estimated cost) of single claims from CPT code 77412, and ( printed page 53596) all of the claims from CPT code 77385. For CPT code 77412, we are imputing the highest 50 percent (with respect to total estimated cost) of claims in 77412, and all of the claims from 77386. As a result of this crosswalking of claims, we are continuing to assign CPT code 77407 to APC 5622, which now has a geometric mean cost of around $397 and reassigning CPT code 77412 to APC 5623, which now has a geometric mean cost of around $569. We note that, while CPT code 77407 is assigned to the same APC it was assigned in the CY 2026 OPPS/ASC proposed rule, as a result of the crosswalk described above, the payment rate for that APC is much greater than it was in the CY 2026 OPPS/ASC proposed rule. With respect to 77402, we disagree with commenters that reassignment to APC 5622 is appropriate. Unlike CPT codes 77407 and 77412, CPT code 77402 was not revised to incorporate IMRT which is the basis for the crosswalk/APC reassignment for CPT codes 77407 and 77412 above.
We note that we received a few comments on our proposal that, due to the proposed deletion of radiation therapy G-codes (G6001-G6017), nonexcepted off-campus PBDs use the revised CPT codes 77402, 77407 and 77412 to continue our existing policy of paying the PFS-equivalent rate for radiation therapy to these departments. For a summary of and response to those comments, we refer readers to section III.G. of this final rule with comment period.
After consideration of the public comments we received, we are: (1) finalizing as proposed to continue to assign CPT code 77402 to APC 5621 and CPT code 77407 to APC 5622, while noting that the payment rate for APC 5622 is much greater than in the CY 2026 OPPS/ASC proposed rule as a result of the above code crosswalk; and (2) reassigning CPT code 77412 to APC 5623. We note that we review the APC assignments for all items and services paid under the OPPS on an annual basis. We will reevaluate the APC assignments for CPT codes 77402, 77407 and 77412 in the next rulemaking cycle.
Table 74 shows the finalized status indicator and APC assignment for these procedure codes. We refer readers to Addendum B to this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
47. Radiofrequency Ablation of Bone Tumors, CPT 20982 (APC 5116)
CPT code 20982 ( Ablation therapy for reduction or eradication of 1 or more bone tumors (e.g., metastasis) including adjacent soft tissue when involved by tumor extension, percutaneous, including imaging guidance when performed; radiofrequency) describes a primarily palliative procedure that reduces the size of bone tumors and addresses the pain from the tumors. For the CY 2026 OPPS/ASC proposed rule, CPT code 20982 had a geometric mean cost of around $18,375 and we proposed to continue to assign the procedure to APC 5115 (Level 5 Musculoskeletal Procedures), which has a proposed payment rate of around $13,254.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter requested that CMS reassign CPT code 20982 from APC 5115 to APC 5116 (Level 6 Musculoskeletal Procedures) with a payment rate of around $18,057. The commenter noted that according to the CY 2026 OPPS/ASC proposed rule cost statistics file, CPT code 20982 has a geometric mean cost of approximately $18,375, which exceeds the overall APC cost of $13,461 by nearly $5,000 or 36 percent. In contrast, the commenter pointed out that the overall cost of APC 5116 is $18,338, which very closely aligns with the cost of CPT code 20982. The commenter also noted that the cost of the bone tumor ablation procedure exceeds that of 10 procedures proposed for assignment to APC 5116 with claims data, including two of the three procedures with significant volume in the APC (described by CPT codes 22612 ( printed page 53597) and 27279), which have costs that are over $1,000 less than CPT code 20982. The third procedure with significant volume in APC 5116, described by CPT code 23472, has a geometric mean cost of $18,452, which is nearly identical to CPT code 20982.
Response: After reviewing the information provided by the commenter, the claims data and input from our CMS Medical Officers, we agree with the commenter that it is appropriate to reassign CPT code 20982 from APC 5115 to 5116 based on the resource costs related to CPT code 20982.
After consideration of the public comments we received, we are assigning CPT code 20982 to APC 5116 (Level 6 Musculoskeletal Procedures). Table 75 shows the finalized status indicator and APC assignment for this procedure code. We refer readers to Addendum B to this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
48. Scalp Cooling, CPT Codes 97007, 97008, and 97009 (APC 1517)
For CY 2025, the scalp cooling service is described by temporary CPT codes 0662T and 0663T. CPT code 0662T ( Scalp cooling, mechanical; initial measurement and calibration of cap) became effective on July 1, 2021, to describe initial measurement and calibration of a scalp cooling device for use during chemotherapy administration to prevent hair loss. According to Medicare's National Coverage Determination (NCD) policy, specifically, NCD 110.6 (Scalp Hypothermia During Chemotherapy to Prevent Hair Loss), the scalp cooling cap itself is classified as an incident to supply to a physician service, and would not be paid under the OPPS; however, interested parties have indicated that there are substantial resource costs of around $1,900 to $2,400 associated with calibrating and fitting the cap. CPT guidance states that CPT code 0662T should be billed once per chemotherapy session, which we interpret to mean once per course of chemotherapy. Therefore, if a course of chemotherapy involves, for example, 6 or 18 sessions, HOPDs should report CPT 0662T only once for those 6 or 18 therapy sessions. We note that CPT code 0663T (Scalp cooling, mechanical; placement of device, monitoring, and removal of device (List separately in addition to code for primary procedure)) describes an ancillary service and is assigned to status indicator “N” to indicate that OPPS payment is packaged into the payment for the primary service. We assigned CPT code 0662T to APC 1519 (New Technology—Level 19 ($1,701-$1,800)) with a payment rate of $1,750.50 and CPT code 0663T to status indicator “N” for CY 2025.
Beginning January 1, 2026, CPT codes 0662T and 0663T will be deleted and replaced with three new Category I CPT codes:
- 97007 (formerly placeholder 9XX01)—Mechanical scalp cooling, including individual cap supply with head measurement, fitting, and patient education.
- 97008 (formerly placeholder 9XX02)—Mechanical scalp cooling; including hair preparation, individual cap placement, therapy initiation, and precooling period).
- 97009 (formerly placeholder 9XX03)—Mechanical scalp cooling; provided after discontinuation of chemotherapy, each 30 minutes (List separately in addition to code for primary procedure).
In the CY 2026 OPPS/ASC proposed rule, we stated that we believed that CPT code 97007 most closely describes the primary service currently described by CPT code 0662T, while CPT codes 97008 and 97009 describe ancillary services for which payment would be packaged in the primary service. Therefore, for CY 2026, we proposed to assign CPT code 97007 to APC 1517 (New Technology—Level 17 ($1,501-$1,600)) with a $1,550.50 payment rate based on existing claims data for CPT code 0662T. We also proposed to assign status indicator “N” to CPT codes 97007 and 97009 to align with our current packaging policies generally, and specifically with regard to our current packaging of CPT code 0663T. Finally, we noted that because CPT is deleting CPT codes 0662T and 0663T, we would similarly delete the temporary codes under the OPPS/ASC payment systems.
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: A few commenters expressed concerns with the proposal to assign the new CPT codes to lower payment rates based on low claim volumes and requested that we withhold any payment reductions until the volume of claims increases. Other commenters supported the proposed APC assignment for CPT code 97007 based on claims data for CPT code 0662T but took issue with the proposal to package payment for CPT code 97008. These commenters requested that we assign CPT code 97008 to APC 1506 (New Technology—Level 6 ($401-$500)) and status indicator “S” instead of the proposed status indicator “N.” The commenter stated that there is no service to which CPT code 97008 can be packaged into, while also explaining that it is reported once for each chemotherapy session for an average of 5 to 7 sessions per patient. Per the commenters, scalp cooling is a ( printed page 53598) standalone treatment that requires specialized nursing resources and significant chair time, with an average cost of $500 outside of the chemotherapy service. The commenters emphasized that there is no service to which scalp cooling can be appropriately packaged, as it is not ancillary to any other procedure. They also highlighted certain concerns about patient access if the proposal to package payment for CPT code 97008 were to be finalized, noting that packaging this service would create financial barriers for Medicare beneficiaries and limit hospitals' ability to offer this treatment.
Response: With regard to the proposed APC assignment for CPT code 97007, we thank the commenters for their support to utilize claims data for CPT code 0662T and assign CPT code 97007 to APC 1517. Since the CY 2026 OPPS/ASC proposed rule was released, 15 additional CY 2024 claims for CPT code 0662T have been processed upon which to base the APC assignment for CPT code 97007. The revised geometric mean cost for CPT code 97007 is approximately $1,410. Therefore, we are finalizing our proposal to use available claims data for CPT code 0662T to finalize the APC assignment for CPT code 97007 to APC 1516 (New Technology—Level 16 ($1,401-$1,500) with a payment rate of $1,450.50 and status indicator “S.”
With regard to comments on the status indicator assignment for CPT code 97008, we disagree with comments asserting that there is no service to which scalp cooling can be appropriately packaged. Scalp cooling is always furnished in conjunction with chemotherapy administration, for the purpose of addressing a side effect of chemotherapy treatment. Unlike CPT code 97007 describing the fitting of the cap, which is performed on a date of service distinct from the chemotherapy administration and for which we proposed separate payment, CPT codes 97008 and 97009 would always be performed on the same date of service as the chemotherapy treatment. We acknowledge commenters' concerns regarding the resources required to furnish scalp cooling and potential impacts to patient access; however, we believe that the costs associated with the scalp cooling service are appropriately captured within the packaged payment for chemotherapy treatment. Therefore, we believe it is appropriate to package payment for CPT codes 97008 and 97009 with the primary chemotherapy service.
In summary, for CY 2026, we are finalizing our proposal to use available claims data for CPT code 0662T to finalize the APC assignment for CPT code 97007 to APC 1516 (New Technology—Level 16 ($1,401-$1,500) with a payment rate of $1,450.50 and status indicator “S.” We are also finalizing our proposals to assign CPT codes 97008 and 97009 to status indicator “N” for CY 2026. These changes are reflected in Table 76.
The final CY 2026 OPPS payment rate for all the codes payable under the OPPS can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 to this final rule with comment period for the SI meanings for all codes reported under the OPPS. Addendum D1 is available via the internet on the CMS website.
Respiratory Related Services and Procedures
49. Group Respiratory Therapy, HCPCS G0239 (APC 5732)
HCPCS code G0239 ( Therapeutic procedures to improve respiratory function or increase strength or endurance of respiratory muscles, two or more individuals (includes monitoring)) describes a medical service for two or more patients to improve their breathing and strengthen their respiratory muscles, conducted in a group setting under the supervision of a healthcare professional. For the CY 2026 OPPS/ASC proposed rule, HCPCS code G0239 had a geometric mean cost of around $43.00, and we proposed to continue to assign the procedure to APC 5732 (Level 2 Minor Procedures), which has a payment rate of around $39.00.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter expressed concern that the payment amount for APC 5732 dropped by 2.5 percent, from $39.96 in CY 2025 to $38.91 in CY 2026. The commenter requested that CMS provide further explanation for what is driving the decrease in payment for APC 5732 and, specifically, for HCPCS code G0239.
Response: There are a number of factors pertinent to the OPPS that may cause geometric mean costs to change from one year to the next. Some of these are a reflection of hospital behavior, and some of them are a reflection of fundamental characteristics of the OPPS, as defined in statute. For example, the OPPS payment rates are based on hospital cost report and claims data. However, hospital costs and charges change each year and this results in both changes to the cost-to- ( printed page 53599) charge ratios (CCRs) taken from the most currently available cost reports and also differences in the charges on the claims that are the basis of the calculation of the geometric mean costs on which OPPS rates are based. Similarly, hospitals adjust their mix of services from year to year by offering new services, and ceasing to furnish services, or changing the proportion of the various services they furnish, which has an impact on the CCRs that we derive from their cost reports. CMS cannot stabilize these hospital-driven fundamental inputs to the calculation of OPPS payment rates. Moreover, there are other essential elements of the OPPS which contribute to the changes in relative weights each year. These include, but are not limited to, reassignments of HCPCS codes to APCs to rectify 2 times violations as required by the law, to address the costs of new services, to address differences in hospitals' costs that may result from changes in medical practice, and to respond to public comments. In summary, after consideration of the public comment, we are finalizing our proposal without modification. Specifically, we are continuing to assign HCPCS code G0239 to APC 5732 (Level 2 Minor Procedures) with a payment rate of $38.16. We note that we review the APC assignments for all items and services paid under the OPPS on an annual basis.
Table 77 shows the finalized status indicator and APC assignment for this HCPCS code. We refer readers to Addendum B to this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
50. Insertion of Endobronchial Valves, CPT Code 31647 (APC 5155)
CPT code 31647 (Bronchoscopy, rigid or flexible, including fluoroscopic guidance, when performed; with balloon occlusion, when performed, assessment of air leak, airway sizing, and insertion of bronchial valve(s), initial lobe) describes a procedure used for conditions such as severe emphysema or persistent bronchopleural air leaks, where a device is placed to control airflow into a portion of the lung. For the CY 2026 OPPS/ASC proposed rule, CPT code 31647 had a geometric mean cost of $11,385.22 and we proposed to continue to assign the procedure to APC 5155 (Level 5 Airway Endoscopy) and status indicator J1, which has a payment rate of around $7,269.
We note that at the August 25, 2025, HOP Panel Meeting, a presentation was made requesting the reassignment of CPT code 31647 from APC 5155 to New Technology APC 1575 (New Technology Level 38). Based on the information presented at the meeting, the Panel recommended this reassignment.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: Commenters recommended that CMS reassign CPT code 31647 from APC 5155 to New Technology APC 1575 (New Technology Level 38).
The commenters noted that the cost of the procedure exceeds payment by approximately $4,000 and expressed concern that this discrepancy would result in patient access to the procedure being limited, particularly considering that the alternatives for severe emphysema patients involve much more invasive and expensive treatment options.
A commenter stated that the reassignment to a New Technology APC was necessary because (1) there is no higher-level APC in the Airway Endoscopy APC family and (2) there is no other clinical APC that would be an appropriate clinical and resource fit.
The commenter claimed that the geometric mean costs for CPT code 31647 have been stable and significantly more than the geometric mean cost for the APC into which it maps (APC 5155). While acknowledging that there is not a 2-times rule violation (given the number of single frequency claims for the procedure), the commenter noted that the GMC for CPT code 31647 is significantly above the GMC of the other procedures assigned to APC 5155 and more than double that for many procedures in the APC.
The commenter stated that they considered an alternative clinical APC but could not identify a clinically and resource cohesive clinical APC into which to move it.
The commenter stated that with no appropriate clinical APC to use as an alternative, CMS should assign the procedure to a new technology APC that more closely aligns with its GMC. The commenter concluded that APC 1575 is the appropriate new technology APC for the service given that the GMC for CPT code 31647 has been in the $11,000-$12,000 range for the past 3 plus years and is $11,385 in the 2024 claims data that CMS released.
Response: As recognized by one of the commenters, APC 5155 does not currently have a 2 times rule violation in the final rule data and APC 5155 appears to be the best clinical fit for CPT code 31647 of any existing clinical APC.
After consideration of the public comments we received and discussion and input from our Medical Officers, we are finalizing our proposal without modification to assign CPT code 31647 to APC 5155. We note that we review the APC assignments for all items and services paid under the OPPS on an annual basis. We will reevaluate the APC assignment for CPT code 31647 in the next rulemaking cycle. Table 78 shows the finalized status indicator and APC assignment for this procedure code. We refer readers to Addendum B to this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
( printed page 53600)51. Non-Invasive Gas Exchange and Cardiorespiratory Status (AGM 100), CPT Code 0893T (APC 5734)
Effective July 1, 2024, the AMA CPT Editorial Panel established CPT code 0893T ( Noninvasive assessment of blood oxygenation, gas exchange efficiency, and cardiorespiratory status, with physician or other qualified health care professional interpretation and report) to describe a non-invasive method for assessing a patient's blood oxygenation, gas exchange efficiency, and overall cardiorespiratory status using a special breathing device and monitor, with the resulting data interpreted and reported by a healthcare professional. We assigned CPT code 0893T to APC 5733 (Level 3 Minor Procedures) in the CY 2025 OPPS/ASC final rule. For the CY 2026 OPPS/ASC proposed rule, we did not have any claims for rate setting, so we proposed to continue to assign the procedure to APC 5733 (Level 3 Minor Procedures), which has a proposed payment rate of around $61.00 and status indicator Q1 (STV-Packaged Codes; Paid under OPPS).
We note that at the August 25, 2025, HOP Panel Meeting, a presentation was made requesting the reassignment to APC 5723 (Level 3 Diagnostic Tests and Related Services) for CPT code 0893T. Based on the information presented at the meeting, the Panel made no recommendation on the APC assignment for the code.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comments: Commenters requested that CPT code 0893T be reassigned from APC 5733 (Level 3 Minor Procedures) to APC 5723 (Level 3 Diagnostic Tests and Related Services) with a proposed payment rate of around $382.00. The commenters stated that the cost of the procedure is approximately $350 to $360 and therefore the proposed payment rate of $61.00 falls far short of adequate reimbursement and threatens patient access to the procedure. Some commenters additionally stated that the current APC assignment is inappropriate because there are no clinically similar procedures in that classification. These commenters stated that CPT 0893T involves exhaled gas analysis of oxygen, carbon dioxide, non-invasive blood oxygenation (PO2) and A-a gradient calculations and state that this procedure is comparable to CPT 94681 (oxygen uptake with exhaled gas analysis including carbon dioxide to assess lung function). According to these commenters, these two procedures utilize the same methodology and measure the physiological impairment parameters involving integrated cardiopulmonary assessment, supporting reclassification to APC 5723 (Level 3 Diagnostic Tests and Related Services).
Response: While we note that we do not have any claims for rate setting, based upon the input provided by commenters, we agree that CPT code 0893T should be reassigned to an APC that better reflects the costs of the procedure. However, we do not agree that the appropriate APC is 5723 (Level 3 Diagnostic Tests and Related Services). We are also not persuaded that the device is diagnostic in nature. While it provides additional information about a patient's oxygen level and other factors that may be helpful in formulating a diagnosis, it is not itself diagnosing a specific condition.
Accordingly, after consideration of the public comments we received and discussion and input from our Medical Officers, we have decided to reassign 0893T to APC 5734 (Level 4 Minor Procedures) with a payment amount of around $136. We note that we review the APC assignments for all items and services paid under the OPPS on an annual basis.
Table 79 shows the finalized status indicator and APC assignment for this procedure code. We refer readers to Addendum B to this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
( printed page 53601)SaaS Imaging
52. 3D Anatomical Segmentation Imaging Software Service, HCPCS Code C8001 (APC 5721)
In the January 2025 update, we established HCPCS code C8001 to describe the 3D anatomical segmentation imaging intended as software for preoperative surgical planning and as software for the intraoperative display of multi-dimensional digital images. We initially assigned HCPCS code C8001 to APC 5521 (Level 1 Imaging without Contrast) with a status indicator of `S' (Procedure or Service, Not Discounted When Multiple; Paid under OPPS; separate APC payment). After receiving feedback from external parties, we reassigned HCPCS code C8001 to APC 5721 (Level 1 Diagnostic Tests and Related Services) for the April 2025 quarterly update.
For CY 2026, we proposed to continue to assign HCPCS code C8001 to APC 5721 which has a proposed rate of around $132. We note that because this is a new service, we do not have any claims data.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter requested for HCPCS code C8001 to be reassigned from APC 5721 (Level 1 Diagnostic Tests and Related Services) to APC 5723 (Level 3 Diagnostic Tests and Related Services). The commenter cited the technical expertise required by the technologist and the additional hardware and software costs as justification for the higher-level APC assignment. The code describes 3D anatomical segmentation imaging for preoperative planning, data preparation, and transmission obtained from previous diagnostic computed tomographic or magnetic resonance examination of the same anatomy.
Response: After careful review of the request to reassign C8001 from APC 5721 to APC 5723, we do not believe it would be appropriate to reassign the APC for C8001 until we have claims data for this service. We will continue to monitor claims and utilization patterns for C8001 and may reconsider the APC assignment in future notice-and-comment rulemaking.
In summary, after consideration of the public comment we received, we are finalizing the APC assignment for HCPCS code C8001 without modification. The final CY 2026 payment rates for this code can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 to this final rule with comment period for the SI definitions for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website. Refer to Table 80 for code descriptor, APC assignment and status indicator assignment for HCPCS code C8001 for CY 2026.
53. 3D Image Generation Used in Surgical Planning and Navigation for Placement of Implants and Devices (BoneMRI), HCPCS Code G0566 (APC 5721)
In CY 2025, we established HCPCS code G0566 ( 3D radiodensity-value bone imaging, algorithm derived, from previous magnetic resonance examination of the same anatomy ) to describe the BoneMRI software as a service (Change Request 13993).[26] This service provides 3D radiodensity-value bone imaging from previous magnetic resonance images. For CY 2025, HCPCS code G0566 was assigned to APC 5721 (Level 1 Diagnostic Tests and Related Services) with the status indicator “S” (separate APC payment).
As HCPCS code G0566 is a new code in 2025, we have no claims data. For CY 2026, we proposed to assign HCPCS Code G0566 to APC 5721 (Level 1 Diagnostic Tests and Related Services).
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: Several commenters expressed concern with the low payment rate for this service which commenters stated may provide a financial barrier for providers to adopt this new technology into their practice and limit access. A commenter stated the proposed payment rate is not comparable to other SaaS technologies payment, such as EchoGo Heart Failure (CPT code 0932T) or HeartFlow FFRct (CPT code 75580), which ranges from about $316 to $879. The commenter stated the payment rate for BoneMRI does not reflect the value and resources for this service. The commenters recommended that HCPCS code be reassigned to APC 5723 (Level 3 Diagnostic Tests and Related Services) as that APC reflects the clinical value and cost of this service.
Response: In determining the appropriate APC placement for CPT/HCPCS codes, we rely on input from a variety of sources, including, but not limited to, review of the resource costs and clinical similarity of the service to existing procedures; input from CMS medical advisors; and information from interested specialty societies. We evaluated the recommendations, modeled the suggestions, analyzed the cost results of the suggested APC reassignments, and received additional input from our medical advisors. We note the SaaS codes that the commenter mentioned were assigned to their respective APCs based on similar clinical and resource similarity. While we recognize that there is not currently a one-to-one match to crosswalk to the new codes, we based the proposed APC assignments for HCPCS code G0566 on crosswalks to CPT/HCPCS codes that have similar service and resource ( printed page 53602) elements to the new codes. Based on our review of the service compared to other services assigned to the Diagnostic Tests and Related Services Series, we believe HCPCS code C8001 ( 3d anatomical segmentation imaging for preoperative planning, data preparation and transmission, obtained from previous diagnostic computed tomographic or magnetic resonance examination of the same anatomy) is an appropriate crosswalk code for HCPCS code G0566, which is currently assigned to APC 5721 (Level 1 Diagnostic Tests and Related Services).
After consideration of the public comments we received, we are finalizing our proposed APC assignment and status indicator for HCPCS code G0566 to APC 5721 without modification. Refer to Table 81 for the final OPPS APC and status indicator assignment for HCPCS code G0566 for CY 2026. We refer readers to Addendum B to this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
54. Augmentative Analysis of CT Imaging Data for Interstitial Lung Disease, CPT Code 0877T (APC 1508)
In the CY 2026 OPPS/ASC proposed rule, we proposed to continue assigning CPT code 0877T ( Augmentative analysis of chest computed tomography (ct) imaging data to provide categorical diagnostic subtype classification of interstitial lung disease; obtained without concurrent ct examination of any structure contained in previously acquired diagnostic imaging) to APC 1508 (New Technology—Level 8 ($601-$700)) with a proposed payment rate of $650.50 and status indicator “S”.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter requested that CMS remove CPT Code 0877T from the current OPPS rule and fee schedule because according to the commenter, this service is best categorized as an ambulatory service, not an outpatient service to be reviewed and priced under the OPPS. That is, there are currently no hospitals that can provide the type of service described in CPT code 0877T. For this reason, any claims billed for this service in the hospital setting were billed in error and are the result of miscoding.
They also stated that CPT code 0877T is currently designated as contractor-priced under the Medicare Physician Fee Schedule (PFS), meaning that the local Medicare contractors determine payment rates for the service within the PFS geographic areas in their jurisdiction. Since IMVARIA's IDTF is located in Texas, Novitas Solutions, Inc., the local Medicare Administrative Contractor (MAC), is solely responsible for evaluating coverage and payment for CPT 0877T.
Finally, they stated that the rate posted by OPPS for 0877T appears potentially based on an inaccurate and unrelated crosswalk to the published rate for CPT code 0721T, which bears no clinical, procedural, or technological relationship to the Fibresolve test or the commenter's area of focus.
Another commenter supported CMS' proposal and urged CMS to finalize the proposed payment structure for CPT code 0877T which enables facilities to bill for state-of-the-art ILD diagnostic services.
They also noted that it came to their attention that there was a comment submitted to CMS that claimed that there are currently no hospitals that can provide the type of service described in CPT code 0877T. For this reason, any claims billed for this service in the hospital setting were billed in error and are the result of miscoding. The commenter noted that their company is not operating as an Independent Diagnostic Testing Facility (IDTF) but rather entering into contractual agreements with facilities to offer e-Lung as a software-as-a-medical-device (SaMD) for which the hospitals will then submit claims as the provider. Therefore, another commenter's claim that existing claims were billed in error, and the result of miscoding is not accurate.
Response: Based on our review and input from our medical advisors, we believe that there may be other services that could be described by CPT code 0877T and therefore, we continue to believe that the current assignment of CPT code 0877T to APC 1508 with status indicator “S” is appropriate. Therefore, we are finalizing without modification our proposal to assign CPT code 0877T to APC 1508 for CY 2026. The final payment rates for the codes can be found in Addendum B to this final rule with comment period.
In addition, we refer to Addendum D1 to this final rule with comment period for the status indicator (SI) meanings for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
55. Noninvasive Arterial Plaque Analyses, CPT Code 0712T (5722)
Established in 2022, Category III CPT code 0712T is related to noninvasive arterial plaque analysis and describes the steps required for a software as a service (SaaS) imaging service that uses data from computed tomography angiography (CTA) to produce clinical information about arterial plaque for providers. See Table 82 for the CPT code, its long descriptor, and the proposed payment assignments.
( printed page 53603)For CY 2026, we proposed to assign CPT code 0712T to APC 5521 (Level 1 Imaging without Contrast), which has a proposed payment rate of approximately $89. The issue of payment for CPT code 0712T was brought to the Advisory Panel on Hospital Outpatient Payment (also known as HOP Panel) in August 2025 for CY 2026 rulemaking. At the 2025 HOP Panel, several presenters provided information to the Panel regarding 0712T and advised the Panel to request that CMS reassign 0712T to New Technology APC 1511 with a payment rate of $950.50. Based on the information presented, the HOP Panel did not make a recommendation to CMS to reassign 0712T to a different APC for CY 2026.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: Several commenters requested that CMS reassign CPT code 0712T to a New Technology APC 1511 with a payment rate of $950.50. Commenters detailed the procedural methodology and stated that the procedure involves segmentation, quantification, and assessment of high-risk biomarkers such as lipid-rich necrotic core plaque, which are critical for predicting strokes and other cardiovascular events. Several commenters recommended using CPT code 0625T ( Automated quantification and characterization of coronary atherosclerotic plaque to assess severity of coronary disease, using data from coronary computed tomographic angiography; computerized analysis of data from coronary computed tomographic angiography) as a crosswalk code for CPT code 0712T, as they believe that CPT code 0625T and CPT code 0712T resemble each other in methodology, clinical purpose, and resource demands. Additionally, commenters stated that the current APC assignment fails to cover the cost of performing noninvasive arterial plaque analysis from CTA data and that hospitals cannot absorb the financial loss associated with this service. Further, commenters stated that undervaluing the complexity and resource cost of CPT code 0712T would discourage adoption, limit data collection, and impede CMS's ability to monitor utilization and outcomes.
Response: After consideration of the public comments, additional review of the procedures, and input from our CMS Medical Officers, we do not agree with the recommended crosswalk code of CPT code 0625T. Based on our review of the clinical characteristics of the procedure and input from our medical advisors, we believe that CPT code 0712T is more similar clinically and in terms of resource requirements and procedure costs to the procedures assigned to APC 5722 (Level 2 Diagnostic Tests).
We remind hospitals that we review, on an annual basis, the APC assignments for all items and services paid under the OPPS and we will continue to monitor the claims data as they become available.
In summary, after consideration of the public comments we received, we are finalizing our proposal with modification and reassigning CPT code 0712T to APC 5722 (Level 2 Diagnostic tests). The final CY 2026 payment rates for these codes can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 to this final rule with comment period for the status indicator definitions for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
Refer to Table 83 for code descriptors, APC assignments and status indicator assignments for CPT code 0712T.
( printed page 53604)Urology Related Services
56. Aquabeam Waterjet Ablation Procedure, CPT Code 52597 (APC 5376)
CPT code 0421T ( Transurethral waterjet ablation of prostate, including control of post-operative bleeding, including ultrasound guidance, complete (vasectomy, meatotomy, cystourethroscopy, urethral calibration and/or dilation, and internal urethrotomy are included when performed) describes the Aquabeam waterjet ablation procedure. According to the manufacturer, Aquabeam is for treating lower urinary tract symptoms (LUTS) due to benign prostatic hyperplasia (BPH) by using a high-velocity water stream to ablate and remove tissue from enlarged prostates. Effective January 1, 2026, CPT code 0421T will be replaced with CPT code 52XX1 (52597), Transurethral robotic-assisted waterjet resection of prostate, including intraoperative planning, ultrasound guidance, control of postoperative bleeding, complete, including vasectomy, meatotomy, cystourethroscopy, urethral calibration and/or dilation, and internal urethrotomy, when performed. We will assign the underlying claims associated with HCPCS code 0421T to CPT code 52XX1 (52597).
For the CY 2026 OPPS/ASC proposed rule, we estimated the geometric mean cost for CPT code 0421T to be $10,342 based on 7,557 single claims and proposed to assign the service to APC 5376 (Level 6 Urology and Related Services), which has a geometric mean cost of $9,746.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: Several commenters requested CMS reassign CPT code 52597 (0421T) from APC 5376 (Level 6 Urology and Related Services) to APC 5377 (Level 7 Urology and Related Services) in the CY 2026 OPPS/ASC final rule with comment period. The commenters stated that CPT code 52597 (0421T) requires greater complexity and more resource than other CPT codes 55880 ( Ablation of malignant prostate tissue, transrectal, with high intensity-focused ultrasound (hifu), including ultrasound guidance) and 55873 ( Cryosurgical ablation of the prostate (includes ultrasonic guidance and monitoring)) that are currently in APC 5376 (Level 6 Urology and Related Services). The commenters stated that CPT code 52597 (0421T) is similar both clinically and in resource use to the procedures assigned to APC 5377 (Level 7 Urology and Related Services), citing CPT code 55882 ( Ablation of prostate tissue, transurethral, using thermal ultrasound, including magnetic resonance imaging guidance for, and monitoring of, tissue ablation; with insertion of transurethral ultrasound transducer for delivery of thermal ultrasound, including suprapubic tube placement and placement of an endorectal cooling device, when performed).
Response: We appreciate the commenter's recommendation regarding the APC assignment of CPT code 52597 (0421T) but we disagree that the clinical complexity and resource required for CPT code 52597 (0421T) is comparable to CPT code 55882. We believe that CPT code 52597 (0421T) fits more appropriately in APC 5376 rather than in APC 5377 based on resource cost and clinical similarity and to the procedures in APC 5376. We note that while both CPT code 52597 (0421T) and CPT code 55882 CPT provides imaging guidance, we believe that the use of magnetic resonance imaging guidance in CPT code 55882 necessitates greater resource than ultrasound guidance.
After consideration of the public comment we received, we are finalizing our proposal without modification to assign CPT code 52597 (0421T) to APC 5376. Refer to Table 84 for the final OPPS APC and status indicator assignment for CPT code 52597 (0421T) for CY 2026. We refer readers to Addendum B to this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
57. Enhanced Lithotripsy System, CPT Code 0991T (APC 5376)
CPT code 0991T ( Cystourethroscopy, with low-energy lithotripsy and acoustically actuated microspheres, including imaging) describes the lithotripsy with acoustically actuated microspheres in an approved investigational device exemption (IDE) study (NCT06942949). According to the manufacturer, this prospective, single arm study utilizes low pressure ultrasound to actuate proprietary microbubble to fragment urinary stones. The AMA CPT Editorial Panel established CPT code 0991T effective January 1, 2026.
As this is a new code in 2026, we have no claims data for CPT code 0991T. For CY 2026, we proposed to assign CPT Code 0991T with the status indicator “E1.”
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A few commenters stated that CPT code X432T has transitioned to CPT code 0991T, effective July 1, 2025 and that CMS incorrectly assigned status indicator “E1” to CPT code X432T in the CY 2026 OPPS/ASC proposed rule. Additionally, the commenters requested the assignment of CPT code 0991T to APC 5376 (Level 6 Urology and Related Services), based on the commenter's resource cost of ( printed page 53605) $10,131, and be designated as device intensive with a device offset of 44 percent.
Response: We note that according to the AMA, CPT code 0991T will replace CPT code X432T, effective January 1, 2026. While CPT code 0991T was released on the AMA website on July 1, 2025, the code is not effective until January 1, 2026. We also note that our status indicator assignment of “E1” for CPT code 0991T (X432T) was appropriate in the CY 2026 OPPS/ASC proposed rule. During our CY 2026 OPPS/ASC proposed rule assessment of CPT code X432T, we determined that the enhanced lithotripsy service involved a non-FDA approved device not excepted by any IDE status and therefore not payable under the OPPS/ASC because non-FDA approved devices are considered not reasonable and necessary under Section 1862(a)(1)(A) of the Act. The Evaluation of Enhanced Lithotripsy System (ELS) in the Treatment of Urinary Stones (NCT06942949) did not receive CMS category B IDE approval until August 1, 2025. Therefore, status indicator “E1” was an appropriate assignment for a service involving a non-FDA approved device. The definition of the OPPS status indicators can be found in Addendum D to this final rule with comment period via the internet on the CMS website.
In determining the appropriate APC placement for new codes, we rely on input from a variety of sources, including, but not limited to, review of the resource costs and clinical similarity of the service to existing procedures; input from CMS medical advisors; and information from interested specialty societies. We evaluated the recommendations, modeled the suggestions, analyzed the cost results of the suggested APC reassignments, and received additional input from our medical advisors. We agree with the commenter's request to assign CPT code 0991T (X432T) to APC 5376 (Level 6 Urology and Related Services). While we recognize that there is not currently a one-to-one match to crosswalk to the new codes, we based our APC assignment for CPT code 0991T (X432T) on crosswalks to CPT codes that have similar service and resource elements, as well as required staff, to the new codes. Based on our review of the enhanced lithotripsy service compared to other services assigned to the Urology and Related Services series, we believe HCPCS code C9761 (Cystourethroscopy, with ureteroscopy and/or pyeloscopy, with lithotripsy, and ureteral catheterization for steerable vacuum aspiration of the kidney, collecting system, ureter, bladder, and urethra if applicable (must use a steerable ureteral catheter)) is an appropriate crosswalk code for CPT code 0991T (X432T), which is currently assigned to APCs 5376 (Level 6 Urology and Related Services).
For this service's device offset, we refer readers to section IV.B. of this final rule with comment period.
After consideration of the public comment we received, we are finalizing our proposal with modification to assign CPT code 0991T (X432T) to APCs 5376 (Level 6 Urology and Related Services) with a status indicator of “J1”. Refer to Table 85 for the final OPPS APC and status indicator assignment for CPT code 0991T (X432T) for CY 2026. We refer readers to Addendum B to this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
58. Insertion of Permanent Urethral Stent, CPT Code 52282 (APC 5374)
CPT code 52282 ( Cystourethroscopy, with insertion of permanent urethral stent) describes the insertion of a permanent urethral stent using an endoscope procedure. CPT code 52282 became effective January 1, 1998. For the CY 2026 OPPS/ASC proposed rule, we estimated the geometric mean cost for CPT code 52282 to be approximately $3,382 based on 111 single claims and proposed to continue to assign the service to APC 5374 (Level 4 Urology and Related Services), which had a proposed geometric mean cost of $3,686.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: Several commenters requested the APC reassignment of CPT code 52282 to APC 5376 (Level 6 Urology and Related Services). The commenter stated that the current payment rate of APC 5374 (Level 4 Urology and Related Services) and the lack of device intensive status do not cover the cost of the stent and the procedure. The commenters stated CPT code 52282 is similar in device intensity to CPT code 53865. The commenters further stated that the inappropriate low payment creates a patient access issue which may push patients to other higher cost treatment options for both Medicare and beneficiaries. Furthermore, the low payment in ASCs for this treatment drives patients to HOPDs which can be more costly for the both Medicare and beneficiaries.
Another commenter stated that there has not been a permanent urethral stent device on the US market for the past decade. The commenter stated the 2024 claims showed CPT code 52282 had a mean device cost of $485, which is far below the price that will be available in the Q4 2025 and 2026. The commenter reported that about 33 percent of the 2024 claims are female patients, which indicate that these were incorrect ( printed page 53606) billings and that they should be removed from CY 2026 claims accounting. The commenter further stated that their analysis of the OPPS CPT 52282 claims revealed diagnosis codes do not correspond to treatment with a permanent urethral stent. The commenter also reported that their analysis of the CPT 52282 claims included device codes such as HCPCS code C2617 ( Stent, non-coronary, temporary, without delivery system) and HCPCS code C1758 ( Catheter, ureteral) that do not correspond to the treatment represented by CPT code 52282. The commenter suggested the following: (1) CMS exclude all CPT code 52282 claims for ratesetting, and device offset calculations, reassign CPT code 52282 to APC 5376, and use the manufacturer's price as a device offset or use CPT code 53865 that has a temporary urethral stent) until accurate claims are available; (2) Create a new device code for a permanent urethral stent with the suggested descriptor (Stent, prostatic, permanent with integrated transurethral delivery system); (3) Conduct annual reviews of device offset and review all surgical procedures; and (4) Coordinate with the FDA and AMA CPT Editorial Panel when devices are withdrawn from the market.
Response: We appreciate the commenters' suggestions regarding CPT code 52282. We rely on hospitals and providers to accurately report the use of HCPCS codes in accordance with their code descriptors, and CPT and CMS instructions, and to report services accurately on claims and charges and costs for the services on their Medicare hospital cost report. We defer to the clinicians and providers to provide the appropriate course of treatment, at the appropriate site of treatment. We will take the commenter's concerns regarding the US market availability of a permanent urethral stent and the claims surrounding CPT code 52282 into consideration for future rulemaking. We note that all comments requesting the reassignment of CPT code 52282 to APC 5376 were related to a device that has not received FDA approval or any exceptions such as an Investigational Device Exemptions (IDE) or PMA/510K Exemptions, and so those comments are out of the scope for this final rule with comment period.
For this service's device offset, we refer readers to section IV.B of this final rule with comment period.
After consideration of the public comments we received, we are finalizing our proposed APC assignment and status indicator for CPT code 52282 without modification. Refer to Table 86 for the final OPPS APC and status indicator assignment for CPT code 52282 for CY 2026. We refer readers to Addendum B to this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
59. Penile Prosthesis, CPT Code 54417 (APC 5377)
CPT code 54417 (Removal and replacement of non-inflatable (semi-rigid) or inflatable (self-contained) penile prosthesis through an infected field at the same operative session, including irrigation and debridement of infected tissue) describes the removal and replacement of penile implant and debridement of infected tissue.
For the CY 2026 OPPS/ASC proposed rule, we calculated the geometric mean for CPT code 54417 to be about $18,732, and we proposed to assign the service to APC 5377 (Level 7 Urology and Related Services), which has a geometric mean cost of $13,635. There were 11 single claims used to calculate the geometric mean cost for CPT code 54417.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter requested that CMS reassign CPT code 54417 to APC 5478. The commenter stated CPT code 54417 is similar to CPT codes 54411 (Removal and replacement of all components of a multi-component inflatable penile prosthesis through an infected field at the same operative session, including irrigation and debridement of infected tissue) and 54416 (Removal and replacement of non-inflatable (semi-rigid) or inflatable (self-contained) penile prosthesis at the same operative session) both clinically and in resource utilization. The commenter stated CPT code 54417 is a low frequency procedure, which is the expected utilization pattern, but its geometric mean has been increasing year over year. The commenter stated all three procedures involve the similar operative complexity of removing and replacing a penile prosthesis, while CPT codes 54411 and 54417 also includes irrigation and debridement of an infected tissue. The commenter stated that CPT code 54417 has the highest geometric mean cost in APC 5377, that exceeds CPT code 54416 and is closer to the geometric mean cost of APC 5378.
Response: We agree with the commenter that CPT code 54417 is similar clinically and in resource utilization to CPT codes 54411 and 54416. APC 5378 (Level 8 Urology and Related Services) is a more appropriate placement for CPT code 54417 due to the clinical work and resources utilized to furnish the removal and replacement of penile implant and/or debridement of infected tissue irrigation, which is clinically similar to CPT codes 54411 and 54416. We also agree with the commenters that the geometric mean cost of CPT code 54417 more closely matches the geometric mean cost of APC 5378 than APC 5377. Therefore, we will be reassigning CPT code 54417 to APC 5378 for CY 2026.
After consideration of the public comment we received, we are finalizing ( printed page 53607) a modification to our proposal to assign CPT code 54417 to APC 5378 (Level 8 Urology and Related Services) with a status indicator of “J1”. Refer to Table 87 for the final OPPS APC and status indicator assignment for CPT code 54417 for CY 2026. We refer readers to Addendum B to this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
60. Prostate Biopsy Codes, CPT Codes 55712, 55713, 55714, and 55715
For 2026, the AMA CPT Editorial Panel established four new codes to describe the various services related to report prostate biopsy to replace CPT code 55770 ( Biopsy, prostate; needle or punch, single or multiple, any approach) which will be retired December 31, 2025. The four new codes are effective January 1, 2026, and describe the biopsy of the prostate with various imaging guidance. The codes and their complete long descriptors are listed in Table 88. When determining the proposed status indicators and APC assignments for CY 2026, we reviewed the clinical and resource characteristics of the procedures, we considered input from our medical advisors and reviewed existing APC classifications to identify similar and closely related procedures.
We note that CPT codes 55712, 55713, 55714, and 55715 were listed as placeholder codes 5XX07, 5XX08, 5XX09, and 5XX10 respectively, in OPPS Addendum B and Addendum O that were released with the CY 2026 OPPS/ASC proposed rule. Because we had not received the final CPT code numbers from AMA for the new codes that would be effective January 1, 2026, in time for the publication of the CY 2026 OPPS/ASC proposed rule, we listed the new CPT codes with their respective placeholder codes in OPPS Addendum B and Addendum O.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: Some commenters sought clarification on the new prostate biopsy CPT codes with more detailed code descriptors for high-field and low-field MRI technologies, including in-office MRI systems.
Response: We thank the commenters for their input, but these comments are out of scope of this OPPS/ASC final rule with comment period. We note the CPT code descriptors are copyright property of the American Medical Association and we do not have the ability to modify CPT codes.
After consideration of the public comments we received, we are finalizing our proposal without modification for CPT codes 55712 through 55715. Table 88 shows the finalized status indicators and APC assignments for all the four prostate biopsy codes. The final CY 2026 payment rates for these codes can be found in Addendum B to this final rule with comment period. We also refer readers to Addendum D1 to this final rule with comment period for the SI meanings for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
( printed page 53608)61. Ureteroscopy, HCPCS Code C9761 (APC 5376)
The ureteroscopy procedure addresses kidney stones and may apply catheterization to vacuum aspirate the fragmented kidney stones. HCPCS code C9761 ( Cystourethroscopy, with ureteroscopy and/or, with lithotripsy, and ureteral catheterization for steerable vacuum aspiration of the kidney, collecting system, ureter, bladder, and urethra if applicable (must use a steerable ureteral catheter) became effective October 1, 2020.
For CY 2026, the OPPS payment rates were proposed based on available CY 2024 claims data. For the CY 2026 OPPS/ASC proposed rule, we found a total of 721 single frequency claims and a geometric mean cost of approximately $9,470 for HCPCS code C9761. For CY 2026, we proposed to continue to assign HCPCS code C9761 to APC 5376 (Level 6 Urology and Related Services) with a proposed payment rate of approximately $9,746.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: Commenters requested CMS provide coding guidance on the appropriate device billed under HCPCS code C9761.
Response: Comments on coding guidance are out of scope of this OPPS/ASC final rule with comment period. We note that if hospitals have questions about appropriate coding that they cannot resolve on their own, the appropriate first step would be to review the HCPCS codes or consult a Medicare Administrative Contractor (MAC).
62. Water Vapor Thermotherapy CPT Code 0582T (APC 5377)
CPT code 0582T ( Transurethral ablation of malignant prostate tissue by high-energy water vapor thermotherapy, including intraoperative imaging and needle guidance) describes an Category B IDE study where water vapor is used to ablate localized prostate cancer. It was designated a Category B IDE study (NCT05683691) on October 12, 2023. The purpose of this prospective, single arm study is to evaluate the safety and effectiveness of utilizing water vapor ablation delivered transurethrally in patients with immediate risk, localized prostate cancer.
For CY 2025, CPT code 0582T was assigned status indicator “E1.” For the CY 2026 OPPS/ASC proposed rule, we proposed to continue assigning CPT code 0582T with the status indicator “E1.”
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter requested CPT code 0582T be reassigned to APC 5378 (Level 8 Urology and Related Services) with a status indicator of J1 where other clinically and cost similar procedures are assigned. The commenter stated that this service is currently an approved Category B IDE study, Water Vapor Ablation for Localized Intermediate Risk Prostate Cancer (VAPOR 2) [G220303-NCT05683691]. Furthermore, the commenter stated the procedure cost ranges between $21,672 to $22,233, including the device cost of $16,415 and requested device intensive status and a device offset percentage of 74 percent.
Response: We thank the commenter for the notification that CPT code 0582T represents a Category B IDE study. While we recognize that there is not currently a one-to-one match to crosswalk to the new codes, we based the APC assignment on crosswalks to CPT codes that have similar service and resource elements to the new codes. Based on our review of the service compared to other services assigned to the Urology and Related Services APC series, we believe CPT code 55882 ( Ablation of prostate tissue, transurethral, using thermal ultrasound, including magnetic resonance imaging guidance for, and monitoring of, tissue ablation; with insertion of transurethral ultrasound transducer for delivery of thermal ultrasound, including suprapubic tube placement and placement of an endorectal cooling device, when performed) is an appropriate crosswalk code for CPT code 0582T. CPT code 55882 is currently assigned to APC 5377 (Level 7 Urology and Related Services).
After consideration of the public comment we received, we are finalizing our proposed APC assignment and status indicator for CPT code 0582T with modification to assign CPT code 0582T to APC 5377 (Level 7 Urology and Related Services) with a status indicator of “J1”. Refer to Table 89 for the final OPPS APC and status indicator assignment for CPT code 0582T for CY 2026. We refer readers to Addendum B to this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
Vascular Procedures
63. Arteriovenous Fistula (AVF) Creation Procedures, CPT Codes 36836 and 36837 (APC 5194)
CPT codes 36836 ( Percutaneous arteriovenous fistula creation, upper extremity, single access of both the peripheral artery and peripheral vein, including fistula maturation procedures (eg, transluminal balloon angioplasty, coil embolization) when performed, including all vascular access, imaging guidance and radiologic supervision and interpretation) and 36837 ( Percutaneous arteriovenous fistula creation, upper extremity, separate access sites of the peripheral artery and peripheral vein, including fistula ( printed page 53609) maturation procedures (e.g., transluminal balloon angioplasty, coil embolization ) when performed, including all vascular access, imaging guidance and radiologic supervision and interpretation) describe a percutaneous arteriovenous fistula creation of an upper extremity. CPT code 36836 became effective January 1, 2023 and replaced HCPCS codes C9754 and G2170, while CPT code 36837 became effective January 1, 2023, and replaced HCPCS codes C9755 and G2171.
In the CY 2020 OPPS/ASC final rule with comment period (84 FR 61246 through 61247), in response to public comment, input from our medical advisors, and the available claims data, we reassigned C9754 and C9755 from APC 5193 to APC 5194. In the CY 2021 OPPS/ASC final rule with comment period (85 FR 85954 through 95955), we used our equitable adjustment authority to maintain the assignment of HCPCS codes G2170 and G2171 to APC 5194 (Level 4 Endovascular Procedures), given that both procedures are for ESRD patients that need dialysis, the predecessor codes had very similar median costs, and there were low claims data available. We continued the assignment of CPT codes 36836 and 36837 to APC 5194, with commenter support, for CY 2022.
In the CY 2023 OPPS/ASC final rule with comment period (87 FR 71863 through 71864), we assigned the newly established CPT codes 36836 and 36837 to APC 5194 based on our assessment of the CY 2023 geometric mean cost of predecessor codes HCPCS codes G2170 ($12,055.90) and G2171 ($13,486.08) and their APC assignment. For CYs 2024 and 2025, we continued assignment to APC 5194 for CPT codes 36836 and 36837, with commenter support.
For CY 2026, we proposed to continue assignment of both CPT codes 36836 and 36837 to APC 5194 (Level 4 Endovascular) with a proposed payment rate of $18,791.32. For the CY 2026 OPPS/ASC proposed rule, CPT code 36836 had 170 single claims for ratesetting, a geometric mean cost (GMC) of around $11,260, and CPT code 36837 had 77 single claims for ratesetting and a GMC of around $19,615.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter requested that CMS reassign CPT code 36836 to APC 5193 (Level 3 Endovascular Procedures) stating that there is a significant cost differential between CPT codes 36836 and 36837.
Response: At this time, we continue to believe that both CPT codes 36836 and 36837 are assigned to appropriate APCs. We will continue to monitor the geometric mean costs and claims data and revise their APC assignments as appropriate.
In summary, after consideration of the public comment we received, we are finalizing the APC assignments for CPT codes 36836 and 36837 without modification. Specifically, we are finalizing the APC assignment of CPT codes 36836 and 36837 to APC 5194 (Level 4 Endovascular Services).
Refer to Table 90 for the code descriptor, proposed and final status indicator (SI) assignment, and proposed and final APC assignment. The final CY 2026 payment rates for the codes can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 to this final rule with comment period for the SI definitions for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
64. Atherectomy With Angioplasty, CPT Code 92924 (APC 5193)
CPT code 92924 ( Percutaneous transluminal coronary atherectomy, with coronary angioplasty when performed; single major coronary artery or branch) describes a procedure to treat coronary artery disease by removing plaque from the coronary vessels.
For the CY 2026 OPPS/ASC proposed rule, CPT code 92924 had a geometric mean cost (GMC) of around $16,262 based on 498 single frequency claims. We proposed to maintain the APC assignment to APC 5193 (Level 3 Endovascular Procedures), with a proposed payment of around $11,873.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: Commenters requested that CMS reassign CPT code 92924 to APC 5194 (Level 4 Endovascular Procedures). The commenters stated that CPT code 92924 is a technically demanding complex procedure and is more clinically similar to other procedures assigned to APC 5194 (Level 4 Endovascular Procedures). They note that in the CY 2026 OPPS/ASC proposed 2 Times Rule File, CPT code 92924 has the 3rd highest GMC in its current APC assignment (APC 5193) and has a higher GMC than 7 other procedures assigned to APC 5194. Based on the GMC of CPT code 92924, the commenters believe it is more appropriate to assign CPT code 92924 to ( printed page 53610) APC 5194 (Level 4 Endovascular Procedures).
Response: After reviewing the comments, we believe that CPT code 92924 is more clinically similar to the procedures assigned to APC 5193. As with many APC families, there may be services and procedures that have higher GMCs than other procedures in the next level. We determine APC assignments based on resource and clinical homogeneity and review and revise the services within each APC group and the APC assignments under the OPPS.
In summary, after consideration of the public comments we received, we are finalizing our proposal without modification, to assign CPT code 92924 to APC 5193 (Level 3 Endovascular Procedures).
The final CY 2026 payment rates for this code can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 to this final rule with comment period for the SI definitions for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
Refer to Table 91 for code descriptor, APC assignment and status indicator assignment for CPT 92924 code for CY 2026.
65. Coronary Therapeutic Services and Procedures CPT Codes 92930, 92945 (APCs 5194, 5193)
The CPT Editorial Panel created 2 new Category I codes 92930 and 92945, effective January 1, 2026. The new final CPT codes (along with their placeholder codes) are as follows:
- 92930 (92X01):Percutaneous transcatheter placement of intracoronary stent(s), with coronary angioplasty when performed, single major coronary artery and/or its branch(es); 2 or more distinct coronary lesions with 2 or more coronary stents deployed in 2 or more coronary segments, or a bifurcation lesion requiring angioplasty and/or stenting in both the main artery and the side branch; and
- 92945 (92X02):Percutaneous transluminal revascularization of chronic total occlusion, single coronary artery, coronary artery branch, or coronary artery bypass graft, and/or subtended major coronary artery branches of the bypass graft, any combination of intracoronary stent, atherectomy and angioplasty; combined antegrade and retrograde approaches.
For CY 2026, we proposed to assign both of these procedures to APC 5193 (Level 3 Endovascular Procedures) and a status indicator (SI) of `J1' with a proposed payment of $11,873.70.
We note that at the August 25, 2025, HOP Panel Meeting, a presentation was made requesting the APC reassignment of CPT codes 92930 (92X01) and 92945 (92X02). Based on the information presented at the meeting, the HOP Panel recommended that we reassign both CPT codes 92930 and 92945 from APC 5193 (Level 3 Endovascular Procedures) to APC 5194 (Level 4 Endovascular Procedures).
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: Commenters requested that CMS reassign CPT codes 92930 and 92945 to APC 5194 (Level 4 Endovascular Procedures) to better reflect the increased complexity of these procedures and resource utilization.
The commenters noted that while CMS crosswalked these codes to the straightforward versions of these procedures, the new codes include additional procedural work and resources that warrant a higher APC assignment. They note that CPT code 92930 includes 2 or more lesions with 2 or more stents deployed in 2 or more coronary segments versus the crosswalk code CPT code 92928 ( Percutaneous transcatheter placement of intracoronary stent(s), with coronary angioplasty when performed; single major coronary artery or branch) which only refers to a single coronary artery or branch.
Commenters also pointed to the multiple procedural approaches (antegrade and retrograde) with CPT code 92945, whereas the crosswalk CPT code 92943 ( Percutaneous transluminal revascularization of chronic total occlusion, coronary artery, coronary artery branch, or coronary artery bypass graft, any combination of intracoronary stent, atherectomy and angioplasty; single vessel) is only a single approach (antegrade).
A commenter suggested using C9607 ( Percutaneous transluminal revascularization of chronic total occlusion, coronary artery, coronary artery branch, or coronary artery bypass graft, any combination of drug-eluting intracoronary stent, atherectomy and angioplasty; single vessel) as a crosswalk code for CPT code 92945. Commenters urged CMS to not undervalue these services from the outset and that CMS should reassign these codes to APC 5194 (Level 4 Endovascular Procedures and monitor the claims data to ensure that these codes are assigned to an appropriate APC based on clinical and resource homogeneity.
Response: After review of the public comments and input from our Medical Officers, we agree with the commenters that CPT code 92930 is more complex and utilizes more resources (that is, additional stents) than its crosswalk code. However, we disagree with commenters that more resources are used with CPT 92945. As we do every year, we will monitor the claims data and reevaluate the APC assignments for these codes in the next rulemaking cycle to determine if a more appropriate APC assignment is warranted.
In summary, after consideration of the public comments we received, input from our Medical Officers, and the HOP Panel recommendation, we are finalizing the APC and status indicators for CPT codes 92930 and 92945 with modification. Specifically, we are assigning CPT 92930 to APC 5194 and continuing to assign CPT 92945 to APC 5193.
The final CY 2026 payment rates for these codes can be found in Addendum B to this final rule with comment period. In addition, we refer readers to ( printed page 53611) Addendum D1 to this final rule with comment period for the SI definitions for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
Refer to Table 92 for code descriptor, APC assignment and status indicator assignments for CPT codes 92930 and 92945 for CY 2026.
66. Lower Extremity Revascularization, CPT Codes 37254 Through 37299 (APCs 5192, 5193, and 5194)
For CY 2026, the CPT Editorial Panel deleted 16 CPT codes that described lower extremity revascularization and replaced them with 46 new codes. The new codes establish an additional peripheral vessel territory as well as being more granular in describing the procedures. The 16 revascularization codes will be deleted December 31, 2025, and replaced with the new CPT codes effective January 1, 2026.
The deleted revascularization CPT codes are listed in Table 93, along with their current APC and status indicator assignments. Table 94 lists the new revascularization CPT code (along with its placeholder), the long descriptor, status indicator, APC assignment, and the crosswalk code that was used to determine the proposed APC assignment.
( printed page 53612) ( printed page 53613) ( printed page 53614) ( printed page 53615) ( printed page 53616) ( printed page 53617) ( printed page 53618) ( printed page 53619) ( printed page 53620) ( printed page 53621) ( printed page 53622) ( printed page 53623)We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: Commenters were supportive that CMS used the corresponding codes that are being deleted as crosswalks for the new codes, noting that we have extensive claims history for the codes that are being deleted, and this approach provides payment stability.
A commenter requested that CMS reassign CPT codes 37263, 37265, and 37269 to APCs 5193, 5194, and 5194, respectively because of the complexity of these codes and the additional resources used during these procedures.
Several of the commenters had comments related to complexity adjustments and device offset percentages involving these new codes. We refer the readers to the applicable sections in this final rule with comment period for responses to those comments.
Response: We reviewed the commenter's request to reassign CPT codes 37263, 37265, and 37269 to APCs 5193, 5194, and 5194 respectively. After review and discussion with our CMS Medical Officers, we believe that we assigned these codes to appropriate APCs utilizing the predecessor codes as crosswalks.
In summary, after consideration of the public comments we received, we are finalizing our proposals without modification, to assign CPT codes 37254 through 37299 to the APCs and status indicators noted in Table 94. The final CY 2026 payment rates for these codes can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 to this final rule with comment period for the SI definitions for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
67. Percutaneous Transcatheter Therapeutic Drug Delivery (CPT Code 0913T)
The CPT Editorial Panel created CPT code 0913T ( Percutaneous transcatheter therapeutic drug delivery by intracoronary drug-delivery balloon (e.g., drug-coated, drug-eluting), including mechanical dilation by nondrug-delivery balloon angioplasty, endoluminal imaging using intravascular ultrasound (IVUS) or optical coherence tomography (OCT) when performed, imaging supervision, interpretation, and report, single major coronary artery or branch) effective January 1, 2025. For CY 2026, we proposed to assign CPT code 0913T to APC 5192 (Level 2 Endovascular Procedures) based on the crosswalk code CPT 92920 (Percutaneous transluminal coronary angioplasty; single major coronary artery or branch) which is assigned to APC 5192 (Level 2 Endovascular Procedures) and a status indicator of “J1” (Hospital Part B Services Paid through a Comprehensive APC; Paid under OPPS).
For the CY 2026 OPPS/ASC proposed rule, the OPPS payment rates were proposed based on available CY 2024 claims data. Because CPT code 0913T became effective January 1, 2025, we have no claims for ratesetting. We also note that the device associated with this procedure (AGENTTM Paclitaxel-Coated Balloon Catheter) received device pass-through status effective January 1, 2025 (89 FR 91434 through 91439).
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: Commenters requested that CMS reassign CPT code 0913T to APC 5193 (Level 3 Endovascular Procedures). Commenters stated that CPT code 0913T was more clinically coherent with procedures assigned to Level 3 Endovascular Procedures than Level 2 Endovascular Procedures. To support their requested APC changes, commenters suggested that CMS consider crosswalking CPT code 0913T to HCPCS C9600 (Percutaneous transcatheter placement of drug eluting intracoronary stent(s), with coronary angioplasty when performed; single major coronary artery or branch). Commenters stated that both services are performed in addition to angioplasty, suggesting that both drug-coated balloon and stent placement serve a separate and distinct purpose, as compared to coronary angioplasty alone. Commenters also provided an analysis of 294 single frequency claims and indicated that the geometric mean cost of those claims was $11,000. Moreover, commenters stated that assignment of CPT code 0913T to APC 5192 would pose a two times rule violation. In addition to the APC reassignment request, commenters also requested a complexity adjustment for CPT code 0913T and procedure code 93459.
Response: We note that APC 5192 does not currently have a two times rule violation in the final rule data. In addition, CPT code 0913T does not meet the requirements for cost significance for 2 times rule violation purposes, under the requirements described in section III.B.2. of this final rule with comment period. After consideration of the public comments, additional review of the procedures, and input from our CMS Medical Officers, we believe that the resource costs of CPT code 0913T is more aligned with the procedures in APC 5193, which includes HCPCS code C9600 (Percutaneous transcatheter placement of drug eluting intracoronary stent(s), with coronary angioplasty when performed; single major coronary artery or branch).
We remind hospitals that we review, on an annual basis, the APC assignments for all items and services paid under the OPPS and we will continue to monitor the claims data for APC 5193 as they become available. Additionally, we refer readers to XIII.C. of this final rule with comment period for a discussion of the complexity adjustment policies.
In summary, after consideration of the public comments we received, we are finalizing our proposal with modification and reassigning CPT code 0913T to APC Level 5193. The final CY 2026 payment rates for these codes can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 to this final rule with comment period for the status indicator definitions for all codes reported under the OPPS. ( printed page 53624) Addenda B and D1 are available via the internet on the CMS website.
Refer to Table 95 for code descriptors, APC assignments and status indicator assignments for CPT code 0913T.
68. APC Specific Comments That Support the Proposed APC Assignment
Each year, in accordance with section 1833(t)(9)(A) of the Act, we review and revise the services within each APC group and the APC assignments under the OPPS. As a result of our annual review of the services and the APC assignments, we may reassign some services to another APC or maintain the current APC assignment.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: Commenters supported the proposed CY 2026 assignments for various HCPCS and CPT codes and requested that CMS finalize the payment assignments for CY 2026 as proposed.
Response: We thank the commenters for their input.
Therefore, for CY 2026, we are finalizing without modification the APCs and status indicators for the CPT codes listed in Table 96. The final CY 2026 payment rates for the codes listed below can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 to this final rule with comment period for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
( printed page 53625) ( printed page 53626)69. Dialysis Related Services and Technologies
a. Dialysis-Related Amyloidosis (DRA) Treatment With Lixelle® Apheresis Columns
LIXELLE® β2-microglobulin Apheresis Column is indicated for use in the treatment of dialysis-related amyloidosis (DRA), a disease that affects people with end-stage renal disease (ESRD). DRA is a metabolic disorder from the failure of the kidney to filter and remove β2-microglobulin, typically from chronic hemodialysis (typically 5 years or longer). The LIXELLE® device is used in an apheresis procedure that selectively removes β2-microglobulin from circulating blood and is used in accordance with a physician prescription in conjunction with hemodialysis. It is intended to be used at each hemodialysis session (that is, frequency of treatment is expected to be 3 times per week). In March 2015, FDA approved LIXELLE® as a Class III Humanitarian Use Device (HUD) with an approved Humanitarian Device Exemption (HDE). For CY 2026, there are currently no specific HCPCS or CPT codes that represent the LIXELLE® apheresis service.
We received public comments on this topic. The following is a summary of the comments we received and our responses.
Comment: Several interested parties commented requesting coverage for Lixelle® apheresis columns for treating dialysis-related amyloidosis (DRA). Commenters noted that Lixelle® has FDA Humanitarian Use Device approval and has been successfully utilized in Japan.
Response: We recognize the clinical need for effective treatments for dialysis-related amyloidosis and appreciates the information provided regarding Lixelle® apheresis columns. We acknowledge that DRA represents a serious complication for long-term dialysis patients and that treatment options have been limited. We are actively reviewing coverage pathways for innovative treatments that address unmet medical needs in the ESRD population, including those with FDA Humanitarian Use Device designations. We note this complex, ongoing issue is still under consideration and continues to merit a thorough evaluation to ensure an appropriate Medicare benefit category and payment pathway for the service is determined.
b. Dialysis Technologies
We received public comments on this topic. The following is a summary of the comments we received and our responses.
Comment: A commenter supported the payment of dialysis technologies not included in the End Stage Renal Disease (ESRD) bundle through the OPPS. The commenter noted that CMS continues to evaluate potential avenues of payment for treatments using medical devices administered during dialysis procedures that are not considered “renal dialysis services” under the Medicare statute and requested that CMS provides payment for such services through the OPPS.
Response: We appreciate the input regarding coverage and payment for medical device technologies used in conjunction with dialysis services. We recognize the importance of ensuring appropriate payment pathways for innovative treatments that may be administered during dialysis but fall outside the definition of a renal dialysis service under § 413.171 and thus are not paid for under the ESRD PPS. We will continue to evaluate the appropriate payment mechanisms for such technologies for future rulemaking. ( printed page 53627)
c. Open Surgical Fistula Creation
CPT code 36821 (Arteriovenous anastomosis, open; direct, any site (e.g., Cimino type) (separate procedure) describes an open surgical fistula creation for hemodialysis procedures. CPT code 36821 is currently assigned to APC 5183 (Level 3 Vascular Procedures) with a proposed payment of around $3,254, and a status indicator of `J1' (Hospital Part B Services Paid Through a Comprehensive APC; Paid under OPPS).
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter expressed concern about payment disparities between traditional surgical arteriovenous fistula (AVF) creation and endovascular procedures. The commenter noted that ASC payment for endovascular creation can be substantially higher than traditional fistula creation, despite the benefits that surgical AVFs may offer to both patients and providers, particularly the greater versatility that surgical AVFs can offer in placement locations when compared to endovascular AVFs. The commenter expressed concern that low payment may cause skilled surgeons to preferentially choose better-paying procedures and potentially diminish the availability of AVF creation.
Response: We acknowledge the concerns regarding payment disparities between surgical and endovascular fistula creation procedures. We recognize that both approaches serve important clinical roles in vascular access for dialysis patients. We will continue to evaluate payment rates for these procedures to ensure appropriate payment that reflects the clinical value and resource costs associated with each approach.
d. Peritoneal Dialysis (PD) Catheter Placement
There are a number of CPT codes that describe placing a catheter for peritoneal dialysis. These codes and their payment assignments include the following:
- 49324—Laparoscopy, surgical; with insertion of tunneled intraperitoneal catheter; assigned to APC 5361 (Level 1 Laparoscopy and Related Services) and status indicator `J1' (Hospital Part B Services Paid Through a Comprehensive APC; Paid under OPPS);
- 49418—Insertion of tunneled intraperitoneal catheter (e.g., dialysis, intraperitoneal chemotherapy instillation, management of ascites), complete procedure, including imaging guidance, catheter placement, contrast injection when performed, and radiological supervision and interpretation, percutaneous; assigned to APC 5341 (Level 1 Abdominal/Peritoneal/Biliary and Related Procedures) and status indicator `J1' (Hospital Part B Services Paid Through a Comprehensive APC; Paid under OPPS); and
- 49421—Insertion of tunneled intraperitoneal catheter for dialysis, open; assigned to APC 5341 (Level 1 Abdominal/Peritoneal/Biliary and Related Procedures) and status indicator `J1' (Hospital Part B Services Paid Through a Comprehensive APC; Paid under OPPS).
For CY 2026, we proposed to continue to assign these codes to their current payment assignments.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter expressed concern that current CMS payment for peritoneal dialysis (PD) catheter placement may have created a disincentive for performing the procedure. The commenter specifically noted that low payment for PD catheter placement relative to vascular access procedures may create barriers to patients receiving more convenient home-based treatment. The commenter requested that CMS equalize payment between PD catheter procedures and vascular access procedures to avoid possible disincentives to home treatment and provide patients with additional quality care options.
Response: We support policies that facilitate appropriate home-based dialysis care when clinically appropriate. We recognize the importance of ensuring that payment policies do not inadvertently create barriers to home dialysis modalities, including peritoneal dialysis. We will review the current payment rates for PD catheter placement procedures and consider any adjustments that better align with the clinical value and resource requirements of these services for future notice-and-comment rulemaking.
70. Mobile Stroke Units
Mobile stroke units (MSU) are specialized ambulances equipped with various skilled healthcare personnel, specialized equipment, including imaging capability to diagnose and treat acute stroke in the prehospital setting.[27]
We received public comments on this topic. The following is a summary of the comments we received and our responses.
Comment: A commenter requested that MSU services be reimbursed as hospital outpatient services rather than ambulance transport services. The commenter noted that the current Ambulance Fee Schedule (AFS) payment does not cover the cost of CT scans and other essential components of an MSU, leading to financial challenges for MSU operators. The commenter cited clinical evidence pointing to improved outcomes associated with the use of MSUs.
Response: We appreciate the detailed comment regarding MSU payment challenges. We appreciate the thoughtful recommendations and will consider them for future rulemaking. We note that MSUs are currently paid under the AFS.
F. Comment Solicitation on Payment Policy for Software as a Service (SaaS)
In recent years, there have been rapid developments in the use of software-based technologies with new functionalities, including artificial intelligence, to support clinical decision-making in the outpatient and physician office settings. Medicare refers to these software-based technologies as software as a service (SaaS). Prior to CY 2018, SaaS procedures were considered supportive or ancillary services, and therefore, payment for the SaaS was packaged into the payment for the underlying clinical service. For example, payment for image processing software that visually enhances an image in an existing MRI, would be packaged into the payment for the MRI service. In recent years, CMS has paid separately for SaaS procedures under the OPPS through New Technology APCs, which are cost bands that allow us to provide appropriate and consistent payment for designated new procedures that are not yet reflected in our claims data, and various clinical APCs based on clinical and resource similarity to existing services, including Imaging APCs and Diagnostic Tests and Related Services APCs. We currently do not have a payment methodology specifically for SaaS, and as these technologies have continued to evolve and diversify, some interested parties have stated that the lack of a consistent payment policy for SaaS can be an impediment to patient access when these services are otherwise approved by the FDA. Interested parties have requested that CMS consider development of a payment policy for these services that is stable and ( printed page 53628) consistent across settings of care, payment systems, and types of SaaS.
In the CY 2026 OPPS/ASC proposed rule, we welcomed public comment as we consider how to appropriately pay for these services, including any applicable lessons or best practices from risk-bearing payment arrangements, how we can determine that Medicare payments for SaaS truly reflect the value of the technologies to medical practice, and how to ensure that any payment policies on this topic demonstrate fiscal responsibility and good stewardship by promoting high-value, cost-effective care. For pricing new technologies where we do not have substantive supporting data, there are ambiguities regarding the service costs for purposes of setting a payment rate. For example, we have observed wide variations in the purported costs of clinically similar SaaS technologies. The various costs that manufacturers consider when pricing their technologies, including research and development as well as software maintenance, are often not publicly verifiable. It is also unclear to what extent Medicare should pay for the research and development costs of SaaS that could be frequently used by non-Medicare beneficiaries in hospital outpatient departments and ambulatory surgical center settings. Additionally, due to the novel and evolving nature of these technologies, there are rarely existing medical items or services that can be utilized for comparison purposes to determine clinical and resource similarity. Finally, while there has been a rapid increase in the development and coding of these services in recent years, there is a very limited amount of Medicare claims data for these services.
Given these issues and our interest in developing payment policies that seek to reflect the underlying value of a service or technology to the practice of medicine, we requested public comment on future SaaS payment ideas, including:
- What factors could Medicare consider when setting payment rates for SaaS?
- What APCs, existing or new, should we use to pay for SaaS?
- How should we assess the costs of SaaS, and how can we account for hospital acquisition costs?
- What cost or claims data should be used to establish the payment rates for the services?
- Why are the geometric mean costs, as provided in our claims data, for SaaS currently assigned to APCs (both clinical and New Technology APCs) consistently lower than the manufacturers' purported costs of the technologies?
- Is there an alternative data source outside of the limited Medicare claims data currently available and hospital invoices provided by manufacturers, which may not fully depict total hospital acquisition costs, that can accurately reflect the costs of the SaaS?
- What kinds of efficiencies, if any, would SaaS provide for services performed in hospital outpatient departments and ambulatory surgical centers?
- In the context of setting Medicare payment rates, how can CMS best reflect the quality and efficacy of SaaS technologies?
We welcomed input from interested parties on these questions as well as any additional suggestions that would enhance our ability to provide accurate and consistent payment for SaaS procedures. Finally, we noted that there is a similar comment solicitation on a payment policy for SaaS under the Physician Fee Schedule, and directed readers to the CY 2026 PFS proposed rule to provide comments.
We received public comments on this topic. The following is a summary of the comments we received and our responses.
Comment: Several commenters requested that CMS more explicitly define the technologies we would consider to be SaaS. Some commenters suggested that CMS use an alternative term to refer to these technologies, such as “Software as a Medical Device” in conformance with FDA definitions, or “clinical decision support tools.” Some commenters provided suggestions on how CMS should categorize these types of services in terms of payment. Suggestions included classifying the technologies by the type of solution they provide, such as diagnostic, therapeutic, augmentative, patient-facing, operational. Commenters encouraged CMS to work with interested parties to establish definitions to help clarify which technologies are payable, while maintaining flexibility to adopt definitions as technologies evolve.
Generally, commenters supported a dedicated payment policy for innovative technologies. Commenters requested that CMS create an evidence-based, flexible framework that encourages the use and innovation of SaaS while maintaining fair and adequate payment for the services in which they are incorporated. Commenters offered ideas for potential payment policies. For example, most commenters recommended that CMS assign all SaaS to New Technology APCs for a certain amount of time, usually between 3 to 5 years, to allow for CMS to gather sufficient claims data upon which to make an appropriate clinical APC assignment. Some commenters supported and referenced the payment pathway outlined in The Health Tech Investment Act (S. 1399). Another suggestion was for CMS to treat SaaS technologies as ancillary items and package payment with the related primary services as long as patient access was not adversely affected. In general, commenters encouraged CMS to adopt a payment approach that balances innovation, patient access, and value to the Medicare program.
Many commenters urged CMS to exercise caution and consider several factors in creating a payment methodology specifically for SaaS. Several commenters explained that costs for these technologies are difficult to determine. For example, some commenters explained the different variations in pricing models for SaaS, noting that some services are invoiced as a subscription or per-click fee as opposed to a one-time purchase of software or equipment. Commenters also stated that appropriately accounting for research and development costs in Medicare payments would be challenging. Many commenters addressed why they believe even current Medicare claims data may be unreliable for setting payment rates for SaaS, including that hospitals may be reporting various revenue center codes with different cost-to-charge ratios. To address the lack of price transparency across vendors and practice types, and to collect real-world cost data, a commenter encouraged CMS to work with the Congress to establish authority to incentivize standardized vendor disclosures, stratified by practice or facility size, setting, and geography. Other commenters suggested that CMS consider creating new revenue codes to facilitate more accurate hospital cost reporting for AI-based services. Others requested that CMS establish a unique cost center for AI-based diagnostic and decision-support services within the cost reporting structure to enable the collection of accurate, disaggregated cost and charge data specific to these technologies. Many commenters requested detailed guidance regarding charging and cost reporting practices for these services given the various pricing models available.
Some commenters were concerned about the potential for costs to increase if CMS were to finalize a new payment policy specific to SaaS. A commenter explained that providing separate payment for SaaS items under the OPPS, rather than packaging payment for SaaS with the related primary service for which it is used, would not ( printed page 53629) provide any downward pricing pressures on SaaS developers to provide value to the Medicare program. Commenters urged that, if not carefully designed, such a policy for SaaS could create significant financial risk, administrative burden, and access issues, without delivering clear clinical value. To that end, some commenters suggested CMS package payment when tools are integral to a service or pursue value-based payment models and risk-based contracts that reward outcomes and efficient care delivery.
Regarding coding, some commenters believe CMS should develop broad HCPCS codes to describe multiple SaaS technologies. Commenters also encouraged CMS to pursue future code development for SaaS and valuation through the American Medical Association CPT/RUC process, which allows for transparency and dialogue with involved interested parties. Commenters also requested new codes for certain SaaS technologies.
Finally, many commenters recommended that CMS adopt a site-neutral payment policy for SaaS across the facility and non-facility settings.
Response: The comments received illustrate the complexity intrinsic to paying for SaaS, and we will proceed with awareness of these challenges. We continue to recognize the need for a payment policy that accounts for the unique and heterogenous characteristics of SaaS, and we remain interested in ensuring that any such payment policy reflects the value provided to Medicare providers and beneficiaries. We intend to take the comments submitted into consideration as we develop our proposals for future rulemaking.
G. Continuation of Payment Policy for Radiation Therapy Services Furnished at Nonexcepted Off-Campus Provider Based Departments (PBDs)
1. Background on Section 603 of the Bipartisan Budget Act of 2015 and the PFS Relativity Adjuster
Section 603 of the Bipartisan Budget Act of 2015 (Pub. L. 114-74) (BBA, 2015) (hereinafter referred to as “section 603”) amended section 1833(t) of the Act by adding a new clause (v) to paragraph (1)(B) and adding a new paragraph (21). As a general matter, under sections 1833(t)(1)(B)(v) and (t)(21) of the Act, applicable items and services furnished by certain off-campus outpatient departments of a provider on or after January 1, 2017, are not considered covered OPD services as defined under section 1833(t)(1)(B) of the Act for purposes of payment under the OPPS. Instead such items are paid “under the applicable payment system” under Medicare Part B if the requirements for such payment are otherwise met. Section 603 amended section 1833(t)(1)(B) of the Act by adding a new clause (v), which excludes from the definition of “covered OPD services” applicable items and services (defined in paragraph (21)(A) of the section) that are furnished on or after January 1, 2017, by an off-campus PBD, as defined in paragraph (21)(B) of the section.
In the CY 2017 OPPS/ASC final rule with comment period (81 FR 79699 through 79719), we adopted a number of policies to implement section 603. Broadly, we: (1) defined applicable items and services in accordance with section 1833(t)(21)(A) of the Act for purposes of determining whether such items and services are covered OPD services under section 1833(t)(1)(B)(v) of the Act or whether payment for such items and services will instead be made under the applicable payment system designated under section 1833(t)(21)(C) of the Act; (2) defined off-campus PBD for purposes of sections 1833(t)(1)(B)(v) and (t)(21) of the Act; and (3) established policies for payment for applicable items and services furnished by an off-campus PBD (nonexcepted items and services) under section 1833(t)(21)(C) of the Act. To do so, we finalized policies that define whether certain items and services furnished by a given off-campus PBD may be considered excepted and, thus, continue to be paid under the OPPS; established the requirements for the off-campus PBDs to maintain excepted status (both for the excepted off-campus PBDs and for the items and services furnished by such excepted off-campus PBDs); and described the applicable payment system for nonexcepted items and services (generally, the PFS).
To effectuate payment for nonexcepted items and services, in the CY 2017 interim final rule with comment period (81 FR 79720 through 79729), we established a new set of payment rates under the PFS that reflected the relative resource costs of furnishing the technical component of a broad range of services to be paid under the PFS specific to the nonexcepted off-campus PBDs of a hospital. Specifically, we established a PFS Relativity Adjuster that is applied to the OPPS rate for the billed nonexcepted items and services furnished in a nonexcepted off-campus PBD in order to calculate payment rates under the PFS. The PFS Relativity Adjuster reflects the estimated overall difference between the payment that would otherwise be made to a hospital under the OPPS for the nonexcepted items and services furnished in nonexcepted off-campus PBDs and the resource-based payment under the PFS for the technical aspect of those services with reference to the difference between the facility and nonfacility (office) rates and policies under the PFS. Nonexcepted items and services furnished by nonexcepted off-campus PBDs are generally paid under the PFS at the applicable OPPS payment rate adjusted by the PFS Relativity Adjuster of 40 percent (that is, 60 percent less than the OPPS rate) (82 FR 53030).
In the CY 2017 OPPS/ASC final rule with comment period (81 FR 79719 and 79725), we created modifier “PN” to collect data for purposes of implementing section 603 but also to trigger payment under the newly adopted PFS-equivalent rates for nonexcepted items and services. Nonexcepted off-campus PBDs bill for nonexcepted items and services on the institutional claim utilizing modifier “PN” to indicate that an item or service is a nonexcepted item or service.
For a full discussion of our initial implementation of section 603, we refer readers to the CY 2017 OPPS/ASC final rule with comment period (81 FR 79699 through 79719) and the interim final rule with comment period (79720 through 79729). For a detailed discussion of the current PFS Relativity Adjuster related to payments under section 603, we refer readers to the CY 2018 OPPS/ASC final rule with comment period (82 FR 52356 through 52637) and the CY 2019 PFS final rule with comment period (82 FR 59505 through 59513).
2. Payment for Radiation Therapy Services Furnished at Nonexcepted Off-Campus PBDs
The PFS Relativity Adjuster is not applied to radiation therapy services (radiation treatment delivery and related imaging guidance services) furnished by nonexcepted off-campus PBDs. Due to section 1848(c)(2)(K) of the Act, which required maintenance of the CY 2016 coding and payment inputs for these services for CY 2017 and CY 2018 under the PFS, when the section 603 requirements were implemented in the CY 2017 final OPPS rule, we instructed nonexcepted off-campus PBDs to bill the PFS G-codes for these services. As we explained in that rule:
“. . . [S]everal radiation treatment delivery and imaging guidance services also are reported using different codes under the MPFS and the OPPS. CMS established HCPCS Level II G-codes to describe radiation treatment delivery services when furnished in the physician office setting (79 FR 67666 ( printed page 53630) through 67667). However, these HCPCS G-codes are not recognized under the OPPS; rather, CPT codes are used to describe these services when furnished in the HOPD. Both sets of codes were implemented for CY 2015 and were maintained for CY 2016. Under the MPFS, there is a particular statutory provision under section 1848(c)(2)(K) of the Act that requires maintenance of the CY 2016 coding and payment inputs for these services for CY 2017 and CY 2018. Accordingly, the finalized CY 2017 MPFS rates for these services were calculated based on the maintenance of the CY 2016 coding payment inputs. On that basis, we are establishing payment amounts for nonexcepted items and services consistent with the payments that would be made to other facilities under the MPFS. That is, an off-campus PBDs submitting claims for nonexcepted items and services will bill the HCPCS G-codes established under the MPFS to describe radiation treatment delivery procedures. However, the off-campus PBD must append modifier “PN” to each applicable claim line for nonexcepted items and services. The payment amount for these services will be set to reflect the technical component rate for the code under the MPFS.” (81 FR 79726).”
As discussed in the CY 2026 Physician Fee Schedule (PFS) final rule, we are finalizing our proposal to delete radiation therapy G-codes (G6001 through G6017) that describe imaging guidance for radiation treatment (G6001, G6002, G6017) and radiation treatment delivery (G6003-G6015) because CPT codes 77402, 77407, and 77412 have been revised and may be used to report these services instead. See Table 97 for the long descriptors of the G codes that we are deleting effective January 1, 2026 and Table 98 for the current and revised long descriptors for CPT codes 77402, 77407, and 77412. The final CY 2026 payment rates for the radiation treatment delivery codes can be found in Addendum B to this final rule via the internet on the CMS website.
As we noted in the CY 2026 OPPS/ASC proposed rule, nonexcepted off-campus PBDs currently use the above referenced G codes to report radiation therapy services and, were we to finalize our proposal to delete them, they would no longer be available after December 31, 2025. To continue paying the PFS-equivalent rate for these services to these departments, we proposed that, effective January 1, 2026, nonexcepted off-campus PBDs would use the revised CPT codes described in the CY 2026 PFS proposed rule. In other words, because the G codes are being eliminated, we proposed that the revised CPT codes be used to preserve the existing policy of paying nonexcepted off-campus PBDs a specific radiation treatment rate, which is the technical component for the code under the Medicare PFS (MPFS). Crosswalk information between the G codes and the revised CPT codes is available under the Downloads section [28] of the CY 2026 PFS proposed rule, under “CY 2025 Analytic Crosswalk to CY 2026.” We additionally proposed that nonexcepted off-campus PBDs would continue to append the “PN” modifier to each applicable claim line for these services. We emphasized in the CY 2026 OPPS/ASC proposed rule that this was not a new policy but rather a continuation of current policy adjusting for the newly revised CPT codes and the corresponding deletion of the G codes.
( printed page 53631) ( printed page 53632)We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter opined that the continuation of the policy was essential to preserve access for patients with cancer who might otherwise face long travel burdens and encouraged CMS to monitor visit utilization rates after implementation to ensure that beneficiaries with multiple chronic conditions do not lose timely access to complex management in outpatient settings.
Response: We thank the commenter for their support.
Comment: A commenter supported the deletion of the G codes and the use of the revised CPT codes by all radiation treatment providers. However, the commenter requested that CMS not apply the PFS Relativity Adjustor to the revised codes when billed by nonexcepted off-campus provider-based departments. The commenter expressed concern that doing so would have an “untenable impact on payment” to these departments because, based on the proposed radiation treatment delivery APCs in CY 2026, the reimbursement for IMRT in these locations would fall to $110 per treatment which is well below cost. The commenter contended that it is not logical to use OPPS data to set PFS rates for offices and then reduce those rates by 60 percent. The commenter recommended that, since CMS plans to use OPPS data for developing MPFS Practice Expense Relative Value Units (PE RVUs), CMS should not apply the PFS adjustor to the new radiation treatment codes and simply pay the MPFS PE rates for these services when billed with modifier PN.
Response: We appreciate the commenter's concerns for the application of the PFS Relativity Adjustor to the new radiation treatment codes when billed by nonexcepted off-campus PBDs. We note that, while the off-campus PBD must append modifier “PN” to each applicable claim line for the new radiation treatment codes, the payment amount for these services when billed with modifier “PN” will be set to reflect the technical component rate for the code under the MPFS. We do not intend for radiation therapy services furnished by nonexcepted off-campus PBDs to be paid any differently than they were paid previously when billed as G codes. We believe that the commenter's concerns for adequate payment to these departments for codes involving IMRT is further addressed by our final APC assignments for CPT codes 77407 and 77412 discussed in section III.E. of this final rule with comment period.
After consideration of the public comments we received, we are finalizing, without modification, our proposal that, effective January 1, 2026, nonexcepted off-campus PBDs use the revised radiation treatment CPT codes described in the CY 2026 PFS proposed rule and append modifier “PN” to each applicable claim line for nonexcepted items and services. The payment amount for these services when billed with the “PN” modifier will be set to reflect the technical component rate for the code under the MPFS.
IV. OPPS Payment for Devices
A. Pass-Through Payment for Devices
1. Beginning Eligibility Date for Device Pass-Through Status and Quarterly Expiration of Device Pass-Through Payments
a. Background
The intent of transitional device pass-through payment, as implemented at 42 CFR 419.66, is to facilitate access for beneficiaries to the advantages of new and truly innovative devices by allowing for adequate payment for these new devices while the necessary cost data is collected to incorporate the costs for these devices into the procedure APC rate (66 FR 55861). Under section 1833(t)(6)(B)(iii) of the Act, the period for which a device category eligible for transitional pass-through payments under the OPPS can be in effect is at least 2 years but not more than 3 years.
In the CY 2017 OPPS/ASC final rule with comment period, in accordance with section 1833(t)(6)(B)(iii)(II) of the Act, we amended § 419.66(g) to provide that the pass-through eligibility period for a device category begins on the first date on which pass-through payment is made under the OPPS for any medical device described by such category (81 FR 79654). In addition, in the CY 2017 OPPS/ASC final rule with comment period, we finalized a policy to allow for quarterly expiration of pass-through payment status for devices to afford a pass-through payment period that is as close to a full 3 years as possible for all pass-through payment devices (81 FR 79655). We also established a policy to package the costs of the devices that are no longer eligible for pass-through ( printed page 53633) payments into the costs of the procedures with which the devices are reported in the claims data used to set the payment rates (67 FR 66763).
We refer readers to the CY 2017 OPPS/ASC final rule with comment period (81 FR 79648 through 79661) for a full discussion of the current device pass-through payment policy.[29]
In the CY 2023 OPPS/ASC final rule with comment period, we finalized our policy to publicly post online OPPS device pass-through applications received on or after March 1, 2023, beginning with the issuance of the CY 2025 proposed rule and for each OPPS rulemaking thereafter. We refer readers to the CY 2023 OPPS/ASC final rule with comment period (87 FR 71934 through 71938) for a full discussion of the policy to publicly post OPPS device pass-through applications.
b. Expiration of Transitional Pass-Through Payments for Certain Devices
As stated earlier, section 1833(t)(6)(B)(iii) of the Act requires that, under the OPPS, a category of devices be eligible for transitional pass-through payments for at least 2 years, but not more than 3 years. Currently, there are 20 device categories eligible for pass-through payment. These devices are listed in Table 99, previously published as Table 46 of the CY 2026 OPPS/ASC proposed rule (90 FR 33570 and 33571), where we detail the expiration dates of pass-through payment status for each of the 20 devices currently receiving device pass-through payment.
( printed page 53634) ( printed page 53635)2. New Device Pass-Through Applications for CY 2026
a. Background
Section 1833(t)(6) of the Act provides for pass-through payments for devices, and section 1833(t)(6)(B) of the Act requires CMS to use categories in determining the eligibility of devices for pass-through payments. As part of implementing the statute through regulations, we continue to believe that it is important for hospitals to receive pass-through payments for devices that offer substantial clinical improvement in the treatment of Medicare beneficiaries to facilitate access by beneficiaries to the advantages of the new technology. Conversely, we have noted that the need for additional payments for devices that offer little or no clinical improvement over previously existing devices is less apparent. In such cases, these devices can still be used by hospitals, and hospitals will be paid for them through appropriate APC payment. Moreover, a goal is to target pass-through payments for those devices where cost considerations are most likely to interfere with patient access (66 FR 55852; 67 FR 66782; and 70 FR 68629).
As specified in regulations at § 419.66(b)(1) through (3), to be eligible for transitional pass-through payment under the OPPS, a device must meet the following criteria:
- If required by FDA, the device must have received FDA approval or clearance and FDA marketing authorization (except for a device that has received an FDA investigational device exemption (IDE) and has been classified as a Category B device by FDA), or meet another appropriate FDA exemption; and the pass-through payment application must be submitted within 3 years from the date of the initial FDA marketing authorization, if required, unless there is a documented, verifiable delay in U.S. market availability after FDA marketing authorization is granted, in which case CMS will consider the pass-through payment application if it is submitted within 3 years from the date of market availability;
- The device is determined to be reasonable and necessary for the diagnosis or treatment of an illness or injury or to improve the functioning of a malformed body part, as required by section 1862(a)(1)(A) of the Act; and
- The device is an integral part of the service furnished, is used for one patient only, comes in contact with human tissue, and is surgically implanted or inserted (either permanently or temporarily), or applied in or on a wound or other skin lesion.
In addition, according to § 419.66(b)(4), a device is not eligible to be considered for device pass-through payment if it is any of the following: (1) equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciable assets as defined in Chapter 1 of the Medicare Provider Reimbursement Manual (CMS Pub. 15-1); or (2) a material or supply furnished incident to a service (for example, a suture, customized surgical kit, or clip, other than a radiological site marker).
Separately, we use the following criteria, as set forth under § 419.66(c), to determine whether a new category of pass-through payment devices should be established. The device to be included in the new category must—
- Not be appropriately described by an existing category or by any category previously in effect established for transitional pass-through payments, and was not being paid for as an outpatient service as of December 31, 1996; ( printed page 53636)
- Have an average cost that is not “insignificant” relative to the payment amount for the procedure or service with which the device is associated as determined under § 419.66(d) by demonstrating: (1) the estimated average reasonable cost of devices in the category exceeds 25 percent of the applicable APC payment amount for the service related to the category of devices; (2) the estimated average reasonable cost of the devices in the category exceeds the cost of the device-related portion of the APC payment amount for the related service by at least 25 percent; and (3) the difference between the estimated average reasonable cost of the devices in the category and the portion of the APC payment amount for the device exceeds 10 percent of the APC payment amount for the related service (with the exception of brachytherapy and temperature-monitored cryoablation, which are exempt from the cost requirements as specified at § 419.66(c)(3) and (e)); and
- Demonstrate a substantial clinical improvement, that is, substantially improve the diagnosis or treatment of an illness or injury or improve the functioning of a malformed body part compared to the benefits of a device or devices in a previously established category or other available treatment, or, for devices for which pass-through payment status will begin on or after January 1, 2020, as an alternative pathway to demonstrating substantial clinical improvement, a device is part of the FDA's Breakthrough Devices Program and has received marketing authorization for the indication covered by the Breakthrough Device designation.
In the CY 2016 OPPS/ASC final rule with comment period, we changed our device pass-through evaluation and determination process. Device pass-through applications are still submitted to CMS through the quarterly process, but the applications are subject to notice and comment rulemaking in the next applicable OPPS annual rulemaking cycle. Under this process, all applications that are preliminarily approved upon quarterly review will automatically be included in the next applicable OPPS annual rulemaking cycle, while submitters of applications that are not approved upon quarterly review will have the option of being included in the next applicable OPPS annual rulemaking cycle or withdrawing their application from consideration. Under this notice-and-comment process, applicants may submit new evidence, such as clinical trial results published in a peer-reviewed journal or other materials, for consideration during the public comment process for the proposed rule. This process allows those applications that we are able to determine meet all of the criteria for device pass-through payment under the quarterly review process to receive timely pass-through payment status, while still allowing for a transparent, public review process for all applications (80 FR 70417 through 70418).
In the CY 2020 OPPS/ASC final rule with comment period, we finalized an alternative pathway for devices that are granted a Breakthrough Device designation (84 FR 61295) and receive FDA marketing authorization for the indication covered by the Breakthrough Device designation. Under this alternative pathway, devices that are granted an FDA Breakthrough Device designation are not evaluated in terms of the current substantial clinical improvement criterion at § 419.66(c)(2) for the purposes of determining device pass-through payment status, but do need to meet the other requirements for pass-through payment status in our regulation at § 419.66. Devices that are part of the Breakthrough Devices Program, have received FDA marketing authorization for the indication covered by the Breakthrough Devices designation, and meet the other criteria in the regulation can be approved through the quarterly process and announced through that process (81 FR 79655). Proposals regarding these devices and whether pass-through payment status should continue to apply are included in the next applicable OPPS rulemaking cycle. This process promotes timely pass-through payment status for innovative devices, while also recognizing that such devices may not have a sufficient evidence base to demonstrate substantial clinical improvement at the time of FDA marketing authorization.
More details on the requirements for device pass-through payment applications are included on the CMS website in the application form itself at https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/HospitalOutpatientPPS/passthrough_payment.html, in the “Downloads” section. In addition, CMS is amenable to meeting with applicants or potential applicants to facilitate information sharing to support the evaluation of an OPPS device pass-through payment application or discuss general application criteria, including the substantial clinical improvement criterion.
In accordance with section V.B.9. of this final rule with comment period, skin substitutes with an approved Biologics License Application (BLA) will be considered under transitional drug pass-through payment status and skin substitutes with FDA Premarket approval (PMA) or FDA 510(k) clearance will continue to be evaluated under transitional device pass-through payment status.
b. Applications Received for Device Pass-Through Status for CY 2026
We received eight complete applications by the March 3, 2025, quarterly deadline, which was the last quarterly deadline for applications to be received in time to be included in the CY 2026 OPPS/ASC proposed rule. Of the complete applications, we received one application in the second quarter of 2024, three applications in the third quarter of 2024, one application in the fourth quarter of 2024, and three applications in the first quarter of 2025. One application was withdrawn. Two of the applications were approved for device pass-through payment during the quarterly review process: VasQ, which was preliminarily approved upon quarterly review under the alternative pathway effective October 1, 2024, and the SCOUT MDTM Surgical Guidance System which was preliminarily approved upon quarterly review under the alternative pathway effective January 1, 2025. As previously stated, all applications that are preliminarily approved upon quarterly review will automatically be included in the next applicable OPPS annual rulemaking cycle. Therefore, VasQ and the SCOUT MDTM Surgical Guidance System were discussed in section IV.2.b.1. of the CY 2026 OPPS/ASC proposed rule and within this section.
Applications received for the later deadlines for the remaining 2025 quarters (the quarters beginning June 1, September 1, and December 1 of 2025), if any, will be discussed in the CY 2027 OPPS/ASC proposed rule. We note that the quarterly application process and requirements have not changed because of the addition of rulemaking review. Detailed instructions on submission of a quarterly device pass-through payment application are included on the CMS website at https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/HospitalOutpatientPPS/Downloads/catapp.pdf.
Discussions of the applications we received by the March 3, 2025, deadline are included in section IV.2.b.1. of the CY 2026 OPPS/ASC proposed rule and in this section. We do not include in this final rule with comment period the description and discussion of new device category for transitional pass-through payment status applications ( printed page 53637) which were included in the CY 2026 OPPS/ASC proposed rule that were withdrawn for consideration for the upcoming calendar year. We also do not summarize or respond to public comments received regarding these withdrawn or ineligible applications.
Comment: One commenter opined that all of the nominated devices included in the CY2026 OPPS/ASC proposed rule meet the criteria for device pass-through. Another commenter encouraged CMS to approve or finalize all qualifying applications that meet the statutory criteria, especially those demonstrating clear clinical improvements. This commenter further noted that the substantial clinical improvement criterion should be applied rigorously and that innovations should earn supplemental payment only if they offer a real patient-care advantage, not merely convenience. This commenter commended CMS's careful scrutiny, particularly in regard to the single-use endoscope application, and urged CMS to continue evidence-based evaluations.
Response: We appreciate the commenters' input. We have taken these comments into consideration in our final determination for pass-through status for the nominated devices included in the CY 2026 OPPS/ASC proposed rule.
(1) Alternative Pathway Device Pass-Through Applications
As discussed in the CY 2026 OPPS/ASC proposed rule, we received four device pass-through applications by the March 2025 quarterly application deadline for devices that have received Breakthrough Device designation from FDA and FDA marketing authorization for the indication for which they have a Breakthrough Device designation and, therefore were eligible to apply under the alternative pathway.
(a) aprevo® Cervical ACDF System, aprevo® Cervical ACDF-X System, aprevo® Cervical ACDF-X NO CAM System
Carlsmed, Inc. submitted an application for a new device category for transitional passthrough payment status for the -aprevo® Cervical ACDF system, aprevo® Cervical ACDF-X system, and aprevo® Cervical ACDF-X NO CAM system (herein after collectively referred to as the aprevo® Cervical ACDF System) for CY 2026. Per the applicant, the aprevo® Cervical ACDF System is designed to stabilize the cervical spinal column and facilitate fusion. The applicant further explained that the personalized aprevo® Cervical ACDF System devices incorporate patient-specific features to allow the clinician to tailor the deformity correction to the individual needs of the patient and include an aperture for the packing of bone graft. Per the applicant, the aprevo® Cervical ACDF System includes the following components: (1) aprevo® implant, which includes two implants with slightly different heights for each vertebral level, (2) aprevo® insertion instrument, and (3) for the aprevo® Cervical ACDF-X system only, integrated fixation screws. The applicant further stated that the aprevo® Cervical ACDF-X NO CAM system does not have a part that blocks screws from backing out.
Please refer to the online application posting for the aprevo® Cervical ACDF System, available at https://mearis.cms.gov/public/publications/device-ptp/DEP250303GJ8LW.
Comment: The applicant noted that, after FDA authorization, it changed the devices' names from aprevo® Cervical ACDF system and aprevo® Cervical ACDF-X system to aprevo® Cervical ACDF interbody and aprevo® Cervical ACDF-X interbody, respectively.
Response: We appreciate the applicant's clarification. Based on the information available to us, we referred to the nominated technology as the aprevo® Cervical ACDF system, the aprevo® Cervical ACDF-X system, the aprevo® Cervical ACDF-X NO CAM system, and collectively as the aprevo® Cervical ACDF System in the CY 2026 OPPS/ASC proposed rule and will continue to refer to the technology as the aprevo® Cervical ACDF System in this final rule with comment period for clarity and consistency.
Comment: The applicant and a commenter expressed their support for approval of transitional pass-through payment for the aprevo® Cervical ACDF System, stating that they believe the device meets all the transitional pass-through payment criteria. The applicant also asserted that beneficiary access is at risk if CMS payment policies do not reflect the actual cost of providing custom-made, anatomically-designed interbody fusion device technology, like the aprevo® Cervical ACDF System, to more Medicare beneficiaries.
Response: We appreciate the commenters' input and support for the aprevo® Cervical ACDF System application for transitional pass-through payment. We have taken these comments into consideration in our final determination for pass-through status for the aprevo® Cervical ACDF System.
As stated previously, to be eligible for transitional pass-through payment under the OPPS, a device must meet the criteria at § 419.66(b)(1) through (4). With respect to the newness criterion at § 419.66(b)(1), the aprevo® Cervical ACDF System received FDA Breakthrough Device designation effective September 15, 2023, under the name aprevo®-C cervical interbody fusion device. The approved FDA indication for the aprevo® Cervical ACDF System is:
- For use in skeletally mature patients with degenerative cervical conditions including cervical disc degeneration, stenosis, deformity, and/or instability of the cervical spine (C2-T1) at one or more levels. DDD [30] is defined as discogenic pain with degeneration of the disc confirmed by history and radiographic studies. These patients should have had at least six (6) weeks of non-operative treatment. These devices are to be filled with autograft bone and/or allogenic bone graft composed of cancellous, cortical, and/or cortico-cancellous bone. The aprevo®-C cervical interbody fusion devices can be used with supplemental fixation, such as an anterior plate, or as a standalone construct to be used [with the] integrated bone screw fixation.
FDA granted the applicant 510(k) clearance for the aprevo® Cervical ACDF System on November 15, 2024, with separate indications for the aprevo® Cervical ACDF system and the aprevo® Cervical ACDF-X system (with and without CAM).[31] We note that while the indication for the FDA Breakthrough Device designation and the indication for the FDA 510(k) clearance for the aprevo® Cervical ACDF System vary, per FDA, the FDA 510(k) clearance indication is covered by the Breakthrough Device designation. We received the application for a new device category for transitional pass-through payment status for the aprevo® Cervical ACDF System on March 3, 2025, which is within 3 years of the date of the initial FDA marketing authorization.
We stated in the CY 2026 OPPS/ASC proposed rule that it was unclear to us whether the aprevo® Cervical ACDF system and the aprevo® Cervical ACDF-X system (with and without CAM) are different devices such that they should be evaluated separately for OPPS pass-through payment status. We noted that the aprevo® Cervical ACDF-X system ( printed page 53638) (with and without CAM) includes additional components, such as the integrated fixation screws, and has a different indicated use as stated in the November 15, 2024, FDA 510(k) clearance letter (K242260). Specifically, based on the FDA 510(k) clearance indication, we noted that a key difference of the aprevo® Cervical ACDF-X system (with and without CAM)'s interbody implant is that it incorporates integrated screw fixation and may be used as a standalone system for certain indications. We also noted that, for deformity procedures to correct coronal angulation or any use of hyperlordotic correction (≥20°), the aprevo® Cervical ACDF-X system (with and without CAM) must include supplemental fixation such as posterior cervical screw fixation or anterior plating.
We invited public comments on whether the aprevo® Cervical ACDF system and aprevo® Cervical ACDF-X system should be evaluated separately for OPPS pass-through payment status. Separately, we invited public comments on whether the aprevo® Cervical ACDF System meets the newness criterion at § 419.66(b)(1).
Comment: The applicant and a commenter expressed support for evaluating the aprevo® Cervical ACDF system and the aprevo® Cervical ACDF-X system together for pass-through payment status, stating that two configurations are part of the same system and noting that the difference between these two configurations is limited to supplemental fixation requirements. Specifically, the applicant stated that the aprevo® Cervical ACDF system requires supplemental fixation that is not provided in the kit, whereas the aprevo® Cervical ACDF-X system's kit includes interfixation screws. In addition, the applicant noted that the incremental cost of each screw is an additional 1.3 percent of the price of the single-level aprevo® Cervical ACDF system. The applicant claimed that this difference does not justify a separate pass-through category code.
Response: We appreciate the applicant's and commenter's clarification. We agree with the applicant that the aprevo® Cervical ACDF system and the aprevo® Cervical ACDF-X system should not be evaluated separately for transitional pass-through payment status. Therefore, we will review the aprevo® Cervical ACDF system and the aprevo® Cervical ACDF-X system together as the aprevo® Cervical ACDF System.
Comment: The applicant stated it agreed with CMS that the aprevo® Cervical ACDF systems are FDA Breakthrough-designated devices that meet the newness criterion. Further, the applicant stated that it submitted its transitional pass-through payment application for aprevo® Cervical ACDF System on March 3, 2025, which is within 3 years of the FDA marketing authorization date of November 15, 2024.
Response: We appreciate the applicant's input. We received the application for a new device category for transitional pass-through payment status for the aprevo® Cervical ACDF System within 3 years of the date of FDA 510(k) clearance. After consideration of the public comments we received and our review of the application, we have determined that the aprevo® Cervical ACDF System meets the newness criterion at § 419.66(b)(1).
With respect to the eligibility criteria at § 419.66(b)(3), the device must be an integral part of the service furnished, be used for one patient only, come in contact with human tissue, and be surgically inserted or implanted, or applied in or on a wound or other skin lesion. Per the applicant, the aprevo® Cervical ACDF System meets the requirements at § 419.66(b)(3).
With respect to the aprevo® Cervical ACDF System, in the CY 2026 OPPS/ASC proposed rule, we questioned whether the aprevo® insertion instrument, the integrated fixation screws, and/or the CAM components or parts are integral to the service furnished. We noted that, in the CY 2014 OPPS/ASC final rule with comment period (78 FR 75005), we stated that we have interpreted the term “integral” to mean that the device is necessary to furnish or deliver the primary procedure with which it is used. For example, a pacemaker is integral to the procedure of implantation of a pacemaker. Given our interpretation of integral, we questioned whether these components and parts of the aprevo® Cervical ACDF System are integral to the service furnished as it remains unclear which of these components and parts are utilized during the primary procedure and we questioned whether some of these components or parts may be purely additive in nature and not necessary to furnish the service. Specifically, we noted that it is unclear whether other available insertion instruments may be used to implant the aprevo® implant, and, for the aprevo® Cervical ACDF-X system, whether any or all of the integrated fixation screws may be replaced with other commercially available screws. In addition, we stated it was unclear whether the CAM is part of the aprevo® implant, part of the integrated fixation screws, or is a separate part altogether. We stated it was also unclear whether the CAM can be removed and replaced by other products, and whether there are any requirements for its utilization. We stated that we were interested in additional information about these components and parts of the aprevo® Cervical ACDF System, including how and when they are used, and whether they can be substituted with other products.
We invited public comments on whether the aprevo® Cervical ACDF System meets the eligibility criterion at § 419.66(b)(3).
Comment: In response to our concerns that all components or parts of the aprevo® Cervical ACDF System may not be integral parts of the service furnished because it is unclear how and when these components or parts are used and whether they can be substituted with other products, the applicant commented that the aprevo® insertion instrument, integrated fixation screws, and the CAM components are all part of the 510(k) cleared aprevo® Cervical ACDF System's kit, are integral to the implant and arthrodesis services furnished, and are not supplies that are incident to the procedure. The applicant stated that the aprevo® insertion instrument is an FDA class II instrument integral to the device systems and that other commercially available inserters are not FDA-authorized for use with or compatible with the aprevo® Cervical ACDF System. The applicant also noted that only the aprevo® insertion instrumentation, cleared by FDA, is authorized to implant the aprevo® Cervical interbody device, and these instruments are not available to be purchased separately for use with other devices. Further, the applicant stated that other commercially available screws are not authorized for use with or compatible with the aprevo® Cervical ACDF System; only the FDA-cleared aprevo® screws are authorized for use with the aprevo® Cervical ACDF-X system, and therefore, the fixation screws are integral to the device systems. The applicant also noted that these screws may not be purchased separately for use with other devices. Additionally, the applicant stated that the CAM is a non-separable, integral component of the FDA-cleared aprevo® Cervical ACDF-X system, cannot be removed from the interbody device, cannot be replaced by other products, and is designed to prevent screw backout. The applicant added that, once the integrated fixation screws have been inserted through the interbody device ( printed page 53639) and properly placed into the vertebral body, the CAM lock is rotated to cover a portion of the fixation screw's head, which is designed to prevent the screw from backing out. The applicant stated that these items cannot be substituted with other items.
Response: We appreciate the applicant's clarification. Based on the information provided in the comment, we agree with the applicant that all components of the aprevo® Cervical ACDF System, including the aprevo® insertion instrument, integrated fixation screws, and CAM, are integral parts of the service furnished. After consideration of the public comments we received and our review of the application, we have determined that the aprevo® Cervical ACDF System meets the eligibility criterion at § 419.66(b)(3).
With respect to the exclusion criteria at § 419.66(b)(4), a device is not eligible to be considered for pass-through payment if it is any of the following: (1) equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciable assets as defined in Chapter 1 of the Medicare Provider Reimbursement Manual (CMS Pub. 15-1); or (2) a material or supply furnished incident to a service (for example, a suture, customized surgical kit, or clip, other than a radiological site marker). Per the applicant, the aprevo® Cervical ACDF System is (1) not considered equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciable assets, and is (2) not a material or supply furnished incident to a service, and, therefore, is eligible to be considered for pass-through payment.
With respect to the aprevo® Cervical ACDF System, in the CY 2026 OPPS/ASC proposed rule, we questioned whether the aprevo® insertion instrument, the integrated fixation screws, and/or the CAM components or parts may be considered a material or supply furnished incident to the service. Specifically, as discussed previously with respect to criteria at § 419.66(b)(3), we noted that it is unclear whether other available insertion instruments may be used to implant the aprevo® implant and, for the aprevo® Cervical ACDF-X system, whether any or all of the integrated fixation screws may be replaced with other commercially available screws. In addition, we stated that we are unclear about whether the CAM is part of the aprevo® implant, part of the integrated fixation screws, or is a separate part altogether, and whether the CAM can be removed and replaced by other products. We requested clarification about each of these components and parts, including how and when they are used and whether they can be substituted with other commercially available products. We questioned whether these components or parts of the aprevo® Cervical ACDF System may be considered a supply or material furnished incident to a service and excluded from device pass-through payment eligibility under § 419.66(b)(4).
We invited public comments on whether the aprevo® Cervical ACDF System meets the criterion at § 419.66(b)(4).
Comment: The applicant submitted a response to our concerns that some components or parts of the aprevo® Cervical ACDF System, specifically the aprevo® insertion instrument, the integrated fixation screws, and/or the CAM, may be considered a material or supply furnished incident to the service, because, as previously discussed with respect to criteria at § 419.66(b)(3), it is unclear how and when these components or parts are used, and whether they can be substituted with other products. The applicant commented that the components of aprevo® Cervical ACDF System (insertion instrument, the integrated fixation screws, and the CAM) are part of the 510(k)-cleared aprevo® Cervical ACDF System and other commercially available insertion instruments and screws are not FDA authorized or compatible with the aprevo® Cervical ACDF System. In addition, the applicant commented that the CAM is a non-separable component of the aprevo® Cervical ACDF-X System and cannot be substituted with other items. Finally, the applicant noted that insertion instruments and screws are not available for purchase separately for use with other devices. Therefore, the applicant asserted that the components of aprevo® Cervical ACDF System are integral to the implant services furnished and are not supplies that are incident to the procedure.
Response: We appreciate the applicant's clarification. Based on the information provided in the comment and our review of the application, we agree with the applicant that the aprevo® Cervical ACDF System meets the criterion because the referenced components are only intended to be used with the aprevo® Cervical ACDF System, commercially available insertion instruments or screws cannot be utilized with the device, and the use of these components is necessary and/or required to furnish or deliver the primary procedure(s) with which it is used, and are not supplies incident to the procedure. As such, we have determined that the aprevo® Cervical ACDF System meets the criterion at § 419.66(b)(4).
In addition to the criteria at § 419.66(b)(1) through (4), the criteria for establishing new device categories are specified at § 419.66(c). The first criterion, at § 419.66(c)(1), provides that CMS determines that a device to be included in the category is not appropriately described by any of the existing categories or by any category previously in effect, and was not being paid for as an outpatient service as of December 31, 1996. Per the applicant, the existing pass-through code C1831 [32] (Interbody cage, anterior, lateral or posterior, personalized (implantable)) does not appropriately describe the aprevo® Cervical ACDF System because C1831 was created for the original (lumbar-specific) aprevo® product. According to the applicant, the aprevo® Cervical ACDF System device is different from C1831 because (1) the original (lumbar-specific) aprevo® and the nominated aprevo® Cervical ACDF System are separate and distinct products that have no overlap in anatomical indications for use or patient population; (2) the original (lumbar-specific) aprevo® and the aprevo® Cervical ACDF System are billed with different primary procedure CPT codes, are indicated for a different set of surgical approaches, are typically assigned to different places of service, and are mapped to different payment classifications; and (3) CMS transmittals state that C1831 is limited to lumbar procedures.
We noted, in the CY 2026 OPPS/ASC proposed rule, that based on the description provided by the applicant, that the aprevo® Cervical ACDF System is a personalized interbody cage that is implanted using an anterior surgical approach, and therefore, could be appropriately described by C1831. Specifically, we stated that C1831 may appropriately describe the aprevo® Cervical ACDF System because it describes any device that is a personalized interbody cage, designed for anterior, lateral, or posterior procedures. We noted that CMS does not establish pass-through device categories for the purposes of describing ( printed page 53640) specific devices, but rather, device categories which are intended to encompass all devices that can be appropriately described by a category. In this context, we stated that we believed that the aprevo® Cervical ACDF System may be similar to devices described by C1831 and therefore, the aprevo® Cervical ACDF System may be appropriately described by C1831.
We invited public comment on whether the aprevo® Cervical ACDF System meets the device category criterion at § 419.66(c)(1).
Comment: In response to our concerns that the aprevo® Cervical ACDF System may be appropriately described by C1831, the applicant and a commenter stated that they believe the aprevo® Cervical ACDF System meets all the criteria for establishing a new device category and asserted that CMS has used C1831 to describe personalized lumbar interbody fusion devices and that C1831 should not be used to describe the personalized cervical interbody fusion device that is the subject of this transitional pass-through payment application. The applicant and commenter further contended that CMS guidance indicates that CMS intended for C1831 to be lumbar-specific. In addition, the applicant noted that there are differences in the FDA indications, applicable CPT codes, and APC assignments for the nominated device and the original (lumbar-specific) aprevo® device. A commenter stated that the device categories differ in applicable CPT codes, APCs and FDA indications.
The applicant further summarized evidence included in its application to support its assertion that the aprevo® Cervical ACDF System is substantively distinct and significantly different from the expired device category C1831. The applicant reiterated that it believes that the original (lumbar-specific) aprevo® and the aprevo® Cervical ACDF System are separate and distinct products that have no overlap in FDA anatomical indications for use or patient population. The applicant stated that CMS has previously made similar distinctions between device categories with anatomical indications when it created device category codes C1748 (Endoscope, single-use ( i.e., disposable), upper GI, imaging/illumination device (insertable)), effective July 1, 2020, and subsequently created both C1747 (Endoscope, single use ( i.e., disposable), urinary tract, imaging/illumination device (insertable)), effective January 1, 2023, and C1601 (Endoscope, single-use ( i.e., disposable), pulmonary, imaging/illumination device (insertable)), effective January 1, 2024. Additionally, the applicant asserted that CMS previously made distinct device categories for anatomical locations when it created device category codes C1888 (Catheter, ablation, non-cardiac, endovascular (implantable)), effective July 1, 2002, and C1886 (Catheter, extravascular tissue ablation, any modality (insertable)), effective January 1, 2012.
The applicant and commenter also stated that the original (lumbar-specific) aprevo® and the aprevo® Cervical ACDF System are billed with different primary procedure CPT codes, are indicated for different surgical approaches, and are assigned to different APCs. Moreover, the applicant noted that anterior lumbar approaches are currently assigned to the Medicare Inpatient Only list and may not be performed in the outpatient setting.
In addition, the applicant and commenter noted that several prior CMS transmittals (#R10997CP, #R11004CP, and #R11801CP) have stated that C1831 is to only be used in lumbar fusion procedures. The applicant and commenter stated that the criteria to apply an existing category to a new device, as stated in § 419.66(f)(2), requires that the new device conforms to CMS guidance relating to the definition of terms and other information in conjunction with the category descriptors and codes. Furthermore, the applicant claimed that no CMS transmittals have stated that C1831 can be appropriately billed with cervical fusion CPT codes. The applicant and commenter asserted that the aprevo® Cervical ACDF System does not conform to CMS guidance related to the category codes, and therefore, they believe that C1831 is specific to the original (lumbar-specific) aprevo® and does not apply to the aprevo® Cervical ACDF System.
Response: We appreciate the commenters' input. After consideration of the public comments we received and our review of the application, we continue to believe that C1831 appropriately describes the aprevo® Cervical ACDF System, because the aprevo® Cervical ACDF System is a personalized interbody cage that is implanted using an anterior surgical approach, and C1831 describes any device that is a personalized interbody cage, designed for anterior, lateral, or posterior procedures.
We note that the applicant asserted that the aprevo® Intervertebral Fusion Device (IFD) (referred to as the original (lumbar-specific) aprevo® by the applicant) and the aprevo® Cervical ACDF System are separate and distinct products. We reiterate that CMS does not establish pass-through device categories for the purposes of describing specific devices. Rather, device categories are intended to encompass any device that can be appropriately described by the category, while ensuring that no medical device is described by more than one category in accordance with section 1833(t)(6)(B)(ii)(II) of the Act. When we evaluate a potential pass-through device to determine whether it meets the device category criterion at § 419.66(c)(1), we compare the nominated device to the device category descriptor rather than to the specific device for which the device category was created. Section 419.66(f) states that a device is described by a category if, (1) it matches the long descriptor of the category code established by CMS, and (2) conforms to the guidance issued by CMS relating to the definition of terms and other information in conjunction with the category descriptors and codes. Per the applicant, the aprevo® Cervical ACDF System is a personalized interbody cage that is implanted using an anterior surgical approach, which matches the descriptor for C1831 (interbody cage, anterior, lateral or posterior, personalized (implantable)). We also believe that the aprevo® Cervical ACDF System conforms to the guidance issued on C1831 as discussed in the following paragraphs.
We disagree with the applicant's assertion in its application and comment that C1831 is “de facto lumbar-specific” and that CMS intended to limit C1831 to lumbar procedures. We note that CMS establishes the reportable procedure codes for each device pass-through category based on the information available at the time in which the code is established and, unless specified in the descriptor, device category codes are not anatomically specific. CMS has included specific anatomic language in the descriptors of previous device category codes when such language was necessary based on the unique circumstances surrounding the establishment of a particular device category code, as acknowledged in the CY 2025 OPPS/ASC final rule with comment period (89 FR 94137). When CMS established C1831 as a new device category based on the approval of the aprevo® (IFD) application for transitional pass-through payment status, there were no circumstances which warranted the use of vertebrae-specific language in the long descriptor, as such, none was included (87 FR 71894 and 71895). The applicant acknowledged this in its device pass- ( printed page 53641) through payment application for the aprevo® Cervical ACDF System, stating that when CMS established C1831 there was no need to make anatomical distinctions as it was the first and only personalized interbody cage. We agree with the applicant that no vertebral distinctions were warranted because C1831 was established to describe any device that is a personalized interbody cage, designed for anterior, lateral, or posterior procedures. The aprevo® Cervical ACDF System is a device that is a personalized interbody cage, designed for anterior procedures, and as such we believe that the aprevo® Cervical ACDF System is described by C1831.
As noted by the applicant and acknowledged, CMS has established anatomically specific device category codes in the past, specifically for endoscopes and some ablation catheters. However, the device category codes referenced by the applicant were intentionally defined with anatomically specific descriptors from their inception ( e.g., C1748 (Endoscope, single-use ( i.e., disposable), upper GI, imaging/illumination device (insertable)) followed by C1747 (Endoscope, single use ( i.e., disposable), urinary tract, imaging/illumination device (insertable)), and C1601 (Endoscope, single-use ( i.e., disposable), pulmonary, imaging/illumination device (insertable)), as well as C1888 (Catheter, ablation, non-cardiac, endovascular (implantable)) followed by C1886 (Catheter, extravascular tissue ablation, any modality (insertable))). The anatomical specificity included in these device descriptors demonstrated our intention that these device category codes be anatomically specific. We further note that the descriptor for C1831 contains no such anatomical distinction within the spinal column, which demonstrates that CMS did not intend C1831 to be anatomically specific within the spinal column as the applicant and commenter believe.
We also disagree with the applicant and commenter's assertion that C1831 does not describe the aprevo® Cervical ACDF System because the aprevo® (IFD) and the aprevo® Cervical ACDF System are billed with different primary CPT procedure codes and assigned to different APCs. First, as previously stated, the procedure codes with which a device category code may be reported are approved when the device category code is established and are based on the current indication of the subject device and information available to us at the time. For the reasons discussed, the list of reportable procedure codes approved for a device category code is not intended to create a de facto determination regarding other devices or exclude devices that may also be appropriately described by the device category code descriptor. It is also not intended to change, restrict, or redefine the device category code in any manner that is inconsistent with the device category code descriptor.
Second and relatedly, the list of procedure codes with which device category codes may be reported is not unalterable. The list of approved procedure codes with which the device category code may be reported can, and do, change as required throughout the device pass-through payment eligibility period. Reportable procedures are updated for many reasons, including FDA approval of a new indication for an existing device or of a new device with an indication that includes additional procedures that are reportable with an existing device category code, necessitating the inclusion of those additional procedure codes in the reportable procedures list. For example, when CMS established C2623 as a device category code effective April 1, 2015, the procedure codes with which C2623 could be reported (HCPCS codes 37224 and 37226) were limited to use in the femoral or popliteal arteries (89 FR 94136). However, based on FDA approval of a new indication for an existing device (a drug-coated balloon catheter for use with dialysis circuit procedures for the treatment of patients with dysfunctional arteriovenous fistulae), CMS added two procedure codes, HCPCS codes 36902 and 36903 (transluminal balloon angioplasty procedures in peripheral dialysis segments), with which C2623 could be reported effective August 25, 2017.[33] As another example, after we established C1748 as a device category code effective July 1, 2020, we updated the list of procedure codes (HCPCS codes 43260 through 43265 and HCPCS codes 43274, 43276 through 43278) associated with HCPCS code C1748 in 2022 to include transnasal services (HCPCS codes 0652T, 0653T, 0654T, 43197, and 43198).[34] In line with these examples, had other devices that were appropriately described by C1831 received marketing authorization during the time C1831 was eligible for device pass-through payments, CMS likely would have added the appropriate additional procedure codes with which C1831 could be reported. In this instance, we believe HCPCS codes 22551 and 22554 (the HCPCS codes with which the aprevo® Cervical ACDF System is reported) could have been included in the list of reportable procedure codes for C1831 either when the device category code was first established or during the device category code status eligibility period, if the nominated device had received FDA marketing authorization. Third, beginning October 1, 2024, upon the expiration of device pass-through payment status for C1831, CMS packaged the payment for the costs of each of the devices described by C1831 into the payment for the costs related to the procedure with which each device is reported in the hospital claims data.[35 36] We note that upon becoming packaged for payment, C1831 effectively became reportable with other musculoskeletal procedure codes, including procedure codes for cervical-specific procedures. Additionally, we note that since payment has been packaged for C1831, cervical procedure codes, including HCPCS code 22551 (Arthrodesis, anterior interbody, including disc space preparation, discectomy, osteophytectomy and decompression of spinal cord and/or nerve roots; cervical below C2), one of the two procedure codes with which the aprevo® Cervical ACDF System may be billed, have been performed and billed with C1831. As such, we believe that the aprevo® Cervical ACDF System is appropriately described by C1831 and that CMS is already collecting cost data for the nominated device in the appropriate APC. Regarding the APC assignment, while we acknowledge that the aprevo® Cervical ACDF System and the aprevo® (IFD) are currently assigned to different APCs, we note that many device category codes are reportable with multiple APCs and HCPCS (procedure) codes, and therefore, we do not agree with the applicant or the commenter that this warrants the establishment of a new device category code. We do not believe, in this case, that the procedure codes and APC ( printed page 53642) assignments distinguish the aprevo® Cervical ACDF System from the aprevo® (IFD) device for purposes of the device category code determination.
Similarly, we disagree with the applicant and commenters' assertion that the CMS transmittals limit C1831 to lumbar procedures. The purpose of CMS transmittals is to describe changes to, and billing instructions for, various payment policies implemented for a specified time period. The guidance for device pass-through category codes is based on the current applicable indications and is not intended to restrict or redefine a device category code descriptor. Since device pass-through payment status expired for C1831, CMS transmittals have appropriately described the changes to, and updated billing instructions for C1831, and explained that C1831 would remain an active code and its payment would be included in the primary service. We believe hospitals are very familiar with the approval and expiration of transitional pass-through status for device category codes and how to correctly report device costs under the OPPS.
We also disagree with the applicant's assertion that the aprevo® (IFD) and the nominated aprevo® Cervical ACDF System are separate and distinct products that have no overlap in anatomical indications for use or patient population. We note that a commenter indicated that the device categories differ in FDA indications. While we are not clear to which other device categories the commenter is referring, we believe there is only one relevant device category code, C1831, we believe the FDA indications are similar and note that both aprevo® devices treat many of the same conditions, including degenerative disc disease, deformity (kyphosis or scoliosis), spinal stenosis, and failed previous fusion, at different levels of the spine.[37] Moreover, the aprevo® Cervical ACDF System and the aprevo® (IFD) appear to be intended for use in the same, or very similar procedures, differentiated only by the section of the spinal column in which the procedure is performed. As such, we disagree with the applicant that the devices have no overlap in anatomical indication and that they treat a different patient population.
We note that for the purposes of transitional pass-through payments, if we determine that a device is not appropriately described by any of the existing device categories or by any category previously in effect, then we issue a new category code.[38] Unlike previously established device category codes where CMS has made an anatomical specification based on a clear anatomical distinction in the device indication, such as a ureter and the bronchi, a different indication such as drug-delivery rather than angioplasty, or different mechanisms of action like radiofrequency versus ultrasound, with regard to the nominated technology, we did not believe at the time the code was established, nor do we believe at this time, that such an anatomical distinction is necessary for C1831. As such, we do not believe that the anatomical indications for different regions of the spinal column sufficiently distinguish the aprevo® Cervical ACDF System from the aprevo® (IFD) for purposes of the device category code determination.
Further, even though the aprevo® Cervical ACDF System is only placed through an anterior surgical approach and the aprevo® (IFD) may be placed through multiple surgical approaches, we note that per the device category code descriptor, C1831 is intended to cover all surgical approaches to the spine: anterior, lateral, or posterior. In the CY 2023 OPPS/ASC final rule with comment period (87 FR 71894 through 71895), we updated the device descriptor for C1831, effective January 1, 2023, to include the posterior/transforaminal approach at the request of the applicant.[39] Moreover, we declined the applicant's request to remove the anterior and lateral approaches from the descriptor and, instead, stated that the anterior and lateral approaches should remain in the descriptor and clarified our intent that the descriptor address all potential surgical approaches. We note, that while the C1831 descriptor was updated effective January 1, 2023, the anterior surgical approach—the surgical approach for which the aprevo® Cervical ACDF System is indicated—has been included in the category's descriptor since its establishment and, as previously noted, cervical-specific procedures using the anterior surgical approach have been reported with C1831 in the outpatient setting. HCPCS codes 22630 and 22633 (which can be reported with C1831) and HCPCS codes 22551 and 22554 (the aprevo® Cervical ACDF System procedure codes) are procedures that can be performed in the outpatient setting.
Finally, we disagree with the applicant's assertion that the healthcare setting in which procedures utilizing the aprevo® Cervical ACDF System device and the aprevo® (IFD)are typically performed has relevance to the device category discussion. We note that both devices may be used in the outpatient setting, as such, we do not believe that the surgical approach or healthcare setting distinguishes the aprevo® Cervical ACDF System from the aprevo® (IFD) for purposes of the device category code assignment.
After consideration of the public comment we received and our review of the device pass-through application, we have determined that the aprevo® Cervical ACDF System does not meet the device category eligibility criterion at § 419.66(c)(1) because it is appropriately described by an existing category or a category previously in effect. Therefore, in this final rule with comment period, we will not address whether the technology meets the other remaining criteria required for transitional pass-through payment for devices. We are not approving the aprevo® Cervical ACDF System for transitional pass-through payment status for CY 2026 because the technology does not meet the device category eligibility criterion at § 419.66(c)(1).
Comment: The applicant commented that CMS assigned a $0.00 device offset amount associated with the device pass-through payment for the original (lumbar-specific) aprevo® and requested that CMS be consistent with this policy determination and assign a $0.00 to the aprevo® Cervical ACDF System device pass-through payment.
Response: We appreciate the applicant's input regarding the device offset amount associated with the ( printed page 53643) aprevo® Cervical ACDF System; however, as the device pass-through application for the aprevo® Cervical ACDF System has not been approved in this final rule with comment period, we will not address the device offset assignment.
Comment: The applicant expressed concern that the placement of the anterior and lateral procedures associated with the original (lumbar-specific) aprevo® on the IPO list effective January 1, 2022 following the approval of device pass-through payment status for C1831 effective October 1, 2021 prevented CMS from collecting sufficient and proper cost data to assign the procedures to an appropriate APC following the expiration of device pass-through payment status on September 30, 2024. The applicant commented that it was unable to market the original (lumbar-specific) aprevo® in the outpatient setting due to this change and that limited applicable use of the anterior and lateral surgical approaches and inconsistent coding guidance for the posterior surgical approach disincentivized adoption. As such, the applicant requested that CMS reinstate C1831 to ensure a full 3 years of device pass-through payment status and to capture the necessary cost data.
Response: We thank the commenters for their input. We appreciate the applicant's concern with the timing of the placement of the anterior and lateral procedures on the IPO list, however, we cannot reinstate the pass-through payment status of HCPCS code C1831, neither do we agree we should. Consistent with section 1833(t)(6)(B)(iii) of the Act and § 419.66(g), the period for which a device category for transitional pass-through payments under the OPPS can be in effect is at least 2 years, but not more than 3 years, beginning on the first date on which pass-through payment is made. Once 3 years has passed since a device category first received transitional pass-through payments, the device category is no longer eligible for pass-through payments and we utilize the established policy (first described in the CY 2003 OPPS/ASC final rule, 67 FR 66763) to package the costs of the devices that are no longer eligible for pass-through payments into the costs of the procedures with which the devices are reported in the claims data used to set the payment rates. We note that device pass-through payment status is intended to be temporary, and we consider the cost data to be included in the payment rates regardless of whether the technology's use in the Medicare population has been frequent or infrequent during the time period under which a device was receiving transitional pass-through payments. The C1831 device category was made effective in the OPPS on October 1, 2021, and expired on September 30, 2024, as such, we cannot reinstate the pass-through payment status of C1831 because reinstatement would make the pass-through payment status effective longer than the maximum 3-year period permitted under section 1833(t)(6)(B)(iii) of the Act and § 419.66(g).
(b) SCOUT MDTM Surgical Guidance System
Merit Medical Systems submitted an application for a new device category for transitional pass-through payment status for the SCOUT MDTM Surgical Guidance System for CY 2026. According to the applicant, the SCOUT MDTM Surgical Guidance System communicates the location of tumor tissue during a tumor excision procedure. Per the applicant, the SCOUT MDTM Surgical Guidance System consists of the SCOUT MDTM Delivery System, SCOUT MDTM Guide, SCOUT MDTM Handpiece, and SCOUT MDTM Console.
The applicant stated that it is only seeking a new device category for transitional pass-through payment status for the SCOUT MDTM Delivery System component of the SCOUT MDTM Surgical Guidance System. The SCOUT MDTM Delivery System consists of the SCOUT MDTM Reflectors and the SCOUT MDTM Delivery Device, a plastic, molded handle attached to a 16 GA introducer needle with a SCOUT MDTM Reflector preloaded inside. According to the applicant, the SCOUT MDTM Delivery System is used to implant each of the SCOUT MDTM Reflectors, which identify the location of the tumor tissue to be excised and/or the boundaries of the region of tissue to be excised during a separately scheduled procedure. The applicant further explained that there are four unique configurations of the SCOUT MDTM Reflectors, which return a detectable signal within surrounding tissue when illuminated by the micro-impulse radar signal from the SCOUT MDTM Guide and Handpiece used during the tumor excision procedure. Per the applicant, each single-use SCOUT MDTM Delivery System contains one SCOUT MDTM Delivery Device with one preloaded SCOUT MDTM Reflector.
Please refer to the online application posting for the SCOUT MDTM Surgical Guidance System, available at https://mearis.cms.gov/public/publications/device-ptp/DEP240830W9M8U.
As stated previously, to be eligible for transitional pass-through payment under the OPPS, a device must meet the criteria at § 419.66(b)(1) through (4). With respect to the newness criterion at § 419.66(b)(1), the SCOUT MDTM Surgical Guidance System received FDA Breakthrough Device designation effective February 1, 2023. The approved FDA indication for the SCOUT MDTM Surgical Guidance System is:
- The SCOUT MD Reflectors are intended to be placed percutaneously in soft tissue, (>30 days) [40] to mark a biopsy site or a soft tissue site intended for surgical removal. Using imaging guidance (such as ultrasound, MRI, or radiography) or aided by non-imaging guidance (SCOUT MD System), the SCOUT MD Reflector is located and surgically removed with the target tissue. The SCOUT MDTM Delivery System is intended only for the non-imaging detection and localization of the SCOUT MD Reflector that has been implanted in a soft tissue biopsy site or a soft tissue site intended for surgical removal.
FDA granted 510(k) clearance for the SCOUT MDTM Surgical Guidance System on February 12, 2024, for the same indication as the one covered by the Breakthrough Device designation. We received the application for a new device category for transitional pass-through payment status for the SCOUT MDTM Surgical Guidance System on August 30, 2024, which is within 3 years of the date of the initial FDA marketing authorization.
We invited public comments on whether the SCOUT MDTM Surgical Guidance System, inclusive of the SCOUT MDTM Delivery System meets the newness criterion at § 419.66(b)(1).
Comment: The applicant reiterated that the SCOUT MDTM Surgical Guidance System received FDA clearance on February 12, 2024, and the application for transitional pass-through payment status for the SCOUT MDTM Surgical Guidance System was submitted on August 30, 2024 which is within 3 years from the date of FDA clearance.
Response: We appreciate the applicant's input. We agree that we received the application for a new device category for transitional pass-through payment status for the SCOUT MDTM Surgical Guidance System within 3 years of the date of FDA 510(k) clearance. After consideration of the public comment we received and our ( printed page 53644) review of the application, we have determined that the SCOUT MDTM Surgical Guidance System meets the newness criterion at § 419.66(b)(1).
As previously noted, the applicant is only seeking a new device category for transitional pass-through payment status for the SCOUT MDTM Delivery System component of the SCOUT MDTM Surgical Guidance System; as such, the eligibility and exclusion criteria will evaluate SCOUT MDTM Delivery System.
With respect to the eligibility criteria at § 419.66(b)(3), the device must be an integral part of the service furnished, be used for one patient only, come in contact with human tissue, and be surgically inserted or implanted, or applied in or on a wound or other skin lesion. Per the applicant, the SCOUT MDTM Delivery System meets the requirements at § 419.66(b)(3).
We invited public comments on whether the SCOUT MDTM Delivery System meets the eligibility criterion at § 419.66(b)(3).
Comment: The applicant reiterated that the SCOUT MDTM Delivery System meets all the eligibility criteria.
Response: We appreciate the applicant's input. After consideration of the public comment we received and our review of the application, we agree with the applicant and have determined that the SCOUT MDTM Delivery System meets the eligibility criterion at § 419.66(b)(3).
With respect to the exclusion criteria at § 419.66(b)(4), a device is not eligible to be considered for pass-through payment if it is any of the following: (1) equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciable assets as defined in Chapter 1 of the Medicare Provider Reimbursement Manual (CMS Pub. 15-1); or (2) a material or supply furnished incident to a service (for example, a suture, customized surgical kit, or clip, other than a radiological site marker). Per the applicant, the SCOUT MDTM Delivery System is (1) not considered equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciable assets, and is (2) not a material or supply furnished incident to a service, and, therefore, is eligible to be considered for pass-through payment.
We invited public comments on whether the SCOUT MDTM Delivery System meets the exclusion criterion at § 419.66(b)(4).
Comment: The applicant reiterated that the SCOUT MDTM Delivery System is not a depreciating asset as defined in Chapter 1 of the Medicare Provider Reimbursement Manual (CMS Pub. 15-1) nor is it a material or supply furnished incident to a service.
Response: We appreciate the applicant's input. After consideration of the public comment we received and our review of the application, we agree with the applicant and have determined that the SCOUT MDTM Delivery System meets the criterion at § 419.66(b)(4).
In addition to the criteria at § 419.66(b)(1) through (4), the criteria for establishing new device categories are specified at § 419.66(c). The first criterion, at § 419.66(c)(1), provides that CMS determines that a device to be included in the category is not appropriately described by any of the existing categories or by any category previously in effect, and was not being paid for as an outpatient service as of December 31, 1996. Per the applicant, the existing pass-through codes C1879 [41] (Tissue marker (implantable)) and C1819 (Tissue localization excision device) do not appropriately describe the SCOUT MDTM Delivery System because the SCOUT MDTM Delivery System is different than other wire-free localization/fiducial devices used for breast conserving surgery and is the only device that: (1) incorporates application-specific integrated circuit (ASIC) technology customized for use with the SCOUT MDTM Surgical Guidance System; (2) uses radar technology to detect, locate and identify the implanted reflector(s) within ±1 millimeter (mm) of accuracy; (3) utilizes up to four uniquely shaped reflectors for a more clearly defined radiographic image of the area of interest to be excised; (4) incorporates differentiated radar signatures and detection cadences specific to each reflector; (5) can detect up to four unique reflectors simultaneously or individually to more precisely identify pre-defined surgical margins; and (6) has no significant MRI artifact. The applicant further explained that the SCOUT MDTM Delivery System includes four distinct implant (SCOUT MDTM Reflector) shapes, each with a unique radar signature that enables clear detection and identification of the multiple localization devices previously placed to mark the desired surgical margins during the excision procedure. Upon review, we stated in the CY 2026 OPPS/ASC proposed rule that we did not identify an existing pass-through payment category that describes the SCOUT MDTM Delivery System.
We invited public comment on whether the SCOUT MDTM Delivery System meets the device category criterion at § 419.66(c)(1).
Comment: The applicant reiterated that the SCOUT MDTM Delivery System is not appropriately described by any of the existing categories or by any category previously in effect, and that it has not been paid for as an outpatient service as of December 31, 1996.
Response: We appreciate the applicant's input. After consideration of the public comment we received and our review of the application, we continue to believe that there is no existing category or category previously in effect that appropriately describes the SCOUT MDTM Delivery System. Therefore, we have determined that the SCOUT MDTM Delivery System meets the device category eligibility criterion at § 419.66(c)(1).
The second criterion for establishing a device category, at § 419.66(c)(2), provides that CMS determines either of the following: (1) that a device to be included in the category has demonstrated that it will substantially improve the diagnosis or treatment of an illness or injury or improve the functioning of a malformed body part compared to the benefits of a device or devices in a previously established category or other available treatment; or (2) for devices for which pass-through status will begin on or after January 1, 2020, as an alternative to the substantial clinical improvement criterion, the device is part of the FDA's Breakthrough Devices Program and has received FDA marketing authorization for the indication covered by the Breakthrough Device designation. The SCOUT MDTM Surgical Guidance System has a Breakthrough Device designation and marketing authorization from FDA for the indication covered by the Breakthrough Device designation (as explained in more detail of the newness criterion) and therefore was not evaluated for substantial clinical improvement.
We invited public comment on whether the SCOUT MDTM Delivery System meets the device category criterion at § 419.66(c)(2).
Comment: The applicant confirmed that the SCOUT MDTM Delivery System received a Breakthrough Device designation from FDA. The applicant also asserted that it believes that, even if the SCOUT MDTM Delivery System did not have Breakthrough Device designation, the device would still meet ( printed page 53645) the device category criterion at § 419.66(c)(2).
Response: We appreciate the applicant's input. The SCOUT MDTM Surgical Guidance System, inclusive of the SCOUT MDTM Delivery System, has a Breakthrough Device designation effective February 1, 2023, and marketing authorization from FDA effective February 12, 2024, for the indication covered by the Breakthrough Device designation. Therefore, the SCOUT MDTM Surgical Guidance System meets the criterion at 419.66(c)(2)(ii) and is not evaluated for substantial clinical improvement at § 419.66(c)(2)(i). After consideration of the public comment we received and our review of the application, we have determined that the SCOUT MDTM Delivery System meets the device category criterion at § 419.66(c)(2).
The third criterion for establishing a device category, at § 419.66(c)(3), requires CMS to determine that the cost of the device is not insignificant, as described in § 419.66(d). Section 419.66(d) includes three cost significance criteria that must each be met. The applicant stated that the SCOUT MDTM Delivery System would be reported with HCPCS codes as shown in Table 100, previously published as Table 49 of the CY 2026 OPPS/ASC proposed rule (90 FR 33579).
To meet the cost criterion for device pass-through payment status, a device must pass all three tests of the cost criterion for at least one APC. As we explained in the CY 2005 OPPS final rule with comment period (69 FR 65775), we generally use the lowest APC payment rate applicable for use with the nominated device when we assess whether a device meets the cost significance criterion, thus increasing the probability the device will pass the cost significance test. Beginning in CY 2017, we calculate the device offset amount at the HCPCS/CPT code level instead of the APC level (81 FR 79657). We noted in the CY 2026 OPPS/ASC proposed rule that the applicant utilized the CY 2025 payment rates for the three tests of the cost criterion. For our calculations, we used APC 5071, which had a CY 2025 payment rate of $703.59 at the time the application was received. HCPCS code 19287 in APC 5071 had a device offset amount of $240.56 at the time the application was received.[42] Per the applicant, an average of 1.95 SCOUT MDTM Reflectors are placed per procedure with a selling price of $550.00 for each single-use SCOUT MDTM Delivery System containing a SCOUT MDTM Delivery Device with one preloaded SCOUT MDTM Reflector. Therefore, according to the applicant, the average cost per procedure for the SCOUT MDTM Delivery System is $1,072.00.
Section 419.66(d)(1), the first cost significance requirement, provides that the estimated average reasonable cost of devices in the category must exceed 25 percent of the applicable APC payment amount for the service related to the category of devices. The average reasonable cost of $1,072.00 for the SCOUT MDTM Delivery System is 152.36 percent of the applicable APC payment amount for the service related to the category of devices, of $703.59 (($1,072.00/$703.59 × 100 = 152.36 percent). Therefore, we stated in the CY 2026 OPPS/ASC proposed rule that we believe that the SCOUT MDTM Delivery System meets the first cost significance requirement.
The second cost significance requirement, at § 419.66(d)(2), provides that the estimated average reasonable cost of the devices in the category must exceed the cost of the device-related portion of the APC payment amount for the related service by at least 25 percent, which means that the device cost needs to be at least 125 percent of the offset amount (the device-related portion of the APC found on the offset list). The estimated average reasonable cost of $1,072.00 for the SCOUT MDTM Delivery System is 445.63 percent of the cost of the device-related portion of the APC payment amount for the related service, of $240.56 ($1,072.00/$240.56 × 100 = 445.63 percent). Therefore, we stated in the CY 2026 OPPS/ASC ( printed page 53646) proposed rule that we believe that the SCOUT MDTM Delivery System meets the second cost significance requirement.
The third cost significance requirement, at § 419.66(d)(3), provides that the difference between the estimated average reasonable cost of the devices in the category and the portion of the APC payment amount for the device must exceed 10 percent of the APC payment amount for the related service. The difference between the estimated average reasonable cost of $1,072.00 for the SCOUT MDTM Delivery System and the portion of the APC payment amount for the device of $240.56 is 118.17 percent of the APC payment amount for the related service, of $703.59 ((($1,072.00−$240.56)/$703.59) × 100 = 118.17 percent). Therefore, we stated in the CY 2026 OPPS/ASC proposed rule that we believe that the SCOUT MDTM Delivery System meets the third cost significance requirement.
We invited public comment on whether the SCOUT MDTM Delivery System meets the cost criterion at § 419.66(c)(3).
Comment: The applicant reiterated that the SCOUT MDTM Delivery System meets all three of the cost significance criteria and confirmed that the cost analysis in the CY 2026 OPPS/ASC proposed rule is accurate.
Response: We appreciate the applicant's input. We agree with the applicant and continue to believe that the SCOUT MDTM Delivery System meets the first, second, and third cost significance tests described at § 419.66(d).
As discussed previously in this section, we preliminarily approved the SCOUT MDTM Delivery System application for transitional pass-through payment under the alternative pathway effective January 1, 2025. After consideration of the public comment we received and our review of the device pass-through application, we have determined that the SCOUT MDTM Delivery System meets the requirements for device pass-through status described at § 419.66. We are finalizing approval for device pass-through payment status for the SCOUT MDTM Delivery System under the alternative pathway for devices that have an FDA Breakthrough Device designation and have received FDA marketing authorization for the indication covered by the Breakthrough Device designation.
Comment: The applicant requested that CMS withdraw C1739, the device category code established upon the preliminary approval of the SCOUT MDTM Delivery System application for device pass-through payment status and issue a new HCPCS code with descriptor language including, “radar detectable implantable reflectors”, effective January 1, 2026, to ensure that the SCOUT MDTM Delivery System receives the full 3 years of pass-through status. The applicant asserted that the code descriptor for C1739, “tissue marker, probe detectable any method (implantable), with delivery system,” is overly broad, describes numerous legacy tissue marker systems that the applicant believes are not eligible for device pass-through, and does not appropriately differentiate the SCOUT MDTM Delivery System or other similar systems from the wide range of existing tissue markers currently on the market. The applicant stated it believes this change is necessary so that only those devices truly eligible for device pass-through status can use the HCPCS code and so that the claims data appropriately reflects accurate billing.
The applicant further asserted that the code description for C1739 has allowed thousands of claims to be submitted by hospitals for devices not eligible for pass-through payment, resulting in millions of dollars of waste and abuse. Specifically, the applicant asserted that as of the end of June 2025, over 2,000 claims have been submitted using C1739 for tissue markers which were commercialized before the issuance of C1739. The applicant clarified that not one of those claims have been for the use of the SCOUT MDTM Delivery System, because the device is not yet commercially available. The applicant estimated that those 2,000 claims would have been paid under APC 5071, but for the creation of C1739, and resulted in $2,000,000 of waste. The applicant further projected that continued use of the current descriptor would result in $4,000,000-$5,000,000 of waste by the end of 2025 and could represent as much as $32,000,000 to $64,000,000 of waste over the next 2 years.
Finally, the applicant stated that, under the regulation at § 419.66(a), CMS makes a pass-through payment for a medical device that meets the requirements in paragraph (b) of that section and that is described by a category of devices established by CMS under the criteria in paragraph (c) of that section, and that, further, according to § 419.66(g), the pass-through period begins on the first date on which pass-through payment is made. The applicant asserted that while CMS has been paying for devices submitted using C1739, none of those payments are pass-through payments because CMS has not paid for the SCOUT MDTM Delivery System or any other devices that would be described by a device pass-through category code with an appropriately specific descriptor. Because the device pass-through category code descriptor is overly broad, according to the applicant, the agency has been paying for devices that do not meet the eligibility requirements in paragraph (b) or the criteria for establishing device categories in paragraph (c) and thus, has not been making pass-through payments. As such, the applicant requested that CMS withdraw C1739 and issue a new device pass-through category code effective January 1, 2026 with a descriptor that describes only the SCOUT MDTM Delivery System and any devices legitimately eligible for pass-through status.
Response: Although we acknowledge the applicant's concerns regarding the descriptor for C1739, we do not agree that the device category code should be withdrawn and replaced with a new code effective January 1, 2026, for the following reasons:
First, per the applicant's request prior to CY 2026 rulemaking, CMS revised the descriptor for C1739 to read, effective October 1, 2025: Tissue marker, uniquely detectable and identifiable with probe/sensor, any method (implantable), with delivery system. The short descriptor was also revised to: Marker unique detect w/probe. These revisions were retroactive to January 1, 2025.[43] We believe that the updated descriptor accurately reflects the devices intended to be described by C1739, inclusive of the SCOUT MDTM Delivery System, and addresses the applicant's concerns that the prior descriptor was overly broad.
Second, we wish to reiterate that device category codes are not device specific; rather, CMS establishes device categories that are intended to encompass all devices that can be appropriately described by the category under § 419.66. We disagree with the applicant's assertion that CMS has not made any pass-through payments for C1739. Any device described by the descriptor associated with a currently payable device pass-through category code qualifies for pass-through payment. The fact that no claims have purportedly been submitted for the SCOUT MDTM Delivery System does not mean that no appropriate pass-through payments have been made for C1739. As such, we believe that appropriate pass-through payments for C1739 have been made in accordance with § 419.66(g). ( printed page 53647)
We further note that, consistent with section 1833(t)(6)(B)(iii) of the Act and § 419.66(g), the period for which a device category for transitional pass-through payments under the OPPS can be in effect is at least 2 years, but not more than 3 years, beginning on the first date on which pass-through payment is made. Once 3 years has passed since a device category first received transitional pass-through payments, the device category is no longer eligible for pass-through payments and we utilize the established policy (first described in the CY 2003 OPPS/ASC final rule, 67 FR 66763) to package the costs of the devices that are no longer eligible for pass-through payments into the costs of the procedures with which the devices are reported in the claims data used to set the payment rates. Moreover, device pass-through payment status is intended to be temporary, and we consider the cost data to be included in the payment rates regardless of whether the technology's use in the Medicare population has been frequent or infrequent during the time period under which a device was receiving transitional pass-through payments.
Finally, we agree with the applicant that the pass-through period begins on the first date on which pass-through payment is made in accordance with § 419.66(g), which we finalized in the CY 2017 OPPS/ASC final rule with comment period (81 FR 79654 and 79655). Therefore, for the purposes of C1739, CMS considers the start of the device pass-through payment status eligibility period to be January 1, 2025, and, as such, CMS does not agree with the applicant that the device category code for C1739 should be withdrawn, and a new code issued effective January 1, 2026. We believe that the updated device category code descriptor for C1739 issued on October 1, 2025, addresses the applicants' concerns and that the necessary cost data will be collected to incorporate the costs for these devices into the procedure APC rate during the device pass-through payment status eligibility period. CMS will continue to monitor utilization and payment trends for C1739 to ensure accurate and appropriate payment.
(c) VasQTM
Laminate Medical submitted an application for a new device category for transitional pass-through payment status for VasQTM for CY 2026. Per the applicant, VasQTM is a nitinol implant which is surgically placed outside and/or around an artery and/or vein to provide external support to arteriovenous fistulas created for vascular access by means of vascular surgery. The applicant further explained that VasQTM reinforces the juxta-anastomotic region against increased wall tension in the newly arterialized vein, guides a more laminate hemodynamic profile of flow with its tapered configuration, and maintains the structural integrity of the anastomotic configuration.
Please refer to the online application posting for VasQTM, available at https://mearis.cms.gov/public/publications/device-ptp/DEP2405312T1JR.
As stated previously, to be eligible for transitional pass-through payment under the OPPS, a device must meet the criteria at § 419.66(b)(1) through (4). With respect to the newness criterion at § 419.66(b)(1), VasQTM received FDA Breakthrough Device designation effective June 5, 2020. The approved FDA indication for VasQTM is:
- For use as an external support for upper extremity arteriovenous fistulas created for vascular access by means of vascular surgery.
FDA granted De Novo classification for VasQTM on September 26, 2023, for the same indication as the one covered by the Breakthrough Device designation. We received the application for a new device category for transitional pass-through payment status for VasQTM on May 31, 2024, which is within 3 years of the date of the initial FDA marketing authorization.
We invited public comments on whether VasQTM meets the newness criterion at § 419.66(b)(1).
We did not receive public comments regarding whether VasQTM meets the newness criterion at § 419.66(b)(1). We received the application for a new device category for transitional pass-through payment status for VasQTM within 3 years of the date of the FDA De Novo classification. Based on our review of the application, we have determined that VasQTM meets the newness criterion at § 419.66(b)(1).
With respect to the eligibility criteria at § 419.66(b)(3), the device must be an integral part of the service furnished, be used for one patient only, come in contact with human tissue, and be surgically inserted or implanted, or applied in or on a wound or other skin lesion. Per the applicant, VasQTM meets the requirements at § 419.66(b)(3).
We invited public comments on whether VasQTM meets the eligibility criterion at § 419.66(b)(3).
We did not receive public comments regarding whether VasQTM meets the eligibility requirements of § 419.66(b)(3). Based on our review of the application, we agree with the applicant, and have determined that VasQTM meets the eligibility criterion at § 419.66(b)(3).
With respect to the exclusion criteria at § 419.66(b)(4), a device is not eligible to be considered for pass-through payment if it is any of the following: (1) equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciable assets as defined in Chapter 1 of the Medicare Provider Reimbursement Manual (CMS Pub. 15-1); or (2) a material or supply furnished incident to a service (for example, a suture, customized surgical kit, or clip, other than a radiological site marker). Per the applicant, VasQTM is (1) not considered equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciable assets, and is (2) not a material or supply furnished incident to a service, and, therefore, is eligible to be considered for pass-through payment.
We invited public comments on whether VasQTM meets the exclusion criterion at § 419.66(b)(4).
We did not receive public comments regarding whether VasQTM meets the exclusion requirements of § 419.66(b)(4). Based on our review of the application, we agree with the applicant and have determined that VasQTM meets the criterion at § 419.66(b)(4).
In addition to the criteria at § 419.66(b)(1) through (4), the criteria for establishing new device categories are specified at § 419.66(c). The first criterion, at § 419.66(c)(1), provides that CMS determines that a device to be included in the category is not appropriately described by any of the existing categories or by any category previously in effect, and was not being paid for as an outpatient service as of December 31, 1996. Per the applicant, no existing (current or previous) device categories for pass-through payment appropriately describe VasQTM . According to the applicant, pass-through code: C1877 (Stent, non-coated/non-covered, without delivery system) does not appropriately describe VasQTM because VasQTM is not a stent and does not come in contact with blood. The applicant also stated that pass-through code C1768 (Graft, vascular) does not appropriately describe VasQTM because VasQTM is not a dialysis graft, is not permitted to be cannulated, and does not have direct contact with blood. The applicant asserted that pass-through code C1881 (Dialysis access system (implantable)) does not appropriately describe VasQTM because VasQTM is not a dialysis access system, is not ( printed page 53648) permitted to be cannulated, and does not have direct contact with blood. Upon review, we stated in the CY 2026 OPPS/ASC proposed rule that we did not identify an existing pass-through payment category that describes VasQTM.
We invited public comment on whether VasQTM meets the device category criterion at § 419.66(c)(1).
We did not receive public comments regarding whether VasQTM meets the eligibility requirements at § 419.66(c)(1). Based on our review of the application, we continue to believe there is no existing category or category previously in effect that appropriately describes VasQTM . Therefore, we have determined that VasQTM meets the device category eligibility criterion at § 419.66(c)(1).
The second criterion for establishing a device category, at § 419.66(c)(2), provides that CMS determines either of the following: (1) that a device to be included in the category has demonstrated that it will substantially improve the diagnosis or treatment of an illness or injury or improve the functioning of a malformed body part compared to the benefits of a device or devices in a previously established category or other available treatment; or (2) for devices for which pass-through status will begin on or after January 1, 2020, as an alternative to the substantial clinical improvement criterion, the device is part of the FDA's Breakthrough Devices Program and has received FDA marketing authorization for the indication covered by the Breakthrough Device designation. VasQTM has a Breakthrough Device designation and marketing authorization from FDA for the indication covered by the Breakthrough Device designation (as explained in more detail of the newness criterion) and therefore was not evaluated for substantial clinical improvement.
We invited public comment on whether VasQTM meets the device category criterion at § 419.66(c)(2).
We did not receive public comments regarding whether VasQTM meets the eligibility requirements at § 419.66(c)(2). VasQTM has a Breakthrough Device designation effective June 5, 2020, and De Novo classification from FDA effective September 26, 2023, for the indication covered by the Breakthrough Device designation. Therefore, VasQTM meets the criterion at 419.66(c)(2)(ii) and is not evaluated for substantial clinical improvement at § 419.66(c)(2)(i). Based on our review of the application, we have determined that VasQTM meets the device category criterion at § 419.66(c)(2).
The third criterion for establishing a device category, at § 419.66(c)(3), requires CMS to determine that the cost of the device is not insignificant, as described in § 419.66(d). Section 419.66(d) includes three cost significance criteria that must each be met. The applicant stated that VasQTM would be reported with HCPCS codes as shown in Table 101, previously published as Table 50 of the CY 2026 OPPS/ASC proposed rule (90 FR 33581).
To meet the cost criterion for device pass-through payment status, a device must pass all three tests of the cost criterion for at least one APC. As we explained in the CY 2005 OPPS final rule (69 FR 65775), we generally use the lowest APC payment rate applicable for use with the nominated device when we assess whether a device meets the cost significance criterion, thus increasing the probability the device will pass the cost significance test. Beginning in CY 2017, we calculate the device offset amount at the HCPCS/CPT code level instead of the APC level (81 FR 79657). We noted that the applicant used the CY 2024 payment rates for the three tests of the cost criterion. For our calculations in the CY 2026 OPPS/ASC proposed rule, we used APC 5183, which had a CY 2024 payment rate of $3,037.01 at the time the application was received. HCPCS code 36821 in APC 5183 had a device offset amount of $49.81 at the time the application was received. According to the applicant, the cost of VasQTM is $4,900.00.
Section 419.66(d)(1), the first cost significance requirement, provides that the estimated average reasonable cost of devices in the category must exceed 25 percent of the applicable APC payment amount for the service related to the category of devices. The average reasonable cost of $4,900.00 for VasQTM is 161.34 percent of the applicable APC payment amount for the service related to the category of devices of $3,037.01 (($4,900.00/$3,037.01) × 100 = 161.34 percent). Therefore, we stated in the CY 2026 OPPS/ASC proposed rule that we believe that VasQTM meets the first cost significance requirement.
The second cost significance requirement, at § 419.66(d)(2), provides that the estimated average reasonable cost of the devices in the category must exceed the cost of the device-related portion of the APC payment amount for the related service by at least 25 percent, which means that the device cost needs to be at least 125 percent of the offset amount (the device-related portion of the APC found on the offset list). The estimated average reasonable cost of $4,900.00 for VasQTM is 9,837.38 percent of the cost of the device-related portion of the APC payment amount for the related service of $49.81 (($4,900.00/$49.81) × 100 = 9,837.38 percent). Therefore, we stated in the CY 2026 OPPS/ASC proposed rule that we ( printed page 53649) believe that VasQTM meets the second cost significance requirement.
The third cost significance requirement, at § 419.66(d)(3), provides that the difference between the estimated average reasonable cost of the devices in the category and the portion of the APC payment amount for the device must exceed 10 percent of the APC payment amount for the related service. The difference between the estimated average reasonable cost of $4,900.00 for VasQTM and the portion of the APC payment amount for the device of $49.81 is 159.70 percent of the APC payment amount for the related service of $3,037.01 ((($4,900.00−$49.81)/$3,037.01) × 100 = 159.70 percent). Therefore, we stated in the CY 2026 OPPS/ASC proposed rule that we believe that VasQTM meets the third cost significance requirement.
We invited public comment on whether VasQTM meets the cost criterion at § 419.66(c)(3).
We did not receive any public comments regarding whether VasQTM meets the cost significance criteria as described at § 419.66(d). Based on our findings from the first, second, and third cost significance tests, we continue to believe that VasQTM meets the cost significance criteria specified at § 419.66(d).
As discussed, the VasQTM pass-through application was preliminarily approved for transitional pass-through payment under the alternative pathway effective October 1, 2024. After our review of the device pass-through application, we have determined that VasQTM meets the requirements for device pass-through status described at § 419.66. We are finalizing approval for device pass-through payment status for VasQTM under the alternative pathway for devices that have an FDA Breakthrough Device designation and FDA marketing authorization for the indication for which the device has Breakthrough Device designation.
(2) Traditional Device Pass-Through Applications
(a) Axoguard HA+ Nerve ProtectorTM
Axogen Corporation submitted an application for a new device category for transitional pass-through payment status for the Axoguard HA+ Nerve ProtectorTM for CY 2026. Per the applicant, the Axoguard HA+ Nerve ProtectorTM is a porcine small intestinal submucosa (SIS) decellularized extracellular matrix (ECM), with a dry coating of sodium hyaluronate and sodium alginate applied to both sides of the device that forms a thin layer of lubricious hydrogel when hydrated. According to the applicant, the Axoguard HA+ Nerve ProtectorTM is designed to be a protective interface between the nerve and the surrounding tissue to minimize the potential for soft tissue attachments and tethering that restricts the nerve's ability to glide and move through the tissue structures during anatomic movement.
Please refer to the online application posting for the Axoguard HA+ Nerve ProtectorTM, available at https://mearis.cms.gov/public/publications/device-ptp/DEP240830YUKGT.
Comment: The applicant expressed its general support for approving transitional pass-through status for the Axoguard HA+ Nerve ProtectorTM. The applicant asserted its belief that the nominated device offers a meaningful advancement for patients with challenging peripheral nerve injuries and that it meets the regulatory criteria for a transitional pass-through payment under § 419.66.
Response: We appreciate the applicant's input. We have taken these comments into consideration in making our determination for pass-through status for the Axoguard HA+ Nerve ProtectorTM.
As stated previously, to be eligible for transitional pass-through payment under the OPPS, a device must meet the criteria at § 419.66(b)(1) through (4). With respect to the newness criterion at § 419.66(b)(1), FDA granted the applicant 510(k) clearance for the Axoguard HA+ Nerve ProtectorTM on April 7, 2023, and then granted a second 510(k) clearance for an expanded indication on October 12, 2023. The approved FDA indications for the Axoguard HA+ Nerve ProtectorTM are:
- For the management of peripheral nerve injuries where there is no gap;
- For the management and protection of peripheral nerve injuries where there is no gap or following closure of the gap.
We received the application for a new device category for transitional pass-through payment status for the Axoguard HA+ Nerve ProtectorTM on August 30, 2024, which is within 3 years of the date of the initial FDA marketing authorization.
Per the applicant, the OPPS pass-through application for the Axoguard HA+ Nerve ProtectorTM is only for the protection of peripheral nerve injuries where there is no nerve gap, specifically for protecting a nerve following a revision (secondary) carpal tunnel (CT) or cubital tunnel (CuT) nerve decompression procedure.
We invited public comments on whether the Axoguard HA+ Nerve ProtectorTM meets the newness criterion at § 419.66(b)(1).
We did not receive public comments regarding whether the Axoguard HA+ Nerve ProtectorTM meets the newness criterion at § 419.66(b)(1). We received the application for a new device category for transitional pass-through payment status for the Axoguard HA+ Nerve ProtectorTM within 3 years of the date of the initial FDA 510(k) clearance. Based on our review of the application, we have determined that the Axoguard HA+ Nerve ProtectorTM meets the newness criterion at § 419.66(b)(1).
With respect to the eligibility criteria at § 419.66(b)(3), the device must be an integral part of the service furnished, be used for one patient only, come in contact with human tissue, and be surgically inserted or implanted, or applied in or on a wound or other skin lesion. Per the applicant, the Axoguard HA+ Nerve ProtectorTM meets the requirements at § 419.66(b)(3).
We invited public comments on whether the Axoguard HA+ Nerve ProtectorTM meets the eligibility criterion at § 419.66(b)(3).
We did not receive public comments regarding whether the Axoguard HA+ Nerve ProtectorTM meets the eligibility requirements of § 419.66(b)(3). Based on our review of the application, we agree with the applicant and have determined that the Axoguard HA+ Nerve ProtectorTM meets the eligibility criterion at § 419.66(b)(3).
With respect to the exclusion criteria at § 419.66(b)(4), a device is not eligible to be considered for pass-through payment if it is any of the following: (1) equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciable assets as defined in Chapter 1 of the Medicare Provider Reimbursement Manual (CMS Pub. 15-1); or (2) a material or supply furnished incident to a service (for example, a suture, customized surgical kit, or clip, other than a radiological site marker). Per the applicant, the Axoguard HA+ Nerve ProtectorTM is (1) not considered equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciable assets and is (2) not a material or supply furnished incident to a service, and therefore, is eligible to be considered for pass-through payment.
We invited public comments on whether the Axoguard HA+ Nerve ProtectorTM meets the exclusion criterion at § 419.66(b)(4).
We did not receive public comments regarding whether the Axoguard HA+ Nerve ProtectorTM meets the exclusion requirements of § 419.66(b)(4). Based on ( printed page 53650) our review of the application, we agree with the applicant and have determined that the Axoguard HA+ Nerve ProtectorTM meets the criterion at § 419.66(b)(4).
In addition to the criteria at § 419.66(b)(1) through (4), the criteria for establishing new device categories are specified at § 419.66(c). The first criterion, at § 419.66(c)(1), provides that CMS determines that a device to be included in the category is not appropriately described by any of the existing categories or by any category previously in effect and was not being paid for as an outpatient service as of December 31, 1996. According to the applicant, no existing device categories for pass-through payment appropriately describe the Axoguard HA+ Nerve ProtectorTM because the existing device categories C1763 (Connective tissue, non-human (includes synthetic)), C1765 (Adhesion barrier), and C1781 (Mesh (implantable)) describe similar, but distinct products. The applicant stated that the existing pass-through code C1763 does not appropriately describe the Axoguard HA+ Nerve ProtectorTM because the devices described by C1763 are used for treating urinary incontinence or for implantation to reinforce soft tissues where weakness exists in the urological or musculoskeletal anatomy, whereas the Axoguard HA+ Nerve ProtectorTM is indicated for the management of peripheral nerve injuries. In addition, the applicant asserted that the existing pass-through code C1765 does not appropriately describe the Axoguard HA+ Nerve ProtectorTM because the devices described by C1765 are bioresorable substances and principally used in spinal surgeries, while the Axoguard HA+ Nerve ProtectorTM is indicated for peripheral nerves. Moreover, the applicant stated that the existing pass-through code C1781 does not appropriately describe the Axoguard HA+ Nerve ProtectorTM because the nominated device is indicated specifically for management of peripheral nerve injuries, whereas devices described by C1781 are for use in hernia repair. The applicant further asserted that C1765 and C1781 do not describe the Axoguard HA+ Nerve ProtectorTM because the device's porcine SIS ECM is not simply resorbed but is remodeled into a meso/epineurium-like tissue, while its sodium hyaluronate and sodium alginate coating reduces friction and promotes nerve gliding.
We noted in the CY 2026 OPPS/ASC proposed rule that based on the description the applicant provided, the Axoguard HA+ Nerve ProtectorTM is a porcine SIS ECM with hyaluronate-alginate coating used for the management and protection of peripheral nerve injuries where there is no gap or following closure of a gap, and thus, could be encompassed by the descriptors C1763 and C1765. Specifically, we stated that we believe that the description the applicant provided for the C1763 category definition is incomplete. The applicant stated that C1763 is indicated for treating urinary incontinence resulting from hypermobility or Intrinsic Sphincter Deficiency (ISD), pelvic floor repair, [or implantation] to reinforce soft tissues where weakness exists in the urological or musculoskeletal anatomy. However, we noted that, in reference to C1763, section 60.4.3, Chapter 4 of the Medicare Claims Processing Manual provides that these tissues include a natural, acellular collagen matrix typically obtained from porcine or bovine small intestinal submucosa, or pericardium. This bio-material is intended to repair or support damaged or inadequate soft tissue. They are used to treat urinary incontinence resulting from hypermobility or Intrinsic Sphincter Deficiency (ISD), pelvic floor repair, or for implantation to reinforce soft tissues where weakness exists in the urological or musculoskeletal anatomy. [This excludes those items that are used to replace skin.] Thus, because the Axoguard HA+ Nerve ProtectorTM is an ECM obtained from porcine SIS and intended to support a damaged or inadequate soft tissue (nerve), we stated that we believe that the pass-through payment category C1763 may appropriately describe the Axoguard HA+ Nerve ProtectorTM.
Additionally, we stated in the CY 2026 OPPS/ASC proposed rule that we believe that the pass-through payment category C1765 may also appropriately describe the Axoguard HA+ Nerve ProtectorTM because the device, as described by the applicant, is designed to be placed on and around neural structures to be a protective interface between a nerve and the surrounding tissue, to minimize the potential for soft tissue attachments, and to ensure the nerve's ability to glide through tissue structures during anatomic movement, and therefore may be appropriately described as an adhesion barrier consistent with devices described by C1765.
We further noted in the CY 2026 OPPS/ASC proposed rule that the two neuroplasty procedure codes that could be used with the Axoguard HA+ Nerve ProtectorTM (CPT® codes 64718 and 64721) have previously been used with both categories C1763 and C1765 and that FDA has previously approved devices described by C1763 and C1765 and billed using CPT® codes 64718 or 64721 for neuroplasty and/or transposition of the ulnar nerve at elbow or median nerve at carpal tunnel. We also noted that the inclusion of these neuroplasty devices in categories C1763 and C1765 appears contradictory to the applicant's assertion that the categories are inapplicable for devices indicated for peripheral nerves. In this context, we stated that we believe the Axoguard HA+ Nerve ProtectorTM may be similar to the devices described by C1763 and C1765, and therefore, the Axoguard HA+ Nerve ProtectorTM may be appropriately described by C1763 and C1765.
We invited public comment on whether the Axoguard HA+ Nerve ProtectorTM meets the device category criterion at § 419.66(c)(1).
Comment: In response to our concern that the Axoguard HA+ Nerve ProtectorTM may be appropriately described by C1763 and C1765, the applicant commented that although the Axoguard HA+ Nerve ProtectorTM, on a superficial level, shares some attributes with products billed to those HCPCS codes, the existing codes were established for different materials and clinical uses, and thus, do not accurately describe the nominated device or its intended application. The applicant asserted that a new, distinct device category is warranted. The applicant acknowledged that C1763 (Connective tissue, non-human (includes synthetic)) generally covers acellular collagen matrices derived from animal sources, which sounds similar to Axoguard HA+ Nerve ProtectorTM 's extracellular matrix component. The applicant noted, however, that the Medicare Claims Processing Manual's definition for C1763 clarifies that these matrices are “intended to repair or support damaged or inadequate soft tissue” and are typically used for “urinary incontinence ( e.g., pelvic floor repair) or to reinforce soft tissues where weakness exists in the urological or musculoskeletal anatomy,” explicitly excluding products used as skin replacements. The applicant further asserted that CMS created C1763 to describe implants like surgical grafts or slings for pelvic floor and orthopedic reinforcement, not nerve protectors. The applicant stated that the Axoguard HA+ Nerve ProtectorTM is indicated for the management and protection of peripheral nerves, which is neither a urological nor a musculoskeletal application. In addition, the applicant asserted its belief that a peripheral nerve's needs ( i.e., gliding in a tissue ( printed page 53651) bed and protection from adhesions) are very different from the structural support typically provided by C1763 devices used in incontinence or hernia repairs. The applicant stated that using C1763 for a nerve protector is a clinical stretch beyond the code's intended scope, even if the base material (porcine SIS) is similar. In response to our statement that some neuroplasty procedures (CPT 64718 and 64721) have previously been billed for devices in the C1763 category, the applicant opined that such practice reflects the absence of a nerve-specific device code rather than a proper fitting of nerve products into C1763. The applicant stated that a new code would eliminate ambiguity and ensure more precise coding for nerve repair technologies.
In regard to the applicability of C1765 (Adhesion barrier), the applicant stated that, although one might consider Axoguard HA+ Nerve ProtectorTM an adhesion barrier since it aims to prevent scar adhesion around nerves, C1765 is defined as a bioresorbable substance placed on or around neural structures, which inhibits fibroblast migration and minimizes scar tissue formation, principally used in spine surgeries (laminectomies, discectomies). The applicant also stated that paradigmatic C1765 devices are spinal dura shields or gels that prevent epidural fibrosis after back surgery. The applicant asserted that the Axoguard HA+ Nerve ProtectorTM differs in both composition and clinical context, as it is a biological matrix implant (not just a synthetic or biochemical barrier), which is used in peripheral nerve surgeries of the limbs, not in the spine. The applicant further added that Axoguard HA+ Nerve ProtectorTM 's ECM is intended to integrate into the nerve's outer tissue layer over time (providing long-term support), rather than resorb away like typical adhesion barriers. The applicant added that, as with C1763, any device assigned to C1765 currently receives no transitional pass-through payment, as C1765 was a pass-through category long ago and is now a packaged supply code, and continuing to use C1765 for a new nerve-specific technology would perpetuate the lack of appropriate payment, contrary to the purpose of establishing new device categories for innovations.
The applicant also stated that C1781 (Mesh, implantable) is used for hernia repair, which is different from the Axoguard HA+ Nerve ProtectorTM 's indication. The applicant asserted that the Axoguard HA+ Nerve ProtectorTM does not fit any existing mesh or patch category used for general surgical repair because it is a specialized nerve protector and stands apart from surgical meshes used in abdominal or orthopedic procedures. The applicant concluded that, given these distinctions, no existing HCPCS Level II code adequately describes Axoguard HA+ Nerve ProtectorTM 's combination of material, function, and clinical indication. The applicant stated that assigning the nominated device to C1763, C1765, or C1781 would not only be inappropriate from a descriptive standpoint but would also fail to facilitate tracking of the device's utilization and outcomes since its use would be obscured under codes that include many other dissimilar products. The applicant asserted that, by creating a new device category and code, CMS will ensure that claims for the Axoguard HA+ Nerve ProtectorTM can be accurately identified, and thus, improve transparency and allow the collection of meaningful data regarding the device's cost and performance during the transitional pass-through period, as intended under § 419.66.
Finally, the applicant also stated that CMS regulations stipulate that a new device category may be established if the device is not appropriately described by any of the existing categories. For these reasons, the applicant believes that the Axoguard HA+ Nerve ProtectorTM meets the device category criterion. The applicant reiterated its belief that the Axoguard HA+ Nerve ProtectorTM satisfies the requirements described at § 419.66(c)(1) regarding distinct category status. According to the applicant, the alternative to establishing a new category code—forcing the device into a category like C1763 or C1765—would continue the current disincentive for adoption, contradicting CMS's goal of enabling access to worthwhile new therapies, whereas creating a new category code would signal to hospitals that the Axoguard HA+ Nerve ProtectorTM is recognized as an innovation and encourage appropriate use in patients who need it. Finally, the applicant stated that finalizing the creation of a new code would accurately describe the device's unique characteristics and clinical use.
Response: We appreciate the applicant's input. We agree with the applicant that C1781 does not appropriately describe the Axoguard HA+ Nerve ProtectorTM . However, based on the information available to us, we continue to believe that C1763 and C1765 may describe the Axoguard HA+ Nerve ProtectorTM . We have taken this into consideration in making our determination for device pass-through payment status for the Axoguard HA+ Nerve ProtectorTM.
The second criterion for establishing a device category, at § 419.66(c)(2), provides that CMS determines either of the following: (1) that a device to be included in the category has demonstrated that it will substantially improve the diagnosis or treatment of an illness or injury or improve the functioning of a malformed body part compared to the benefits of a device or devices in a previously established category or other available treatment; or (2) for devices for which pass-through status will begin on or after January 1, 2020, as an alternative to the substantial clinical improvement criterion, the device is part of the FDA's Breakthrough Devices Program and has received FDA marketing authorization for the indication covered by the Breakthrough Device designation. The applicant asserted that the Axoguard HA+ Nerve ProtectorTM represents a substantial clinical improvement over existing technologies in the management of peripheral nerve injuries where there is no nerve gap, specifically in protecting a nerve following a revision (secondary) CT or CuT nerve decompression procedure.
The applicant provided three redacted manufacturer internal reports to support these claims, as well as eight background articles/documents about the predicate device, the Axoguard Nerve ProtectorTM . We noted in the CY 2026 OPPS/ASC proposed rule that the predicate device differs from the Axoguard HA+ Nerve ProtectorTM in that the nominated device has a dry coating of sodium hyaluronate and sodium alginate applied to both sides that forms a thin layer of lubricious hydrogel when hydrated. We stated that the addition of the dry coating of sodium hyaluronate and sodium alginate to the Axoguard Nerve ProtectorTM appears to be the distinguishing feature of the device that is the subject of this application. In addition, the applicant submitted 32 supplemental background articles describing topics including general disease processes and disease prevalence.
The applicant's assertions regarding the substantial clinical improvement criterion are shown in Table 102, previously published as Table 51 of the CY 2026 OPPS/ASC proposed rule (90 FR 33584 through 33588). Please see the online posting for the Axoguard HA+ Nerve ProtectorTM for the applicant's complete statements regarding the substantial clinical improvement ( printed page 53652) criterion and the supporting evidence provided.
( printed page 53653) ( printed page 53654) ( printed page 53655) ( printed page 53656)After review of the information provided by the applicant, we noted in the CY 2026 OPPS/ASC proposed rule that we have the following concerns regarding whether the Axoguard HA+ Nerve ProtectorTM meets the substantial clinical improvement criterion.
The applicant asserted that the Axoguard HA+ Nerve ProtectorTM demonstrates clinical improvement in: (1) nerve health in an injured tissue bed through less adhesion, extraneural scarring, and inflammatory markers, (2) nerve health in an injured tissue bed through decreased friction between the nerve and surrounding tissue to allow for gliding and to minimize potential for soft tissue attachment, (3) device performance due to its sodium hyaluronate and sodium alginate gel layer that allows for nerve gliding, and (4) sensory and motor symptoms. We noted that the applicant provided redacted internal studies of animal models (Axogen Corporation, 2024; Axogen Corporation, n.d.; Axogen Corporation, 2022) and an abstract (Alsmadi et al., 2025) on Axoguard HA+ Nerve ProtectorTM in rats. We noted that the applicant did not submit studies assessing the Axoguard HA+ Nerve ProtectorTM in humans. Therefore, we questioned whether data from animal studies is sufficient to extrapolate to human populations for the purposes of demonstrating substantial clinical improvement.
For the other claims, the applicant provided only background evidence, specifically retrospective studies, which describe findings for a predicate device, the Axoguard Nerve ProtectorTM , which received FDA 510(k) clearance on January 10, 2014, not the nominated device, the Axoguard HA+ Nerve ProtectorTM . We noted that the applicant stated that the nominated Axoguard HA+ Nerve ProtectorTM improved on the predicate device, but the applicant did not provide any additional information or evidence to support this claim. We also noted that the application did not include comparative outcome data between the Axoguard HA+ Nerve ProtectorTM and its predicate device. We stated that we welcome additional information that compares outcome data from the Axoguard HA+ Nerve ProtectorTM and the predicate device, the Axoguard Nerve ProtectorTM , to help inform our assessment of whether the Axoguard HA+ Nerve ProtectorTM demonstrates a substantial clinical improvement.
In addition, we stated in the CY 2026 OPPS/ASC proposed rule that we are concerned that the provided evidence did not directly support the applicant's 10 claims that the Axoguard HA+ Nerve ProtectorTM demonstrates substantial clinical improvement over existing technologies. We noted that no evidence was provided comparing the Axoguard HA+ Nerve ProtectorTM to other currently available treatments for the indicated condition including autologous flaps/fat pads and xenografts or off-the-shelf wraps that include materials sourced from human amniotic membrane, bovine, porcine, and plants. We stated that we welcome further evidence that compares the Axoguard HA+ Nerve ProtectorTM to currently available treatments in the clinical setting where it is most likely to be used. To demonstrate substantial clinical improvement over currently available treatments, we stated that we consider supporting evidence, preferably published peer-reviewed clinical trials, that show improved clinical outcomes, such as reduction in mortality, complications, subsequent interventions, future hospitalizations, recovery time, pain, or a more rapid beneficial resolution of the disease process compared to the standard of care. We stated that additional supporting evidence demonstrating these improved clinical outcomes would help inform our assessment of whether the Axoguard HA+ Nerve ProtectorTM demonstrates substantial clinical improvement over existing technologies.
We invited public comment on whether the Axoguard HA+ Nerve ProtectorTM meets the device category criterion at § 419.66(c)(2).
Comment: The applicant acknowledged that CMS applies a high evidentiary standard for substantial clinical improvement, typically requiring robust clinical data showing improved patient outcomes such as reduced complications, fewer reoperations, faster recovery, or better functional results compared to the current standard of care. The applicant further acknowledged the concerns CMS ( printed page 53657) included in the CY 2026 OPPS/ASC proposed rule regarding the Axoguard HA+ Nerve ProtectorTM application and agreed that demonstrating improved clinical outcomes in patients is essential to meeting the substantial clinical improvement criterion.
In response to our concerns, the applicant commented that it is committed to providing the clinical evidence necessary to satisfy the substantial clinical improvement standard and indicated that it is currently conducting an ongoing prospective, multicenter, open-label, single-arm clinical study[44] to evaluate the Axoguard HA+ Nerve ProtectorTM in patients undergoing a first revision cubital tunnel decompression procedure for recurrent or recalcitrant cubital tunnel syndrome. The applicant explained that the study has enrolled 19 study participants with recurrent cubital tunnel syndrome following a failure to maintain the functional improvements from their primary decompression procedure, allowing participants to serve as their own in-patient controls for analysis of the treatment effect of repeating the decompression procedure with the addition of the Axoguard HA+ Nerve ProtectorTM . The applicant noted that the study is currently in the participant follow-up phase, with the intent for each participant to complete 18 months of follow-up. The applicant reported that although the study is not complete, it has completed a planned interim analysis of the study that it believes directly addresses the previous lack of human outcomes data for the Axoguard HA+ Nerve ProtectorTM . The applicant asserted that the preliminary results demonstrate a decrease in pain, improvements in grip strength and sensory testing, and no adverse effects.
Specifically, the applicant stated that 84.2 percent of study participants reported a level of none to mild (0-30 mm on 0-100 mm in the Visual Analog Scale (VAS)) pain at 6 months post-operatively. Further, the study found a mean VAS score at first pre-operative visit of 73.6 mm (SD = 18.3) and a post-operative score of 13.5 mm at 6 months (SD = 23.8), representing an 81.7 percent decrease in mean VAS-reported pain. The applicant also stated that, 26.3 percent of participants reported a VAS of zero at the 6-month post-operative timepoint. Moreover, the applicant added that the participants demonstrated functional recovery of the ulnar nerve as measured by improvements in grip strength (68.4 percent of participants) and sensory testing (63.2 percent of participants) from baseline to 6 months. The applicant finally asserted that the study data showed improvement in investigator-rated ulnar neuropathy assessed by the Modified McGowan Classification of Ulnar Nerve Neuropathy and that there have been no second revision procedures performed, no reports of recurrences of ulnar neuropathy, and no safety issues. The applicant noted that these interim findings are descriptive in nature, and the final analysis will be available upon completion of the 18-month follow-up period, database lock, and final statistical review. Finally, the applicant highlighted that, while the study is ongoing and data remains subject to final investigator approval and statistical analysis, preliminary, 100 percent source-verified observations to date are consistent with the FDA-cleared intended use and function of the Axoguard HA+ Nerve ProtectorTM . The applicant stated that it believes that the nominated device meets the substantial clinical improvement threshold, given its novel mechanism addressing unmet need for recurrent nerve compression injuries, and requested that CMS consider the provided clinical data and recognize the Axoguard HA+ Nerve ProtectorTM as an innovative device offering improved clinical outcomes.
Response: We appreciate the applicant's input; however, we maintain some of our concerns listed in the CY 2026 OPPS/ASC proposed rule (90 FR 33588 through 35589), including the lack of comparative data between the Axoguard HA+ Nerve ProtectorTM and the predicate device and other currently available treatments for the indicated condition. While the new information from the preliminary findings of the 19-patient clinical trial currently underway may address our concern about the lack of human clinical trial evidence, we believe that the data provided does not sufficiently demonstrate substantial clinical improvement. Specifically, we believe that the preliminary findings may contain uncontrolled confounding variables specific to the study participants' initial procedures that may cast doubt on the validity of the study's outcomes. We note that the new evidence provided is preliminary data gathered during the first 6 months of an 18-month long clinical trial. The applicant specifically stated that these interim findings are descriptive in nature, and the final analysis will be conducted upon completion of follow-up, database lock, and final statistical review. At this time, the provided summary of the preliminary data is insufficient to draw a conclusion that the clinical benefits purportedly resulting from use of the Axoguard HA+ Nerve ProtectorTM are substantial when compared to existing treatments.
In addition, we note that the small sample size may affect the quality and reliability of the data provided in support of the Axoguard HA+ Nerve ProtectorTM and may limit the statistical significance and generalizability of the results. Further, we question whether using patients as their own controls for subsequent decompression procedure with Axoguard HA+ Nerve ProtectorTM demonstrates a substantial clinical improvement when compared to other available treatments. Finally, it is unclear based on the data submitted by the applicant which prior procedures were performed.
For the reasons discussed, we do not believe that the Axoguard HA+ Nerve ProtectorTM represents a substantial clinical improvement relative to existing therapies currently available. Therefore, after consideration of the public comment we received and our review of the device pass-through application, we are not approving the Axoguard HA+ Nerve ProtectorTM for transitional pass-through payment status for CY 2026 because the technology does not meet the substantial clinical improvement criterion at § 419.66(c)(2). Because we have determined that the Axoguard HA+ Nerve ProtectorTM does not meet the substantial clinical improvement criterion, we will not address in this final rule with comment period whether the technology meets the cost criterion required for transitional pass-through payment for devices.
(b) LithoVueTM Elite Digital Flexible Ureteroscope System With Pressure Monitoring
Boston Scientific Corporation submitted an application for a new device category for transitional pass-through payment status for the LithoVueTM Elite Digital Flexible Ureteroscope System with Pressure Monitoring (the LithoVueTM Elite System) for CY 2026. Per the applicant, the LithoVueTM Elite System consists of a single-use, disposable flexible ureteroscope (the LithoVueTM Elite Ureteroscope) and a workstation (the StoneSmart Connect Console), that provide real-time intraluminal pressure monitoring in the kidney and ureter during ureteroscopy and can be used in conjunction with endoscopic accessories to perform various diagnostic and therapeutic procedures in the urinary tract. The applicant stated that the distal tip of the LithoVueTM ( printed page 53658) Elite Ureteroscope's shaft includes the working channel, the illumination optics, the digital imaging sensor, and a Micro-Electro-Mechanical Systems (MEMS) pressure sensor for monitoring the real-time intraluminal pressure during ureteroscopy.
The applicant is only seeking a new device category for transitional pass-through payment status for the LithoVueTM Elite Ureteroscope, a component of the LithoVueTM Elite System.
Please refer to the online application posting for the LithoVueTM Elite Digital Flexible Ureteroscope System with Pressure Monitoring, available at https://mearis.cms.gov/public/publications/device-ptp/DEP2503038TF22.
Comment: A few commenters expressed support for approval of transitional pass-through payment for the LithoVueTM Elite Ureteroscope. The commenters expressed their belief that the LithoVueTM Elite Ureteroscope benefits patients by minimizing the post-operative risks of pyelonephritis, urosepsis, and pain.
However, a few commenters expressed that the LithoVueTM Elite Ureteroscope should not receive approval for transitional pass-through payments, because the applicant did not provide sufficiently robust, peer-reviewed clinical evidence that demonstrates a clear benefit to patient outcomes over existing ureteroscopes.
Response: We appreciate the commenters' input and acknowledge the commenters' support for and against the approval of the LithoVueTM Elite Ureteroscope for transitional pass-through status. We have taken these comments into consideration in our final determination for pass-through status for the LithoVueTM Elite Ureteroscope.
As stated previously, to be eligible for transitional pass-through payment under the OPPS, a device must meet the criteria at § 419.66(b)(1) through (4). With respect to the newness criterion at § 419.66(b)(1), FDA granted the applicant 510(k) clearance for the LithoVueTM Elite System on February, 2023. The approved FDA indication for the LithoVueTM Elite System is:
- To be used to visualize organs, cavities, and canals in the urinary tract (urethra, bladder, ureter, calyces and renal papillae) via transurethral or percutaneous access routes. It can also be used in conjunction with endoscopic accessories to perform various diagnostic and therapeutic procedures in the urinary tract.
On July 1, 2024, FDA granted the applicant Special 510(k) clearance for the LithoVueTM Elite Ureteroscope (with pressure monitoring) with a redesigned distal tip to improve its durability during a ureteroscopy for this same indication. We received the application for a new device category for transitional pass-through payment status for the LithoVueTM Elite System on March 3, 2025, which is within 3 years of the date of the initial FDA marketing authorization.
We invited public comments on whether the LithoVueTM Elite System meets the newness criterion at § 419.66(b)(1).
Comment: With respect to the newness criterion at § 419.66(b)(1), the applicant reiterated that FDA granted 510(k) clearance for the LithoVueTM Elite Ureteroscope on February 2, 2023. The applicant also noted that it submitted an application for transitional pass-through payment on March 3, 2025, which is within 3 years of the initial market authorization, and therefore, the LithoVueTM Elite Ureteroscope meets the criterion at § 419.66(b)(1).
Response: We appreciate the applicant's input. We agree with the applicant that we received the application for a new device category for transitional pass-through payment status for the LithoVueTM Elite Ureteroscope within 3 years of the date of FDA 510(k) clearance. After consideration of the public comments we received and our review of the application, we have determined that the LithoVueTM Elite System meets the newness criterion at § 419.66(b)(1).
As previously noted, the applicant is only seeking a new device category for transitional pass-through payment status for the LithoVueTM Elite Ureteroscope component of the LithoVueTM Elite System, and as such, the eligibility and exclusion criteria will evaluate the LithoVueTM Elite Ureteroscope.
With respect to the eligibility criteria at § 419.66(b)(3), the device must be an integral part of the service furnished, be used for one patient only, come in contact with human tissue, and be surgically inserted or implanted, or applied in or on a wound or other skin lesion. Per the applicant, the LithoVueTM Elite Ureteroscope meets the requirements at § 419.66(b)(3).
With respect to the LithoVueTM Elite Ureteroscope, we questioned in the CY 2026 OPPS/ASC proposed rule whether the MEMS pressure sensor is integral to the service furnished. In the CY 2014 OPPS final rule with comment period (78 FR 75005), we stated that we have interpreted “integral” to mean that the device is necessary to furnish or deliver the primary procedure with which it is used. For example, a pacemaker is integral to the procedure of implantation of a pacemaker. Per the applicant, the LithoVueTM Elite Ureteroscope differs from other currently available ureteroscopes, because the device includes the MEMS pressure sensor which is located at the distal tip of the ureteroscope and enables continuous, real-time monitoring of intrarenal pressure (IRP) during ureteroscopy. We noted that neither the FDA 510(k) indication nor the FDA Special 510(k) indication includes the MEMS pressure sensor, and the cleared indications appear to be consistent with the indications for other FDA approved ureteroscopes. In addition, as discussed in more detail in the § 419.66(c)(2) discussion in the CY 2026 OPPS/ASC proposed rule, we questioned whether there is sufficient evidence to support the assertion that continuous pressure monitoring is necessary and/or required to furnish or deliver the primary procedure (ureteroscopy) with which it is used. While we did not question whether the ureteroscope itself is integral to the service furnished, we questioned whether the MEMS pressure sensor, the mechanism which the applicant asserts is the distinguishing feature of the LithoVueTM Elite Ureteroscope, is integral to the service furnished in accordance with § 419.66(b)(3), because pressure monitoring during ureteroscopy procedures appears to be purely additive and not necessary to furnish the ureteroscopy.
We invited public comments on whether the LithoVueTM Elite Ureteroscope meets the eligibility criterion at § 419.66(b)(3).
Comment: The applicant reiterated that the LithoVueTM Elite Ureteroscope is used for one patient only, comes in contact with human tissue, and is surgically inserted during a ureteroscopy procedure.
In response to our concern whether the LithoVueTM Elite Ureteroscope's MEMS pressure sensor is integral to the services furnished, specifically whether continuous pressure monitoring is required to perform ureteroscopy, the applicant commented that it disagrees with our interpretation of integral and believes that the MEMS pressure sensor is an intrinsic part of the LithoVueTM Elite Ureteroscope because it is not a detachable or an adjunctive component. The applicant further stated that the LithoVueTM Elite Ureteroscope's unique pressure sensing capability is the device's key feature and the basis for its substantial clinical improvement claim. Specifically, the applicant asserted that ( printed page 53659) the ability to measure IRP makes ureteroscopy safer because the surgeon is able (through use of this new technology) to avoid dangerously high IRPs, which the surgeon has no way of detecting with a conventional ureteroscope. The applicant also asserted that CMS's literal application of its example that “a pacemaker is integral to the procedure of implantation of a pacemaker” is overly simplistic and suggests that any feature beyond the basic function of a device could be considered “non-integral.” In addition, the applicant asserted that CMS has previously deviated from this approach when determining transitional pass-through payment status. For example, according to the applicant, pass-through categories distinguish between neurostimulators with rechargeable and non-rechargeable batteries. The applicant noted that different power supplies have different clinical advantages for neurostimulators, but the type of power supply does not affect the basic function of the neurostimulator. The applicant stated that the LithoVueTM Elite Ureteroscope with the MEMS pressure sensor is integral to the ureteroscopy codes listed in Table 54 of the CY 2026 OPPS/ASC proposed rule (90 FR 33597 and 33598) in that it is a ureteroscope with a novel capability (IRP measurement) that allows for fewer complications associated with high (and unchecked) IRP during ureteroscopy.
In contrast, a few commenters stated that they do not believe that the LithoVueTM Elite Ureteroscope with the MEMS pressure sensor is integral to the services furnished. One commenter asserted that the LithoVueTM Elite Ureteroscope is not integral to the procedures listed in the application and uses technology in a manner like device applications previously rejected on this basis. The commenter also stated that it agrees with CMS that the pressure sensor technology that is the basis of the LithoVueTM Elite Ureteroscope application is not integral to the procedures presented in the application. According to the commenter, CMS has consistently applied its interpretation of integral to mean that the device is necessary to furnish or deliver the primary procedure with which it is used. The commenter noted that interested parties rely on consistent and reliable rulemaking to make informed decisions about when to engage with CMS and what to expect. The commenter claimed that CMS made a similar “decision” when it “rejected” the CANARY CanturioTM Tibial Extension (CTE) with CHIRP® System application in the CY 2025 OPPS/ASC proposed rule after concluding that the CTE implant within the CHIRP® system was not integral to the arthroplasty procedure identified in the application. The commenter stated that the CHIRP® System's CTE implant included technology that provided kinematic data, and in this manner, is similar to the LithoVueTM Elite Ureteroscope's pressure sensor providing data. The commenter noted that, in the CY 2025 OPPS/ASC proposed rule, CMS questioned if the CTE implant was integral to the service provided because the utilization appeared to be purely additive and unnecessary to furnish or deliver the underlying procedure. The commenter stated that the LithoVueTM Elite Ureteroscope pressure sensor is of a similar nature to the CHIRP® System's CTE implant because ureteroscopy can be completed without IRP monitoring. The commenter expressed its belief that CMS has articulated, in the CY 2026 OPPS/ASC proposed rule, a position on the LithoVueTM Elite Ureteroscope that is consistent with prior rulemaking related to § 419.66(b)(3). Another commenter asserted that monitoring a physiological parameter does not inherently translate to improved outcomes unless accompanied by actionable thresholds and validated interventions. Further, the commenter asserted that without consensus on what constitutes a safe IRP level or how to respond to elevated readings, the utility of the sensor remains theoretical.
Response: We appreciate the applicant's clarification and the commenters' input. First, we agree with the applicant that the LithoVueTM Elite Ureteroscope is used for one patient only, comes in contact with human tissue, and is surgically implanted or inserted or applied in or on a wound or other skin lesion. However, we disagree with the applicant's assertion that we deviated from our approach of assessing whether a device is integral regarding neurostimulators with rechargeable and non-rechargeable batteries because neurostimulators require a power source to operate and the batteries are, therefore, integral to the device regardless of how they are charged. We also note that while we did express concerns in the CY2025 OPPS/ASC proposed rule that the CHIRP® System's CTE implant may not be integral (89 FR 59294), we cannot comment further on the CHIRP® System's CTE implant because the applicant for that device withdrew the application prior to the final rule and we made no final determination of the eligibility of the CHIRP® System's CTE implant. We also disagree with the applicant's interpretation of the pacemaker example. Pacemakers are used as an example of a device being utilized in an integral manner for illustrative purposes. The example is not intended to suggest or imply any standard as the applicant asserts. Finally, while we maintain our concern regarding the overall utility of the MEMS pressure sensor, we agree with the applicant that the LithoVueTM Elite Ureteroscope, inclusive of the MEMS pressure sensor, meets the criterion because the MEMS pressure sensor is not a detachable or an adjunctive component to the ureteroscope itself, which we previously agreed is necessary and/or required to furnish or deliver the primary procedure (ureteroscopy) with which it is used. After consideration of the public comments we received and our review of the application, we have determined that the LithoVueTM Elite Ureteroscope meets the eligibility criterion at § 419.66(b)(3).
With respect to the exclusion criteria at § 419.66(b)(4), a device is not eligible to be considered for pass-through payment if it is any of the following: (1) equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciable assets as defined in Chapter 1 of the Medicare Provider Reimbursement Manual (CMS Pub. 15-1); or (2) a material or supply furnished incident to a service (for example, a suture, customized surgical kit, or clip, other than a radiological site marker). Per the applicant, the LithoVueTM Elite Ureteroscope, the component nominated in this application, is (1) not considered equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciable assets, and (2) not a material or supply furnished incident to a service, and therefore, is eligible to be considered for pass-through payment.
We invited public comments on whether the LithoVueTM Elite Ureteroscope meets the exclusion criterion at § 419.66(b)(4).
Comment: The applicant reiterated that the LithoVueTM Elite Ureteroscope is a single-use ureteroscope, is not subject to capital equipment depreciation schedules, and is therefore not excluded under the criterion at § 419.66(b)(4).
Response: We appreciate the applicant's input. After consideration of the public comments we received and our review of the application, we agree with the applicant and have determined ( printed page 53660) that the LithoVueTM Elite Ureteroscope meets the criterion at § 419.66(b)(4).
In addition to the criteria at § 419.66(b)(1) through (4), the criteria for establishing new device categories are specified at § 419.66(c). The first criterion, at § 419.66(c)(1), provides that CMS determines that a device to be included in the category is not appropriately described by any of the existing categories or by any category previously in effect, and was not paid for as an outpatient service as of December 31, 1996. Per the applicant, the existing pass-through code C1747 (Endoscope, single-use ( i.e., disposable), urinary tract, imaging/illumination device (insertable)) does not appropriately describe the LithoVueTM Elite Ureteroscope because the category description does not include the LithoVueTM Elite Ureteroscope's pressure monitoring feature. The applicant also stated that the existing pass-through code C2624 (Implantable wireless pulmonary artery pressure sensor with delivery catheter, including all system components) does not appropriately describe the LithoVueTM Elite Ureteroscope because the nominated device is an insertable ureteroscope that measures IRP, whereas C2624 is specific to sensors that measure pulmonary artery pressure.
We noted in the CY 2026 OPPS/ASC proposed rule that, based on the description the applicant provided, the LithoVueTM Elite Ureteroscope is a single-use, disposable ureteroscope inserted into the urinary tract for imaging and illumination, and thus, could be appropriately described by C1747. Specifically, we stated that we believe that C1747 may appropriately describe the LithoVueTM Elite Ureteroscope because it describes any device that is a single-use ( i.e., disposable) endoscope with imaging/illumination capabilities intended for use in the urinary tract to perform ureteroscopy procedures. We noted that the descriptor for C1747 does not reference device features that would exclude the inclusion of a pressure monitoring feature. Further, we noted that the HCPCS procedure codes with which the applicant has stated the LithoVueTM Elite Ureteroscope would be reported are consistent with the HCPCS codes approved for C1747. In this context, we stated that we believe that the LithoVueTM Elite Ureteroscope may be similar to the devices described by C1747, and therefore, the LithoVueTM Elite Ureteroscope may also be appropriately described by C1747.
We invited public comment on whether the LithoVueTM Elite Ureteroscope meets the device category criterion at § 419.66(c)(1).
Comment: In response to our concern that the LithoVueTM Elite Ureteroscope may be appropriately described by C1747, the applicant stated that while C1747 does describe a single-use ureteroscope, the descriptor is too broad and does not include the key feature—IRP monitoring—that makes the LithoVueTM Elite Ureteroscope unique. The applicant stated it strongly believes that the IRP monitoring capabilities that make the LithoVueTM Elite Ureteroscope unique, merit a new pass-through device category.
In contrast, a few commenters stated their belief that the LithoVueTM Elite Ureteroscope is appropriately described by the existing code C1747. One commenter agreed with CMS that the LithoVueTM Elite Ureteroscope is appropriately described by existing device category C1747 and that the presence of the additional feature of a pressure sensor is not, by itself, a reason to exclude the LithoVueTM Elite Ureteroscope from C1747. The commenter also stated that, from its experience with providers, billers, facilities, and other interested parties in the field of urology, C1747 has already been used for the LithoVueTM Elite Ureteroscope. Therefore, the commenter stated it supports a conclusion that C1747 remains the appropriate device category for the LithoVueTM Elite Ureteroscope, and no new device category is warranted.
Another commenter stated that, per the criteria described at § 419.66(c), CMS has previously established this device category in the CY 2023 OPPS/ASC final rule with comment period. Specifically, the commenter noted that CMS approved a device category code for single-use ureteroscopes—C1747 (Endoscope, single-use (that is, disposable), urinary tract, imaging/illumination device (insertable))—and asserted that CMS created this device category for Uretero1TM . The commenter also noted that 1 month after C1747 was established based on the approval of the STERIS Uretero1TM device pass-through application, FDA granted the LithoVueTM Elite System 510(k) clearance, on February 2, 2023. The commenter asserted that, therefore, the nominated device has been eligible for transitional pass-through payment for 35 out of the full 36-month eligibility period, which expires December 31, 2025. The commenter stated its belief that the applicant is clearly aware that the LithoVueTM Elite System has already benefited from transitional pass-through status for single-use ureteroscopes.
In addition, the commenter noted that advertisement on the applicant's website recommends billing C1747 for single-use ureteroscopes, including the LithoVue[TM] Elite Single-Use Digital Flexible Ureteroscope. Furthermore, the commenter stated that, in the new study by Bhojani et al. (2025),[45] the methods indicate that the authors used claims data to ascertain the study's conclusions. The commenter stated that for the Bhojani et al. (2025) study, the authors queried Medicare claims data submitted with C1747 alongside either CPT code 52353 or 52356. According to the commenter, in using the C1747 data for the Bhojani et al. (2025) study alongside the LithoVueTM Elite Ureteroscope, the authors are demonstrating that this device has previously been identified by the established device category code and has already benefited from its use. Moreover, the commenter noted that the initial LithoVueTM Elite Ureteroscope application cited literature that suggests IRP relevance is limited to kidney stone procedures. The commenter further highlighted that the Bhojani et al. (2025) study only references lithotripsy procedures. The commenter asserted that, despite this, the LithoVueTM Elite Ureteroscope application does not appropriately narrow the associated CPT code set to reflect this clinical specificity; instead, the applicant generalizes the clinical claims to all ureteroscopy procedures. Per the commenter, if the LithoVueTM Elite Ureteroscope pressure sensor were integral to improved outcomes in stone management, as the applicant claims, then the applicable CPT codes should have been refined accordingly, as not all ureteroscopy procedures are performed for the treatment of kidney stones.
Additionally, the commenter stated that in the CY 2026 OPPS/ASC proposed rule, CMS appropriately acknowledged that the LithoVueTM Elite Ureteroscope may fall within the scope of the existing device category C1747, which includes all single-use ureteroscopes and their associated procedures. The commenter asserted that creating a new device category code without clear differentiation risks redundancy and confusion in coding practices. The commenter further asserted that it may also set a precedent for incremental innovations to seek separate reimbursement pathways ( printed page 53661) without sufficient clinical justification. The commenter stated that, therefore, it agrees that the LithoVueTM Elite Ureteroscope may be appropriately described by C1747.
Response: We appreciate the applicant's and commenters' input. After consideration of the public comments we received and our review of the application, we continue to believe that C1747 appropriately describes the LithoVueTM Elite Ureteroscope because C1747 includes any device that is a single-use (that is, disposable) endoscope with imaging/illumination capabilities intended for use in the urinary tract to perform ureteroscopy procedures. We also continue to believe that the procedure codes with which the applicant has stated that the LithoVueTM Elite Ureteroscope would be reported are consistent with the CPT codes approved for C1747.
We disagree with the applicant's assertions that the IRP monitoring capabilities that make the LithoVueTM Elite Ureteroscope unique merit a new pass-through device category because the LithoVueTM Elite Ureteroscope is consistent with C1747, regardless of the presence of the additional pressure sensing feature, as a few commenters noted. We agree with the commenter that the nominated device has already benefited from transitional pass-through status for single-use ureteroscopes and that CMS is already collecting cost data for the nominated device in the appropriate APC. Therefore, we have determined that the LithoVueTM Elite Ureteroscope does not meet the device category eligibility criterion at § 419.66(c)(1), because it is appropriately described by an existing category or a category previously in effect.
We conclude that the LithoVueTM Elite Ureteroscope does not meet the device category eligibility criterion to be considered as a device for transitional pass-through payment. Therefore, in this final rule with comment period, we will not address whether the technology meets the other remaining criteria required for transitional pass-through payment for devices. We are not approving the LithoVueTM Elite Ureteroscope for transitional pass-through payment status for CY 2026 because the technology does not meet the device category eligibility criterion at § 419.66(c)(1).
(c) VersaVueTM Single-Use Flexible Cystoscope
Boston Scientific Corporation submitted an application for a new device category for transitional pass-through payment status for the VersaVueTM Single-Use Flexible Cystoscope for CY 2026. Per the applicant, the VersaVueTM Single-Use Flexible Cystoscope is used in cystoscopy procedures to diagnose or treat diseases of the lower urinary tract. According to the applicant, the VersaVueTM Single-Use Flexible Cystoscope is a single-use, disposable flexible cystoscope intended to be operated with its compatible display system, the VersaVueTM Tablet (a tablet where the image is present directly on the tablet) or the VersaVueTM Video Box (a standalone imaging transfer system which can be connected to a computer to project live imaging), that provides live imaging of the lower urinary tract.
Please refer to the online application posting for the VersaVueTM Single-Use Flexible Cystoscope, available at https://mearis.cms.gov/public/publications/device-ptp/DEP250211C4HRV.
Comment: A commenter expressed its support for approval of transitional pass-through payment for the VersaVueTM Single-Use Flexible Cystoscope. The commenter stated that single-use, disposable cystoscopes provide clinical benefit in the diagnosis and treatment of lower urinary tract diseases and the elimination of reprocessing steps, not only enhancing safety but also streamlining workflow and improving operational efficiency for providers.
Response: We appreciate the commenter's input. We have taken this comment into consideration in our final determination for pass-through status for the VersaVueTM Single-Use Flexible Cystoscope.
As stated previously, to be eligible for transitional pass-through payment under the OPPS, a device must meet the criteria at § 419.66(b)(1) through (4). With respect to the newness criterion at § 419.66(b)(1), FDA granted the applicant 510(k) clearance for the VersaVueTM Single-Use Flexible Cystoscope on October 6, 2023. The approved FDA indication for the VersaVueTM Single-Use Flexible Cystoscope is:
- The VersaVueTM Single-Use Flexible Cystoscope is a sterile, single-use, and flexible device intended to be operated with its compatible display system (VersaVueTM Tablet or VersaVueTM Video Box). The device provides endoscopic procedure and surgical treatment within the lower urinary tract. The Cystoscope is intended to provide visualization via [the] displaying unit. The Cystoscope is intended for use in a hospital environment or medical office environment. It is designed for use in adults.
We received the application for a new device category for transitional pass-through payment status for the VersaVueTM Single-Use Flexible Cystoscope on February 11, 2025, which is within 3 years of the date of the initial FDA marketing authorization.
We invited public comments on whether the VersaVueTM Single-Use Flexible Cystoscope meets the newness criterion at § 419.66(b)(1).
Comment: With respect to the newness criterion at § 419.66(b)(1), the applicant reiterated that the VersaVueTM Single-Use Flexible Cystoscope received 510(k) clearance from FDA on October 6, 2023, and that it submitted an application for pass-through status on February 11, 2025, which is within 3 years of the initial market authorization.
Response: We appreciate the applicant's input. We received the application for a new device category for transitional pass-through payment status for the VersaVueTM Single-Use Flexible Cystoscope on February 11, 2025, which is within 3 years of October 6, 2023, the date of FDA 510(k) clearance. After consideration of the public comment we received and our review of the application, we have determined that the VersaVueTM Single-Use Flexible Cystoscope meets the newness criterion at § 419.66(b)(1).
With respect to the eligibility criteria at § 419.66(b)(3), the device must be an integral part of the service furnished, be used for one patient only, come in contact with human tissue, and be surgically inserted or implanted, or applied in or on a wound or other skin lesion. Per the applicant, the VersaVueTM Single-Use Flexible Cystoscope meets the requirements at § 419.66(b)(3).
We invited public comments on whether the VersaVueTM Single-Use Flexible Cystoscope meets the eligibility criterion at § 419.66(b)(3).
Comment: The applicant reiterated that the VersaVueTM Single-Use Flexible Cystoscope is an integral part of a cystoscopy procedure, used for one patient only, comes in contact with human tissue, and is surgically inserted during a cystoscopy procedure.
Response: We appreciate the applicant's input. We agree with the applicant that the VersaVueTM Single-Use Flexible Cystoscope is an integral part of the service furnished, used for one patient only, comes in contact with human tissue, and is surgically implanted or inserted. After consideration of the public comment we received, and our review of the ( printed page 53662) application, we have determined that the VersaVueTM Single-Use Flexible Cystoscope meets the eligibility criterion at § 419.66(b)(3).
With respect to the exclusion criteria at § 419.66(b)(4), a device is not eligible to be considered for pass-through payment if it is any of the following: (1) equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciable assets as defined in Chapter 1 of the Medicare Provider Reimbursement Manual (CMS Pub. 15-1); or (2) a material or supply furnished incident to a service (for example, a suture, customized surgical kit, or clip, other than a radiological site marker). Per the applicant, the VersaVueTM Single-Use Flexible Cystoscope is (1) not considered equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciable assets, and is (2) not a material or supply furnished incident to a service, and, therefore, is eligible to be considered for pass-through payment.
We invited public comments on whether the VersaVueTM Single-Use Flexible Cystoscope meets the exclusion criterion at § 419.66(b)(4).
Comment: The applicant reiterated that the VersaVueTM Single-Use Flexible Cystoscope is not a supply furnished incident to the service, is a single-use cystoscope, is not subject to capital equipment depreciation schedules, and cannot be reprocessed.
Response: We appreciate the applicant's input. We agree with the applicant that the VersaVueTM Single-Use Flexible Cystoscope is not equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciable assets, or a material or supply furnished incident to a service. After consideration of the public comment we received and our review of the application, we have determined that the VersaVueTM Single-Use Flexible Cystoscope meets the criterion at § 419.66(b)(4).
In addition to the criteria at § 419.66(b)(1) through (4), the criteria for establishing new device categories are specified at § 419.66(c). The first criterion, at § 419.66(c)(1), provides that CMS determines that a device to be included in the category is not appropriately described by any of the existing categories or by any category previously in effect, and was not being paid for as an outpatient service as of December 31, 1996. Per the applicant, the existing pass-through code C1747 (Endoscope, single-use ( i.e., disposable), urinary tract, imaging/illumination device (insertable)) does not appropriately describe the VersaVue[TM] Single-Use Flexible Cystoscope because cystourethroscopy procedures are not encompassed by this pass-through device category.[46] The applicant also stated that the existing code C1889 (Implantable/insertable device, not otherwise classified) does not appropriately describe the VersaVueTM Single-Use Flexible Cystoscope. We noted in the CY 2026 OPPS/ASC proposed rule that C1889 is not a device pass-through category code and therefore would not describe the VersaVueTM Single-Use Flexible Cystoscope for the purposes of device pass-through status. Upon review, we stated that we did not identify an existing pass-through payment category that describes the VersaVueTM Single-Use Flexible Cystoscope.
We invited public comment on whether the VersaVueTM Single-Use Flexible Cystoscope meets the device category criterion at § 419.66(c)(1).
Comment: The applicant commented that FDA first issued the VersaVueTM Single-Use Flexible Cystoscope 510(k) clearance in 2023, that the VersaVueTM Single-Use Flexible Cystoscope was not receiving payment as an outpatient service as of December 31, 1996, and that it is not described by any current or previous device categories. The applicant stated that, in correspondence between Boston Scientific and CMS from September 2023, CMS determined that C1747 (Endoscope, single-use ( i.e., disposable), urinary tract, imaging/illumination device (insertable)) does not include cystoscopy or cystourethroscopy devices.
Response: We appreciate the applicant's input. After consideration of the public comment we received and our review of the application, we continue to believe that there is no existing category or category previously in effect that appropriately describes the VersaVueTM Single-Use Flexible Cystoscope. Therefore, we have determined that the VersaVueTM Single-Use Flexible Cystoscope meets the device category eligibility criterion at § 419.66(c)(1).
The second criterion for establishing a device category, at § 419.66(c)(2), provides that CMS determines either of the following: (1) that a device to be included in the category has demonstrated that it will substantially improve the diagnosis or treatment of an illness or injury or improve the functioning of a malformed body part compared to the benefits of a device or devices in a previously established category or other available treatment; or (2) for devices for which pass-through status will begin on or after January 1, 2020, as an alternative to the substantial clinical improvement criterion, the device is part of the FDA's Breakthrough Devices Program and has received FDA marketing authorization for the indication covered by the Breakthrough Device designation. The applicant asserted that single-use, disposable cystoscopes, including the VersaVueTM Single-Use Flexible Cystoscope, represent a substantial clinical improvement over reusable cystoscopes.
The applicant provided five documents to support these claims, which included three studies and two FDA communications concerning reusable, reprocessed urological endoscopes. The applicant's assertions regarding the substantial clinical improvement criterion are shown in Table 103, previously published as Table 55 of the CY 2026 OPPS/ASC proposed rule (90 FR 33600 and 33601). Please see the online posting for the VersaVueTM Single-Use Flexible Cystoscope for the applicant's complete statements regarding the substantial clinical improvement criterion and the supporting evidence provided.
( printed page 53663) ( printed page 53664)After review of the information provided by the applicant, we stated in the CY 2026 OPPS/ASC proposed rule that we had the following concerns regarding whether the VersaVueTM Single-Use Flexible Cystoscope meets the substantial clinical improvement criterion.
Overall, we noted that the applicant indicated that the technology does not offer a treatment option for patients unresponsive to or ineligible for currently available treatments, stating that the same patient population could be treated using a reusable, reprocessed cystoscope. Further, the applicant did not claim that the nominated device, the VersaVueTM Single-Use Flexible Cystoscope, offers a substantial clinical improvement over other single-use, disposable cystoscopes available on the market. Specifically, the applicant stated that no claim is being made that a specific disposable device offers a substantial clinical improvement over other disposable devices in the same category. Rather, the applicant stated that it presented evidence to support its claim that single-use, disposable cystoscopes (as a group) demonstrate substantial clinical improvement over reusable cystoscopes. We noted that for the purposes of the device pass-through evaluation process, CMS evaluates the nominated device that is the subject of an application to determine if the device meets the eligibility criteria described in § 419.66.
Further, for the purposes of our substantial clinical improvement evaluation in the CY 2026 OPPS/ASC proposed rule, we considered both reusable, reprocessed cystoscopes and single-use, disposable cystoscopes as available treatment options for this patient population and noted that single-use, disposable cystoscopes appear to be widely accessible and well utilized in the outpatient setting. According to the applicant, of the 2.2 million flexible cystoscopy procedures furnished annually across all payers, 23 percent are performed with single-use, disposable cystoscopes. As discussed in more detail in this section, we stated in the CY 2026 OPPS/ASC proposed rule that we were interested in additional evidence that demonstrates substantial clinical improvement with the use of the VersaVueTM Single-Use Flexible Cystoscope over other available treatment options (both single-use, disposable cystoscopes and reusable, reprocessed cystoscopes). In order to evaluate substantial clinical improvement over currently available treatments to meet the transitional pass-through payment criterion at § 419.66(c)(2), we stated that we consider supporting evidence, preferably published peer-reviewed clinical trials, that demonstrates improved clinical outcomes, such as reduction in mortality, complications, subsequent interventions, future hospitalizations, recovery time, pain, or a more rapid beneficial resolution of the disease process comparing the nominated device to the standard of care (88 FR 81733).
We stated in the CY 2026 OPPS/ASC proposed rule that, first, the evidence provided did not include data demonstrating that the use of the VersaVue[TM] Single-Use Flexible Cystoscope compared to other available single-use, disposable cystoscopes for this patient population results in substantial clinical improvement. The applicant identified other devices it believes are closely related or similar to the VersaVue[TM] Single-Use Flexible Cystoscope, including the following: (1) Ambu® aScope 4[TM] Cysto manufactured by AMBU A/S, (2) Ambu® aScope 5[TM] Cysto manufactured by AMBU A/S, (3) WiScope® SingleUse- Digital Flexible Cystoscope manufactured by OTU Medical AnQing, (4) Medical Single Use Flexible Cystoscope manufactured by Shanghai AnQing Medical Instrument Company, and (5) Pusen Single Use Flexible Video Cystoscope System manufactured by Zhuhai Pusen Medical Technology Company. We noted that the VersaVue[TM] Single-Use Flexible Cystoscope was determined to be substantially equivalent to a legally marketed device, the Ambu® aScope 4[TM] Cysto (K193095), which received 510(k) clearance on April 2, 2020.[47] The ( printed page 53665) FDA 510(k) summary for the VersaVueTM Single-Use Flexible Cystoscope stated that both devices have the same intended use and similar specifications, and that there are no significant differences. According to the applicant, these five similar devices would also become eligible for transitional pass-through payment under the additional category proposed by the applicant. We reiterated that we consider other single-use, disposable cystoscopes as available treatment options for this patient population and that the devices appear to share similar technological and/or procedural characteristics. We noted that none of the studies the applicant included reference another single-use, disposable device as a comparator against which to evaluate and assess the VersaVueTM Single-Use Flexible Cystoscope. While we found that the source articles provided background information about multiple risks associated with reprocessing reusable devices, we stated that we would welcome additional evidence demonstrating a comparison of the VersaVueTM Single-Use Flexible Cystoscope's performance against other similar single-use, disposable devices. We questioned whether the VersaVueTM Single-Use Flexible Cystoscope offers a substantial clinical improvement over other single-use, disposable cystoscopes currently on the market. We stated that we would welcome evidence that demonstrates substantial clinical improvement with the use of the VersaVueTM Single-Use Flexible Cystoscope over other single-use, disposable cystoscopes.
Second, in the CY 2026 OPPS/ASC proposed rule, we questioned whether the supporting evidence submitted by the applicant demonstrates substantial clinical improvement of the VersaVue[TM] Single-Use Flexible Cystoscope over reusable, reprocessed cystoscopes for this patient population. In the first claim, the applicant asserted that the use of single-use, disposable cystoscopes decreases post-procedure encounters and infections compared to reusable cystoscope devices. However, while Geldmaker et al. (2023) reported some improved clinical outcomes with the use of a specific single-use, disposable cystoscope when compared to the use of a specific reusable cystoscope,[48] we noted that the study does not assess, evaluate, or review clinical outcomes associated with the use of the VersaVueTM Single-Use Flexible Cystoscope or compare clinical outcomes associated with the use of the VersaVueTM Single-Use Flexible Cystoscope to reusable cystoscopes. Rather, we stated that the evidence provided compared clinical outcomes associated with another device, the single-use, disposable Ambu aS4C cystoscope [Ambu® aScope 4TM Cysto] to the reuseable Olympus® CYF-5 V2 Flexible cystoscope. Therefore, we questioned whether the use of the VersaVueTM Single-Use Flexible Cystoscope resulted in substantial clinical improvement as compared to reusable, reprocessed cystoscopes.
In addition, we noted that, as a retrospective study, Geldmaker et al. (2023) fails to establish that the differences in the observed clinical outcomes are caused by using reusable, reprocessed cystoscopes versus single-use, disposable cystoscopes. We noted that retrospective studies can only suggest associations between variables and cannot establish cause and effect relationships. While the propensity score matching did an adequate job of balancing the two groups (reusable, reprocessed cystoscope procedures versus single-use, disposable cystoscope procedures) and yielded statistically significant results, we questioned whether the propensity score matching variables used in the study adequately account for patient factors that may impact the outcomes, such as the reason for the cystoscopy, positive preoperative UTI, and other comorbid conditions. We noted that data were collected during different time periods (reusable, reprocessed cystoscope data were collected in 2020, and single-use, disposable cystoscope data were collected in 2021), which may introduce systematic errors in the measurement due to retrospective data collection or confounders not accounted for, such as changes in clinical practice between the 2 study years. Further, per the study authors, we noted that urine cultures were ordered more frequently in the reusable cystoscope group, potentially increasing the likelihood of a UTI diagnosis in the reusable cystoscope group. We stated that we would be interested in whether equivalent pre- and post-procedure urine cultures from patients in both groups would have yielded different results. We noted that the evidence is not conclusive to support whether the use of single-use, disposable cystoscopes results in improved clinical outcomes compared to reusable, reprocessed cystoscopes.
Moreover, while not included in the evidence submitted by the applicant in support of the substantial clinical improvement claims for the VersaVue[TM] Single-Use Flexible Cystoscope, we noted two studies, Anderson et al. (2024) and Johnson et al. (2023), provide notable evidence directly related to the use of single-use, disposable cystoscopes versus reusable cystoscopes.[49 50] Anderson et al. (2024), a systematic review (using meta-analyses techniques) comparing the clinical outcomes of all single-use, disposable endoscopes used in urology with those of reusable endoscopes across a range of urological procedures, found that of the seven studies that reported the rate of postoperative infections, none found a statistically significant difference in postoperative infection rates between single-use, disposable endoscopes and reusable endoscopes.[51] Further, we noted that the Anderson et al. (2024) sub-group analysis of cystoscopes found no difference in overall complication rates or postoperative infection rates between the single-use, disposable cystoscopes and the reusable cystoscope subgroups. Similarly, Johnson et al. (2023) found no statistically significant difference in adverse events in a multicenter, randomized trial comparing single-use, disposable cystoscopes (Ambu® aScope 4[TM] Cysto) with reusable cystoscopes for ureteral stent removal in 102 patients.[52] Given the evidence in these additional studies, we questioned in the CY 2026 OPPS/ASC proposed rule whether the totality of available evidence establishes that the use of a single-use, disposable cystoscope results in substantial clinical improvement when compared to reprocessed cystoscopes, and furthermore, whether the use of the VersaVueTM Single-Use Flexible Cystoscope compared to reusable, reprocessed cystoscopes results in decreased adverse events, including post-procedure encounters and infections. We stated that we would welcome studies that evaluate whether the use of the VersaVueTM Single-Use Flexible Cystoscope results in ( printed page 53666) substantial clinical improvement over reusable, reprocessed cystoscopes, such as a reduction in mortality, complications, subsequent interventions, future hospitalizations, recovery time, pain, or a more rapid beneficial resolution of the disease process compared to reusable, reprocessed cystoscopes.
Third, in the second, third, fourth and fifth claims, the applicant asserted that the use of single-use, disposable cystoscopes avoids post-cystoscopy infections, device malfunctions, and contamination problems associated with reusable devices, and eliminates the need for reprocessing and avoids the risk of infection associated with improper reprocessing. In support of these claims, the applicant provided two retrospective reviews (Lee et al., 2022 and Muscarella, 2022) of medical device reports (MDRs) from the FDA Manufacturer and User Facility Device Experience (MAUDE) database, an FDA News Release (2021, April 1) concerning infection and contamination risks associated with reusable urological endoscopes, and an FDA Update (2025, January 31) communicating the recall of endoscope accessories from Olympus® reusable urological endoscopes as supporting evidence.[53 54 55 56] First, we questioned whether, per the applicant, the avoidance of device malfunctions, contamination problems, and the elimination of the need for reprocessing demonstrates substantial clinical improvement, as these are not clinical outcome metrics. Second, while we concurred that avoiding post-cystoscopy infections is important, we noted that none of the studies the applicant submitted as evidence evaluated or assessed the VersaVue[TM] Single-Use Flexible Cystoscope and that none of the studies compared clinical outcomes, such as adverse events (including post-cystoscopy infection) associated with the use of the VersaVue[TM] Single-Use Flexible Cystoscope to clinical outcomes associated with reusable cystoscopes. Third, while these studies discuss potential adverse events from reusable cystoscope procedures, we noted that FDA states that the FDA MAUDE database's MDR data are not intended to be used to evaluate rates of adverse events, evaluate a change in event rates over time, or compare adverse event occurrence rates across devices.[57] FDA explains that the MAUDE database is a passive surveillance system, and that incidence, prevalence, or cause of an event cannot be determined from this surveillance system alone due to under-reporting of events, inaccuracies in reports, lack of verification that the device caused the reported event, and lack of information about frequency of device use.[58] FDA further explains that the submission of an MDR itself does not necessarily demonstrate that the device caused or contributed to the adverse outcome or event.[59] Therefore, we questioned whether these reports can substantiate that the use of single-use, disposable cystoscopes, like the VersaVue[TM] Single-Use Flexible Cystoscope, would result in substantial clinical improvements over currently available reusable, reprocessed cystoscopes. Fourth, while the applicant asserted that the FDA News Release (2021, April 1) encouraged manufacturers to transition to single-use, disposable devices, we noted that this FDA News Release does not specifically reference single-use, disposable cystoscopes but, rather encouraged manufacturers to transition to devices with features that eliminate the need for reprocessing and provided information to manufacturers on how to modify and validate their reprocessing instructions. As such, we questioned the assertion that this FDA communication encouraged manufacturers to transition to single-use, disposable cystoscopes, such as the VersaVue[TM] Single-Use Flexible Cystoscope. We further noted that FDA stated that the risk of infection from reusable, reprocessed urological endoscopes was low based on its data.[60] We also noted that the FDA Update (2025, January 31) communicated a medical device recall of the Olympus® endoscope accessory (MAJ-891 Forceps/Irrigation Plug) that is attached to the instrument channel port of a certain endoscope, due to the risk of infection that may result from improper reprocessing, but that this communication made no mention of the use of the nominated device or single-use, disposable cystoscopes, instead it appears to be a concern related to a particular reusable device component.[61]
While the applicant asserted that the use of single-use, disposable cystoscopes avoids risk of infection associated with improper reprocessing, the applicant did not submit any FDA safety communications directly related to single-use, disposable cystoscopes. We questioned whether the evidence provided by the applicant directly supports this claim.
Finally, we noted in the CY 2026 OPPS/ASC proposed rule that the intent of transitional device pass-through payment, as implemented at § 419.66, is to facilitate access for beneficiaries to the advantages of new and truly innovative devices by allowing for adequate payment for these new devices while the necessary cost data is collected to incorporate the costs for these devices into the procedure APC rate (66 FR 55861). Based on the information provided by the applicant, approximately 510,600 units of single-use, disposable cystoscope devices, like those that would be included in the proposed device category for single-use, disposable cystoscopes, are estimated to have sold annually in the U.S. Moreover, the applicant provided that, of the 2.2 million flexible cystoscopy procedures furnished annually, 23 percent are performed with single-use, disposable cystoscopes, further, single-use, disposable cystoscopes are used in at least 500 hospitals and clinics, including 35 to 50 academic medical centers. Based on the information provided in the application, we stated that it appears as though single-use, disposable cystoscopes are widely available and consistently utilized for the purposes of performing cystoscopy procedures in outpatient facilities. As such, we questioned whether the creation of a device pass-through payment category code for single-use, disposable cystoscopes is consistent ( printed page 53667) with the intent of transitional device pass-through payment and necessary to appropriately incorporate adequate cost data of these devices into the applicable procedure APC.
We questioned whether the evidence submitted by the applicant demonstrates that the use of single-use, disposable cystoscopes results in improved patient outcomes and reduced patient risk compared to the use of reusable devices. Further, we questioned whether the VersaVueTM Single-Use Flexible Cystoscope offers a substantial clinical improvement in the treatment of Medicare beneficiaries over other available treatment and whether a transitional device pass-through category for single-use, disposable cystoscopes is in alignment with the intent of the transitional device pass-through payment program policy.
We invited public comment on whether the VersaVueTM Single-Use Flexible Cystoscope meets the device category criterion at § 419.66(c)(2).
Comment: The applicant provided responses to concerns raised by CMS in the CY 2026 OPPS/ASC proposed rule. In response to the concern that the data provided did not compare the VersaVueTM Single-Use Flexible Cystoscope to other single-use, disposable cystoscopes, the applicant stated its belief that such evidence would not be relevant to this application, and that CMS did not explain why such evidence is necessary or how it could apply to the application under review. Further, the applicant explained that it did not assert that the VersaVueTM Single-Use Flexible Cystoscope offers a substantial clinical improvement compared to other single-use cystoscopes, stating that the substantial clinical improvement claims and evidence provided were in support of the category of single-use cystoscopes compared to reusable cystoscopes rather than the VersaVueTM Single-Use Flexible Cystoscope compared to other single-use, disposable cystoscopes or reusable cystoscopes. The applicant stated its belief that CMS has recognized similar claims for categories of other types of disposable endoscopes, including upper GI endoscopes, ureteroscopes, and bronchoscopes. As an example, the applicant cited language from the Uretero1TM decision in which CMS agreed that the device met the criterion for substantial clinical improvement, which stated that while comparative studies between Uretero1TM and other disposable devices would be helpful, CMS agreed that the evidence demonstrating improved outcomes and reduced patient risk associated with the disposable device in comparison with reusable devices represented substantial clinical improvement (87 FR 71932). The applicant asserted that the VersaVueTM Single-Use Flexible Cystoscope evidence is directly equivalent to the Uretero1TM evidence in both type and claims. Additionally, the applicant asserted that the Uretero1TM decision makes clear that the lack of such evidence comparing the nominated device to other disposable devices does not and should not preclude an applicant from meeting the substantial clinical improvement standard. The applicant stated that CMS should consider the VersaVueTM Single-Use Flexible Cystoscope application using the same standard applied to other recent decisions for disposable devices and determine that the improved patient outcomes and reduced patient risk associated with the disposable nominated device in comparison with reusable devices is a substantial clinical improvement.
Response: We appreciate the applicant's input. However, we maintain our concern regarding the lack of data comparing the VersaVueTM Single-Use Flexible Cystoscope to other single-use, disposable and reusable, reprocessed cystoscopes. First, regarding the applicant's statement that the evidence provided in support of the category of single-use cystoscopes should suffice for a demonstration of substantial clinical improvement, we reiterate that, CMS evaluates transitional pass-through applications using the substantial clinical improvement criterion described at § 419.66(c)(2)(i), which requires CMS to determine if “the device” that has been nominated has demonstrated that “it will substantially improve the diagnosis or treatment of an illness or injury or improve the functioning of a malformed body part compared to the benefits of a device or devices in a previously established category or other available treatment.” The regulatory provision refers to “the” device (singular) being compared to “a device or devices” for purposes of substantial clinical improvement. For the purposes of the device pass-through evaluation process, CMS evaluates the nominated device that is the subject of an application, not a category of devices, to determine if the device meets the eligibility criteria described in § 419.66. The applicant referenced studies comparing the category of single-use cystoscopes to reusable cystoscopes but did not provide evidence that its specific device demonstrates a substantial clinical improvement.
Second, we disagree with the applicant that CMS has recognized similar claims for other types of disposable endoscopes, including upper GI endoscopes, ureteroscopes, and bronchoscopes. For example, while we concluded that Uretero1TM met the criterion for substantial clinical improvement, we note that CMS agreed that the evidence demonstrated improved outcomes for “the disposable device” (singular), i.e., Uretero1TM (87 FR 71932). In response to additional comments, we explain more about our evaluation of the evidence submitted for both applications.
Finally, we disagree with the applicant that the evidence for the VersaVueTM Single-Use Flexible Cystoscope is equivalent to that used for Uretero1TM , and, as we previously explained in the CY 2024 and CY 2025 OPPS/ASC final rules with comment period for Ambu® aScopeTM 5 Broncho HD (88 FR 81736) and Ambu® aScopeTM Gastro (89 FR 94182), and as discussed in more detail in our responses to comment, we evaluate all evidence submitted for each device pass-through application as it applies to the nominated device. Due to inherent differences in the devices themselves and the supporting documentation submitted, CMS may have different concerns as they relate to the nominated device. As discussed in the following responses, we continue to believe that the evidence submitted for the VersaVueTM Single-Use Flexible Cystoscope application fails to demonstrate a substantial clinical improvement as required.
Comment: In response to our concerns that the single-use device included in the Geldmaker et al. (2023) study was not the VersaVueTM Single-Use Flexible Cystoscope, the applicant commented that the Geldmaker et al. (2023) study provides important evidence about the impact of disposable cystoscope use on clinical practice and demonstrates one of the substantial clinical improvement claims for single-use cystoscopes. The applicant acknowledged that the device involved in this study was not the VersaVueTM Single-Use Flexible Cystoscope but asserted that this situation is identical to the Uretero1TM transitional pass-through evaluation and approval. The applicant asserted that the Uretero1TM application used a study by Bozzini et al. (2021) to support the claims that Uretero1TM reduced hospitalization rates, antibiotic therapy, complication rates, and post-operative infection rates. The applicant further asserted that the Bozzini et al. (2021) study did not utilize Uretero1TM but the study findings were directly applicable ( printed page 53668) to the Uretero1TM application. The applicant stated its belief that CMS appropriately accepted that study in support of the Uretero1TM substantial clinical improvement claims, allowing the findings to be directly applicable to Uretero1TM . The applicant opined that CMS should apply the same standard to the VersaVueTM Single-Use Flexible Cystoscope application and recognize that Geldmaker et al. (2023) demonstrates the substantial clinical improvement afforded by use of disposable cystoscopes, including the VersaVueTM Single-Use Flexible Cystoscope.
In response to our concerns regarding the Geldmaker et al. (2023) study's design, the applicant commented that the study is a high-quality, retrospective analysis of a large outpatient urology practice's real-world experience following conversion from reusable to disposable cystoscopes and stated that it strongly disagrees with CMS's assessment of the study in the CY 2026 OPPS/ASC proposed rule. Additionally, the applicant asserted that the specific substantial clinical improvement claim addressed by Geldmaker et al. (2023) is the impact that the type of scope has on post-procedural health care resource utilization—that is, whether the type of scope affects unplanned interactions or the incidence of UTIs. The applicant further asserted that a large study population is essential for meaningful findings and this retrospective review allowed the authors to include 1,000 patients.
Regarding our question whether the propensity score matching variables adequately account for patient factors, such as the reason for cystoscopy, positive preoperative UTI, and other comorbid conditions, the applicant stated that the reason for cystoscopy, specifically including UTI, was the basis for the propensity score weighting. The applicant also noted that after propensity-score weighting, the study considered all differences in the reason(s) for cystoscopy to be negligible and collected data from a single institution's integrated electronic medical record. The applicant asserted its belief that the specific focus on equivalent pre- and post-procedure urine cultures suggests that CMS may have misunderstood the intent of the Geldmaker et al. (2023) study. The applicant stated that the study included urine cultures which were only ordered based on post-procedure, patient-reported lower urinary tract symptoms and noted that Geldmaker et al. (2023) found patients in the reusable scope group were three times as likely to have an encounter, which would likely include reporting of symptoms, and therefore, it is not surprising that the group also had more urine cultures ordered. The applicant agreed that systematic urine cultures would have produced different evidence related to the incidence of UTIs, but it would also have eliminated key data on health resource utilization. The applicant asserted its belief that the study was appropriately designed and provides strong evidence that the use of disposable scopes is associated with reduced post-procedure encounters.
Response: We appreciate the applicant's input. However, we maintain our concerns listed in the CY 2026 OPPS/ASC proposed rule regarding the Geldmaker et al. (2023) study. First, the device involved in this study was not the VersaVue[TM] Single-Use Flexible Cystoscope, and second, the Geldmaker et al. (2023) study has design limitations as discussed. As previously stated, we disagree with the applicant that this situation is identical to the Uretero1[TM] transitional pass-through evaluation and approval. First, no two applications are identical in nature given the fact that the basic premise of device pass-through is to provide additional payment for novel technologies and, as such, these innate differences for each technology requires a robust evaluation of each application on its own merits. As the applicant indicated, CMS approved Uretero1[TM 62] for transitional pass-through payment status in the CY 2023 OPPS/ASC final rule with comment period. We note that we expressed similar concerns relating to the lack of comparative studies between the single-use Uretero1TM device and other disposable devices and indicated that, while we ultimately agreed that the totality of evidence demonstrated improved patient outcomes and reduced patient risk associated with the disposable device in comparison with reusable devices represents substantial clinical improvement, we stated it would have been helpful to see comparative studies (87 FR 71932). The applicant seems to suggest that because we determined that the Uretero1TM device demonstrated substantial clinical improvement despite providing a study which did not include the nominated device as a comparator, that we must similarly determine that the type of evidence submitted for the VersaVueTM Single-Use Flexible Cystoscope represents substantial clinical improvement. We do not believe that this implied approach to application evaluation is appropriate. Rather, we continue to believe that our current process, wherein we evaluate all evidence submitted for each device pass-through application as it applies to the nominated device, is appropriate and consistent with the regulatory requirements. We reiterate that, due to inherent differences in the devices themselves and the supporting documentation submitted, CMS may have different concerns as they relate to the nominated device. In addition, we are not precluded from evaluating evidence, expressing concerns, or making a determination on the applicability or validity regarding evidence submitted in support of an application, simply because that type of evidence has been submitted in support of a previous application.
Furthermore, we maintain our concerns listed in the CY 2026 OPPS/ASC proposed rule that the Geldmaker et al. (2023) study has design limitations. Specifically, we note that retrospective studies can only suggest associations between variables and cannot establish causation. While we appreciate the applicant's explanation about the propensity score matching in Geldmaker et al. (2023), we question whether the propensity score matching variables used in the study adequately account for all patient factors that may impact outcomes, such as the reason for cystoscopy and other comorbid conditions. We also maintain our interest in whether equivalent pre- and post-procedure urine cultures from patients in both study groups would have yielded different results, which the applicant agreed would have produced different evidence related to UTI incidence. Finally, we continue to maintain concerns over potential systematic errors in measurement due to retrospective data collection and unaccounted for confounders, such as changes in clinical practice between the 2 study years.
The Geldmaker et al. (2023) study did not assess, evaluate, review, or compare clinical outcomes associated with the use of the VersaVueTM Single-Use Flexible Cystoscope to reusable cystoscopes. Likewise, none of the other evidence provided compared the benefits of the VersaVueTM Single-Use Flexible Cystoscope to currently available treatments, including other single-use cystoscopes. For these reasons, we do not believe that the Geldmaker et al. (2023) study supports a demonstration of substantial clinical improvement. ( printed page 53669)
Based on our review, we continue to believe that the current application lacks sufficient evidence to demonstrate that the VersaVueTM Single-Use Flexible Cystoscope significantly improves clinical outcomes in patients receiving cystoscopy using either reusable scopes or other single-use scopes.
Comment: In response to our request for additional evidence of substantial clinical improvement, the applicant stated its belief that the Anderson et al. (2024) and Johnson et al. (2023) studies, which we identified in the CY 2026 OPPS/ASC proposed rule, have substantial limitations. Specifically, the applicant asserted that while the Anderson et al. (2024) meta-analysis included 12 studies, only 4 of them involved cystoscopes, which makes the subgroup analysis particularly challenging. The applicant also stated that the cystoscope studies were not specifically intended to identify post-procedure complications and that two of the included studies provided no data on those outcomes. Additionally, the applicant noted that only one of the two remaining cystoscopy studies reported the overall complication rate, and the other cystoscopy study reported the post-operative infection rate. Therefore, the applicant asserted that when Anderson et al. (2024) reported a disposable cystoscopes subgroup analysis for each of these outcomes, the study was simply reporting the result of a single study from its meta-analysis. The applicant also noted Anderson et al. (2024) supports the understanding that disposable scope use reduces the risk of infection and explains why data is unavailable to document that conclusion, stating:
The use of single-use endoscopes should reduce the risk of transmission of these important public health infections to a negligible level. However, the absolute risk of transmission of these pathogens with reusable endoscopes is so low that it is unlikely any study will be sufficiently powered to show a difference when compared to [single-use flexible ureteroscopes].
In addition, the applicant stated that the Johnson et al. (2023) study, a multicenter, randomized trial that compared disposable cystoscopes with reusable cystoscopes for ureteral stent removal, found no statistically significant difference in adverse events. The applicant asserted that based on the study design, this outcome is unsurprising and should not be considered evidence that no difference exists in adverse events for single-use versus multiple-use cystoscopes. The applicant noted that Johnson et al. (2023) was a randomized, dual-arm post-market clinical trial that compared single-use cystoscopes (Ambu® aScopeTM 4 Cysto; Ambu A/S) for removal of ureteral stents to routine flexible reusable cystoscopes (combination of digital and fiberoptic). The applicant further opined that, although not explicitly stated, the paper presumably calculated the sample size based on the primary endpoint of stent removal, not to detect any clinical differences and, while Johnson et al. (2023) collected patient-reported adverse event data as a secondary endpoint, the study would have been severely underpowered to detect any group differences in adverse events. The applicant added that the single serious adverse event observed during the study occurred in the standard of care reusable arm and that the authors noted that patients in the reusable cystoscope group reported significantly higher pain with urination post-procedure.
The applicant submitted a new study, Chew et al. (2025), with its public comment. According to the applicant, Chew et al. (2025),[63] is a retrospective study that compared 30-day health care utilization between patients treated with single-use cystoscopes and patients treated with reusable cystoscopes. The applicant stated that the Chew et al. (2025) study used propensity score matching (1:5 ratio) to control for demographics, comorbidities, clinical history, and prior utilization and performed a pre-defined subgroup analysis in patients aged 65 years. The applicant also noted that of the study's 62,965 eligible encounters, 1,473 (2.3 percent) used single-use cystoscopes and 61,492 (97.7 percent) used reusable cystoscopes. The applicant explained that, after propensity score matching, Chew et al. (2025) compared 1,473 single-use procedures with 7,365 reusable procedures and found that single-use devices resulted in lower 30-day health care utilization (5.2 percent vs. 13.0 percent; hazard ratio (HR)=0.34; 95 percent confidence interval (CI): 0.26, 0.45; p<0.001). The applicant further noted that the study's component analyses favored single-use cystoscopes for acute care events (p<0.001), emergency department visits (p=0.03), same-day surgery (p=0.01), and clinic visits (p<0.001). The applicant reported that Chew et al. (2025) found that single-use cystoscopes resulted in lower complication rates (1.8 percent vs. 4.3 percent; HR=0.40; 95 percent CI: 0.27, 0.60; p<0.001) and serious complications (HR=0.65, 95 percent CI: 0.49, 0.86; p=0.003), and that these findings were similar in patients aged 65 years. The applicant asserted that this study provides important new evidence that use of disposable cystoscopes is associated with a lower risk of serious complications and found comparable infection rates between disposable and reusable cystoscopes. The applicant acknowledged that Chew et al. (2025) has the same underpowering issue identified in Anderson et al. (2024) and stated that the study's authors speculate that mechanical factors associated with reprocessing may influence morbidity following cystoscopy. The applicant expressed its belief that the totality of the evidence, particularly with the addition of the Chew et al. (2025) study, strongly supports that single-use, disposable cystoscope use reduces complication rates, post-procedure encounters, and health care utilization.
Response: We appreciate the applicant's input. However, we maintain our concerns listed in the CY 2026 OPPS/ASC proposed rule that additional evidence is needed to establish the VersaVueTM Single-Use Flexible Cystoscope's substantial clinical improvement over reusable, reprocessed cystoscopes or other single-use cystoscopes, such as that which shows a reduction in mortality, complications, subsequent interventions, future hospitalizations, recovery time, pain, or a more rapid beneficial resolution of the disease process.
We appreciate the applicant's critique of the Anderson et al. (2024) and Johnson et al. (2023) studies and for sharing the Chew et al. (2025) study. While we agree that all studies have their limitations, we note that Anderson et al. (2024) is reportedly the largest study to compare outcomes of single-use urologic endoscopes to those of reusable endoscopes and presents evidence suggesting that single-use urologic endoscopes provide no improvement with regard to postoperative infection rates or overall complication rates compared to reusable urologic endoscopes. This is contradictory to the applicant's assertions that the VersaVueTM Single-Use Flexible Cystoscope or any other single-use disposable cystoscope represents a substantial clinical improvement compared to a reusable cystoscope.
Moreover, we have concerns regarding the Chew et al. (2025) study. First, similar to our concern with ( printed page 53670) Geldmaker et al. (2023), Chew et al. (2025) does not compare the VersaVueTM Single-Use Flexible Cystoscope to other single-use or reusable devices, and retrospective studies can only suggest associations between variables, not establish causation. Second, the methodology used by Chew et al. (2025) for selecting and matching cases retrospectively is unclear, specifically, how the 7,365 matched reusable procedures were selected from the potential pool of 62,965. We also question if the study's results demonstrate substantial clinical improvement of single-use cystoscopes compared to reusable cystoscopes. While we acknowledge that Chew et al. (2025) reports an association between single-use cystoscopes and significantly lower 30-day complication rates (cumulative incidence: 1.8 percent vs. 4.3 percent) with a 60 percent relative risk reduction (HR=0.40; 95 percent CI: 0.27, 0.60; p<0.001), we note that there were no statistical differences between the single-use device group and the reusable device group in regard to recatheterization (47 percent risk reduction; p=0.11), UTI (6 percent risk increase; p=0.85), or sepsis/bacteremia (87 percent risk reduction; p=0.17). Finally, we note that the Chew et al. (2025) study cited the systematic review by Anderson et al. (2024) of 21 studies that reported no differences in overall complications or infection rates between single-use and reusable urologic endoscopes.
Comment: In response to our question whether the avoidance of device malfunctions, contamination problems, and the elimination of the need for reprocessing are clinical outcome metrics, the applicant commented that these are items that put patients at risk of complications and additional healthcare encounters, and that avoidance of these outcomes is clearly a clinical benefit. The applicant stated that CMS has recognized the complication risk in consideration of previous device pass-through applications. Specifically, the applicant pointed to CMS's consideration of the Utereo1TM pass-through application in which CMS concluded that the evidence demonstrated that the improved patient outcomes and reduced patient risk associated with the disposable device in comparison to reusable devices represented a substantial clinical improvement. The applicant asserted that many of the substantial clinical improvement claims for Uretero1TM related to these same outcomes, including infection transmission prevention, contamination risk reduction, procedure delays reduction, and increased patient safety and education. The applicant further expressed its belief that CMS should consider reduction in patient risk as a clinical outcome metric and recognize this evidence as demonstrating a substantial clinical improvement for the VersaVueTM Single-Use Flexible Cystoscope.
Response: We appreciate the applicant's input. We agree that risk of device malfunction, device contamination, and faulty reprocessing are important considerations that may affect device integrity and subsequent clinical outcomes. However, we note that these risks may not be the same between different types of devices, such as a ureteroscope and a cystoscope, and they may not even be the same between different models of similar devices, such as two cystoscopes. Further, for some devices, these risks may be so small that they render no significant clinical impact on a patient population. Thus, we believe it is not sufficient to simply argue that the elimination of risk must result in a substantial clinical improvement, especially if that risk is small. Therefore, we do not believe the applicant has sufficiently established that avoidance of device malfunctions, contamination problems, and the elimination of the need for reprocessing demonstrates substantial clinical improvement.
We further note that when there are currently-available treatment options for a patient population (as is the case for the VersaVue[TM] Single-Use Flexible Cystoscope), substantial clinical improvement is demonstrated when the candidate device demonstrates significantly improved clinical outcomes compared to the currently available treatments.[64] We evaluate all evidence submitted for each device pass-through application as it applies to the nominated device that is the subject of the application. In this context, the submitted evidence in support of the VersaVueTM Single-Use Flexible Cystoscope's substantial clinical improvement must demonstrate that the VersaVueTM Single-Use Flexible Cystoscope results in substantial clinical improvement when compared to available reusable and single-use devices for the treatment of the patient population. Inferences that the device may improve clinical outcomes because it may obviate complications associated with other available treatments are insufficient to demonstrate substantial clinical improvement. Therefore, the inferences derived from submitted evidence here do not establish substantial clinical improvement.
Comment: In response to our concerns regarding the use of FDA data in support of substantial clinical improvement claims, the applicant asserted that CMS cited two concerns with the use of FDA information in the VersaVueTM Single-Use Flexible Cystoscope application. Per the applicant, CMS cited guidance from FDA about use of MDRs compiled in the MAUDE database and questioned whether a study that includes analysis of that data can be used to demonstrate substantial clinical improvement. The applicant stated that CMS also questioned the appropriateness of an FDA News Release concerning infection and contamination risks associated with reusable urological endoscopes, including cystoscopes, because the release did not specifically mention single-use devices.
The applicant commented that these concerns appear to represent a reversal of CMS's position taken 3 years prior when evaluating the Uretero1TM application. The applicant further asserted that one of the primary studies, Ofstead et al. (2022), used for the Uretero1TM application's literature review included 892 MDRs submitted to FDA, and that CMS raised no concerns about use of MAUDE data in that application.
Regarding the FDA News Release, the applicant stated that FDA encouraged manufacturers to transition to devices with features that eliminate the need for reprocessing and helped manufacturers modify and validate their reprocessing instructions. The applicant opined that single-use devices are not reprocessed, thus eliminating the need for reprocessing. The applicant further commented that the Uretero1TM application included the same release and that CMS noted at that time that the release did not specifically mention single-use devices. The applicant stated that, despite receiving no comments as to whether Uretero1TM met the substantial clinical improvement criteria, CMS approved Uretero1TM for device pass-through and therefore, the applicant inferred that CMS concluded that this concern, while noted, was not significant. The applicant acknowledged that CMS's intent is to evaluate applications by consistently applying the standards for each criterion, and that each pass-through application presents its own unique circumstances, but, according to the applicant, the similarities between the Uretero1TM and the VersaVueTM Single-Use Flexible Cystoscope applications are ( printed page 53671) overwhelming. According to the applicant, both devices are urological endoscopes, both applications include evidence that is not specific to the nominated device, and both applications include claims of substantial clinical improvement related to avoidance of patient risk, documented in part through analysis of FDA MAUDE data. The applicant stated its belief that this level of evidence appropriately supported the Uretero1TM application and also supports the substantial clinical improvement provided by VersaVueTM Single-Use Flexible Cystoscope and other disposable cystoscopes. The applicant asked that if CMS reaches a different conclusion, the agency provide a detailed rationale as to what requirements have changed since the rulemaking cycle for 2023.
Response: We appreciate the applicant's input. As an initial matter, we note that the requirements under § 419.66(c)(2) have not changed since we revised the alternative pathway provision effective January 1, 2021 (85 FR 86303), and CMS has consistently explained that its approach is to evaluate all evidence submitted for each device pass-through application as it applies to the nominated device (88 FR 81736, 89 FR 94182). In this case, we maintain our concerns listed in the CY 2026 OPPS/ASC proposed rule about whether the use of data that solely rely on MDRs compiled in the MAUDE database can be used to demonstrate that the use of single-use, disposable cystoscopes, like the VersaVueTM Single-Use Flexible Cystoscope, would result in substantial clinical improvement. Additionally, we continue to be concerned that the FDA News Release (2021, April 1) does not specifically reference single-use, disposable cystoscopes or encourage manufacturers to transition to single-use, disposable cystoscopes, such as the VersaVueTM Single-Use Flexible Cystoscope.
With respect to the concern that the use of MDRs compiled in the MAUDE database can be used to demonstrate substantial clinical improvement, we acknowledge that, since the Uretero1TM approval, we have more critically evaluated and questioned the utility of studies and reports using data solely collected from the MAUDE database, which claim associations between infections and devices for the purposes of demonstrating substantial clinical improvement. For example, in our decision for Ambu® aScopeTM Gastro (89 FR 94179 through 94182), we found that the applicant's self-sponsored analyses of FDA adverse event reports and the FDA MAUDE report did not provide evidence on the prevalence of infection, establish a clear relationship between infection risk and reprocessing procedures, or substantiate that single-use disposable scopes, or the nominated device specifically, would be a substantial clinical improvement over currently-available devices. As we discussed in the CY 2026 OPPS/ASC proposed rule, the FDA MAUDE database's MDR data are not intended to be used to evaluate rates of adverse events, evaluate a change in event rates over time, or compare adverse event occurrence rates across devices. Further, FDA describes the MAUDE database as a passive surveillance system, and that incidence, prevalence, or cause of an event cannot be determined from this surveillance system alone due to underreporting of events, inaccuracies, or bias in reports; untimely reports; lack of verification that the device caused the reported event; and lack of information about frequency of device use. Additionally, MDRs submitted to MAUDE have not been independently reviewed and the reported adverse events, by themselves, are not definitive evidence of faulty or defective medical devices. Finally, we note that the MAUDE database lacks the clinical granularity, such as comorbidities, procedure details, or the etiology of an adverse event, needed to calculate the true incidence of any post-cystoscopy complication. These same limitations of the MAUDE database are also acknowledged in the Lee et al. (2022) and Muscarella (2022) articles, which the applicant submitted as part of the VersaVueTM Single-Use Flexible Cystoscope application. For these reasons, we maintain our concerns that data from MAUDE alone cannot substantiate that the VersaVueTM Single-Use Flexible Cystoscope exhibits substantial clinical improvement over currently available devices.
With respect to the MAUDE data included as evidence in the Uretero1TM application, we note that, in addition to 892 MDRs, the Ofstead et al. (2022) study also included five articles describing reprocessing breaches, contamination, infections, or injuries related to fully reprocessed flexible ureteroscopes, as well as conference abstracts. We note that the inclusion of Ofstead et al. (2022), which contained additional studies and data sources, provided evidence other than MAUDE data for the Uretero1TM application. In contrast to Ofstead et al. (2022), the Lee et al. (2022) and Muscarella (2022) articles included in the VersaVueTM Single-Use Flexible Cystoscope application rely solely on MAUDE data and do not evaluate other data or sources. Moreover, the Muscarella (2022) article is not peer-reviewed and is self-published. For these reasons, we maintain our concerns that the use of studies that solely rely on MDRs compiled in the MAUDE database cannot substantiate that the use of single-use, disposable cystoscopes, like VersaVueTM Single-Use Flexible Cystoscope, results in substantial clinical improvement over currently available devices.
Regarding the FDA News Release (April 1, 2021), we note that within the document, FDA does not make specific recommendations for the use of single-use, disposable cystoscopes, including the VersaVueTM Single-Use Flexible Cystoscope, over reusable, reprocessed devices. Rather, FDA stated that its Letter to Health Care Providers issued recommendations for reprocessing and using these devices, such as following the reprocessing instructions and developing schedules for routine device inspection and maintenance. Additionally, FDA stated that it believes the risk of infection from reusable, reprocessed urological endoscopes was low based on its data. We also note that the FDA Update (January 31, 2025) communicated a medical device recall of a specific device component, the Olympus® endoscope accessory (MAJ-891 Forceps/Irrigation Plug) that is attached to the instrument channel port of a certain endoscope, due to the risk of infection that may result from improper reprocessing; however, it made no mention of, or recommendation for, the use of single-use, disposable devices, including the VersaVueTM Single-Use Flexible Cystoscope, over reusable, reprocessed devices. For these reasons, we maintain our concern that the FDA News Release (April 1, 2021) does not specifically reference single-use, disposable devices, including the nominated device.
In response to the applicant's comment comparing the Uretero1TM application summary with the nominated device's application summary, we note that we expressed a similar concern for Uretero1TM , specifically that the FDA advisory letter regarding ureteroscopes did not mention single-use devices and that it was unclear how the news release's recommendations supported Uretero1TM 's claims of substantial clinical improvement (87 FR 71932). While we ultimately determined that evidence submitted as part of the Uretero1TM pass-through application was sufficient to demonstrate substantial clinical improvement at that ( printed page 53672) time, we would like to reiterate, again, that we evaluate all evidence submitted for each device pass-through application as it applies to the nominated device at the time the current application is submitted. As previously noted, we have gained additional knowledge about the limitations of what is reported and included in the MAUDE database.
Based on the totality of the evidence, we do not believe that the applicant has submitted documentation demonstrating that the use of the VersaVueTM Single-Use Flexible Cystoscope results in a substantial clinical improvement compared to other existing technologies. Further, we disagree with the assertion that our acceptance of FDA guidance documents for previous applications controls the outcome of our evaluation of evidence of substantial clinical improvement for the VersaVueTM Single-Use Flexible Cystoscope. The ultimate determination of whether evidence demonstrates substantial clinical improvement for one application is not controlling on future determinations because, due to inherent differences in the devices themselves and the supporting documentation submitted, CMS may have different concerns as they relate to the nominated device. We do not believe our evaluation of the totality of the evidence provided by the applicant, including the FDA data and News Release, represents a change in requirements for evaluating transitional pass-through applications.
Comment: In response to our concern that the utilization data submitted as part of the VersaVueTM Single-Use Flexible Cystoscope application suggests that disposable cystoscopes are widely available and consistently used in outpatient facilities, the applicant commented that neither the statute nor the regulations describe a standard related to whether a device is widely available or consistently used. The applicant stated its belief that CMS has never applied such a standard in the past and should not apply such a requirement to an individual application. The applicant asserted that, while it opposes such an approach, if CMS intends to limit pass-through categories for devices it determines are widely available, then CMS must go through rulemaking to establish that standard.
Further, the applicant stated that it understood the utilization data CMS collects is requested to calculate the estimated total utilization across pass-through items in a given year. Finally, the applicant clarified that some of the marketing data provided on its application was inaccurate and that the Chew et al. (2025) study indicates that, based on review of outpatient hospital claims data, 2.3 percent of encounters involved single-use cystoscopes, rather than the estimated 23 percent initially provided.
Response: We appreciate the applicant's input. The applicant is correct that volume and utilization data are used to calculate the estimated cost of pass-through items. However, we may use any information submitted as part of a device pass-through application for the purposes of evaluating that application, if such information is relevant to other eligibility criteria. In addition, we continue to believe that our approach, including the question raised regarding the creation of a device pass-through payment category code for certain categories of devices, is consistent with both the statutory intent of section 1833(t)(6)(B) of the Act and with the implementing regulation at § 419.66. As explained in the CY 2002 OPPS interim final rule with comment period (66 FR 55852 and 55853) and in other final and proposed rules (see, for example, the CY 2023 final rule with comment period, 87 FR 71886) thereafter, the transitional pass-through payment provision is intended as an interim measure to facilitate beneficiary access by allowing for adequate payment of new, innovative technology while we collect the necessary data to incorporate the costs for these items into the base APC rates. The statute and regulations specifically limit the payment for individual pass-through items to at least 2 years but no more than 3 years, with the intention that the costs for these items should be incorporated into the APC rates for the procedures associated with these items after that period. We believe that in if CMS has already collected cost data in the appropriate APC for devices that would be described by a potential device category code, then establishing such a device category code would be unnecessary and contrary to the intent of the statute. Further, CMS is not establishing a new standard as the applicant suggests; rather, the standard is set by the statute and regulation and our discussion of volume and utilization is part of our review of the evidence to determine compliance with the requirements of § 419.66(c)(2), which we believe is consistent with both the language and intent of transitional pass-through payment provisions. We continue to believe that, consistent with the statute and regulations, transitional pass-through payment should be limited to those new and innovative technologies for which the necessary data to appropriately incorporate adequate cost data of these technologies into the applicable procedure APC have not been collected.
Because of the reasons discussed in this section, the VersaVueTM Single-Use Flexible Cystoscope does not meet the substantial clinical improvement criterion to be considered as a device for transitional pass-through payment. Therefore, in this final rule with comment period, we will not address whether the technology meets the cost criterion required for transitional pass-through payment for devices. We are not approving the VersaVueTM Single-Use Flexible Cystoscope for transitional pass-through payment status for CY 2026 because the technology does not meet the substantial clinical improvement criterion at § 419.66(c)(2).
(d) Other Comments
Comment: Multiple commenters recommended CMS calculate the device-related portion of APCs for purposes of determining transitional pass-through payment status eligibility and the device offset using only the cost of the devices replaced by the proposed device pass-through payment status device category. Specifically, the commenters recommended CMS eliminate the evaluation of device pass-through cost significance criteria when a proposed device does not replace any existing devices used in a procedure. For devices that replace some, but not all, of the devices used in a procedure, the commenters requested CMS revise the methodology of device pass-through cost significance criteria to assess the cost of proposed device using only the cost of the devices the proposed device replaces in the associated procedure. For approved pass-through devices, the commenters recommended CMS identify and apply an offset amount that reflects only the costs of the replaced devices for devices that replace only a portion of device-related costs within a procedure. The commenters asserted that the refinement of the device offset calculation would ensure a more accurate and fair evaluation of device costs and reduce unnecessary barriers for a procedure to perform safely and successfully. In addition, one commenter proposed that CMS consider a new policy to set the device offset at the lesser of the APC offset or the HCPCS-level offset for purposes of calculating transitional pass-through payments when insufficient claims data are available to demonstrate that the costs of a pass-through device are meaningfully reflected in the associated APC payment for the procedure. The commenter opined that this approach would prevent excessive and ( printed page 53673) unwarranted device costs from being deducted from hospital payments.
Response: We appreciate the commenters' recommendations regarding the revision of the methodology for calculating the device-related portion of APCs for the purpose of determining the transitional pass-through payment status eligibility and the device offset of a nominated device. We will continue to consider the issues and any additional public comments related to them.
Comment: We received multiple comments supporting the OPPS transitional device pass-through payment pathway and highlighting the important role the program plays in the treatment of Medicare beneficiaries. One commenter noted that the program bridges the gap between FDA approval of novel devices and their full integration into OPPS rates and encourages manufacturers to invest in innovation without delaying patient access.
Response: We appreciate the commenters' input and ongoing support of the device pass-through payment program.
Comment: We received several public comments requesting changes to the device pass-through payment policies, such as, but not limited to: modifying or removing the requirement that a device be surgically inserted or implanted. We also received multiple public comments requesting updates to the device offset amounts for existing device category codes. In addition, we received comments on devices that were not under consideration for device pass-through payment status for CY 2026.
Response: These comments were outside the scope of the proposals included in the CY 2026 OPPS/ASC proposed rule and we are therefore not addressing them in this final rule with comment period.
B. Device-Intensive Procedures
1. Background
Under the OPPS, prior to CY 2017, device-intensive status for procedures was determined at the APC level for APCs with a device offset percentage greater than 40 percent (79 FR 66795). Beginning in CY 2017, CMS began determining device-intensive status at the HCPCS code level. In assigning device-intensive status to an APC prior to CY 2017, the device costs of all the procedures within the APC were calculated and the geometric mean device offset of all of the procedures had to exceed 40 percent. Almost all of the procedures assigned to device-intensive APCs utilized devices, and the device costs for the associated HCPCS codes exceeded the 40-percent threshold. The no cost/full credit and partial credit device policy (79 FR 66872 through 66873) applies to device-intensive procedures and is discussed in detail in section IV.B.4. of the CY 2026 OPPS/ASC proposed rule. A related device policy was the requirement that certain procedures assigned to device-intensive APCs require the reporting of a device code on the claim (80 FR 70422) and is discussed in detail in section IV.B.3. of the CY 2026 OPPS/ASC proposed rule. For further background information on the device-intensive APC policy, we refer readers to the CY 2016 OPPS/ASC final rule with comment period (80 FR 70421 through 70426).
a. HCPCS Code-Level Device-Intensive Determination
As stated earlier, prior to CY 2017, under the device-intensive methodology we assigned device-intensive status to all procedures requiring the implantation of a device that were assigned to an APC with a device offset greater than 40 percent and, beginning in CY 2015, that met the three criteria as listed. Historically, the device-intensive designation was at the APC level and applied to the applicable procedures within that APC. In the CY 2017 OPPS/ASC final rule with comment period (81 FR 79658), we changed our methodology to assign device-intensive status at the individual HCPCS code level rather than at the APC level. Under this policy, a procedure could be assigned device-intensive status regardless of its APC assignment, and device-intensive APC designations were no longer applied under the OPPS or the ASC payment system.
We believe that a HCPCS code-level device offset is, in most cases, a better representation of a procedure's device cost than an APC-wide average device offset based on the average device offset of all of the procedures assigned to an APC. Unlike a device offset calculated at the APC level, which is a weighted average offset for all devices used in all of the procedures assigned to an APC, a HCPCS code-level device offset is calculated using only claims for a single HCPCS code. We believe that this methodological change results in a more accurate representation of the cost attributable to implantation of a high-cost device, which ensures consistent device-intensive designation of procedures with a significant device cost. Further, we believe a HCPCS code-level device offset removes inappropriate device-intensive status for procedures without a significant device cost that are granted such status because of their APC assignment.
Under our existing policy, procedures that meet the criteria listed in section IV.C.1.b. of this final rule with comment period are identified as device-intensive procedures and are subject to all the policies applicable to procedures assigned device-intensive status under our established methodology, including our policies on device edits and no cost/full credit and partial credit devices discussed in sections IV.C.3. and IV.C.4. of this final rule with comment period.
b. Use of the Three Criteria To Designate Device-Intensive Procedures
We clarified our established policy in the CY 2018 OPPS/ASC final rule with comment period (82 FR 52474), where we explained that device-intensive procedures require the implantation of a device and additionally are subject to the following criteria:
- All procedures must involve implantable devices that would be reported if device insertion procedures were performed.
- The required devices must be surgically inserted or implanted devices that remain in the patient's body after the conclusion of the procedure (at least temporarily); and
- The device offset amount must be significant, which is defined as exceeding 40 percent of the procedure's mean cost.
We changed our policy to apply these three criteria to determine whether procedures qualify as device-intensive in the CY 2015 OPPS/ASC final rule with comment period (79 FR 66926), where we stated that we would apply the no cost/full credit and partial credit device policy—which includes the three criteria listed previously—to all device-intensive procedures beginning in CY 2015. We reiterated this position in the CY 2016 OPPS/ASC final rule with comment period (80 FR 70424), where we explained that we were finalizing our proposal to continue using the three criteria established in the CY 2007 OPPS/ASC final rule with comment period for determining the APCs to which the CY 2016 device intensive policy will apply. Under the policies we adopted in CYs 2015, 2016, and 2017, all procedures that require the implantation of a device and meet the previously described criteria are assigned device-intensive status, regardless of their APC placement.
2. Device-Intensive Procedure Policy
As part of our effort to better capture costs for procedures with significant device costs, in the CY 2019 OPPS/ASC final rule with comment period (83 FR ( printed page 53674) 58944 through 58948), for CY 2019, we modified our criteria for device-intensive procedures. We had heard from interested parties that the criteria excluded some procedures that interested parties believed should qualify as device-intensive procedures. Specifically, we were persuaded by interested party arguments that procedures requiring expensive surgically inserted or implanted devices that are not capital equipment should qualify as device-intensive procedures, regardless of whether the device remains in the patient's body after the conclusion of the procedure. We agreed that a broader definition of device-intensive procedures was warranted, and made two modifications to the criteria for CY 2019 (83 FR 58948). First, we allowed procedures that involve surgically inserted or implanted single-use devices that meet the device offset percentage threshold to qualify as device-intensive procedures, regardless of whether the device remains in the patient's body after the conclusion of the procedure. We established this policy because we no longer believe that whether a device remains in the patient's body should affect a procedure's designation as a device-intensive procedure, as such devices could, nonetheless, comprise a large portion of the cost of the applicable procedure. Second, we modified our criteria to lower the device offset percentage threshold from 40 percent to 30 percent, to allow a greater number of procedures to qualify as device intensive. We stated that we believed allowing these additional procedures to qualify for device-intensive status would help ensure these procedures receive more appropriate payment in the ASC setting, which would help encourage the provision of these services in the ASC setting. In addition, we stated that this change would help to ensure that more procedures containing relatively high-cost devices are subject to the device edits, which leads to more correctly coded claims and greater accuracy in our claims data. Specifically, for CY 2019 and subsequent years, we finalized that device-intensive procedures will be subject to the following criteria:
- All procedures must involve implantable devices assigned a CPT or HCPCS code;
- The required devices (including single-use devices) must be surgically inserted or implanted; and
- The device offset amount must be significant, which is defined as exceeding 30 percent of the procedure's mean cost (83 FR 58945).
In addition, to further align the device-intensive policy with the criteria used for device pass-through payment status, we finalized, for CY 2019 and subsequent years, that for purposes of satisfying the device-intensive criteria, a device-intensive procedure must involve a device that:
- Has received FDA marketing authorization, has received an FDA investigational device exemption (IDE), and has been classified as a Category B device by FDA in accordance with §§ 405.203 through 405.207 and 405.211 through 405.215, or meets another appropriate FDA exemption from premarket review;
- Is an integral part of the service furnished;
- Is used for one patient only;
- Comes in contact with human tissue;
- Is surgically implanted or inserted (either permanently or temporarily); and
- Is not either of the following:
++ Equipment, an instrument, apparatus, implement, or item of the type for which depreciation and financing expenses are recovered as depreciable assets as defined in Chapter 1 of the Medicare Provider Reimbursement Manual (CMS Pub. 15-1); or
++ A material or supply furnished incident to a service (for example, a suture, customized surgical kit, scalpel, or clip, other than a radiological site marker) (83 FR 58945).
In addition, for new HCPCS codes describing procedures requiring the implantation of devices that do not yet have associated claims data, in the CY 2017 OPPS/ASC final rule with comment period (81 FR 79658), we finalized a policy for CY 2017 to apply device-intensive status with a default device offset set at 41 percent for new HCPCS codes describing procedures requiring the implantation or insertion of a device that did not yet have associated claims data until claims data are available to establish the HCPCS code-level device offset for the procedures. This default device offset amount of 41 percent was not calculated from claims data; instead, it was applied as a default until claims data were available upon which to calculate an actual device offset for the new code. The purpose of applying the 41-percent default device offset to new codes that describe procedures that implant or insert devices was to ensure ASC access for new procedures until claims data become available.
As discussed in the CY 2019 OPPS/ASC proposed rule and final rule with comment period (83 FR 37108 through 37109 and 83 FR 58945 through 58946, respectively), in accordance with our policy stated previously to lower the device offset percentage threshold for procedures to qualify as device-intensive from greater than 40 percent to greater than 30 percent, for CY 2019 and subsequent years, we modified this policy to apply a 31-percent default device offset to new HCPCS codes describing procedures requiring the implantation of a device that do not yet have associated claims data until claims data are available to establish the HCPCS code-level device offset for the procedures. In conjunction with the policy to lower the default device offset from 41 percent to 31 percent, we continued our current policy of, in certain rare instances (for example, in the case of a very expensive implantable device), temporarily assigning a higher offset percentage if warranted by additional information such as pricing data from a device manufacturer (81 FR 79658). Once claims data are available for a new procedure requiring the implantation or insertion of a device, device-intensive status is applied to the code if the HCPCS code-level device offset is greater than 30 percent, according to our policy of determining device-intensive status by calculating the HCPCS code-level device offset.
In addition, in the CY 2019 OPPS/ASC final rule with comment period, we clarified that since the adoption of our policy in effect as of CY 2018, the associated claims data used for purposes of determining whether or not to apply the default device offset are the associated claims data for either the new HCPCS code or any predecessor code, as described by CPT coding guidance, for the new HCPCS code. Additionally, for CY 2019 and subsequent years, in limited instances where a new HCPCS code does not have a predecessor code as defined by CPT, but describes a procedure that was previously described by an existing code, we use clinical discretion to identify HCPCS codes that are clinically related or similar to the new HCPCS code but are not officially recognized as a predecessor code by CPT, and to use the claims data of the clinically related or similar code(s) for purposes of determining whether or not to apply the default device offset to the new HCPCS code (83 FR 58946). Clinically related and similar procedures for purposes of this policy are procedures that have few or no clinical differences and use the same devices as the new HCPCS code. In addition, clinically related and similar codes for purposes of this policy are codes that either currently or previously describe the procedure described by the ( printed page 53675) new HCPCS code. Under this policy, claims data from clinically related and similar codes are included as associated claims data for a new code, and where an existing HCPCS code is found to be clinically related or similar to a new HCPCS code, we apply the device offset percentage derived from the existing clinically related or similar HCPCS code's claims data to the new HCPCS code for determining the device offset percentage. We stated that we believe that claims data for HCPCS codes describing procedures that have minor differences from the procedures described by new HCPCS codes will provide an accurate depiction of the cost relationship between the procedure and the device(s) that are used, and will be appropriate to use to set a new code's device offset percentage, in the same way that predecessor codes are used. If a new HCPCS code has multiple predecessor codes, the claims data for the predecessor code that has the highest individual HCPCS-level device offset percentage is used to determine whether the new HCPCS code qualifies for device-intensive status. Similarly, in the event that a new HCPCS code does not have a predecessor code but has multiple clinically related or similar codes, the claims data for the clinically related or similar code that has the highest individual HCPCS level device offset percentage is used to determine whether the new HCPCS code qualifies for device-intensive status.
In the CY 2025 OPPS/ASC final rule with comment period (89 FR 94214 through 92419), we finalized a change to our methodology for applying default device offset percentages for new device-intensive procedures. Under our previous policy, if a new CPT/HCPCS code did not have available claims data, either from the new HCPCS code or any predecessor code or clinically-similar code that uses the same device, and the CPT/HCPCS code otherwise met our criteria for device-intensive status, we would apply a default device offset percentage of 31 percent. However, we were aware of certain situations where the default device offset amount might not adequately reflect the existing device portion of the procedure's costs when compared to the cost of similar devices. A potential large difference between the default device offset amount and the device portion of similar devices might impede our ability to accurately remove device offset amounts from new device-intensive procedures under the OPPS and to set payment rates for device-intensive procedures under the ASC payment system. Therefore, for CY 2025 and subsequent CYs, we finalized our proposal to modify our default device offset percentage policy for new device-intensive procedures. Specifically, for new CPT/HCPCS codes that both describe a procedure that requires the surgical implantation or insertion of a single-use device that exceeds 30 percent of the procedure's cost and that meets our requirements of a device as described here and lack claims data (from either the new HCPCS code or any predecessor code or clinically-similar code that uses the same device), we would apply a default device offset percentage that is the greater of 31 percent or the device offset percentage of the APC to which the procedure has been assigned. We stated that we still believe that a HCPCS code-level device offset is, in most cases, a more accurate representation of a procedure's device cost than an APC-wide average device offset based on the average device offset of all the procedures assigned to an APC. However, because newer device-intensive procedures lack claims data, we believe the APC-wide average device offset percentage is, in many cases, a better reflection of the estimated device costs of the procedure than a default 31 percent offset. Additionally, there can be instances where the typical device costs of procedures in an APC can be significantly greater than the 31 percent default device offset. For these reasons, we finalized our modification to our default device offset percentage for new device-intensive procedures. This methodological change was finalized for both the OPPS and ASC Payment System for CY 2025 and subsequent CYs and applies to new procedures assigned to clinical APCs, but not to new procedures assigned to New Technology APCs.
Additionally, in the CY 2025 OPPS/ASC final rule with comment period (89 FR 92414 through 92419), we stated that we were persuaded by commenters that the lack of a device edit for device-intensive procedures, particularly new technologies, might lead to an underreporting of device costs and total procedure costs and potentially impede beneficiary access to such new technologies over time. Therefore, in addition to finalizing a modification to our device edits policy for CY 2025, we finalized a modification to our device offset percentage calculation. For procedures subject to our modified device edits policy for CY 2025 that cannot report modifier “CG” to bypass this claims processing edit, the device offset percentages calculated (for the CPT/HCPCS code or its predecessor code) are based on hospital claims that reported a device code. We stated that we believed that hospital outpatient claims that report a device code with such procedures provide, in general, a more accurate representation of the procedures' total costs. We also finalized, for purposes of determining device offset percentages, that we will not use claims data from procedures that had a status indicator of “E1” during the calendar year we are using for ratesetting and determining device offset percentages. Lastly, we refined our process for applying device offset percentages to use available claims data from predecessor codes annually, rather than the first year of the successor code's activation date, until we have available claims data from the successor code.
In the CY 2026 OPPS/ASC proposed rule (90 FR 33609), we proposed to continue these policies for CY 2026. As we indicated in the CY 2019 OPPS/ASC proposed rule and final rule with comment period, additional information for our consideration of an offset percentage higher than the default of 31 percent (or the APC-wide default offset percentage) for new HCPCS codes describing procedures requiring the implantation (or, in some cases, the insertion) of a device that do not yet have associated claims data, such as pricing data or invoices from a device manufacturer, should be directed to the Division of Outpatient Care, Mail Stop C4-01-26, Centers for Medicare & Medicaid Services, 7500 Security Boulevard, Baltimore, MD 21244-1850, or electronically outpatientpps@cms.hhs.gov. We stated in the CY 2026 OPPS/ASC proposed rule that additional information can be submitted prior to issuance of an OPPS/ASC proposed rule or as a public comment in response to an issued OPPS/ASC proposed rule. Device offset percentages will be set in each year's final rule.
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: Commenters supported our assignment of device-intensive status and the proposed device offset percentages for the following CPT/HCPCS codes:
- CPT code 0671T (Insertion of anterior segment aqueous drainage device into the trabecular meshwork, without external reservoir, and without concomitant cataract removal, one or more);
- CPT code 0970T (Ablation, benign breast tumor (e.g., fibroadenoma), percutaneous, laser, including imaging guidance when performed, each tumor); ( printed page 53676)
- CPT code 0971T (Ablation, malignant breast tumor(s), percutaneous, laser, including imaging guidance when performed, unilateral);
- CPT code 52284 (Cystourethroscopy, with mechanical urethral dilation and urethral therapeutic drug delivery by drug-coated balloon catheter for urethral stricture or stenosis, male, including fluoroscopy, when performed);
- CPT code 52443 (placeholder code 52XX2) (Cystourethroscopy with initial transurethral anterior prostate commissurotomy with a nondrug-coated balloon catheter followed by therapeutic drug delivery into the prostate by a drug-coated balloon catheter, including transrectal ultrasound and fluoroscopy, when performed);
- CPT codes 66989 (Extracapsular cataract removal with insertion of intraocular lens prosthesis (1-stage procedure), manual or mechanical technique (e.g., irrigation and aspiration or phacoemulsification), complex, requiring devices or techniques not generally used in routine cataract surgery ( e.g., iris expansion device, suture support for intraocular lens, or primary posterior capsulorrhexis) or performed on patients in the amblyogenic developmental stage; with insertion of intraocular ( e.g., trabecular meshwork, supraciliary, suprachoroidal) anterior segment aqueous drainage device, without extraocular reservoir, internal approach, one or more),
- CPT code 66991 (Extracapsular cataract removal with insertion of intraocular lens prosthesis (1 stage procedure), manual or mechanical technique (e.g., irrigation and aspiration or phacoemulsification); with insertion of intraocular ( e.g., trabecular meshwork, supraciliary, suprachoroidal) anterior segment aqueous drainage device, without extraocular reservoir, internal approach, one or more), and
- HCPCS code C9781 (Arthroscopy, shoulder, surgical; with implantation of subacromial spacer (e.g., balloon), includes debridement ( e.g., limited or extensive), subacromial decompression, acromioplasty, and biceps tenodesis when performed);
Response: We appreciate the commenters' support for our proposed device-intensive assignments and proposed device offset percentages based on CY 2024 claims data available for the CY 2026 OPPS/ASC proposed rule. Final device-intensive assignments are based on CY 2024 claims data available found in Addendum P of this final rule with comment period.
Comment: One commenter recommended a nomination process for device-intensive procedures.
Response: We thank the commenter for their recommendation. We believe it is important for our current methodology to rely on cost information from claims data for determining device-intensive status for CPT/HCPCS codes. Additionally, we believe our current methodology of determining device-intensive status and device offset percentages in the absence of claims data and seeking public comments on our determinations in our notice and comment rulemaking remains an appropriate methodology that relies on engagement with healthcare interested parties. Therefore, we do not see a need to create a nomination process for device-intensive procedures at this time.
Comment: One commenter requested that CMS update Addendum P for the CY 2026 OPPS/ASC final rule to include the default device offset of 31 percent for CPT codes 0956T (Partial craniectomy, channel creation, and tunneling of electrode for sub-scalp implantation of an electrode array, receiver, and telemetry unit for continuous bilateral electroencephalography monitoring system, including imaging guidance) and 0960T (Replacement of sub-scalp implanted electrode array, receiver, and telemetry unit with tunneling of electrode for continuous bilateral electroencephalography monitoring system, including imaging guidance) as these are device-intensive procedures requiring sophisticated implantable devices with external wearable components and cloud services.
Response: We thank the commenter and note that we inadvertently omitted CPT codes 0956T and 0960T from Addendum P in the CY 2026 OPPS/ASC proposed rule, although we did propose in the CY 2026 OPPS/ASC proposed rule that such procedures were device-intensive in the ASC setting in Addenda AA and FF. We are therefore accepting the commenter's recommendation and assigning device-intensive status to CPT codes 0956T and 0960T with a device offset percentage of 31 percent in this CY 2026 OPPS/ASC final rule with comment period.
Comment: Some commenters recommended that we modify our definition of device-intensive procedures to include non-insertable or implantable devices. The CPT/HCPCS codes commenters recommended assigning device-intensive status include:
- CPT code 0686T (Histotripsy (i.e., non-thermal ablation via acoustic energy delivery) of malignant hepatocellular tissue, including image guidance);
- CPT code 0888T (Histotripsy (i.e., non-thermal ablation via acoustic energy delivery) of malignant renal tissue, including imaging guidance);
- CPT code 15013 (Preparation of skin cell suspension autograft, requiring enzymatic processing, manual mechanical disaggregation of skin cells, and filtration; first 25 sq cm or less of harvested skin); and
- HCPCS code C8002 (Preparation of skin cell suspension autograft, automated, including all enzymatic processing and device components (do not report with manual suspension preparation)).
The commenters asserted that failure to assign device-intensive status to these procedures will result in ASC payment rates that do not accurately reflect the resource costs of furnishing these services, potentially limiting beneficiary access to innovative skin cell suspension autograft procedures and certain histotripsy procedures in the ASC setting.
Response: We appreciate the commenters' recommendations. We do not believe we should expand our definition of devices to include the costs of skin substitute products or capital-intensive equipment, as our current ratesetting methodology adequately captures the costs for such items and services for developing OPPS/ASC payment rates. Therefore, we are not accepting the commenter's recommendation.
Comment: One commenter disagreed with our proposed device offset percentage for CPT code 0202T (Posterior vertebral joint(s) arthroplasty ( e.g., facet joint[s] replacement), including facetectomy, laminectomy, foraminotomy, and vertebral column fixation, injection of bone cement, when performed, including fluoroscopy, single level, lumbar spine) at 59.21 percent. The commenter was unclear how we determined this figure, and stated it is unclear since this procedure was previously on the IPO list and the APC-wide device offset percentage is 59.63 percent.
Response: While CPT code 0202T was on the inpatient-only list for CY 2024, we did receive one claim for CY 2026 OPPS/ASC ratesetting from CY 2024. Based on this CY 2024 claim available for this final rule with comment period, we are not relying on our default device offset methodology and assigning device-intensive status to CPT code 0202T using the CY 2024 claim data available. The final device offset percentage for CPT code 0202T under the OPPS can be found in Addendum P to this final rule with comment period. ( printed page 53677)
Comment: Two commenters requested that CMS assign device-intensive status to HCPCS code C9779 (Endoscopic submucosal dissection (esd), including endoscopy or colonoscopy, mucosal closure, when performed). The commenters noted that endoscopic submucosal dissection requires specialized, expensive single-use devices and equipment that represent a significant portion of the total procedure cost, warranting device-intensive recognition to ensure appropriate reimbursement. Both commenters asserted that without proper device-intensive status, the payment may be insufficient to cover the actual costs of performing this complex procedure, potentially limiting patient access to this advanced endoscopic technique in both hospital outpatient and ASC settings.
Response: For this final rule with comment period, the device offset percentage for HCPCS code C9779 is below our device-intensive threshold based on 963 claims for this service in the CY 2024 data and, therefore, does not qualify for device-intensive for CY 2026.
Comment: Some commenters recommended that we establish the device offset percentage for CPT code 47384 (placeholder code 4001X) (Ablation, irreversible electroporation, liver, one or more tumors, including imaging guidance, percutaneous) and CPT code 55877 (placeholder code 5XX11) (Ablation, irreversible electroporation, prostate, one or more tumors, including imaging guidance, percutaneous) using CPT code 0600T (Ablation, irreversible electroporation; one or more tumors per organ, other than liver or prostate, including imaging guidance, when performed, percutaneous). The commenters argue that CPT code 0600T was the predecessor code to CPT codes 47384 and 55877 and therefore under our policy for establishing device offset percentages for new device-intensive procedures, we should rely on claims data from CPT code 0600T to establish the device offset percentages for the new procedures.
Response: We agree with the commenters' recommendation. We inadvertently did not use claims data from predecessor CPT code 0600T for establishing the proposed device offset percentages for CPT codes 47384 and 55877 for the CY 2026 OPPS/ASC proposed rule. We are accepting the commenters' recommendations and establishing the device offset percentage for CPT codes 47384 and 55877 using the claims data from CPT code 0600T for CY 2026.
Comment: Multiple commenters requested that CMS classify CPT code 52282 (Cystourethroscopy, with insertion of permanent urethral stent) as device-intensive. The commenters believed that CMS's claims data analysis is fundamentally flawed because no permanent urethral stents have been available in the U.S. market since April 2016, making any recent claims data implausible and not reflective of actual procedural activity, with analysis showing that over 33 percent of the 111 claims used were inappropriately billed to female patients despite the procedure being indicated only for males. The commenters urged CMS to exclude all existing hospital outpatient claims for CPT 52282 from payment calculations, designate the procedure as device-intensive using manufacturer pricing as a device offset, and ensure adequate reimbursement to support patient access to upcoming clinically proven permanent urethral stent technologies for BPH treatment. Separately, a commenter requested that CMS consider granting device-intensive status for CPT code 62287 (Decompression procedure, percutaneous, of nucleus pulposus of intervertebral disc, any method utilizing needle based technique to remove disc material under fluoroscopic imaging or other form of indirect visualization, with discography and/or epidural injection(s) at the treated level(s), when performed, single or multiple levels, lumbar) due to a lack of consistent reporting of device costs associated with this procedure. The commenter stated that if the invoice cost of devices used in the procedure were compared to the reimbursement rate, the device-related portion of the reimbursement would exceed the 30 percent threshold required for device-intensive status.
Response: We appreciate the concerns the commenters have raised but note that hospitals have reported these CPT codes across a significant number of claims in total over the past several years and the device offset percentages of such claims has not exceeded our device-intensive threshold of 30 percent in each of the past several years. We believe it would be inappropriate to disregard the entirety of such claims data for determining device-intensive status as we rely on hospitals to accurately report device costs for OPPS/ASC ratesetting. Therefore, we are not accepting the commenters' recommendation to assign device-intensive status to CPT codes 52282 and 62287 for CY 2026.
Comment: Commenters requested that CMS retain the predecessor device offset percentages for the newly restructured lower extremity revascularization CPT codes (placeholder codes 37X02-37X46) rather than adopting the APC device offset percentage, arguing that this approach aligns with CMS's standard policy of maintaining historical device offset data when codes are revised or bifurcated.
Response: We agree with the commenters that the newly-restructured lower extremity revascularization CPT codes have a suitable predecessor code for which we can use claims data for determining device offset percentages. Therefore, for such procedures that are separately payable, we are accepting the commenters' recommendation and will assign a device offset percentage to the new lower extremity revascularization CPT codes based on claims data from the predecessor code. Specifically, for CY 2026, we rely on the mapping that follows:
- Claims data from CPT code 37220 (Revascularization, endovascular, open or percutaneous, iliac artery, unilateral, initial vessel; with transluminal angioplasty) for determining the device offset percentage for CPT codes 37254 (Revascularization, endovascular, open or percutaneous, iliac vascular territory, with transluminal angioplasty, including all maneuvers necessary for accessing and selectively catheterizing the artery and crossing the lesion, including all imaging guidance and radiological supervision and interpretation necessary to perform the angioplasty within the same artery, unilateral; straightforward lesion, initial vessel) and 37256 (Revascularization, endovascular, open or percutaneous, iliac vascular territory, with transluminal angioplasty, including all maneuvers necessary for accessing and selectively catheterizing the artery and crossing the lesion, including all imaging guidance and radiological supervision and interpretation necessary to perform the angioplasty within the same artery, unilateral; complex lesion, initial vessel) (placeholder codes 37XX1 and 37X03);
- Claims data from CPT code 37221 (Revascularization, endovascular, open or percutaneous, iliac artery, unilateral, initial vessel; with transluminal stent placement(s), includes angioplasty within the same vessel, when performed) for determining the device offset percentage for CPT codes 37258 (Revascularization, endovascular, open or percutaneous, iliac vascular territory, with transluminal stent placement, including transluminal angioplasty when performed, including all maneuvers necessary for accessing and selectively catheterizing the artery and crossing the lesion, including all imaging guidance and radiological ( printed page 53678) supervision and interpretation necessary to perform the stent placement and angioplasty when performed, within the same artery, unilateral; straightforward lesion, initial vessel) and 37260 (Revascularization, endovascular, open or percutaneous, iliac vascular territory, with transluminal stent placement, including transluminal angioplasty when performed, including all maneuvers necessary for accessing and selectively catheterizing the artery and crossing the lesion, including all imaging guidance and radiological supervision and interpretation necessary to perform the stent placement and angioplasty when performed, within the same artery, unilateral; complex lesion, initial vessel) (placeholder codes 37X05 and 37X07);
- Claims data from CPT code 37224 (Revascularization, endovascular, open or percutaneous, femoral, popliteal artery(s), unilateral; with transluminal angioplasty) for determining the device offset percentage for CPT codes 37263 (Revascularization, endovascular, open or percutaneous, femoral and popliteal vascular territory, with transluminal angioplasty, including all maneuvers necessary for accessing and selectively catheterizing the artery and crossing the lesion, including all imaging guidance and radiological supervision and interpretation necessary to perform the angioplasty within the same artery, unilateral; straightforward lesion, initial vessel) and 37265 (Revascularization, endovascular, open or percutaneous, femoral and popliteal vascular territory, with transluminal angioplasty, including all maneuvers necessary for accessing and selectively catheterizing the artery and crossing the lesion, including all imaging guidance and radiological supervision and interpretation necessary to perform the angioplasty within the same artery, unilateral; complex lesion, initial vessel) (placeholder codes 37X10 and 37X12);
- Claims data from CPT code 37225 (Revascularization, endovascular, open or percutaneous, femoral, popliteal artery(s), unilateral; with atherectomy, includes angioplasty within the same vessel, when performed) for determining the device offset percentage for CPT codes 37271 (Revascularization, endovascular, open or percutaneous, femoral and popliteal vascular territory, with transluminal atherectomy, including transluminal angioplasty when performed, including all maneuvers necessary for accessing and selectively catheterizing the artery and crossing the lesion, including all imaging guidance and radiological supervision and interpretation necessary to perform the atherectomy and angioplasty when performed, within the same artery, unilateral; straightforward lesion, initial vessel) and 37273 (Revascularization, endovascular, open or percutaneous, femoral and popliteal vascular territory, with transluminal atherectomy, including transluminal angioplasty when performed, including all maneuvers necessary for accessing and selectively catheterizing the artery and crossing the lesion, including all imaging guidance and radiological supervision and interpretation necessary to perform the atherectomy and angioplasty when performed, within the same artery, unilateral; complex lesion, initial vessel) (placeholder codes 37X18 and 37X20);
- Claims data from CPT code 37226 (Revascularization, endovascular, open or percutaneous, femoral, popliteal artery(s), unilateral; with transluminal stent placement(s), includes angioplasty within the same vessel, when performed) for determining the device offset percentage for CPT codes 37267 (Revascularization, endovascular, open or percutaneous, femoral and popliteal vascular territory, with transluminal stent placement, including transluminal angioplasty when performed, including all maneuvers necessary for accessing and selectively catheterizing the artery and crossing the lesion, including all imaging guidance and radiological supervision and interpretation necessary to perform the stent placement and angioplasty when performed, within the same artery, unilateral; straightforward lesion, initial vessel) and 37269 (Revascularization, endovascular, open or percutaneous, femoral and popliteal vascular territory, with transluminal stent placement, including transluminal angioplasty when performed, including all maneuvers necessary for accessing and selectively catheterizing the artery and crossing the lesion, including all imaging guidance and radiological supervision and interpretation necessary to perform the stent placement and angioplasty when performed, within the same artery, unilateral; complex lesion, initial vessel) (placeholder codes 37X14 and 37X16);
- Claims data from CPT code 37227 (Revascularization, endovascular, open or percutaneous, femoral, popliteal artery(s), unilateral; with transluminal stent placement(s) and atherectomy, includes angioplasty within the same vessel, when performed) for determining the device offset percentage for CPT codes 37275 (Revascularization, endovascular, open or percutaneous, femoral and popliteal vascular territory, with transluminal stent placement, with transluminal atherectomy, including transluminal angioplasty when performed, including all maneuvers necessary for accessing and selectively catheterizing the artery and crossing the lesion, including all imaging guidance and radiological supervision and interpretation necessary to perform the stent placement, atherectomy, and angioplasty when performed, within the same artery, unilateral; straightforward lesion, initial vessel) and 37277 (Revascularization, endovascular, open or percutaneous, femoral and popliteal vascular territory, with transluminal stent placement, with transluminal atherectomy, including transluminal angioplasty when performed, including all maneuvers necessary for accessing and selectively catheterizing the artery and crossing the lesion, including all imaging guidance and radiological supervision and interpretation necessary to perform the stent placement, atherectomy, and angioplasty when performed, within the same artery, unilateral; complex lesion, initial vessel) (placeholder codes 37X22 and 37X24);
- Claims data from CPT code 37228 (Revascularization, endovascular, open or percutaneous, tibial, peroneal artery, unilateral, initial vessel; with transluminal angioplasty) for determining the device offset percentage for CPT codes 37280 (Revascularization, endovascular, open or percutaneous, tibial and peroneal vascular territory, with transluminal angioplasty, including all maneuvers necessary for accessing and selectively catheterizing the artery and crossing the lesion, including all imaging guidance and radiological supervision and interpretation necessary to perform the angioplasty within the same artery, unilateral; straightforward lesion, initial vessel), 37282 (Revascularization, endovascular, open or percutaneous, tibial and peroneal vascular territory, with transluminal angioplasty, including all maneuvers necessary for accessing and selectively catheterizing the artery and crossing the lesion, including all imaging guidance and radiological supervision and interpretation necessary to perform the angioplasty within the same artery, unilateral; complex lesion, initial vessel), 37296 (Revascularization, endovascular, open or percutaneous, inframalleolar vascular territory, with transluminal angioplasty, including all maneuvers necessary for accessing and selectively catheterizing the artery and ( printed page 53679) crossing the lesion, including all imaging guidance and radiological supervision and interpretation necessary to perform the angioplasty within the same artery, unilateral; straightforward lesion, initial vessel), 37298 (Revascularization, endovascular, open or percutaneous, inframalleolar vascular territory, with transluminal angioplasty, including all maneuvers necessary for accessing and selectively catheterizing the artery and crossing the lesion, including all imaging guidance and radiological supervision and interpretation necessary to perform the angioplasty within the same artery, unilateral; complex lesion, initial vessel) (placeholder codes 37X27, 37X29, 37X43, and 37X45);
- Claims data from CPT code 37229 (Revascularization, endovascular, open or percutaneous, tibial, peroneal artery, unilateral, initial vessel; with atherectomy, includes angioplasty within the same vessel, when performed) for determining the device offset percentage for CPT codes 37288 (Revascularization, endovascular, open or percutaneous, tibial and peroneal vascular territory, with transluminal atherectomy, including transluminal angioplasty when performed, including all maneuvers necessary for accessing and selectively catheterizing the artery and crossing the lesion, including all imaging guidance and radiological supervision and interpretation necessary to perform the atherectomy and angioplasty when performed, within the same artery, unilateral; straightforward lesion, initial vessel) and 37290 (Revascularization, endovascular, open or percutaneous, tibial and peroneal vascular territory, with transluminal atherectomy, including transluminal angioplasty when performed, including all maneuvers necessary for accessing and selectively catheterizing the artery and crossing the lesion, including all imaging guidance and radiological supervision and interpretation necessary to perform the atherectomy and angioplasty when performed, within the same artery, unilateral; complex lesion, initial vessel) (placeholder codes 37X35 and 37X37);
- Claims data from CPT code 37230 (Revascularization, endovascular, open or percutaneous, tibial, peroneal artery, unilateral, initial vessel; with transluminal stent placement(s), includes angioplasty within the same vessel, when performed) for determining the device offset percentage for CPT codes 37284 (Revascularization, endovascular, open or percutaneous, tibial and peroneal vascular territory, with transluminal stent placement, including transluminal angioplasty when performed, including all maneuvers necessary for accessing and selectively catheterizing the artery and crossing the lesion, including all imaging guidance and radiological supervision and interpretation necessary to perform the stent placement and angioplasty when performed, within the same artery, unilateral; straightforward lesion, initial vessel) and 37286 (Revascularization, endovascular, open or percutaneous, tibial and peroneal vascular territory, with transluminal stent placement, including transluminal angioplasty when performed, including all maneuvers necessary for accessing and selectively catheterizing the artery and crossing the lesion, including all imaging guidance and radiological supervision and interpretation necessary to perform the stent placement and angioplasty when performed, within the same artery, unilateral; complex lesion, initial vessel) (placeholder codes 37X31 and 37X33);
- Claims data from CPT code 37231 (Revascularization, endovascular, open or percutaneous, tibial, peroneal artery, unilateral, initial vessel; with transluminal stent placement(s) and atherectomy, includes angioplasty within the same vessel, when performed) for determining the device offset percentage for CPT codes 37292 (Revascularization, endovascular, open or percutaneous, tibial and peroneal vascular territory, with transluminal stent placement, with transluminal atherectomy, including transluminal angioplasty when performed, including all maneuvers necessary for accessing and selectively catheterizing the artery and crossing the lesion, including all imaging guidance and radiological supervision and interpretation necessary to perform the stent placement, atherectomy, and angioplasty when performed, within the same artery, unilateral; straightforward lesion, initial vessel) and 37294 (Revascularization, endovascular, open or percutaneous, tibial and peroneal vascular territory, with transluminal stent placement, with transluminal atherectomy, including transluminal angioplasty when performed, including all maneuvers necessary for accessing and selectively catheterizing the artery and crossing the lesion, including all imaging guidance and radiological supervision and interpretation necessary to perform the stent placement, atherectomy, and angioplasty when performed, within the same artery, unilateral; complex lesion, initial vessel) (placeholder codes 37X39 and 37X41);
After consideration of the public comments we received, we are finalizing our proposed continued use of HCPCS code-level device-intensive determination and three criteria to designate device-intensive procedures, in accordance with existing policies as discussed in the CY 2026 OPPS/ASC proposed rule section IV.B.1.a (90 FR 33607). We are also finalizing our proposed continuation of our device-intensive procedure policy, proposed use of CY 2024 claims information for determining device offset percentages and assigning device-intensive status, and our proposed default device offset policy for determining device offset percentages in the absence of claims data for device-intensive procedures, in accordance with existing policies as discussed in the CY 2026 OPPS/ASC proposed rule section IV.B.1.b (90 FR 33607).
Comments and our responses related to device-intensive procedure policy and the procedures we proposed for device-intensive status under the ASC payment system for CY 2026 can be found in section XIII.C.4. of this final rule with comment period. Our responses and final determinations related to device-intensive status for certain procedures under the ASC payment system are also applied to our final determinations of device-intensive status under the OPPS. The full listing of the final CY 2026 device-intensive procedures can be found in Addendum P to this final rule with comment period (which is available via the internet on the CMS website). Further, our claims accounting narrative contains a description of our device offset percentage calculation. Our claims accounting narrative for this final rule with comment period can be found under supporting documentation for the CY 2026 OPPS/ASC final rule with comment period on our website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient.
3. Device Edit Policy
In the CY 2015 OPPS/ASC final rule with comment period (79 FR 66795), we finalized a policy and implemented claims processing edits that require any of the device codes used in the previous device-to-procedure edits to be present on the claim whenever a procedure code assigned to any of the APCs listed in Table 5 of the CY 2015 OPPS/ASC final rule with comment period (the CY 2015 device-dependent APCs) was reported on the claim. In addition, in the CY 2016 OPPS/ASC final rule with ( printed page 53680) comment period (80 FR 70422), we modified our previously existing policy and applied the device coding requirements exclusively to procedures that require the implantation of a device assigned to a device-intensive APC. In the CY 2016 OPPS/ASC final rule with comment period, we also finalized our policy that the claims processing edits are such that any device code, when reported on a claim with a procedure assigned to a device-intensive APC (listed in Table 42 of the CY 2016 OPPS/ASC final rule with comment period (80 FR 70422)), will satisfy the edit.
In the CY 2017 OPPS/ASC final rule with comment period (81 FR 79658 through 79659), we changed our policy for CY 2017 and subsequent years to apply the CY 2016 device coding requirements to the newly defined device-intensive procedures. For CY 2017 and subsequent years, we also specified that any device code, when reported on a claim with a device-intensive procedure, will satisfy the edit. In addition, we created HCPCS code C1889 to recognize devices furnished during a device-intensive procedure that are not described by a specific Level II HCPCS Category C-code. Reporting HCPCS code C1889 with a device-intensive procedure will satisfy the edit requiring a device code to be reported on a claim with a device-intensive procedure. In the CY 2019 OPPS/ASC final rule with comment period, we revised the description of HCPCS code C1889 to remove the specific applicability to device-intensive procedures (83 FR 58950). For CY 2019 and subsequent years, the description of HCPCS code C1889 is “Implantable/insertable device, not otherwise classified.”
In the CY 2024 OPPS/ASC final rule with comment period (88 FR 81758 through 81759), we finalized our proposal to establish a procedure-to-device edit for the procedures assigned to APC 5496 (Level 6 Intraocular Procedures) and require hospitals to report the correct device HCPCS codes when reporting any of the four procedures—CPT codes 0308T and 0616T, 0617T, and 0618T. (We note that CPT codes 0617T and 0618T were deleted effective January 1, 2025 and CPT code 0616T was deleted effective January 1, 2025 and replaced with new CPT code 66683.) We have noted that interested parties have previously recommended in past rulemaking that we reestablish all our previous procedure-to-device edits, but we do not expect to extend this policy beyond the procedures assigned to APC 5496 (Level 6 Intraocular Procedures). This APC represents a unique situation—the APC (which was the Level 5 Intraocular APC in previous years) had been a Low Volume APC (fewer than 100 claims in a claims year) since we established our Low Volume APC policy, the procedures associated with this APC have significant procedure costs often greater than $15,000, and the procedures associated with this APC require the implantation of a high-cost intraocular device. In the CY 2025 OPPS/ASC final rule, we finalized to continue this policy for APC 5496 (Level 6 Intraocular Procedures) for CY 2025 and subsequent years.
In the CY 2025 OPPS/ASC final rule with comment period (89 FR 92419 through 92422), we finalized a modification to our device edits policy. While historically our device edits policy has only applied to procedures that are device-intensive based on the most recent claims data available, commenters had raised concerns about hospitals underreporting device costs in years when certain device-intensive procedures had lost device-intensive status because the device portion of a procedure can fluctuate above and below our device-intensive threshold of 30 percent. Commenters indicated to us that the presence of the device edit requirement can have a significant impact on the device portion and geometric mean cost of a procedure, particularly for newer technologies. Therefore, for CY 2025 and subsequent CYs, we finalized a policy to apply our device edits policy permanently once a procedure is designated as a device-intensive procedure in a given year. Additionally, we finalized a policy to reinstate our device edits policy for procedures that have been device-intensive since we began assigning device-intensive status at the HCPCS code level on January 1, 2017. We believed that by applying our device edit policy to procedures that were device-intensive on or after January 1, 2017, we might continue to receive device cost information for relatively new procedures with limited claims data, which may have been impacted by our policy to require that only existing device-intensive procedures be subject to our device edits policy. For CY 2026, under our modified device edits policy, our device edits requirement will apply to procedures that are designated as device-intensive in CY 2026 and will apply in subsequent years.
We did not propose any changes to our device edit policy for CY 2026 in the CY 2026 OPPS/ASC proposed rule. We received public comments. The following is a summary of the comments we received and our responses.
Comment: Some commenters supported CMS's current device edits policy.
Response: We appreciate commenters support of our device edits policy.
Comment: One commenter recommended we reinstate procedure-to-device edits, particularly for joint replacement procedures.
Response: We believe hospitals have adequate experience in coding and billing for joint replacement procedures, which are subject to our device edits policy, and do not believe it is necessary to reinstate specific procedure-to-device edits for joint replacement procedures. Therefore, we are not accepting the commenter's recommendation.
Comment: Commenters recommended that CMS establish specific new device category HCPCS C-codes to improve capturing device costs for certain procedures under the OPPS/ASC ratesetting methodologies. Specifically, commenters requested we establish a C-code for a bone-anchored annular implant, C-codes for devices used with procedures we are finalizing to remove from the inpatient-only list for CY 2026, and C-codes to describe the drug-coated balloon catheter and guidewire used for urethral stricture or prostatic hyperplasia.
Response: While we may create new device category C-codes for device categories approved under transitional pass-through status to be paid at charges reduced to cost, we do not believe it is appropriate or necessary to create additional device category C-codes outside of the transitional pass-through approval process. We believe hospitals have sufficient experience in coding and reporting significant device costs correctly on hospital claims using the existing device category HCPCS C-codes as well as uncoded revenue codes
After consideration of public comments, we are finalizing our proposal, without modification, to continue our device edits policy for CY 2026.
4. Adjustment to OPPS Payment for No Cost/Full Credit and Partial Credit Devices
a. Background
To ensure equitable OPPS payment when a hospital receives a device without cost or with full credit, in CY 2007, we implemented a policy to reduce the payment for specified device-dependent APCs by the estimated portion of the APC payment attributable to device costs (that is, the device offset) when the hospital receives a specified device at no cost or with full credit (71 FR 68071 through 68077). ( printed page 53681) Hospitals were instructed to report no cost/full credit device cases on the claim using the “FB” modifier on the line with the procedure code in which the no cost/full credit device is used. In cases in which the device is furnished without cost or with full credit, hospitals were instructed to report a token device charge of less than $1.01. In cases in which the device being inserted is an upgrade (either of the same type of device or to a different type of device) with a full credit for the device being replaced, hospitals were instructed to report as the device charge the difference between the hospital's usual charge for the device being implanted and the hospital's usual charge for the device for which it received full credit. In CY 2008, we expanded this payment adjustment policy to include cases in which hospitals receive partial credit of 50 percent or more of the cost of a specified device. Hospitals were instructed to append the “FC” modifier to the procedure code that reports the service provided to furnish the device when they receive a partial credit of 50 percent or more of the cost of the new device. We refer readers to the CY 2008 OPPS/ASC final rule with comment period for more background information on the “FB” and “FC” modifiers payment adjustment policies (72 FR 66743 through 66749).
In the CY 2014 OPPS/ASC final rule with comment period (78 FR 75005 through 75007), beginning in CY 2014, we modified our policy of reducing OPPS payment for specified APCs when a hospital furnishes a specified device without cost or with a full or partial credit. For CY 2013 and prior years, our policy had been to reduce OPPS payment by 100 percent of the device offset amount when a hospital furnishes a specified device without cost or with a full credit and by 50 percent of the device offset amount when the hospital receives partial credit in the amount of 50 percent or more of the cost for the specified device. For CY 2014, we reduced OPPS payment, for the applicable APCs, by the full or partial credit a hospital receives for a replaced device. Specifically, under this modified policy, hospitals are required to report on the claim the amount of the credit in the amount portion for value code “FD” (Credit Received from the Manufacturer for a Replaced Device) when the hospital receives a credit for a replaced device that is 50 percent or greater than the cost of the device. For CY 2014, we also limited the OPPS payment deduction for the applicable APCs to the total amount of the device offset when the “FD” value code appears on a claim. For CY 2015, we continued our policy of reducing OPPS payment for specified APCs when a hospital furnishes a specified device without cost or with a full or partial credit and to use the three criteria established in the CY 2007 OPPS/ASC final rule with comment period (71 FR 68072 through 68077) for determining the APCs to which our CY 2015 policy will apply (79 FR 66872 through 66873). In the CY 2016 OPPS/ASC final rule with comment period (80 FR 70424), we finalized our policy to no longer specify a list of devices to which the OPPS payment adjustment for no cost/full credit and partial credit devices would apply and instead apply this APC payment adjustment to all replaced devices furnished in conjunction with a procedure assigned to a device-intensive APC when the hospital receives a credit for a replaced specified device that is 50 percent or greater than the cost of the device.
b. Policy for No Cost/Full Credit and Partial Credit Devices
In the CY 2017 OPPS/ASC final rule with comment period (81 FR 79659 through 79660), for CY 2017 and subsequent years, we finalized a policy to reduce OPPS payment for device-intensive procedures, by the full or partial credit a provider receives for a replaced device, when a hospital furnishes a specified device without cost or with a full or partial credit. Under our current policy, Under our current policy, hospitals continue to be required to report on the claim the amount of the credit in the amount portion for value code “FD” when the hospital receives a credit for a replaced device that is 50 percent or greater than the cost of the device.
In the CY 2014 OPPS/ASC final rule with comment period (78 FR 75005 through 75007), we adopted a policy of reducing OPPS payment for specified APCs when a hospital furnishes a specified device without cost or with a full or partial credit by the lesser of the device offset amount for the APC or the amount of the credit. We adopted this change in policy in the preamble of the CY 2014 OPPS/ASC final rule with comment period and discussed it in subregulatory guidance, including chapter 4, section 61.3.6 of the Medicare Claims Processing Manual. Further, in the CY 2021 OPPS/ASC final rule with comment period (85 FR 86017 through 86018, 86302), we made conforming changes to our regulations at § 419.45(b)(1) and (2) that codified this policy.
We did not propose any changes related to our policies regarding payment for no cost/full credit and partial credit devices for CY 2026 in the CY 2026 OPPS/ASC proposed rule, and we did not receive public comments. We are maintaining our current policy for CY 2026.
V. OPPS Payment for Drugs, Biologicals, and Radiopharmaceuticals
A. OPPS Transitional Pass-Through Payment for Additional Costs of Drugs, Biologicals, and Radiopharmaceuticals
1. Background
Section 1833(t)(6) of the Act (42 U.S.C. 1395 l (t)(6)) provides for temporary additional payments or “transitional pass-through payments” for certain drugs and biologicals. A “biological” as used in this final rule with comment period, and as codified at 42 CFR 414.802 and 414.902 includes a “product licensed under section 351 of the PHS [Public Health Service] Act”. As enacted by the Medicare, Medicaid, and SCHIP Balanced Budget Refinement Act of 1999 (BBRA) (Pub. L. 106-113), this pass-through payment provision requires the Secretary to make additional payments to hospitals for: current orphan drugs for rare diseases and conditions, as designated under section 526 of the Federal Food, Drug, and Cosmetic Act; current drugs and biologicals and brachytherapy sources used in cancer therapy; and current radiopharmaceutical drugs and biologicals. “Current” refers to those types of drugs or biologicals mentioned above that are hospital outpatient services under Medicare Part B for which transitional pass-through payment was made on the first date the hospital OPPS was implemented.
Transitional pass-through payments also are provided for certain “new” drugs and biologicals that were not being paid for as a Hospital Outpatient Department (HOPD) service as of December 31, 1996, and whose cost is “not insignificant” in relation to the OPPS payments for the procedures or services associated with the new drug or biological. For pass-through payment purposes, radiopharmaceuticals are included as “drugs.” As required by statute, transitional pass-through payments for a drug or biological described in section 1833(t)(6)(C)(i)(II) of the Act can be made for a period of at least 2 years, but not more than 3 years, after the payment was first made for the drug as a hospital outpatient service under Medicare Part B. Final CY 2026 pass-through drugs and biologicals and their designated APCs are assigned status indicator “G” in Addenda A and ( printed page 53682) B to this final rule with comment period (which are available on the CMS website).[65]
Section 1833(t)(6)(D)(i) of the Act specifies that the pass-through payment amount, in the case of a drug or biological, is the amount by which the amount determined under section 1842(o) of the Act (42 U.S.C. 1395u(o)) for the drug or biological exceeds the portion of the otherwise applicable Medicare Outpatient Department (OPD) fee schedule that the Secretary determines is associated with the drug or biological. The methodology for determining the pass-through payment amount is set forth in regulations at 42 CFR 419.64. In accordance with section V.B.9. of the CY 2026 OPPS/ASC proposed rule, skin substitutes with an approved Biologics License Application (BLA) would be considered under transitional drug pass-through payment status. As such, we proposed to amend our regulation at § 419.64 to remove paragraph (a)(4)(iv), which currently reads “A biological that is not a skin substitute or similar product that aids wound healing.” The regulations at 42 CFR 419.64(d) specify that the pass-through payment equals the amount determined under section 1842(o) of the Act minus the portion of the Ambulatory Payment Classification (APC) payment that CMS determines is associated with the drug or biological.
Section 1847A of the Act (42 U.S.C. 1395w-3a) establishes the average sales price (ASP) methodology, which is used for payment for drugs and biologicals described in section 1842(o)(1)(C) of the Act furnished on or after January 1, 2005. The ASP methodology, as applied under the OPPS, uses several sources of data as a basis for payment, including the ASP, the wholesale acquisition cost (WAC), and the average wholesale price (AWP). In this final rule with comment period, the term “ASP methodology” and “ASP-based” are inclusive of all data sources and methodologies described therein. Additional information on the ASP methodology can be found on our website at https://www.cms.gov/medicare/payment/fee-for-service-providers/part-b-drugs/average-drug-sales-price.
The pass-through application [66] and review process for drugs and biologicals is described on our website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/pass-through-payment-status-new-technology-ambulatory-payment-classification-apc.
We received a comment on our proposal.
Comment: We received a comment requesting that CMS codify in regulation that biologicals classified and approved by the FDA under section 351 will be evaluated for coding and pass-through payment under CMS' drugs and biologics pathways and applications.
Response: While under this policy, skin substitute products approved as a drug or biological product (via the BLA pathway) under section 351 of the PHS Act may be eligible for drug pass-through, we note that, the policy does not apply to the evaluation for coding or transitional pass-through payment for other products that are not skin substitute products, including other drugs and biological products approved under section 351 of the PHS Act.
In accordance with section V.B.9. of the CY 2026 OPPS/ASC final rule with comment period, skin substitutes with an approved BLA will be considered under the transitional drug pass-through payment process. As such, we are finalizing our proposal to amend our regulation at § 419.64 to remove paragraph (a)(4)(iv) “A biological that is not a skin substitute or similar product that aids wound healing.” For more information on our payment policy for skin substitute products, we refer readers to section V.B.9 of this final rule with comment period.
2. Transitional Pass-Through Payment Period for Pass-Through Drugs, Biologicals, and Radiopharmaceuticals and Quarterly Expiration of Pass-Through Status
As required by statute, transitional pass-through payments for a drug or biological described in section 1833(t)(6)(C)(i)(II) of the Act can be made for a period of at least 2 years, but not more than 3 years, after the payment was first made for the drug or biological as a hospital outpatient service under Medicare Part B. Drugs and biologicals pass-through applications are accepted and approved on a quarterly basis in which pass-through payments for approved applications could begin on the next available OPPS quarterly update. Furthermore, our current policy, which was finalized in CY 2017 OPPS/ASC final rule with comment period (81 FR 79662), is to allow for quarterly expiration of pass-through payment status for drugs, biologicals, and radiopharmaceuticals to afford a pass-through payment period that is as close to a full 3 years as possible to allow, on a prospective basis, for the maximum pass-through payment period without exceeding the statutory limit of 3 years. Notice of drugs for which pass-through payment status is ending during the calendar year is included in the quarterly OPPS Change Request transmittals.
3. Drugs and Biologicals With Expiring Pass-Through Payment Status in CY 2025
There are 28 drugs and biologicals for which pass-through payment status expires by December 31, 2025, as listed in Table 104. These drugs and biologicals will have received OPPS pass-through payment for 3 years during the period of April 1, 2022 through December 31, 2025. In accordance with the policy finalized in the CY 2017 OPPS/ASC final rule with comment period (81 FR 79662) and described earlier, pass-through payment status for drugs and biologicals approved in CY 2017 and subsequent years will expire on a quarterly basis, with a pass-through payment period as close to 3 years as possible.
With the exception of those groups of drugs and biologicals that are always packaged when they do not have pass-through payment status (specifically, anesthesia drugs; drugs, biologicals, and radiopharmaceuticals [67] that function as supplies when used in a diagnostic test or procedure; and drugs and biologicals that function as supplies when used in a surgical procedure), our standard methodology for providing payment for drugs and biologicals with expiring pass-through payment status in an upcoming calendar year is to determine the product's estimated per day cost and compare it with the OPPS drug packaging threshold for that calendar year, which is proposed to be $140 for CY 2026 for all drugs, biologicals, and therapeutic radiopharmaceuticals (for high-cost diagnostic radiopharmaceuticals, we would provide separate payment when their per day cost greater than the finalized threshold of $655). These policies are discussed further in section V.B.1. of this final rule with comment period. If the estimated per day cost for the drug or biological is less than or equal to the applicable OPPS drug packaging threshold, we package payment for the ( printed page 53683) drug or biological into the payment for the associated procedure in the upcoming calendar year. If the estimated per day cost of the drug or biological is greater than the OPPS drug packaging threshold, we provide separate payment at the applicable ASP methodology-based payment amount (which is generally ASP plus 6 percent), as discussed further in section V.B.2. of this final rule with comment period.
( printed page 53684) ( printed page 53685)4. Drugs, Biologicals, and Radiopharmaceuticals With Pass-Through Payment Status Expiring in CY 2026
We proposed to end pass-through payment status in CY 2026 for 52 drugs and biologicals. These drugs and biologicals, which were initially approved for pass-through payment status between April 1, 2023 and January 1, 2024, are listed in Table 105. The APCs and Healthcare Common Procedure Coding System (HCPCS) codes for these drugs and biologicals, which have pass-through payment status that will end by December 31, 2026, are assigned status indicator “G” (Pass-Through Drugs and Biologicals) in Addenda A and B to this final rule with comment period (which are available on the CMS website).[68] The APCs and HCPCS codes for these drugs and biologicals are assigned status indicator “G” only for the duration of their pass-through status.
Section 1833(t)(6)(D)(i) of the Act sets the amount of pass-through payment for pass-through drugs and biologicals (the pass-through payment amount) as the difference between the amount authorized under section 1842(o) of the Act and the portion of the otherwise applicable OPD fee schedule that the Secretary determines is associated with the drug or biological. For CY 2026, we are continuing our policy to pay for pass-through drugs and biologicals using the ASP methodology, meaning a payment rate based on ASP, WAC, or AWP, as applicable. This payment rate is generally ASP plus 6 percent, equivalent to the payment rate these drugs and biologicals would receive in the physician's office setting in CY 2026. We note that, under the OPD fee schedule, separately payable drugs assigned to an APC are generally payable at ASP plus 6 percent. Therefore, a $0 pass-through payment amount will continue to be paid for pass-through drugs and biologicals under the CY 2026 OPPS because the difference between the amount authorized under section 1842(o) of the Act, which is generally ASP plus 6 percent, and the portion of the otherwise applicable OPD fee schedule that the Secretary determines is appropriate, which is generally ASP plus 6 percent, is $0.
In the case of policy-packaged drugs (which include the following: anesthesia drugs; drugs, biologicals, and radiopharmaceuticals [69] below the applicable cost threshold that function as supplies when used in a diagnostic test or procedure; and drugs and biologicals that function as supplies when used in a surgical procedure), their pass-through payment amount will continue to be equal to a payment rate calculated using the ASP methodology, meaning a payment rate based on ASP, WAC, or AWP. This payment rate will generally continue to be ASP plus 6 percent for CY 2026, minus a payment offset for the portion of the otherwise applicable OPPS payment that the Secretary determines is associated with the drug or biological. We note that if not for the pass-through payment status of these policy-packaged products, payment for these products would be packaged into the associated procedure and therefore, there are associated OPPS payment amounts for them.
We note that in the CY 2025 OPPS/ASC final rule with comment period (89 FR 93948 through 93963), we modified the regulation text at 42 CFR 419.2(b)(15) to specify that only those ( printed page 53686) diagnostic radiopharmaceuticals with per-day costs at or below the per-day diagnostic radiopharmaceutical packaging threshold for the applicable year are policy-packaged. Meaning, for those diagnostic radiopharmaceuticals that are below the diagnostic radiopharmaceutical packaging threshold, for purposes of pass-through co-insurance calculations, they are treated like policy packaged drugs. For those diagnostic radiopharmaceuticals above the diagnostic radiopharmaceutical packaging threshold, they are not packaged, and are not considered policy packaged; therefore, for purposes of pass-through co-insurance calculations, they are treated like separately payable drugs assigned to an APC. Accordingly, a $0 pass-through payment amount is assigned consistent with our policy described previously in this section for separately payable drugs assigned to an APC.
We will continue our policy to update pass-through payment rates on a quarterly basis on the CMS website during CY 2026 if later quarter ASP submissions (or more recent WAC or AWP information, as applicable) indicate that adjustments to the payment rates for these pass- through payment drugs or biologicals are necessary. For a full description of this policy, we refer readers to the CY 2006 OPPS/ASC final rule with comment period (70 FR 68632 through 68635).
For CY 2026, consistent with our CY 2025 policy for diagnostic and therapeutic radiopharmaceuticals, we will continue to provide payment for both diagnostic and therapeutic radiopharmaceuticals that are granted pass-through payment status based on the ASP methodology. As stated earlier, for purposes of pass-through payment, we consider radiopharmaceuticals to be drugs under the OPPS. Therefore, if a diagnostic or therapeutic radiopharmaceutical receives pass-through payment status during CY 2026, we will continue to follow the standard ASP methodology to determine the pass-through payment rate that drugs receive under section 1842(o) of the Act, which is generally ASP plus 6 percent. If ASP data are not available for a radiopharmaceutical, we will continue to provide pass-through payment at WAC plus 3 percent (consistent with our policy in section V.B.2.a. of this final rule with comment period), the equivalent payment provided for pass-through drugs and biologicals without ASP information. Additional detail on the WAC plus 3 percent payment policy can be found in section V.B.2.a. of this final rule with comment period. If WAC information also is not available, we will continue to provide payment for the pass-through radiopharmaceutical at 95 percent of its most recent AWP.
We refer readers to Table 105 for the list of drugs and biologicals with pass-through payment status expiring during CY 2026.
( printed page 53687) ( printed page 53688) ( printed page 53689) ( printed page 53690)We did not receive public comments on our proposal, and therefore, we are finalizing our proposal to end pass-through payment status in CY 2026 for 52 drugs and biologicals. However, we did receive comment regarding our clarification included in the CY 2026 OPPS/ASC proposed rule regarding the co-insurance calculations for diagnostic radiopharmaceuticals (90 FR 33614).
In the CY 2025 OPPS/ASC final rule with comment period (89 FR 93948 through 93963), we modified the regulation text at 42 CFR 419.2(b)(15) to specify that only those diagnostic radiopharmaceuticals with per-day costs at or below the per-day diagnostic radiopharmaceutical packaging threshold for the applicable year are policy-packaged. Meaning, for those diagnostic radiopharmaceuticals that are below the diagnostic radiopharmaceutical packaging threshold, for purposes of pass-through co-insurance calculations, they are treated like policy packaged drugs. For those diagnostic radiopharmaceuticals above the diagnostic radiopharmaceutical packaging threshold, they are not packaged, and are not considered policy packaged; therefore, for purposes of pass-through co-insurance calculations, they are treated like separately payable drugs assigned to an APC. Accordingly, a $0 pass-through payment amount is assigned consistent with our policy described previously in this section for separately payable drugs assigned to an APC.
Comment: A few commenters asked for additional clarification from CMS regarding the calculation of beneficiary copayments for diagnostic radiopharmaceuticals while on drug pass-through. Commenters requested that CMS clarify that diagnostic radiopharmaceuticals that exceed the applicable cost threshold while on transitional pass-through status would have a beneficiary copayment listed. Commenters also asked for clarification regarding the beneficiary co-insurance for specific product HCPCS codes, such as HCPCS Code A9608 (Flotufolastat f18 diag 1 mci) and HCPCS code A9611 (flurpiridaz F 18).
Commenters also requested clarification that, for any diagnostic radiopharmaceutical that is separately paid specifically because it is on transitional drug pass-through status, and whose payment is expected to be below the diagnostic radiopharmaceutical cost threshold, no beneficiary copayment should apply, which the commenter states is consistent with the principle that such costs are packaged.
Commenters contend that when a diagnostic radiopharmaceutical is applying for drug pass-through status, CMS is not making a fair comparison if it uses the diagnostic radiopharmaceutical's ASP, WAC, or AWP to determine if the diagnostic radiopharmaceutical would otherwise be packaged, because if the product had claims data, CMS would use the MUC, which is often lower than ASP, WAC, and AWP, to determine if the product was above or below the packaging threshold. Therefore, in the commenter's view, CMS is incorrectly determining that pass-through applicants would otherwise be above the packaging threshold, and thus that the product would otherwise be separately paid and that the pass-through amount is $0, resulting in applicable co-insurance for the beneficiary.
One commenter believed CMS should calculate the co-insurance for all diagnostic radiopharmaceuticals under ( printed page 53691) the policy packaged method. Additionally, this commenter stated that it is impossible for interested parties or CMS to know whether a product will exceed the cost threshold under the MUC methodology in future claims data and therefore whether a product will or will not be considered policy packaged.
Response: We thank the commenters for engaging with CMS on this issue. Only those diagnostic radiopharmaceuticals with estimated per-day costs at or below the per-day diagnostic radiopharmaceutical packaging threshold for the applicable year are policy-packaged. Meaning, for these diagnostic radiopharmaceuticals that are estimated to be below the diagnostic radiopharmaceutical packaging threshold, for purposes of pass-through co-insurance calculations, they are treated like policy packaged drugs. These diagnostic radiopharmaceuticals would have an overall $0 co-insurance payment. If a drug is policy packaged, meaning absent pass-through status there would be no separate payment for the drug, the pass-through payment amount is generally equal to a payment rate calculated using the ASP methodology (89 FR 94226). Per statute, 1833(t)(8) of the Act, there is no copayment on the pass-through payment amount. Therefore, policy packaged drugs have a $0 copayment amount, since the entirety of their separate payment amount is due to their passthrough payment status.
For those diagnostic radiopharmaceuticals estimated to be above the diagnostic radiopharmaceutical packaging threshold, their payment is not policy packaged, and they are not considered policy packaged; therefore, for purposes of pass-through co-insurance calculations, they are treated like a separately payable drug assigned to an APC. Accordingly, a $0 pass-through payment amount is assigned consistent with our policy described previously in this section for separately payable drugs assigned to an APC. The pass-through amount is the amount that is not subject to co-insurance. The non-pass-through payment amount is still subject to co-insurance provisions, including 1833(t)(3)(B) of the Act and § 419.41(d). These diagnostic radiopharmaceuticals would have a co-insurance payment.
Estimating the per day costs of radiopharmaceuticals with HCPCS codes, but without hospital claims data, is consistent with our longstanding policy regarding payment for non-pass-through drugs, biologicals, and radiopharmaceuticals with HCPCS codes but without OPPS Hospital Claims Data where CMS estimates the average number of units of each product that would typically be furnished to a patient during one day in the hospital outpatient setting and utilizes the available payment rate for the product, to determine whether their payment will be packaged.
Specifically for HCPCS Codes A9608 (Flotufolastat f18 diag 1 mci) and A9611 (flurpiridaz F 18), these HCPCS codes have co-insurance assigned during CY 2025 as these products were determined to have an estimated per day cost above the diagnostic radiopharmaceutical packaging threshold.
Comment: A commenter asked CMS to confirm that multiple offsets are not applied to the pass-through payment for diagnostic radiopharmaceuticals. This commenter was concerned that CMS may be inappropriately subtracting both the ASP-based payment under the separately payable drug methodology and subtracting the policy-packaged drug amount under the policy-packaged drug methodology.
Response: We can confirm that the payment should not have multiple offsets subtracted, either the policy-packaged offset or the threshold-packaged offset depending on the calculations performed as discussed earlier in this section to determine co-insurance status.
5. Drugs, Biologicals, and Radiopharmaceuticals With Pass-Through Payment Status Continuing Through CY 2026
We proposed to continue pass-through payment status in CY 2026 for 61 drugs and biologicals. These drugs and biologicals, which were approved for pass-through payment status with effective dates beginning between April 1, 2024 and October 1, 2025, are listed in Table 106. The APCs and HCPCS codes for these drugs and biologicals, which have pass-through payment status that would continue after December 31, 2026, are assigned status indicator “G” in Addenda A and B to the CY 2026 OPPS/ASC proposed rule (which are available on the CMS website).[70]
Section 1833(t)(6)(D)(i) of the Act sets the amount of pass-through payment for pass-through drugs and biologicals (the pass-through payment amount) as the difference between the amount authorized under section 1842(o) of the Act and the portion of the otherwise applicable OPD fee schedule that the Secretary determines is associated with the drug or biological. For CY 2026, we are continuing our policy to pay for pass-through drugs and biologicals at a payment rate based on the ASP methodology, which may be based on ASP, WAC, or AWP, but is generally ASP plus 6 percent, which is equivalent to the payment rate these drugs and biologicals would receive in the physician's office setting in CY 2026. We will continue with our policy of paying a $0 pass-through payment amount for pass-through drugs and biologicals that are not policy-packaged under the CY 2026 OPPS, because the difference between the amount authorized under section 1842(o) of the Act, which would generally be ASP plus 6 percent, and the portion of the otherwise applicable OPD fee schedule that the Secretary determines is appropriate, which would also generally be ASP plus 6 percent, is $0.
In the case of policy-packaged drugs (which include the following: anesthesia drugs; drugs, biologicals, and radiopharmaceuticals [71] that function as supplies when used in a diagnostic test or procedure; and drugs and biologicals that function as supplies when used in a surgical procedure), their pass-through payment amount would continue to be equal to a payment rate based on the ASP methodology, which may be based on ASP, WAC, or AWP, but would generally be ASP plus 6 percent for CY 2026, minus a payment offset for any predecessor drug products contributing to the pass-through payment. We note if not for the pass-through payment status of these policy-packaged products, payment for these products would be packaged into the associated procedure and therefore, there are associated OPD fee schedule amounts for them.
We are continuing our policy to update pass-through payment rates on a quarterly basis on our website during CY 2026 if later quarter ASP submissions (or more recent WAC or AWP information, as applicable) indicate that adjustments to the payment rates for these pass-through payment drugs or biologicals are necessary. For a full description of this policy, we refer readers to the CY 2006 OPPS/ASC final rule with comment period (70 FR 68632 through 68635).
For CY 2026, consistent with our CY 2025 policy for diagnostic and therapeutic radiopharmaceuticals, we proposed to continue our policy to provide payment for both diagnostic and therapeutic radiopharmaceuticals ( printed page 53692) that are granted pass-through payment status based on the ASP methodology. As stated earlier, for purposes of pass-through payment, we consider radiopharmaceuticals to be drugs under the OPPS. Therefore, if a diagnostic or therapeutic radiopharmaceutical receives pass-through payment status during CY 2026, we will continue to follow the standard ASP methodology to determine the pass-through payment rate that drugs receive under section 1842(o) of the Act, which would generally be ASP plus 6 percent. If ASP data are not available for a radiopharmaceutical, we would provide pass-through payment at WAC plus 3 percent (consistent with our policy in section V.B.2.a. of this final rule with comment period), the equivalent payment provided for pass-through drugs and biologicals without ASP information. Additional detail on the WAC plus 3 percent payment policy can be found in section V.B.2.a. of this final rule with comment period. If WAC information also is not available, we would provide payment for the pass-through radiopharmaceutical at 95 percent of its most recent AWP.
The drugs and biologicals that would have pass-through payment status expire after December 31, 2026, are shown in Table 106.
( printed page 53693) ( printed page 53694) ( printed page 53695) ( printed page 53696)We received public comments on these proposals. The following is a summary of the comments we received and our responses. ( printed page 53697)
Comment: A commenter expressed support for maintaining pass-through status for iDose® TR for all of CY 2026.
Response: We thank the commenter for their comment. We are maintaining drug pass-through status for iDose TR, which may be described by HCPCS Code J7355, for all of CY 2026.
Comment: A commenter expressed support for our inclusion of IMDELLTRA® and PAVBLU® in the List of Drugs with Pass-Through Payment Status Expiring After 2026 (Table 106) and acknowledged that we did not include drugs that were granted pass-through status after April 1, 2025 in Table 106 in the CY 2026 OPPS/ASC proposed rule, due to the timing of preparation of the CY 2026 OPPS/ASC proposed rule. The commenter requested that BKEMVTM , which was granted pass-through status effective July 1, 2025, be included in Table 106 in the CY 2026 OPPS/ASC final rule with comment period.
Response: We thank the commenter for their comment. BKEMVTM is included in Table 106 in this final rule final rule with comment period.
After consideration of public comments, we are finalizing as proposed to continue pass-through payment status in CY 2026 for 61 drugs and biologicals.
B. OPPS Payment for Drugs, Biologicals, and Radiopharmaceuticals Without Pass-Through Payment Status
We note that several commenters made comments on the Inflation Reduction Act Medicare Drug Price Negotiation Program, provider status for pharmacists under Medicare Part B, Most Favored Nation-style reference pricing, among other topics that were not discussed in the CY 2026 OPPS/ASC proposed rule. These topics are out of scope for purposes of this notice and comment rulemaking, but we will consider these comments as we consider these issues for future rulemaking.
1. Criteria for Packaging Payment for Drugs, Biologicals, and Radiopharmaceuticals
a. Packaging Threshold
In accordance with section 1833(t)(16)(B) of the Act, the threshold for establishing separate APCs for payment of drugs and biologicals was set to $50 per administration during CYs 2005 and 2006. In CY 2007, we used the four-quarter moving average Producer Price Index (PPI) levels for Pharmaceutical Preparations (Prescription) to trend the $50 threshold forward from the third quarter of CY 2005 (when the Pub. L. 108-173 mandated threshold became effective) to the third quarter of CY 2007. We then rounded the resulting dollar amount to the nearest $5 increment to determine the CY 2007 threshold amount of $55. Using the same methodology as that used in CY 2007 (which is discussed in more detail in the CY 2007 OPPS/ASC final rule with comment period (71 FR 68085 through 68086)), we set the packaging threshold for establishing separate APCs for drugs and biologicals at $140 for CY 2025 (89 FR 94237).
Following the CY 2007 methodology, for the CY 2026 OPPS/ASC proposed rule, we proposed to use the most recently available four quarter moving average PPI levels to trend the $50 threshold forward from the third quarter of CY 2005 to the third quarter of CY 2026 and round the resulting dollar amount ($141.67) to the nearest $5 increment, which yields a figure of $140. In performing this calculation, we used the most recent forecast of the quarterly index levels for the PPI for Pharmaceuticals for Human Use (Prescription) (Bureau of Labor Statistics series code WPUSI07003) from IGI. IGI is a nationally recognized economic and financial forecasting firm with which CMS contracts to forecast various price indexes including the PPI Pharmaceuticals for Human Use (Prescription). Based on these calculations, we proposed a packaging threshold for CY 2026 of $140 for drugs, biologicals, and therapeutic radiopharmaceuticals. We also proposed that if more recent data subsequently become available after the publication of the CY 2026 OPPS/ASC proposed rule, we would use such updated data, if appropriate, to determine the final CY 2026 OPPS drug packaging threshold amount in the CY 2026 OPPS/ASC final rule with comment period.
We finalized in section II.A.3.c. of the CY 2025 OPPS/ASC final rule with comment period (89 FR 94238 through 94241) to pay separately for diagnostic radiopharmaceuticals with a per-day cost above the packaging threshold for CY 2025 of $630. We also finalized that starting in CY 2026 and subsequent years, we would update this threshold by the PPI for Pharmaceuticals for Human Use (Prescription) (Bureau of Labor Statistics series code WPUSI07003) from IHS Global, Inc (IGI). For the diagnostic radiopharmaceutical packaging threshold, we finalized using the same methodology as that used in CY 2007 (which is discussed in more detail in the CY 2007 OPPS/ASC final rule with comment period (71 FR 68085 and 68086)) to calculate the update to the OPPS drug packaging threshold. Specifically, we finalized that, starting for the CY 2026 rulemaking, we would use the most recently available four quarter moving average PPI levels to trend the final current year (CY 2025) threshold forward from the third quarter of the data year (CY 2024) to the third quarter of the current year (CY 2025) and round the resulting dollar amount to the nearest $5 increment. In the CY 2026 OPPS/ASC proposed rule, we proposed a technical refinement to this policy to use the most recently available four-quarter moving average PPI levels to trend the CY 2025 final threshold forward from the third quarter of CY 2025 to the third quarter of the payment year (CY 2026) and round the resulting dollar amount to the nearest $5 increment. We believed using the most recently available four quarter moving average PPI levels more appropriately updates the packaging threshold from CY 2025 for payment in CY 2026. For the CY 2026 OPPS/ASC proposed rule, we used the most recently available four quarter moving average PPI levels to trend the $630 diagnostic radiopharmaceutical packaging threshold forward from the third quarter of CY 2025 to the third quarter of CY 2026 and we rounded the resulting dollar amount ($654.23) to the nearest $5 increment, which yielded a figure of $655. We also proposed that if more recent data subsequently becomes available after the publication of the CY 2026 OPPS/ASC proposed rule, we would use such updated data, if appropriate, to determine the final CY 2026 diagnostic radiopharmaceutical packaging threshold amount in the CY 2026 OPPS/ASC final rule with comment period. For CY 2027 and subsequent updates, we therefore proposed to trend the CY 2025 threshold of $630 forward using the four-quarter moving average PPI levels for Pharmaceuticals for Human Use, Prescription for CY 2025 (third quarter) forward using the PPI for Pharmaceuticals for Human Use, Prescription for the applicable payment year (third quarter).
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: Some commenters supported the drug packaging threshold. One commenter stated that a static threshold would fail to account for rapid drug inflation. This same commenter urged CMS to implement a transparent and predictable methodology for adjusting the threshold annually to reflect drug price inflation.
Response: We thank the commenters for their support. We believe using the ( printed page 53698) four-quarter moving average PPI levels for Pharmaceuticals for Human Use, Prescription to adjust the OPPS drug packaging threshold through notice and comment rulemaking is a transparent and predictable methodology that has been in use under the OPPS for many years.
Comment: Several commenters did not support CMS following the proposed methodology to update the OPPS drug packaging threshold. These commenters had concerns that the packaging threshold approach introduces financial and operational concerns, especially when multiple packaged drugs are used during a procedure. Commenters recommended using a different inflation factor, maintaining the current threshold, or rolling back the threshold.
Response: We appreciate these perspectives; however, we believe that the PPI update factor provides aggregate changes in the selling prices of pharmaceuticals, which makes it an appropriate factor with which to update the OPPS drug packaging threshold to ensure that as costs change over time, the threshold continues to identify products appropriate for payment packaging, a fundamental principle of a prospective payment system. For CY 2026, the update to the inflation factor results in the same rounded final OPPS drug packaging threshold for CY 2026 compared to CY 2025.
After consideration of public comments, we are finalizing our proposal to use the most recently available four-quarter moving average PPI levels to trend the $50 threshold forward from the third quarter of CY 2005 to the third quarter of CY 2026 and round the resulting dollar amount to the nearest $5 increment for the OPPS drug packaging threshold. We proposed that if more recent data subsequently become available after the publication of the CY 2026 OPPS/ASC proposed rule, we would use such updated data, if appropriate, to determine the final CY 2026 OPPS drug packaging threshold amount in the CY 2026 OPPS/ASC final rule with comment period. Using the most recent forecast of the quarterly index levels for the PPI for Pharmaceuticals for Human Use (Prescription) (Bureau of Labor Statistics series code WPUSI07003) from IGI, we trended the $50 threshold forward from the third quarter of CY 2005 to the third quarter of CY 2026 and round the resulting dollar amount ($140.13) to the nearest $5 increment, which yields a figure of $140.
We refer readers to section II.A.3.c.(2). of this final rule with comment period for additional details regarding the diagnostic radiopharmaceutical packaging threshold comment and response discussion. We are finalizing our proposal to update the CY 2025 $630 threshold amount by the four-quarter moving average PPI levels for Pharmaceuticals for Human Use, Prescription to trend the $630 threshold forward. Specifically, we are using the most recently available forecast of the four-quarter moving average PPI levels for Pharmaceutical for Human Use, Prescription from the third quarter of 2025 to the third quarter of 2026, and to round the resulting dollar amount to the nearest $5 increment. We also proposed, and are subsequently finalizing, that if more recent data are subsequently available (for example, a more recent estimate of the PPI for Pharmaceuticals for Human Use, Prescription), we would use such data, if appropriate, to determine the CY 2026 diagnostic radiopharmaceutical packaging threshold in the final rule. Based on this methodology, using the most recent data available for this final rule, we trended the $630 threshold forward and rounded the resulting dollar amount ($656.65) to the nearest $5 increment, which yields a final diagnostic radiopharmaceutical packaging threshold figure of $655 per day for CY 2026.
b. Packaging of Payment for HCPCS Codes That Describe Certain Drugs, Certain Biologicals, and Certain Radiopharmaceuticals Under the Cost Thresholds
To determine the proposed CY 2026 packaging status for all nonpass-through drugs, biologicals, diagnostic and therapeutic radiopharmaceuticals that are not policy packaged, we calculated, on a HCPCS code-specific basis, the per day cost of all drugs, biologicals, and therapeutic radiopharmaceuticals that had a HCPCS code in CY 2024 and were paid (via packaged or separate payment) under the OPPS. We used data from CY 2024 claims processed through December 31, 2024, for this calculation. However, we did not perform this calculation for those drugs and biologicals with multiple HCPCS codes that include different dosages, as described in section V.B.1.d. of this final rule with comment period, or for the following policy-packaged items that we proposed to continue to package in CY 2026: anesthesia drugs; drugs, biologicals, and contrast agents and other drugs that function as supplies when used in a diagnostic test or procedure; and drugs and biologicals that function as supplies when used in a surgical procedure. Consistent with our policy described in section V.B.5. of this final rule with comment period, in situations where we have no claims data and must determine if these products exceed the per-day cost threshold, we estimated the average number of units of each product that would typically be furnished to a patient during one day in the hospital outpatient setting and utilized the ASP methodology to determine whether their payment will be packaged as well as their payment status indicators.
To calculate the per day costs for drugs, biologicals, diagnostic radiopharmaceuticals, and therapeutic radiopharmaceuticals to determine their proposed packaging status in CY 2026, we used the methodology that was described in detail in the CY 2006 OPPS proposed rule (70 FR 42723 through 42724) and finalized in the CY 2006 OPPS final rule with comment period (70 FR 68636 through 68638). For each drug and biological HCPCS code, we used an estimated payment rate based on the ASP methodology, which is generally ASP plus 6 percent (which is the payment rate we proposed for separately payable drugs and biologicals) for CY 2026, as discussed in more detail in section V.A.1. and V.B.2. of the CY 2026 OPPS/ASC proposed rule to calculate the CY 2026 proposed rule per day costs. We used the manufacturer-submitted ASP data from the fourth quarter of CY 2024 (data that were used for payment purposes in the physician's office setting, effective April 1, 2025) to determine the CY 2026 OPPS/ASC proposed rule per day cost.
As is our standard methodology, for CY 2026, we proposed to use payment rates based on the ASP data from the fourth quarter of CY 2024 for budget neutrality estimates, packaging determinations, impact analyses, and completion of Addenda A and B to the CY 2026 OPPS/ASC proposed rule (which are available via the internet on the CMS website) because these are the most recent data available for use at the time of development of the CY 2026 OPPS/ASC proposed rule. These data also are the basis for drug payments in the physician's office setting, effective April 1, 2025. Exceptions to our standard methodology include:
- For therapeutic radiopharmaceuticals that do not have pass-through status as of April 1, 2025, and do not have an ASP-based payment rate, we did not use a payment rate based on WAC or AWP for those items, consistent with our policy described in section V.B.3.a. of this final rule with comment period. Instead, we used their arithmetic mean unit cost derived from the CY 2024 hospital claims data to determine their per day cost. ( printed page 53699)
- For diagnostic radiopharmaceuticals that do not have pass-through status as of April 1, 2025, we used their mean unit cost derived from the CY 2024 hospital claims data to determine their per day cost. We did not use an ASP-based, WAC-based, or AWP-based payment rate for those items unless there was no mean unit cost reported for the product, consistent with our finalized policy described in section V.B.3.b of this final rule with comment period.
- For items other than diagnostic or therapeutic radiopharmaceuticals that did not have either an ASP-based payment rate, a payment rate based on WAC, or a payment rate based on AWP, we used the arithmetic mean unit cost of the items derived from the CY 2024 hospital claims data to determine their per day cost.
We proposed to package drugs, biologicals, and therapeutic radiopharmaceuticals with a per day cost less than or equal to $140 and identify items with a per day cost greater than $140 as separately payable unless they are policy-packaged. For diagnostic radiopharmaceuticals, we proposed to package those items with a per day cost less than or equal to $655 and identify items with a per day cost greater than $655 as separately payable. Consistent with our past practice (72 FR 667580), we cross-walked historical OPPS claims data from the CY 2024 HCPCS codes that were reported to the CY 2024 HCPCS codes that we display in Addendum B to the CY 2026 OPPS/ASC proposed rule (which is available on the CMS website) [72] for proposed payment in CY 2026.
Our policy during previous cycles of OPPS rulemaking has been to use updated ASP and claims data to make final determinations of the packaging status of HCPCS codes for drugs, biologicals, and therapeutic radiopharmaceuticals for the OPPS/ASC final rule with comment period (71 FR 68086; 78 FR75022; 89 FR 94238). We note that it is also our policy to make an annual packaging determination for a HCPCS code only when we develop the OPPS/ASC final rule with comment period for the update year (71 FR 68086). Only HCPCS codes that are identified as separately payable in the final rule with comment period are subject to quarterly updates. For our calculation of per day costs of HCPCS codes for drugs, biologicals, and radiopharmaceuticals in the CY 2026 OPPS/ASC proposed rule, we proposed to use ASP data from the fourth quarter of CY 2024, which is the basis for calculating payment rates for drugs and biologicals in the physician's office setting using the ASP methodology, effective April 1, 2025, along with updated hospital claims data from CY 2024. We note that we also proposed to use these data for budget neutrality estimates and impact analyses for the CY 2026 OPPS/ASC proposed rule.
We proposed that payment rates for HCPCS codes for separately payable drugs and biologicals included in Addenda A and B of the CY 2026 OPPS/ASC final rule with comment period would be based on ASP data from the second quarter of CY 2025. These data are the basis for calculating payment rates for drugs and biologicals in the physician's office setting using the ASP methodology, effective October 1, 2025. These payment rates would then be updated in the January 2026 OPPS update, based on the most recent ASP data to be used for physicians' office and OPPS payment as of January 1, 2026. For drugs and biologicals that do not currently have a payment rate based on ASP, WAC, or AWP, for therapeutic radiopharmaceuticals that do not currently have an ASP payment rate, and for all diagnostic radiopharmaceuticals, we will calculate their arithmetic mean unit cost from all of the CY 2024 claims data and updated cost report information available for the CY 2026 final rule with comment period to determine their final per day cost.
Consequently, the final rule with comment period packaging status of some HCPCS codes for drugs, biologicals, and radiopharmaceuticals in the CY 2026 OPPS/ASC proposed rule may be different from the same drugs' HCPCS codes' packaging status determined based on the data used for this final rule with comment period. Under such circumstances, we proposed to continue to follow the established policies initially adopted for the CY 2005 OPPS final rule with comment period (69 FR 65780) is in order to more equitably pay for those drugs whose costs fluctuate relative to the proposed CY 2026 OPPS drug packaging threshold and the drug's payment status (packaged or separately payable) in CY 2026. These established policies have not changed for many years and are the same as described in the CY 2016 OPPS/ASC final rule with comment period (80 FR 70434). Specifically, for CY 2026 and subsequent years, consistent with our historical practice, we proposed to apply the following policies to those HCPCS codes for drugs, biologicals, and therapeutic radiopharmaceuticals whose relationship to the drug packaging threshold changes based on the updated drug packaging threshold and on the final updated data:
- HCPCS codes for drugs, biologicals, and radiopharmaceuticals that were paid separately in CY 2025 and that are proposed for separate payment in CY 2026, and that then have per day costs equal to or less than the CY 2026 final rule drug packaging threshold or diagnostic radiopharmaceutical packaging threshold, based on the updated ASPs and hospital claims data used for the CY 2026 final rule, would continue to receive separate payment in CY 2026.
- HCPCS codes for drugs, biologicals, and radiopharmaceuticals that were packaged in CY 2025 and that are proposed for separate payment in CY 2026, and that then have per day costs equal to or less than the CY 2026 final rule drug packaging threshold or diagnostic radiopharmaceutical packaging threshold, based on the updated ASPs and hospital claims data used for the CY 2026 final rule, would remain packaged in CY 2026.
- HCPCS codes for drugs, biologicals, and radiopharmaceuticals for which we proposed packaged payment in CY 2026 but that then have per-day costs greater than the CY 2026 final rule drug packaging threshold or diagnostic radiopharmaceutical packaging threshold, based on the updated ASPs and hospital claims data used for the CY 2026 final rule, would receive separate payment in CY 2026.
We did not receive public comments on this provision, and therefore, we are finalizing as proposed.
c. Policy-Packaged Drugs, Biologicals, and Radiopharmaceuticals
As mentioned earlier in this section, under the OPPS, we package several categories of nonpass-through drugs, biologicals, and radiopharmaceuticals, regardless of the cost of the products. Because the products are packaged according to the policies in 42 CFR 419.2(b), we refer to these packaged drugs, biologicals, and radiopharmaceuticals as “policy-packaged” drugs, biologicals, and radiopharmaceuticals. These policies are either longstanding or based on longstanding principles and inherent to the OPPS and are currently as follows:
- Anesthesia, certain drugs, biologicals, and other pharmaceuticals; medical and surgical supplies and equipment; surgical dressings; and devices used for external reduction of fractures and dislocations (§ 419.2(b)(4));
- Intraoperative items and services (§ 419.2(b)(14)); ( printed page 53700)
- Drugs, biologicals, and radiopharmaceuticals that function as supplies when used in a diagnostic test or procedure (including but not limited to, diagnostic radiopharmaceuticals with per-day costs at or below the per-day diagnostic radiopharmaceutical packaging threshold for the applicable year, contrast agents, and pharmacologic stress agents) (§ 419.2(b)(15)); and
- Drugs and biologicals that function as supplies when used in a surgical procedure (including, but not limited to, skin substitutes and similar products that aid wound healing and implantable biologicals) (§ 419.2(b)(16)).
The policy at § 419.2(b)(16) is broader than the policy at § 419.2(b)(14). As we stated in the CY 2015 OPPS/ASC final rule with comment period: “We consider all items related to the surgical outcome and provided during the hospital stay in which the surgery is performed, including postsurgical pain management drugs, to be part of the surgery for purposes of our drug and biological surgical supply packaging policy” (79 FR 66875). The category described by § 419.2(b)(15) is large and includes diagnostic radiopharmaceuticals that have a per day cost below the finalized diagnostic radiopharmaceutical packaging threshold that we discuss in section II.A.3. of this final rule with comment period, contrast agents, stress agents, and some other products. The category described by § 419.2(b)(16) currently includes skin substitutes and some other products. We believe it is important to reiterate that cost consideration is not a factor when determining whether an item is a surgical supply (79 FR 66875).
We received public comments on these policies. The following is a summary of the comments we received and our responses.
Comment: One commenter suggested CMS should develop a policy that covers drugs that are administered at the time of ophthalmic surgery, that are direct substitutes for postoperative medications, or that have an FDA-approved indication to treat/prevent post-operative issues, such as pain, inflammation, or infection, separately under Medicare Part B.
Response: We thank the commenter for their suggestion. We believe such a policy already exists under 42 CFR 419.2(b)(16)), in which drugs and biologicals that function as supplies when used in a surgical procedure (including, but not limited to, skin substitutes and similar products that aid wound healing and implantable biologicals) have their payment policy-packaged into the procedures in which the product is used. We believe these drugs function as supplies and are supportive to the procedures in which they are used, which maintains the important packaging principles of the OPPS to encourage efficiencies.
We did not propose any changes to our policy for policy-packaged drugs, biologicals, and radiopharmaceuticals; therefore, after consideration of comments received, we are continuing the policies at 42 CFR 419.2(b).
d. Packaging Determination for HCPCS Codes That Describe the Same Drug or Biological but Different Dosages
In the CY 2010 OPPS/ASC final rule with comment period (74 FR 60490 through 60491), we finalized a policy to make a single packaging determination for a drug, rather than an individual HCPCS code, when a drug has multiple HCPCS codes describing different dosages because we believe that adopting the standard HCPCS code-specific packaging determinations for these codes could lead to inappropriate payment incentives for hospitals to report certain HCPCS codes instead of others. We continue to believe that making packaging determinations on a drug-specific basis eliminates payment incentives for hospitals to report certain HCPCS codes for drugs and allows hospitals flexibility in choosing to report all HCPCS codes for different dosages of the same drug or only the lowest dosage HCPCS code. Therefore, we proposed to continue our policy to make packaging determinations on a drug-specific basis, rather than a HCPCS code-specific basis, for those HCPCS codes that describe the same drug or biological but different dosages in CY 2026.
To propose a packaging determination that is consistent across all HCPCS codes that describe different dosages of the same drug or biological, we aggregated both our CY 2024 claims data and our pricing information, which is based on the ASP methodology, generally ASP plus 6 percent, across all of the HCPCS codes that describe each distinct drug or biological in order to determine the mean units per day of the drug or biological in terms of the HCPCS code with the lowest dosage descriptor. The following drugs did not have pricing information available for the ASP methodology for the CY 2026 OPPS/ASC proposed rule; and, as is our current policy for determining the packaging status of other drugs, we used the mean unit cost available from the CY 2024 claims data to make the proposed packaging determinations for them: HCPCS code J3472 (Injection, hyaluronidase, ovine, preservative free, per 1000 usp units); HCPCS code J7100 (Infusion, dextran 40,500 ml); and HCPCS code J7110 (Infusion, dextran 75,500 ml).
For all other drugs and biologicals that have HCPCS codes describing different doses, we then multiplied the proposed weighted average ASP methodology based payment rate, which is generally ASP plus 6 percent, per-unit payment amount across all dosage levels of a specific drug or biological by the estimated units per day for all HCPCS codes that describe each drug or biological from our claims data to determine if the estimated per day cost of each drug or biological is less than or equal to the proposed CY 2026 drug packaging threshold of $140 (in which case all HCPCS codes for the same drug or biological would be packaged) or greater than the proposed CY 2026 drug packaging threshold of $140 (in which case all HCPCS codes for the same drug or biological would be separately payable). The proposed and final packaging status of each drug and biological HCPCS code to which this methodology would apply in CY 2026 is displayed in Table 107.
( printed page 53701)We proposed that our policy to make packaging determinations on a drug-specific basis, rather than a HCPCS code-specific basis, for those HCPCS codes that describe the same drug or biological but different dosages in CY 2026 would also apply to diagnostic radiopharmaceuticals. This is because, as with drugs and biologicals, we believe that adopting standard HCPCS code-specific packaging determinations for radiopharmaceutical codes could lead to inappropriate payment incentives for hospitals to report certain HCPCS codes instead of others. To propose a packaging determination that is consistent across all HCPCS codes that describe different dosages of the same diagnostic radiopharmaceutical, we would aggregate our CY 2024 claims data across all the HCPCS codes that describe each distinct diagnostic radiopharmaceutical to determine the mean units per day of the diagnostic radiopharmaceutical in terms of the HCPCS code with the lowest dosage descriptor. We would then analyze the aggregate per day cost of the diagnostic radiopharmaceutical to determine if the per day cost is less than or equal to the proposed CY 2026 diagnostic radiopharmaceutical packaging threshold of $655 (in which case all HCPCS codes for the same diagnostic radiopharmaceutical would be packaged) or greater than the proposed CY 2026 diagnostic radiopharmaceutical packaging threshold of $655 (in which case all HCPCS codes for the same diagnostic radiopharmaceutical would be separately payable). There are currently no diagnostic radiopharmaceuticals that this policy would apply to.
We did not receive any public comments on our proposal, and we are finalizing our proposal without modification, and confirm that the final CY 2026 drug packaging threshold remains at $140 per day as described in section V.B.1.a. of this final rule with comment period. All parts of the proposal are finalized without modification.
2. Payment for Drugs and Biologicals Without Pass-Through Status That Are Not Packaged
a. Payment for Specified Covered Outpatient Drugs (SCODs) and Other Separately Payable Drugs and Biologicals
Section 1833(t)(14) of the Act defines certain separately payable radiopharmaceuticals, drugs, and biologicals and mandates specific payments for these items. Under section 1833(t)(14)(B)(i) of the Act, a “specified covered outpatient drug” (known as a SCOD) is defined as a covered outpatient drug, as defined in section 1927(k)(2) of the Act, for which a separate APC has been established and that either is a radiopharmaceutical agent or a drug or biological for which payment was made on a pass-through basis on or before December 31, 2002.
Under section 1833(t)(14)(B)(ii) of the Act, certain drugs and biologicals are designated as exceptions and are not included in the definition of SCODs. These exceptions are—
- A drug or biological for which payment is first made on or after ( printed page 53702) January 1, 2003, under the transitional pass-through payment provision in section 1833(t)(6) of the Act.
- A drug or biological for which a temporary HCPCS code has not been assigned.
- During CYs 2004 and 2005, an orphan drug (as designated by the Secretary).
Section 1833(t)(14)(A)(iii) of the Act requires that payment for SCODs in CY 2006 and subsequent years be equal to the average acquisition cost for the drug for that year as determined by the Secretary, subject to any adjustment for overhead costs and considering the hospital acquisition cost survey data collected by the Government Accountability Office (GAO) in CYs 2004 and 2005, and later periodic surveys conducted by the Secretary as set forth in the statute. If hospital acquisition cost data are not available, the law requires that payment be equal to payment rates established under the methodology described in section 1842(o), section 1847A, or section 1847B of the Act, as calculated and adjusted by the Secretary as necessary for purposes of paragraph (14). We refer to this alternative methodology as the “statutory default.” Most physician Part B drugs are paid at ASP plus 6 percent in accordance with section 1842(o) and section 1847A of the Act.
Section 1833(t)(14)(E)(ii) of the Act provides for an adjustment in OPPS payment rates for SCODs to consider overhead and related expenses, such as pharmacy services and handling costs. Section 1833(t)(14)(E)(i) of the Act required MedPAC to study pharmacy overhead and related expenses and to make recommendations to the Secretary regarding whether, and if so how, a payment adjustment should be made to compensate hospitals for overhead and related expenses. Section 1833(t)(14)(E)(ii) of the Act authorizes the Secretary to adjust the weights for ambulatory procedure classifications for SCODs to consider the findings of the MedPAC study.[73]
It has been our policy since CY 2006 to apply the same treatment to all separately payable drugs and biologicals, which include SCODs, and drugs and biologicals that are not SCODs. Therefore, we apply the payment methodology in section 1833(t)(14)(A)(iii) of the Act to SCODs, as required by statute, but we also apply it to separately payable drugs and biologicals that are not SCODs, which is a policy determination rather than a statutory requirement. For CY 2023 and subsequent years, we finalized a policy to apply section 1833(t)(14)(A)(iii)(II) of the Act to all separately payable drugs and biologicals, including SCODs. Although we do not distinguish SCODs in this discussion, we note that we are required to apply section 1833(t)(14)(A)(iii)(II) of the Act to SCODs; but we also are applying this provision to other separately payable drugs and biologicals, consistent with our history of using the same payment methodology for all separately payable drugs and biologicals.
For a detailed discussion of our OPPS drug payment policies from CY 2006 to CY 2012, we refer readers to the CY 2013 OPPS/ASC final rule with comment period (77 FR 68383 through 68385). In the CY 2013 OPPS/ASC final rule with comment period (77 FR 68386 through 68389), we first adopted the statutory default policy to pay for separately payable drugs and biologicals at ASP plus 6 percent based on section 1833(t)(14)(A)(iii)(II) of the Act. We have continued this policy of paying for separately payable drugs and biologicals at the statutory default for CYs 2014 through 2025.
In the case of a drug or biological during an initial sales period in which data on the prices for sales of the drug or biological are not sufficiently available from the manufacturer, section 1847A(c)(4) of the Act permits the Secretary to make payments that are based on WAC. Under section 1833(t)(14)(A)(iii)(II) of the Act, the amount of payment for a separately payable drug equals the average price for the drug for the year established under, among other authorities, section 1847A of the Act. As explained in greater detail in the CY 2019 PFS final rule, under section 1847A(c)(4) of the Act, although payments may be based on WAC, unlike section 1847A(b) of the Act (which specifies that payments using ASP or WAC must be made with a 6 percent add-on), section 1847A(c)(4) of the Act does not require that a particular add-on amount be applied to WAC-based pricing for this initial period when ASP data are not available. Consistent with section 1847A(c)(4) of the Act, in the CY 2019 PFS final rule (83 FR 59661 to 59666), we finalized a policy that, effective January 1, 2019, WAC-based payments for Part B drugs made under section 1847A(c)(4) of the Act will utilize a 3 percent add-on in place of the 6 percent add-on that was being used according to our policy in effect as of CY 2018. For the CY 2019 OPPS, we followed the same policy finalized in the CY 2019 PFS final rule (83 FR 59661 to 59666). Since CY 2020, we have continued to utilize a 3 percent add-on instead of a 6 percent add-on for drugs that are paid based on WAC pursuant to our authority under section 1833(t)(14)(A)(iii)(II) of the Act (84 FR 61318 and 85 FR 86039), which provides, in part, that the amount of payment for a SCOD is the average price of the drug in the year established under section 1847A of the Act. We also apply this provision to non-SCOD separately payable drugs, biologicals, and certain radiopharmaceuticals. Because we establish the average price for a drug paid based on WAC under section 1847A of the Act as WAC plus 3 percent instead of WAC plus 6 percent, we believe it is appropriate to price separately payable drugs paid based on WAC at the same amount under the OPPS. Our policy to pay for drugs and biologicals at WAC plus 3 percent, rather than WAC plus 6 percent, applies whenever WAC-based pricing is used for a drug, biological, or radiopharmaceutical under section 1847A(c)(4). When WAC-based pricing is used for a drug, biological, or radiopharmaceutical, but not under section 1847A(c)(4), the payment of WAC plus 6 percent would apply. We refer readers to the CY 2019 PFS final rule (83 FR 59661 to 59666) for additional background on this policy.
Consistent with our current policy, payments for separately payable drugs, biologicals, and radiopharmaceuticals are included in the budget neutrality adjustments, under the requirements in section 1833(t)(9)(B) of the Act. Also, the budget neutral weight scalar is not applied in determining payments for these separately payable drugs and biologicals.
Separately payable drug, biological, and radiopharmaceutical payment rates were listed in Addenda A and B to the CY 2026 OPPS/ASC proposed rule (available on the CMS website).[74] These addenda provided the proposed CY 2026 payment rates based on the ASP methodology for separately payable nonpass-through drugs, biologicals, and radiopharmaceuticals, with exceptions for certain radiopharmaceuticals previously discussed, and the ASP methodology for pass-through drugs, biologicals, and radiopharmaceuticals. Except for proposed payment rates for certain radiopharmaceuticals, these rates were based either on ASP information that is the basis for calculating payment rates for drugs and biologicals in the physician's office ( printed page 53703) setting effective April 1, 2025, or WAC, AWP, or mean unit cost from CY 2024 claims data and updated cost report information available for the proposed rule. For nonpass-through therapeutic radiopharmaceuticals, payment rates were based on ASP data or mean unit cost. We proposed to pay separately at mean unit cost for diagnostic radiopharmaceuticals with per day costs above the proposed threshold; the payment rates proposed for qualifying diagnostic radiopharmaceuticals are entirely mean unit cost if available. See section II.A.3.c.(3) for the finalized policy regarding payment of qualifying diagnostic radiopharmaceuticals. In general, these published proposed payment rates will not be the same as the actual January 2026 payment rates. This is because payment rates for drugs, biologicals, and therapeutic radiopharmaceuticals with ASP information for January 2026 will be determined through the standard quarterly process where ASP data submitted by manufacturers for the third quarter of CY 2025 (July 1, 2025, through September 30, 2025) will be used to set the payment rates that are released for the quarter beginning in January 2026 in December 2025. In addition, in Addenda A and B to the CY 2026 OPPS/ASC proposed rule, payment rates for drugs, biologicals, and therapeutic radiopharmaceuticals for which there was no ASP, WAC, or AWP information available for April 2025, as well as all separately payable diagnostic radiopharmaceuticals, were based on mean unit cost in the available CY 2024 claims data.
If new pricing information becomes available for payment for the quarter beginning in January 2026, we will price payment for these drugs, biologicals, therapeutic radiopharmaceuticals, and diagnostic radiopharmaceuticals based on their newly available information. Finally, there may be drugs, biologicals and therapeutic radiopharmaceuticals that had ASP, WAC, or AWP information available for the proposed rule (reflecting April 2025 ASP data) that do not have ASP, WAC, or AWP information available for the quarter beginning in January 2026. These drugs, biologicals and therapeutic radiopharmaceuticals would then be paid based on arithmetic mean unit cost data derived from CY 2024 hospital claims. Therefore, the proposed payment rates listed in Addenda A and B to the CY 2026 OPPS/ASC proposed rule were not for January 2026 payment purposes and were only illustrative of the CY 2026 OPPS payment methodology using the most recently available information at the time of issuance of the CY 2026 OPPS/ASC proposed rule.
As previously discussed, we proposed that payment rates for HCPCS codes for separately payable drugs and biologicals included in Addenda A and B of this CY 2026 OPPS/ASC final rule with comment period would be based on ASP data from the second quarter of CY 2025. These data are the basis for calculating payment rates for drugs and biologicals in the physician's office setting using the ASP methodology, effective October 1, 2025. These payment rates would then be updated in the January 2026 OPPS update, based on the most recent ASP data to be used for physicians' office and OPPS payment as of January 1, 2026. For drugs and biologicals that do not currently have a payment rate based on ASP, WAC, or AWP, for therapeutic radiopharmaceuticals that do not currently have an ASP payment rate, and for all diagnostic radiopharmaceuticals, we would calculate their arithmetic mean unit cost from all of the CY 2024 claims data and updated cost report information available for the CY 2026 final rule with comment period to determine their final per day cost.
We note that payment amounts for most drugs separately payable under Medicare Part B are determined using the methodology in section 1847A of the Act, and in many cases, payment is based on the average sales price (ASP) plus a statutorily mandated 6 percent add-on.
In CY 2025, we clarified that only ASP data or, if ASP data are not available, mean unit cost data, would be used to set payment rates for separately payable nonpass-through therapeutic radiopharmaceuticals under the OPPS. For CY 2026, we did not propose any changes to our policies for payment for separately payable therapeutic or diagnostic radiopharmaceuticals other than the technical update that was proposed to the diagnostic radiopharmaceutical packaging threshold update factor as discussed in section V.B.1. of this final rule with comment period.
For CY 2026, we did not propose any additional changes to our policies for payment for separately payable drugs, biologicals, and radiopharmaceuticals. We proposed to continue our payment policy that has been in effect since CY 2013 to pay for separately payable drugs and biologicals in accordance with section 1833(t)(14)(A)(iii)(II) of the Act (the statutory default).
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: Several commenters supported our proposals related to the separate payment for separately payable non-passthrough drugs and biologicals under the OPPS.
Response: We thank the commenters for their support.
Comment: Several commenters suggested technical improvements for CMS to consider regarding the operationalization of drug payment. Suggestions included improving transparency regarding updates to the CMS Average Sales Price Pricing Files, the OPPS/ASC Addenda Files, and the NCCI Medically Unlikely Edits. A commenter also suggested enabling a timelier issuance of J-codes to improve transparency for new drugs and to accelerate the rate at which ASP data is utilized following ASP reporting to CMS by the manufacturer. Two commenters requested CMS designate additional resources, including career staff and guidance, for communication with the pharmaceutical industry to increase transparency and ensure timely inclusion of new products in the OPPS payment files. One commenter indicates that uncertainty regarding Medicare payment often causes providers to hesitate or delay adoption of new therapies. Additionally, one commenter suggested CMS develop a drug-intensive policy for ASC rate setting similar to the current device-intensive policy. Additionally, two commenters recommended CMS increase transparency regarding packaging status post pass-through expiration and include the expected packaging status of a drug as part of the rulemaking process.
Response: We thank the commenters for their feedback and recommendations. While these comments are generally out of scope for purposes of this rulemaking, we will take these comments into consideration for future rulemaking, future issuance of sub-regulatory guidance, and future internal process improvements. Based on these comments we may also consider publishing additional files, documents, and data as part of the rulemaking process in future years.
After consideration of public comments, we are finalizing, without modification, our proposals to continue our payment policy for separately payable drugs and biologicals in accordance with section 1833(t)(14)(A)(iii)(II) of the Act.
b. Biosimilar Biological Products
For CY 2024, we finalized the exception of biosimilars from the OPPS threshold packaging policy when their ( printed page 53704) reference products are separately paid (88 FR 81783 through 81785). This policy allows for separate payment for biosimilars even if the biosimilar's per-day cost is below the packaging threshold if the biosimilar's reference product is separately paid. This policy removes the financial incentive to use a more expensive separately payable biological and promotes biosimilar use as a lower cost alternative to higher cost reference products.
Payment rates for drugs and biologicals (including biosimilars) under Medicare Part B are determined using the methodology in section 1847A of the Act, and in many cases, payment is based on the average sales price (ASP) plus a statutorily mandated 6 percent add-on. Additionally, section 11403 of the IRA requires that a qualifying biosimilar be paid at ASP plus 8 percent of the reference product's ASP rather than 6 percent during the applicable 5-year period. Section 1847A(b)(8)(B)(ii) of the Act defines the applicable 5-year period for a qualifying biosimilar for which payment has been made using ASP (that is, payment under section 1847A(b)(8) of the Act) as of September 30, 2022, as the 5-year period beginning on October 1, 2022. For a qualifying biosimilar for which payment is first made using ASP during the period beginning October 1, 2022, and ending December 31, 2027, the statute defines the applicable 5-year period as the 5-year period beginning on the first day of such calendar quarter of such payment (88 FR 81783). These payment rates are published in the quarterly release of Addendum B or ASP pricing files.
Comment: A few commenters supported the continuation of our biosimilar packaging exception policy that was implemented in CY 2024. The commenters stated they support policies that encourage a competitive marketplace and do not penalize price competition. Furthermore, a commenter suggested CMS expand the current biosimilar packaging exception policy to all biosimilars regardless of the reference product's packaging status. Another commenter expressed concerns regarding the OPPS packaging policy and believed it hinders competition against the reference products.
Response: We thank the commenters for their support of continuing our biosimilar packaging exception policy. We believe the current policy supports both our intent to promote the use of biosimilars as a less expensive alternative to the reference products while also preserving our longstanding OPPS packaging intent to create incentives for efficiency. We continue to believe packaging is an essential component of a prospective payment system and do not support categorically excepting all biosimilars from the threshold packaging policy.
After consideration of public comments we received, we are finalizing our proposal without modification to continue the exception of biosimilars from the OPPS threshold packaging policy when their reference products are separately paid. For CY 2026, we will pay separately for these biosimilars even if their per-day cost is below the threshold packaging policy when their reference products are separately paid.
c. Invoice Drug Pricing for CY 2026
In the CY 2025 OPPS/ASC final rule with comment period (89 FR 94243 to 94244), we finalized that, for separately payable drugs or biologicals for which CMS does not provide a payment rate in Addendum B, which would indicate to MACs that CMS does not have pricing information (specifically, that ASP, WAC, AWP, and mean unit cost information is not available to determine a payment rate), MACs would calculate the payment based on provider invoices. The drug or biological invoice cost would be the net acquisition cost minus any rebates, chargebacks, or post-sale concessions. Before calculating an invoice-based payment amount, MACs would use the provider invoice to determine that: (a) the drug is not policy packaged; and (b) the per-day cost of the drug, biological, therapeutic radiopharmaceutical or diagnostic radiopharmaceutical is above the threshold packaging amount, as applicable. If both conditions are met, the MACs would use the provider invoice amount to set a payment rate for the separately payable drug, biological, or radiopharmaceutical until its payment amount becomes available to CMS. We generally expect invoice pricing to be temporary, lasting two to three quarters, for qualified drugs required to report ASP under section 1847A of the Act. For drug products that are not required to report ASP under section 1847A of the Act (that is, diagnostic pharmaceuticals), invoice pricing may be used on a longer-term basis until a MUC can be calculated. We finalized the invoice pricing policy for drugs to be effective January 1, 2026, with the intent to make technical updates to outpatient hospital claims and to allow providers time to prepare for any operational changes. We noted that the National Uniform Billing Committee (NUBC) created a value code that would allow for the reporting of invoice prices of drugs, biologicals, and radiopharmaceuticals for CY 2026 for the purpose of this policy. The NUBC value code created is 92 (Drug/Biologic Invoice Cost), with the definition of: “Invoice Cost of drug/biologic. For use with Revenue Category 0636 when required by federal regulation.” We proposed a technical clarification to this policy in the CY 2026 OPPS/ASC proposed rule. Previously, we stated that MACs would use the provider invoice to determine that: (1) the drug is not policy packaged; and (2) the per-day cost of the drug, biological, therapeutic radiopharmaceutical or diagnostic radiopharmaceutical is above the threshold packaging amount, as applicable. However, we proposed to clarify that CMS will determine whether the first condition is met, whether the drug is not policy packaged; however, the MAC will continue to determine whether the second condition is met, whether the per-day cost of the drug, biological, therapeutic radiopharmaceutical or diagnostic radiopharmaceutical is above threshold packaging amount, as applicable.
Comment: A commenter sought clarification regarding “invoice pricing”. The commenter stated that “invoice pricing” by nature does not reflect post sale adjustment and that the CMS text on this subject, “The drug or biological invoice cost would be the net acquisition cost minus any rebates, chargebacks, or post-sale concessions” may create confusion and result in unintended consequences.
Response: We thank the commenter for seeking clarification on this subject. The invoice pricing policy aims to enhance transparency and accuracy in the determination of payment rates for drugs and biologicals when CMS lacks pricing information. By using the net acquisition cost and accounting for various post-sale adjustments, this policy ensures that the payment rates reflect the true cost incurred by providers. Therefore, in the CY 2026 OPPS/ASC proposed rule, we defined “invoice pricing” to be net acquisition cost minus any rebates, chargebacks, or post-sale concessions. To illustrate the calculation of the net acquisition cost: if the acquisition cost of a unit of drug X is $10, and the rebate was $2. The net acquisition cost of drug X per unit is $8 ($10−$2). Therefore, the invoice price to submit to the MAC is $8 per unit.
After consideration of the public comments we received, we are implementing this policy without modification in CY 2026. We refer readers to the previous discussion when we finalized the policy in the CY 2025 OPPS/ASC final rule with comment period (89 FR 94243 to 94244). ( printed page 53705)
3. Payment Policy for Radiopharmaceuticals
For a complete history of the OPPS payment policy for radiopharmaceuticals, we refer readers to the CY 2005 OPPS final rule with comment period (69 FR 65811), the CY 2006 OPPS final rule with comment period (70 FR 68655), and the CY 2010 OPPS/ASC final rule with comment period (74 FR 60524).
We received public comments on our payment policy for radiopharmaceuticals in general. The following is a summary of the comments we received and our responses.
Comment: One commenter requested that CMS determine HCPCS specific radiopharmaceutical offsets and include preliminary nuclear medicine APC offset data with the proposed rule in order to inform public comments. Additionally, this commenter requested that CMS restore the radiopharmaceutical edits used in nuclear medicine procedures to increase the accuracy of claims data used to set rates for both separately payable diagnostic radiopharmaceuticals as well as the nuclear medicine APCs.
Response: We appreciate the commenter's feedback; however, we are not reinstating the radiolabeled product edits for nuclear medicine procedures, which required a diagnostic radiopharmaceutical to be present on the same claim as a nuclear medicine procedure for payment to be made under the OPPS. As previously discussed in the CY 2020 OPPS/ASC final rule with comment period (85 FR 86033 and 86034), the edits were in place between CY 2008 and CY 2014 (78 FR 75033). We believe the period of time in which the edits were in place was sufficient for hospitals to gain experience reporting procedures involving radiolabeled products and to become accustomed to ensuring that they code and report charges so that their claims fully and appropriately reflect the costs of those radiolabeled products. As with all other items and services recognized under the OPPS, we expect hospitals to code and report their costs appropriately, regardless of whether there are claims processing edits in place. We welcome ongoing dialogue and engagement from interested parties regarding suggestions for payment changes for consideration in future rulemaking. We will take into consideration additional files and data that may be helpful to readers as supplements to future notice and comment rulemaking.
a. Payment Policy for Therapeutic Radiopharmaceuticals
In the CY 2023 OPPS/ASC final rule with comment period, we adopted as final our proposal to continue our longstanding payment policy for therapeutic radiopharmaceuticals for CY 2023 and subsequent years. Accordingly, this payment policy for therapeutic radiopharmaceuticals will continue to apply in CY 2026.
Specifically, our policy of paying for separately payable pass-through therapeutic radiopharmaceuticals under the ASP methodology adopted for separately payable drugs and biologicals described in section V.A.1. of this CY 2026 OPPS/ASC final rule will continue to apply for CY 2026. We will pay for separately payable nonpass-through therapeutic radiopharmaceuticals through a modified ASP methodology where we pay at ASP plus 6 percent if ASP data are available. However, if ASP information is unavailable for a separately payable nonpass-through therapeutic radiopharmaceutical, we will continue to base the payment rate on arithmetic mean unit cost data derived from hospital claims. Our policy not to use WAC or AWP to establish payment for separately payable nonpass-through therapeutic radiopharmaceuticals if ASP is not available will continue for CY 2026. We explained our rationale in the CY 2010 OPPS/ASC final rule with comment period (74 FR 60524 through 60525) when we first adopted our policy to apply the principles of separately payable drug pricing to therapeutic radiopharmaceuticals.
For a full discussion of ASP-based payment for therapeutic radiopharmaceuticals, we refer readers to the CY 2010 OPPS/ASC final rule with comment period (74 FR 60520 through 60521). We will rely on CY 2024 mean unit cost data derived from hospital claims data for payment rates for separately payable nonpass-through therapeutic radiopharmaceuticals for which ASP data are unavailable and update the payment rates for these products according to our usual process for updating the payment rates for separately payable drugs and biologicals on a quarterly basis if updated ASP information becomes available.
The CY 2026 payment rates for separately payable nonpass-through therapeutic radiopharmaceuticals are included in Addenda A and B of this final rule with comment period (which are available on the CMS website).[75]
Though we made no proposal regarding payment for therapeutic radiopharmaceuticals, we received public comments that were generally in support of these policies. We thank the commenters for their support. We are continuing our policies unchanged for CY 2026.
b. Payment Policy for Diagnostic Radiopharmaceuticals Without Claims Data
For CY 2025, we finalized, as described in the CY 2025 OPPS/ASC final rule (89 FR 93948 through 93963), to pay separately at arithmetic mean unit cost for diagnostic radiopharmaceuticals with a per day cost above our diagnostic radiopharmaceutical packaging threshold ($655 for CY 2026). We also finalized our policy to pay for pass-through diagnostic radiopharmaceuticals based on ASP, WAC, and AWP.
We continue to believe that paying for nonpass-through diagnostic radiopharmaceuticals using arithmetic mean unit cost would appropriately pay for the average price of a nonpass-through separately payable diagnostic radiopharmaceutical, as discussed in section II.A.3.c of this final rule with comment period. In our view, MUC is an appropriate proxy for the average price for a diagnostic radiopharmaceutical for a given year, as it is calculated based on the average costs for a particular year and is directly reflective of the actual cost data that hospitals submit to CMS. As we stated in the CY 2010 OPPS/ASC final rule with comment period (74 FR 60523), we believe that WAC or AWP is not an appropriate proxy to provide OPPS payment for radiopharmaceuticals because these pricing methodologies do not include discounts. Specifically, the absence of appropriate ASP reporting could result in payment for a separately payable diagnostic radiopharmaceutical based on WAC or AWP indefinitely, a result which we believe would be inappropriate, as these pricing metrics do not capture all of the pricing discounts that may be reflected in the ASP.
Additionally, in the CY 2025 OPPS/ASC final rule with comment period (89 FR 93948 through 93963), we finalized to base the initial payment for new diagnostic radiopharmaceuticals with HCPCS codes that do not have pass-through status or claims data on ASP, and on the WAC for these products if ASP data for these diagnostic radiopharmaceuticals are not available. To further clarify, these products will be paid based on ASP plus 6 percent, and ( printed page 53706) at WAC plus 3 or 6 percent according to the policy in section V.B.2.a. of this final rule with comment period if ASP data are not available.
If the WAC also is unavailable, we proposed to make payment for new diagnostic radiopharmaceuticals at 95 percent of the products' most recent AWP. We believe the volume of products in this category will typically be very low; however, in these rare situations, we believe it would be appropriate to use ASP, WAC, or AWP until a MUC is established for new diagnostic radiopharmaceuticals with HCPCS codes that do not have passthrough status or claims data.
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: Commenters were generally supportive of our policy regarding payment for non-passthrough diagnostic radiopharmaceuticals without claims data. While commenters had suggestions regarding payment for diagnostic radiopharmaceuticals with claims data and while on pass-through status, commenters generally supported the policies to pay for diagnostic radiopharmaceuticals without claims data according to the existing methodology.
Response: We thank the commenters for their support.
After consideration of public comments, we are continuing our policy, without modification, to base the initial payment for new diagnostic radiopharmaceuticals with HCPCS codes that do not have pass-through status or claims data on ASP, at ASP plus 6 percent, and on the WAC for these products if ASP data for these diagnostic radiopharmaceuticals are not available. To further clarify, these products will be paid based on ASP plus 6 percent, and at WAC plus 3 or 6 percent according to the policy in section V.B.2.a. of this final rule with comment period if ASP data are not available.
If the WAC also is unavailable, payment for new diagnostic radiopharmaceuticals will be made at 95 percent of the products' most recent AWP. We believe the volume of products in this category will typically be very low; however, in these rare situations, we believe it would be appropriate to use ASP, WAC, or AWP until a MUC is established for new diagnostic radiopharmaceuticals with HCPCS codes that do not have passthrough status or claims data.
Please refer to section II.A.3.c of this final rule with comment period for information regarding our broader payment policies for diagnostic radiopharmaceuticals, including our policy to pay separately for diagnostic radiopharmaceuticals above a certain cost threshold, our policy to pay for separately payable diagnostic radiopharmaceuticals with claims data based on mean unit cost data derived from hospital claims, and a list of the final qualifying diagnostic radiopharmaceuticals with per day costs exceeding the $655 threshold for CY 2026 in Table 7. The final CY 2026 payment rates for separately payable nonpass-through diagnostic radiopharmaceuticals are included in Addenda A and B of this final rule with comment period (which are available on the CMS website).[76]
4. Payment for Blood Clotting Factors
For CY 2026, we proposed to continue our established policy to provide payment for blood clotting factors using the same methodology as other separately payable drugs and biologicals under the OPPS and to continue to pay a furnishing fee. For a full discussion of our established payment policy for blood clotting factors, please refer to the CY 2023 OPPS/ASC final rule with comment period (87 FR 71969 through 71970). In accordance with our policy as finalized in the CY 2008 OPPS/ASC final rule with comment period (72 FR 66765), we will announce the actual figure of the percent change in the applicable CPI and the updated furnishing fee calculation based on that figure through the applicable program instructions and posting on the CMS website at https://www.cms.gov/medicare/payment/fee-for-service-providers/part-b-drugs/average-drug-sales-price.
Comment: Commenters supported our proposal to continue to provide payment for the for blood clotting factors using the same methodology as other separately payable drugs and biologicals under the OPPS and to continue to pay a furnishing fee.
Response: We thank the commenters for their support of our policy.
After consideration of public comments, we are finalizing our proposal without modification to pay for clotting factors using the same methodology as other separately payable drugs and biologicals under the OPPS, and to provide a clotting factor furnishing fee. For CY 2026, the blood clotting factor furnishing fee is $0.265. The actual percentage change in the applicable CPI and the updated furnishing fee calculation based on that figure through the applicable program instructions are posted on the CMS website.[77]
5. Payment for Nonpass-Through Drugs, Biologicals, and Radiopharmaceuticals With HCPCS Codes But Without OPPS Hospital Claims Data
In the CY 2023 OPPS/ASC final rule with comment period, we adopted as final our proposal to continue our longstanding payment policy for nonpass-through drugs, biologicals, and radiopharmaceuticals with HCPCS codes but without OPPS hospital claims data for CY 2023 and subsequent years. Therefore, for CY 2026, this policy will continue to apply. For a detailed discussion of the payment policy and methodology, we refer readers to the CY 2016 OPPS/ASC final rule with comment period (80 FR 70442 through 70443). Consistent with our policy, because we have no claims data and must determine if these products, drugs, biologicals, therapeutic radiopharmaceuticals, and diagnostic radiopharmaceuticals, exceed the per-day cost threshold, we estimated the average number of units of each product that would typically be furnished to a patient during one day in the hospital outpatient setting and utilized the payment rate for the product, typically the ASP methodology, to determine whether their payment will be packaged as well as their payment status indicators.
We did not make a proposal on this policy. This policy did not receive public comments on this provision. We are continuing this policy unchanged for CY 2026.
6. Requirement in the CY 2026 PFS Proposed Rule for HOPDs and ASCs To Report Discarded Amounts of Certain Single-Dose or Single-Use Package Drugs
Section 90004 of the Infrastructure Investment and Jobs Act (Pub. L. 117-9, November 15, 2021) (“the Infrastructure Act”) amended section 1847A of the Act to re-designate subsection (h) as subsection (i) and insert a new subsection (h), which requires manufacturers to provide a refund to CMS for certain discarded amounts from a refundable single-dose container or single-use package drug. We explained in the CY 2026 OPPS/ASC proposed rule (90 FR 33631) that the CY 2026 Physician Fee Schedule (PFS) proposed rule (90 FR 32538 through 32540) includes proposals related to the discarded drug refund ( printed page 53707) policy, including proposals that may impact hospital outpatient departments (HOPDs) and ambulatory surgical centers (ASCs). Similar to our past notices in OPPS/ASC proposed rules, such as in the CY 2025 OPPS/ASC proposed rule (89 FR 59370), we wanted to ensure interested parties were aware of these proposals and knew to refer to the CY 2026 PFS proposed rule for a full description of the proposed policy. Interested parties were asked to submit comments on any proposals to implement section 90004 of the Infrastructure Act to the CY 2026 PFS proposed rule. We noted that public comments on these proposals would be addressed in the CY 2026 PFS final rule with comment period.
We refer readers to the CY 2026 PFS final rule with comment period for a summary of comments, our responses, and the finalized policy for CY 2026.
7. CY 2026 Prospective Adjustment to Payments for Non-Drug Items and Services To Offset the Increased Payments for Non-Drug Items and Services Made in CY 2018 Through CY 2022 as a Result of the 340B Payment Policy
a. Overview
Under the OPPS, we generally set payment rates for separately payable drugs, and biologicals (hereinafter referred to collectively as “drugs” in this section) under section 1833(t)(14)(A) of the Act). Section 1833(t)(14)(A)(iii)(II) of the Act provides that, if hospital acquisition cost data are not available, the payment amount is the average price for the drug in a year established under sections 1842(o), 1847A, or 1847B of the Act, as the case may be. Payment rates for drugs have usually been established under section 1847A of the Act, which generally sets a default rate of the average sales price (ASP) plus 6 percent. Section 1833(t)(14)(A)(iii)(II) of the Act also provides that the average price for the drug in the year as established under section 1847A of the Act, is calculated and adjusted by the Secretary as necessary for purposes of paragraph (14).
In the CY 2018 OPPS/ASC final rule with comment period (82 FR 59353 through 59371), CMS reexamined the appropriateness of paying the ASP plus 6 percent for drugs acquired through the 340B Drug Pricing Program (hereinafter referred to as the “340B Program”), a Health Resources and Services Administration (HRSA)-administered program that allows covered entities to purchase certain covered outpatient drugs at discounted prices from drug manufacturers. Based on findings of the Government Accountability Office (GAO),[78] the HHS Office of the Inspector General (OIG),[79] and the Medicare Payment Advisory Commission (MedPAC) [80] that 340B hospitals were acquiring drugs at a significant discount under the 340B Program, CMS adopted a policy beginning in 2018 generally to pay an adjusted amount of ASP minus 22.5 percent for certain separately payable drugs or biologicals acquired through the 340B Program. This adjustment amount was based on our concurrence with an analysis by MedPAC that concluded that the estimated average minimum discount of 22.5 percent of ASP adequately represented the average minimum discount that a 340B participating hospital received for separately payable drugs under the OPPS (82 FR 59354 through 59371). Our intent in implementing this payment reduction was to reflect more accurately the actual costs incurred by participating hospitals in acquiring 340B drugs. We stated our belief that such changes would allow Medicare beneficiaries and the Medicare program to pay a more appropriate amount when hospitals participating in the 340B Program furnished drugs to Medicare beneficiaries that were purchased under the 340B Program (82 FR 59353 through 59371).
b. Payment for 340B Drugs and Biologicals in CYs 2018 Through 2022
From January 1, 2018 through September 27, 2022, under the OPPS we generally paid for certain separately payable drugs acquired through the 340B Program at ASP minus 22.5 percent. In the CY 2018 OPPS/ASC final rule with comment period (82 FR 59369 through 59370), we finalized our proposal to adjust the payment rate for separately payable drugs (other than drugs with pass-through payment status and vaccines) acquired under the 340B Program from ASP plus 6 percent to ASP minus 22.5 percent. We also noted that critical access hospitals are not paid under the OPPS and therefore were not subject to the OPPS 340B drug payment adjustment policy. For ease of reference, the OPPS 340B drug payment adjustment policy is hereinafter referred to as the “340B Payment Policy” and refers both to the adjustments made to payment rates for 340B-acquired drugs described here and the corresponding rate adjustment for non-drug services and items described later in section V.B.7.c. of this final rule with comment period. We note that rural sole community hospitals, children's hospitals, and PPS-exempt cancer hospitals were exempted from the adjustments made to payment rates for 340B-acquired drugs primarily due to these hospitals receiving special payment adjustments under the OPPS. In addition, as stated in the CY 2018 OPPS/ASC final rule with comment period, this policy change did not apply to drugs with pass-through payment status, which are required to be paid based on the ASP methodology, or vaccines, which are excluded from the 340B Program.
In the CY 2019 OPPS/ASC final rule with comment period (83 FR 58981), we continued the Medicare 340B payment policies that were implemented in CY 2018 and adopted a policy to pay for non-pass-through 340B-acquired biosimilars at ASP minus 22.5 percent of the biosimilar's ASP, rather than the reference biological product's ASP. Additionally, in the CY 2019 OPPS/ASC final rule with comment period (83 FR 59015 through 59022), we finalized a policy to pay ASP minus 22.5 percent for 340B-acquired drugs furnished in non-exempted off-campus provider-based departments (PBDs) paid under the PFS. We adopted this payment policy for CY 2019 and subsequent years. Also, during the CY 2019 OPPS/ASC rulemaking cycle, we clarified that the 340B payment adjustment applied to drugs priced using either wholesale acquisition cost (WAC) or average wholesale price (AWP), and since the policy was first adopted, we applied the 340B payment adjustment to 340B-acquired drugs priced using these pricing methodologies. The 340B payment adjustment for WAC-priced drugs was WAC minus 22.5 percent. 340B-acquired drugs that were priced using AWP were paid an adjusted amount of 69.46 percent of AWP (83 FR 37125).[81]
( printed page 53708)As discussed further in section V.B.7.f. of the CY 2026 OPPS/ASC proposed rule, the results of this policy meant that hospitals received an estimated $10.6 billion less in 340B drug payments (including money that would have been paid by Medicare and money that would have come from beneficiaries as copayments) than they would have for drugs provided in CY 2018 through September 27th of 2022 had the 340B Payment Policy not been implemented (88 FR 77162). These reduced payments are detailed in Table 108 and are derived from Addendum AAA published with the Final Remedy rule (88 FR 77150).
For more detailed descriptions of our OPPS payment policy for drugs acquired under the 340B Program during this timeframe, we refer readers to the CY 2018 OPPS/ASC final rule with comment period (82 FR 59353 through 59371); the CY 2019 OPPS/ASC final rule with comment period (83 FR 59015 through 59022); the CY 2020 OPPS/ASC final rule with comment period (84 FR 61321 through 61327); the CY 2021 OPPS/ASC final rule with comment period (85 FR 86042 through 86055); the CY 2022 OPPS/ASC final rule with comment period (86 FR 63640 through 63649); the CY 2023 OPPS/ASC final rule with comment period (87 FR 71972 through 71973); and the CY 2024 OPPS/ASC final rule with comment period 88 FR 81789 through 81792).
c. Payment for Non-Drug Items and Services in CY 2018 Through CY 2022
In the CY 2018 OPPS/ASC final rule with comment period (82 FR 59216, 59258), to comply with the statutory budget neutrality requirements under sections 1833(t)(9)(B) and (t)(14)(H) of the Act, we finalized our proposal to redistribute our estimated reduction in payments for separately payable drugs as a result of the 340B Payment Policy by increasing the conversion factor used to determine the payment amounts for non-drug items and services. As further described in the CY 2018 OPPS/ASC final rule with comment period, we used updated CY 2016 claims data and a list of 340B-eligible providers to calculate an estimated impact of $1.6 billion based on the final CY 2018 policy to pay for OPPS 340B-acquired drugs at a payment rate of generally ASP minus 22.5 percent. To effectuate the budget neutrality provisions of the OPPS for CY 2018, we redistributed an estimated $1.6 billion in reduced drug payments from adoption of the final 340B payment methodology to all hospitals paid under the OPPS by increasing the payment rates by 3.19 percent for nondrug items and services furnished by all hospitals paid under the OPPS for CY 2018. We carried through this conversion factor adjustment from CYs 2019 through 2022, increasing payments for non-drug items and services in these CYs. This resulted in approximately $7.769 billion, which for ease of reference in this rule we hereafter refer to as $7.8 billion, in additional spending on non-drug items and services from CYs 2018 through 2022.
d. Litigation History of the 340B Payment Policy
The 340B Payment Policy was the subject of extensive litigation. See the Proposed Remedy for the 340B-Acquired Drug Payment Policy for Calendar Years 2018-2022 (hereinafter referred to as the “proposed remedy rule”) for a more comprehensive summary of the litigation history (88 FR 44079 through 44080).
On June 15, 2022, the Supreme Court held that because we had not conducted a survey of hospitals' acquisition costs, we could not vary the payment rates for outpatient prescription drugs by hospital group. See Am. Hosp. Ass'n v. Becerra, 142 S. Ct. 1896, 1906 (2022). The Supreme Court declined to opine on the appropriate remedy, id. at 1903, and on September 28, 2022, the district court vacated the reimbursement rate for 340B-acquired drugs for the remainder of 2022. See Am. Hosp. Ass'n v. Becerra, 1:18-cv-2084-RC, 2022 WL 4534617, at *5.[82] On January 10, 2023, the district court remanded without vacatur to give the agency the opportunity to determine the proper remedy for the reduced payment amounts to 340B hospitals under the payment rates in the final OPPS rules for CY 2018 through CY 2022. See Am. Hospital Ass'n v. Becerra, 1:18-cv-2084-RC, 2023 WL 143337, at *6.[83]
e. Payment for 340B-Acquired Drug Claims for September 28, 2022 Through CY 2025
The agency complied with the district court's September 28, 2022 decision by uploading revised OPPS drug files to pay the default rate (generally ASP plus 6 percent) for all CY 2022 claims for 340B-acquired drugs paid from September 28, 2022, through the end of CY 2022.
In the CY 2023 OPPS/ASC final rule with comment period (87 FR 71970), we finalized a policy reversing the 340B Payment Policy so that going forward we would pay for 340B acquired drugs no differently than we pay for drugs that are not acquired through the 340B program. To do so, we first provided that drugs acquired through the 340B Program would be paid at the statutory default rate (generally ASP plus 6 ( printed page 53709) percent) for CY 2023. Second, to ensure budget neutrality for CY 2023 OPPS payment rates as required by statute, we finalized a reduction of 3.09 percent to the 2023 OPPS conversion factor. This one-time adjustment to the conversion factor removed the effect of this aspect of the 340B Payment Policy, as originally adopted in CY 2018, for CY 2023 and subsequent years. This adjustment to the conversion factor reduced the conversion factor to the conversion factor that would have been in place in CY 2023 if the 340B payment policy had never been implemented. For more detail on the payment rate for drugs acquired under the 340B Program for CY 2023 and the corresponding adjustment to the conversion factor to maintain budget neutrality as a result of reversing the 340B adjustment and paying for all separately payable drugs at ASP plus 6 percent (or WAC plus 3 or 6 percent or 95 percent of AWP), we refer readers to the CY 2023 OPPS/ASC final rule with comment period (87 FR 71973 through 71976).
For CYs 2024 and 2025, consistent with our policy finalized for CY 2023, we continued to pay the statutory default rate for 340B acquired drugs (88 FR 81789 through 81791).
f. Remedy Payment Adjustment for 340B-Acquired Drugs From CY 2018 Through September 27, 2022
The agency complied with the district court's January 10, 2023, remand order by issuing the Final Remedy for the 340B-Acquired Drug Payment Policy for Calendar Years 2018-2022 (hereinafter referred to as the “Final Remedy rule”) on November 8, 2023 (88 FR 81540). The purpose of this rule was to address the reduced payment amounts to 340B hospitals under the reimbursement rates in effect for CY 2018 through September 27, 2022 and to comply with the statutory requirement to maintain budget neutrality under the OPPS.
To address the reduced payment amounts to 340B hospitals under the reimbursement rates in effect for CY 2018 through September 27, 2022, CMS made one-time lump sum payments to affected 340B covered entity hospitals, calculated as the difference between what an affected 340B covered entity hospital received for 340B-acquired drugs from CY 2018 through September 27, 2022 and what they would have received for those drugs if the 340B adjustment had not been in place. These one-time lump sum payments were issued in early 2024. For more information on the calculation and distribution of the one-time lump sum payments, see the Final Remedy rule (88 FR 77156 through 77170).
g. Prospective Adjustment to Payments for Non-Drug Items and Services To Offset the Increased Payments for Non-Drug Items and Services Made in CY 2018 Through CY 2022
As we described under section V.B.7.c. of the CY 2026 OPPS/ASC proposed rule and section I.A.3 of the Final Remedy rule, to comply with statutory budget neutrality requirements, the decreased payments made to 340B hospitals for drugs in CY 2018 through September 27, 2022 were budget neutralized by corresponding increased payments to all hospitals for non-drug items and services starting in CY 2018 through CY 2022. When these past payments were subsequently increased through the one-time lump sum payments in 2024, the same budget neutrality requirements correspondingly required us to decrease the non-drug item and services payments made from CY 2018 through CY 2022.
To reduce the burden on providers of immediately offsetting the estimated $7.8 billion of increased non-drug item and services payments made from CY 2018 through CY 2022, we decided to implement the offset prospectively over the course of several years. As we explained in the CY 2026 OPPS/ASC proposed and Final Remedy rules (88 FR 44088, 88 FR 77172), this approach was similar to the original budget neutrality adjustment in the 340B Payment Policy that increased the payment for every non-drug item and service for CY 2018 through CY 2022 to offset the downward adjustment in the payment rate for drugs acquired under the 340B Program. We finalized in the Final Remedy rule that, beginning in CY 2026, we would reduce the conversion factor for non-drug items and services to all OPPS providers—except any hospital that enrolled in Medicare after January 1, 2018 (as described further below)—by 0.5 percent each year until the total offset was reached (which we estimated would take approximately 16 years (88 FR 77181)).
As we stated in the CY 2026 OPPS/ASC proposed and Final Remedy rule, we believed an annual reduction in the conversion factor would be appropriate because it would balance the need to address the past payments for non-drug items and services to ensure budget neutrality while also ensuring that the offset was not immediately overly financially burdensome on impacted entities, which we believed would be the case if we were to apply an adjustment for the full offset amount in a single year. (88 FR 44087, 88 FR 77170).
Accordingly, the Final Remedy rule finalized changes to the calculation of the OPPS conversion factor applicable to non-drug items and services beginning in CY 2026. Specifically, we codified a 0.5 percent reduction in the OPPS conversion factor applicable to non-drug items and services in the regulations by adding new paragraph (b)(1)(iv)(B)( 12) to § 419.32. This 0.5 percent reduction would remain in effect until the estimated payment reduction reached $7.8 billion, which we estimated would occur in CY 2041. For a fuller discussion of the CY 2026 adjustment to the conversion factor for non-drug items and services in the Final Remedy rule, see the Final Remedy rule (88 FR 77156 through 77170).
In finalizing our policy to apply a prospective adjustment, we recognized that any hospital that enrolled in Medicare after January 1, 2018 (hereinafter referred to as a “new provider”) received less than the full amount of the increased non-drug item and service payments made during that time than they otherwise would have received if enrolled prior to that date (88 FR 44080). We therefore exempted these providers from the prospective rate reduction, which was predominantly designed to account for non-drug item and service payments made during CY 2018 through CY 2022. As we explained, that meant that we would calculate payment rates for new providers using the conversion factor before applying the 0.5 percent annual reduction to the conversion factor for non-drug items and services that would apply for hospitals that are not “new providers” for purposes of this policy. For the purpose of designating a new provider, we defined the date of enrollment in Medicare as the provider's CMS certification number (CCN) effective date. We codified the exclusion of these new providers from the prospective payment adjustment to the conversion factor for the duration of its application in the regulations by adding new paragraph (b)(1)(iv)(B)( 12) to § 419.32.
In the CY 2026 OPPS/ASC proposed rule we indicated that we had reviewed our provider enrollment and OPPS billing records, and based on that data, the providers that would be subject to the proposed payment reduction were listed in Addendum R—340B Remedy Offset Providers to the CY 2026 OPPS/ASC proposed rule. We welcomed comment on the providers listed in this Addendum, and based upon those comments, we proposed to publish a final Addendum R—340B Remedy Offset Providers for CY 2026 in the CY 2026 OPPS/ASC final rule with ( printed page 53710) comment period. We indicated in the CY 2026 OPPS/ASC proposed rule that providers not included on this list (providers that began billing Medicare under the OPPS after January 1, 2018) would not be subject to the proposed payment reduction. For a complete discussion of our exclusion of new providers from the prospective payment adjustment, we refer readers to the Final Remedy rule (88 FR 77182 through 77185).
We did not receive public comments on Addendum R—List of Providers Subject to the Reduction to Non-Drug Item and Service Payments as a Result of the 340B Payment Policy Remedy, and therefore, we are finalizing as proposed.
h. CY 2026 Prospective Payment Adjustment
When we considered how to recover the estimated $7.8 billion in increased estimated payments made for non-drug items and services from 2018 through 2022, we considered several alternatives, including those that would fully recover that amount in a single year. For example, in the Proposed Remedy rule, we rejected an aggregate payment approach that would have implemented budget neutrality requirements through an immediate lump sum recoupment that would mirror the lump sum remedy payment because “[s]uch an approach would require immediate, and in many cases large, retroactive recoupments from the majority of OPPS hospitals and would impose a substantial, immediate burden on these hospitals as well as an uncertain impact on beneficiaries” (88 FR 44083). To avoid imposing such a burden, we elected to reduce payments prospectively until the total offset was reached, which we estimated would take approximately 16 years.
As we discussed in the CY 2026 OPPS/ASC proposed rule, we considered various methods to implement this prospective payment reduction. In the Final Remedy rule, we made the prospective payment reduction by applying an annual 0.5-percentage point downward adjustment to the OPPS conversion factor. We stated in the CY 2026 OPPS/ASC proposed rule that we continued to believe that a downward adjustment to the OPPS conversion factor was a fair way to apportion the $7.8 billion reduction amongst hospitals, because relative hospital utilization of non-drug items and services beginning in 2026 would approximately track the relative hospital utilization for non-drug items and services each hospital received from CY 2018 through CY 2022. We stated in the CY 2026 OPPS/ASC proposed rule that the future payment reductions would thus roughly offset the windfall those hospitals received from increased payments from CY 2018 through CY 2022. And we noted our statement in the final rule that the approach of tethering future payments for each non-drug item and service for each hospital “was similar to the original budget neutrality adjustment in the 340B Payment Policy that increased the payment for every non-drug item and service for CY 2018 through CY 2022 to offset the downward adjustment in the payment rate for drugs acquired under the 340B Program” (88 FR 77172). Finally, the methodology does so with minimal administrative burden to hospitals and beneficiaries, because we can effectuate the offset by calculating the appropriate payment reduction in annual rulemaking without requiring any subsequent action by hospitals. Other methodologies—like delivering a series of demand letters to each hospital for a share of the $7.8 billion—would not only require us to recalculate the proper amount to apportion to each hospital but would most likely require large lump-sum payments from hospitals. We expressed concern that hospitals might find it financially disruptive to promptly write such one-time checks depending on their financial circumstances when we issue the demand letters, whereas implementing a percentage reduction in their Medicare OPPS payments over a number of years would be less disruptive. Such one-time payments would impose greater administrative burden on hospitals and possibly introduce complications to our collections efforts if hospitals delay payments.
We stated in the CY 2026 OPPS/ASC proposed rule that while we continued to believe that a reduction to the OPPS conversion factor was the best way to effectuate budget neutrality, we had reconsidered whether the timing we selected—a 0.5-percentage point annual reduction for approximately 16 years—best achieved the overarching goal of the Final Remedy rule, which is to restore hospitals to as close to the financial position they would have been in had the 340B Payment Policy never been implemented as is reasonably feasible. In particular, we indicated that the further away from CY 2018 through CY 2022 the adjustments extend, the less likely that relative hospital utilization of non-drug items and services would correlate to the relative hospital utilization of non-drug items and services from 2018 through 2022. In other words, a hospital's utilization of non-drug items and services is likely going to diverge more from CY 2018 utilization in CY 2041 than it would in CY 2031 or CY 2026. And the more a hospital's utilization of non-drug items and services diverge, the less hospitals would be restored to as close as possible to the approximate financial position as they would have been in had the 340B Payment Policy never been implemented. By beginning the decrease to non-drug item and service payments in CY 2026, there is already an 8-year delay between the first year of the OPPS 340B payment policy and the first year of the prospective offset. Thus, the longer it takes for us to fully recover the $7.8 billion, the less likely that the relative burden on hospitals from the adjustments will match the relevant benefits those hospitals previously received. In addition, it is possible that at least some hospitals that benefited from the increased payments from CY 2018 through CY 2022 will leave the market before 2041, increasing the risk that the remaining hospitals might ultimately account for a larger share of the payment reductions than they would have if the annual reduction to the OPPS conversion factor concluded sooner. We noted that the $7.8 billion dollar figure calculated in the Final Remedy rule (88 FR 77150) does not and will not account for inflation and does not contain interest even though the prospective offset is occurring many years after both the start of the 340B payment policy in CY 2018 as well as the lump sum remedy payments made in CY 2024.
Accordingly, effective January 1, 2026, we proposed to revise the annual reduction to the OPPS conversion factor under § 419.32(b)(1)(iv)(B)( 12) used to determine the payment amounts for non-drug items and services from 0.5 percent to 2 percent. Under this revised rate, we expected it would take approximately 6 years to reach the total offset of $7.8 billion (see Table 62 in the CY 2026 OPPS/ASC proposed rule (90 FR 33636)). Consistent with the Final Remedy rule, we noted, this reduction would not apply to new providers. We also included on Table 62 in the CY 2026 OPPS/ASC proposed rule, and Table 109 in this final rule with comment period, an alternative policy option with an annual reduction of 5 percent which would reach the total offset of $7.8 billion in approximately 3 years.
We acknowledged that this revised annual reduction would be a change to the approach we finalized in the Final Remedy rule and that, at that time, we considered but did not adopt a ( printed page 53711) suggestion from a commenter requesting that we recover the amount over a shorter timeframe than 16 years. (88 FR 77179.) We indicated that our basis for not accepting the suggestion in the Final Remedy rule was that the 0.5 percent rate/16-year timeframe “properly reverses the increased payments for non-drug items and services to comply with statutory budget neutrality requirements while at the same time accounting for any reliance interests and ensuring that the offset is not overly burdensome to impacted entities”. We stated in the CY 2026 OPPS/ASC proposed rule that we now thought that this balancing insufficiently accounted for the main premise of the Final Remedy rule, which is to implement the budget neutrality requirement in a manner that restores affected 340B covered entity hospitals to the financial position they would have been in had the 340B Payment Policy not been implemented in 2018. For the reasons explained in the CY 2026 OPPS/ASC proposed rule, we believed that a 6-year time frame better achieved that main goal. We also stated that we believed this time frame balanced better that goal and our budget neutrality obligations against hospital burden and reliance interests. We provided as an example that the 16-year timeframe is more than three times longer than the 5-year period the 340B Payment Policy was in place. The 6 years we expect that the revised policy would be in effect, by contrast, is closer to the timeframe the 340B Payment Policy was in place, and the 2 percent payment reduction we proposed is still well below the 3.19 percent payment increase hospitals received for that time period (82 FR 52624 through 52625). We also stated that because we proposed this policy in advance of CY 2026 and before any rate reductions go into effect for OPPS and Medicare Fee for Service payments, any reliance interests hospitals have in a policy that has not been implemented yet for these payment systems would be minimal and outweighed by the other considerations discussed in the CY 2026 OPPS/ASC proposed rule.
( printed page 53712)We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: Most commenters opposed our proposal. Many of these commenters referenced previous comments they or others made to the Final Remedy rule alleging that CMS lacks the statutory authority to budget neutralize the remedy and that CMS' approach to budget neutrality is inconsistent with its past practices. Based on these reasons, these commenters requested that we abandon the proposal and rescind the regulation codified in that rule, 42 CFR 419.32(b)(1)(iv)(B)( 12) to implement the offset.
Response: We direct readers to our responses to those comments in the ( printed page 53713) Final Remedy rule at 88 FR 77150 through 77194.
Comment: One commenter stated that CMS does not have the legal authority to apply an additional 2.0-percentage point reduction to OPPS rates because, under section 1833(t)(3)(C)(iv) of the Act, CMS is required to update OPPS rates by the same update that applied under the IPPS. Since CMS has already finalized an update of 2.6 percent for the FY 2026 IPPS, the commenter stated that CMS must apply the same update to the OPPS for CY 2026 without further changes.
Response: We disagree with the commenter, who conflates two distinct statutory obligations. Under section 1833(t)(3)(C) of the Act, the Secretary updates the conversion factor used to determine the payment rates under the OPPS on an annual basis by applying the OPD fee schedule increase factor. The commenter is correct that we have implemented that fee schedule increase factor under section 1833(t)(3)(C)(iv) of the Act by applying the hospital inpatient market basket percentage increase applicable to hospital discharges under section 1886(b)(3)(B)(iii) of the Act, subject to sections 1833(t)(17) and (t)(3)(F) of the Act. As we explain in section XXVI. of this final rule with comment period, we continue to do so by increasing the OPD fee schedule for CY 2026 by a factor of 2.6 percent—the same as applicable to hospital discharges for fiscal year 2026 under section 1886(b)(3)(B)(iii) of the Act.
The commenter is incorrect, however, that the statute makes the fee schedule increase factor the only update the Secretary can make to the OPPS conversion factor each year. Section 1833(t)(3)(C)(iii) of the Act, for example, authorizes the Secretary further to modify the conversion factor in certain circumstances based on adjustments to service mix. And many parts of the OPPS statute require the Secretary to budget neutralize particular payments, including sections 1833(t)(2)(E), (t)(9)(B), and (t)(14)(H) of the Act. Since the very first payment rule implementing OPPS, we have implemented OPPS's budget neutrality requirements by adjusting the conversion factor. (65 FR 18476 (implementing budget-neutrality requirement)). We have long rejected the commenter's suggestion that section 1833(t)(3)(C)(iv) of the Act means that the conversion factor cannot account for these budget neutrality requirements, explaining that the “[s]tatute requires us to ensure that a conversion factor for covered OPD services in subsequent years is an amount equal to the conversion factor applicable to the previous year before any increases due to the market-basket increase.” (67 FR 66788.) We accordingly applied that same policy when we first decreased payments for 340B-acquired drugs in the CY 2018 OPPS/ASC final rule with comment period, which we budget neutralized through an upward adjustment of 1.0319 to the OPPS conversion factor on top of the annual OPD fee schedule increase factor under 1833(t)(3)(C)(iv) of the Act. (82 FR 59353 through 59371). Even on the commenter's statutory theory, then, that increase—in place from CY 2018 through CY 2022—was itself unlawful and so should be repaid.
Comment: One commenter stated that the statute does not authorize CMS to impose what the commenter characterized as a compressed repayment schedule or heightened offset and disagreed with CMS' stated justification for it, which the commenter characterizes as “that a shorter offset period is preferable because it more closely aligns with the duration of the unlawful 340B payment policy”. The commenter contended that the statute does not authorize CMS to “calibrate budget neutrality offsets based on temporal symmetry or administrative convenience” and that such an approach ignores the disproportionate impact that a 2 percent annual reduction will have on hospitals that did not receive full remedy payments, including those with high Medicare Advantage penetration or limited exposure to the original 340B cuts.
Response: We disagree with the commenter that the statute prohibits the proposed budget neutrality adjustment schedule or amount, or that our rationale for that proposed schedule is flawed based on hospitals' reduced payments for drug acquired under the 340B program from CY 2018 through 2022. As we explained in the CY 2026 OPPS/ASC proposed rule, the main reason we proposed to shorten the timeframe for the adjustment was to recognize that OPPS utilization changes over time. As we have explained, the purpose of any rate reduction is to unwind the 3.19 percent increase budget neutrality hospitals received from CY 2018 through 2022. (For example, 88 FR 77170.) Current OPPS utilization for a hospital is a reasonable proxy for past OPPS utilization for a hospital, but it becomes a less accurate proxy the longer the recoupment timeframe. The commenter is wrong to suggest that our proposed policy would pursue symmetry for symmetry's sake. By pointing out that adjusting rates by 2 percent for 6 years is similar to the initial policy of adjusting rates by 3.19 percent for 4 years, we meant to illustrate both that the proposed larger decrease would still be the type of “adjustment” to payment rates authorized by sections 1833(t)(2)(E), (9)(B), and (14)(H) of the Act (88 FR 77158 through 77159) and that the reduction would still fall within the type of year-to-year rate fluctuations in OPPS rates that hospitals should reasonably expect in annual ratemaking. Nor was it inherently unreasonable for us to consider how long it will take fully to implement the adjustment; no statute requires us to implement our budget neutrality obligations so that a policy in place only from CY 2018 through 2022 still drives payment rates in 2040. Finally, the commenter is correct that not all hospitals received 340B remedy payments and might have been impacted differently by the payment reductions for 340B-acquired drugs from CY 2018 through 2022. But the commenter is wrong that we should treat those hospitals differently here. We accounted for the fact that some hospitals received payment reductions for 340B-acquired drugs from CY 2018 through 2022 by making remedy payment to those hospitals. By contrast, and as noted above, the payment cuts in 42 CFR 419.32 (b)(1)(iv)(B)( 12) unwinds the 3.19 percent budget neutrality increase hospitals received from CY 2018 through 2022. We are applying this cut only to the hospitals who received the full increase. Because hospitals received the 3.19 percent increase from CY 2018 through 2022 regardless of whether they also were paid less for 340B-acquired drugs, we disagree that whether hospitals were paid less for 340B-acquired drugs is relevant here.
Comment: Many commenters noted that in the Final Remedy rule we rejected annual percent reductions greater than 0.5 based on our conclusion that a 0.5 percent reduction “properly reverses the increased payments for non-drug items and services to comply with statutory budget neutrality requirements while at the same time accounting for any reliance interests and ensuring that the offset is not overly burdensome to impacted entities”. (88 FR 77179). These commenters critiqued our subsequent determination in the CY 2026 OPPS/ASC proposed rule that the 0.5 percent reduction “insufficiently accounted for the main premise of the Final Remedy rule which is to implement the budget neutrality requirement in a manner that restores affected 340B covered entity hospitals to the financial position they would have been in had the 340B Payment Policy ( printed page 53714) not been implemented in 2018” and our conclusion that “a 6 year time frame better achieves that main goal” and “balances better that goal and our budget neutrality obligations against hospital burden and reliance interests”. These commenters stated that CMS does not sufficiently explain how it reassessed the relationship between budget neutrality obligations, hospital reliance interests, and the financial burden on providers and that CMS has attributed insufficient weight to the reliance interest and hospital burden side of the equation. One commenter accused CMS of a “bait-and-switch” because it changed its position after initially rejecting the proposal for a quicker recoupment and faults CMS for not offering any new facts or circumstances not known to CMS when it published the Final Remedy rule that would warrant a change in policy. That commenter suggested that CMS failed to provide a rational basis for its position change, because another policy best achieves CMS' stated goal: reprocessing of all claims for the period in which the unlawful policy was in effect.
Commenters also raised several alleged reliance interests. They emphasized that we finalized the 0.5 percent reduction in the Final Remedy rule and that they have relied on that amount in good faith in the nearly 2 years since to engage in financial planning and long-term investment decisions. These commenters challenged CMS' contention that any reliance interests hospitals have in a policy that has not been implemented yet would be minimal because the proposal to increase the offset was made in advance of CY 2026 and before any rate reductions go into effect for OPPS and Medicare Fee for Service payments. These commenters stated that this fails to account for the long-term, multi-year nature of hospital budgeting and they described the many expenditures (opening new facilities, buying new medical equipment, hiring staff and expanding services lines, etc.) they have allegedly made based on the 0.5 percent reduction. Commenters maintained that CMS recognized their reliance interests in the Final Remedy rule when it delayed the start of the offset to 2026 so that hospitals could “assess and prepare for the new payment rates that will be calculated using a reduced conversion factor”. One commenter stated that the reliance interest discussed in the Final Remedy rule is “clearly the reliance interest on continued, stable payments in the OPPS” and that CMS took this interest into account by deciding not to impose the 0.5 percent reduction until 2026 “allowing adequate time for impacted parties to assess and prepare for the new payment rates that will be calculated using a reduced conversion factor”. The commenter contended that the CY 2026 OPPS/ASC proposed rule not only entirely fails to address hospitals' reliance interest as envisioned in the Final Remedy rule but also undermines the separate reliance interests that CMS allowed to be built from the date of the Final Remedy rule's publication to the CY 2026 OPPS/ASC rule's publication. The commenter stated that the budget process for CY 2026 is already materially complete and a roughly 2-month planning period (assuming a November OPPS/ASC final rule with comment period release) is simply not reasonable and would have drastic consequences without the ability to plan or adjust. Another commenter argued that CMS cannot ignore these considerations, “which informed CMS' choice in 2023 to adopt a lengthy recovery period and to extend the implementation of even a 0.5 percent reduction from 2025 to 2026, giving hospitals over 2 years to prepare.”
Response: While we disagree with many of the arguments these commenters raise, we are persuaded by the commenters to the extent that we will not finalize in CY 2026 our proposal to increase to 2 percent the 0.5 percent adjustment in 42 CFR 419.32(b)(1)(iv)(B)( 12). We currently anticipate delaying a change for just 1 year. Thus, while we will retain the original 0.5 percent adjustment in the conversion factor in CY 2026, hospitals should anticipate that we will implement a larger adjustment (such as 2 percent or other adjustment greater than 0.5 percent) beginning in CY 2027. Any change to the adjustment in 42 CFR 419.32(b)(1)(iv)(B)( 12) that applies beginning in CY 2027 would go through the usual annual rulemaking process.
We do so based on the unique circumstances here. We finalized outside the standard annual rulemaking cycle a 0.5 percent reduction to payment rates that would not begin for 2 years and explained that we were giving hospitals that second year before implementing the payment reduction in part “to provide entities additional time to prepare for the new payment rate” (88 FR 77180). It might not have been unreasonable for hospitals to do just as we suggested and used that additional year to prepare for only a 0.5 percent payment decrease in CY 2026—particularly because our decision to announce the rate adjustment outside the usual calendar year rulemaking process and then delay that rate adjustment was atypical. While hospitals may be correct that they often plan their budgets in advance, section 1833(t)(9)(A) of the Act requires us to update OPPS rates annually, and section 1871(e)(1)(B) of the Act requires only that changes to Medicare payment rates be finalized at least 30 days before they take effect. We therefore often implement policy changes with significant financial impacts for the upcoming year through the annual rulemaking process, and nothing here should be construed to suggest a change to that general practice, nor should hospitals expect us to generally give them additional time to prepare for policy changes beyond what the Congress prescribes in the statute. As we noted above, we changed hospital payments by over 3 percent through the annual ratemaking process for CY 2018 as part of our budget neutralization obligations, and so a 1.5 percentage point change through those same rulemaking procedures falls within the type of annual rate fluctuations hospitals can reasonably expect. Neither the APA nor the Medicare statute imposes a one-way ratchet in which we may increase payments through annual rulemaking but not decrease them through annual rulemaking.
Also important to our decision is that giving hospitals an additional year before any increase to the adjustment still fulfills the rationale underlying the Final Remedy rule that motivated our proposed change: to implement the budget neutrality requirement in a manner that restores affected 340B covered entity hospitals to the financial position they would have been in had the 340B Payment Policy not been implemented in 2018. We proposed to increase the adjustment to 2 percent so that the hospital utilization that will ultimately determine each hospital's payment reduction would better map onto payment increase from CY 2018 through 2022. We are still beginning the reductions this year, and we doubt that 1 year of reduced reductions will materially undermine that rationale. Nor do we agree that because reprocessing every claim would more perfectly place hospitals in the position they would have been in absent the 340B Payment Policy than the repayment policy we adopt here, it makes relying on that general approach irrational. We already explained the flaws with claims reprocessing (88 FR 77153 through 77154), including significant delays and administrative burden on us and hospitals alike. We anticipate that adopting in future years a larger percent prospective payment reduction will ( printed page 53715) better serve that goal than a 0.5 percent reduction while still avoiding the pitfalls of full claims reprocessing.
We also disagree with commenters to the extent that they suggest more explanation would be required, including any suggestion that we need to define precisely the relationship between budget neutrality obligations, hospital reliance interests, and the financial burden on providers. As the U.S. Supreme Court recently explained, when changing positions, “the agency does not need to show that the reasons for the new policy are better than the reasons for the old one” or “provide a more detailed justification than what would suffice for a new policy created on a blank slate” so long as it remains “cognizant” that “longstanding” policies might have engendered reliance interests. Food & Drug Admin. v. Wages & White Lion Invs., L.L.C., 604 U.S. 542, 570 (2025) (internal quotation marks omitted and emphasis in original). We identified in the CY 2026 OPPS/ASC proposed rule that we were proposing to change our policy, and we gave an explanation for why we believed our proposed new policy reasonably balanced the relevant interests. We specifically considered reliance interests in the CY 2026 OPPS/ASC proposed rule, and in this final rule with comment period we are finalizing to delay by an additional year any increase in payment reductions to further respect any reliance interest. Any change to the adjustment in 42 CFR 419.32(b)(1)(iv)(B)( 12) that applies beginning in CY 2027 would go through the usual annual rulemaking process. Nothing more is required.
Comment: One commenter suggested that CMS failed in the CY 2026 OPPS/ASC proposed rule to adequately to grapple with why CMS did not implement a commenter's suggestion to impose percent reductions greater than 0.5 in the Final Remedy rule. In the CY 2026 OPPS/ASC proposed rule, we explained that “[o]ur basis for not accepting the suggestion was that the 0.5 percent rate/16-year timeframe `properly reverses the increased payments for non-drug items and services to comply with statutory budget neutrality requirements while at the same time accounting for any reliance interests and ensuring that the offset is not overly burdensome to impacted entities”. (90 FR 33635 [quoting 88 FR 77179]) In the commenter's estimation, this incorrectly suggests that CMS' response to these comments as being the entire rationale for selecting the 0.5 percent reduction when the Final Remedy rule had additional rationales. The commenter identified these rationales as “potential impact on vulnerable providers and their communities”, “standard remedial principles” and “basic fairness”. The commenter stated that CMS fails to account for these concerns in the CY 2026 OPPS/ASC proposed rule “leaving affected communities without any information about how the proposed rule is lawful, much less fair”.
Response: We disagree that our proposal failed to properly account for the potential impact on vulnerable providers and their communities, standard remedial principles, and basic fairness when proposing a new schedule for the budget neutrality adjustment. As an initial matter, we did not cite any of those rationales as the basis for rejecting a higher reduction rate in the Final Remedy rule. Instead, as we noted in the CY 2026 OPPS/ASC proposed rule, our explanation turned on the fact that a 0.5 percent reduction complies with budget neutrality requirements while accounting for reliance interests and burden on providers. We cited remedial principles and basic fairness when rejecting a suggestion that we recoup the full $10.6 billion payment we were also making to providers who had previously acquired drugs through the 340B program instead of simply unwinding the $7.8 billion payment increase to hospitals from CY 2018 through 2022 to place hospitals in as close to a position as they would have been absent the 340B Payment Policy. We also addressed vulnerable patients and communities in response to a suggestion that we exempt from the budget neutrality policy hospital groups that serve certain communities. We explained in part that the payment reductions were the mirror image of prior payment increases that would otherwise be a windfall to providers—windfalls that those communities would share in the cost of funding through taxes, premiums, and cost sharing. We did not propose to revisit either policy and so there was no reason to revisit those rationales.
In any event, our proposal was consistent with those principles. As we noted, “standard remedial principles and basic fairness support situating hospitals as closely as possible to the financial situation they would have been in absent the 340B Payment Policy” (88 FR 77179)—the same rationale we explained in the CY 2026 OPPS/ASC proposed rule supports a quicker recoupment period (90 FR 33635). And we continue to be sensitive to the potential impact on vulnerable patients, their communities, and providers. The proposed decreases were the mirror image of prior payment increases—in fact, they would be even more so if we ultimately implement a quicker recoupment period. Additionally, as we noted in the Final Remedy rule, the statute authorizes transitional outpatient payments to cancer and children's hospitals that insulate them from the payment impact of policies like these. (88 FR 77181)
Comment: One commenter, reiterating their comments to the Final Remedy rule, suggested that CMS abandon reliance on “inapt” payment rules and its budget neutrality proposal altogether and instead invoke section 1870 of the Act (42 U.S.C. 1395gg), which describes when and how CMS may recover incorrect payments it makes on behalf of an individual. The commenter explained that this authority would allow CMS to forgo recovery where the individual for whom the incorrect payment was made was without fault and making the adjustment would “defeat the purposes of subchapter II or subchapter XVIII or would be against equity and good conscience” and that it would be appropriate for CMS to exercise this discretion as “[c]learly the beneficiaries for whom providers received increased payments from 2018 to 2022 were without fault”. The commenter additionally contended that CMS recovering from recipient hospitals is also against equity and good conscience from a broader economic standpoint. The commenter stated that the increased OPPS payments hospitals received have been incorrectly characterized as a “windfall” and such a characterization “does not square with the modest 3.19 percent adjustment that was in place from 2018 to 2022. Over that time period, even a compounded 3.19 percent adjustment would not have kept up with the rate of inflation.” Finally, the commenter alleged that recoupment is bad policy because it excuses statutory non-compliance. By setting a precedent of financing its remedy payments, the commenter stated that CMS removes an incentive to engage in thoughtful, judicious and textually grounded rulemaking. In the absence of clear statutory authority to offset its remedy payments, the commenter argued that CMS should choose not to do so, and allow the Congress to intervene if it chooses to do so.
Response: We refer readers to our response in the Final Remedy rule to the same commenter's suggestion that we rely on section 1870 of the Act (42 U.S.C. 1395gg) (88 FR 77178). As we explained there, section 1870 of the Act specifies when providers can shift liability to beneficiaries for overpayments, which can in turn be ( printed page 53716) waived when, among other requirements, liability would “defeat the purposes of . . . subchapter XVIII or would be against equity and good conscience.” Section 1870 is silent about the situation here where CMS adjusts future payments through its budget neutrality authority, and the commenter does not suggest we are required to invoke section 1870 of the Act in this circumstance.
We disagree with the commenter's suggestion that the payment statute that we have invoked to make the lump-sum remedy payments—section 1833(t)—is comparatively “inapt.” That is the payment statute governing the OPPS, and as we explained in the Final Remedy rule, sections 1833(t)(2)(E) and (14) of the Act authorize the $10.6 billion in payments that compensate hospitals for the reduced payments they received. (88 FR 77156 through 77161.) Those authorities, however, have budget neutrality consequences, which we have implemented through prospective payment decreases. (88 FR 77169 through 77182.) The commenter does not explain how section 1870 of the Act interacts with those authorities, or how abandoning section 1833(t) of the Act in favor of section 1870 of the Act would have allowed us to make billions of dollars in lump-sum remedy payments. And even if the commenter could, we do not find under section 1870 of the Act that honoring the budget neutrality requirements under section 1833 of the Act through our policy here would “defeat the purposes of . . . subchapter XVIII” of the Act. Disregarding the Congress' instruction that OPPS generally be budget neutral here would instead defeat the purposes of subchapter XVIII—one of which is sustainability.
Nor do we find under section 1870 of the Act that honoring the budget neutrality requirements under section 1833 of the Act through the budget neutrality policy we proposed would be against equity and good conscience. It is incongruous to suggest that the 3.19 percent increase from 2018 through 2022 is “modest”, but our proposed 2 percent decrease or finalized 0.5 percent decrease to unwind that increase would be against equity and good conscience. As we have repeatedly stated, even a 2 percent decrease would be within the usual annual payment fluctuations. With regards to inflation, the OPPS primarily accounts for increased costs through other mechanisms like the annual market basket increase and wage index, not through the payment changes for hospitals from budget neutralization requirements. We maintain that allowing hospitals to keep past payment increases due to the 340B Payment Policy would be a windfall in the sense that hospitals would be retaining payment increases after we unwound the corresponding payment decreases that both justified and authorized them. See, for example, windfall, Mirriam Webster Online (“an unearned . . . gain or advantage”).[84]
Finally, we disagree with the commenter's policy statements against budget neutrality. As an initial matter, such arguments cannot overcome the text of the statute. And the commenter is wrong that we are financing remedy payments to the extent the commenter means that we are charging hospitals for the cost of the Final Remedy rule. We rejected comments suggesting that we budget neutralize the full $10.6 billion remedy payments in the Final Remedy rule. Instead, we are simply unwinding the $7.8 billion in payment increases from CY 2018 through 2022 predicated on the invalidated payment decreases to place all parties as close as we can to the situation they would have been in if the 340B Payment Policy had never been adopted. Doing so does not excuse statutory non-compliance or disincentivize us from seeking the best reading of the statute. Rather, as we have noted, it ensures that the only money actually spent is money authorized to be spent by the statute and avoids strategic behavior on behalf of regulated entities. (88 FR 77176.)
Comment: The same commenter stated that section 1833(t)(14)(A) of the Act creates a “workaround” to section 1833(t)(14)(D) of the Act, which requires CMS to conduct acquisition cost surveys to assess in setting the drug APC payment rates, that “absolves” the Secretary from actually using data resulting from the acquisition cost surveys. In the commenter's view, this allows the Secretary to “game the system” for any rates set under section 1395l(t)(14) of the Act. The commenter alleged that there is no information in the CY 2026 OPPS/ASC proposed rule to suggest the pricing information was set with data obtained from compliance with section 1833(t)(14)(D) of the Act or its “workaround” in 1833(t)(14)(A) of the Act. The commenter claimed that this lack of transparency prevents meaningful comment and does not adequately explain the agency's authority to budget neutralize. The commenter concluded that “[a]s the requisite survey is just being rolled out in this same Proposed Rulemaking, these `budget neutrality offset adjustments' should not be allowed to continue because the agency is under no obligation to neutralize the effects of its 340B payments cuts that the Supreme Court found to be in violation of another statute.”
Response: The commenter correctly identifies that 1833(t)(14) of the Act authorizes two options to set drug APC payment rates, depending on the circumstance, though we would not necessarily characterize either as a “workaround” for the other. As the Supreme Court explained, section 1833(t)(14)(A)(III)(i) of the Act “applies if [CMS] collects `hospital acquisition cost survey data' from hospitals” under paragraph (D), and section 1833(t)(14)(A)(III)(ii) of the Act applies if we “do not conduct a survey of hospitals' acquisition costs and if acquisition cost data are therefore `not available.' ” Am. Hosp. Ass'n v. Becerra, 596 U.S. 724, 734 (2022). Because we have not yet completed a survey under paragraph (D), we have set payment rates under section 1833(t)(14)(A)(III)(ii) of the Act, just as we have for two decades. We explained that policy and how we set rates under section 1833(t)(14)(A)(III)(ii) of the Act in the CY 2026 OPPS/ASC proposed rule (88 FR 33628 through 33629) and published for comments the proposed rates in Appendices A and B to that proposed rule.[85] The commenter does not identify any flaw with those explanations, and we disagree that doing so inadequately explains those rates or failed to provide adequate information for comment, or that by following the statutory process we have “game[d] the system.” As we have previously explained, we do not rely on section 1833(t)(14)(A)(III)(i) or (ii) of the Act for the payment adjustment here, but instead our budget neutralization authority.[86] We explained at length how we arrived at the reduction and the amount of the total reduction, and the commenter does not identify any flaws with that methodology besides reiterating its disagreement with our statutory interpretation. We therefore disagree that we have provided insufficient information for comment.
Comment: One commenter stated that CMS has not provided meaningful notice of its intent to recoup because CMS has not provided affected hospitals with concrete information for review and consideration. To support this contention, the commenter focuses on CMS' use of the words “estimated” and ( printed page 53717) “approximately” in the CY 2026 OPPS/ASC proposed rule's discussion of payment for non-drug items and services in CY 2018 through CY 2022 and stated that “3 years later the agency is still talking in estimates and approximations”. The commenter alleged a discrepancy between an estimated impact of $1.6 billion that is listed in the narrative and an approximate impact of $1.9B noted for FY 2018 in Table 61 on the same page. The commenter also compared Table 4 in the Final Remedy rule, describing the repayment schedule, scheduled to begin in CY 2026, that would take back 0.5 percent per year from 2026-2041 and Table 62 in the CY 2026 OPPS/ASC proposed rule, describing the repayment schedule that would take back 2 percent per year from 2026-2031 and alleged that “[s]eemingly, something has changed in the math. The 2023 0.5 percent reduction dollar amounts in Table 4 multiplied by 4 yield very different numbers than what appears in Table 62. Yet, no meaningful explanation walks the public through these numbers, nor is there any confirmation that the $7.769 billion was the actual reinstatement. It is referred to as an “estimate” in the Table 62”. The commenter stated that 42 U.S.C. 1395hh(a)(2) requires the agency to undergo notice and comment rulemaking regarding anything that affects a provider's payment and that this is separate from its other obligations regarding notice and comment requirements in the Administrative Procedure Act (APA) (5 U.S.C. 553). The commenter stated that in order for there to be proper notice under either statute, the notice must provide sufficient detail for the reviewer to fully understand the substantive change to be made and that that is not possible because the commenter does not know why CMS is using estimates for data that was completed more than a year ago or why CMS' numbers changed from the 2023 Final Remedy rule to the CY 2026 OPPS/ASC proposed rule.
Response: We disagree that using the terms “estimated” and “approximately” means that this rulemaking fails to meet any requirement to engage in notice-and-comment rulemaking. The commenter does not suggest that we failed adequately to explain how we arrived at our estimations or approximations or that we gave the commenter inadequate information to comment on our methodology or data. Instead, the commenter appears to read into sections 1870 of the Act and the APA a categorical ban on setting policy based on estimations or approximations. We are unaware of any such requirement, nor would one make sense in a prospective payment system where the statute requires us routinely to set policy based on future predictions. We routinely approximate the results of our calculation rather than describe every calculation to the cent. However, for the readers awareness, in the Final Remedy rule, we state that our estimate of the total amount of additional spending on non-drug item and service spending “rounds to $7.8 billion, but is more precisely $7,768,568, 239” (88 FR 77153). And here, it was more precise to refer to estimations when we did. We are unwinding payment increases for non-drug items and services between CY 2018 and 2022 based on the rate increase for those items and services that CMS estimated in 2017 would redistribute the amount it estimated it would save for drugs acquired through the 340B program. We are not recouping the actual amount CMS saved by decreasing payments for drugs acquired through the 340B program, which, as we explained in the Final Remedy rule, exceeded our 2017 expectations. ( See88 FR 77177 and 77187.)
That difference between estimated payments and actual payments also explains the alleged discrepancy between the estimated impact of $1.6 billion for CY 2018 that is listed in the narrative and the approximate impact of $1.9 billion for that year listed in Table 61 on the same page of the CY 2026 OPPS/ASC proposed rule. As we explained in the CY 2026 OPPS/ASC proposed rule, to effectuate the budget neutrality provisions of the OPPS in CY 2018 rulemaking, CMS redistributed the $1.6 billion it estimated in 2017 that it would save in CY 2018 in reduced drug payments to increase nondrug item and service payments (90 FR 33632). That is the $1.6 billion number we reference in the narrative and that contributes to the $7.8 billion we intend to recoup through budget neutrality policy. Again, actual savings on drug payments in CY 2018 through CY 2022 exceeded our projections. (88 FR 77177 and 77187.) Table 61 of the CY 2026 OPPS/ASC proposed rule summarizes the actual reduced 340B drug payment amounts, which we derived from Addendum AAA published with the Final Remedy rule. (90 FR 33632). Thus, adding the amounts in Table 61 together totals to $10.6 billion—the total remedy payments we made to hospitals that acquired drugs through the 340B program at a reduced payment rate. Nor has anything changed in the math between Table 4 in the Final Remedy rule and the projected recoupments in Table 62 in the 2026 OPPS/ASC proposed rule. Since Table 4 was published in 2023, CMS has updated its payment projections for Medicare based on additional data. Table 62 uses those updated projections when estimating the impact of a 2 percent reduction, rather than just multiplying the original 0.5 percent reduction dollar amount by 4 like the commenter.
Comment: One commenter referenced CMS' statement in the CY 2026 OPPS/ASC proposed rule that the main premise of the Final Remedy rule was to implement the budget neutrality requirement in a manner that restored affected 340B covered entity hospitals to the financial position they would have been in had the 340B Payment Policy not been implemented in 2018. The commenter stated that there is no need to restore affected 340B entity hospitals to the financial position they would have been in had the 340B payment policy not been implemented in 2018 because that restoration already occurred with the early 2024 lump sum payments.
Response: We do not agree that the lump sum payments paid to hospitals in 2024 restored 340B covered entity hospitals, or any OPPS-paid hospital, to the financial position they would have been in had the 340B Payment Policy not been implemented from 2018 through 2022. The lump sum payment does not account for the additional payments for non-drug items and services that were made to all hospitals from CY 2018 through CY 2022 to achieve budget neutrality for the reduced 340B drug payments.
Comment: Many commenters stated that CMS' proposal failed to appreciate the financial strain the proposed increased reduction would impose on hospitals that are already operating on tight margins. These commenters claimed that CMS' proposal failed to account for adverse financial trends that have occurred since 2023, such as increased costs of labor, supplies and pharmaceuticals, aging hospital infrastructure, inflation, inadequate government reimbursements that lag behind inflation, eroding margins, supply chain disruptions, an aging population with more complex, chronic conditions, the lingering effects of the COVID-19 PHE and impending economic strains such as the OBBBA's reduction to Medicaid and Health Insurance Marketplace payments, IRA drug reductions, Medicare sequestration, and HRSA's recently noticed 340B Rebate Model. These commenters stated that the financial burden imposed by the proposal would threaten the services that they provide. ( printed page 53718)
Response: We recognize that hospitals may have experienced financial strain in recent years, but other statutory provisions address many of the issues hospitals raise like inflation or the medical complexity of the Medicare population, and we do not believe that relaxing OPPS's budget neutrality provisions is the proper policy response. With respect to interest specifically, we note that the first of the increased payment amounts occurred in 2018, whereas the last of the recoupment amounts may not be until after 2030—in effect a more than 10-year loan without any attached interest. Ultimately, though, we cannot ignore the financial windfall that hospitals received from 2018 to 2022 and our statutory obligation to recover that windfall. We hope that delaying for a year any increase to the 0.5 percent reduction will allow hospitals to do any necessary planning and help to mitigate any financial strain
Comment: One commenter contended that the CY 2026 OPPS/ASC proposed rule fails to address how a 2 percent reduction is not “overly burdensome” when it previously concluded in the Final Remedy rule that reductions of 1.25 percent, 2.25 percent, and 3 percent would have been.
Response: We have addressed above why we reevaluated in the CY 2026 OPPS/ASC proposed rule our burden analysis in the Final Remedy rule and believed a larger offset percentage was more appropriate and better achieved the overarching goal of the Final Remedy rule, which is to restore hospitals as close to the financial position they would have been in had the 340B Payment Policy never been implemented. However, also for the reasons we have described above, we are maintaining the 0.5 percent reduction for CY 2026.
Comment: Many commenters characterized the offset, whether increased or not, as a penalty, arguing that it is unfair for hospitals to be penalized for mistakes or past unlawful actions by CMS.
Response: The offset is not a penalty on hospitals. Rather, it is a rate adjustment under section 1833(t) of the Act that accomplishes an incremental and interest free recovery of windfall payments to hospitals. It is calibrated to the amount of extra money hospitals received and thus achieves payment precision, not punishment. It returns hospitals to the position they would have been absent the unlawful 340B Payment Policy and ensures that the only money ultimately spent is the money authorized to be spent by the statute.
As we stated in the Final Remedy rule, in determining the specific annual percent reduction by which to recover the funds from hospitals, our goal was to appropriately balance our statutory budget neutrality obligations against hospitals' burden and reliance interests. The adjustment we proposed in the CY 2026 OPPS/ASC proposed rule to the percent reduction we finalized in the Final Remedy rule is our attempt to more precisely balance these elements.
Comment: Multiple commenters expressed concern about the disproportionate financial effect the increased rate of recoupment would have on safety-net providers, rural providers, 340B hospitals and teaching hospitals and stated that the recoupment would reduce resources available to provide services to their patients, particularly to low-income, rural, underserved and vulnerable populations.
Response: As we said in response to similar concerns expressed by commenters in the Final Remedy rule, we recognize that our proposal to decrease future payments will have a financial impact across all hospitals paid under the OPPS, except for new providers, and we are particularly mindful of the impact on vulnerable patients and communities. But, as we also stated in the Final Remedy rule, future decreases are, on aggregate, the mirror image of prior payment increases that would otherwise be a windfall to providers and such windfalls are not cost-free; the costs are ultimately borne by beneficiaries and taxpayers—including the vulnerable patients and communities served by the hospitals to which commenters themselves refer. In fact, this remedy will reduce any beneficiary cost sharing obligations, and incrementally reduce beneficiary Part B premiums, for all Medicare beneficiaries, including the vulnerable patients to which the commenters refer. (88 FR 77180 through 77181)
Comment: Many commenters expressed concern that, if implemented, the 2 percent reduction would effectively wipe out the CY 2026 OPPS rate increase. Others referenced recent estimates from the Congressional Budget Office predicting a new 4 percent Medicare sequestration to begin in January 2026 and argued that, coupled with the proposed 2 percent reduction, outpatient hospital services would be reduced by as much as 8 percent in CY 2026.
Response: Commenters are referencing distinct statutory obligations or potential future statutory obligations. Section 1833(t)(3)(C)(ii) of the Act requires the Secretary to update the conversion factor used to determine the payment rates under the OPPS on an annual basis by applying the OPD fee schedule increase factor. Sections 1833(t)(9)(B), (t)(14)(H) and (t)(2)(E) of the Act require that the OPPS be a budget neutral system, and we decline the commenters' invitation to implement those budget neutrality provisions in a way that defeats the purpose of other statutory policies.
Comment: Many commenters recognized CMS' statutory obligation to implement a budget neutral recoupment but, based on concern about the financial impact of a two percent reduction, requested the reduction to remain at 0.5 percent. Some commenters requested that the reduction be no larger than one percent while others requested 0.25 percent. One commenter suggested that if we could not maintain the 0.5 percent reduction then we should exempt non-340B hospitals entirely and specifically, physician owned hospitals. Nearly all commenters opposed our suggested alternative of a 5 percent reduction.
Response: We appreciate commenters' acknowledgement of our statutory obligation to budget neutralize the recoupment and their suggestions for alternative annual percent reductions to do so. With respect to a 0.25 percent reduction, we do not believe that an approximately 40 year recoupment timeframe would appropriately implement the budget neutrality requirement in a manner that restores affected 340B covered entity hospitals to the financial position they would have been in had the 340B Payment Policy not been implemented in 2018. This would be over 6 times the timeframe that the 340B Payment Policy was in place and exacerbates the concern that was driving the proposed 2 percent reduction—that the longer it takes to complete the recoupment, the more likely hospital utilization or other payment factors will change from CY 2018 through 2022, and so the less each hospital's total payment reduction will correspond with that hospitals' payment increase from CY 2018 through 2022.
As for the other suggestions, we are this year finalizing a 0.5 percent reduction for the reasons we discussed previously. That is consistent with these commenters' suggestion of a 0.5 percent reduction, and less than the alternative 1 percent reduction. As we noted above, however, we anticipate implementing a larger adjustment (such as 2 percent or other adjustment greater than 0.5 percent) in next year's rulemaking, and we can consider additional alternatives at that time. ( printed page 53719)
Comment: One commenter stated that CMS is required to pay interest under 42 U.S.C. 1395 l (j) for the remedy payments from the Final Remedy rule. The commenter stated that section 1395 l (j) of the Act provides that interest is due when a provider received an OPPS payment “in excess of or less than the amount of payment that is due” that is not corrected within 30 days and that CMS paid 340B hospitals “less than the amount of payment that [was] due”. The commenter alleged that although the Supreme Court's decision applied only to 2018 and 2019, since CMS has never disputed that the rule was just as unlawful in 2020, 2021, and 2022, CMS must pay affected hospitals interest at the rate determined under 42 U.S.C. 1395 l (j), with the clock on interest beginning no later than July 15, 2022.
Response: The amount of the remedy payments, including interest on the remedy payment, is outside the scope of this rulemaking. In any event, we have addressed the issue of interest under 42 U.S.C. 1395 l (j) in the Final Remedy rule and in subsequent court briefing in Board of Trustees of University of Alabama v. Becerra, 22-cv-3367 (D.D.C.), which we incorporate here. (88 FR 77167 through 77168.) At least one court has agreed with our interpretation. See Bd. of Trs. of Univ. of Alabama v. Becerra, No. CV 22-3367 (RC), 2025 WL 2239289 (D.D.C. Aug. 6, 2025).
Comment: One commenter stated that for the same reasons we exempted new providers from the adjustment, we should exempt new procedures from the adjustment. The commenter stated that a procedure that did not exist or was not yet billable in CY 2018 through 2022 should be excluded from the reduction. The commenter suggested that recoupment be done “through individual hospital payment terms, not a nonspecific process linked to ongoing or future services”. In the event CMS does not adopt the commenter's suggestion, the commenter alternatively suggested that CMS adopt the 5 percent reduction to expedite repayment.
Response: A similar request was made by a commenter in the Final Remedy rule. As we said in our response to that comment, “exempting new items and services from this payment adjustment may distort providers' incentives to prescribe items and services based on whether they existed between CY 2018 and 2022 rather than whether they are medically appropriate, potentially impacting the care providers give to beneficiaries. And the more exceptions we create, the more complicated we make the payment reduction. Complications increase the risk of delays or errors in implementing this final rule”. (88 FR 77180.) This continues to be true. With respect to the commenter's recommendation that we consider adopting a 5 percent reduction to expedite repayment, we do not believe that it would be appropriate to implement such a reduction in CY 2026 for the same reasons that we are not implementing a 2 percent reduction in CY 2026, as we have explained previously.
Comment: Several commenters offered an alternative method of recoupment in which CMS would incrementally increase the 340B repayment percentage to shorten the overall repayment period but do so in a way that accommodates other financial pressures in the industry. In such a scenario, the commenters suggested, the CY 2026 reduction would remain at 0.5 percent since there are other planned cuts for Medicare providers due to sequestration, and CMS could plan to move to 0.75 and then one percent in future years if the sequester cuts are mitigated by Congress.
Response: We thank the commenter for their suggestions. While we disagree that we should implement our budget neutrality obligations in a way that frustrates other Congressional payment directives such as sequestration, we have partially adopted this commenter's proposed phased approach by retaining a 0.5 percent reduction for CY 2026 while delaying any larger reduction until CY 2027. As we note, we believe an additional year is sufficient time for providers to adequately prepare for the change in policy.
Comment: Many commenters posited that CMS failed to consider a sufficient number of alternative timeframes and adequately explain why it selected 3 years as the one alternative it did consider. As a result of this failure, these commenters stated that the 6-year timeframe CMS did propose is arbitrary, inadequately justified, and does not satisfy the APA's requirement to consider reasonable alternatives. These commenters stated that CMS needs to consider more alternatives, including those longer than 16 years, and explain why 6 years is the appropriate timeframe compared to them.
Response: For the reasons we have explained, we believe that our proposal properly acknowledged that it was shifting course and adequately explained the rationale behind the proposed shift. We disagree that we needed to propose more options to consider. Ultimately, commenters ask us to, but neither the APA nor the Act requires us to, consider every possible alternative. We are just required to consider significant ones. In the CY 2026 OPPS/ASC proposed rule, we discussed the initial 0.5 percent reduction for 16 years, a 2 percent reduction for 6 years, and a 5 percent reduction for 3 years. (90 FR 33636.) We proposed that a 2 percent reduction for 6 years adequately balanced budget neutrality against hospital burden and reliance. (90 FR 33635.) Hospitals have not raised additional interests we failed to consider, but convinced us that we could better account for their reliance interests, which we have done by retaining the 0.5 percent reduction for CY 2026 this while planning to raise the reduction to a larger percentage (such as 2 percent) beginning in CY 2027. In the course of finalizing this rule, we also considered commenters' suggestion that we extend the budget neutrality policy, such as implementing a 0.25 percent reduction for 40 years. As we explained above, any extension would likely exacerbate differences between how much hospitals received in excess payments from CY 2018 through 2022 and their total reductions in CY 2026 and thereafter, thus undermining our goal of returning hospitals as close as reasonably possible to the position they would have been absent the 340B Payment Policy. We have rejected this option.
While there are many possible specific payment reductions, the particular amount is necessarily an exercise in line-drawing. Commenters raise no specific reduction range that we fail to address in this final rule with comment period, and we disagree that there are significant other options commenters do not identify that we have not considered.
Comment: Several commenters requested that CMS establish an appeals process for hospitals that disagree with cost assignments. One commenter indicated that the CY 2026 OPPS/ASC proposed rule states that CMS will direct MACs to remedy the hospital 340B drug underpayments with budget neutrality adjustments between 2026-2031 but it does not clearly state how those remedy payments fit into the existing claims, reimbursement, and appeal structures. The commenter recommended that CMS state in the final rule that (1) dissatisfied hospitals will have a clear path to appeal the amount of CMS' budget neutrality adjustment recoupments and describe the appeals process; and (2) that CMS intends the final rule to be subject to judicial review. Specifically, the commenter stressed, CMS should state that reliance on section 1833(t)(2)(E) of the Act as authority for its proposed adjustments is not intended to create any implication that the adjustments are ( printed page 53720) not subject to judicial review under section 1833 (t)(12) of the Act.
Response: With respect to a process for hospitals to appeal the amount of CMS' budget neutrality adjustment recoupments, we believe these adjustments, like the initial adjustments in 2018, ultimately adjudicate claims for payment and so any available appeal would follow the procedures set out under section 1869 of the Act and its implementing regulations. We respectfully decline the commenter's request to opine in advance on how the jurisdictional provisions of section 1833(t)(12) of the Act might impact the Departmental Appeals Board's or courts' jurisdiction. Those bodies will adjudicate their jurisdiction in specific cases in the ordinary course with the benefit of appropriate briefing.
Comment: One commenter suggested that if we proceed with the two percent reduction that we should conduct robust monitoring for any unintended consequences and consideration of flexibilities or targeted supports for safety-net and teaching hospitals that may be disproportionately affected.
Response: We agree that it is important to monitor the effects of the increase for any unintended consequences and will take the commenter's suggestion under consideration for future rulemaking. We also appreciate the commenter's suggestion with respect to future consideration of flexibilities or targeted supports for hospitals based on that monitoring. We will consider this for future rulemaking.
Comment: One commenter recommended that CMS consider stronger regulation of the pricing by pharmaceutical companies to allow hospitals to get more of a discount “since pharmaceutical companies continue to have high profit margins while non-profit health systems continue to struggle to provide 340B drugs to many underserved patients.”
Response: This rule implements budget neutralization requirements in section 1833(t)(14) of the Act based on the Final Remedy rule, and the CY 2026 OPPS/ASC proposed rule did not propose additional regulations of pharmaceutical prices. This comment is therefore out of its scope.
Comment: A few commenters supported our proposal to revise the annual reduction to the OPPS conversion factor under § 419.32(b)(1)(iv)(B)( 12) used to determine the payment amounts for non-drug items and services from 0.5 percent to 2 percent. One commenter stated that faster repayment would ensure greater certainty in repayment amounts and also, due to the time value of money, reduce the strain on the Federal budget.
Response: We thank commenters for their support of our proposal. As we note, while we are not finalizing that proposal at this time, we anticipate proposing a larger offset beginning in CY 2027.
Comment: Several commenters expressed concern about the implications of the proposed 2 percent reduction in OPPS non-drug payments for providers contracting with Medicare Advantage (MA) organizations. Commenters stated that CMS has provided remedy payments under traditional Medicare following AHA v. Becerra in accordance with the Medicare Program; Hospital Outpatient Prospective Payment System: Remedy for the 340B-Acquired Drug Payment Policy for calendar years 2018-2022 OPPS/ASC rule.[87] Further, commenters shared that MA organizations, who adopted a similar reimbursement rate for 340B-acquired drugs during that same time period, may not have made the corresponding remedy payments to providers. Commenters emphasized that this could leave hospitals disadvantaged as they may not have received the benefit of a remedy payment from MA organizations, and they may be subject to the new prospective reductions to provider reimbursement for non-drug payments. Several commenters stated that this dynamic unfairly shifts resources to MA organizations and compounds the financial strain on hospitals, particularly given high and growing MA enrollment.
Several commenters recommended that CMS take additional steps in this CY 2026 OPPS/ASC final rule with comment period to mitigate the impact of the proposed adjustment. Commenters specifically urged CMS to clarify that MA organizations are expected to make hospitals whole for 340B-acquired drugs administered between 2018-2022 and to prevent the prospective reductions from being passed through to providers absent repayment by MA organizations.
Response: We appreciate commenters' feedback on this issue. Under the MA program, CMS provides a capitated prospective payment to MA organizations to provide coverage to enrollees, and the MA organizations pay providers for this care. CMS calculated and paid CY 2018-2022 MA rates under the Advance Notice and Rate Announcement,[88] and those MA rates reflected the FFS policies as of the time they were finalized.
We also appreciate commenters' concerns regarding the potential implications of the Final Remedy rule for MA organizations' payments to providers. We understand from commenters that many MA organizations base their privately contracted reimbursement rates with providers on FFS rates set by CMS and that existing MA contracts may not account for the remedy provisions established in the Final Remedy rule. However, CMS establishes payment policies and payment rates for services payable under FFS through a separate, distinct process that is not directly related to the terms of private contracting arrangements between MA organizations and providers. Further, section 1854(a)(6)(B)(iii) of the Act prohibits CMS from requiring an MA organization to contract with a particular hospital, physician, or other entity to furnish items and services, including 340B-acquired drugs, or requiring a particular price structure for payment under such a contract. Providers and MA organizations may engage in any contract negotiations or re-negotiations independently of CMS.
Section 1852(a)(2) of the Act mandates that MA organizations reimburse non-contract providers at least the amount they would have received under Medicare FFS. We expect that MA organizations will comply with this statutory requirement. We note that the Final Remedy rule excluded providers that enrolled in Medicare after January 1, 2018, from the prospective rate reduction.
After consideration of public comments received, we are finalizing our proposal to adopt Addendum R—340B Remedy Offset Providers for CY 2026. For the reasons discussed above, we are not finalizing for CY 2026 our proposal to revise the reduction to the OPPS conversion factor under 42 CFR 419.32(b)(1)(iv)(B)( 12) used to determine the payment amounts for non-drug items and services for hospitals for whom this adjustment applies from 0.5 percent to 2 percent. However, also for the reasons discussed above and in the 2026 OPPS/ASC proposed rule, we anticipate implementing a larger percent reduction (such as 2 percent or other reduction greater than 0.5 percent) beginning in CY 2027. Any change to the adjustment ( printed page 53721) in 42 CFR 419.32(b)(1)(iv)(B)( 12) that applies beginning in CY 2027 would go through the usual annual rulemaking process. Please see Table 110, for an estimate of the impacts of the offset for CY 2026.
We note that the status indicators impacted by this finalized policy include, SI = J1, J2, P, Q1, Q2, Q3, R, S, S1, T, U, V. These status indicators generally capture the non-drug items and services impacted by a change in the OPPS conversion factor. Status indicator S1 will be newly effective starting in CY 2026 per our policy finalized in section V.B. of this final rule with comment period. The new “S1” status indicator represents products that were once packaged into procedures assigned to a status indicator of “S”. This aligns with our goal of reducing non-drug item and service spending to situate hospitals in the approximate financial position they would have been in absent the 340B payment policy. Additionally, we note that although New Technology APCs are assigned to a status indicator of “S” or “T” they are assigned to fixed payment rates that are unaffected by this reduction to the OPPS conversion factor.
i. Impact of the Prospective Offset to the OPPS Conversion Factor on the ASC Payment System
As we noted in the CY 2023 OPPS/ASC final rule with comment period (87 FR 71975), budget neutrality adjustments to the OPPS conversion factor do not impact the ASC conversion factor. However, we also noted in that rule that revisions to the OPPS conversion factor can have an indirect impact on the ASC payment system because the ASC standard rate setting methodology adopts OPPS payment rates and the device portion (or device offset amount). Specifically, because the device portion for device-intensive procedures is held constant with the OPPS and is not calculated with the ASC conversion factor, a reduction to the OPPS conversion factor will lower the device portion for device-intensive procedures, including the payment rates for device-intensive procedures under the ASC payment system. We further clarified, however, that any decline in expenditures for device portions under the ASC payment system would be fully offset through the ASC weight scalar, which would increase payment for the non-device portions of all covered surgical procedures and certain covered ancillary services. Together, that means that reducing the OPPS conversion factor can mean that we pay relatively less for device-intensive procedures and relatively more for other surgical procedures.
In the Final Remedy rule (88 FR 77179), a commenter referenced this discussion in the CY 2023 OPPS/ASC final rule with comment period and requested that CMS provide an analysis of the impact of the remedy's proposed OPPS conversion factor reduction on ASC payment rates. Specifically, the commenter requested additional details on the magnitude of the change in payments for device-intensive procedures with and without the OPPS conversion factor reduction. As further discussed in section XIII. of the CY 2026 OPPS/ASC proposed rule, historically, the ASC payment system has generally adopted the final OPPS conversion factor for a calendar year in determining the OPPS payment rates that are used for determining the device portions for device-intensive procedures under the ASC payment system. A 2 percent reduction in OPPS payment rates would otherwise reduce ASC payments for device-intensive procedures by approximately 1 percent; the non-device portions for all covered surgical procedures would otherwise be increased to offset reduction to device portions for device-intensive procedures. For CY 2026, we estimated that the reduction to device portions would be approximately $42 million and would otherwise increase the ASC weight scalar by 0.1 percent.
However, we proposed to set ASC payment rates based on the OPPS payment rates without the remedy's 2 percent prospective offset. In other words, we proposed that these payment rates would be based on OPPS payment rates for hospitals that enrolled in Medicare after January 1, 2018. We acknowledged that in the CY 2023 OPPS/ASC proposed rule we stated that “the revised OPPS conversion factor will have an impact on the ASC payment system”, but we were responding to a comment asking about how unwinding the 340B Payment Policy would reduce the OPPS conversion factor prospectively beginning in CY 2023, not about how we should approach any temporary reduction in the OPPS conversion factor to unwind the 340B Payment Policy in place from CY 2018 through 2022 (87 FR 71975). In this context, we believed that selecting the higher OPPS payment rate is more consistent with the history ( printed page 53722) and logic of both the ASC payment system as well as the Final Remedy rule.
As for the ASC payment system, including the 2-percent prospective offset would not be an accurate reflection of the device costs of covered surgical procedures in the ASC setting. Further, we are concerned beneficiaries could have access issues to certain device-intensive procedures in the ASC setting, such as total knee arthroplasty and total hip arthroplasty, if we maintained a 2 percent reduction to the payment rates for device-intensive procedures for each calendar year we applied the prospective offset. The total payment for device portions of device-intensive procedures under the ASC payment system is roughly 27 percent of total ASC payments.
We stated in the CY 2026 OPPS/ASC proposed rule that this proposed policy would also be consistent with the logic of the Final Remedy rule. As we explained in that rule, the reduction to the OPPS payment rate is intended to comply with statutory budget neutrality requirements and was implemented in a manner to place hospitals in as close to the financial position they would have been in had this policy not been implemented in CY 2018 as is reasonably feasible. By contrast, it would not satisfy any similar statutory budget neutrality requirements to pass through this reduction to ASC payment rates. Nor would changing ASC payment rates for the next several years help place hospitals affected by the 340B Payment Policy in the same position as they have been absent that policy. Even if the agency wanted to extend the Final Remedy rule's logic to ASCs and try to place ASCs—none of whom ever challenged the 340B Payment Policy—in the same position as they would have been absent that policy, we doubt that passing through the 2 percent OPPS payment reduction to the device portion of ASC payment rates would do so. That is because, as discussed in the CY 2026 OPPS/ASC proposed rule, doing so would have a purely distributional impact on ASC payment rates that financially favors procedures that are less device-intensive. Therefore, as discussed in section XIII.C.4. of the CY 2026 OPPS/ASC proposed rule, we proposed that the OPPS payment rates used for rate setting under the ASC payment system for CY 2026 and subsequent years would not include the two percent prospective offset to the OPPS conversion factor as a result of the 340B remedy offset that we proposed to implement in that rule.
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: Two commenters supported our proposal to set ASC payment rates based on OPPS payment rates without the two percent prospective offset. Both commenters stated that not doing so would result in inaccurate payments for device costs in covered surgical procedures in the ASC setting.
Response: We thank commenters for their support.
After consideration of public comments, we are finalizing, without modification, our proposal to set ASC payment rates based on the OPPS payment rates without the remedy's prospective offset.
8. All-Inclusive Rate (AIR) Add-On Payment for High-Cost Drugs Provided by Indian Health Service and Tribal Facilities
a. Background
In the CY 2000 OPPS final rule (65 FR 18434), CMS implemented the PPS for hospital outpatient services furnished to Medicare beneficiaries, as set forth in section 1833(t) of the Act. In the CY 2000 OPPS final rule, we noted that the OPPS applies to covered hospital outpatient services furnished by all hospitals participating in the Medicare program with a few exceptions. We identified one of these exceptions as “outpatient services provided by hospitals of the Indian Health Service (IHS).” We stated that these services would “continue to be paid under separately established rates which are published annually in the Federal Register ” and, in the CY 2002 OPPS/ASC final rule (66 FR 59856), we finalized a revision to § 419.20 (Hospitals subject to the hospital outpatient prospective payment system) by adding paragraph (b)(4), which specifies that hospitals of the IHS are excluded from the OPPS.
In the intervening years, IHS and tribal facilities have been paid under the separately established All-Inclusive Rate (AIR). On an annual basis, the IHS calculates and publishes, in the Federal Register , calendar year reimbursement rates.[89] Due to the higher cost of living in Alaska, separate rates are calculated for Alaska and the lower 48 States. For CY 2025, the Medicare Outpatient per visit rate is $718 for the lower 48 States (hereinafter referred to as “the lower 48 AIR”) and $1,193 for Alaska.[90]
In the CY 2025 OPPS/ASC final rule with comment period (89 FR 94280 through 94286), we finalized a policy to separately pay IHS and tribal hospitals for high-cost drugs, biologicals, and radiopharmaceuticals (hereinafter referred to as “drugs” for the purpose of this section) furnished in hospital outpatient departments through an add-on payment in addition to the AIR using the authority under which the AIR is calculated.[91] We note that the AIR and the add-on payment are paid out of the Part B trust fund and are not subject to OPPS budget neutrality.
We defined high cost drugs (that is, drugs qualifying for the add-on payment) for the purpose of the policy as all drugs covered under Medicare Part B and for which payment would otherwise be made under the OPPS whose per day cost exceeds two times the lower 48 AIR amount in effect at the time of the release of each year's OPPS/ASC final rule. In the CY 2025 OPPS/ASC final rule with comment period, this amount was identified as $1,334 (2) times the CY 2024 lower 48 AIR of $667).
To determine the calculated per day cost for each drug HCPCS code, we employed a methodology similar to our longstanding methodology used to calculate the per day cost of drugs for OPPS payment purposes. Specifically, to calculate the per day cost for CY 2025, we used an estimated payment rate based on the ASP methodology payment rate, which for purposes of the policy was generally ASP plus 0 percent (which is the payment rate for separately payable IHS drugs under the policy). We then used the manufacturer-submitted ASP data from the fourth quarter of CY 2023 to determine the per day cost. For drugs that did not have either an ASP-based payment rate or a payment rate based on WAC, we used mean unit cost (MUC) of the items derived from the CY 2023 hospital claims data to determine their per day cost.
We finalized that the amount of the add-on payment for a high-cost drug would be the average sales price (ASP) for the drug with no additional payment (that is, ASP plus zero percent). We note that this add-on payment was implemented on a per-dose basis. In the event ASP pricing information was not available for a particular drug, we paid the WAC plus 0 percent and if WAC pricing information was not available, we paid 89.6 percent of average ( printed page 53723) wholesale price (AWP). We also adopted a drug packaging threshold exception for biosimilars in which the add-on payment is made for biosimilars whose per-day costs do not exceed the threshold of two times the lower 48 AIR but whose reference products do exceed the threshold.
To implement this policy, we finalized in the CY 2025 OPPS/ASC final rule with comment period a recurring annual process in which the lower 48 AIR in effect at the time of the release of each year's OPPS/ASC final rule with comment period would be used to create a list of drugs qualifying for the add-on payment for the following calendar year. Once the drugs qualifying for the add-on payment were determined, the payment rate for a unit of the drug would be determined in accordance with the above described pricing hierarchy. The results of that process for CY 2025 were displayed in Addendum Q to the CY 2025 OPPS/ASC final rule with comment period. We additionally finalized that during the calendar year, the list of drugs would be modified on a quarterly basis to add new-to-market drugs with per-day costs that exceeded two times the lower 48 AIR and to update qualifying drugs' ASPs. For a full discussion of the AIR add-on payment for high cost drugs provided by IHS and tribal hospitals, we refer readers to the CY 2025 OPPS/ASC final rule with comment period (89 FR 94280 through 94286).
b. AIR Add-On Payment for High-Cost Drugs Provided by IHS and Tribal Facilities Policy for CY 2026
For CY 2026, we proposed to continue to separately pay IHS and tribal hospitals for high-cost drugs furnished in hospital outpatient departments through an add-on payment in addition to the AIR using the authorities under which the AIR is calculated.
We proposed to continue to define high cost drugs (that is, drugs qualifying for the add-on payment) for the purpose of the policy as any drugs covered under Medicare Part B and for which payment would otherwise be made under the OPPS which have per day costs exceeding two times the lower 48 AIR amount in effect at the time of the release of the CY 2026 OPPS/ASC final rule with comment period. For CY 2026, we proposed that if the CY 2025 lower 48 AIR amount was still in effect at the time of the release of the CY 2026 OPPS/ASC final rule with comment period, this amount would be $1,436 (2 times the CY 2025 lower 48 AIR of $718).
To determine the calculated per day cost for each drug HCPCS code, we proposed to continue using an estimated payment rate based on the ASP methodology payment rate (generally ASP plus 0 percent) and then using the manufacturer-submitted ASP data from the fourth quarter of CY 2024 to determine the per day cost. For drugs that do not have either an ASP-based payment rate or a payment rate based on WAC, we proposed to continue to use the MUC of the items derived from the CY 2024 hospital claims data to determine their per day cost.
With respect to the amount of the add-on payment, we proposed to use the same pricing hierarchy that we adopted in the CY 2025 OPPS/ASC final rule with comment period. For CY 2025, we explained that we adopted a practice of paying the MUC when AWP pricing is not available for a particular drug, and we proposed to continue that practice for CY 2026. We proposed for CY 2026 that the amount of the add-on payment for each dose of a high-cost drug would continue to be the average sales price (ASP) for the drug with no additional payment (that is, ASP plus zero percent). In the event ASP pricing information is not available for a particular drug, we proposed to continue to pay the wholesale acquisition cost (WAC) plus 0 percent. If WAC pricing information is not available, we proposed to continue to pay 89.6 percent of AWP. And, consistent with our practice for purposes of CY 2025, if AWP pricing information is not available, we proposed to pay the MUC. Finally, we proposed to continue the drug packaging threshold exception for biosimilars in which the add-on payment is made for biosimilars whose per-day costs do not exceed the threshold of two times the lower 48 AIR but whose reference products do exceed the threshold.
c. List of Drugs Qualifying for the Add-On Payment for CY 2026
Using two times the lower 48 AIR amount of $718 that is in effect for CY 2025 and applying the above described per-day cost methodology and pricing hierarchy, we included as Addendum Q to the CY 2026 OPPS/ASC proposed rule a preliminary list of the drugs qualifying for the proposed add-on payment and their proposed add on payment rates for CY 2026.
We proposed to create a final Addendum Q in the CY 2026 OPPS/ASC final rule with comment period using the claims data (units used per day) and ASPs available at that time. We also proposed that for HCPCS codes for drugs that are proposed for separate payment in CY 2026, but then have per day costs equal to or less than $1,436 (2 times $718) in the CY 2026 OPPS/ASC final rule with comment period, based on the updated ASPs and hospital claims data used for the CY 2026 OPPS/ASC final rule with comment period, those drugs would still receive separate payment in CY 2026.
Finally, during CY 2026, as we did during CY 2025, we proposed to modify the list on a quarterly basis (January, April, July, October) to add new-to-market drugs with per-day costs that exceed two times the lower 48 AIR and to update qualifying drugs' ASPs.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: All commenters supported our proposal to continue to separately pay IHS and tribal hospitals for high-cost drugs furnished in hospital outpatient departments through an add-on payment in addition to the AIR using the authorities under which the AIR is calculated.
Response: We thank commenters for their support.
Comment: One commenter suggested that CMS consider whether the current threshold and payment methodology adequately supports access to the full range of cell and gene therapies (CGTs) that may become available for outpatient administration. The commenter recommends that as experience with CGT delivery in these settings grows, CMS should be prepared to make additional adjustments to ensure that payment policies continue to support access to innovative therapies for underserved populations. The commenter stated that this may include evaluating whether additional reimbursement support is needed for the specialized infrastructure and training requirements associated with CGT administration.
Response: We thank the commenter for their input and will keep it in mind for future rulemaking.
After consideration of the public comments we received, we are finalizing our proposed policy without modification to continue to separately pay IHS and tribal hospitals for high-cost drugs furnished in hospital outpatient departments through an add-on payment in addition to the AIR using the authorities under which the AIR is calculated.
9. Payment for Skin Substitutes
For the public's awareness: as the policies discussed in this section are closely aligned with those addressed in the CY 2026 PFS final rule, the preamble language is largely consistent ( printed page 53724) across both rules. Because we proposed to apply these policies across both the HOPD, ASC, and non-facility setting, we believe it is important for us to address the issues raised by commenters in a comprehensive way and provide the public with a unified understanding of how these policies would apply under both the PFS and OPPS/ASC payment systems.
While the majority of the discussion is the same in both rules, we have also included additional information specific to technical payment issues that commenters raised in response to the OPPS proposals and did not include certain highly technical issues unique to the PFS that were not proposed or included in the CY 2026 OPPS/ASC proposed rule. That said, our approach here is to provide a comprehensive discussion of the payment for skin substitute products in both the facility and non-facility settings. This approach promotes transparency, consistency, and reflects CMS' strong interest in aligning payment policies across care settings when appropriate. Finally, because the proposals under both rules are substantively the same apart from technical differences arising from the distinct characteristics of each payment system, such as geometric mean costs of APCs and Practice Expense RVUs, we believe it is reasonable and efficient to present a unified discussion of the policy.
a. Background
The CY 2014 Hospital Outpatient Prospective Payment System (OPPS)/Ambulatory Surgical Center (ASC) final rule with comment period describes skin substitutes as “a category of products that are most commonly used in outpatient settings for the treatment of diabetic foot ulcers and venous leg ulcers” (78 FR 74930 through 74931). When a procedure utilizing a skin substitute product is performed, providers bill one or more Healthcare Common Procedure Coding System (HCPCS) codes to describe the preparation of the wound, the use of at least one skin substitute product, and application of the skin substitute product through suturing or various other techniques. Specifically, CPT codes 15271 through 15278 describe the application of skin substitutes to various size wounds and anatomical locations.
Recently, several novel industry practices have come to our attention, likely driving substantial and unusual increases in the number of available skin substitute products, the sales and distribution structure for these products, and the rapidity of products changing manufacturer ownership. These industry changes are causing a significant increase in spending under Medicare Part B for skin substitute products in the non-facility setting. According to Medicare claims data, Part B spending for these products rose from approximately $250 million in 2019 to over $10 billion in 2024, a nearly 40-fold increase, while the number of patients receiving these products only doubled. Increases in payment rates, and launch prices for skin substitutes, especially newer products, account for the majority of observed Medicare spending increases on these products. Of note, as part of its workplan, the U.S. Department of Health and Human Services' Office of the Inspector General announced, in November 2024, plans to review Medicare Part B claims for skin substitutes to identify payments that were at risk for noncompliance with Medicare requirements with an expected issue date of fiscal year 2026.[92]
We outlined our HCPCS Level II coding and payment policy objectives for skin substitutes in the CY 2023 OPPS/ASC proposed rule (87 FR 71985) because we concluded it would be beneficial for interested parties to understand our priorities as we work to create a consistent approach for the suite of products we have referred to as skin substitutes. As discussed in the CY 2023 OPPS/ASC proposed rule, we have a number of objectives related to refining our Medicare policies in this area, including: (1) ensuring a consistent payment approach for skin substitute products across the physician office and hospital outpatient department settings; (2) ensuring that appropriate HCPCS codes describe skin substitute products; (3) employing a uniform benefit category across products within the physician office setting, regardless of whether the product is synthetic or comprised of human- or animal-based material, so we can incorporate payment methodologies that are more consistent; and (4) promoting clarity for interested parties on CMS skin substitutes policies and procedures. Interested parties have asked CMS to address what they have described as inconsistencies in our payment and coding policies, indicating that treating clinically similar products (for example, animal-based and synthetic skin products) differently for purposes of payment is confusing and problematic for healthcare providers and patients. These concerns exist specifically within the non-facility setting; however, interested parties have also indicated that further alignment of our policies across the non-facility and hospital outpatient department settings would reduce confusion.
On April 25, 2024, the Medicare Administrative Contractors (MACs) released a proposed Local Coverage Determination (LCD) to provide appropriate coverage for skin substitute grafts used for chronic non-healing diabetic foot and venous leg ulcers. The MACs issued the collaborative proposed Skin Substitute Grafts/Cellular and Tissue-Based Products for the Treatment of Diabetic Foot Ulcers and Venous Leg Ulcers LCD to make sure that Medicare covers, and people with Medicare have access to, skin substitute products that are supported by evidence that shows that they are reasonable and necessary for the treatment of diabetic foot and venous leg ulcers in the Medicare population and that coverage aligns with professional guidelines for appropriately managing these wounds. All of the MACs have delayed the effective date of the final LCDs for cellular and tissue-based products for wounds, or skin substitutes, in diabetic foot ulcers and venous leg ulcers, moving the implementation date across all MAC jurisdictions to January 1, 2026. For details, please see the final LCD, titled: Skin Substitute Grafts/Cellular and Tissue-Based Products for the Treatment of Diabetic Foot Ulcers and Venous Leg Ulcers at: https://www.cms.gov/medicare-coverage-database/basket/basket.aspx?loadBasketLink=Y&basketLinkId=552. We note that additional coverage determinations may apply to skin substitute products.
The Medicare statute, regulations, and manual provisions empower the Medicare program to determine if a product is reasonable and necessary for the treatment of a beneficiary's condition and safe and effective, not experimental or investigational, and appropriate and therefore eligible for coverage under Part B. ( See, for example, section 1833(e) of the Act (42 U.S.C. 1395l(e)), section 1862(a)(1)(A) of the Act (42 U.S.C. 1395y(a)(1)(A)), 42 CFR 411.15(k)(1), 424.5(a)(6), Medicare Program Integrity Manual section 3.6.2.2, Medicare Benefit Policy Manual chapter. 15, section 50.4.1-50.4.3, and Medicare Program Integrity Manual, chapterch.13 section 13.5.3, 13.5.4.) Coverage is a threshold determination that must be satisfied before payment considerations arise. The inclusion of a product in this payment rule or in any payment file does not necessarily imply that a determination has been made by CMS or its contractors that it is reasonable and necessary and meets the other preconditions to Medicare coverage. Any skin substitute could not ( printed page 53725) be covered if it were determined to be unreasonable or unnecessary for a particular beneficiary. Similarly, the use of short descriptors and associated FDA regulatory categories [93] may reflect current FDA regulation but are not intended to imply that FDA has determined that a product meets any specific FDA statutory or regulatory requirements. FDA's statutory and regulatory framework, including, for example, FDA's findings that a product is “safe and effective,” is not controlling of Medicare's determination under its own authorities of whether a product is “reasonable and necessary” for a Medicare beneficiary and meets all preconditions for Medicare coverage and payment. FDA does not make Medicare coverage or payment determinations, nor do FDA statutes and regulations govern Medicare coverage or payment determinations.
Medicare coverage and payment are also governed under separate statutory authorities and serve fundamentally different purposes. Coverage determinations under section 1862(a)(1)(A) of the Act (and related provisions) of the Act establish whether a service is reasonable and necessary while payment methodologies under section 1848 of the Act (and other applicable payment provisions) of the Act establish the amount Medicare will pay for covered physician services based on considerations such as resource similarity. CMS has determined that setting payment rates on a prospective basis is a different inquiry and exercise with a different set of considerations and that it makes sense here to consider how FDA regulates skin substitute products as a factor in grouping those products in various categories as described below.
We continue to believe that our existing payment policies are unsatisfactory, unsustainable over the long term, and rooted in historical practice established two decades ago prior to significant evolutions in medical technology and practice. After holding a town hall [94] to provide an opportunity for public input, including discussion of potential approaches to the methodology for payment of skin substitute products, as well as reviewing several years of comments in response to CY rulemaking in 2023, 2024, and 2025 on this subject, we developed a proposal that addressed our stated objectives as well as many of the comments we have received.
b. Medicare Part B Payment for Skin Substitutes
(1) Payment for Skin Substitutes When Used During a Covered Application Procedure Under the PFS in the Non-Facility Setting
CMS has historically considered skin substitutes to be biologicals for payment purposes under Medicare Part B. The Medicare Prescription Drug, Improvement, and Modernization Act of 2003 (Pub. L. 108-173) (MMA) established a payment methodology for drugs and biologicals under section 1847A of the Act. Under this methodology, a vast majority of drugs and biologicals separately paid under Medicare Part B are paid at the Average Sales Price (ASP) plus six percent. Section 303 of the MMA, titled “Payment reform for covered outpatient drugs and biologicals,” amended Title XVIII of the Act by adding new section 1847A of the Act. In part, this section established the use of the ASP to determine the payment limit for drugs and biologicals described in section 1842(o)(1)(C) of the Act (42 U.S.C. 1395u(o)(1)(C)) (that is, drugs or biologicals billed by a physician, supplier, or any other person and not paid on a cost or prospective payment basis) furnished on or after January 1, 2005. Because Medicare is currently paying for most skin substitutes as biologicals using the methodology under section 1847A of the Act, each skin substitute product receives a unique billing code (typically, a Level II HCPCS code) and payment limit.
Section 401 of Division CC, Title IV of the Consolidated Appropriations Act, 2021 (Pub. L. 116-260) (CAA, 2021) amended section 1847A of the Act to add new section 1847A(f)(2) of the Act, which requires certain manufacturers without a Medicaid drug rebate agreement, such as certain manufacturers of skin substitutes, to report ASP data to CMS for calendar quarters beginning on January 1, 2022, for drugs or biologicals payable under Medicare Part B and described in sections 1842(o)(1)(C), (E), or (G) or 1881(b)(14)(B) of the Act (42 U.S.C. 1395rr(b)(14)(B)), including items, services, supplies, and products that are payable under Part B as a drug or biological. Because most skin substitutes are currently paid as biologicals using the methodology described in section 1847A of the Act, manufacturers of these products are currently required to report their ASP data to CMS every quarter. Prior to this, section 1927(b)(3)(A)(iii)(I) of the Act only required manufacturers with a Medicaid drug rebate agreement to report ASP data to CMS for drugs or biologicals described in section 1842(o)(1)(C) of the Act.
Section 1847A of the Act also includes several relevant definitions. While the definition of “single-source drug or biological” provided at section 1847A(c)(6)(D) of the Act includes “a biological,” sections 1847A(c)(6)(H) and (I) of the Act offer more insight into the meaning of the term for purposes of this section. Subparagraph (I) of such section defines the term “reference biological product” as a biological product licensed under section 351 of the PHS Act (42 U.S.C. 262). Subparagraph (H) of section 1847A(c)(6) defines the term “biosimilar biological product” as “a biological product approved under an abbreviated application for a license of a biological product that relies in part on data or information in an application for another biological product licensed under section 351 of the Public Health Service Act.”
Section 1927 of the Act (42 U.S.C. 1396r-8), which is referred to multiple times in section 1847A of the Act, also references section 351 of the PHS Act when referencing biologicals. The title of section 303 of the MMA, which added section 1847A to the Act, refers to “covered outpatient drugs,” defined in section 1927(k)(2) of the Act. Subparagraph (B) of section 1927(k)(2) adds biological products to this definition when those products are licensed under section 351 of the PHS Act, among other requirements.
In the CY 2022 PFS final rule, to address the need to establish a payment mechanism for synthetic skin substitutes in the physician office setting and to be responsive to feedback received from commenters, we finalized an approach for payment of each synthetic skin substitute for which we had received a HCPCS Level II coding application. We finalized that those products would be payable in the physician office setting and billed separately from the procedure to apply them using HCPCS A-codes (86 FR 65120). ( printed page 53726)
(2) Payment for Skin Substitutes Under the Outpatient Prospective Payment System (OPPS)
Prior to CY 2014, all products considered to be skin substitutes were separately paid under the OPPS as if they were biologicals according to the ASP methodology (78 FR 74930 through 74931). In the CY 2014 OPPS/ASC final rule with comment period (78 FR 74938), we unconditionally packaged skin substitute products furnished in the hospital outpatient setting into their associated application procedures as part of a broader policy to package all drugs and biologicals that function as supplies when used in a surgical procedure. As part of the policy to package skin substitutes, we also finalized a methodology that divides the skin substitutes into a high-cost group and a low-cost group, to ensure adequate resource homogeneity among Ambulatory Payment Classification (APC) assignments for the skin substitute application procedures (78 FR 74933). In the CY 2015 OPPS/ASC final rule with comment period (79 FR 66886), we stated that skin substitutes are best characterized as either surgical supplies or devices because of their required surgical application and because they share significant clinical similarity with other surgical devices and supplies.
Skin substitutes assigned to the high-cost group are described by CPT codes 15271 through 15278. Skin substitutes assigned to the low-cost group are described by HCPCS codes C5271 through C5278. Claims billed with primary CPT codes 15271, 15273, 15275, or 15277 are used to calculate the geometric mean costs for procedures assigned to the high-cost group, and claims billed with primary HCPCS codes C5271, C5273, C5275, or C5277 are used to calculate the geometric mean costs for procedures assigned to the low-cost group (78 FR 74935). The graft skin substitute administration add-on codes, which include “each additional 25 sq cm” in the description (that is, CPT codes 15272, 15274, 15276, and 15278; HCPCS codes C5272, C5274, C5276, and C5278), are packaged into the payment rates for the primary administration codes.
For CY 2025, each of the HCPCS codes described earlier are assigned to one of the following three skin procedure APCs according to the geometric mean cost for the code: APC 5053 (Level 3 Skin Procedures): HCPCS codes C5271, C5275, and C5277; APC 5054 (Level 4 Skin Procedures): HCPCS codes C5273, 15271, 15275, and 15277; or APC 5055 (Level 5 Skin Procedures): HCPCS code 15273. In CY 2025, the payment rate for APC 5053 (Level 3 Skin Procedures) is $612.13, the payment rate for APC 5054 (Level 4 Skin Procedures) is $1,829.23, and the payment rate for APC 5055 (Level 5 Skin Procedures) is $3,660.97. Table 111 lists the APC assignments and CY 2025 payment rates for the HCPCS codes describing the skin substitute application procedures. This information is also available in Addenda A and B of the CY 2025 final OPPS/ASC rule with comment period (the Addenda A and B are available on the CMS website https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices).
Beginning in CY 2016, we adopted a policy where we determine the high-cost/low-cost status for each skin substitute product based on either a product's geometric mean unit cost (MUC) exceeding the geometric MUC threshold or the product's per day cost (PDC), which is calculated as the total units of a skin substitute multiplied by the mean unit cost and divided by the total number of days, exceeding the PDC threshold. We assign each skin substitute that exceed either the MUC threshold or the PDC threshold to the high-cost group. In addition, we assign any skin substitute with a MUC or a PDC that does not exceed either the MUC threshold or the PDC threshold to the low-cost group (87 FR 71976). We also assign skin substitutes with pass-through payment status to the high-cost category.
We assign skin substitutes with some pricing information but without claims data for which to calculate a geometric MUC or PDC to either the high-cost or low-cost category based on the product's ASP plus 6 percent payment rate as compared to the MUC threshold. If ASP is not available, we use the wholesale acquisition cost (WAC) plus 3 percent to assign a product to either the high-cost or low-cost category. Finally, if neither ASP nor WAC is available, we use 95 percent of average wholesale price (AWP) to assign a skin substitute to either the high-cost or low-cost category.
In the CY 2021 OPPS/ASC final rule with comment period, after the first entirely synthetic skin substitute products were introduced into the market, we revised our description of skin substitutes to include both biological and synthetic products (85 FR 86064 through 86067). Any skin substitute product that is assigned to a code in the HCPCS A2XXX series is assigned to the high-cost skin substitute group, including new products without pricing information. New skin substitutes without pricing information that are not assigned a code in the HCPCS A2XXX series are assigned to the low-cost category until pricing information is available to compare to ( printed page 53727) the MUC and PDC thresholds (89 FR 94247).
In the CY 2014 OPPS/ASC final rule, we also noted that several skin substitute products are applied as either liquids or powders per milliliter or per milligram and are employed in procedures outside of CPT codes 15271 through 15278. We stated that these products “. . . will be packaged into the surgical procedure in which they are used.” (78 FR 74930 through 74931).
We also clarified that our definition of skin substitutes does not include bandages or standard dressings, and that, under the OPPS, these items cannot be assigned to either the high-cost or low-cost skin substitute groups or be reported with either CPT codes 15271 through 15278 or HCPCS codes C5271 through C5278 (85 FR 86066).
c. Current FDA Regulation of Products CMS Considers To Be Skin Substitutes
The FDA regulates products that CMS considers to be skin substitutes based on a variety of factors, including product composition, mode of action, and intended use. Relevant categories of FDA regulation for skin substitute products include the following:
(1) Self-Determination Under Section 361 of the PHS Act and the Regulations in 21 CFR 1271 (361 HCT/Ps)
Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps) are defined in 21 CFR 1271.3(d) as articles containing or consisting of human cells or tissues that are intended for implantation, transplantation, infusion, or transfer into a human recipient. Examples include bone, ligament, skin, dura mater, heart valve, cornea, hematopoietic stem/progenitor cells derived from peripheral and cord blood, manipulated autologous chondrocytes, epithelial cells on a synthetic matrix, and semen or other reproductive tissue. Pursuant to section 361 of the Public Health Service (PHS) Act, FDA promulgated regulations at 21 CFR 1271, et seq. that create an electronic registration and listing system for establishments that manufacture HCT/Ps, regulate donor eligibility, and establish current good tissue practice and other procedures to prevent the introduction, transmission, and spread of communicable diseases by HCT/Ps.
A subset of HCT/Ps are those that are regulated solely under section 361 of the PHS Act and the regulations in 21 CFR 1271 (361 HCT/Ps). The FDA has taken a risk-based, tiered approach in regulating HCT/Ps; as the potential risk posed by a product increases, so does the level of oversight (63 FR 26745). Although FDA is authorized to apply the applicable requirements in the Federal Food, Drug, and Cosmetic Act (FD&C Act) and/or the PHS Act to those products that meet the definition of drug, biological product, or device, under a tiered, risk-based approach, HCT/Ps that meet specific criteria or fall within detailed exceptions do not require premarket review and approval. HCT/Ps that do not meet all the criteria in 21 CFR 1271.10(a) are not regulated solely under section 361 of the PHS Act and the regulations in 21 CFR part 1271. Unless an exception in 21 CFR 1271.15 applies, such products are regulated as drugs, devices, and/or biological products under the FD&C Act and/or the PHS Act and are subject to additional regulation, including applicable premarket review and approval. An HCT/P is regulated solely under section 361 of the PHS Act and 21 CFR part 1271 if it meets all of the following criteria (21 CFR 1271.10(a)):
- The HCT/P is minimally manipulated;
- The HCT/P is intended for homologous use only, as reflected by the labeling, advertising, or other indications of the manufacturer's objective intent;
- The manufacture of the HCT/P does not involve the combination of the cells or tissues with another article, except for water, crystalloids, or a sterilizing, preserving, or storage agent, provided that the addition of water, crystalloids, or the sterilizing, preserving, or storage agent does not raise new clinical safety concerns with respect to the HCT/P; and
Either:
++ The HCT/P does not have a systemic effect and is not dependent upon the metabolic activity of living cells for its primary function; or
++ The HCT/P has a systemic effect or is dependent upon the metabolic activity of living cells for its primary function, and:
-- Is for autologous use;
-- Is for allogeneic use in a first-degree or second-degree blood relative; or
-- Is for reproductive use.
Establishments that manufacture 361 HCT/Ps, as defined by 21 CFR 1271.3(e), must register and list their 361 HCT/Ps in the FDA's electronic Human Cell and Tissue Establishment Registration System (eHCTERS), but premarket review and approval by FDA is not needed. However, FDA acceptance of an establishment registration and 361 HCT/P listing form does not constitute a determination that an establishment is compliant with applicable FDA rules and regulations, that the FDA has agreed with the manufacturer's self-determination as a 361 HCT/P, or that the HCT/P is licensed or approved by FDA (21 CFR 1271.27(b)). When this proposed rule refers to 361 HCT/Ps, it generally refers to products where an establishment has self-determined that their product is a 361 HCT/P.[95] If an HCT/P does not meet the criteria set out in 21 CFR 1271.10(a), and the establishment that manufactures the HCT/P does not qualify for any of the exceptions in 21 CFR 1271.15, the HCT/P will be regulated as a drug, device, and/or biological product under the FD&C Act, and/or section 351 of the PHS Act (42 U.S.C. 262), and applicable regulations, including 21 CFR part 1271, and premarket review generally is required.
(2) 510(k) Premarket Notification Submissions, Premarket Approval Applications, and De Novo Requests
“Devices,”, as defined under 21 U.S.C. 321(h)(1), do not achieve their primary intended purposes through chemical action and are not dependent upon being metabolized for the achievement of their primary intended purposes. Devices may be subject to premarket review through: (1) a 510(k) premarket notification submission (510(k)) in accordance with section 510(k) of the FD&C Act and implementing regulations in subpart E of 21 CFR part 807; (2) a premarket approval application (PMA) under section 515 of the FD&C Act and regulations in 21 CFR part 814; or, potentially, (3) a De Novo classification request (De Novo request) under section 513(f)(2) of the FD&C Act and regulations in subpart D of 21 CFR part 860. A 510(k) is a premarket submission made to the FDA to demonstrate that the device to be marketed is substantially equivalent to a legally marketed device that is not subject to premarket approval (sections 510(k) and 513(i) of the FD&C Act). Premarket approval is the most rigorous type of review and generally is required for class III medical devices. Class III devices are those devices for which insufficient information exists to determine that general controls and special controls would provide a reasonable assurance of safety and effectiveness and are purported or represented to be for a use in supporting or sustaining human life or for a use which is of substantial importance in preventing impairment of human health, or present potential unreasonable risk of illness or injury (section 513(a)(1)(C) of the FD&C Act). De Novo classification is a marketing ( printed page 53728) pathway for novel medical devices for which general controls alone (class I), or general and special controls (class II), provide reasonable assurance of safety and effectiveness, but for which there is no legally marketed predicate device. Devices that are classified into class I or class II through a De Novo request may be marketed and used as predicates for future premarket notification (that is, 510(k)) submissions, when applicable.
(3) Biologics License Application
To lawfully introduce or deliver for introduction into interstate commerce a drug that is a biological product, a valid biologics license application (BLA) must be in effect under section 351(a)(1) of the PHS Act, 42 U.S.C. 262(a)(1), unless exempted under 42 U.S.C. 262(a)(3). Such licenses are issued only after showing that the product is safe, pure, and potent. Approval of a biologics license application or issuance of a biologics license shall constitute a determination that the establishment(s) and the product meet applicable requirements to ensure the continued safety, purity, and potency of such products (21 CFR 601.2(d)). Potency has long been interpreted to include effectiveness (21 CFR 600.3(s)).
The definition of the term “biological product” in section 351(i) of the PHS Act is: “a virus, therapeutic serum, toxin, antitoxin, vaccine, blood, blood component or derivative, allergenic product, protein, or analogous product . . . applicable to the prevention, treatment, or cure of a disease or condition of human beings.” (42 U.S.C. 262(i)). In contrast to the registration and listing requirements for a 361 HCT/P or the substantial equivalence requirements for 510(k)s, products licensed under section 351 of the PHS Act are required to meet stringent pre-and post-market requirements to ensure the products' safety and efficacy when marketed. Table 112 lists several other notable differences between the relevant FDA regulatory categories for products CMS considers to be skin substitutes.
d. Payment of Skin Substitute Products Under the PFS and OPPS
1. Payment for Skin Substitute Products as Incident-to Supplies
We have carefully considered our policy objectives, which include: (1) ensuring a consistent payment approach for skin substitute products across the physician office, hospital outpatient department, and ambulatory surgical center (ASC) settings; (2) ensuring that appropriate HCPCS codes describe skin substitute products; (3) employing a uniform approach across products within the physician office setting, regardless of whether the product is synthetic or comprised of human- or animal-based material; and (4) providing clarity for interested parties on CMS skin substitutes policies and procedures. We proposed, starting January 1, 2026, to separately pay for the provision of certain groups of skin substitute products as incident-to supplies when, for those products that are coverable under Medicare's rules, they are used during a covered application procedure paid under the PFS in the non-facility setting or under the OPPS. This proposal does not apply to biological products licensed under section 351 of the PHS Act, which would continue to be paid as biologicals under the ASP methodology in section 1847A of the Act. While we considered proposing to pay separately for skin substitutes initially under just the PFS in non-facility settings consistent with current practice, one of our primary policy objectives is to ensure a consistent payment approach for skin substitute products across the physician office and hospital outpatient department settings; and so, we ultimately determined that the suite of products referred to as skin substitutes should be treated in a uniform manner across different outpatient care settings, to the extent permitted by applicable law, such as section 1833(t)(2)(B) of the Act. The physician, in consultation with his or her patient, decides the site of service for treatment. While many factors are considered as a part of that decision, substantial differences in payment for the application of the same skin substitute product in one site of service versus another, or between similar skin substitute products, should not be one of them. Establishing a consistent framework for how these products are treated within the non-facility and hospital outpatient settings would empower providers to make the best treatment decisions for their patients, ensure equitable access to needed services, and pay appropriately for these services. We also considered bundling payment for skin substitute products in both the PFS and OPPS as part of this proposal. While supplies are generally bundled into the payment of the service in both the physician office and hospital outpatient departments, for many years skin substitute products ( printed page 53729) have been paid separately in the physician office setting, where the majority of these products are currently applied. So, we have determined that bundling payment for skin substitute products with their administration procedures across both settings under this new proposal, before efforts are made to address improper utilization patterns, would be premature. Depending on the outcomes of a final policy, we may consider packaging skin substitute products with the related application procedure in both the hospital outpatient setting and non-facility setting in future rulemaking. We sought comments on our proposal to separately pay for the provision of certain groups of skin substitute products as well as on our proposal to implement this policy in both the non-facility and hospital outpatient settings.
In the CY 2014 OPPS/ASC final rule with comment period, we finalized a policy to package the payment for skin substitutes into high- and low-cost administration codes (see 78 FR 74930 through 74931 and 42 CFR 419.2(b)(16)). Under the proposal in the CY 2026 OPPS/ASC proposed rule, the payment for skin substitutes would no longer be packaged into the administration procedures under the OPPS, when performed in the outpatient hospital setting. Rather, we proposed to remove skin substitutes from the list of packaged items and services at 42 CFR 419.2(b)(16) and specify that we will continue to package payment for products that aid wound healing that are not skin substitute products. Accordingly, the C-codes describing the low-cost group, HCPCS codes C5271 through C5278, would be deleted; and skin substitutes assigned to the high-cost group, described by HCPCS codes 15271 through 15278, would remain to describe skin substitute administration procedures. As a result of the unbundling of the skin substitute products from HCPCS codes 15271 through 15278, the costs associated with the HCPCS codes may be impacted, resulting in changes in APC assignments. We refer readers to Addendum B to the CY 2026 OPPS/ASC proposed rule for the APC assignments and associated payment rates for HCPCS codes 15271 through 15278. We also proposed to combine the existing claims data available for the two sets of current OPPS codes, the low-cost and the high-cost administration groups, to set the initial payment rate for the proposed skin substitute administration procedures described by HCPCS codes 15271 through 15278. We believe it is appropriate to combine the available claims data from both the low-cost and high-cost administration groups to calculate the payment rate for the proposed skin substitute administration procedures as both the low-cost and high-cost groups describe skin substitute administration. While HCPCS add-on administration codes 15272, 15274, 15276, and 15278 would still be packaged in the hospital outpatient setting, because add-on codes are generally packaged in the hospital outpatient setting, we anticipate that many of the concerns expressed by presenters at previous meetings of the Advisory Panel on Hospital Outpatient Payment (HOP Panel) and by public commenters on previous rules that providers are discouraged from treating larger wounds in the hospital outpatient setting (89 FR 94247) would be addressed by our proposal to pay separately for codes describing provision of skin substitute products from their associated administration codes. We sought comment on our proposal to pay separately for provision of skin substitutes as incident-to supplies when used as part of an administration procedure in the hospital outpatient setting.
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: Many commenters expressed strong support for CMS' goal of aligning payment and coding policies across different sites of care. They agree that a consistent, site-neutral payment system empowers providers to choose the most clinically appropriate setting based on patient needs rather than financial incentives. Commenters noted this would reduce the confusion, administrative burden, and disparities in care caused by the current system. They see this policy as a way to establish a fair and uniform standard that centers treatment decisions around the patient.
Many commenters also supported CMS' proposal to pay for skin substitutes separately from the application procedure. Several stated that these products add significant clinical value. They noted that separate payment ensures consistent treatment availability, preserves access for patients, and avoids disincentives for treating larger wounds. Several commenters expressed appreciation that CMS did not move forward with a previous proposal to bundle the products, which they believe would have harmed patients with large wounds. They regarded the proposed policy as a more clinically and operationally sound approach.
Several commenters specifically supported discontinuing the bundling of skin substitute payments in the Hospital Outpatient Department (HOPD) and Ambulatory Surgical Center (ASC) settings. They argued the current system, which bundles payment in the HOPD and ASC settings, but pays separately in the office, has created a disincentive to treat larger wounds in facilities, pushing those cases disproportionately into the physician's office or even more expensive inpatient settings. They applauded the per-square-centimeter methodology for aligning payment with wound size and expanding access across all sites.
Response: We thank commenters for their support.
Comment: Other commenters opposed the move away from bundled payments. MedPAC, for example, did not support unbundling skin substitutes in the facility setting, arguing that paying for items separately undermines payment bundles, can lead to overuse, and shifts financial burden from providers to Medicare and its beneficiaries. Another commenter supported maintaining a bundled approach to align with longstanding policy and statutory authority.
Several commenters recommended that CMS delay any changes to the HOPD payment methodology. They suggested that CMS should first assess the impact of the payment reforms in the non-facility setting before applying them to the hospital outpatient setting to avoid unintended consequences, such as shifting care to more expensive settings.
Response: While we acknowledge the concerns about unbundling raised by commenters, ensuring a consistent payment approach for skin substitute products across the physician office and hospital outpatient department settings has been a long-stated policy objective. As noted above, we determined that it would be premature to bundle payment for skin substitute products with their administration procedures across both settings before efforts are made to address improper utilization patterns. Depending on the outcomes of this policy, we may consider packaging skin substitute products with the related application procedures in the hospital outpatient department setting, ambulatory surgical center setting, and non-facility setting in future rulemaking.
Comment: Many commenters warned that the proposed payment rate for the application codes create a new, major disparity between care settings. They pointed out the large gap between the proposed physician application ( printed page 53730) payment rate of ~$150 in an office/mobile setting and the combined facility and physician facility setting payment rates of over $800 in an HOPD. They stated this disparity will create a strong financial incentive to shift patient care to the more expensive HOPD setting, which could strain hospital capacity, create access issues for rural and underserved patients, and cause physician offices and mobile practices to shutter. Several commenters highlighted the unique challenges and higher costs faced by providers serving rural and homebound patients. They recommended CMS offer financial incentives, enhance telemedicine reimbursement, and provide add-on payments or grants to ensure these vulnerable populations do not lose access to care. To achieve true site neutrality, they strongly suggested CMS increase the application payment for clinicians in non-facility settings to close this gap.
Other commenters supported the proposed RVUs and payment rates for the application procedure codes in both the HOPD and MPFS settings, finding them to be a fair assessment of clinical resource utilization.
Another commenter stated that CMS should increase the proposed APC payment rates for these procedures in the HOPD setting, as the proposal demotes some codes to lower-paying APCs, which exacerbates existing disincentives for treating wounds in the hospital. The commenter also stated that we had not provided any rationale for reassigning skin substitute application procedures to APCs with lower payment rates. The commenter requested that we revert the APC assignments for CPT code 15271, 15273 and 15275 to the APC assignments finalized in the CY 2025 OPPS/ASC final rule. Specifically, the commenter requested that we assign CPT codes 15271 and 15275 to APC 5054 and assign CPT code 15273 to APC 5055.
One commenter sought clarification on whether APC rates for the skin substitute application procedures, such as CPT codes 15271 through 15278 would be adjusted to maintain budget neutrality.
Response: We acknowledge the disparities in the payment rates for the application codes between settings. As described in section II.B. of this final rule with comment period, we are open to exploring alternative data sources, including use of OPPS cost data, to inform PFS rate setting for certain services in future rulemaking. We also recognize the possibility raised by interested parties that some of the excessive payment for the skin substitute products may have been useful in subsidizing costs associated with providing these services in beneficiaries' home. We look forward to continued dialogue on this point as well as on the point of access to care for homebound and other beneficiaries for whom care is reasonable and necessary.
With regard to specific APC assignments for some of the skin substitute application procedures (HCPCS codes 15271-15278), we remind commenters that we provided a rationale for potential APC reassignments. Specifically, we explained in the CY 2026 OPPS/ASC proposed rule that, as a result of the unbundling of the skin substitute products from HCPCS codes 15271-15278, the costs associated with the HCPCS codes may be impacted, resulting in changes in APC assignments. Such was the case when we ran an updated analysis of the geometric mean costs of HCPCS codes 15271-15278, upon which we update APC assignments, when we unbundled the skin substitute products from the application codes. Once we removed the costs of the skin substitute products, we saw approximately a 50 percent decrease in the geometric mean cost of each of the HCPCS application codes. Stated differently, when skin substitute products were packaged into the payment for the application procedure, they accounted for a significant portion of the cost of the procedure. With the product no longer packaged and instead separately paid, it would be expected that the cost of the procedure would decrease, because the procedure no longer includes the cost of the skin substitute product. Table 113 illustrates this in more granular detail.
We note that, while additional claims have been processed since the CY 2026 OPPS/ASC proposed rule was released, there have not been significant changes to the geometric mean costs of these codes as outlined in the costs statistics files to this final rule available on the CMS OPPS website.
Finally, we are clarifying that the prospective payments for skin substitute products and skin substitute administration procedures based on our CY 2026 OPPS payment policy would be included in budget neutrality as it is typically applied. For more detail, we refer readers to section II.A.4 of this final rule with comment period.
After careful consideration of the comments, we are finalizing our proposal to pay separately for the ( printed page 53731) provision of certain groups of skin substitute products as well as our proposal to implement this policy in both the non-facility and hospital outpatient settings as proposed. We are also finalizing the proposed APC assignments for HCPCS codes 15271, 15273, 15275, and 15277. Specifically, we are finalizing our proposal to assign HCPCS codes 15271 and 15272 to APC 5053 (Level 3 Skin Procedures) and HCPCS codes 15273 and 15277 to APC 5054 (Level 4 Skin Procedures). The final CY 2026 payment rates can be found in Addendum B to this final rule with comment via the internet on the CMS website. In addition, we refer readers to Addendum D1 of this final rule with comment period for the status indicator meanings for all codes reported under the OPPS. Addendum D1 can also be found via the internet on the CMS website.
In the CY 2014 OPPS/ASC final rule with comment period, we finalized a policy to treat skin substitutes as biologicals that function as supplies when used in a surgical procedure. Similarly, under this proposal, most skin substitutes would be considered incident-to supplies in accordance with section 1861(s)(2)(A) of the Act. Supplies are a large category of items that typically are either for single use or have a shorter use life span than equipment. Supplies can be anything that is not equipment and include not only minor, inexpensive, or commodity-type items but also include a wide range of products used in outpatient settings, including certain implantable medical devices. “Incident-to supplies” refers to supplies that are furnished as an integral, although incidental, part of the physician's personal professional services in the course of diagnosis or treatment of an injury or illness, among other requirements at 42 CFR 410.26(b). Because a skin substitute must be used to perform any of the procedures described by a CPT code in the range 15271 through 15278, and the procedure of treating the wound and applying a covering to the wound is the independent service, skin substitute products serve as a necessary supply for these surgical repair procedures.
One purpose of the new proposed policy was to limit some of the current profiteering practices occurring in this industry. For example, as reflected in the last several years of CMS' ASP Pricing Files, we have observed a dramatic increase in launch prices. It is unclear how these prices could be attached to realistic changes in resource costs as many of these new products are allegedly minimally manipulated tissues. Our policy is likely to disincentivize this practice, as well as several other novel industry practices that have come to our attention, by preventing exploitation of skin substitute pricing under section 1847A of the Act, overuse of expensive skin substitute products, and waste resulting from use of more expensive skin substitute products over clinically-appropriate, less expensive alternatives. Notably, there has not been significant growth in payments for skin substitutes in the OPPS, due in part to our packaging principles. We note that the relevant statutory provisions, when considered together, do not require all of these kinds of products to be paid as biologicals under section 1847A of the Act. Therefore, under this policy, unless a skin substitute is approved as a biological product under section 351 of the PHS Act, in which case we would continue to pay for it consistent with section 1847A of the Act, we would consider it a supply for payment purposes under the OPPS with definitions and rates described below. For Medicare purposes, we proposed to codify the definition of “biological” as “a product licensed under section 351 of the Public Health Service Act” at §§ 414.802 and 414.902. We sought comments on our proposal to limit the application of section 1847A of the Act to skin substitutes that are approved as a biological product under section 351 of the PHS Act and our proposed edits to the regulations.
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: Many commenters opposed classifying skin substitutes, particularly amniotic and placental-derived products, as supplies. They stated this classification is a misstep that diminishes the products' clinical value, calling the products advanced, life-changing biologic therapies, not simple bandages or dressings. Some stated CMS lacks legal authority, as skin substitutes do not meet the statutory definition of an “incident-to supply.” They stated the reclassification is legally, clinically, and definitionally incorrect, as these products are the primary intervention, not an incidental part of a service, and interact directly with body tissues.
Response: Section 1861(s)(2)(A) of the Act defines “medical and other health services” as services and supplies furnished incident to a physician's professional service that are commonly furnished in physicians' offices and provided either without charge or as part of a physician's bill. As stated above, supplies are a large category of items that typically are either for single use or have a shorter use life span than equipment. Supplies can be anything that is not equipment and include not only minor, inexpensive, or commodity-type items but also include a wide range of products used in outpatient settings, including certain implantable medical devices such as class III medical devices requiring premarket approval, which is the most rigorous review required of a skin substitute that is also a medical device. “Incident-to supplies” refers to supplies that are furnished as an integral part of the physician's professional services in the course of diagnosis or treatment of an injury or illness, among other requirements at 42 CFR 410.26(b). Because a skin substitute must be used to perform any of the procedures described by a CPT code in the range 15271 through 15278, and the procedure of treating the wound and applying a covering to the wound is the independent service, skin substitute products serve as a necessary supply for these surgical repair procedures.
Comment: Many commenters strongly supported the classification of non-BLA skin substitutes as incident-to supplies. They see it as an essential, rational approach to realign incentives away from overuse and toward more clinically appropriate care. They stated that this classification is expected to curb the significant fraud, waste, and abuse that has plagued the current system.
Response: We appreciate the commenters for their feedback on our proposal to treat and pay for all skin substitute products as incident-to supplies. We agree that this proposal will help to resolve issues such as exorbitant price increases and curb fraud, waste, and abuse.
After careful consideration of the comments, we are finalizing our proposal to pay separately for the provision of skin substitutes as incident-to supplies under the OPPS in the hospital outpatient department as proposed.
We received divided comments on our proposal to codify the definition of “biological” in regulation. The following is a summary of the comments we received and our responses.
Comment: Several commenters stated that CMS's proposal to narrow the definition of “biological” to only include products licensed under section 351 of the PHS Act is a misapplication of the law and conflicts with the Act. They contend that the of the Act itself provides a broader definition in section 1861(t)(1) of the Act, which includes products listed in the U.S. Pharmacopoeia (USP) or approved by hospital P&T committees—a definition ( printed page 53732) that many skin substitutes meet. Commenters state that if Congress had intended to limit the term “biological” to section 351 of the PHS Act products in the payment statute (section 1847A of the Act), it would have done so explicitly, as it has in other parts of the law. Furthermore, they noted that the Consolidated Appropriations Act of 2021 referred to these products as “drugs and biologicals,” signaling that the Congress considers them as such for payment purposes. Commenters stated that for decades CMS has appropriately classified and paid for CTPs/skin substitutes as drugs or biologicals under section 1847A of the Act. They state the proposal to abruptly reclassify them is an unexplained reversal of this long-standing policy and is therefore “arbitrary and capricious.”
Response: Section 1861(t)(1) of the Act states, in relevant part, that the term “drugs” and the term “biologicals” include only products that are included (or approved for inclusion) in the U.S. Pharmacopoeia, the National Formulary, or the United States Homeopathic Pharmacopoeia, or in New Drugs or certain products listed in the Accepted Dental Remedies, or as are approved by the pharmacy and drug therapeutics committee (or equivalent committee) of the medical staff of the hospital furnishing such drugs and biologicals for use in such hospital; subparagraph (t)(2) adds that the term “drugs” includes any drugs or biologicals used in certain anticancer chemotherapeutic regimens in the definition. The definition of these terms does not include certain medical supplies. Reliance on this provision to determine that a skin substitute is a biological payable under section 1847A of the Act is problematic for several reasons. To begin, we note that this provision does not require that all products included in the listed compendia are deemed drugs and biologicals. Instead, it states that “drugs” and “biologicals” include only such drugs and biologicals as are included in the compendia. Second, only one of the listed compendia in section 1861(t)(1) of the Act that is still in publication, a combination compendium containing USP and the National Formulary (USP-NF), which contains standards for medicines, dosage forms, drug substances, excipients, biologics, compounded preparations, medical devices, dietary supplements and other therapeutics.[101] The compendia issued a statement in 2018 that it would no longer develop new monographs for biologics unless there is other interested parties consensus supporting its creation, including the support of FDA.[102] As a result, very few of CMS' paid biologicals actually have product-specific monographs in that compendium. Instead of product-specific monographs, the USP primarily develops performance standards and general guidelines to support the quality assessment of biologics. This is also true in the case of skin substitutes. References to these types of products in the USP are not product-specific monographs. Instead, these references are general descriptions of product types. As the source and manufacture of products with biological activity can dramatically change their safety and efficacy, these general references are not sufficient to describe any product with specificity. Therefore, we rely on the language in section 1847A of the Act to authorize payment for products described therein.
The Consolidated Appropriations Act, 2021, Public Law 116-260, division CC, section 401(c), amended section 1847A(f)(A) to state that, manufacturers of drugs or biologicals including items, services, supplies, and products that are payable under Medicare Part B as a drug or biological, that have not entered into a National Medicaid Drug Rebate Agreement are required to report ASP (and WAC) data to CMS. Under this policy, as finalized, skin substitute products (other than those approved via BLA under section 351 of the PHS Act) will no longer be paid as biologicals and will no longer be required to report ASP to us.
Finally, as noted previously, we outlined our HCPCS Level II coding and payment policy objectives for skin substitutes in the CY 2023 PFS proposed rule (87 FR 46249) and stated we believed that our existing payment policies were unsatisfactory, unsustainable over the long term, and rooted in historical practice established two decades ago prior to significant evolutions in medical technology and practice. CMS also hosted a town hall to provide an opportunity for public input, including discussion of potential approaches to the methodology for payment of skin substitute products, and reviewed several years of comments in response to CY rulemaking in 2023, 2024, and 2025 on this subject before developing this proposal to address our stated objectives as well as many of the comments we have received.
Comment: Other commenters agreed with CMS' interpretation. They supported the proposal to reserve the payment methodology under section 1847A of the Act for products that have undergone the rigorous FDA Biologics License Application (BLA) process under section 351 and agreed that non-BLA products do not meet the statutory definition of biologicals contemplated for payment under section 1847A of the Act, making the “incident-to supply” classification a rational approach. They believe this accurately reflects the statutory definition of a biological and rightly rewards manufacturers who invest in the highest level of regulatory review. Other commenters requested that CMS clarify that skin substitute products do not fall under the discarded drug or inflation rebate policies.
Response: We appreciate the commenters for their support. We clarify that skin substitute products that are not regulated as biological products under section 351 of the PHS Act and that are paid as incident to supplies are not subject to the Medicare discarded drug policy. At this time, skin substitutes are excluded from Part B inflation rebates as described at § 427.101(b)(5) and as finalized in the CY 2025 PFS final rule (89 FR 98235).
Comment: A commenter warned that continuing to pay for the few BLA-approved products under section 1847A of the Act while moving all others to a flat rate would create a perverse incentive for those manufacturers to continue increasing prices.
Response: As previously described, we believe that the payment methodology described in section 1847A of the Act applies to drugs and biological products approved under a BLA. Further, such licenses are issued only after showing that the product is safe, pure, potent, and may justify a higher payment rate. Finally, between the longer time required to bring these products to market, potential rebate requirements, and the changes to ASP reporting described in the CY 2026 PFS final rule, we believe opportunities for dramatic pricing increases will be significantly curtailed. However, we will continue to monitor pricing trends for products approved under a BLA.
Comment: A commenter requested that CMS codify in regulation that biological products licensed by the FDA under the section 351 BLA process are not considered skin substitutes, are not considered incident-to supplies under the CY 2026 OPPS/ASC proposed rule, and will continue to be eligible for separate payment under section 1847A of the Act (generally, ASP plus 6 percent). ( printed page 53733)
Response: While products licensed under section 351 of the PHS Act may properly be used along with skin substitute application codes, they will continue to be separately paid as biological products using the methodology described in section 1847A of the Act. We also received many comments on the growth in payments for this class of products.
Comment: Many commenters, including ACOs, primary care providers, and health systems, stated that they have witnessed an explosive and unsustainable growth in skin substitute spending, which they attribute to fraud, waste, and abuse. They report seeing products used in clinically questionable circumstances, often by third-party mobile wound clinics that operate without coordination with the patient's primary care team. The commenters identified the ASP-based payment limits as a primary driver of abuse, as it creates financial incentives to use more expensive products, regardless of clinical need. They described a system where manufacturers can launch new, clinically undifferentiated products at inflated prices and offer deep discounts to providers, who then profit from the spread between their acquisition cost and the high Medicare reimbursement rate. Commenters provided examples of significant patient harm resulting from this misuse, including failure to treat the underlying causes of wounds, unnecessary applications, severe infections, sepsis, and even death. They noted a troubling pattern of skin substitutes being applied to vulnerable and terminally ill patients, including those on hospice, where such treatment is inappropriate.
Response: We appreciate the commenters for their input. We believe this policy will dramatically reduce these problematic behaviors in both the physician office and hospital outpatient settings. We also believe this policy has the potential to prevent these harmful practices from occurring in different settings of care, including hospice and home health.
Comment: Several commenters referenced analyses indicating that the vast majority of the spending is driven by a very small number of outlier providers. Commenters referenced one analysis by Tettelbach et al.[103] that found that in 2023, fewer than 3 percent of providers accounted for nearly two-thirds of all Medicare spending on these products. The commenters suggested the problem is not broad utilization but isolated misuse by a few bad actors. Based on the concentration of abuse, some commenters stated that CMS should use targeted program integrity measures, such as audits of outlier providers, NPI-level analytics, and stricter enforcement—rather than implementing sweeping payment cuts that penalize all providers and risk harming patient access. Beyond targeted enforcement, commenters recommended systematic oversight mechanisms. A commenter suggested CMS recognize site accreditation through a self-regulatory organization (SRO) to verify adherence to standard of care, documentation protocols, and product handling, analogous to DMEPOS and CLIA accreditation models. The SRO would conduct inspections, publish outcomes dashboards, and impose sanctions. The commenters contend that slashing payment rates is not a fraud control measure and will disproportionately harm compliant providers and the patients who need these products.
Some commenters caution that increased use of skin substitutes is not, in itself, a negative trend. They stated that the growth also reflects expanded access to care for previously underserved populations (like homebound patients) and increased provider awareness of the products' efficacy in healing chronic wounds.
Response: We agree that not all increased use of skin substitutes is improper. However, it is clear that the dramatic growth in spending is not statutorily required and comes without a clear, consistent, and corresponding benefit. The Agency has a responsibility to the public to be good stewards to the Medicare Trust Fund, so CMS has implemented a coordinated effort across several Centers, such as the Center for Medicare, the Center for Clinical Standards and Quality, and the Center for Program Integrity, to address this issue.
Comment: Several commenters offered feedback on skin substitute billing as it relates to value-based care programs such as the Medicare Shared Savings Program. These comments acknowledged support for the proposals relating to the changes in skin substitute payment policy, but expressed concern related to negative impacts to some ACOs who may be disproportionately impacted by skin substitute billing compared to the national trend. Some commenters also made recommendations for revisions to value-based care programs that could address skin substitute billing and similar future scenarios that may not be captured by the Significant, Anomalous, and Highly Suspect (SAHS) billing activity policy which was finalized in the CY2025 PFS.
Response: As the commenters referenced, on November 1, 2024, we issued the CY 2025 Medicare PFS final rule (89 FR 97710), which included policies discussed under the “Mitigating the Impact of Significant, Anomalous, and Highly Suspect (SAHS) Billing Activity on Shared Savings Program Financial Calculations in Calendar Year 2024 or Subsequent Calendar Years” section of the final rule (89 FR 98191). These policies give us the ability to determine that the billing of one or more HCPCS or CPT codes represents significant, anomalous, and highly suspect billing activity for a calendar year that warrants adjustment to calculations made under 42 CFR part 425. Generally, a level of billing for a given HCPCS or CPT code is considered SAHS billing activity when a given HCPCS or CPT code exhibits a level of billing that represents a significant claims increase, either in the volume or dollars, with national or regional impact, and represents a deviation from historical utilization trends that is unexpected and is not clearly attributable to reasonably explained changes in policy or the supply or demand for covered items or services. The billing level must be significant and represent billing activity that would cause significantly inaccurate and inequitable payments and repayment obligations in the Shared Savings Program if not addressed (89 FR 98195).
We assessed the impact of an increase in billing to Medicare for skin substitutes and determined that the billing activity for these services does not represent SAHS billing activity for Performance Year (PY) 2024. Skin Substitute billing can have varying impacts on ACOs' performance and could either contribute to increasing or decreasing shared savings and losses, dependent on ACO-level expenditures and national/regional billing activity impacts.
We established the SAHS billing policy to address certain unexplained billing anomalies that could impact program wide calculations, to be invoked in rare and extreme cases when we identify a code that meets the high bar to be defined as SAHS billing activity (89 FR 98196). Payments that are not excluded under the SAHS policy are also reviewable at the ACOs' request if improper payments are identified after the initial determination is made under the reopening policy (42 CFR 425.315). ( printed page 53734)
We will continue to monitor this area with our program integrity partners and to explore options that could mitigate extreme deviations in costs that are outside of the ACOs' control and not addressed through the SAHS policy.
Comment: Some commenters acknowledged that truncation in Medicare Shared Savings Program calculations greatly mitigates a significant amount of outlier billing for skin substitutes. A portion of these commenters also suggested that CMS should apply a lower stop loss truncation threshold in the Medicare Shared Savings Program to address skin substitutes.
Response: To minimize variation in catastrophically large claims, the Medicare Shared Savings Program truncates an assigned beneficiary's total annual Medicare Parts A and B fee-for-service (FFS) per capita expenditures at the 99th percentile of national Medicare Parts A and B FFS expenditures as determined for the applicable performance year for assignable beneficiaries identified for the 12-month calendar year corresponding to the performance year (42 CFR 425.605(a)(3) and 425.610(a)(4)(ii)).
For all benchmark years and performance years, CMS provides ACOs with the number of assigned beneficiaries with truncated expenditures, the total dollar amount truncated, and the percentage of total annualized expenditures truncated. We also provide this truncation information for the National Assignable FFS population. We have examined the impact of the truncation policy on skin substitute expenditures and found that over 50 percent of PY 2024 Part B expenditures for skin substitutes were addressed by truncation. Skin substitute expenditures, on average for PY 2024, represent roughly 1 percent of total Parts A and B expenditures for ACOs, and with truncation applied, the average skin substitute expenditures equate to less than 0.5 percent of total Parts A and B expenditures for ACOs. This information indicates that truncation effectively mitigates large spending associated with skin substitute billing.
Comment: Some commenters also provided feedback requesting a formal process and direct channel for ACOs to report fraud.
Response: Medicare Shared Savings Program ACOs are encouraged to report potential fraud or abuse by submitting a complaint to the CMS Center for Program Integrity (CPI), Fraud Investigations Group (FIG), Division of Provider Investigations (DPI) at dpi.intake@cms.hhs.gov. ACOs can also report potential fraud or abuse by submitting a complaint to the Office of Inspector General (OIG) website at https://oig.hhs.gov/fraud/report-fraud/, OIG hotline at 1-800-HHS-TIPS (1-800-447-8477), TTY at 1-800-377-4950, by fax at 1-800-223-8164, or by mailing to: Office of Inspector General ATTN: OIG HOTLINE OPERATIONS P.O. Box, 23489 Washington, DC 20026. ACOs suspecting healthcare fraud, waste, or abuse are encouraged to visit the CMS Center for Program Integrity (CPI) website at https://www.cms.gov/medicare/medicaid-coordination/center-program-integrity for more information.
After careful consideration of public comments, we are finalizing our proposal to limit application of section 1847A of the Act to skin substitutes that are approved as a biological product under section 351 of the PHS Act and we are finalizing our proposed edits to the regulations as proposed.
2. Payment Categories Based on FDA Regulatory Pathways
Paying separately for skin substitutes in the non-facility setting has led to dramatic price increases for these products, as noted above. Grouping similar products or services into a single billing code and using a single payment amount for them, as we do with many services under the OPPS, some services under the PFS, and all multiple-source drugs under section 1847A of the Act, incentivizes hospitals and prescribers to make more cost-efficient, clinically effective decisions. However, we recognize that grouping dissimilar products and/or services to set payment rates, can limit beneficiaries' access to appropriate care, especially when some groups encompass products and services with significant clinical and resource variability. In the case of skin substitutes, no single product among the wide range of products stands out as typical; so, we have reviewed several methods to group or classify skin substitutes to determine which best reflects clinical and resource similarities between these products.
We proposed that only skin substitute products licensed under section 351 of the PHS Act will be considered drugs and biologicals for Medicare payment purposes. Furthermore, we proposed that, to reflect relevant product characteristics, we would group skin substitutes that are not drugs or biologicals (that is, anything that is not a section 351 product) using three CMS payment categories based on FDA regulatory categories ((PMAs, 510(k)s, and 361 HCT/Ps) to set payment rates. We have previously noted in rulemaking that CMS has no obligation to categorize products based on the FDA's current regulatory framework (74 FR 60476); but, in this case, we have determined that the FDA regulatory categories provide an appropriate level of distinction for a heterogeneous category of products that exhibit clinical and resource variability and that categorizing products based on these categories can ultimately improve the accuracy of payment under the OPPS. Proposing a payment policy that aligns with FDA's current regulatory framework also provides predictability and efficiency for purposes of Medicare payment. Payment for new products, as discussed below, could be achieved quickly and consistently by CMS' capacity to immediately recognize the FDA regulatory categories.
a. 361 HCT/Ps
As described previously, 361 HCT/Ps are a subset of HCT/Ps that are regulated solely under section 361 of the PHS Act and the regulations in 21 CFR 1271 and listed in the FDA's eHCTERS. Currently, registered 361 HCT/Ps generally are dressings intended only to cover and protect a wound. They are not intended to act on the wound to mediate, facilitate, or accelerate wound healing. Their activity is typically limited to that of a physical covering or wrap. A structural tissue intended for wound care is generally limited to the homologous use of cover and protect in order to be a 361 HCT/P.[104] Intended uses such as wound treatment, promotion or acceleration of wound healing, or serving as a skin substitute would generally be non-homologous uses of structural tissues. Instead, products for such intended uses (for example, the treatment of wounds) generally are subject to PMA or BLA requirements.
b. Devices Requiring 510(k) Clearance
A 510(k) is a premarket submission made to the FDA generally by the manufacturer of a device to demonstrate that the device to be marketed is substantially equivalent to legally marketed device that is not subject to premarket approval. (FD&C Act sections 510(k),513(i)). Currently, 510(k)-cleared devices that we are considering for purposes of this proposal generally are dressings intended only to cover and protect a wound, to absorb exudate, and to maintain appropriate moisture balance within the wound. They are not intended to act on the wound to mediate, facilitate, or accelerate wound ( printed page 53735) healing. Their activity is typically limited to that of a physical covering or wrap. When intended only to cover and protect a wound, to absorb exudate, and to maintain appropriate moisture balance within the wound and otherwise meeting the device definition, generally the FDA's Center for Devices and Radiological Health (CDRH) regulates wound dressings composed of natural biomaterials, including animal and human derived tissue as devices, and they are currently subject to 510(k) requirements. At this time, wound dressings have not been 510(k) cleared by FDA for indications such as wound treatment, promotion or acceleration of wound healing, or serving as a skin substitute.[105] Instead, products for such intended uses generally are subject to PMA or BLA requirements.
For the purposes of this policy, we proposed to group any skin substitutes authorized through the De Novo pathway with those cleared under 510(k)s. Similar to products cleared under 510(k)s, De Novo classification is a marketing pathway for medical devices for which general controls alone (class I), or general and special controls (class II), provide reasonable assurance of safety and effectiveness. While products authorized through the De Novo pathway have no legally marketed predicate device, devices that are classified into class I or class II through a De Novo request may be marketed and used as predicates for future premarket notification (that is, 510(k)) submissions, when applicable. Because of this, we would expect skin substitutes authorized through the De Novo pathway and those cleared under 510(k)s to be similar for payment purposes. We sought comment on our proposal to group skin substitutes into three FDA approval categories, PMA, 510(k), and 361 HCT/P, to set payment rates and our proposal to group any skin substitutes authorized through the De Novo pathway with those cleared under 510(k)s for payment purposes.
c. Products Subject to PMAs
Premarket approval is the most rigorous type of review of a device and generally is required for class III medical devices. Similar to BLA-approved wound care products, PMA-approved wound care products generally are intended to go beyond a simple wound cover to provide some type of direct treatment effect. The FDA has not defined the term “skin substitute.” However, the term has been used as a descriptor for certain wound care constructs that are currently approved under a BLA or PMA for treatment of burns or skin ulcers, including ulcers that appear to have failed to heal after standard of care. The intended uses of these products may include scaffold claims, reference to matrix attributes that promote endogenous cell binding, migration, differentiation, or proliferation, and/or activities mediated by matrix-associated regulatory factors that facilitate wound healing. Currently, wound care products intended to interact with the wound to facilitate, promote, or accelerate wound healing generally require approval of a BLA or, in some instances, a PMA. Approval of these products requires demonstration of safety and efficacy for the intended use, which generally requires the performance of clinical studies. So PMA-approved devices can be readily distinguished from 510(k)-cleared devices and 361 HCT/P products, which are intended mainly to cover and protect the wound. They are clinically different, provide different benefits, and would theoretically be used for patients presenting with different clinical scenarios. As discussed, PMA-approved devices also go through a much more rigorous review process before marketing as compared to the substantial equivalence requirements for 510(k)s and lack of premarket review for registered 361 HCT/Ps. This more rigorous review for PMAs, as well as differences in clinical utility, and the associated costs to manufacturers, suggests that the resources involved in furnishing these products could be distinct from 361 HCT/Ps and 510(k)s. We sought comment on our proposal to group skin substitutes (other than those approved via BLA under section 351 of the PHS Act) into three FDA categories, PMA, 510(k), and 361 HCT/P, to set payment rates.
Section 1833(t)(2) requires the Secretary to establish groups so that services classified within each group are comparable clinically and with respect to the use of resources. To effectuate this categorization into a payment policy under the OPPS, we proposed to create three new APCs for HCPCS codes that describe skin substitute products organized by clinical and resource similarity. These three APCs would divide skin substitutes by their FDA regulatory pathway. Specifically, we proposed to create: APC 6000 (PMA Skin Substitute Products); APC 6001 (510(k) Skin Substitute Products); and APC 6002 (361 HCT/P Skin Substitute Products). In addition, as noted previously, we proposed to assign any skin substitutes approved through the De Novo pathway to APC 6001 (510(k) Skin Substitute Products) based on our proposed policy of categorizing products with these two regulatory statuses together. We also proposed to create three new unlisted Q-codes, one to describe skin substitute products in each approval pathway, for new skin substitute products that have received FDA approval or clearance but do not yet have their own code in effect. We proposed to create HCPCS codes Q4431 (formerly placeholder code QXXX1) (Unlisted PMA skin substitute product) and assign it to APC 6000 (PMA Skin Substitute Products); Q4432 (formerly placeholder code QXXX2) (Unlisted 510(k) skin substitute product) and assign it to APC 6001 (510(k) Skin Substitute Products); and Q4433 (formerly placeholder code QXXX3) (Unlisted 361 HCT/P skin substitute product) and assign it to APC 6002 (361 HCT/P Skin Substitute Products). We proposed to create these unlisted codes to prevent delays in Medicare payments for new FDA-approved or cleared skin substitute products. We note that unlisted codes should only be reported when there is no other existing CPT or HCPCS code that adequately describes the service being performed.
We note that device pass-through payment status would still be available to new skin substitutes that meet the pass-through payment criteria in the hospital outpatient setting. However, while skin substitutes approved under device pass-through payment status are currently assigned to the high-cost category, because our proposal would eliminate the low- and high-cost groups, we proposed to pay for skin substitutes approved under device pass-through payment status consistent with other devices approved under that payment pathway. For the purposes of eligibility of skin substitutes for transitional drug pass-through payment, we proposed to define the term “biological” consistent with our interpretation of the term under section 1847A of the Act. Under this proposal, skin substitutes with an approved BLA would be considered under transitional drug pass-through payment status and skin substitutes with PMA or 510(k) clearance would continue to be evaluated under transitional device pass-through payment status. See section IV.A. of this final rule with comment period for more information on device pass-through payments under the OPPS and see ( printed page 53736) section V.A. of this final rule with comment period for more information on drug pass-through payments under the OPPS.
Comment: Many commenters support grouping skin substitutes based on their FDA regulatory categories, viewing it as a clear, logical, and transparent approach. They believe this method acknowledges the different levels of scientific rigor and evidence required for each pathway and can serve as a surrogate for CMS' own evidence review. This framework would allow CMS to differentiate payment over time based on product characteristics and clinical value, which could incentivize competition and innovation. Some suggest a tiered payment structure where products with more rigorous review (like PMA) receive the highest rates, followed by 510(k) and then 361 HCT/P products. One commenter noted that utilizing FDA's existing regulatory paths and associated compliance activities avoids unnecessary duplication of product assessment resources.
Response: We appreciate the commenters for their support.
Comment: Many commenters opposed using FDA pathways to determine payment, stating that regulatory status does not correlate with clinical effectiveness, outcomes, or resource use. Some commenters noted that none of the skin substitute products approved via the PMA pathway have indications for wound healing in their Instructions for Use or FDA intended use/indications. They point out that some older PMA devices have outdated clinical data and may not be superior to newer 361 HCT/P products or products cleared via the 510(k) pathway. Several commenters stated that 510(k) clearance follows a less intensive pathway than PMA but emphasized that the FDA would never permit any device for market use unless the 510(k) submission sufficiently demonstrates patient safety and clinical efficacy. Some commenters noted that receiving 510(k) clearance does not imply inferior quality to a PMA product, and that it is inappropriate to consider a PMA product superior simply because it went through more testing. Another commenter explained that a 510(k) device can sometimes be superior to a PMA device for the same indication despite the less rigorous approval process, due to technological advancements, improved materials and design, real-world data, and improved usability and safety features. Several commenters stated that establishing payment based on regulatory pathways creates potential for instability over time because FDA pathway choice is not voluntary—FDA determines which pathway is appropriate for which product. A commenter stated that CMS already determined that FDA approval pathways are not appropriate for Medicare payment policy decisions in its CY 2014 OPPS/ASC final rule with comment period, referencing the discussion at 78 FR 74933 regarding CMS' decision not to use the FDA regulatory pathway to determine OPPS skin substitute payment policy. Overall, some commenters believed this policy could entrench outdated classifications, create perverse incentives to choose products based on reimbursement instead of clinical evidence, and penalize innovative products that use newer, more streamlined regulatory pathways.
Response: We disagree. The FDA's regulatory framework in this context provides an objective and consistent basis on which to group these products for purposes of development payment rates. Each regulatory path is distinct and provides a specific level/type of information regarding product content and activity that CMS can leverage to inform payment rate decisions. For example, registered 361 HCT/Ps are not approved, cleared or licensed by FDA. There is no premarket review, and manufacturing controls are focused on prevention of infectious disease transmission. These products are often dressings generally intended only to cover and protect a wound. They are not intended to act on the wound to mediate, facilitate, or accelerate wound healing. Similarly, 510(k)-cleared devices [106] relevant to this policy generally are dressings intended only to act as a physical cover to protect a wound, to absorb exudate, and to maintain appropriate moisture balance within the wound. As for 361 HCT/Ps relevant to this policy, activity claims are typically limited to that of a physical covering or wrap. They are also not intended to act on the wound to mediate, facilitate, or accelerate wound healing. The 510(k) review assesses equivalence to other 510(k) products and generally does not evaluate activities that otherwise require a PMA or BLA. PMA-approved wound care products generally are intended to go beyond a simple wound cover to provide some type of direct treatment effect. The intended uses of these products may include physical scaffold claims or reference to structural matrix attributes that promote endogenous cell binding, migration, differentiation, or proliferation. Currently, wound care products intended to interact with the wound to facilitate, promote, or accelerate wound healing generally require approval of a BLA or a PMA when it meets the statutory definition of a device. As an example, Integra® Wound Matrix is indicated for the treatment of certain wounds. Approval of these products requires demonstration of safety and efficacy for the intended use, which generally requires the performance of clinical studies. A determination of pathway is informed, in part, by a sponsor's desired indications and ability to prove them.
Notably, unless a product has obtained approval through a BLA, non-homologous use marketing claims are not allowed. Such claims would directly contradict the regulatory status of registered 361 HCT/Ps, because of the criteria in 21 CFR 1271.10(a)(2). Similarly, 510(k)-cleared devices relevant to this policy generally are dressings intended only to act as a physical cover to protect a wound, to absorb exudate, and to maintain appropriate moisture balance within the wound. Descriptions of purported biological healing factors in these products have not been evaluated by FDA; and there is no guarantee that these factors are present or active and, if present, their concentration because these products are not requires to have undergone purity or potency assessment performed by FDA. Biological products can differ greatly based on their source material and manufacturing, and it is therefore difficult to generalize any conclusions about their safety and effectiveness beyond those allowed by FDA.
Finally, while we have no obligation to categorize products based on the FDA's current regulatory framework, in this case, we have determined that the FDA regulatory categories provide an appropriate level of distinction for a heterogeneous category of products that exhibit clinical and resource variability for purposes of setting payment rates. This methodology can ultimately improve the accuracy of the relative value units under the PFS while also being predictable and efficient.
Comment: Several commenters emphasized that CMS should acknowledge post-FDA clinical investment if considering FDA pathways as a basis for categorization. One commenter noted investing more than $7.5 million over 6 plus years for two seminal RCTs, stating such investment in post-FDA studies alone ( printed page 53737) exceeds application fees of both PMA and BLA products but is not captured in CMS' FDA pathway analysis. Commenters stated that if an overly generalized distinction is drawn between products approved under different pathways, CMS risks disincentivizing manufacturers from further developing clinical evidence and continuing to innovate improved skin substitute products.
Response: We encourage those entities that have made investments in clinical research to work with the FDA to determine if these studies are sufficient to support approval or clearance through the appropriate FDA regulatory pathway and to ensure that essential manufacturing information and controls are available to support approval of a PMA or BLA.
Comment: Several commenters noted that regulatory pathways for skin substitutes have evolved significantly over time, and since CMS does not oversee FDA regulatory pathways, future changes by FDA could inadvertently affect categorization.
Response: We understand that refinements in categorizations for this policy could be warranted in the future and would, at a minimum, address any potential changes through notice and comment rulemaking.
Comment: Commenters generally supported the use of transitional pass-through (TPT) and other payment pathways, like New Technology Add-on Payments (NTAP), to pay for eligible skin substitute products. One commenter requested that we establish a dedicated pass-through pathway for tribally manufactured skin substitute products.
With regard to the non-facility setting, several commenters urged CMS to leverage existing programs like NTAP and TPT or create a new, parallel program to NTAP/TPT that would provide temporary add-on payments for innovative products used in the physician office setting to ensure site neutrality for new technologies.
Response: We appreciate these comments. We reiterate that drug and device transitional pass-through are available pathways for eligible skin substitute products. With regard to exploring additional payment pathways, we will take commenters' suggestions into consideration as we consider how to incentivize innovation in future rulemaking.
After careful consideration of the comments, we are finalizing our proposal to group skin substitutes (other than those approved via BLA under section 351 of the PHS Act) into three FDA categories, PMA, 510(k), and 361 HCT/Ps, for purposes of developing payment rates in future notice and comment rulemaking, as proposed.
We are also finalizing our proposals regarding drug and device transitional pass-through payments. Specifically, we are finalizing our proposal to pay for skin substitutes approved for device pass-through payment status consistent with other devices approved for device pass-through payment. For the purposes of eligibility of skin substitutes for transitional drug pass-through payment, we are finalizing our proposal to define the term “biological” consistent with our interpretation of the term under section 1847A of the Act.
We are also finalizing our proposals to create three new APCs (APCs 6000-6002) and assign skin substitute products, unless the product is licensed under a BLA pursuant to section 351 of the PHS Act, to one of the three clinical APCs based on their FDA regulatory category. We are finalizing our proposal to create three new unlisted codes (HCPCS codes Q4431-Q4433) to describe skin substitute products that are FDA authorized or cleared but have not yet received a specific individual HCPCS or CPT code.
3. Alternative Payment Categories
As a conceptually possible alternative to our proposal to group skin substitutes based on FDA regulatory categories for purposes of payment, we considered aligning these products based on their composition, for example, whether they are non-synthetic or synthetic. Two examples provided by interested parties include grouping the products as allografts (for example, amniotic products, cellular products), xenografts (for example, collagen products derived from animals), synthetics (for example, artificial products made from various biomaterials) and grouping the products as human living/cryopreserved tissue, dehydrated human/amniotic tissue, animal xenografts, and synthetics/polymers. However, as noted previously, skin substitutes are a heterogenous group with an increasing intersection between tissue, bioengineered, and synthetic components. With many products now including both non-synthetic and synthetic components, clear categorization of skin substitutes by composition is no longer feasible. This alternative categorization would be extremely complex to implement because it would be necessary to determine which category would be most appropriate for each individual product based on the components of its composition and an assessment of the importance of each component. In addition, it is unclear if grouping products based solely on their composition would provide accurate differentiation with respect to resource or clinical similarity for the purposes of setting an appropriate payment rate.
Other alternatives we considered include grouping all products together to set a single payment rate or creating two or more categories reflecting product cost, similar to the groupings used currently to set payment rates for skin substitutes in hospital outpatient departments. While these options may offer certain operational advantages for their simplicity, neither recognizes the clinical differences among skin substitutes as reflected by their different intended uses. Paying for similar items and services at a comparable rate is a foundational aspect of our payment systems, but hospital outpatient departments paid under the OPPS and physicians and other practitioners paid under the PFS could potentially have a financial incentive to use the least expensive skin substitute or the product offering the greatest discount, which could negatively affect patient outcomes and disincentivize innovation in this space if clinical differences are not recognized and differential payments rates are not set. In addition, dividing products by cost relies on pricing set by manufacturers. Especially in light of the dramatic growth of skin substitutes' ASP-based payment limits, this method is unlikely to accurately reflect skin substitute resource costs or clinical similarity.
We sought comment on whether adding certain subcategories to the three proposed FDA categories would improve clinical or resource similarity. One potential example is creating certain subcategories for payment based on one or more FDA device product codes, which is a categorization process that FDA uses to group similar products together. Other examples that have come to our attention include setting unique payment rates for 361 HCT/Ps based on the number of tissue layers (for example, one layer, two layers, and three or more tissue layers) or entirely synthetic products versus non-synthetic products for 510(k)s. If significant clinical or resource differences were identified between products in one or more of these categories, CMS could create a separate payment grouping for these products for payment purposes.
We received public comments on this comment solicitation. The following is a summary of the comments we received and our responses.
Comment: Many commenters advocated establishing a single payment rate for all non-BLA skin substitutes. ( printed page 53738) They stated that a single group creates a level playing field, encouraging product selection based on clinical evidence and patient need rather than on which category receives the highest reimbursement. This approach is seen as simpler and avoids the perverse incentives created by past high/low-cost buckets.
Response: While a single rate would result in an administratively simpler policy and likely would result in the most savings, the differentiation of the products in this space supports subgrouping to better ensure access to products in each group. A flat payment rate also reduces the incentive to innovate, perform relevant studies, and seek an FDA approval requiring proof of wound treatment or healing. However, we can continue to evaluate the appropriateness of maintaining a single rate in future rulemaking.
Comment: Many commenters oppose a single flat rate, characterizing it as a “one-size-fits-all” approach that fails to recognize the clinical complexity and diversity of products. They believe it would force providers to use less effective products, stifle innovation, and lead to worse patient outcomes, such as higher amputation rates. They argue that a single rate below acquisition costs for many products will destabilize office-based care and restrict access.
The commenters suggested various alternative categorization schemes:
- Several commenters suggested a tiered system with 2-3 tiers based on product technology, clinical evidence, or cost thresholds. For example, a basic collagen matrix could be in a lower tier, while a cellular product with strong RCT data could be in a higher tier. Another proposal suggested tiers based on whether a product requires one or multiple applications to achieve wound closure.
- Several commenters suggested grouping products based on their composition (for example, human tissue, animal-derived, synthetic) rather than just their FDA pathway. Specific proposals included creating a distinct category for amniotic/placental tissue products or sub-categorizing 361 HCT/P products based on the number of tissue layers (for example, single-layer vs. multi-layer) to better reflect complexity and resource costs.
- A commenter suggested greater aggregation into broader categories like “synthetic” vs. “non-synthetic” would be sufficient.
- A commenter requested a reimbursement framework that provides higher payment for products supported by product-specific randomized clinical trial (RCT) data.
- A commenter suggested implementing a tiered system based on the strength of clinical evidence (for example, number of RCTs) rather than just regulatory pathway.
- Another commenter suggested using the product's FDA cleared/approved label information (for example, product description, including mechanism of action, and indications for use) as well as supporting level 1 human clinical data.
Response: We agree that long-term use of a single, flat rate has the potential to create access issues for specific types of products and reduces the incentive to innovate. However, the options suggested here are also problematic. As noted previously, skin substitutes are a heterogenous group with an increasing intersection between tissue, bioengineered, and synthetic components. With many products now including both non-synthetic and synthetic components, clear categorization of skin substitutes by composition is no longer feasible. This makes this alternative extremely complex to implement because it would be necessary to determine which category would be most appropriate for each individual product based on the components of its composition and an assessment of the importance of each. In addition, it is unclear if grouping products based solely on their composition would provide accurate differentiation with respect to resource or clinical similarity for the purposes of setting an appropriate payment rate.
In addition, we have concerns about the quality of many of the studies that are being produced as well as whether the results can accurately be extrapolated more broadly. As previously discussed, unless a product has obtained approval through a BLA, non-homologous use marketing claims are not allowed. Such claims would directly contradict the regulatory status of registered 361 HCT/Ps, because of the criteria in 21 CFR 1271.10(a). Similarly, 510(k)-cleared devices relevant to this policy generally are dressings intended only to act as a physical cover to protect a wound, to absorb exudate, and to maintain appropriate moisture balance within the wound. Descriptions of purported biological healing factors in these products have not been evaluated by FDA; and there is no guarantee that these factors are present and active or, if present, their concentrations, because these products have undergone no purity or potency assessment. Biological products can differ greatly based on their source and manufacturing, and it is therefore difficult to generalize any conclusions about their safety and effectiveness beyond those allowed by FDA. While section 1862(a)(1)(A) of the Act directs CMS to make determinations about what is reasonable and necessary for Medicare coverage. FDA's statutorily-defined mandate includes determining the safety, purity, and potency of products such as these. We neither have the resources nor authority to replicate these functions for payment purposes and we believe it would be an inefficient administration of government resources to duplicate them.
Comment: A few commenters suggested ways to further subdivide the three FDA categories such as:
- Creating subcategories for 361 HCT/P products based on tissue composition or number of layers to better reflect resource costs.
- Creating a separate category for amniotic/placental tissue products.
- Creating separate categories for products using one or more of FDA device product codes.
Response: We will take these comments into consideration for future rulemaking as we implement this policy and begin to gather new cost data.
We also sought comments on whether products that are not in sheet form are appropriately considered skin substitutes for the purposes of providing separate payment under this policy. Examples include gel, powder, ointment, foam, liquid, or injected products listed in the nontraditional units of cc, mL, mg, and cm3 . We requested feedback on whether these products could be appropriately used as part of the CPT administration codes in the range 15271 through 15278, despite existing CPT coding guidelines limiting their use, and how these units could be paid using the FDA regulatory category groups. For example, assuming these products were appropriate to administer using the noted CPT administration codes or other administration codes, CMS could include products listed in units of cc, mL, or cm3 in the applicable FDA categories and equate a single cm2 unit to each cc, mL, or cm3 for payment purposes. We sought comments on whether other administration codes could be used to appropriately describe services performed using products with units other than cm2 .
We received public comments on this comment solicitation. The following is a summary of the comments we received and our responses.
Comment: Many commenters recommended CMS include non-sheet product forms (gels, powders, liquids, injectables, 3D-printed constructs etc.) in the definition of skin substitutes eligible for separate payment. They argue these products perform similar ( printed page 53739) functions to sheets, offer additional treatment options for irregularly shaped or tunneling wounds, and excluding them from separate payment would stifle innovation and limit physician choice.
The commenters highlighted the following significant challenges with billing for non-sheet products under the proposed framework.
- The proposed per-cm2 payment does not align with products billed by volume (mL) or weight (mg). Commenters stressed the need for a standardized and fair unit conversion methodology (for example, mL to cm2 of coverage) to ensure equitable payment and prevent reimbursement misalignment. One commenter suggested that 1 mL of a particular flowable product should be paid at the rate equivalent to 10 cm2 of a sheet product.
- It is unclear if current surgical application codes (CPT 15271-15278) can be used for non-sheet products, creating a risk they may not be payable at all. The CPT manual explicitly excludes powders and injectables from these codes. Commenters recommended CMS either confirm their eligibility, create alternative CPT/HCPCS G-codes for their application, or develop a crosswalk framework pairing product form with appropriate procedure codes.
Response: We agree that it is important to maintain access to non-sheet products performing similar functions to sheet skin substitutes, in cases where application of these products is part of reasonable and necessary care. These products have the potential to be payable as skin substitutes; but we agree that units, as expressed in a product's coding, are difficult to standardize for payment purposes. Therefore, we will maintain the current coding mechanism for these products and will direct the MACs to determine appropriate payment in the physician office, which is generally consistent with how these products are currently paid. In the OPPS, these products will continue to be paid through their current packaged payment mechanism. However, we will continue to evaluate payments for these products to determine if an alternative payment methodology might be better suited to non-sheet products. For now, we are also revising HCPCS code A4100 (Non-sheet form skin substitute, FDA cleared as a device, not otherwise specified (list in addition to primary procedure) to allow billing for non-sheet form skin substitute products that do not yet have a more specific code.
Comment: A commenter recommended that CMS not pay separately for non-sheet products as skin substitutes. They stated that procedures for these products are reported with different CPT codes and including them could introduce new opportunities for gaming the system. Another commenter stated that for products cleared via the 510(k) pathway that are classified as gels, liquids, or particulates, reimbursement should follow the existing DME pathway, as these products are not skin substitutes and should not be reimbursed under the skin substitute payment framework.
Response: We disagree that form should be the singular determinant of payment for these products.
Comment: Several commenters stated that CMS has never established a formal definition of “skin substitutes,” leading to inconsistent policy, and recommended the agency to develop a comprehensive, clinically grounded definition. Several commenters advocated for defining products based on their clinical function rather than their initial physical form. The commenters cited the cellular, acellular, and matrix-like products (CAMPs) initiative, which defines products by their ability to support tissue regeneration. They stated that a product that forms a “sheet scaffolding for skin growth” in situ (in the wound bed) should be considered functionally equivalent to a product pre-packaged as a sheet. Several commenters recommended that the CPT definition and CAMPS definition be adopted as standard references for skin substitute classification, eliminating outdated distinctions based on initial product form, aligning with scientific consensus, and supporting value-based care focused on clinical outcomes and regenerative functionality. Many commenters stated that the American Medical Association CPT definition of skin substitute grafts explicitly includes “biological products that form a sheet scaffolding for skin growth,” specifying the operative standard as whether the product forms a sheet scaffolding, not whether it is originally formulated as a sheet.
Response: We recognize that skin substitutes have been described but not defined in previous rulemaking. While a formal definition would provide certain advantages, it might also prematurely and unnecessarily limit an evolving category of products. For example, definitions that require products to obtain claims of treatment or healing of wounds or scaffold claims would exclude large numbers of even sheet-form products currently considered by CMS to be skin substitutes for payment purposes. However, we will continue to consider whether a definition or one or more defining characteristics should be identified in future rulemaking.
After careful consideration of the comments and given the significant challenges with billing for non-sheet products under the proposed framework, we are not finalizing changes to the current payment arrangement under the OPPS for products that are not sheet form at this time. While non-sheet products will continue to be packaged with a separately payable service for CY 2026, we will continue to explore the issue and consider whether to provide separate payment for non-sheet products under the OPPS for future rulemaking.
4. Establishing Initial Payment Rates
We proposed to establish initial payment rates for the three FDA regulatory categories based on the volume-weighted average ASP, with no additional markup, for skin substitute products in each category as submitted by manufacturers, when available. We developed initial payment rates for each group based on the weighted, per-unit average of ASPs for the fourth quarter of calendar year 2024. These initial payment rates were listed in the file titled “CY 2026 PFS Proposed Rule Skin Substitute Products by FDA Regulatory Category—Updated 08/11/2025” on the CMS website under downloads for the CY 2026 PFS proposed rule at https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/PhysicianFeeSched/PFS-Federal-Regulation-Notices.html. When ASP was not available, we used the MUC, which we currently use to determine the high-cost/low-cost status for each skin substitute product in the hospital outpatient setting to calculate the proposed initial rates. We considered using only the MUC data to calculate payment rates for these products. However, when ASP is reported, it may serve as a better estimate of cost across both settings as the ASP reflects sales to physicians as well as hospitals. We sought comment on our proposal to calculate payment rates for skin substitute products in each of the three FDA regulatory categories using ASP, or MUC when ASP is not available, using per-unit averaged pricing data from the fourth quarter of 2024. We also sought comments on whether these calculations, if finalized, should be updated with the most recently available data at the time the final rule is drafted.
We received public comments on these proposals. The following is a ( printed page 53740) summary of the comments we received and our responses.
Comment: A few commenters supported prioritizing ASP data but raised concerns about the reliability of MUC data as a fallback option. These commenters stated that hospital outpatient MUC is less accurate than ASP data submitted in compliance with statute and regulation, citing longstanding challenges with hospital charge compression where reported charges often do not reflect actual acquisition costs. A commenter noted that in some cases CMS did not use ASP data reported to CMS to set Medicare payment limits and recommended the agency to rely on reported ASP data whenever available, given that manufacturers submit this information quarterly in accordance with statutory requirements established by the Consolidated Appropriations Act, 2021.
Response: When ASP is available and accurately reported, we generally agree that it may serve as a better estimate of acquisition cost across both settings as the ASP reflects sales to physicians as well as hospitals and is net of certain discounts. However, as discussed in the CY 2026 PFS final rule, we have had concerns with the accuracy of some reported ASP data; specifically, that manufacturers could be classifying certain costs as bona fide service fees (BFSFs) when they should instead be classified as price concessions, which would artificially inflate ASP. Additionally, we cannot expect that all manufacturers of skin substitute products will continue to report ASP data to CMS each quarter. We disagree with the statement that the outpatient hospital MUC is an inaccurate measure of actual acquisition costs. We use cost-to-charge ratios specifically to account for variations in hospital markup, and our use of MUC in this case is consistent with how we price other products in hospital outpatient departments. MUC is a useful alternative based on actual claims data when the ASP is not available. Generally, with limited exceptions, when ASP data was available for a product, it was used to calculate a Medicare Part B payment limit and published on the ASP drug pricing files. An exception is for diagnostic radiopharmaceuticals that are above the packaging threshold, which use MUC. However, for the purposes of this policy, payment rates were determined using pricing for the 361 HCT/P products only.
Comment: A few commenters expressed concern that using ASP without markup removes appropriate overhead and handling costs. They noted that drugs and biologicals payable under Medicare Part B are statutorily paid at ASP+6 percent, and using ASP alone eliminates legitimate indirect costs from both OPPS and MPFS systems.
Response: We appreciate these comments and note that overhead costs related to application of these products are included in the facility fees or PE RVUs for the application procedures. A recent Office of the Inspector General report [107] found that, in the third quarter of 2024, a typical beneficiary received 82 units of skin substitutes, meaning that the typical $74 add-on amount per unit alone was worth over $6,000 per patient. Notably, hospitals have been managing these products without separate markup for years through bundled payments.
Comment: A commenter recommended using the arithmetic mean unit cost (AMUC) for products without an ASP, rather than the geometric mean (MUC), to better align with the ASP calculation methodology. Conversely, another recommended using a volume-weighted geometric mean for the overall calculation because it is less influenced by extreme outliers.
Response: We appreciate the commenters for the additional information and may consider these alternatives in future rulemaking.
Comment: A few commenters supported using Q4 2024 ASP data as the foundation for CY 2026 rate calculations.
Response: CMS thanks commenters for their support.
Comment: Several commenters criticized the use of Q4 2024 ASP data. Some suggested using data from before the recent dramatic increases in price (for example, CY 2019, 2022, 2023, or Q4 2023) to establish a more reasonable baseline free from market distortions. Other commenters stated that more current data (for example, Q3 2025) should be used to reflect real-time market conditions, reasoning that using older data could introduce a systemic underpayment.
Response: We agree that much earlier datasets (for example, CY 2019) do not reflect a significant portion of the recent growth in products and payments for this class of products. However, these datasets also do not reflect many new products that might represent quality additions to the market. To avoid this issue, we instead calculated initial rates using hospital outpatient utilization to weight how much each product's price contributes to the final payment rates for skin substitutes. In this setting, skin substitutes are currently paid in two groups (high- and low-cost) to incentivize cost-effective product selection. No similar incentive currently exists in the non-facility setting for physicians and other suppliers billing under the PFS. That is why we consider the hospital outpatient utilization a better source to weight the average among the products. We used the fourth quarter of 2024 because it was the most recent, substantially complete quarter of data and the most complete ASP reporting is typically in the fourth quarter of each year. Finally, use of a later quarter's file would not have allowed us to match up time periods for utilization patterns, and interested parties were given an opportunity to review and comment on that proposed rate.
After careful consideration of the comments, we are finalizing our proposal to establish PE RVUs and initial payment rates for skin substitute products in each of the three FDA regulatory categories using ASP, or MUC when ASP is not available, using per-unit averaged pricing data from the fourth quarter of 2024 as proposed.
As we proposed to implement this policy for CY 2026 in a site-neutral manner across both the non-facility setting under the PFS and hospital outpatient setting under the OPPS, we included all products used in either setting to calculate the rates. However, when product-specific utilization across both settings is used to calculate volume-weighted average payments, the result is an apparent rank order anomaly; despite having a more rigorous regulatory review process and receiving indications to treat and heal wounds, the PMA category has the lowest average payment. We are concerned that the use of the novel pricing practices noted above has resulted in a decoupling of actual resource costs from the ASP. To address this, as a short-term measure, we proposed to weight the product-specific utilization in calculating the proposed rates using the proportions from only the hospital OPPS data and establish, for CY 2026, a single payment rate that would apply to all skin substitute products in the three FDA regulatory categories, or APCs. We believe the OPPS utilization data may better predict utilization patterns under our proposed policies for non-facility settings because, similar to our proposals, these products are already grouped together for payment purposes under the OPPS. By grouping skin substitutes into high- and low-cost groups in the OPPS, hospitals are ( printed page 53741) incentivized to choose either the lowest-cost, clinically appropriate product in the low-cost group or the lowest-cost, clinically appropriate product in the high-cost group. No similar incentive currently exists in the non-facility setting for physicians and other suppliers billing under the PFS. As the proposed policies are intended to mitigate the current patterns of use in the non-facility setting by establishing payment rates for the products in groups instead of individually, we do not believe it would reflect the expected resource costs involved in providing care if we were to base the initial rates on utilization data from the non-facility setting that may have been skewed by aggressive and/or improper billing practices that would be less likely to exist under our policies. For these reasons, we proposed to initially use hospital outpatient utilization to weigh how much each product's price contributes to the proposed payment rates for skin substitutes cleared through the 510(k) pathway, registered 361 HCT/Ps, or approved under a PMA. We sought comments on the use of these product utilization patterns to set payment rates.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: Several commenters, including MedPAC, strongly support using only OPPS utilization data to set the initial rate. They agree with CMS that utilization data from the physician office setting (PFS) has been distorted by profiteering practices of certain skin substitute product manufacturers. They believe OPPS data is a better predictor of use patterns under a grouped payment approach because hospitals are already incentivized to choose lower-cost products within the existing high/low-cost categories.
Response: We thank commenters for their support.
Comment: A majority of commenters strongly opposed the exclusion of physician office data, arguing that this methodology is flawed, biased, and not representative of real-world practice. They make several key points:
- The vast majority of skin substitute utilization (nearly 90% in Q4 2024) occurs in the physician office setting, so excluding data from the predominant site of service is unreasonable.
- OPPS data is itself skewed. The current bundled payment system in hospitals disincentivizes the treatment of larger wounds in the hospital setting and the use of more advanced, higher-cost products, artificially driving down the average cost in that setting.
- Hospitals can negotiate lower prices through GPOs, which are unavailable to smaller physician offices. Basing a national rate on hospital acquisition costs will create a payment rate that is unsustainable for non-facility providers.
- Using data from only one setting to create a site-neutral rate for all settings is methodologically unsound and works against the stated goal of a consistent payment approach.
The prevailing recommendation from those opposed to the OPPS-only method is to use a blended approach that incorporates utilization data from both the physician office and hospital outpatient settings. To address CMS' concerns about distorted office data, they suggest using safeguards like trimming outliers or using data from a time period before the recent dramatic spending increases (for example, 2019 or 2022).
Response: Although skin substitute products are more commonly used in the physician office setting, as we stated in the proposed rule, we believe that separate payment for these products in the non-facility setting has led to problematic practices that are mitigated by the current OPPS policy of paying for skin substitute products in either high-cost or low-cost groupings. We believe OPPS utilization data better reflects the utilization associated with grouping these products into categories for purposes of payment. As we stated in the CY 2026 OPPS/ASC proposed rule, we do not believe it would reflect the expected resource costs involved in providing care if we were to base the initial rates on utilization data from the non-facility setting. Once updated use patterns reflecting this policy are available to calculate rates, we proposed to use all relevant products and the combined product utilization patterns (OPPS and non-facility) to determine a weighted average per-unit cost by category to set separate payment rates for each of the three categories.
Comment: A few commenters recommended using guardrails to ensure the methodology does not inadvertently embed misaligned historical incentives. These commenters believed CMS has several options to mitigate fraudulent and abusive practices, including trimming for outliers that may signal falsely high utilization, using data only from claims that meet established criteria for completeness and accurate coding, and capping counted square centimeters at 120-150 percent of the CPT-implied wound size.
Response: Because we are using OPPS utilization patterns and the OPPS does not currently stratify payment for each individual product, and, by doing so, promote more efficient care, we believe many of these problematic claims will be mitigated. However, as this policy unbundles skin substitutes from their application codes and pays for them separately, we are concerned about the potential for overuse and waste. We invite additional thoughts about how best to mitigate these issues while products are separately paid and note that we will be monitoring usage as compared to CPT coding moving forward.
Comment: A commenter questioned why all outpatient facility volumes were not used for this policy, including critical access hospitals and other providers, noting that coding for products would be on those claims as well.
Response: We appreciate this comment. We developed the proposed rate using hospital OPPS data since, as we stated above, we believe the structure of payment under the OPPS with payment for these products grouped into two categories best reflects use not potentially influenced by the previous system. Using OPPS data only allows for a consistent data set that reflects the majority of Medicare hospital outpatient services, across a wide variety of geographies and areas of the country.
After careful consideration of the comments, we are finalizing our proposal on the use of the hospital outpatient product utilization patterns to set payment rates for these products under the PFS as proposed.
We also proposed for CY 2026 to establish the same initial APC payment rate for each group of skin substitutes, including 510(k)-cleared devices, registered 361 HCT/Ps, and PMA-approved devices. To ensure we are not underestimating the resources involved in using these products in furnishing care, we proposed to use the highest of the calculated volume-weighted average payment amounts for 510(k) cleared devices, 361 HCT/Ps, and PMA-approved devices to set initial payment valuations. As the 361 HCT/Ps have the highest volume-weighted average payment amount, this average payment rate is reflected in the proposed initial payment rate below. However, we note that, in future notice and comment rulemaking, we intend to propose using claims data to set payment rates for products in these three categories, which would likely result in payment valuations that diverge based on the updated data. Another alternative is to set the payment rate for products in these categories at the volume-weighted average for all three categories, resulting ( printed page 53742) in a lower initial payment rate for all three groups of products. We sought comment on our proposal to use the 361 HCT/P volume-weighted average payment amount to set the initial payment rates for products in all three categories as well as the alternative of using a pooled average of the three categories to set the initial payment rates.
Alternatively, while the ASP Pricing Files show that skin substitutes across all three of the FDA regulatory categories have increased in cost substantially since 2019, unlike the self-determined 361 HCT/Ps and 510(k)-cleared devices, there has not been a substantial increase in the number of skin substitutes with approved PMAs. Consequently, it is possible that the non-facility utilization of the skin substitutes with approved PMAs is not as distorted as the utilization of the other kinds of skin substitutes. Setting a separate payment rate for this category using combined product utilization patterns (from both OPPS and non-facility settings), would result in a higher initial payment rate for the PMA category. This would rationally order the FDA regulatory categories, based on clinical considerations and some indicators of resource cost, until pricing data removed from these aberrant financial incentives can be incorporated. We seek comment on this alternative policy option.
The proposed calculation methodology would result in an initial payment rate of $125.38/cm2 for all three proposed new APCs based on the FDA categories including PMA-approved devices, 361 HCT/Ps, and 510(k) devices. Specifically, this proposal would result in an initial payment rate of $125.38/cm2 for each HCPCS code assigned to APC 6000 “PMA Skin Substitute Products,” APC 6001 “510(k) Skin Substitute Products,” and APC 6002 “361 HCT/P Skin Substitute Products.” We sought comments on these proposed initial payment rates.
We determined these proposed rates using product pricing and volume for skin substitutes from paid claims with dates of service in the fourth quarter of 2024 because it was the most recent, substantially complete quarter of data available at the time of the CY 2026 OPPS/ASC proposed rule. For professional claims, we excluded claims without a positive line-level allowed amount, so that we did not inadvertently include volume without presumed costs in the calculation. In addition, in reviewing the ASP pricing files from the first quarter of 2017 through the first quarter of 2025, the most complete ASP reporting is in the fourth quarter of each year. To determine the payment rates, we first used a product's ASP if it was available. If the ASP rate was missing, we used the 2024 MUC for the HCPCS code. We then calculated a single rate for each FDA category by taking the volume-weighted average of the rates for the applicable codes using the hospital outpatient utilization to weight each category. We note that if rather than using the final quarter of CY 2024, we alternatively, were to use pricing and volume from all four quarters of 2024 to determine proposed rates, the rate for all categories would be approximately $114.87/cm2 . Using a pooled payment rate across all three categories would result in a rate of approximately $65.85/cm2 , while splitting the categories to pay the PMA category using the combined product utilization patterns and the 510(k) and 361 HCT/P categories using the OPPS utilization patterns would result in rates of approximately $259.47/cm2 and $125.38/cm2 respectively. We sought comment on our proposed process to calculate initial payment rates as well as these alternatives.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: Many commenters stated that the proposed payment rate of approximately $125/cm2 is far too low and unsustainable. The commenters stated this rate is well below the actual acquisition and operational costs for most providers, especially smaller practices and mobile units without hospital purchasing power. If implemented, they warn it will make offering these therapies financially impossible, forcing practices to close and severely restrict patient access to care, particularly for homebound, rural, and underserved populations. This could lead to worse outcomes, including more amputations, infections, and hospitalizations, ultimately increasing overall Medicare costs.
Instead, many commenters proposed higher rates. A frequently cited alternative, based on independent analyses, was a payment range of approximately $478 to $704/cm2 . Commenters stated that this range would still generate over $100 billion in Medicare savings over a decade while preserving patient access and innovation. Other suggestions fell within a similar range, such as $500/cm2 , $500-$640/cm2 , or up to $973/cm2 . Another commenter suggested that CMS calculate the payment rate for skin substitute products using a trimmed mean approach—excluding the top and bottom 10% of reported prices of 130 skin substitute products—to eliminate outliers and better reflect fair value. They suggested that the resulting average price of $865/cm2 would reflect a fair market average and urged CMS to set a higher allowed amount of approximately $1,080/cm2 to ensure providers can sustain services under Medicare's 80 percent payment structure. A few commenters also suggested that the 20 percent copayments should be waived for treatment with skin substitute products. Several commenters supported the reimbursement methodology outlined in Senate Bill 2561 (Skin Substitute Access and Payment Reform Act of 2025), which would establish rates based on Q4 2023 ASP data volume-weighted according to actual utilization in both the professional and hospital outpatient settings. Commenters stated that the methodology from this legislation would result in a payment range of approximately $500-700/cm2 . Some commenters proposed tiered pricing structures for HCT/P 361 products based on configuration, with recommendations ranging from $400-$500/cm2 for single-layer amniotic products to $800-$900/cm2 for full-thickness grafts. Other commenters recommended tiered frameworks incentivizing manufacturers to pursue rigorous FDA pathways with reimbursement increases of 25-100 percent over base pricing of $400-$900/cm2 . Several commenters cited crosswalks to similar products as valid data points. Commenters noted that CPT codes for placing amniotic membrane on ocular surfaces (65778 and 65779) use amniotic membrane allograft supplies reimbursed at $835 and $1,149 in CY 2025. Since typical amniotic tissue grafts placed in eyes are 14mm diameter discs with surface area of approximately 1.5 cm2 , commenters calculated these rates equal $557-$776 per square centimeter, stating it would be arbitrary and capricious for CMS to price amniotic tissue grafts for eyes at significantly higher rates than same tissues used for chronic wounds. Some suggest using the higher rate CMS calculated for one of the other FDA categories ($259.47/cm2 ) as a more reasonable starting point.
Response: We appreciate the many detailed comments we received on this issue, but we do not agree that higher payment rates are warranted at this time for several reasons. First, most of these payment rates use non-facility utilization patterns that have been significantly distorted in recent years by problematic practices. Incorporating this ( printed page 53743) data into the payment rate would embed these practices into the policy. These practices have largely been mitigated in the OPPS utilization patterns, which is why they were used to develop an initial rate for this policy. Second, manufacturers have demonstrated the ability to offer products well below current ASP levels, which indicates that current prices have considerable room for compression without risking product availability or access. Third, there are a significant number of products with current ASPs below this policy's payment rate already. Finally, many of the more expensive products on the market fall into our 361 HCT/P and 510(k) product categories, but their activity is typically limited to that of a physical covering or wrap. These products are not intended to act on the wound to mediate, facilitate, or accelerate wound healing and the justification for these rates is unclear. However, we do not agree that a product used to cover the cornea and prevent scarring and others used to prevent infection should necessarily be priced at the same rate. Once updated use patterns reflecting this policy are available to calculate rates, we will use all relevant products and the combined product utilization patterns (OPPS and non-facility) to determine a weighted average per-unit cost by category to set separate payment rates for each of the three categories, and we will continue to monitor this product class and propose additional adjustments to the policy as necessary in future rulemaking.
Comment: Many commenters expressed concern that a low, uniform payment rate would stifle innovation. They argued that it would discourage investment in research and development for next-generation products, as manufacturers would be unable to recoup the significant costs associated with bringing novel therapies to market. They stated that this could penalize innovative therapies, favor older and lower-cost alternatives, and ultimately limit patient access to more effective technologies. In contrast, some commenters believe the proposed changes will rightly shift the focus to value-based innovation. They stated that true innovation lies in making highly efficacious products at a reasonable price, not just launching increasingly expensive ones. They contend that manufacturers should be required to show robust clinical evidence to justify payment, and the current proposal encourages this shift.
Response: We agree that our policy should incentivize innovation in this class of products. As noted above, in future notice and comment rulemaking, we intend to use claims data to set separate payment rates for products in the three categories, which would likely result in payment valuations that diverge based on the updated data. As the PMA category is by far the smallest, we would anticipate less competition and potentially higher payment rates for those products. In addition, products marketed under the BLA pathway would continue to receive separate payment under the ASP methodology described in section 1847A of the Act. We also welcome continued dialogue on ways to differentiate a truly innovative product from another that offers no true clinical advance as well as on how to properly recognize innovative products through payment policy under the PFS as we continue to assess how best to identify and value innovative products under the PFS.
Comment: A smaller group of commenters supported the proposed rate, and some even advocated for a lower one. Some commenters cited published cost-effectiveness analyses. A commenter noted a study concluding that interventions using skin substitutes with payment limits below $140/cm2 were dominant (less costly, better outcomes) compared to typical care; and, based on cost-effectiveness threshold of $100,000 per quality-adjusted life year, interventions were cost-effective with payment limits up to $430/cm2 . A commenter stated that a rate of $125/cm2 seems “more than reasonable” given that manufacturing costs for some products are much lower. Several ACOs and provider groups recommended that CMS use a “pooled” average across all product categories, which would result in an even lower rate of ~$65/cm2 , to more accurately reflect resource costs and further curb overspending. Another commenter recommended a rate of $75/cm2 .
Response: We appreciate the commenters for their feedback. We disagree that further reduction of the initial payment rate is prudent before updated claims data reflecting the results of this policy can be gathered and evaluated.
Comment: There was a split on whether to use a single rate for all products. Many commenters supported finalizing a single, standardized payment rate for all non-BLA products. They argue this creates a level playing field, where clinical choice is driven by evidence and patient need, not by which category has the highest reimbursement. Commenters believe this approach is simpler and less likely to repeat the high-cost/low-cost bucket system of the past. Some commenters requested that after setting an initial rate for 2026, CMS should maintain that single rate for two additional rulemaking cycles before recommending any changes, allowing time to carefully evaluate claims data. Some comments requested moving to tiered payment rates in CY 2027 after setting an initial flat payment rate for CY 2026. Other commenters argued for immediate implementation of a tiered or differentiated payment system in 2026, rather than waiting until 2027. They believe this better reflects product complexity and clinical evidence and avoids treating all products as if they are the same.
Response: While a single rate would result in an administratively simpler policy and likely would result in the most savings, the differentiation of the products in this space argues for subgrouping to better ensure access to products in each group. A flat payment rate also reduces the incentive to innovate, perform relevant studies, and seek an FDA approval requiring proof of wound treatment or healing. While we do intend to use claims data to set separate payment rates for products in the three categories in future notice and comment rulemaking, we are concerned that use of the novel pricing practices noted above has resulted in a decoupling of actual resource costs from the ASP. To address this, as a short-term measure, we proposed to weight the product-specific utilization in calculating the rates using the proportions from only the hospital OPPS data and establish for CY 2026 a single payment rate that would apply to all skin substitute products in the three FDA regulatory categories. We will propose rates for these three categories through notice and comment rulemaking once updated use patterns reflect this policy.
Comment: To avoid sudden market disruption and access issues, some commenters recommended phasing in the payment reduction over several years (for example, 3 years). This would blend the old ASP plus 6 percent payment with the new flat rate over time, giving providers and manufacturers a chance to adapt.
Response: We do not agree that a phased-in approach is necessary. The nearly 40-fold increase in spending on these products has been concentrated in just the past several years. To allow these damaging practices to continue, even in part, for years longer may only serve to unnecessarily delay efficient purchasing, appropriate utilization, and product selection based on clinical need.
Comment: Several commenters opposed applying geographic ( printed page 53744) adjustments to skin substitute product payments in any setting, arguing that product costs do not vary by location as manufacturer pricing remains consistent regardless of clinical site or geography. Several commenters stated that geographic adjustments would create access disparities, particularly harming rural and underserved communities where patients may have greater need but providers face lower reimbursement rates. A commenter explained that, given the standard OPPS payment methodology, 60 percent of the payment rate for these products would be geographically adjusted, based on the application of the hospital wage index to the labor-related portion of the OPPS conversion factor prior to the application of the APC relative weight. Per the commenter, this would be a significant departure from current policy, where the supply portion of the APC is not geographically adjusted, and would similarly have negative impacts on patients' ability to access products in areas with lower wage index values. Commenters also recommended that CMS exclude skin substitutes from geographic RVU adjustments, noting that while professional service costs may vary by location, product costs do not.
Response: We understand commenters' concerns and are not applying a geographic adjustment to skin substitute products under the OPPS and ASC, where we are not required by statute to do so. With regard to the non-facility setting, section 1848(e) of the Act requires that the PFS include geographic adjustment factors and account for geographic variations in the costs of furnishing services. We refer commenters to the CY 2026 PFS final rule for more information regarding geographic adjustment factors under the PFS.
Comment: Several commenters raised concerns about maintaining payment consistency between PFS and OPPS. Commenters noted that OPPS receives annual market basket adjustments while PFS does not, questioning how CMS would prevent the rates from diverging over time and creating site-of-service incentives.
Response: We appreciate commenters bringing these concerns to our attention, and while we remind readers that variations in the annual update mechanisms for Medicare payment systems are generally determined by statute, we may consider the interaction between the update factors and the development of future proposed rates for these products for future rulemaking.
Comment: Several commenters raised concerns about transparency in CMS's rate calculation. The commenters stated that the information provided was insufficient to understand or recreate CMS' calculations. Some commenters were unable to replicate the proposed $125.38/cm2 rate, while others replicated it within 1 percent. A commenter noted that a supplemental document posted August 11, 2025, created additional confusion by describing steps the proposed rule indicated were not performed and contradicting information in the rule itself. Commenters stated CMS did not publish comparison files showing ASP versus MUC values for each product, which are important for evaluating the proposal. Many commenters recommended CMS provide complete methodology details to enable meaningful comment, with some stating that the lack of transparency violates APA requirements for well-reasoned analysis.
Response: We disagree with the commenters' statement that CMS was unclear in the description of the methodology used to calculate the proposed initial payment rate of $125.38/cm2 . We provided a substantial amount of detail on our calculations in the proposed rule, including the HCPCS codes that were pulled and the data sources to provide even more detail. We released a supplemental document, “Additional Description of Calculation of Proposed Payment Rates for Skin Substitutes” on the CMS website at https://www.cms.gov/medicare/payment/fee-schedules/physician/federal-regulation-notices/cms-1832-p, which further explains the methodology used to calculate the payment rates for skin substitute products Several commenters were able to replicate our methodology and stated that they were able to approximate the proposed payment rate based on the information provided.
Comment: Commenters identified several products that CMS had incorrectly categorized in its published lists, suggesting the agency correct these errors before finalizing any rate-setting based on the categories. Commenters noted that HCPCS codes Q4107, Q4108, Q4116, Q4122, Q4142, Q4146, Q4147, Q4159, Q4198, Q4201, Q4225, Q4226, Q4232, Q4238, Q4276, Q4282, and Q4297 were misclassified into the wrong FDA categories. The commenters stated these errors lead to inaccurate payment rates for all products in affected categories and suggested CMS correct all assignments before finalizing the rule.
Response: We appreciate the comments. We reviewed the information provided and acknowledge there were some discrepancies in the grouping of some HCPCS codes for particular products in the CY 2026 OPPS/ASC proposed rule. We conducted additional analyses shifting those FDA categorizations, including those suggested by commenters, some of which caused volatility resulting in significant reductions compared to the proposed rule rates.
After careful consideration of the comments, for CY 2026 we are finalizing rates consistent with the groupings illustrated in the CY 2026 OPPS/ASC proposed rule in the interest of transparency and consistency. Prospectively, however, we will designate these HCPCS to the groupings suggested by commenters. We are finalizing our proposal to use the 361 HCT/P volume-weighted average payment amount, based on the grouping of HCPCS codes in the CY 2026 OPPS/ASC proposed rule, to set the initial payment rates for products in all three categories and calculate the initial payment rates as proposed. We also note, that consistent with the proposed rule, we maintained use of hospital claims volume from services incurred during the fourth quarter 2024. We used an updated version of the hospital claims volume as this is the most recent data available as of the time of drafting this final rule with comment period. We also used fourth quarter 2024 ASP data and 2026 MUC data (based on 2024 claims data), when ASP data was not available, as proposed. Given the volatility around skin substitute products due in part to gaming and the proliferation of coding already discussed in this final rule with comment period, even small changes to the methodologies, such as using a different quarter of ASP data or updating the regulatory categories of only 17 of over 200 skin substitute products, can result in significant changes to the payment rate. Accordingly, we believe maintaining the same framework for setting the payment rate that was used for the CY 2026 OPPS/ASC proposed rule, including the FDA regulatory categorizations and the same time periods for pricing, is consistent with the proposed rate on which the public had an opportunity to comment on compared to the volatility that could occur using different definitions. Prospectively, one of our policy goals is to promote greater stability in the payment for skin substitute products. Therefore, consistent with the framework for the methodology proposed in the CY 2026 OPPS/ASC proposed rule, we are ( printed page 53745) finalizing a final payment rate for CY 2026 of $127.14/cm2 .
The full list of codes with the payment groupings used in developing the proposed and final rate calculations are available on the CMS website. Likewise, the full list of codes and their payment groupings prospectively are available on the CMS website and in Addendum B to this final rule with comment period.
After careful consideration of the comments, we are finalizing our proposal to use the 361 HCT/P volume-weighted average payment amount to set the initial payment rates for products in all three categories and calculate the initial payment rates as proposed with the exception of updating the calculation with more recent utilization data available as of the time of drafting this final rule with comment period, which resulted in a final payment rate for CY 2026 of $127.14/cm2 . Specifically, we also used fourth quarter 2024 ASP data and 2026 MUC data (based on 2024 claims data), when ASP data was not available, weighted by more up to date hospital outpatient claims volume.
We proposed to maintain the current structure of HCPCS codes for skin substitutes, including a process to introduce new product-specific codes and proposed initial valuation based on the typical resource costs (that is, those reflected in ASP and MUC data) of the groups associated with each skin substitute's HCPCS code. We proposed to assign each current HCPCS code that describes an individual skin substitute product to one of the three new APCs based on the product's appropriate FDA regulatory category. For a complete list of codes and FDA categories, please see file entitled “Skin Substitute Products by FDA Regulatory Category” available on the CMS website under downloads for the CY 2026 PFS final rule at https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/PhysicianFeeSched/PFS-Federal-Regulation-Notices.html. Individual HCPCS coding remains necessary to provide identification on claims and track each product's cost. This will also allow effectuation of any applicable coverage policies and improve our ability to determine if any refinements in payment categories would be appropriate in future rulemaking.
For the most part, materials or supplies furnished incident to a service (for example, a suture, customized surgical kit, scalpel, or clip, other than a radiological site marker) are not paid separately under the OPPS. However, separate payment for products is not novel, since Medicare pays for various components of services through the use of separate HCPCS codes and/or payment modifiers. The most obvious examples of these kinds of payment and coding splits occur in diagnostic tests, radiation treatment services, and blood product services. For example, under the OPPS, blood products receive specific HCPCS codes and are paid separately from other services. In this case, the procedure of applying or administering a skin substitute product would not be described or paid for by a single code. Rather, when a skin substitute product is applied, both the application code as well as the HCPCS code of the skin substitute that is being applied would be billed under the OPPS. For example, when CPT Code 15271 (application of skin substitute graft, leg or ankle) is billed, we would expect for the hospital outpatient department to also report a skin substitute HCPCS code, which would be paid at a payment rate that includes the resources involved in using the skin substitute product. We proposed for skin substitute products to receive a separate payment independent from the payment for the application procedure. To effectuate this proposed payment policy under the OPPS, we proposed to create a new status indicator for HCPCS codes describing skin substitutes that are assigned to one of the three new APCs for skin substitutes based on FDA regulatory pathway. Specifically, we proposed to create status indicator “S1” to indicate that the skin substitute product is paid separately from other procedure codes under the OPPS. We proposed to assign all existing HCPCS codes describing skin substitute products to status indicator “S1” for CY 2026. The proposed status indicator “S1,” along with its descriptor and payment status, is listed in Table 114.
We also sought comments on whether we should consider treating the codes describing skin substitute products as add-on codes to the current CPT administration codes (CPT codes 15271-15278). This would more clearly indicate that the only skin substitute products to be paid for and treated as supplies by Medicare are those used in conjunction with the already existing CPT administration codes. If we were to treat these codes as add-on codes to the administration codes, we would effectuate this by revising the code descriptors of the skin substitute products to state “list separately in addition to the primary procedure.” While we would normally assign the add-on codes to a status indicator that indicates that payment is packaged (that is, status indicator “N” (Items and Services Packaged into APC Rates)), given our proposal to pay separately for skin substitute products as incident-to supplies, we would still propose to assign status indicator “S1,” to the skin substitute codes.
We received comments on whether we should treat the skin substitute product codes as add-on codes to the current CPT administration codes. The following is a summary of the comments we received and our responses.
Comment: Some commenters suggested that CMS' alternative approach of establishing payment for skin substitute products as an add-on code to current CPT application codes could have the added benefit of eliminating the bifurcated HCPCS coding system for skin substitute products (that is, Q-codes vs. A-codes), which has created significant administrative burdens and confusion for physicians and MACs, and streamline administrative requirements when physicians utilize and submit claims for skin substitute products. One commenter supported paying for skin substitute products as an add-on service ( printed page 53746) rather than as a standalone service to allow for packaged payments.
Response: We appreciate the information provided by commenters as to whether skin substitute codes should be considered add-on codes. We note that, for the non-facility setting, we are finalizing conversion of all skin substitute products to add-on codes. To ensure alignment across payment systems, we are also finalizing this change to classify skin substitute products as add-on codes in the hospital outpatient setting. Operationally, this would have the effect of requiring providers to bill an administration code when a skin substitute product is furnished. While we are converting skin substitute product codes to add-on codes, given the proposal to pay separately for skin substitute products as incident-to supplies that we are finalizing, we are assigning skin substitute products to status indicator “S1” to indicate separate payment.
Based on the input we received from commenters, we are finalizing classifying skin substitute products as add-on codes in the hospital outpatient setting. Since we are making this change to align with the corresponding change in the non-facility setting but we are still finalizing separate payment for skin substitute products, we are revising 42 CFR 419.2(b)(18) to clarify that skin substitute product add-on codes are not among the “certain services” described by add-on codes for which packaged payment is made.
We proposed that new HCPCS codes describing skin substitutes would be categorized based on whether they are PMA-approved, 510(k)-cleared, or registered 361 HCT/Ps and the payment rates that apply to that category would be applied to the new code at the next quarterly update. Currently, HCPCS Level II coding applications are submitted and reviewed during our quarterly and biannual coding cycles. We post our coding determinations for drugs and biologicals on a quarterly basis, and do not routinely review those applications at a HCPCS public meeting. For non-drugs and non-biologicals, we post our coding decisions on a biannual basis. For our biannual cycles for non-drugs and non-biologicals, we post preliminary coding determinations then invite feedback on those preliminary coding determinations at a biannual HCPCS public meeting; final coding determinations are posted following the HCPCS public meeting. CMS has been reviewing skin substitutes marketed as 361 HCT/Ps in the quarterly drugs and biologicals coding cycle and 510(k)-cleared skin substitutes in the biannual, non-drugs and non-biologicals coding cycle. Beginning January 1, 2026, we proposed to review HCPCS Level II coding applications for all skin substitutes marketed as 361 HCT/Ps through our biannual coding cycle for non-drugs and non-biological products, rather than on a quarterly basis. Skin substitutes that received a 510(k) clearance, PMA approval, or a granted De Novo request would continue to be evaluated in the biannual HCPCS Level II coding cycles. Therefore, under this proposal, CMS would evaluate all complete HCPCS Level II applications for skin substitutes in our biannual cycles. Should any products come to market under the BLA, NDA, or ANDA pathways that could potentially be considered skin substitutes, CMS would instead review them in a quarterly HCPCS Level II drugs and biologicals coding cycle.
Before a code is assigned to describe the skin substitute product, not otherwise classified (NOC) codes would be used and the CMS MACs would assign the appropriate payment based on the product's FDA category. We proposed to create three new unlisted codes to describe skin substitute products that are FDA authorized or cleared but have not yet received a specific individual HCPCS or CPT code: HCPCS codes Q4431 (Unlisted PMA skin substitute product), Q4432 (Unlisted 510(k) skin substitute product), and Q4433 (Unlisted 361 HCT/P skin substitute product). We proposed to assign the unlisted HCPCS codes to the appropriate APCs based on the product's FDA approval or clearance. Specifically, we proposed to assign HCPCS code Q4431 to APC 6000 (PMA Skin Substitute Products); Q4432 to APC 6001 (Unlisted 510(k) Skin Substitute Products); and HCPCS Code Q4433 to APC 6002 (Unlisted 361 HCT/P Skin Substitute Products). We proposed to create these unlisted codes to prevent delays in Medicare payments for new FDA-approved or cleared skin substitute products that do not yet have a specific HCPCS or CPT code.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: Multiple commenters expressed support for CMS' proposal to maintain the current structure of HCPCS codes for skin substitutes. Several commenters recommended that CMS eliminate the bifurcated coding system where some products have “Q” codes and others have “A” codes. They urged CMS to reassign all skin substitute products to “Q” codes to reduce administrative burden and confusion for providers and MACs.
Response: We appreciate the feedback. We will take comments about the coding differences between the products into consideration for future rulemaking.
Comment: Several commenters opposed CMS's proposal to shift the HCPCS Level II coding application review for 361 HCT/P products from a quarterly to a biannual cycle. Commenters argued that maintaining the quarterly cycle is essential for timely patient access to new therapies, supporting innovation, and aligning with the faster review cadence used for drugs and biologics. Commenters stated a biannual cycle would create lengthy delays (18 months or more) for new products to get a code and be reimbursed, creating a barrier to market entry. Commenters stated that all skin substitutes, regardless of regulatory status, should have the same quarterly review process to maintain a level playing field.
Response: We disagree. We post our coding decisions for all non-drugs and non-biologicals on a biannual basis. While CMS has been reviewing skin substitutes marketed as 361 HCT/Ps in the quarterly drugs and biologicals coding cycle, under this policy, unless a skin substitute is approved as a biological product under section 351 of the PHS Act, we would consider it an incident-to supply for payment and coding purposes under the PFS. Beginning January 1, 2026, CMS will review HCPCS Level II coding applications for all skin substitutes marketed as 361 HCT/Ps through our biannual coding cycle for non-drugs and non-biological products, rather than on a quarterly basis. Skin substitutes that received a 510(k) clearance, PMA approval, or a granted De Novo request will continue to be evaluated in the biannual HCPCS Level II coding cycles.
After careful consideration of the comments, we are finalizing our proposal to evaluate all complete HCPCS Level II applications for skin substitutes in our biannual cycles as proposed.
If skin substitutes that are not licensed under section 351 of the PHS Act are no longer paid as biologicals using the methodology under section 1847A of the Act, as proposed, then the manufacturers of these products would no longer be required to report ASP data to CMS under section 1847A(f)(2) of the Act. However, when ASP data is reported, it may serve as a better estimate of resources across the hospital outpatient and non-facility settings than hospital outpatient MUC data. We proposed to update the rates for the skin substitute categories annually through ( printed page 53747) rulemaking using the most recently available calendar quarter of ASP data, when available, to set the rates. However, we have concerns that using a single, scheduled quarter of ASP data to set payment rates could encourage gaming. We sought comment on the use of a longer timeframe, such as the most recently available four calendar quarters, to set payment rates in future years. In the event ASP is not available for a particular product, we proposed to use the MUC data. If MUC is not available, we proposed to use the product's WAC or 89.6 percent of AWP if WAC is also unavailable, similar to other products for which ASP is used to calculate a payment rate.[108] Once updated use patterns reflecting this policy are available to calculate rates, we proposed using all relevant products and the combined product utilization patterns (OPPS and non-facility) to determine a weighted average per-unit cost by category to set separate payment rates for each of the three new APC groups. We sought comments on our proposed methodology to set and update the payment rates for skin substitutes as well as the rates themselves.
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: The most common recommendation was that CMS should update the payment rates annually using an inflation index, such as the Consumer Price Index for Urban Consumers (CPI-U) or the Producer Price Index (PPI), rather than recalculating them based on new ASP data. Commenters stated that an inflationary update would provide stability and predictability for providers and manufacturers, avoiding the significant rate variability that would occur with annual ASP recalculations, and reduce the regulatory burden on both manufacturers and CMS. Also, since the proposal would no longer require manufacturers to report ASP data for these products, commenters stated there would be very little reliable data to use for future updates. They stated that relying on ASP would perpetuate the same system that has been susceptible to gaming and abuse.
Several commenters recommended CMS to abandon any reliance on ASP data for setting future payment rates. They stated that the ASP system itself is flawed and has led to the pricing distortions and “profiteering” that CMS is trying to correct. Because the data is distorted, using it to set future rates—even if based on claims—will perpetuate the problem. In contrast, one commenter encouraged CMS to continue collecting ASP data for transparency and to support future reimbursement refinements, even if it is not the primary basis for payment.
Response: We appreciate the commenters for their input. We believe that, over time, the ASP data will more accurately reflect the market impacts of our policy to treat skin substitute products as incident-to supplies. By relying on ASP, payment updates will be responsive to changes in the actual cost of skin substitute products as a result of market pressures, whereas an index like the CPI-U is a general inflation measure that does not account for pricing dynamics. Therefore, we believe that updating the payment rates, based on ASP data will more likely result in payment rates that reflect the changing dynamics of the market rather than anchoring the cost to the initial payment rate for CY 2026 based on data collected prior to the implementation of this policy.
Comment: Several commenters expressed concern that using a single, scheduled quarter of ASP data for updates would encourage gaming. MedPAC recommended that if ASP data is used, it should be from a longer timeframe, such as four calendar quarters, to avoid manipulation.
Response: We agree that, moving forward, using a longer timeframe for collection of ASP data would reduce the opportunity for manipulation. While we may be unable to use a longer timeframe if we propose updated rates for 2027, we will look to use additional quarters of data to set rates in future years.
Comment: Several commenters argued the proposal may violate the Administrative Procedure Act (5 U.S.C. 706(2)(A)), which requires well-reasoned analysis for major policy changes. A commenter argued the drastic reimbursement reduction constitutes a de facto adverse National Coverage Determination without following statutory NCD requirements: public notice, consultation with advisory committees, evidence consideration, and clear basis statements (42 U.S.C. 1395y(l)(3)-(4)).
Response: This rule finalizes a proposed change to payment policy. It neither constitutes, nor is akin to, a national coverage determination as it does not make any coverage determinations but merely makes a change to the amount of payment made for certain covered skin substitutes. Section 1869(f)(1)(B) of the Act (that is, 42 U.S.C. 1395ff(f)(1)(B)) defines the term “national coverage determination” as “a determination by the Secretary with respect to whether or not a particular item or service is covered nationally under this subchapter, but does not include a determination of what code, if any, is assigned to a particular item or service covered under this subchapter or a determination with respect to the amount of payment made for a particular item or service so covered”. The provision the commenters cite, 42 U.S.C. 1395y (section 1862 of the Act) expressly incorporates this definition of the term. See 42 U.S.C. 1395y(a)(25) (section 1862(a)(25) of the Act). Because this is not a “national coverage determination”, the requirements set forth in 42 U.S.C. 1395y(l)(3)-(4) do not apply on their own terms.
Moreover, we believe our proposal satisfies the requirements of the Administrative Procedure Act and is not arbitrary and capricious because we have, for example: (1) Explicitly considered and responded to a dramatic increase in Medicare spending for this class of products, (2) Provided a reasoned explanation for our classification of non-section 351 products as incident-to supplies rather than biologicals separately payable under section 1847A of the Act, (3) Articulated our rationale for using OPPS utilization data and excluding non-facility utilization because of the profit-maximizing incentives distorting the latter, (4) Explained the methodology for calculating the final payment rate with sufficient detail to allow verification by the public, (5) Offered and analyzed several alternatives and explained why our final approach better serves statutory objectives, (6) Acknowledged potential access concerns while reasonably concluding that a sufficient number of manufacturers have demonstrated the ability to provide these products at or below the final payment rate and the assurance that we will monitor and adjust the policy in future rulemaking as necessary, and, finally, (7) Described and responded to the comments CMS received in response to the CY 2026 OPPS/ASC proposed rule.
Several comments, including those related to coverage and the skin substitute LCDs and requests to change how the FDA regulates products CMS considers skin substitutes for payment purposes were out of scope for purposes of this rulemaking. ( printed page 53748)
5. Summary
To implement this policy, we are finalizing, starting January 1, 2026, to separately pay for skin substitute products as incident-to supplies in both the non-facility and hospital outpatient settings. Under the OPPS, we are finalizing our proposal to unpackage the skin substitute product from the payment for the administration of the skin substitute product and pay for the administration of the skin substitute product separately from the skin substitute product itself. Accordingly, we are deleting the C-codes describing the skin substitutes assigned to the low-cost group, HCPCS codes C5271 through C5278. We did not propose to delete the HCPCS codes assigned to the high-cost group, described by CPT codes 15271 through 15278, as they would remain to describe skin substitute administration procedures. We are specifying that CPT add-on administration codes 15272, 15274, 15276, and 15278 would still be packaged in the outpatient hospital setting.
We are finalizing our proposal to pay separately for certain groups of skin substitute products as incident-to supplies involved in furnishing services under both the physician non-facility and outpatient hospital settings. Unless a skin substitute meets the definition of a biological in section 1847A, in which case the payment methodology under section 1847A would continue to apply, we are finalizing our proposal to assign sheet-form skin substitute products to one of three clinical APCs we are creating based on FDA regulatory categories: APC 6000 (PMA Skin Substitute Products), APC 6001 (510(k) Skin Substitute Products), and APC 6002 (361 HCT/P Skin Substitute Products). We are finalizing our policy to calculate initial payment rates for the APCs using the volume-weighted average ASP for skin substitute products in each group as submitted by manufacturers, when available, and the MUC when ASP is not available. We are finalizing to use the hospital outpatient utilization patterns to set the payment rates for all three categories of skin substitute products, which we are finalizing to pay at a single rate for CY 2026. For CY 2026, this finalized policy would result in an initial payment rate of $127.14/cm2 for APCs 6000 (PMA Skin Substitute Products), 6001 (510(k) Skin Substitute Products), and 6002 (361 HCT/P Skin Substitute Products).
We are converting the current HCPCS codes for skin substitute products to add-on codes, for the purpose of ensuring that an administration code is always billed with a product, and assigning the sheet-form codes to the three APCs based on the product's FDA regulatory category. We are also creating three new unlisted codes to describe skin substitute products that are FDA authorized or cleared but have not yet received a specific individual HCPCS or CPT code: HCPCS codes Q4431 (Unlisted PMA skin substitute product), Q4432 (Unlisted 510(k) skin substitute product), and Q4433 (Unlisted 361 HCT/P skin substitute product). We are finalizing our proposal to assign the unlisted HCPCS codes to the appropriate APCs based on the product's FDA approval or clearance. Specifically, we are finalizing our proposal to assign HCPCS code Q4431 to APC 6000 (PMA Skin Substitute Products); Q4432 to APC 6001 (Unlisted 510(k) Skin Substitute Products); and HCPCS Code Q4433 to APC 6002 (Unlisted 361 HCT/P Skin Substitute Products). We are creating these unlisted codes to prevent delays in Medicare payments for new FDA-approved or cleared skin substitute products that do not yet have a specific HCPCS or CPT code. We are creating a new status indicator, “S1,” for skin substitute products to allow for separate payment under the OPPS. We are assigning status indicator “S1” to all skin substitute products assigned to APCs 6000, 6001, and 6002.
We are finalizing our proposal to update the rates for the skin substitute categories annually through rulemaking using the most recently available calendar quarter of ASP data, when available, to set the rates. In the event ASP data is not available for a particular product, we are finalizing to use the outpatient hospital MUC data. If MUC is not available, we will use the product's WAC or 89.6 percent of AWP if WAC is also unavailable. We are finalizing to include all skin substitute products used across both settings as well as the combined product utilization patterns, as soon as data is available that reflects the results of this policy, to determine a weighted average per-unit cost by group to set the payment rates for each of the three categories. We will evaluate all complete HCPCS Level II applications for skin substitutes in our biannual cycles. We are finalizing our proposal to codify the definition of “biological” as “a product licensed under section 351 of the Public Health Service Act” at §§ 414.802 and 414.902. Finally, we note that these finalized changes for the CY 2026 OPPS skin substitute policy will affect prospective CY 2026 OPPS payments and weights, and as a result will be budget neutralized through the OPPS weight scaler, which accounts for prospective changes in OPPS payments and payment weight. For a discussion of the OPPS budget neutral weight scaler, see section II.A.5. of the CY 2026 OPPS/ASC final rule. We direct readers to section XIII. of the CY 2026 OPPS/ASC final rule for more information on our proposal for payment for skin substitute products applied in the ASC setting.
10. CY 2026 PFS Proposal Regarding Cell and Gene Therapies
In the CY 2026 PFS proposed rule (90 FR 32546 through 32547), CMS proposed that (1) preparatory procedures for tissue procurement required for manufacturing an autologous cell-based immunotherapy or gene therapy be included in the payment of the product itself and (2) that, beginning January 1, 2026, any preparatory procedures for tissue procurement required for manufacturing an autologous cell-based immunotherapy or gene therapy that were paid for by the manufacturer be included in the calculation of the manufacturer's ASP.
As stated in the CY 2026 OPPS/ASC proposed rule (90 FR 33649 and 33650), we made readers aware of this proposal as it may impact therapies paid under the OPPS and ASC payment system. We directed readers to submit their comments on this topic to the CY 2026 PFS proposed rule. We stated that comments submitted to the PFS rule will be responded to in the CY 2026 PFS final rule with comment period along with the finalized policy.
We refer readers to the CY 2026 PFS final rule with comment period for a summary of comments, our responses, and the finalized policy for CY 2026.
11. Medicare Part B Drugs Without a Medicaid National Drug Rebate Agreement (NDRA)
CMS has reviewed drugs, biologicals, and radiopharmaceuticals with HCPCS codes that meet the definition of a covered outpatient drug (defined at 42 CFR 447.502) and are receiving payment under Medicare Part B. In accordance with section 1927(a)(1) of the Act, for payment to be available under Medicare Part B for covered outpatient drugs of a manufacturer, the manufacturer must have entered into and have in effect a Medicaid NDRA. As of the writing of the notice in the CY 2026 OPPS/ASC proposed rule (90 FR 33650 through 33651), our records indicated that the manufacturers of the single source drugs, biologicals, and radiopharmaceuticals listed in Table 115 did not currently have a Medicaid NDRA in effect. Accordingly, we stated ( printed page 53749) that if the manufacturers, or labelers, of these products did not promptly enter into a Medicaid NDRA, Medicare Part B payment would no longer be available for these products, which includes payment under the OPPS and ASC payment system. We stated that these HCPCS codes would be assigned to an OPPS status indicator of E1, which indicates a non-payable status by Medicare when submitted on outpatient claims (any outpatient bill type). Similarly, we stated that these HCPCS codes would be assigned to an ASC payment indicator of B5.
We received public comments on this notice. The following is a summary of the comments we received and our responses.
Comment: A few commenters supported CMS publishing this notice ( printed page 53750) and ensuring manufacturer compliance with statutory obligations. Specifically, one commenter supported CMS actions as they believed it promoted fairness, accountability and consistency across Federal health programs.
Response: We thank commenters for their support.
Comment: Several commenters explained that enforcement of the statute would reduce Medicare Beneficiary access to drug therapies, including those that commenters said had few, or inferior, clinical alternatives. Some of these commenters communicated that participation in a Medicaid NDRA, and the associated 340B agreement, would be financially unsustainable for the manufacturers of these products. A few commenters asked CMS to delay enforcement so CMS could further consider their concerns and so that manufacturers could consider how they may come into compliance, such as considerations regarding implementing the government price reporting infrastructure required and other contractual obligations. Additionally, a commenter stated that they do not believe the 2003 Medicare Prescription Drug and Modernization Act ties Medicare Part B reimbursement to NDRA participation, that past statements from several government agencies do not support that a NDRA is required for Medicare Part B payment, that, in their view, the plain language of the 2021 Consolidated Appropriations Act confirms that Medicare Part B payment is available for manufacturers without NDRAs, and that, in their view, CMS is reinterpreting the statute which places an unconstitutional condition on Medicare Part B payment.
Response: We acknowledge the concerns raised by commenters. However, in accordance with section 1927(a)(1) of the Act, for payment to be available under Medicare Part B for covered outpatient drugs of a manufacturer, the manufacturer must have entered into and have in effect a Medicaid NDRA. For the manufacturers who have not come into compliance with this statutory requirement, Medicare Part B payment for the HCPCS codes that describe their products will be stopped effective April 1, 2026. We note, that if manufacturers satisfy the statutory requirement, Medicare Part B payment will be resumed. We do not believe that an additional postponement is appropriate as this is an existing statutory requirement that manufacturers should have been aware of and already compliant with. In addition, notices were provided to manufacturers in the spring of 2025, and through the CY 2026 OPPS/ASC proposed rule, which have provided manufacturers with more than sufficient time to initiate a Medicaid NDRA.
Comment: One commenter requested additional information from CMS on future enforcement of this statutory requirement and if enforcement will be carried out through notice and comment rulemaking in subsequent years.
Response: Section 1927(a)(1) of the Social Security Act states that for payment to be available under Medicare Part B for covered outpatient drugs of a manufacturer, the manufacturer must have entered into and have in effect a Medicaid NDRA. We believe this requirement is clear, self-implementing, and does not require notice and comment rulemaking to enforce. CMS may, at its discretion, continue to alert manufacturers to any non-compliance we become aware of through notice and comment rulemaking, direct communication to manufacturers, or other means. However, we expect manufacturers that want Medicare Part B payment for their covered outpatient drugs to promptly enter into an NDRA when their product enters the market and maintain that NDRA to continue to receive Part B payment.
Comment: The manufacturer of the product, MACI, stated that MACI is not regulated as a Human Cells, Tissues, and Cellular and Tissue-Based Products by the FDA, and is actually approved by the FDA pursuant to a Biologics License Application under Section 351 of the Public Health Service Act. This commenter believed that HCPCS code J7330 describes their product and is erroneously not being paid by CMS, and they request that CMS make their product a separately payable OPPS drug starting in CY 2026. This commenter did not mention the issue of Medicaid NDRA, rather they generally requested OPPS payment for the product MACI.
Response: We thank the commenter for their engagement. For CMS to pay for this product as a drug under Medicare Part B, the manufacturer must enter into a Medicaid NDRA. As listed in the CY 2026 OPPS/ASC proposed rule, another product manufactured by Vericel was listed as lacking a Medicaid NDRA. This product can be described by HCPCS code J7353 (Anacaulase-bcdb, 8.8 percent gel, 1 gram). Our records do not indicate that the manufacturer, Vericel, of this product, currently has an active NDRA. Therefore, none of the covered outpatient drugs manufactured by this manufacturer can receive payment under Medicare Part B.
After consideration of public comments, we are adhering to the statutory requirement, and CMS is stopping Medicare Part B Payment for HCPCS codes describing products whose manufacturers did not promptly enter into a Medicaid NDRA for their covered outpatient drugs. As of the writing of this CY 2026 OPPS/ASC final rule with comment period, our records indicated that the manufacturers of the single source drugs, biologicals, and radiopharmaceuticals listed in Table 116 do not currently have a Medicaid NDRA in effect. Accordingly, effective April 1, 2026, Medicare Part B payment would no longer be available for these products, which includes payment under the OPPS and ASC payment system. In the April 1, 2026 quarterly OPPS/ASC update, these HCPCS codes will be assigned to an OPPS status indicator of E1, which indicates a non-payable status by Medicare when submitted on outpatient claims (any outpatient bill type). Similarly, these HCPCS codes would be assigned to an ASC payment indicator of B5.
We note that since the notice included in the proposed rule, three of the listed manufacturers came into compliance, describing HCPCS code J2850 (Inj secretin synthetic human), HCPCS codes A9592 (Fluoroestradiol f 18), and J9248(Inj melphalan (hepzato) 1 mg). Therefore, for these three products, we are finalizing a status indicator assignment of “K” indicating nonpass-through drugs and nonimplantable biologicals, including radiopharmaceuticals, that are paid under the OPPS through a separate APC payment. We will continue to closely monitor for continued compliance of these products, and all products, with their Medicaid NDRA obligations and will adjust Medicare Part B payment status accordingly. Upon the publication of this final rule, if a manufacturer immediately contacts CMS with the intent to enter into an NDRA, we will aim to maintain their separate payment for January 1, 2026.
However, if a manufacturer does not enter promptly into an NDRA, then Medicare Part B payment will be stopped as discussed in this rule. If a manufacturer comes into compliance and enters into an NDRA, Medicare Part B payment will be reinstated retroactive to the date that the signed and completed NDRA has been received and accepted by CMS. For clarity and transparency to the public, we will also indicate on the CMS ASP Pricing Files [109] and the OPPS Addendum B 110 ( printed page 53751) which drugs are non-payable under Medicare Part B due to the manufacturer's lack of effectuation of a Medicaid NDRA. We encourage manufacturers to immediately contact CMS regarding their intent to enter into an NDRA.
12. Add-On Payment for Technetium-99m (Tc-99m) Derived From Domestically Produced Molybdenum-99 (Mo-99)
As discussed in prior rulemaking, in collaboration with the Department of Energy, National Nuclear Security Administration (DOE/NNSA), CMS provided an additional payment of $10 for the marginal cost for Tc-99m produced by non-Highly Enriched Uranium (HEU) sources in an effort to support access to Technetium-99m (Tc-99m), a critical radioisotope used in the majority of diagnostic imaging services, and produced through the radioactive decay of molybdenum-99 (Mo-99) from CY 2013 to CY 2025. We stated that we believed such an adjustment would ensure equitable payments in light of market influences, cost differentials, and cost variations and their potential to create significant payment inequities among hospitals (77 FR 68323). Accordingly, we established HCPCS ( printed page 53752) code Q9969 (Tc-99m from non-highly enriched uranium source, full cost recovery add-on per study dose) to describe the Tc-99m radioisotope produced by non-HEU methods and used in a diagnostic procedure.
In January 2022, the Secretary of Energy issued a certification that stated that there is a sufficient global supply of non-HEU-sourced Mo-99 to meet the needs of patients in the United States (86 FR 73270). Additionally, in March 2023, the last major global producer of Mo-99 converted from HEU to low-enriched uranium (LEU) targets, and all Mo-99 used in the United States is now produced without HEU. As such, in the CY 2024 OPPS final rule with comment period (88 FR 81803), we adopted a policy to end the additional $10 add-on payment described by HCPCS code Q9969 (Tc-99m from non-highly enriched uranium source, full cost recovery add-on per study dose) for non-HEU-sourced Tc-99m at the end of CY 2025. We explained that given that all Mo-99 used in the U.S. is now produced without HEU, we believed that starting in CY 2026 the Medicare claims data utilized to set payment rates would only include claims for diagnostic radiopharmaceuticals that utilized non-HEU-sourced Tc-99m, meaning the data would reflect the full cost of the Tc-99m diagnostic radiopharmaceuticals that providers would use in CY 2026. That being the case, we believed there would no longer be a need for the additional $10 add-on payment for non-HEU-sourced Tc-99m for CY 2026 or future years.
In the CY 2025 OPPS/ASC proposed rule with comment period (89 FR 59377 through 59378), we shared that the Department of Energy and other interested parties raised another issue affecting the domestic supply chain for Mo-99 and Tc-99 that, left unaddressed, could also cause payment inequity among outpatient hospital providers. We noted that foreign Mo-99 production has historically been subsidized by foreign governments, resulting in prices below the true cost of production. These artificially low, foreign government-subsidized prices have created a disincentive for domestic investments in Mo-99 production infrastructure and a barrier to entry for new producers, including U.S. companies, which in turn has resulted in unreliable production and periodic shortages. Subsequently, using our equitable adjustment authority under section 1833(t)(2)(E) of the Act, we proposed and finalized a policy to address the payment inequity resulting from the higher cost of Tc-99m derived from domestically produced Mo-99 by establishing a new add-on payment of $10 per dose for radiopharmaceuticals that use Tc-99m derived from domestically produced Mo-99 starting on January 1, 2026. Furthermore, we stated that the Department of Energy, National Nuclear Security Administration (DOE/NNSA) would establish the criteria to certify whether the Tc-99m radiopharmaceutical dose is derived from domestically produced Mo-99 and eligible for the add-on payment, which would be included in the CY 2026 OPPS/ASC proposed rule with comment period. We also stated that we would continue to develop the technical implementation of this additional payment after a definition for domestically produced Tc-99m radiopharmaceutical was established, including any requirements for providers to document that the Tc-99m radiopharmaceutical used in a procedure was domestically produced and could qualify to receive the add-on payment. Additionally, we stated that our intent was to have the regulatory framework for this policy in place prior to the domestic production of Mo-99 and Tc-99m, to increase provider awareness of the availability of additional payments for domestically sourced Tc-99m radiopharmaceuticals. Likewise, producers of domestic Mo-99 would have certainty that the Medicare OPPS payment policy takes into account the additional costs of Tc-99m derived from domestically produced Mo-99.
a. Criteria for Classifying a Tc-99m Radiopharmaceutical Dose as Domestically Produced
As mentioned above, in the CY 2025 OPPS/ASC proposed and final rules with comment period, CMS stated that DOE/NNSA would establish the criteria to certify whether the Tc-99m radiopharmaceutical dose is derived from domestically produced Mo-99 and eligible for the add-on payment, and that the criteria would be included in the CY 2026 OPPS/ASC proposed rule.
In the CY 2026 OPPS proposed rule with comment period (90 FR 33651 through 33653), for purposes of this provision, DOE/NNSA recommended, and CMS proposed, the following definitions:
- Domestically produced dose of Tc-99m: A dose of Tc-99m generated from domestically produced Mo-99;
- Domestically produced Mo-99: Mo-99 that was both irradiated and processed in the U.S.;
- Irradiated: The process of bombarding a uranium or molybdenum target with radiation in order to produce Mo-99, and to specify that irradiation is typically performed with a nuclear reactor or particle accelerator
- Processed: In this context, processed refers to the purification of Mo-99 from irradiated material.
Additionally, DOE/NNSA recommended, and we proposed that a dose of Tc-99m generated from Mo-99 that was irradiated or processed outside the U.S. would not qualify for this add-on payment, even if the Mo-99 was loaded into a Tc-99m generator in the U.S. or if the Tc-99m was eluted [111] at a radiopharmacy in the U.S.
As an example, we noted that Mo-99 imported and shipped separately to a U.S-based generator manufacturer or radiopharmacy, and then loaded into a generator stateside, would not be considered domestically produced Mo-99 for the purposes of this add-on payment. To elaborate, we noted that although the Mo-99 was loaded into a generator or eluted in the U.S., the Mo-99 was irradiated and processed abroad, imported, and then loaded into the domestic generator, and would therefore be excluded from this add-on payment.
Additionally, in the CY 2026 OPPS/ASC proposed rule, as part of the implementation process for the add-on payment for Tc-99m derived from domestically produced Mo-99, per DOE/NNSA's recommended definitions for the purpose of this add-on payment, we proposed to codify the aforementioned definitions and references for domestically produced Mo-99 to § 419.49 to specify when a dose of Tc-99m generated from domestically produced Mo-99 could be eligible for the add-on payment. We received public comments on these proposals.
Comment: We received public comments on DOE/NNSA's recommended definitions for the purpose of this add-on payment and its codification to § 419.49 to specify when a dose of Tc-99m generated from domestically produced Mo-99 would be eligible for the add-on payment. Most commenters, including manufacturers, producers, trade associations, health systems, and providers, were largely supportive of the add-on payment for Tc-99m derived from domestically produced Mo-99 and also supported the proposed technical implementation plan, including the recommended definition, the stated threshold levels, and the codification of eligibility for this add-on payment to § 419.49.
Response: We thank the commenters for their support. ( printed page 53753)
Comment: We received a few comments requesting that we delay implementation of this add-on payment for Tc-99m derived from domestically produced Mo-99 until domestic production of Mo-99 begins.
Response: As we stated above and in prior rulemaking (89 FR 94258 through 94259), while we recognize that there may not be domestic production of Mo-99 in CY 2026, we believe it is better to have a regulatory framework for this policy in place for when domestic production of Mo-99 begins. This way, providers will be knowledgeable about the availability of additional payments for domestically sourced Tc-99m radiopharmaceuticals, and producers of domestic Mo-99 will be certain that the Medicare OPPS payment policy will take the additional costs of domestic production of Mo-99 into account. We further note that the technical steps that we have taken to operationalize this policy do not mean that payment will be made for Tc-99m derived from domestically produced Mo-99 in CY 2026. We recognize that domestically produced Mo-99 is not yet available.
After review of the public comments, for the purposes of this add-on payment, we are finalizing our proposal without modification to define a domestically produced dose of Tc-99m as a dose of Tc-99m generated from domestically produced Mo-99. Whereas domestically produced Mo-99 means Mo-99 that was both irradiated and processed in the U.S., and “irradiated” refers to the process of bombarding a uranium or molybdenum target with radiation in order to produce Mo-99. Specifying that irradiation is typically performed with a nuclear reactor or particle accelerator. Lastly, DOE/NNSA recommended, and we proposed here, to define “processed” in this context to refer to the purification of Mo-99 from irradiated material. We are also finalizing our proposal without modification to codify the aforementioned definitions and references for domestically produced Mo-99 to § 419.49 to specify when a dose of Tc-99m generated from domestically produced Mo-99 could be eligible for the add-on payment.
We refer readers to section V.B.12.c of this final rule with comment period for additional discussion of commenters' feedback on policy modifications, including documentation requirements, the add-on payment amount, and implementation guidance.
b. Coding and Documentation for the Add-On Payment for Tc-99m Derived From Domestically Produced Mo-99
As discussed above and in the CY 2026 OPPS/ASC proposed rule, from CY 2013 to CY 2025, CMS provided an additional payment of $10 for the marginal cost for radioisotopes produced by non-HEU sources (77 FR 68323). Under this non-HEU add-on payment policy, hospitals reported HCPCS code Q9969 (Tc-99m from non-highly enriched uranium source, full cost recovery add-on per study dose) once per dose, along with any diagnostic scan or scans furnished using Tc-99m, as long as the Tc-99m doses used could be certified by the hospital to be at least 95 percent derived from non-HEU sources (77 FR 68323).
Consistent with the non-HEU add-on payment policy implementation plan, in the CY 2026 OPPS proposed rule with comment period (90 FR 33651 through 33653), CMS proposed to establish new HCPCS C-code C9176 (Tc-99m from domestically produced non-HEU Mo-99, [minimum 50 percent], full cost recovery add-on, per study dose), effective January 1, 2026. CMS stated that hospitals could report new HCPCS C-code C9176 once per dose, along with any diagnostic scan or scans furnished using Tc-99m derived from domestically produced Mo-99, if they could certify that at least 50 percent of the Mo-99 in the Tc-99m generator to produce the Tc-99m was domestically produced Mo-99.
Similarly, we also stated that we would align the certification and tracking requirements to the non-HEU add-on payment certification and tracking requirements and that we would expect hospitals requesting this additional payment to perform standard due diligence to ensure that their claims are supported by internal records.
We received comments on the establishment of new HCPCS C-code C9176 to facilitate the add-on payment for Tc-99m derived from domestically produced Mo-99.
Comment: Commenters largely supported our proposed establishment of new HCPCS C-code C9176 (Tc-99m from domestically produced non-HEU Mo-99, [minimum 50 percent], full cost recovery add-on, per study dose) to facilitate the add-on payment for Tc-99m derived from domestically produced Mo-99, effective January 1, 2026.
Response: We thank the commenters for their support.
After review of the public comments, for the purposes of this add-on payment, we are finalizing the establishment of new HCPCS C-code C9176 (Tc-99m from domestically produced non-HEU Mo-99, [minimum 50 percent], full cost recovery add-on, per study dose), effective January 1, 2026.
We refer readers to section V.B.12.c of this final rule with comment period for additional discussion of commenters' feedback on policy modifications, including the certification and tracking requirements, the amount of the add-on payment, and requests for additional guidance.
c. Comment Solicitation on the Add-On Payment for Tc-99m Derived From Domestically Produced Mo-99
As stated in prior rulemaking, CMS aims to account for the per-dose cost of Tc-99m derived from domestically produced Mo-99 while limiting administrative burden for hospitals and protecting the Medicare trust fund. In the CY 2026 OPPS/ASC proposed rule, we sought comment on the following:
- We asked commenters to provide any insight regarding the irradiation and processing required to produce Mo-99 that may be relevant.
- As we mentioned in the CY 2025 OPPS/ASC final rule with comment period, available information from the Organization for Economic Co-operation and Development, Nuclear Energy Agency (OECD/NEA) supports an add-on payment amount of $10 as appropriate to address the cost of Tc-99m derived from domestically produced Mo-99 for hospital outpatient departments. However, we sought additional information that could further inform the cost differentials between radioisotopes derived from domestic and foreign-produced Mo-99, including, but not limited to, production, transportation, and storage costs that outpatient hospital departments may incur that would not be accounted for in historical claims data. We stated that CMS may consider reevaluating the amount of the add-on payment if we receive new, substantial information to inform these cost differential factors of Tc-99m derived from domestically produced Mo-99.
- In the CY 2026 OPPS/ASC proposed rule, CMS proposed that 50 percent of the Mo-99 used in the Tc-99m generator to produce the Tc-99m must be domestically produced Mo-99 to qualify for the add-on payment. We sought feedback on the appropriateness of the proposed threshold and additional factors that CMS may consider when defining the eligibility threshold.
- We asked what forms and levels of documentation would be most viable and efficient for tracking and certifying ( printed page 53754) if the Mo-99 and Tc-99m were domestically produced?
- We asked what additional steps CMS could take to reduce administrative burden or improve tracking of domestically produced Mo-99 for purposes of this add-on payment?
The following is a summary of the comments we received and our responses.
Comment: As stated above, several commenters supported the proposed add-on payment, the intent to support domestic production of Mo-99, and the proposed definitions pertaining to domestically produced Tc-99m. However, while noting their overall support, several commenters also shared feedback regarding potential implementation challenges, including transparency, regulatory guidance, and the potential for administrative burden.
Specifically, some commenters asked CMS to provide clear operational guidance on the documentation that providers must maintain to verify that at least 50 percent of the Mo-99 in Tc-99m generators is domestically sourced and to ensure that this documentation and tracking requirements are straightforward, administratively feasible, and do not create barriers to access. Some commenters cited the guidance related to code Q9969 as an example of clear operational guidance that minimizes the potential for administrative burden. Other commenters requested that CMS continue to engage with industry and other Federal counterparts in the development of future guidance. A few commenters shared high-level workflows and information flows that could pose as challenges or potential gaming opportunities, including opportunities to game the threshold amount. Commenters also suggested potential collaborations and engagement opportunities that could help inform future payment strategies and policy refinements.
Response: We thank commenters for their input. In the CY 2025 OPPS/ASC final rule with comment period (89 FR 94256 through 94259), we stated that we would continue to develop the technical implementation of this additional payment after we established a definition for domestically produced Tc-99m radiopharmaceutical, including any requirements for providers to document that the Tc-99m radiopharmaceutical used in a procedure was derived from domestically produced Mo-99 and could qualify to receive the add-on payment. We will consider the comments received in response to this comment solicitation for future guidance.
Comment: We received several comments regarding the amount of this add-on payment. Most commenters offered support for the initial add-on amount of $10 in addressing the payment inequity resulting from the higher cost of Tc-99m derived from domestically produced Mo-99. Several commenters disagreed with the appropriateness of an additional payment amount of $10 in addressing the payment inequity resulting from the higher cost of Tc-99m derived from domestically produced Mo-99. One commenter stated that there would not be a payment inequity and that the $10 add-on amount was unnecessary. Per this commenter, costs of Tc-99m derived from domestically produced Mo-99 should actually be less expensive than non-domestically produced Mo-99, as the commenter believes that suppliers would not be able to price the domestically produced Mo-99 higher than non-domestically produced Mo-99 given current industry competition and contracts. Other commenters shared concerns that an add-on amount of $10 would be insufficient. These commenters stated that $10 would not be enough to address cost differentials associated with domestic Mo-99 supply, including costs associated with production, transport, and storage. These commenters requested that CMS provide additional analysis to support the amount of the add-on payment and to continue to work with interested parties to evaluate whether $10 continues to be an adequate level of reimbursement to offset increased costs of sourcing domestically produced radioisotopes. Of those commenters who found the proposed add-on amount insufficient, a few of those commenters offered recommendations to improve the amount, including suggestions to increase the Tc-99m add-on amount to $15 per dose and various other suggestions for adjustment methodologies.
Response: We thank commenters for their feedback on this proposal. As previously stated above and in prior rulemaking, we welcome commenters to provide data to inform rate setting and to support assertions that the price differential between domestically produced and foreign produced Mo-99 will be more or less than $10. We reviewed the submitted information and available data on cost differentials associated with domestic Mo-99 supply. We did not find that the information provided was sufficient to demonstrate that the $10 add-on payment was inappropriate to address the additional cost to hospital outpatient departments of utilizing domestically produced Tc-99m radiopharmaceuticals.
We find that $10 remains an appropriate estimate of the incremental per-dose cost for Tc-99m derived from domestically produced Mo-99 given the uncertainties of the true cost of Tc-99m derived from domestically produced Mo-99 at this time. We note that we review our claims data on an annual basis to establish the OPPS payment rates, and we will continue to monitor data related to this add-on payment and potentially adjust the payment amount if warranted in future years.
Comment: Numerous commenters requested an expansion of this add-on payment for Tc-99m derived from domestically produced Mo-99 as a means to extend support for other radiopharmaceuticals, such as Xe-133 and I-131, and other domestically manufactured products, including N95 masks, nitrile gloves, medical gowns, syringes, needles, and other items considered as medical essentials. Commenters stated that such add-on payments would align with Executive Orders and protect beneficiaries by alleviating concerns associated with foreign-made medical products.
Response: We thank the commenters for their input. However, we note that comments related to domestically produced radioisotopes beyond Tc-99m and domestically produced medical products are out of scope for the purposes of the proposed Tc-99m add-on payment policy and comment solicitation.
In conclusion, in accordance with our review and comments from interested parties, using our equitable adjustment authority under section 1833(t)(2)(E) of the Act, we are finalizing our proposed implementation plan for the add-on payment for radiopharmaceuticals that use Tc-99m derived from domestically produced Mo-99 without modification. For the purposes of this add-on payment, we are finalizing our proposed definition of a domestically produced dose of Tc-99m, codification of the definition under § 419.49, and the establishment of new HCPCS C-code C9176 (Tc-99m from domestically produced non-HEU Mo-99, [minimum 50 percent], full cost recovery add-on, per study dose), effective January 1, 2026.
C. Notice of Intent To Conduct Medicare OPPS Drugs Acquisition Cost Survey
Section 1833(t)(14)(A)(iii) of the Act requires the Secretary to set payment rates for specified covered outpatient ( printed page 53755) drugs (SCODs) [112] beginning in 2006 at the amount the Secretary determines to be the average acquisition cost for the drug for that year, at least when certain hospital acquisition cost survey data is available. To collect the cost survey data for the Secretary to use for 2006 payment rates, section 1833(t)(14)(D)(i)(I) of the Act required the Comptroller General of the U.S. to conduct a survey in each of 2004 and 2005 to determine the hospital acquisition cost for each SCOD. To inform payment rates in later years, section 1833(t)(14)(D)(ii) of the Act requires the Secretary periodically to conduct surveys of hospital acquisition costs for each SCOD. In developing that survey, section 1833(t)(14)(D)(i)(II) of the Act requires the Secretary to take into account certain recommendations from the Comptroller General regarding frequency and methodology of subsequent surveys.
The GAO conducted the required surveys in 2004 and 2005, and, in reporting the results in 2006, recommended that the Secretary thereafter validate, “on an occasional basis—possibly every 5 or 10 years—ASP data that manufacturers report to CMS for developing SCOD payment rates.” [113] CMS has not yet conducted a survey of the acquisition costs for each SCOD for all hospitals paid under the OPPS. Additionally, on April 18, 2025, President Trump signed Executive Order (E.O.) 14273, “Lowering Drug Prices by Once Again Putting Americans First.” [114] Section 5 of the E.O., “Appropriately Accounting for Acquisition Costs of Drugs in Medicare,” directing the Secretary of HHS to publish in the Federal Register a plan to conduct a survey under section 1833(t)(14)(D)(ii) of the Act so he can determine the hospital acquisition cost for covered outpatient drugs at hospital outpatient departments.
Accordingly, in the CY 2026 OPPS/ASC proposed rule, we announced that under section 1833(t)(14)(D)(ii) of the Act we would be conducting a survey of the acquisition costs for each separately payable drug acquired by all hospitals paid under the OPPS, including SCODs, and drugs and biologicals CMS historically treats as SCODs. We indicated that this survey would be open starting at the end of CY 2025 to early CY 2026. We also stated that we had reviewed and taken into account the Comptroller General's recommendations regarding the frequency and methodology of these surveys in developing our proposed survey, and that we intended for the survey to be completed in time for the survey results to be used to inform policy making beginning with the CY 2027 OPPS/ASC proposed rule. We indicated that we intended to propose and seek comment on any payment rates for SCODs based on the survey results in CY 2027 rulemaking.
Comment: Most commenters opposed our announcement that we will be conducting a survey of the acquisition costs for each separately payable drug acquired by all hospitals paid under the OPPS, including SCODs, and drugs and biologicals CMS historically treats as SCODs and urged CMS to reconsider the necessity and timing of the proposed survey.
Many of these commenters stated that, given the complexity and scale of the required data collection and analysis, conducting the survey will impose a significant burden on hospitals and that CMS' estimate of that burden grossly underestimates the cost, time and resources that will be necessary to complete the survey. Commenters also claimed that CMS is wrong to assume that the reporting will be done by pharmacy technicians and that the survey will actually be completed by pharmacists that will cost more. One commenter opined that completing the survey would demand the concerted effort of multi-disciplinary teams, including pharmacy, supply chain, finance, legal, and reimbursement professionals, far beyond what pharmacy technicians alone could provide. The commenter stated that CMS' own Information Collection Review document “acknowledges the breadth of the undertaking but fails to account for the extensive coordination needed with vendors and suppliers, from whom much of the requested data must be sourced. The time and resources required to collect, clean, analyze, and accurately report thousands of distinct drug prices would divert critical personnel from patient care and other essential hospital functions”. Many other commenters emphasized that the cost of completing the survey would come at the expense of the hospital's ability to provide essential care to patients and quoted the GAO's 2006 report to Congress in which the GAO concluded that the surveys it conducted “created a considerable burden for hospitals” and that to submit the required price data hospitals “had to divert staff from their normal duties, thereby incurring additional costs.” Many commenters stated that the financial burden of completing the survey would be exacerbated by upcoming Medicaid and Medicare reductions under the Inflation Reduction Act.
Response: We take the burdens on hospitals of completing the survey and the GAO's conclusions relating to that burden very seriously in the design and implementation of the survey. We have created a survey instrument and survey process that we think will minimize, to the extent reasonably possible, the staffing and financial burden on hospitals of collecting and reporting the necessary information to CMS. The survey instrument consists of a streamlined online portal, where hospitals can either directly enter acquisition costs or download and reupload an excel template of acquisition costs. There will be technical assistance available for any hospital staff with any issues that arise during the submission process. We have taken the feedback from commenters into careful consideration when finalizing the survey tool.
We are also engaging in robust education and outreach efforts with hospitals to reduce the burden of completing the survey. These activities consist of:
- Obtaining user feedback from hospitals. We worked with a small group of hospital representatives to learn about their operations and business practices so that we could thoughtfully incorporate that knowledge into the survey design.
- Providing hospitals with a comprehensive communications plan. We are outlining thorough communications to support hospital representatives and system users throughout the onboarding, training, and implementation phases of the survey process.
- Providing hospitals with webinar training sessions. We plan to host two webinar sessions to provide more details on the registration and data submission processes for the survey, and to offer hospital representatives an opportunity to ask questions in a dedicated question-and-answer session.
- Providing hospitals with accessible resources. We have created a survey website (https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient-pps/outpatient-prospective-payment-system-opps-drug-acquisition-cost-survey) to house resources for hospital representatives and system users, including user guides, frequently asked questions, fact sheets, and a data collection template for ease of use. ( printed page 53756)
- Creating a survey instrument with an intuitive design and application support. The survey instrument is being built with ease of use in mind. The screens are simple, featuring enabled tooltips, descriptive error messages, downloadable copies of the user guide, and access to dedicated helpdesk contact information.
- Providing hospitals with dedicated technical support. There is a dedicated Helpdesk email (OPPSdrugsurvey@cms.hhs.gov) to support users with any technical issues they may have with the system and a Helpdesk phone number is forthcoming.
But many of commenters' arguments appear to rest on a belief that CMS should never conduct a survey. We cannot reconcile that position with the statute, which requires the Secretary to “conduct periodic surveys,” or GAO's advice to conduct those surveys every five or 10 years. The Secretary has not yet conducted any such survey. We reiterate that future surveys will be periodic in nature, which will limit the burden that hospitals face on a reoccurring basis.
Comment: Many commenters stated that the reason CMS has never previously conducted a survey of the acquisition costs for each SCOD for all hospitals paid under the OPPS is because of the immense administrative burden it would impose on both hospitals and CMS. One commenter stated that that “CMS has historically opted out of conducting this survey for several reasons—including undue burdens and inaccuracy of results—and these realities have not changed”. The commenter claimed that the burden of conducting the survey was so significant that GAO recommended that the survey be used only to validate “ASP data that manufacturers report to CMS for developing SCOD rates” and that “CMS declined to conduct a survey between 2006 and 2019, influenced by GAO's analysis, and cited the survey's burden on both hospital staff and the agency.” The commenter additionally noted that “[a]s recently as November 2021, HHS publicly acknowledged the significant burden the survey has on the agency and hospitals. During a Supreme Court oral argument, HHS stated that the surveys are `very burdensome on the study takers', `very burdensome on the hospitals', and `do not produce results that are all that accurate.' ” The hospital acquisition cost survey, the commenter concluded, “would divert resources from patient care, contribute to inefficiencies, and create financial strain on hospitals at a time when it is a CMS priority to actively reduce such inefficiencies and burdens.” Similarly, several other commenters questioned the choice of conducting a survey that imposes a significant burden on hospitals “given the Administration's goal of reducing administrative burdens”.
Response: We acknowledge, as we did in the CY 2026 OPPS/ASC proposed rule, that we have not previously conducted a survey of the acquisition costs for each SCOD for all hospitals paid under the OPPS. We also acknowledge that surveys can be administratively burdensome both to recipients and administrators. However, as discussed in our response to the previous comment, we believe that the survey instrument and accompanying process that we have devised will alleviate to the greatest extent possible the staffing and financial burdens identified by the GAO and HHS. Again, regardless of the burden involved, we are required to comply with section 1833(t)(14)(D)(ii) of the Act which requires “the Secretary to conduct periodic . . . surveys to determine the hospital acquisition cost for each specified covered outpatient drug for use in setting the payment rates.” E.O. 14273 accordingly directs the Secretary to publish in the Federal Register a plan to conduct a survey under section 1833(t)(14)(D)(ii) of the Act so he can determine the hospital acquisition cost for covered outpatient drugs at hospital outpatient departments.
Comment: One commenter stated that the burden of completing the survey would be even greater for 340B covered entities not only because they typically have limited administrative resources, but because the survey would require 340B hospitals to report twice the amount of data as other hospitals.
Response: We understand the commenter's concerns; however, we believe it is prudent to collect all discounts, rebates, and other price concessions that are provided to hospitals, including 340B discounts, so that we have data that reveals the actual acquisition costs of the drugs that hospitals purchase.
Comment: One commenter stated that the survey is broader than permitted by law because it includes drugs that are not included in the definition of specified covered outpatient drugs under section 1833(t)(14)(B) of the Act. The commenter also expressed surprise that CMS is proposing to exclude radiopharmaceuticals from the survey when those drugs are explicitly included within the statutory definition of SCODs. Conversely, one commenter agreed with CMS' decision to exclude radiopharmaceuticals, as the commenter stated that these products present unique characteristics that make their inclusion in a broad hospital acquisition cost survey problematic. They stated that radiopharmaceuticals are used in highly specialized clinical settings, often with low-volume distribution. Another commenter requested that CMS exclude all separately payable radiopharmaceuticals from the survey. The commenter recommended that doing so while using targeted alternative data sources would ensure that OPPS payment rates remain accurate, support continued patient access, and minimize administrative burden on hospitals.
Response: We acknowledge, as we did in the CY 2026 OPPS/ASC proposed rule, that the proposed survey includes both SCODs and drugs and biologicals that we have historically treated as SCODs for payment purposes. As we explained, we have historically treated non-SCOD drugs as SCODs for payment purposes since CY 2006. We believe that drug pricing and payment is a complex and dynamic issue; therefore, CMS, beneficiaries, and taxpayers would benefit from the additional details that the survey will provide on acquisition costs for non-SCODs, which represent the majority of drugs currently separately paid under the OPPS.
As discussed in the draft Supporting Statement A for this data collection, for the NDCs included in the survey template we excluded radiopharmaceutical NDCs from our proposed list of NDCs. Radiopharmaceuticals are unique products with various forms in which they can be purchased—The GAO explicitly acknowledged these unique characteristics in the original survey. While GAO still collected information, and analyzed data, for radiopharmaceuticals, it specifically acknowledged that “their complex nature as compared with drugs poses challenges for collecting and interpreting cost data” and the “GAO found that the diversity of forms in which they can be purchased—ready-to-use unit doses, multidoses, or separately purchased radioactive and non-radioactive substances—complicates CMS' efforts to select a data source that can provide reasonably accurate price data efficiently”.[115] We believe that, due to these unique characteristics and challenges, the burdens of collection for radiopharmaceuticals outweigh the benefits, and so we should not include radiopharmaceuticals in the survey at ( printed page 53757) this time. This will also reduce hospital reporting burden. We thank the one commenter for their support regarding our proposal, and finalized policy, to exclude radiopharmaceuticals from the OPPS drug acquisition cost survey at this time. With respect to the commenter that suggested that we exclude all radiopharmaceuticals from the survey, we confirm that we are doing so.
Comment: One commenter, noting that the survey does not have an OMB approval number, indicated it had already received email communications regarding the survey and cautioned CMS that it should not be taking efforts to conduct the survey before it has been finalized via rulemaking.
Response: We can assure the commenter that CMS has not begun collecting drug acquisition cost data prior to OMB approval of the survey package. CMS has alerted hospitals paid under the OPPS of our intent to conduct the survey so that they would be aware of the survey if finalized in this rulemaking and assigned an approved OMB control number.
Comment: One commenter stated that CMS' survey methodology contradicts GAO guidance and will consequently lead to unreliable results. The commenter stated that when conducting the 2004 survey, the GAO worked with knowledgeable interested parties to develop a survey instrument before sending it to its sample population. According to the commenter, the GAO pretested the survey instrument, made adjustments based on hospital feedback, and engaged a data collection contractor to pilot the revised instrument with a larger group. As a result of the pilot, the commenter contended, GAO adjusted the instructions and changed its procedures before engaging in a multifaceted effort to collect data from about half as many hospitals as CMS intends to survey. The commenter stated that CMS' survey methodology, instrument, and instructions do not compare, “although it intends to undertake a larger survey under the same statutory authority, with the same goal, but far more serious consequences.” To illustrate the alleged deficiencies of CMS' survey as compared to the GAO's, the commenter provided the following example: CMS instructs hospitals to report purchase prices on an NDC-by-NDC basis, net of all discounts, rebates, and price concessions. The commenter pointed out that discounts, rebates and price concessions are not aways reflected in hospitals' purchasing systems. When the GAO faced this same problem in 2004 and 2005, the commenter contended, it determined that “it was generally not feasible to allocate rebates to specific drugs” and limited its review solely to hospital purchase price information, excluding any rebates paid after the hospital's receipt of the product. In contrast, the commenter alleged, CMS instructs hospitals to “estimate the approximate discount received for NDC, deduct that amount from the NDC cost, and report the final cost with all deductions in the template.” CMS, the commenter concluded, provides no further instructions regarding the estimation methodology or even a field for hospitals to indicate which of their reported prices include estimated discounts.
Response: We thank the commenter for their feedback. As we create a survey instrument, we are engaging with hospitals and experts within the hospital and drug purchasing industry, and we are incorporating the valuable feedback received through the notice and comment rulemaking process. The survey will go through extensive user testing, and there will be a thorough onboarding process once an OMB control number is received, which will involve webinars, user guides, fact sheets, additional communications as well as a dedicated email inbox, phone number, and website in order to ensure hospitals are able to respond to the survey. We disagree with commenters to the extent they suggest the statute requires us to mimic the GAO survey in every particular. The statute instead requires us to craft our survey “taking into account” the GAO's recommendations. We have done so. We also note that the world has changed since 2004 and 2005, when GAO conducted its survey tool. For example, hospital systems in 2026 have technology far more advanced and sophisticated than the technology hospitals had over 20 years ago in 2004 and 2005. Similarly, CMS is able to create a more streamlined, sophisticated, and modern survey tool more than 20 years after GAO created its survey tool, while still adhering to relevant principles that GAO used when crafting the original survey in 2004. We thus believe it is reasonable, for example, to require hospitals to report drug discounts to us. Drug pricing and discounts have also drastically changed since 2004 and 2005. For example, according to a GAO Report [116] on Prescription Drug Spending, the amount of money spent on prescription drugs has increased dramatically over the past two decades. They specifically list pharmaceutical rebates as one of the several factors that affect prescription drug prices in the U.S. The rapid growth of drug spending and drug discounts and rebates since the original GAO survey emphasizes the need and increased importance for today's survey to incorporate these factors into the acquisition cost.
Comment: The same commenter faulted the CY 2026 OPPS/ASC proposed rule for failing to recognize the importance of a well-developed survey instrument, clear instructions, and accessible technical support. The commenter found this perplexing, given GAO's experience. The commenter stated that despite the GAO's careful planning and testing, its 2004 survey required extensive, continuous engagement with respondent hospitals: “On average, [contracted] interviewers called each hospital 8 times before receiving a complete data submission”. It was only after this “rigorous, labor intensive” activity by the GAO that the GAO reported that the survey produced accurate hospital drug price data. The commenter stated that it is reasonable to expect that hospitals will similarly need ongoing guidance to navigate the complexities of responding to CMS' proposed survey but states that CMS' proposal gives no indication that hospitals will have any opportunity to seek clarification or technical support during the survey process. This lack of robust instructions and practical support, the commenter alleged, will almost certainly result in inconsistent data, undermining the reliability of the survey results.
Response: We agree with the commenter that it is reasonable to expect that hospitals will need ongoing guidance to navigate the complexities of responding to our proposed survey similarly to how they did with the GAO's survey. However, we disagree that our proposal gives no indication that hospitals will have any opportunity to seek clarification or technical support during the survey process. We also do not agree with the commenter's conclusion that our survey lacks a well-developed survey instrument, clear instructions, and accessible technical support. We have created a survey instrument that we believe minimizes, to the extent reasonably possible, the amount of effort necessary for hospitals to collect and report the required information to CMS. We have established a communication method for the hospitals subject to the survey. We have also established extensive written instructions and several real-time training sessions to provide ( printed page 53758) guidance and receive feedback. For technical assistance we have multiple guidance documents (FAQs, user guides, fact sheets, among others), which will be available on our new dedicated website ( https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient-pps/outpatient-prospective-payment-system-opps-drug-acquisition-cost-survey), as well as a dedicated phone line (forthcoming, to be announced on the above referenced website) and email address ( OPPSdrugsurvey@cms.hhs.gov) that hospitals can contact for assistance before and throughout the survey process. There will be additional assistance tools available within the survey itself to facilitate the completion of the survey by hospitals.
Comment: The same commenter expressed concern that if CMS performs a “deficient survey”, it could undermine the purpose of the 340B program. The commenter cited a finding from GAO's 2005 report that it “could not fully account for rebates or payments from group purchasing organizations”. The commenter stated that many 340B-participating hospitals are subject to a prohibition on purchasing covered outpatient drugs through a group purchasing organization or other group purchasing arrangement, “so the GPO payment issue may not substantially affect them” but “non-340B hospitals may be unable to reasonably allocate GPO payments on an NDC-by-NDC basis, leading to overreporting of their acquisition prices, and creating an artificial gap between 340B and non-340B providers”. The commenter listed other consequences that they alleged would occur as a result of a “deficient survey”. “For example, the price discerned through a retrospective acquisition cost survey will not reflect the true net cost to 340B covered entities for any drugs that, in the future, are subject to both the Medicare Drug Price Negotiation Program (DPNP) under Part B and HRSA's proposed 340B rebate pilot program. For such drugs, HRSA has proposed that 340B covered entities will be required to purchase the drugs at the WAC price, then wait for manufacturers to pay rebates to bring the price down to the 340B ceiling price.” The commenter stated that “[u]nless every manufacturer pays every covered entity every requested rebate—which seems unlikely for the reasons stated in our letter to HRSA regarding its rebate pilot program, which we incorporate here by reference—covered entities' costs for these drugs will be, on average, higher than the 340B ceiling price. Because the CMS survey is retrospective and based on historical acquisition costs, it will systematically understate the actual net cost to 340B covered entities for these drugs. Any payment rates set using such survey data will therefore fail to accurately reimburse covered entities for their true acquisition costs”. The commenter requested that CMS acknowledge this limitation and, if it proceeds with its survey proposal and anticipated reimbursement cuts, clarify how it will ensure that payment rates for Part B drugs subject to both the DPNP and the 340B rebate pilot will fully account for the additional rebate liability imposed on 340B covered entities. The commenter alleged that the complexities associated with the CY 2026 OPPS/ASC proposed survey have only increased since the GAO's report and that the approach proposed by CMS is the wrong solution to resolve budget shortfalls. “It serves only to harm 340B covered entities—threatening this country's safety net front line—in contravention of clear Congressional intent. This approach therefore functionally guts the 340B Program's benefits.”
Response: We thank the commenter for their detailed comment outlining their concerns. We note that HRSA's 340B pilot program is out of scope for purposes of this rulemaking and CMS defers to HRSA for specifics on this program. This pilot program has yet to go into effect, and the period of the survey does not overlap with the pilot program, but we can consider how to incorporate any potential intersection between that program and Medicare payment policy in future rulemaking or future drug surveys released by us. We acknowledge that drug pricing and payment is dynamic and evolving, but that this survey will provide valuable insight into this topic, and will help address some of the same issues that this commenter raises. The survey will bring greater transparency to drug acquisition costs, which also will help address some of the same broader issues that the commenter raises. Whereas not conducting a survey because it might prove difficult only serves to leave the issue opaque for all interested parties.
Comment: Many commenters stated that the survey results would be unrepresentative or quickly obsolete given the volatility in drug pricing and policy due to various factors such as proposed pharmacy benefit manager reform, the implementation of negotiated drug prices under the IRA, 340B drug pricing program rebate policies, the “most favored-nation” policy and pharmaceutical tariffs. They stated that this volatility will preclude a survey conducted in CY 2026 collecting data from 2024 to 2025 from serving as an accurate basis for future Medicare reimbursement policies. Some of these commenters suggested that CMS should use the proposed survey timeframe to instead conduct a test survey, incorporate interested parties' feedback, and learn from GAO's findings of technical challenges while these changes are implemented. Other commenters went further and suggested that the constantly fluctuating nature of drug prices generally would render survey results questionable regardless of when the survey was conducted. These commenters stated that any survey will only yield a point-in-time estimate of a drug's acquisition cost, which can change wildly quarter to quarter and will fail to account for new, expensive drugs that enter the market after the survey is completed.
Response: We acknowledge that surveys are inherently a snapshot of data at a given point in time (indeed, that is their purpose) and that there are multiple factors which can affect drug costs at any given point in time. To a significant extent, the value of a survey is providing a snapshot of data at given point of time which reflects all of the factors that are affecting the data at that point of time. The statute requires that to conduct a survey of drug acquisition costs to inform drug payment amounts to do so on a periodic basis. We note that commenters' observations cut both ways: if drug pricing changes significantly over time, then that supports our decision to collect data now instead of continuing to rely on drug pricing policies set based on GAO surveys from two decades ago. The statute also provides that we do not have to rely solely on survey data to inform our drug pricing policies. For example, as discussed in the CY 2026 OPPS/ASC proposed rule (90 FR 33654), we could consider pricing from the Federal Supply Schedule (FSS), 340B ceiling pricing, ASP plus 6 percent, zero percent or another percentage; or other recognized drug pricing to inform future CMS payment policy. Such policies have been, and will continue to be, set in advance every year through notice and comment rulemaking.
Comment: Some commenters supported our announcement that we will be conducting a survey. These commenters stated that doing so would promote transparency and bring greater alignment of payment with acquisition costs.
Response: We thank commenters for their support. ( printed page 53759)
Comment: Many commenters suggested refinements to various aspects of the proposed survey. Several commenters supported CMS conducting the survey but were concerned about using its results for CY 2027. These commenters cautioned CMS against making “abrupt changes” to the 340B payment policy as a result of the survey. These commenters maintained that CY 2027 is too soon to make changes and 1 year of data is an insufficient basis upon which to make changes. These commenters argued that careful consideration of data validity and analysis is necessary before making changes to payment policy and that inaccurate data collection could lead to reimbursement policies that directly impact Medicare beneficiaries rather than correct any inequities in hospital payment.
Response: We agree with commenters that a careful consideration of data validity and analysis of that data is necessary before making changes to payment policy and, as we stated in the CY 2026 OPPS/ASC proposed rule, we intend to propose and seek comment on any payment rates for SCODs, and drugs historically treated as SCODs, after taking into consideration the survey results in CY 2027 rulemaking. We do not agree, however, that 1 year of data is necessarily an insufficient basis upon which to make changes. The commenter cites no statistical support for such a categorical conclusion, which is likely overbroad. And under the statute, we adjust payment rates for drugs on an annual basis, suggesting Congress anticipated year-to-year policy updates. Indeed, the further we get from the survey to make payment policy the more likely it is that we run into the problem of there being too long of a lag. We have to strike an appropriate balance, and 1 full year of data from the most recent year seems reasonable.
Comment: One commenter stated that the proposed start of the survey is too soon. The commenter stated that hospitals need time to identify, validate and coordinate drug cost information across internal departments and that without additional advance notice and technical guidance hospitals may be unable to respond comprehensively within the proposed survey window. The commenter requested that CMS extend the timeline for survey development and hospital response to ensure robust participation and accurate data collection.
Response: We appreciate commenters' concerns about hospitals having sufficient time to identify, validate and coordinate drug cost information across internal departments and the need to provide hospitals with advance notice of the survey and technical guidance with respect to it. However, we do not believe it is necessary to delay the proposed start of the survey to address these concerns. As we have stated in our previous responses to comments, our survey has been designed to minimize the amount of effort required by hospitals to collect and report the information. With respect to notice, in addition to the notice provided by the CY 2026 OPPS/ASC proposed rule itself, we have alerted all hospitals subject to the survey to make them aware of the potential for a survey. With respect to technical assistance, in addition to multiple guidance documents on our website, we will conduct training sessions for hospitals, hold webinars, publish extensive onboarding materials, and we have established a dedicated helpline to provide one-on-one support and assistance.
Comment: Several commenters expressed concern about the length of the survey window, arguing that 90 days was not enough time given that survey completion will require a multi-disciplinary team at any hospital, including but not limited to pharmacy, supply chain, finance, and reimbursement. One commenter argued that smaller/independent 340B-eligible hospitals that lack the appropriate infrastructure or the support of system resources would struggle to meet a 90-day deadline.
Response: As discussed in the previous comment, we have designed our survey to require as little effort as possible on the part of the hospital to collect and report the required data. Consequently, we believe that 90 days is enough time for hospitals to collect and report this data to us. Hospitals have been on notice regarding the fundamentals of the survey since the proposed rule publication in July of 2025, and the associated PRA packaged contains the outline of the survey, which is nearly identical to the final survey. Therefore, in July of 2025 hospitals were aware of the approximate drugs that would be surveyed as well as the information that they would be required to provide. Even if hospitals had not been provided this notice, we still believe 90 days is more than adequate time given that this acquisition cost information should already be at the hospitals disposal and can easily be compiled into the minimal data fields of the survey.
Comment: One commenter supported the CY 2026 OPPS/ASC proposed scope of data to be collected, stating that CMS' proposal to collect data on purchases during a 1-year timeframe of July 1, 2024 through June 30, 2025 was an “optimal” timeframe. The commenter stated that since the acquisition costs of drugs frequently change, an average acquisition cost calculated over a shorter timeframe might be skewed by temporary supply-chain issues or other factors that would cause the average acquisition cost to be under-or over-stated. In contrast, the commenter contended, a 1-year acquisition timeframe would appropriately capture the range of acquisition costs for each surveyed drug in that year. The commenter also stated that responding to the survey with acquisition data for a 1-year timeframe would be no more burdensome than responding with acquisition data for a shorter timeframe.
Response: We thank the commenter for their support.
Comment: One commenter also supported CMS' statement that it anticipated conducting the survey no more than every 4 years, noting that this would minimize survey burden. The commenter additionally noted that a revised survey is not necessary to adjust the payment amount under section 1833(t)(14)(A)(iii)(I) of the Act. By statute, the commenter pointed out, the average acquisition costs for the drug and hospital group must be determined “taking into account the hospital acquisition cost survey data,” and consequently CMS can use other data to keep payment amounts current. The commenter concluded, “Thus, if the survey indicates that the average acquisition cost for a particular drug for non-340B hospitals is equal to 3percent more than the ASP, the average acquisition cost should automatically update with CMS' quarterly updates to its ASP data so that it does not fall below or increase above the survey-determined amount of 3 percent more than the ASP.”
Response: We thank the commenter for their support and input on potential ways to adjust payment amounts following the survey.
Comment: One commenter provided input on balancing data validation with confidentiality. The commenter acknowledged that some data transparency is necessary to ensure the integrity of the payment system and appropriateness of payment but expressed concern that if data is identifiable to individual hospitals, it might suppress responses in light of the sensitivity of drug pricing data. To promote a broad response while still ensuring that third parties can replicate CMS' acquisition cost estimates by drug and class of hospital, the commenter recommended that all four data fields ( printed page 53760) (total units and net acquisition costs for 340B and non-340B drugs) be made available at the hospital level with pseudonymized provider numbers, but that all hospital identifiers (for example, state or location data, NPIs, etc.) be excluded from the data sets that can be accessed by the public or those with data use agreements.
Response: We agree with the commenter that it is necessary to balance data transparency with confidentiality and appreciate the suggestion as to how we might do so. We believe that the survey we have designed achieves this balance. We of course intend to follow relevant privacy laws. Only the Department of Health and Human Services (HHS) and relevant component agencies, their contractors and representatives, and the Executive Office of the President (EOP) will have access to these data unless such data are otherwise required to be released to other parties by law. Following the collection of data and its analysis, the aggregate results of the survey will be considered when determining proposed payment rates. The proposed payment rates and calculations with aggregate data could appear in notice and comment rulemaking to ensure transparency and replicability; however, the confidentiality of individual hospitals and proprietary information would be maintained to the extent permitted by law. To the extent that acquisition costs are deemed sensitive and/or confidential, we do not intend to make such prices available in an individually identifiable manner.
Comment: Another commenter requested that Disproportionate Share (DSH) eligible Covered Entities be allowed to submit unified responses via their system Chain Home Office, be exempted from any consequences of not responding to the survey (as the commenter expects all covered entities will participate) and “limit the survey to only the information necessary to carry out the Agency's immediate policy objectives.”
Response: We are not treating any hospital type or group differently for purposes of conducting this survey. All hospitals paid under the OPPS are required to respond to the survey. We note that Critical Access Hospitals are not included in the survey pool since they are not paid under the OPPS.
Comment: Many commenters opined that limiting data collection to hospitals would provide an incomplete picture of acquisition costs and that to ensure accuracy and comprehensiveness, CMS should use supplemental data sources and engage drug manufacturers and distributors and other entities involved in the supply chain to incorporate data points such as the cost of goods sold and any applicable discounts and rebates.
Response: We agree with commenters to the extent that CMS should incorporate information from supplemental data sources into the survey, and we have designed our survey to allow for the incorporation of information from supplemental data sources, including manufacturers and distributors, that are available to the hospitals. We encourage hospitals to engage with their partners, such as wholesale distributors, to ensure the accuracy of their survey submission. Additionally, we note that when considering any future payment policy, we will consider taking into account the results of the survey, as well as any additional appropriate external sources of information, such as those suggested by the commenter.
Comment: One commenter requested that CMS provide clear, detailed technical instructions well in advance of the survey opening and asked the following questions about the survey methodology:
- What specific acquisition cost elements will be required (for example, invoice price, discounts, rebates, administrative fees)?
- How will CMS ensure comparability across hospitals with different purchasing arrangements (for example, group purchasing organizations, 340B participation, direct contracts)?
- How will CMS protect sensitive hospital purchasing data from public disclosure?
- How will CMS reconcile acquisition cost data with existing manufacturer reported ASP to avoid duplication or conflicting datasets?
Response: We are asking hospitals to incorporate all rebates and discounts in their acquisition cost for each NDC—including discounts directly applicable to an individual NDC—but also those discounts that are not necessarily linked to a single NDC. That discount could be one linked to a certain invoice, or discounts linked to purchases made over a certain time period such as prompt pay discounts, wholesaler discounts, or other discounts. We understand that certain discounts may depend on whether an eligible patient receives the drug. That is true, for example, for drugs acquired through the 340B program. We are therefore asking for hospitals to separately list their acquisition costs for drug NDCs acquired through the 340B program and those drug NDCs acquired outside of the 340B program in order to ensure that all of the discounts are accurately captured and represent the hospital's acquisition costs.
We will follow all privacy laws. Only the Department of Health and Human Services (HHS) and relevant component agencies, their contractors and representatives, and the Executive Office of the President (EOP) will have access to these data unless release of such data to other parties is otherwise required by law. In accordance with section 1833(t)(14)(A)(iii)(I) of the Act, the agency plans to use the data collected to determine acquisition costs for SCODs, and drugs historically treated as SCODs. Following the collection of data and its analysis, the aggregate results of the survey will be considered when determining proposed payment rates. The proposed payment rates and calculations with aggregate data could appear in notice and comment rulemaking; however, the confidentiality of individual hospitals and proprietary information would be maintained to the extent permitted by law. To the extent that acquisition prices for certain SCODs, and drugs historically treated as SCODs, are deemed sensitive and/or confidential, we do not intend to make such prices available in an individually identifiable manner. Acquisition cost data collected through a survey is directed by sections 1833(t)(14)(A)(iii)(I) and 1833(t)(14)(D)(ii) of the Act. We are authorized to obtain the data to inform payment for drugs paid under the OPPS.
Finally, we note this data collection is separate from ASP data collection where manufacturers submit data to CMS regarding their average sales price.
Comment: One commenter stated that CMS does not need to collect data from all institutions when it could instead use a statistical sampling method and that CMS could avoid the entire survey process by using the 340B ceiling price as the proxy for hospital acquisition costs. Another commenter suggested CMS adopt one of two sampling approaches that would reduce survey burden while ensuring accurate results. Under the first suggested approach, CMS would survey a representative sample of drugs and use the survey data and appropriate statistical analysis to determine the hospital acquisition cost for each drug.
Under the second proposed approach, CMS would confine the survey to those drugs that account for the vast majority of OPPS spending. Under this approach, CMS would use the resulting data to set payments only for the surveyed drugs based on the average acquisition cost for 340B hospitals and for non-340B hospitals pursuant to section ( printed page 53761) 1833(t)(14)(A)(ii) of the Act. Because hospital acquisition cost data for the non-surveyed drugs would not be available to CMS, CMS would be required to continue to set payment for these non-surveyed drugs for all hospitals using its current average sales price methodology pursuant to section 1833(t)(14)(A)(iii)(II) of the Act.
Response: We appreciate commenters' suggested alternatives to conducting a survey of all hospitals paid under the OPPS. As mentioned previously in this section, drug acquisition cost and payment is dynamic, complex, and of great importance. No drug acquisition cost survey has been conducted for decades. Therefore, for this initial survey, we will start by surveying the universe to obtain a comprehensive data set. We intend to conduct surveys periodically, and could consider some of the sampling methodologies suggested by commenters for future surveys once we've established a baseline dataset.
Comment: One commenter referenced the following observation from the GAO's 2006 report: “Other options available to CMS for validating price data could include audits of manufacturers' price submissions or an examination of proprietary data the agency considers reliable for validation purposes.” The commenter requested that CMS make it explicit in the final rule as to whether they considered these alternatives and why CMS decided to propose a hospital survey versus those alternatives.
Response: We did consider these alternatives. We decided to proceed with conducting a survey because that is the rate-setting methodology prescribed by statute. We do not believe the statute envisions that the Secretary will rely on surveys from 2004 and 2005 to set payment rates for SCODs forever. As we have stated previously, we intend to conduct surveys periodically, and could consider some of these additional GAO suggestions for future data validation exercises and survey design once we've established a baseline dataset.
Comment: One commenter recommended that CMS refine its instructions on the calculation of the total units purchased and total net acquisition cost to account for certain situations that are not informative of the actual acquisition cost. The commenter indicated that they believed that the reporting of total units purchased should be net of units returned to the manufacturer and that such returns should also be removed from the total net acquisition costs. The commenter alleged that the proposed instructions do not address returned units, and the commenter recommended clarifying that returned units are removed from the total units purchased and from the total net acquisition cost when finalizing the survey. The commenter contended that “it would be particularly inappropriate to retain returned units in the total unit count but then apply the manufacturer's refund for those units to reduce the net acquisition cost for those drugs. Rather, the data for returned units should be removed from both data points.”
Response: We refer this reader to the Draft Survey Template,[117] where the draft instructions address this scenario. Specifically, the draft instructions stated for “Total Units Purchased—Non-340B” to exclude the following: “Exclude purchases intended only for inpatient use; Exclude purchases that were returned; Exclude purchases made under the 340B Program.”
Comment: Several commenters requested that in the final rule CMS provide transparency regarding survey methodology, intended use of data, and potential impacts on policymaking. The commenters stated that this approach will give interested parties additional insight into the survey and may also encourage broader participation by hospitals. One commenter encouraged CMS to ensure that any survey findings are applied in a manner that is transparent, methodologically sound and reflective of the real-world costs of acquiring and delivering oncology drugs.
Response: We agree with commenters about the importance of providing transparency regarding survey methodology, intended use of data, and potential impacts on policymaking in the final rule. With respect to our survey methodology, as we explained in the CY 2026 OPPS/ASC proposed rule, we will be conducting a survey of the acquisition costs for each separately payable drug acquired by all hospitals paid under the OPPS, including SCODs, and drugs and biologicals CMS historically treats as SCODs. With respect to the intended use of data and potential impacts on policy making, as we stated in the CY 2026 OPPS/ASC proposed rule, we intend for the survey to be completed in time for the survey results to be used to inform policymaking beginning with the CY 2027 OPPS/ASC proposed rule. Any policy that is informed by the survey data will be subject to notice and comment rulemaking. We also appreciate the commenter's suggestion to apply survey findings in a manner that is transparent, methodologically sound and reflective of the real-world costs of acquiring and delivering oncology drugs.
Comment: Several commenters requested that CMS make the survey findings public to provide transparency for policymakers, taxpayers, and providers across care settings. Other commenters requested that time be allowed for commentary by interested parties to review the survey data prior to implementation in CY 2027.
Response: As we indicated in section 10 of Supporting Statement A [118] to our Information Collection Request under the Paperwork Reduction Act (PRA), the proposed payment rates and calculations with aggregate data could appear in future notice and comment rulemaking; however, the confidentiality of individual hospitals and proprietary information would be maintained to the extent permitted by law.
Comment: One commenter urged CMS to revise the proposed survey design to capture other hospital characteristics that might influence drug acquisition costs, rather than the singular focus on the difference between 340B and non-340B acquired drug costs.
Response: We thank the commenter for their suggestion, and we intend to consider many different hospital characteristics when analyzing the results of the data, such as hospital size, location, urban vs rural status, teaching hospital status, etc. These are all hospital characteristics that we routinely consider and analyze for various policies under the OPPS. As such, these hospital characteristics are already known; therefore, to reduce burden on hospitals submitting data, we are not asking them to submit information we already have.
Comment: Another commenter suggested that to improve transparency, hospitals should be required to submit claims data that identifies when 340B-purchased drugs are administered to Medicare patients. A commenter suggested that CMS require either a 340B or non-340B modifier to be appended to a claim in order for it to be considered complete and eligible for payment. They recommended that CMS require this in both the OPPS and PFS and that CMS create a clearinghouse to validate 340B units.
Response: We note that based on the CY 2024 OPPS/ASC final rule with comment period, we require that all ( printed page 53762) 340B covered entity hospitals paid under the OPPS report the “TB” modifier effective January 1, 2025, for 340B-acquired drugs and biologicals (88 FR 81791 through 81792). We may consider modifications to this policy in future rulemaking.
Comment: One commenter suggested that CMS (1) use a stratified approach with separate, volume-weighted averages for different types of hospitals when analyzing cost data to ensure payment adjustments reflect actual cost differences and do not rely on broad averages; (2) consider implementing blended or graduate payment adjustments rather than applying uniform reductions across all hospitals; and (3) establish mechanisms to monitor how payment changes affect patient access and hospital financial stability, particularly for those serving vulnerable populations.
Response: We thank the commenter for their suggestions. We'll take this feedback into consideration when analyzing and implementing future payment policy informed by the results of the survey.
Comment: One commenter suggested that CMS should further consider and address the methodology recommendations and challenges identified by the Comptroller General. The commenter maintains that while CMS claims to have “reviewed and taken into account the Comptroller General's recommendations regarding the frequency and methodology of these surveys in developing [its] proposed survey,” “[s]uch consideration is not reflected in the proposed OPPS rule.” The commenter stated that when GAO conducted the survey it did not require 340B costs to be reported separately. According to the commenter, GAO identified several relevant characteristics that impacted acquisition costs, and 340B status was not included in that list. The commenter opined that “[a] large number of factors outside of covered entity status implicate drug acquisition costs; it's unreasonable to base payments on 340B status alone”. The commenter also contended that CMS fails to address a critical flaw in the GAO survey that would undermine the accuracy of results and misleadingly inflate the difference in relative acquisition cost between 340B and non-340B hospitals. The commenter stated that CMS proposes that hospitals would be required to report the total acquisition cost of each drug, net of rebates and discounts but that in its report GAO stated “we found that we could not obtain data that would permit calculation of hospitals' acquisition costs, because, in general, hospitals were unable to report accurately or comprehensively on rebates”. The commenter stated that CMS does not acknowledge this issue or address how it proposes to assist hospitals in more accurately reporting on rebates. To the extent non-340B hospitals are more likely to receive discounts through rebates, the survey results will reflect higher than actual acquisition costs for this group as well as an overestimate of the difference from 340B hospital acquisition costs. A survey that does not address this flaw should be questioned by policymakers as a basis for rate setting. The GAO also recommended that the survey be conducted only once or twice per decade and be used only to validate “ASP data that manufacturers report to CMS for developing SCOD rates”. The commenter remarked it appears that CMS' intention is to conduct the survey periodically and use the hospital acquisition cost data on its own to set payment rates rather than use the survey to validate ASP data that is already used for payments. One commenter indicated that the rebates component of the survey would be challenging to report, ensuring only rebates that were actually paid are considered.
Response: We disagree with the commenter's contention that we have failed to review or take into account the Comptroller General's recommendations regarding the frequency and methodology of these surveys in developing our proposed survey. With respect to methodology, we agree with the commenter that factors other than 340B covered entity status can affect drug acquisition costs and our intent in conducting the survey is to collect drug acquisition cost data which reflects those other factors. We disagree with the commenter's allegation that we are proposing to base future drug payments on 340B status alone. As we have said throughout this rule, the results of the survey will be used to inform future payment policy. We are not prejudging the results of the survey or what we may do with the results of the survey. We are asking hospitals to incorporate all rebates and discounts in their acquisition cost for each NDC—including discounts directly applicable to an individual NDC—but also those discounts that are not necessarily linked to a single NDC. That discount could be one linked to a certain invoice, or discounts linked to purchases made over a certain time period such as prompt pay discounts, wholesaler discounts, or other discounts. With respect to discounts not linked to an individual NDC, we expect hospitals to report these discounts to us as part of this survey. Hospitals are most familiar with their acquisition costs and business practices and are best positioned to determine the method of incorporating discounts received into their NDC specific acquisition costs. However, if hospitals are unable to reasonably incorporate these non-NDC based discounts, rebates, and price concessions, such as those received through a GPO, we are asking hospitals to (1) indicate whether they have such discounts for the reporting time period and, if so, (2) identify what Group Purchasing Organization (GPO) they are a member of and (3) provide the total dollar amount of all non-NDC based discounts for the reporting time period, which will then allow CMS to best determine how to incorporate these non-NDC based discounts We acknowledge that the GAO was unable to obtain data that would permit calculation of hospitals' acquisition costs because, in general, hospitals were unable to report accurately or comprehensively on rebates at that time. However, we believe all of these discounts should be readily available in hospitals' purchasing systems, and with technology advancing considerably in the 20 plus years since the GAO survey we believe it is reasonable for hospitals to determine these types of discounts. Similarly to how there have been significant advances in technology over the past 20 years, there has also been an explosion in 340B drug utilization and spending. When the GAO conducted this survey this type of discount may not have been as prevalent; however, now that the prevalence has increased over the years we believe 340B discounts are required to be tracked similar to other discounts, rebates, and price concessions. With respect to frequency, our proposal to conduct the survey every 4 years is consistent with the GAO's recommendation that the Secretary validate, “on an occasional basis—possibly every 5 or 10 years—average sales price (ASP) data that manufacturers report to CMS for developing SCOD payment rates.” [119] Finally, with respect to the commenter's allegation that we might use the hospital acquisition cost data on its own to set payment rates rather than use the survey to validate ASP data that is already used for payments, we emphasize that were we to use survey data to set payment rates in future rulemaking, doing so would be consistent with section 1833(t)(14)(D)(ii) of the Act which requires “the Secretary to conduct periodic . . . surveys to determine the ( printed page 53763) hospital acquisition cost for each specified covered outpatient drug for use in setting the payment rates.”
Comment: Many commenters opposed our intent to propose and seek comment on any payment rates for SCODs based on the survey results in CY 2027 rulemaking. These commenters expressed concern about future payment reductions based on the results of the survey and urged CMS to abandon the survey to avoid its results being used to reduce Medicare reimbursements in 2027 and beyond. In these commenters' view, Medicare payment is already too low and further reductions resulting from the survey would be unsustainable. Many commenters expressed concern that future payment reductions would come at the expense of services provided to patients.
Response: We thank the commenters for their input. It is premature to consider the payment consequences of a survey we have not yet completed, and commenters' policy concerns of those payment consequences are therefore out of scope for purposes of this final rule with comment period regarding CY 2026.
Comment: Many commenters expressed concern about the disproportionate effect future payment reductions resulting from the survey would have on safety-net hospitals and rural providers. Some of these commenters asked CMS to exempt all rural hospitals, including SCHs, MDHs, Low-Volume Hospitals (LVHs), Rural Emergency Hospitals (REHs) and small rural hospitals with 100 beds or less from any such reductions. One commenter requested an exemption from any payment reductions resulting from the survey for the three freestanding cancer centers that currently participate in the 340B program.
Response: We thank the commenters for their input. It is premature to consider the payment consequences of a survey we have not yet completed, and commenters' policy concerns of those payment consequences are therefore out of scope for purposes of this final rule with comment period regarding payment amounts for CY 2026.
Comment: One commenter stated that CMS should not use the proposed survey to set OPPS rates because the CY 2026 OPPS/ASC proposed rule discusses use of the survey results in future rate-setting in ways that could be inconsistent with the OPPS statute. The commenter states that while CMS acknowledges the statutory requirement to conduct a survey of acquisition costs on a periodic basis, CMS does not clarify how it would adjust payments outside of survey periods. By contrast, current policy updates payments quarterly based on Medicare's ASP, ensuring payment rates reflect current market drug costs. The CY 2026 OPPS/ASC proposed rule includes no such similar adjustment for routine adjustments to costs. The commenter states that section 1833(t)(14)(A)(iii)(I) of the Act requires that the payment for a specified covered outpatient drug should be equal to the average acquisition cost for the drug for that year. Given the significant changes being implemented by and further proposed by this Administration—such as changes resulting from proposed Pharmacy Benefit Manager (PBM) reform, Medicare fair price negotiations, and international price matching, as noted above—a survey fielded at the start of CY 2026 is unlikely to reflect the acquisition cost even in CY2027, never mind future years.
Response: We thank the commenters for their input. It is premature to consider the payment consequences of a survey we have not yet completed, and commenters' policy concerns of those payment consequences are therefore out of scope for purposes of this final rule with comment period regarding payment amounts for CY 2026.
Comment: Several commenters stated that their experience from the 2018-2022 period demonstrated that reducing 340B payment rates set a dangerous precedent as private pharmacy benefit managers and third-party payers could use such a reduction to justify additional reductions for 340B drugs, further weakening the healthcare safety net.
Response: We thank the commenter for their input. It is premature to consider the payment consequences of a survey we have not yet completed, and commenters' policy concerns of those payment consequences is therefore out of scope for purposes of this final rule with comment period regarding payment amounts for CY 2026.
Comment: Several commenters requested that CMS provide a clear methodology for data use and protections against retroactive payment adjustments based on survey data.
Response: We are not sure what these commenters are requesting. It is premature to speculate on how a survey we have not yet completed might best be used to set future payment policies, and so commenters' concerns about hypothetical uses of the survey is therefore out of scope for purposes of this final rule with comment period regarding payment amounts set without reference to that survey.
Comment: One commenter stated that using the results of the survey as the sole basis to set drug reimbursement rates contravenes the guidance of the U.S. Comptroller General and the GAO as stated in the GAO's 2006 report to Congress: “A key lesson for CMS that we learned from conducting the 2004 MMA-mandated hospital survey is that such a survey would not be practical for collecting the data needed to set and update SCOD rates routinely. However, it would be useful, on occasion, for CMS to survey hospitals so that the rate-setting data it obtained from other sources could be validated by an independent source.” The commenter contended that, despite refencing this report in the proposed rule, CMS provides no substantive explanation for disregarding the recommendation and appears poised to use the survey not as a validation mechanism but as the foundation for setting payment rates. The commenter alleged that CMS' failure to address or justify its departure from this key lesson raises serious questions about the methodological soundness of the proposed survey and the legality of this rulemaking exercise. This will ultimately lead to a circumvention of Congressional intent in implementing the 340B Program, which is indisputably intended to support safety net providers—not to reallocate savings intended for them to all Medicare OPPS providers. The commenter stated that other mechanisms, such as surveying wholesaler distributors or using drug manufacturer data based on chargeback and other data commonly used by these entities, should first be used to collect baseline data which should then be validated.
Response: As we note, we anticipate that the results of the survey will be used to inform future payment policy. We also note that we may not rely solely on survey data to inform our drug pricing policies. Any policy to alter OPPS payment rates for drugs informed by this survey would be included in the CY 2027 OPPS/ASC rulemaking, and would be subject to notice and comment rulemaking.
Comment: Several commenters stated that the Congress has not directed the executive branch to reduce payments to 340B providers and did not direct CMS to introduce new policies that seek to reduce the Federal government's commitment to serving low-income Americans.
Response: We thank the commenters for their input. We have not proposed reductions to 340B providers in this final rule with comment period, and so these comments are out of scope. ( printed page 53764)
Comment: Many commenters implored CMS not to use the survey to lower payments to 340B hospitals. One commenter acknowledged that the CY 2026 OPPS/ASC proposed rule does not explicitly state CMS' intent to use the survey to lower payments to 340B-acquired drugs only but alleges that “the proposed structure of the survey foreshadows CMS' future actions.” The commenter stated that a reduction in Medicare payment rates to 340B hospitals significantly erodes the intent of the 340B program and that the 340B program is critical to ensuring that low-income and other disadvantaged people have access to the types of services best provided by essential hospitals. The commenter alleges that hospitals participating in the 340B program operate on margins significantly narrower than margins of other hospitals, with many operating at a loss. These hospitals, the commenter contends, which serve a high number of low-income individuals, are already struggling under insufficient Medicare and Medicaid payments. Given the fragile financial position of essential hospitals, policy changes that jeopardize any piece of the patchwork of support on which they rely, including the 340B program, can threaten their ability to maintain critical services. Reducing Medicare payments for 340B hospitals would have many negative consequences for patients and providers and would not save the Medicare program any money. Any changes to OPPS must be made in a budget-neutral manner. Thus, a cut in funding for 340B hospitals does not go back to the Medicare program or directly to beneficiaries; instead, the funds would be redistributed to non-340B hospitals at the expense of 340B hospitals and their patients.
Response: We thank the commenters for their input. We have not proposed reductions to 340B providers in this final rule with comment period, and so these comments, including speculation about legislative intent and policies that could be enacted by future legislation are out of scope for purposes of this CY 2026 OPPS/ASC final rule with comment period.
Comment: Some commenters supported our intent to propose and seek comment on payment rates for SCODs based on survey results in CY 2027 rulemaking. One commenter encouraged CMS to ensure a transparent process in analyzing information obtained from the survey, to the extent possible, in advance of CY 2027 proposed rulemaking.
Response: We appreciate commenters' support.
We noted in the CY 2026 OPPS/ASC proposed rule that under section 1833(t)(14)(D)(iii) of the Act, the surveys must have a large sample of hospitals that is sufficient to generate a statistically significant estimate of the average hospital acquisition cost for each specified covered outpatient drug. Consequently, we stated that we would seek an adequate response rate to the survey and that surveyed hospitals had an obligation to respond to the survey. We indicated that hospitals had ample notice in the CY 2026 OPPS/ASC proposed rule regarding the intent and the details of the OPPS Drug Acquisition Cost Survey so we expected all hospitals would submit their acquisition costs in a timely manner to CMS. We stated that we understood that hospitals have significant drug acquisition costs, and so, consistent with the Comptroller's General experience conducting earlier drug acquisition cost surveys in which 83 percent of the hospitals surveyed provided usable data,[120] we anticipated hospitals would want to respond to the survey to demonstrate to CMS these costs. We requested comment from readers on whether we should make responding to the survey a mandatory requirement of all hospitals paid under the OPPS through 1833(t)(14)(D)(iii) of the Act.
Comment: Many commenters stated that CMS lacks the statutory authority to make the survey mandatory and requested that CMS state in the final rule that the survey is voluntary. These commenters stated that section 1833(t)(14)(D)(iii) simply sets forth the requirements for a survey and does not provide the agency with the authority to mandate hospital responses. These commenters contended that if the Congress wanted to require hospital participation in a drug acquisition cost survey or allow HHS to take action for a non-response, it would have done so, as it has in other contexts. One commenter, referencing the survey previously conducted by GAO, suggested that the level of response to that survey demonstrates that making completion of the survey mandatory is not necessary to ensure sufficient responses. The commenter stated that when GAO previously conducted the survey it provided no incentives or penalties to encourage participation. Even without any incentives in place, GAO received usable data from 83 percent of the hospitals.
Other commenters supported making the survey mandatory, arguing that doing so was critical to ensuring that the wide range of hospital acquisition costs is appropriately accounted for and reflected in the survey results and in CY 2027 OPPS rates. Several commenters stated that making the survey mandatory is the only way for CMS to ensure it collects complete and usable data.
Response: Under section 1833(t)(14) (D) of the Act, the Secretary “shall conduct periodic surveys” which “shall have a large sample of hospitals that is sufficient to generate a statistically significant estimate of the average hospital acquisition cost” and which the Secretary shall under section 1833(t)(140(A) of the Act “tak[e] into account” when determining the average acquisition cost of drugs for a year to set payment policy. Hospitals attempt to read a conditional into the text of the statute: the Secretary may use cost acquisition survey data to set policy “only if” that data generates a statistically significant estimate of the average hospital acquisition. But that is not what the statute says. Rather, it provides that the survey sample “shall” generate such an estimate. We read that instruction from the Congress to impose obligations on both the Secretary to design such a survey and on hospitals generally to respond the survey, just as they did when GAO conducted the survey in 2004 and 2005. We agree that section 1833(t)(14)(D) of the Act does not itself mandate specific consequences either on CMS for failing to design the survey it describes or on hospitals for failing to respond to that survey. We reiterate the reciprocal obligations imposed by section 1833(t)(14)(D) of the Act on CMS and hospitals alike, however, in light of certain comments suggesting that some hospitals might be planning to coordinate non-responses. The lack of a response to this required survey is still meaningful data to CMS which can be taken into consideration to inform future payment rates in future rulemaking.
In the CY 2026 OPPS/ASC proposed rule, we welcomed comment on how we might propose to interpret non-responses to the survey. For example, since a failure on the part of a hospital to respond to the survey could suggest that the hospital has minimal acquisition costs, or has lower acquisition costs than an otherwise similar hospital that responds to the survey and so is withholding its response strategically, we stated in the CY 2026 OPPS/ASC proposed rule that we might, if the data so suggested, determine that groups of hospitals who do not respond to the survey have lower ( printed page 53765) acquisition costs for SCODs than their otherwise similar counterparts under section 1833(t)(14)(A)(iii)(I) of the Act. In such instances, we stated that we would consider various appropriate ways, taking into account the hospital acquisition cost survey data, to determine the average acquisition cost. One method we said we might consider, depending on the cost survey data, could be to use the lowest acquisition cost reported among otherwise similar responding hospitals as a proxy for the average acquisition costs for hospitals that do not respond to the survey. We also stated that we might also consider supplemental data sources to inform our determination of average acquisition costs for hospitals for whom we lacked cost acquisition survey data. For example, we said we might consider using, as available, pricing from the FSS; [121] 340B ceiling price; [122] ASP plus 6 percent, zero percent or another percentage; or other recognized drug pricing for payment of hospitals that do not respond to the survey.
We stated that we could also consider a hospital's non-response to the survey when determining how to package drug costs for particular hospital groups. We noted that under section 1833(t)(2)(B) of the Act, the OPPS establishes groups of covered HOPD services, namely APC groups, and uses them as the basic unit of payment. In the case of much of the care paid under the OPPS, we indicated that we viewed a complete service as potentially being reported by a combination of two or more HCPCS codes, rather than a single code, and establish payment policies that support this view. We stated that ideally we would consider a complete HOPD service to be the totality of care furnished in a hospital outpatient encounter or in an episode of care. We noted that we generally package payment for items and services that are typically ancillary and supportive into the payment for the primary diagnostic or therapeutic modalities in which they are used rather than pay for them separately. We indicated that as we noted previously in the CY 2026 OPPS/ASC proposed rule, if a hospital does not submit its acquisition cost data, it could suggest that the hospital has minimal acquisition costs. We also noted that if the hospital has minimal acquisition costs for drugs, that could support viewing those costs as ancillary or supportive, and we might, if the data supported it, conclude that hospitals who do not report their drug acquisition costs lack meaningful additional, marginal costs related to their acquisition of these drugs and, as such, their drugs costs should not be paid separately but rather should be packaged into the payment for the associated service.
We sought comment broadly on how to approach payment to hospitals for drugs usually paid under the OPPS absent a hospital's response to the survey.
We received many comments in response to our request for comment.
Comment: Most commenters opposed CMS modifying its payment approach based on non-responses to the survey. Many of these commenters stated that CMS lacks the statutory authority to do so. One commenter stated that the statute does not contemplate CMS “filling in gaps from non-responses”. In the commenter's view, the statute instructs the agency to collect data from “a large sample of hospitals that is sufficient to generate a statistically significant estimate of the average hospital acquisition cost for each specified covered drug”. The commenter stated that CMS cannot “manipulate an otherwise insufficient sample by using other hospitals' proxy data for hospitals that do not respond”. The commenter also stated that the methodology CMS posits to interpret non-responses lacks reasoning. The commenter alleged that CMS points to no evidence for any conclusion that groups of hospitals that do not respond to the survey have lower acquisition costs, and certainly no justification for why “us[ing] the lowest acquisition cost reported among otherwise similar responding hospitals as a proxy” would be a remotely accurate methodology. The commenter concludes that the Congress, in section 1833(t)(14)(A)(iii) of the Act, dictates drug reimbursement be based on either hospital acquisition cost data, or the average price of the drug. The commenter contends that substituting data such as “pricing from the FSS; 340B ceiling price; ASP plus 6 percent, zero percent or another percentage; or other recognized drug pricing for payment of hospitals” would contravene explicit congressional intent.
Another commenter stated that the statute authorizes CMS to conduct surveys to inform payment rates for specified covered outpatient drugs, but it does not authorize CMS to “penalize” hospitals by reducing reimbursement or applying alternative payment methodologies based solely on survey non-participation.” The commenter claimed that CMS' suggestion that it may pay non-responding hospitals at rates below ASP plus 6 percent or exclude them from future rate-setting processes is not supported by the statutory text and represents an overreach of administrative authority. The commenter explained that hospitals may have legitimate operational, legal, or resource-based reasons for being unable to complete the survey within the prescribed timeframe and that “imposing financial penalties in response to non-compliance with a voluntary or burdensome survey process, particularly one that lacks finalized rulemaking and OMB approval, would be both unlawful and inequitable.” The commenter urged CMS to clarify that survey participation would not be used as a basis for punitive reimbursement decisions and to ensure that any future payment methodologies are grounded in statutory authority and fair process. Many other commenters also claimed that CMS' imputing data to a non-response would unfairly penalize hospitals for simply lacking the resources to complete the survey. These commenters pointed out that a non-response is not necessarily indicative of lower acquisition costs as it could reflect data collection challenges or unclear instructions rather than lower costs. One commenter alleged that CMS does not cite any evidence that non-response means minimal acquisition costs. The commenter stated that both GAO and CMS acknowledge the burden of this survey, which could clearly be a reason for non-response. In the commenter's view, interpreting non-responses is an attempt to make the survey effectively mandatory, even though the commenter does not believe CMS has the authority to make the survey mandatory. The commenter contended that CMS is aware that the drugs at issue can be prohibitively expensive, particularly for hospitals serving disproportionate shares of low income individuals, and that if CMS does not pay adequately for hospitals' drug costs it will not only undermine the financial stability of hospitals but will impact Medicare patients' access to critical medications. Moreover, the OPPS statute requires CMS to pay for these drugs either based on the survey or based on ASP; CMS does not have the authority to eliminate payment for these drugs.
Many commenters stated that CMS' imputing data to a non-response would result in a survey that would fail to satisfy the requirement under section 1833(t)(14)(D)(iii) of the Act that the ( printed page 53766) survey “. . . have a large sample of hospitals that is sufficient to generate a statistically significant estimate of the average hospital acquisition cost for each specified covered outpatient drug”. Several commenters stated that absent a statistically significant number of responses, CMS cannot implement changes to reimbursement rates that vary by groups of hospitals for separately payable drugs under the OPPS.
One commenter noted that section 1833(t)(14)(D)(i)(II) of the Act requires the Secretary to take into account recommendations from the Comptroller General regarding the methodology of the survey and that when the GAO conducted its survey, it explicitly excluded non-responses from its price calculations stating it was “not appropriate for [its] purpose.” Several commenters requested that non-responding hospitals be excluded from the survey dataset.
One commenter supported using the FSS and other information as a mechanism of determining hospital costs for drugs. Another expressed support for the proposal to use the lowest reported acquisition cost as a proxy for non-responding hospitals.
Several commenters suggested that in instances where hospital pricing information is not sufficiently provided, CMS should (1) for non-responsive 340B hospitals, assume the price of the drug to be the 340B ceiling price and (2) for non-responsive non-340B hospitals, assume the price of the drug to be the average acquisition cost from a similarly situated non-340B hospital (similar size, location, etc.) that responded to the survey. Other commenters suggested that for non-responding non-340B covered entities, CMS should assume the lesser of the median cost or mean cost of similarly situated non-340B hospitals.
Several commenters recommended that just as any estimations used to address non-responses must consider 340B status, any findings derived from this survey should be stratified by whether the reporting hospital participates in the 340B Program.
Response: We thank commenters for their feedback regarding what we might do in the scenario in which we do not receive responses from all hospitals paid under the OPPS. Under section 1833(t)(14)(A)(iii)(I) of the Act, the Secretary may set average drug prices—which may vary by hospital group—by “taking into account” the results of survey. We have made no final decision on how, if at all, we will “tak[e] into account” non-responses, which is premature before we analyze the data we receive. Any policies based on this discussion will be included in future rulemaking, as soon as CY 2027, if we adopt payments rates based on the results of the survey.
After consideration of public comments, we are finalizing our proposal outlining our intent to conduct a required OPPS drug acquisition cost survey to all hospitals paid under the OPPS, pending final approval from OMB.
Consistent with E.O. 14273 and PRA requirements, additional details of this survey can be found in section XXIII. of this final rule with comment period as well as the Information Collection Request that can be found on reginfo.gov. This new information collection request will be submitted to OMB for review under control number 0938-1487 (CMS-10931). The OMB control number will not be valid until formally approved by OMB.
VI. Estimate of OPPS Transitional Pass-Through Spending for Drugs, Biologicals, Radiopharmaceuticals, and Devices
A. Amount of Additional Payment and Limit on Aggregate Annual Adjustment
Section 1833(t)(6)(E) of the Act limits the total projected amount of transitional pass-through payment for drugs, biologicals, and categories of devices for a given year to an “applicable percentage,” currently not to exceed 2.0 percent of total program payments estimated to be made for all covered services under the OPPS furnished for that year. If we estimate before the beginning of the calendar year that the total amount of pass-through payments in that year would exceed the applicable percentage, section 1833(t)(6)(E)(iii) of the Act requires a uniform prospective reduction in the amount of each of the transitional pass-through payments made in that year to ensure that the limit is not exceeded. We estimate the pass-through spending to determine whether payments exceed the applicable percentage and the appropriate pro rata reduction to the conversion factor for the projected level of pass-through spending in the following year to ensure that total estimated pass-through spending for the prospective payment year is budget neutral, as required by section 1833(t)(6)(E) of the Act.
For devices, developing a proposed estimate of pass-through spending in CY 2026 entails estimating spending for two groups of items. The first group of items consists of device categories that are currently eligible for pass-through payment and that will continue to be eligible for pass-through payment in CY 2026. The CY 2008 OPPS/ASC final rule with comment period (72 FR 66778) describes the methodology we have used in previous years to develop the pass-through spending estimate for known device categories continuing into the applicable update year. The second group of items consists of devices that we know are newly eligible, or project may be newly eligible, for device pass-through payment in the remaining quarters of CY 2025 or beginning in CY 2026. The sum of the proposed CY 2026 pass-through spending estimates for these two groups of device categories equals the proposed total CY 2026 pass-through spending estimate for device categories with pass-through payment status. We determined the device pass-through estimated payments for each device category based on the amount of payment as required by section 1833(t)(6)(D)(ii) of the Act, and as outlined in previous rules, including the CY 2025 OPPS/ASC final rule with comment period (89 FR 94259 through 94261). We note that, beginning in CY 2010, the pass-through evaluation process and pass-through payment methodology for implantable biologicals newly approved for pass-through payment beginning on or after January 1, 2010, that are surgically inserted or implanted (through a surgical incision or a natural orifice) use the device pass-through process and payment methodology (74 FR 60476). As has been our past practice (76 FR 74335), we include an estimate of any implantable biologicals eligible for pass-through payment in our estimate of pass-through spending for devices. Similarly, we finalized a policy in CY 2015 that applications for pass-through payment for skin substitutes and similar products be evaluated using the medical device pass-through process and payment methodology (76 FR 66885 through 66888). Therefore, as we did beginning in CY 2015, for CY 2026, we also proposed to include an estimate of any skin substitutes and similar products in our estimate of pass-through spending for devices. However, in accordance with the finalized policy in this final rule with comment period to align payment policy for skin substitutes with FDA's regulatory categories, we have separated the pass-through spending estimates into the appropriate pathway, device or drug pass-through. We note that since there are no current skin substitutes eligible for pass-through payments, this estimate for both pathways is zero. ( printed page 53767)
For drugs and biologicals eligible for pass-through payment, section 1833(t)(6)(D)(i) of the Act establishes the pass-through payment amount as the amount by which the amount authorized under section 1842(o) of the Act (or, if the drug or biological is covered under a competitive acquisition contract under section 1847B of the Act, an amount determined by the Secretary equal to the average price for the drug or biological for all competitive acquisition areas and year established under such section as calculated and adjusted by the Secretary) exceeds the portion of the otherwise applicable fee schedule amount that the Secretary determines is associated with the drug or biological. Consistent with current policy, we proposed to apply a rate of ASP plus 6 percent to most drugs and biologicals for CY 2026, and therefore our estimate of drug and biological pass-through payment for CY 2026 for this group of items was $15.2 million.
Payment for certain drugs,[123] specifically contrast agents without pass-through payment status, is packaged into payment for the associated procedures, and these products are not separately paid. In addition, we policy-package non-pass-through drugs and biologicals that function as supplies when used in a diagnostic test or procedure unless a high-cost diagnostic radiopharmaceutical with a per-day cost greater than the finalized per-day threshold, finalized to be $655 for CY 2026 as outlined in section II.A.3.c. of this final rule with comment period, is used for the test or procedure. We policy-package all drugs and biologicals that function as supplies when used in a surgical procedure or for anesthesia, and other categories of drugs and biologicals, as described in section V.B.1.c. of the CY 2026 OPPS/ASC proposed rule (90 FR 33625 through 33626). Consistent with current policy, for CY 2026, we proposed that policy-packaged drugs and biologicals with pass-through payment status will be paid at ASP+6 percent, like other pass-through drugs and biologicals less the policy-packaged drug APC offset amount described below. Our estimate of pass-through payment for policy-packaged drugs and biologicals with pass-through payment status approved prior to CY 2026 is not $0. This is because the pass-through payment amount and the fee schedule amount associated with the drug or biological will not be the same, unlike for separately payable drugs and biologicals. In the CY 2024 OPPS/ASC final rule with comment period (88 FR 81774 through 81776), we discussed our policy to determine if the costs of certain policy-packaged drugs or biologicals are already packaged into the existing APC structure. If we determine that a policy-packaged drug or biological approved for pass-through payment resembles predecessor drugs or biologicals already included in the costs of the APCs that are associated with the drug receiving pass-through payment, we offset the amount of pass-through payment for the policy-packaged drug or biological. For these drugs or biologicals, the APC offset amount is the portion of the APC payment for the specific procedure performed with the pass-through drug or biological, which we refer to as the policy-packaged drug APC offset amount. Consistent with current policy described in section V.A.5. of the CY 2026 OPPS/ASC proposed rule (90 FR 33619 to 33620), if we determine that an offset is appropriate for a specific policy-packaged drug or biological receiving pass-through payment, we proposed to reduce our estimate of pass-through payments for these drugs or biologicals by the APC offset amount.
Similar to pass-through spending estimates for devices, the first group of drugs and biologicals requiring a pass-through payment estimate consists of those products that were recently made eligible for pass-through payment and that will continue to be eligible for pass-through payment in CY 2026. The second group contains drugs and biologicals that we know are newly eligible, or project will be newly eligible, in CY 2026. The sum of the CY 2026 pass-through spending estimates for these two groups of drugs and biologicals equals the total CY 2026 pass-through spending estimate for drugs and biologicals with pass-through payment status.
We did not receive public comments on this provision, and therefore, we are finalizing as proposed.
B. Final Estimate of Pass-Through Spending for CY 2026
For CY 2026, we proposed to set the applicable pass-through payment percentage limit at 2.0 percent of the total projected OPPS payments for CY 2026, consistent with section 1833(t)(6)(E)(ii)(II) of the Act and our OPPS policy from CY 2004 through CY 2025 (89 FR 94260). The pass-through payment percentage limit is calculated using pass-through spending estimates for devices and for drugs and biologicals.
For the first group of devices, consisting of device categories that are currently eligible for pass-through payment and will continue to be eligible for pass-through payment in CY 2026, there are 14 active categories for CY 2026. The active categories are described by HCPCS codes C1600, C1601, C1602, C1603, C1604, C1605, C1606, C8000, C1735, C1736, C1737, C1738, C1739, and C9610. Based on CY 2024 Medicare hospital outpatient claims data available by the time of the proposed rule and information from the device manufacturers provided in their respective pass-through applications regarding the device cost and the projected CY 2026 OPPS utilization, we estimated that HCPCS code C1600 would cost $0.3 million in pass-through expenditures in CY 2026, HCPCS code C1601 would cost $5.0 million in pass-through expenditures in CY 2026, HCPCS code C1602 would cost $0.2 million in pass-through expenditures in CY 2026, HCPCS code C1603 would cost $0.1 million in pass-through expenditures in CY 2026, HCPCS code C1604 would cost $2.0 million in pass-through expenditures in CY 2026, HCPCS code C1605 would cost $113.0 million in pass-through expenditures in CY 2026, HCPCS code C1606 would cost $0.3 million in pass-through expenditures in CY 2026, HCPCS code C8000 would cost $2.9 million in pass-through expenditures in CY 2026, HCPCS code C1735 would cost $16.0 million in pass-through expenditures in CY 2026, HCPCS code C1736 would cost $32.8 million in pass-through expenditures in CY 2026, HCPCS code C1737 would cost $34.1 million in pass-through expenditures in CY 2026, HCPCS code C1738 would cost $0.8 million in pass-through expenditures in CY 2026, HCPCS code C1739 would cost $8.5 million in pass-through expenditures in CY 2026, and HCPCS code C9610 would cost $36.0 million in pass-through expenditures in CY 2026. Therefore, we proposed an estimate for the first group of devices of $252.0 million.
We did not receive any public comments on our proposed estimate. Based on updated CY 2024 Medicare hospital outpatient claims data available for this final rule with comment period and information from the device manufacturers provided in their respective pass-through applications regarding the device cost and the projected CY 2026 OPPS utilization, our final estimated pass through costs are as follows: HCPCS code C1600 will cost ( printed page 53768) $0.3 million in pass-through expenditures in CY 2026, HCPCS code C1601 will cost $4.7 million in pass-through expenditures in CY 2026, HCPCS code C1602 will cost $0.3 million in pass-through expenditures in CY 2026, HCPCS code C1603 will cost $0.1 million in pass-through expenditures in CY 2026, HCPCS code C1604 will cost $2.1 million in pass-through expenditures in CY 2026, HCPCS code C1605 will cost $113.0 million in pass-through expenditures in CY 2026, HCPCS code C1606 will cost $0.3 million in pass-through expenditures in CY 2026, HCPCS code C8000 will cost $2.9 million in pass-through expenditures in CY 2026, HCPCS code C1735 will cost $16.0 million in pass-through expenditures in CY 2026, HCPCS code C1736 will cost $32.8 million in pass-through expenditures in CY 2026, HCPCS code C1737 will cost $34.1 million in pass-through expenditures in CY 2026, HCPCS code C1738 will cost $0.8 million in pass-through expenditures in CY 2026, HCPCS code C1739 will cost $8.5 million in pass-through expenditures in CY 2026, and HCPCS code C9610 will cost $36.0 million in pass-through expenditures in CY 2026.[124] Therefore, we have finalized the CY 2026 spending estimate for the first group of devices of approximately $251.9 million.
In estimating our proposed CY 2026 pass-through spending for device categories in the second group, we included the following: (1) device categories that we assumed at the time of the development of the CY 2026 OPPS/ASC proposed rule would be newly eligible for pass-through payment in CY 2026; (2) additional device categories that we estimated could be approved for pass-through status after the development of the CY 2026 OPPS/ASC proposed rule and before January 1, 2026; and (3) contingent projections for new device categories established in the second through fourth quarters of CY 2026. For CY 2026, we proposed to use the general methodology described in the CY 2008 OPPS/ASC final rule with comment period (72 FR 66778), while also taking into account recent OPPS experience in approving new pass-through device categories. For the CY 2026 OPPS/ASC proposed rule (90 FR 33656), the proposed estimate of CY 2026 pass-through spending for this second group of device categories was $319.8 million.
We did not receive any public comments on this proposed estimate. As stated earlier in section IV.A.2. of this final rule with comment period, we are approving two devices for pass-through payment status in the CY 2026 rulemaking cycle: VasQ and the SCOUT MDTM Surgical Guidance System.[125] In addition, we note that HCPCS codes C1740, C1741 and C1742 were preliminarily approved as part of the device pass-through quarterly review process with an effective date of October 1, 2025.[126 127] For this final rule with comment period based on information from the device manufacturers provided in their respective pass-through applications regarding the projected CY 2026 OPPS utilization, we estimate that HCPCS code C1740 will cost $31.7 million in pass-through expenditures in CY 2026, HCPCS code C1741 will cost $7.5 million in pass-through expenditures in CY 2026, and HCPCS code C1742 will cost $0.7 million in pass-through expenditures in CY 2026. Therefore, for the reasons explained more below, we are finalizing an estimate of $39.9 million for this second group of devices for CY 2026.
The estimated amount of pass-through spending of $39.9 million for the second group of devices in this final rule with comment period is substantially different from the estimated amount of pass-through spending in the CY 2026 OPPS/ASC proposed rule (90 FR 33656) of $319.8 million because, in the CY 2026 OPPS/ASC proposed rule, we consider that all of the device applications included in that CY 2026 OPPS/ASC proposed rule may receive pass-through status approval in the corresponding CY 2026 OPPS/ASC final rule with comment period, and as such, we include the estimated amount of pass-through spending for each device. For the final rule with comment period, the estimate reflects the estimated amount of pass-through spending for only the devices that were approved for pass-through status. In addition, we updated the estimates to include the estimated pass-through expenditures for any devices that are preliminarily approved as part of the device pass-through quarterly review process after the development of the current CY 2026 OPPS/ASC proposed rule.
To estimate proposed CY 2026 pass-through spending for drugs and biologicals in the first group, specifically those drugs and biologicals recently made eligible for pass-through payment and continuing on pass-through payment status for at least one quarter in CY 2026, we proposed to use the CY 2024 Medicare hospital outpatient claims data regarding their utilization, information provided in their respective pass-through applications, other historical hospital claims data, pharmaceutical industry information, and clinical information regarding these drugs and biologicals to project the CY 2026 OPPS utilization of the products.
For the known drugs and biologicals (excluding policy-packaged contrast agents, drugs, biologicals, radiopharmaceuticals with per-day costs at or below the packaging threshold that function as supplies when used in a diagnostic test or procedure, and drugs and biologicals that function as supplies when used in a surgical procedure) that will be continuing on pass-through payment status in CY 2026, we estimated the pass-through payment amount as the difference between the general payment rate of ASP+6 percent and the payment rate for non-pass- ( printed page 53769) through drugs and biologicals that would be separately paid. Because we proposed to utilize a payment rate of ASP+6 percent for most separately payable drugs and biologicals in the CY 2026 OPPS/ASC proposed rule, the proposed payment rate difference between the pass-through payment amount and the non-pass-through payment amount was $0 for this group of drugs.
Because payment for policy-packaged drugs and biologicals is packaged if the product is not paid separately due to its pass-through payment status, we proposed to include in the CY 2026 pass-through estimate the difference between payment for the policy-packaged drug or biological at ASP+6 percent (or WAC+6 percent, or 95 percent of AWP, if ASP or WAC information is not available) and the policy-packaged drug APC offset amount, if we determine that the policy-packaged drug or biological approved for pass-through payment resembles a predecessor drug or biological already included in the costs of the APCs that are associated with the drug receiving pass-through payment. Diagnostic radiopharmaceuticals that currently have pass-through status, but would likely be paid separately because of the policy initially established in the CY 2025 OPPS/ASC final rule with comment period (89 FR 93953) to separately pay for diagnostic radiopharmaceuticals with per-day costs greater than the per-day cost threshold and which we are continuing as discussed in section II.A.3.c. of this final rule with comment period, are not considered to be policy-packaged and therefore are not included in this group. For this first group of policy-packaged drugs and biologicals, we estimated a pass-through spending for CY 2026 of $5.2 million.
We did not receive any public comments on our proposed estimate. Using our methodology for this final rule with comment period, we are finalizing the CY 2026 spending estimate for this first group of drugs and biologicals to be $5.2 million.
To estimate proposed CY 2026 pass-through spending for drugs and biologicals in the second group (that is, drugs and biologicals that we knew at the time of development of the proposed rule were newly eligible or recently became eligible for pass-through payment in CY 2025, additional drugs and biologicals that we estimated could be approved for pass-through status subsequent to the development of the proposed rule and before January 1, 2026, and projections for new drugs and biologicals that could be initially eligible for pass-through payment in the second through fourth quarters of CY 2026), we proposed to use utilization estimates from pass-through applicants, pharmaceutical industry data, clinical information, recent trends in the per unit ASPs of hospital outpatient drugs, and projected annual changes in service volume and intensity as our basis for making the CY 2026 pass-through payment estimate. We also proposed to consider the most recent OPPS experience in approving new pass-through drugs and biologicals. Using our proposed methodology for estimating CY 2026 pass-through payments for this second group of drugs, we calculated a proposed spending estimate for this second group of drugs and biologicals of approximately $10 million.
We did not receive any public comments on our proposed estimate. Using our methodology for this final rule with comment period, we are finalizing our estimate of pass-through spending for the second group of drugs and biologicals to be $10 million.
We estimated for the CY 2026 OPPS/ASC proposed rule (90 FR 33656) that the amount of pass-through spending for the device categories and the drugs and biologicals that are continuing to receive pass-through payment in CY 2026 and the amount of pass-through spending for those device categories, drugs, and biologicals that first become eligible for pass-through payment during CY 2026 would be approximately $587.0 million (approximately $571.8 million for device categories and approximately $15.2 million for drugs and biologicals), which represents only 0.59 percent of total projected OPPS payments for CY 2026 (approximately $100 billion). Therefore, we estimated that pass-through spending in CY 2026 will not exceed the 2.0 percent of total projected OPPS CY 2026 program spending limit provided for in section 1833(t)(6)(E) of the Act.
We are finalizing our estimate for this final rule with comment period that the amount of pass-through spending for the device categories and the drugs and biologicals that are continuing to receive pass-through payment in CY 2026 and the amount of pass-through spending for those device categories, drugs, and biologicals that first become eligible for pass-through payment during CY 2026 will be approximately $307.0 million (approximately $291.8 million for device categories and approximately $15.2 million for drugs and biologicals), which represents only 0.30 percent of total projected OPPS payments for CY 2026 (approximately $101.0 billion). Therefore, we estimate that pass-through spending in CY 2026 will not exceed the 2.0 percent of total projected OPPS CY 2026 program spending limit provided for in section 1833(t)(6)(E) of the Act.
The estimated amount of $307.0 million of pass-through spending for the device categories and the drugs and biologicals that are continuing to receive pass-through payment in CY 2026 and the amount of pass-through spending for those device categories, drugs, and biologicals that first become eligible for pass-through payment during CY 2026 in this final rule is substantially different from the estimated amount of pass-through spending in the CY 2026 OPPS/ASC proposed rule (90 FR 33656) of $587.0 million mainly because, the updated estimated amount of pass-through spending for the second group of devices only includes the estimated pass-through spending for the devices that were approved for pass-through status in this final rule and the estimated pass-through expenditures for devices that are preliminarily approved as part of the device pass-through quarterly review process after the development of the CY 2026 OPPS/ASC proposed rule.
Comment: A commenter stated CMS's estimate of aggregate pass-through spending and application of statutory limits should not be allowed to restrict patient access to high-value therapies. The commenter additionally noted that to ensure safeguards are appropriately calibrated, CMS should publish analyses showing how spending caps affect specific categories such as oncology, rare diseases, and advanced diagnostics. The commenter elaborated that this would help interested parties assess whether limits create unintended barriers and inform any mid-course corrections that might be needed.
Response: We note the spending limit for pass-through is not considered during the pass-through application evaluation process and therefore, would not impact our final determination of pass-through payment status for any eligible technology. The spending limit is considered for the purposes of assessing the total amount of pass-through payments in a given calendar year for technologies which are currently eligible for pass-through payment status. If, before the beginning of the calendar year, we estimate that the total amount of pass-through payments in that year will exceed the applicable percentage, section 1833(t)(6)(E)(iii) of the Act requires a uniform prospective reduction in the amount of each of the transitional pass-through payments made in that year to ensure that the limit is not exceeded. ( printed page 53770)
VII. OPPS Payment for Hospital Outpatient Visits and Critical Care Services
For CY 2026, we proposed to continue our current clinic and emergency department (ED) hospital outpatient visit payment policies. For a description of these policies, we referred readers to the CY 2016 OPPS/ASC final rule with comment period (80 FR 70298). We also proposed to continue our payment policy for critical care services for CY 2026. For a description of this policy, we referred readers to the CY 2016 OPPS/ASC final rule with comment period (80 FR 70298), and for the history of this payment policy, we referred readers to the CY 2014 OPPS/ASC final rule with comment period (78 FR 75043).
As we stated in the CY 2022 OPPS/ASC final rule with comment period (86 FR 63663), the volume control method for clinic visits furnished by excepted off-campus provider-based departments (PBDs) applies for CY 2022 and subsequent years. More specifically, we finalized a policy to continue to utilize a PFS-equivalent payment rate for the hospital outpatient clinic visit service described by HCPCS code G0463 when it is furnished by these departments for CY 2022 and subsequent years. The PFS-equivalent rate for CY 2026 is 40 percent of the proposed OPPS payment. Under this policy, these departments will be paid approximately 40 percent of the OPPS rate for the clinic visit service in CY 2026. For CY 2026, we proposed to implement a volume control method for additional services furnished by excepted PBDs. For more information on this policy, we refer readers to section X.A. of this final rule with comment period.
In the CY 2023 OPPS/ASC final rule with comment period (87 FR 71748), we finalized a policy that excepted off-campus PBDs (departments that bill the modifier “PO” on claim lines) of rural Sole Community Hospitals (SCHs), as described under 42 CFR 412.92 and designated as rural for Medicare payment purposes, are exempt from the clinic visit payment policy that applies a PFS-equivalent payment rate for the clinic visit service, as described by HCPCS code G0463, when provided at an off-campus PBD excepted from section 1833(t)(21) of the Act. For the full discussion of this policy, we refer readers to the CY 2023 OPPS/ASC final rule with comment period (87 FR 72047 through 72051). For CY 2026, we proposed to exempt excepted off-campus PBDs (departments that bill the modifier “PO” on claim lines) of rural SCHs, as described under 42 CFR 412.92 and designated as rural for Medicare payment purposes, from any additional services subject to our volume control method payment policy. For more information on this policy, we refer readers to section X.A. of this final rule with comment period.
We did not receive specific public comment on our existing clinic and ED hospital outpatient visits payment policies outside of the context of the proposed expansion of our volume control method to other services, and therefore, we are finalizing as proposed to continue these policies. For discussion of the existing clinic visit policy in the context of the proposed expansion of our volume control method to other services, please see section X.A. of this final rule with comment period.
VIII. Payment for Partial Hospitalization and Intensive Outpatient Services
This section discusses payment for partial hospitalization services as well as intensive outpatient services. Since CY 2000, Medicare has paid for partial hospitalization services under the OPPS. Beginning in CY 2024, as authorized by section 4124 of the Consolidated Appropriations Act (CAA), 2023 (Pub. L. 117-328), Medicare began paying for intensive outpatient services furnished by hospital outpatient departments, community mental health centers, Federally qualified health centers, and rural health clinics in addition to opioid treatment programs. Additional background on the partial hospitalization and intensive outpatient benefits is included in the following paragraphs.
A. Background
1. Partial Hospitalization
A partial hospitalization program (PHP) is an intensive outpatient program of psychiatric services provided as an alternative to inpatient psychiatric care for individuals who have an acute mental illness, which includes, but is not limited to, conditions such as depression, schizophrenia, and substance use disorders (SUD). Section 1861(ff)(1) of the Act defines partial hospitalization services as the items and services described in paragraph (2) prescribed by a physician and provided under a program described in paragraph (3) under the supervision of a physician pursuant to an individualized, written plan of treatment established and periodically reviewed by a physician (in consultation with appropriate staff participating in such program), which sets forth the physician's diagnosis, the type, amount, frequency, and duration of the items and services provided under the plan, and the goals for treatment under the plan. Section 1861(ff)(2) of the Act describes the items and services included in partial hospitalization services. Section 1861(ff)(3)(A) of the Act specifies that a PHP is a program furnished by a hospital to its outpatients or by a community mental health center (CMHC), as a distinct and organized intensive ambulatory treatment service, offering less than 24-hour-daily care, in a location other than an individual's home or inpatient or residential setting. Section 1861(ff)(3)(B) of the Act defines a CMHC for purposes of this benefit. We refer readers to sections 1833(t)(1)(B)(i), 1833(t)(2)(B), 1833(t)(2)(C), and 1833(t)(9)(A) of the Act and 42 CFR 419.21, for additional information regarding PHP.
PHP policies and payment have been addressed under OPPS since CY 2000. In CY 2008, we began efforts to strengthen the PHP benefit through extensive data analysis, along with policy and payment changes, by implementing two refinements to the methodology for computing the PHP median. For a detailed discussion on these policies, we refer readers to the CY 2008 OPPS/ASC final rule with comment period (72 FR 66670 through 66676). In CY 2009, we implemented several regulatory, policy, and payment changes. For a detailed discussion on these policies, we refer readers to the CY 2009 OPPS/ASC final rule with comment period (73 FR 68688 through 68697). In CY 2010, we retained the two-tier payment approach for partial hospitalization services and used only hospital-based PHP data in computing the PHP APC per diem costs, upon which PHP APC per diem payment rates are based (74 FR 60556 through 60559). In CY 2011 (75 FR 71994), we established four separate PHP APC per diem payment rates: two for CMHCs (APC 0172 and APC 0173) and two for hospital-based PHPs (APC 0175 and APC 0176). We also instituted a 2-year transition period for CMHCs to the CMHC APC per diem payment rates. For a detailed discussion, we refer readers to section X.B. of the CY 2011 OPPS/ASC final rule with comment period (75 FR 71991 through 71994). In CY 2012, we determined the relative payment weights for partial hospitalization services provided by CMHCs based on data derived solely from CMHCs and the relative payment weights for partial hospitalization services provided by ( printed page 53771) hospital-based PHPs based exclusively on hospital data (76 FR 74348 through 74352). In the CY 2013 OPPS/ASC final rule with comment period, we finalized our proposal to base the relative payment weights that underpin the OPPS APCs, including the four PHP APCs (APCs 0172, 0173, 0175, and 0176), on geometric mean costs rather than on the median costs. For a detailed discussion on this policy, we refer readers to the CY 2013 OPPS/ASC final rule with comment period (77 FR 68406 through 68412).
In the CY 2014 OPPS/ASC proposed rule (78 FR 43621 and 43622) and CY 2015 OPPS/ASC final rule with comment period (79 FR 66902 through 66908), we continued to apply our established policies to calculate the four PHP APC per diem payment rates based on geometric mean per diem costs using the most recent claims data for each provider type. For a detailed discussion on this policy, we refer readers to the CY 2014 OPPS/ASC final rule with comment period (78 FR 75047 through 75050). In the CY 2016 OPPS/ASC final rule with comment period (80 FR 70453 through 70467), we described our extensive analysis of the claims and cost data and ratesetting methodology, corrected a cost inversion that occurred in the final rule with comment period data with respect to hospital-based PHP providers, and renumbered the PHP APCs. In the CY 2017 OPPS/ASC final rule with comment period (81 FR 79687 through 79691), we continued to apply our established policies to calculate the PHP APC per diem payment rates based on geometric mean per diem costs and finalized a policy to combine the Level 1 and Level 2 PHP APCs for CMHCs and for hospital-based PHPs. We also implemented an eight-percent outlier cap for CMHCs to mitigate potential outlier billing vulnerabilities. For a comprehensive description of PHP payment policy, including a detailed methodology for determining PHP per diem amounts, we refer readers to the CY 2016 and CY 2017 OPPS/ASC final rules with comment period (80 FR 70453 through 70455 and 81 FR 79678 through 79680, respectively).
In the CYs 2018 and 2019 OPPS/ASC final rules with comment period (82 FR 59373 through 59381 and 83 FR 58983 through 58998, respectively), we continued to apply our established policies to calculate the PHP APC per diem payment rates based on geometric mean per diem costs, designated a portion of the estimated 1.0 percent hospital outpatient outlier threshold specifically for CMHCs, and proposed updates to the PHP allowable HCPCS codes. We finalized these proposals in the CY 2020 OPPS/ASC final rule with comment period (84 FR 61352).
In the CY 2020 OPPS/ASC final rule with comment period (84 FR 61339 through 61350), we finalized a proposal to use the calculated CY 2020 CMHC geometric mean per diem cost and the calculated CY 2020 hospital-based PHP geometric mean per diem cost, but with a cost floor equal to the CY 2019 final geometric mean per diem costs as the basis for developing the CY 2020 PHP APC per diem rates. Also, we continued to designate a portion of the estimated 1.0 percent hospital outpatient outlier threshold specifically for CMHCs, consistent with the percentage of projected payments to CMHCs under the OPPS, excluding outlier payments.
In the April 30, 2020 interim final rule with comment (85 FR 27562 through 27566), effective as of March 1, 2020 and for the duration of the COVID-19 Public Health Emergency (PHE), hospital and CMHC staff were permitted to furnish certain outpatient therapy, counseling, and educational services (including certain PHP services), incident to a physician's services, to beneficiaries in temporary expansion locations, including the beneficiary's home, as long as the location met all conditions of participation to the extent not waived. A hospital or CMHC could furnish such services using telecommunications technology to a beneficiary in a temporary expansion location if that beneficiary was registered as an outpatient. In the CY 2023 OPPS/ASC final rule with comment period (87 FR 72247), we confirmed that these provisions applied only for the duration of the COVID-19 PHE. On May 11, 2023, the COVID-19 PHE ended, and accordingly, these flexibilities ended as well.
In the CY 2021 OPPS/ASC final rule with comment period (85 FR 86073 through 86080), we continued our current methodology to utilize cost floors, as needed. In the CY 2022 OPPS/ASC final rule with comment period (86 FR 63665 and 63666), as a result of the COVID-19 PHE, we finalized our proposal to calculate the PHP per diem costs using the year of claims consistent with the calculations that would be used for other OPPS services, by using the CY 2019 claims and the cost reports that were used for CY 2021 final rulemaking to calculate the CY 2022 PHP per diem costs. In addition, for CY 2022 and subsequent years, we finalized our proposal to use cost and charge data from the Hospital Cost Report Information System (HCRIS) as the source for the CMHC cost-to-charge ratios (CCRs), instead of using the Outpatient Provider Specific File (OPSF) (86 FR 63666).
In the CY 2023 OPPS/ASC final rule with comment period (87 FR 71995), we finalized our proposal to use the latest available CY 2021 claims but use the cost information from prior to the COVID-19 PHE for calculating the CY 2023 CMHC and hospital-based PHP APC per diem costs. The application of the OPPS standard methodology, including the effect of budget neutralizing all other OPPS policy changes unique to CY 2023, resulted in the final calculated CMHC PHP APC payment rate being unexpectedly lower than the CY 2022 final CMHC PHP APC rate. Therefore, we finalized utilizing the equitable adjustment authority of section 1833(t)(2)(E) of the Act to appropriately pay for CMHC PHP services at the same payment rate as for CY 2022, that is, $142.70. In addition, we clarified the payment under the OPPS for new HCPCS codes that designate non-PHP services provided for the purposes of diagnosis, evaluation, or treatment of a mental health disorder and are furnished to beneficiaries in their homes by clinical staff of the hospital that would not be recognized as PHP services; however, none of the PHP regulations would preclude a patient that is under a PHP plan of care from receiving other reasonable and medically necessary non-PHP services from a hospital (87 FR 72001 and 72002).
In the CY 2024 OPPS/ASC final rule with comment period (88 FR 81811), we revised the regulation at § 424.24(e)(1)(i) to require the physician certification for PHP services to include a certification that the patient requires such services for a minimum of 20 hours per week, as required by section 1861(ff)(1) of the Act, as amended by section 4124(a) of Division FF of the CAA, 2023. In addition, we modified the regulations for PHP at § 410.43 to include references to SUD. In the same CY 2024 OPPS/ASC final rule with comment period, we also established separate payment rates for PHP days with 3 services and days with 4 or more services. Accordingly, we established four separate PHP APC per diem payment rates: one for CMHCs for 3-service days and another for CMHCs for 4-service days (APC 5853 and APC 5854, respectively), and one for hospital-based PHPs for 3-service days and another for hospital-based PHPs for 4-service days (APC 5863 and APC 5864, respectively). We also finalized a policy to utilize the separate CMHC rates for 3-service and 4-service PHP days as the Medicare Physician Fee Schedule (MPFS) rates, depending upon whether a nonexcepted off-campus ( printed page 53772) hospital outpatient department furnishes 3 or 4 PHP services in a day. Lastly, we finalized several changes beginning in CY 2024 to align coding, billing, and payment between PHPs and intensive outpatient programs.
In the CY 2025 OPPS/ASC final rule with comment period (89 FR 94266 through 94268), we maintained the coding and billing policies for PHP as established in the CY 2024 OPPS/ASC final rule with comment period.
2. Intensive Outpatient Program Services
Section 4124(b) of the CAA, 2023, amended section 1861(ff) of the Act, establishing Medicare coverage for intensive outpatient services effective for items and services furnished on or after January 1, 2024. An intensive outpatient program (IOP) is a distinct and organized program of psychiatric services for individuals who have an acute mental illness, which includes, but is not limited to, conditions such as depression, schizophrenia, and SUD. Intensive outpatient services are not required to be provided in lieu of inpatient hospitalization. Section 1861(ff)(4) of the Act defines intensive outpatient services as the items and services described in paragraph (2) of section 1861(ff) prescribed by a physician for an individual determined (not less frequently than every other month) by a physician to have a need for such services for a minimum of 9 hours per week and provided under a program described in paragraph (3) under the supervision of a physician pursuant to an individualized, written plan of treatment established and periodically reviewed by a physician (in consultation with appropriate staff participating in such program), which plan sets forth the physician's diagnosis, the type, amount, frequency, and duration of the items and services provided under the plan, and the goals for treatment under the plan. Section 1861(ff)(2) of the Act describes the items and services included in intensive outpatient services. Section 1861(ff)(4)(C) of the Act specifies that an IOP is a program furnished by a hospital to its outpatients, by a CMHC, by a Federally qualified health center (FQHC), or by a rural health clinic (RHC) as a distinct and organized intensive ambulatory treatment service, offering less than 24-hour-daily care, in a location other than an individual's home or inpatient or residential setting. Section 1861(ff)(3)(B) of the Act defines a CMHC for purposes of this benefit. We refer readers to sections 1833(t)(1)(B)(i), 1833(t)(2)(B), 1833(t)(2)(C), and 1833(t)(9)(A) of the Act and 42 CFR 419.21, for additional information regarding IOP.
In the CY 2024 OPPS/ASC final rule with comment period (88 FR 81812 through 81857), we established payment and program requirements for the IOP benefit furnished by a hospital to its outpatients, or by a CMHC, an FQHC, or an RHC. In addition, we established Medicare Part B coverage for IOP services provided by Opioid Treatment Programs (OTPs) for the treatment of opioid use disorder (OUD).
Consistent with the statutory definition of intensive outpatient services under section 1861(ff)(4) of the Act, we finalized regulations at 42 CFR 410.44 to set forth the conditions and exclusions applicable for intensive outpatient services, and at § 424.24 to set forth the content of the certification and plan of treatment requirements for intensive outpatient services. We also revised certain existing regulations at §§ 410.2, 410.3, 410.10, 410.27, 410.150, and 419.21 to add a regulatory definition of intensive outpatient services and to include intensive outpatient services in the regulations for medical and other health services paid for under Medicare Part B, and in the case of § 419.21, under the OPPS. Additionally, we created regulations at § 410.111 to establish the requirements for coverage of IOP services furnished in CMHCs, and at § 410.173 to establish conditions of payment for IOP services furnished in CMHCs. Lastly, we revised § 410.155 to exclude IOP services from the outpatient mental health treatment limitation, consistent with the statutory requirement of section 1833(c)(2) of the Act, as amended by section 4124(b)(3) of the CAA, 2023.
In addition, as discussed in greater detail in the following sections, we established coding, billing, and payment policies for IOP that align with the policies established for PHP provided in the same settings. Specifically, we established four separate IOP APC per diem payment rates at the same rates we proposed for the PHP APCs: one for CMHCs for 3-service days and another for CMHCs for 4-service days (APC 5851 and APC 5852, respectively), and one for hospital-based IOPs for 3-service days and another for hospital-based IOPs for 4-service days (APC 5861 and APC 5862, respectively). Similar to the policy finalized for PHP, we finalized a policy to utilize the CMHC rates for 3-service and 4-service IOP days as the MPFS rates, depending upon whether a nonexcepted hospital outpatient department furnishes 3 or 4 IOP services in a day.
For IOP services provided by an RHC or FQHC, we established a 3-service per day payment rate based on the same rate as APC 5861, which is the 3-service hospital-based IOP rate (§ 405.2462(j)). In the CY 2025 PFS final rule, we established a 4 or more services per day payment rate for an IOP provided by an RHC or FQHC based on the same rate as APC 5862, which is the 4 or more services hospital-based IOP rate (89 FR 98017 and 98018). Information regarding payment policies for IOP services furnished by FQHCs and RHCs, including information regarding proposed CY 2026 policies for those settings, can be found in the MPFS proposed rule, which is published elsewhere in the Federal Register .
Furthermore, in the CY 2024 OPPS/ASC final rule with comment period, we established a payment adjustment for IOPs provided by an OTP based on three times the payment rate for APC 5861 beginning in CY 2024 (§ 410.67(d)(4)(i)(F)). We finalized regulations at § 410.67(d)(4)(ii) to add that the payment amount for OTP intensive outpatient services will be geographically adjusted using the Geographic Adjustment Factor (GAF) described in § 414.26. Lastly, we amended § 410.67(d)(4)(iii) to add that payment for OTP intensive outpatient services is updated annually using the Medicare Economic Index described in § 405.504(d). Payment rates for IOP provided in the OTP setting are updated as part of the OTP fee schedule and are not addressed in this CY 2026 OPPS/ASC proposed rule.
Lastly, in the CY 2025 OPPS/ASC final rule with comment period (89 FR 94266 through 94268), we maintained the coding and billing policies for IOP as established in the CY 2024 OPPS/ASC final rule with comment period.
B. Coding and Billing for PHP and IOP Services Under the OPPS
In the CY 2024 OPPS/ASC final rule with comment period, we finalized a billing requirement that all providers use condition code 41 to indicate that a claim is for partial hospitalization services and use condition code 92 to identify intensive outpatient claims, effective January 1, 2024. Since the statutory definitions of both IOP and PHP generally include the same types of items and services covered, we stated in the CY 2024 OPPS/ASC final rule with comment period that we believe it is appropriate to align the programs using a consistent list of services, so that level of intensity would be the only differentiating factor between partial hospitalization services and intensive outpatient services. The use of condition codes 41 for PHP claims and 92 for IOP claims allows us to ( printed page 53773) differentiate between these services for billing purposes.
We recognize that the level of intensity of mental health services that a patient requires may vary over time; therefore, we believe utilizing a consolidated list of HCPCS codes to identify services under both the IOP and PHP benefits supports a smooth transition for patients when a change in the intensity of their services is necessary to best meet their needs. For example, a patient receiving IOP services may experience an acute mental health need that necessitates more intense services through a PHP. Alternatively, an IOP patient that no longer requires the level of intensity provided by the IOP can access less intense mental health services, such as individual mental health services. The full list of HCPCs codes recognized under the PHP and IOP benefits can be found in the Medicare Claims Processing Internet Only Manual, Chapter 4, sections 260.1 and 261.1, respectively, and their subsections, available at https://www.cms.gov/regulations-and-guidance/guidance/manuals/downloads/clm104c04.pdf.
To qualify for payment for the IOP APC (5851, 5852, 5861, or 5862) or the PHP APC (5853, 5854, 5863, or 5864), one service provided that day must be from the Partial Hospitalization and Intensive Outpatient Primary list. We refer readers to the CY 2024 OPPS final rule with comment period for further discussion regarding our expectation that at least one of the services on the PHP and IOP Primary list will be indicated per day for patients who need the level of care offered by a PHP or IOP program. The PHP and IOP Primary List can be found in the CY 2024 OPPS/ASC final rule with comment period at 88 FR 81821.
Beginning in CY 2024, we recognized caregiver training services and Principal Illness Navigation (PIN) services as PHP and IOP services. We explained that the reported costs associated with providing such services are included when we calculate the PHP and IOP payment rates; however, these services do not count toward the determination of whether a PHP or IOP day is paid at the 3-service or 4-service rate. We refer readers to the CY 2024 OPPS/ASC final rule with comment period for a detailed discussion of this policy (88 FR 81823 through 81825).
As finalized in the CY 2024 OPPS/ASC final rule with comment period (88 FR 81821 and 81822), if new codes are established that represent the PHP and IOP services described under §§ 410.43(a)(4) and 410.44(a)(4), respectively, such codes are added to the list of codes recognized for payment for PHP or IOP through sub-regulatory guidance. We note that coding updates frequently occur outside of the standard rulemaking timeline. We adopted this sub-regulatory process to pay expeditiously when new codes are created that describe any of the services enumerated at §§ 410.43(a)(4) and 410.44(a)(4), which PHPs and IOPs, respectively, would provide. We explained that this policy applies to new codes that are cross walked to a previously included code, or whose code descriptor is substantially similar to a descriptor for a code on the list or describes a service on the list. We stated that any additional services not described at § 410.43(a)(4) or § 410.44(a)(4) would be added to the lists in regulation through notice and comment rulemaking.
In the CY 2025 OPPS/ASC final rule with comment period (89 FR 94266 through 94268), we did not add any new services not described at § 410.43(a)(4) or § 410.44(a)(4) to the list of PHP and IOP services.
We did not propose to add any new services to the list of PHP and IOP services for CY 2026. The following is a summary of the comments we received regarding these policies and our responses.
Comment: One commenter expressed support for our coding policies for occupational therapy services furnished in PHPs and IOPs, noting that these policies reinforce the importance of occupational therapy services in PHPs and IOPs. This commenter also advocated for including measures specific to occupational therapy in quality reporting programs.
Response: We thank the commenter for their support of our coding policies for occupational therapy services furnished in PHPs and IOPs. We will take the commenter's suggestions into consideration to potentially inform future rulemaking about quality measures.
Comment: One commenter advocated for coverage for virtual PHPs and IOPs to increase access to services, particularly for patients in rural areas who may have to travel for several hours to access behavioral health or SUD services.
Response: As we have previously discussed in the CY 2023 OPPS/ASC final rule with comment period (87 FR 72000), section 1861(ff)(3)(A) of the Act, which defines partial hospitalization services, specifies that a PHP is a program furnished by a hospital to its outpatients or by a community mental health center (CMHC), as a distinct and organized intensive ambulatory treatment service, offering less than 24-hour-daily care, in a location other than an individual's home or inpatient or residential setting. Section 1861(ff)(4) of the Act defines IOP with reference to that is, IOP services must be furnished in a location other than an individual's home or inpatient or residential setting.
C. CY 2026 Payment Rates for PHP and IOP
We proposed for CY 2026 to maintain the current payment rate methodology that we use for calculating PHP and IOP payment rates for hospital-based providers. For CMHCs, we proposed to revise our methodology for calculating PHP and IOP payment rates. Specifically, we proposed to apply the 40 percent MPFS Relativity Adjuster to calculate PHP and IOP payment rates for CMHCs. In the CY 2026 OPPS/ASC proposed rule (90 FR 33660), we explained that we would multiply the CY 2026 rates for the hospital-based PHP and IOP APCs by 0.4 to calculate the payment rates for the CMHC PHP and IOP APCs.
1. Background on the Current Payment Rate Methodology for PHP and IOP
Beginning in CY 2024, we established four separate PHP APC per diem payment rates: one for CMHCs for 3-service days and another for CMHCs for 4-service days (APC 5853 and APC 5854, respectively), and one for hospital-based PHPs for 3-service days and another for hospital-based PHPs for 4-service days (APC 5863 and APC 5864, respectively). In addition, for hospital-based PHPs, we finalized a policy to calculate payment rates using the broader OPPS data set, instead of using hospital-based PHP data only. We explained that using the broader OPPS data set allows CMS to capture data from claims not identified as PHP, but that also include the service codes and intensity required for a PHP day. Because we established consistent coding and payment between the PHP and IOP benefits, we considered all OPPS data for PHP days and non-PHP days that include 3 or more of the same service codes. We established four separate IOP APC per diem payment rates at the same rates we proposed for the PHP APCs: one for CMHCs for 3-service days and another for CMHCs for 4-service days (APC 5851 and APC 5852, respectively), and one for hospital-based IOPs for 3-service days and another for hospital-based IOPs for 4-service days (APC 5861 and APC 5862, respectively).
In the CY 2024 OPPS/ASC final rule with comment period (88 FR 81829 and 81830), we noted that the standard PHP ( printed page 53774) day is typically four services or more per day. We explained that we have historically provided payment for three services a day for extenuating circumstances when a beneficiary would be unable to complete a full day of PHP treatment. As we stated in the CY 2008 OPPS/ASC final rule with comment period (72 FR 66672), it was never our intention that days with only three units of service should represent the number of services provided in a typical PHP day. Our intention was to cover days that consisted of three units of service only in certain limited circumstances. For example, as we noted in the CY 2009 OPPS/ASC proposed rule (73 FR 41513), we believe 3-service days may be appropriate when a patient is transitioning towards discharge (or days when a patient is at the beginning of his or her PHP stay). Another example of when it may be appropriate for a program to provide only three units of service in a day is when a patient is required to leave the PHP early for the day due to an unexpected medical appointment.
In the same CY 2024 OPPS/ASC final rule with comment period, we also explained that prior to CY 2024, we historically prepared the data by first applying PHP-specific trims and data exclusions and assessing CCRs. We direct the reader to the CY 2016 OPPS/ASC final rule with comment period (80 FR 70463 through 70465) for a more complete discussion of these trims, data exclusions, and CCR adjustments. In prior rules, we typically included a discussion of PHP-specific data trims, exclusions, and CCR adjustments; we did not include that discussion in the CY 2024 OPPS/ASC proposed rule or final rule with comment period. We stated that these PHP-specific data trims and exclusions addressed limitations as well as anomalies in the PHP data. However, as noted earlier, we finalized a methodology for CY 2024 to calculate hospital-based PHP payment rates for 3 services per day and 4 services per day based on cost per day using the broader OPPS data set. Accordingly, we did not apply PHP-specific trims and data exclusions, but rather we applied the same trims and data exclusions consistent with the OPPS.
We stated in the CY 2024 OPPS/ASC final rule with comment period (88 FR 81830) that while no IOP benefit existed prior to the CAA, 2023, the types of items and services included in IOP had been, and were, paid for by Medicare either as part of the PHP benefit or under the OPPS more generally. Additionally, we stated that prior to the CAA, 2023, CMS had begun gathering information from interested parties on IOP under Medicare. In the CY 2023 OPPS/ASC proposed rule (87 FR 44679), we issued a comment solicitation on intensive outpatient mental health treatment, including SUD treatment furnished by IOPs, to collect information regarding whether there are any gaps in coding that may be limiting access to needed levels of care for treatment of mental health disorders or SUDs for Medicare beneficiaries, and specific information about IOP services, such as the settings of care in which these programs typically furnish services, the range of services typically offered, and the range of practitioner types that typically furnish these services.
In addition, in the same CY 2024 OPPS/ASC final rule with comment period, we explained that along with the requirements for IOP mandated by the CAA, 2023, we took into consideration the information we received from the comment solicitation to construct an appropriate data set to develop proposed rates for IOP. Since IOPs furnish the same types of services as PHP, just at a lower intensity, we stated that we believe it was appropriate to use the same data and methodology for calculating payment rates for both PHP and IOP for CY 2024. We explained that although PHP claims can be specifically identified, there was no specific identifier or billing code to indicate IOP services that may have been provided before CY 2024. However, we noted that hospitals have been permitted to furnish and bill for many of these services as outpatient services under the OPPS. Thus, we analyzed a broader set of data that included both PHP and non-PHP days with 3 or more services in order to calculate proposed payment for PHP services. To establish consistent payment between PHP and IOP, we set IOP payment rates at the same rates as PHP. We stated that the primary goal in developing the payment rate methodology for IOP and PHP services was to pay providers an appropriate amount relative to the patients' needs, and to avoid cost inversion in future years. We stated that setting the IOP payment rates equal to the PHP payment rates was appropriate because IOP was a newly established benefit, and we did not have definitive data on utilization. However, we explained that both programs utilize the same services, but furnish them at different levels of intensity, with different numbers of services furnished per day and per week, depending on the program. Therefore, we stated that we expect it would be appropriate to pay the same per diem rates for IOP and PHP services unless future data analysis supports calculating rates independently.
In the CY 2024 OPPS/ASC final rule with comment period (88 FR 81833) we established a policy of applying the 4-service day payment rate (that is, payment for PHP APCs 5854 for CMHCs and 5864 for hospitals, and IOP APCs 5852 for CMHCs and 5862 for hospitals) for days with 4 or more services. For days with three or fewer services, we apply the 3-service day payment rate (that is, payment for PHP APCs 5853 for CMHCs and 5863 for hospitals, and IOP APCs 5851 for CMHCs and 5861 for hospitals). As we noted in the CY 2024 OPPS/ASC final rule with comment period, we expect days with fewer than three services would be very infrequent, and we intend to monitor the provision of these days among providers and individual patients.
In the CY 2025 OPPS/ASC final rule with comment period (89 FR 94269), for beneficiaries in a PHP or IOP, we maintained the payment rate methodology finalized in the CY 2024 OPPS/ASC final rule with comment period.
2. Analysis of PHP and IOP Costs Under the Current Methodology
In the CY 2026 OPPS/ASC proposed rule (90 FR 33661), we explained that when following the current cost structure, the calculated CY 2026 geometric mean per diem cost for hospital-based PHP and IOP providers that provide 3 services per day would be $340.90, which we proposed to use for calculating the payment rate for the 3-service day hospital-based PHP APC 5863 and the 3-service day hospital-based IOP APC 5861, as discussed in the following section. Likewise, the calculated CY 2026 geometric mean per diem cost for hospital-based PHP and IOP providers that provide 4 services per day would be $424.60, which we proposed to use for calculating the payment rate for the 4-service day hospital-based PHP APC 5864 and the 4-service day hospital-based IOP APC 5862, as discussed in the following section.
In the CY 2026 OPPS/ASC proposed rule (90 FR 33661), we explained that the calculated CY 2026 geometric mean per diem cost for CMHC PHP and IOP providers would result in an inversion, with the CMHC 3-service geometric mean per diem costs equaling $191.83 and the CMHC 4-service geometric mean per diem costs equaling $110.39. We stated that we believe the inverted geometric mean per diem costs were influenced by the small number of CMHCs that bill Medicare for PHP and IOP services, as well as CMHCs with ( printed page 53775) low costs that first began billing Medicare for services in CY 2024. Table 117 summarizes the PHP and IOP geomean costs calculated using the current methodology as set forth in the CY 2026 OPPS/ASC proposed rule.
3. CY 2026 Payment Rate Methodology for PHP and IOP
For CY 2026, we proposed to maintain our current methodology of calculating separate rates for hospitals and CMHCs. For the four hospital-based PHP and IOP APCs (that is, APCs 5861, 5862, 5863, and 5864), we proposed using the latest available cost information, from cost reports beginning three fiscal years prior to the year that is the subject of the rulemaking, and CY 2024 OPPS claims to update the payment rates. We explained that this proposal was consistent with the overall proposed use of cost data for the OPPS, which is discussed in section II.A.1.a. of the CY 2026 OPPS/ASC proposed rule (90 FR 33485 and 33486).
In accordance with the methodology finalized in the CY 2024 OPPS/ASC final rule with comment period, we proposed to base the payment rate for each hospital-based PHP APC on the geometric mean per diem cost for days with three services and four or more services. We proposed to use the broader set of OPPS data to calculate the geometric mean costs for hospital outpatient departments, and we proposed to apply the same trims and exclusions consistent with the OPPS. We also proposed to set the payment rates for the hospital-based IOP APCs based on the geometric mean per diem cost for PHP days with three services and four or more services.
For the four CMHC PHP and IOP APCs (that is, APCs 5851, 5852, 5853, and 5854), we proposed to calculate the CY 2026 geometric mean per diem costs based on 40 percent of the corresponding hospital-based PHP and IOP APCs (APCs 5861, 5862, 5863, and 5864, respectively). We proposed this change in methodology for calculating the four CMHC PHP and IOP APCs because using the current methodology would result in inverted costs for CMHCs (that is, the cost for 3-service days would be greater than the cost for 4-service days), as discussed in the preceding section. As we discuss further in the following section of this final rule with comment period, we believe this methodology would be generally appropriate for estimating CMHC costs and would align with the methodology that is used for other nonexcepted OPPS services furnished by a nonexcepted off-campus hospital outpatient department.
Lastly, we proposed that if more recent hospital cost data subsequently became available after the publication of the CY 2026 OPPS/ASC proposed rule (90 FR 33476, July 17, 2025), we would consider using such updated data to determine the CY 2026 payment rates for the four PHP APCs and the four IOP APCs.
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: A few commenters generally supported our proposal to maintain the current methodology of calculating hospital-based and CMHC-based PHP and IOP payment rates for 3 services per day and 4 services per day. One commenter encouraged CMS to continue to pay for those days of service when patients receive only 3 services per day.
Response: We thank commenters for their support. We did not propose any changes to the structure of our payment rate methodology for CY 2026, and we are not finalizing any changes in this final rule with comment period. As we discussed in section VIII.C.1. of this final rule with comment period, we believe utilizing the three-service payment rate (that is, payment for PHP APCs 5853 for CMHCs and 5863 for hospitals, and IOP APCs 5851 for CMHCs and 5861 for hospitals) for days with three or fewer services would accommodate occasional instances when a patient is unable to complete a full day of PHP or IOP.
Comment: Multiple commenters stated that CMHC cost estimates do not reflect the actual costs of providing PHP and IOP and that the resources involved are comparable to those required for hospital-based programs. Commenters stated that CMS' policy of paying separate rates for hospitals and CMHCs creates arbitrary incentives for PHP and IOP services in settings other than CMHCs.
Response: We disagree with the commenters who believe CMS policy creates an incentive for PHP and IOP services in settings other than CMHCs. As discussed earlier in this section and in the CY 2026 OPPS/ASC proposed rule (90 FR 33662), our longstanding payment policies reflect the cost ( printed page 53776) differences between the CMHC and hospital settings, based on our observation of CMHCs incurring significantly different costs than hospitals in the provision of PHP services. We believe that the stark difference between hospital and CMHC costs for PHP and IOP services that we continue to observe in the data reflects the actual cost structure differences between facility types.
Final Decision: After consideration of public comments, we are finalizing the payment rate methodology for CY 2026 as proposed.
4. CY 2026 PHP and IOP APC Geometric Mean Per Diem Costs
In the CY 2024 OPPS/ASC final rule with comment period (88 FR 81831), we anticipated there would be significant differences between CMHCs' and hospitals' costs of furnishing IOP, based on our observation of CMHCs incurring significantly different costs than hospitals in the provision of PHP services. Our longstanding payment policies reflect those differences. In the CY 2026 OPPS/ASC proposed rule (90 FR 33662), we noted that for CY 2026, we continue to observe significant cost structure differences between hospitals and CMHCs in the provision of PHP and IOP services. That is, we continue to see lower PHP and IOP costs in the CMHC setting as compared to the hospital setting. However, as we noted earlier in this final rule with comment period, if we were to apply our current methodology for calculating the CY 2026 geometric mean per diem costs for CMHC PHP and IOP APCs, those costs would be inverted (that is, the cost for 3-service days would be greater than the cost for 4-service days).
We believe it is appropriate to continue to recognize the differences in cost structures for different providers of PHP and IOP. This is of particular importance not only to the Medicare program, but also for the Medicare beneficiaries that CMHCs serve, who are subject to a 20 percent coinsurance requirement on all PHP and IOP services under Part B. However, as we previously explained, one of our goals is to avoid cost inversion because we would expect that the geometric mean per diem costs when providing three services per day would be lower than the geometric mean per diem costs when providing four or more services per day. We note that our current estimates are significantly impacted by a small number of CMHCs with low estimated costs who first began billing Medicare for services in CY 2024.[128] In the CY 2026 OPPS/ASC proposed rule (90 FR 33662), we explained that we were concerned that these cost estimates may not best reflect the costs of providing PHP and IOP in CY 2026. As such, we stated that we believe that using CMHC data to establish the CMHC payment rates is not appropriate for CY 2026, given the cost inversion. For CY 2026, we considered alternative methodological approaches to estimate the costs for PHP and IOP services furnished by CMHCs.
Section 1833(t)(9)(A) of the Act requires the Secretary to annually review and revise the relative payment weights by considering new cost data, and other relevant information and factors. We note that in creating the original APC for PHP services (APC 0033), the initial relative payment weight for PHP services provided in hospital-based and CMHC-based settings was based on hospital data only. Subsequently, we have, in prior rulemaking, exercised our authority under section 1833(t)(9)(A) of the Act to change the data source for the relative payment weights for hospital-based and CMHC-based PHP services as new cost data became available. We refer readers to the CY 2012 OPPS/ASC final rule with comment period (76 FR 74350 and 74351) for more details on the history of changes in the data sources for relative payment weights for PHP services.
For CY 2026, we considered alternative methodological approaches for calculating the CMHC costs that could avoid cost inversions and provide greater stability for CMHC payment rates. We believe the stability of CMHC payment rates and the avoidance of cost inversions are important for CMHCs to more easily anticipate future payments associated with the PHP and IOP benefits. For CY 2026, we considered whether the 40 percent MPFS Relativity Adjuster would appropriately estimate CMHC PHP and IOP costs.
First, we considered the similarities between CMHCs and nonexcepted off-campus hospital outpatient departments. CMHCs are freestanding entities that are not part of a hospital, but they provide the same PHP and IOP services as hospital-based PHP and IOPs. As we noted in the CY 2017 OPPS/ASC final rule with comment period (81 FR 79717), this is similar to the differences between freestanding entities paid under the MPFS that furnish other services also provided by hospital-based entities. Similarly, to other entities currently paid for their technical component services under the MPFS, we believe CMHCs would typically have lower cost structures than hospital-based PHP and IOPs, largely due to lower overhead costs and other indirect costs such as administration, personnel, and security.
The 40 percent MPFS Relativity Adjuster was established in the CY 2018 PFS final rule (82 FR 53030) and applies to payments for nonexcepted items and services furnished in nonexcepted off-campus provider-based departments, including a hospital outpatient department. In that same final rule, we discussed our rationale for finalizing the MPFS Relativity Adjuster at 40 percent (82 FR 53026 through 53030). We explained that we believe a 40 percent adjuster would reflect a middle ground between the CY 2017 PFS Relativity Adjuster of 50 percent (selected to ensure adequate payment to hospitals) and the proposed CY 2018 PFS Relativity Adjuster of 25 percent (selected to ensure that hospitals are not paid more than others would be paid through the PFS non-facility rate).
Application of the 40 percent MPFS Relativity Adjuster to determine the CMHC geometric mean per diem costs bases the relative payment weights for PHP and IOP services furnished by CMHCs on hospital cost data, which we note has been more stable than CMHC cost data in recent years. In the CY 2026 OPPS/ASC proposed rule (90 FR 33662), we explained that the stability of the hospital cost data is primarily driven by the larger number of providers and the fact that hospital-based providers more consistently bill for PHP and IOP services from one year to the next. We stated that we believe this methodology would appropriately stabilize CMHC payment rates by setting them relative to hospital-based rates, while avoiding cost inversions in future years. We also stated that we believe applying the 40 percent MPFS Relativity Adjuster to calculate payment rates for the CMHC PHP and IOP APCs would support our longstanding goal to pay providers an appropriate amount relative to the patients' needs.
Application of the 40 percent MPFS Relativity Adjuster to the hospital-based PHP and IOP geometric mean per diem costs resulted in proposed CY 2026 CMHC costs of $136.36 for a 3-service day and $169.84 for a 4-service day. These proposed CY 2026 CMHC costs are generally in line with the CY 2025 CMHC costs, which were $112.59 for a 3-service day and $170.37 for a 4-service ( printed page 53777) day. Additionally, we observed on average, the CY 2024 and CY 2025 geometric mean costs for the CMHC PHP and IOP APCs were 40 percent of the CY 2024 and CY 2025 geometric mean costs for the hospital-based PHP and IOP APCs. Therefore, we stated in the CY 2026 OPPS/ASC proposed rule (90 FR 33663), that applying the 40 percent Relativity Adjuster to hospital-based PHP and IOP costs would better approximate CMHC cost structures than the latest available CMHC cost data would. As we previously noted, the latest available CMHC cost data is influenced by the small number of CMHCs that bill Medicare for PHP and IOP services, as well as by CMHCs with low costs that first began billing Medicare for services in CY 2024.
Therefore, for the reasons discussed in the prior paragraphs, we proposed to apply the 40 percent MPFS Relativity Adjuster to the hospital-based PHP and IOP costs for the purposes of calculating the proposed geometric mean per diem costs for the CMHC PHP and IOP APCs for CY 2026 and subsequent years.
Given the requirements of section 1833(t)(9)(A) of the Act, we stated that we believe it would be appropriate to revise our methodology for setting the relative payment weights for the OPPS rates for PHP and IOP services furnished by CMHCs based on new cost data and other relevant information and factors. Specifically, we proposed to base this calculation on hospital cost data and the observed relationship between PHP and IOP costs in the hospital and CMHC settings, which as we noted earlier has been approximately 40 percent in recent years.
We noted in the CY 2026 OPPS/ASC proposed rule that we intend to monitor the provision of services in both PHP and IOP programs to better understand utilization patterns and would reevaluate our payment rate methodology if necessary. We also explained that if more recent data became available for the CY 2026 OPPS/ASC final rule that mitigated the cost inversion for CMHC geometric mean per diem costs, we may consider using such data as a basis for finalizing a payment rate methodology based on CMHC costs, rather than based on hospital costs adjusted by the 40 percent MPFS Relativity Adjuster.
We solicited comments on our current and proposed payment rate methodologies for PHP and IOP services furnished by CMHCs. We also solicited comments on potential methodological changes or changes to data that could avoid or mitigate future cost inversions and instability for CMHC payment rates. We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: A few commenters generally supported the proposed calculated geometric mean per diem costs for hospital-based PHP and IOP APCs.
Response: We thank commenters for their support.
Comment: Multiple commenters urged CMS to implement a site-neutral payment for CMHCs and hospital-based providers for PHP and IOP services. Some commenters stated that CMHCs are entitled under sections 1832(a)(2)(J) and 1833(a)(2)(B)(iii) of the Act to receive payment for PHP/IOP under the same methodology as hospital outpatient departments. A few commenters cited 1833(t)(2)(B) and stated that CMS has the discretion to create APC codes for different classes of services, but not to create distinct APC codes based on where a service is provided. Some commenters further stated that because of the small number of CMHCs billing for PHP and IOP services, paying CMHCs at the same higher rate as hospitals for these services would have a negligible impact on the Medicare program.
Response: It has been our longstanding policy since CY 2011 to pay separate PHP APC per diem payment rates for CMHCs and hospital-based PHPs. In the CY 2011 OPPS/ASC final rule with comment period (75 FR 71992), we stated that section 1833(t)(9)(A) of the Act requires the Secretary to “review not less often than annually and revise the groups, the relative payment weights, and the wage and other adjustments described in paragraph (2) to take into account changes in medical practice, changes in technology, the addition of new services, new cost data, and other relevant information and factors.” We stated that we believe we have authority to revise the groups and relative payment weights and to make other adjustments to the payment rates for PHP services, including basing rates on hospital-based PHP data only, combined hospital-based PHP and CMHC data, or CMHC data only, to take into account relevant information and factors that would allow us to more appropriately pay providers for the resource costs associated with providing PHP services. Accordingly, we finalized separate PHP APC per diem payment rates for hospital OPDs and CMHCs, and we have maintained that structure for payment since CY 2011. Beginning in CY 2024, we applied this payment structure to IOP because we expected (and subsequently have observed) differences in resource use between CMHCs and hospital OPDs for the provision of both PHP and IOP services.
In response to the commenters who cited to section 1833(t)(2)(B) of the Act, we believe we have authority to create APC codes as shown by our longstanding methodology for PHP and IOP. CMS data shows that PHP and IOP services furnished at hospital OPDs differ with respect to the use of resources from PHP and IOP services furnished at CMHCs; therefore, payment rates that recognize these resource differences are consistent with the statutory requirements under section 1833(t) of the Act.
We also disagree that paying CMHCs at the same higher rate as hospitals for these services would have a negligible impact on the Medicare program. Despite the small number of CMHCs billing Medicare, the volume of PHP and IOP services that they provide is significant. Our data from CY 2024 OPPS claims shows that CMHCs furnished approximately 38 percent of all PHP and IOP days that were paid under the OPPS. If we were to increase payment for these PHP and IOP days by 150 percent, as commenters have suggested, it would cause a significant increase in the total amount of PHP and IOP payments, both from the Medicare program and from Medicare beneficiaries in the form of coinsurance payments.
Comment: Some commenters stated that, given inflation and increased labor costs, the proposed rate is too low for CMHCs to effectively and appropriately staff and deliver PHPs and IOPs. The commenters stated that applying the 40 percent relativity adjuster could reduce CMHC payment rates compared to the hospital-based PHP and IOP payment rates.
Response: We appreciate the commenters' concerns; however, we disagree that the proposed rates are inadequate. We note that the OPPS payment rates are adjusted annually for inflation. In the CY 2026 OPPS/ASC proposed rule, we explained that the proposed CY 2026 CMHC costs were generally in line with the CY 2025 CMHC costs and noted that the proposed geometric mean per diem cost for CMHC IOP days with 4 or more services was $169.84, calculated by applying the 40 percent MPFS Relativity Adjuster (90 FR 33663). We performed further analysis of CMHC PHP and IOP claims which showed that CMHCs furnish PHP and IOP days with 4 or more services more frequently than days with 3 or fewer services. According to our analysis, the proposed CMHC geometric mean per diem cost for days ( printed page 53778) with 4 or more services would reimburse CMHCs at or above cost for approximately 75 percent of days with 4 or more services. Therefore, we believe applying the 40 percent MPFS Relativity Adjuster to the hospital-based PHP and IOP costs for the purposes of calculating the proposed geometric mean per diem costs for the CMHC PHP and IOP APCs results in adequate payment rates for CMHCs. We remind readers that due to the OPPS budget neutrality adjustments and the application of the final market basket update for CY 2026, the final APC payment rates may be higher or lower than their estimated APC geometric mean costs.
Comment: Some commenters stated that they do not believe the 40 percent MPFS Relativity Adjuster is relevant for calculating the geometric mean per diem costs for the CMHC PHP and IOP APCs rates.
Response: We disagree with commenters that the 40 percent MPFS Relativity Adjuster is not relevant for calculating the geometric mean per diem costs for the CMHC PHP and IOP APC rates. In the CY 2026 OPPS/ASC proposed rule (90 FR 33662), we noted the similarities between CMHCs and nonexcepted off-campus hospital outpatient departments. Additionally, we observed on average, the CY 2024 and CY 2025 geometric mean costs for the CMHC PHP and IOP APCs were 40 percent of the CY 2024 and CY 2025 geometric mean costs for the hospital-based PHP and IOP APCs. Therefore, we believe that applying the 40 percent Relativity Adjuster to hospital-based PHP and IOP costs better approximates CMHC cost structures than the latest available CMHC cost data does. We continue to believe this methodology appropriately stabilizes CMHC payment rates by setting them relative to hospital-based rates, while avoiding cost inversions in future years.
We note that the OPPS is subject to budget neutral adjustments to the weight scaler as described in section II.A.5. of this final rule with comment period, and APC payment rates are calculated using the OPPS conversion factor described in section II.B. of this final rule with comment period. As a result of the OPPS budget neutrality adjustments, the proposed and final APC payment rates may be higher or lower than their estimated APC geometric mean costs.
The final payment rates for the PHP and IOP APCs can be found in Addendum A of this CY 2026 OPPS/ASC final rule with comment period.
Comment: Commenters expressed concern that reduced CMHC payments could impact access to PHP and IOP services to patients in underserved communities. In addition, one commenter urged CMS to monitor utilization and outcomes stratified by geography and population characteristics such as age, disability status, and language proficiency to ensure that CMS payment policies translate into improved access to PHP and IOP services for all Medicare beneficiaries.
Response: We appreciate the concerns that commenters raised about Medicare beneficiaries' access to behavioral and mental health services. However, we do not believe that this policy reduces access to behavioral health services, because similarly other entities currently paid for their technical component services under the MPFS, we believe CMHCs typically have lower cost structures than hospital-based PHPs and IOPs, largely due to lower overhead costs and other indirect costs such as administration, personnel, and security. As we noted earlier, we believe the proposed payment rates for CMHCs provide adequate payment that reflects the level of cost in that setting. We intend to continue monitoring access to PHP and IOP services and may consider changes in future rulemaking, as appropriate.
Comment: Multiple commenters voiced concerns that administrative barriers (specifically, conditions of participation), contribute to the low number of CMHCs participating in the PHP and IOP benefits. The commenters suggested CMS change its interpretation, and clarify where needed, to state that the conditions of participation apply only to services that are billed by the CMHC.
Response: We thank commenters for their feedback. However, these comments are outside the scope of this rulemaking.
Final Decision: After consideration of the public comments we received, we are finalizing the payment rate methodology for CY 2026 as proposed. We will monitor the effects of this policy and will consider proposing modifications in future rulemaking if appropriate. Table 118 shows the final calculated geometric mean per diem costs for hospital-based PHP and IOP APCs, and the final geometric mean per diem costs for CMHC PHP and IOP APCs based on the application of the 40 percent MPFS Relativity Adjuster for this CY 2026 OPPS/ASC final rule with comment period. Additional information about the data trims, data exclusions, and CCR adjustments applicable to the data used for this final rule can be found online at https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/HospitalOutpatientPPS/index.html .[129]
( printed page 53779)D. Outlier Policy for CMHCs
For CY 2026, we proposed to maintain the calculations of the CMHC outlier percentage, cutoff point and percentage payment amount, outlier reconciliation, outlier payment cap, and fixed dollar threshold according to previously established policies to include PHP and IOP services. We refer readers to the CY 2024 OPPS/ASC final rule with comment period (88 FR 81834 through 81836) for more details on CMHC outlier policies, and to section II.G. of this final rule with comment period for our general policies for hospital outpatient outlier payments.
1. Background
As discussed in the CY 2004 OPPS final rule with comment period (68 FR 63469 and 63470), we created a separate outlier policy specific to the estimated costs and OPPS payments provided to CMHCs. We designated a portion of the estimated OPPS outlier threshold specifically for CMHCs, consistent with the percentage of projected payments to CMHCs under the OPPS each year, excluding outlier payments, and established a separate outlier threshold for CMHCs.
2. CMHC Outlier Percentage
In the CY 2018 OPPS/ASC final rule with comment period (82 FR 59267 and 59268), we described the current outlier policy for hospital outpatient payments and CMHCs. We note that we also discussed our outlier policy for CMHCs in more detail in section VIII.C. of that same final rule (82 FR 59381). We set our projected target for all OPPS aggregate outlier payments at 1.0 percent of the estimated aggregate total payments under the OPPS (82 FR 59267). This same policy was also reiterated in the CY 2019 OPPS/ASC final rule with comment period (83 FR 58996), the CY 2020 OPPS/ASC final rule with comment period (84 FR 61350), and the CY 2021 OPPS/ASC final rule with comment period (85 FR 86082). We did not propose any changes to the CMHC outlier percentage policy for CY 2026 and did not receive any public comments on this provision.
3. Cutoff Point and Percentage Payment Amount
Also described in the CY 2018 OPPS/ASC final rule with comment period (82 FR 59381), our policy has been to pay CMHCs for outliers if the estimated cost of the day exceeds a cutoff point. In CY 2006, we set the cutoff point for outlier payments at 3.4 times the highest CMHC PHP APC payment rate implemented for that calendar year (70 FR 68551). For CY 2018, the highest CMHC PHP APC payment rate was the payment rate for CMHC PHP APC 5853. In addition, in CY 2002, the final OPPS outlier payment percentage for costs above the multiplier threshold was set at 50 percent (66 FR 59889). In CY 2018, we continued to apply the same 50 percent outlier payment percentage that applies to hospitals to CMHCs and continued to use the existing cutoff point (82 FR 59381). Therefore, for CY 2018, we continued to pay for partial hospitalization services that exceeded 3.4 times the CMHC PHP APC payment rate at 50 percent of the amount of CMHC PHP APC geometric mean per diem costs over the cutoff point. This same policy was also reiterated in the CY 2019 OPPS/ASC final rule with comment period (83 FR 58996 and 58997), the CY 2020 OPPS/ASC final rule with comment period (84 FR 61351), the CY 2021 OPPS/ASC final rule with comment period (85 FR 86082 and 86083), the CY 2022 OPPS/ASC final rule with comment period (86 FR 63670), the CY 2023 OPPS/ASC final rule with comment period (87 FR 72004), and the CY 2024 OPPS/ASC final rule with comment period (88 FR 81835). In the CY 2024 OPPS/ASC final rule with comment period, we extended this policy to intensive outpatient services. We did not propose any changes to the cutoff point and payment amount policy for CY 2026 and did not receive any public comments on this provision.
4. Outlier Reconciliation
In the CY 2009 OPPS/ASC final rule with comment period (73 FR 68594 through 68599), we established an outlier reconciliation policy to address charging aberrations related to OPPS outlier payments. We addressed vulnerabilities in the OPPS outlier payment system that led to differences between billed charges and charges included in the overall CCR, which are used to estimate cost and apply to all hospitals and CMHCs paid under the OPPS. We initiated steps to ensure that outlier payments appropriately account for the financial risk when providing an extraordinarily costly and complex service but are only being made for services that legitimately qualify for the additional payment.
For a comprehensive description of outlier reconciliation, we refer readers to the CY 2023 OPPS/ASC and CY 2019 OPPS/ASC final rules with comment period (83 FR 58874 and 58875 and 81 FR 79678 through 79680, respectively). We did not propose any changes to the outlier reconciliation policy for CY 2026 ( printed page 53780) and did not receive any public comments on this provision.
5. Outlier Payment Cap
In the CY 2017 OPPS/ASC final rule with comment period, we implemented a CMHC outlier payment cap to be applied at the provider level, such that in any given year, an individual CMHC will receive no more than a set percentage of its CMHC total per diem payments in outlier payments (81 FR 79692 through 79695). Our analysis of CY 2014 claims data found that CMHC outlier payments began to increase similarly to the way they had prior to CY 2004. This was due to inflated costs from three CMHCs that accounted for 98 percent of all CMHC outlier payments that year and received outlier payments that ranged from 104 percent to 713 percent of their total per diem payments. To balance our concern about disadvantaging CMHCs with our interest in protecting the benefit from excessive outlier payments and to mitigate potential inappropriate outlier billing vulnerabilities, we finalized the CMHC outlier payment cap at 8 percent of the CMHC's total per diem payments (81 FR 79694 and 79695) to limit the impact of inflated CMHC charges on outlier payments. This cap was established after detailed analysis of claims data, which showed that a cap set at 8 percent would effectively address excessive outlier payments while minimally impacting CMHCs with legitimate high-cost cases. The cap applies to each CMHC's total per diem payments, which include both the Medicare payment portion and the beneficiary cost-sharing amount. The 8 percent cap continues to be calculated and applied on a calendar year basis, with outlier payments monitored throughout the year to ensure compliance with the cap.
This outlier payment cap only affects CMHCs; it does not affect other provider types (that is, hospital-based PHPs) and is in addition to and separate from the current outlier policy and reconciliation policy in effect. We did not propose any changes to the outlier payment cap for CY 2026 and did not receive any public comments on this provision.
6. Fixed-Dollar Threshold
In the CY 2018 OPPS/ASC final rule with comment period (82 FR 59267 and 59268), for the hospital outpatient outlier payment policy, we set a fixed-dollar threshold in addition to an APC multiplier threshold. Fixed-dollar thresholds are typically used to drive outlier payments for very costly items or services, such as cardiac pacemaker insertions. Currently, for CY 2025, CMHC PHP APCs (5853 or 5854) and IOP APCs (5851 or 5852) are the only APCs for which CMHCs may receive payment under the OPPS, and these APCs are for providing a defined set of services that are relatively low cost when compared to other OPPS services. Because of the relatively low cost of CMHC services that are used to comprise the structure of CMHC PHP APCs (5853 or 5854) and IOP APCs (5851 or 5852), it is not necessary to also impose a fixed-dollar threshold on CMHCs. Therefore, in the CY 2018 OPPS/ASC final rule with comment period, we did not set a fixed-dollar threshold for CMHC outlier payments (82 FR 59381). This same policy was also reiterated in the CY 2020 OPPS/ASC final rule with comment period (84 FR 61351), the CY 2021 OPPS/ASC final rule with comment period (85 FR 86083), the CY 2022 OPPS/ASC final rule with comment period (86 FR 63508), the CY 2023 OPPS/ASC final rule with comment period (87 FR 72004), the CY 2024 OPPS/ASC final rule with comment period (88 FR 81836), and the CY 2025 OPPS/ASC final rule with comment period (89 FR 94271). We did not propose any changes to the fixed-dollar threshold policy for CY 2026 and did not receive any public comments on this provision.
IX. Services That Will Be Paid Only as Inpatient Services
A. Background
The Inpatient Only (IPO) list was established in rulemaking as part of the initial implementation of the Outpatient Prospective Payment System (OPPS) in 2000, pursuant to the Secretary's authority under section 1833(t)(1)(B)(I) of the Act (65 FR 18455). The IPO list was created to identify services for which Medicare will make payment only when furnished in the inpatient hospital setting because of the invasive nature of the procedures, the underlying physical condition of the Medicare patient, or the need for at least 24 hours of postoperative recovery time or monitoring before the patient can be safely discharged (70 FR 68695). The creation of the IPO list was based on the premise (rooted in the practice of medicine at that time) that Medicare should not pay for procedures furnished as outpatient services which are performed on an inpatient basis virtually all of the time for the Medicare population because performing these procedures on an outpatient basis would not be safe or appropriate, and therefore not reasonable and necessary under Medicare rules (86 FR 63671; 63 FR 47571). Designation of a service as inpatient only does not preclude the service from being furnished in a hospital outpatient setting but means that Medicare will not make payment for the service if it is furnished to a Medicare beneficiary in the hospital outpatient setting (65 FR 18443). Conversely, the absence of a procedure from the list should not be interpreted as identifying that procedure as appropriately performed only in the hospital outpatient setting (70 FR 68696). Rather, from the beginning, we have emphasized our expectation that, in every case, the physician or surgeon and hospital will exercise their professional judgment and assess the risk of the procedure or service to the individual patient, taking into account the site of service and act in that patient's best interest (65 FR 18456). We have also previously stated that for procedures that are not included on the inpatient list, we rely on the practitioner's judgment to determine on a patient-by-patient basis whether or not a particular procedure would be most appropriately performed in the inpatient setting (70 FR 68698).
The IPO list policy has elicited both opposition and support in public comments since its establishment in CY 2000. In 2000, some commenters stated that they believed that CMS (then, the Health Care Financing Administration) was making decisions, such as the site of service for a particular medical procedure, which should be left to the discretion of surgeons and their patients (65 FR 18455). In 2011, certain comments suggested that regulations should not supersede the physician's level of knowledge and assessment of the patient's condition, and that the physician can appropriately determine whether a procedure can be performed in a hospital outpatient setting, and many commenters suggested that the inpatient only list be eliminated in its entirety (76 FR 74354). Again, in 2013, some commenters requested that the IPO list be eliminated in its entirety (78 FR 75055). From the beginning, several interested parties have also stated that the exclusion of services from payment under the OPPS is unnecessary and could have an adverse effect on advances in surgical care (65 FR 18442). Others have noted that the existence of the IPO list suggests that services that are not on the list or have been removed from the list should be/must be provided in the outpatient setting, regardless of the clinical judgment of the physician or the needs of the patient (85 FR 86084). Other commenters have defended the need for the list, stating that the IPO list serves as an important programmatic safeguard and maintains a ( printed page 53781) common standard in the Medicare program (85 FR 86086).
In the CY 2021 OPPS/ASC final rule with comment period, published in the Federal Register on December 29, 2020 (85 FR 86084 through 86088), we finalized a policy to eliminate the IPO list over the course of 3 years (85 FR 86093). We revised our regulation at 42 CFR 419.22(n) to state that, effective January 1, 2021, the Secretary shall eliminate the list of services and procedures designated as requiring inpatient care through a 3-year transition. As part of the first phase of this elimination of the IPO list, we removed 298 codes, including 266 musculoskeletal-related services, from the list beginning in CY 2021.
In the 2022 OPPS/ASC final rule with comment period, published on November 16, 2021, we halted the elimination of the IPO list and, after clinical review of the services removed from the IPO list in CY 2021 as part of the first phase of eliminating the IPO list, we returned most services removed from the IPO list in 2021 back to the IPO list beginning in CY 2022 (86 FR 63671 through 63736). We amended the regulation at § 419.22(n) to remove the reference to the elimination of the list of services and procedures designated as requiring inpatient care through a 3-year transition. We also finalized our proposal to codify the five longstanding criteria for determining whether a service or procedure should be removed from the IPO list in the regulation at § 419.23 (86 FR 63678). For CY 2023 through CY 2025, we maintained the IPO list and continued to evaluate services brought forth by interested parties for removal using the five longstanding criteria (87 FR 72004 through 72012; 88 FR 81858 through 81863; and 89 FR 94271 through 92475).
B. Current Methodology for Identifying Appropriate Changes to the IPO List
Currently, there are approximately 1,731 services on the IPO list. Under our longstanding policy and current regulations, we annually review the IPO list to identify any services that should be removed from, or added to, the list, based on the most recent data and medical evidence available. We have established five criteria to determine whether a procedure should be removed from the IPO list (65 FR 18455), which we codified in the CY 2022 OPPS/ASC final rule with comment period (86 FR 63676). As noted in the CY 2012 OPPS/ASC final rule with comment period (76 FR 74353), we assess whether a procedure or service met these criteria to determine if it should be removed from the IPO list and assign to an APC group for payment under the OPPS when provided in the hospital outpatient setting. We have explained that while we only require a service to meet one criterion to be considered for removal, satisfying only one criterion does not guarantee that the service will be removed; instead, the case for removal is strengthened with the more criteria the service meets. The criteria for assessing procedures for removal from the IPO list are:
- Most outpatient departments are equipped to provide the service or procedure to the Medicare population.
- The simplest service or procedure described by the code may be performed in most outpatient departments.
- The service or procedure is related to codes that CMS has already removed from the Inpatient Only list.
- CMS determines that the service or procedure is being performed in numerous hospitals on an outpatient basis.
- CMS determines that the service or procedure can be appropriately and safely performed in an ambulatory surgical center, and is specified as a covered ambulatory surgical procedure, or CMS has proposed to specify it as a covered ambulatory surgical procedure.
We encouraged interested parties, including professional societies, hospitals, surgeons, hospital associations, and beneficiary advocacy groups, to evaluate the IPO list and determine whether services should be added to or removed from the list. We requested that they submit corresponding evidence in support of their claims that a code or group of codes met the longstanding criteria for removal from the IPO list and is safe to perform on the Medicare population in the hospital outpatient setting—including, but not limited to case reports, operative reports of actual cases, peer-reviewed medical literature, medical professional analysis, clinical criteria sets, and patient selection protocols (67 FR 66740). Our clinicians thoroughly reviewed all information submitted within the context of the established criteria and if, following this review, we determined that there was sufficient evidence to confirm that the medical procedure represented by the code could be safely and appropriately performed on an outpatient basis, we assigned the service to an APC and included it as a payable procedure under the OPPS (67 FR 66740). We determined the APC assignment for services removed from the IPO list by evaluating the clinical similarity and resource costs of the service compared to other services paid under the OPPS and reviewing the Medicare Severity Diagnosis Related Groups (MS-DRG) rate for the service under the IPPS. It should be noted, however, that we would generally expect the cost to provide a service in the outpatient setting to be less than the cost to provide the service in the inpatient setting (67 FR 66740).
As we have stated in prior rulemaking, over time, given advances in technology and surgical technique, we will continue to evaluate services to determine whether they should be removed from the IPO list. We have made it clear that, insofar as advances in medical practice mitigate concerns about these procedures being performed on an outpatient basis, we are prepared to remove procedures from the IPO list and provide for payment for them under the OPPS (65 FR 18443).
C. CY 2026 Changes to IPO List
1. CY 2026 Proposal To Eliminate the IPO List
Since the IPO list was established in 2000, it has been our policy that, regardless of how a procedure is classified for the purposes of payment, we expect in every case the surgeon and the hospital will assess the risk of a procedure or service to the individual patient, taking site of service into account, and will act in that patient's best interests (65 FR 18456). We have reiterated this expectation in rulemaking over the years, including in our discussion of the removal of total knee arthroplasty (TKA) from the IPO list in the CY 2018 OPPS/ASC final rule with comment period, total hip arthroplasty (THA) from the IPO list in the CY 2020 OPPS/ASC final rule with comment period, and lumbar spine fusion, shoulder joint reconstruction, and ankle reconstruction in CY 2021 (82 FR 59383; 84 FR 61354; 85 FR 86093). In those rules, we stated that the decision regarding the most appropriate care setting for a given surgical procedure is a complex medical judgment made by the physician based on the beneficiary's individual clinical needs and preferences and on the general coverage rules requiring that any procedure be reasonable and necessary.
Over the course of the years since the establishment of the IPO list, we have received comments from some interested parties who believe that we should eliminate the IPO list entirely and, instead, defer to the clinical judgment of physicians for decisions regarding site of service. For example, in the CY 2000 final rule with comment period, in response to the establishment of the IPO list, certain commenters stated that they believed CMS was ( printed page 53782) making decisions, such as the appropriate site of service for a particular medical procedure, that should be left to the discretion of surgeons and their patients (65 FR 18442 and 18455). In its 2001 and 2002 public meetings, the Advisory Panel on APC Groups supported eliminating the IPO list (67 FR 66722). We refer readers to the CY 2021 OPPS/ASC final rule with comment period for additional discussion of the opposition to the IPO list, including its lack of deference to physician judgment, its adverse effect on advances in surgical care, and the expectation it can create that non-IPO services must be furnished in the outpatient setting (85 FR 86084 through 86089).
Other interested parties have supported maintaining the IPO list and consider it an important tool to indicate which services are appropriate to furnish in the outpatient setting and to ensure that Medicare beneficiaries receive quality care. They have stated that many of the procedures that we currently designate as “inpatient only” are currently performed appropriately and safely only in the inpatient setting (65 FR 18442). We refer readers to the CY 2022 OPPS/ASC final rule with comment period for a summary of recent commenter concerns related to patient safety and quality of care in the absence of the IPO list (86 FR 63674).
Interested parties have also supported the use of the IPO list because services included on the IPO list are an exception to the 2-midnight rule and, as such, are considered appropriate for payment under Medicare Part A, regardless of the expected length of stay. As a result, many procedures are not subject to medical review by Medicare review contractors for “patient status” (that is, site-of-service). We note that, in the CY 2020 OPPS/ASC final rule with comment period, we finalized a policy to exempt procedures that have been removed from the IPO list from certain medical review activities for 2 calendar years following their removal from the IPO list. In the CY 2021 OPPS/ASC final rule with comment period, we finalized a policy to indefinitely exempt such procedures from those medical review activities while the IPO list was eliminated over 3 years.
For CY 2026 and subsequent years, we proposed to eliminate the IPO list through a 3-year transition, completing the elimination by January 1, 2029. While we agreed with commenters in previous rulemakings that the IPO list was necessary, and that it would be inappropriate for us to establish payment rates for those services under the OPPS (78 FR 75055, 86 FR 63673), we have reconsidered the various comments from interested parties requesting that we eliminate the IPO list, and reevaluated the need for CMS to restrict payment for certain procedures in the hospital outpatient setting. As a result of that reconsideration, we no longer believe there is a need for the IPO list to identify services that require inpatient care. We agree with past commenters that the physician should use clinical knowledge and judgment, together with consideration of the beneficiary's specific needs, to determine whether a procedure can be performed appropriately in a hospital outpatient setting or whether inpatient care is required for the beneficiary, subject to the general coverage rules requiring that any procedure be reasonable and necessary. We believe that this change would ensure maximum availability of services to beneficiaries in the outpatient setting.
Although we decided to halt the elimination of the IPO list in the 2022 OPPS/ASC final rule with comment period, for the reasons we discussed in the CY 2026 OPPS/ASC proposed rule and later in this section, we have come to believe with greater certainty that, since the IPO list was established, there have been significant developments in the practice of medicine that have allowed numerous services to now be provided safely and effectively in the outpatient setting. We acknowledged in the CY 2000 OPPS/ASC final rule with comment period that we believed that emerging new technologies and innovative medical practice were blurring the difference between the need for inpatient care and the sufficiency of outpatient care for many services (65 FR 18456). We also stated in the CY 2001 OPPS/ASC interim final rule with comment period that, over time, given advances in technology and surgical technique, many of the procedures that were on the IPO list at the time may eventually be performed safely in a hospital outpatient setting and that we would continue to evaluate services to determine whether they should be removed from the IPO list (65 FR 67826). Specifically, we stated that, insofar as advances in medical practice mitigate concerns about these services being furnished on an outpatient basis, we would be prepared to remove them from the IPO list and provide for payment under the OPPS (65 FR 67826).
Over the course of the last 25 years, these expectations have been borne out. There have been many new technologies and advances in surgical techniques and surgical care protocols, including the use of minimally invasive surgical procedures such as laparoscopy, improved perioperative anesthesia, expedited rehabilitation protocols, as well as significant enhancements to postoperative processes such as improvements in pain management, that have reduced the inpatient length of stay and the need for postoperative care following a surgical service. In consideration of these advancements, we have removed certain services from the IPO list that were previously considered to require inpatient care, including musculoskeletal procedures such as TKA in CY 2018 (82 FR 59385), THA in CY 2020 (84 FR 61355), and lumbar spine fusion, shoulder joint reconstruction, and ankle reconstruction in CY 2021 (85 FR 86093).
Since we previously considered elimination of the IPO list in the CY 2021 OPPS/ASC rule final rule with comment period, there have also been other innovations in the practice of medicine; for example, innovations in infection control spurred by the COVID-19 PHE (87 FR 72194). During that time, CMS issued flexibilities in the furnishing of acute hospital services at different locations, including the patient's home. While this Acute Hospital at Home Initiative was originally spurred by the necessity of expanding hospital capacity, it has demonstrated an increased ability to deliver certain services outside of the traditional inpatient setting. These advances have heightened awareness of practices that can increase patient safety across provider types, ensuring that clinicians emphasize these considerations in the practice of medicine, including site of service decisions. As medical practice continues to develop, we believe that the difference between the need for inpatient care and the appropriateness of outpatient care will continue to be less and less distinct for many services. Therefore, we stated in the CY 2026 OPPS/ASC proposed rule that we believe that the IPO list is no longer necessary to identify services that require inpatient care.
In recent years, there have also been certain procedures which we have decided to remove from the IPO list multiple times. For example, we removed several maxillofacial procedures in CY 2023, after originally removing them from the IPO list in CY 2021 and adding them back in CY 2022 (87 FR 72009). This frequency of change in policy can cause an uncertain regulatory landscape in which hospitals and providers are unclear on the policy and whether or not certain procedures are paid for in the hospital outpatient setting, potentially impacting access to ( printed page 53783) care for beneficiaries. Eliminating the IPO list and the related annual review process would mitigate this issue, offering more regulatory certainty for Medicare beneficiaries and providers.
Enabling IPO list services to be delivered outside of the inpatient setting, when clinically appropriate, can also advance important goals related to access to care. In the past, we have noted longstanding concerns over the closures of rural hospitals, which has prompted other policy actions to maximize access to care in rural or underserved areas (87 FR 72160). The experience of the COVID-19 PHE has also highlighted the importance of assisting areas and populations that suffer from a lower supply of medical services, which we addressed with temporary flexibilities during the COVID-19 PHE. Allowing for a greater exercise of clinical judgment will increase the ability of hospitals to provide Medicare-reimbursed services on an outpatient basis when clinically appropriate, while preserving inpatient beds for individual patients who truly need to be admitted. This will increase the availability of such services and additionally provide facilities with greater experience and flexibility that can be particularly crucial during future public health emergencies that constrict the supply of medical care.
We acknowledge the seriousness of the concerns regarding patient safety and quality of care that various interested parties have expressed regarding removing procedures from the IPO list or eliminating the IPO list altogether. However, as stated in the CY 2026 OPPS/ASC proposed rule, we believe that the evolving nature of the practice of medicine has mitigated and continues to mitigate patient safety and quality of care risks. That allows more procedures to be performed on an outpatient basis with a shorter recovery time. This trend, combined with physician judgment, State and local licensure requirements, accreditation requirements, hospital conditions of participation (CoPs), medical malpractice laws, and CMS quality and monitoring initiatives and programs, will continue to ensure the safety of beneficiaries in both the inpatient and outpatient settings, even in the absence of the IPO list. As mentioned previously in this section, we have consistently believed that it is important for physicians to exercise their clinical expertise based on the circumstances of individual patients and in light of these protections. We refer readers to the CY 2021 OPPS/ASC final rule with comment period for a full discussion of how these factors provide extensive safeguards for patients receiving services from Medicare enrolled providers, including hospital CoPs in 42 CFR part 482 (such as the requirement at § 482.30 that hospitals conduct a utilization review on medical necessity of admission, length of stay, and services rendered, and the most efficient use of available health facilities and services) (85 FR 48910).
2. CY 2026 Proposal To Use a 3-Year Transition To Eliminate the IPO List
We proposed to eliminate the IPO list over a 3-year transition period, beginning in CY 2026. We also proposed eliminating the criteria for removing procedures from the IPO list currently codified at § 419.23, as a conforming change.
Given the significant number of services on the list and that we would establish new reimbursement rates for those services under the OPPS, we recognize that interested parties may need time to adjust to the removal of procedures from the list. Providers may need time to prepare to furnish newly removed procedures on an outpatient basis, update their billing systems, and gain experience with newly removed procedures eligible to be paid under either the IPPS or OPPS. Therefore, we proposed to transition services off the IPO list over a 3-year period, with the list completely eliminated by CY 2029. In accordance with this proposal, we proposed to amend § 419.22(n) to state that effective beginning on January 1, 2026, the Secretary shall eliminate the list of services and procedures designated as requiring inpatient care through a 3-year transition, with the full list eliminated in its entirety by January 1, 2029.
For CY 2026, we proposed that musculoskeletal services would be the first group of services that would be removed from the IPO list, as we had done in the CY 2021 OPPS/ASC final rule with comment period. We stated in the CY 2026 OPPS/APS proposed rule that we believe it is appropriate to remove this group of services first for several reasons. In recent years, due to new technologies and advances in surgical care protocols, expedited rehabilitation protocols, improved infection control practices, and significant enhancements to postoperative processes, we have removed TKA and THA, both musculoskeletal services, from the IPO list. During the COVID-19 PHE, there was an accelerated decrease in short-length inpatient stays associated with musculoskeletal procedures—a decrease which was about four times faster than before the COVID-19 PHE (14.5 percent from 2020 to 2021). The number of Medicare ASCs specializing in orthopedic or musculoskeletal services also roughly doubled between 2016 and 2021. These trends suggest a shift in musculoskeletal services from inpatient to outpatient settings.[130]
Furthermore, during the notice and comment process of removing TKA and THA from the IPO list, interested parties continually requested that CMS remove other musculoskeletal services from the IPO list as well, citing shortened length of stay times, advancements in technologies and surgical techniques, and improved postoperative processes. Additionally, we note that, more often than not, interested parties' historical requests for removals of medical procedures from the IPO list were for musculoskeletal services. Further, there is already a set of C-APCs for musculoskeletal services for payment in the outpatient setting, which facilitates the removal of these types of services from the IPO list for CY 2026. Specifically, because we have previously removed codes corresponding to musculoskeletal services from the IPO list that are similar clinically and in terms of resource cost and assigned them to these C-APCs, these APCs generally describe appropriate ranges and placements for these musculoskeletal codes proposed for removal in CY 2026, which will allow for appropriate payment. As discussed in section III.E.3. of the CY 2026 OPPS/ASC proposed rule, we also proposed to establish a 7 level Musculoskeletal Procedures APC series, which will allow for the assignment of musculoskeletal procedures removed from the IPO list to an APC with an applicable range of estimated costs. We had previously finalized the removal of 266 musculoskeletal procedures from the IPO list in the 2021 OPPS/ASC final rule with comment period. Although we largely reversed this action in the 2022 OPPS/ASC final rule based on our halting of the elimination of the IPO list and re-evaluation of our removal criteria at the time, we maintained the removal of seven musculoskeletal procedures, and their related anesthesia services, from the IPO list. In the CY 2023 OPPS/ASC final rule with comment period, we removed 11 more musculoskeletal services from the IPO list. As we explained in the CY 2026 OPPS/ASC proposed rule, we now believe the entire IPO list should be eliminated over a 3-year transition period. Our previous consideration of removing ( printed page 53784) musculoskeletal procedures from the IPO list, and the continued removal of such procedures from it, suggests that we should begin the elimination and transition from the IPO list with the removal of these procedures from the list.
For CY 2026, we identified 285 mostly musculoskeletal services that we proposed to remove from the IPO list, including 16 non-musculoskeletal services that were recommended by the 2020 HOP Panel and removed from the IPO list in CY 2021 (85 FR 86089 through 86092). These 16 services, which include cardiovascular, lymphatic, digestive, gynecological, and endovascular procedures, were added back to the IPO list when the elimination of the IPO list was halted in CY 2022. The 285 services that we proposed to remove from the IPO list for CY 2026 and subsequent years, including the CPT/HCPCS codes, long descriptors, and the proposed CY 2026 payment indicators, are included in Table 69 of the CY 2026 OPPS/ASC proposed rule. These services and their proposed status indicators and APC assignments (if applicable) are included in Addendum B of the CY 2026 OPPS/ASC proposed rule. The complete list of codes that describe services that were proposed to be paid by Medicare in CY 2026 as inpatient only services is included as Addendum E to the CY 2026 OPPS/ASC proposed rule, which is available on the CMS website.[131]
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: Many commenters supported our proposal to eliminate the IPO list and defer to physicians' judgment on site of service determinations. These commenters stated that CMS' proposal would provide patients with greater flexibility to receive affordable care and allows for more patient-centered care. The commenters also believed the proposed change could potentially decrease overall healthcare costs and improve clinical outcomes for patients. Some commenters stated that there is no clinical difference between a surgery performed in an inpatient setting and an outpatient setting, and that eliminating the IPO list would create more flexibility for physicians and beneficiaries. These commenters also stated that there were already safeguards in place to ensure patient safety, such as State and local regulations, hospital conditions of participation (CoPs), and quality and monitoring initiatives. We also received comments in support of the removal of several specific codes from the IPO list for CY 2026, stating that these procedures can be or are already being performed safely in the outpatient setting.
Response: We thank the commenters for their support.
Comment: Numerous commenters, including hospital associations and systems, opposed the elimination of the IPO list due to patient safety concerns, stating that the IPO list serves as an important programmatic safeguard. These commenters stated that high-risk, invasive procedures that require post-operative monitoring that are currently included on the IPO list would not be safe to perform on Medicare beneficiaries in the outpatient setting. Additionally, commenters stated that there are several procedures on the IPO list that could never be performed in the outpatient setting, due to the complexity or need for postoperative inpatient care. Commenters were also concerned that we were removing services from the IPO list without data or literature to support that these could be safely performed in the outpatient setting and that there was not an evidence-based review process to determine which procedures to remove from the IPO list. These commenters requested that CMS maintain its current process and criteria for evaluating and removing procedures from the IPO list through rulemaking.
Response: We thank the commenters for their input. As we previously stated in the CY 2021 OPPS/ASC final rule with comment period (85 FR 86087), we continue to believe that physicians can and should use their clinical knowledge and judgment to appropriately determine whether a procedure can be performed in a hospital outpatient setting or whether inpatient care is required for the beneficiary based on the beneficiary's specific needs and preferences, subject to the general coverage rules requiring that any procedure be reasonable and necessary, and that payment should be made pursuant to the otherwise applicable payment policies. We believe that patient safety and quality of care will be safeguarded by the physician's assessment of the risk of a procedure or service to the individual beneficiary and their selection of the most appropriate setting of care based on this risk, in addition to State and local licensure requirements, accreditation requirements, hospital conditions of participation (CoPs), medical malpractice laws, and CMS quality and monitoring initiatives and programs. In addition, as we have stated in previous rulemaking (82 FR 59384; 84 FR 61354), the removal of a service from the IPO list does not require the service to be performed only on an outpatient basis. Rather, it allows for payment under the OPPS when the service is performed in an outpatient setting. Services that are removed from the IPO list can be and are performed on individuals who are admitted as inpatients (as well as individuals who are registered hospital outpatients), based on the physician's clinical knowledge and judgment. We also continue to believe that there have been significant developments in the practice of medicine that have allowed numerous services that were previously provided on an inpatient basis to now be provided safely and effectively in the outpatient setting. Therefore, at this time, we do not believe it is necessary for CMS to maintain an IPO list, nor do we currently believe that it is necessary to require specific HCPCS codes to remain payable only when furnished in the inpatient setting.
Comment: Some commenters were also concerned about the financial impact and administrative burden that this proposal would have on hospitals. Commenters had concerns that eliminating the IPO list could push healthier and less complex patients into outpatient settings, skewing the inpatient population to become a higher percentage of more complex patients that the existing MS-DRG reimbursement rates would not accurately cover. A few commenters also expressed concerns on the impact of this proposal on payment for Graduate Medical Education (GME) if inpatient stays and procedures decrease. Many commenters were also concerned with the potential burden of increased claim denials for inpatient claims for procedures that are no longer on the IPO list, along with potential breakdowns in the current administrative and pre-operative workflow for current inpatient-only procedures. Many commenters also shared concerns that physicians may be pressured by hospital administrations and commercial payors to perform formerly IPO procedures in lower cost settings, instead of prioritizing patient safety.
Response: As stated previously, we have repeatedly recognized that the decision regarding the most appropriate care setting for a given surgical procedure is a complex medical judgment made by the physician based on the beneficiary's individual clinical needs and on the general coverage rules ( printed page 53785) requiring that any procedure be reasonable and necessary. We continue to believe that deference should be given to physicians and medical professionals in these determinations. In accordance with section 1801 of the Act, we do not control or supervise the practice of medicine or the manner in which medical services are provided. We also reiterate that we do not require services that are no longer included on the IPO list to be performed solely in the outpatient setting and that, following elimination of the IPO list, services that were previously identified as inpatient only can continue to be performed in the inpatient setting. It is not CMS' policy to require services that are removed from the IPO list to be performed only in the outpatient setting. Instead, we aim to offer providers enhanced flexibility and choice in determining the safest, most efficient setting of care for Medicare beneficiaries, whether that is the inpatient or outpatient setting. It would be a misinterpretation of CMS payment policy for providers to create policies or guidelines that establish the outpatient setting as the baseline or default site of service for a procedure based on its removal from the IPO list or the elimination of the IPO list. As stated in previous rulemaking (84 FR 61354; 82 FR 59384; 81 FR 79697), services that are no longer included on the IPO list are payable in either the inpatient or outpatient setting subject to the general coverage rules requiring that any procedure be reasonable and necessary, and payment should be made pursuant to the otherwise applicable payment policies.
We believe there are sufficient guardrails, including State and local regulations, hospital CoPs, accreditation requirements, and medical malpractice laws, to ensure that physicians are able to prioritize patient safety when determining the site of service. In regard to concerns about increased administrative burden due to inpatient claims denials, as discussed below and in section X.D. of this final rule with comment period, we are continuing to exempt procedures that have been removed from the IPO list from certain medical review activities to assess compliance with the 2-midnight rule until the Secretary determines that the service or procedure is more commonly performed in the Medicare population in the outpatient setting. Regarding changes made by commercial insurance providers and site selection for outpatient services as a result of CMS eliminating the IPO list, while we believe that these comments are outside the scope of the OPPS/ASC proposed rule, we note that commercial providers establish their own rules regarding payment for services.
Comment: Multiple commenters requested, if CMS finalizes the proposal to eliminate the IPO list, that CMS provides guidance on the patient selection criteria, including clinical and social factors, for determining the appropriate site of service for procedures removed from the IPO list. Commenters also recommended that CMS monitor the safety impacts and surgical outcomes of moving procedures off the IPO list, including surgical complications and hospital readmissions.
Response: We thank the commenters for their feedback. As we have previously stated (86 FR 63675), we note the balance between several factors on this important issue, namely, the prohibition on CMS interfering with the practice of medicine in Section 1801 of the Act, the need to provide clear information about CMS billing and payment rules that ensure hospitals, physicians, and other interested parties can understand and operate within them, and our belief that the specific decision about the most appropriate care setting for a given surgical procedure is a complex medical judgment made by the physician based on the beneficiary's individual clinical needs and preferences and on the general coverage rules requiring that any procedure be reasonable and necessary.
We also thank the commenters for their recommendations on monitoring safety data related to the elimination of the IPO list. We would welcome and use any patient outcomes data, analyses, or recommendations that interested parties would like to share with us to help inform IPO decision-making going forward. We will take these recommendations into consideration for future rulemaking and are open to receiving any relevant data in the interim.
Comment: We received comments requesting that we assign services newly removed from the IPO list to New Technology APCs until sufficient data is collected to assign these services to clinical APCs. One commenter suggested that we make an appropriate New Technology APC assignment for their technology based upon inpatient charges converted to cost and then maintain the APC assignment for at least two years. We also received comments with alternative methodologies for making APC assignment determinations, including crosswalking procedures to ICD-10-PCS codes and using IPPS claims with shorts stays to calculate payment rates.
Response: We thank the commenters for their input. As we previously stated in the CY 2021 OPPS/ASC final rule with comment period (85 FR 86093), consistent with our regulation at 42 CFR 419.31(a)(1), we classify outpatient services and procedures that are comparable clinically and in terms of resource use into APC groups. As we stated in the CY 2012 OPPS/ASC final rule with comment period (76 FR 74224), the OPPS is a prospective payment system that provides payment for groups of services that share clinical and resource use characteristics. It should be noted that for all codes newly paid under the OPPS, including codes removed from the IPO list, our policy has been to assign the service or procedure to an APC based on feedback from a variety of sources, including but not limited to, review of the clinical similarity of the service to existing procedures; advice from CMS medical advisors; information from interested specialty societies; and review of all other information available to us, including information provided to us by the public, whether through meetings with interested parties or additional information that is mailed or otherwise communicated to us (84 FR 61229). Therefore, we believe assigning procedures removed from the IPO list to existing clinical APCs that are similar in clinical characteristics and resource costs is appropriate. We note that procedures assigned to New Technology APCs cannot be placed in clinical APCs due to insufficient clinical and cost data, unlike the procedures that are transitioning from the IPO list.
While we disagree with assigning these procedures to New Technology APCs at this time, we will take commenters' suggestions regarding alternative methodologies for assigning former IPO procedures to clinical APCs under consideration for future rulemaking.
Comment: Commenters disagree with certain proposed APC assignments for procedures proposed to be removed from the IPO list and stated that CMS did not provide sufficient detail as to how the proposed APC placements were determined. Some commenters also believed that the proposed APC payments did not adequately reflect the resource costs associated with providing the procedure in the outpatient setting. We received the following APC reassignment requests:
- Reassign CPT codes 0202T, 22610, 22800, 22802, 22808, and 27703 to APC 5117 (Level 7 Musculoskeletal Procedures). ( printed page 53786)
- Reassign CPT codes 23474, 27132, 27134, 27137, 27138, 27450, 27486, 27487 to APC 5116 (Level 6 Musculoskeletal Procedures).
- Reassign CPT codes 23334, 23335, 27236, 27244, 27245, 27248, 27254, 27269, 27450, 27506, 27507, 27511, 27513, 27514, 27535, 27536, and 27540 to APC 5115 (Level 5 Musculoskeletal Procedures).
- Reassign CPT codes G0413, G0414, and G0415 to APC 5114 (Level 4 Musculoskeletal Procedures) or higher.
- Reassign CPT code 61624 to APC 1577 (New Technology—Level 40 ($20,001-$25,000)).
- Reassign CPT code 37182 to APC 5194 (Level 4 Endovascular Procedures) or 1576 (New Technology—Level 39 ($15,001-$20,000)).
Response: As we have previously stated, we determine the APC assignment for services removed from the IPO list by evaluating the clinical similarity and resource costs of the service compared to other services paid under the OPPS and by reviewing the MS-DRG rate for the service under the IPPS, though we note we would generally expect the cost to provide a service in the outpatient setting to be less than the cost to provide the service in the inpatient setting. After further clinical review and review of the limited available claims data for these procedures, we believe that we have appropriately assigned the codes in the APC reassignment requests listed above, with the exception of the following updates:
- We are reassigning CPT codes 23334 and 23335 from APC 5073 (Level 3 Excision/Biopsy/Incision and Drainage) to APC 5114 (Level 4 Musculoskeletal Procedures).
- We are reassigning CPT code 27254 from APC 5113 (Level 3 Musculoskeletal Procedures) to APC 5114 (Level 4 Musculoskeletal Procedures).
- We are reassigning CPT code 27487 from APC 5115 (Level 5 Musculoskeletal Procedures) to APC 5116 (Level 6 Musculoskeletal Procedures).
- We are reassigning CPT codes 27511, 27513, and 27514 from APC 5114 (Level 4 Musculoskeletal Procedures) to APC 5115 (Level 5 Musculoskeletal Procedures).
- We are reassigning CPT code 37182 from APC 5193 (Level 3 Endovascular Procedures) to APC 5194 (Level 4 Endovascular Procedures).
Final APC assignments, status indicator assignments, and long descriptors for all procedures removed from the IPO list for CY 2026 can be found in Table 119.
Comment: Numerous commenters raised concerns about the impact of the elimination of the IPO list on the 3-day stay requirement for skilled nursing facility (SNF) care. By statute, beneficiaries must have a prior inpatient hospital stay of no fewer than three consecutive days to be eligible for Medicare coverage of inpatient SNF care. Commenters expressed concerns that the elimination of the IPO list may have a significant impact on Medicare beneficiaries' ability to satisfy the 3-day inpatient stay requirement to qualify for SNF care.
Response: While we believe that these comments are outside the scope of the OPPS/ASC proposed rule, we reiterate that removal of procedures from the IPO list does not require the procedures to be performed only on an outpatient basis. Removal of procedures from the IPO list allows for payment of the procedure in either the inpatient setting or the outpatient setting. A prior 3-day inpatient hospital stay remains a statutory requirement for SNF coverage. As stated in the CY 2018 final rule with comment period (82 FR 59384), in our discussion of the removal of TKA from the IPO list, we stated that we would expect that those Medicare beneficiaries identified as appropriate candidates to receive a surgical procedure in the outpatient setting would not be expected to require SNF care following surgery. Instead, we expect that these beneficiaries would be appropriate for discharge to home (with outpatient therapy) or home health care. Therefore, we do not anticipate that Medicare beneficiaries receiving the procedures we are proposing to remove from the IPO list for CY 2026 in the outpatient setting would require SNF care following the procedure.
Comment: We received comments regarding the potential impact of eliminating the IPO list on our transitional device pass-through policy. One commenter requested clarification on if devices associated with procedures removed from the IPO list would be eligible for device pass-through status. Other commenters requested that CMS reinstate certain expired device category codes, arguing that associated procedures being on the IPO list created a “verifiable delay in U.S. market availability” for these devices as referenced in § 419.66(b)(1) for the outpatient hospital setting.
Response: We thank the commenters for their feedback. Devices that are reported only with codes that are removed from the IPO list will not be precluded from applying for or receiving transitional device pass-through payment status. However, we note that all devices must meet the eligibility criteria described in § 419.66 to be eligible for transitional device pass-through payment status, including the requirement that the pass-through payment application must be submitted within 3 years from the date of the initial FDA marketing authorization. We do not believe that an IPO designation for procedures associated with devices creates a “verifiable delay in U.S. market availability” for these devices as referenced in § 419.66(b)(1), because these devices have been available on the market, even if the associated procedures have not been performed, or the associated procedures are performed in low volumes in the outpatient setting. We do not believe the elimination of the IPO list warrants changes to our device pass-through policy. We also do not agree with commenters' suggestions to reinstate device pass-through payment status for certain device codes due to procedures being removed from the IPO list because. consistent with section 1833(t)(6)(B)(iii) of the Act and 42 CFR 419.66(g), the period for which a device category for transitional pass-through payments under the OPPS can be in effect is at least 2 years, but not more than 3 years, beginning on the first date on which pass-through payment is made. Once 3 years has passed since a device category first received transitional pass-through payments, the device category is no longer eligible for pass-through payments, and we utilize the established policy (67 FR 66763) to package the costs of the devices that are no longer eligible for pass-through payments into the costs of the procedures with which the devices are reported in the claims data used to set the payment rates. We note that device pass-through payment status is intended to be temporary, and we consider the cost data to be included in the payment rates regardless of whether the technology's use in the Medicare population has been frequent or infrequent during the time period under which a device was receiving transitional pass-through payments. We cannot reinstate the pass-through payment status of expired device category codes where reinstatement would make the pass-through payment status effective longer than the maximum 3-year period permitted under section 1833(t)(6)(B)(iii) of the Act and § 419.66(g).
3. Effect on Beneficiary Cost-Sharing
As noted in the CY 2021 OPPS/ASC final rule with comment period, some interested parties have shared concerns with us that removing procedures from the IPO list and allowing them to be ( printed page 53787) paid under the OPPS when performed in the outpatient setting may result in an increased financial burden for beneficiaries for certain complex services (85 FR 86086). Under current law, the OPPS cost-sharing for a service is capped at the applicable Part A hospital inpatient deductible amount for that year for each service. This cap applies to individual services, and some commenters have expressed concern in the past that if a Medicare beneficiary receives multiple separately payable OPPS services, it is possible that the aggregate cost-sharing for a beneficiary may be higher for services provided in the outpatient setting than it would be had the services been furnished during an inpatient stay. However, as we stated in the CY 2026 OPPS/ASC proposed rule, we emphasize that services included on the IPO list tend to be surgical procedures that would typically be the focus of the hospital outpatient stay and would likely be assigned to a comprehensive APC (C-APC) when they are removed from the IPO list. As such, these services would likely be considered a single episode of care with one payment rate and one copayment amount, instead of multiple copayments for each individual service. In most instances, we expect that beneficiaries will not be responsible for multiple copayments for individual ancillary services removed from the IPO list since, because of their assignment to C-APCs, the inpatient deductible cap will apply to the entire hospital claim which is billed and paid as a comprehensive service or procedure. In the event there are separately payable OPPS services included on a claim with a service assigned to a C-APC, the policy that the OPPS cost-sharing for an individual service is capped at the applicable Part A hospital inpatient deductible amount for that year for each service remains applicable, which is that the OPPS cost-sharing for an individual service is capped at the applicable Part A hospital inpatient deductible amount for that year for each service. For further information regarding beneficiary copayments, please refer to section II.I. of this final rule with comment period.
Comment: We received one comment that expressed concern about the potential for increased beneficiary cost-sharing when complex procedures are performed in outpatient settings. The commenter stated that beneficiaries may still face unexpected financial burdens, particularly if multiple services are required or complications arise.
Response: As stated previously, services included on the IPO list tend to be surgical procedures that, if performed on an outpatient basis, would typically be the focus of the hospital outpatient stay and would likely be assigned to a comprehensive APC (C-APC) when they are removed from the IPO list. As such, these services would likely be considered a single episode of care with one payment rate and one copayment amount. In most instances, we expect that beneficiaries will not be responsible for multiple copayments for individual ancillary services associated with services removed from the IPO list, because the primary service will be assigned to a C-APC and the inpatient deductible cap will apply to the entire hospital claim, which is paid as a comprehensive service. The majority of procedures being removed from the IPO list for CY 2026 are assigned to C-APCs or packaged into payment for other services, which will result in beneficiaries paying one copayment amount. Therefore, we do not believe that beneficiaries will be significantly impacted through increased cost sharing for services that were removed from the IPO list and are furnished in the hospital outpatient department setting.
Also, as stated previously, in the event there are separately payable OPPS services included on a claim with a service assigned to a C-APC, OPPS cost-sharing for an individual service is capped at the applicable Part A hospital inpatient deductible amount for that year for each service.
4. Exemption From Certain Medical Review Activities for Services Removed From the IPO List
To further address concerns from interested parties, we proposed to continue to exempt procedures that have been removed from the IPO list from certain medical review activities to assess compliance with the 2-midnight rule until the Secretary determines that the service or procedure is more commonly performed in the Medicare population in the outpatient setting. Specifically, we proposed to continue the indefinite exemption from site-of-service claim denials, referrals to Recovery Audit Contractors (RACs), and RAC reviews for “patient status” for procedures that are removed from the IPO list under the OPPS beginning on January 1, 2021, as part of the transition away from the IPO list (85 FR 86120). Pursuant to this exemption, initial medical review contractors may continue to review claims for procedures previously on the IPO list to provide education for practitioners and providers regarding compliance with the 2-midnight rule, but will not deny claims identified as noncompliant with respect to the site-of-service under Medicare Part A. We proposed that this exemption will continue for all services or procedures removed from the IPO list until the Secretary determines that the exemption is no longer appropriate for each specific service or procedure because it is more commonly performed in the outpatient setting. We also sought comment on whether other exemption periods may be more warranted. For more information on the 2-midnight rule and comment responses, please refer to section X.D. of this final rule with comment period.
As stated in the CY 2026 OPPS/ASC proposed rule, although we believe it is important to pause certain medical review activities related to patient status to allow providers time to adjust to the proposed changes to the IPO list, we note that initial medical review contractors routinely address, and will continue to address, any beneficiary quality of care complaints that include concerns about treatment as a hospital inpatient or outpatient, not receiving expected services, early discharge, and discharge planning. CMS' case management system currently allows initial medical review contractors and CMS to monitor the frequency and status of beneficiary quality of care complaints and other beneficiary appeals by topic, provider type, and geographic area. These numbers are currently compiled by the BFCC-QIO national coordinating and oversight review contractor and reported to the QIOs and CMS leadership on a weekly basis for monitoring purposes. As previously noted, although we proposed to continue to indefinitely exempt procedures removed from the IPO list beginning on January 1, 2021, from site-of-service claim denials, referrals to RACs, and RAC reviews of “patient status,” medical review contractors would continue to conduct initial medical reviews concerning the medical necessity of both the services and the site of service, and will continue to be permitted and expected to deny claims if the service itself is determined not to be reasonable and medically necessary, as noted in the CY 2021 OPPS/ASC final rule with comment period (85 FR 86118). Therefore, given CMS' increasing ability to measure the safety of procedures performed in the outpatient setting and to monitor the quality of care, in addition to the other safeguards detailed previously in this section, we stated in the CY 2026 OPPS/ASC proposed rule that we now believe that quality of care is unlikely to be negatively affected by the elimination of the IPO list. However, we requested that commenters submit evidence on what effect, if any, they believe eliminating ( printed page 53788) the IPO list may have on the quality of care.
We received several comments on our proposal to continue to exempt procedures that have been removed from the IPO list from certain medical review activities to assess compliance with the 2-midnight rule until the Secretary determines that the service or procedure is more commonly performed in the Medicare population in the outpatient setting. Please refer to section X.D. of this final rule with comment period for a summary of these comments and our responses.
5. Comment Solicitation on Order of Removal of Additional Clinical Families From the IPO List During the Transition to Complete Elimination of the IPO List
As stated previously in this section, we proposed to eliminate the current IPO list of 1,731 services, starting with the 285 mostly musculoskeletal-related services as provided in Table 69 of the CY 2026 OPPS/ASC proposed rule. We requested comments from the public on whether 3 years is an appropriate time frame for the transition, whether there are other services that would be ideal candidates for removal from the IPO list in the near term, given known technological and other advances in care, and the order of removal of additional clinical families of services, and/or specific services, for each of the CY 2027 and CY 2028 rulemakings, until the IPO list is completely eliminated. Additionally, we sought comment on whether we should restructure or create any new APCs or C-APCs to allow for efficient OPPS payment for services that are removed from the IPO list. As discussed in section III.E.3. of the CY 2026 OPPS/ASC proposed rule, we proposed to establish a 7 level Musculoskeletal Procedures APC series for CY 2026.
Comment: A few commenters supported the 3-year transition period, stating that it would give hospitals and physicians sufficient time to adjust.
Response: We thank the commenters for their support.
Comment: A few commenters disagreed with our 3-year transition period. Some commenters recommended that we proceed with the elimination of the IPO list at a slower pace, with a few commenters recommending at least 4 years. One commenter recommended we proceed with eliminating the IPO list in total, stating that a transition over multiple years could cause confusion regarding which procedures have or have not been removed.
Response: We thank the commenters for their feedback. We continue to believe, as discussed previously, that gradually removing services from the IPO list over the 3-year phase-out period will allow for adequate time for providers to prepare to furnish newly removed procedures on an outpatient basis, update their billing systems, and gain experience with newly removed procedures eligible to be paid under either the IPPS or OPPS.
Comment: One commenter recommended that CMS remove CPT codes 58548 (Laparoscopy, surgical, with radical hysterectomy, with bilateral total pelvic lymphadenectomy and para-aortic lymph node sampling (biopsy), with removal of tube(s) and ovary(s), if performed) and 58575 (Laparoscopy, surgical, total hysterectomy for resection of malignancy (tumor debulking), with omentectomy including salpingo-oophorectomy, unilateral or bilateral, when performed) from the IPO list for CY 2026, stating that they are safe to perform in outpatient settings.
Response: We thank the commenter for their suggestion. We will take this into consideration in future rulemaking.
Comment: Some commenters suggested that we review procedures on the IPO list for procedures that have claims data that show a high volume of 1-2 day stays and remove those procedures first. One commenter requested that we wait to remove certain invasive procedures involving a craniectomy, craniotomy, and/or burr holes until the last phase. Another commenter recommended that we remove cardiovascular procedures during the last phase.
Response: We thank the commenters for their suggestions. We will take these into consideration in future rulemaking.
Comment: Several commenters expressed support for our proposal to create a Level 7 Musculoskeletal APC. A few of those commenters requested that we continue to review those APCs for cost and clinical homogeneity, and the need for additional APCs.
Response: We appreciate the commenters' support. After consideration of the public comments, we are finalizing our proposal to create a Level 7 Musculoskeletal APC. We note that we will continue to monitor the APC series as updated claims data continues to be available. The complete list of codes assigned to APC 5117 (Level 7 Musculoskeletal Procedures) can be found in Addendum B of this final rule with comment period, which is available on the CMS website.
6. Comment Solicitation on Changes to IPO List Removal Criteria
In addition to our proposal to eliminate the IPO list over a 3-year period, we proposed to eliminate the codified criteria for removing procedures from the IPO list at § 419.23. As mentioned previously in this section, we finalized the adoption of these longstanding criteria for removal procedures from the list in the CY 2022 OPPS/ASC final rule with comment period when we decided to halt the elimination of the IPO list (86 FR 63678). However, as we noted in the CY 2026 OPPS/ASC proposed rule, if we finalize our proposal to eliminate the IPO list in its entirety, there would no longer be any need to maintain a list of criteria for removing individual procedures from the list in any given year. However, we acknowledged that some commenters may disagree with our proposed approach to the IPO list. Therefore, we wished to consider other methods to provide greater deference to the medical judgment of clinicians besides eliminating the IPO list completely. We solicited comment on other approaches to provide greater flexibility in making site-of-service decisions, such as updating the list of criteria for removing procedures from the IPO list.
Comment: We received comments that requested, as an alternative to removing the five criteria at § 419.23(b), that CMS establish criteria that factor in clinical and social factors, such as comorbidities, age, and home support. Some commenters suggested updating the criteria to allow for a smaller version of the IPO list or to maintain a shortened list with just procedures that would never be performed in the outpatient setting, such as organ transplants. Some commenters suggested that instead of eliminating the IPO list, CMS instead provide a process to allow physicians to override the IPO designation in certain situations.
Response: We will take this into consideration in future rulemaking.
7. Summary of IPO List Changes
In summary, and after consideration of the comments received, given recent developments in surgical technique and technological advances in the practice of medicine, innovations in infection control and site of service shifts to the outpatient setting spurred by the COVID-19 PHE, as well as the various safeguards discussed previously in this section, we finalize eliminating the IPO list over the course of the next 3 years, starting with the removal of 285 mostly musculoskeletal-related services, as ( printed page 53789) provided in Table 119, for CY 2026. We finalize our proposal eliminating the criteria for removing procedures from the IPO list currently codified at § 419.23, as a conforming change. We are also finalizing amending § 419.22(n) to state that, effective on January 1, 2026, the Secretary shall eliminate the list of services and procedures designated as requiring inpatient care through a 3-year transition period, with the list eliminated in its entirety by January 1, 2028. We believe that there are a number of safety mechanisms that will continue to ensure the safety of our beneficiaries and the quality of care, including physician judgment, State and local regulations, accreditation requirements, medical malpractice laws, hospital conditions of participation, and other CMS initiatives.
The services removed from the IPO list for CY 2026 and their final status indicators and APC assignments (if applicable) are included in Addendum B of this final rule with comment period, which is available on the CMS website. The complete list of codes that describe services that are finalized to be paid by Medicare in CY 2026 as inpatient only services is included as Addendum E to this CY 2026 OPPS/ASC final rule with comment period, which is available on the CMS website.
( printed page 53790) ( printed page 53791) ( printed page 53792) ( printed page 53793) ( printed page 53794) ( printed page 53795) ( printed page 53796) ( printed page 53797) ( printed page 53798) ( printed page 53799) ( printed page 53800) ( printed page 53801) ( printed page 53802)X. Nonrecurring Policy Changes
A. Method To Control Unnecessary Increases in the Volume of Outpatient Services Furnished in Excepted Off-Campus Provider-Based Departments (PBDs)
1. Background
In the CY 2019 OPPS/ASC final rule with comment period (83 FR 59004 through 59014), we adopted a method to control unnecessary increases in the volume of clinic visit services furnished in excepted off-campus provider-based departments (PBDs). We refer readers to the CY 2019 OPPS/ASC final rule with comment period for a detailed discussion of the background, legislative provisions, and payment policies we developed to address increases in the volume of covered outpatient department (OPD) services. Below we discuss the policy we finalized in the CY 2019 OPPS/ASC final rule with comment period and its application under the OPPS for CY 2020 and subsequent years.
In the CY 2019 OPPS/ASC final rule with comment period, we finalized a policy to use our authority under section 1833(t)(2)(F) of the Act to adopt a method to control unnecessary increases in the volume of covered outpatient department services. We applied an amount equal to the site-specific Medicare Physician Fee Schedule (PFS) payment rate for nonexcepted items and services furnished by a nonexcepted off-campus PBD (the PFS payment rate) for the clinic visit service, as described by HCPCS code G0463, when provided at an off-campus PBD excepted from section 1833(t)(21) of the Act (departments that bill the modifier “PO” on claim lines). However, we phased in the application of the reduction in payment for the clinic visit service described by HCPCS code G0463 in the excepted provider-based department setting over 2 years. For CY 2019, the payment reduction was phased-in by applying 50 percent of the total reduction in payment that would have applied if these departments were paid the site-specific PFS rate for the clinic visit service. The PFS equivalent rate was 40 percent of the OPPS payment for CY 2019 (that is, 60 percent less than the OPPS rate). We provided for a 2-year phase-in of this policy under which one-half of the total 60 percent payment reduction (a 30 percent reduction) was applied in CY 2019. These departments were paid approximately 70 percent of the OPPS rate (100 percent of the OPPS rate minus the 30 percent payment reduction that was applied in CY 2019) for the clinic visit service in CY 2019.
For CY 2020, the second year of the 2-year phase-in, we stated that we would apply the total reduction in payment that is applied if these departments (departments that bill the modifier “PO” on claims lines) are paid the site specific PFS rate for the clinic visit service described by HCPCS code G0463. For CY 2020 and subsequent years, the PFS-equivalent rate was 40 percent of the proposed OPPS payment (that is, 60 percent less than the OPPS rate).
In addition, as we stated in the CY 2019 OPPS/ASC final rule with comment period (83 FR 58818), we implemented this policy in a non-budget neutral manner. We did so to ensure that our method for controlling the unnecessary growth in the volume of clinic visits furnished by excepted off-campus PBDs does not simply increase other unnecessary expenditures within the OPPS, thus driving different utilization-distorting decisions.
In the CY 2023 OPPS/ASC final rule with comment period (87 FR 71748), we finalized a policy which provided that off-campus PBDs (departments that bill the modifier “PO” on claim lines) of rural Sole Community Hospitals (SCHs), as described under 42 CFR 412.92 and designated as rural for Medicare payment purposes, are exempt from the clinic visit payment policy that applies a Physician Fee Schedule-equivalent payment rate for the clinic visit service, as described by HCPCS code G0463, when provided at an off-campus PBD excepted from section 1833(t)(21) of the Act. For the full discussion of this ( printed page 53803) policy, we refer readers to the CY 2023 OPPS/ASC final rule with comment period (87 FR 72047 through 72051). For CY 2024 and CY 2025, we continued to exempt excepted off-campus PBDs of rural SCHs from the clinic visit payment policy.
We noted in the CY 2026 OPPS/ASC proposed rule that we continued to believe that section 1833(t)(2)(F) of the Act provides authority to implement this policy. The U.S. Court of Appeals for the District of Columbia Circuit held in American Hospital Association v. Azar that a service-specific, non-budget-neutral reduction of the reimbursement rate for OPD services “qualifies as a `method for controlling unnecessary increases in the volume of covered [outpatient] services' ” under that provision. 964 F.3d 1230, 1245 (D.C. Cir. 2020) (quoting 42 U.S.C. 1395 l (t)(2)(F)). The D.C. Circuit reasoned in part that “[t]he lower the reimbursement rate for a service, the less the incentive to provide it, all else being equal[,]” and “[r]educing particular the reimbursement rate . . . is naturally suited to addressing unnecessary increases in the overall volume of a service provided by hospitals.” Id. at 1241. It ultimately concluded that the policy “falls comfortably within the plain text” of section 1833(t)(2)(F) of the Act, id. at 1241, “and `fits the design of the statute as a whole and its object and policy,' ” id. at 45 (quoting Good Samaritan Hosp. v. Shalala, 508 U.S. 402, 418 (1993)). We noted in the CY 2026 OPPS/ASC proposed rule that we continued to believe that our interpretation of the Act was the best one, and that this policy falls well within the Act's delegation to the Secretary to “develop a method for controlling unnecessary increases in the volume of covered OPD services”.
2. Expanding the Method To Control Unnecessary Increases in the Volume of Outpatient Services Furnished in Excepted Off-Campus Provider-Based Departments
As described in the CY 2019 OPPS/ASC final rule with comment period, we previously found that earlier rulemaking efforts were insufficient to control the unnecessary growth of certain covered OPD services and as a result we implemented a method to control for unnecessary growth in covered OPD services by adjusting the payment rate for clinic visits in excepted off-campus PBDs to be at the PFS-equivalent rate rather than the higher OPPS rate. While this regulatory change had a positive impact, we noted in CY 2026 OPPS/ASC proposed rule that there is evidence of continued growth in the volume of OPD services driven by site of service payment differentials rather than clinical need. We continued to be concerned that beneficiaries are being driven into a higher cost setting of care because of financial incentives when they could safely receive care in a lower cost setting. This creates greater financial burden both for Medicare and for the beneficiary in the form of increased coinsurance. Volume increases that seek to take advantage of financial incentives created by payment policy rather than clinical need are unnecessary and therefore warrant policy changes to halt and address these increases. As the D.C. Circuit explained, “[i]t is reasonable to think that Congress . . . would have wanted the agency to avoid causing unnecessary volume growth with its own reimbursement practices”. Am. Hosp. Ass'n, 964 F.3d at 1245. Accordingly, we proposed to remove this differential for drug administration services delivered in excepted PBDs.
Many healthcare services can be performed in multiple settings. Even when there is little variation in the service provided across settings, the Medicare Trust Fund and Medicare beneficiaries typically pay more when that service is performed in an OPD than when the same service is performed in a physician office. That payment differential creates an incentive for providers to shift the care of beneficiaries to an OPD rather than a physician office or ASC, even if the services can be safely performed in the physician office or an ASC. Generally, 20 percent of any increased payment is the responsibility of the beneficiary in the form of coinsurance. Taking into account that any payment differential occurs across millions of claims for drug administration and other services each year, this threatens to create a significant source of unnecessary spending both by Medicare beneficiaries in the form of unnecessarily high copayments and by Medicare in the form of unnecessarily high Medicare payments for services that can be performed safely in a different setting.
In the CY 2019 OPPS/ASC final rule with comment period, we discussed vertical consolidation and the practice of hospitals purchasing freestanding physician practices and converting the billing from the PFS to higher paying OPD visits. These conversions shift market share from freestanding physician offices to OPDs. We stated that we believed there was a correlation among the increasing volume of OPD clinic visits, vertical integration, and the higher OPPS payment rates for clinic visits. More favorable reimbursement for hospital-owned sites compared to physician-owned sites has been shown to encourage hospitals' acquisition of physician practices.[132 133] Once a practice is acquired and designated as an outpatient department, physician services can be billed at higher hospital-based rates. This type of consolidation has been associated with higher Medicare spending and more intense treatment patterns.[134 135 136] The impact of vertical integration and the increases in volume of outpatient services extends beyond just the clinic visit. In the CY 2019 OPPS/ASC final rule with comment period, we cited our concern that beneficiaries receiving chemotherapy administration, a high-volume service within the drug administration APC family, receive more sessions on average when treated in the OPD. Chemotherapy days per beneficiary were an estimated 9 to 12 percent higher in the hospital outpatient department than the physician office setting.[137] From 2003-2015 the rate of hospital or health system ownership of cancer care practices doubled from about 30 percent to about 60 percent.[138] For some drug administration services for cancer care, provider consolidation increases the cost of outpatient chemotherapy treatment.[139]
Our policy in the CY 2019 OPPS/ASC final rule with comment period to pay for clinic visits in excepted off-campus PBDs at the PFS-equivalent rate addressed the financial incentive for only one type of service in one outpatient setting. However, the share of other ambulatory services billed under the OPPS has continued to increase. For example, in its 2023 report, MedPAC stated that the share of chemotherapy services furnished in OPDs has grown from 35.2 percent in 2012 to 51.9 percent in 2021. HCPCS code 96413—which describes chemotherapy administration, intravenous infusion technique; up to 1 hour, single or initial substance/drug—is one of the most frequently billed drug administration codes in the OPPS. In 2025 this service ( printed page 53804) has a physician office payment rate of around $119 dollars and an OPPS payment rate of approximately $341, making the same chemotherapy infusion service almost three times more expensive in the OPD than in the physician office. Similarly, between 2012 and 2021, the OPD share of nuclear cardiography services has grown from 33.9 percent to 47.6 percent, and the OPD share of echocardiography services has grown from 31.6 percent to 43.1 percent.[140]
We are not aware of any clinical or other substantive change in the services provided that would have led to these increases in the share of these services furnished in OPDs, as opposed to in other settings. Thus, we noted in the CY 2026 OPPS/ASC proposed rule that we believed that these changes are in large part a result of financial incentives and therefore represent unnecessary increases in the volume of OPD services.
We stated in the CY 2026 OPPS/ASC proposed rule that we believe that financial incentives have driven volume from the office setting to the higher paying OPD setting, creating unnecessary increases in the volume of OPD services. We also stated that we believe that this problem is pervasive and exists across a number of services. Any time a service is provided in the higher cost OPD when it could be provided safely in the physician office, but it is not because of financial incentives, that potentially represents unnecessary utilization of the OPD setting. In CY 2019, we chose to start tackling this problem by addressing the clinic visit when provided in excepted PBDs. In that case, it was practical to address only a single code, G0463, the clinic visit. For CY 2026, we proposed to address drug administration services provided at excepted PBDs a bit more generally. We proposed to address payment for these services across the APC family, as we believe this volume control method should apply to all drug administration services at excepted PBDs.
Our authority under section 1833(t)(2)(F) of the Act to adopt a method to control unnecessary increases in the volume of covered outpatient department services authorizes us to address the consequences of these payment inequalities. Given these continued disparities, we proposed in the CY 2026 OPPS/ASC to further examine and refine our volume control method by identifying additional covered OPD services at high risk of unnecessarily shifting to the hospital setting based on financial incentives rather than medical necessity. We analyzed several families of services paid under the OPPS and presented our findings on the utilization and payment of drug administration services, which we discuss in the later in this section.
3. Utilization of Drug Administration Services
The high volume of drug administration services and the fact that Medicare pays approximately 200-300 percent more for the administration of drugs in the OPD setting than in the physician office setting incentivizes providers to migrate this family of services to the OPD setting.
Drug administration includes the intravenous or intramuscular administration of a range of medicines. Drug administration can be performed in either physician offices or OPDs. The process of administering a drug does not meaningfully differ between a physician office or OPD. In the OPPS, drug administration is categorized into four levels of complexity. Payments are set at a category level, called an Ambulatory Payment Classification (APC). The APCs for drug administration are 5691, 5692, 5693, and 5694. Currently, 61 Healthcare Common Procedure Coding System (HCPCS) codes make up the four drug administration APCs. HCPCS codes that are similar in terms of cost and clinical attributes are placed in the same APC. All HCPCS codes in the same APC have the same OPPS payment rate. The individual HCPCS and APC assignments are available in Addendum B to this final rule with comment period.
We evaluated the growth in volume and spending for multiple families of APCs in OPDs across multiple years of claims data. Should commenters wish to replicate any of our analyses, the CMS website includes information about obtaining the “Limited Data Set,” https://www.cms.gov/data-research/files-for-order/data-disclosures-and-data-use-agreements-duas/limited-data-set-lds through which OPPS claims data is available for purchase. We found that the volume of services paid through the drug administration APCs (5691-5694) has increased over time, which would indicate that these services have migrated to the OPD setting. Specifically, from 2011 to 2019 the volume of drug administration services paid under these APCs grew by almost 35 percent. This growth persisted even with the introduction of the PFS-equivalent rate for PBDs subject to section 603 of the Bipartisan Budget Act of 2015 (BBA) starting in 2017. The growth also persisted after the COVID-19 Public Health Emergency (PHE). Between 2018 and 2024 the number of beneficiaries enrolled in fee-for-service Medicare decreased by over 14 percent.[141] But since 2020 we have simultaneously seen increases in the volume of drug administration services provided in OPDs utilized per beneficiary.[142] Between 2020 and 2023 the utilization of drug administration services grew per beneficiary by over 30 percent. That means that while there are now fewer Medicare fee-for-service beneficiaries than there were prior to the COVID-19 PHE, each beneficiary on average receives more drug administration services in the OPD setting than they were prior to the COVID-19 PHE. There was also growth prior to the COVID-19 PHE. The utilization of drug administration services per beneficiary also grew by 30 percent between 2011 and 2019.
In addition to looking at the growth in volume and spending at the APC level, we looked at the growth in volume at the HCPCS code-level and found that some HCPCS codes within the drug administration APCs have experienced particularly significant growth. As we stated earlier, HCPCS code 96413—which describes chemotherapy administration, intravenous infusion technique; up to 1 hour, single or initial substance/drug—is the most frequently billed HCPCS code within any of the drug administration APCs at excepted PBDs. This code has seen an almost 70 percent increase in volume from 2011 to 2023.[143] In 2025 this service has a physician office payment rate of around $119 dollars and an OPPS payment rate of approximately $341. That makes the same chemotherapy infusion service 186 percent more expensive in the OPD than in the physician office. We concluded that this 70 percent increase in excepted hospital outpatient department volume over a 10-year period was at least partially driven by the payment differential between the physician office and OPD setting. The HCPCS codes representing chemotherapy administration grew more generally in volume by 64 percent in the OPPS between 2011 and 2023.[144] The chemotherapy administration codes ( printed page 53805) represent some of the highest-cost and most-frequently billed services within the drug administration APCs. MedPAC found that from 2015 to 2021, the volume of chemotherapy administration in freestanding clinician offices, the ambulatory setting for which payment rates are usually lowest, fell 14.2 percent.[145] We concluded that if there was not a difference in payment rates, fewer of these services would have shifted to the hospital outpatient setting and the corresponding increase in Medicare payments and beneficiary cost-sharing would not have occurred.
We were also concerned about beneficiaries who pay higher cost sharing because of the payment incentives driving them to OPDs. Drug administration services are skewed toward a small portion of the population with high utilization. Cancer patients receiving chemotherapy are among the highest utilizers of these services. The administration of chemotherapy highlights that a small portion of the population is disproportionately harmed by the current state of drug administration payment in the OPPS. A meaningful number of beneficiaries in this cohort are paying substantially more per year in cost sharing than they would had they received the same treatments at freestanding facilities or non-excepted off-campus PBDs.[146] Focusing on the cost sharing of chemotherapy patients demonstrates how this cohort is disproportionately impacted by the current payment structure and is uniquely positioned to benefit from an appropriate application of our authority to control for unnecessary increases in the volume of OPD services. Indeed, one study found that “in 2021, approximately 74,000 Medicare FFS chemotherapy patients utilized excepted off-campus OPDs and would have had cost sharing expenses that were $292 lower per patient had site neutrality applied. For the highest utilizing 5,000 patients who received chemotherapy most frequently at excepted off-campus OPDs, cost sharing would have been $1,055 lower per patient if payments had been site neutral”.[147]
While there have been increases in the volume of drug administration services in the hospital outpatient setting in recent years, drug administration services are still frequently provided in freestanding facilities. One study found that in 2023, 68 percent of drug administration services occurred in physician offices, indicating that they are (and can be) safely performed in multiple settings.[148] In its 2023 report, MedPAC examined APCs for which it might be appropriate to make a site neutral payment. To identify appropriate APCs they compared the volume of services in each APC that was provided in OPDs, ASCs, and freestanding offices over the period of 2016 through 2021, but omitted 2020 because the coronavirus pandemic affected the volume of care in ambulatory settings. If freestanding offices had the highest volume for an APC, they concluded that the services in that APC could be provided safely in freestanding offices for most beneficiaries and that beneficiaries would be able to access the services in that APC. Therefore, for those services, it would be reasonable to align the OPPS payment rates with the PFS payment rates. MedPAC found that all four of the drug administration APCs had higher volume in freestanding facilities than in OPDs, indicating that these services can be safely provided to beneficiaries in a lower cost setting of care. We believed MedPAC's analysis aligns well with the rationale CMS adopted in the CY 2019 OPPS/ASC final rule with comment period: we consider OPPS utilization unnecessary if the beneficiary can safely receive the same services in a lower cost setting but instead receives care in the hospital outpatient setting because of payment differentials that result in the unnecessary consumption of OPD services.
In our review of the utilization of drug administration services in excepted PBDs we found increases in the volume of services over time, increases in the volume of services provided per beneficiary, and cases of significant volume growth for some individual HCPCS codes within the drug administration APC family. We believed that these changes represent unnecessary increases in the volume of covered outpatient department drug administration services and that it would be appropriate to apply our volume control method to these services.
4. Payment for Drug Administration Services at PBDs
As discussed in the CY 2017 OPPS/ASC final rule with comment period (81 FR 33648), we established a PFS relativity adjuster that is applied to the OPPS rate for the billed non-excepted items and services furnished in a non-excepted off-campus PBD to calculate payment rates under the PFS. The PFS relativity adjuster reflects the estimated overall difference between the payment that would otherwise be made to a hospital under the OPPS for the non-excepted items and services furnished in non-excepted off-campus PBDs and the resource-based payment under the PFS for the technical aspect of those services with reference to the difference between the facility and nonfacility (office) rates and policies under the PFS. The current PFS relativity adjuster is set at 40 percent of the amount that would have been paid under the OPPS (82 FR 53028). Non-excepted PBDs are required to use the modifier “PN” so that the PFS relativity adjuster is applied to the payment of their claim. Excepted PBDs use the modifier “PO” on their claims to indicate that the service was provided at an excepted off-campus PBD and that payment should generally be made at the OPPS rate.
In the CY 2019 OPPS/ASC final rule with comment period, we stated that we consider the shift of services from the physician office to the hospital outpatient department unnecessary if the beneficiary can safely receive the same services in a lower cost setting but is instead receiving services in the higher paid setting due to payment incentives.[149] In the CY 2026 OPPS proposed rule (90 FR 33476), to better understand the migration of services in OPDs we analyzed claims data for drug administration services to assess whether increases in volume and spending could be driven by payment incentives. We examined the top twenty most frequently billed HCPCS codes in the drug administration APC family at both excepted and non-excepted off-campus PBDs. Twenty HCPCS codes account for over 98 percent of the volume of drug administration services in off-campus PBDs. We found that the top twenty most frequently billed HCPCS codes in the drug administration APCs when provided at an off-campus PBD excepted from section 1833(t)(21) of the Act (departments that bill the modifier “PO” on claim lines) and off-campus PBDs that are not excepted from ( printed page 53806) section 603 of the BBA (departments that bill the modifier “PN”), are the same with slight variations in the order based on volume. We knew that the overwhelming majority of HCPCS codes in the drug administration APCs were being billed with both the “PO” and “PN” modifiers. Meaning that these drug administration services were being provided in both excepted and non-excepted PBDs. That indicates that the payment rate in non-excepted PBDs is sufficient and can support the provision of these services in an off-campus PBD. We further used the PFS payment rates for the top twenty most frequently billed drug administration HCPCS codes by excepted PBDs (departments that bill the modifier “PO” on claim lines) and volume weighted them to create a PFS proxy APC payment rate for each of the four drug administration APCs. We found that for each of the four APC payment levels, the same services were paid 200-300 percent higher under the OPPS than under the PFS. The volume-weighted PFS payment for the drug administration APCs ranged from 24 percent to 33 percent of the OPPS payment.
We concluded that the differential in our payment rates had created a payment incentive that had led to unnecessary growth for the services in the drug administration APCs. If the PFS payment rate for drug administration APCs ranges from 24 percent to 33 percent of the OPPS payment, then payment using the PFS relativity adjuster of 40 percent should sufficiently cover the cost of these services. We considered the shift of services from the physician office to the hospital outpatient department unnecessary if the beneficiary can safely receive the same services in a lower cost setting but is instead routinely receiving services in the higher paid setting due to payment incentives. We believed the OPPS payment rate for drug administration APCs being several times greater than the PFS rate provides this payment incentive and that the growth in drug administration services paid under the OPPS over time is unnecessary.
5. Patient Severity and Cost of Care
In comments to the CY 2019 OPPS/ASC proposed rule and subsequent rulemaking, we heard from commenters that the higher payments for services in hospital outpatient settings are justified by the level of care patients need, the higher costs of providing care in hospitals, and the costs of maintaining emergency care and standby capacity. We recognize that OPDs serve unique patient populations and provide services to medically complex beneficiaries; however, there is no evidence to demonstrate the need for higher payment for services provided in OPDs that could also be provided in lower-cost settings. In general, despite marked differences in payment rates for a range of services, identical services are being delivered to very similar patients across physicians' offices, hospital outpatient departments, and ASCs.[150 151] Moreover, a 2023 literature review found no peer-reviewed evidence that shows differences in the quality of services delivered across hospital outpatient departments and physicians' offices.[152] In their 2023 report, MedPAC evaluated risk scores from the CMS hierarchical condition category (CMS-HCC) risk-adjustment model to compare the medical complexity of OPD patients with patients in freestanding offices. They found that, on average, OPD patients have higher risk scores, which suggests that OPD patients are potentially more medically complex than those in physician offices. However, they also found substantial overlap in the CMS-HCC risk scores of patients in these two settings, which suggests that the difference in patient severity between settings is small. Their analysis showed that the effects of patient severity on cost of care for the aligned services is not statistically significant as the services, like drug administration, are generally of low complexity. In addition, if there is a need to bill for more complex cases, under the OPPS providers can often bill separately for additional services that a patient might need.
6. Impact of Unnecessary Increases in Volume on the OPPS
Our concern with unnecessary increases in the volume of drug administration services was tied to the health and sustainability of the OPPS. In the CY 2019 OPPS/ASC final rule with comment period, we found that the mean and median annual increase in the volume and intensity of hospital outpatient services was about 5.5 percent and 5.4 percent, respectively, from 2011 to 2019. During this time period, the estimated increase in aggregate annual hospital reimbursements incurred through Medicare Fee for Service (FFS) Part B was $28.2 billion.[153] As stated in the CY 2026 OPPS/ASC proposed rule (90 FR 33476) and in Table 120, we projected that between 2019 and 2027, the cost of outpatient hospital services per FFS enrollee would grow at a mean of about 7.3 percent per year and a median of 8.1 percent per year. This accounts for a $27.2 billion increase in aggregate annual incurred reimbursements for hospitals in FFS Part B during that time, far exceeding the growth of other categories of Part B services in FFS in dollar terms.[154]
( printed page 53807)There was evidence that increased volume and intensity of certain covered OPD services was likely driven by financial incentives to furnish services in hospitals in order to receive higher reimbursement, rather than making site-of-service decisions based on medical necessity. The OPPS was originally designed to manage Medicare spending growth by replacing a cost-based system with a prospective payment system. Contrary to this Congressional purpose, the OPPS had continued to be the one of the fastest growing sectors of Medicare payments out of all payment systems under Medicare Parts A and B.[156] Furthermore, we were concerned that the persistent rate of growth relative to other payment systems suggests that payment incentives, rather than patient acuity or medical necessity, continue to affect site-of-service decision-making. This site-of-service selection had an impact on not only the Medicare program, but also on Medicare beneficiary out-of-pocket spending. Therefore, to the extent that there were lower-cost sites-of-service available, we continued to believe that beneficiaries and the physicians treating them should have that choice and not be encouraged to receive or provide care in higher paid settings solely for financial reasons. Our authority to implement volume control methods was an important tool in combating unnecessary OPPS utilization. We had seen success in stemming the unnecessary growth in the volume of off-campus clinic visits and believed off-campus drug administration services were in need of similar examination.
Therefore, given the unnecessary increases in the volume of drug administration services in hospital outpatient departments, for the CY 2026 OPPS, we proposed to use our authority under section 1833(t)(2)(F) of the Act to apply an amount equal to the site-specific PFS payment rate for nonexcepted items and services furnished by a non-excepted off-campus PBD (the PFS payment rate) for any HPCPCs codes assigned to the drug administration services APCs, when provided at an off-campus PBD excepted from section 1833(t)(21) of the Act (departments that bill the modifier “PO” on claim lines). Table 121 shows the specific APCs that we proposed to identify for this policy, which are APCs 5691-5694. Off-campus PBDs that are not excepted from section 603 of the BBA (departments that bill the modifier “PN”) already received a PFS-equivalent payment rate for any HCPCS codes assigned to the drug administration services APCs. Additionally, we noted that this proposal aligned with President Trump's Executive Order (E.O.) 14273, “Lowering Drug Prices by Once Again Putting Americans First”.[157] Section 11 of the E.O., “Reducing Costly Care for Seniors”, directs the Secretary to “evaluate and, if appropriate and consistent with applicable law, proposed regulations to ensure that payment within the Medicare program is not encouraging a shift in drug administration volume away from less costly physician office settings to more expensive hospital outpatient departments”.
We proposed to implement this proposed method to address the unnecessary increases in utilization of drug administration services in the OPD setting in a non-budget neutral manner, similar to the CY 2019 OPPS/ASC final rule with comment period approach to address unnecessary increases in utilization of clinic visits (83 FR 58818). We proposed to continue our interpretation that while section 1833(t)(9)(B) of the Act requires that ( printed page 53808) certain changes made under the OPPS be made in a budget neutral manner, this section does not apply to the volume control method under section 1833(t)(2)(F) of the Act. In particular, section 1833(t)(9)(A) of the Act, titled “Periodic review,” provides, in part, that the Secretary must annually review and revise the groups, the relative payment weights, and the wage and other adjustments described in paragraph (2) to take into account changes in medical practice, changes in technology, the addition of new services, new cost data, and other relevant information and factors” (emphasis added). Section 1833(t)(9)(B) of the Act, titled “Budget neutrality adjustment” provides that if “the Secretary makes adjustments under paragraph (A), then the adjustments for a year may not cause the estimated amount of expenditures under this part for the year to increase or decrease from the estimated amount of expenditures under this part that would have been made if the adjustments had not been made” (emphasis added). However, a volume-control method under section 1833(t)(2)(F) of the Act is not an “adjustment” under paragraph (2). Unlike the wage adjustment under section 1833(t)(2)(D) of the Act and the outlier, transitional pass-through, and equitable adjustments under section 1833(t)(2)(E) of the Act, section 1833(t)(2)(F) of the Act refers to a “method” for controlling unnecessary increases in the volume of covered OPD services, not an adjustment. Likewise, sections 1833(t)(2)(D) and (E) of the Act also explicitly require the adjustments authorized by those paragraphs to be budget neutral, while the volume control method authority at section 1833(t)(2)(F) of the Act does not. Therefore, the volume control method proposed under section 1833(t)(2)(F) of the Act is not one of the adjustments under section 1833(t)(2) of the Act that is referenced under section 1833(t)(9)(A) of the Act that must be included in the budget neutrality adjustment under section 1833(t)(9)(B) of the Act. Moreover, section 1833(t)(9)(C) of the Act specifies that if the Secretary determines under methodologies described in paragraph (2)(F) that the volume of services paid for under this subsection increased beyond amounts established through those methodologies, the Secretary may appropriately adjust the update to the conversion factor otherwise applicable in a subsequent year. We interpreted this provision to mean that the Secretary can implement a volume control method under section 1833(t)(2)(F) of the Act in a nonbudget neutral manner in the year in which the method is implemented, and that the Secretary may then make further adjustments to the conversion factor in a subsequent year to account for volume increases that are beyond the amounts estimated by the Secretary under the volume control method.
We stated in the CY 2019 OPPS/ASC final rule with comment period (83 FR 58818) that we believed implementing a volume control method in a budget neutral manner would not appropriately reduce the overall unnecessary volume of covered OPD services, and instead would simply shift the movement of the volume within the OPPS system in the aggregate, a concern similar to the one we discussed in the CY 2008 OPPS/ASC final rule with comment period (72 FR 66613). We believed that concern applies to drug administration services just the same. The estimated payment impact for various provider classifications is displayed in Table 167: Estimated Impact of the Final CY 2026 Changes for Services Provided in the Hospital Outpatient Prospective Payment System of this final rule with comment period. The 10-year estimated impact of this policy is displayed in Table 166: Estimated Effect of Changes to Drug Administration Services when Furnished at Excepted Off-Campus Providers. For CY 2026, the estimated savings are $290 million, with $220 million of the savings accruing to Medicare, and $70 million saved by Medicare beneficiaries in the form of reduced beneficiary coinsurance.[158] To effectively establish a method for controlling the unnecessary growth in the volume of drug administration services furnished by excepted off-campus PBDs that does not simply reallocate expenditures that are unnecessary within the OPPS, we believed that this method must be adopted in a nonbudget neutral manner. The impact associated with this proposal is further described in section XXV. of this final rule with comment period.
While we were refining our method to control for unnecessary increases in the volume of hospital outpatient department services, we continued to recognize the importance of not impeding development or beneficiary access to new innovations. We solicited public comments on other ways to exercise the Secretary's statutory authority under section 1833(t)(2)(F) of the Act:
- Are there other services for which CMS should develop a method to control unnecessary increases in the volume of covered OPD services by paying a PFS-equivalent rate for services provided at excepted off-campus PBDs?
- Of particular concern for us are the services within the imaging without contrast APCs (APCs 5521-5524). Imaging without contrast services are some the most costly and frequently provided services at excepted PBDs. We believe that there is a high likelihood that there has been unnecessary growth in this space and that a volume control method would be appropriate to apply here in the future. Would it be appropriate to apply this method to the Imaging Without Contrast APCs?
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: We received a number of comments in response to our comment solicitation on other services for which the Secretary's should exercise his statutory authority under section 1833(t)(2)(F) of the Act. Commenters shared their views on whether there have been unnecessary increases in volume for certain other services and whether it would be appropriate to apply our volume control method to those services.
Response: We thank commenters for their interest and engagement on this important issue. Given the wide array of information presented through this public comment process, we may take the technical recommendations and other detailed feedback provided regarding other services that could potentially have unnecessary increases in volume under consideration for future notice and comment rulemaking.
Comment: Numerous commenters, including organizations representing private health insurance plans, physician associations, specialty medical associations, and individual Medicare beneficiaries, supported the proposal to apply a volume control method for drug administration APCs. Some commenters commended CMS for its proposal, which they believed will help to control costs for both beneficiaries and the Medicare program, as well as foster greater competition in the physician services market. Commenters agreed that this method will help to control the unnecessary increases in volume of services in excepted off-campus PBDs when beneficiaries can generally safely receive these same services in a lower ( printed page 53809) cost setting but instead receive care in a higher cost setting due to payment incentives. Commenters stated that higher payments to OPDs for lower-complexity services directly has translated into higher costs for Medicare beneficiaries and taxpayers, as hospitals shift care towards higher-paid OPDs. Commenters cited studies showing that this has indirectly driven up Medicare spending and out-of-pocket costs for beneficiaries by incentivizing consolidation of independent physician practices into hospitals, which is associated with higher prices.[159 160 161] Commenters noted that consolidation has the downstream effect of shifting more billing from the lower physician office rates to higher OPD rates, further increasing total Medicare spending and beneficiary cost-sharing. Several commenters flagged that consolidation is further driven by incentives in the 340B program, where participating hospitals profit by acquiring physician offices, converting them into outpatient clinics, purchasing discount drugs and funneling drug administration services to this site of care.
Commenters wrote that our proposed policy would help to address long-standing payment disparities between OPDs and office-based infusion centers, where commenters believe the same services are delivered at lower cost to Medicare and beneficiaries without compromising quality. Others believed that addressing the unnecessary increases in the volume of these services would help address the ramifications of market distortions that restrict the availability of lower-cost care and increase costs for patients and taxpayers without adding quality improvements. Commenters advocated that non-hospital settings of care often deliver the same or better quality of care at lower cost and rebalancing the effects of unnecessary utilization would enhance the overall value of Medicare spending and direct resources toward lower-cost and effective care models. Commenters asserted that off-campus PBDs are often indistinguishable from physician offices aside from the hospital signage; commenters stated that nothing about the site of care justifies the higher payment rates. Commenters stated that off-campus PBDs are often indistinguishable from physician offices aside from the hospital signage and nothing about the site of care has meaningfully changed to justify their higher payment rates. Because of this, commenters stated that off-campus PBDs are not providing true hospital-based care and do not require additional payment for 24/7 capacity or emergency standby services. Other commenters claimed that the office setting is the most cost-effective and patient-centered site of service for drug administration services. They wrote that delivering drug therapies in the office setting not only saves the health care system money, but also avoids unnecessary risks such as hospital-acquired infections—risks that are particularly concerning for immune-compromised patients receiving drug administration services.
Several commenters also supported implementing this policy in a non-budget neutral manner, as this would allow CMS to protect and preserve the Part B trust fund rather than simply redistributing funds. Commenters state that by aligning payment for routine services with the PFS rate CMS could help discourage site-of-service shifts driven by financial incentives rather than clinical need, promote competition, and create a more level playing field for providers across care settings. Many commenters encouraged CMS to consider expansions of this policy for additional services in excepted off-campus PBDs and to on-campus OPDs in the future.
Response: We appreciate the commenters' support. As mentioned in the CY 2026 OPPS/ASC proposed rule, we share the commenters' concern that payment incentives, rather than patient acuity or medical necessity, are affecting site-of-service decision-making and the downstream effect these payment disparities create. As we noted in the CY 2019 OPPS/ASC proposed rule (83 FR 37138 through 37143), “[a] large source of growth in spending on services furnished in hospital outpatient departments (OPDs) appears to be the result of the shift of services from (lower cost) physician offices to (higher cost) OPDs”. We continue to believe that these shifts in site of service are unnecessary if the beneficiary can safely receive the same services in a lower cost setting but instead receives care in a higher cost setting due to payment incentives. In addition to the concern that the difference in payment is leading to unnecessary increases in the volume of covered outpatient department services, we remain concerned that this shift in care setting increases beneficiary cost-sharing liability because Medicare payment rates for the same or similar services are generally higher in hospital outpatient departments than in physician offices.
We appreciate the comments supporting the implementation of this policy in a nonbudget neutral manner. As we stated in the CY 2026 OPPS/ASC proposed rule (83 FR 37138 through 37143), we believe implementing a volume control method in a budget neutral manner would not appropriately reduce the overall unnecessary volume of covered OPD services, and instead would simply shift the volume of services within the OPPS system in the aggregate. As detailed later in this section, we are finalizing our proposal, without modification, in response to public comments. We will continue to take information submitted by the commenters into consideration for future study.
Comment: In their comment letter the Medicare Payment Advisory Commission (MedPAC) noted their support for expanding the PFS equivalent payment rate for drug administration services that are provided in excepted off-campus PBDs. They highlighted their June 2023 Report to Congress in which they included a framework they suggested could be helpful in identifying services for which it might be safe and appropriate to align payment rates. They noted that CMS should consult with clinicians, industry, and other interested parties when determining which services to choose.
MedPAC stated that payment parity between the OPPS and PFS should not adversely affect hospitals' ability to be available 24/7 for emergency care, with particular attention paid to safety-net and rural providers. Additionally, they recommended maintaining the packaging of ancillary items in the OPPS. MedPAC reiterated a point from their June 2023 report to the Congress, in which they noted some benefits they see in making budget-neutral adjustments for some OPPS services in conjunction with site-neutral payments. For example, applying a budget-neutral payment adjustment with a site-neutral policy would increase OPPS payment rates for other services including emergency department visits, which would support hospitals' emergency care and standby capacity.
Response: We thank MedPAC for its comment and support of this policy. We have continued to find their work ( printed page 53810) valuable as we further identify services that may have experienced unnecessary increases in volume in the OPPS. In the CY 2026 OPPS/ASC proposed rule, we requested information from interested parties on other services for which CMS should develop a method to control unnecessary increases in the volume of covered OPD services by paying a PFS-equivalent rate for services provided at excepted off-campus PBDs. We will continue to work with clinicians and industry interested parties when making choices about which services to examine in the future.
We believe that our volume control method will not adversely impact hospitals' ability to provide 24/7 emergency care and, as discussed later in this section, we are finalizing our proposal to exempt rural Sole Community Hospitals from this expanded policy, which we believe addresses MedPAC's concern that a non-budget neutral volume control method will adversely affect rural providers. We are applying our volume control method to a limited set of services provided in a limited set of OPD departments and do not believe that the financial impact of this provision will significantly impact hospitals' ability to provide 24/7 emergency care. Our final policy will only impact drug administration services billed with the “PO” modifier.
We understand that MedPAC's June 2023 recommendation proposed to make some site-neutral adjustments in a budget neutral manner. However, we are using our authority under section 1833(t)(2)(F) of the Act to implement a volume control method to control unnecessary utilization in drug administration. Under sections 1833(t)(2)(F) and (9)(C) of the Act, we are not required to budget neutralize the volume-control method here, and as we stated in the CY 2019 OPPS ASC final rule with comment period (83 FR 59013), we believe implementing a volume control method in a budget neutral manner would not reduce the overall unnecessary volume of covered OPD services, and instead would simply shift services within the OPPS system because of payment rather than medical necessity.
We received several comments which provided detailed legal rationales as to why CMS lacks the statutory authority to reduce payments to excepted off-campus PBDs, particularly in a non-budget neutral manner. We discuss these comments below.
Comment: Commenters suggested that legal developments since the U.S. Court of Appeals for the District of Columbia decision in American Hospital Association v. Azar, 964 F. 3d 1230 (D.C. Cir. 2020), which had supported the agency's interpretation of section 1833(t)(2)(F) of the Act, now may not be viable and undermine the agency's reliance on section 1833(t)(2)(F) of the Act for the proposed volume-control methodology.
Commenters argued that the D.C. Circuit in American Hospital Association v. Azar reviewed HHS' interpretation “under Chevron' s two-step framework.” Id. at 1241. But Chevron has since been overruled. See Loper Bright Enterprises v. Raimondo, 603 U.S. 369, 412 (2024). Consequently, “courts need not and under the [Administrative Procedures Act (APA)] may not defer to an agency interpretation of the law simply because a statute is ambiguous”. Id. at 413. Commenters assert that is what the D.C. Circuit impermissibly did in American Hospital Association v. Azar. See 964 F.3d at 1244.
Commenters contend that the agency's reading of section 1833(t)(2)(F) of the Act is not the best interpretation of the law. For reasons previously offered through public comment and litigation, commenters described that the best interpretation of the law is that section 1833(t)(2)(F) of Act does not authorize HHS to lower payments only for certain services performed by certain providers, including and especially those grandfathered under the law.
Commenters go on to argue that other precedential developments cast doubt on the D.C. Circuit's decision in American Hospital Association v. Azar. Commenters explained that the D.C. Circuit did not adequately address what they believed was the district court's correct conclusion that CMS' interpretation of section 1833(t)(2)(F) of the Act assumes the authority to “supersede Congress' carefully crafted relative payment system” based on a single sentence in the U.S. Code. Id. at 158.
Commenters posit that, under HHS' view, a provision that gives the agency authority to adopt a “method[s] for controlling unnecessary increases in the volume” of covered outpatient services permits it to ignore the entire OPPS system and make non-budget-neutral reductions to particular services. Echoing the district court, commenters stated this would massively “upend” the OPPS system (rejecting HHS' attempt to “acquire unilateral authority to pick and choose what to pay for OPD services, which clearly was not Congress' intention”).
Commenters argue that the Supreme Court rulings in West Virginia v. EPA and Biden v. Nebraska “double-down” on the legal principles described above. Commenters describe HHS as claiming unfettered power to depart from the OPPS based on a vague provision buried elsewhere in the statute. They contend that HHS will not be able to do so because the Congress does not “use oblique or elliptical language to empower an agency to make a `radical or fundamental change' to a statutory scheme”. West Virginia, 597 U.S. at 723 (quoting MCI Telecommunications Corp. v. American Telephone & Telegraph Co., 512 U.S. 218, 229 (1994)).
Response: We continue to believe that section 1833(t)(2)(F) of the Act gives the Secretary authority to develop a method for controlling unnecessary increases in the volume of covered OPD services, including a method that controls unnecessary volume increases by removing a payment differential that is driving a site-of-service decision and, as a result, is unnecessarily increasing service volume.
As we noted in the CY 2019 OPPS/ASC final rule with comment period (83 FR 58818) and in our decision to complete the two-year phase of this policy in the CY 2020 OPPS/ASC final rule with comment period (84 FR 61142), “[a] large source of growth in spending on services furnished in hospital outpatient departments (OPDs) appears to be the result of the shift of services from (lower cost) physician offices to (higher cost) OPDs”. We continue to believe that these shifts in the sites of service are unnecessary if the beneficiary can safely receive the same services in a lower cost setting but instead receives care in a higher cost setting due to payment incentives. In most cases, the difference in payment is leading to unnecessary increases in the volume of covered outpatient department services, and we remain concerned that this shift in care setting increases beneficiary cost-sharing liability because Medicare payment rates for the same or similar services are generally higher in hospital outpatient departments than in physician offices. We continue to believe that our method addresses the concerns described in the CY 2019 OPPS/ASC final rule with comment period (83 FR 59005).
As we stated in the CY 2019 OPPS ASC final rule with comment period (83 FR 59013), we believe implementing a volume control method in a budget neutral manner would not appropriately reduce the overall unnecessary volume of covered OPD services, and instead would simply shift services within the OPPS system because of payment rather than medical necessity. We also outlined in the CY 2019 OPPS/ASC ( printed page 53811) final rule with comment period (83 FR 59013) that while section 1833(t)(9)(B) of the Act requires that certain changes made under the OPPS be made in a budget neutral manner, this section does not apply to the volume control method under section 1833(t)(2)(F) of the Act.
As noted, the D.C. Circuit previously held that our regulation was a reasonable interpretation of section 1833(t)(2)(F)'s of the Act authority to adopt a method to control for unnecessary increases in the volume of the relevant service. See AHA v. Azar, 964 F.3d at 1241-45. Loper Bright does not change the result. As an initial matter, while Loper Bright changed certain aspects of the interpretative framework the D.C. Circuit used, the Supreme Court cautioned that the “holdings of those cases that specific agency actions are lawful . . . are still subject to statutory stare decisis despite our change in interpretive methodology”. 144 S. Ct. at 2273. AHA' s holding that our volume-control methodology complies with section 1833(t)(2)(F) of the Act is therefore still good law.
Even interpreting section 1833(t)(2)(F) of the Act anew under the new Loper Bright framework, the Supreme Court clarified that the Congress “often” enacts statutes that “delegate[ ] discretionary authority to an agency.” Id. at 2263. Section 1833(t)(2)(F) of the Act is just such a statute. By instructing the Secretary to “develop a method for controlling unnecessary increases in the volume of covered OPD services” without specifying the method, the statute delegates discretionary authority to the Secretary. Because, as the D.C. Circuit explained, our policy “falls comfortably within the plain text of subparagraph (2)(F)” and is further supported by its structure, 964 F.3d at 1241, it does not fall outside the “outer statutory bounds” of the delegation, as discussed in Loper Bright. We thus read AHA to apply the Chevron framework because that was the law at the time and to suggest that if the court had analyzed this matter under the new Loper Bright framework, the result would have been the same. That aspect of the D.C. Circuit's holding, particularly when coupled with the stare decisis principles discussed previously in the final rule with comment period, indicate that there is no reason to doubt the continued validity of our policy. 144 S. Ct. at 2268.
Nor do we agree that the major question doctrine or related interpretation principles undermine our interpretation of section 1833(t)(2)(F) of the Act. As the Supreme Court recently reiterated, those principles do not apply in the “ordinary” case; only in “extraordinary cases” will courts apply a “different approach” from usual to arrive at the best meaning of a statute. W. Virginia, 597 U.S. at 721. Here, neither the “`history and the breadth of the authority that [the agency] has asserted,” nor the “economic and political significance” of that authority militate against our reading of section 1833(t)(2)(F) of the Act. Unlike the novel debt-relief authority the court rejected in Biden v. Nebraska, 600 U.S. at 500-01, we have not offered in this final rule with comment period a novel interpretation of section 1833(t)(2)(F) of the Act, or even a novel methodology to control volume increases. Instead, we extend a judicially-endorsed methodology to another context. We are not “restructur[ing] the American energy market,” like the agency in West Virginia, 597 U.S. at 724, but preventing a particular payment system manipulation—unnecessary volume increases—that the Congress expressly required us to address. And section 1833(t)(2)(F) of the Act lacks any term carrying “a connotation of increment or limitation” like the statute in Biden v. Nebraska, 600 U.S. at 494 (internal quotation marks omitted). Here, the Congress authorized the agency to put in place a “methodology” that accomplishes the goal of controlling unnecessary volume-increases. Following the Congress' instruction to accomplish its stated goal does not “upend” the payment system the Congress crafted or make radical changes to it; it helps to vindicate it. By focusing on the length of section 1833(t)(2)(F) of the Act, commenters confuse the number of words the Congress used with the substantive authority those words communicate. “Brevity,” whether in regulatory documents or legislation, “should not be mistaken for lack of detail.” Seven Cnty. Infrastructure Coal. v. Eagle Cnty., Colorado, 145 S. Ct. 1497, 1512 (2025).
Here, the agency has identified an unnecessary increase in volume in a set of OPPS payment codes based on payment differentials between the OPPS and the PFS. It has tailored a methodology to eliminate that unnecessary volume increase by eliminating its cause. As the D.C. Circuit already found, that falls comfortably within the agency's delegated authority under the best reading of section 1833(t)(2)(F) of the Act.
Comment: Commenters outlined that the D.C. Circuit failed to account for section 603 of the BBA. Commenters explained that the BBA of 2015 created two categories of provider-based departments (PBDs): (1) those established before November 2015 and (2) those established after November 2015. For those PBDs in existence prior to November 2015, commenters stated that the Congress required CMS to continue paying off-campus PBDs (referred to in the statute as “excepted” off-campus PBDs) at the same rate as hospitals; for post-November 2015 PBDs, the Congress required CMS to pay PBDs at the same rate as independent physicians' offices. Commenters believed that the Congress reinforced this policy choice by providing that “mid-build” PBDs should be paid at the same rates as existing, pre-November 2015 off-campus PBDs, that is, the same rate as hospitals. Some commenters asserted that section 603 of the BBA, represents the Congress' thoughtful consideration of MedPAC's recommendation to eliminate the difference in payment between hospital outpatient departments and physician's offices in certain circumstances, and CMS' proposal undermines the balance struck by the Congress. Again, commenters state the D.C. Circuit ruled incorrectly and that the agency should not be able to use the decision in American Hospital Association v. Azar to deflect from addressing this issue. They take issue with the fact the D.C. Circuit treated section 603 of the BBA as an “alternative” argument and relegated its analysis of that provision to a separate section of its opinion from its analysis of section 1833(t)(2)(F) of the Act. Commenters opined that the court of appeals answered the section 1833(t)(2)(F) of the Act question both as if it were wholly distinct from the section 603 of the BBA question and with a heavy thumb on the scale due to Chevron deference.
Going forward, they proclaim that the agency cannot rely solely on American Hospital Association v. Azar and must instead provide a more fulsome legal explanation than the D.C. Circuit for why the text and history of section 603 of the BBA do not foreclose its authority here.
Response: As we stated in the CY 2019 OPPS/ASC final rule with comment period (83 FR 59005) we continue to believe the changes required by section 603 of the BBA made in section 1833(t) of the Act had the effect of addressing some of the concerns related to shifts in settings of care and overutilization of services in the hospital outpatient setting for new off-campus PBDs that began billing Medicare for OPPS services after November 1, 2015. In passing section 603 of the BBA, Congress addressed the specific circumstances that lead to the proliferation of new off-campus ( printed page 53812) provider-based departments. As we stated in the CY 2017 OPPS/ASC final rule with comment period (81 FR 79562), we believe Congress may have been trying to address the incentive for hospitals to purchase physician's offices and convert them to OPDs without changing their location or patient population with section 603 of the BBA. In 2019 and now in 2025, we are addressing new and separate circumstances, the over utilization of certain services in the OPPS.
We disagree with commenters that by passing legislation a decade ago that does not reference section 1833(t)(2)(F) of the Act, Congress impliedly meant to circumscribe our authority under that provision. As the D.C. Circuit explained when rejecting a similar argument, “[n]othing in the text of section 603 of the BBA indicates that preexisting off-campus PBDs are forever exempt from adjustments to their reimbursement” and instead “leav[es] the exempted providers subject to all the provisions of the OPPS statute, including subparagraph (2)(F).” AHA, 946 F.3d at 1246.
Additionally, as the D.C. Circuit explained, even assuming that section 603 of the BBA could be read to judge increases in volume at preexisting off-campus PBDs are not “unnecessary” under section 1833(t)(2)(F) of the Act, that judgment would extend only to 2015 and “would not mean that Congress considered acceptable the continued volume increases later taking place” in other years. Id. When the Congress passed the BBA of 2015, Medicare OPPS expenditures were $56 billion and growing at an annual rate of about 7.3 percent. In addition, the percentage increase in volume and intensity of outpatient services was increasing at 3.4 percent. In 2019 without the volume control method, OPPS expenditures would have been approximately $74.5 billion, growing at a rate of 9.1 percent, with the volume and intensity of outpatient services increasing at 5.4 percent. For 2026, we estimate that, without an expansion of this policy to drug administration services, OPPS expenditures would be $101.0 billion, growing at a rate of 8.6 percent, with the volume and intensity of outpatient services increasing at 6.8 percent. A review of claims processed in CY 2022 showed that only 2.3 percent of payments for outpatient services are made at the PFS equivalent rate for off-campus PBDs.[162] We would not be able to adequately address the unnecessary increases in the volume of clinic visits in OPDs after 2015 if we did not apply this policy to all off-campus OPDs.
Finally, the hospitals are also wrong that Loper Bright undermines the D.C. Circuit's analysis of section 603 of the BBA. Nothing in the court's analysis of section 603 of the BBA suggests that it found the provision ambiguous or incorrectly deferred to the agency's interpretation of it. See id. And contrary to the commenters' suggestion, the court fully considered any impact of section 603 of the BBA on the agency's authority under the OPPS statute, even if it characterized the section 603 argument as “alternative”.
Comment: Commenters reiterated their objections from the CY 2019 OPPS/ASC proposed and final rules with comment period that these existing and proposed 60 percent payment cuts constitute “adjustments” that are subject to the budget neutrality requirements set forth in section 1833(t)(9)(B) of the Act, and urged CMS to return the OPPS to the budget neutral payment system required by the Congress.
Response: As we stated in the CY 2019 OPPS/ASC final rule with comment period (83 FR 59005), we maintain that while section 1833(t)(9)(B) of the Act does require that many changes made under the OPPS be made in a budget neutral manner, this provision does not apply to the volume control method under section 1833(t)(2)(F) of the Act as outlined through our proposals in CY 2019 and CY 2026 rulemakings. As we noted in the CY 2019 OPPS/ASC proposed rule (83 FR 37138 through 37143), unlike the wage adjustment under section 1833(t)(2)(D) of the Act and the outlier, transitional pass-through, and equitable adjustments under section 1833(t)(2)(E) of the Act, section 1833(t)(2)(F) of the Act refers to a “method” for controlling unnecessary increases in the volume of covered OPD services, not an adjustment. Likewise, sections 1833(t)(2)(D) and (E) of the Act also explicitly require the adjustments authorized by those paragraphs to be budget neutral, while the volume control method authority at section 1833(t)(2)(F) of the Act does not include such a requirement. Therefore, we maintain that the volume control method proposed under section 1833(t)(2)(F) of the Act is not one of the adjustments under section 1833(t)(2) of the Act that is referenced under section 1833(t)(9)(A) of the Act that must be included in the budget neutrality adjustment under section 1833(t)(9)(B) of the Act. Moreover, section 1833(t)(9)(C) of the Act specifies that if the Secretary determines under methodologies described in paragraph (2)(F) of section 1833(t) of the Act that the volume of services paid for under this subsection increased beyond amounts established through those methodologies, the Secretary may appropriately adjust the update to the conversion factor otherwise applicable in a subsequent year. We continue to interpret this provision to mean that the Secretary will have implemented a volume control method under section 1833(t)(2)(F) of the Act in a nonbudget neutral manner in the year in which the method is implemented. Further, as we stated in the CY 2019 OPPS/ASC proposed rule (83 FR 37138 through 37143), we believe that implementing a volume control method in a budget neutral manner would not appropriately reduce the overall unnecessary volume of covered OPD services, and instead would simply shift the volume within the OPPS system in the aggregate.
In addition, the D.C. Circuit has held not only that budget neutrality is not required under a volume control method, making that method budget neutral would be contrary to the purpose and intent of the Medicare statute. For example, the court emphasized that subparagraph (2)(F) of the Act “says nothing about budget-neutrality,” and specifically rejected the hospitals' argument that volume-control methods must be budget neutral. AHA, 964 F.3d at 1241. After all, it would be “anomalous” for the statute to require a rate cut made for purposes of volume control to be “implemented budget-neutrally” because, if we were required “to redistribute the costs traceable to the provisions of unnecessary services throughout the OPPS,” the result would be “no net savings to Medicare,” “largely negating the point of reducing reimbursement in the first place”. Id. at 1241 through 1242. ( See83 FR 37142 through 37143). This common-sense interpretation is thus supported by the text and structure of the OPPS statute, as both we and the D.C. Circuit have explained.
Comment: Some commenters were particularly concerned with using only the most recent claims data as this does not address the vertical consolidation trend whereby hospitals acquired many physician offices that were later reclassified as hospital outpatient departments (OPDs). Commenters stated that only using more recent data “bakes in” the higher number of services provided by OPDs as a result of this consolidation resulting in more claims paid at the higher OPPS rate. Commenters suggest using a time period or a range of years prior to the ( printed page 53813) consolidation trend (for example, 2010 through 2014), as this captures the volume of service before the vertical consolidation trend. Commenters stated this timeframe provides a more appropriate baseline for analyzing the site of care and corresponding fee schedule since it better illustrates the landscape before consolidation trends resulted in shifts to care settings for the financial benefit of the hospitals. Other commenters suggested that in order for CMS to understand which services can be appropriately provided in freestanding office settings a reasonable place to begin is with understanding where procedures have been historically performed. But in doing so, commenters stated, CMS must not permit the provider consolidation dynamic that has driven the need for site neutrality to hobble CMS' efforts to address the consequences of that dynamic.
Conversely some commenters raised concerns related to the data we presented in the CY 2026 OPPS/ASC proposed rule. Specifically, commenters stated a belief that the timeframe we used to analyze the volume of drug administration services, which largely was periods of time between 2012-2024, was not appropriate because that timeframe encompasses growth trends that are not relevant to current utilization. Commenters contend that utilization prior to the implementation of section 603 of the BBA should not be evaluated because the BBA fully addressed any overutilization that may have been occurring.
Some commenters stated that their analysis contradicted our findings of volume increases for drug administration services. Commenters cited the CMS finding that there has been a 70 percent increase in the volume of chemotherapy claims billed with HCPCS code 96413 (chemotherapy administration, intravenous infusion technique; up to 1 hour, single or initial substance/drug) for services furnished in excepted PBDs between 2011 and 2023. Commenters review of claims data indicated that claims for this service in excepted, off-campus PBDs are declining. In 2020 they believe that approximately 550,000 units of this code were reported for chemotherapy administrations in excepted PBDs, and this number fell to 546,000 units in 2024. Their data analysis shows that there was not a single year in this 5-year period where excepted PBD claims for this HCPCS code have exceeded 2020 utilization.
Response: We thank the commenters for their input. Based on these comments, we have looked at a wider timeframe to determine whether it changes our conclusion that the unnecessary volume of a service has increased. OPPS spending by traditional Medicare and its beneficiaries increased 71 percent from 2012 to 2022, and spending per Medicare Part B beneficiary under the OPPS grew rapidly, at an annual rate of 6.9 percent, during this period.[163] Disregarding the growth from this time period and how it has impacted the baseline of OPPS spending would be unwise and would not account for the factors that led to this extreme and often unnecessary growth. We do not believe that it would be appropriate to only examine data from 2020 and later years as some commenters did. In our CY 2026 OPPS?ASC proposed rule analysis, we started our claims review in CY 2011 in order to examine what volume looked like prior and during the intense period of consolidation prior to the passage of the BBA of 2015 and in the subsequent years. Commenters indicated that data spanning the roll-out of section 603 of the BBA and the mid-build exception in 2017 through at least 2019 would naturally report growth arising from providers' acquisition or construction of off-campus PBDs. Rather than a reason to exclude this data, we believe fully understanding how the growth of excepted off-campus PBDs impacted the volume of drug administration services is crucial in examining unnecessary increases in the volume of outpatient services. We note that MedPAC has also used 2012 as a baseline year for their analyses.
Looking only at data from CYs 2020-2024 removes the context for the changes that were occurring in the OPPS over the prior decade. Even if volume leveled off for a particular service in recent years, we would assert that if utilization grew unnecessarily from 2011-2019 and that overutilization is built into the baseline of the OPPS services being provided in recent years, then recent year utilization still reflects unnecessary increases in volume. Stated differently, we do not believe it would matter for purposes of this provision if an increase in volume over ten years happened steadily at the same rate each year, or if the volume fluctuated over those ten years, but ended up at the same increased amount in the final year.
Looking at a wider timeframe only further supports our analysis as presented in the CY 2026 OPPS/ASC proposed rule. As we discussed earlier, we found that there has been an increase in volume of services paid through the drug administration APCs (5691-5694) over time, which would indicate that there has been migration of these services to the OPD setting. From 2011 to 2019 the volume of drug administration services paid under these APCs grew by almost 35 percent. This growth persisted even with the introduction of the PFS-equivalent rate for PBDs subject to section 603 of the BBA starting in 2017. The COVID-19 PHE did impact utilization across the OPPS, but we have seen the volume of drug administration services rebound and return to this pattern of growth and also believe that overutilization has become entrenched in the baseline of the OPPS for some services and APCs. Between 2018 and 2024 the number of beneficiaries enrolled in fee-for-service Medicare decreased by over 14 percent.[164] Since 2022, we have simultaneously seen increases in the volume of drug administration services provided in OPDs utilized per beneficiary.[165] Between 2020 and 2023 the utilization of drug administration services per beneficiary grew by over 30 percent. Meaning that while there are now fewer Medicare fee-for-service beneficiaries than there were prior to the COVID-19 PHE, each beneficiary on average is receiving more drug administration services in the OPD setting than they were prior to the COVID-19 PHE. There was also growth prior to the COVID-19 PHE. The utilization of drug administration services per beneficiary also grew by 30 percent between 2011 and 2019.
Again, as stated earlier, some HCPCS codes within the drug administration APCs have experienced significant growth. HCPCS code 96413—which describes chemotherapy administration, intravenous infusion technique; up to 1 hour, single or initial substance/drug—is the most frequently billed HCPCS code within any of the drug administration APCs at excepted PBDs. This code has seen an almost 70 percent increase in volume from 2011 to 2023.[166] In 2025, this service has a physician office payment rate of around $119 dollars and an OPPS payment rate of approximately $341. That makes the same chemotherapy infusion service 186 percent more expensive in the OPD than in the physician office. We conclude that this 70 percent increase in excepted hospital outpatient department volume over a 10-year period was at least partially driven by the payment ( printed page 53814) differential between the physician office and OPD setting. The HCPCS codes representing chemotherapy administration grew in volume by 64 percent in the OPPS between 2011 and 2023.[167] These changes and the context in which they occurred, illustrate our continued belief that shifts in the sites of service are inherently unnecessary if the beneficiary can safely receive the same services in a lower cost setting but instead receives care in a higher cost setting due to the payment incentives created by the difference in payment amounts.
Comment: Commenters also examined physician billing across from CYs 2020 to 2024 for HCPCS code 96413 and their findings indicate that approximately 1.85 million units of this service were furnished in the physician office setting in 2020, and that physician office utilization grew to 1.87 million units in 2024. Thus they conclude, over the same 5-year period, there is stable growth in physician office utilization (+0.6 percent) and stable to declining utilization of the same service in excepted PBDs (−0.6 percent). They ultimately conclude that the distribution of this service among ambulatory settings has remained stable over the past 5 years. They state that despite our emphasis on HCPCS code 96413 in the CY 2026 OPPS/ASC proposed rule, they believe that recent utilization data on HCPCS code 96413 directly undercuts CMS' assertion that there is “unnecessary growth” in utilization justifying the proposed payment cut.
In other cases, commenters cited data that shows growth in utilization in the physician office setting alongside more modest growth in excepted PBDs. Commenters gave the example of CPT code 96405 (chemotherapy administration; intralesional, up to and including 7 lesions) where utilization increased by approximately 156 percent in the physician office setting, but only increased by approximately 34 percent in excepted PBDs.
In their data analysis, commenters found contractions and growth in drug administration services furnished in excepted PBDs and physician offices. Commenters thus conclude that you could not support a finding of unnecessary growth in drug administration services furnished in excepted PBDs. Commenters also contend that the drug claims billed along with these drug administration codes reflect significant medical advancements and changes in treatment protocols, with new drugs being approved by the FDA and used in hospitals and physician offices for the first time during this five-year period. Where CMS states that “increases in the volume of drug administration services provided in OPDs utilized per beneficiary” since 2022 are an example of unnecessary utilization, commenters contend that there may be medical reasons for these changes (including new drugs and new drug regimens).
Commenters also note that the proposed payment reduction would apply to some services that are not currently furnished in excepted off-campus PBDs. Between 2020 and 2023, there were no Medicare claims for excepted PBDs for the following HCPCS codes: 37195, 90473, 95144, 95170, 96371, 96422, 96423, 96440, and G0012. Commenters state that even if a payment reduction can constitute a “method for controlling unnecessary increases in the volume of covered OPD services”, the payment reductions cannot be applied to services with negligible utilization. Furthermore, commenters cited that some of the HCPCS codes had negligible to no utilization in the physician office setting, including HCPCS codes 96446, 96542, and C8957, which might indicate that it is not reasonable to expect that these services would be provided more often in the physician office setting but for the higher payment rate for these services in the OPPS.
Response: We thank the commenters for their input. We draw a different conclusion from the cases made above. As stated earlier, using 2020-2024 as a time frame for utilization review, even if just for comparison, would not allow us to adequately evaluate whether there has been unnecessary utilization in the OPPS. It would be difficult to fully appreciate the trends in the physician office without acknowledging the baseline for what the volume of services would have been absent the impact of consolidation. We would reason that stability in the physician market shows that the mechanisms in force are persistent and not driving care into the lower cost setting. As commenters note, the provision of these services in the physician office is stable, safe, and being done at a substantial volume. This furthers our belief that some services in the OPD are inherently unnecessary if the beneficiary can safely receive the same services in a lower cost setting but instead receives care in a higher cost setting due to the payment incentives created by the difference in payment amounts.
We do not believe that the inference that growth in utilization in the physician office setting in comparison to more modest growth in excepted PBDs indicates that there is no unnecessary utilization in the excepted PBD. Again, taking the data out of its full historical context does not give you an accurate gauge of the actual growth of that service in excepted PBDs.
As we stated in the CY 2026 OPPS/ASC proposed rule, our review of the utilization of drug administration services in excepted PBDs found increases in the volume of services over time, increases in the volume of services provided per beneficiary, and cases of significant volume growth for some individual HCPCS codes within the drug administration APC family. We believe that these changes represent unnecessary increases in the volume of covered outpatient department drug administration services and that it would be appropriate to apply our volume control method to these services.
In the CY 2026 OPPS/ASC proposed rule we discussed how we examined the twenty most frequently billed HCPCS codes in the drug administration APC family at both excepted and non-excepted off-campus PBDs. We found that twenty HCPCS codes account for over 98 percent of the volume of drug administration services in off-campus PBDs. We found that the twenty most frequently billed HCPCS codes in the drug administration APCs when provided at an off-campus PBD excepted from section 1833(t)(21) of the Act (departments that bill the modifier “PO” on claim lines) and off-campus PBDs that are not excepted from section 603 of the BBA (departments that bill the modifier “PN”), are the same with slight variations in the order based on volume. We know that there is claims volume for the overwhelming majority of HCPCS codes in the drug administration APCs with both the “PO” and “PN” modifiers. There are approximately 61 HCPCS codes within the four levels of the drug administration APCs. Meaning that the remaining 41 codes account for only 2 percent of the volume of drug administration services for PBDs. We have evaluated unnecessary utilization at the APC level and found unnecessary increases in utilization. In the CY 2026 OPPS/ASC proposed rule, we noted specific HCPCS codes that experienced significant increases in volume. We believe this data supports the proposed method to control unnecessary increases in volume for all codes in the drug administration APCs. ( printed page 53815)
We intentionally chose to apply the volume control method for drug administration services at the APC level, rather than at the service level. While we exercised our volume control authority for the first time by applying it to a single code, G0463 (clinic visit), we believe that the most practical and efficient way to apply this method for drug administration services is to apply it to all drug administration services at the APC level. As such, even if a few less frequently used codes in those APCs are rarely utilized in either the outpatient setting or the physician office setting, that does not detract from the larger point that the drug administration services in these APCs have seen unnecessary growth in the volume of outpatient services and that it would be appropriate to apply our volume control method to the services in these APCs.
Comment: A few commenters suggested that, to control unnecessary volume increases, CMS would need to identify a necessary rate of increase for the volume of drug administration services. Absent that benchmark, commenters stated that this is payment reduction rather than a method to control volume under section 1833 (t)(2)(F) of the Act. Commenters were further concerned that CMS has excluded year-to-year monitoring of volume from the proposed policy.
Response: We disagree that we need to determine a specific increase in volume that is necessary before determining that there has been unnecessary increases in volume under section 1833(t)(2)(F) of the Act. Because the effects of site-of-service payment differentials are pervasive throughout the OPPS it would be extremely difficult, on a HCPCs or APC level, to measure what volume should have been absent that influence. Retroactively creating a benchmark would not substantively improve our efforts to measure unnecessary increases in volume. Using a benchmark could lead to similar issues as seen with the Medicare sustainable growth rate (SGR) formula that was used under the Medicare Physician Fee Schedule (MPFS). This formula set annual benchmarks for Medicare physician spending based on growth in the gross domestic product (GDP). The Congress chose to eliminate in the SGR in 2015 after multiple years where they intervened to delay cuts and provide increases to physician payments. Setting a benchmark and then retroactively holding providers to it may actually be harder on providers than simply using a volume control method to prospectively set OPPS rates for these services at a PFS equivalent rate.
We continue to believe shifts of services to the off-campus OPD setting are unnecessary if the beneficiary can safely receive the same services in a lower cost setting but instead receives care in a higher cost setting due to the payment incentives created by the difference in payment amounts. As we noted in the CY 2019 OPPS/ASC final rule with comment period (83 FR 37138 through 37143), we have developed many payment policies, such as packaging policies and comprehensive APCs, to address the rapid growth of services in the OPPS. However, these policies have not been able to control for unnecessary increases in volume that are due to site-of-service payment differentials, which create an incentive to furnish a service in the OPD that could be furnished in a lower cost setting based solely on the higher payment amount available under the OPPS. We previously made the case that the clinic visit service had experienced this shift in site-of-service. We likewise believe that drug administration services furnished in excepted off-campus PBDs are the same as drug administration services furnished in nonexcepted off-campus PBDs. We believe that applying an amount equal to the site-specific PFS payment rate for nonexcepted items and services furnished by a nonexcepted off-campus PBD (the PFS payment rate) for drug administration services, as described by APCs 5691-5694, when provided at an off-campus PBD excepted from section 1833(t)(21) of the Act, is an appropriate method to control the unnecessary increase in the volume of outpatient services. Just as we have monitored the impacts on volume for the clinic visit volume control policy, we will monitor the impacts of this policy on drug administration services.
Comment: A few commenters noted that CMS has previously acknowledged the risk that payment cuts could unintentionally increase volume. Comments cited the CY 2008 OPPS/ASC final rule (72 FR 66580), where CMS declined to adopt a sustainable growth rate (SGR) like methodology. They quoted that final rule:
“implementing such a system could have the potentially undesirable effect of escalating service volume as payment rates stagnate and hospital costs rise, thus actually resulting in a growth in volume rather than providing an incentive to control volume. Therefore, this approach to addressing the volume growth under the OPPS could inadvertently result in the exact opposite of our desired outcome”.
Commenters concluded that subsection 1833 (t)(2)(F) of the Act only authorizes a method for controlling unnecessary increases in volume, following CMS' logic, they stated a policy that increases volume through price reductions would not be a permissible volume-control method.
Response: As discussed above, the Medicare SGR formula was used under the Medicare (MPFS and set annual targets for Medicare physician spending based on growth in the GDP. Under the SGR, if physician spending exceeded its target in a given year, payment rates would be cut the following year, while spending that was below the target led to increased rates. The Medicare Access and CHIP Reauthorization Act of 2015 permanently eliminated the SGR formula for the PFS. In the CY 2008 OPPS/ASC rulemaking we did not propose to adopt a sustainable growth rate (SGR) like methodology for the OPPS. We were referring to the September 8, 1998 proposed rule, proposing the establishment of the OPPS (63 FR 47585), where we did consider creating a system that mirrors the SGR methodology applied to the MPFS update to control unnecessary growth in service volume. While in 1998 we may have contemplated the effects that implementing an SGR methodology might have had on the OPPS, that does not mean that we viewed that any future payment reductions would necessarily result in increases in the volume of services.
In 2008 we were concerned about the continued double digit increases in the growth in expenditures in the OPPS from CY 2001-2008. This was coupled with increases in the volume and intensity of services in that same timeframe that ranged from 3.5 percent to 10.1 percent. In 2008, we stated we were hopeful that expanded packaging and, ultimately, greater bundling under the OPPS may result in sufficient moderation of growth in volume and spending that further volume controls would not be needed. However, we noted that if spending were to continue to escalate at the current rates, even after we have exhausted our options for increased packaging and bundling, we are considering multiple options under our authority to address these issues, including the possibility of imposing external controls that could link growth in volume to reduced payments under the OPPS in the future.
As we noted in the CY 2026 OPPS/ASC proposed rule, from 2011-2019, the time of the greatest provider consolidation and growth of excepted PBDs, the mean and median annual increase in the volume and intensity of hospital outpatient services was about 5.5 percent and 5.4 percent, ( printed page 53816) respectively, from 2011 to 2019. During this time period, the estimated increase in aggregate annual hospital reimbursements incurred through Medicare Fee for Service (FFS) Part B was $28.2 billion.[168] We would contend that spending has continued to increase, especially from 2011 through 2019, and we have done exactly as we stated we would. We have considered and implemented other options under 1833(t)(2)(F) of the Act authority to address unnecessary growth.
Comment: Some commenters stated that the volume control method for clinic visits was not an effective method to control volume as their data analysis indicated that volume did not decrease. One commenter cited a study which found that only 1.5 percent of OPPS payments from 2017 through 2020 were to non-excepted PBDs as evidence that policies that align payment across sites of care do not reduce volume. Commenters opined that since the clinic visit policy did not effectively control volume increases, the drug administration policy should be abandoned. Other commenters asserted that it is not a “method” at all and that it addresses a purported volume increase that the Congress had already addressed under section 603 of the BBA.
Response: Our data indicates that clinic visit volume in excepted off-campus PBDs has decreased since 2021 both in absolute volume and in relative volume compared to the total of OPPS and PFS volume. Additionally, we note that it is difficult to ascertain the impact of a policy such as this without a clear counterfactual. That is to say, we cannot state what the trend of clinic visits at excepted off-campus PBDs would be outside of the existence of this policy. However, given both on-campus and non-excepted PBDs have experienced increases in volume for the clinic visit, while excepted off-campus has experienced a decrease, we believe that our clinic visit volume control method has reduced the unnecessary increases in off-campus PBDs and, likely, overall volume. As we discussed in the CY 2019 OPPS/ASC final rule with comment period, we continue to believe that section 1833(t)(2)(F) of the Act gives the Secretary broad authority to develop a method for controlling unnecessary increases in the volume of covered OPD services, including a method that controls unnecessary volume increases by removing a payment differential that is driving a site-of-service decision, and as a result, is unnecessarily increasing service volume. We believe that applying an amount equal to the site-specific PFS payment rate for nonexcepted items and services furnished by a nonexcepted off-campus PBD (the PFS payment rate) for the clinic visit service, as described by HCPCS code G0463, when provided at an off-campus PBD excepted from section 1833(t)(21) of the Act is an appropriate method to control the unnecessary increase in the volume of outpatient services. Further, we believe this method can be applied to control the unnecessary increases in the volume of drug administration services.
Comment: Some commenters raised concerns related to the use of the PFS relativity adjuster for this volume control method. Commenters indicated that it was not an appropriate unit of payment for services furnished at OPDs as the OPPS payments include far more packaged costs than similar services paid under the PFS. Specifically, commenters indicated that, because the OPPS packages costs, OPPS payment includes things other than the strict payment for the CPT/HCPCS code available under the PFS, and indicated a belief that a relativity adjuster based on a direct comparison would not be appropriate. One commenter stated that over 98.7 percent of the OPPS payment rate is for packaged services and that it would be more accurate to consider the OPPS payment for the chemotherapy administration alone as approximately $5 compared to the $119 paid to physicians' offices. One commenter noted that the estimates presented in the CY 2026 OPPS/ASC proposed rule for the relative difference between the OPPS and PFS payment rate for drug administration codes were generally lower than the 40 percent PFS equivalent payment rate and instead suggested we directly use the technical component of the PFS payment, with some exceptions for packaging.
Other commenters took issue with the overall comparison between OPPS and PFS payment rates as commenters believe they reflect fundamentally different approaches to valuing services. Commenters believe that there is no basis for substituting payment rates from one system to the other. Some commenters were concerned that the Medicare PFS payment rates are not sustainable for physicians. One commenter, a large medical association, stated that while it generally supported site neutral payments, it did not “believe that it is possible to sustain a high-quality health care system if site neutrality is defined as shrinking all payments to the lowest amount paid in any setting”. The commenter went on to state that “CMS should not implement site neutrality in a way that reduces payment to the lowest common denominator and should reinvest savings from lowering facility payments to other Part B services, including payments under the physician fee schedule”.
Response: We appreciate the commenters' concerns about the PFS relativity adjuster. We conducted an analysis for the CY 2026 OPPS/ASC proposed rule which indicated that the 40 percent PFS relativity adjuster was a reasonable equation of OPPS to PFS payments for the same services. This analysis included accounting for packaged services which are separately payable under the PFS. Specifically, when comparing the OPPS and PFS relative payment rates, we reduced the OPPS payment rate used for the analysis by the percentage of that payment rate which we identified as attributable to other HCPCS frequently billed on the same day which would be packaged in the APC. We are uncertain where the one commenter's assertion that 98.7 percent of the OPPS payment rate is packaged costs, our analysis indicated that the amount is generally between 0 and 50 percent based on the specific HCPCS code. Accordingly, we reduced the OPPS payment rate by the percentage attributable to packaged codes which are separately payable under the PFS for our analysis.
While there was some variation in PFS payments for different HCPCS codes (as there is variation in geometric mean costs for HCPCS codes within a single APC under the OPPS) the range of relative costs between the OPPS and the PFS overall for each of the four drug administration APCs was between 24 and 33 percent. This would indicate to us that 40 percent is a reasonable PFS equivalence factor for the purposes of our volume control method. We do not believe it would be appropriate to use the technical component of the PFS payment rate directly as we believe that accounting for frequently packaged services for each APC would be unnecessarily complex. We believe that aligning the payment rate under our volume control method with the payment rate for nonexcepted off-campus PBDs is appropriate as our data indicates that the payment rate is sufficient for off-campus PBDs to continue to provide drug administration services while removing the payment incentives that drive the unnecessary volume this policy is intended to control.
As we stated in the CY 2019 OPPS/ASC final rule with comment period (83 ( printed page 53817) FR 59005), to the extent that similar services can be safely provided in more than one setting, we do not believe it is prudent for the Medicare program to pay more for these services in one setting than another. We believe the increase in the volume of clinic visits, in particular, was due to the payment incentive that exists to provide this service in the higher cost setting. Because these services could likely be safely provided in a lower cost setting, we believed that the growth in clinic visits paid under the OPPS was unnecessary. Further, we believed that setting the OPPS payment at the PFS-equivalent rate would be an effective method to control the volume of these unnecessary services because the payment differential that is driving the site-of-service decision would be removed.
We note that the overall amount of Medicare payments to physicians and other entities made under the PFS is determined by the PFS statute, and the rates for individual services are determined based on the resources involved in furnishing these services relative to other services paid under the PFS. To the extent the commenter believes that the PFS rate for a particular service is misvalued relative to other PFS services, we encourage the commenter to nominate the service for review as a potentially misvalued service under the PFS.
Comment: One commenter discussed the Oncology Care Model and stated a belief that the model's results indicated that payment incentives would not change the volume of drug administration services.
Response: The Center for Medicare & Medicaid Innovation (CMS Innovation Center) develops payment and delivery models designed to improve the effectiveness and efficiency of specialty care. Among those specialty models was the Oncology Care Model, which aimed to provide higher quality, more highly coordinated oncology care at the same or lower cost to Medicare. Under the Oncology Care Model (OCM), physician practices entered into payment arrangements that included financial and performance accountability for episodes of care surrounding chemotherapy administration to cancer patients. CMS also partnered with commercial payers in the model. We do not believe that the structured payment incentives of the OCM serve as a reasonable comparison for the drug administration volume control policy. We believe the clinic visit volume control policy is a more reasonable direct comparison and, as discussed previously, we have strong evidence to suggest that policy did have a material impact on clinic visit volume at excepted off-campus PBDs.
Comment: We received several comments attesting to the continuing negative effects of vertical consolidation, which is the practice of hospitals acquiring physician practices. Commenters stated that the current payment differentials have fueled consolidation by hospitals and health systems, which frequently acquire independent practices to shift infusion services into higher-paying outpatient departments. Commenters believe that this consolidation is further driven by incentives in the 340B program. Between 2012 and 2024, commenters noted, the share of physicians working in a physician-owned practice declined from over 60 percent to 42 percent.[169] Commenters were concerned with the direct impact this shift has continued to have on patients and beneficiaries. Commenters believe that consolidation has led to markets across the country becoming increasingly concentrated, or dominated by a limited number of enterprises. This, they contend, has reduced competition for healthcare services and greater market share by large hospitals and health systems is associated with greater out-of-pocket costs for patients and greater costs to Federal programs, without any improvements in quality.[170] Consolidation, they stated, has been associated with a 4.9 percent increase in Medicare enrollee spending, and a 14.1 percent increase in the price of services in the commercial market. One insurer was particularly concerned about the impact of consolidation on their State's market. They noted that in their State there is a 47 percent higher use of hospital outpatient visits, on a per capita basis, compared to the U.S. average.
Response: We thank the commenters for their thoughts and share their concerns about the ongoing negative effects of vertical integration on the OPPS. Similar to clinic visits, we believe there is a correlation among the increasing volume of OPD drug administration services, vertical integration, and the higher OPPS payment rates for drug administration services.
A 2021 report by the American Medical Association found that more than 50 percent of U.S. physicians are employed by a hospital or health system, a roughly 20 percent increase from 2012.[171] The percentage of physicians in practices owned by hospitals or health systems increased from 23 to 29 percent in 2010 to 44 to 48 percent in 2018, depending on physicians' specialties.[172]
Multiple studies have found increases in prices after vertical consolidation by hospitals acquiring or sharing ownership with physician practice groups.[173 174 175 176 177 178] A key driver of higher prices among vertically consolidated entities is a shift in the site-of-service to higher-cost settings, specifically the on-campus hospital setting or OPD where the hospital can bill for both professional and facility fees. Results from multiple studies suggest that physicians working in hospital-owned practices are more likely to refer patients to hospitals than freestanding facilities.[179 180] Furthermore, studies have found that physicians integrated with hospitals changed their referral patterns, steering more patients to the owning hospitals.[181 182] Results from one study also suggest that those owning hospitals are more likely to be higher-cost, less convenient, and lower-quality options.[183]
( printed page 53818)Similar to the studies of the effect of vertical consolidation on price, multiple studies have found increases in spending associated with vertical consolidation.184 185 186 187 188 189 190 191 192 One study found higher Medicare expenditures for physicians working at acquired practices and for acquiring hospitals following consolidation.[193] Another study found that physicians who primarily practice at hospitals had higher Medicare reimbursement amounts.[194] There continues to be a link between integration and increased costs. Coupling the increases in integration with the migration and growth of drug administration services leads us to believe that the growth of drug administration services in the OPPS represents unnecessary utilization. As we discuss below, we believe that the 340B program has added an additional incentive, outside of OPPS payment rates, for hospitals to acquire physician office practices and shift drug administration services from the lower cost physician office setting to the higher cost OPD.
Comment: Several commenters suggested that drug administration services in particular have experienced shifts in the site-of-service and unnecessary increases in volume due in part to the combination of vertical integration and the 340B program. Commenters asserted that hospital-physician vertical integration is not just fueled by the payment incentives that exist between the two settings. Consolidation, commenters stated, is further driven by incentives in the 340B program, where hospitals profit by acquiring physician offices and converting them into PBDs. Commenters cited studies that found that hospitals newly participating in the 340B program shifted the site of cancer drug administration to OPDs and increased spending on other cancer care.[195] Commenters stated that the shift in the delivery of chemotherapy drugs to outpatient settings also implicates the 340B Drug Pricing program and potential interactions it may have with this proposed policy. This commenter noted a study, which currently is under peer review, that found hospitals newly participating in 340B, compared to non-participating hospitals, led to a higher volume of drug administration services in off-campus OPDs, even though they were subject to site-neutral payments. This could indicate that 340B participation offers hospitals an additional incentive to increase drug administration services at off-campus OPDs even when site-neutral payment is in place.
One commenter stated that 340B eligibility should be prohibited for future off-campus PBDs. This commenter explained that many 340B hospitals have purchased physician practices, retained their pre-existing off-site locations (often even the name of the practice), and commenced providing drug administration services at these “child sites” or off-campus outpatient facilities, which are then eligible for 340B drug discounts from manufacturers. This, the commenter explains, generates revenue for the hospital but does not necessarily benefit patients. They conclude that just as the Congress has closed the loophole for new off-campus PBDs with respect to Medicare reimbursement, it should also close the loophole that allows these off-campus outpatient facilities to benefit from 340B drug pricing—a major driver of hospital acquisition of physician practices and infusion clinics. Specifically, they recommend that any future acquisitions of physician practices by a 340B eligible hospital be ineligible for the 340B program if they continue to treat patients in an off-campus facility that is the same location where the practice provided care prior to the acquisition.
Response: We thank the commenters for their input. The 340B Drug Pricing Program requires pharmaceutical manufacturers to sell outpatient prescription drugs to participating health care facilities at discounted prices. Facilities that participate in the 340B program are hospitals, clinics, and other providers of health care services as well as other organizations that purchase drugs, such as those affiliated with State and local governments. About 90 percent of health care facilities that participate in the 340B program also participate in the Prime Vendor Program (PVP). Through that program, the Health Resources and Services Administration contracts with an external organization, known as the prime vendor, to support 340B operations. These off-site outpatient clinics are often off-campus provide-based departments.
Commenters mainly suggest statutory changes for Congress. They do, however, raise that policies like the 340B program could be contributing to growth in the volume of drug administration services. Our initial review revealed studies that suggest that the 340B program shifted the site of cancer drug administration to OPDs and increased spending on other cancer care.[196] But neither that study nor commenters suggest that the 340B program is responsible for the entire shift in volume from physician offices to OPDs. Instead, this information suggests at most that for drug administration services, the payment incentives might be layered. We address in this rule the incentive to shift volume away from physician practices and into hospital OPDs for drug administration services that could be safely provided in either setting based just on the differences between PFS and OPPS payment for drug administration services. Concerns that the 340B program might further incentivize an additional shift in volume away from physicians practices and towards more expensive hospital OPDs are outside the scope of this rulemaking and would need to be considered in future rulemaking.
Comment: One commenter noted a possible error in CMS' calculation of the estimated effect of the proposed drug administration policy. Commenters stated that they believe there may have been in error in the savings shown in Table 111 of the CY 2026 OPPS/ASC proposed rule. This table shows the estimated annual and 10-year impacts of the proposed drug administration services policy. In particular, commenters were unable to understand the substantial savings that accrue to the Part B Trust Fund for 2027 and future years.
Response: Beginning in 2027, the savings from this policy begin to flow into the baseline for Medicare Advantage rates, thus resulting in a significant increase in savings for 2027 compared to 2026. For 2026, the Medicare Advantage rates had already been calculated at the time of the CY 2026 OPPS/ASC proposed rule and thus the 2026 Medicare Advantage rates are not impacted by this proposed policy.
Comment: Several commenters asserted that CMS fails to consider other explanations for increases in the volume of drug administration services in PBDs. ( printed page 53819) Commenters disagreed that that higher payments for drug administration services under the OPPS are incentivizing hospital acquisition of independent physician offices and, therefore, leading to an “unnecessary increase in the volume of services”. Commenters offered that when hospitals acquire independent physician offices, it is not because there is a financial incentive to do so, but is instead because physician practices are failing. Commenters placed the blame on poor payer mix, increasing regulatory and administrative burden, and declines in payment. Commenters also believe disproportionate attention has been placed on hospitals' acquisition of physician practices. They note that other entities, such as commercial insurers and private equity have invested heavily in physician practice acquisitions. Commenters cite one study that contends that private equity, physician groups, and health insurers, acquired the vast majority of physician practices from 2019 to 2023, while hospitals and health systems accounted for only 6 percent of acquisitions during this period.
Response: As discussed earlier in this section and in the CY 2019 OPPS/ASC final rule with comment period, we continue to believe there is sufficient evidence that consolidation and the payment disparity between the OPPS and PFS rates has driven unnecessary utilization in the OPPS. Hospitals did, in large quantities, acquire physician practices and convert them into PBDs. A financial incentive to make these conversions did exist. While physician practices may be acquired by private equity or commercial insurers, only hospitals are able to move payments to the practice from the PFS to the OPPS as a result of the acquisition. The OPPS was intended to pay for services provided in the hospital outpatient setting, it has no mandate to subsidize care or the cost of providing that care as a result of broader economic forces or the perceived inadequacy of other forms of payment.
We have seen providers react to the financial incentives created by these payment differentials. In January 2019, we began the phase-in of our volume control method for the clinic visit (G0463) and began paying the PFS equivalent rate to excepted PBDs for this service. Non-excepted PBDs were already being paid at the PFS equivalent rate for this service. We found that between 2018-2024 the volume of clinic visit services provided in excepted PBDs billing with modifier “PO” (excluding rural SCHs) decreased by almost 27 percent. This indicates to us that our volume control method was successful and can address unnecessary increases in the volume of outpatient services.
Comment: Commenters stated that CMS inappropriately equates drug administration services provided in OPDs with what they described as less comprehensive and complex care provided in freestanding physician offices. Commenters explained that hospital and health system costs are higher than physician office costs because hospitals invest significant resources to meet the stricter regulatory requirements and safety standards to which they are subject. Commenters contend that the factors below contribute to the increased operating costs for off-campus PBDs compared to physician offices:
- Standby services and emergency care.
- Comprehensive staff and equipment.
- Licensing, accreditation regulatory requirements.
- Drug acquisition and storage.
- Complex patient care.
Therefore, they contend that such care is not equivalent, and current OPPS payment rates appropriately account for these significant differences.
Commenters cited specific measures that hospitals must take to ensure that medications are prepared and administered safely while also providing important care coordination services for their patients. Commenters noted that hospitals must take steps to ensure that a licensed pharmacist supervises drug preparation, rooms are cleaned with positive air pressure to prevent microbial contamination, and employees are protected from exposure to hazardous drugs. In addition they note that hospitals must remain in compliance with important safety standards. Commenters also highlighted the different requirements for safe preparation, administration, care coordination, and oversight that applies to hospitals and physician offices who administer drugs.
Commenters stressed that hospitals and health systems invest significant resources to provide essential benefits and advanced levels of healthcare to their communities. They note hospitals must cover their costs through direct patient care revenue and that OPPS payment rates must be sufficient to support the higher standards of care that CMS and other regulators require to be met in hospital-based settings. Commenters state that these obligations incur additional costs that are not reflected in the MPFS rate, making site-neutral payments inequitable and unsustainable.
Response: We appreciate that some off-campus hospitals outpatient departments sometimes incur costs that physician offices do not. But the issue we are addressing here is whether the difference between OPPS payment rates and PFS payment rates for drug administration services distorts the market and incentivizes providers to shift volume to the higher-paying setting unnecessarily. We note again that we found that drug administration services were not just paid more in the OPD setting, but that the same services were paid 200-300 percent higher under the OPPS than under the PFS. We would note that aspects of the OPD setting that the commenters identify—standby and emergency services; comprehensive staff and equipment; licensing, accreditation regulatory requirements; drug acquisition and storage; and complex patient care—do not change the fact that a specific drug administration service provided in an off-campus provider-based department of a hospital is clinically similar to the provision of that same drug administration service when provided in a physician office. We believe that these drug administration services are safe to perform in the physician office setting and that physician offices also incur costs to adhere to regulatory and safety standards and ensure that they are fully equipped to provide drug administration services. While some hospital off-campus hospital outpatient departments may have additional costs that physician offices do not, we believe that hospitals also have some efficiencies physician offices do not, such as greater purchasing power than physician offices, and likely are able to achieve cost savings through those efficiencies that may offset some of the additional costs identified by commenters. And we do not agree that because hospitals might use profits stemming from increases in the volume of drug administration services to indirectly subsidize other activities that this renders the increase in volume in drug administration services necessary.
Comment: Commenters stated that CMS failed to consider that OPDs are more likely to serve Medicare patients who are sicker, more clinically complex, and more likely to be disabled or living in poorer, rural communities than patients treated in independent physician offices.
Commenters cited two studies which were prepared for the American Hospital Association. The first study commenters cited stated that when comparing beneficiaries treated in OPDs ( printed page 53820) to beneficiaries treated in independent physician offices, beneficiaries receiving care in OPDs are:
- 54 percent more likely to be under 65 and disabled.
- 60 percent more likely to reside in a rural county.
- 61 percent more likely to be dually eligible for Medicare and Medicaid.
- 67 percent more likely to have multiple serious chronic conditions, including heart disease, diabetes, and cancer.
- More likely to have recently used hospital care, including:
++ 73 percent more likely to have prior emergency department visits.
++ 114 percent more likely to have prior inpatient hospitalizations.
The second study cited by commenters compared beneficiaries with cancer treated in OPDs to beneficiaries with cancer treated in physician offices. This study concluded that beneficiaries with cancer receiving care in OPDs are:
- 131 percent more likely to be under 65 and disabled,
- 75 percent more likely to reside in a rural county,
- 125 percent more likely to be dually eligible for Medicare and Medicaid,
- 63 percent more likely to have multiple serious chronic conditions, and
- More likely to have recently used hospital care, including:
++ 63 percent more likely to have prior emergency department visit.
++ 113 percent more likely to have prior inpatient hospitalizations.
Commenters assert that these sicker beneficiaries, who they note are more commonly treated in OPDs, require a greater level of care. Therefore, commenters stated, that to the extent that these differences result in variations in the cost of care, site-neutral payments would have adverse effects on patient access to care.
Response: We appreciate that OPDs serve unique patient populations and provide services to medically complex beneficiaries, however the data provided by the commenter does not demonstrate how these factors necessitate a higher payment for all drug administration services provided in OPDs. While commenters indicate that these patients are more frequently treated in OPDs, they do not show how OPDs alone are clinically capable of treating complex patients or how physician offices would be unequipped to safely treat these same beneficiaries. As we stated in the CY 2019 OPPS/ASC final rule with comment period, we continue to believe shifts in the sites of service described in the preceding paragraphs are inherently unnecessary if the beneficiary can safely receive the same services in a lower cost setting but instead receives care in a higher cost setting due to the payment incentives created by the difference in payment amounts. Further, the OPPS contains mechanisms to pay providers for complex care. Hospitals can receive outlier payments to help hospitals mitigate financial risks associated with complex and costly procedures that exceed the normal payment amount. We also use complexity adjustments to provide increased payment for certain comprehensive services.
Comment: Commenters state that CMS fails to provide any deference to physician's judgment as to the clinical necessity of the OPD setting. Commenters explained that our discussion of the proposed payment reduction for chemotherapy and other drug administration services furnished in excepted, off-campus PBDs too heavily equates outpatient services furnished in physicians' offices and those furnished in hospital outpatient departments.
Commenters stated that aligning payment would treat an OPD and a physician's office as virtually interchangeable. This approach, commenters stated, fails to acknowledge the difference in the resources and level of care offered by hospital outpatient departments and the variability in the acuity and needs of patients undergoing chemotherapy or other drug administrations represented by these codes.
Commenters contend that there are certain services that cannot be furnished in a physician's office, as demonstrated by the fact that there is no non-facility payment rate under the PFS for those services, and there are instances where a physician, in his or her judgment, would determine that a hospital outpatient department, not a physician's office, is the appropriate setting for a particular patient. This decision may be based on the patient's needs, the presence of comorbidities, or a desire for the resources available in an outpatient department. Commenters stated that with respect to drug administration services more specifically, a physician may refer a patient to the hospital for a particular infusion or other drug administration because of the acquisition cost of the drugs, the equipment needed to safely store and mix the drugs, and the inherent danger and complexity that the handling and mixing of chemotherapy drugs poses to the patient and staff.
Response: We thank the commenters for their input; however we believe that payment should have no influence on what setting of care a physician decides is most appropriate for their patient. That decision should be based on the medical needs of the patient. Our proposed policy does not prevent beneficiaries from receiving these services in an OPD if it is medically necessary. Studies examining the role of vertical integration on physicians' choice of care setting show that physicians are much more likely to admit a given patient to their acquiring hospital and that these same hospitals tend to be higher cost, less convenient, and lower quality than nearby options—suggesting negative patient welfare effects.[197] We are simply removing an incentive that unnecessarily increases outpatient volume for drug administration services. Across the OPPS we are finalizing policies, like the elimination of the Inpatient Only List and changes to the Ambulatory Surgical Center Covered Procedures List, to empower physicians to have the ultimate say in what site of service is best for their patients.
Comment: Some commenters were concerned about the financial impact this proposal would have on providers. This reduction, commenters stated, threatens the financial viability of hospitals, particularly those serving underserved communities. Commenters stated that CMS should monitor the impact of this policy on hospitals and the availability of impacted services and develop additional exemption processes if needed.
Response: We appreciate commenters concerns about the financial viability of hospitals and their ability to serve vulnerable populations. As discussed below, we are finalizing our proposal to exempt rural Sole Community Hospitals from this policy as we have previously determined that rural Sole Community Hospitals have higher costs than other outpatient hospitals. We will continue to monitor the effect of this change in Medicare payment policy, including the volume of these types of OPD services, in the future and may revisit this policy in future rulemaking.
Comment: We received several comments suggesting that CMS should phase-in this policy over multiple years and not implement the full payment reduction in CY 2026. Commenters suggested that this would be consistent with the precedent for clinic visit.
Response: While we did phase in the clinic visit payment reduction over 2 years, we do not believe that is necessary for drug administration services. At the time we stated we ( printed page 53821) would use a phase-in to balance the immediate need to address the unnecessary increases in the volume of clinic visits with providers' need for time to adjust to these payment changes. The clinic visit is the most frequently billed service in the OPPS and that weighed heavily in our decision to phase in the implementation of this policy. We believe that the provider community is capable of adapting to the change in payment for drug administration services, which is smaller in magnitude than the clinic visit payment reduction and, as a result, we do not believe that a phase-in is necessary.
After consideration of the public comments we received, we are finalizing our proposal to use our authority under section 1833(t)(2)(F) of the Act to apply an amount equal to the site-specific PFS payment rate for nonexcepted items and services furnished by a nonexcepted off-campus PBD (the PFS payment rate) for HCPCs codes assigned to the drug administration services APCs, when provided at an off-campus PBD excepted from section 1833(t)(21) of the Act (departments that bill the modifier “PO” on claim lines) without modification. In addition, we are finalizing our proposal to implement this policy in a nonbudget neutral manner without modification. We will monitor the impacts of this policy to ensure that beneficiaries continue to have access to quality care.
7. Request for Information: Expanding the Method To Control for Unnecessary Increases in the Volume of Covered OPD Services to On-Campus Clinic Visits
As discussed above, we finalized a method to control unnecessary increases in the volume of covered OPD services under section 1833(t)(2)(F) of the Act in the CY 2019 OPPS/ASC final rule with comment period. This method was to pay the PFS-equivalent payment rate for clinic visit services furnished by excepted off-campus PBDs, removing the payment incentive to furnish clinic visit services in these PBDs. In the above discussion, we note that the volume of covered OPD services is still unnecessarily high for other services, and we proposed a similar policy for drug administration services furnished by excepted off-campus PBDs. For the reasons explained above, we believe that drug administration is the next most appropriate service to include in our method for volume at excepted off-campus PBDs. However, we recognized that the clinic visit is still the most utilized service across the OPPS and over 60 percent of clinic visits furnished under the OPPS are furnished on-campus. These on-campus clinic visits are not impacted by the existing volume control policy. Given the volume for clinic visits is so significant, we requested information on whether it would be appropriate to address unnecessary increases in the volume of covered OPD services by expanding the method to control unnecessary increases in volume to on-campus clinic visits. We requested information on the potential impact of a policy to pay the PFS-equivalent rate of 40 percent of the OPPS rate for clinic visit services furnished in on-campus OPDs. We noted that we intended to use the responses to this request to inform future rulemaking. Specifically, we requested feedback on the following topics:
- Given clinic visits can safely be performed in other, lower cost settings, to what extend are clinic visits performed at OPDs “necessary” or “unnecessary”? Is it appropriate to include on-campus clinic visits when considering how to address unnecessary volume increases at OPDs? How would commenters suggest that CMS could identify which clinic visits may be necessary to be provided on-campus at an OPD? Are there such clinic visits?
- What would be the impact on providers of such a policy? Would any category of hospital be impacted more than others, for example, those in rural areas? Would such a policy result in lower on-campus OPD volume for clinic visits?
- What would be the impact on beneficiaries of such a policy? To what extent would removing any payment incentive from site-of-service determination provide beneficiaries with greater access at sites other than on-campus? To what extent would lower payments for on-campus clinic visits reduce beneficiary access at on-campus OPDs? To what extent would lower co-payments for on-campus clinic visits improve beneficiary access by reducing cost as a potential barrier to care?
- Are there additional costs associated with on-campus clinic visits? If there are additional costs associated with on-campus clinic visits, to what extent could these clinic visits be furnished in a lower-cost setting, for example an off-campus PBD or a physician's office?
- Rural SCHs are excluded from the off-campus clinic visit policy. Should rural SCHs be excluded from any similar on-campus policy? Should any other type of hospital be excluded? Are there any types of hospitals where clinic visits would be more likely to represent “necessary” volume despite being able to be furnished in a lower-cost setting?
We thank commenters for their detailed comments regarding expanding the method to control for unnecessary increases in the volume of covered OPD services. We received a range of comments on expanding the method to the on-campus setting for clinic visits, imagining without contrast in off-campus PBDs, and other services that may have experienced unnecessary growth. We intend to take these comments into consideration as we develop our proposals for future rulemaking.
8. Exemption for Rural Sole Community Hospitals
We proposed to expand our method to control unnecessary increases in the volume of covered OPD services by paying a PFS-equivalent payment rate for drug administration services furnished in excepted off-campus PBDs. As discussed earlier in this section, we believe that this policy is an appropriate method for controlling unnecessary volume of these drug administration services in excepted off-campus PBDs because beneficiaries can generally safely receive these same services in a lower cost setting but instead receive care in a higher cost setting due to payment incentives. In these cases, we explained that, similar to the clinic visit policy established in the CY 2019 OPPS/ASC final rule with comment period (83 FR 37142), to the extent similar services can be safely provided in more than one setting, we do not believe it is prudent for the Medicare program to pay more for these services in one setting than another. We continue to believe the difference in payment for these services is a significant factor in the shift in services from the physician's office setting to the hospital outpatient department.
In the CY 2023 OPPS/ASC final rule with comment period (87 FR 72047 through 72051), we stated that we believe that the volume of the clinic visit service in PBDs of rural Sole Community Hospitals (SCHs) has been driven by factors other than the payment differential for that service. In that rule, we finalized an exemption to our clinic visit volume control method and to instead pay the full OPPS payment rate, rather than the PFS-equivalent rate, when the clinic visit is furnished in excepted PBDs. In that rule, we explained that rural SCHs have historically received special payment treatment to account for their higher costs and the disproportionately harmful impact that payment reductions could have on them. Because we ( printed page 53822) proposed a volume control payment policy for drug administration services, we have additionally considered whether a similar policy for rural SCHs or other provider types would be appropriate.
a. Special Payment Treatment for Rural SCHs
Across the various Medicare payment systems, CMS has established a number of special payment provisions for rural providers to ensure access to high quality care for beneficiaries in rural areas. CMS administers five statutory hospital payment designations in which rural or isolated hospitals that meet specified eligibility criteria receive higher reimbursement for hospital services than they otherwise would receive under Medicare's standard payment methodologies. A rural hospital may qualify as a Critical Access Hospital (CAHs),[198] Sole Community Hospital (SCH),[199] Rural Emergency Hospital (REH),[200] or Medicare Dependent Hospital [201] —each of which has different eligibility criteria and payment methodologies. With the exception of CAHs, rural hospitals may also qualify as Low Volume Hospitals [202] and Rural Referral Centers (RRCs),[203] which qualify these hospitals for additional payments or exemptions. Not all rural or isolated hospitals receive special payment treatment under the OPPS. For instance, CAHs are not paid under the OPPS and are reimbursed at 101 percent of reasonable costs for outpatient services. PBDs of CAHs are not subject to section 603 of the BBA.
Rural SCHs are a hospital type that has received special payment treatment under the OPPS to account for their higher costs and the disproportionately harmful impact that payment reductions could have on them. In the CY 2006 OPPS final rule with comment period (70 FR 68556 through 68561), we finalized a payment increase for rural SCHs of 7.1 percent for all services and procedures paid under the OPPS, excluding separately payable drugs and biologicals, items paid at charges reduced to costs, and devices paid under the pass-through payment policy. This policy was adopted under section 1833(t)(13)(B) of the Act, which required the Secretary, by January 1, 2006, to provide for an appropriate adjustment under paragraph (t)(2)(E) to reflect the higher costs of hospitals in rural areas if the Secretary determined, pursuant to a study required by section 1833(t)(13)(A) of the Act, that the costs to rural hospitals by APC exceeded those costs for hospitals in urban areas. Our analysis revealed that rural SCHs had significantly higher costs per unit than urban hospitals. We have continued to adjust payments for rural SCHs by 7.1 percent each year since 2006. As discussed in section II.E. of this final rule with comment period, for CY 2026 we proposed to continue the current policy of utilizing a 7.1 percent payment adjustment for rural SCHs.
As noted above, in the CY 2023 OPPS/ASC final rule with comment period we finalized an exemption to our policy to pay the PFS-equivalent rate for the clinic visit service at excepted off-campus PBDs to control unnecessary increases in the volume of covered OPD services. Commenters were generally supportive of this proposal and noted that rural SCHs are typically the chief, if not sole, source of community outpatient care for rural residents and opined that this exemption would be vital to ensuring continued access to the care they need. Some commenters stated that the exemption should be extended to other types of hospitals, including urban SCHs. In that rule, we explained that our analysis did not find that urban SCHs had the additional resource costs for covered outpatient department services that rural SCHs have, and only finalized applying the clinic visit policy exemption to rural SCHs.
b. Utilization of Drug Administration Services in Off-Campus Provider-Based Departments of Rurals SCHs
Earlier in this section, where we proposed the volume control method policy for drug administration services, we stated that to the extent there are lower-cost sites of service available, beneficiaries and the physicians treating them should be able to choose the appropriate care setting and not be encouraged to receive or provide care in settings for which payment rates are higher solely for financial reasons. However, many rural providers, and rural SCHs in particular, are often the only source of care in their communities,[204] which means beneficiaries and providers are not choosing between a higher paying off-campus PBD of a hospital and a lower paying physicians' office setting. The closure of inpatient departments of hospitals and the shortage of primary care providers in rural areas likely further drives utilization to off-campus PBDs in areas where rural SCHs are located.
We have reviewed utilization data for drug administration services at rural SCHs and have not found strong evidence that drug administration services are being utilized at an unnecessary volume at excepted off-campus PBDs of rural SCHs. As with clinic visits, we do not believe that rural SCH site-of-service decisions for drug administration are being made solely based on payment rates. Rural areas often experience lower availability of health care professionals and hospitals than urban areas.[205] Hospital closures in rural communities are associated with lower access to health care and worse health outcomes.[206] Access to outpatient services, particularly in rural areas, is vital to keeping beneficiaries from being admitted as an inpatient because beneficiaries in rural settings face unique challenges that impact their health. In the CY 2023 OPPS/ASC final rule with comment period, we explained that we believe that exempting rural SCHs from the clinic visit policy would help to maintain access to care in rural areas by ensuring rural providers are paid for clinic visit services provided at off-campus PBDs at rates comparable to those paid at on-campus departments (87 FR 72049). We believe that a similar exemption is warranted for the drug administration policy for similar reasons. Specifically, we proposed to exempt rural SCHs from payment of the site-specific PFS-equivalent payment for drug administration services, as described by APC family 569X, when furnished at an off-campus PBD exempted from section 1833(t)(21) of the Act (departments that bill the modifier “PO” on claim lines). Under this proposed policy, a rural SCH would continue to bill services in APC family 569X with the “PO” modifier for CY 2026 and the payment rate for such services would continue be the full OPPS payment without the PFS relativity adjustment.
This exemption results in higher payments to excepted off-campus PBDs of rural SCHs compared to if it were not finalized and rural SCHs were subject to the proposed volume control method. The proposed CY 2026 OPPS full payment rates for drug administration APCs 5691, 5692, 5693, and 5694 were $47.83, $74.57, $216.49, $341.52, ( printed page 53823) respectively. The PFS-equivalent rates for these APCs, calculated by applying the 40 percent relativity adjuster to the OPPS payment rates for rural SCHs, would have been $19.13, $29.83, $86.60, $136.61, respectively. By exempting rural SCHs, the Medicare payments for these services would remain at the OPPS level. We estimate that exempting rural SCHs from the method to control unnecessary volume of drug administration services is a difference of approximately $16 million for CY 2026. Per treatment, exempting rural SCHs from this policy results in beneficiary cost sharing remaining between $5.74 and $40.98 higher than it would be should we not finalize this exemption, depending on the service. We note, however, that these figures do not represent increases in costs to Medicare or the beneficiaries above the current policy, as our proposed exemption would maintain current payment rates at excepted off-campus PBDs of rural SCHs of 107.1 percent of the OPPS payment rate for these services. These figures were solely for the purpose of comparing potential savings should we implement a method to control unnecessary volume in drug administration services without such an exemption.
We invited comments on all aspects of the proposed exemption for rural SCHs from the method to control unnecessary volume of drug administration services. Specifically, we requested comments on whether such an exemption is appropriate for rural SCHs, what the impact on SCHs would be should we finalize the method without an exemption for rural SCHs, and whether we should consider any other hospital types for an exemption to either of the policies to control unnecessary volume of outpatient services at off-campus PBDs. Additionally, we requested comments on whether the current exemption for rural SCHs from the method to control unnecessary volume of clinic visit services remains appropriate.
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: The majority of commenters supported our proposal to exempt rural Sole Community Hospitals (rural SCHs) from payment of the site-specific Medicare PFS-equivalent payment for any HCPCs codes assigned to the drug administration services APCs, when furnished at an off-campus PBD excepted from section 1833(t)(21) of the Act (departments that bill the modifier “PO” on claim lines). Several commenters opposed the policy in general but stated that should CMS finalize the volume control method for drug administration services, we should also finalize the proposed exemption for rural SCHs. Some commenters opined on the appropriateness of this exemption and noted that rural SCHs have higher costs and are important for access in rural areas. One commenter discussed urban-rural reclassifications and supported reclassified-rural hospitals receiving the exemption. Only one commenter expressed explicit opposition to the exemption for rural SCHs; this commenter stated that any exemptions to the volume control methodology should be targeted for specific, independent hospitals.
Response: We thank the commenters for their support of the exemption for rural SCHs. As discussed in the CY 2026 OPPS/ASC proposed rule, our historical analysis indicated that rural SCHs have higher costs and we agree that their financial health is vital to ensure continued access to care in rural areas. We acknowledge the nuanced perspective that many commenters took in supporting the proposed exemption while disagreeing with the volume control methodology overall. We appreciate the discussion of reclassifications in regard to this policy and agree that any exemption for rural SCHs would include those SCHs that have reclassified as rural, consistent with our rural SCH exception for the clinic visit volume control method. While we appreciate the one commenter's opinion that any exemption should be targeted, we believe the rural SCH designation generally indicates a hospital has additional costs and is vital for access in rural areas. We disagree with the commenters that exemptions should only be made for independent hospitals, as we believe that rural SCHs, which can be part of a larger chain, also face similarly heightened costs, according to our analysis. We intend to continue to monitor costs at rural SCHs and to ensure that this exemption continues to be warranted both for the clinic visit and drug administration policies.
Comment: Several commenters suggested additional types of hospitals for which they believed further exemptions would be warranted. Such hospital types included urban SCHs, Medicare Dependent Hospitals (MDHs), urban and rural safety-net hospitals, REHs, FQHCs, and all rural hospitals. One commenter noted that there were very few urban SCHs which were not reclassified as rural. Some commenters discussed how hospitals other than rural SCHs can be important for access, generally in rural areas. A few commenters requested a new CMS designation for essential hospitals and requested we except those from the volume control method.
Response: As we discussed in the CY 2026 OPPS/ASC proposed rule, our historical analysis has found that rural SCHs have higher costs which we believe is appropriate to consider when developing a payment policy such as this. Rural SCHs have historically received special treatment under the OPPS due to these higher costs. As we stated in the CY 2023 OPPS/ASC final rule with comment period (87 FR 72047 through 72051) we believe exempting rural SCHs, which have demonstrated additional resource costs, is appropriate to ensure these hospitals can remain open to serve the beneficiaries who rely on them for their care. We share commenters' concerns about the financial difficulties associated with maintaining access to care in medically vulnerable communities. However, in each of these cases, the Congress has not previously determined that any of these hospital types required additional payments for outpatient services.
We proposed a narrow exception to our clinic visit policy largely based upon the historical treatment and documented additional resource costs of rural SCHs under the OPPS. We are only excepting rural SCHs because we continue to believe in the underlying principles of the clinic visit policy—that same rationale continues to justify application of the volume control method for clinic visits to other services. These same principles apply to the exemption for drug administration services. Where the difference in payment is leading to unnecessary increases in the volume of covered outpatient department services, we remain concerned that this shift in care setting increases beneficiary cost-sharing liability because Medicare payment rates for the same or similar services are generally higher in hospital outpatient departments than in physician offices. Further, we do not believe that commenters provided sufficient reasoning or data to show that the other provider types suggested (Medicare Dependent Hospitals, Urban Sole Community Hospitals, Rural Referral Centers, Medicaid DSH, Medicare DSH, and Low-Volume Adjustment Hospitals) demonstrate the additional resource costs that rural SCHs do and should therefore also be exempted from this OPPS payment policy. We share commenters' concerns about maintaining access to care in urban and rural settings and enhancing access to care in medically vulnerable communities. We will continue to study ( printed page 53824) access and cost to see if further exemptions to the volume control policy are appropriate in the future.
Comment: Some commenters suggested other criteria for exemptions which would not be based on hospital type. Several commenters discussed certain drug administration services which they believe should qualify for an exemption, such as biologics which were only given to high acuity patients or cell and gene therapy. One commenter suggested that CMS should exempt drug administration services associated with oncology, immunotherapy, and other high-acuity treatments where they contend immediate access to hospital resources is essential. A different commenter requested exemptions for high-risk patients. Another commenter suggested the volume control methodology should be targeted only for hospitals which do not maintain the same comprehensive services as hospital outpatient departments.
Response: We appreciate the thoughtful suggestions and will consider them for potential future rulemaking. We are concerned that exempting a small subset of services from the volume control methodology could inadvertently incentivize an increase in the provision of those services in the higher cost setting of care. Furthermore, there is no clear clinical justification for exempting certain services while leaving others non-exempted, and we believe it would be most transparent to propose any service-specific exceptions through notice and comment rulemaking. As for the other suggestions related to high risk patients and low service hospitals, we are not certain of how we would identify such patients or hospitals; however, we would appreciate further input these suggestions in the future.
Comment: Several commenters discussed the impact of the proposed method to control volume for drug administration on cancer and children's hospitals. Commenters highlighted that payment for cancer hospitals is based on a payment-to-cost ratio and that lowering payment would indirectly impact these cancer hospitals.
Response: We appreciate the discussion of the impact of this policy on cancer and children's hospitals. We note that any reduced initial OPPS payment to PPS-exempt cancer hospitals as a result of this policy would lower a PPS-exempt cancer hospital's payment-to-cost ratio and would result is an offsetting increased adjustment at cost report settlement. We believe that the impact of this policy on the target payment-to-cost ratio, by lowering non-cancer hospital's payment-to-cost ratios, would be relatively small and that, as we believe the reduced payment for non-cancer hospitals for these services is appropriate, likewise the lower target payment-to-cost ratio would be appropriate.
After consideration of public comments we received, we are finalizing our proposal to exempt rural Sole Community Hospitals (rural SCHs) from payment of the site-specific Medicare PFS-equivalent payment for any HCPCs codes assigned to the drug administration services APCs, when furnished at an off-campus PBD excepted from section 1833(t)(21) of the Act (departments that bill the modifier “PO” on claim lines). We believe that exempting rural SCHs from the volume control policy for drug administration services will help to maintain access to care in rural areas by ensuring rural providers are paid for drug administration services provided at off-campus PBDs at same rate paid when the service is furnished in on-campus departments. Finalizing this policy also aligns with the special payment treatment rural SCHs receive under the OPPS. We will continue to monitor the effects of this change in Medicare payment policy.
B. Request for Information: Adjusting Payment Under the OPPS for Services Predominately Performed in the Ambulatory Surgical Center or Physician Office Settings
In general, Medicare payments to hospital outpatient departments under the Outpatient Prospective Payment System (OPPS) are higher than payments made to ASCs under the ASC payment system or to physician offices under the Physician Fee Schedule (PFS) for the same services. As discussed in section X.A. of this final rule with comment period, CMS has taken steps to address payment disparities between hospital outpatient departments and the physician office setting. While we believe that our regulatory efforts to control for unnecessary utilization in Medicare payments for OPD services has had a positive impact, there is evidence of continued growth in the volume of OPD services driven by site-of-service payment differentials. Building on the CY 2019 OPPS/ASC final rule with comment period policy for clinic visits, in section X.A. of this final rule with comment period we are finalizing our proposal to pay off-campus PBDs otherwise excepted under section 603 of the BBA at the equivalent of the site-specific PFS rate for drug administration services.
While we have implemented volume control policies to pay for certain hospital outpatient clinic visits at a rate closer to that under the PFS and proposed to expand this policy to drug administration services, we sought feedback in the CY 2026 OPPS/ASC proposed rule for future rulemaking on the development of a more systematic process for identifying ambulatory services at high risk of shifting to the hospital setting based on financial incentives rather than medical necessity and adjusting payments according. Specifically, we sought feedback on the following questions:
1. What items and services paid under the OPPS may have experienced unnecessary increases in volume? Should any policies that address those increases be more targeted to those services that have the most notable increases in volume indicative of shifting care from the ASC or physician office setting to the hospital OPD setting?
2. Should we limit OPPS payment for certain services to the payment made for that service under the ASC payment system or the PFS—depending on the setting where the service is performed most frequently? We note that the OPPS currently does not have a payment policy to limit OPPS payment rates to the rate under ASC payment system for procedures that are predominantly performed in an ASC. For example, while a simple cataract removal with insertion of an intraocular lens is a commonly-performed hospital outpatient surgical procedure, 82 percent of all such procedures that Medicare beneficiaries receive are performed in an ASC setting. In general, ASC payment rates are roughly 55 percent of the payment rate under the OPPS (86 FR 63485).
3. If we were to adjust payment based on the setting-specific volume of ambulatory services, should we pay the ASC payment amount if the service is predominantly performed in the ASC setting; and if the service is predominantly performed in the physician office setting, should we continue to calculate the PFS-equivalent rate using a PFS relativity adjuster that we would periodically update?
4. In determining the setting in which a service is performed most frequently, should we use the most recent data available or should we use data that is 5 or even 10 years prior to the rate-setting year? For example, as noted above, the share of chemotherapy administration services billed under the OPPS increased from 35.2 percent to 51.9 percent between 2012 and 2021, so using only more recent data may lead to ( printed page 53825) the conclusion that most of these services take place in the hospital OPD setting, even if that was not historically true. For services that experienced this type of migration, we believe it may be prudent to attempt to address the accumulation of past unnecessary increases in volume rather than allow that shift and the underlying financial incentive that caused it to remain permanent. Should we use solely Medicare FFS data for our analysis or should we explore and potentially incorporate Medicare Advantage data into our work (to the extent feasible and practicable)?
5. How could we account for the availability of OPDs, ASCs, and physician offices in a geographic area when determining the setting in which a service is most frequently performed? If there is a shortage of one of these settings of care in a geographic area, would it be appropriate to tie payment for a service to a setting of care that may not be readily available to a beneficiary?
6. What are the best ways to address different packaging and bundling policies across ambulatory payment systems? The PFS has less packaging of ancillary items than the OPPS and ASC payment system and tends to provide separate payment more frequently. Conversely, certain surgical procedures that have a global code in the PFS may not be packaged in the OPPS or ASC payment systems and the packaging policies of the OPPS and ASC payment system are not based on the period of time elapsed before or after the procedure or service. We could consider retaining the original payment rate that would apply absent any expanded site neutral policies, or we could apply a payment adjustment that approximates the impact of the packaging policies in the payment system whose rate would apply to the item or service under the proposed ambulatory payment adjustment.
7. Should we exempt certain services from a larger site neutral policy if such services are delivered in relation to emergent care, trauma-related care, or other care where the hospital is the most appropriate setting regardless of whether the item or service is typically furnished in a different setting? We note that physicians are appropriately responsible for making site-of-service decisions based on their clinical expertise and may determine that the hospital OPD setting is most appropriate for their patient's circumstances regardless of the level of Medicare payment. We solicited comment on the best way to designate items and services as being emergent or trauma-related and whether to include other categories of care or circumstances where certain items or services would be most appropriately paid at the OPPS rate regardless of the typical setting of care where they are furnished.
8. Should we apply OPPS site neutral policies more broadly to all hospital OPDs or should we instead consider applying this payment adjustment to only certain hospital OPDs, such as excepted off-campus hospital PBDs?
9. Should we exempt certain types of hospitals from a larger site neutral policy, such as rural Sole Community Hospitals, Medicare Dependent Hospitals, or Rural Emergency Hospitals? Currently, rural Sole Community Hospitals are exempted from the clinic visit site neutrality policy and instead are paid the full OPPS rate when such visits are furnished in excepted off-campus PBDs of these hospitals.
10. What other methods may be warranted to control unnecessary increases in the volume of outpatient services besides changes to payment rates, including prior authorization or other utilization management policies?
11. What impact would the proposed ambulatory payment adjustment have on beneficiaries and the health care market, including the development of or beneficiary access to new health care innovations?
We received approximately 43 pieces of correspondence that were submitted in response to the RFI questions. We thank all interested parties for their comments and may take them into consideration for future rulemaking.
C. Virtual Direct Supervision of Cardiac Rehabilitation (CR), Intensive Cardiac Rehabilitation (ICR), Pulmonary Rehabilitation (PR) Services and Diagnostic Services Furnished to Hospital Outpatients
1. Background
a. Virtual Direct Supervision of CR, ICR and PR Services Furnished to Hospital Outpatients (42 CFR 410.27(a)(1)(iv)(B)( 1))
In the interim final rule with comment period titled “Policy and Regulatory Provisions in Response to the COVID-19 Public Health Emergency,” published on April 6, 2020 (the April 6th COVID-19 IFC) (85 FR 19230, 19246, 19286), we changed the regulation at 42 CFR 410.27(a)(1)(iv)(D) [207] to provide that, during a Public Health Emergency (PHE) as defined in 42 CFR 400.200, the presence of the physician for purposes of the direct supervision requirement for PR, CR, and ICR services includes virtual presence through audio/video real-time communications technology when use of such technology is indicated to reduce exposure risks for the beneficiary or health care provider. Specifically, the required direct physician supervision can be provided through virtual presence using audio/video real-time communications technology (excluding audio-only) subject to the clinical judgment of the supervising practitioner. We further amended § 410.27(a)(1)(iv)(B) [208] in the CY 2021 OPPS/ASC final rule with comment period to provide that this flexibility continues until the later of the end of the calendar year in which the PHE as defined in § 400.200 ends or December 31, 2021 (85 FR 86113 and 86299). In the CY 2021 OPPS/ASC final rule with comment period we also clarified that this flexibility excluded the presence of the supervising practitioner via audio-only telecommunications technology (85 FR 86113).
In the CY 2023 OPPS/ASC final rule with comment period, we finalized a policy to extend the revised definition of direct supervision of CR, ICR, and PR services to include the presence of the supervising physician through two-way, audio/video telecommunications technology (excluding audio-only) until December 31, 2023 (87 FR 72019 and 72020).
In the CY 2024 OPPS/ASC final rule with comment period, we finalized a policy to further revise § 410.27(a)(1)(iv)(B)( 1) to continue to allow for the direct supervision requirement for CR, ICR, and PR services to include the virtual presence of the physician through audio-video real-time communications technology (excluding audio-only) through December 31, 2024 and to extend this policy to the nonphysician practitioners, that is NPs, PAs, and CNSs, who were eligible to supervise these services beginning in CY 2024 (88 FR 81863 through 81867).
In the CY 2025 OPPS/ASC final rule with comment period, we finalized a policy to continue to allow for the direct supervision of CR, ICR, PR services to include the virtual presence of the physician (or other nonphysician practitioner) through audio-video real- ( printed page 53826) time communications technology (excluding audio-only) through December 31, 2025 (89 FR 94280).
b. Virtual Direct Supervision of Diagnostic Services Furnished to Hospital Outpatients (42 CFR 410.28(e)(2)(iii))
In the April 6th, 2020 COVID-19 IFC, for consistency with the revisions made to 42 CFR 410.27(a)(1)(iv)(D) [209] described above and § 410.32(b)(3)(ii) (revising the definition of direct supervision of diagnostic services furnished in a physician's office to include virtual supervision for the duration of the PHE), we changed the regulation at 42 CFR 410.28(e) to provide that, during a PHE as defined in 42 CFR 400.200, the presence of the physician for purposes of the direct supervision requirement for diagnostic services includes virtual presence through audio/video real-time communications technology when use of such technology is indicated to reduce exposure risks for the beneficiary or health care provider (85 FR 19245 and 19246).
To ensure consistency with additional revisions made to 42 CFR 410.27(a)(1)(iv)(B)( 1) and 410.32(b)(3)(ii) extending the end date of the flexibility allowing for the virtual supervision of the services governed by those regulations, the CY 2023 OPPS/ASC final rule with comment period (87 FR 72024 through 72026), CY 2024 OPPS/ASC final rule with comment period (88 FR 81866 and 81867), and CY 2025 OPPS/ASC final rule with comment period (89 FR 94278 and 94280) subsequently extended the end date of the flexibility allowing for direct supervision to include the virtual supervision of outpatient diagnostic services through audio/video real-time communications technology (excluding audio-only) through December 31, 2025.
2. CY 2026 Virtual Direct Supervision of CR, ICR, PR Services and Diagnostic Services Furnished to Hospital Outpatients
In the CY 2026 PFS proposed rule (90 FR 32393 through 32395), we proposed to revise the definition of direct supervision at § 410.26(a)(2) and § 410.32(b)(3)(ii) to make permanent the availability of virtual direct supervision of therapeutic and diagnostic services under the PFS, except for services that have a global surgery indicator of 010 or 090. This information can be found in the PFS PPRVU public use file ( https://www.cms.gov/medicare/payment/fee-schedules/physician/pfs-relative-value-files). These global surgery indicators are defined in IOM Pub. 100-04, chapter 23, section 50.6 as 010 “Minor procedure with preoperative relative values on the day of the procedure and postoperative relative values during a 10-day postoperative period included in the fee schedule amount; evaluation and management services on the day of the procedure and during this 10-day postoperative period generally not payable” and 090 “Major surgery with a 1-day preoperative period and 90-day postoperative period included in the fee schedule payment amount”. As explained in that rule, this proposal was made in response to overwhelming support and requests to extend this policy permanently for a wider set of services than the ones that were finalized in the CY 2025 PFS final rule and would build on the incremental approach of making the virtual supervision of certain services permanent which we began in the CY 2025 PFS final rule. As noted in the CY 2026 PFS proposed rule, we believed that adopting this approach would recognize that virtual supervision has been available and widely utilized since the beginning of the PHE while excluding certain services to ensure quality of care and patient safety, and in particular, the ability of the supervising practitioner to intervene if complications arise, particularly in complex, high-risk instances where unexpected or adverse events may occur or for procedures that may be riskier or more intense since a patient's clinical status can quickly change. For the complete discussion of the proposed revisions to § 410.26(a)(2) and § 410.32(b)(3)(ii), we refer readers to the CY 2026 PFS proposed rule (90 FR 32393 through 32395).
In addition to desiring uniformity under the PFS and OPPS in how regulations are applied to similarly situated clinicians and providers, we agreed in the CY 2026 OPPS proposed rule that the approach proposed in the CY 2026 PFS proposed rule struck the appropriate balance between recognizing that the virtual supervision of diagnostic services has been available and widely utilized since the beginning of the PHE and ensuring quality of care and patient safety. Consequently, we proposed to revise § 410.27(a)(1)(iv)(B)( 1) and § 410.28(e)(2)(iii) to make the availability of the direct supervision of CR, ICR, PR services and diagnostic services via audio-video real-time communications technology (excluding audio-only) permanent, except for diagnostic services that have a global surgery indicator of 010 or 090. We noted in the CY 2026 OPPS/ASC proposed rule that permanently adopting a definition of direct supervision that allows “immediate availability” of the supervising practitioner using audio/video real-time communications technology (excluding audio-only), for CR, ICR, PR and diagnostic services described under § 410.28, except for diagnostic services that have a global surgery indicator of 010 or 090 did not mean that it was appropriate to allow virtual presence for every service for every Medicare beneficiary in every clinical scenario. As always, we stated, the physician or nonphysician practitioner should use his or her complex professional judgment to determine the appropriate supervision modality on a case-by-case basis.
We received public comments on this proposal. The following is a summary of the comments we received and our responses to those comments.
Comment: Nearly all commenters supported our proposal to revise § 410.27(a)(1)(iv)(B)( 1) and § 410.28(e)(2)(iii) to make the availability of the direct supervision of CR, ICR, PR services and diagnostic services via audio-video real-time communications technology (excluding audio-only) permanent, except for diagnostic services that have a global surgery indicator of 010 or 090. These commenters stated that virtual direct supervision does not inherently give rise to patient safety issues and indicated that making the availability of virtual direct supervision permanent would improve patient access to historically underutilized services, particularly in rural and other underserved areas where workforce shortages remain acute, enhance workforce efficiency, reduce burden on providers, provide the certainty needed to maintain existing programs and encourage new adoption by rural providers who may have been hesitant. Several of these commenters suggested additional refinements to the proposal such as retiring the requirement for a service-level modifier to identify when direct supervision is provided via appropriate telehealth technology, more precisely defining “immediate availability” in the context of virtual direct supervision, adding a requirement that virtual supervision of CR and ICR services be preceded by an in-person E/M consultation for the development of a care plan for which CR or ICR services will be rendered and ensuring that trained and authorized ( printed page 53827) staff are present in the event of an adverse reaction to injected contrast.
Response: We thank commenters for their support and suggestions of how we might refine our policy and will take their input under consideration for future rulemaking.
Comment: One commenter opposed the proposal. Reiterating concerns previously expressed in comments to the CY 2025 OPPS/ASC final rule with comment period, the commenter opposed our proposal to permanently allow for the virtual direct supervision of CR, ICR, PR and diagnostic services because doing so would increase the amount of physician “incident to” billing (a Medicare billing provision that applies in the office or clinic setting and allows medical services to be performed by auxiliary personnel as an incident to the services of the billing practitioner and under their supervision) for services provided by Physician Assistants (PAs) and Nurse Practitioners (NPs), which would obscure the extent to which PAs and NPs are actually performing the services. This commenter suggested that CMS allow for virtual supervision only of medical professionals who are unauthorized to bill Medicare or, alternatively, establish a method through which CMS is able to collect the information of the health professional actually providing the service under “incident to.”
Response: We appreciate the commenter's input regarding the appropriate attribution of services performed by PAs and NPs when those services are billed “incident to” a physician's service. However, we note that these nonphysician practitioners (as well as clinical nurse specialists (CNSs)) are authorized by statute to supervise these services and we think that any potential obscuration of the extent to which PAs and NPs are providing virtual direct supervision resulting from incident to billing is outweighed by the flexibility and enhanced access to services resulting from allowing these practitioners to provide virtual direct supervision. We are also not persuaded that the commenter's concerns warrant CMS establishing a method through which we would collect the information of the health professional actually providing the service under “incident to.”
Comment: Several commenters requested that CMS make clear that Advanced Practice Registered Nurses can conduct virtual direct supervision of rehabilitation services in hospital outpatient settings.
Response: PAs, NPs and CNSs can conduct virtual direct supervision of CR, ICR and PR in hospital outpatient settings. Section 410.27(a)(1)(iv)(B)( 1) defines direct supervision for the purpose of therapeutic services provided in the outpatient setting and states that “[f]or pulmonary rehabilitation, cardiac rehabilitation, and intensive cardiac rehabilitation services, direct supervision must be furnished as specified in §§ 410.47 and 410.49, respectively.” Sections 410.47(a) and 410.49(a), specifically list the individual practitioners (PA, NP, and CNS) that are included in the term “nonphysician practitioner” for the purposes of the supervision of ICR, CR and PR.
Comment: One commenter requested that CMS continue to monitor the impact of virtual direct supervision to ensure it supports high quality, safe and accessible diagnostic care for all beneficiaries. Another commenter suggested that CMS monitor outcomes such as patient satisfaction, program completion rates, and safety indicators to ensure quality is maintained.
Response: We appreciate the commenters' concerns about monitoring the impact of virtual direct supervision to ensure quality of care. We plan on continuing to do so.
Comment: Several commenters, as they have in comments to previous proposed rules, advocated for the removal of what they refer to as referral barriers to certain non-physician practitioners ordering CR, ICR, PR. These commenters point out that while NPs, PAs and CNSs can supervise CR, ICR and PR services, they cannot order those services for Medicare patients. These commenters identify CMS' previous interpretation of “physician-prescribed exercise” under section 1861(fff)(2)(A) of the Act (for PR) and section 1861(eee)(3)(A) of the Act (for CR/ICR) as the barrier preventing NA, PAs and CNSs from referring patients to CR, ICR, and PR services and stated that CMS should change its interpretation of these sections of the statute to allow these practitioners to order CR, ICR, and PR services. One commenter stated that CMS can interpret “physician-prescribed” to allow NAs, PAs and CNSs to order CR, ICR and PR services because there is a distinction between the referral for physician-prescribed exercise and the physician's prescription for exercise.
Response: As we have explained previously in the CY 2024 OPPS/ASC final rule with comment period (88 FR 81864 through 81865) and CY 2024 PFS final rule (88 FR 79088), we do not believe that there is a reasonable interpretation of the statute that would allow NAs, PAs and CNSs to order CR, ICR, and PR services. We encourage interested parties to work with the Congress to explore further statutory changes to support these requests.
Comment: Several commenters encouraged CMS to consider additional services that could be safely supervised virtually in future years, additional policies that might support virtual care in the hospital outpatient setting and the possibility of reinstating the virtual delivery of CR, ICR, and PR services furnished by hospital outpatient departments.
Response: We thank the commenter for their input and will consider taking it under consideration in future rulemaking.
Comment: One commenter requested that CMS extend the definition of direct supervision to include real-time virtual presence via audio/video technology for all therapeutic radiation therapy services for sites of service paid under the OPPS and MPFS.
Response: We thank the commenter for their comment and note that for therapeutic services under § 410.27, ICR, CR and PR, are the only services that are subject to direct supervision requirements when furnished to hospital outpatients.
After consideration of the public comments we received, we are finalizing, without modification, our proposal to revise § 410.27(a)(1)(iv)(B)( 1) and § 410.28(e)(2)(iii) to make the availability of the direct supervision of CR, ICR, PR services and diagnostic services via audio-video real-time communications technology (excluding audio-only) permanent, except for diagnostic services that have a global surgery indicator of 010 or 090.
D. Medical Review of Certain Inpatient Hospital Admissions Under Medicare Part A for CY 2026 and Subsequent Years
1. Background on the 2-Midnight Rule
In the FY 2014 IPPS/LTCH PPS final rule (78 FR 50944 through 50952), we clarified our policy regarding when an inpatient admission is considered reasonable and necessary for purposes of Medicare Part A payment. Under this policy, we established a benchmark providing that surgical procedures, diagnostic tests, and other treatments would be generally considered appropriate for payment under Medicare Part A when the physician expects the patient to require a stay that crosses at least 2 midnights and admits the patient as an inpatient based upon that expectation. Conversely, when a beneficiary enters a hospital for a surgical procedure not designated as an ( printed page 53828) inpatient-only (IPO) procedure as described in 42 CFR 419.22(n), a diagnostic test, or any other treatment, and the physician expects to keep the beneficiary in the hospital for only a limited period of time that does not cross 2 midnights, the services would be generally inappropriate for payment under Medicare Part A, regardless of the hour that the beneficiary came to the hospital or whether the beneficiary used a bed. With respect to services designated under the OPPS as IPO procedures, we explained that because of the intrinsic risks, recovery impacts, or complexities associated with such services, these procedures would continue to be appropriate for payment under Medicare Part A regardless of the expected length of stay. We also indicated that there might be further “rare and unusual” exceptions to the application of the benchmark, which would be detailed in subregulatory guidance.
In the FY 2014 IPPS/LTCH PPS final rule (78 FR 50944 through 50952), we also finalized the 2-midnight presumption, which is related to the 2-midnight benchmark but is a separate medical review policy. The 2-midnight benchmark represents guidance to reviewers to identify when an inpatient admission is generally reasonable and necessary for purposes of Medicare Part A payment, while the 2-midnight presumption relates to instructions to medical reviewers regarding the selection of claims for medical review. Specifically, under the 2-midnight presumption, inpatient hospital claims with lengths of stay greater than 2 midnights after the formal admission following the order are presumed to be appropriate for Medicare Part A payment and are not the focus of medical review efforts, absent evidence of systematic gaming, abuse, or delays in the provision of care in an attempt to qualify for the 2-midnight presumption. We refer readers to the CY 2021 OPPS/ASC final rule with comment period for additional discussion about the distinction between the 2-midnight presumption and benchmark (85 FR 86113 through 86114).
In the CY 2016 OPPS/ASC final rule with comment period (80 FR 70538 through 70545), we revisited the previous rare and unusual exceptions policy and finalized a proposal to allow for case-by-case exceptions to the 2-midnight benchmark, whereby Medicare Part A payment may be made for inpatient admissions where the admitting physician does not expect the patient to require hospital care spanning 2 midnights, if the documentation in the medical record supports the physician's determination that the patient nonetheless requires inpatient hospital care. We stated that the following criteria would be relevant to determining whether an inpatient admission with an expected length of stay of less than 2 midnights is nonetheless appropriate for Medicare Part A payment:
- Complex medical factors such as history and comorbidities;
- The severity of signs and symptoms;
- Current medical needs; and
- The risk of an adverse event.
In other words, for purposes of Medicare payment, an inpatient admission is payable under Part A if the documentation in the medical record supports either the admitting physician's reasonable expectation that the patient will require hospital care spanning at least 2 midnights, or the physician's determination based on factors such as those identified previously that the patient nonetheless requires care on an inpatient basis. The exceptions for procedures on the IPO list and for “rare and unusual” circumstances designated by CMS as national exceptions were unchanged by the CY 2016 OPPS/ASC final rule with comment period.
As we stated in the CY 2016 OPPS/ASC final rule with comment period, the decision to formally admit a patient to the hospital is subject to medical review. Specifically, for inpatient admissions not related to a surgical procedure specified by Medicare as an IPO procedure under § 419.22(n) and for which there is not a national exception, payment of the claim under Medicare Part A is subject to the clinical judgment of the medical reviewer to determine whether the medical record supports a reasonable expectation of the need for hospital care crossing at least 2 midnights or otherwise supports a need for inpatient care. The medical reviewer's clinical judgment involves the synthesis of all submitted medical record information (for example, progress notes, diagnostic findings, medications, nursing notes, and other supporting documentation) to make a medical review determination on whether the clinical requirements in the relevant policy have been met. In addition, Medicare review contractors must abide by CMS' policies in making payment determinations. While Medicare review contractors may continue to use commercial screening tools to help evaluate the inpatient admission decision for purposes of payment under Medicare Part A, such tools are not binding on the hospital, CMS, or its review contractors. This type of information also may be appropriately considered by the physician as part of the complex medical judgment that guides his or her decision to keep a beneficiary in the hospital and formulation of the expected length of stay.
2. Current Policy for Medical Review of Inpatient Hospital Admissions for Procedures Removed From the Inpatient Only List
In the CY 2020 OPPS/ASC final rule with comment period, we finalized a policy to exempt procedures that have been removed from the IPO list from certain medical review activities to assess compliance with the 2-midnight rule within the 2 calendar years following their removal from the IPO list. We stated that these procedures would be exempted from site-of-service claim denials under Medicare Part A, eligibility for Beneficiary and Family-Centered Care Quality Improvement Organizations (BFCC-QIOs) referrals to Recovery Audit Contractors (RACs) for noncompliance with the 2-midnight rule, and RAC reviews for “patient status” (that is, site-of-service). We explained that during this 2-year period, BFCC-QIOs would have the opportunity to review such claims in order to provide education for practitioners and providers regarding compliance with the 2-midnight rule, but claims identified as noncompliant would not be denied with respect to the site-of-service under Medicare Part A.
For CY 2021, in conjunction with our proposal to eliminate the IPO list, we modified our proposal to continue the 2-year exemption, and instead finalized a policy under which procedures removed from the IPO list on or after January 1, 2021, would be indefinitely exempted from the above described medical review activities. We explained that the elimination of the IPO list was a large-scale change that created brand new considerations for providers regarding site-of-service determinations. We believed a change of this significance required us to reevaluate our stance on the exemption period for procedures removed from the IPO list, resulting in our decision to finalize an indefinite exemption period rather than continuing the previous 2-year exemption period. We stated that this exemption would last with respect to each procedure removed from the IPO list until we had Medicare claims data indicating that the procedure was more commonly performed in the outpatient setting than the inpatient setting. Thus, for the exemption to end for a specific procedure, in a single calendar year we would need to have Medicare claims ( printed page 53829) data indicating that the procedure was performed more than 50 percent of the time in the outpatient setting. We noted that the end of the exemption period for each procedure removed from the IPO list on or after January 1, 2021 would be announced via rulemaking.
Consequently, in the CY 2021 OPPS/ASC final rule with comment period, we amended 42 CFR 412.3(d)(2) to clarify when a procedure removed from the IPO list is exempt from the identified medical review activities. To account for the previous exemption policy that was in effect for CY 2020, we added § 412.3(d)(2)(i) which stated that for “those services and procedures removed between January 1 and December 31, 2020, this exemption will last for 2 years from the date of such removal.” To implement the change to an indefinite exemption period that we finalized in CY 2021, we added § 412.3(d)(2)(ii) which stated that for “those services and procedures removed on or after January 1, 2021, this exemption will last until the Secretary determines that the service or procedure is more commonly performed in the outpatient setting.”
In the CY 2022 OPPS/ASC final rule with comment period (86 FR 63736 through 63740), given our decision in that rule to halt the elimination of the IPO list, and the fact that we were accordingly no longer removing an unprecedented number of procedures from the list at one time, we proposed to return to the 2-year exemption period from the specified medical review activities for procedures removed from the IPO list. Under the circumstances of that final rule with comment period, we believed that a 2-year exemption period was adequate to enable providers to gain experience with the application of the 2-midnight rule to those procedures that have been newly removed from the IPO list. We also stated that we believed that a 2-year exemption from the medical review activities was also sufficient time for providers and BFCC-QIOs to understand the documentation necessary to support Part A payment for those patients for which the admitting physician determines that the procedures should be furnished in an inpatient setting.
In the preamble to the CY 2022 OPPS/ASC final rule with comment period (86 FR 63739), we stated that we were amending § 412.3(d)(2) to clarify “that for all services and procedures removed after January 1, 2020, this exemption would last for 2 years from the date of such removal. This would include those services and procedures removed on or after January 1, 2021, for which this exemption would also be for 2 years from the date of such removal”. Accordingly, § 412.3(d)(2)(i) was revised to read “for those services and procedures removed on or after January 1, 2020, the exemption in this paragraph (d)(2) will last for 2 years from the date of such removal.” However, due to a drafting oversight, we failed to correspondingly remove § 412.3(d)(2)(ii): “For those services and procedures removed on or after January 1, 2021, the exemption in this paragraph (d)(2) will last until the Secretary determines that the service or procedure is more commonly performed in the outpatient setting.” As a result of this error, the exemption period was not changed to two years as we intended and instead remained the indefinite exemption period that was finalized in the CY 2021 OPPS/ASC final rule with comment period.
3. Medical Review of Inpatient Hospital Admissions for Procedures Removed From the Inpatient Only List for CY 2026 and Subsequent Years
As stated earlier in this section, services on the IPO list are not subject to the 2-midnight rule for purposes of determining whether payment is appropriate under Medicare Part A. However, the 2-midnight rule is applicable once services have been removed from the IPO list. Outside of the exemption periods discussed above, services that have been removed from the IPO list are currently subject to initial medical reviews of claims for short-stay inpatient admissions conducted by Medicare review contractors.[210]
MACs' current typical process is to perform reviews on a sample of Medicare pre-payment Part A claims as part of the MAC Targeted Probe and Educate (TPE) program. For more details on this program see: https://www.cms.gov/data-research/monitoring-programs/medicare-fee-service-compliance-programs/medical-review-and-education/targeted-probe-and-educate-tpe. MACs may refer any provider that fails to improve after three rounds of targeted audits and education to CMS for next steps. CMS actions may include additional prepay review, extrapolation, referral to a Recovery Auditor, or other action.
However, as finalized in the CY 2021 OPPS/ASC final rule with comment period, procedures that have been removed from the IPO list on January 1, 2021 or later were indefinitely exempted from site-of-service claim denials under Medicare Part A, eligibility for referrals to RACs for noncompliance with the 2-midnight rule, and RAC reviews for “patient status” (that is, site-of-service). We stated that this exemption would last for each procedure until we have Medicare claims data indicating that the procedure is more commonly performed in the outpatient setting than the inpatient setting.
As stated in section IX. of the CY 2026 OPPS/ASC proposed rule, we proposed to eliminate the IPO list in CY 2026 with a transitional period of 3 years. For CY 2026, we proposed to remove all musculoskeletal procedures from the IPO list. Prior to the CY 2020 exemption for services removed from the IPO list, the elimination of the IPO list would have meant that procedures currently on the IPO list would be subject to the 2-midnight rule (both the 2-midnight benchmark and 2-midnight presumption) upon removal from the IPO list.
We believe that with the elimination of the IPO list, which we are finalizing, as discussed in section IX. of this final rule with comment period, the 2-midnight benchmark remains an important metric to help guide when Part A payment for inpatient hospital admissions is appropriate. As technology advances and more services may be safely performed in the hospital outpatient setting and paid under the OPPS, it is increasingly important for physicians to exercise their clinical judgment in determining the appropriate clinical setting for their patient to receive a procedure, whether that be as an inpatient or on an outpatient basis. Importantly, removal of a service from the IPO list has never meant that a beneficiary cannot receive the service as a hospital inpatient—as always, the physician should use his or her complex medical judgment to determine the appropriate setting on a case-by-case basis.
As finalized in the CY 2021 OPPS/ASC final rule with comment period, procedures removed from the IPO list after January 1, 2021, were indefinitely exempted from site-of-service claim denials under Medicare Part A, ( printed page 53830) eligibility for Medicare review contractor referrals to RACs for noncompliance with the 2-midnight rule, and RAC reviews for “patient status” (that is, site-of-service). These procedures are not considered by the Medicare review contractors in determining whether a provider exhibits persistent noncompliance with the 2-midnight rule for purposes of referral to the RAC nor will claims for these procedures be reviewed by RACs for “patient status.” During the exemption period, Medicare review contractors have the opportunity to review such claims in order to provide education for practitioners and providers regarding compliance with the 2-midnight rule, but claims identified as noncompliant are not denied with respect to the site-of-service under Medicare Part A. Again, information gathered by the Medicare review contractor when reviewing procedures as they are newly removed from the IPO list can be used for educational purposes but will not result in a claim denial during the exemption period.
When we previously finalized elimination of the IPO list in the CY 2021 OPPS/ASC final rule with comment period, we received numerous comments that suggested a longer exemption period would be appropriate, due to the unprecedented volume of procedures becoming subject to the 2-midnight rule. Therefore, we finalized an indefinite exemption period for procedures removed from the IPO list during the 3-year transition from the list to allow providers to become more familiar with how to comply with the 2-midnight rule and with the availability of payment under both the hospital inpatient and outpatient payment systems for procedures removed from the IPO list. Our proposal in the CY 2026 OPPS/ASC proposed rule to eliminate the IPO list over a 3-year period warranted similar considerations. Accordingly, we proposed to maintain the indefinite exemption period under 42 CFR 412.3(d)(2)(ii) for procedures that are removed from the IPO list that is currently in effect. In the interest of clarity, we proposed to delete § 412.3(d)(2)(i) and (ii) and revise § 412.3(d)(2) to read “An inpatient admission for a surgical procedure specified by Medicare as inpatient only under § 419.22(n) of this chapter is generally appropriate for payment under Medicare Part A regardless of the expected duration of care. Procedures no longer specified as inpatient only under § 419.22(n) of this chapter are appropriate for payment under Medicare Part A in accordance with paragraph (d)(1) or (3) of this section. Claims for services and procedures removed from the inpatient only list under § 419.22 of this chapter on or after January 1, 2021 are exempt from certain medical review activities until the Secretary determines that the service or procedure is more commonly performed in the outpatient setting.” As indicated in the CY 2021 OPPS/ASC final rule with comment period, the determination of the Secretary that a service or procedure is more commonly performed in the outpatient setting is based on claims data that demonstrates that the service or procedure is being performed more than 50 percent of the time in the outpatient setting in a single calendar year (85 FR 86117 and 86119). We noted in the CY 2026 OPPS/ASC proposed rule, that this would be an exemption from certain medical review activities, not an exception to the 2-midnight rule. Providers are still required to comply with the 2-midnight rule during the exemption period, and CMS or its contractors may still conduct patient status medical review in cases in which there is evidence of systemic fraud or abuse occurring. Additionally, we noted that other types of medical review, unrelated to patient status, would not be impacted by the proposed exemption. We proposed to announce in subregulatory guidance when the exemption is ending for a particular service or procedure prior to the effective date of the end of the exemption for the particular service or procedure. We invited commenters to indicate whether and why they believed an indefinite exemption period, or another time period, would be most appropriate.
In summary, for CY 2026 and subsequent years, we proposed to continue the indefinite exemption from site-of-service claim denials, initial medical review contractor referrals to RACs, and RAC reviews for “patient status” (that is, site-of-service) finalized in the CY 2021 OPPS/ASC final rule with comment period for procedures that are removed from the IPO list in CY 2021 or later under the OPPS. We also proposed to remove § 412.3(d)(2)(i) and (ii) and revise § 412.3(d)(2) to clarify that claims for services and procedures removed from the IPO list on or after January 1, 2021 are exempt from certain medical review activities until the Secretary determines that the service or procedure is more commonly performed in the outpatient setting than the inpatient setting. Finally, we sought comment on whether other exemption periods may be warranted.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: Many commenters supported our proposal to continue the indefinite exemption from site-of-service claim denials, initial medical review contractor referrals to RACs, and RAC reviews for “patient status” (that is, site-of-service) finalized in the CY 2021 OPPS/ASC final rule with comment period for procedures that are removed from the IPO list in CY 2021 or later under the OPPS. Some of these commenters stated that maintaining this exemption would help ensure that providers are not penalized for exercising clinical discretion and alleviate concerns regarding 2-midnight audits and claims denials from influencing the site of-surgery. One of these commenters stated that by acknowledging the foremost expertise of physicians in determining the most appropriate site of care through this indefinite exemption along with other existing policies related to the 2-midnight rule, CMS is taking an important step toward increasing choice and improving quality of care. Other commenters noted that the proposal would facilitate a smoother transition for procedures newly eligible for outpatient reimbursement, allow time for practice patterns and infrastructure to adapt, reduce confusion and avoid additional, unnecessary administrative burden on physicians. One commenter indicated that the exemption period was necessary to ensure that physicians are appropriately educated on the change of policy and to inform facilities and their compliance departments on the totality of the 2-midnight rule and all of its exceptions. Another commenter underscored the importance of the proposal because it appropriately recognizes the unprecedented volume of procedures that would newly be subject to the 2-midnight rule if the IPO list is eliminated.
Response: We thank commenters for their support.
Comment: One commenter supported an indefinite exemption but expressed concern that the exemption would end when CMS determines that the service or procedure is more commonly performed in the Medicare population in the outpatient setting. The commenter's concern was that certain patients may require inpatient level care, including intensive care unit (ICU) level care, even if some patients may appropriately receive the service in a lower-acuity setting.
Response: We appreciate the commenter's concern and emphasize, as ( printed page 53831) we did in the CY 2016 final OPPS/ASC rule, that that the 2-midnight benchmark does not override the clinical judgment of the physician regarding the need to keep the beneficiary at the hospital, to order specific services, or to determine appropriate levels of nursing care or physical locations within the hospital. This physician judgment is recognized in 42 CFR 412.3(d)(3) which states that “[w]here the admitting physician expects a patient to require hospital care for only a limited period of time that does not cross 2 midnights, an inpatient admission may be appropriate for payment under Medicare Part A based on the clinical judgment of the admitting physician and medical record support for that determination.” Even when a procedure loses its exemption from certain medical review activities as a result of being determined by the Secretary to be more commonly performed in the outpatient setting, it can still be deemed appropriate for payment under Medicare Part A under 42 CFR 412.3(d)(3) for patients the physician has determined need to receive the service in an inpatient setting despite the physician not having the expectation that the inpatient stay will cross 2 midnights.
Comment: Many commenters asked for additional clarification on the proper application of the case-by-case exception to the 2-midnight benchmark under 42 CFR 412.3(d)(3). This exception allows for Medicare Part A payment on a case-by-case basis for inpatient admissions that do not satisfy the 2-midnight benchmark, if the documentation in the medical record supports the admitting physician's determination that the patient requires inpatient hospital care despite an expected length of stay that is less than 2 midnights. Most of these commenters emphasized that this additional clarity was necessary due to Medicare Advantage plans historically ignoring the 2-midnight rule.
Response: CMS has recently reminded the Medicare Administrative Contractors (MACs) responsible for conducting inpatient short stay medical reviews of CMS' longstanding policy that recognizes the important role of physician judgment and individual patient needs in the hospital admission decision-making process. Providers should ensure that they clearly articulate in the medical record their rationale for admission to assist reviewers in understanding why admission for inpatient care is appropriate despite an expected length of stay that is less than two midnights. In other words, the medical record should reflect exactly what the current medical needs and/or other complex medical factors are that the physician/qualified practitioner is concerned about which would justify a case-by-case exception to the two-midnight rule (that is, that inpatient care is needed, despite an expected length of stay that is less than two midnights).
CMS has also instructed the MACs to continue to follow other longstanding guidance that they review the reasonableness of the inpatient admission for the purposes of Part A payment based on the information known to the physician at the time of admission. We have informed the MACs that the expectation of time and the determination of the underlying need for inpatient care despite an expected length of stay that is less than two midnights should be supported by patient-specific complex medical factors such as history and comorbidities, the severity of signs and symptoms, current medical needs, and the risk of an adverse event. MACs will expect such factors to be documented in the medical record. The entire medical record may be reviewed to support or refute the reasonableness of the physician's/qualified practitioner's expectation, but entries after the point of the admission order are only used in the context of interpreting what the physician/qualified practitioner knew and expected at the time of admission. Additionally, we have provided guidance to MACs that comorbidities and other complex medical factors are to be considered in the context of their contribution to the need for hospital services.
With respect to commenters' concerns regarding Medicare Advantage plans ignoring the 2-midnight rule, we emphasize here, as we have previously, that Medicare Advantage plans are required to comply with the 2-midnight benchmark. In the final rule titled `Medicare Program; Contract Year 2024 Policy and Technical Changes to the Medicare Advantage Program, Medicare Prescription Drug Benefit Program, Medicare Cost Plan Program, and Programs of All-Inclusive Care for the Elderly” which appeared in the Federal Register on April 12, 2023 (88 FR 22120), we clarified that “MA plans must comply with general coverage and benefit conditions included in Traditional Medicare laws, unless superseded by laws applicable to MA plans” and that “this includes coverage criteria for inpatient admissions at 42 CFR 412.3 . . .” (88 FR 22191). We additionally stated that “[i]n regards to inpatient admissions at § 412.3, we confirm that the criteria listed at § 412.3(a)-(d) apply to MA.” (88 FR 22191). In that final rule, we codified this requirement by revising 42 CFR 422.101(b)(2) to state that each MA organization must comply “with general coverage and benefit conditions included in Traditional Medicare laws, unless superseded by laws applicable to MA plans. This includes criteria for determining whether an item or service is a benefit available under Traditional Medicare. For example, this includes payment criteria for inpatient admissions at 42 CFR 412.3 . . .”
Comment: Several commenters offered suggestions about how CMS should provide notice that an exemption period is ending for a service when the service has been determined by the Secretary to be more commonly performed in the outpatient setting. One commenter suggested a “transparent process of advance notification” for providers when a service has been determined to be more commonly performed in the outpatient setting. Two commenters stated that CMS should use notice and comment rulemaking, not sub-regulatory guidance as proposed, to end exemption periods. These commenters did not provide a reason for this recommendation, other than one commenter's statement that “[w]e oppose audit policy changes to procedures removed from the IPO list being issued through CMS or Medicare Area Contractor `guidance.' ” Two of these commenters additionally suggested that CMS include in this notice and comment rulemaking the quality and patient safety data it relied on to determine that the procedure can be safely performed in the outpatient setting. Two commenters urged CMS not to end the exemption until at least two years after the Secretary determines that the service is more commonly performed in the outpatient setting to allow hospitals time to update their billing systems and gain experience with respect to the newly removed procedures consistent with CMS' past practice with respect to procedures removed from the IPO list based on clinical considerations.
Response: We agree with the need for a transparent process of advance notification to the public when a service has been determined to be more commonly performed in the outpatient setting. We appreciate commenters' suggestion that such notice be provided through rulemaking rather than subregulatory guidance as well as their request for the exemption period to extend at least 2 years after the service has been determined to be more commonly performed in the outpatient setting. Since we have not yet gone ( printed page 53832) through the process of ending the exemption period for an indefinitely exempted procedure, we will establish that process, including the effective date of the end of the exemption period, through notice and comment rulemaking the first year that the Secretary determines that a procedure is more commonly performed in the outpatient setting than the inpatient setting. We will address the process for subsequent years at that time.
Comment: Three commenters supported the proposal but urged CMS to exempt hospitals that utilize certain clinical decision support tools from patient status review for the 2-midnight policy. The commenters argue that clinical decision support technology exists to achieve the same program integrity and patient safety goals as the 2-midnight rule without the uncertainty around coverage for patients.
Response: We appreciate the commenters' suggestion and enthusiasm for clinical decision support technology, however we do not think clinical decision support tools are an appropriate substitute for patient status review by CMS or Medicare review contractors for compliance with the 2-midnight rule.
Comment: One commenter noted that in our summary of major provisions section discussing our proposal to continue the indefinite exemption from the 2-midnight rule for certain medical review activities we referred to “Beneficiary and Family-Centered Care Quality Improvement Organization (BFCC-QIO) referrals to Recovery Audit Contractor (RAC).” The commenter indicated that since, as of September 1, 2025, those 2-midnight reviews will be performed by the MACs and not the BFCC-QIOs, the reference to BFCC-QIOs should be replaced with MACs.
Response: We thank the commenter for pointing this out and the change has been made.
After consideration of the public comments we received, we are finalizing our proposal with modification. We are finalizing our proposal to (1) continue the indefinite exemption from site-of-service claim denials, initial medical review contractor referrals to RACs, and RAC reviews for “patient status” (that is, site-of-service) finalized in the CY 2021 OPPS/ASC final rule with comment period for procedures that are removed from the IPO list in CY 2021 or later under the OPPS; and (2) remove § 412.3(d)(2)(i) and (ii) and revise § 412.3(d)(2) to clarify that claims for services and procedures removed from the IPO list on or after January 1, 2021 are exempt from certain medical review activities until the Secretary determines that the service or procedure is more commonly performed in the outpatient setting than the inpatient setting. We are not finalizing our proposal to announce in subregulatory guidance when an exemption is ending for a particular service or procedure. Instead, the first time that the Secretary determines that a service or procedure is more commonly performed in the outpatient setting than the inpatient setting, we will make the announcement that the exemption is ending for the service or procedure through notice and comment rulemaking. We will address the process for subsequent years at that time.
E. Coding and Payment for Category B IDE Devices and Studies
We proposed to revise the section heading and paragraph (a) introductory text at § 419.47 to correct two errors that occurred when this regulation was revised in the CY 2025 OPPS/ASC final rule with comment period (89 FR 94304 through 94307).
In the CY 2025 OPPS/ASC final rule with comment period, we finalized our proposal to codify our coding and payment policy for Category B Investigational Device Exemption (IDE) clinical trials with control arms through revisions to § 419.47. Specifically, we revised § 419.47's paragraph (a) introductory text to specify that our policy only applies to IDE studies with a placebo control arm and where a payment adjustment is necessary to preserve the scientific validity of such a study. However, in making these revisions, we inadvertently deleted existing regulatory text that was not changed in the CY 2025 OPPS/ASC final rule with comment period. Specifically, we inadvertently deleted § 419.47(a)(1) “The Medicare coverage IDE study criteria in § 405.212 of this chapter are met” and paragraph (2) “A new or revised code is necessary to preserve the scientific validity of such a study, such as by preventing the unblinding of the study.” Therefore, effective January 1, 2026, we proposed to amend the regulatory text at § 419.47(a) to restore these two inadvertently removed paragraphs.
Additionally, in the CY 2025 OPPS/ASC final rule with comment period, we did not finalize our CY 2025 OPPS/ASC proposal to extend our coding and payment policy to drugs and devices that are being studied in clinical trials under a Coverage with Evidence Development (CED) National Coverage Determination (NCD) [211] for which the trial includes a treatment and control arm for CY 2025. However, despite our intent to remove all proposed revisions relating to this extension of the policy in the final rule, we inadvertently revised the section heading at § 419.47 to state that the policy applied to “devices/drugs studies.” Since we did not finalize the policy for CY 2025, we proposed, effective January 1, 2026, to delete “and devices/drugs studies” from the section heading at § 419.47.
We note that the revisions described previously in this section to § 419.47 were inadvertently omitted from the proposed regulation text in the CY 2026 OPPS/ASC proposed rule but that regulatory text has been included in this final rule with comment period.
We did not receive public comments on this proposal, and therefore, we are finalizing as proposed for CY 2026.
XI. CY 2026 OPPS Payment Status and Comment Indicators
A. CY 2026 OPPS Payment Status Indicator Definitions
Payment status indicators (SIs) that we assign to HCPCS codes and APCs serve an important role in determining payment for services under the OPPS. They indicate whether a service represented by a HCPCS code is payable under the OPPS or another payment system and whether particular OPPS policies apply to the code.
For CY 2026 and subsequent years, we proposed to create a new status indicator “S1”. We proposed this new status indicator to indicate that a skin substitute product is paid separately from other procedure codes under the OPPS. We proposed to assign all existing HCPCS codes describing skin substitute products to status indicator “S1” for CY 2026. This policy is further discussed in section V.B.10. of this final rule with comment period. We solicited public comments on the proposed definitions of the OPPS payment status indicators for CY 2026. We did not propose to make any additional changes to the existing definitions of status indicators that were listed in Addendum D1 to the CY 2026 OPPS/ASC proposed rule, which is available on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices.
We did not receive any public comments related to the definition of status indicator “S1”. Therefore, we are finalizing our proposed definition for status indicator “S1” without modification for CY 2026. The final definition and payment status of status ( printed page 53833) indicator “S1” can be found in Table 122.
The complete list of CY 2026 payment status indicators and their definitions is displayed in Addendum D1 to this final rule with comment period, which is available on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices. CY 2026 payment status indicator assignments for APCs and HCPCS codes are shown in Addendum A and Addendum B, respectively, to this final rule with comment period, which are available on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices.
B. CY 2026 Comment Indicator Definitions
We proposed to use four comment indicators for the CY 2026 OPPS. These comment indicators, “CH,” “NC,” “NI,” and “NP,” are in effect for CY 2025; and we proposed to continue their use in CY 2026. The proposed CY 2026 OPPS comment indicators are as follows:
- “CH”—Active HCPCS code in current and next calendar year, status indicator and/or APC assignment has changed; or active HCPCS code that will be discontinued at the end of the current calendar year.
- “NC”—New code for the next calendar year or existing code with substantial revision to its code descriptor in the next calendar year, as compared to current calendar year for which we requested comments in the CY 2026 OPPS/ASC proposed rule; final APC assignment; comments will not be accepted on the final APC assignment for the new code.
- “NI”—New code for the next calendar year or existing code with substantial revision to its code descriptor in the next calendar year, as compared to current calendar year, interim APC assignment; comments will be accepted on the interim APC assignment for the new code.
- “NP”—New code for the next calendar year or existing code with substantial revision to its code descriptor in the next calendar year, as compared to current calendar year, proposed APC assignment; comments will be accepted on the proposed APC assignment for the new code.
We solicited public comments on our proposed definitions of the OPPS comment indicators for CY 2026.
We did not receive public comments on this provision, and therefore, we are finalizing those definitions without modification for CY 2026. The definitions of OPPS comment indicators for CY 2026 are listed in Addendum D2 to this final rule with comment period, which is available on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices.
XII. MedPAC Recommendations
The Medicare Payment Advisory Commission (MedPAC) was established under section 1805 of the Act in large part to advise the U.S. Congress on issues affecting the Medicare program. As required under the statute, MedPAC submits reports to the Congress no later than March and June of each year that present its Medicare payment policy recommendations. The March report typically provides discussion of Medicare payment policy across different payment systems and the June report typically discusses selected Medicare issues. We are including this section to make interested parties aware of certain MedPAC recommendations for the OPPS and ASC payment systems as discussed in its March 2025 report.
Comments received from MedPAC for other OPPS or ASC payment system policies are discussed in the applicable sections of this final rule with comment period.
A. OPPS Payment Rates Update
The March 2025 MedPAC “Report to the Congress: Medicare Payment Policy,” recommended that the Congress update Medicare OPPS payment rates by the amount specified in current law plus 1 percent. We refer readers to the March 2025 report for a complete discussion of this recommendation.[212] We appreciate MedPAC's recommendation and, as discussed further in section II.B. of this final rule with comment period, we are finalizing our proposal to increase the OPPS payment rates by the amount specified in current law.
B. Medicare Safety Net Index
In the March 2025 MedPAC “Report to the Congress: Medicare Payment Policy,” MedPAC stated that their recommended update to IPPS and OPPS payment rates of current law plus 1 percent may not be sufficient to ensure the financial viability of some Medicare safety-net hospitals with a poor payer mix. MedPAC recommended redistributing the current Medicare safety-net payments (disproportionate share hospital and uncompensated care payments) using the MedPAC-developed Medicare Safety-Net Index (MSNI) for hospitals. In addition, MedPAC recommended adding $4 billion to this MSNI pool of funds to help maintain the financial viability of Medicare safety-net hospitals and recommended to the Congress transitional approaches for an MSNI policy. The FY 2026 IPPS/LTCH proposed and final rule with comment period (90 FR 18002 and 90 FR 36536) provides additional information regarding statutory requirements for disproportionate share hospital and uncompensated care payments. We look forward to working with the Congress on these matters. ( printed page 53834)
XIII. Updates to the Ambulatory Surgical Center (ASC) Payment System
A. Background, Legislative History, Statutory Authority, and Prior Rulemaking for the ASC Payment System
For a detailed discussion of the legislative history and statutory authority related to payments to ASCs under Medicare, we refer readers to the CY 2012 OPPS/ASC final rule with comment period (76 FR 74377 through 74378) and the June 12, 1998 proposed rule (63 FR 32291 through 32292). For a discussion of prior rulemaking on the ASC payment system, we refer readers to the CYs 2012 to 2025 OPPS/ASC final rules with comment period (76 FR 74378 through 74379; 77 FR 68434 through 68467; 78 FR 75064 through 75090; 79 FR 66915 through 66940; 80 FR 70474 through 70502; 81 FR 79732 through 79753; 82 FR 59401 through 59424; 83 FR 59028 through 59080; 84 FR 61370 through 61410; 85 FR 86121 through 86179; 86 FR 63761 through 63815; 87 FR 72054 through 72096; 88 FR 81900 through 81961; and 89 FR 94309 through 94367).
B. ASC Treatment of New and Revised Codes
1. Background on Process for New and Revised HCPCS Codes
We update the lists and payment rates for covered surgical procedures and covered ancillary services in ASCs in conjunction with the annual proposed and final rulemaking process to update the OPPS and the ASC payment systems (§ 416.173; 72 FR 42535). We base ASC payment and policies for most covered surgical procedures, drugs, biologicals, and certain other covered ancillary services on the OPPS payment policies, and we use quarterly change requests (CRs) to update services paid for under the OPPS. We also provide quarterly update CRs for ASC covered surgical procedures and covered ancillary services throughout the year (January, April, July, and October). We release new and revised Level II HCPCS codes and recognize the release of new and revised CPT codes by the American Medical Association (AMA) and make these codes effective (that is, the codes are recognized on Medicare claims) via these ASC quarterly update CRs. We recognize the release of new and revised Category III CPT codes in the July and January CRs. These updates implement newly created and revised Level II HCPCS and Category III CPT codes for ASC payments and update the payment rates for separately paid drugs and biologicals based on the most recently submitted ASP data. New and revised Category I CPT codes, except vaccine codes, are released only once a year, and are implemented only through the January quarterly CR update. New and revised Category I CPT vaccine codes are released twice a year and are implemented through the January and July quarterly CR updates. We refer readers to Table 41 in the CY 2012 OPPS/ASC proposed rule for an example of how this process is used to update HCPCS and CPT codes, which we finalized in the CY 2012 OPPS/ASC final rule with comment period (76 FR 42291; 76 FR 74380 through 74384).
In our annual updates to the ASC list of covered surgical procedures and covered ancillary services, we undertake a review of excluded surgical procedures, new codes, and codes with revised descriptors, to identify any that we believe meet the criteria for designation as ASC covered surgical procedures or covered ancillary services. Updating the lists of ASC covered surgical procedures and covered ancillary services, as well as their payment rates, in association with the annual OPPS rulemaking cycle, is particularly important because the OPPS relative payment weights and, in some cases, payment rates, are used as the basis for the payment of many covered surgical procedures and covered ancillary services under the revised ASC payment system. This joint update process ensures that the ASC updates occur in a regular, predictable, and timely manner.
Payment for ASC procedures, services, and items are generally based on medical billing codes, specifically, HCPCS codes, that are reported on ASC claims. The HCPCS is divided into two principal subsystems, referred to as Level I and Level II. Level I is comprised of CPT (Current Procedural Terminology) codes, a numeric and alphanumeric coding system maintained by the AMA, and includes Category I, II, and III CPT codes. Level II of the HCPCS, which is maintained by CMS, is a standardized coding system that is used primarily to identify products, supplies, and services not included in the CPT codes. Together, Level I and II HCPCS codes are used to report procedures, services, items, and supplies under the ASC payment system. Specifically, we recognize the following codes on ASC claims:
- Category I CPT codes, which describe surgical procedures, diagnostic and therapeutic services, and vaccine codes;
- Category III CPT codes, which describe new and emerging technologies, services, and procedures; and
- Level II HCPCS codes (also known as alpha-numeric codes), which are used primarily to identify drugs, devices, supplies, temporary procedures, and services not described by CPT codes.
We finalized a policy in the August 2, 2007 ASC final rule with comment period (72 FR 42533 through 42535) to evaluate each year all new and revised Category I and Category III CPT codes and Level II HCPCS codes that describe surgical procedures, and to make preliminary determinations during the annual OPPS/ASC rulemaking process regarding whether or not they meet the criteria for payment in the ASC setting as covered surgical procedures and, if so, whether or not they are office-based procedures. In addition, we identify new and revised codes as ASC covered ancillary services based upon the final payment policies of the revised ASC payment system. In prior rulemakings, we refer to this process as recognizing new codes. However, this process has always involved the recognition of new and revised codes. We consider revised codes to be new when they have substantial revision to their code descriptors that necessitate a change in the current ASC payment indicator. To clarify, we refer to these codes as new and revised in this CY 2026 OPPS/ASC proposed rule.
We have separated our discussion below based on when the codes are released and whether we propose to solicit public comments in the proposed rule (and respond to those comments in this final rule with comment period) or whether we will be soliciting public comments in this CY 2026 OPPS/ASC final rule with comment period (and responding to those comments in the CY 2027 OPPS/ASC final rule with comment period).
2. April 2025 HCPCS Codes Proposed Rule Comment Solicitation
For the April 2025 update, there were no new CPT codes; however, there were several new Level II HCPCS codes. In the April 2025 ASC quarterly update (Transmittal 13181, dated April 25, 2025, CR 14017), we added one new Level II HCPCS code to the list of covered ancillary services. Table 73 (New Level II HCPCS Codes for ASC Covered Surgical Procedures and Ancillary Services Effective April 1, 2025) of the CY 2026 OPPS/ASC proposed rule (90 FR 33701) displayed the new Level II HCPCS codes that were implemented April 1, 2025. These new codes that were effective April 1, 2025, were assigned to comment indicator ( printed page 53835) “NP” in Addendum BB to the CY 2026 OPPS/ASC proposed rule to indicate that the codes were assigned to an interim APC assignment and that comments would be accepted on their interim APC assignments. In addition, we note that the entire ASC addenda, which consist of the addenda listed below, are available via the internet on the CMS website, specifically, at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices:
ASC Addendum AA: ASC Covered Surgical Procedures (Including Surgical Procedures for Which Payment is Packaged).
ASC Addendum BB: ASC Covered Ancillary Services Integral to Covered Surgical Procedures (Including Ancillary Services for Which Payment is Packaged).
ASC Addendum DD1: ASC Payment Indicators (PI).
ASC Addendum DD2: ASC Comment Indicators (CI).
ASC Addendum EE: Surgical Procedures to be Excluded from Payment in ASCs.
ASC Addendum FF: ASC Device Offset Percentages.
- Addendum O: Long Descriptors for New Category I CPT Codes, Category III CPT Codes, C-Codes, and G-Codes Effective January 1, 2026.
We invited public comments on the proposed interim payment indicators for the new HCPCS codes that were recognized as ASC covered ancillary services in April 2025 through the quarterly update CRs, as listed in Table 123 (New Level II HCPCS Codes for ASC Covered Surgical Procedures and Ancillary Services Effective April 1, 2025). The new codes that were effective April 1, 2025, were assigned to comment indicator “NP” in ASC Addendum BB to the CY 2026 OPPS/ASC proposed rule to indicate that the codes are assigned to interim payment indicators and comments would be accepted on their interim assignments. We proposed to finalize the payment indicators in this final rule with comment period. We did not receive any comments on the proposed ASC payment indicator assignments for the new Level II HCPCS codes implemented in April 2025 and are finalizing the proposed ASC payment indicator assignments for these codes. We note in prior years we included the final ASC payment indicators in the coding tables in the preamble, but because we include the same information in the ASC addenda, we have not included them in Table 123. Therefore, readers are advised to refer to the ASC addenda for the final ASC payment indicators and payment rates for all codes reported under the ASC payment system. The list of ASC payment indicators and definitions used under the ASC payment system can be found in the ASC addenda. We note that the ASC addenda (AA, BB, DD1, DD2, EE, and FF) are available via the internet on the CMS website.
( printed page 53836)3. July 2025 HCPCS Codes Proposed Rule Comment Solicitation
In the July 2025 ASC quarterly update (Transmittal 13344, Change Request 14101, August 1, 2025), we added several separately payable CPT and Level II HCPCS codes to the list of covered surgical procedures and covered ancillary services. Table 74 (New HCPCS Codes for ASC Covered Surgical Procedures and Ancillary Services Effective July 1, 2025) of the CY 2026 OPPS/ASC proposed rule (90 FR 33702), displayed the new HCPCS codes that are effective July 1, 2025. We invited public comments on the proposed payment indicators for these Level II HCPCS codes, and indicated that the proposed comment indicators, payment indicators, and payment rates for these codes were listed in ( printed page 53837) Addendum AA and Addendum BB of the proposed rule. These new codes that were effective July 1, 2025, were assigned to comment indicator “NP” in ASC Addendum AA and Addendum BB to the CY 2026 OPPS/ASC proposed rule to indicate that the codes were assigned to interim payment indicators and comments would be accepted on their interim assignments. We further stated that we proposed to finalize the payment indicators in final rule with comment period. We note in prior years we included the final ASC payment indicators in the coding preamble tables, however, because the same information can be found in Addendum AA and Addendum BB, we are no longer including them in Table 124. Therefore, readers are advised to refer to the ASC addenda for the final ASC payment indicators and payment rates for all codes reported under the ASC payment system.
We did not receive any comments on the proposed interim ASC payment indicator assignments for the new CPT and Level II HCPCS codes that were added to the list of covered surgical procedures and ancillary services implemented in July 2025. Therefore, we are finalizing the proposed ASC payment indicator assignments for the codes.
( printed page 53838) ( printed page 53839) ( printed page 53840)4. October 2025 HCPCS Codes Final Rule Comment Solicitation
For CY 2026, consistent with our established policy, we proposed in the CY 2026 OPPS/ASC proposed rule (90 FR 33704) that the Level II HCPCS codes that will be effective October 1, 2025, would be flagged with comment indicator “NI” in Addendum BB to this final rule with comment period to indicate that we have assigned the codes an interim ASC payment status for CY 2026. In the October 2025 ASC quarterly update (Transmittal1349, Change Request 14246, dated September 22, 2025), we added several separately payable Level II HCPCS codes to the list of covered surgical procedures and covered ancillary services. Table 125 lists the codes that were effective October 1, 2025. We are inviting public comments on this final rule with comment period on the interim payment indicators, which would be finalized in the CY 2027 OPPS/ASC final rule with comment period. We note these codes will be subject to comment in the CY 2027 OPPS/ASC proposed rule with comment period, which would be finalized in the CY 2027 OPPS/ASC final rule with comment period.
( printed page 53841)5. January 2026 HCPCS Codes
a. New Level II HCPCS Codes Final Rule Comment Solicitation
As has been our practice in the past, we incorporate those new Level II HCPCS codes that are effective January 1 in the final rule with comment period, thereby updating the ASC payment system for the calendar year. We note that unlike the CPT codes that are effective January 1 and are included in the OPPS/ASC proposed rules, and except for the G-codes listed in Addendum O to the CY 2026 OPPS/ASC proposed rule, most Level II HCPCS codes are not released until sometime around November to be effective January 1. Because these codes are not available until November, we are unable to include them in the OPPS/ASC proposed rule; however, the codes are flagged with comment indicator “NI” in ASC Addendum AA and Addendum BB to this final rule with comment period to indicate that we are assigning them an interim payment status, which is subject to public comment. Therefore, as we stated in the CY 2025 OPPS/ASC proposed rule, these Level II HCPCS codes that will be effective January 1, 2026 are included in this final rule with comment period and will also be released to the public through the January 2026 ASC Update CR and the CMS HCPCS website. We are inviting public comments in this final rule with comment period on the payment indicator assignments, which would be finalized in the CY 2027 OPPS/ASC final rule with comment period. Similar to the codes effective October 1, 2025, these new Level II HCPCS codes that will be effective January 1, 2026 will be subject to comment in the CY 2027 OPPS/ASC proposed rule, which would be finalized in the CY 2027 OPPS/ASC final rule with comment period.
b. New CY 2026 CPT Codes Proposed Rule Comment Solicitation
For the CY 2026 ASC update, we received the CPT codes that will be effective January 1, 2026, from the AMA in time to be included in the CY 2026 OPPS/ASC proposed rule. The new, revised, and deleted CPT codes can be found in ASC Addendum AA and Addendum BB to the CY 2026 OPPS/ ( printed page 53842) ASC proposed rule, which are available via the internet on the CMS website. We note that the new and revised CPT codes are assigned to comment indicator “NP” in ASC Addendum AA and Addendum BB of the CY 2026 OPPS/ASC proposed rule to indicate that the code is new for the next calendar year, or the code is an existing code with substantial revision to its code descriptor in the next calendar year as compared to the current calendar year with a proposed payment indicator assignment. We stated that we would accept comments and finalize the payment indicators in this final rule with comment period. Further, we remind readers that the CPT code descriptors that appeared in Addendum AA and Addendum BB are short descriptors and do not describe the complete procedure, service, or item described by the CPT code. Therefore, we included the 5-digit placeholder codes and their long descriptors for the new CY 2026 CPT codes in Addendum O to the CY 2026 OPPS/ASC proposed rule (which is available via the internet on the CMS website) so that the public could comment on our proposed payment indicator assignments. The 5-digit placeholder codes were listed in Addendum O to the CY 2026 OPPS/ASC proposed rule, specifically under the column labeled “CY 2026 OPPS/ASC Proposed Rule 5-Digit Placeholder Code.” We also stated that we would include the final CPT code numbers in this CY 2026 OPPS/ASC final rule with comment period.
We did not receive any comments on the proposed ASC payment indicators for the new CPT codes effective January 1, 2026, so we are finalizing these codes as proposed.
Finally, in Table 126, we summarize our process for updating codes through our ASC quarterly update CRs, seeking public comments, and finalizing the treatment of these new codes under the ASC payment system.
6. ASC Payment and Comment Indicators
a. Background
In addition to the payment indicators that we introduced in the August 2, 2007 ASC final rule with comment period, we created final comment indicators for the ASC payment system in the CY 2008 OPPS/ASC final rule with comment period (72 FR 66855). We created Addendum DD1 to define ASC payment indicators that we use in Addenda AA and BB to provide payment information regarding covered surgical procedures and covered ancillary services, respectively, under the revised ASC payment system. The ASC payment indicators in Addendum DD1 are intended to capture policy-relevant characteristics of HCPCS codes that may receive packaged or separate payment in ASCs, such as whether they were on the ASC CPL prior to CY 2008; payment designation, such as device-intensive or office-based, and the corresponding ASC payment methodology; and their classification as separately payable ancillary services, including radiology services, brachytherapy sources, OPPS pass-through devices, corneal tissue acquisition services, drugs or biologicals, NTIOLs, or qualifying non-opioid devices.
We also created Addendum DD2 that lists the ASC comment indicators. The ASC comment indicators included in Addenda AA and BB to the proposed rules and final rules with comment period serve to identify, for the revised ASC payment system, the status of a specific HCPCS code and its payment indicator with respect to the timeframe when comments will be accepted. The comment indicator “NI” is used in the OPPS/ASC final rule with comment period to indicate new codes for the next calendar year for which the interim payment indicator assigned is subject to comment. The comment indicator “NI” also is assigned to existing codes with substantial revisions to their descriptors such that we consider them to be describing new services, and the interim payment indicator assigned is subject to comment, as discussed in the CY 2010 OPPS/ASC final rule with comment period (74 FR 60622).
The comment indicator “NP” is used in the OPPS/ASC proposed rule to indicate new codes for the next calendar year for which the proposed payment indicator assigned is subject to comment. The comment indicator “NP” also is assigned to existing codes with substantial revisions to their descriptors, such that we consider them to be describing new services, and the ( printed page 53843) proposed payment indicator assigned is subject to comment, as discussed in the CY 2016 OPPS/ASC final rule with comment period (80 FR 70497).
The “CH” comment indicator is used in Addenda AA and BB to the proposed rule (these addenda are available via the internet on the CMS website) to indicate that the payment indicator assignment has changed for an active HCPCS code in the current year and the next calendar year, for example, if an active HCPCS code is newly recognized as payable in ASCs or an active HCPCS code is discontinued at the end of the current calendar year. The “CH” comment indicators that are published in the final rule are provided to alert readers that a change has been made from one calendar year to the next, but do not indicate that the change is subject to comment.
In the CY 2021 OPPS/ASC final rule with comment period, we finalized the addition of ASC payment indicator “K5”—Items, Codes, and Services for which pricing information and claims data are not available. No payment made—to ASC Addendum DD1 (which is available via the internet on the CMS website) to indicate those services and procedures that CMS anticipates will become payable when claims data or payment information becomes available.
In CY 2024 OPPS/ASC final rule with comment period, we finalized the addition of two ASC payment indicators, “D1”—“Ancillary dental service/item; no separate payment made” and “D2”—“Non office-based dental procedure added in CY 2024 or later”, for new dental codes for CY 2024 and subsequent calendar years to indicate potentially payable dental services and procedures in the ASC setting (88 FR 81907). We added these two codes to Addendum DD1 (which is available via the internet on the CMS website).
In CY 2025 OPPS/ASC final rule with comment period, we finalized the modification of the descriptor of ASC payment indicator “L6” to “Special payment; New Technology Intraocular Lens (NTIOL) or qualifying non-opioid devices”, to account for non-opioid devices paid for under the ASC payment system pursuant to section 4135 of the Consolidated Appropriations Act (CAA), 2023 (89 FR 94317). We added this code to Addendum DD1 (which is available via the internet on the CMS website).
b. Final ASC Payment and Comment Indicators for CY 2026
For CY 2026, we proposed new and revised Category I and III CPT codes as well as new and revised Level II HCPCS codes. Proposed Category I and III CPT codes that are new and revised for CY 2026 and any new and existing Level II HCPCS codes with substantial revisions to the code descriptors for CY 2026, compared to the CY 2025 descriptors, are included in ASC Addenda AA and BB to the CY 2026 OPPS/ASC proposed rule and labeled with comment indicator “NP” to indicate that these CPT and Level II HCPCS codes were open for comment as part of the CY 2026 OPPS/ASC proposed rule.
As discussed in section III. of the CY 2026 OPPS/ASC proposed rule, we proposed to create APC groups to pay separately for certain skin substitutes under the OPPS and, as discussed in section XIII.D. of the CY 2026 OPPS/ASC proposed rule, we also proposed to pay separately for skin substitute supplies in the ASC payment system and add such supplies to the ancillary items and services list for CY 2026.
Under the ASC payment system, skin substitute products are currently packaged and assigned an ASC payment indicator of “N1” (Packaged service/item; no separate payment made). We do not believe there is an existing payment indicator available that would adequately describe these supplies and provide the correct separate payment under the ASC payment system. Under this new policy, payment under the ASC payment system for separately payable skin substitute products would be based on the OPPS conversion factor, not on the ASC conversion factor. Additionally, payment for these skin substitute products would not be subject to the ASC wage index. Therefore, for CY 2026 and subsequent years, we proposed to create a new ASC payment indicator “S2”—(Skin substitute supply group; paid separately when provided integral to a surgical procedure on ASC list; payment based on OPPS rate)—to Addendum DD1 to this final rule with comment period to describe skin substitute products paid separately in an ASC. This “S2” payment indicator would indicate a separately payable ancillary skin substitute supply when provided integral to a separately payable ASC covered surgical procedure.
We did not receive public comments on our proposal to create a new “S2” payment indicator. Therefore, we are finalizing our proposal to indicate a separately payable ancillary skin substitute supply when provided integral to a separately payable ASC covered surgical procedure beginning CY 2026. We refer readers to Addenda DD1 and DD2 of this final rule with comment period (these addenda are available via the internet on the CMS website) for the complete list of ASC payment and comment indicators finalized for the CY 2026 update.
C. Payment Policies Under the ASC Payment System
1. Final ASC Payment for Covered Surgical Procedures
a. Background
Our ASC payment policies for covered surgical procedures under the revised ASC payment system are described in the CY 2008 OPPS/ASC final rule with comment period (72 FR 66828 through 66831). Under our established policy, we use the ASC standard ratesetting methodology of multiplying the ASC relative payment weight for the procedure by the ASC conversion factor for that same year to calculate the national unadjusted payment rates for procedures with payment indicators “G2” and “A2.” Payment indicator “A2” was developed to identify procedures that were included on the list of ASC covered surgical procedures in CY 2007 and, therefore, were subject to transitional payment prior to CY 2011. Although the 4-year transitional period has ended and payment indicator “A2” is no longer required to identify surgical procedures subject to transitional payment, we have retained payment indicator “A2” because it is used to identify procedures that are exempted from the application of the office-based designation.
Payment rates for office-based procedures (payment indicators “P2,” “P3,” and “R2”) are the lower of the PFS nonfacility PE RVU-based amount or the amount calculated using the ASC standard rate setting methodology for the procedure. As detailed in section XIII.C.3.b. of this final rule with comment period, we update the payment amounts for office-based procedures (payment indicators “P2,” “P3,” and “R2”) using the most recent available PFS and OPPS data. We compare the estimated current year rate for each of the office-based procedures, calculated according to the ASC standard rate setting methodology, to the PFS nonfacility PE RVU-based amount to determine which is lower and, therefore, would be the current year payment rate for the procedure under our final policy for the revised ASC payment system (§ 416.171(d)).
The rate calculation established for device-intensive procedures (payment indicator “J8”) is structured so only the service (non-device) portion of the rate is subject to the ASC conversion factor. We update the payment rates for device-intensive procedures to incorporate the most recent device offset percentages calculated under the ASC standard ( printed page 53844) ratesetting methodology, as discussed in section XIII.C.4. of this final rule with comment period.
In the CY 2014 OPPS/ASC final rule with comment period (78 FR 75081), we finalized our proposal to calculate the CY 2014 payment rates for ASC covered surgical procedures according to our established methodologies, with the exception of device removal procedures. For CY 2014, we finalized a policy to conditionally package payment for device removal procedures under the OPPS. Under the OPPS, a conditionally packaged procedure (status indicators “Q1” and “Q2”) describes a HCPCS code where the payment is packaged when it is provided with a significant procedure but is separately paid when the service appears on the claim without a significant procedure. Because ASC services always include a covered surgical procedure, HCPCS codes that are conditionally packaged under the OPPS are generally packaged (payment indicator “N1”) under the ASC payment system. Under the OPPS, device removal procedures are conditionally packaged and, therefore, would be packaged under the ASC payment system. There is no Medicare payment made when a device removal procedure is performed in an ASC without another surgical procedure included on the claim; therefore, no Medicare payment would be made if a device was removed but not replaced. To ensure that the ASC payment system provides separate payment for surgical procedures that only involve device removal—conditionally packaged in the OPPS (status indicator “Q2”)—we have continued to provide separate payment since CY 2014 and assign the current ASC payment indicators associated with these procedures.
b. Update to ASC Covered Surgical Procedure Payment Rates for CY 2026
We proposed to update ASC payment rates for CY 2026 and subsequent years using the established rate calculation methodologies under § 416.171 and using our definition of device-intensive procedures, as discussed in section XIII.C.4. of this final rule with comment period. As the proposed OPPS relative payment weights are generally based on geometric mean costs, we proposed that the ASC payment system will generally use the geometric mean cost to determine proposed relative payment weights under the ASC standard methodology. We proposed to continue to use the amount calculated under the ASC standard ratesetting methodology for procedures assigned payment indicators “A2” and “G2”.
We proposed to calculate payment rates for office-based procedures (payment indicators “P2,” “P3,” and “R2”) and device-intensive procedures (payment indicator “J8”) according to our established policies and to identify device-intensive procedures using the methodology discussed in section XIII.C.4. of this final rule with comment period. Therefore, we proposed to update the payment amount for the service portion (the non-device portion) of the device-intensive procedures using the standard ASC ratesetting methodology and the payment amount for the device portion based on the proposed CY 2026 device offset percentages that have been calculated using the standard OPPS APC ratesetting methodology. We proposed that payment for office-based procedures would be at the lesser of the proposed CY 2026 PFS nonfacility PE RVU-based amount or the proposed CY 2026 ASC payment amount calculated according to the ASC standard ratesetting methodology.
As we did for CYs 2014 through 2025, for CY 2026, we proposed to continue our policy for device removal procedures, such that device removal procedures that are conditionally packaged in the OPPS (status indicators “Q1” and “Q2”) will be assigned the current ASC payment indicators associated with those procedures and will continue to be paid separately under the ASC payment system.
c. Final Payment for ASC Add-On Procedures Eligible for Complexity Adjustments Under the OPPS
In this section, we discuss the policy to provide increased payment under the ASC payment system for combinations of certain “J1” service codes and add-on procedure codes that are eligible for a complexity adjustment under the OPPS.
(1) OPPS C-APC Complexity Adjustment Policy
Under the OPPS, complexity adjustments are utilized to provide increased payment for certain comprehensive services. As discussed in section II.A.2.b. of this final rule with comment period, we apply a complexity adjustment by promoting qualifying paired “J1” service code combinations or paired code combinations of “J1” services and add-on codes from the originating Comprehensive APC (C-APC) (the C-APC to which the designated primary service is first assigned) to the next higher paying C-APC in the same clinical family of C-APCs. A “J1” status indicator refers to a hospital outpatient service paid through a C-APC. We package payment for all add-on codes, which are codes that describe a procedure or service always performed in addition to a primary service or procedure, into the payment for the C-APC. However, certain combinations of primary service codes and add-on codes may qualify for a complexity adjustment.
We apply complexity adjustments when the paired code combination represents a complex, costly form or version of the primary service when the frequency and cost thresholds are met. The frequency threshold is met when there are 25 or more claims reporting the code combination, and the cost threshold is met when there is a violation of the 2 times rule, as specified in section 1833(t)(2) of the Act and described in section III.A.2.b. of this final rule with comment period, in the originating C-APC. These paired code combinations that meet the frequency and cost threshold criteria represent those that exhibit materially greater resource requirements than the primary service. After designating a single primary service for a claim, we evaluate that service in combination with each of the other procedure codes reported on the claim that are either assigned to status indicator “J1” or add-on codes to determine if there are paired code combinations that meet the complexity adjustment criteria. Once we have determined that a particular combination of “J1” services, or combinations of a “J1” service and add-on code, represents a complex version of the primary service because it is sufficiently costly, frequent, and a subset of the primary comprehensive service overall according to the criteria described previously, we promote the claim to the next higher cost C-APC within the clinical family unless the primary service is already assigned to the highest cost APC within the C-APC clinical family or assigned to the only C-APC in a clinical family. We do not create new C-APCs with a comprehensive geometric mean cost that is higher than the highest geometric mean cost (or only) C-APC in a clinical family just to accommodate potential complexity adjustments. Therefore, the highest payment for any claim including a code combination for services assigned to a C-APC would be the highest paying C-APC in the clinical family (79 FR 66802).
As previously stated, we package payment for add-on codes into the C-APC payment rate. If any add-on code reported in conjunction with the “J1” primary service code does not qualify for a complexity adjustment, payment for the add-on service continues to be packaged into the payment for the ( printed page 53845) primary service and the primary service code reported with the add-on code is not reassigned to the next higher cost C-APC. We list the final complexity adjustments for “J1” and add-on code combinations for CY 2026, along with all of the other final complexity adjustments, in Addendum J to this final rule with comment period (which is available via the internet on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices).
(2) CY 2026 ASC Special Payment Policy Proposal for OPPS Complexity-Adjusted C-APCs
For CY 2026, we proposed to continue the special payment policy and methodology for OPPS complexity-adjusted C-APCs that was finalized in the CY 2023 OPPS/ASC final rule with comment period (87 FR 72078 through 72080).
For those ASC complexity adjustment codes for which we have claims data, we proposed to use the claims data to calculate the code combination utilization and estimated payments for the ASC payment system budget neutrality calculations for CY 2026. The ASC complexity adjustment budget neutrality calculations are discussed further in section XIII.H.2.a. of this final rule with comment period. The full list of the proposed ASC complexity adjustment codes for CY 2026 can be found in the CY 2026 proposed ASC Addendum AA and the supplemental policy file, which also includes both the existing ASC complexity adjustment codes and proposed additions and published on the CMS website at https://www.cms.gov/medicare/medicare-fee-for-service-payment/ascpayment/asc-regulations-and-notices. Since the complexity adjustment assignments change each year under the OPPS, the proposed list of ASC complexity adjustment codes eligible for the proposed payment policy changed slightly from the previous year. Additionally, since complexity adjustment assignments may change between the proposed rule and final rule under the OPPS, the final list of ASC complexity adjustment codes eligible for this payment policy may be slightly different than the proposed list of ASC complexity adjustment codes.
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: A commenter recommended adjustments to our device-intensive determination for ASC complexity adjustment codes. The commenter stated that the current methodology fails to recognize the device-intensive status of add-on codes associated with significant device costs and that failure to capture these costs could present a financial challenge for ASCs who wish to offer these services in the setting that is preferred by many beneficiaries.
Response: We appreciate the commenter's recommendation. We did not propose to calculate the device portion of an ASC complexity adjustment code based on both the device costs of packaged add-on procedures and the device costs of the primary procedure but we will take this suggestion into consideration for future rulemaking.
After consideration of public comments we received, we are finalizing the ASC special payment policy for OPPS complexity-adjusted C-APCs, as proposed. The final ASC complexity adjustment codes, based on the most recent data available for this final rule with comment period, can be found on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/ambulatory-surgicalcenter-asc/annual-policy-files. Existing ASC complexity adjustment codes that do not meet our criteria for separate payment for CY 2026 because the code combination is not eligible for a complexity adjustment under the OPPS for CY 2026 will be non-payable and assigned a status indicator of “B5”—“Alternative code may be available; no payment made”—for CY 2026. Additionally, proposed ASC complexity adjustment codes that met our criteria based on data available for the CY 2026 OPPS/ASC proposed rule but do not meet our criteria based on claims data available for this final rule with comment period will not be finalized.
d. Final Low Volume APCs and Limit on ASC Payment Rates for Procedures Assigned to Low Volume APCs
As stated in section XIII.D.1.b. of the CY 2025 OPPS/ASC proposed rule, the ASC payment system generally uses OPPS geometric mean costs under the standard methodology to determine proposed relative payment weights under the standard ASC ratesetting methodology.
In the CY 2022 OPPS/ASC final rule with comment period (86 FR 63743 through 63747), we adopted a universal Low Volume APC policy for CY 2022 and subsequent calendar years. Under our policy, we expanded the low volume adjustment policy that is applied to procedures assigned to New Technology APCs to also apply to clinical and brachytherapy APCs. Specifically, a clinical APC or brachytherapy APC with fewer than 100 claims per year would be designated as a Low Volume APC. For items or services assigned to a Low Volume APC, we use up to 4 years of claims data to establish a payment rate for the APC as we currently do for low volume services assigned to New Technology APCs. The payment rate for a Low Volume APC or a low volume New Technology procedure would be based on the highest of the median cost, arithmetic mean cost, or geometric mean cost calculated using multiple years of claims data.
Based on claims data available for the CY 2026 OPPS/ASC proposed rule, we proposed to designate six brachytherapy APCs and four clinical APCs as Low Volume APCs under the ASC payment system. The four clinical APCs and six brachytherapy APCs met our criteria of having fewer than 100 single claims in the relevant claims year (CY 2024 for the CY 2026 OPPS/ASC proposed rule) and therefore, we proposed that they would be subject to our universal Low Volume APC policy and the APC cost metric would be based on the greater of the median cost, arithmetic mean cost, or geometric mean cost using up to 4 years of claims data. Nine of the ten APCs were designated as Low Volume APCs in CY 2025. Based on data for the CY 2026 OPPS/ASC proposed rule, APC 2645 (Brachytx, non-stranded, gold-198) had 103 single claims and no longer met our criteria to be designated as a Low Volume APC; however, APC 2643 (Brachytx, non-stranded, c-131) had only 88 single claims and met our criteria to be designated as a Low Volume APC.
We did not receive any comments on our proposal. Based on claims data available for this final rule with comment period, we are finalizing our proposal to designate the four clinical APCs and six brachytherapy APCs shown in Table 127 as Low Volume APCs under the ASC payment system, because they continue to meet our criteria of having fewer than 100 single claims in the relevant claims year (CY 2024). Table 127 includes the CY 2024 claims available for ratesetting for each of the APCs we are finalizing to be designated as a Low Volume APCs for CY 2026. The cost statistics for our Low Volume APCs, such as the median, arithmetic mean, and geometric mean cost are available for download with this final rule with comment period on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/ambulatory-surgical-center-asc/ ( printed page 53846) ;ascregulations-and-notices; click on the relevant regulation to download the Low Volume APC cost statistics under the standard (ASC) ratesetting methodology in the “Downloads” section of the web page.
2. Payment for Covered Ancillary Services
a. Background
Our payment policies under the ASC payment system for covered ancillary services generally vary according to the particular type of service and its payment policy under the OPPS. Our overall policy provides separate ASC payment for certain ancillary items and services integrally related to the provision of ASC covered surgical procedures that are paid separately under the OPPS and provides packaged ASC payment for other ancillary items and services that are packaged or conditionally packaged (status indicators “N,” “Q1,” and “Q2”) under the OPPS.
In the CY 2013 OPPS/ASC rulemaking (77 FR 45169 and 77 FR 68457 through 68458), we further clarified our policy regarding the payment indicator assignment for procedures that are conditionally packaged in the OPPS (status indicators “Q1” and “Q2”). Under the OPPS, a conditionally packaged procedure describes a HCPCS code where the payment is packaged when it is provided with a significant procedure but is separately paid when the service appears on the claim without a significant procedure. Because ASC services always include a surgical procedure, HCPCS codes that are conditionally packaged under the OPPS are generally packaged (payment indicator “N1”) under the ASC payment system (except for device removal procedures, as discussed in the CY 2022 OPPS/ASC proposed rule (86 FR 42083)). Thus, our policy generally aligns ASC payment bundles with those under the OPPS (72 FR 42495). In all cases, in order for ancillary items and services also to be paid, the ancillary items and services must be provided integral to the performance of ASC covered surgical procedures for which the ASC bills Medicare.
Our ASC payment policies generally provide separate payment for drugs and biologicals that are separately paid under the OPPS at the OPPS rates and package payment for drugs and biologicals for which payment is packaged under the OPPS. However, as discussed in the CY 2022 OPPS/ASC final rule with comment period, for CY 2022, we finalized a policy to unpackage and pay separately at ASP plus 6 percent for the cost of non-opioid pain management drugs and biologicals that function as a supply when used in a surgical procedure as determined by CMS under § 416.174 (86 FR 63483).
We generally pay for separately payable radiology services at the lower of the PFS nonfacility PE RVU-based (or technical component) amount or the rate calculated according to the ASC standard ratesetting methodology (72 FR 42497). However, as finalized in the CY 2011 OPPS/ASC final rule with comment period (75 FR 72050), payment indicators for all nuclear medicine procedures (defined as CPT codes in the range of 78000 through 78999) that are designated as radiology services that are paid separately when provided integral to a surgical procedure on the ASC list are set to “Z2” so that payment is made based on the ASC standard ratesetting methodology rather than the PFS nonfacility PE RVU amount (“Z3”), regardless of which is lower (§ 416.171(d)(1)).
Similarly, we also finalized our policy to set the payment indicator to “Z2” for radiology services that use contrast agents so that payment for these procedures will be based on the OPPS relative payment weight using the ASC standard ratesetting methodology and, therefore, will include the cost for the contrast agent (§ 416.171(d)(2)).
ASC payment policy for brachytherapy sources mirrors the payment policy under the OPPS. ASCs are paid for brachytherapy sources provided integral to ASC covered surgical procedures at prospective rates adopted under the OPPS or, if OPPS rates are unavailable, at contractor-priced rates (72 FR 42499). Since December 31, 2009, ASCs have been paid for brachytherapy sources provided integral to ASC covered surgical procedures at prospective rates adopted under the OPPS.
Our ASC policies also provide separate payment for: (1) certain items and services that CMS designates as contractor-priced, including, but not limited to, the procurement of corneal tissue; and (2) certain implantable items that have pass-through payment status under the OPPS. These categories do not have prospectively established ASC payment rates according to ASC payment system policies (72 FR 42502 and 42508 through 42509; § 416.164(b)). Under the ASC payment system, we have designated corneal tissue acquisition and hepatitis B vaccines as contractor-priced. Corneal tissue ( printed page 53847) acquisition is contractor-priced based on the invoice costs for acquiring the corneal tissue for transplantation. Hepatitis B vaccines are contractor-priced based on invoiced costs for the vaccine.
Devices that are eligible for pass-through payment under the OPPS are separately paid under the ASC payment system and are contractor-priced. Under the revised ASC payment system (72 FR 42502), payment for the surgical procedure associated with the pass-through device is made according to our standard methodology for the ASC payment system, based on only the service (non-device) portion of the procedure's OPPS relative payment weight if the APC weight for the procedure includes other packaged device costs. We also refer to this methodology as applying a “device offset” to the ASC payment for the associated surgical procedure. This ensures that duplicate payment is not provided for any portion of an implanted device with OPPS pass-through payment status.
In the CY 2015 OPPS/ASC final rule with comment period (79 FR 66933 through 66934), we finalized that, beginning in CY 2015, certain diagnostic tests within the medicine range of CPT codes for which separate payment is allowed under the OPPS are covered ancillary services when they are integral to an ASC covered surgical procedure. We finalized that diagnostic tests within the medicine range of CPT codes include all Category I CPT codes in the medicine range established by CPT, from 90000 to 99999, and Category III CPT codes and Level II HCPCS codes that describe diagnostic tests that crosswalk or are clinically similar to procedures in the medicine range established by CPT. In the CY 2015 OPPS/ASC final rule with comment period, we also finalized our policy to pay for these tests at the lower of the PFS nonfacility PE RVU-based (or technical component) amount or the rate calculated according to the ASC standard ratesetting methodology (79 FR 66933 through 66934). We finalized that the diagnostic tests for which the payment is based on the ASC standard ratesetting methodology be assigned to payment indicator “Z2” and revised the definition of payment indicator “Z2” to include a reference to diagnostic services and those for which the payment is based on the PFS nonfacility PE RVU-based amount be assigned payment indicator “Z3,” and revised the definition of payment indicator “Z3” to include a reference to diagnostic services.
b. Final Payment for Covered Ancillary Items and Services for CY 2026
We proposed to update the ASC payment rates and to make changes to ASC payment indicators, as necessary, to maintain consistency between the OPPS and ASC payment system regarding the packaged or separately payable status of services and the proposed CY 2026 OPPS and ASC payment rates and subsequent years' payment rates. We proposed to continue to set the CY 2026 ASC payment rates and subsequent years' payment rates for brachytherapy sources and separately payable drugs and biologicals equal to the OPPS payment rates for CY 2026 and subsequent years' payment rates.
Comment: A commenter stated that there is currently no CMS-level guidance on how MACs should establish separate payment under the ASC payment system for devices that are eligible for pass-through payment under the OPPS.
Response: Devices that are eligible for pass-through payment under the OPPS may be separately paid under the ASC payment system and are contractor-priced. Currently, MACs have been instructed to pay for such devices in the ASC setting based on invoice or cost of the approved transitional pass-through device category. While we understand that many products with different price points may use the same approved transitional pass-through device category, we rely on the MACs to efficiently process these claims such that ASCs can be paid in a timely manner for the services provided and that payment for these pass-through devices is justified with supporting documentation. We believe the current guidance provided in Chapter 14, section 50 of the Medicare Claims Processing Manual (Pub. 100-04) is sufficient.
After consideration of the public comments we received, we are finalizing our proposal to update the ASC payment rates and to make changes to ASC payment indicators, as necessary, to maintain consistency between the OPPS and ASC payment system regarding the packaged or separately payable status of services and the final CY 2026 OPPS and ASC payment rates and subsequent years' payment rates. We are also finalizing our proposal without modification to continue to set the CY 2026 ASC payment rates for brachytherapy sources and separately payable drugs and biologicals equal to the OPPS payment rates for CY 2026 and subsequent years.
Covered ancillary services and their final payment indicators for CY 2026 are listed in Addendum BB of this final rule with comment period (which is available via the internet on the CMS website). For those covered ancillary services where the payment rate is the lower of the rate under the ASC standard rate setting methodology and the PFS proposed rates (similar to our office-based payment policy), the final payment indicators and rates set forth in this final rule with comment period are based on a comparison using the final PFS rates effective January 1, 2026. For a discussion of the PFS rates, we refer readers to the CY 2026 PFS final rule with comment period which is available on the CMS website at https://www.cms.gov/medicare/payment/fee-schedules/physician/federal-regulation-notices.
3. Covered Surgical Procedures Designated as Office-Based Procedures
a. Background
In the August 2, 2007 ASC final rule with comment period, we finalized our policy to designate as “office-based” those procedures that are added to the ASC Covered Procedures List (CPL) in CY 2008 or later years that we determine are furnished predominantly (more than 50 percent of the time) in physicians' offices based on consideration of the most recently available volume and utilization data for each individual procedure code and/or, if appropriate, the clinical characteristics, utilization, and volume of related codes. In that final rule, we also finalized our policy to exempt all procedures on the CY 2007 ASC list from application of the office-based classification (72 FR 42512). The procedures that were added to the ASC CPL beginning in CY 2008 that we determined were office-based were identified in Addendum AA to that final rule with payment indicator “P2” (Office-based surgical procedure added to ASC list in CY 2008 or later with PFS nonfacility PE RVUs; payment based on OPPS relative payment weight); “P3” (Office-based surgical procedures added to ASC list in CY 2008 or later with PFS nonfacility PE RVUs; payment based on PFS nonfacility PE RVUs); or “R2” (Office-based surgical procedure added to ASC list in CY 2008 or later without PFS nonfacility PE RVUs; payment based on OPPS relative payment weight), depending on whether we estimated the procedure would be paid according to the ASC standard ratesetting methodology based on its OPPS relative payment weight or at the PFS nonfacility PE RVU-based amount.
Consistent with our final policy to annually review and update the ASC ( printed page 53848) CPL to include all covered surgical procedures eligible for payment in ASCs, each year we identify covered surgical procedures as either temporarily office-based (these are new procedure codes with little or no utilization data that we have determined are clinically similar to other procedures that are permanently office-based), permanently office-based, or nonoffice-based, after taking into account updated volume and utilization data.
b. CY 2026 Final Office-Based Procedures
In developing the CY 2026 OPPS/ASC proposed rule, we followed our policy to annually review and update the covered surgical procedures for which ASC payment is made and to identify new procedures that may be appropriate for ASC payment, including their potential designation as office-based. Historically, we also review the most recent claims volume and utilization data (CY 2024 claims) and the clinical characteristics for all covered surgical procedures that are currently assigned a payment indicator in CY 2025 of “G2” (Non office-based surgical procedure added in CY 2008 or later; payment based on OPPS relative payment weight) as well as for those procedures assigned one of the temporary office-based payment indicators, specifically “P2,” “P3,” or “R2” in the CY 2025 OPPS/ASC final rule with comment period (89 FR 94322 through 94326).
Our review of the CY 2024 volume and utilization data of covered surgical procedures currently assigned a payment indicator of “G2” (Non office-based surgical procedure added in CY 2008 or later; payment based on OPPS relative payment weight) resulted in the identification of one surgical procedure—CPT code 21930 (Excision, tumor, soft tissue of back or flank, subcutaneous; less than 3 cm)—that we believed met the criteria for designation as permanently office-based. The data indicated that this procedure is performed more than 50 percent of the time in physicians' offices, and the services are of a level of complexity consistent with other procedures performed routinely in physicians' offices. We have included CPT code 21930 in our list of surgical procedures to permanently designate as office-based for CY 2026 in Table 128.
As discussed in the August 2, 2007 ASC final rule with comment period (72 FR 42533 through 42535), we finalized our policy to designate certain new surgical procedures as temporarily office-based until adequate claims data are available to assess their predominant sites of service, whereupon if we confirm their office-based nature, the procedures are permanently assigned to the list of office-based procedures. In the absence of claims data, we use other available information, including our clinical advisors' judgment, predecessor CPT and Level II HCPCS codes, information submitted by representatives of specialty societies and professional associations, and information submitted by commenters during the public comment period.
In Table 153 of the CY 2025 OPPS/ASC final rule with comment period, we finalized assigning temporary office-based designations to nine surgical procedures for CY 2025 (89 FR 94325 through 94326). As discussed in section XIII.B. of the CY 2026 OPPS/ASC proposed rule (90 FR 33710 through 33713), two of the nine procedures were deleted effective April 2025—HCPCS codes G0564 and G0565. For two of the remaining seven surgical procedures, interested parties submitted information that suggested CPT code 15013 (Preparation of skin cell suspension autograft, requiring enzymatic processing, manual mechanical disaggregation of skin cells, and filtration; first 25 sq cm or less of harvested skin) and its automated counterpart HCPCS C8002 (Preparation of skin cell suspension autograft, automated, including all enzymatic processing and device components (do not report with manual suspension preparation)) are not most similar to CPT code 11310 (Shaving of epidermal or dermal lesion, single lesion, face, ears, eyelids, nose, lips, mucous membrane; lesion diameter 0.5 cm or less) as we stated in the CY 2025 OPPS/ASC final rule with comment period (89 FR 94322 through 94324) since CPT code 15013 must be performed with other skin cell suspension autograft procedure codes and the entirety of the procedure—harvesting of skin, preparation and application of the skin cell suspension autograft—is not expected to be predominantly performed in an office setting. After reviewing the information and consulting with our medical officers, we agree that the entirety of the procedure is not expected to be performed in a physician office setting and that CPT code 11310 would not be an accurate crosswalk for site-of-service utilization. Therefore, as shown in Table 78 of the CY 2026 OPPS/ASC proposed rule (90 FR 33712), we proposed to permanently remove the temporarily office-based designation for CPT code 15013 and HCPCS code C8002.
We reviewed CY 2024 volume and utilization data for the remaining five surgical procedures designated as temporarily office-based in the CY 2025 OPPS/ASC final rule with comment period. As shown in Table 77 and Table 78 of the CY 2026 OPPS/ASC proposed rule (90 FR 33711 through 33712), for one of the five surgical procedures—CPT code 0864T—there are greater than 50 claims available and the volume and utilization indicated this procedure was performed predominantly in the office setting. Therefore, we proposed to no longer designate this procedure as temporarily office-based and to permanently designate this procedure as office-based and assign one of the office-based payment indicators, specifically “P2”, “P3”, or “R2.”
We did not receive any public comments on our proposed permanent office-based designations and proposal to no longer designate the procedures in Table 129 as temporarily office-based; therefore, we are finalizing our proposal to designate the procedures in Table 128 as permanently office-based beginning in CY 2026 and to no longer designate the procedures in Table 129 as temporarily office-based.
( printed page 53849)For the remaining four procedures that were designated as temporarily office-based in the CY 2025 OPPS/ASC final rule with comment period and temporarily assigned one of the office-based payment indicators, specifically “P2,” “P3,” or “R2,” there were fewer than 50 claims; therefore, there was an insufficient number of claims to determine if the office setting was the predominant setting of care for these procedures. Therefore, as shown in Table 79 of the CY 2026 OPPS/ASC proposed rule (90 FR 33713), we proposed to continue to designate such procedures as temporarily office-based for CY 2026 and assign one of the office-based payment indicators. Additionally, for CY 2026, we did not propose to designate any new CY 2026 CPT codes for ASC covered surgical procedures as temporarily office-based.
We did not receive public comments on this provision, and therefore, we are finalizing our proposed temporarily office-based designation to the procedures shown below in Table 130. The procedures for which the final office-based designation for CY 2026 is temporary are also indicated by an asterisk in Addendum AA to this final rule with comment period (which is available via the internet on the CMS website at https://www.cms.gov/Medicare/Medicare-Feefor-Service-Payment/ASCPayment/ASCRegulations-and-Notices).
( printed page 53850)4. Device-Intensive ASC Covered Surgical Procedures
a. Background
We refer readers to the CY 2019 OPPS/ASC final rule with comment period (83 FR 59040 through 59041), for a summary of our existing policies regarding ASC covered surgical procedures that are designated as device-intensive.
In the CY 2019 OPPS/ASC final rule with comment period (83 FR 59040 through 59043), we modified our criteria for device-intensive procedures to better capture costs for procedures with significant device costs. We adopted a policy to allow procedures that involve surgically inserted or implanted, high-cost, single-use devices to qualify as device-intensive procedures. In addition, we modified our criteria to lower the device offset percentage threshold from 40 percent to 30 percent. The device offset percentage is the percentage of device costs within a procedure's total costs. Specifically, for CY 2019 and subsequent years, we adopted a policy that device-intensive procedures would be subject to the following criteria:
- All procedures must involve implantable or insertable devices assigned a CPT or HCPCS code;
- The required devices (including single-use devices) must be surgically inserted or implanted; and
- The device offset amount must be significant, which is defined as exceeding 30 percent of the procedure's mean cost. Corresponding to this change in the cost criterion, we adopted a policy that the default device offset for new codes that describe procedures that involve the implantation of medical devices will be 31 percent beginning in CY 2019. For new codes describing procedures that are payable when furnished in an ASC and involve the implantation of a medical device, we adopted a policy that the default device offset would be applied in the same manner as the policy we adopted in section IV.B.2 of the CY 2019 OPPS/ASC final rule with comment period (83 FR 58944 through 58948). We amended § 416.171(b)(2) of the regulations to reflect these new device criteria.
In addition, as also adopted in section IV.B.2. of the CY 2019 OPPS/ASC final rule with comment period, to further align the device-intensive policy with the criteria used for device pass-through status, we specified, for CY 2019 and subsequent years, that for purposes of satisfying the device-intensive criteria, a device-intensive procedure must involve a device that:
- Has received FDA marketing authorization, has received an FDA investigational device exemption (IDE) and has been classified as a Category B device by FDA in accordance with42 CFR 405.203 through 405.207 and 405.211 through 405.215, or meets another appropriate FDA exemption from premarket review;
- Is an integral part of the service furnished;
- Is used for one patient only;
- Comes in contact with human tissue;
- Is surgically implanted or inserted (either permanently or temporarily); and
- Is not any of the following: ( printed page 53851)
++ Equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciable assets as defined in Chapter 1 of the Medicare Provider Reimbursement Manual (CMS Pub. 15-1); or
++ A material or supply furnished incident to a service (for example, a suture, customized surgical kit, scalpel, or clip, other than a radiological site marker).
In the CY 2022 OPPS/ASC final rule with comment period (86 FR 63773 through 63775), we modified our approach to assigning device-intensive status to surgical procedures under the ASC payment system. First, we adopted a policy of assigning device-intensive status to procedures that involve surgically inserted or implanted, high-cost, single-use devices if their device offset percentage exceeds 30 percent under the ASC standard ratesetting methodology, even if the procedure is not designated as device-intensive under the OPPS. Second, we adopted a policy that if a procedure is assigned device-intensive status under the OPPS, but has a device offset percentage below the device-intensive threshold under the standard ASC ratesetting methodology, the procedure will be assigned device-intensive status under the ASC payment system with a default device offset percentage of 31 percent. The policies were adopted to provide consistency between the OPPS and ASC payment system and provide a more appropriate payment rate for surgical procedures with significant device costs under the ASC payment system.
In the CY 2023 OPPS/ASC final rule with comment period (87 FR 72078 through 72080), we finalized our policy to create certain C-codes, or ASC complexity adjustment codes that describe certain combinations of a primary covered surgical procedure as well as a packaged (payment indicator = “N1”) procedure that are otherwise eligible for a complexity adjustment under the OPPS (as listed in Addendum J). Each ASC complexity adjustment code's APC assignment is based on its corresponding OPPS complexity adjustment code's APC assignment. In the CY 2023 OPPS/ASC final rule with comment period, we stated our belief that it would be appropriate for these ASC complexity adjustment codes to qualify for device-intensive status under the ASC payment system if the primary procedure of the code was also designated as device-intensive. Under our current policy, the ASC complexity adjustment code retains the device portion of the primary procedure (also called the “device offset amount”) and not the device offset percentage. Therefore, for device-intensive ASC complexity adjustment codes, we set the device portion of the combined procedure equal to the device portion of the primary procedure and calculate the device offset percentage by dividing the device portion by the ASC complexity adjustment code's APC payment rate. Further, we apply our standard ASC payment system ratesetting methodology to the non-device portion of the ASC complexity adjustment code's APC payment rate; that is, we multiply the OPPS relative weight by the ASC budget neutrality adjustment and the ASC conversion factor and sum that amount with the device portion to calculate the ASC payment rate.
In the CY 2025 OPPS/ASC final rule with comment period, we finalized a modification to our policy regarding default device offset percentages for new codes that meet our criteria for device-intensive status. Under both the OPPS and ASC payment system, for new device-intensive procedures that lack claims data, or lack claims data from a predecessor code or a clinically-similar code that uses the same device, we apply the greater of the APC-wide device offset percentage or 31 percent (the previous default device offset percentage). We believe that an APC-wide average device offset percentage is, in most cases, a better reflection of device costs when the typical device costs of procedures assigned to such APC are significantly greater than 31 percent. This policy does not apply to new device-intensive procedures assigned to New Technology APCs.
b. CY 2026 Final Device Intensive Procedures
In section V.B. of this final rule with comment period, we discuss the implementation of the Final Remedy for the 340B-Acquired Drug Payment Policy for Calendar Years 2018-2022 rule and the impact of the OPPS conversion factor on the ASC payment system. Since most ASC payment rates for surgical procedures are constructed from OPPS relative weights or the PFS unadjusted nonfacility PE RVU-based amount, the remedy's proposed prospective offset to the OPPS conversion has a very limited impact on the ASC payment system. The only impact of the proposed reduction to the OPPS conversion factor is the payment rate for device-intensive procedures under the ASC payment system. Since the ASC payment system holds device portions constant between the two settings, the device portion is the device offset percentage multiplied by the OPPS payment rate.
Historically, in our proposed rules, device portions for device-intensive procedures would be based on the proposed prospective OPPS conversion factor multiplied by the proposed prospective OPPS relative weights. However, for the CY 2026 OPPS/ASC proposed rule, we believed it would be inaccurate and inappropriate to use OPPS payment rates that have been reduced by the remedy's prospective offset since this could accumulate to have a potentially noticeable impact on ASC payment rates for certain device-intensive procedures over time. Since the ASC payment system would otherwise set the device portion in the ASC setting at the amount without the proposed offset reduction to OPPS payment rates, we believe it would not be an accurate reflection of the device costs of covered surgical procedures in the ASC setting if we were to incorporate the prospective offset that we proposed in the CY 2026 OPPS/ASC proposed rule. Further, we are concerned beneficiaries could have access issues to certain device-intensive procedures in the ASC setting if we maintained a reduction to the payment rates for device-intensive procedures for each calendar year we applied the prospective offset. Therefore, we proposed that the OPPS payment rates used for ratesetting under the ASC payment system for CY 2026 and subsequent years would not incorporate the prospective offset to the OPPS conversion factor as a result of the 340B remedy offset that we proposed to implement in the proposed rule. For the proposed CY 2026 device offset percentages, which include device offset percentages based on CY 2024 claims processed through March 31, 2025, we refer readers to Addendum FF of the CY 2026 OPPS/ASC proposed rule. Final CY 2026 device offset percentages may differ from the proposed percentages, as we rely on the most recently available claims data for the CY 2026 OPPS/ASC final rule with comment period (CY 2024 claims data processed through June 30th).
We received public comments on our proposal. The following is a summary of the comments we received and our responses.
Comment: A commenter requested that CMS use manufacturer invoices to set the OPPS and ASC device offset amounts/percentages for CPT code 64728 (placeholder code 647XX) (Decompression; median nerve at the carpal tunnel, percutaneous, with intracarpal tunnel balloon dilation, including ultrasound guidance), stating ( printed page 53852) that the device offset percentage should be significantly higher than the proposed default of 31 percent to ensure appropriate ASC payment and Medicare beneficiary access to CTR-US in ASCs.
Response: We are not accepting the commenter's recommendation. Notwithstanding the rare circumstances of an extremely costly device, we believe the application of our default device offset percentage policy for device-intensive procedures is a reasonable and appropriate approach for determining the device portions of procedures without claims data. Our default device offset percentage policy allows us to both estimate the approximate cost of the device but also supports cost containment objectives that our packaging policies aim to achieve under both the OPPS and ASC payment systems.
Comment: Commenters requested that CMS designate CPT code 43889 (Gastric restrictive procedure, transoral, endoscopic sleeve gastroplasty (ESG), including argon plasma coagulation, when performed) as device-intensive by assigning status indicator “J8” instead of the proposed “G2” status, stating that the procedure includes multiple single-use devices with hospital acquisition costs of $4,000 to $7,000 per patient, including an endoscopic suturing system, tissue helix, polypropylene sutures, and suture anchors. The commenter noted that CMS should not rely on the aberrant and very low volume data set from predecessor HCPCS code C9784 (Gastric restrictive procedure, endoscopic sleeve gastroplasty, with esophagogastroduodenoscopy and intraluminal tube insertion, if performed, including all system and tissue anchoring components), which showed inconsistent device offset percentages ranging from 12.03 percent to 52.94 percent across different years that do not approximate actual device costs. The commenter requested that CMS apply the default device offset amount of 31 percent to CPT code 43889 based on documented invoice cost of the device rather than unreliable claims data.
Response: We are not accepting the commenter's recommendation. CPT code 43889 has available CY 2024 claims data from predecessor code C9784. The device offset percentage of such claims data does not exceed our device-intensive threshold; therefore, we are not accepting the commenter's recommendation to assign device-intensive status to CPT code 43889.
Comment: A commenter recommended that CPT codes 22802 (Arthrodesis, posterior, for spinal deformity, with or without cast; 7 to 12 vertebral segments), 22804 (Arthrodesis, posterior, for spinal deformity, with or without cast; 13 or more vertebral segments), 22810 (Arthrodesis, anterior, for spinal deformity, with or without cast; 4 to 7 vertebral segments), and 22812 (Arthrodesis, anterior, for spinal deformity, with or without cast; 8 or more vertebral segments) receive device-intensive status when added to the ASC CPL. The commenter requested that CMS apply device offsets of 55.46 percent to CPT codes 22802 and 22804 (equivalent to clinically similar CPT code 22800) and 56.97 percent to CPT codes 22810 and 22812 (equivalent to clinically similar CPT code 22808), stating that CMS should use device offsets from clinically similar codes that use the same devices when claims data is not available for procedures recently removed from the IPO list. The commenter emphasized that device offset data frequently may not be available for procedures recently removed from the IPO list, and recommended that CMS use the greater of the APC-wide device offset percentage or the default device offset percentage of 31 percent if clinically similar code data is not available.
Response: We agree with the commenters that such procedures would have significant device costs. We will rely on CY 2024 claims data, where available, for establishing the device portions for CPT codes 22802, 22804, 22810, and 22812 for CY 2026. Where claims data is unavailable, we will rely on default device offset percentage methodology for determining the device portion. We are accepting the commenter's recommendations to assign device-intensive status to CPT codes 22802, 22804, 22810, and 22812 for CY 2026.
Comment: Some commenters recommended that we maintain the device offset percentage for CPT code 0621T (Trabeculostomy ab interno by laser).
Response: Where available, we rely on hospital claims data for determining the device offset percentages for procedures under the ASC payment system. Based on CY 2024 claims data available for this final rule with comment period, we are finalizing device-intensive status for CPT code 0621T as this procedure meets our criteria for device-intensive status. The device offset percentages for CPT code 0621T are based on CY 2024 claims data and can be found in Addendum FF to this final rule with comment period.
Comment: A commenter recommended that we revise the ASC payment indicators for CPT codes 19281 (Placement of breast localization device(s) ( e.g., clip, metallic pellet, wire/needle, radioactive seeds), percutaneous; first lesion, including mammographic guidance) and 19285 (Placement of breast localization device(s) ( e.g., clip, metallic pellet, wire/needle, radioactive seeds), percutaneous; first lesion, including ultrasound guidance) from their current designation to J8—“Device-intensive procedure added to ASC list in CY 2008 or later; paid at adjusted rate”—to permit Medicare ASC payment for these services. The commenter noted these codes have significant device costs (51.01 percent and 53.87 percent respectively), but current lack of ASC payment prevents Medicare patients from accessing wire-free, nonradioactive soft tissue localization services in the cost-effective ASC setting.
Response: We are not accepting the commenter's recommendation. As we stated in previous rulemaking (78 FR 75081), under the OPPS, a conditionally packaged procedure (status indicators “Q1” and “Q2) describes a HCPCS code where the payment is packaged when it is provided with a significant procedure but is separately paid when the service appears on the claim without a significant procedure. Because ASC services always include a covered surgical procedure, HCPCS codes that are conditionally packaged under the OPPS are generally packaged (payment indicator “N1”) under the ASC payment system and their costs are packaged into the cost for the covered surgical procedure and reflected in the payment rate.
Comment: A few commenters supported the designation of CPT code 31295 (Nasal/sinus endoscopy, surgical, with dilation ( e.g., balloon dilation); maxillary sinus ostium, transnasal or via canine fossa) as “device intensive,” stating this accurately recognizes device costs and results in appropriate payment. However, commenters expressed concerns regarding the removal of CPT code 31298 (Nasal/sinus endoscopy, surgical, with dilation ( e.g., balloon dilation); frontal and sphenoid sinus ostia) from the device intensive list, stating that this procedure continues to involve high device costs and should retain its classification to maintain predictable ASC payments and patient access. The commenter requested CMS reexamine Medicare claims data for accuracy, noting that CPT 31298 should have a device offset greater than 30 percent since it is more device intensive than CPT 31296 (which has a 28.5 percent offset) because it includes both frontal and sphenoid sinuses rather than just frontal. ( printed page 53853)
Response: We appreciate the commenter's recommendation. Based on our review of CY 2024 claims data available for this final rule with comment period, the device offset percentage for CPT code 31298 does not exceed our device-intensive threshold; therefore, we are not assigning device-intensive status to CPT code 31298 for CY 2026.
Comment: A few commenters requested that CMS verify the accuracy of its device offset calculations for CPT code 66174 (Transluminal dilation of aqueous outflow canal ( e.g., canaloplasty); without retention of device or stent) and assign device-intensive status (J8) if the device offset exceeds the 30 percent threshold. Both commenters expressed concerns about under-reporting of hospital device costs and inappropriate coding processes that have resulted in denial of device-intensive status despite the procedure requiring advanced microcatheters or permanently implanted devices, which limits Medicare patient access to these procedures in the cost-effective ASC setting.
Response: Based on our review of CY 2024 claims data available for this final rule with comment period, the device offset percentage for CPT code 66174 does not exceed our device-intensive threshold; therefore, we are not accepting the commenter's recommendation to assign device-intensive status to CPT code 66174 for CY 2026.
Comment: A few commenters requested that CMS assign device-intensive status to HCPCS code C9785 (Endoscopic outlet reduction, gastric pouch application, with endoscopy and intraluminal tube insertion, if performed, including all system and tissue anchoring components) and HCPCS code C9901 (Endoscopic defect closure within the entire gastrointestinal tract, including upper endoscopy (including diagnostic, if performed) or colonoscopy (including diagnostic, if performed), with all system and tissue anchoring components) instead of the proposed G2 payment indicator, stating that the procedures includes multiple single-use devices with hospital acquisition costs of $4,000 to $7,000 per patient, including an endoscopic suturing system, tissue helix, polypropylene sutures, cobalt chromium and stainless-steel alloy suture anchors, and polyetheretherketone (PEEK) suture cinches. The commenters noted that the current claims data is insufficient due to extremely low volume (fewer than 10 single frequency claims in 2025 and fewer than 50 in 2026) and should not be used to determine device intensive status. Both commenters requested that CMS apply the default device offset of 31 percent.
Response: We rely on hospitals to accurately report device costs for OPPS/ASC ratesetting and for determining device offset percentages. Based on our review of CY 2024 claims data available for this final rule with comment period, the device offset percentage for HCPCS codes C9785 does not exceed our device-intensive threshold; therefore, we are not accepting the commenter's recommendations to assign device-intensive status to HCPCS codes C9785 for CY 2026. However, the device offset percentage for C9901 based on claims data available for this final rule with comment period does exceed our device-intensive threshold. Based on the more recent claims data, we are finalizing device-intensive status under the ASC payment system for HCPCS code C9901 for CY 2026.
Comment: A commenter requested that CMS re-evaluate device-intensive status to CPT code 55880 (Ablation of malignant prostate tissue, transrectal, with high intensity-focused ultrasound (hifu), including ultrasound guidance) stating that the procedure is as device-intensive as other procedures that treat malignancies through the prostate.
Response: Based on our review of CY 2024 claims data available for this final rule with comment period, the device offset percentages for CPT code 55880 does not exceed our device-intensive threshold; therefore, we are not assigning device-intensive status to CPT code 55880 for CY 2026.
Comment: Commenters recommended adding CPT codes 0582T (Transurethral ablation of malignant prostate tissue by high-energy water vapor thermotherapy, including intraoperative imaging and needle guidance), 0786T (Insertion or replacement of percutaneous electrode array, sacral, with integrated neurostimulator, including imaging guidance, when performed), and 0991T (Cystourethroscopy, with low-energy lithotripsy and acoustically actuated microspheres, including imaging) to the ASC CPL and assigning device-intensive status to each of the CPT codes.
Response: We have reviewed the clinical characteristics and devices required of these procedures and are accepting the commenter's recommendations. As shown in Table 131 we are adding CPT codes 0582T, 0786T, and 0991T to the ASC CPL. We believe these procedures have significant device costs and are assigning these device-intensive status for CY 2026. In the absence of claims data for these three procedures, we are using our default device offset methodology to assign the device offset percentages to CPT codes 0582T, 0786T, and 0991T for CY 2026.
Comment: A commenter requested that we assign device-intensive status to CPT codes 0951T (Totally implantable active middle ear hearing implant; initial placement, including mastoidectomy, placement of and attachment to sound processor), 0952T (Totally implantable active middle ear hearing implant; revision or replacement, with mastoidectomy and replacement of sound processor), 0953T (Totally implantable active middle ear hearing implant; revision or replacement, without mastoidectomy and replacement of sound processor), and 0954T (Totally implantable active middle ear hearing implant; replacement of sound processor only, with attachment to existing transducers) under the ASC payment system.
Response: These codes are not separately payable under the OPPS; therefore, these procedures are not eligible to be added to the ASC CPL and be assigned device-intensive status under the ASC payment system. We are not accepting the commenters' recommendations to assign device-intensive status to CPT codes 0951T, 0952T, 0953T, or 0954T under the ASC payment system for CY 2026.
Comment: A commenter recommended that CMS establish a drug-intensive policy for ASC rate setting, similar to the current device-intensive policy, stating that the logic behind device-intensive procedures should equally apply to situations where a costly drug is packaged into a procedure payment since ASCs typically do not pay less than hospitals for expensive drugs. The commenter noted that just as the device-intensive policy allows ASC payment rates to better reflect device costs by applying the ASC conversion factor only to the non-device portion of the OPPS rate, a similar drug-intensive mechanism would ensure ASC rates more accurately represent the costs of costly packaged drugs.
Response: We are not accepting this recommendation. Unlike device costs, which are always packaged into the primary procedure (with the exception of devices on transitional pass-through status), many costly drugs that may be packaged under the OPPS into a Comprehensive APC are separately payable under the ASC payment system.
After consideration of public comments, we are finalizing our proposal to not incorporate the prospective offset to the OPPS conversion factor or device-related ( printed page 53854) portion as a result of the 340B remedy offset that we are finalizing to implement in this final rule with comment period. For final CY 2026 device offset percentages based on claims data for this final rule with comment period, CY 2024 claims processed through June 30, 2025, we refer readers to Addendum FF of this final rule with comment period.
c. Adjustment to ASC Payments for No Cost/Full Credit and Partial Credit Devices
Our ASC payment policy for costly devices implanted or inserted in ASCs at no cost/full credit or partial credit is set forth in § 416.179 of our regulations and is consistent with the OPPS policy that was in effect until CY 2014. We refer readers to the CY 2008 OPPS/ASC final rule with comment period (72 FR 66845 through 66848) for a full discussion of the ASC payment adjustment policy for no cost/full credit and partial credit devices. ASC payment is reduced by 100 percent of the device offset amount when a hospital furnishes a specified device without cost or with a full credit and by 50 percent of the device offset amount when the hospital receives partial credit in the amount of 50 percent or more of the cost for the specified device.
Effective CY 2014, under the OPPS, we finalized our proposal to reduce OPPS payment for applicable APCs by the full or partial credit a provider receives for a device, capped at the device offset amount. Although we finalized our proposal to modify the policy of reducing payments when a hospital furnishes a specified device without cost or with full or partial credit under the OPPS, in the CY 2014 OPPS/ASC final rule with comment period (78 FR 75076 through 75080), we finalized our proposal to maintain our ASC policy for reducing payments to ASCs for specified device-intensive procedures when the ASC furnishes a device without cost or with full or partial credit. Unlike the OPPS, there is currently no mechanism within the ASC claims processing system for ASCs to submit to CMS the amount of the actual credit received when furnishing a specified device at full or partial credit. Therefore, under the ASC payment system, we finalized our proposal for CY 2014 to continue to reduce ASC payments by 100 percent or 50 percent of the device offset amount when an ASC furnishes a device without cost or with full or partial credit, respectively.
Under current ASC policy, all ASC device-intensive covered surgical procedures are subject to the no cost/full credit and partial credit device adjustment policy. Specifically, when a device-intensive procedure is performed to implant or insert a device that is furnished at no cost or with full credit from the manufacturer, the ASC appends the HCPCS “FB” modifier on the line in the claim with the procedure to implant or insert the device. The contractor reduces payment to the ASC by the device offset amount that we estimate represents the cost of the device when the necessary device is furnished without cost or with full credit to the ASC. We continue to believe that the reduction of ASC payment in these circumstances is necessary to pay appropriately for the covered surgical procedure furnished by the ASC.
In the CY 2019 OPPS/ASC final rule with comment period (83 FR 59043 through 59044) we adopted a policy to reduce the payment for a device-intensive procedure for which the ASC receives partial credit by one-half of the device offset amount that would be applied if a device was provided at no cost or with full credit if the credit to the ASC is 50 percent or more (but less than 100 percent) of the cost of the new device. The ASC will append the HCPCS “FC” modifier to the HCPCS code for the device-intensive surgical procedure when the facility receives a partial credit of 50 percent or more (but less than 100 percent) of the cost of a device. To report that the ASC received a partial credit of 50 percent or more (but less than 100 percent) of the cost of a new device, ASCs have the option of either: (1) submitting the claim for the device-intensive procedure to their Medicare contractor after the procedure's performance, but prior to manufacturer acknowledgment of credit for the device, and subsequently contacting the contractor regarding a claim adjustment, once the credit determination is made; or (2) holding the claim for the device implantation or insertion procedure until a determination is made by the manufacturer on the partial credit and submitting the claim with the “FC” modifier appended to the implantation procedure HCPCS code if the partial credit is 50 percent or more (but less than 100 percent) of the cost of the device. Beneficiary coinsurance would be based on the reduced payment amount. As finalized in the CY 2015 OPPS/ASC final rule with comment period (79 FR 66926), to ensure our policy covers any situation involving a device-intensive procedure where an ASC may receive a device at no cost or receive full credit or partial credit for the device, we apply our “FB”/“FC” modifier policy to all device-intensive procedures.
In the CY 2019 OPPS/ASC final rule with comment period (83 FR 59043 through 59044) we stated we would reduce the payment for a device-intensive procedure for which the ASC receives partial credit by one-half of the device offset amount that would be applied if a device was provided at no cost or with full credit, if the credit to the ASC is 50 percent or more (but less than 100 percent) of the cost of the device. In the CY 2020 OPPS/ASC final rule with comment period, we finalized continuing our existing policies for CY 2020. We note that we inadvertently omitted language that this policy would apply not just in CY 2019 but also in subsequent calendar years. We intended to apply this policy in CY 2019 and subsequent calendar years. Therefore, we finalized our proposal to apply our policy for partial credits specified in the CY 2019 OPPS/ASC final rule with comment period (83 FR 59043 through 59044) in CY 2022 and subsequent calendar years (86 FR 63775 through 63776). Specifically, for CY 2022 and subsequent calendar years, we would reduce the payment for a device-intensive procedure for which the ASC receives partial credit by one-half of the device offset amount that would be applied if a device was provided at no cost or with full credit, if the credit to the ASC is 50 percent or more (but less than 100 percent) of the cost of the device. To report that the ASC received a partial credit of 50 percent or more (but less than 100 percent) of the cost of a device, ASCs have the option of either: (1) submitting the claim for the device intensive procedure to their Medicare contractor after the procedure's performance, but prior to manufacturer acknowledgment of credit for the device, and subsequently contacting the contractor regarding a claim adjustment, once the credit determination is made; or (2) holding the claim for the device implantation or insertion procedure until a determination is made by the manufacturer on the partial credit and submitting the claim with the “FC” modifier appended to the implantation procedure HCPCS code if the partial credit is 50 percent or more (but less than 100 percent) of the cost of the device. Beneficiary coinsurance would be based on the reduced payment amount.
We did not receive any comments on our policies related to no cost/full credit or partial credit devices, and we are finalizing the continuation of our existing policies for CY 2026 without modification. ( printed page 53855)
5. Requirement in the Physician Fee Schedule CY 2026 Final Rule for HOPDs and ASCs To Report Discarded Amounts of Certain Single-Dose or Single-Use Package Drugs
Section 90004 of the Infrastructure Investment and Jobs Act (Pub. L. 117-9, November 15, 2021) (“the Infrastructure Act”) amended section 1847A of the Act to re-designate subsection (h) as subsection (i) and insert a new subsection (h), which requires manufacturers to provide a refund to CMS for certain discarded amounts from a refundable single-dose container or single-use package drug.
The CY 2026 PFS proposed rule included proposals related to the discarded drug refund policy, including proposals that may impact hospital outpatient departments (HOPDs) and ambulatory surgical centers (ASCs). Similar to our CY 2023, CY 2024, and CY 2025 notices in the OPPS/ASC proposed rules (87 FR 71988, 88 FR 49760, and 89 FR 59421 through 59422), we included a notice in the CY 2026 OPPS/ASC proposed rule to ensure interested parties were aware of these proposals and knew to refer to the CY 2026 PFS proposed rule for a full description of the proposed policy. Interested parties were asked to submit comments on any proposals to further implement section 90004 of the Infrastructure Act to the CY 2026 PFS proposed rule. Public comments on these proposals are addressed in the CY 2026 PFS final rule with comment period. We note that this same notice appeared in section V.B. of the CY 2025 OPPS/ASC proposed rule (90 FR 33716).
We refer readers to the CY 2026 PFS final rule with comment period for a summary of comments, our responses, and the finalized policy for CY 2026.
D. Final Additions to ASC Covered Surgical Procedures and Covered Ancillary Services Lists
1. Current Review Process for the List of ASC Covered Surgical Procedures
Section 1833(i)(1) of the Act requires us, in part, to specify, in consultation with appropriate medical organizations, surgical procedures that are appropriately performed on an inpatient basis in a hospital but that can also be safely performed in an ASC, a CAH, or an HOPD, and to review and update the list of ASC covered surgical procedures at least every 2 years. We evaluate the ASC covered procedures list (ASC CPL) each year to determine whether procedures should be added to or removed from the list, and changes to the list are often made in response to specific concerns raised by interested parties.
Under our regulations at §§ 416.2 and 416.166, covered surgical procedures furnished on or after January 1, 2022, are surgical procedures that meet the general standards specified in § 416.166(b) and are not excluded under the general exclusion criteria specified in § 416.166(c). Specifically, under § 416.166(b), the general standards provide that covered surgical procedures are surgical procedures specified by the Secretary and published in the Federal Register and/or via the internet on the CMS website that are separately paid under the OPPS, that would not be expected to pose a significant safety risk to a Medicare beneficiary when performed in an ASC, and for which standard medical practice dictates that the beneficiary would not typically be expected to require active medical monitoring and care at midnight following the procedure.
Section 416.166(c) sets out the general exclusion criteria used under the ASC payment system to evaluate the safety of procedures for performance in an ASC. The general exclusion criteria provide that covered surgical procedures do not include those surgical procedures that: (1) generally result in extensive blood loss; (2) require major or prolonged invasion of body cavities; (3) directly involve major blood vessels; (4) are generally emergent or life-threatening in nature; (5) commonly require systemic thrombolytic therapy; (6) are designated as requiring inpatient care under § 419.22(n); (7) can only be reported using a CPT unlisted surgical procedure code; or (8) are otherwise excluded under § 411.15.
In the CY 2019 OPPS/ASC final rule with comment period (83 FR 59029 through 59030), we defined a surgical procedure under the ASC payment system as any procedure described within the range of Category I CPT codes that the CPT Editorial Panel of the AMA defines as “surgery” (CPT codes 10000 through 69999) (72 FR 42476), as well as procedures that are described by Level II HCPCS codes or by Category I CPT codes or by Category III CPT codes that directly crosswalk or are clinically similar to procedures in the CPT surgical range that we determined met the general standards established in previous years for addition to the ASC CPL.
For a detailed discussion of the history of our policies for adding surgical procedures to the ASC CPL, we refer readers to the CY 2021 through CY 2025 OPPS/ASC final rules with comment period (85 FR 86143 through 86145; 86 FR 63777 through 63805; 87 FR 72068 through 72076; 88 FR 81923 through 81945; and 89 FR 94331 through 94334).
2. Final Changes to the List of ASC Covered Surgical Procedures for CY 2026
Historically, we have reviewed the clinical characteristics of procedures and consulted with appropriate medical organizations, other interested parties, and our clinical advisors to determine if those procedures would meet our existing regulatory criteria under 42 CFR 416.2 and 42 CFR 416.166.
In the CY 2021 OPPS/ASC final rule with comment period, we significantly revised our policy for adding surgical procedures to the ASC CPL. Specifically, we revised our regulatory criteria by removing certain general standard and general exclusion criteria at 42 CFR 416.166(b) and (c), moving them to a new section as nonbinding physician considerations for patient safety (85 FR 86143 through 86153). We also stated that we would add surgical procedures when we identified a surgical procedure that met general standards criteria or when we were notified of a surgical procedure that could meet general standards criteria and we confirmed that the procedure met those requirements.
In the CY 2022 OPPS/ASC final rule with comment period, we reinstated the general standard and general exclusion criteria as part of the review process, rather than safety factors for physicians to consider, and renamed the notifications process finalized in the CY 2021 rule as a nominations process, later re-named the “Pre-Proposed Rule CPL Recommendation Process” (86 FR 63776 through 63782). Under this process, which became effective in CY 2024, an external party can recommend a surgical procedure by March 1 of a calendar year for the list of ASC covered surgical procedures for the following calendar year. As a result of the reinstatement of the general standard and general exclusion criteria, we finalized the removal of 255 procedures that had been added to the ASC CPL in CY 2021. We also maintained these criteria and the Pre-Proposed Rule CPL Recommendation Process during the CY 2023 through CY 2025 rulemaking cycles.
In the CY 2022 OPPS/ASC final rule with comment period, commenters were largely split on the issue of reinstating the general standard and general exclusion criteria at § 416.166 that were in place prior to CY 2021. Many commenters opposed this proposal and recommended that CMS not re-adopt these criteria. Commenters contended ( printed page 53856) that this policy may substitute administrative criteria for physician clinical judgment, reduce beneficiary choice, and increase costs since the lack of payment in the ASC setting may push these procedures to be performed in the higher-cost hospital setting.
For CY 2026, we proposed to revise our regulatory criteria at 42 CFR 416.166 to evaluate potential additions to the ASC CPL, similar to the changes we finalized in the CY 2021 OPPS/ASC final rule with comment period. Specifically, we proposed to revise our regulatory criteria by removing certain general standard and general exclusion criteria at 42 CFR 416.166(b) and (c), moving them to a new section as nonbinding physician considerations for patient safety. Under the revised criteria, we proposed to add certain surgical procedures to the ASC CPL, beginning in CY 2026, in order to expand access, while maintaining the safety for Medicare beneficiaries through the nonbinding physician considerations for patient safety.
a. ASC CPL Review Process for CY 2026
(1) Final Changes to General Standards and Exclusion Criteria for CY 2026
For CY 2026, we continued to build on our efforts to maximize patient and physician choice and access to care by exploring broader approaches to adding procedures to the ASC CPL in order to further increase the availability of ASCs as an alternative and often lower cost site of care for Medicare beneficiaries, while maintaining patient safety as required by section 1833(i) of the Act. An expansion of the ASC CPL would maximize the ability of ASCs to divert patients that can be safely treated in an ASC setting away from the hospital setting, which would preserve the capacity of hospitals to treat more acute patients. Expanding the procedures placed on the ASC CPL would also build on the policy changes we have made in recent years to further site neutrality between the HOPD and ASC settings.
In light of these objectives, we proposed to modify the existing general standard criteria under 42 CFR 416.166(b) that currently require covered surgical procedures to be surgical procedures specified by the Secretary and published in the Federal Register and/or via the internet on the CMS website, separately paid under the OPPS, not expected to pose a significant safety risk to a Medicare beneficiary when performed in an ASC, and for which standard medical practice dictates that the beneficiary would not typically be expected to require active medical monitoring and care at midnight following the procedure. We retained the condition that procedures be separately paid under the OPPS and moved the latter two standards to a new section outlining possible physician considerations in making site-of-service decisions.
We also proposed to eliminate five of the current general exclusion criteria at 42 CFR 416.166(c)(1) through (c)(5) and move them to the new physician considerations section. We believe these five exclusionary criteria may no longer be necessary to determine what procedures can be safely added to the ASC CPL because many ASCs are currently able to safely provide services with these characteristics, based on prior interested parties' feedback and public comments we have received. This would also allow physicians practicing in the ASC setting, who have the greatest familiarity and insight into the needs of individual beneficiaries, to use their complex medical judgement to determine whether they can safely perform a procedure in the ASC, given the entirety of the circumstances, including the clinical profile of the patient, the surgical back-up available at the ASC, and the ability to safely and timely respond to unexpected complications.
Under this proposal, we would keep the remaining three general exclusion criteria at § 416.166(c)(6) through (c)(8) because the original reasons we adopted them in CY 2008 continue to exist, subject to the proposed modifications to § 416.166(c)(6). These criteria would continue to exclude certain procedures from the ASC CPL, namely those that are designated as requiring inpatient care under 42 CFR 419.22(n), can only be reported using a CPT unlisted surgical procedure code, or are otherwise excluded under 42 CFR 411.15. We believe that these proposed criteria are sufficient guardrails to ensure, along with appropriate patient selection and complex medical judgement of the physician, that the procedure can be performed safely on an ambulatory basis, including procedures that involve these five currently excluded characteristics. We believe that this proposal could advance the goals of increasing physician and patient choice and expanding site neutral options in conjunction with patient safety considerations.
With respect to the existing general exclusion at § 416.166(c)(6), which excludes procedures designated as requiring inpatient care under § 419.22(n) from classification as covered surgical procedures, this proposal would modify this standard since the IPO list is proposed for elimination beginning in CY 2026 with a 3-year transition period, as described in section IX. of this final rule with comment period. While we recognize the need to revisit the criterion at § 416.166(c)(6) following the elimination of the IPO list, we believe that maintaining this criterion for CY 2026 would allow for consistency between the two lists during the 3-year phaseout period. We note that if a service comes off the IPO list at any time, then the general exclusion at § 416.166(c)(6) would cease to apply to the service.
We acknowledge that this approach is a departure from the existing criteria that we established effective beginning in 2008, and from our policy finalized in the CY 2022 OPPS/ASC rule. However, we believe that this approach would expand and build upon our 2008 policy intent. Although there are some differences when comparing our CY 2008 criteria and the proposed CY 2026 criteria, such as removing the general standards and several of the original general exclusion criteria, permitting the addition of procedures to the ASC CPL that would have been prohibited by those criteria, and the different accreditation requirements and conditions of participation requirements between HOPDs and ASCs, these concerns have largely been addressed by the progress in medical practice and ASC capabilities in the 17 years since the criteria were developed as previously noted. In particular, given advances in the practice of medicine and the evolving nature of ASCs, we believe ASCs are now better equipped to safely perform procedures that were once too complex or risky to be performed safely on Medicare beneficiaries in the ASC setting. As previously mentioned, although ASCs and hospitals have different health and safety requirements, many ASCs often undergo accreditation as a condition of State licensure and share some similar licensure and compliance requirements with hospitals. Each of these requirements provides additional safeguards for the health and safety of Medicare beneficiaries receiving surgical procedures in an ASC. Additionally, in the CY 2022 OPPS/ASC final rule with comment period, when we reinstated the ASC CPL criteria that were in effect during CY 2020, we stated that many of the surgical procedures added to the list in CY 2021 may pose a significant safety risk to a typical Medicare beneficiary when performed in an ASC (86 FR 63777). However, we believe that these procedures are safe to ( printed page 53857) perform in an ASC setting because all procedures identified are already payable in the HOPD setting and, therefore, are already safely performed on an ambulatory basis, consistent with the statutory requirement under section 1833(i)(1) of the Act. In addition, while several of the identified procedures may typically require hospital care that lasts beyond midnight, we expect that appropriately selected patient populations in the ASC setting would be healthier and less complex and would likely not require active monitoring or medical care past midnight beyond the procedure.
Comment: Several commenters, including ASCs, ambulatory surgery associations, and professional societies supported our proposal to revise the ASC CPL criteria under § 416.166 and add 547 surgery or surgery-like procedures to the ASC CPL for CY 2026. Some commenters believed that clinical and technological advances have enabled both outpatient hospital departments and ambulatory surgical centers to deliver equivalent patient outcomes and maintain similar standards of care quality, and allow for procedures to be performed in the ASC setting that previously could not. There was also support for allowing the physician's clinical judgment to make the site of service determinations for these procedures. Some commenters also stated that expanding the ASC CPL would allow for certain procedures to shift from higher-cost inpatient settings to lower-cost outpatient settings, which would also increase access and beneficiary choice. This would also lower hospital burden and preserve hospital capacity for higher acuity patients.
Response: We thank the commenters for their support.
Comment: Many commenters, including hospital systems, hospital associations, and professional societies were opposed to our proposal to expand the ASC CPL by revising the ASC CPL criteria under § 416.166, with patient safety being the primary concern. Some commenters urged CMS not to treat ASCs as the equivalent of hospital outpatient departments because they are not regulated as hospitals and do not have the necessary resources on site to provide the higher level of care necessary to perform many of the surgical procedures proposed for addition to the ASC CPL. Commenters also stated that the ASC CPL serves as an important guardrail for patient safety. A commenter stated that while recognizing a physician's clinical judgement is important, the ASC CPL and the criteria at § 416.166 act as a roadmap for the physician's decision-making. Commenters were concerned removing certain general standards and exclusion criteria could lead to procedures being inappropriately performed in the ASC setting. Commenters supported maintaining the current criteria and review process, as they stated it allowed the ASC CPL to expand while restricting procedures that cannot be safely performed in the ASC setting. One commenter gave the example that for exclusion criteria (5) “commonly require systemic thrombolytic therapy”, despite advances in care, there are still significant safety risks with performing these procedures in the ASC setting. A commenter was also concerned that CMS was not fulfilling the statutory obligation to limit procedures on the ASC CPL to those that can be safely performed in an ASC.
Commenters were also concerned about our proposal to add procedures removed from the IPO list to the ASC CPL, as we proposed to eliminate the IPO list over a 3-year period. Commenters suggested that we implement a 2-year period between removing a code from the IPO list and adding it to the ASC CPL or wait until the procedure was being performed at least 50 percent of the time in the outpatient setting. Commenters also suggested waiting until we have more data on these procedures in the outpatient setting before adding them to the ASC CPL.
Response: We thank the commenters for their input. As we previously stated in the CY 2021 OPPS/ASC final rule with comment period, while these are important considerations in determining whether a surgical procedure may be safely performed in an ASC, we are finalizing the proposed approach because ASCs are currently and increasingly able to safely provide services that would be covered under the revised criteria when considered appropriate for a patient by their physician. Since these general standard and exclusionary criteria will simply be moved to a different section of § 416.166, we believe these criteria will still act as a roadmap for physician decision-making. We also believe there are sufficient guardrails for ASCs, including State and local regulations, Conditions for Coverage (CfCs), accreditation requirements, and medical malpractice laws, to ensure that physicians are able to prioritize patient safety when determining the site of service.
We have previously recognized the importance of maintaining flexibility in our review of procedures that can be safely performed in the ASC setting. In recent years, we have been able to add surgical procedures to the ASC CPL that were once considered solely hospital inpatient procedures, including, for example, total knee arthroplasty, which is currently one of the most frequently performed procedures in ASCs, or certain coronary intervention procedures involving major blood vessels. We believe it is important that we continue to adapt the ASC CPL in light of the significant advances in medical practice, surgical techniques, and ASC capabilities that have enabled some ASCs to safely perform procedures involving major blood vessels and other general exclusion criteria that were once too complex for the ASC setting. As we have heard from several interested parties, many procedures that are currently only payable as hospital outpatient services under Medicare are safely performed in the ASC setting for other payors. We acknowledge that non-Medicare patients tend to be younger and have fewer comorbidities than the Medicare population, but believe that careful patient selection can identify Medicare beneficiaries who are suitable candidates to receive these services in the ASC setting. We have long recognized the importance of ensuring that the health care system has as many access points and patient choices for all Medicare beneficiaries as possible, and we believe it is important that we continue to support both greater flexibility for physicians and patients to choose ASCs as the site of care and patient safety in supporting those important goals.
We recognize commenters' concerns regarding our proposal to add procedures to the ASC CPL that were proposed for removal from the IPO list for CY 2026 as part of the first phase of the elimination of the IPO list. We believe that adding these procedures to the ASC CPL as the IPO list is phased-out allows for greater consistency and less confusion between the two lists. We also believe that current guardrails in place are sufficient to ensure patient safety.
Comment: Some commenters also had concerns about the financial and administrative burden this proposal would have on hospitals. Commenters were concerned that allowing certain procedures to be performed in the ASC setting could increase the burden on hospitals if they face increased transfers from ASC facilities if there are increases in complications from ASCs performing procedures that are not safe in that setting. Commenters were also concerned that this proposal would allow certain higher paying procedures to move over to the ASC setting, ( printed page 53858) negatively affecting hospitals and their ability to offset lower paying procedures. Additionally, many commenters stated that physicians may face financial and administrative pressure to prioritize performing procedures in lower-cost settings over patient safety.
Response: We disagree that this proposal would negatively impact hospitals due to higher paying procedures being shifted to the ASC setting. Given that physicians will be exercising their judgment to determine the cases and procedures that may be safely performed in the ASC setting, we anticipate that hospitals will continue to provide high paying procedures that will offset lower paying procedures. We have also heard from interested parties that certain procedures we proposed to add to the ASC CPL setting are already being performed safely in the ASC setting. Therefore, given that physicians will be using their clinical judgment to determine the appropriate site of service, we believe that any shifts to the ASC setting that may occur with the expansion of the ASC CPL will be clinically appropriate and reflect ongoing trends. We believe that this proposal will incentivize efficiency and foster access, while continuing to maintain patient safety. Additionally, we would welcome any data or analyses on the effects of potential shifts of high cost procedures to the ASC setting that interested parties would like to share.
We also emphasize that the fact that a procedure is added to the ASC CPL should not be interpreted to mean the procedure is only appropriately performed in ASC setting or that the ASC setting is the most appropriate setting for a beneficiary and instead defer to the clinical judgment of physician. We believe that current safeguards, such as facility accreditations, State licensures, CfCs, medical malpractice laws, and professional guidelines, will prevent inappropriate shifts in the site of service and allow physicians to prioritize patient safety.
Comment: Multiple commenters requested that, if we were to finalize our proposal to revise the criteria and expand the ASC CPL, that CMS provide additional monitoring and guidance. Commenters requested that CMS monitor site of service data to observe any shifts as the ASC CPL is expanded. Commenters also requested that CMS monitor for safety and quality concerns for beneficiaries, such as emergency department admissions following procedures performed in ASCs. Additionally, commenters requested that CMS provide guidance on determining the appropriate site of service selection for procedures being added to the ASC CPL, including considerations for clinical and social factors.
Response: We thank the commenters for their feedback. We agree with the value of monitoring this data, including tracking the complications or readmission rates as procedures are adding the ASC CPL. We would welcome any patient outcomes data, analyses, or recommendations that interested parties would like to share. We will take these recommendations into consideration for future rulemaking.
In regards to providing further guidance, we note the balance between several factors on this important issue, namely, the prohibition on CMS interfering with the practice of medicine in section 1801 of the Act, the need to provide clear information about CMS billing and payment rules that ensure hospitals, physicians, and other interested parties can understand and operate within them, and our belief that the specific decision about the most appropriate care setting for a given surgical procedure is a complex medical judgment made by the physician based on the beneficiary's individual clinical needs and preferences and on the general coverage rules requiring that any procedure be reasonable and necessary. Additionally, we believe that input received from interested parties suggests physicians are sufficiently capable of making the requisite site of service determinations for their patients, and as such, further guidance is not needed.
Comment: Some commenters expressed concern about the potential for increased coinsurance obligations for beneficiaries if we finalize our proposal to expand the ASC CPL. These commenters also recommended CMS ensure beneficiaries are informed in advance that, unlike under the OPPS, ASC cost-sharing is not capped at the inpatient deductible and could exceed cost sharing in the hospital outpatient setting for the same procedure.
Response: We are aware that beneficiaries may incur greater cost-sharing for procedures in an ASC setting that we proposed to add to the ASC CPL, but note that this is not an occurrence that is unique to these procedures. As we stated in the CY 2018 OPPS/ASC final rule with comment period (82 FR 59389), section 4011 of the 21st Century Cures Act (Pub. L. 114-255) amended section 1834 of the Act by adding a new subsection (t), which requires the Secretary to make available to the public via a searchable website, with respect to an appropriate number of items and services, the estimated payment amount for the item or service under the OPPS and ASC payment system and the estimated beneficiary liability applicable to the item or service. We implemented this provision by providing our Outpatient Procedure Price Lookup tool available via the internet at https://www.medicare.gov/procedure-price-lookup. This web page allows beneficiaries to compare their potential cost-sharing liability for procedures performed in the hospital outpatient setting versus the ASC setting. We believe this tool helps inform beneficiaries of potential cost-sharing amounts for receiving a service in the ASC setting compared to the outpatient setting, and note that this tool would include a comparison of cost-sharing liability for procedures in the outpatient hospital and ASC settings in the future.
(2) Final Procedure Additions for CY 2026
CMS will add surgical procedures to the ASC CPL in rulemaking as we become aware of new surgical procedures that meet the four criteria requirements at § 416.166(b)(2). A member of the public may also notify CMS of a surgical procedure they believe meets the requirements at new § 416.166(b)(2) through the pre-proposed rule recommendation process or the public comment period. CMS will confirm whether the procedure does meet those requirements and will add it to the ASC CPL if it does meet that criteria. In accordance with the new proposed regulatory text at § 416.166(d), physicians would then assess whether their specific patients can or cannot safely receive such covered surgical procedure in the ASC setting, based on patient-specific considerations.
For CY 2026, we proposed to update the ASC CPL by adding 276 potential surgery or surgery-like codes to the list that we believe would meet the proposed revised ASC CPL criteria under 42 CFR 416.166. This includes procedures submitted through our pre-proposed rule nominations process for addition to the ASC CPL under the proposed revised criteria. Additionally, we proposed to add 271 surgery or surgery-like codes to the CPL that are currently on the IPO list, if we finalize our proposal to remove these services from the IPO list for CY 2026.
Comment: We received comments in support of specific codes that we proposed to add to the ASC CPL for CY 2026, with many commenters supporting the addition of cardiac ablation codes. ( printed page 53859)
Response: We thank the commenters for their support.
Comment: Multiple commenters recommended specific codes to be added to the ASC CPL including, echocardiography, electrophysiological studies, and percutaneous coronary interventions. We received 44 procedure recommendations for the CPL, listed in Table 155, below.
Response: We thank commenters for their recommendations. We individually assessed each of the recommended procedures to determine whether these procedures meet each of the revised regulatory criteria at the § 416.166. Based on our review of the clinical characteristics of the procedures and their similarity to other procedures that are currently on the ASC CPL, we believe that 13 procedures out of the 44 procedure recommendations we received meet the general standards and exclusion criteria for the ASC CPL as set forth in the revised § 416.166(b) and (c), respectively. These procedures, listed in Table 131, are:
CPT code 92960 (Cardioversion, elective, electrical conversion of arrhythmia; external);
CPT code 92961 (Cardioversion, elective, electrical conversion of arrhythmia; internal (separate procedure));
CPT code 92924 (Percutaneous transluminal coronary atherectomy, with coronary angioplasty when performed; single major coronary artery or branch);
CPT code 92933 (Percutaneous transluminal coronary atherectomy, with intracoronary stent, with coronary angioplasty when performed; single major coronary artery or branch);
CPT code 92937 (Percutaneous transluminal revascularization of or through coronary artery bypass graft (internal mammary, free arterial, venous), any combination of intracoronary stent, atherectomy and angioplasty, including distal protection when performed; single vessel);
CPT code 92943 (Percutaneous transluminal revascularization of chronic total occlusion, coronary artery, coronary artery branch, or coronary artery bypass graft, any combination of intracoronary stent, atherectomy and angioplasty; single vessel);
CPT code 92973 (Percutaneous transluminal coronary thrombectomy mechanical (list separately in addition to code for primary procedure));
CPT code 92974 (Transcatheter placement of radiation delivery device for subsequent coronary intravascular brachytherapy (list separately in addition to code for primary procedure));
CDT code D7440 (Excision of malignant tumor-lesion diameter up to 1.25 cm);
CDT code D7441 (Excision of malignant tumor-lesion diameter greater than 1.25 cm);
CPT code 0582T (Transurethral ablation of malignant prostate tissue by high-energy water vapor thermotherapy, including intraoperative imaging and needle guidance);
CPT code 0786T (Insertion or replacement of percutaneous electrode array, sacral, with integrated neurostimulator, including imaging guidance, when performed); and
CPT code 0991T (Cystourethroscopy, with low-energy lithotripsy and acoustically actuated microspheres, including imaging).
In the upcoming section, we explain our rationale for not including the 26 recommended procedures, organized by category. We note that while we did not add these codes to the ASC CPL, we did determine some of these codes are appropriate to add to list of covered ancillary procedures, as discussed in section XIII.D.3. of this final rule with comment period.
13 cardiovascular codes, including electrophysiological studies, transesophageal echocardiography, and 3D predictive modeling for procedure planning. As these procedures are non-surgical, they would not qualify as separately payable surgical procedures on the ASC CPL. Additionally, 2 cardiovascular procedures requested are currently packaged procedures on the ASC CPL. As these procedures are add-on codes, they would not qualify for separate payment on the ASC CPL.
2 circulatory assist and one percutaneous coronary intervention procedures. As these procedures will remain on the IPO list for CY 2026, they would not qualify as payable surgical procedures on the ASC CPL.
4 percutaneous coronary intervention and 2 thrombolysis procedures. The codes associated with these procedures are scheduled to be deleted for CY 2026.
1 pharmacologic agent administration procedure. As this procedure is non-surgical, it would not qualify as a separately payable surgical procedures on the ASC CPL.
1 neurostimulator analysis procedure. As this procedure is non-surgical, it would not qualify as a separately payable surgical procedures on the ASC CPL.
5 middle ear implant procedures. These procedures are currently nonpayable under the OPPS and would not qualify as payable surgical procedures on the ASC CPL.
After consideration of the public comments, we are finalizing our proposal, with modification, to revise the ASC CPL criteria under § 416.166 by modifying the general standard criteria and eliminating five of the general exclusion criteria and to add the proposed 547 procedures to the ASC CPL for CY 2026. We are also finalizing adding an additional 13 codes recommended by commenters to the ASC CPL for CY 2026. These codes, along with their long descriptors and final payment indicator assignments, are listed in Tables 131, 132, and 133. We believe that these finalized policies will increase the flexibility for physicians to exercise their complex medical judgment, factoring in patient safety considerations, and for patients have more choice in which setting to receive surgical procedures.
( printed page 53860) ( printed page 53861) ( printed page 53862) ( printed page 53863) ( printed page 53864) ( printed page 53865) ( printed page 53866) ( printed page 53867) ( printed page 53868) ( printed page 53869) ( printed page 53870) ( printed page 53871) ( printed page 53872) ( printed page 53873) ( printed page 53874) ( printed page 53875) ( printed page 53876) ( printed page 53877) ( printed page 53878) ( printed page 53879) ( printed page 53880) ( printed page 53881) ( printed page 53882) ( printed page 53883) ( printed page 53884) ( printed page 53885) ( printed page 53886) ( printed page 53887)3. Covered Ancillary Services
Covered ancillary services are specified in § 416.164(b) and, as stated previously, are eligible for separate ASC payment. As provided at § 416.164(b), we make separate ASC payments for ancillary items and services when they are provided integral to ASC covered surgical procedures that include the following: (1) brachytherapy sources; (2) certain implantable items that have pass-through payment status under the OPPS; (3) certain items and services that we designate as contractor-priced, including, but not limited to, procurement of corneal tissue; (4) certain drugs and biologicals for which separate payment is allowed under the OPPS; (5) certain radiology services for which separate payment is allowed under the OPPS; and (6) non-opioid pain management drugs that function as a supply when used in a surgical procedure. Payment for ancillary items and services that are not paid separately under the ASC payment system is packaged into the ASC payment for the covered surgical procedure.
In the CY 2019 OPPS/ASC final rule with comment period (83 FR 59062 through 59063), consistent with the established ASC payment system policy (72 FR 42497), we finalized the policy to update the ASC list of covered ancillary services to reflect the payment status for the services under the OPPS and to continue this reconciliation of packaged status for subsequent calendar years. As discussed in prior rulemaking, maintaining consistency with the OPPS may result in changes to ASC payment indicators for some covered ancillary services. For example, if a covered ancillary service was separately paid under the ASC payment system in CY 2024, but will be packaged under the CY 2025 OPPS, we would also package the ancillary service under the ASC payment system for CY 2025 to maintain consistency with the OPPS. Comment indicator “CH” is used in Addendum BB (which is available via the internet on the CMS website) to indicate covered ancillary services for which we proposed a change in the ASC payment indicator to reflect a proposed change in the OPPS treatment of the service for CY 2025.
In the CY 2022 OPPS/ASC final rule with comment period, we finalized our proposal to revise § 416.164(b)(6) to include, as ancillary items that are integral to a covered surgical procedure and for which separate payment is allowed, non-opioid pain management drugs and biologicals that function as a supply when used in a surgical procedure as determined by CMS (86 FR 63490).
Comment: A commenter requested that CMS work with dental and ASC interested parties to expand the number of dental procedures that can be performed with HCPCS code G0330. They also requested that CMS remove the requirement that HCPCS code G0330 must be billed along with a covered but non-payable dental ancillary service.
Response: We thank the commenter for their input. While we are not adding any additional dental procedures that can be billed with G0330 to the list of covered ancillary services at this time, we will take this into consideration for future rulemaking.
Comment: Multiple commenters recommended specific codes to be added to the list of covered ancillary services for CY 2026. We received a total of 24 recommended procedures, which included esophageal motility, gastrointestinal tract imaging, and magnetic resonance safety procedures.
Response: We agreed with commenters that these recommendations could be added to the list of covered ancillary procedures for CY 2026, with the exception of two procedures. We did not find HCPCS codes G0562 (Therapeutic radiology simulation-aided field setting; complex, including acquisition of pet and ct imaging data required for radiopharmaceutical-directed radiation therapy treatment planning ( i.e., modeling)) and G0563 (Stereotactic body radiation therapy, treatment delivery, per fraction to one or more lesions, including image guidance and real-time positron emissions-based delivery adjustments to one or more lesions, entire course not to exceed 5 fraction) to be appropriate to add to the list of covered ancillary services because we did not find them to be integral to any procedures on the ASC CPL. Therefore, we are adding an additional 22 procedures recommended by commenters to the list of ASC covered ancillary services for CY 2026. In addition to these 22 procedures, we also found that 13 procedures recommended by commenters that did not qualify for addition to the ASC CPL, due to being nonsurgical in nature, could be appropriately placed on the list of ASC covered ancillary services.
In summary, we are finalizing adding 35 procedures recommended by interested parties to the list of ASC covered ancillary services for CY 2026. New CPT and HCPCS codes for covered ancillary services for CY 2026 can be found in section XIII.B. of this final rule.
E. Skin Substitute Changes to the List of ASC Covered Items and Services for CY 2026
As we discussed in section III. of the CY 2026 OPPS/ASC proposed rule, beginning January 1, 2026, we proposed to remove skin substitutes from the list of packaged items and services at § 419.2(b)(16) under the OPPS and under § 416.164(a)(5) under the ASC payment system. Our proposal intended to establish a consistent and uniform framework for how these products are treated across different outpatient settings of care to help ensure equitable access and appropriate payment for these services. While we do not believe these products are commonly used in the ASC setting, we believe extending our uniform framework from the physician office and hospital outpatient setting to the ASC setting will help ensure equitable access to these products in the future across the different sites of outpatient care.
Our payment policies under the ASC payment system for covered ancillary services generally vary according to the particular type of item or service and its payment policy under the OPPS. Drugs and biologicals that are separately paid under the ASC payment system are paid at the prospective rates adopted under the OPPS. Similar to how ASCs are paid for brachytherapy sources provided integral to ASC covered surgical procedures at prospective rates adopted under the OPPS, we proposed to pay for groups of skin substitute products at annual prospective rates adopted under the OPPS, effective January 1, 2026. Additionally, these prospective rates would not be subject to the ASC wage index adjustment and beneficiaries would be responsible for 20 percent coinsurance.
To separately pay for the provision of certain groups of skin substitute products when used during a covered surgical procedure, we proposed to revise § 416.164(b) to include groups of skin substitute products as covered ancillary items and services that are integral to a covered surgical procedure. As discussed in section XIII.B.6. of the CY 2026 OPPS/ASC proposed rule, we proposed to identify HCPCS skin substitute codes which may be separately payable with our proposed payment indicator of “S2”—Skin substitute supply group paid separately when provided integral to a surgical procedure on ASC list; payment based on OPPS rate. Therefore, for those existing skin substitute products for which we proposed to separately pay for, we revised the payment indicator from “N1”—Packaged service/item; no ( printed page 53888) separate payment made—to payment indicator “S2” effective January 1, 2026. Additionally for new skin substitute products which we proposed to add to the list of ASC covered ancillary items and services, we proposed to assign these skin substitute products an ASC payment indicator of “S2”.
We did not receive public comments on this provision, and therefore, we are finalizing as proposed.
All ASC covered ancillary services and their final payment indicators for CY 2026 are also included in Addendum BB to this final rule (which is available via the internet on the CMS website).
F. Final CY 2026 Non-Opioid Policy for Pain Relief Under the OPPS and ASC Payment System
1. Background
The Consolidated Appropriations Act (CAA), 2023 (Pub. L. 117-328), was signed into law on December 29, 2022. Section 4135(a) and (b) of the CAA, 2023, titled Access to Non-Opioid Treatments for Pain Relief, amended section 1833(t)(16) and section 1833(i) of the Act, respectively, to provide for temporary additional payments for non-opioid treatments for pain relief (as that term is defined in section 1833(t)(16)(G)(i) of the Act). In particular, section 1833(t)(16)(G) of the Act provides that with respect to a non-opioid treatment for pain relief furnished on or after January 1, 2025, and before January 1, 2028, the Secretary shall not package payment for the non-opioid treatment for pain relief into payment for a covered OPD service (or group of services) and shall make an additional payment for the non-opioid treatment for pain relief as specified in clause (ii) of that section. Clauses (ii) and (iii) of section 1833(t)(16)(G) of the Act provide for the amount of additional payment and set a limitation on that amount.
Paragraph (10) of section 1833(i) of the Act cross-references the OPPS provisions about the additional payment amount and payment limitation for non-opioid treatments for pain relief and applies them to payment under the ASC payment system. In particular, paragraph (A) of paragraph (10) of section 1833(i) of the Act, as added by section 4135(b) of the CAA, 2023, provides that in the case of surgical services furnished on or after January 1, 2025, and before January 1, 2028, additional payments shall be made under the ASC payment system for non-opioid treatments for pain relief in the same amount provided in clause (ii) and subject to the limitation in clause (iii) of section 1833(t)(16)(G) of the Act for the OPPS. Paragraph (B) of section 1833(i)(10) of the Act provides that a drug or biological that meets the requirements of 42 CFR 416.174 and is a non-opioid treatment for pain relief shall also receive additional payment in the amount provided in clause (ii) and subject to the limitation in clause (iii) of section 1833(t)(16)(G) of the Act.
Additional payments under this policy began on January 1, 2025. As stated in the CY 2025 OPPS/ASC final rule with comment period (89 FR 94343 through 94344), the statute directs CMS to provide “additional payment”, and for purposes of this policy, we interpret this language to be equivalent to “separate payment,” since CMS provides an additional payment by unpackaging the product and then making a separate payment. “Separate payment” is the more commonly used terminology in the OPPS rule and likely more familiar to readers. To avoid confusion, we will continue to use “separate payment” throughout the rest of this section, which we believe to be synonymous with “additional payment.” Under section 1833(t)(2)(E) of the Act, the temporary separate payments must be made in a budget neutral manner.
For background information on the ASC Payment Policy for Non-Opioid Post-Surgery Pain Management Drugs and Biologicals prior to CY 2025, please see the summary provided in the CY 2025 OPPS/ASC final rule with comment period (89 FR 94342 through 94343).
2. Final CY 2025 Non-Opioid Policy Implementation of Section 4135 of the CAA, 2023
In CY 2025, CMS finalized our implementation methodology for section 4135 of CAA, 2023 (89 FR 94343 through 94361) to provide for temporary separate payments for certain non-opioid treatment for pain relief in the hospital outpatient department and ambulatory surgical center settings on a temporary basis from January 1, 2025 through December 31, 2027. CMS also finalized regulation text at 42 CFR 416.174 and 42 CFR 419.43(k), which outline the payment for non-opioid pain management drugs, biologicals, and medical devices under both the ASC payment system and OPPS, respectively.
a. Drugs and Biologicals Subject to the ASC Non-Opioid Policy (42 CFR 416.174)
Section 1833(i)(10)(B), titled “Transition”, provides that a drug or biological that meets the requirements of the regulation at 42 CFR 416.174, the current ASC non-opioid policy, and also meets the definition of a non-opioid treatment for pain relief at section 1833(t)(16)(G)(iv) of the Act shall receive separate payments under section 4135 of the CAA, 2023, subject to the payment limitation. In light of this requirement, we finalized in the CY 2025 OPPS/ASC final rule with comment period that drugs and biologicals that meet the definition of a non-opioid treatment for pain relief for purposes of section 4135 of the CAA, 2023 that were subject to the ASC policy for non-opioid treatments authorized by section 6082 of the SUPPORT Act in CY 2024, would instead receive separate payments, subject to the limitation, for the duration of the payment period for section 4135 of the CAA, 2023 (89 FR 94344). The policy was finalized to be in effect for the duration of the payment period for section 4135 of the CAA, 2023.
b. Definition of Non-Opioid Treatment for Pain Relief
Section 1833(t)(16)(G)(iv) of the Act defines a non-opioid treatment for pain relief for a drug, biological product, or medical device, and requires, in part, that such a treatment not receive transitional pass-through payment and has payment packaged into a payment for a covered OPD service (or group of services). In addition, in order for a drug or biological product to qualify as a non-opioid treatment for pain relief, pursuant to section 1833(t)(16)(G)(iv)(I), the product must have “a label indication approved by the Food and Drug Administration to reduce postoperative pain, or produce postsurgical or regional analgesia, without acting upon the body's opioid receptors”. In order for a medical device to qualify as a non-opioid treatment for pain relief, pursuant to section 1833(t)(16)(G)(iv)(II) of the Act, it must, in part, be “used to deliver a therapy to reduce postoperative pain, or produce post-surgical or regional analgesia”. A medical device must also, pursuant to section 1833(t)(16)(G)(iv)(II)(aa) and (bb) of the Act have both “an application under section 515 of the Federal Food, Drug, and Cosmetic Act that has been approved with respect to the device, been cleared for market under section 510(k) of such Act, or is exempt from the requirements of section 510(k) of such Act pursuant to subsection (l) or (m) or section 510 of such Act or section 520(g) of such Act” and “demonstrated the ability to replace, reduce, or avoid intraoperative or postoperative opioid use or the quantity of opioids prescribed ( printed page 53889) in a clinical trial or through data published in a peer-reviewed journal”.
c. Evidence Requirement for Medical Devices
To determine whether a medical device fulfills the statutory requirement that it has demonstrated the ability to replace, reduce, or avoid intraoperative or postoperative opioid use or the quantity of opioids prescribed in a clinical trial or through data published in a peer-reviewed journal, we finalized in the CY 2025 OPPS/ASC final rule with comment period (89 FR 94345) a policy to review all data submitted during the public comment period to determine if the device demonstrates the ability to replace, reduce, or avoid intraoperative or postoperative opioid use or the quantity of opioids. In CY 2025, we encouraged interested parties submitting non-opioid device recommendations to submit any relevant literature that demonstrates that the named medical device replaces, reduces, or avoids opioid use per this statutory provision with their public comments. We review any literature submitted and determine whether it meets this evidence criterion. There is no requirement that commenters submit any data or literature with their device recommendations. If there is no data or literature submitted for a medical device, or if the materials submitted do not demonstrate any ability of the medical device to replace, reduce, or avoid opioids, the medical device would not meet this evidence criterion and would not qualify for separate payment under section 4135 of the CAA, 2023.
d. Non-Opioid Product Indications
(1) FDA-Approved Indications for Drugs and Biologicals
Section 1833(t)(16)(G)(iv)(I) of the Act specifies that to meet the definition of a non-opioid treatment for pain relief and to be eligible for separate payment, a drug or biological product must have a label indication approved by the Food and Drug Administration to reduce postoperative pain, or produce postsurgical or regional analgesia, without acting upon the body's opioid receptors.
Given these statutory requirements, we finalized a policy in the CY 2025 OPPS/ASC final rule with comment period (89 FR 94345 through 94346) only to approve separate payment for drug or biological products with an FDA-approved indication that closely aligns with the statutorily required indication language to reduce post-operative pain or produce post-surgical or regional analgesia. We noted that products with an indication that does not meet this statutory requirement would not qualify. We specifically stated that products with only a general pain indication will not qualify.
As discussed in the CY 2025 OPPS/ASC final rule with comment period (89 FR 94345 through 94346), we note that the Congress specifically included language at section 1833(t)(16)(G)(iv)(I) of the Act requiring that drugs or biologicals have “a label indication approved by the Food and Drug Administration to reduce postoperative pain, or produce postsurgical or regional analgesia, without acting upon the body's opioid receptors”. Therefore, products with an indication that does not meet the statutory requirement will not qualify.
(2) Intended Use for Medical Devices
Regarding medical devices, section 1833(t)(16)(G)(iv)(II) of the Act specifies that such a device must be used to deliver a therapy to reduce postoperative pain or produce post-surgical or regional analgesia to qualify for separate payment under section 4135 of the CAA, 2023. It also must have an application approved under section 515 of the Federal Food, Drug, and Cosmetic Act (the FD&C Act), have been cleared for market under section 510(k) of the FD&C Act, or be exempt from the requirements of section 510(k) of the FD&C Act pursuant to section 510(l) or (m) or 520(g) of the FD&C Act. For CY 2025, for medical devices, we finalized without modification our proposal that a device must be used to deliver a therapy to reduce postoperative pain or produce post-surgical or regional analgesia to qualify for separate payment under section 4135 of the CAA, 2023 (89 FR 94346 through 94347). We also finalized that the medical device must have an application approved under section 515 of the FD&C Act, which has been cleared for market under section 510(k) of the FD&C Act, or be exempt from the requirements of section 510(k) of the FD&C Act pursuant to section 510(l) or (m) or 520(g) of the FD&C Act. (89 FR 94346 through 94347). This is consistent with the regulation text at 42 CFR 419.43(k)(2)(i) through (iv).
e. Amount of Payment
Section 1833(t)(16)(G)(ii)(I) of the Act provides that, for a non-opioid treatment for pain relief that is a drug or biological product, the amount of separate payment is the amount of payment for such product determined under section 1847A of the Act that exceeds the portion of the otherwise applicable Medicare OPD fee schedule that the Secretary determines is associated with the drug or biological, subject to a limitation, as described in the next section. Section 1833(t)(16)(G)(ii)(II) of the Act provides that, for a non-opioid treatment for pain relief that is a medical device, the amount of separate payment is the amount of the hospital's charges for the device, adjusted to cost, that exceeds the portion of the otherwise applicable Medicare OPD fee schedule that the Secretary determines is associated with the device, subject to a limitation, as described in the next section.
In the CY 2025 OPPS/ASC final rule with comment period, we finalized a policy to assign a payment offset of zero dollars for the qualifying drugs, biologicals, and devices for CY 2025 (89 FR 94347 through 94348). A zero dollar offset means that we would not offset or remove the amount that the non-opioid product represents from the procedure payment rate when setting payment rates. We finalized a zero dollar offset for the initial year of the policy as some of these products are new products or newly separately paid in the OPPS setting and their costs may not be fully reflected yet in the cost of procedures in which they may be used. Therefore, we stated that the separate payment for a drug or biological would be determined by subtracting from the amount calculated using the methodology outlined in section 1847A of the Act the portion of the otherwise applicable Medicare OPD fee schedule associated with the drug or biological, which as previously discussed, we finalized to be zero dollars for CY 2025. For the amount of payment for a medical device, since we are unable to reduce charges to costs for ASCs, we stated that the separate payment amount would be contractor-priced by the ASC's Medicare Administrative Contractor reduced by the portion of the otherwise applicable Medicare OPD fee schedule amount associated with the medical device, which as previously discussed, we finalized to be zero dollars for CY 2025. These separate payment amounts are all subject to the payment limitation, described in the subsequent section.
Section 1833(i)(10) of the Act establishes the same separate payment for the ASC setting as for hospital outpatient departments, as described in section 1833(t)(16)(G)(ii) of the Act. Both separate payments are subject to the limitation in section 1833(t)(16)(G)(iii) of the Act, which specifies that the separate payment amount shall not exceed the estimated average of 18 percent of the OPD fee schedule amount for the OPD service (or ( printed page 53890) group of services) with which the non-opioid treatment for pain relief is furnished. Given this statutory requirement, starting on January 1, 2025 through December 31, 2027, we finalized paying the same separate payment amount for qualifying non-opioid products in both the HOPD and ASC settings.
As the statute requires separate payment for these non-opioid treatments for pain relief, these products cannot be packaged into the procedure payment. Under our current threshold packaging policy, if the estimated per day cost for a drug or biological is less than or equal to the applicable OPPS drug packaging threshold, we package payment for the drug or biological into the payment for the associated procedure. Similarly, under our comprehensive APC (C-APC) policy, we package all payments for services integral, ancillary, supportive, dependent, and adjunctive to the primary service into a single payment for the primary comprehensive service. For CY 2025, we finalized that non-opioid treatments for pain relief would not be subject to the threshold packaging policy and would also be separately paid when used during a comprehensive APC (C-APC) procedure in the HOPD setting (89 FR 94347 through 94348). See section V.B.1.a. of this final rule with comment period for more information regarding the drug packaging threshold. Section II.A.2.b. of this final rule with comment period contains further information on C-APC packaging.
f. Payment Limitation
Section 1833(t)(16)(G)(iii) of the Act states that the separate payment amount specified in clause (ii), (which is described in the previous section) shall not exceed the estimated average of 18 percent of the OPD fee schedule amount for the OPD service (or group of services) with which the non-opioid treatment for pain relief is furnished, as determined by the Secretary.
In the CY 2025 OPPS/ASC final rule with comment period, we finalized a policy to base the 18 percent payment limitation on the volume weighted average of the payment rates of the top five primary procedures by volume into which a non-opioid treatment for pain relief would have their payment packaged, absent this policy. We also finalized applying the 18 percent payment limitation per date of service billed (89 FR 94349).
g. Payment Limitation With No Claims Data
For drugs, biologicals, and devices with no claims data, such as for newly FDA-approved and marketed products or products that did not previously have their own product-specific HCPCS code by which to track payment and utilization data, we finalized in the CY 2025 OPPS/ASC final rule with comment period (89 FR 94350) a policy where CMS will utilize the services with which a product would be expected to be furnished and would typically be packaged absent this policy, to calculate the payment limitation based on expected clinical use patterns. The finalized policy stated that CMS will determine the service, or group of services, to use to calculate the payment limitation through engagement with interested parties and a review by CMS Medical Officers and clinical staff during annual rulemaking. In the absence of engagement from interested parties, we will determine clinically appropriate procedures with which we would expect the drug or device to be frequently used in order to determine the payment limitation, including review of FDA approval materials, procedures identified in literature available to CMS, and other relevant materials. We noted that we may update the payment limitation amount in future rulemaking as we gather additional claims data on the utilization of and payment for this product.
3. Final CY 2026 Non-Opioid Policy Implementation of Section 4135 of the CAA, 2023
For CY 2026, we proposed to continue the policies finalized in the CY 2025 OPPS ASC final rule without modification (90 FR 33745).
We stated in the CY 2026 OPPS/ASC proposed rule that we continue to believe a zero-dollar offset is appropriate for all qualifying products regulated under the non-opioid policy as some of these products are new products or newly separately paid in the OPPS setting and their costs may not be fully reflected yet in the cost of procedures in which they may be used. Additionally, the data used for CY 2026 ratesetting is derived from CY 2024 claims, which was prior to the effective date of this policy in CY 2025. Accordingly, we proposed to edit the regulation text at 42 CFR 416.174(c)(1) to remove the following text: “which is determined to be zero dollars for calendar year 2025”. We proposed to remove this language pertaining to the portion of the otherwise applicable Medicare OPD fee schedule amount for CY 2025, as we will discuss the appropriate amount in each year's rulemaking.
We noted that the final payment limitation calculation in the CY 2026 OPPS/ASC final rule with comment period would be based on the proposed procedure payment rates and utilization data available in the CY 2026 OPPS/ASC proposed rule. Therefore, the values included in Table 133, previously published as Table 83 in the CY 2026 OPPS/ASC proposed rule (90 FR 33748 and 33749), are approximate payment limitations based on the best data available at the time of writing the CY 2026 OPPS/ASC proposed rule. We note that the final payment limitations for the CY 2026 OPPS/ASC final rule with comment period are also based on the proposed payment rates in the CY 2026 OPPS/ASC proposed rule.
Table 131, previously published as Table 82 in the CY 2026 OPPS/ASC proposed rule (90 FR 33746 and 33747), includes citations to the indications of the drugs and biologicals proposed to have met the statutory requirements and qualify for separate payment for the CY 2026 OPPS/ASC proposed rule. We welcomed public comment on all of these policies, including the procedures used to determine the payment limitations that are detailed in Table 133.
We welcomed comments regarding additional drugs or devices that readers believe meet the criteria at 42 CFR 416.174 and 42 CFR 419.43(k) and should qualify as non-opioid treatments for pain relief. In the CY 2026 OPPS/ASC proposed rule, we stated we would review those comments, evaluate the products against the criteria, and, if appropriate, would finalize additional drugs and devices that meet these criteria as non-opioid treatments for pain relief in the CY 2026 OPPS/ASC final rule with comment period to begin payment in CY 2026.
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: Commenters were generally supportive of CMS paying for drugs paid under the ASC non-opioid treatment policy authorized by section 6082 of the SUPPORT Act under the policy authorized by section 4135 of the CAA, 2023 for the second year, including the C-APC exclusion of qualifying products. Commenters recommended that CMS begin to assess its authority for continuing a policy for the payment of non-opioid treatments for pain relief in future rulemaking, starting in CY 2028 and beyond, including making the current policy ( printed page 53891) permanent. Several commenters provided suggestions on how CMS could craft a policy to pay for non-opioid treatments for pain relief starting in CY 2028 and beyond.
Response: We thank commenters for their support. Comments about possible future funding, payments, and policies are outside the scope of this CY 2026 rule.
Comment: A commenter requested that CMS approve products for this policy off-cycle in order to provide more immediate access to new non-opioid products. This commenter suggested that adding new products upon approval will guarantee that patients have access to new non-opioid options as soon as possible. Many commenters stated that there needs to be access to all qualifying non-opioid treatments for pain relief, and noted the general importance of the availability of non-opioid alternatives.
Response: We thank the commenters for their insights regarding the importance of access to non-opioid treatments for pain relief, as well as the commenter's suggestion for us to approve products off-cycle to provide more immediate access. As we have noted in past rules, it is our priority to address the opioid epidemic and we are committed to evaluating and adjusting our current processes, if necessary, in order to ensure appropriate care or access for beneficiaries amidst the current opioid epidemic. We would expect the majority of non-opioid products that meet the statutory criteria to have already been addressed under this policy; however, we acknowledge there could be a small number of products that could newly meet the established criteria throughout the calendar year. Therefore, given our priorities in addressing the opioid epidemic, and the potential development of new non-opioid products, we believe it may be reasonable to consider a pathway to approve new products or products newly meeting the statutory requirements on a quarterly basis. We welcome engagement from the public on products that newly meet the qualifying criteria and should be paid according to the statute under this policy. Such engagement could include: (1) documentation verifying appropriate FDA status, including the required FDA-approved indication for drugs; (2) qualifying peer-reviewed literature for medical devices and that the medical device is used to deliver a therapy to reduce postoperative pain, or produce postsurgical or regional analgesia; (3) suggestions for the top 5 most frequent procedures for purposes of calculating a payment limitation; (4) verification that the product does not have transitional pass-through status; (5) confirmation that the product has payment that is packaged into a payment for a covered OPD service, and (6) the current HCPCS code describing the product or recommendations for a new HCPCS code to describe the product. Please see 42 CFR 419.43(k) and 416.174 for the full established criteria. As we are considering evaluating and updating the CY 2026 qualifying non-opioid treatments for pain relief, we are modifying the regulation text accordingly. Specifically, we are removing the phrase “through that year's rulemaking” and “for an applicable calendar year” from 42 CFR 419.43(k)(1), 419.43(k)(2), 416.174(a), and 416.174(b).
We anticipate that if any non-opioid treatments for pain relief qualify during CY 2026, our review process would be similar to the other existing quarterly approval processes, such as OPPS New Technology APC process or the CMS HCPCS application process, where CMS evaluates and approves products on a more frequent basis than annual rulemaking. A quarterly implementation process will be necessary to create new HCPCS codes and implement the necessary claims processing changes to effectuate the calculated payment rates as well as the required payment limitations. We will make payment, according to the statute, retroactively effective to the date that we determine that a product meets the criteria, with the earliest effective payment date being January 1, 2026. Once these determinations are made by us, we will make them publicly available on the CMS OPPS website.[213] Payment will be made according to the statute, and established regulation text, and will be effectuated in the applicable quarterly OPPS/ASC update.
Comment: We received many comments recommending CMS increase access to non-opioid products including commenters emphasizing the benefits of a broader interpretation of the statutory language requiring drugs and biologicals to have an FDA-approved indication to reduce postoperative pain, or produce postsurgical or regional analgesia, without acting upon the body's opioid receptors. Several commenters requested that we allow drugs and biologicals, including those that are orally administered, that have FDA-approved indications for “acute pain” or other pain indications to qualify under this policy, rather than only those with the FDA-approved label indications to “reduce postoperative pain” or to “produce postsurgical or regional analgesia” that is prescribed by the Congress. Some commenters argued that products may receive a broader label that indicates their ability to treat moderate or severe acute pain with the intention of being used in every setting in which pain presents, including postoperatively. Commenters asserted that requiring non-opioid drugs to have a specific indication to reduce postoperative pain, or produce post-surgical or regional analgesia limits patient access to FDA-approved non-opioid treatments with broader indications, including in rural settings.
Several commenters stated their belief that FDA guidance provides that general pain indications can encompass situations in which analgesia or pain management is provided post operatively, and that general indications are often approved based on clinical trials in patients with postoperative pain. One commenter stated that they had discussed with the FDA and were not able to revise their FDA-approved indication. They believed that FDA did not consider it appropriate to add an express reference to postoperative or postsurgical use, due to the fact that adding additional specific language to the FDA-approved indication could be viewed as narrowing the approved indication. Commenters also argued that CMS does not require that the FDA-approved label state that the product must not act upon the body's opioid receptors and, therefore, should not require the indication to have an express reference to postoperative or postsurgical use.
Commenters also noted their belief that the FDA has issued guidance, which CMS understands to be labeled as draft guidance by the FDA, on the approval and labeling of general acute pain therapies, which states that such an indication is appropriate for a product that is supported by at least two successful clinical trials, including trials in postoperative pain. Commenters believed this FDA guidance made it clear that there was a need for a more expansive interpretation of the NO PAIN Act to ensure that products with broader labeling are taken fully into consideration by CMS for purposes to satisfying the criteria set forth by the NO PAIN Act. Commenters encouraged CMS consider FDA-approved labeling that includes products that have been studied in two successful clinical trials in postoperative pain.
Overall, commenters did not think it was appropriate for CMS to require the ( printed page 53892) indications specified in section 1833(t)(16)(G)(iv) of the Act strictly, and they believed that a broader label indication would still satisfy the requirements. Several commenters believed that drugs with more general pain indications, or indications that did not align with those as listed in the NO PAIN Act, satisfied the criteria of the Act and of the criteria CMS finalized. The drugs mentioned by commenters included Sprix, Caldolor, Journavx, gabapentinoids, Prialt, Celecoxib, XIFYRM, and acetaminophen.
Response: We thank the commenters for their input. As we discussed in section XIII.F.2. of the CY 2025 OPPS/ASC final rule with comment period (89 FR 94344 through 94347), the postoperative pain or postsurgical requirement, referred to in the public comments, is in section 1833(t)(16)(G)(iv) of the Act. The Congress specifically included language stating that, to meet the definition of a “non-opioid treatment for pain relief,” drugs or biologicals must have “a label indication approved by the Food and Drug Administration to reduce postoperative pain, or produce postsurgical or regional analgesia, without acting upon the body's opioid receptors”. We codified this provision at § 419.43(k)(1)(i).
As we stated in the CY 2025 OPPS/ASC final rule with comment period (89 FR 94246), we “only approve separate payment for drug or biological products with an FDA-approved indication to reduce post-operative pain or produce post-surgical or regional analgesia. Products such as those with only a general pain indication will not qualify”. As we explained, if there is no mention of postoperative or post-surgical use in the FDA-approved indications for the products being evaluated, they do not meet the statutory requirement. For CY 2026, we continue to believe this approach conforms with the statute. While some commenters have asserted that a broad pain indication could include a more specific one, we do not agree that we can infer from a broad acute, or general, pain indication that the FDA also intended that the drug is indicated to reduce post-operative pain or produce post-surgical or regional analgesia, as that specific pain indication is not provided. We defer to the FDA to approve indications for drugs and biological products, based on the relevant statutory and regulatory authorities, and it is not within our purview to infer alternative or more specific pain indications that have not been specifically finalized in the FDA's review. Therefore, while we understand the commenters' concerns regarding revising the payment indication, determining whether the specific pain indications is met under a broader indication is not within our purview.
Because the statute is clear that an indication to reduce post-operative pain or product post-surgical or regional analgesia is required for separate Medicare payment under section 4135 of the CAA, 2023, we do not believe that we can look outside of the specific indication provided or to other sources that are not the FDA-approved Drug Label. Therefore, given the current labeling for the following products, we are not finalizing separate payment for the following products under the OPPS and ASC payment systems in this rule: Sprix, Caldolor, Journavx, gabapentinoids, Prialt, Celecoxib, XIFYRM, and acetaminophen. We note that these products may still be able to be paid under the OPPS/ASC payment system, for example, through packaged payments, but they do not currently qualify for additional separate payment under this provision.
After consideration of public comments, we are finalizing our proposal to continue the policies finalized in the CY 2025 OPPS ASC final rule with comment period with a minor modification to permit more timely consideration of payment requests.
Comment: Commenters supported our proposal to continue to set a zero-dollar offset for CY 2026. Additionally, we received a comment requesting that we keep the zero-dollar offset for qualifying products for CY 2026 and CY 2027. The commenter stated that by extending the policy, CMS removes potential financial barriers that could discourage the use of safer, non-opioid pain control options, which are particularly important in health outcomes for women.
Response: We thank commenters for their support. We will evaluate the need to continue to assign a zero-dollar offset in future rulemaking.
Comment: A commenter provided CMS suggestions and recommendations on how to best complete the Report to Congress assessing the impact of the Act, mandated by subsection C of section 4135 of the CAA, 2023.
Response: We appreciate this engagement on this component of the statute and look forward to continued collaboration with interested parties regarding this issue. We will take this information into consideration for future rulemaking.
a. Qualifying Products for CY 2026
The following table, Table 134, previously published as Table 82 in the CY 2026 OPPS/ASC proposed rule (90 FR 33746 and 33747), lists the non-opioid alternatives that we proposed would receive separate payment as a non-opioid pain management drug or device under section 4135 criteria for CY 2026.
CMS routinely receives public comments with detailed rationales on why commenters believe a particular drug, biological, medical device, or other item or service should receive separate payment. As such, we solicited comment in the CY 2026 OPPS/ASC proposed rule on whether there are any additional drugs, biologicals, or medical devices that meet the statutory requirements outlined in sections 1833(t)(16)(G) and 1833(i)(10) of the Act. In addition to soliciting comment on the actual product and how it meets the criteria at 42 CFR 416.174 and 42 CFR 419.43(k), we solicited comment on the top five procedures used to calculate the payment limitation, as well as HCPCS coding for the product, which CMS could use to establish the payment rate, if CMS determines that the product discussed in the comment qualifies as a non-opioid treatment for pain relief, listed in Table 135.
As discussed previously in this section, there are specific requirements that must be met in order for the product to qualify for separate payment. In the CY 2026 OPPS/ASC proposed rule, we stated that interested parties that believed that a product not addressed in the proposed rule met the statutory requirements were encouraged to submit information during the comment period indicating how the product meets the statutory eligibility requirements. In the proposed rule, we stated that if CMS determines that the product(s) does in fact meet the statutory eligibility requirements, we would finalize separate payment for the product(s) in the CY 2026 OPPS/ASC final rule with comment period.
We stated that for drugs and biological products not addressed in the CY OPPS/ASC proposed rule, if no comment was submitted that outlines how that drug or biological meets the statutory criteria, then CMS would not finalize separate payment for such product for CY 2026. Additionally, for medical devices not addressed in the CY 2026 OPPS/ASC proposed rule, unless a comment was submitted that both outlines how that device meets the statutory criteria, including literature that demonstrates that the device has the ability to replace, reduce, or avoid intraoperative or postoperative opioid use or the quantity of opioids prescribed in a clinical trial or through data published in a peer-reviewed journal, ( printed page 53893) CMS would not finalize separate payment for such device for CY 2026.
We proposed that the HCPCS codes describing the qualifying devices and drugs in Table 134 would be placed on the ASC covered ancillary procedures list. We noted that Medicare Administrative Contractors (MACs) determine whether a drug, device, procedure, or other service meets all program requirements and conditions for coverage and payment. HOPDs and ASCs only receive payment for qualifying drugs, biologicals, and medical devices when the appropriate MAC determines that the service meets the relevant conditions for coverage and payment. As we have consistently stated in past OPPS/ASC final rules with comment period (see, for example, 87 FR 71879 and 88 FR 81660 through 81661), the fact that a drug, device, procedure or service is assigned a HCPCS code and a payment rate under the OPPS does not imply coverage by the Medicare program, but indicates only how the product, procedure, or service may be paid if covered by the program (see, for example, Pub 100-04 Medicare Claims Processing, Transmittal 11937).
( printed page 53894) ( printed page 53895) ( printed page 53896)We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: Commenters were generally supportive of our proposals on the qualifying products in Table 82 of the CY 2026 OPPS/ASC proposed rule, republished as Table 134 in this final rule with comment period, and they recommended that we finalize those products for separate payment for CY 2026. These commenters were also generally supportive of most aspects of the implementation of this policy, including the payment amounts for these products and the payment limitations.
Response: We thank the commenters for their support, and we are finalizing those products for separate payment, with updates to their product specific payment limitations as listed in Table 137.
Comment: A commenter requested clarification regarding the established descriptor for HCPCS code C9806 (Rotary peristaltic infusion pump ( e.g., ambIT Pump), including catheter and all disposable system components, non-opioid medical device (must be a qualifying Medicare non-opioid medical device for post-surgical pain relief in accordance with Section 4135 of the CAA, 2023) and whether it could be used to describe both reusable and disposable versions of the ambIT pump. This commenter stated that the literature provided concludes lowered opioid usage with both versions of the pump, disposable and reusable.
Response: We thank the commenter for their question and input. We believe that it is appropriate to create a new C-code to describe the reusable version of the ambIT pump. Specifically, we are creating C9816 (Rotary peristaltic infusion pump ( e.g., reusable ambIT Pump) including all disposable system components, reusable non-opioid medical device (must be a qualifying Medicare non-opioid medical device for post-surgical pain relief in accordance with section 4135 of the CAA, 2023)) to describe the reusable version of the pump. Please see Table 137 for the payment limitation calculation and the new C-code used to describe the device.
Comment: A commenter supported the continuation of the products described by HCPCS Code C9808 (Nerve cryoablation probe ( e.g., cryoice, ( printed page 53897) cryosphere, cryosphere max, cryoice cryosphere, cryoice cryo2), including probe and all disposable system components, non-opioid medical device (must be a qualifying medicare non-opioid medical device for post-surgical pain relief in accordance with section 4135 of the CAA, 2023)) as qualifying non-opioid treatments for pain relief. This commenter requested that CMS add the CryoXT cryoablation probe to that HCPCS code descriptor. This commenter stated that this product was not widely available on the market at the time that the CY 2025 OPPS/ASC final rule with comment period was published, but they believe that this device should also meet the qualifying criteria. They state this device has the same mechanism of action as the cryoNB probes that were studied in the clinical literature, previously submitted by this same commenter, and since this new device is substantially equivalent, and is a predicate device, to devices that were studied in the literature, the commenter believed that the CryoXT should also qualify for separate payment.
Response: Based on the comment received, we find that the CryoXT medical device is used to deliver a therapy, cryoablation, as stated in the device's 510k summary, to reduce postoperative pain or produce post-surgical or regional analgesia. Accordingly, we confirmed that this device has FDA clearance.[214] However, the supporting literature does not specifically name the CryoXT medical device or demonstrate that this device, specifically, has the ability to replace, reduce, or avoid intraoperative or postoperative opioid use or the quantity of opioids prescribed in a clinical trial or through data published in a peer-reviewed journal.[215] [216] [217] [218] [219] [220] [221] We believe that the device demonstrating the ability to reduce, replace, or avoid opioid use in a clinical trial or peer-reviewed journal must be the same device as the one being studied in order to qualify for separate payment under section 4135. Therefore, we are not finalizing a change to the descriptor of HCPCS code C9808 to include CryoXT as requested by this commenter for CY 2026.
SapphireTM Multi-Therapy Infusion System
Comment: Commenters requested that we evaluate and approve the SapphireTM infusion system manufactured by Eitan Medical, under our non-opioid treatment for pain relief separate payment policy. Per the manufacturer, among other uses, the SapphireTM infusion system is used to deliver therapies to reduce post-surgical pain and/or product post-surgical or regional analgesia in the outpatient setting. The manufacturer provided that the SapphireTM infusion system has been cleared by the FDA under 510(k) as a Class II device (K192860) [222] and has supporting literature. The manufacturer explained that a range of surgeries requiring postoperative analgesia including shoulder, knee and ankle arthroplasties, thoracic surgery, gynecological surgeries, and spine surgery have been supported with anesthesia and postoperative pain control with the SapphireTM infusion system. The manufacturer provided four HCPCS procedure codes with which they believed the SapphireTM infusion system could be used for post-surgical pain management.
Response: Based on comments received, we find that this medical device is used to deliver a therapy to reduce postoperative pain or produce post-surgical or regional analgesia. We confirmed that this product is FDA cleared [223] and has supporting literature demonstrating the ability to replace, reduce, or avoid intraoperative or postoperative opioid use or the quantity of opioids prescribed in a clinical trial or through data published in a peer reviewed journal.[224] The medical device does not currently receive transitional passthrough status and we believe it would have payment that is otherwise packaged into a payment for a covered OPD service, absent this policy. Therefore, we believe that the SapphireTM medical device meets the statutory requirements at section 1833(t)(16)(G)(iv) of the Act and we are finalizing that it will be paid separately under this provision. We appreciate the commenter's suggestions regarding the top five procedures on which to base the payment limitation. Based on our review of the device, as well as the volume of services in which the device is most likely to be used, we believe that the top five procedures to base the payment limitation on are: HCPCS codes 27447 (Arthroplasty, knee, condyle and plateau; medial AND lateral compartments with or without patella resurfacing (total knee arthroplasty)), 27130 (Arthroplasty, acetabular and proximal femoral prosthetic replacement (total hip arthroplasty), with or without autograft or allograft), 23472 (Arthroplasty, glenohumeral joint; total shoulder (glenoid and proximal humeral replacement (e.g., total shoulder))), 29827 (Arthroscopy, shoulder, surgical; with rotator cuff repair), and 29881 (Arthroscopy, knee, surgical; with meniscectomy (medial OR lateral, including any meniscal shaving) including debridement/shaving of articular cartilage (chondroplasty), same or separate compartment(s), when performed). We will assume an equal utilization of the device with these five procedures for the payment limitation calculation, as shown in Table 137 at the end of this section. We note that in the absence of utilization data for the top five procedures, we believe it is reasonable to assume equal utilization for purposes of calculating the payment limitation until claims data are available. Please see Table 137 for the payment limitation calculation and the new C-code used to describe the device.
( printed page 53898)AvosetTM Infusion System
Comment: Commenters requested that we evaluate and approve the AvosetTM infusion system manufactured by Eitan Medical, under our non-opioid treatment for pain relief separate payment policy. They noted that the AvosetTM infusion system is a compact ambulatory infusion pump, similar to Eitan Medical's SapphireTM infusion system. Like the SapphireTM infusion system, the AvosetTM infusion system is used to deliver therapies to reduce post-surgical pain and/or product post-surgical or regional analgesia in the outpatient setting. The AvosetTM infusion system has been cleared by the FDA under 510(k) as a Class II device. The manufacturer provided four HCPCS procedure codes with which they believed the AvosetTM infusion system could be used for post-surgical pain management.
The manufacturer pointed to the clinical evidence submitted for the SapphireTM infusion system as evidence that the AvosetTM infusion system has the ability to replace, reduce, or avoid intraoperative or postoperative opioid use in a clinical trial or through data published in a peer-reviewed journal. Per the commenter, the evidence for the SapphireTM device also provides evidence for the AvosetTM device since SapphireTM was one of the predicate devices deemed substantially equivalent to the AvosetTM device by the FDA. The manufacturer noted their plans to submit data on the AvosetTM device to the November 2025 American Society of Regional Anesthesia 24th Annual Pain Medicine Meeting. The manufacturer also submitted articles providing supportive evidence for electronic infusion pumps, generally, which do not name either the SapphireTM or AvosetTM infusion systems. The commenter urged us to interpret the statutory language in section 4135 of the CAA, 2023 to allow for evidence of the effectiveness of therapies using infusion devices to reduce or eliminate opioid use for postoperative pain, even if the device named in the trial or data is not the same infusion device for which separate payment is requested. The commenter believes that an expanded interpretation of the literature requirement will allow for greater access to non-opioid postoperative pain therapies and lower costs to the Medicare program.
Response: Based on the comments received, we find that the Avoset medical device is used to deliver a therapy to reduce postoperative pain or produce post-surgical or regional analgesia. We confirmed that the device has FDA clearance.[225] However, the supporting literature does not demonstrate that the AvosetTM device specifically has the ability to replace, reduce, or avoid intraoperative or postoperative opioid use or the quantity of opioids prescribed in a clinical trial or through data published in a peer reviewed journal. While we acknowledge that SapphireTM may be the predicate device for the AvosetTM infusion system, we believe that the device demonstrating the ability to reduce, replace, or avoid opioid use in a clinical trial or peer-reviewed journal must be the same device as the one being studied in order to qualify for separate payment under section 4135 of the CAA, 2023. Therefore, we believe that the AvosetTM medical device does not meet the statutory requirements at section 1833(t)(16)(G)(iv)(II) of the Act and are not finalizing separate payment for it under this provision. We understand that peer reviewed literature may be forthcoming for this device, but it was not available at the time of writing of this final rule with comment period. We can take new clinical evidence regarding the named device into consideration for future rulemaking.
Ultrasound-Visible Nerve Block Needles (SonoPlex, SonoBlock, SonoTap)
Comment: A commenter recommended CMS evaluate and approve ultrasound-visible nerve block needles, specifically the SonoPlex, SonoBlock, and SonoTap medical devices manufactured by PAJUNK, under our non-opioid treatment for pain relief separate payment policy. The commenter stated that the nerve block needles are cleared by the FDA as 510(k) devices to inject local anesthetic to achieve regional analgesia and have supporting literature.
The commenter provided five HCPCS codes with which they requested CMS establish the payment limitation. These CPT codes are: 23472 (Arthroplasty, glenohumeral joint; total shoulder (glenoid and proximal humeral replacement (e.g., total shoulder))), 27447 (Arthroplasty, knee, condyle and plateau; medial and lateral compartments with or without patella resurfacing (total knee arthroplasty)), 27130 (Arthroplasty, acetabular and proximal femoral prosthetic replacement (total hip arthroplasty), with or without autograft or allograft), 27792 (Open treatment of distal fibular fracture (lateral malleolus), includes internal fixation, when performed), and 19301 (Mastectomy, partial (e.g., lumpectomy, tylectomy, quadrantectomy, segmentectomy)).
Response: Based on comments received, we find that ultrasound-visible nerve block needles are used to deliver a therapy to reduce postoperative pain or produce post-surgical or regional analgesia. We confirmed that the three named ultrasound-visible nerve block needles, SonoPlex, SonoBlock, and SonoTap, are FDA cleared [226 227 228] and have supporting literature demonstrating the ability to replace, reduce, or avoid intraoperative or postoperative opioid use or the quantity of opioids prescribed in a clinical trial or through data published in a peer reviewed journal.[229 230 231] The medical device does not currently receive transitional passthrough status and we believe it would have payment that is otherwise packaged into a payment for a covered OPD service, absent this policy. Therefore, we believe that the three named ultrasound-visible nerve block needles, specifically the SonoPlex, SonoBlock, and SonoTap medical devices, meet the statutory requirements at section 1833(t)(16)(G)(iv) of the Act and are finalizing that they will be paid separately under this provision. We appreciate the commenter's suggestions regarding the top five procedures on which to base the payment limitation. Based on our review of the devices, as well as the volume of services in which the devices are most likely to be used, we believe that the top five procedures to base the payment limitation on are: HCPCS codes 27447 (Arthroplasty, knee, condyle and plateau; medial AND lateral compartments with or without patella resurfacing (total knee arthroplasty)), 27130 (Arthroplasty, acetabular and proximal femoral prosthetic replacement (total hip arthroplasty), with or without autograft ( printed page 53899) or allograft), 23472 (Arthroplasty, glenohumeral joint; total shoulder (glenoid and proximal humeral replacement (e.g., total shoulder))), 29827 (Arthroscopy, shoulder, surgical; with rotator cuff repair), and 29881 (Arthroscopy, knee, surgical; with meniscectomy (medial OR lateral, including any meniscal shaving) including debridement/shaving of articular cartilage (chondroplasty), same or separate compartment(s), when performed). We will assume an equal utilization of the devices with these 5 procedures for the payment limitation calculation, as shown in Table 137 at the end of this section. We note that in the absence of utilization data for the top 5 procedures, we believe it is reasonable to assume equal utilization for purposes of calculating the payment limitation until claims data are available. Please see Table 137 for the payment limitation calculation and the new C-code used to describe the ultrasound-visible nerve block needle devices.
Perforated Continuous Infusion Catheter Set (InfiltraLong)
Comment: A commenter recommended CMS evaluate and approve a perforated continuous infusion catheter set, specifically the InfiltraLong medical device manufactured by PAJUNK, under our non-opioid treatment for pain relief separate payment policy. The commenter stated that the InfiltraLong device is a self-contained system intended for continuous or intermittent pre-operative, peri-operative, or post-operative delivery of a local anesthetic at the surgical site. Per the commenter, the device's distribution of local anesthetic optimizes patient analgesia and improves outcomes while reducing opioid consumption. The commenter stated that the InfiltraLong device is cleared by the FDA and has supporting literature.
The commenter provided five HCPCS codes with which they requested CMS establish the payment limitation. These CPT codes are: 23472 (Arthroplasty, glenohumeral joint; total shoulder (glenoid and proximal humeral replacement (e.g., total shoulder))), 27447 (Arthroplasty, knee, condyle and plateau; medial and lateral compartments with or without patella resurfacing (total knee arthroplasty)), 49650 (Laparoscopy, surgical; repair initial inguinal hernia), 15847 (Excision, excessive skin and subcutaneous tissue (includes lipectomy), abdomen (e.g., abdominoplasty) (includes umbilical transposition and fascial plication) (list separately in addition to code for primary procedure)), and 21601 (Excision of chest wall tumor including rib(s)).
Response: Based on comments received, we find that this medical device is used to deliver a therapy to reduce postoperative pain or produce post-surgical or regional analgesia. We confirmed that this product is FDA cleared [232] and has supporting literature demonstrating the ability to replace, reduce, or avoid intraoperative or postoperative opioid use or the quantity of opioids prescribed in a clinical trial or through data published in a peer reviewed journal.[233] The medical device does not currently receive transitional passthrough status and we believe it would have payment that is otherwise packaged into a payment for a covered OPD service, absent this policy. Therefore, we believe that the InfiltraLong medical device meets the statutory requirements at section 1833(t)(16)(G)(iv) of the Act and are finalizing that it will be paid separately under this provision. We appreciate the commenter's suggestions regarding the top five procedures on which to base the payment limitation. Based on our review of the device, as well as the volume of services in which the device is most likely to be used, we believe that the top five procedures to base the payment limitation on are: HCPCS codes 27447 (Arthroplasty, knee, condyle and plateau; medial AND lateral compartments with or without patella resurfacing (total knee arthroplasty), 27130 (Arthroplasty, acetabular and proximal femoral prosthetic replacement (total hip arthroplasty), with or without autograft or allograft), 23472 (Arthroplasty, glenohumeral joint; total shoulder (glenoid and proximal humeral replacement (e.g., total shoulder))), 29827 (Arthroscopy, shoulder, surgical; with rotator cuff repair), and 29881 (Arthroscopy, knee, surgical; with meniscectomy (medial OR lateral, including any meniscal shaving) including debridement/shaving of articular cartilage (chondroplasty), same or separate compartment(s), when performed). We will assume an equal utilization of the device with these five procedures for the payment limitation calculation, as shown in Table 137 at the end of this section. We note that in the absence of utilization data for the top five procedures, we believe it is reasonable to assume equal utilization for purposes of calculating the payment limitation until claims data are available. Please see Table 137 for the payment limitation calculation and the new C-code used to describe the device.
Atraumatic Spinal Anesthesia Needle (Sprotte)
Comment: A commenter recommended CMS evaluate and approve an atraumatic spinal anesthesia needle, specifically the Sprotte medical device manufactured by PAJUNK, under our non-opioid treatment for pain relief separate payment policy. The commenter stated that the Sprotte device is used to administer anesthetic agents to the subarachnoid space in spinal anesthesia procedures. Per the commenter, the Sprotte device enables physicians to more effectively use spinal anesthesia, which allows for longer, controlled pain relief compared to post-surgical IV opioids. The commenter stated that the Sprotte device is cleared by the FDA[ 234 235] and has supporting literature.
The commenter provided five HCPCS codes with which they requested CMS establish the payment limitation. These CPT codes are: 27447 (Arthroplasty, knee, condyle and plateau; medial and lateral compartments with or without patella resurfacing (total knee arthroplasty)), 27130 (Arthroplasty, acetabular and proximal femoral prosthetic replacement (total hip arthroplasty), with or without autograft or allograft), 52601 (Transurethral electrosurgical resection of prostate, including control of postoperative bleeding, complete (vasectomy, meatotomy, cystourethroscopy, urethral calibration and/or dilation, and internal urethrotomy are included)), 52235 (Cystourethroscopy, with fulguration (including cryosurgery or laser surgery) and/or resection of; medium bladder tumor(s) (2.0 to 5.0 cm)), and 52000 (Cystourethroscopy (separate procedure)).
Response: Based on the comments received, we find that this medical device is used to deliver a therapy to reduce postoperative pain or produce post-surgical or regional anesthesia. We confirmed that this device has 510(k) approval by the FDA.[236] However, the ( printed page 53900) supporting literature does not demonstrate that the Sprotte device specifically has the ability to replace, reduce, or avoid intraoperative or postoperative opioid use or the quantity of opioids prescribed in a clinical trial or through data published in a peer-reviewed journal. In one of the articles submitted, it is not possible to verify whether the patients in the intervention group were using the Sprotte device or an alternative needle, called the Quincke. Therefore, we cannot attribute the study results to either device. The other article that was submitted only compared the efficacy between the Sprotte and Quincke needles, and therefore, we similarly could not find clinical evidence to demonstrate that the Sprotte device specifically has the ability to replace, reduce, or avoid intraoperative or postoperative opioid use or the quantity of opioids prescribed.
Therefore, we believe that the atraumatic spinal anesthesia needle, specifically the Sprotte medical device, does not meet the statutory requirements at section 1833(t)(16)(G)(iv)(II) of the Act and are not finalizing separate payment for it under this provision.
Continuous Anesthesia Conduction Catheter Sets (SonoLong, E-Cath)
Comment: A commenter recommended CMS evaluate and approve continuous anesthesia conduction catheter sets, specifically the SonoLong and E-Cath medical devices manufactured by PAJUNK, under our non-opioid treatment for pain relief separate payment policy. Per the commenter, the SonoLong and E-Cath devices are similar to the SonoPlex, SonoBlock, and SonoTAP nerve block needles, which improve needle and catheter placement, except that SonoLong and E-Cath stay in the nerve block site to provide pain relief days after surgery. The commenter provided that the catheter sets are cleared by the FDA. Per the commenter, the intended use of the SonoLong device is the delivery of continuous conduction anesthesia and/or analgesia of peripheral nerves for up to 72 hours, while the intended use of the E-Cath device is the delivery of medication for regional anesthesia and pain management. The commenter submitted literature to advance their claim that both devices have the ability to replace, reduce, or avoid intraoperative or postoperative opioid use or the quantity of opioids prescribed in a clinical trial or through data published in a peer-reviewed journal.
The commenter provided 5 HCPCS codes with which they requested CMS establish the payment limitation. These CPT codes are: 23472 (Arthroplasty, glenohumeral joint; total shoulder (glenoid and proximal humeral replacement ( e.g., total shoulder))), 27447 (Arthroplasty, knee, condyle and plateau; medial and lateral compartments with or without patella resurfacing (total knee arthroplasty)), 27130 (Arthroplasty, acetabular and proximal femoral prosthetic replacement (total hip arthroplasty), with or without autograft or allograft), 27792 (Open treatment of distal fibular fracture (lateral malleolus), includes internal fixation, when performed), and 21811 (Open treatment of rib fracture(s) with internal fixation, includes thoracoscopic visualization when performed, unilateral; 1-3 ribs).
Response: Based on comments received, we find that both of these medical devices are used to deliver a therapy to reduce postoperative pain or produce post-surgical or regional analgesia. We confirmed that the products are FDA cleared.[237 238] We also confirmed that the SonoLong device has supporting literature demonstrating the ability to replace, reduce, or avoid intraoperative or postoperative opioid use or the quantity of opioids prescribed in a clinical trial or through data published in a peer reviewed journal.[239] However, we were not able to confirm that the E-Cath device had supporting literature. While one of the two articles submitted did not study the E-Cath device specifically,[240] the other article submitted by the commenter did not differentiate between the two studied devices.[241] We cannot attribute any result to the E-Cath device because it was either not studied at all in one study, or unclear whether it was utilized over another device in the other study. Therefore, we believe that the E-Cath medical device does not meet the statutory requirements at section 1833(t)(16)(G)(iv)(II) of the Act and are not finalizing separate payment for it under this provision.
The rest of this discussion is only applicable to the SonoLong device. The SonoLong device does not currently receive transitional passthrough status and we believe it would have payment that is otherwise packaged into a payment for a covered OPD service, absent this policy. Therefore, we believe that the SonoLong medical device meets the statutory requirements at section 1833(t)(16)(G)(iv) of the Act and are finalizing that it will be paid separately under this provision. We appreciate the commenter's suggestions regarding the top five procedures on which to base the payment limitation. Based on our review of the device, as well as the volume of services in which the device is most likely to be used, we believe that the top five procedures to base the payment limitation on are: HCPCS codes 27447 (Arthroplasty, knee, condyle and plateau; medial AND lateral compartments with or without patella resurfacing (total knee arthroplasty), 27130 (Arthroplasty, acetabular and proximal femoral prosthetic replacement (total hip arthroplasty), with or without autograft or allograft), 23472 (Arthroplasty, glenohumeral joint; total shoulder (glenoid and proximal humeral replacement ( e.g., total shoulder))), 29827 (Arthroscopy, shoulder, surgical; with rotator cuff repair), and 29881 (Arthroscopy, knee, surgical; with meniscectomy (medial OR lateral, including any meniscal shaving) including debridement/shaving of articular cartilage (chondroplasty), same or separate compartment(s), when performed). We will assume an equal utilization of the device with these five procedures for the payment limitation calculation, as shown in Table 137 at the end of this section. We note that in the absence of utilization data for the top five procedures, we believe it is reasonable to assume equal utilization for purposes of calculating the payment limitation until claims data are available. Please see Table 137 for the payment limitation calculation and the new C-code used to describe the device.
CADD-Solis Ambulatory Infusion Pump
Comment: Commenters recommended CMS evaluate and approve the CADD-Solis infusion pump manufactured by ICU Medical, under our non-opioid treatment for pain relief separate payment policy. Per the commenters, the CADD-Solis infusion pump is a medical device designed to deliver medication to reduce postoperative pain or produce postsurgical or regional analgesia. One commenter provided that the CADD-Solis infusion pump has FDA clearance and is indicated for intravenous, intra-arterial, subcutaneous, intraperitoneal, in close ( printed page 53901) proximity to nerves, into an intraoperative site (soft tissue, body cavity/surgical wound site), epidural space or subarachnoid space. The pump is intended for therapies that require a continuous rate of infusion, and/or an intermittent bolus, and/or with patient-controlled demand doses. The manufacturer submitted literature to demonstrate that the CADD-Solis infusion pump has the ability to replace, reduce, or avoid intraoperative or postoperative opioid use or the quantity of opioids prescribed in a clinical trial or through data published in a peer reviewed journal.
The commenter provided five HCPCS codes with which they requested CMS establish the payment limitation. These CPT codes are: 27447 (Arthroplasty, knee, condyle and plateau; medial and lateral compartments with or without patella resurfacing (total knee arthroplasty)), 23472 (Arthroplasty, glenohumeral joint; total shoulder (glenoid and proximal humeral replacement ( e.g., total shoulder))), 29827 (Arthroscopy, shoulder, surgical; with rotator cuff repair), 49505 (Repair initial inguinal hernia, age 5 years or older; reducible), and 27130 (Arthroplasty, acetabular and proximal femoral prosthetic replacement (total hip arthroplasty), with or without autograft or allograft).
Response: Based on comments received, we find that this medical device is used to deliver a therapy to reduce postoperative pain or produce post-surgical or regional analgesia. We confirmed that this product is FDA cleared [242] and has supporting literature demonstrating the ability to replace, reduce, or avoid intraoperative or postoperative opioid use or the quantity of opioids prescribed in a clinical trial or through data published in a peer reviewed journal.[243] The medical device does not currently receive transitional passthrough status and we believe it would have payment that is otherwise packaged into a payment for a covered OPD service, absent this policy. Therefore, we believe that the CADD-Solis infusion pump meets the statutory requirements at section 1833(t)(16)(G)(iv) of the Act and are finalizing that it will be paid separately under this provision. We appreciate the commenter's suggestions regarding the top five procedures on which to base the payment limitation. Based on our review of the device, as well as the volume of services in which the device is most likely to be used, we believe that the top five procedures to base the payment limitation on are: HCPCS codes 27447 (Arthroplasty, knee, condyle and plateau; medial AND lateral compartments with or without patella resurfacing (total knee arthroplasty), 27130 (Arthroplasty, acetabular and proximal femoral prosthetic replacement (total hip arthroplasty), with or without autograft or allograft), 23472 (Arthroplasty, glenohumeral joint; total shoulder (glenoid and proximal humeral replacement ( e.g., total shoulder))), 29827 (Arthroscopy, shoulder, surgical; with rotator cuff repair), and 29881 (Arthroscopy, knee, surgical; with meniscectomy (medial OR lateral, including any meniscal shaving) including debridement/shaving of articular cartilage (chondroplasty), same or separate compartment(s), when performed). We will assume an equal utilization of the device with these 5 procedures for the payment limitation calculation, as shown in Table 137 at the end of this section. We note that in the absence of utilization data for the top five procedures, we believe it is reasonable to assume equal utilization for purposes of calculating the payment limitation until claims data are available. Please see Table 137 for the payment limitation calculation and the new C-code used to describe the device.
Altius System
Comment: We received a comment recommending CMS evaluate and approve the Altius System® manufactured by Neuros, under our non-opioid treatment for pain relief separate payment policy. Per the commenter, the Altius System® is designed for the treatment of chronic post-amputation pain. The commenter stated that the Altius System® has FDA approval [244] through the premarket process and is intended as an aid in the management of chronic intractable phantom and residual lower limb post-amputation pain in adult amputees. Per the commenter, FDA approval for the Altius System® was supported by the QUEST study, which was a prospective, multicenter, double-blind, randomized, active-sham-controlled clinical trial designed to evaluate the safety and efficacy of the Altius System® for the treatment of severe chronic post-amputation pain relief in lower limb adult amputees in the U.S. The commenter explained that the QUEST study demonstrated that the use of a high frequency nerve block system provides significant improvement in pain and functional outcomes in patients with amputations experiencing chronic pain.[245]
Response: Based on the comment received, we do not believe that this medical device is used to deliver a therapy to reduce postoperative pain or produce post-surgical or regional analgesia because the supporting information provided demonstrates that this therapy is used to treat chronic rather than post-operative or post-surgical or regional analgesia. For example, in the QUEST study, the key inclusion criteria specified individuals with chronic post-amputation pain, defined as “6-months with exacerbations lasting 60-minutes with a frequency of 4 episodes per week with >5 Numerical Rating Scale (NRS).[246] Additionally, as the commenter noted, the device's FDA-approved indication specifies that the device is intended as an aid in the management of chronic intractable phantom and residual lower limb post-amputation pain. Therefore, we believe that Altius System® does not meet the statutory requirements at section 1833(t)(16)(G)(iv)(II) of the Act and are not finalizing separate payment for it under this provision.
Reactiv8 Implantable Neurostimulation System
Comment: We received one comment recommending CMS evaluate and approve ReActiv8 Implantable Neurostimulation System, manufactured by Mainstay Medical, under our non-opioid treatment for pain relief separate payment policy. Per the commenter, ReActiv8 addresses the root cause of pain for patients experiencing multifidus muscle dysfunction through efferent stimulation of the medial branch nerve of the dorsal ramus, triggering the multifidus muscle to contract, which over time, improves the multifidus' ability to stabilize the spine. The commenter stated that the ReActiv8 ( printed page 53902) has FDA approval [247] through the premarket process and is indicated for bilateral stimulation of the L2 medial branch of the dorsal ramus as it crosses the transverse process at L3 as an aid in the management of intractable chronic low back pain associated with multifidus muscle dysfunction, as evidenced by imaging or physiological testing in adults who have failed therapy including pain medications and physical therapy and are not candidates for spine surgery. The commenter submitted clinical literature to demonstrate that ReActive8 therapy drives a reduction in opioid use.
Response: Based on the comment received, we do not believe that this medical device is used to deliver a therapy to reduce postoperative pain or produce post-surgical or regional analgesia because the supporting information provided demonstrates that this therapy is used to treat chronic low back pain associated with multifidus muscle dysfunction, rather than post-operative or post-surgical or regional analgesia. For example, in a study submitted by the commenter, the study participants were adults “with a diagnosis of moderate-to-severe, disabling, refractory, predominantly mechanical chronic low back pain . . . with pain on at least half of the days in the year before baseline”.[248] Further, the study participants had persistent chronic low back pain, lasting “a minimum of 90 days of conservative medical management that included at least medication and physical therapy, and they were not considered candidates for spine surgery”. Therefore, we believe that ReActiv8 does not meet the statutory requirements at section 1833(t)(16)(G)(iv)(II) of the Act and are not finalizing separate payment for it under this provision.
Game Ready System
Comment: We received a comment recommending CMS evaluate and approve the Game Ready System manufactured by Avanos Medical, Inc., under our non-opioid treatment for pain relief separate payment policy. Per the commenter, the Game Ready System is an electronically controlled, integrated cryotherapy and intermittent pneumatic compression system designed for clinical use. The manufacturer provided that the Game Ready System has FDA clearance and is indicated for to treat post-surgical and acute injuries to reduce edema, swelling, and pain where cold and compression are indicated. The manufacturer submitted literature to demonstrate that the Game Ready System has the ability to replace, reduce, or avoid intraoperative or postoperative opioid use or the quantity of opioids prescribed in a clinical trial or through data published in a peer reviewed journal.
The commenter provided five HCPCS codes with which they requested CMS establish the payment limitation. These CPT codes are: 27447 (Arthroplasty, knee, condyle and plateau; medial and lateral compartments with or without patella resurfacing (total knee arthroplasty)), 27130 (Arthroplasty, acetabular and proximal femoral prosthetic replacement (total hip arthroplasty), with or without autograft or allograft), 23472 (Arthroplasty, glenohumeral joint; total shoulder (glenoid and proximal humeral replacement ( e.g., total shoulder))), 29888 (Arthroscopically aided anterior cruciate ligament repair/augmentation or reconstruction), and 29827 (Arthroscopy, shoulder, surgical; with rotator cuff repair).
Response: Based on the comment received, we find that this medical device is used to deliver a therapy to reduce postoperative pain or produce post-surgical or regional analgesia. We confirmed that this product is FDA cleared [249] and has supporting literature demonstrating the ability to replace, reduce, or avoid intraoperative or postoperative opioid use or the quantity of opioids prescribed in a clinical trial or through data published in a peer reviewed journal.[250] The medical device does not currently receive transitional passthrough status and we believe it would have payment that is otherwise packaged into a payment for a covered OPD service, absent this policy. Therefore, we believe that the Game Ready System meets the statutory requirements at section 1833(t)(16)(G)(iv) of the Act and are finalizing that it will be paid separately under this provision.
We appreciate the commenter's suggestions regarding the top five procedures on which to base the payment limitation. Based on our review of the device, as well as the volume of services in which the device is most likely to be used, we believe that the top five procedures to base the payment limitation on are: HCPCS codes 27447 (Arthroplasty, knee, condyle and plateau; medial AND lateral compartments with or without patella resurfacing (total knee arthroplasty), 27130 (Arthroplasty, acetabular and proximal femoral prosthetic replacement (total hip arthroplasty), with or without autograft or allograft), 23472 (Arthroplasty, glenohumeral joint; total shoulder (glenoid and proximal humeral replacement ( e.g., total shoulder))), 29827 (Arthroscopy, shoulder, surgical; with rotator cuff repair), and 29881 (Arthroscopy, knee, surgical; with meniscectomy (medial OR lateral, including any meniscal shaving) including debridement/shaving of articular cartilage (chondroplasty), same or separate compartment(s), when performed). We will assume an equal utilization of the device with these 5 procedures for the payment limitation calculation, as shown in Table 137 at the end of this section. We note that in the absence of utilization data for the top five procedures, we believe it is reasonable to assume equal utilization for purposes of calculating the payment limitation until claims data are available. Please see Table 137 for the payment limitation calculation and the new C-code used to describe the device.
Additional Non-Opioid Device Comments
Comment: A few commenters requested payments related to physician office payments and long-term care payments to ensure the continued advancement of these safer and more effective techniques related to non-opioid treatments for pain relief.
Response: We appreciate the feedback from commenters, but note that these payment systems are outside the scope of the statutory provision at section 1833(t)(16)(G)(iv) of the Act and this final rule with comment period.
Comment: A commenter suggested that CMS revise the code descriptors of qualifying non-opioid treatment for pain relief products to ensure that it was clear that only the qualifying products were to be appropriately described by these HCPCS codes. This commenter stated, that because “ e.g.” is used to identify an example of an inclusive universe, rather than narrowing or defining the general category of device to that specifically named device, providers suggest that other devices ( printed page 53903) beyond the brand name product may qualify—despite the additional parenthetical stating the device must be a qualifying device under section 4135 of the CAA, 2023.
Response: We thank the commenter for their suggestion. We believe the language of the long descriptors makes it clear that the device being billed by the HCPCS code must be a device reviewed and approved by CMS through notice and comment rulemaking, particularly the language CMS includes within all long code descriptors stating that the specific product “. . . must be a qualifying Medicare non-opioid medical device for post-surgical pain relief in accordance with section 4135 of the CAA, 2023” clarifies which devices that should be billing that particular HCPCS code. The qualifying Medicare non-opioid medical devices for post surgical pain relief with separate payment starting or continuing on January 1, 2026, are those listed in table 136 of this final rule with comment period. However, we appreciate the commenter's suggestions and we may consider modifications to the descriptors in the future.
Comment: Commenters also recommended other products to CMS but did not provide any supporting documentation or information on the products. Some commenters suggested services or general practices that should be supported as the commenters believed they provided appropriate pain management and could decrease opioid usage. Other commenters requested that we consider alternative non-opioid treatments, such as acupuncture, occupational therapy, physical therapy, and cognitive behavioral therapy for separate payment under section 4135 of the CAA, 2024.
Response: For those products suggested by commenters who did not provide supplemental information, we are unable to fully evaluate the product against the statutory criteria for inclusion in this policy and therefore are not finalizing separate payment for any of these products. This policy covers drugs, biologicals, and medical devices; therefore, many services suggested by commenters are out of scope for purposes of this final rule with comment period. However, in general, if an item or service discussed by commenters does not qualify for separate payment under this provision, that does not mean that there are not other appropriate methods of coverage and payment available under the Medicare Act.
Comment: A commenter mentioned our proposed regulation text edits and noted that the changes discussed in the preamble did not align with what was included in the regulation text at the end of the proposed rule and suggested that there may have been an inadvertent drafting error.
Response: We thank the commenter for their suggested edit. As discussed in the CY 2026 OPPS/ASC proposed rule, we proposed to edit the regulation text at 42 CFR 416.174(c)(1) to remove the following text: “which is determined to be zero dollars for calendar year 2025” (90 FR 33745). This information is correct; however, the commenter is also correct that there was an inadvertent drafting error in the proposed rule where this information was not carried over correctly to the proposed rule regulation text (90 FR 33862). We thank the commenter for identifying this drafting error, and we are updating the regulation text to be consistent with our proposal.
After consideration of public comments, for CY 2026, we are finalizing the proposed lists of qualifying products and payment limitations, with modifications. See Table 136 for a list of the final qualifying products for CY 2026, and please see Table 135 for a list of the final payment limitations for qualifying products for CY 2026. This final list of payment limitations contains updated claims data as available for this final rule with comment period, and is using the CY 2026 OPPS/ASC proposed rule payment rates.
Additionally, we are finalizing our proposed edit to the regulation text at 42 CFR 416.174(c)(1) to remove the following text: “which is determined to be zero dollars for calendar year 2025.” We are removing this language pertaining to the portion of the otherwise applicable Medicare OPD fee schedule amount for CY 2025, as we will discuss the appropriate amount in each year's rulemaking. Additionally, as we are considering evaluating and updating the CY 2026 qualifying non-opioid treatments for pain relief, we are modifying the regulation text accordingly. Specifically, we are removing the phrase “through that year's rulemaking” and “for an applicable calendar year” from 42 CFR 419.43(k)(1), 419.43(k)(2), 416.174(a), and 416.174(b).
We note that we are placing the HCPCS codes describing the qualifying devices and drugs in Table 136 on the ASC covered ancillary procedures list. We note that Medicare Administrative Contractors (MACs) determine whether a drug, device, procedure, or other service meets all program requirements and conditions for coverage and payment. HOPDs and ASCs only receive payment for qualifying drugs, biologicals, and medical devices when the appropriate MAC determines that the service meets the relevant conditions for coverage and payment.
As discussed in greater detail earlier in this section, we believe it may be reasonable to consider a pathway to approve new products or products newly meeting the statutory requirements on a quarterly basis. We welcome engagement from the public, including on topics relating to products that newly meet the qualifying criteria and should be paid according to the statute under this policy. Such engagement could include: (1) documentation verifying appropriate FDA status, including the required FDA-approved indication for drugs; (2) qualifying peer-reviewed literature for medical devices and that the medical device is used to deliver a therapy to reduce postoperative pain, or produce postsurgical or regional analgesia; (3) suggestions for the top five most frequent procedures for purposes of calculating a payment limitation; (4) verification that the product does not have transitional pass-through status; (5) confirmation that the product has payment that is packaged into a payment for a covered OPD service, and (6) the current HCPCS code describing the product or recommendations for a new HCPCS code to describe the product. Please see 42 CFR 419.43(k) and 416.174 for the full established criteria.
We anticipate that if any non-opioid treatments for pain relief qualify during CY 2026, any potential CMS review process would be similar to the other existing quarterly approval processes, such as OPPS New Technology APC process or the CMS HCPCS application process, where CMS evaluates and approves products on a more frequent basis than annual rulemaking. A quarterly implementation process will be necessary to create new HCPCS codes and implement the necessary claims processing changes to effectuate the calculated payment rates as well as the required payment limitations. We will make payment, according to the statute, retroactively effective to the date that we determine that a product meets the criteria, with the earliest effective payment date being January 1, 2026. Once these determinations are made by CMS, we will make them publicly available on the CMS OPPS website.[251] Payment will be made according to the statute, and established regulation text, ( printed page 53904) and will be effectuated in the applicable quarterly OPPS/ASC update.
( printed page 53905) ( printed page 53906) ( printed page 53907) ( printed page 53908) ( printed page 53909)G. New Technology Intraocular Lenses (NTIOLs)
New Technology Intraocular Lenses (NTIOLs) are intraocular lenses that replace a patient's natural lens that has been removed in cataract surgery and that also meet the requirements listed in § 416.195.
1. NTIOL Application Cycle
Our process for reviewing applications to establish new classes of NTIOLs is as follows:
- Applicants submit their NTIOL requests for review to CMS by the annual deadline which is announced in the annual OPPS/ASC final rule with comment period. For a request to be considered complete, we require submission of the information requested in the guidance document titled “Application Process and Information Requirements for Requests for a New Class of NTIOLs or Inclusion of an IOL in an Existing NTIOL Class” posted on the CMS website athttps://www.cms.gov/medicare/payment/prospective-payment-systems/ambulatory-surgical-center-asc/new-technology-intraocular-lenses-ntiols.
- We announce annually, in the CY OPPS/ASC proposed rule updating the ASC and OPPS payment rates for the following calendar year, a list of all requests to establish new NTIOL classes accepted for review during the calendar year in which the proposal is published. In accordance with section 141(b)(3) of Pub. L. 103-432 and our regulations at § 416.185(b), the deadline for receipt of public comments is 30 days following publication of the list of requests to establish a new NTIOL class as published in the proposed rule.
- In the final rule with comment period updating the ASC and OPPS payment rates for the following calendar year, we—
++ Provide a list of determinations made as a result of our review of all new NTIOL class requests and public comments.
++ When a new NTIOL class is created, identify the predominant characteristic of NTIOLs in that class that sets them apart from other IOLs (including those previously approved as members of other expired or active NTIOL classes) and that is associated with an improved clinical outcome.
++ Set the date of implementation of a payment adjustment in the case of approval of an IOL as a member of a new NTIOL class prospectively as of 30 days after publication of the ASC payment update final rule, consistent with the statutory requirement.
++ Announce the deadline for submitting requests for review of an application for a new NTIOL class for the following calendar year.
2. Requests To Establish New NTIOL Classes for CY 2026
We did not receive any requests for review to establish a new NTIOL class for CY 2026 by March 1, 2025, the due date published in the CY 2025 OPPS/ASC final rule with comment period (89 FR 94361).
3. Payment Adjustment
The current payment adjustment for a 5-year period from the implementation date of a new NTIOL class is $50 per lens. Since implementation of the process for adjustment of payment amounts for NTIOLs in 1999, we have not revised the payment adjustment amount, and we did not propose to revise the payment adjustment amount for CY 2026.
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: Multiple commenters requested that CMS increase the NTIOL ( printed page 53910) add-on payment from the current $50 per lens (established in 1999) to between $95.69-$100 for CY 2026, stating that the payment has not been adjusted for over 2 decades despite significant inflation and increased research and development costs associated with bringing new IOL technologies to market. The commenters noted that in real dollar terms, the flat rate payment for NTIOLs has significantly lagged the overall economic inflation rate, with consumer inflation increasing by 138.6 percent since 2010 and manufacturing costs rising due to labor inflation and increased material costs. The commenters also requested that CMS update the NTIOL payment annually going forward.
Response: We responded to this comment in the CY 2024 OPPS/ASC final rule with comment period (88 FR 81955 through 81956). We refer readers to that final rule with comment period for our response on an increase to the NTIOL $50 per lens payment adjustment.
4. Announcement of CY 2027 Deadline for Submitting Requests for CMS Review of Applications for a New Class of NTIOLs
In accordance with § 416.185(a) of our regulations, CMS announces that in order to be considered for payment effective beginning in CY 2027, request for review of applications for a new class of new technology IOLs must be received by 5 p.m. Eastern Standard Time, on March 1, 2026. Send requests via email to outpatientpps@cms.hhs.gov or by mail to ASC/NTIOL, Division of Outpatient Care, Mailstop C4-05-07, Centers for Medicare and Medicaid Services, 7500 Security Boulevard, Baltimore, MD 21244-1850. To be considered, requests for NTIOL reviews must include information requested on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/ambulatory-surgical-center-asc/newtechnology-intraocular-lenses-ntiols.
H. Calculation of the ASC Payment Rates and the ASC Conversion Factor
1. Background
In the August 2, 2007 ASC final rule with comment period (72 FR 42493), we established our policy to base ASC relative payment weights and payment rates under the revised ASC payment system on APC groups and the OPPS relative payment weights. Consistent with that policy and the requirement at section 1833(i)(2)(D)(ii) of the Act that the revised payment system be implemented so that it would be budget neutral, the initial ASC conversion factor (CY 2008) was calculated so that estimated total Medicare payments under the revised ASC payment system in the first year would be budget neutral to estimated total Medicare payments under the prior (CY 2007) ASC payment system (the ASC conversion factor is multiplied by the relative payment weights calculated for many ASC services in order to establish payment rates). That is, application of the ASC conversion factor was designed to result in aggregate Medicare expenditures under the revised ASC payment system in CY 2008 being equal to aggregate Medicare expenditures that would have occurred in CY 2008 in the absence of the revised system, taking into consideration the cap on ASC payments in CY 2007, as required under section 1833(i)(2)(E) of the Act (72 FR 42522). We adopted a policy to make the system budget neutral in subsequent calendar years (72 FR 42532 through 42533; § 416.171(e)).
In the CY 2008 OPPS/ASC final rule with comment period (72 FR 66857 through 66858), we set out a step-by-step illustration of the final budget neutrality adjustment calculation based on the methodology finalized in the August 2, 2007 ASC final rule (72 FR 42521 through 42531) and as applied to updated data available for the CY 2008 OPPS/ASC final rule with comment period. The application of that methodology to the data available for the CY 2008 OPPS/ASC final rule with comment period resulted in a budget neutrality adjustment of 0.65.
For CY 2008, we adopted the OPPS relative payment weights as the ASC relative payment weights for most services and, consistent with the final policy, we calculated the CY 2008 ASC payment rates by multiplying the ASC relative payment weights by the final CY 2008 ASC conversion factor of $41.401. For covered office-based surgical procedures, covered ancillary radiology services (excluding covered ancillary radiology services involving certain nuclear medicine procedures or involving the use of contrast agents, as discussed in section XIII.D.2. of the CY 2023 OPPS/ASC proposed rule (87 FR 44715 through 44716)), and certain diagnostic tests within the medicine range that are covered ancillary services, the established policy is to set the payment rate at the lower of the PFS unadjusted nonfacility PE RVU-based amount or the amount calculated using the ASC standard ratesetting methodology. Further, as discussed in the CY 2008 OPPS/ASC final rule with comment period (72 FR 66841 through 66843), we also adopted alternative ratesetting methodologies for specific types of services (for example, device-intensive procedures).
As discussed in the August 2, 2007 ASC final rule with comment period (72 FR 42517 through 42518) and as codified at § 416.172(c) of the regulations, the revised ASC payment system accounts for geographic wage variation when calculating individual ASC payments by applying the pre-floor and pre-reclassified IPPS hospital wage indexes to the labor-related share, which is 50 percent of the ASC payment amount based on a GAO report of ASC costs using 2004 survey data. Beginning in CY 2008, CMS accounted for geographic wage variation in labor costs when calculating individual ASC payments by applying the pre-floor and pre-reclassified hospital wage index values that CMS calculates for payment under the IPPS, using updated Core Based Statistical Areas (CBSAs) issued by OMB in June 2003.
The reclassification provision in section 1886(d)(10) of the Act is specific to acute care hospitals. We believe that using the most recently available pre-floor and pre-reclassified IPPS hospital wage indexes result in the most appropriate adjustment to the labor portion of ASC costs. We continue to believe that the pre-floor, pre-reclassified hospital wage indexes, which are updated yearly and are used by several other Medicare payment systems, appropriately account for geographic variation in labor costs for ASCs (89 FR 23424). Therefore, the wage index for an ASC is the pre-floor and pre-reclassified hospital wage index for the fiscal year under the IPPS of the CBSA that maps to the CBSA where the ASC is located.
On July 21, 2023, OMB issued OMB Bulletin No. 23-01, which provided the delineations of all Metropolitan Statistical Areas, Metropolitan Divisions, Micropolitan Statistical Areas, Combined Statistical Areas, and New England City and Town Areas in the U.S. and Puerto Rico based on the standards published on July 16, 2021, in the Federal Register (86 FR 37770) and 2020 Census Bureau data. (A copy of this bulletin may be obtained at https://www.whitehouse.gov/wp-content/uploads/2023/07/OMB-Bulletin-23-01.pdf.) As discussed in the FY 2025 IPPS/LTCH PPS final rule with comment period (89 FR 69253 through 69266), we finalized our proposal to use the new CBSAs delineations issued by OMB in OMB Bulletin 23-01 for the IPPS hospital wage index beginning in CY 2025. Therefore, because the ASC wage indexes for the calendar year are ( printed page 53911) the pre-floor and pre-reclassified IPPS hospital wage indexes for the fiscal year, in the CY 2025 OPPS/ASC final rule with comment period (89 FR 94362 through 94363) we finalized our proposal to incorporate the new OMB delineations into CY 2025 ASC wage indexes. We believe that using the revised delineations based on OMB Bulletin No. 23-01 will increase the integrity of the ASC wage index system by creating a more accurate representation of current geographic variations in wage levels. In addition to adopting the revised delineations based on OMB Bulletin No. 23-01, we also finalized our proposal to limit year-to-year ASC wage index value changes to no more than a 5-percent decrease, similar to the policy of other Medicare payment systems under Parts A and B. This 5-percent cap, implemented in a budget neutral manner through the wage index scalar, mitigates any large negative impacts of adopting the new delineations and prevents large year-to-year declines in wage index values as a means to reduce volatility in Medicare payments.
The proposed CY 2026 ASC wage indexes reflect the OMB labor market area delineations (including the revisions to the OMB labor market delineations discussed previously, as set forth in OMB Bulletin No. 23-01). We note that, in certain instances, there might be urban or rural areas for which there is no IPPS hospital that has wage index data that could be used to set the wage index for that area. When all of the areas contiguous to the urban CBSA of interest are rural and there is no IPPS hospital that has wage index data that could be used to set the wage index for that area, our policy has been to determine the ASC wage index by calculating the average of all wage indexes for urban areas in the State (75 FR 72058 through 72059). For example, for CY 2026, we propose to apply a proxy wage index based on this methodology to ASCs located in CBSA 35 (Rural North Dakota). In other situations, where there are no IPPS hospitals located in a relevant labor market area, we apply our current policy of calculating an urban or rural area's wage index by calculating the average of the wage indexes for CBSAs (or metropolitan divisions where applicable) that are contiguous to the area with no wage index. For example, for CY 2026, we proposed that we continue to apply a proxy wage index based on this methodology to ASCs located in CBSA 25980 (Hinesville, GA). Further, the proposed CY 2026 ASC wage index includes our policy finalized in the CY 2025 OPPS/ASC final rule with comment period that limits wage index changes to decrease by no more than 5 percent from the final CY 2025 ASC wage index value. As we discussed in the April 2025 Update to the Ambulatory Surgical Center Payment System (Change Request 14017), to limit wage index changes by no more than 5 percent from the final CY 2025 ASC wage index, some counties may require a transition CBSA before being fully reflected in the OMB labor market delineations as set forth in OMB Bulletin No. 23-01.
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: A few commenters recommended that we apply OPPS geographic reclassifications and wage index floor policies to ASCs to further harmonize payment systems and mitigate the impact of inadequate wage indices on rural ASCs, noting that hospitals can seek geographic reclassifications and receive wage index floors of 1.0 in frontier States while ASCs are excluded from these protections, despite competing in the same labor markets. The commenters highlighted specific inequities such as in South Dakota, where hospitals receive a wage index floor of 1.0 while ASCs face a much lower rural wage index of 0.8203, creating serious implications for ASCs' ability to recruit and retain staff in areas already facing workforce shortages. Both organizations emphasized that applying these wage index policies to ASCs would advance CMS's goal of aligning outpatient payment systems and ensure that beneficiaries in rural and frontier areas retain access to safe, high-quality surgical care.
Response: We appreciate the concerns the commenter has raised. The reclassification provision in section 1886(d)(10) of the Act is specific to acute care hospitals. We believe that using the most recently available pre-floor and pre-reclassified IPPS hospital wage indexes result in the most appropriate adjustment to the labor portion of ASC costs. However, we are sensitive to payment disparities the commenter raises between the OPPS and ASC payment system and will continue to monitor this issue and consider the commenter's recommendation in future rulemaking.
After consideration of public comments we received, we are finalizing our proposal to use the most recently available pre-floor and pre-reclassified IPPS hospital wage indexes to the labor-related share to create the final CY 2026 ASC wage indexes and to limit wage index decreases by no more than 5 percent from the final CY 2025 ASC wage index for a particular CBSA. The final CY 2026 ASC wage indexes reflect the OMB labor market area delineations (including the revisions to the OMB labor market delineations discussed previously, as set forth in OMB Bulletin No. 23-01) unless the county is otherwise in a transition CBSA. Additionally, based on the absence of IPPS hospitals, we are also finalizing our proposal to apply a proxy wage index based to ASCs located in CBSA 25980 (Hinesville, GA) and in CBSA 35 (Rural North Dakota).
2. Calculation of the ASC Payment Rates
a. Updating the ASC Relative Payment Weights for CY 2026 and Future Years
We update the ASC relative payment weights each year using the national OPPS relative payment weights (and PFS nonfacility PE RVU-based amounts, as applicable) for that same calendar year and uniformly scale the ASC relative payment weights for each update year to make them budget neutral (72 FR 42533). The OPPS relative payment weights are scaled to maintain budget neutrality for the OPPS. We then scale the OPPS relative payment weights again to establish the ASC relative payment weights. To accomplish this, we hold estimated total ASC payment levels constant between calendar years for purposes of maintaining budget neutrality in the ASC payment system. That is, we apply the weight scalar to ensure that projected expenditures from the updated ASC payment weights in the ASC payment system are equal to what would be the current expenditures based on the scaled ASC payment weights. In this way, we ensure budget neutrality and that the only changes to total payments to ASCs result from increases or decreases in the ASC payment update factor.
As discussed in section II.A.1.a. of this final rule with comment period, we are using the CY 2024 claims data to be consistent with the OPPS claims data for this final rule with comment period. Consistent with our established policy, we proposed to scale the CY 2026 relative payment weights for ASCs according to the following method. Holding ASC utilization, the ASC conversion factor, and the mix of services constant from CY 2024, we proposed to compare the estimated total payment using the CY 2025 ASC relative payment weights with the estimated total payment using the CY 2026 ASC relative payment weights to ( printed page 53912) take into account the changes in the OPPS relative payment weights between CY 2025 and CY 2026.
In consideration of our policy to provide a higher ASC payment rate with ASC complexity adjustment codes for certain primary procedures when performed with add-on packaged services, we incorporated estimated total spending and estimated utilization for these codes in our budget neutrality calculation for CYs 2023 and 2024. For the CY 2026 OPPS/ASC proposed rule, our proposed ASC complexity adjustment codes for CY 2026 did not impact the ASC weight scalar. Similarly, for this final rule with comment period, our estimated change in ASC spending related to our final ASC complexity adjustment codes for CY 2026 did not impact the ASC weight scalar.
Additionally, as discussed in section XIII.E. of the CY 2025 OPPS/ASC final rule with comment period (89 FR 94342 through 94361), section 4135(a) and (b) of the CAA, 2023, titled “Access to Non-Opioid Treatments for Pain Relief”, amended sections 1833(t)(16) and 1833(i) of the Act, respectively, to provide for temporary separate payments for non-opioid treatments for pain relief. As discussed in further detail in section XIII.E. of the CY 2025 OPPS/ASC final rule with comment period, for qualifying non-opioid products, we finalized applying an 18 percent payment limitation on the volume weighted payment average of the top 5 services associated with the use of the qualifying non-opioid product. In CY 2024, four of these qualifying nonopioid products were separately payable without the 18 percent payment limitation—HCPCS Codes C9089 (Bupivacaine implant, 1 mg), J0666 (Inj, bupivacaine liposome), J1096 (Dexametha opth insert 0.1 mg), and J1097 (Phenylep ketorolac opth soln). Therefore, to maintain budget neutrality, we estimated the total anticipated reduction in ASC spending for these qualifying non-opioid products for CY 2025 as a result of the 18 percent payment limitation required by section 4135 of the CAA, 2023. Based on the updated 18 percent payment limitations and CY 2024 utilization, we estimated that the proposed CY 2026 payment limitations will not impact the ASC weight scalar. Similarly, based on the updated 18 percent payment limitations and CY 2024 utilization for this final rule with comment period, we estimate that the final CY 2026 payment limitations will not impact the ASC weight scalar.
In section XIII.C.2.b. of this final rule with comment period, we discuss our proposal to unpackage and pay separately for groups of skin substitute products under the ASC payment system beginning January 1, 2026. Currently, these products are packaged into payment for the primary covered surgical procedures. To maintain budget neutrality under the OPPS, the reduction in any APC's relative weights from the loss of skin substitute costs in the APC's geometric mean cost will be offset by an increase in the OPPS weight scalar. To maintain budget neutrality, this increase in the OPPS weight scalar will be offset by a reduction in estimated new OPPS payment for skin substitute APC groups.
Since we request ASCs not to report packaged items and services on ASC claims, we are unable to perform a similar adjustment and determine existing utilization of skin substitute products from ASC claims. To resolve this limitation but maintain budget neutrality within the ASC payment system, we multiplied the change in the geometric mean costs of covered surgical skin procedures in the ASC setting from unpackaging skin substitute products by the utilization of such skin procedures in the ASC setting to approximate the estimated skin substitute payments in the ASC setting. Based on existing surgical procedure utilization and our estimated utilization of skin substitute products in the ASC setting, our estimated separate payments for skin substitutes in the ASC setting did not impact the ASC weight scalar.
We proposed to use the ratio of estimated CY 2025 to estimated CY 2026 total payments (the weight scalar) to scale the ASC relative payment weights for CY 2026. The proposed CY 2026 ASC weight scalar was 0.872. The CY 2026 OPPS/ASC proposed rule inadvertently stated the proposed CY 2026 ASC weight scalar was 0.842 (90 FR 33752 and 90 FR 33846). The ASC payment system payment rates that were displayed in Addendum AA, BB, and FF to the CY 2026 OPPS/ASC proposed rule used the correct proposed ASC weight scalar, 0.872, in the proposed payment rate calculations.
As discussed further in the CY 2025 OPPS/ASC final rule with comment period (89 FR 94363 through 94364), we have historically displayed this figure rounded to the nearest ten thousandth; however, we believe this level of specificity is unnecessarily burdensome for an ASC payment system that is less than one-tenth the size of the OPPS (in which the weight scalar is rounded to the nearest ten-thousandth). Consistent with historical practice, we proposed to scale, using this method (with an ASC weight scalar rounded to the nearest thousandth), the ASC relative payment weights of covered surgical procedures, covered ancillary radiology services, and certain diagnostic tests within the medicine range of CPT codes, which are covered ancillary services for which the ASC payment rates are based on OPPS relative payment weights.
We proposed that we would not scale ASC payment for separately payable covered ancillary services that have a predetermined national payment amount (that is, their national ASC payment amounts are not based on OPPS relative payment weights), such as drugs and biologicals that are separately paid or services that are contractor-priced or paid at reasonable cost in ASCs. Any service with a predetermined national payment amount, which includes the device portion of device-intensive procedures, would be included in the ASC budget neutrality comparison, but scaling of the ASC relative payment weights would not apply to those services or the portion of those services. The ASC payment weights for those services without predetermined national payment amounts would be scaled to eliminate any difference in the total payment between the current year and the update year.
However, as discussed in sections V.B.8.i. and XIII.C.4. of the CY 2025 OPPS/ASC proposed rule, we proposed that the OPPS payment rates used for ratesetting under the ASC payment system for CY 2026 and subsequent years would not incorporate the prospective offset to the OPPS conversion factor, as a result of the 340B remedy offset. Historically, the ASC payment system has generally adopted the OPPS conversion factor used for determining the OPPS payment rates and determining the device portions for device-intensive procedures under the ASC payment system.
We estimated that a 2 percent reduction in the OPPS conversion factor would otherwise reduce ASC payments for device-intensive procedures by approximately one percent; the non-device portions for all covered surgical procedures would otherwise be increased to offset reduction to device portions for device-intensive procedures. For CY 2026, we estimated the reduction to device portions from a 2 percent prospective offset would have reduced proposed CY 2026 ASC expenditures for device-intensive procedures by approximately $42 million and would have otherwise increased the ASC weight scalar by 0.1 percent to offset such reduction.
For any given year's ratesetting, we typically use the most recent full calendar year of claims data to model ( printed page 53913) budget neutrality adjustments. We proposed to use the CY 2024 claims data to model our budget neutrality adjustment for CY 2026.
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: Many commenters reiterated their longstanding recommendation that CMS discontinue the ASC weight scalar, stating that the secondary rescaling process applied to maintain budget neutrality has created an inappropriate and growing payment disparity between ASCs and HOPDs, with ASC reimbursement rates now averaging 50 percent less than HOPD rates for the same procedures despite no evidence of increased differences in capital and operating costs between settings. The commenters noted that this antiquated cost containment mechanism penalizes migration to lower-cost ASC settings and threatens outpatient access to care, with ASC relative weights decreasing by an average of 7 percent each year since 2010 and the proposed 2026 scalar representing the largest projected cut to ASC weights since payment system alignment in 2008. The commenters recommended that we either eliminate the ASC weight scalar or alternatively combine OPPS and ASC utilization to establish a single weight scalar that would improve payment system alignment and more accurately scale for outpatient volume across both sites of service.
Response: We thank the commenters for their input. We disagree that the ASC weight scalar has impeded beneficiary access to outpatient surgical procedures in an ASC setting. In fact, utilization data demonstrates the opposite trend. While the ASC weight scalar has generally declined each year to maintain budget neutrality in the payment system on a year-to-year basis, the data shows that ASC market share of commonly-performed outpatient surgical procedures has steadily increased. For example, CPT code 66984 (Extracapsular cataract removal with insertion of intraocular lens prosthesis (one stage procedure), manual or mechanical technique ( e.g., irrigation and aspiration or phacoemulsification); without endoscopic cyclophotocoagulation), which has been the most commonly-performed surgical procedure in an ASC setting, was reported 73.4 percent of the time in an ASC in CY 2014 when comparing the ASC, HOPD, and physician office settings. This procedure was reported 16.7 percent of the time in a HOPD in CY 2014. For CY 2024, we estimate the share of ASC utilization of CPT code 66984 increased and was reported 81.5 percent of the time in an ASC setting while it declined to 9.2 percent of the time in the HOPD setting. The increase in ASC utilization share across the three outpatient settings and corresponding decline in HOPD utilization share over a 10-year period suggests that the budget neutrality adjustment, and any increase in payment disparity between the OPPS and ASC payment system, does not itself create access to care issues. Therefore, while we will continue to look for changes to our payment methodologies that might improve access to care for Medicare beneficiaries by improving payment accuracies and encourage greater efficiencies in care, we are not accepting the commenters' recommendation to discontinue the ASC weight scalar for CY 2026.
Comment: Many commenters expressed a strong concern with the proposed 4.7 percent reduction in payment for CPT code 66984. Commenters stated that the proposed payment would not keep up with inflation and would have a significant negative financial impact on ASCs that perform such procedures.
Response: After reviewing our OPPS/ASC ratesetting programs, we identified an issue that incorrectly calculated the geometric mean cost for procedures that use intraocular lenses. Specifically, our ratesetting programs inadvertently did not import the HCPCS codes that describe intraocular lens devices and disregarded all intraocular lens costs as not related to the OPPS or ASC payment system. Therefore, such IOL costs had not been packaged into the costs of the primary procedure. After correcting this issue, we observed a 7.3 percent increase in the geometric mean cost of CPT code 66984 as well as a 6.5 percent increase in the geometric mean cost of CPT code 66982 (Extracapsular cataract removal with insertion of intraocular lens prosthesis (1-stage procedure), manual or mechanical technique ( e.g., irrigation and aspiration or phacoemulsification), complex, requiring devices or techniques not generally used in routine cataract surgery ( e.g., iris expansion device, suture support for intraocular lens, or primary posterior capsulorrhexis) or performed on patients in the amblyogenic developmental stage; without endoscopic cyclophotocoagulation). Since the Level 1 Intraocular APC is largely influenced by the cost of CPT code 66984, we have seen a similarly large increase from our proposed national unadjusted ASC payment rate from $1,156.71. For final CY 2026 surgical procedure payment rates under the ASC payment system, we refer readers Addendum AA of this final rule with comment period.
Comment: A commenter expressed concern about a growing national trend of reduced ASC access for retina surgery, noting that a majority of retina specialists in a recently-performed survey report difficulty accessing sufficient OR time for emergent cases such as retinal detachment repair, with many having their ASC surgical block time reduced or eliminated due to inadequate facility reimbursement that results in significant financial losses for procedures like retinal detachment repair (CPT code 67108 (Repair of retinal detachment; with vitrectomy, any method, including, when performed, air or gas tamponade, focal endolaser photocoagulation, cryotherapy, drainage of subretinal fluid, scleral buckling, and/or removal of lens by same technique)) and complex retinal detachment repair (CPT code 67113 (Repair of complex retinal detachment ( e.g., proliferative vitreoretinopathy, stage c-1 or greater, diabetic traction retinal detachment, retinopathy of prematurity, retinal tear of greater than 90 degrees), with vitrectomy and membrane peeling, including, when performed, air, gas, or silicone oil tamponade, cryotherapy, endolaser photocoagulation, drainage of subretinal fluid, scleral buckling, and/or removal of lens)). The commenter emphasized that facilities cannot afford to allocate adequate time to meet patient demand when losing several hundred dollars per case, leading to ASCs limiting retina cases or suspending the service entirely, ultimately moving cases to higher-cost settings contrary to CMS's goal of providing care in the most cost-effective setting.
Response: We appreciate the concerns the commenter has raised and will continue to monitor the payment adequacy of retina detachment procedures and other surgical procedures in the Intraocular Procedures APC clinical family going forward.
Comment: Multiple commenters requested that CMS adjust the ASC payment for CPT code 64590 (insertion or replacement of peripheral neurostimulator pulse generator) from the proposed $16,502.60 to $18,330.98 to maintain consistency with historical methodology where ASC payments typically represent 110 percent-113 percent of the device offset, rather than the proposed 102 percent. The commenters also requested that CMS limit the proposed 16.1 percent payment reduction, stating that such a dramatic decrease will halt patient access to neurostimulation therapies in ( printed page 53914) ASC facilities and goes against site neutrality goals. Some commenters suggested any payment change, particularly for device-intensive procedures, should be capped at no more than 10 percent in any given year, consistent with the IPPS.
Response: We are aware of the reduction in the OPPS and ASC payment rates for CPT code 64590 but believe our APC assignment changes represent an improvement in our payment for high-cost neurostimulator-related procedures. We note that with the reassignment of CPT code 61885 to APC 5465 Level 5 Neurostimulator and Related Procedures, 95 percent of the claims for APC 5464 Level 4 Neurostimulator and Related Procedures contain CPT code 64590 as the primary procedure. Therefore, the proposed geometric mean cost and payment rate of APC 5464 is almost entirely based on the geometric mean cost of CPT code 64590. For this reason, we do not believe it would be appropriate to limit the payment rate decline for CPT code 64590 and are not accepting the commenters' recommendation.
Comment: A commenter recommended a cap on year-to-year reductions, limiting payment decreases, especially for device-intensive procedures in the ASC payment system, to no more than 10 percent.
Response: We did not propose such a policy in our CY 2026 OPPS/ASC proposed rule. We appreciate the recommendation and may take this comment into consideration in future rulemaking.
After consideration of public comments we received, we are finalizing our proposal to use scaled OPPS relative weights to establish ASC relative payment weights and to use the ratio of CY 2025 to CY 2026 total payments (the weight scalar) to scale the ASC relative payment weights for CY 2026. The final CY 2026 ASC weight scalar is 0.872. Consistent with historical practice, we are finalizing our proposal to scale the ASC relative payment weights of covered surgical procedures, covered ancillary radiology services, and certain diagnostic tests within the medicine range of CPT codes, which are covered ancillary services for which the ASC payment rates are based on OPPS relative payment weights.
b. Updating the ASC Conversion Factor
Under the OPPS, we typically apply a budget neutrality adjustment for provider-level changes, most notably a change in the wage index values for the upcoming year, to the conversion factor. Consistent with our final ASC payment policy, for the CY 2017 ASC payment system and subsequent years, in the CY 2017 OPPS/ASC final rule with comment period (81 FR 79751 through 79753), we finalized our policy to calculate and apply a budget neutrality adjustment to the ASC conversion factor for supplier-level changes in wage index values for the upcoming year, just as the OPPS wage index budget neutrality adjustment is calculated and applied to the OPPS conversion factor.
For CY 2026, we calculated the proposed adjustment for the ASC payment system by using the most recent CY 2024 claims data available and estimating the difference in total payment that would be created by introducing the proposed CY 2026 ASC wage indexes. Specifically, holding CY 2024 ASC utilization, service-mix, and the proposed CY 2026 national payment rates after application of the weight scalar constant, we calculated the total adjusted payment using the CY 2025 ASC wage indexes and the total adjusted payment using the proposed CY 2026 ASC wage indexes which included the 5-percent cap on wage index declines. We used the 50 percent labor-related share for both total adjusted payment calculations. We then compared the total adjusted payment calculated with the CY 2025 ASC wage indexes to the total adjusted payment calculated with the proposed CY 2026 ASC wage indexes and applied the resulting ratio of 0.9999 (the proposed CY 2026 ASC wage index budget neutrality adjustment) to the CY 2025 ASC conversion factor to calculate the proposed CY 2026 ASC conversion factor.
Section 1833(i)(2)(D)(v) of the Act requires that the ASC conversion factor be reduced by a productivity adjustment in each calendar year. Section 1886(b)(3)(B)(xi)(II) of the Act defines the productivity adjustment to be equal to the 10-year moving average of changes in annual economy-wide private nonfarm business multifactor productivity (MFP). We finalized the methodology for calculating the productivity adjustment in the CY 2011 PFS final rule with comment period (75 FR 73394 through 73396) and revised it in the CY 2012 PFS final rule with comment period (76 FR 73300 through 73301) and the CY 2016 OPPS/ASC final rule with comment period (80 FR 70500 through 70501). The proposed productivity adjustment for CY 2026 was projected to be 0.8 percentage point, as published in the FY 2026 IPPS/LTCH PPS proposed rule (90 FR 18266) based on IGI's 2024 fourth quarter forecast.
Section 1833(i)(2)(C)(i) of the Act requires that, if the Secretary has not updated amounts established under the revised ASC payment system in a calendar year, the payment amounts shall be increased by the percentage increase in the Consumer Price Index for all urban consumers (CPI-U), U.S. city average, as estimated by the Secretary for the 12-month period ending with the midpoint of the year involved. The statute does not mandate the adoption of any particular update mechanism, but it requires the payment amounts to be increased by the CPI-U in the absence of any update. Because the Secretary updates the ASC payment amounts annually, we adopted a policy, which we codified at § 416.171(a)(2)(ii)), to update the ASC conversion factor using the CPI-U for CY 2010 and subsequent calendar years.
In the CY 2019 OPPS/ASC final rule with comment period (83 FR 59075 through 59080), we finalized a policy to apply the hospital market basket update (which is the inpatient hospital market basket percentage increase reduced by the productivity adjustment) to ASC payment system rates for an interim period of 5 years (CY 2019 through CY 2023), during which we would assess whether there was a migration of the performance of procedures from the hospital setting to the ASC setting as a result of the use of a hospital market basket update, as well as whether there were any unintended consequences, such as less than expected migration of the performance of procedures from the hospital setting to the ASC setting. At that time, the most recently available full year of claims data to assess the expected migration applying the productivity-adjusted hospital market basket update during the interim period was within the period from CY 2019 through CY 2022. However, the impact of the COVID-19 PHE on health care utilization, CY 2020 in particular, was tremendously profound, particularly for elective surgeries, because many beneficiaries avoided healthcare settings, when possible, to avoid possible infection from the SARS-CoV-2 virus. As a result, it was nearly impossible to disentangle the effects from the COVID-19 PHE in our analysis of whether the higher update factor for the ASC payment system caused increased migration to the ASC setting. To analyze whether procedures migrated from the hospital setting to the ASC setting, we needed to use claims data from a period during which the COVID-19 PHE had less of an impact on health care utilization. Therefore, for CY 2024, we finalized our proposal to extend the 5-year interim period an additional 2 years through CY 2024 and ( printed page 53915) CY 2025. We believed hospital outpatient and ASC utilization data from CYs 2023 and 2024 would enable us to more accurately analyze whether the application of the hospital market basket update to the ASC payment system had an effect on the migration of services from the hospital setting to the ASC setting. We revised our regulations at § 416.171(a)(2)(iii), (iv), (vi), (vii), and (viii) which establish the annual update to the ASC conversion factor, to reflect this 2-year extension.
For the CY 2026 OPPS/ASC proposed rule, we proposed to extend our utilization of the hospital market basket update factor in the ASC payment system for one additional year, through CY 2026, as we continue to review and evaluate hospital outpatient and ASC utilization data, as well as the migration of surgical procedures between settings. In conjunction with our proposal, we are revising our regulations at § 416.171(a)(2)(iii), (iv), (vi), (vii), and (viii), which establish the annual update to the ASC conversion factor, the 2.0 percentage point reduction for ASCs that fail to meet the standards for reporting ASC quality measures, and the productivity adjustment, to reflect this one year extension.
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: Multiple commenters supported our continued use of the hospital market basket as the annual update mechanism for ASC payments through CY 2026, with several organizations noting that this policy helps align cost and resource calculations between hospital outpatient departments and ambulatory surgery centers and supports migration of services to lower-cost settings. Many of these commenters recommended we permanently update the ASC payment system with the productivity-adjusted hospital market basket update factor. However, a major hospital industry organization opposed our proposal and recommended that CMS work expeditiously with ASC interested parties to develop a minimally burdensome way to collect ASC-specific cost data that could be used to calculate an appropriate update mechanism, stating that hospitals and ASCs have different costs and serve different patients. The commenters noted that while the hospital market basket update is an improvement over previous methods, evidence indicates it may not accurately reflect ASCs' actual cost structure, and MedPAC has recommended since 2010 that CMS collect ASC cost data to develop an ASC-specific market basket.
Response: We appreciate the feedback from commenters. We did not propose to collect ASC-specific cost data in our CY 2026 OPPS/ASC proposed rule but may take this comment into consideration in future rulemaking. We agree with commenters that providing the same annual update mechanism under the OPPS and ASC payment system can support the migration of services to the lower-cost ASC setting. We are finalizing our proposal to apply the productivity-adjusted hospital market basket update to the ASC payment system for CY 2026.
2. CY 2026 Final ASC Conversion Factor
For CY 2026, we are finalizing our proposal to utilize inpatient hospital market basket percentage increase of 3.3 percent reduced by the productivity adjustment of 0.7 percentage point, resulting in a final hospital market basket update of 2.6 percent for ASCs meeting the quality reporting requirements. Therefore, we are finalizing a 2.6 percent hospital market basket update factor to the CY 2025 ASC conversion factor for ASCs meeting the quality reporting requirements to determine the CY 2026 ASC payment amounts. The ASCQR Program affected payment rates beginning in CY 2014 and, under this program, there is a 2.0 percentage point reduction to the hospital market basket update factor for ASCs that fail to meet the ASCQR Program requirements. We refer readers to section XIV.E. of the CY 2019 OPPS/ASC final rule with comment period (83 FR 59138 through 59139) and section XIV.E. of the CY 2026 OPPS/ASC proposed rule for a detailed discussion of our policies regarding payment reduction for ASCs that fail to meet ASCQR Program requirements.
For CY 2026, we are adjusting the CY 2025 ASC conversion factor ($54.895) by a wage index budget neutrality factor of 1.0000 in addition to the productivity-adjusted hospital market basket update of 2.6 percent, discussed previously, which results in a final CY 2026 ASC conversion factor of $56.322 for ASCs meeting quality reporting requirements. For ASCs not meeting quality reporting requirements, we are adjusting the CY 2025 ASC conversion factor ($54.895) by the wage index budget neutrality factor of 01.0000 in addition to the reduced productivity-adjusted hospital market basket update of 0.6 percent, discussed above, which results in a final CY 2026 ASC conversion factor of $55.224 for ASCs not meeting the quality reporting requirements.
3. Display of the Final CY 2026 ASC Payment Rates
Addenda AA and BB to this final rule with comment period (which are available on the CMS website) display the final ASC payment rates for CY 2026 for covered surgical procedures and covered ancillary services, respectively. The final payment rates included in Addenda AA and BB to this final rule with comment period reflect the full ASC payment update and not the reduced payment update used to calculate payment rates for ASCs not meeting the quality reporting requirements under the ASCQR Program.
These Addenda contain several types of information related to the proposed CY 2026 payment rates. Specifically, in Addendum AA, a “Y” in the column titled “To be Subject to Multiple Procedure Discounting” indicates that the surgical procedure would be subject to the multiple procedure payment reduction policy. As discussed in the CY 2008 OPPS/ASC final rule with comment period (72 FR 66829 through 66830), most covered surgical procedures are subject to a 50 percent reduction in the ASC payment for the lower-paying procedure when more than one procedure is performed in a single operative session.
The values displayed in the column titled “Final CY 2026 Payment Weight” are the final relative payment weights for each of the listed services for CY 2026. The final relative payment weights for all covered surgical procedures and covered ancillary services where the ASC payment rates are based on OPPS relative payment weights were scaled for budget neutrality. Therefore, scaling was not applied to the device portion of the device-intensive procedures; services that are paid at the PFS nonfacility PE RVU-based amount; separately payable covered ancillary services that have a predetermined national payment amount, such as drugs and biologicals and brachytherapy sources that are separately paid under the OPPS; or services that are contractor-priced or paid at reasonable cost in ASCs. This includes separate payment for non-opioid pain management drugs.
To derive the final CY 2026 payment rate displayed in the “Final CY 2026 Payment Rate” column, each ASC payment weight in the “Final CY 2026 Payment Weight” column was multiplied by the final CY 2026 conversion factor. The conversion factor includes a budget neutrality adjustment for changes in the wage index values and the annual update as reduced by the ( printed page 53916) productivity adjustment. The final CY 2026 ASC conversion factor uses the final CY 2026 productivity-adjusted hospital market basket update factor of 2.6 percent (which is equal to the inpatient hospital market basket percentage increase of 3.3 percent reduced by the productivity adjustment of 0.7 percentage point).
Comment: A commenter requested that we remove the Multiple Procedure Payment Reduction (MPPR) assignment from CPT code 64596 (insertion of neurostimulator electrode array, peripheral nerve) and reassign it from “Y” to “N” status in ASC Addendum AA for CY 2026, stating that the current multi-procedure discount results in inequitable payment access since bilateral implantation or multiple nerve targeting requires separate StimRouter devices with individual incision and tunneling procedures. The commenter noted that CPT code 64596 is an outlier compared to other percutaneous implantation codes (64555, 64575, 64590) that do not have MPPR assignment, and emphasized that the clinical need for multiple integrated neurostimulators is similar to other peripheral nerve stimulation devices where each implant requires additional work and separate device costs. The commenter also noted confusion among providers since StimRouter was previously reimbursed under CPT 64555 (which has no multi-procedure discount) prior to the creation of CPT 64596 in January 2024.
Response: We do not agree that CPT code 64596 is an outlier in assigning the MPPR status to “Y” in that many other surgical procedures with significant device costs in the Neurostimulator and Related Procedures APC clinical family are also discounted under the MPPR policy, such as CPT code 64583 (Revision or replacement of hypoglossal nerve neurostimulator array and distal respiratory sensor electrode or electrode array, including connection to existing pulse generator). However, we appreciate the concerns the commenter has raised with respect to the impacts of our MPPR policy on access to surgical procedures with significant device costs. We will continue to monitor the concerns the commenter has raised and may take the commenter's recommendation into consideration in future rulemaking.
In Addendum BB, there are no relative payment weights displayed in the “Final CY 2026 Payment Weight” column for items and services with predetermined national payment amounts, such as separately payable drugs and biologicals. The “Final CY 2026 Payment” column displays the final CY 2026 national unadjusted ASC payment rates for all items and services. The final CY 2026 ASC payment rates listed in Addendum BB for separately payable drugs and biologicals are based on the most recently available data used for payment in physicians' offices. For CY 2021, we finalized adding a new column to ASC Addendum BB titled “Drug Pass-Through Expiration during Calendar Year” where we flag through the use of an asterisk each drug for which pass-through payment is expiring during the calendar year (that is, on a date other than December 31st).
Addendum EE to this final rule with comment period provides the HCPCS codes and short descriptors for surgical procedures that are to be excluded from payment in ASCs for CY 2026.
Addendum FF to this final rule with comment period displays the OPPS payment rate (based on the standard ratesetting methodology), the APC device offset percentage, the device offset percentage for determining device-intensive status (based on the standard ratesetting methodology), and the device portion of the ASC payment rate for CY 2026 for covered surgical procedures.
XIV. Cross-Program Measures for the Hospital Outpatient Quality Reporting (OQR), Rural Emergency Hospital Quality Reporting (REHQR), and Ambulatory Surgical Center Quality Reporting (ASCQR) Programs
A. Background
We refer readers to sections XV., XVI., and XVII. of this final rule with comment period for program-specific background information, including the statutory authorities and previously finalized and newly proposed measure sets, for the Hospital Outpatient Quality Reporting (OQR), Rural Emergency Hospital Quality Reporting (REHQR), and Ambulatory Surgical Center Quality Reporting (ASCQR) Programs, respectively.
B. Measure Concepts Under Consideration for Future Years in the Hospital OQR, REHQR, and ASCQR Programs—Request for Information (RFI): Well-Being and Nutrition
In the CY 2026 OPPS/ASC proposed rule (90 FR 33754), we sought input on well-being and nutrition measures for consideration in future rulemaking for the Hospital OQR, REHQR, and ASCQR Programs. Well-being is a comprehensive approach to disease prevention and health promotion, as it integrates mental and physical health while emphasizing preventative care to proactively address potential health issues.[252] This comprehensive approach emphasizes person-centered care by promoting the well-being of patients and family members. We sought comments on tools and measures that assess overall health, happiness, and satisfaction in life, which could include aspects of emotional well-being, social connections, purpose, and fulfillment. We also requested input and comments on the applicability of tools and constructs that assess the integration of complementary and integrative health, skill building, and self-care.
We also sought comments on tools and measures that assess optimal nutrition and preventive care in the Hospital OQR, REHQR, and ASCQR Programs. Assessments for nutritional status may include strategies, guidelines, and practices that promote healthy eating habits and ensure individuals receive the necessary nutrients for maintaining health, growth, and overall well-being. Such assessments may also include aspects of health that support or mediate nutritional status, such as physical activity and sleep. In this context, preventive care plays a vital role by proactively addressing factors that may lead to poor nutritional status or related health issues. These efforts not only support optimal nutrition but also work to prevent conditions that could otherwise hinder an individual's health and nutritional needs.
Comment: Many commenters supported the inclusion of well-being and nutrition measures in the Hospital OQR, REHQR, and ASCQR Programs. Many commenters emphasized that including evidence-based, actionable metrics on these topics in quality reporting programs is essential for managing chronic illness, improving patient outcomes, and advancing health equity. Several commenters encouraged CMS to adopt malnutrition quality measures, such as the Malnutrition Care Score (MCS, formerly known as the Global Malnutrition Composite Score) currently used in the Hospital Inpatient Quality Reporting (IQR) program, for the Hospital OQR, REHQR, and ASCQR programs. A few commenters emphasized the importance of implementing measures that use validated, flexible, and interdisciplinary tools, such as the Patient-Reported Outcomes Measurement Information System (PROMIS), World Health Organization Well-Being Index (WHO-5), Patient Health Questionnaire-9 ( printed page 53917) (PHQ-9), Canadian Occupational Performance Measure (COPM), Occupational Self-Assessment (OSA), and Self-Management Ability Scale (SMAS), that capture individualized outcomes including participation, resilience, and quality of life.[253 254 255 256 257 258] Other commenters recommended Functional Communication Measures (FCMs) for swallowing to ensure adequate nutrition, hydration, and recovery.[259] A few commenters recommended incorporating measures that assess care transitions, patient activation, and personalized goals to support pathways to well-being. A few commenters expressed the importance of integrating evidence-based measures that target the aging population and incorporating protective factors such as food access, community safety, financial security, and health literacy. Other commenters encouraged developing new measures that address the topic of maternal recovery in the outpatient setting, as well as measures that recognize specific interventions such as Medical Nutrition Therapy, food insecurity screenings, and chronic care interventions.
A few commenters urged CMS to consider the applicability of any well-being or nutrition measures to the outpatient setting, noting that facilities should not be held accountable for factors beyond their control, such as lack of access to healthy foods. Many commenters expressed concern that implementing measures related to well-being and nutrition in healthcare facilities, particularly in rural or resource-limited settings, may be administratively burdensome. A few commenters stated that, although ASCs play a vital role in delivering surgical and procedural care, the short-term, episodic nature of ASC services is not optimal for addressing broader well-being or nutritional needs, which require longitudinal interventions and follow-up beyond the ASC setting. A few commenters encouraged CMS to engage with primary care providers, patients, and caregivers to ensure that new domains reflect both clinical relevance and patient experience. A few commenters urged CMS to ensure adequate reimbursement for nutrition care measures that reflects true costs and supports equitable patient access.
Many commenters expressed concern over the proposal to remove SDOH measures, arguing that these screenings provide critical insights into patient needs and support holistic care delivery. Some of these commenters recommended aligning any future well-being and nutrition measures with existing social determinants of health screening tools and identified food insecurity screening as a foundational tool for addressing nutrition and well-being. A few commenters emphasized the importance of aligning nutrition measures with clinical workflows and addressing both food insecurity and diet quality.
Several commenters stated that barriers to nutrition and well-being, such as food insecurity and social isolation, should be addressed through targeted interventions and community partnerships. A few commenters stressed the need to address resource gaps through Federally funded programs that impact nutrition and well-being while others recommended incentivizing facilities to partner with community organizations to expand access to nutrition services, including medically tailored meals and food pharmacies. A few commenters emphasized the importance of ensuring continuity of care through discharge planning and community referrals. A few commenters recommended expanding facility-based measures to include post-discharge follow-up and integration with community resources to support long-term health outcomes.
A few commenters recommended the use of standardized tools and existing data sources, such as electronic health records (EHRs), to simplify administration and integration into clinical workflows. A few commenters recommended pilot testing in diverse settings and populations to ensure reliability, practicality, and applicability of new measures before full implementation. A commenter suggested that new measures related to well-being and nutrition should be electronic clinical quality measures (eCQMs) to align submission formats across CMS quality reporting programs. A commenter urged CMS to leverage the Gravity Project's United States Core Data for Interoperability (USCDI) and Fast Healthcare Interoperability Resources® (FHIR®) standards to improve interoperability and integrate community-based supports.[260 261]
Response: We thank the commenters for their input. While we are not responding to specific comments in response to the RFI in this final rule with comment period, we will take this feedback into consideration for our future measure development efforts for the Hospital OQR, REHQR, and ASCQR Programs.
C. Changes to the Hospital OQR, REHQR, and ASCQR Program Measure Sets
1. Removal of the COVID-19 Vaccination Coverage Among Healthcare Personnel (HCP) Measure From the Hospital OQR and ASCQR Programs Beginning With the CY 2024 Reporting Period/CY 2026 Payment Determination
We refer readers to the CY 2022 OPPS/ASC final rule with comment period where we adopted the COVID-19 Vaccination Coverage Among HCP measure into the Hospital OQR and ASCQR Programs (86 FR 63824 through 63833 and 86 FR 63875 through 63883, respectively) and the CY 2024 OPPS/ASC final rule with comment period where we modified the COVID-19 Vaccination Coverage Among HCP measure to account for updated vaccine guidance (88 FR 81963 through 81968 and 88 FR 82013 through 82017, respectively).
In the CY 2026 OPPS/ASC proposed rule (90 FR 33754 through 33755), for the Hospital OQR and ASCQR Programs, we proposed to remove the COVID-19 Vaccination Coverage Among HCP measure beginning with the CY 2024 reporting period/CY 2026 payment determination under removal Factor 8, as the costs associated with the measure outweigh the benefit of its continued use in the program (42 CFR 419.46(i)(3)(i)(H) and 416.320(c)(2)(viii), respectively). Reporting on this measure currently requires reporting data on COVID-19 Vaccination Coverage ( printed page 53918) Among HCP for at least 1 week every month. This requires healthcare facilities to track current vaccination status for all employees, licensed independent practitioners, adult students/trainers and volunteers, and other contract personnel and log in to the National Healthcare Safety Network (NHSN) system to report the data monthly, either manually in NHSN or by uploading a comma-separated value (CSV) file.[262] The estimated burden of collecting this information annually across all 3,200 hospitals in the Hospital OQR Program is between $1,446,400 and $1,687,680. Across the 4,590 ASCs in the ASCQR Program, the estimated annual burden is between $2,074,680 and $2,420,766. We refer readers to section XXIII. of this final rule with comment period for more details on this estimated burden calculation.
When we first adopted the COVID-19 Vaccination Coverage Among HCP measure for the Hospital OQR and ASCQR Programs, the U.S. was in the midst of a Public Health Emergency (PHE) that incurred millions of cases and over 718,000 COVID-19 deaths (86 FR 63825 and 86 FR 63875 through 63876, respectively). While preventing the spread of COVID-19 remains a public health goal, the PHE ended on May 11, 2023.[263] In addition, the death rate due to COVID-19 in the U.S. has decreased since the adoption of this measure.[264] In August 2021, when this measure was being proposed, the U.S. was averaging over 6,000 deaths related to COVID-19 per week.[265] In April 2023, the last full month of the PHE, weekly number of deaths attributed to COVID-19 averaged around 1,300.[266] With the end of the PHE and the decrease in COVID-19 deaths, we believe the continued costs and burden to healthcare facilities of tracking and monthly reporting on this measure outweigh the benefit of continued information collection on COVID-19 vaccination coverage among HCP. As it may be costly for hospitals and ASCs to continue to report on the COVID-19 Vaccination Coverage Among HCP measure, removal of this measure will allow for the Hospital OQR and ASCQR Programs to focus on other clinical goals.
We stated that if this proposal is finalized, hospitals and ASCs that do not report their CY 2024 reporting period data for the COVID-19 Vaccination Coverage Among HCP measure to CMS would not be considered noncompliant with the measure for their CY 2026 payment determination (that is, hospitals and ASCs that do not report CY 2024 reporting period data would not be penalized for CY 2026 payments due to this measure). Any COVID-19 Vaccination Coverage Among HCP measure data received by CMS would not be used for public reporting or payment purposes.
We also stated that if this proposal is not finalized, hospitals and ASCs that do not report their CY 2024 reporting data for the COVID-19 Vaccination Coverage Among HCP measure to CMS would be considered noncompliant with the measure for their CY 2026 payment determination and would receive a letter of noncompliance. Payment adjustments would apply to CY 2026 payment determination for fee-for-service claims as previously finalized.
We invited public comments on these proposals.
Comment: Many commenters supported the removal of the COVID-19 Vaccination Coverage Among HCP measure and agreed that the burden imposed by tracking COVID-19 vaccination among healthcare personnel outweighs the benefits of its continued use in the Hospital OQR and ASCQR Programs. Many commenters supported removal of this measure because it requires near-continuous monitoring of each employee's vaccination status due to changing vaccination definitions. Commenters asserted that the labor and resource intensiveness of collecting and reporting data for this measure requires significant staff time and resources that are diverted from other clinical priorities, such as direct patient care. Many commenters agreed that this measure no longer aligns with an urgent public health priority or provides meaningful or actionable data for quality improvement. Commenters stated that the measure has become outdated, especially since the COVID-19 PHE declaration ended in May 2023.
Response: We thank commenters for their support.
Comment: Several commenters did not agree with removing this measure, asserting that the benefits of maintaining vaccination data for public health surveillance and promoting vaccine uptake outweigh the costs of tracking vaccination coverage. Commenters stated that systematic reporting of healthcare personnel vaccination rates is essential for monitoring and responding to future infectious disease outbreaks in healthcare settings. Instead of removal, these commenters recommended modifying the measure to reduce administrative burden so that preparedness remains a priority. Commenters expressed concern that removing the measure could reduce institutional accountability and preparedness, ultimately resulting in higher costs associated with preventable infections.
Several commenters asserted that vaccination is a critical strategy to minimize preventable harm and maintain safe healthcare environments, and that removing the measure would contradict the healthcare industry's obligation to uphold high standards of care and infection prevention. Some commenters were concerned that removing this measure could reduce attention to vaccination programs, potentially compromising healthcare system resilience. Commenters stated that removing this measure could lead to decreased vaccination rates among healthcare personnel, thereby increasing the risk of hospital-acquired infections and compromising patient safety for vulnerable patient populations, such as those who are immunocompromised, undergoing cancer treatment, or pregnant. A few commenters stated that vaccination of healthcare personnel is particularly important in ASC settings where many patients are undergoing cardiovascular, oncological, or mediport placement procedures.
Response: We acknowledge commenter concerns about patient safety, protecting vulnerable populations, and maintaining public health surveillance and readiness. We agree that patient safety practices and high-quality healthcare for all patients are a priority, and we expect participating hospitals and ASCs to support safe practices that protect patients from infections and other preventable harm. The removal of the COVID-19 Vaccination Coverage Among HCP measure is not intended to interfere with infection control practices, but rather to alleviate the burden associated with data collection ( printed page 53919) and reporting on a monthly cadence now that the PHE has ended. We note that hospitals and ASCs are not restricted from tracking HCP vaccinations that are appropriate for the setting of care and the population served.
Comment: A few commenters expressed concerns about the proposed applicability date for removing the COVID-19 Vaccination Coverage Among HCP measure, which is the CY 2026 payment determination. Commenters stated that hospitals and ASCs have already invested significant resources to complete the process of submitting CY 2024 quality data used for CY 2026 payment determination. These commenters discouraged the proposal to remove measures applicable to past reporting periods, especially for relatively new measures, as it creates confusion and burden among hospitals and ASCs.
Response: We understand commenters' concern regarding the timing around removal of these measures and the confusion and burden this may impose on hospitals and ASCs that have already submitted CY 2024 quality data. However, because we have determined that the cost of reporting on these measures outweighs the benefits of retaining them in the program, it would place an undue burden on hospitals and ASCs to continue requiring reporting on these measures for an additional year. We note that hospitals and ASCs that do not report their CY 2024 reporting period data for the COVID-19 Vaccination Coverage Among HCP measure to CMS will not be considered noncompliant with the measure for purposes of their CY 2026 payment determination (that is, hospitals and ASCs that do not report CY 2024 reporting period data will not be penalized for CY 2026 payments due to this measure). Any COVID-19 Vaccination Coverage Among HCP measure data received by CMS will not be used for public reporting or payment purposes.
After consideration of the comments received, we are finalizing our proposal to remove the COVID-19 Vaccination Coverage Among HCP measure from the Hospital OQR and ASCQR Programs beginning with the CY 2024 reporting period/CY 2026 payment determination.
2. Removal of the Hospital Commitment to Health Equity (HCHE) Measure From the Hospital OQR and REHQR Programs and the Facility Commitment to Health Equity (FCHE) Measure From the ASCQR Program Beginning With the CY 2025 Reporting Period/CY 2027 Payment or Program Determination
We refer readers to the CY 2025 OPPS/ASC final rule with comment period where we adopted the Hospital Commitment to Health Equity (hereafter referred to as HCHE) measure into the Hospital OQR and REHQR Programs and the Facility Commitment to Health Equity (hereafter referred to as FCHE) measure into the ASCQR Program (89 FR 94368 through 94381).
In the CY 2026 OPPS/ASC proposed rule (90 FR 33755 through 33756), for the Hospital OQR, REHQR, and ASCQR Programs, we proposed to remove the HCHE and FCHE measures beginning with the CY 2025 reporting period/CY 2027 payment or program determination under removal Factor 8, as the costs associated with achieving a high score on the measure outweigh the benefit of its continued use in the program (§§ 419.46(i)(3)(i)(H), 419.95(e)(3)(i)(H), and 416.320(c)(2)(viii), respectively).
When adopted, we intended the collection of data described in the five domains of these measures to provide hospital, REH, and ASC leadership with meaningful and actionable health data to drive quality improvements to eliminate health disparities. We have a renewed focus on measurable clinical outcomes and direct patient care. The HCHE and FCHE measures are structural measures which do not directly assess these areas of interest. Further, based on the feedback received from hospitals, REHs, and ASCs, the burden of collecting these measures may outweigh the benefits. Removal of these measures will alleviate an estimated annual burden of approximately 533 hours, at a cost of $22,518, across all participating hospitals (89 FR 94523); 6 hours, at a cost of $332, across all participating REHs (89 FR 94530); and 746 hours, at a cost of $41,313 across all participating ASCs (89 FR 94534).
An important goal of the Hospital OQR, REHQR, and ASCQR Programs is to move forward in the least burdensome manner possible while maintaining a parsimonious set of meaningful quality measures and continuing to incentivize improvement in the quality of care provided to patients. Removing these measures from the Hospital OQR, REHQR and ASCQR Programs serves this goal. Our priority is to re-focus on measurable clinical outcomes as well as identifying quality measures on topics of prevention, nutrition, and well-being. As such we refer readers to “Measure Concepts under Consideration for Future Years in the Hospital OQR, REHQR, and ASCQR Programs—Request for Information (RFI): Well-Being and Nutrition” in section XIV.B. of this final rule with comment period.
We acknowledge that some hospitals, REHs, and ASCs may have expended resources to implement some or all of the activities described in the HCHE and FCHE measures attestation statements to be able to attest “yes” for measure reporting purposes.
We stated that if this proposal is finalized, hospitals, REHs, and ASCs that do not report their CY 2025 reporting period data for the HCHE or FHCE measure to CMS would not be considered noncompliant with the measure for purposes of their CY 2027 payment or program determination (that is, hospitals, REHs, or ASCs that do not report CY 2025 reporting period data would not be penalized for CY 2027 payments due to this measure, if applicable). Any HCHE or FCHE measure data received by CMS would not be used for public reporting or payment purposes.
We also stated that if this proposal is not finalized, hospitals, REHs, or ASCs that do not report their CY 2025 reporting data for the HCHE or FCHE measures to CMS would be considered noncompliant with the measure for their CY 2027 payment or program determination and would receive a letter of noncompliance. Payment adjustments would apply to CY 2027 payment determination fee-for-service (FFS) claims as previously finalized in the Hospital OQR and ASCQR Programs.
We invited public comments on this proposal.
Comment: Many commenters supported the removal of the HCHE measure from the Hospital OQR and REHQR Programs, and the FCHE measure from the ASCQR Program. Several commenters supported the removal as part of broader efforts to streamline quality reporting programs and reduce regulatory burden. They agreed that eliminating measures like HCHE and FCHE would allow facilities to redirect resources toward higher-priority initiatives and patient care, focusing on more tangible interventions and measurable outcomes rather than attestation-based requirements. A commenter stated that the measure duplicates efforts already met through existing standards, such as The Joint Commission's National Patient Safety Goal NPSG.16.01.01—Improve Health ( printed page 53920) Care Equity. This commenter encouraged CMS to align measures with existing standards.
Many commenters expressed concerns about structural measures, emphasizing their concerns about administrative burden and the limited impact of these measures on improving patient outcomes. These commenters stated that the burden of reporting these measures outweighs the benefits, highlighting challenges in implementation such as a lack of infrastructure, training, and staff capacity to collect and act on the data meaningfully. Several commenters stated these challenges were particularly acute for small and rural facilities, including single-specialty ASCs.
Many commenters stated that the FCHE measure does not reflect the structure of ASCs to provide short-term, episodic care, and ASCs generally do not have the infrastructure nor the resources to meet the requirements of those measures, such as EHR technology. A commenter asserted that the cost of implementation was higher than the CMS estimate for ASCs.
Many commenters supported the removal of these measures as they expressed the measure was not appropriately tailored to the outpatient hospital, ASC, and REH settings. A few commenters supported the removal due to their lack of consensus-based entity (CBE) endorsement, measure testing, and validity. A few commenters expressed concern about the measure scoring methodology, specifically, the complexity of the reporting requirements and the actionability of the data.
Response: We thank the commenters for their support. We agree that the removal of this measure will reduce the administrative burden and the challenges related to implementation in the ASC setting. We note that both the HCHE and FCHE measures went through the rigorous measure development lifecycle outlined at the CMS Measures Management System website,[267] which includes measure testing and reliability analysis. For the Hospital OQR Program and ASCQR Program, we note that section 1833(t)(17) of the Act does not require that each measure adopted for these programs be CBE-endorsed. For the REHQR Program, section 1861(kkk)(7)(C)(i) of the Act generally requires that quality measures specified by the Secretary for the REHQR Program be endorsed by a CBE; however, section 1861(kkk)(7)(C)(ii) of the Act provides an exception to the general CBE-endorsement requirement, stating that in the case of a specified area or medical topic determined appropriate by the Secretary for which a measure has not been endorsed by the entity with a contract under section 1890(a) of the Act, the Secretary may specify a measure that is not endorsed as long as due consideration is given to measures that have been endorsed or adopted by a consensus organization identified by the Secretary. We reviewed CBE-endorsed measures and were unable to identify any other CBE-endorsed measures on this topic, and therefore we believe the exception in section 1861(kkk)(7)(C)(ii) of the Act applies for purposes of this measure for the REHQR Program.
Comment: A few commenters supported the removal of these measures and stated they remain committed to ensuring quality care for all patients and investing in culturally responsive care models.
Response: We appreciate commenters' support and commitment to maintaining quality care for all patients. Despite removal of these measures, facilities will still be able to collect data that is important to their patient care initiatives and reflects the unique needs of their specific patient population.
Comment: Many commenters opposed the removal of the HCHE measure from the Hospital OQR and REHQR Programs, and the FCHE measure from the ASCQR Program, emphasizing the critical role these measures play in advancing health equity and addressing disparities in care delivery. These commenters stated that the measures provide structured accountability for facility leadership to prioritize equity work, collect data on social determinants of health, and implement quality improvement initiatives.
Several commenters asserted that removing the measure would signal a retreat from CMS' stated goals of reducing disparities and improving care for vulnerable populations, including those with severe mental illness, racial and ethnic minorities, rural populations, those with low socioeconomic status, and dual eligible beneficiaries. A commenter stated that removing the HCHE and FCHE measures from the respective programs contradicts the goals of the Make America Healthy Again initiative.
Response: We acknowledge the commenters' concerns. We agree that holding facilities accountable for high-quality healthcare delivery to all beneficiaries is important and remains a priority for the Hospital OQR, REHQR, and ASCQR Programs. We remain focused on identifying measures that balance feasibility, burden, and impact, while aligning with shifting administration priorities as the health system continues to evolve. We are identifying ways to reduce provider reporting burden, while continuing to hold facilities accountable for measurable clinical health outcomes and patient safety. We appreciate the commenters' support for the Make America Healthy Again initiative and will review suggestions received on the new measure RFI in section XIV.B. of this final rule with comment period as we consider relevant measures to introduce in the future.
Comment: Many commenters stated that removal of the measure could result in decreased quality of care, reduce transparency and accountability, and exacerbate gaps in care quality, ultimately resulting in worsened health outcomes and higher costs. Several commenters cited examples of persistent disparities in care, including maternal mortality rates across the population and differences between urban and rural health outcomes. A few commenters asserted removal of this measure would widen the existing gap between medical and behavioral health institutions, emphasizing that addressing social needs such as food insecurity, housing instability, and transportation barriers is essential for improving health outcomes, particularly chronic diseases, and reducing preventable hospital admissions.
Response: We acknowledge commenters' concerns and considered the potential impact on patient outcomes and health disparities when deciding to remove this measure. We urge facilities to continue to incorporate industry standards that may address challenges that could impact safe, high-quality healthcare delivery. Despite removal of these measures, facilities will still be able to collect data that is important to their patient care initiatives and reflects the unique needs of their specific patient population.
Comment: Several commenters stated that the benefits of this measure, which is reducing the costs associated with health inequities, outweigh CMS' estimated burden of implementing the HCHE and FCHE measures. A few commenters stated that structural measures incur a low reporting burden, as facilities do not incur financial penalties so long as they report complete and accurate data. A commenter stated that CMS should prioritize the benefits for Medicare beneficiaries and taxpayers, with ( printed page 53921) burden to providers as a secondary consideration.
Response: We appreciate commenters' input regarding the burden associated with reporting on the HCHE and FCHE measures. We agree with commenters that the reporting burden associated with structural measures is typically small; however, we believe that costs are multi-faceted and include administrative costs to facilities, maintaining information collection systems, and analyzing reported data. At this time, we remain focused on identifying outcome measures that balance feasibility, burden, and impact, while aligning with administration priorities. We are identifying ways to reduce provider reporting burden, while continuing to hold facilities accountable for measurable clinical health outcomes and patient safety. We have considered both the benefits and the burden of these measures, and determined the multi-faceted costs associated with the measures outweigh the benefits of continued use in the program.
Comment: A few commenters stated that facilities have already incurred the burden to create programs to support the HCHE and FCHE measures and have already invested resources in collecting and submitting data for the CY 2025 reporting period. These commenters stated that CMS should delay the removal of the measures so as to not erode the substantial efforts already underway. A commenter expressed concerns about the proposed effective date for the removal of the FCHE measures, which is the CY 2027 payment determination.
Response: We acknowledge commenters' concern regarding the timing around measure removal and the costs of preparing to report on the measures. However, because we have determined that the cost of reporting on this measure outweighs the benefits of retaining it in the program, we are removing these measures at the earliest feasible reporting period of CY 2025, for which the submission period has not yet opened and the deadline would not be until in May 2026. This negates the need for hospitals and ASCs to expend additional resources on reporting a measure for which we have determined that the costs outweigh the benefits.
Comment: Several commenters recommended refining the HCHE and FCHE measures rather than removing them entirely. A few commenters suggested modifications to reduce administrative burden while preserving the measure's intent and improving value. A few commenters proposed adjustments to scoring methodologies, reporting frequency, or voluntary submission to make the measure more feasible for facilities to implement. A few commenters expressed concerns about removing structural measures as clinical-based measures do not incentivize actionable change at the systemic level, citing “measure gaming”, or demonstrating improvements on measures that are disconnected from true improvements in patient care, as a reason to maintain these measures. A few commenters encouraged CMS to explore alternative mechanisms for tracking equity-related efforts and integrating social needs into care delivery, such as voluntary documentation of Z-codes.
Response: We thank the commenters for their recommendations and will consider them as we evaluate any potential future measures on this subject. We note that structural measures, such as the HCHE and FCHE measures, evaluate a hospital or facility's capacity, systems, and processes to deliver high quality care, whereas outcome measures assess the impact of health care services or interventions on patients.[268] We are focused on identifying ways to reduce provider reporting burden while continuing to hold hospitals and facilities accountable for measurable clinical outcomes and patient safety. Hospitals and facilities are encouraged to continue to engage in activities to close gaps in care and collect data that is important to their patient care initiatives and reflect the needs of their patient population regardless of whether it is required for the Hospital OQR, REHQR, and ASCQR Programs.
After consideration of the comments received, we are finalizing our proposal to remove the HCHE measure from the Hospital OQR and REHQR Programs, and the FCHE measure from the ASCQR Program beginning with the CY 2025 reporting period/CY 2027 payment determination.
3. Removal of Two Social Drivers of Health Measures From the Hospital OQR, REHQR, and ASCQR Programs Beginning With the CY 2025 Reporting Period
In the CY 2026 OPPS/ASC proposed rule (90 FR 33756), we proposed to remove two social drivers of health (SDOH) process measures from the Hospital OQR, REHQR, and ASCQR Programs beginning with the CY 2025 reporting period: Screening for Social Drivers of Health (adopted at 89 FR 94381 through 94398) and Screen Positive Rate for Social Drivers of Health (adopted at 89 FR 94398 through 94403), for which the CY 2025 reporting period would be voluntary, and mandatory reporting would begin with the CY 2026 reporting period/CY 2028 payment or program determination.
We proposed to remove the SDOH measures beginning with the CY 2025 reporting period under removal Factor 8 as the costs associated with the measure outweigh the benefit of its continued use in these programs (§§ 419.46(i)(3)(i)(H), 419.95(e)(3)(i)(H), and 416.320(c)(2)(viii), respectively). We heard from some hospitals, REHs, and ASCs concerned with the costs and resources associated with screening patients via manual processes, manually storing such data, training staff, and altering workflows for these measures. In the CY 2025 OPPS/ASC final rule with comment period, we estimated a total annual burden of 6,878,055 hours at a cost of $168,460,032 in the Hospital OQR Program (89 FR 94523 and 94524), 12,984 hours at a cost of $318,163 in the REHQR Program (89 FR 94530 and 94531), and 711,479 hours at a cost of $17,447,164 in the ASCQR Program (89 FR 94534 and 94535), to screen all admitted patients in accordance with the Screening for SDOH measure specifications and report the measure data to CMS. For the Screen Positive Rate for SDOH measure, we estimated a total annual burden of 533 hours at a cost of $29,518 in the Hospital OQR Program (89 FR 94524), 6 hours at a cost of $332 in the REHQR Program (89 FR 94531 and 94532), and 746 hours at a cost of $41,313 in the ASCQR Program (89 FR 94535), to report the measure data. We noted that the HQR system calculates the rate for these two measures, and that hospitals, REHs, and ASCs' responsibility is to report the aggregate number of patients screened, the aggregate number of patients that screened positive, and their total patient population.
We discussed in the CY 2026 OPPS/ASC proposed rule that the costs of the continued use of these measures in the Hospital OQR, REHQR, and ASCQR Programs outweigh the benefits to facilities and patients. Removal of these measures would alleviate the burden on hospitals, REHs, and ASCs to manually screen each patient and submit data each reporting cycle, allowing hospitals, REHs, and ASCs to focus resources on measurable clinical outcomes and direct patient care. This would also remove the patient burden associated with repeated SDOH screenings across multiple healthcare facilities. We refer readers to “Measure Concepts under ( printed page 53922) Consideration for Future Years in the Hospital OQR, REHQR, and ASCQR Programs-Request for Information (RFI): Well-Being and Nutrition” in section XIV.B. of this final rule with comment period for more information regarding our areas of focus for new measures. We acknowledged that some hospitals, ASCs and REHs may have expended resources to implement SDOH screenings, however, facilities that had already implemented such screenings prior to adoption of the measures would not have expended similar resources. The objectives of the Hospital OQR, REHQR, and ASCQR Programs continue to incentivize the improvement of care quality and health outcomes for all patients through transparency and use of appropriate quality measures.
We invited public comments on this proposal.
Comment: Many commenters supported removing the Screening for SDOH and Screen Positive Rate for SDOH measures. These commenters emphasized that the measures require significant resources for data collection, which could distract hospitals, REHs, and ASCs from focusing on direct patient outcomes and other quality improvement initiatives. Many commenters supported removal of these measures because the measures do not show whether facilities are addressing the specific risk factors impacting patients in response to screenings and patients have expressed frustration resulting from duplicative screenings across healthcare settings.
Response: We thank the commenters for their support.
Comment: Several commenters supported removal due to a lack of testing, appropriate tailoring to the outpatient hospital, ASC, and REH settings, and CBE endorsement. Many commenters supported removal of the FCHE measure as ASCs do not provide longitudinal care, and it is therefore unreasonable to expect them to comprehensively address SDOH. A few commenters expressed that ASCs generally do not have the infrastructure nor the resources to meet the requirements of those measures, such as social workers on staff or knowledge of resources in a patient's own community as many patients travel for ASC care.
Response: We note that the two SDOH measures went through the rigorous measure development lifecycle outlined at the CMS Measures Management System website [269] which includes measure testing and reliability analysis. For the Hospital OQR Program and ASCQR Program, we note that section 1833(t)(17) of the Act does not require that each measure adopted for these programs is CBE-endorsed. For the REHQR Program, section 1861(kkk)(7)(C)(i) of the Act generally requires that quality measures specified by the Secretary for the REHQR Program be endorsed by a CBE; however, section 1861(kkk)(7)(C)(ii) of the Act provides an exception to the general CBE-endorsement requirement, stating that in the case of a specified area or medical topic determined appropriate by the Secretary for which a measure has not been endorsed by the entity with a contract under section 1890(a) of the Act, the Secretary may specify a measure that is not endorsed as long as due consideration is given to measures that have been endorsed or adopted by a consensus organization identified by the Secretary. We reviewed CBE-endorsed measures and were unable to identify any other CBE-endorsed measures on this topic, and therefore we believe the exception in section 1861(kkk)(7)(C)(ii) of the Act applies for purposes of this measure for the REHQR Program.
Comment: A few commenters supported the removal of the measure and stated they remain committed to health equity and to operationalizing SDOH screening and intervention in the best manner for their facilities. These commenters mentioned experiencing declined readmissions and emergency department use through connecting patients to community-based services, but that the burden of reporting is significant, so they will continue referring patients voluntarily.
Response: We appreciate commenters' support and commitment to maintaining quality care for all patients.
Comment: Many commenters did not support CMS' proposal to remove the two SDOH measures from the Hospital OQR, REHQR, and ASCQR Programs. Many commenters described how SDOH significantly impacts health outcomes and the types of care and services patients may require in healthcare facilities. These commenters stated that screening for SDOH is fundamental to patient-centered care, including clinical outcomes, treatment adherence, and reducing preventable healthcare utilization (for example, emergency department visits and readmissions). A commenter stated that these SDOH measures intended to create a systematic structure and standardized measurement to support patients with complex needs across all settings, and their removal adds difficulty in scaling care transitions and integrating social care into clinical workflows. Another commenter asserted that ASCs are well-positioned for SDOH screening by serving as a strategic starting point for scaling upstream driver initiatives across the continuum of care.
Response: We acknowledge commenters' concerns and encourage hospitals, REHs, and ASCs to continue to close identified gaps in patient care. Removing these measures from the Hospital OQR, REHQR, and ASCQR Programs does not prevent facilities from measuring and addressing patients' social needs, as clinically appropriate. Further, these SDOH measures are only reported in aggregate and do not measure the extent to which providers are ultimately connecting patients with resources or services and whether patients are benefiting from these screenings.
Comment: Many commenters disagreed that removal of the SDOH measures would reduce burden. Many of these commenters stated that the ultimate cost savings arising from SDOH screening through improved chronic disease management and prevention of avoidable hospitalizations. A few commenters also asserted that facilities have already incurred the cost to set up the systems to collect this data, and that removal now would have minor impacts on costs. Several commenters stated that eliminating these measures without a transition plan could disrupt established care practices, undermine quality, harm partnerships with community-based organizations, and present ethical challenges. A commenter stated that the public deserves to know which facilities are taking a prevention-driven approach to health care.
Response: We thank commenters for their feedback. As previously mentioned, removal of these measures does not prevent hospitals and ASCs from measuring and addressing patients' social needs as clinically appropriate. We acknowledge that hospitals and ASCs may have expended resources to implement SDOH screenings; however, removing these measures at this time will alleviate additional burden with regard to data collection and submission requirements, especially with screening patients via manual processes and other manual collection and data storage mechanisms. We encourage hospitals and facilities to transition in a manner that aligns with existing workflows, operational practices, and community partnerships.
Comment: Several commenters suggested that CMS retain these measures and conduct listening sessions or publish an RFI to gather interested ( printed page 53923) parties' feedback to address burden. Several commenters requested that CMS allow for voluntary reporting of the SDOH measures or pause the measures to avoid disrupting ongoing efforts to collect social risk data. A few commenters suggested a period of confidential reporting to facilitate iterative improvement. A few commenters expressed the importance of identifying and documenting Z codes and noted the importance of SDOH-related screening for capturing applicable Z codes. A few commenters suggested stratifying performance reports based on SDOH-associated ICD-10 diagnoses or patient demographics. A commenter advised CMS to develop a claims-based approach to measure health disparities.
A few commenters recommended maintaining the current SDOH measures and developing an additional measure to encourage facilities to connect patients to community resources. Some commenters encouraged alignment with other initiatives, such as the National Committee for Quality Assurance's (NCQA's) Healthcare Effectiveness Data and Information Set (HEDIS) Social Needs Screening and Interventions (SNS-E) measure, Health Level 7 Gravity Project, and United States Core Data for Interoperability. Some commenters requested that CMS provide technical assistance in integrating screening into workflows and connecting patients with social needs to resources. A commenter recommended an incentive program for community-based organizations that face funding challenges in supporting an influx of demand as a result of SDOH screening.
Response: We appreciate the commenters' concerns and feedback regarding the importance of collecting SDOH data from patients and acknowledge that some patients may face challenges following discharge that may be related to SDOH. We recognize that some clinicians may find value in obtaining SDOH information as part of clinical decision making, such as discharge planning and patient care, and acknowledge feedback from some commenters stating that they value collecting this information. We agree that healthcare outcomes may be different for those experiencing unstable housing or food insecurity. Facilities may find ways to address these concerns in their workflow because they recognize the importance of these items and the removal of the SDOH measure requirements should not, in any way, preclude hospitals, REHs, or ASCs from collecting and using this information.
Comment: Many commenters stated that the SDOH measures align with CMS' broader goals, including ensuring high-quality healthcare for all patients and implementing the Make America Healthy Again initiative. In response to the request for comment, “Measure Concepts under Consideration for Future Years in the Hospital OQR, REHQR, and ASCQR Programs-Request for Information (RFI): Well-Being and Nutrition,” several commenters urged CMS to keep the SDOH measures and invest resources into improving the applicability and actionability of these measures as a way to improve well-being and nutrition.
Response: We appreciate commenters' support for the goals of the Make America Healthy Again initiative and the constructive role that quality measures can play in ensuring quality healthcare for all. Because we have determined that the cost of reporting on these measures outweighs the benefits of retaining them in these programs, it would place an undue burden on hospitals and ASCs to require reporting on these measures as we explore alternative approaches to implementing measures related to well-being and nutrition. We will consider the feedback commenters provided in future policymaking.
After consideration of the comments we received, we are finalizing our proposal to remove the Screening for Social Drivers of Health and Screen Positive Rate for Social Drivers of Health measures from the Hospital OQR, REHQR, and ASCQR Programs beginning with the CY 2025 reporting period.
D. Updates to the Extraordinary Circumstances Exception (ECE) Policy for the Hospital OQR, REHQR, and ASCQR Programs
1. Background
Under our current Extraordinary Circumstances Exception (ECE) regulations, we have granted exceptions to data submission deadlines and requirements for the Hospital OQR, REHQR, and ASCQR Programs in the event of extraordinary circumstances beyond the control of a hospital, REH, or ASC (42 CFR 419.46(e), 419.95(g), 416.310(d), respectively). Extraordinary circumstances may include, but are not limited to, natural disasters or systemic problems with data collection systems.[270] We refer readers to the CY 2022 OPPS/ASC final rule with comment period (86 FR 63873), the CY 2024 OPPS/ASC final rule with comment period (88 FR 82076), and the CY 2018 OPPS/ASC final rule with comment period (82 FR 52614 through 52614) for further background about the ECE policies for the Hospital OQR, REHQR, and ASCQR Programs, respectively. We also refer readers to the QualityNet website for program-specific requirements for submitting an ECE request.[271]
Our ECE policy provides flexibility for Hospital OQR, REHQR, and ASCQR Program participants toward meeting program requirements in the event of an extraordinary circumstance. For instance, we recognize that, in circumstances where a full exception is not applicable, it is beneficial for a hospital, REH, or ASC to report data later than the reporting deadline. Delayed reporting authorized under our ECE policy allows temporary relief for a hospital, REH, or ASC experiencing an extraordinary circumstance while preserving the benefits of data reporting, such as transparency and informed decision-making for beneficiaries and providers alike.
2. Update to the Extraordinary Circumstances Exception (ECE) Policy for the Hospital OQR, REHQR, and ASCQR Programs
In the CY 2026 OPPS/ASC proposed rule (90 FR 33756 through 33757), we proposed to update the current Hospital OQR, REHQR, and ASCQR Program ECE policies codified at 42 CFR 419.46(e); 419.95(g); and 416.310(d), respectively, to include extensions of time as a form of relief and to further clarify the policy. Specifically, we proposed updating the regulations at §§ 419.46(e)(1), 419.95(g)(1), and 416.310(d)(1) to state that CMS may grant an ECE with respect to reporting requirements in the event of an extraordinary circumstance—defined as an event beyond the control of a hospital, REH, or ASC (for example, a natural or man-made disaster such as a hurricane, tornado, earthquake, terrorist attack, or bombing)—that affected the ability of the hospital, REH, or ASC to comply with one or more applicable ( printed page 53924) reporting requirements with respect to a calendar year.
We proposed that the steps for requesting or granting an ECE would remain the same as the current ECE process, detailed by CMS at the QualityNet website or a successor website.[272] However, at §§ 419.46(e)(2)(i), 419.95(g)(2)(i), and 416.310(d)(2)(i), we proposed that a hospital, REH, or ASC, respectively, may request an ECE within 30 calendar days of the date that the extraordinary circumstance occurred. Our current policy allows a request within 90 days; this change was proposed toward aligning the Hospital OQR, REHQR, and ASCQR policy with CMS systems implementation requirements across all quality reporting programs. Under this proposed policy, we clarified that CMS would retain the authority to grant an ECE as a form of relief at any time after the extraordinary circumstance has occurred. For the Hospital OQR, REHQR, and ASCQR Programs, at §§ 419.46(e)(2)(ii), 419.95(g)(2)(ii), and 416.310(d)(2)(ii), respectively, we proposed that CMS would notify the requestor with a decision in writing. If CMS grants an ECE to the hospital, REH, or ASC, the written decision will specify whether the hospital, REH, or ASC is exempted from one or more reporting requirements or whether CMS has granted the hospital, REH, or ASC an extension of time to comply with one or more reporting requirements.
Additionally, at §§ 419.46(e)(3), 419.95(g)(3), and 416.310(d)(3), we proposed that we may grant an ECE to one or more hospitals, REHs, or ASCs that have not requested an ECE if we determine that: a systemic problem with a CMS data collection system directly impacted the ability of the hospital, REH, or ASC to comply with a quality data reporting requirement, or that an extraordinary circumstance has affected an entire region or locale. We further proposed that, as is the case under our current policy, any ECE granted would specify whether the affected hospitals, REHs, or ASCs are exempted from one or more reporting requirements or whether CMS has granted the hospital, REH, or ASC an extension of time to comply with one or more reporting requirements.
We invited public comments on these proposals.
Comment: Many commenters expressed support for CMS' proposals to update and codify the ECE policy across hospital, REH, and ASC quality programs. Many commenters appreciated CMS' efforts to codify its authority to grant reporting deadline extensions or exceptions in response to extraordinary circumstances, recognizing this flexibility as critical for hospitals, REHs, and ASCs facing natural disasters or other emergencies. A few commenters specifically supported the proposal to update and codify CMS' ability to grant ECEs to hospitals, REHs, and ASCs even if those facilities have not requested an exception. A commenter supported the proposal to allow hospitals, REHs, and ASCs 30 days to submit an ECE request.
Response: We thank the commenters for their support.
Comment: Several commenters cautioned CMS to avoid defaulting to extensions in cases where broader relief is warranted and ensure reporting extensions are not disproportionately utilized in place of exceptions. A few commenters also urged CMS to recognize that a mere extension is not always sufficient, as the reliability and integrity of data collected during extraordinary events may be compromised. A few commenters urged CMS to provide details on how the determination of an exception versus an extension will be made to ensure transparency. A commenter stated this transparency would allow hospitals to better prepare for response times and required resources based on whether they are likely to receive an exemption or an extension. A commenter requested that CMS consider granting full exceptions for cyberattacks, specifically, due to the amount of time that the systems would be offline.
Response: We appreciate commenters' concerns regarding the use of extensions for ECE requests. We note that we do not intend to replace exceptions with extensions and acknowledge that extensions are not always appropriate or operationally feasible. We will determine whether to grant an exception versus an extension using the same evaluation approach currently used in ECE determinations, on a case-by-case basis, based on the specific circumstances affecting the hospital, REH, or ASC, including circumstances impacting the operational feasibility of an extension.
Comment: Many commenters did not support the reduced timeframe for hospitals to request an ECE from the current 90-day period to 30 days following an extraordinary circumstance. A few commenters stated that the proposed 30-day window is insufficient for hospitals, REHs, or ASCs to respond to a crisis, assess the impact on data collection and systems, and submit a request for an exception. A few commenters mentioned previously experienced and potential future situations, such as severe flooding and ransomware attacks, where hospitals, REHs, and ASCs would be fully engaged in patient care and operational recovery, leaving little capacity to prioritize administrative tasks like ECE requests. A few commenters expressed concern that the reduced timeframe encourages hospitals, REHs, or ASCs to divert critical staff at a time they are needed most and would force them to prioritize paperwork over patient care, undermining the goals of the CMS quality reporting and value programs. A few commenters requested that CMS review ECE submissions to assess the feasibility for facilities to meet a 30-day response deadline and disclose its justification for reducing the window.
Many commenters urged CMS to retain the current 90-day window but would support 60 days to align with the IPPS and IPF PPS final rules. A few commenters requested CMS retain the 90-day window, despite alignment with the IPPS and IPF PPS final rules; these commenters stated that even 90 days can be an insufficient period of time due to the impacts of extraordinary circumstances on infrastructure and operations.
Response: We appreciate commenters' feedback regarding concerns about reducing the timeframe for hospitals, REHs, and ASCs to submit an ECE request. Due to concerns regarding a hospital's, REH's, or ASC's ability to assess the impact on quality data submissions and complete the necessary ECE request form within 30 days of the extraordinary circumstance, we are modifying the timeframe to allow for 60 days to submit an ECE request. We believe this timeframe will provide sufficient time for hospitals, REHs, and ASCs to assess the impact on quality reporting without disrupting operational and care needs. The 60-day period will also align with the same policy finalized for hospital inpatient providers and inpatient psychiatric facilities (IPF) in the FY 2026 IPPS/LTCH PPS final rule (90 FR 37026) and the FY 2026 IPF PPS final rule (90 FR 37661), respectively.
Comment: A commenter did not support the proposal that CMS may grant an ECE to one or more hospitals, REHs, or ASCs that have not requested ( printed page 53925) an ECE. This commenter expressed concern that granting wholesale exceptions for entire regions or locales is not in the best interest of beneficiaries or the general public who depend on access to quality reporting to assess the safety and quality of the care delivered. Another commenter expressed concern that less publicized extraordinary circumstances will not prompt CMS to issue an ECE without request, and that the process to submit an ECE request via the HQR portal, email, or secure fax may be unavailable if an emergency event severely disrupts operations for months.
Response: We appreciate commenters' concerns regarding our proposal to grant ECEs to hospitals, REHs, and ASCs that have not requested an ECE but were subject to an extraordinary circumstance affecting their region or locale. While we understand the value of quality reporting data to the public, we believe that granting ECEs during certain circumstances where entire regions or locales may be experiencing a collective disruption in normal operations would alleviate some of the immediate administrative burden of submitting an ECE request. In such cases, we would decide whether facilities are excepted from one or more reporting requirements or granted an extension of time so that we can ensure transparent reporting of reliable data. If a facility is not automatically granted an ECE due to an extraordinary circumstance, we encourage the hospital, REH, or ASC to follow the same approach currently used to submit an ECE request. We believe the modified 60-day timeframe will provide sufficient time for a facility to submit an ECE request without further disrupting operations. The 60-day period will also align with the same policy finalized for the Hospital IQR Program and IPFQR Program in the FY 2026 IPPS/LTCH PPS final rule (90 FR 37026) and the FY 2026 IPF PPS final rule (90 FR 37661), respectively.
After consideration of the public comments, we are finalizing our ECE proposals as proposed, except for the proposed 30-day deadline. In lieu of the 30-day deadline, we are finalizing an ECE request deadline of 60 days following an extraordinary circumstance. We are making conforming amendments to our regulation text at §§ 419.46(e), 419.95(g), and 416.310(d) for the Hospital OQR, REHQR, and ASCQR Programs, respectively, to reflect these policy changes.
XV. Hospital Outpatient Quality Reporting (OQR) Program
A. Background and History of the Hospital OQR Program
The Hospital Outpatient Quality Reporting (OQR) Program is a pay-for-reporting program intended to ensure transparency and quality of care furnished at hospital outpatient departments (HOPDs). Section 1833(t)(17)(A) of the Social Security Act (the Act) states that subsection (d) hospitals (as defined under section 1886(d)(1)(B) of the Act) that do not submit data required for measures selected with respect to such a year, in the form and manner required by the Secretary, will incur a 2.0 percentage point reduction to their annual Outpatient Department (OPD) fee schedule increase factor.
We refer readers to the CY 2011 OPPS/ASC final rule with comment period (75 FR 72064 through 72065) for a detailed discussion of the statutory history of the Hospital OQR Program. The Hospital OQR Program requirements are codified at 42 CFR 419.46. We also refer readers to the CMS website at https://www.cms.gov/medicare/quality/initiatives/hospital-quality-initiative/hospital-outpatient-quality-reporting-program for general background on the Hospital OQR Program, as well as the CMS QualityNet Hospital OQR website at https://qualitynet.cms.gov/outpatient for current program requirements and measure specifications.
B. Changes to the Hospital OQR Program Measure Set
In the CY 2026 OPPS/ASC proposed rule (90 FR 33757), we proposed to adopt the Emergency Care Access & Timeliness electronic clinical quality measure (eCQM) beginning, with voluntary reporting for the CY 2027 reporting period followed by mandatory reporting beginning with the CY 2028 reporting period/CY 2030 payment determination. In addition, we proposed to remove the Median Time from Emergency Department (ED) Arrival to ED Departure for Discharged ED Patients (Median Time for Discharged ED Patients) measure and the Left Without Being Seen measure, beginning with the CY 2028 reporting period/CY 2030 payment determination, if the Emergency Care Access & Timeliness eCQM is adopted into the program. We also proposed to modify the Excessive Radiation Dose or Inadequate Image Quality for Diagnostic Computed Tomography (CT) in Adults (Hospital Level—Outpatient) measure (Excessive Radiation eCQM) from mandatory reporting to voluntary reporting, beginning with the CY 2027 reporting period.
We refer readers to section XIV.C. of this final rule with comment period, Cross-Program Measures, where we discuss the removal of the following Hospital OQR Program measures: (1) COVID-19 Vaccination Coverage Among Healthcare Personnel (HCP) measure beginning with the CY 2024 reporting period/CY 2026 payment determination; (2) Hospital Commitment to Health Equity (HCHE) measure beginning with the CY 2025 reporting period/CY 2027 payment determination; (3) Screening for Social Drivers of Health (SDOH) measure beginning with the CY 2025 reporting period; and (4) Screen Positive Rate for SDOH measure beginning with the CY 2025 reporting period.
1. Adoption of the Emergency Care Access & Timeliness eCQM Beginning With Voluntary Reporting for the CY 2027 Reporting Period Followed by Mandatory Reporting Beginning With the CY 2028 Reporting Period/CY 2030 Payment Determination
a. Background
Occupancy and boarding rates in U.S. emergency departments (EDs) continue to worsen and exceed pre-pandemic levels.[273] ED boarding, defined as holding a patient in the ED after the patient is admitted or placed into observation status at a hospital, often occurs due to shortages of inpatient beds and staff and contributes to ED crowding, leading to safety risks for patients and stressful working conditions for healthcare personnel.[274] A recent report from the Agency for Healthcare Research and Quality (AHRQ) characterized patient ED boarding as a growing public health crisis and engaged interested parties to address the strain on the U.S. healthcare system.[275]
Recent studies indicate that delays in the timeliness of ED care are associated ( printed page 53926) with patient harm.[276 277] Long ED wait times are also one of the most cited reasons for patients leaving an ED without being evaluated by a clinician.[278] Increased ED length of stay (LOS) is also a strong predictor of poor timeliness of care and is significantly impacted by ED boarding. One study found that for every patient boarded, the median ED LOS for all admitted patients increased by at least 12 minutes.[279] Furthermore, ED boarding and crowding have been associated with poor patient outcomes, such as increased mortality,[280] delays in needed care,[281] and negative patient and staff experiences.[282 283] For instance, evidence shows that ED crowding can harm patients with sepsis by delaying administration of lifesaving intravenous (IV) fluids and antibiotics.[284]
Due to growing concerns about the quality and timeliness of care in the ED, as well as the burden associated with two chart-abstracted ED measures adopted in the Hospital OQR Program measure set, the Median Time for Discharged ED Patients measure and the Left Without Being Seen measure, CMS assessed additional ways to support efforts that reduce patient harm and improve outcomes for patients requiring emergency care.
b. Measure Overview
The Emergency Care Access & Timeliness eCQM [285] is specified for the hospital setting and calculates the proportion of four outcome metrics that quantify access to and timeliness of care in an ED setting against specified thresholds, including: (1) patient wait time—1 hour; (2) whether the patient left the ED without being evaluated; (3) patient boarding time in the ED (as defined by a Decision to Admit (order) to ED departure for admitted patients)—4 hours; and (4) patient ED LOS (time from ED arrival to ED physical departure, as defined by the ED departure timestamp)—8 hours. The Emergency Care Access & Timeliness eCQM provides HOPDs with data for each of these individual numerator components, which are described in greater detail in section XV.B.1.c. of this final rule with comment period.
The numerator components of the Emergency Care Access & Timeliness eCQM overlap with the patient population and measure specifications of two chart-abstracted measures in the Hospital OQR Program: (1) the Median Time for Discharged ED Patients measure, and (2) the Left Without Being Seen measure. The Median Time for Discharged ED Patients measure assesses the time patients spent in the ED before being sent home, also known as ED throughput. The Left Without Being Seen measure assesses the percentage of patients who leave the ED without being evaluated by a physician/advanced practice nurse/physician's assistant (physician/APN/PA). Numerator component (2) overlaps with the Left Without Being Seen patient population, and numerator component (4) overlaps with the Median Time for Discharged ED Patients measure. In addition to capturing the same data elements as the Median Time for Discharged ED Patients and Left Without Being Seen measures, the Emergency Care Access & Timeliness eCQM measures boarding time in the ED, numerator component (3), and time from arrival to placement in a treatment room, numerator component (1), which are not currently captured by any other measure in the Hospital OQR Program measure set.[286]
In the CY 2026 OPPS/ASC proposed rule (90 FR 33757 through 33762), we proposed that removal of two chart-abstracted measures in conjunction with the proposed adoption of the Emergency Care Access & Timeliness eCQM would reduce HOPD burden by requiring the reporting of one digital quality measure instead of two chart-abstracted measures. While the Median Time for Discharged ED Patients and the Left Without Being Seen measures require manual intervention to retrieve data from clinical documentation, the Emergency Care Access & Timeliness eCQM allows for automated extraction of patient-level data directly from the electronic health record (EHR). In the proposed rule (90 FR 33758), we acknowledged that updating EHRs with new measures requires some initial investment from hospitals, but in the long-term it would automate timely collection of more granular quality information. We referred readers to the eCQI Resource Center for general eCQM implementation guidance at https://ecqi.healthit.gov/oqr/ecqm-resources. We also refer readers to section XV.B.2. of this final rule with comment period for more information on these removals of the chart-abstracted measures.
For more information about the testing, feasibility, scientific acceptability, meaningfulness, and validity of the Emergency Care Access & Timeliness eCQM, we refer readers to https://p4qm.org/measures/4625e. ( printed page 53927)
c. Measure Calculation
The measure denominator includes all ED encounters associated with patients of all ages, for all-payers, during a 12-month period of performance. Patients can have multiple encounters during a period of performance, and each encounter is eligible to contribute to the calculation of the measure.[287]
The measure numerator includes any ED encounter in the denominator where the patient experiences any one of the following: (1) the patient waited longer than 1 hour after arrival to the ED to be placed in a treatment room or dedicated treatment area that allows for audiovisual privacy during history-taking and physical examination; (2) the patient left the ED without being evaluated; (3) the patient boarded in the ED for longer than 4 hours; and (4) the patient had an ED LOS of longer than 8 hours.[288] An encounter is considered part of the numerator if it includes any one of the four numerator events, with events not being mutually exclusive and each contributing only once to the numerator. ED encounters with ED observation stays [289] are excluded from components (3) and (4) but are included in the denominator. Patients who have a “decision to admit” after an ED observation stay remain excluded from criteria (3) calculations.[290]
These four outcomes were selected based on published literature demonstrating that each numerator component is associated with patient harm,[291] as well as input from clinical experts including ED experts and statistical and methodological experts and a Technical Expert Panel (TEP) that was convened by the measure developer.[292] A Patient and Family Engagement (PFE) Work Group provided feedback on experiences with emergency care, noting long wait times to be seen by a provider, long wait times to be transferred, and gaps in the discharge processes.
The numerator thresholds were developed according to evidence and consensus-based clinical guidelines for ED time thresholds, including guidelines developed by The Joint Commission (TJC), the American College of Emergency Physicians (ACEP), and the Emergency Department Benchmarking Alliance as well as input from a TEP, literature reviews, and environmental scans. For example, the 4-hour threshold for numerator component (3), boarding time, was developed according to recommendations from TJC and ACEP.[293 294]
Measure testing for the Emergency Care Access & Timeliness eCQM was conducted by the measure developer across 32 hospital-based EDs, representing a diverse mix of geographic regions, rurality, hospital size, teaching status, trauma level, and EHR vendors, demonstrating that the measure is reliable, valid, and feasible for all required data elements.[295] Measure testing results showed a wide range in overall scores, and across all strata, indicating variation in performance and implying room for quality improvement.[296]
The measure score is first calculated at the individual ED level as the proportion of ED encounters where any one of the four outcomes occurred. Raw measure scores are then standardized by ED case volume using z-scores. The z-score, or standard score, indicates how many standard deviations a data point is from the mean of a normal distribution. It is calculated by subtracting the mean from a data point, then dividing the result by the standard deviation. For the Emergency Care Access & Timeliness eCQM, a volume-adjusted z-score shows how an ED's performance compares to the average for similar-volume EDs, addressing differences in patient population in HOPDs and ensuring fair “like to like” comparisons between EDs of similar size. ED volume strata are defined in volume bands of 20,000 ED visits, and each ED is assigned to only one volume stratum. For CMS Certification Numbers (CCNs) with more than one ED, volume-adjusted z-scores are then combined as a weighted average for that CCN.[297]
The results of the Emergency Care Access & Timeliness eCQM are stratified into four groups, two by age (18 years and older, and under 18 years) and two by mental health diagnoses (with, and without).[298] The stratification of results by age and mental health diagnosis, as well as standardization of measure performance scores by volume, is sufficient to account for differences between hospitals without further need for risk adjustment.
For more detail on the measure specifications, we referred readers to the CMS QualityNet Hospital OQR Program website at https://qualitynet.cms.gov/outpatient, which also takes readers to the electronic specifications available at the eCQI Resource Center: https://ecqi.healthit.gov/ecqm/hosp-outpt/2027/cms1244v1.
d. Pre-Rulemaking Measure Review (PRMR)
As required under section 1890A of the Act, the Secretary must establish and follow a pre-rulemaking process for selection of quality and efficiency measures, including for the Hospital OQR Program. The pre-rulemaking process, which we refer to as the Pre-Rulemaking Measure Review (PRMR), includes a review of measures published on the publicly available “Measures Under Consideration List” (MUC List) by one of several committees convened by the consensus-based entity (CBE), with which we contract in accordance with section 1890 of the Act, for the purpose of providing interested parties input to the Secretary on the selection of quality and efficiency ( printed page 53928) measures under consideration for use in certain Medicare quality programs, including the Hospital OQR Program. We refer readers to the CY 2025 OPPS/ASC final rule with comment period (89 FR 94372) for details on the PRMR process, including the voting procedures used to reach consensus on measure recommendations. We described in the CY 2026 OPPS/ASC proposed rule that the PRMR Hospital Recommendation Group met on January 15 and 16, 2025, to review measures included by the Secretary on the publicly available 2024 MUC List, including the Emergency Care Access & Timeliness eCQM (MUC2024-075), and provided additional recommendations on the potential use of this measure.[299]
The voting results of the PRMR Hospital Recommendation Group for the proposed Emergency Care Access & Timeliness eCQM within the Hospital OQR Program were: 10 members recommended adopting the measure into the Hospital OQR Program; 10 members recommended adoption with conditions; 7 members voted not to recommend the measure for adoption. No voting category reached 75 percent or greater, including the combination of the recommend and the recommend with conditions categories and thus, the Hospital Recommendation Group did not reach consensus.[300]
The PRMR Hospital Recommendation Group noted in their deliberations that the measure will provide important insights into ED wait times which impact experience of care. The Group expressed concern that this measure may cause an increase in cost of care due to patients being transferred from the ED to observation.[301] While we acknowledged that patients transferred from the ED to observation may result in increased short-term costs due to additional monitoring and extended stays, the measure is designed to address significant issues surrounding the access to timely care which have been proven to reduce long-term costs.[302] Hospital Recommendation Group members also expressed concern about the lack of CBE endorsement. We noted that we submitted the Emergency Care Access & Timeliness eCQM for CBE endorsement for review in the Fall 2024 cycle and the CBE endorsed the measure with conditions on February 12, 2025.[303]
The Hospital Recommendation Group discussed conditions specific to the Hospital OQR Program, including changing the name of the measure to better reflect the measure's focus.[304] We agreed with this feedback and changed the name of the measure from the Emergency Care Capacity and Quality eCQM to Emergency Care Access & Timeliness eCQM. Group members also recommended refraining from including the Emergency Care Access & Timeliness eCQM in Overall Hospital Quality Star Ratings (Star Ratings) due to the possible duplication of data with existing measures. We noted that we proposed to remove two existing measures in the Hospital OQR Program, the Median Time for Discharged ED Patients and the Left Without Being Seen measures, to avoid duplicative data collection and reporting. We also noted that the Emergency Care Access & Timeliness measure would only be included in the Star Ratings calculation after the existing measures are removed. We refer readers to section XV.B.2. of this final rule with comment period for more information on the removal of the Median Time for Discharged ED Patients and the Left Without Being Seen measures.
The Hospital Recommendation Group also recommended revising the measure specifications to create separate measure components and explore alternative measures for patient boarding time and patient ED LOS. We acknowledged the Hospital Recommendation Group's concerns and noted that multiple TEPs and interested parties supported the inclusion of more than one numerator component as a strategy for internally balancing the measure and that the time thresholds for patient boarding time and ED LOS are based on more than a decade of consensus work. Lastly, Group members recommended stratifying the measure by factors such as care type, region, and hospital or trauma level designation. We emphasized that the approach to stratification by age and mental health diagnosis, as well as volume standardization of the measure performance scores, is sufficient to account for differences between hospitals without further need for additional stratification.[305]
e. Measure Endorsement
Section 1833(t)(17)(C)(i) of the Act provides that the Hospital OQR Program shall include measures that reflect consensus among affected parties and, to the extent feasible and practicable, shall include measures set forth by one or more national consensus-based entities. A TEP consisting of interested parties, experts, and consumer advocates contributed their input through the Emergency Care Access & Timeliness eCQM measure design process.[306] The Emergency Care Access & Timeliness eCQM was submitted to the CBE for endorsement review in the Fall 2024 cycle (CBE #4625e), and the CBE endorsed the measure with conditions for use in the Hospital OQR Program on February 12, 2025. The conditions include that the measure developer explore within 3 years: (1) the unintended consequences to patients and providers, including burden, by engaging with the patient community and accountable entities (for instance, qualitative assessments and empirical analyses); and (2) the data elements to identify and address where challenges may persist, including engaging accountable entities. We stated in the CY 2026 OPPS/ASC proposed rule that if the proposal to adopt the Emergency Care Access & Timeliness eCQM for the Hospital OQR Program is finalized, we would monitor the burden on patients and providers and identify areas where ( printed page 53929) challenges may persist as part of the standard measure maintenance (90 FR 33760).
f. Data Collection, Submission, and Reporting
The Emergency Care Access & Timeliness eCQM is specified in a standard electronic format, utilizing data extracted electronically from EHRs, with all data coming from defined fields in electronic sources. We noted in the CY 2026 OPPS/ASC proposed rule that eCQMs allow for retrieval of data directly from an EHR, reducing administrative burden on hospitals and minimizing errors due to manual abstraction of data.[307]
We proposed to adopt the Emergency Care Access & Timeliness eCQM beginning with voluntary reporting for the CY 2027 reporting period followed by mandatory reporting beginning with the CY 2028 reporting period/CY 2030 payment determination. We stated we believe this would provide HOPDs sufficient time to test and integrate the eCQM into existing clinical workflows. Additionally, limiting voluntary reporting to 1 year prioritizes addressing long ED wait times and ED boarding as well as removing two chart-abstracted measures from the Hospital OQR Program measure set to reduce HOPD burden. We refer readers to section XV.C.2. of this final rule with comment period for additional information on the Emergency Care Access & Timeliness eCQM form, manner, and timing of data submission and reporting requirements.
We refer readers to section XVI.B.1. of this final rule with comment period where we discuss the adoption of a similar measure for the Rural Emergency Hospital Quality Reporting Program.
We invited public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: Many commenters supported the adoption of the Emergency Care Access & Timeliness eCQM as proposed. These commenters emphasized the importance of measuring emergency room wait times, particularly boarding times. Many commenters stated that ED boarding is a public health emergency and national security and preparedness issue, as it endangers patient outcomes and contributes to ambulance diversion, adverse events, preventable errors, reduced patient satisfaction, ED violence, staff burnout, and higher health care costs. Many commenters stated that boarding reflects broader health system dysfunction and that valid and reliable measurement is essential to identifying, diagnosing, and pursuing strategies that can solve this issue. A few commenters supported CMS for moving to reinstate a boarding-related measure into its quality reporting programs following the previous removal of several measures related to this topic.
Response: We thank commenters for their support. We agree that measurement is essential to addressing the complex issue of ED boarding.
Comment: Several commenters supported the timeline for adoption of the Emergency Care Access & Timeliness eCQM into the Hospital OQR Program, believing the transition from voluntary reporting to mandatory reporting to be appropriate for a new eCQM. These commenters supported the transition for existing ED efficiency measures to the new Emergency Care Access & Timeliness eCQM and urged CMS to finalize the timeline as proposed.
Response: We thank the commenters for their support. The timeline for adoption of the Emergency Care Access & Timeliness eCQM, beginning with voluntary reporting for the CY 2027 reporting period followed by mandatory reporting beginning with the CY 2028 reporting period/CY 2030 payment determination, was developed to balance the immediacy of the ED boarding crisis while providing HOPDs sufficient time to test and integrate the eCQM into existing clinical workflows in preparation for reporting the measure. HOPDs would have approximately 25 months from the publication of this final rule with comment period until the start of the mandatory reporting period in January 2028, and approximately 3.5 years from the publication of this final rule with comment period until the submission deadline for the CY 2028 data in May 2029, to implement any workflow modifications or organizational updates necessary to successfully report on the measure.
Comment: Several commenters recommended publicly reporting the four individual numerator components as well as the overall measure score for the Emergency Care Access & Timeliness eCQM. Several commenters noted that data for the individual numerator components would provide more granular insights and improve hospitals' ability to target specific areas for improvement. A few commenters opposed the use of a composite measure, noting that it would mask performance on the four distinct numerator components.
Response: We agree with commenters that performance data for the four numerator components provides meaningful and actionable information for hospitals, patients, and other members of the public. If the Emergency Care Access & Timeliness eCQM is adopted into the Hospital OQR Program, we would publicly report the overall measure score and rates for the four numerator components once the measure becomes mandatory.
We note that the overall score for the Emergency Care Access & Timeliness eCQM represents the proportion of ED encounters in the denominator where the patient experiences at least one of the four numerator events: (1) the patient waited longer than 1 hour after arrival to the ED to be placed in a treatment room or dedicated treatment area that allows for audiovisual privacy during history-taking and physical examination; (2) the patient left the ED without being evaluated; (3) the patient boarded in the ED for longer than 4 hours; and (4) the patient had an ED LOS of longer than 8 hours.[308] We also note that the four numerator component rates are not blended or combined to obtain the overall score. That is, for each numerator component, the denominator includes all ED encounters associated with patients of all ages, for all-payers, during a 12-month period of performance, and the numerator includes the ED encounters in the denominator that met the criteria for the designated numerator event.
Comment: A few commenters requested clarification on how encounters that meet more than one numerator criteria would be included in the measure calculations. A commenter requested clarification on double counting, benchmarking, and reporting for patients with behavioral health needs.
Response: As specified, if a single ED encounter meets the criteria for more than one numerator event, the encounter would be included in each of the applicable component scores and once in the overall summary score regardless of how many numerator events it met. Because the overall measure score represents the proportion of ED visits where any component outcome occurred, the Emergency Care ( printed page 53930) Access & Timeliness eCQM avoids overlap and double counting that can skew measure results. For the request for clarification on the benchmarks for this measure, we believe the commenter is referring to the numerator thresholds. As discussed in the CY 2026 OPPS/ASC proposed rule, these thresholds were developed according to evidence and consensus-based clinical guidelines for ED time thresholds, including those from The Joint Commission (TJC) and the American College of Emergency Physicians (ACEP). For the request for clarification on reporting for patients with behavioral health needs, we reiterate that we will stratify the data for each of the numerator components and the overall summary score into four groups, two by age (18 years and older, and under 18 years) and two by mental health diagnoses (with, and without).[309]
Comment: A few commenters requested clarification of what constitutes a “treatment room or dedicated treatment area” in the first numerator component, with a commenter stating that some hospitals may interpret this strictly as a dedicated room, while others might consider curtained dividers as meeting the definition. A few commenters requested that CMS consider revising the language to focus on the initiation of treatment, regardless of where it occurs, to accommodate diverse ED layouts and workflows.
Response: We note that measure numerator component (1) does not specify a physical location; it evaluates whether the patient waited longer than 1 hour after arrival to the ED to be placed in a treatment room or dedicated treatment area that allows for audiovisual privacy during history-taking and physical examination. Based on this description, multiple settings, including both curtained dividers and a dedicated room, would qualify if there is audiovisual privacy. Spaces that do not provide both visual privacy and audio privacy sufficient to ensure patient dignity, confidentiality, and quality of care during history-taking and physical examination would not qualify. We believe this approach allows for flexibility so that diverse ED layouts and workflows can be accommodated when hospitals implement the measure while also ensuring that patients' privacy is respected.
Comment: A few commenters requested that CMS consider either defining exclusion criteria for complex ED encounters (for example, specialized care pathways, specialty consultations, forensic patients, or patients requiring advanced imaging) or adding exclusionary language for each of the numerator components. Another commenter requested exclusion criteria for multi-casualty events. According to commenters, these exclusions are necessary to avoid misclassifying high-quality, comprehensive care provided in a single ED visit as poor performance.
Response: Regarding commenters' requests for exclusion criteria, we note that the measure methodology, including consideration of potential exclusions, was informed by extensive literature review, empiric analyses, and feedback from the TEP, PFE Work Group, and the public comment period held during measure development. The measure does not have exclusions beyond observation stays for numerator component (3) and (4) because the numerator events included in the measure are considered access failures regardless of patient complexity or surge scenarios. We believe that excluding patients from the measure would violate its intent to ensure timely access to emergency care for all patients. As described below, we do not anticipate zero scores for all numerator components on every patient, and we believe the hospital comparison logic (such as using standardized z-scores to adjust for ED volume) adequately accounts for differences in performance among hospitals. We will monitor the measure during implementation to determine whether any additional exclusions are appropriate for measure incorporation in future re-specification.
We agree that some patients are clinically complex and require specialized, time-intensive services in the ED, and note that this was considered during measure development. We do not expect hospitals to achieve zero scores for all numerator components. We also note that the Hospital OQR Program is a pay-for-reporting program that penalizes hospitals for not submitting data rather than for performance on the measure. However, collecting and publicly reporting these data will allow for comparisons between hospitals that serve similar communities and support the development of targeted quality improvement strategies.
Comment: A few commenters noted that for small, rural hospitals, delays in the ability to transfer patients are frequently out of their control. A few commenters sought clarification on whether the measure includes transfer patients, with a commenter noting a distinction between patients who are transferred to another facility and patients being admitted to the same facility or discharged home.
Response: We recognize the complexity and interdependence of multiple systems when it comes to attribution of transfer cases. The decision to include transfer patients in the Emergency Care Access & Timeliness eCQM was carefully considered during measure development, particularly through input from the TEP. Capturing transfers in the measure numerator ensures that it is the responsibility of the transferring ED to proactively build processes that maintain care coordination to ensure timely transfer, which patients from the PFE Work Group expressed was important to them. Additionally, we believe including transfers in the numerator limits the incentive to inappropriately transfer patients to other facilities due to ED overcrowding.
In response to commenters' requests for clarification on whether the measure includes transfer patients, we clarify that transfer patients are included and assessed against all numerator criteria. To the extent that transfer patient encounters meet the threshold for each numerator component, they would be included in the measure. During measure testing, the proportion of total encounters with a final disposition of transfer was 2 percent. Therefore, we believe the inclusion of transfers will not have a significant impact on the reliability or validity of the measure.
We note that the two versions of the Emergency Care Access & Timeliness eCQM proposed for the Hospital OQR and REHQR Programs differ in numerator component (3), boarding. The Hospital OQR Program version of the measure captures inpatient boarding time, defined as the time from Decision to Admit (order) to ED departure. The REHQR Program version of the measure captures transfer boarding time in the ED because REHs do not provide inpatient services. We refer readers to section XVI.B.1 of this final rule with comment period for more information on the Emergency Care Access & Timeliness eCQM proposed for the REHQR Program.
We understand commenters' concerns that small and rural hospitals may face factors outside their control when transferring patients and note that we do not expect hospitals will achieve zero scores for all numerator components. However, collecting and publicly reporting these data is an important step towards addressing ED overcrowding ( printed page 53931) and barriers to access, which all parties involved during the measure development process agreed is critical.
Comment: Several commenters recommended refinements to the time thresholds established for the numerator components of the Emergency Care Access & Timeliness eCQM. A few commenters suggested using these timing thresholds as absolute maximum limits rather than as mean or median targets. A few commenters recommended that future performance targets should move towards shorter time periods as the quality gap closes. These commenters expressed concern about boarding times for high-risk groups, such as patients admitted to intensive care units and adults aged 65 and older. Another commenter expressed concern about the strain experienced by hospitals serving the most high-risk patients and recommended against setting targets hospitals may not be able to achieve. A few commenters stated that CMS had not provided clinical data to support the timing thresholds in the measure specifications. A few commenters noted that during peak seasonal spikes in patient volume, particularly during the winter months when influenza and COVID-19 surges occur, it would be unrealistic for hospitals to consistently meet the timing thresholds.
Response: We note that the timing thresholds for each numerator component are maximum limits rather than mean or median targets. These timing thresholds were developed according to evidence-based clinical guidelines, including those developed by The Joint Commission, American College of Emergency Physicians, and the Emergency Department Benchmarking Alliance,[310 311] as well as from input from a TEP, literature reviews, and environmental scans. We acknowledge commenters' concerns that the timing thresholds may be challenging for hospitals serving high-risk patient populations to meet. During measure development, the developer considered ways to address high-risk groups based on risk factor categories, including age, mental health diagnosis, comorbidity, complexity, race, and income.[312] The measure's approach, involving stratification by age and mental health diagnosis and volume standardization to address case mix differences across EDs, is intended to account for differences between hospitals without requiring additional risk adjustment.
We acknowledge that ED volumes fluctuate by seasonal trends. To account for these variations, a full calendar year was selected as the measure's performance period, to allow time to capture seasonal variations in the reported outcomes and allow hospitals and health systems sufficient time to analyze data and implement quality improvement efforts. During the public comment period held during measurement development, commenters strongly agreed with this approach and appreciated its alignment with other CMS reporting periods.[313] While we acknowledge that certain seasons could experience peak volumes, we proposed to require hospitals to report all four calendar quarters (one full calendar year) of data beginning with the CY 2028 reporting period/CY 2030 payment determination (90 FR 33766). The measure would be reported annually, not seasonally, for public reporting purposes. Furthermore, we do not expect the measure rate to be zero.
We also note that hospitals should prepare for predictable volume increases, including ensuring sufficient staffing, as part of emergency preparedness, and note that the measure can help hospitals monitor the adequacy of their staffing plans for anticipated surges. We will consider recommendations regarding the appropriateness of the timing thresholds as part of our measure monitoring and evaluation program.
Comment: A few commenters recommended that CMS assign more weight to numerator component (3) (that is, the patient boarded in the ED for longer than 4 hours), to reflect its importance and the likelihood that it may affect other ED outcomes. A few commenters stated that applying volume standardization for numerator component (3) would be inappropriate because all hospitals must minimize boarding regardless of hospital size.
Response: We appreciate commenters' recommendation to assign more weight to numerator component (3). As previously stated, the overall score for the Emergency Care Access & Timeliness eCQM represents the proportion of ED encounters associated with patients of all ages, for all-payers, that experience at least one of the four numerator events during a 12-month period of performance: (1) the patient waited longer than 1 hour after arrival to the ED to be placed in a treatment room or dedicated treatment area that allows for audiovisual privacy during history-taking and physical examination; (2) the patient left the ED without being evaluated; (3) the patient boarded in the ED for longer than 4 hours; and (4) the patient had an ED LOS of longer than 8 hours.[314] In addition to the overall score, the Emergency Care Access & Timeliness eCQM provides HOPDs with data for each of the individual numerator components; however, the rates for these components are not added or combined to obtain the overall score as they are not mutually exclusive. Therefore, it is not feasible to assign a heavier weight to numerator component (3) without changing the underlying meaning of the measure, or its capacity to provide information that is interpretable or actionable for HOPDs. We recognize, however, that ED boarding is an important issue and we will evaluate the burden and feasibility of the commenters' recommendation as more information on this measure becomes available.
We also acknowledge commenters' concerns about applying volume standardization to numerator component (3). As specified, volume standardization for the Emergency Care Access & Timeliness eCQM is applied after the overall score of the four numerator components is calculated. Since the four numerator components are not mutually exclusive, applying volume standardization to each individual numerator outcome could lead to calculation challenges and undesirable statistical distortions when calculating the overall score. As discussed above, the evidence-based clinical guidelines, input from the TEP, literature reviews, and environmental scans did not provide a basis to support using a different threshold for numerator component (3) based on hospital volume or other factors, since prolonged boarding is associated with patient safety risks, regardless of hospital volume. The application of volume standardization occurs after ( printed page 53932) calculation of the overall score, which allows comparison of hospitals of similar sizes without diminishing the importance of any numerator component.
Comment: A few commenters recommended additional stratification, such as hospital designation or status, hospital-based ED vs. free-standing ED designations, payer type, race, ethnicity, primary language, or hospital teaching status. A commenter recommended replacing the denominator definition of “cases with a mental health diagnosis” with “patients awaiting a psychiatric bed” for the mental health stratification, as the need to locate a psychiatric bed is a well-documented and significant driver of prolonged ED stays.
Response: We note that many of the factors recommended are provider-level characteristics, and as such are not feasible to use as a patient characteristic for stratification. Additionally, the measure collects payer type, race, ethnicity, and sex as supplemental data elements. However, we thank the commenters for their suggestions on additional stratifications for public reporting of the measure and will consider utilizing this information in support of additional future stratifications in the future.
We appreciate the commenter's recommendation regarding the denominator definition for mental health stratification, and note that while psychiatric bed delays were considered, the measure consistently uses a diagnostic based approach when identifying patients for the mental health cohorts. For the denominator definition for the mental health strata, we note that the principal diagnosis (first listed diagnosis at ED discharge), and not the patient disposition, would be used to define strata inclusion. We note that this approach is consistent with the Median Time for Discharged ED Patients measure that we have previously reported in our quality reporting programs.[315]
Comment: A few commenters recommended stratification by ED volume, with a commenter suggesting stratifying ED volume as small, medium, or large, to enable meaningful comparisons across hospitals with varying patient volume and resource levels.
Response: For the Emergency Care Access & Timeliness eCQM, volume standardization is applied to the overall score. Our intention is to improve ED efficiency regardless of hospital size. During measure development, volume bands of 20,000 visits were chosen based on existing literature and actual use within the ED measurement and quality community, such as the ED Benchmarking Alliance.[316] The measure uses volume standardization to address the case mix differences between EDs,[317] and volume standardization offers the simplest approach, as approved by the industry, without the complexities and unintended consequence of statistical modeling. This is aligned with ACEP's measure approach in the Merit-Based Incentive Payment System (MIPS) to measuring patient flow in the ED setting.[318 319] Through volume standardization we allow comparisons across hospitals of all sizes, while accounting for potential variations associated with patient volume, such that large volume EDs will be compared to large volume EDs, while medium and smaller volume EDs will likewise be compared to EDs of similar size.[320] We appreciate commenters' recommendation regarding stratification based on ED volume and note that ED volume is a facility-level characteristic, rather than a patient-case characteristic, although we will take this into consideration as we monitor implementation of this measure and consider any potential future refinements.
Comment: Several commenters expressed concern that factors outside of a hospital's control, such as shortages of beds and providers throughout the healthcare system, can negatively affect ED efficiency. These commenters encouraged CMS to incorporate robust risk adjustment to account for patient case mix and volumes and the availability of post-acute care services within a hospital's service area. A few commenters expressed concern that the Emergency Care Access & Timeliness eCQM does not distinguish between delays that are within a hospital's control and those driven by systemic capacity issues. A few commenters also recommended reporting even more detailed performance data for numerator events, such as performance metrics for the 90th percentile for the timed numerator components.
Response: Although we acknowledge that ED efficiency could be affected by multiple factors, some of which are outside the control of hospitals and health systems, measurement is essential for tracking and addressing these complex systemic issues and their downstream effects. We believe that many hospitals face such concerns and that timely care is a critical aspect of quality of care, directly impacting patient outcomes, particularly for an ED encounter. Collecting and publicly reporting these data will allow comparisons between hospitals that serve similar communities and support the development of targeted quality improvement strategies.
For the risk-adjustment, we note that the measure is not risk-adjusted because the numerator events are considered access failures regardless of patient acuity or clinical comorbidity. This approach focuses on a clear threshold that reflects operational and patient-experience standards for accessible and timely care. We recognize that bed and provider shortages or limited post-acute options can affect ED times but reiterate that the measure is meant to highlight and provide insight to general patterns in access and timeliness. Facilities may supplement this measure internally with percentile-based analyses as part of their ongoing performance improvement efforts. We maintain that the stratification of results by age and mental health diagnosis, as well as standardization of measure performance scores by ED volume, is sufficient to account for differences between hospitals without further need for risk adjustment or additional performance data detail.
Comment: A few commenters expressed concern with the potential for unintended consequences related to the current structure of the measure. The commenters expressed concern that the measure's focus on time thresholds could put undue pressure on ED staff to rush, and that emphasizing speed over quality could compromise patient care to avoid financial penalties. A commenter expressed support for the exclusions of observation stays from numerator components (3) and (4) but ( printed page 53933) sought clarity regarding cases where observation stays lead to inpatient admission. Another commenter noted that the measure could potentially lead to the inappropriate use of “observation” status to circumvent the measure's intent.
Response: We note that the Hospital OQR Program is a pay-for-reporting program, and hospitals are only penalized for not submitting data rather than for their performance on the measure. We also note that because the measure does not track patient status after leaving the emergency department, a patient that is admitted as an inpatient from observation status does not impact this measure. For the concern that some hospitals may inappropriately use observation status to circumvent the measure's intent, we note that clinicians are responsible for adhering to all standards for patient care, including using appropriate clinical decision making to determine which patients should be placed in observation status. We will consider monitoring observation status volumes to determine whether hospitals are increasing use of this status concurrent with measure adoption.
Comment: A few commenters expressed concern about the lack of evidence-based guidelines on how to improve performance on the measure. A few commenters advised CMS to work with interested parties in the hospital community to investigate effective solutions to address the underlying causes of ED boarding instead of implementing a generalized accountability measure.
Response: We refer readers to Table S1 in the supplemental attachment (section 7.1) for the measure on the Partnership for Quality Measurement website.[321] We note that this table details interventions that can improve each of the four numerator components. We also believe that data from this measure could be used as an evidence base to refine interventions and develop additional interventions to improve ED efficiency, for which we will continue to engage with patients, providers, and other interested parties in the hospital community.
Comment: A few commenters stated that measure performance for hospitals caring for larger proportions of Medicare Advantage patients may be affected by delays while waiting for insurance approval for patient admissions or transfers.
Response: We acknowledge that the prior authorization process used by commercial health plans could lead to delays in care or denials in coverage. We note that the Emergency Care Access & Timeliness eCQM includes all payers, which mitigates the potential for national variations in MA coverage to influence measure performance. Additionally, in measure testing the developer examined differences in measure score by patients' payer type composition (that is, proportion of patients with Medicaid, proportion of patients with Private/Other, and proportion of patients with Medicare) and found the strength of the relationship was weak to moderate and not statistically significant across all three of these compositional characteristics.
Comment: A commenter sought CMS guidance on its plans to ensure consistency across the Medicare Shared Savings Program (MSSP) in applying the revised definitions, providing updated technical specifications and training resources, and monitoring the impact of these changes on MSSP performance scores.
Response: We note that this measure is proposed for the Hospital OQR Program and does not affect current or future requirements for the MSSP, which does not currently include any ED boarding measures.
Comment: A few commenters requested excluding this measure from Overall Hospital Star Ratings and performance-based quality reporting programs.
Response: We have only proposed the Emergency Care Access & Timeliness eCQM for adoption in the Hospital OQR Program at this time. If we determine that it is appropriate for inclusion in the Overall Hospital Star Ratings or performance-based quality reporting programs in the future, we would propose it through notice and comment rulemaking.
Comment: Many commenters requested at least 1 or 2 more years of voluntary reporting to give hospitals, especially small and rural hospitals, adequate time to address processes and systems. Specifically, commenters stated that they would need additional time to map the necessary data elements within their EHR system, evaluate the validity of the resulting data, and adjust clinical workflows. A few commenters described their experiences implementing the STEMI (OP-40) eCQM and stated that an early start to mandatory reporting created an unnecessary burden.
Response: We understand commenters' concerns regarding adequate time to update systems and processes to prepare to report the Emergency Care Access & Timeliness eCQM. However, we sought to balance this concern with the urgency of addressing the ED boarding crisis and decided to propose one year of voluntary reporting. HOPDs will have approximately 25 months from the publication of this final rule with comment period until the start of the mandatory reporting period in January 2028, and approximately 3 and a half years from the publication of this final rule until the submission deadline for the CY 2028 data in May 2029. We note this eCQM was developed with the goal of minimizing implementation burden by using data elements that are already captured in EHRs in structured fields. In addition, replacing the current Median Time for Discharged ED Patients and LWBS measures (which require manual abstraction) with the Emergency Care Access & Timeliness eCQM will alleviate burden for hospitals.
Comment: A few commenters requested an indefinite period of voluntary reporting to allow the agency to determine if the measure provides useful data for the public and for hospital quality improvement. A commenter recommended that after voluntary reporting, there should be a period of mandatory reporting that is not publicly reported.
Response: We note that this measure was developed with input from the public regarding its importance. Specifically, these four outcomes were selected based on published literature demonstrating that each numerator component is associated with patient harm,[322] as well as input from clinical experts including ED, statistical, and methodological experts and a TEP that was convened by the measure developer.[323] A PFE Work Group provided feedback on experiences with emergency care, noting long wait times to be seen by a provider, long wait times to be transferred, and gaps in the discharge processes. This demonstrates that the measure would provide useful data for the public and for hospital quality improvement. We strive for transparency in our quality reporting and value-based payment programs, which includes making data available to the public at the first opportunity. We would not publicly report data during ( printed page 53934) the voluntary reporting period to allow time to confirm that publicly reported data are accurate and complete but will begin to publicly report these data beginning with the first mandatory reporting period consistent with our commitment to transparency.
After consideration of the public comments we received, we are finalizing our proposal to adopt the Emergency Care Access & Timeliness eCQM into the Hospital OQR Program, beginning with voluntary reporting in the CY 2027 reporting period followed by mandatory reporting beginning with the CY 2028 reporting period impacting CY 2030 payment determination. As stated previously, because we are finalizing adoption of the Emergency Care Access & Timeliness eCQM, we will publicly report the overall measure score and rates for the four numerator components, as well as the criteria-specific results regarding the age and mental health strata once the measure becomes mandatory.
2. Removals of the Median Time From ED Arrival to ED Departure for Discharged ED Patients (Median Time for Discharged ED Patients) Measure and the Left Without Being Seen Measure Beginning With the CY 2028 Reporting Period/CY 2030 Payment Determination
In the CY 2026 OPPS/ASC proposed rule (90 FR 33761), we proposed that the Emergency Care Access & Timeliness eCQM would serve as a replacement for two existing chart-abstracted measures in the Hospital OQR Program.
The Median Time for Discharged ED Patients measure (75 FR 72086) and the Left Without Being Seen measure (75 FR 72088 through 72089) were adopted in the CY 2011 OPPS/ASC final rule with comment period to promote transparency, improve patient care and access to EDs, and reduce avoidable delays in the emergency care setting. The Median Time for Discharged ED Patients measure assesses the time patients spent in the ED before being sent home, also known as ED throughput. The Left Without Being Seen measure assesses the percentage of patients who leave the ED without being evaluated by a physician/advanced practice nurse/physician's assistant (physician/APN/PA). Both measures are chart-abstracted, requiring human review and manual intervention to extract data elements from clinical documentation.
In the CY 2024 OPPS/ASC final rule with comment period (88 FR 81961), we did not finalize our proposal to remove the Left Without Being Seen measure due in part to public comments emphasizing the importance of the measure in addressing ED overcrowding and boarding. We stated our intention to identify a more granular measure that could replace the Left Without Being Seen measure, which can now be achieved through the adoption of the Emergency Care Access & Timeliness eCQM. We note that Hospital OQR Program measure specifications can be found at https://qualitynet.cms.gov/outpatient.
As stated in section XV.B.1. of this final rule with comment period, the Emergency Care Access & Timeliness eCQM is specified for the hospital setting and calculates the proportion of four outcome metrics that quantify access to and timeliness of care in an ED setting against specified thresholds. The numerator components of the Emergency Care Access & Timeliness eCQM overlap with data elements of the Median Time for Discharged ED Patients and the Left Without Being Seen measures. The numerator of the Emergency Care Access & Timeliness eCQM is comprised of any ED visit in the denominator where the patient experiences any one of the following: (1) waited longer than 1 hour to be placed in a treatment room or a dedicated treatment area that allows for audiovisual privacy history-taking and physical examination; (2) left the ED without being evaluated by a physician/advanced practice nurse/physician's assistant; (3) boarded (defined as time from a Decision to Admit (order) to ED departure for admitted patients) for longer than 4 hours; or (4) had an ED LOS (time from ED arrival to ED physical departure as defined by the ED departure timestamp) of longer than 8 hours. Numerator component (2) overlaps with the Left Without Being Seen patient population and numerator component (4) overlaps with the Median Time for Discharged ED Patients measure. The Emergency Care Access & Timeliness eCQM also incorporates additional metrics to enhance its comprehensiveness and analytic value, including boarding time in the ED, numerator component (3), and time from arrival to placement in a treatment room, numerator component (1), which are not currently captured by any other measure in the Hospital OQR Program.[324]
The Emergency Care Access & Timeliness eCQM therefore provides an alternative approach to quality measurement used to address ED boarding and barriers to emergency care by capturing multiple components of quality and capacity. In addition, the Emergency Care Access & Timeliness eCQM allows for retrieval of patient-level data directly from the EHR. As a result, the Emergency Care Access & Timeliness eCQM, along with our previously adopted eCQMs, advances the Hospital OQR Program toward the use of EHR data for quality measurement, leading to more accurate quality data as well as reduced burden for providers. The adoption of the Emergency Care Access & Timeliness eCQM would allow us to employ a more precise assessment of the timeliness and appropriateness of ED visits and to provide additional information important to patients and hospitals on ED boarding and ED LOS.
Our measure removal policy, codified at 42 CFR 419.46(i)(3), identifies eight factors CMS considers in the removal of quality measures. Removal Factor 4, described at § 419.46(i)(3)(i)(D), is the availability of a more broadly applicable (across settings, populations, or conditions) measure for the topic. Compared to the Median Time for Discharged ED Patients measure and the Left Without Being Seen measure, the Emergency Care Access & Timeliness eCQM is a more broadly applicable measure for the topic. We therefore proposed that, if the Emergency Care Access & Timeliness eCQM is adopted in the Hospital OQR Program, we would remove the Median Time for Discharged ED Patients measure and the Left Without Being Seen measure under removal Factor 4. We proposed that these measure removals would begin with the CY 2028 reporting period/CY 2030 payment determination, when reporting for the Emergency Care Access & Timeliness eCQM becomes mandatory.
We invited public comments on these proposals.
Comment: Many commenters supported the removal of the Median Time for Discharged ED Patients and LWBS measures. Several commenters supported the removal of these measures upon the adoption of the Emergency Care Access & Timeliness eCQM, noting that the eCQM offers a more standardized and improved approach to assessing ED throughput. A commenter supported the removal of the Median Time for Discharged ED Patients and LWBS measures citing that adoption of the Emergency Care Access & Timeliness eCQM would better align with existing technology investments and quality improvement strategy. ( printed page 53935)
Response: We thank the commenters for their support.
Comment: Several commenters supported the replacement of the Median Time for Discharged ED Patients and LWBS measures with the Emergency Care Access & Timeliness eCQM because electronic data sources reduce administrative burden and enhance real time data availability. A few commenters also supported the removal of the Median Time for Discharged ED Patients and LWBS measures to avoid duplicative reporting requirements.
Response: We thank commenters for their support and agree that the removal of the Median Time for Discharged ED Patients and LWBS measures will reduce administrative burden and prevent duplicative reporting.
Comment: A few comments supported the removal of Median Time for Discharged ED Patients and LWBS measures because the measures are no longer endorsed by the CBE. A few commenters supported the removal of the measures because they lack sufficient evidence demonstrating a link to improved patient outcomes. A commenter stated that the measures should be removed even if the Emergency Care Access & Timeliness eCQM is not adopted into the Hospital OQR Program due to concerns regarding the measures' lack of CBE endorsement and poor evidence-base.
Response: We thank the commenters for their support. We note that the Median Time for Discharged ED Patients and LWBS measures went through the measure development lifecycle outlined at the CMS Measures Management System website, which includes measure testing and reliability analysis. Further, section 1833(t)(17) of the Act does not require each measure adopted for the Hospital OQR Program to be CBE-endorsed. Section 1833(t)(17)(C)(i) of the Act provides that the Hospital OQR Program shall, to the extent feasible and practicable, include measures set forth by one or more national consensus building entities. Section 1833(t)(17)(C)(i) of the Act also requires measures developed for the Hospital OQR Program to reflect consensus among affected parties, which may be reflected in ways other than CBE endorsement. We note that the adoption of the Emergency Care Access & Timeliness eCQM is being finalized to replace the existing ED quality measures in the Hospital OQR Program in section XIV.B.1. of this final rule with comment period. This eCQM was submitted to the CBE for endorsement in the Fall 2024 cycle (CBE #4625e) and the CBE endorsed the measure with conditions on February 12, 2025. For more information on CBE endorsement, please refer to section XIV.B.1.e. of this final rule with comment period.
For evidence of impact on patient outcomes, we agree with commenters that it is critical to ensure that quality measures are supported by sufficient evidence. While the Median Time for Discharged ED Patients and LWBS measures have provided useful information on ED throughput, the Emergency Care Access & Timeliness eCQM offers a more robust connection to patient access and safety, as well as to outcome improvement. Therefore, we are finalizing our proposal to adopt the Emergency Care Access & Timeliness eCQM.
Comment: A commenter supported the removal of the Median Time for Discharged ED Patients measure and recommended removing the measure before the CY 2028 reporting period/CY 2030 payment determination.
Response: We acknowledge the commenter's recommendation for earlier removal of the measure but note that the proposed adoption of the Emergency Care Access & Timeliness eCQM begins with voluntary reporting for the CY 2027 reporting period, followed by mandatory reporting beginning with the CY 2028 reporting period/CY 2030 payment determination. Given the importance of ED throughput and its direct impact on patient care, it is essential to maintain mandatory measures within the Hospital OQR Program to ensure continued and reliable assessment of ED performance. This current timeline ensures that there will not be a gap in quality measurement for ED throughput, as there will be a year of overlap between the existing ED throughput measures and the voluntary reporting period for the new eCQM.
Comment: A commenter did not support the removal of the LWBS measure stating that the measure is an indication of ED workload and crowding.
Response: The Emergency Care Access & Timeliness eCQM overlaps with the LWBS measure collecting data on ED workload and crowding for the Hospital OQR Program. We refer readers to section XIV.B.1.b. of this final rule with comment period for additional information on the Emergency Care Access & Timeliness eCQM measure specifications.
After consideration of the comments received, we are finalizing our proposals to remove the Median Time for Discharged ED Patients and LWBS measures from the Hospital OQR Program beginning with the CY 2028 reporting period/CY 2030 payment determination.
3. Modify the Excessive Radiation Dose or Inadequate Image Quality for Diagnostic Computed Tomography (CT) in Adults (Hospital Level—Outpatient) Measure (Excessive Radiation eCQM) From Mandatory Reporting Beginning With the CY 2027 Reporting Period/CY 2029 Payment Determination To Continue Voluntary Reporting in the CY 2027 Reporting Period and Subsequent Years
In the CY 2024 OPPS/ASC final rule with comment period, we finalized the adoption of the Excessive Radiation eCQM in the Hospital OQR Program measure set, with voluntary reporting beginning in the CY 2025 reporting period followed by mandatory reporting beginning with the CY 2027 reporting period/CY 2029 payment determination, one year later than originally proposed (88 FR 81992). We explained our delay in implementing mandatory reporting of the Excessive Radiation eCQM was in response to commenters' concerns regarding the burden associated with implementing the eCQM.
In the CY 2026 OPPS/ASC proposed rule, we proposed to modify the reporting requirements for the Excessive Radiation eCQM in the Hospital OQR Program by maintaining voluntary reporting instead of transitioning to mandatory reporting of the measure, beginning with the CY 2027 reporting period (90 FR 33762). Our proposal to maintain indefinite voluntary reporting of this measure arose from continued feedback expressing concerns about the complex interfaces necessary to develop, maintain, and report the Excessive Radiation eCQM, including the financial burden and operational feasibility needed to translate CT radiology data into standardized eCQM-consumable data used by the measure. In January 2025, we issued a notice to clarify that hospitals and clinicians who choose to report this eCQM can use any vendor's translation software to calculate this measure,[325] consistent with the measure's specifications, and stated our intent to monitor measure results to ensure that all reported data for the Excessive Radiation eCQM are both reliable and valid.[326]
( printed page 53936)We stated that the modification from mandatory to voluntary reporting of the Excessive Radiation eCQM, beginning with the CY 2027 reporting period, would allow HOPDs additional time to integrate, adequately test, and gain experience with implementing the eCQM. This modification would also provide CMS with additional time to monitor implementation progress, including data collection burden and response rates. We stated that we will continue to consider feedback regarding this measure and may propose additional changes in future rulemaking.
We invited public comments on this proposal.
Comment: Many commenters supported the proposed change to maintain voluntary reporting for the Excessive Radiation eCQM. These commenters expressed concern that calculating and reporting the measure creates significant administrative and financial burden and diverts limited resources from patient care without providing commensurate value or improving patient safety. Commenters noted the measure's complexity and reporting requirements create a regulatory burden for which hospitals must divert resources in order to come into compliance. Many commenters noted that the expenses associated with implementing the measure, such as labor and interface costs, impose resource constraints on hospitals that make it difficult to collect and leverage data gained from the measure. Additionally, commenters noted that these expenses are not negated by the software available for free from the measure steward, as the measure requires working with a third-party vendor. Many of these commenters stated that maintaining voluntary reporting appropriately acknowledges the ongoing concerns around provider burden while still allowing hospitals to report data as they are able and avoiding punitive consequences for non-compliance.
Commenters also cited the technical and operational challenges of implementing the Excessive Radiation eCQM as a rationale for supporting voluntary reporting. Several commenters noted the limited health IT infrastructure available to report the measure, particularly for smaller and rural hospitals, which presents significant barriers to implementation. Many commenters noted the measure requires complex integrations across multiple hospital information systems, including Picture Archiving and Communication Systems (PACS), Radiology Information Systems (RIS), CT consoles, and EHRs, and that much of the necessary data reside in imaging software rather than outpatient EMR systems. Several of these commenters stated that extended voluntary reporting is warranted to allow hospitals time to develop the new processes and data extraction methods needed to capture and report this information effectively. Many commenters appreciated the fact that maintaining voluntary reporting allows hospitals time to improve imaging practices. A commenter urged CMS to work closely with hospitals that voluntarily report the measure to determine if the measure is feasible for broader implementation across CMS quality reporting programs. A commenter recommended that CMS align the reporting requirements for the measure across the Hospital OQR and Inpatient Quality Reporting (IQR) Programs to ensure consistency.
Response: We thank the commenters for their support. While we continue to believe in the validity and importance of this measure, and the clinical significance of measuring radiation safety, we acknowledge the current challenges that HOPDs face with respect to the measure's implementation that could necessitate a longer implementation timeframe. We thank the commenter for their recommendation to align reporting requirements across the Hospital OQR and Hospital Inpatient Quality Reporting (IQR) Programs. We will continue to monitor progress during the voluntary reporting period for the Hospital OQR Program, as well as the Hospital IQR Program, where this measure is included on the list of eCQMs from which hospitals may self-select measures to report. We will also continue to engage with hospitals, professional organizations, and health IT interested parties to assess feasibility, interoperability, and opportunities to refine the measure and related implementation pathways. Any further changes to the reporting requirements for this measure would be made in notice and comment rulemaking so that interested parties can prepare. We encourage hospitals to use the voluntary reporting period to improve imaging practices and develop the new processes and data extraction methods needed to capture and report data for this measure effectively, in preparation for future implementation.
Comment: Many commenters expressed concern that the measure relies on a proprietary software platform developed by a single for-profit vendor that also serves as the measure steward. Many commenters stated that this structure creates a potential conflict of interest and could limit transparency, competition, and public trust. Several commenters questioned the vendor's limited experience and technical capacity to oversee the measure, noting issues such as slow responsiveness, nonstandard contracting terms, and inadequate solutions to safeguard patient data. Several commenters raised concerns regarding lack of transparency in the underlying specifications and algorithms, and reliance on non-standardized metrics. Several commenters noted concerns regarding potential privacy and cybersecurity risks associated with transmitting PHI to third-party systems. A commenter highlighted that many current CT systems do not export data in a format that the software can reliably interpret, further complicating implementation.
Due to these concerns, many commenters urged CMS to ensure that any future mandatory implementation of this measure be supported by independent validation, peer-reviewed evidence, and the availability of multiple, vendor-neutral pathways for reporting and compliance. A commenter recommended that CMS explore alternative reporting mechanisms beyond the current single software platform and prioritize development of additional imaging quality measures, including those that recognize the use of emerging technologies such as artificial intelligence, to address existing gaps in diagnostic imaging quality and patient safety. A commenter also recommended that CMS work with the Assistant Secretary for Technology Policy/Office of the National Coordinator for Health Information Technology (ASTP) to address the underlying challenges of this measure to ensure that imaging data can be shared more effectively and broadly.
Response: We acknowledge that the Alara Imaging software for CMS Measure Compliance is proprietary. Regarding commenters' concerns about a conflict of interest, we note that the measure steward makes this software available without charge. In addition, the measure steward has successfully deployed its software across a diverse range of several hundred clinical settings with varied EHR, Picture Archiving and Communication System (PACS), Radiology Information System (RIS), dose monitoring, image quality, and CMS measure submission systems. Furthermore, hospitals are not required to use the Alara Imaging software for ( printed page 53937) CMS Measure Compliance. They may choose to use any software(s) that performs the necessary functions to generate the same standardized data elements necessary to calculate the measure consistent with the measure's specifications.
Pilot testing conducted by the measure steward was reviewed by an independent TEP, which found that the pilot met the CBE standards for reliability and validity testing. For more information on the Excessive Radiation eCQM pilot, we refer readers to the measure submission materials on the Partnership for Quality Measurement website at https://p4qm.org/measures/3663e and the eCQI Resource Center at https://ecqi.healthit.gov/ecqm/eh/pre-rulemaking/2024/cms1074v1.
Comment: Many commenters expressed concern that the two core metrics of the Excessive Radiation eCQM, “Calculated CT Global Noise” and “Calculated CT Size-Adjusted Dose” lack technical validity, clinical relevance, and implementation feasibility. Many commenters stated that the measure specifications have not been vetted or endorsed by the medical and scientific community, including interested parties such as the American Association of Physicists in Medicine (AAPM), the American College of Radiology (ACR), or the International Electrotechnical Commission (IEC). These commenters noted that the calculation methods for both metrics are not transparent or published, limiting peer review, reproducibility, and validation. Many commenters stated that forcing fixed cutoffs, such as the defined thresholds in the Excessive Radiation eCQM's numerator components, can compromise diagnostic accuracy by pushing CT exams toward too little or too much radiation, resulting in missed findings and unnecessary radiation exposure, respectively. These commenters also stated that the “Calculated CT Global Noise” does not reliably reflect diagnostic image quality and that the “Calculated CT Size-Adjusted Dose” is an imprecise measure of patient safety because it fails to account for factors such as clinical indication, protocol, patient size, scanner technology, and reconstruction methods.
Many commenters stated that maintaining voluntary reporting would give professional organizations and the CT imaging community time to identify metrics that better monitor diagnostic quality and patient outcomes. For example, many commenters recommended adopting the Size Specific Dose Estimate, (SSDE) as a validated alternative that is already implemented in commercial dose-monitoring systems and according to commenters, should be used instead of the “Calculated CT Size-Adjusted Dose”. Many commenters also recommended that CMS assess independent studies and peer-reviewed evidence of the eCQM's two core metrics before making the measure mandatory.
Response: We remain committed to the Excessive Radiation eCQM as a patient safety issue and affirm, as we did in the CY 2024 OPPS/ASC final rule (88 FR 81987), that the measure's data elements are scientifically and practically valid. The measure's framework was published in peer reviewed medical literature, and the measure's thresholds for noise and radiation dose were developed with close input from a diverse TEP, including radiologists and medical physicists.[327] The image noise thresholds were designed to maintain the diagnostic value of CT scans and empirically derived by radiologists evaluating image quality across a wide variety of studies and practice settings in experiments whose results were reviewed and approved during the measure review process.[328] Further, the measure is based on evidence and consensus-based clinical guidelines for optimizing CT radiation doses, from organizations such as the ACR,[329] Society of Interventional Radiology,[330] Society of Cardiovascular CT,[331] cardiovascular imaging societies,[332] Image Wisely 2020,[333] and the FDA.[334] In addition, measure testing across 16 inpatient and outpatient hospitals confirmed high availability, accuracy, validity, and reproducibility for all of its required data elements and the variables calculated by the translation software. Multiple bodies under the National Quality Forum (NQF) umbrella, including a Scientific Methods panel, gave the measure high scores for accuracy, validity, and reproducibility which led to a unanimous approval from the NQF.[335]
The Excessive Radiation eCQM aims to ensure CT radiation dose and image quality fall within safe and appropriate thresholds. The measure does not set minimal radiation dose requirements for any CT scan that could inadvertently result in higher doses than are currently used. The radiation dose thresholds are size-adjusted and based on underlying clinical indication, allowing for individualized patient care and clinician discretion for imaging protocol. We note that these dose thresholds are intended as general reference ranges to promote consistent optimization of radiation dose and image quality, not as prescriptive limits that override clinical judgment. We would like to further emphasize that hospitals should use the measure as a CT scan guideline while also adjusting noise and radiation doses when needed to ensure quality care.
The purpose of size adjustment in the Excessive Radiation eCQM is to account for the variation in patient size so that reporting entities are not penalized if they see a different mix of patients. To ensure case mix across reporting entities does not drive outlier values, SSDE is not used for adjustment for several reasons. First, SSDE reflects the average dose per slice rather than the total dose a patient receives during a complete scan, and because multiphase scanning is common, SSDE values may appear reasonable even when total exposure is high. The DLP reflects the total dose to the patient and thus more accurately reflects the radiation used and thus the risk for the study. In addition, SSDE is not consistently reported across CT ( printed page 53938) manufacturers and would require hospitals to upgrade existing CT machines. Lastly using SSDE for size adjustment would not account for the differences in patient case mix across hospitals. Because radiologists frequently use higher doses of radiation per unit of tissue in larger patients, practices who assess larger patients would be unfairly penalized.
Comment: Many commenters did not support the proposed change to maintain voluntary reporting for the Excessive Radiation eCQM, noting that while CT scans provide essential, life-saving diagnostic benefits, they are frequently performed using radiation doses that are higher than necessary for accurate diagnosis. These commenters, which included both patients and healthcare providers, expressed concern that factors increasing personal cancer risk, such as excessive ionizing radiation, are not being adequately addressed. Many commenters also expressed concerns regarding the lack of standardization and oversight in CT imaging, which according to the commenters results in radiation doses that are highly variable, often unoptimized, and frequently higher than needed for diagnostic purposes, thereby undermining national efforts to minimize unnecessary exposure. Many commenters stated that voluntary reporting is insufficient to protect patients from unnecessary radiation exposure and does not adequately hold hospitals accountable for using standardized, evidence-based radiation doses.
Response: We thank the commenters for their input and note that we remain committed to the Excessive Radiation eCQM as a patient safety issue for cancer prevention. We continue to believe, as we did when this measure was adopted in the Hospital OQR Program measure set, there is evidence that excessive radiation leads to harm,[336] and evidence that radiation doses could be lowered in many patients' situations without deteriorating image diagnostic utility.[337 338 339 340 341] A recent study in the New England Journal of Medicine suggested an association between exposure to radiation from medical imaging and a small but significantly increased risk of hematologic cancer.[342] We also agree with commenters about the importance of promoting patient safety by ensuring that patients are exposed to the lowest possible level of radiation while preserving image quality. Reducing unnecessary radiation exposure that leads to increased cancer incidence is also an example of primary prevention, or preventing illness before it occurs, a key priority of this administration. We believe that maintaining voluntary reporting supports patient safety goals by allowing the continued monitoring of hospital performance and progress on dose optimization, while providing hospitals with the opportunity to strengthen their technical capacity, refine dose optimization practices, and enhance data quality and consistency before the measure becomes mandatory. Retaining the measure in the program as voluntary ensures that it remains active and that hospitals may opt to report data during this period. This phased approach helps ensure that hospitals are better prepared to achieve sustainable and meaningful dose reductions, thereby advancing patient safety objectives over time.
Comment: A commenter expressed concern that maintaining voluntary reporting would be unfair to hospitals already investing in compliance and would signal a lack of confidence in the measure.
Response: We acknowledge that some hospitals may have already invested in compliance and reiterate that we remain committed to the Excessive Radiation eCQM as a patient safety issue for cancer prevention. Although we proposed extending voluntary reporting of the measure in response to feedback regarding implementation challenges, we also stated our intent to propose any additional changes to this measure in future rulemaking. As such, we intend to return to rulemaking to propose a date to begin mandatory reporting in the future, and we encourage hospitals to use the voluntary reporting period to improve imaging practices and develop the new processes and data extraction methods needed to capture and report data for this measure in preparation for future implementation.
Comment: Several commenters did not support maintaining voluntary reporting for the Excessive Radiation eCQM as they believe the measure is ready for broader adoption across CMS quality reporting programs. A few commenters highlighted their successful implementation of the measure and noted the measure's technical feasibility. A few commenters emphasized that the measure is scientifically sound and supported by peer-reviewed evidence. These commenters maintained that the measure specifications are validated and appropriate for identifying outliers in CT practice, and that the methodology accounts for variations in imaging protocols and technologies, demonstrating its robustness and readiness for broader implementation.
Response: We thank commenters for their feedback regarding the feasibility of the measure and confidence in its evidence-based specifications. We agree that the measure's data elements are scientifically and practically valid, and that the measure could be ready for broader adoption across CMS quality reporting programs. While we believe the measure is technically feasible for some hospitals, the variation in resource levels among hospitals with different characteristics, such as small and rural facilities, combined with feedback we have received regarding the implementation challenges faced by some HOPDs, indicates that more time is needed before reporting for the measure becomes mandatory. As previously stated, any future transition from voluntary to mandatory reporting ( printed page 53939) would be proposed through notice and comment rulemaking, and we encourage hospitals to use the voluntary reporting period to prepare adequately for future implementation.
While we continue to believe in the validity and importance of this measure in reducing excessive CT radiation dosage as a personal risk factor in developing cancer, we have also received ongoing feedback from interested parties reflecting current significant burden on HOPDs due to implementation challenges, including software integration into facility EHR or EMR systems and the additional processes required to aggregate data components. By keeping reporting voluntary, hospitals will have additional time to integrate and test the measure in their EHR systems effectively. Simultaneously, we can monitor how well hospitals are able to implement the measure over time before reconsidering mandatory reporting requirements in the future. As previously stated, any future transition from voluntary to mandatory reporting would be proposed through notice and comment rulemaking.
After consideration of the comments received, we are finalizing our proposal to modify the Excessive Radiation Dose or Inadequate Image Quality for Diagnostic Computed Tomography (CT) in Adults (Hospital Level—Outpatient) measure (Excessive Radiation eCQM) from mandatory reporting beginning with the CY 2027 reporting period/CY 2029 payment determination to continue voluntary reporting beginning with the CY 2027 reporting period. We encourage hospitals to use the voluntary reporting period to prepare for future implementation by addressing the remaining technical, operational, and data submission challenges needed to support consistent adoption of the measure. We intend to propose a date to begin mandatory reporting at the next feasible opportunity, which we expect to be the CY 2027 OPPS/ASC proposed rule.
4. Summary of Previously Finalized and Newly Finalized Hospital OQR Program Measure Set for CY 2026 to CY 2031 Payment Determinations
Table 138 summarizes the previously finalized and newly finalized Hospital OQR Program measure set for the CY 2026 to CY 2031 payment determinations, which removes the HCHE, Screening for SDOH, Screen Positive Rate for SDOH, and COVID-19 Vaccination Coverage Among HCP measures as discussed in section XIV.C. of this final rule with comment period; modifies reporting requirements for the Excessive Radiation eCQM from mandatory to voluntary reporting beginning with the CY 2027 reporting period, as discussed in section XV.B.3. of this final rule with comment period; removes the Left Without Being Seen and the Median Time for Discharged ED Patients measures as discussed in section XV.B.2. of this final rule with comment period; and adds the Emergency Care Access & Timeliness eCQM as discussed in section XV.B.1. of this final rule with comment period.
( printed page 53940) ( printed page 53941) ( printed page 53942)We refer readers to the QualityNet website at https://qualitynet.cms.gov/outpatient for additional information on the reporting periods and submission deadlines for each measure previously finalized in the Hospital OQR Program.
5. Hospital OQR Program Measures and Topics for Future Consideration
We refer readers to section XIV.B. of this final rule with comment period for ( printed page 53943) our cross-program Request for Information on measure concepts regarding well-being and nutrition for the Hospital OQR Program.
C. Updates to the Form, Manner, and Timing of Hospital OQR Program Data Submission
1. Background on Data Submission and Reporting Requirements for eCQMs
We refer readers to § 419.46(j) and the CY 2025 OPPS/ASC final rule with comment period (89 FR 94418 through 94420) for a discussion of our previously finalized eCQM requirements.
2. Data Submission and Reporting Requirements for the Emergency Care Access & Timeliness eCQM
In section XV.B.1. of this final rule with comment period, we finalized the adoption of the Emergency Care Access & Timeliness eCQM beginning with voluntary reporting for the CY 2027 reporting period followed by mandatory reporting beginning with the CY 2028 reporting period/CY 2030 payment determination. In the CY 2026 OPPS/ASC proposed rule (90 FR 33766), for the CY 2027 reporting period, we proposed that hospitals that voluntarily submit Emergency Care Access & Timeliness eCQM data could submit data for any quarter(s) (that is, up to all four quarters of data).
We also proposed that beginning with the CY 2028 reporting period/CY 2030 payment determination, hospitals would be required to report all four calendar quarters (1 calendar year) of data for the Emergency Care Access & Timeliness eCQM. We also proposed to require Emergency Care Access & Timeliness eCQM data submission by May 15 in the year prior to the affected payment determination year, in alignment with our policies on eCQM submission deadlines, as finalized in the CY 2022 OPPS/ASC final rule with comment period (86 FR 63867 through 63870). For example, for the CY 2028 reporting period/CY 2030 payment determination, hospitals would be required to submit eCQM data by May 15, 2029. All deadlines occurring on a Saturday, Sunday, or legal holiday, or on any other day declared at least in part to be a non-workday for Federal employees by statute or Executive Order, would be extended to the first business day thereafter. All current CMS policies regarding eCQM data submission requirements—including file format, zero denominator declarations, case thresholds, submission deadlines, and EHR certification requirements outlined at § 419.46(j) and finalized in the CY 2022 or CY 2025 OPPS/ASC final rules (86 FR 63867 through 63870 and 89 FR 94418 through 94420, respectively) would apply to the Emergency Care Access & Timeliness eCQM for both the voluntary and mandatory data submission periods.
We invited public comments on this proposal.
We did not receive public comments on these proposals, and therefore, we are finalizing the data submission and reporting requirements for the Emergency Care Access & Timeliness eCQM as proposed.
D. Payment Reduction for Hospitals That Fail To Meet the Hospital OQR Program Requirements for the CY 2026 Payment Determination
1. Background
Section 1833(t)(17) of the Act, which applies to subsection (d) hospitals (as defined under section 1886(d)(1)(B) of the Act), states that hospitals that fail to report data required to be submitted on measures selected by the Secretary, in the form and manner, and at a time, specified by the Secretary will incur a 2.0-percentage point reduction to their OPD fee schedule increase factor; that is, the annual payment update factor. Section 1833(t)(17)(A)(ii) of the Act specifies that any reduction applies only to the payment year involved and will not be taken into account in computing the applicable OPD fee schedule increase factor for a subsequent year.
The application of a reduced OPD fee schedule increase factor results in reduced national unadjusted payment rates that apply to certain outpatient items and services provided by hospitals that are required to report outpatient quality data in order to receive the full payment update factor and that fail to meet the Hospital OQR Program requirements. Hospitals that meet the reporting requirements receive the full OPPS payment update without the reduction. For a more detailed discussion of how this payment reduction was initially implemented, we refer readers to the CY 2009 OPPS/ASC final rule with comment period (73 FR 68769 through 68772).
The national unadjusted payment rates for many services paid under the OPPS equal the product of the OPPS conversion factor and the scaled relative payment weight for the APC to which the service is assigned. The OPPS conversion factor, which is updated annually by the OPD fee schedule increase factor, is used to calculate the OPPS payment rate for services with the following status indicators (listed in Addendum B to this final rule with comment period, which is available via the internet on the CMS website): “J1”, “J2”, “P”, “Q1”, “Q2”, “Q3”, “R”, “S”, “T”, “V”, or “U”. Payment for all services assigned to these status indicators will be subject to the reduction of the national unadjusted payment rates for hospitals that fail to meet Hospital OQR Program requirements, with the exception of services assigned to New Technology APCs with assigned status indicator “S” or “T”. We refer readers to the CY 2009 OPPS/ASC final rule with comment period (73 FR 68770 through 68771) for a discussion of this policy. In the CY 2017 OPPS/ASC final rule with comment period (81 FR 79796), we clarified that the reporting ratio does not apply to codes with status indicator “Q4” because services and procedures coded with status indicator “Q4” are either packaged or paid through the Clinical Laboratory Fee Schedule and are never paid separately through the OPPS.
The OPD fee schedule increase factor is an input into the OPPS conversion factor, which is used to calculate OPPS payment rates. To reduce the OPD fee schedule increase factor for hospitals that fail to meet reporting requirements, we calculate two conversion factors—a full market basket conversion factor (that is, the full conversion factor), and a reduced market basket conversion factor (that is, the reduced conversion factor). We then calculate a reduction ratio by dividing the reduced conversion factor by the full conversion factor. We refer to this reduction ratio as the “reporting ratio” to indicate that it applies to payment for hospitals that fail to meet their reporting requirements. Applying this reporting ratio to the OPPS payment amounts results in reduced national unadjusted payment rates that are mathematically equivalent to the reduced national unadjusted payment rates that would result if we multiplied the scaled OPPS relative payment weights by the reduced conversion factor. For example, to determine the reduced national unadjusted payment rates that applied to hospitals that failed to meet their quality reporting requirements for the CY 2010 OPPS/ASC final rule with comment period, we multiplied the final full national unadjusted payment rate found in Addendum B of the CY 2010 OPPS/ASC final rule with comment period by the CY 2010 OPPS final rule with comment period reporting ratio of 0.980 (74 FR 60642).
We note that the only difference in the calculation for the full conversion factor and the calculation for the reduced conversion factor is that the full ( printed page 53944) conversion factor uses the full OPD update, and the reduced conversion factor uses the reduced OPD update. The baseline OPPS conversion factor calculation is the same since all other adjustments would be applied to both conversion factor calculations. Therefore, our standard approach of calculating the reporting ratio as described earlier in this section is equivalent to dividing the reduced OPD update factor by that of the full OPD update factor. In other words:
Full Conversion Factor = Baseline OPPS conversion factor * (1 + OPD update factor)
Reduced Conversion Factor = Baseline OPPS conversion factor * (1 + OPD update factor − 0.02)
Reporting Ratio = Reduced Conversion Factor/Full Conversion Factor
Which is equivalent to:
Reporting Ratio = (1 + OPD Update factor − 0.02)/(1 + OPD update factor)
In the CY 2009 OPPS/ASC final rule with comment period (73 FR 68771 through 68772), we established a policy that the Medicare beneficiary's minimum unadjusted copayment and national unadjusted copayment for a service to which a reduced national unadjusted payment rate applies would each equal the product of the reporting ratio and the national unadjusted copayment or the minimum unadjusted copayment, as applicable, for the service. Under this policy, we apply the reporting ratio to both the minimum unadjusted copayment and national unadjusted copayment for services provided by hospitals that receive the payment reduction for failure to meet the Hospital OQR Program reporting requirements. This application of the reporting ratio to the national unadjusted and minimum unadjusted copayments is calculated according to § 419.41 of our regulations, prior to any adjustment for a hospital's failure to meet the quality reporting standards according to § 419.43(h). Beneficiaries and secondary payers thereby share in the reduction of payments to these hospitals.
In the CY 2009 OPPS/ASC final rule with comment period (73 FR 68772), we established the policy that all other applicable adjustments to the OPPS national unadjusted payment rates apply when the OPD fee schedule increase factor is reduced for hospitals that fail to meet the requirements of the Hospital OQR Program. For example, the following standard adjustments apply to the reduced national unadjusted payment rates: the wage index adjustment, the multiple procedure adjustment, the interrupted procedure adjustment, the rural sole community hospital adjustment, and the adjustment for devices furnished with full or partial credit or without cost. Similarly, OPPS outlier payments made for high cost and complex procedures will continue to be made when outlier criteria are met. For hospitals that fail to meet the quality data reporting requirements, the hospitals' costs are compared to the reduced payments for purposes of outlier eligibility and payment calculation. We established this policy in the OPPS beginning in the CY 2010 OPPS/ASC final rule with comment period (74 FR 60642). For a complete discussion of the OPPS outlier calculation and eligibility criteria, we refer readers to section II.G. of the CY 2023 OPPS/ASC proposed rule (87 FR 44533 through 44534).
2. Reporting Ratio Application and Associated Adjustment Policy for CY 2026
We proposed to continue our established policy of applying the reduction of the OPD fee schedule increase factor through the use of a reporting ratio for those hospitals that fail to meet the Hospital OQR Program requirements for the full CY 2026 annual payment update factor. For the CY 2026 OPPS/ASC proposed rule, the proposed reporting ratio was 0.9805, which, when multiplied by the proposed full conversion factor of $91.747, equaled a proposed conversion factor for hospitals that fail to meet the requirements of the Hospital OQR Program (that is, the reduced conversion factor) of $89.958. We proposed to continue to apply the reporting ratio to all services calculated using the OPPS conversion factor. We proposed to continue to apply the reporting ratio, when applicable, to all HCPCS codes to which we have proposed status indicator assignments of “J1,” “J2,” “P,” “Q1,” “Q2,” “Q3,” “R,” “S,” “T,” “V,” and “U” (other than New Technology APCs to which we have proposed status indicator assignments of “S” and “T”). We proposed to continue to exclude services paid under New Technology APCs. We proposed to continue to apply the reporting ratio to the national unadjusted payment rates and the minimum unadjusted and national unadjusted copayment rates of all applicable services for those hospitals that fail to meet the Hospital OQR Program reporting requirements. We also proposed to continue to apply all other applicable standard adjustments to the OPPS national unadjusted payment rates for hospitals that fail to meet the requirements of the Hospital OQR Program. Similarly, we proposed to continue to calculate OPPS outlier eligibility and outlier payment based on the reduced payment rates for those hospitals that fail to meet the reporting requirements. In addition to our proposal to implement the policy through the use of a reporting ratio, we proposed to continue to calculate the reporting ratio to four decimals.
We did not receive any public comments on our proposal and are finalizing as proposed. For this final rule with comment period, the final reporting ratio is 0.9805, which, when multiplied by the final full conversion factor of $91.415, equals a final conversion factor for hospitals that fail to meet the requirements of the Hospital OQR Program (that is, the reduced conversion factor) of $89.632. We are finalizing our proposal to continue to calculate OPPS outlier eligibility and outlier payment based on the reduced payment rates for those hospitals that fail to meet the reporting requirements. We are also finalizing our proposals to implement the policy through the use of a reporting ratio, and to continue to calculate the reporting ratio to four decimals to more precisely calculate the reduced adjusted payment and copayment rates for hospitals that fail to meet the Hospital OQR Program requirements for CY 2026 payment.
XVI. Rural Emergency Hospital Quality Reporting (REHQR) Program
A. Background and History of the REHQR Program
The Rural Emergency Hospital Quality Reporting (REHQR) Program is intended to ensure transparency and quality for rural emergency hospitals (REHs), defined at section 1861(kkk)(2) of the Act. Section 1861(kkk)(7)(A) authorizes the Secretary to implement a quality reporting program requiring REHs to submit data on measures in accordance with the Secretary's requirements in section 1861(kkk)(7). Section 1861(kkk)(7)(B)(ii) requires REHs to submit quality measure data to the Secretary “in a form and manner, and at a time, specified by the Secretary.” The Act does not require the Secretary to provide incentives for submitting this data under the REHQR Program, nor does it require the Secretary to impose penalties for failing to comply with this requirement under the REHQR Program. We refer readers to the CY 2024 OPPS/ASC final rule with comment period (88 FR 82046 through 82047) for a detailed discussion of the history of the REHQR Program. The REHQR Program requirements are codified at 42 CFR 419.95. We also refer readers to the CMS QualityNet REHQR ( printed page 53945) Program website at https://qualitynet.cms.gov/reh/rehqr for current program requirements and measure specifications.[343]
B. Changes to the REHQR Program Measure Set
In the CY 2026 OPPS/ASC proposed rule (90 FR 33768 through 337722), we proposed to adopt the Emergency Care Access & Timeliness electronic clinical quality measure (eCQM) beginning with the CY 2027 reporting period/CY 2029 program determination as an alternative to reporting the Median Time from Emergency Department (ED) Arrival to ED Departure for Discharged ED Patients measure. We also refer readers to section XIV.C. of this final rule with comment period where we finalize the following measure removals: (1) Hospital Commitment to Health Equity (HCHE) measure, beginning with the CY 2025 reporting period/CY 2027 program determination; (2) Screening for Social Drivers of Health (SDOH) measure, beginning with the CY 2025 reporting period; and (3) Screen Positive Rate for SDOH measure, beginning with the CY 2025 reporting period.
1. Adoption of the Emergency Care Access & Timeliness eCQM Beginning With Optional Reporting for the CY 2027 Reporting Period/CY 2029 Program Determination
a. Background
Occupancy and boarding rates in United States (U.S.) emergency departments (EDs) continue to worsen and exceed pre-pandemic levels.[344] ED boarding, defined as holding a patient in the ED when there are no available inpatient beds, often occurs due to shortages of inpatient beds and staff. ED boarding time contributes to ED crowding which heightens safety risks for patients and can lead to stressful working conditions for healthcare personnel.[345] A recent report from the Agency for Healthcare Research and Quality (AHRQ) characterized patient ED boarding as a growing public health crisis and engaged interested parties to address the strain on the U.S. healthcare system.[346]
Recent studies indicate that delays in the timeliness of ED care are associated with patient harm.[347] [348] Long ED wait times are also one of the most cited reasons for patients leaving an ED without being evaluated by a clinician.[349] One recent study indicated that ED crowding can harm sepsis patients by delaying administration of lifesaving intravenous (IV) fluids and antibiotics.[350] ED boarding time can lead to an increased length of stay (LOS) which is also a strong predictor of poor timeliness of care. One study found that for every patient boarded, the median ED LOS for all admitted patients increased by at least 12 minutes.[351] While less studied than inpatient boarding, transfer boarding (defined as keeping the patient in the ED after the decision to transfer has been made), can have similar impacts as inpatient boarding, with greater impacts on patients receiving care in rural settings.[352 353] The timeliness of care provided at REHs may be further impacted by transfer boarding due to lack of inpatient beds and resources required to coordinate transfers.[354]
Due to growing concerns about the quality and timeliness of care in the ED as well as the burden associated with manually abstracting the chart-abstracted ED measure adopted in the REHQR Program measure set, the Median Time from ED Arrival to ED Departure for Discharged ED Patients (Median Time for Discharged ED Patients) measure, CMS assessed additional ways to support efforts that reduce patient harm and improve outcomes for patients requiring emergency care while also providing flexibility for REHs. We previously sought comment on eCQM reporting under the REHQR Program in the CY 2024 OPPS/ASC proposed rule (88 FR 49840 through 49841). Adoption of the Emergency Care Access & Timeliness eCQM as an optional measure into the REHQR Program measure set, as discussed in section XVI.C.2.c. of this final rule with comment period, is in response to public comment recommending that CMS add eCQMs as optional measures initially (88 FR 82070). We refer readers to section XVI.C.2. of this final rule with comment period for a discussion of eCQM reporting and submission policies and requirements for the REHQR Program.
b. Measure Overview
An intermediate outcome measure, the Emergency Care Access & Timeliness eCQM [355] as specified for the REH setting calculates the proportion of four outcome metrics that quantify access to and the timeliness of care in an ED setting against specified thresholds, including: (1) patient wait ( printed page 53946) time; (2) whether the patient left the ED without being evaluated; (3) patient transfer boarding time in the ED; and (4) patient ED LOS. The numerator components for the Emergency Care Access & Timeliness eCQM are described in detail in section XVI.B.1.c. of this final rule with comment period. We note that the population and measure specifications for the Median Time for Discharged ED Patients measure overlaps with the Emergency Care Access & Timeliness eCQM for the numerator outcome metric (4), but that the scope of the proposed Emergency Care Access & Timeliness eCQM is broader than the Median Time for Discharged ED Patients measure.[356] The Median Time for Discharged ED Patients measure assesses one component, the time patients spent in the ED before being sent home, also known as ED throughput. The Emergency Care Access & Timeliness eCQM measures four different ED components in a single measure and provides REHs with separate data for each individual component. Additionally, the eCQM measures transfer boarding time in the ED and time from arrival to placement in a treatment room, which is not measured by the Median Time for Discharged ED Patients measure, or any other measure currently in the REHQR Program measure set.[357] As discussed in the CY 2026 OPPS/ASC proposed rule (90 FR 33771 and 33772), to provide flexibility for REHs, we proposed that REHs could elect to report either the Emergency Care Access & Timeliness eCQM or the Median Time for Discharged ED Patients measure beginning with the CY 2027 reporting period/CY 2029 program determination. While CMS proposed that the Emergency Care Access & Timeliness eCQM would not be required to be reported by REHs, we proposed that REHs must nonetheless elect to report either the Emergency Care Access & Timeliness eCQM or the Median Time for Discharged ED Patient measure to meet program requirements, beginning with the CY 2027 reporting period/CY 2029 program determination. We stated our belief that this timeline would provide REHs sufficient time to test and integrate the Emergency Care Access & Timeliness eCQM into existing clinical workflows.
For more information about the testing, feasibility, scientific acceptability, meaningfulness, and validity of the Emergency Care Access & Timeliness eCQM, we refer readers to the “Feasibility” and “Scientific Acceptability” sections of the Emergency Care Access & Timeliness eCQM listing on the Partnership for Quality Measurement website at https://p4qm.org/measures/4625e.
c. Measure Calculation
The measure denominator includes all ED encounters by patients of all ages, for all-payers, during a 12-month period of performance. Patients can have multiple encounters during a period of performance, and each encounter is eligible to contribute to the calculation of the measure.[358]
The measure numerator includes any ED encounter in the denominator where the patient experiences any one of the following: (1) the patient waited longer than 1 hour after arrival to the ED to be placed in a treatment room or dedicated treatment area that allows for audiovisual privacy during history-taking and physical examination; (2) the patient left the ED without being evaluated; (3) the patient, if transferred, boarded in the ED for longer than 4 hours; [359] or (4) the patient had an ED LOS of longer than 8 hours. An encounter is included in the numerator if any one of the four numerator events occurred with events not being mutually exclusive and each contributing only once to the numerator. ED encounters with ED observation stays [360] are excluded from components (3) and (4).
These four outcomes were selected based on published literature demonstrating that each numerator component is associated with patient harm,[361] as well as clinical (including ED), statistical, and methodological expert input. Additionally, a Technical Expert Panel (TEP) was convened by the measure developer.[362] The Patient and Family Engagement (PFE) Work Group provided feedback on emergency care experiences such as long wait times to be seen by a provider, long wait times to be transferred, and gaps in discharge processes.[363]
The numerator thresholds were developed according to evidence and consensus-based clinical guidelines for ED time thresholds from a TEP and environmental scans. For example, the four-hour threshold for numerator component (3), transfer boarding time, was developed per recommendations from The Joint Commission (TJC) and the American College of Emergency Physicians (ACEP).[364] If this proposal to adopt the Emergency Care Access & Timeliness eCQM for the REHQR Program is finalized, CMS would closely monitor the effect of this measure in REHs and may revise thresholds as appropriate.
Measure testing for the Emergency Care Access & Timeliness eCQM was conducted by the measure developer across 32 hospital-based EDs, representing a diverse mix of geographic regions, rurality, hospital size, teaching status, trauma level, and electronic health record (EHR) vendors, demonstrating that the measure is reliable, valid, and feasible for all required data elements.[365] Measure testing results had a wide range in overall scores, and across all strata, indicating variation in performance and implying room for quality improvement.[366] Although data element feasibility testing was performed at only one REH, the measure was also tested at a few rural hospital-based EDs with similar characteristics of pre-conversion REHs (for example, average bed range of under 25).[367] Based on this, we believe ( printed page 53947) these measure testing results are applicable to REHs.
The measure score is calculated at the individual ED level as the proportion of ED encounters where any one of the four outcomes occurred, divided by the number of encounters in a performance period. For CMS Certification Numbers (CCNs) with more than one ED, individual ED scores are then combined as a weighted average for that CCN.[368] The results of the Emergency Care Access & Timeliness eCQM are stratified into four groups, two by age (18 and older, and under 18) and two by mental health diagnoses (with, and without).[369] We note that the approach to stratification by age and mental health diagnosis is sufficient to account for differences between REHs without further need for risk adjustment.
For additional details regarding the measure specifications, we refer readers to the CMS QualityNet REHQR Program website at https://qualitynet.cms.gov/reh/rehqr.
d. Pre-Rulemaking Measure Review (PRMR)
As required under section 1890A of the Act, the Secretary must establish and follow a pre-rulemaking process for selection of quality and efficiency measures, including for the REHQR Program. The pre-rulemaking process, which we refer to as the Pre-Rulemaking Measure Review (PRMR), includes a review of measures published on the publicly available list of Measures Under Consideration (MUC List) by one of several committees convened by the consensus-based entity (CBE), with which we contract in accordance with section 1890 of the Act, for the purpose of providing interested parties input to the Secretary on the selection of quality and efficiency measures under consideration for use in certain Medicare quality programs, including the REHQR Program. We refer readers to the CY 2025 OPPS/ASC final rule with comment period (89 FR 94372) for details on the PRMR process, including the voting procedures used to reach consensus on measure recommendations. The PRMR Hospital Recommendation Group met on January 15 and 16, 2025, to review measures included by the Secretary on the publicly available “2024 Measures Under Consideration List” (MUC List), including the Emergency Care Access & Timeliness eCQM (MUC2024-095), and provided additional recommendations on the potential use of this measure.[370]
The voting results of the PRMR Hospital Recommendation Group for the proposed Emergency Care Access & Timeliness eCQM within the REHQR Program were: 9 members recommended adopting the measure into the REHQR Program; 6 members recommended adoption with conditions; 11 members voted not to recommend the measure for adoption. No voting category reached 75 percent or greater, including the combination of the recommend and the recommend with conditions categories and thus, the Hospital Recommendation Group did not reach consensus.[371]
The PRMR Hospital Recommendation Group noted in their deliberations that the measure will provide important insights into ED wait times which impact experience of care. The Group expressed concern that this measure may cause an increase in cost of care due to patients being transferred from the ED to observation.[372] While we acknowledge that patients transferred from the ED to observation may result in increased short-term costs due to additional monitoring and extended stays, the measure is designed to address significant issues surrounding the access to timely care which have been proven to reduce long-term costs.[373] Hospital Recommendation Group members also expressed concern about the lack of CBE endorsement. We note that we submitted the Emergency Care Access & Timeliness eCQM for CBE endorsement for review in the Fall 2024 cycle and the CBE endorsed the measure with conditions for use in the REHQR Program on February 12, 2025.[374] [375] We refer readers to section XVI.B.1.e. of this final rule with comment period for additional details on CBE endorsement.
The Hospital Recommendation Group discussed a few conditions specific to the REHQR Program, including changing the name of the measure to better reflect the measure's focus.[376] We agree with this feedback and have changed the name of the measure from the Emergency Care Capacity and Quality eCQM to Emergency Care Access & Timeliness. Additionally, Group members recommended stratifying the measure by factors such as care type as well as by region and trauma level designation. We emphasize that the approach to stratification by age and mental health diagnosis is sufficient to account for differences between REHs without further need for additional stratification.[377]
The Hospital Recommendation Group recommended revising the measure specifications to create separate measure components with the encouragement to explore additional measures for patient transfer boarding time and ED LOS as well as conducting further testing to expand the measure's applicability to REHs. We acknowledge the Hospital Recommendation Group's concerns and note that multiple TEPs and interested parties supported the inclusion of more than one numerator component as a strategy for internally balancing the measure and that time thresholds are ( printed page 53948) based on more than a decade of consensus work.[378] [379] [380] If the proposal to adopt the Emergency Care Access & Timeliness eCQM for the REHQR Program is finalized, CMS would closely monitor the effect of this measure in REHs and revise thresholds as appropriate (90 FR 33771). We note that out of the 32 hospital-based EDs that were tested for reliability, validity, and feasibility, approximately 20 percent were rural EDs, although not in the REHQR Program. Finally, Group members recommended implementing the Emergency Care Access & Timeliness eCQM with a phased approach with 2 years of voluntary reporting. We noted that we proposed adoption of the Emergency Care Access & Timeliness eCQM as an optional measure in lieu of the Median Time for Discharged ED Patients measure to provide additional flexibility for REHs given the anticipated implementation burden of this measure.
e. Measure Endorsement and Consideration of Low Case Volumes
Section 1861(kkk)(7)(C)(i) of the Act generally provides that any measure specified by the Secretary for use in the REHQR Program be endorsed by the entity with a contract under section 1890(a) of the Act, also known as the consensus-based entity (CBE).
The Emergency Care Access & Timeliness eCQM was submitted to the CBE for endorsement review in the Fall 2024 cycle (CBE #4625e), and the CBE endorsed the measure for use in the REHQR Program with conditions on February 12, 2025.[381] The conditions include that the measure developer explore: (1) the unintended consequences to patients and providers by engaging with the patient community and accountable entities; and (2) the data elements to identify and address where challenges may persist. If our proposal to adopt the Emergency Care Access & Timeliness eCQM is finalized, CMS would closely monitor the effects of this measure in REHs and data as part of the standard measure maintenance (90 FR 33771).
We believe this measure is appropriate for measuring quality of care under the REHQR Program because ED care is the primary focus of REHs. Furthermore, we believe that this measure meets the selection criteria under section 1861(kkk)(7)(C)(i) of the Act because it is endorsed by the CBE.
The REHQR Program's statute also includes a requirement at section 1861(kkk)(7)(C)(iii) of the Act to consider ways to account for rural emergency hospitals that lack sufficient case volumes when selecting measures to ensure that the performance rates for such measures are reliable. We note that the target population for this measure is comprised of patients of all ages, for all payers, that visit an REH during a 1-year measurement period. As such, we anticipate that the overall number of patients that visit an REH within a given year would be high enough so that there would not be any issues with low case volumes that could undermine the reliability of this measure when used in the REH context. We therefore do not believe the Emergency Care Access & Timeliness eCQM would suffer from low case volumes, and we further believe that we have appropriately considered low case volumes as required by the REHQR statute when selecting this measure.
f. Data Collection, Submission, and Reporting
The Emergency Care Access & Timeliness eCQM is specified in a standard electronic format, utilizing data extracted electronically from EHRs, with all data coming from defined fields in electronic sources. We note that eCQMs allowing for the retrieval of data directly from the EHR will minimize errors due to manual abstraction of data.[382] As discussed in the CY 2026 OPPS/ASC proposed rule (90 FR 33771 and 33772), we proposed that REHs would be required to report either the Emergency Care Access & Timeliness eCQM or the Median Time for Discharged Patients measure. We refer readers to the CY 2026 OPPS/ASC proposed rule (90 FR 33774 through 33776) for a discussion of proposed eCQM form, manner, and timing of data submission and reporting requirements. If an REH chooses to report the Emergency Care Access & Timeliness eCQM, the REH would report the data using the proposed methods and standards specified in section XVI.C.2. of this final rule with comment period.
In the CY 2026 OPPS/ASC proposed rule (90 FR 33772), we proposed to adopt the Emergency Care Access & Timeliness eCQM into the REHQR Program measure set beginning with the CY 2027 reporting period/CY 2029 program determination. However, as discussed later in the CY 2026 OPPS/ASC proposed rule (90 FR 33775 and 33776), we also proposed that the Emergency Care Access & Timeliness eCQM would not be a required measure under the REHQR Program, and that REHs could thus elect to report either the Emergency Care Access & Timeliness eCQM or the Median Time for Discharged ED Patients measure for a given reporting period/program determination. We stated that providing REHs with the option to report either of these measures would provide greater flexibility for REHs to implement EHR infrastructure that meets their individual needs while still prioritizing measurement of the variation in access to and the timeliness of emergency care, the goal of promoting interoperability, and reducing burden for REHs in the long term. We note that adoption of this measure does not change the number of mandatory quality measures required to be reported in the REHQR measure set.
The Median Time for Discharged ED Patients measure assesses the time patients spent in the ED before being sent home, also known as ED throughput. We note that this measure is reported quarterly, compared to the Emergency Care Access & Timeliness eCQM, which is less burdensome as it is reported annually. We further emphasize that the Emergency Care Access & Timeliness eCQM measures four different ED metric components in a single measure and is more comprehensive than the Median Time for Discharged ED Patients measure which measures one ED metric component.[383] Additionally, the eCQM measures transfer boarding time in the ED and time from arrival to placement in a treatment room, which are not currently captured by the Median Time for Discharged ED Patients measure, or ( printed page 53949) any other measure currently in the REHQR Program measure set.[384]
We refer readers to section XV.B.1. of this final rule with comment period where we finalize adoption of a similar version of this eCQM for the Hospital Outpatient Quality Reporting Program. With that finalized adoption, we note that adoption of this eCQM in the REHQR Program would also provide greater alignment of metrics between the two quality reporting programs.
We refer readers to the CY 2024 OPPS/ASC final rule with comment period (88 FR 49832) for more information on the Median Time for Discharged ED Patients measure.[385]
We intend to publicly report data submitted on the Emergency Care Access & Timeliness eCQM and the Median Time for Discharged ED Patients measure on a CMS website after a 30-day preview period. Public reporting for both measure serves to provide similar data on timeliness and encourages REHs to implement process improvements and reduce inefficiencies in ED operations, resulting in better quality of care.
We invited public comment on our proposal to adopt the Emergency Care Access & Timeliness eCQM as an optional measure beginning with the CY 2027 reporting period/CY 2029 program determination.
We note that we are finalizing the adoption of the Emergency Care Access & Timeliness eCQM in the Hospital OQR Program in section XV.B.1. of this final rule with comment period. We refer readers to that section for general comments related to the Emergency Care Access & Timeliness eCQM.
We invited public comments on our proposal.
Comment: Many commenters supported our proposal to adopt the Emergency Care Access & Timeliness eCQM as proposed. A few commenters supported the measure as appropriate and relevant to the REH setting, noting that ED care is a primary focus of REHs and that the measure appropriately addresses the growing challenges of ED occupancy and boarding, challenges which directly affect patient safety and timeliness of care. Some commenters appreciated that the measure represented a meaningful step towards streamlining data collection, improving measure efficiency, and enhancing real-time data availability to support quality improvement efforts. A commenter noted that the measure would enable healthcare organizations to optimize clinical processes, enhance patient safety, and achieve better outcomes in a timely manner.
Response: We thank commenters for their support of our proposal to adopt the Emergency Care Access & Timeliness eCQM and agree that the measure is appropriate and relevant to REHs. We agree that this measure would support healthcare organizations in achieving better outcomes.
Comment: Several commenters supported the Emergency Care Access & Timeliness eCQM as an alternative to reporting the Median Time for Discharged ED Patients measure, noting that this approach recognizes the realities of REHs, such as the technological, monetary, and staffing barriers that may present challenges to eCQM adoption and use for some REHs. Some commenters noted that this approach would enable REHs to select the option with the lower administrative burden of reporting based on their resources and clinical and administrative workflows. Some commenters requested that CMS continue to evaluate reporting for small, rural facilities, including consideration of flexible implementation strategies.
Response: We thank commenters for their support of the Emergency Care Access & Timeliness eCQM. We agree that providing REHs with the option to report either the Emergency Care Access & Timeliness eCQM or the Median Time for Discharged ED Patients measure, as discussed in section XVI.C.2.c. of this final rule with comment period, would provide greater flexibility for REHs. We are committed to ensuring flexibility that supports REHs in meeting program requirements.
Comment: Several commenters recommended publicly reporting the four individual numerator components as well as the overall measure score for the Emergency Care Access & Timeliness eCQM. Several commenters noted that data for the individual numerator components would provide more granular insights and improve hospitals' ability to target specific areas for improvement.
Response: As noted in the CY 2026 OPPS/ASC proposed rule (90 FR 33769), the Emergency Care Access & Timeliness eCQM measures four different ED numerator components in a single measure and provides REHs with separate data for each individual numerator component. We agree that such data provides meaningful and actionable information for REHs, patients, and other members of the public. While we proposed to report data for the Emergency Care Access & Timeliness eCQM as soon as feasible on a CMS website after a 30-day preview period (90 FR 33776), public reporting of these data would be contingent on the measure an REH chooses to satisfy mandatory reporting of one of the two ED-related measures in the REHQR Program measure set. Public reporting of Emergency Care Access & Timeliness eCQM data would begin once an REH has chosen to report and submitted data for the measure. This includes publicly reporting the overall score and four numerator components, in alignment with the approach taken by the Hospital OQR Program for publicly reporting a similar version of Emergency Care Access & Timeliness eCQM. We refer readers to section XV.B.1. of this final rule with comment period for more information on the Hospital OQR Program's adoption of a similar version of the Emergency Care Access & Timeliness eCQM.
Comment: Several commenters requested additional stratification factors for public reporting of the measure. Some commenters stated that they supported the measure but only with public reporting of each of the measure strata.
Response: We thank the commenters for their suggestions for additional stratifications for public reporting of the measure. The measure was evaluated by several stratification factors, and it was determined that age and mental health diagnoses were sufficient to account for differences between hospitals without further need for risk adjustment. We also note that several of the factors recommended are provider-level characteristics, and as such are not feasible to use as a patient characteristic for stratification. Additionally, we note that the Emergency Care Access & Timeliness eCQM does collect payer type, race, ethnicity, and sex as supplemental data elements; we can consider utilizing this information in support of additional future stratifications in the future. We clarify that we intend to publicly report the two mental health strata with the other two age-related strata on a CMS website.
Comment: Several commenters encouraged CMS to incorporate robust risk adjustment to account for patient case mix and volumes and the availability of post-acute care services within a hospital's service area.
Response: We thank the commenters for raising this aspect of the measure. The measure is not risk-adjusted because the numerator events are considered access failures regardless of ( printed page 53950) patient acuity or clinical comorbidity. We believe that the stratification of results by age and mental health diagnosis sufficiently accounts for differences between hospitals without risk adjustment or additional performance data detail based on analyses conducted during measure development.
Additionally, in measure testing the developer examined differences in Emergency Care Access & Timeliness score by patients' payer type composition (that is, proportion of patients with Medicaid, proportion of patients with Private/Other, and proportion of patients with Medicare) and found the strength of the relationship was weak to moderate and not statistically significant across all three of these compositional characteristics.
Comment: Some commenters requested more detailed performance metrics such as percentiles.
Response: We thank the commenters for their interest in the measure. We note that the Emergency Care Access & Timeliness eCQM is intentionally defined as a rate of access and timeliness failures. This approach focuses on a clear threshold that reflects operational and patient experience standards for accessible and timely care. Facilities may supplement this measure internally with percentile-based analyses as part of their ongoing performance improvement efforts.
Comment: Some commenters requested that CMS provide technical assistance to support REHs.
Response: Technical assistance for reporting information is available on the QualityNet website ( https://qualitynet.cms.gov/reh) and questions can be submitted via the Quality Question and Answer Tool ( https://cmsqualitysupport.servicenowservices.com/qnet_qa).
Comment: Several commenters did not support adoption of the Emergency Care Access & Timeliness eCQM, citing its unsuitability for REHs. These commenters noted that many rural facilities operate with limited staff, infrastructure, and technological resources, which can make implementation and reporting of complex eCQMs especially challenging. A commenter noted that most REHs operate with scaled-back electronic medical record (EMR) systems that are not designed to capture this data without costly customization and expressed concern that the reporting requirement would risk diverting scarce financial and human resources away from patient care. A few commenters expressed concern that adding a new layer of reporting would undermine the mission of the REH model, which is to preserve essential access to care in rural communities, particularly those facing severe financial distress. A commenter expressed concern that implementation of the measure beginning with the CY 2026 reporting period would be a challenge for Tribal REHs. The commenter noted that the lack of investment in Tribal REHs would hinder their ability to modernize their EHR system and urged cross-agency collaboration across HHS to modernize EHR systems for REHs. The commenter also requested that CMS make exceptions for Tribal REHs implementing new quality reporting requirements.
Response: As we noted in the CY 2026 OPPS/ASC proposed rule (90 FR 33774), while REHs may have some familiarity and experience with reporting eCQMs when formerly operating as a subsection (d) hospital or a CAH participating in the Medicare Promoting Interoperability Program, we recognize that technological, monetary, and staffing barriers may present challenges to eCQM adoption and use in some REHs. In recognition of these barriers, the anticipated implementation burden, as well as in response to public comments recommending that CMS add eCQMs as optional measures initially (88 FR 82070), we proposed that the Emergency Care Access & Timeliness eCQM would not be a required measure under the REHQR Program, and that REHs could thus elect to report either the Emergency Care Access & Timeliness eCQM or the Median Time for Discharged ED Patient measure for a given reporting period/program determination (90 FR 33771 and 33772). Given this flexibility, we believe that this approach, which we are finalizing, is compatible with the intent of the REH model and will provide REHs, including Tribal REHs, with more flexibility to implement the measure in a way that meets individual REH's needs. We also note that we proposed for this measure to begin with the CY 2027 reporting period, not the CY 2026 reporting period as the commenter noted.
Comment: Some commenters expressed concern that factors outside of an REH's control (such as weather or transportation barriers) may impact measure performance.
Response: Although we acknowledge that measure performance could be affected by multiple factors both inside and outside the control of REHs, measurement is essential for tracking and addressing these complex systemic issues and their downstream effects. By measuring the variation in access to and timeliness of care nationally, the measure builds an evidence base to determine which REHs are meeting benchmarks for emergency care, and which REHs could see improvement. Many REHs face similar challenges with factors such as weather or transportation barriers; however, the information collected through the Emergency Care Access & Timeliness eCQM can help patients and their caregivers compare REH performance and inform REH quality improvement efforts. We also note that we examined Median Time from Arrival to Departure metrics in the CY 2024 OPPS/ASC rulemaking and found that smaller, rural hospitals did not have lower performance than larger, urban hospitals.
Comment: A commenter recommended evaluating receiving facilities instead of REHs for numerator component (3) which evaluates the time from Decision to Transfer (order) to the ED departure.
Response: We thank the commenter for underscoring the importance of the time from Decision to Transfer (order) to the ED departure, one of the numerator components for the measure. We note that REHs are statutorily required to have in effect a transfer agreement with a level I or level II trauma center, such that patients that present at an REH with needs for longer-term inpatient care may receive that care. REHs must, therefore, address issues related to the coordination of care for transferred patients. We note that we intend to publicly report all strata of the measure so that this information is available for evaluation purposes.
Comment: Another commenter noted that CMS should focus instead on measures that rely on data sources already collected and available, such as claims data, cost reports, or Hospital Consumer Assessment of Healthcare Providers and Systems (HCAHPS) surveys.
Response: We note that three out of the four measures in the REHQR Program measure set are claims-based. As REHs are statutorily required to provide ED services, we believe it is important that the REHQR Program include one or more measures related to these services. We have provided flexibility to REHs regarding which ED services measure to report, the Emergency Care Access & Timeliness eCQM or the Median Time for Discharged Patients measure. Regarding the commenter's suggestion to use HCAHPS survey data, we note that the HCAHPS survey is designed for the ( printed page 53951) inpatient setting,[386] and would thus not be suitable for the REH setting.
Comment: A commenter did not support the Emergency Care Access & Timeliness eCQM because it was developed and tested for use in the hospital outpatient setting. The commenter stated that all measures must be tested at the REH level and demonstrate a high level of reliability, feasibility, and validity.
Response: We recognize the value of measures undergoing testing and evaluation of reliability, feasibility, and validity in the setting for which they are being adopted. As part of the PRMR process, the Emergency Care Access & Timeliness eCQM underwent an assessment of the measure's testing data and appropriateness for the REH setting, including a review of the measure's scientific acceptability, feasibility, and usability. As explained in the CY 2026 OPPS/ASC proposed rule (90 FR 33770), although data element feasibility testing was performed at only one REH, the measure was also tested at some rural hospital EDs with similar characteristics of pre-conversion REHs.[387] Based on this, we believe these measure testing results are applicable to REHs. We refer readers to the CY 2026 OPPS/ASC proposed rule (80 FR 33770 through 33771) for further discussion of the measure's review and evaluation.
Comment: A commenter noted that the low volumes treated by these facilities likely would be incompatible with the Emergency Care Access & Timeliness eCQM's calculation methodology.
Response: Regarding the commenter's concern about low case volumes, we refer readers to the CY 2026 OPPS/ASC proposed rule (90 FR 33771) for discussion of our consideration of low case volumes prior to proposing this measure for adoption in the REH setting. Given the denominator includes all ED visits over a 1-year measurement period, we anticipate that there would not be any issues with low case volumes that could undermine the reliability of this measure for REHs.
After consideration of the public comments we received, we are finalizing our proposal to adopt the Emergency Care Access & Timeliness eCQM into the REHQR Program, beginning with the CY 2027 reporting period/CY 2029 program determination as proposed. We also refer readers to section XVI.C.2.c. of this final rule with comment period where we finalize the data submission and reporting requirements for the Emergency Care Access & Timeliness eCQM as proposed, such that the Emergency Care Access & Timeliness eCQM would not be required to be reported by REHs under the REHQR Program, but REHs must nonetheless report either the Emergency Care Access & Timeliness eCQM or the Median Time for Discharged ED Patients measure to meet program requirements, beginning with the CY 2027 reporting period/CY 2029 program determination.
2. Summary of Previously Finalized and Newly Finalized REHQR Program Measure Set for CY 2026 to CY 2031 Program Determinations
Table 139 summarizes the previously finalized and newly finalized REHQR Program measure set for the CY 2026 to CY 2031 program determinations, which removes the Hospital Commitment to Health Equity (HCHE), Screening for SDOH, and Screen Positive Rate for SDOH measures as discussed in section XIV.C. of this final rule with comment period and adds the Emergency Care Access & Timeliness eCQM as discussed in section XVI.B.1. of this final rule with comment period.
( printed page 53952)We refer readers to the QualityNet website at https://qualitynet.cms.gov/reh/rehqr for additional information on the reporting periods and submission deadlines for each measure finalized and proposed in the REHQR Program.
3. REHQR Program Measures and Topics for Future Consideration
We refer readers to section XIV.B. of this final rule with comment period for our cross-program Request for Information on measure concepts regarding well-being and nutrition for consideration in the REHQR Program.
C. Updates to the Form, Manner, and Timing of REHQR Program Data Submission
In the CY 2026 OPPS/ASC proposed rule (90 FR 33774 through 33776), we proposed to update program policies for introducing eCQMs into the REHQR Program by establishing eCQM data submission and reporting requirements which apply to the proposed Emergency Care Access & Timeliness eCQM.
1. Maintenance of Technical Specifications for Quality Measures
CMS maintains technical specifications for adopted REHQR Program measures. The manuals containing the specifications for adopted measures are on the QualityNet website at https://qualitynet.cms.gov/reh/specifications-manuals. We refer readers to the CY 2024 OPPS/ASC final ( printed page 53953) rule with comment period (88 FR 82054) for additional information regarding these specification manuals.
In the CY 2026 OPPS/ASC proposed rule (90 FR 33774), in alignment with the Hospital OQR Program, we proposed that the technical specifications for eCQMs for the REHQR Program would be contained in the CMS Annual Update for the Hospital Quality Reporting Programs (Annual Update). The Annual Update and implementation guidance documents are available on the eCQI Resource Center website at https://ecqi.healthit.gov/. For eCQMs, we would generally update the measure specifications on an annual basis through the Annual Update which includes code updates, logic corrections, alignment with current clinical guidelines, and additional guidance for REHs and EHR vendors to collect and submit data on eCQMs from EHRs.
We invited public comments on this proposal.
We did not receive public comments on this proposal, and therefore, we are finalizing the policy for maintaining technical specifications for eCQMs as proposed.
2. Data Submission and Reporting Requirements for eCQMs for the REHQR Program Beginning With the CY 2027 Reporting Period/CY 2029 Program Determination
a. Background
Collection and reporting of data through health information technology greatly simplifies and streamlines quality reporting, and automated electronic extraction and reporting of clinical quality data would significantly reduce the administrative burden on REHs for the REHQR Program. Certified EHR technology (CEHRT) could effectively assist REHs in a variety of ways, such as by improving coordination of care with receiving hospitals during transfers, facilitating the types of staffing and personnel models required for REHs, and using eCQMs to improve quality and safety. In response to our request for comments in the CY 2024 OPPS/ASC proposed rule (88 FR 49840 through 49841) on eCQM reporting for the REHQR Program, some commenters recommended that CMS should consider adding eCQMs as optional measures initially (88 FR 82070). REHs have some familiarity and experience with reporting eCQMs when formerly operating as a subsection (d) hospital or CAH participating in the Medicare Promoting Interoperability Program, although we acknowledge that technological, monetary, and staffing barriers may present challenges to eCQM adoption and use in some REHs.
We refer readers to section XVI.B.1. of this final rule with comment period, where we finalize adoption of the Emergency Care Access & Timeliness eCQM into the REHQR Program measure set as an optional measure, beginning with the CY 2027 reporting period/CY 2029 program determination. The Emergency Care Access & Timeliness eCQM is the first eCQM in the REHQR Program measure set. Introducing eCQM reporting to the REHQR Program involves establishing related policies and requirements, such as eCQM certification requirements, data standards and formats, submission methods, and other program-specific requirements. In the following sections, to reduce reporting burden for REHs, we proposed eCQM reporting and submission policies and requirements for the REHQR Program, including reporting of the Emergency Care Access & Timeliness eCQM, that align with those of the Hospital OQR Program, Hospital Inpatient Quality Reporting Program, and Medicare Promoting Interoperability Program.
b. General Data Submission Requirements and Reporting Requirements
(1) eCQM Certification Requirements for eCQM Reporting
In the CY 2025 OPPS/ASC final rule with comment period (89 FR 94418 through 94420), the Hospital OQR Program finalized and codified three requirements relating to eCQM certification for the submission of eCQM data, beginning with the CY 2025 reporting period/CY 2027 payment determination. In the CY 2026 OPPS/ASC proposed rule, we proposed adopting the same eCQM certification requirements in the REHQR Program, beginning with the CY 2027 reporting period/CY 2029 program determination, and to likewise codify them by adding new paragraph (h) “Requirements for submission of electronic clinical quality measures (eCQMs) under the REHQR Program” to § 419.95 (90 FR 33774 through 33775). As discussed in section XVI.C.2.c. of this final rule with comment period, REHs would be required to meet these eCQM requirements beginning with the CY 2027 reporting period/CY 2029 program determination if the REH chooses to submit the Emergency Care Access & Timeliness eCQM rather than the Median Time for Discharged ED Patients measure.
Under this approach, we proposed to codify at § 419.95(h)(1) the requirement for REHs to utilize technology certified to Office of the National Coordinator for Health Information Technology's (ONC's) health information technology (IT) certification criteria, as adopted and updated in 45 CFR 170.315, for reporting eCQMs under the REHQR Program. Using the most recent certified health IT, which includes updated standards and criteria, is important as it allows the collection of relevant, accurate, and structured electronic data for electronic clinical quality measurement.
We also proposed to codify at 42 CFR 419.95(h)(2) the requirement that the health IT used for eCQM reporting by REHs must be certified to all eCQMs (that is, tested and validated on each individual eCQM) available to report under the REHQR Program. Additionally, we proposed to codify at § 419.95(h)(3) the requirement that REHs use the most recent version of the eCQM electronic measure specifications for the applicable reporting period available on the Electronic Clinical Quality Improvement (eCQI) Resource Center website at https://ecqi.healthit.gov/ or another website as designated by CMS. We also proposed that certified EHR technology would not need to be recertified each time the eCQMs specifications are updated to a more recent version.
Requiring EHRs to be certified to all available eCQMs under the REHQR Program would produce greater certainty for REHs that their EHR systems are capable of accurately calculating the eCQMs under the REHQR Program because the EHR technology would be up to date and tested on each eCQM. We believe this would reduce burden on REHs by minimizing the need to consult with their EHR and other health information technology vendors each time they report on a new or different eCQM.
Finally, we also proposed to codify at § 419.95(h)(4) that the requirements set forth in paragraphs (h)(1) through (3) apply only where an REH opts to report an eCQM.
We invited public comments on these proposals.
We did not receive public comments on these proposals, and therefore, we are finalizing the eCQM certification requirements for eCQM reporting as proposed and to codify these requirements by adding new paragraph (h) “Requirements for submission of electronic clinical quality measures (eCQMs) under the REHQR Program” to § 419.95. ( printed page 53954)
(2) File Format for EHR Data, Zero Denominator Declarations, and Case Threshold Exemptions
(a) File Format for EHR Data
Data can be collected in EHRs and health information technology systems using standardized formats to promote consistent representation, interpretation, and allowance for systems to compute data without needing human interpretation. As described in the FY 2016 IPPS/LTCH PPS final rule (80 FR 49701), these standards are referred to as content exchange standards because the standard details how data should be represented and the relationships between data elements. This allows the data to be exchanged across EHRs and health IT systems while retaining their meaning. Commonly used content exchange standards include the Quality Reporting Document Architecture (QRDA). The QRDA standard provides a document format and standard structure to electronically report quality measure data. We believe electronically reporting data elements formatted according to the QRDA standard can promote consistent representation and more efficient calculation of eCQM measure results.
To utilize the same file format requirements currently applied in the Hospital IQR, OQR, and Medicare Promoting Interoperability Programs (85 FR 58940, 86 FR 42262, and 80 FR 49706, respectively), in the CY 2026 OPPS/ASC proposed rule (90 FR 33775), we proposed the file format requirements for the REHQR Program beginning with the CY 2027 reporting period/CY 2029 program determination. Specifically, we proposed that REHs: (1) must submit eCQM data via the QRDA Category I (QRDA I) file format; [388] (2) may use third parties to submit QRDA I files on their behalf; and (3) may either use abstraction or pull the data from non-certified sources in order to then input these data into CEHRT for capture and reporting QRDA I. REHs could meet the reporting requirements by submitting data via QRDA I files, zero denominator declaration, or case threshold exemptions. We discuss the zero denominator declaration and case threshold exemptions in the subsequent sections. We also refer readers to section XVI.C.1. of this final rule with comment period where we outline the maintenance of technical specifications including those for eCQMs.
Under this proposal, we expect QRDA I files to reflect data for one patient per file per quarter with five key elements necessary to identify the file: (1) CCN; (2) CMS Program Name; (3) EHR Patient ID; (4) Reporting period specified in the Reporting Parameters Section; and (5) EHR Submitter ID.
(b) Zero Denominator Declarations
We understand there may be situations in which an REH does not have data to report on a particular eCQM. Therefore, we proposed if the REH's EHR is certified to an eCQM, but the REH does not have patients that meet the denominator criteria of that eCQM, the REH could submit a zero in the denominator for that eCQM; submission of a zero in the denominator for an eCQM would qualify as a successful submission for that eCQM (90 FR 33775).
(c) Case Threshold Exemptions
As a general matter, we understand that in some cases, particularly for REHs, an REH may not meet the applicable case threshold of encounters or discharges for a particular eCQM to reliably calculate performance on the measure. In the CY 2026 OPPS/ASC proposed rule (90 FR 33775), we proposed to align with the case threshold exemption from the Medicare Promoting Interoperability Program (77 FR 54080), the Hospital IQR Program (79 FR 50324), and the Hospital OQR Program (86 FR 63869). As stated for the Hospital IQR Program, the case threshold exemption means that for each quality measure where the minimum number of patients that meet the patient population denominator criteria for the relevant reporting period is not met, REHs could declare a “case threshold exemption.” Specifically, for the REHQR Program, we proposed that beginning with the CY 2027 reporting period/CY 2029 program determination, if an REH's EHR system is certified to report an eCQM and the REH has 5 or fewer outpatient encounters or discharges per quarter or 20 or fewer outpatient encounters or discharges per year (Medicare and non-Medicare combined), as defined by an eCQM's denominator population, that REH would be exempt from reporting on that eCQM. Case threshold exemptions would be able to be entered on the Denominator Declaration screen within the Hospital Quality Reporting (HQR) System (formerly referred to as the QualityNet Secure Portal) available during the submission period.[389] The exemption would not have to be used; REHs could report those individual cases if they would like to.
We invited public comments on these proposals.
Comment: A commenter supported our proposals related to zero denominator declarations and case threshold exemptions. The commenter particularly appreciated that the proposed case threshold exemption policy aligns with the Medicare Promoting Interoperability Program, the Hospital IQR Program, and the Hospital OQR Program.
Response: We thank the commenter for their support for our proposals related to zero denominator declarations and case threshold exemptions.
Comment: A commenter requested that CMS align eCQM submission formats across quality reporting programs.
Response: We note that in the CY 2026 OPPS/ASC proposed rule (90 FR 33775), we proposed to utilize the same file format requirements—the QRDA standard—applied in the Hospital IQR, OQR, and Medicare Promoting Interoperability Programs (85 FR 58940, 86 FR 42262, and 80 FR 49706, respectively).
After consideration of the public comments we received, we are finalizing the proposals related to the file format for EHR data, zero denominator declarations, and case threshold exemptions as proposed.
(3) Submission Deadlines for eCQM Data
To align with the Hospital OQR Program, in the CY 2026 OPPS/ASC proposed rule (90 FR 33775), we proposed to adopt a policy to require eCQM data submission by May 15 of the following year for the applicable CY reporting period, beginning with the CY 2027 reporting period/CY 2029 program determination. For example, if an REH elects to report the Emergency Care Access & Timeliness eCQM, the first proposed reporting period would run from January 1, 2027, through December 31, 2027, with a submission deadline of May 15, 2028. We note that the submission deadline may be moved to a subsequent day if it falls on a non-working day for Federal employees such as weekends or Federal holidays. ( printed page 53955)
We invited public comments on this proposal.
We did not receive public comments on this proposal, and therefore, we are finalizing the submission deadlines for eCQM data as proposed.
c. Data Submission and Reporting Requirements for the Emergency Care Access & Timeliness eCQM Beginning With the CY 2027 Reporting Period/CY 2029 Program Determination
In section XVI.B.1. of this final rule with comment period, we finalize the adoption of the Emergency Care Access & Timeliness eCQM into the REHQR Program beginning with the CY 2027 reporting period/CY 2029 program determination as an option for REHs to report instead of the Median Time for Discharged ED Patients measure. In addition to the general data submission and reporting requirements finalized for eCQMs in section XVI.C.2. of this final rule with comment period, in the CY 2026 OPPS/ASC proposed rule (90 FR 33775 through 33776), we also proposed requirements for reporting the Emergency Care Access & Timeliness eCQM under the REHQR Program. Specifically, we proposed that the Emergency Care Access & Timeliness eCQM would not be required to be reported by REHs under the REHQR Program, but that REHs must elect to report either the Emergency Care Access & Timeliness eCQM or the Median Time for Discharged ED Patients measure to meet program requirements, beginning with the CY 2027 reporting period/CY 2029 program determination. We stated that we believe our approach would provide REHs with more flexibility, including the time to plan and budget for the type of EHR infrastructure that meets their needs. Additionally, we noted this approach could contribute to successful participation in the REHQR Program, while still requiring REHs to report timeliness of ED care metrics. We also noted that the Median Time for Discharged ED Patients measure is reported quarterly through the HQR system, compared to the Emergency Care Access & Timeliness eCQM, which is reported annually. Sources of the relevant data for the Median Time for Discharged ED Patients measure may include claims forms, electronic health care data, EHRs, or paper records. We refer readers to the CY 2024 OPPS/ASC final rule with comment period (88 FR 82059 through 82062; 88 FR 82074 through 82075) for additional information on reporting the chart-abstracted Median Time for Discharged ED Patients measure.[390]
In the CY 2026 OPPS/ASC proposed rule (90 FR 33776), we proposed to report data from the REHQR Program as soon as it is feasible on a CMS website after a 30-day preview period.
We invited public comments on these proposals.
Comment: A few commenters recommended that CMS extend voluntary reporting of the Emergency Care Access & Timeliness eCQM in the REHQR Program to support a smoother transition to eCQM reporting and provide REHs with time needed to build capacity, train staff, and prepare systems, without compromising care or financial stability. Another commenter recommended mandatory reporting and retiring the Median Time for Discharged ED Patients, for measure alignment purposes.
Response: In the CY 2026 OPPS/ASC proposed rule (90 FR 33775 through 33776), we proposed that the Emergency Care Access & Timeliness eCQM would not be required to be reported by REHs under the REHQR Program, but that REHs report either the Emergency Care Access & Timeliness eCQM or the Median Time for Discharged ED Patients measure to meet program requirements, beginning with the CY 2027 reporting period/CY 2029 program determination. As discussed in greater depth in section XVI.B.1 of this final rule with comment period, CMS is finalizing its proposal to adopt the Emergency Care Access & Timeliness eCQM as an optional measure that REHs may choose to report instead of the existing Median Time for Discharged ED Patients measure. This approach is intended to ensure flexibility for REHs which may face barriers to implementing eCQMs.
After consideration of the public comments we received, we are finalizing the data submission and reporting requirements for the Emergency Care Access & Timeliness eCQM as proposed, such that the Emergency Care Access & Timeliness eCQM would not be required to be reported by REHs under the REHQR Program, but REHs report either the Emergency Care Access & Timeliness eCQM or the Median Time for Discharged ED Patients measure to meet program requirements, beginning with the CY 2027 reporting period/CY 2029 program determination.
3. Review and Corrections Period for Measure Data Submitted to the REHQR Program
In the CY 2024 OPPS/ASC final rule with comment period (88 FR 82075 through 82076), we finalized and codified at § 419.95(c)(3) a review and corrections period for all measure data submitted to the REHQR Program, which runs concurrently with the data submission period. During the review and corrections period, REHs can review, correct, and change these data up until the close of each submission deadline. However, after the submission deadline, REHs are not allowed to change these data. This policy applies to all measure data submitted to the REHQR Program, so this would include eCQM data.
The review and corrections period is from the time the submission period opens to the submission deadline. In the HQR System, REHs can submit QRDA Category I test and production data files and can correct QRDA Category I test and production data files before production data is submitted for final reporting. We encourage early testing and the use of pre-submission testing tools to reduce errors and inaccurate data submissions in eCQM reporting. We refer readers to the HQR System website (available at https://hqr.cms.gov/hqrng/login) and the CMS eCQI Resource Center (available at https://ecqi.healthit.gov/tool/hospital-quality-reporting-hqr-system) for more resources on eCQM reporting.
We invited public comments on this proposal.
Comment: A commenter supported our proposal to apply the review and corrections period for eCQM data submitted for the REHQR Program. The commenter noted that this policy reflects the workflow of many practices to periodically review progress, adjust their behaviors as necessary, and appreciated the opportunity to review and correct measure during the submission period.
Response: We thank the commenter for their support.
After consideration of the public comments we received, we are finalizing the review and corrections period for eCQM data as proposed.
4. REHQR Program Extraordinary Circumstances Exceptions (ECE) Policy
We refer readers to section XIV.D. of this final rule with comment period where we finalize with modification our cross-program proposal to codify updates to the Extraordinary Circumstances Exceptions (ECE) policy for the REHQR Program. ( printed page 53956)
XVII. Ambulatory Surgical Center Quality Reporting (ASCQR) Program
A. Background and History of the ASCQR Program
The Ambulatory Surgical Center Quality Reporting (ASCQR) Program is a pay-for-reporting program intended to ensure transparency for quality of care provided at ambulatory surgical centers (ASCs). Section 1833(i)(7)(A) of the Act authorizes the Secretary to reduce any annual increase under the revised ambulatory surgical center (ASC) payment system by 2.0 percentage points for such year that an ASC fails to submit required data on quality measures specified by the Secretary in accordance with section 1833(i)(7)(B) of the Act. Section 1833(i)(7)(B) of the Act states that, except as the Secretary may otherwise provide, several of the statutory provisions governing the Hospital Outpatient Quality Reporting (OQR) Program, specifically sections 1833(t)(17)(B) through (E) of the Act, also apply to the services of ASCs under the ASCQR Program in a similar manner to the manner in which they apply to the services of hospital outpatient departments under the Hospital OQR Program.
We refer readers to the CY 2012 OPPS/ASC final rule with comment period (76 FR 74492 through 74494) for a detailed discussion of the statutory authority of the ASCQR Program. The ASCQR Program requirements are codified at 42 CFR part 416, subpart H (§§ 416.300 through 416.330). We refer readers to the CMS website at https://www.cms.gov/medicare/quality/initiatives/asc-quality-reporting for general background on the ASCQR Program, as well as the CMS QualityNet ASCQR website at https://qualitynet.cms.gov/asc for current program requirements and measure specifications.
B. Changes to the ASCQR Program Measure Set
In the CY 2026 OPPS/ASC proposed rule, we proposed to adopt the Patient Understanding of Key Information Related to Recovery After a Facility-Based Outpatient Procedure or Surgery, Patient Reported Outcome-Based Performance Measure (Information Transfer PRO-PM) beginning with voluntary reporting for the CY 2027 and CY 2028 reporting periods followed by mandatory reporting beginning with the CY 2029 reporting period/CY 2031 payment determination (90 FR 33777 through 33779).
We refer readers to section XIV.C. of this final rule with comment period for a discussion of the following measure removals: (1) the COVID-19 Vaccination Coverage Among Healthcare Personnel (HCP) measure, beginning with the CY 2024 reporting period/CY 2026 payment determination; (2) the Facility Commitment to Health Equity (FCHE) measure, beginning with the CY 2025 reporting period/CY 2027 payment determination; (3) the Screening for Social Drivers of Health (SDOH) measure, beginning with the CY 2025 reporting period; and (4) the Screen Positive Rate for SDOH measure, beginning with the CY 2025 reporting period.
1. Proposed Adoption of the Information Transfer PRO-PM Beginning With Voluntary Reporting for the CY 2027 and CY 2028 Reporting Periods Followed by Mandatory Reporting Beginning With the CY 2029 Reporting Period/CY 2031 Payment Determination
a. Background
The volume and complexity of surgical procedures performed in outpatient settings, including ASCs, have steadily increased for over a decade.[391 392 393] As patients can benefit from having a clear understanding of their discharge information to support recovery from such procedures, the communication of discharge information is an important quality of care area for assessing facilities, and this information should be publicly available. A patient's lack of understanding of clinical care instructions provided after a procedure and other aspects of health literacy have been linked to poor adherence to treatment, decreased patient safety, increased return to the emergency department, and lower levels of patient satisfaction; disproportionately increased rates of such adverse effects occur to patients with limited English proficiency and patients over age 65.[394 395] Research in the hospital setting indicates that information provided to patients that is simpler and more complete is associated with fewer follow-up calls to an associated trauma center and less frequent hospital readmissions.[396 397] A study comparing discharge instructions provided to patients who had procedures performed in inpatient and ambulatory settings found that discharge instructions from the inpatient setting contained more complete medication lists and pending diagnostic result elements compared to discharge instructions provided by the hospital ambulatory setting.[398]
b. Measure Overview
The Information Transfer PRO-PM assesses patient understanding of provided discharge information for patients aged 18 years or older who had a procedure (surgical or non-surgical) at an ASC via a 9-item survey.[399] The survey evaluates patient reported understanding of information received across three domains: applicability to patient needs, medication, and daily activities. Survey results provide patient reported outcome (PRO) data measuring ASCs' communication efforts regarding discharge instructions and enable ASCs to reduce future risk of patient harm related to patients not fully understanding their recovery information. The survey was tested and deemed reliable in both English and Spanish versions; for ease of administration, the survey can be completed using a translator, proxy, or caregiver. The measure's testing results ( printed page 53957) are based on data from the hospital outpatient department (HOPD) setting; however, it is reasonably expected that the instrument and methodology apply to the ASC setting regarding patients receiving surgical procedures as both are outpatient surgical settings providing similar services with the supply of discharge instructions. We note that the measure specifications for the Information Transfer PRO-PM require that the survey be administered anonymously to patients and that the survey instrument does not collect any identifiable patient information.
In monitoring implementation of this measure for the Hospital OQR Program, we discovered that the anonymous administration requirement could potentially limit the hospitals' ability to collect data for their patients without working with a third-party vendor. If the survey is required to be fully anonymous, hospitals fielding the survey themselves would not be able to conduct any targeted follow-up with patients during the 65-day response window or use the information provided to develop more targeted quality improvement efforts.
While anonymous surveys can be valuable for gathering candid feedback, these issues of preventing follow-up, targeted action plans, and deeper investigation of specific issues, as well as leading to less serious or misleading responses have been documented.[400]
In the CY 2026 OPPS/ASC proposed rule, we invited public comment on the proposal to utilize this measure as specified with anonymous administration as well as potential data collection options to address the anonymity requirement in both the Hospital OQR Program, where the measure has already been adopted, and the ASCQR Program, where this measure was proposed for adoption (90 FR 33777).
The measure developer conducted pilot testing for this measure in 26 HOPDs in five states and demonstrated that the measure is reliable and meaningful.[401] Reliability of the measure was assessed with the Cronbach alpha score [402] to determine whether the nine survey questions reliably measured the same underlying characteristic; that is, patient's assessment of the clarity and applicability of recovery instructions. The Cronbach alpha score indicated that the survey items are reliable.[403] The measure developer also found the performance scores among facilities in the pilot study to be moderately reliable using a signal-to-noise ratio, which estimated variance among facilities and measured facility-specific standard errors to determine the extent to which variance in facility scores can be attributed to variance in actual performance.[404] More information about the testing, feasibility, scientific acceptability, meaningfulness, and validity of the Information Transfer PRO-PM for the HOPD setting is available at https://p4qm.org/measures/4210.
c. Measure Calculation
The measure numerator is the sum of all the individual scores an ASC receives from eligible respondents, which could be patients or their caregivers. Individual scores are calculated for each respondent by taking the sum of items for which the respondent gave the most positive response (either, “Yes” or “Very Clear”) and dividing by the number of items the respondent deemed applicable to their procedure or surgery. Applicable items are calculated by subtracting the sum of items for which the respondent selected “Does not apply” from the total number of survey items (nine).[405] The measure denominator is the total number of patients 18 years or older who had a procedure or surgery in an ASC, left the ASC alive, and responded to the survey. The cohort of patients for the Information Transfer PRO-PM is standardized with the OAS CAHPS cohort to minimize provider burden and to harmonize between the two surveys. Only fully completed surveys are included in the measure calculation. For additional details regarding the proposed measure specifications, we refer readers to the CMS QualityNet website.[406]
d. Pre-Rulemaking Measure Review (PRMR)
As required under section 1890A of the Act, the Secretary must establish and follow a pre-rulemaking process for the selection of quality and efficiency measures, including for the ASCQR Program. The pre-rulemaking process, which we refer to as the Pre-Rulemaking Measure Review (PRMR), includes a review of measures published on the publicly available list of Measures Under Consideration (MUC List) by one of several committees convened by the consensus-based entity (CBE), with which we contract in accordance with section 1890 of the Act, for the purpose of providing interested parties' input to the Secretary on the selection of quality and efficiency measures under consideration for use in certain Medicare quality programs, including the ASCQR Program. We refer readers to the CY 2025 OPPS/ASC final rule with comment period (89 FR 94372) for details on the PRMR process, including the voting procedures used to reach consensus on measure recommendations.
The PRMR Hospital Recommendation Group met on January 15 and 16, 2025 to review measures included by the Secretary on the publicly available “2024 Measures Under Consideration List” (MUC List), including the Information Transfer PRO-PM, for potential use.[407 408] The voting results of the PRMR Hospital Recommendation Group for the proposed Information Transfer PRO-PM for the ASCQR Program were: 5 members recommended adopting the measure; 14 ( printed page 53958) members recommended adoption with conditions; and 8 members voted not to recommend the measure for adoption. No voting category reached 75 percent or greater, including the combination of the recommend and the recommend with conditions categories. Thus, the PRMR Hospital Recommendation Group did not reach consensus and did not recommend including this measure in the ASCQR Program either with or without conditions.
The PRMR Hospital Recommendation Group noted in their deliberations the importance of measuring patient experience and delivering personalized and clear discharge instructions to prevent unnecessary hospital readmissions. However, the PRMR Hospital Recommendation Group members expressed concerns about lack of testing in the ASC setting given differences between HOPDs as a hospital setting (where testing of the Information Transfer PRO-PM was conducted) and ASCs. This group also highlighted concerns related to patient survey fatigue; potential overlap with the Outpatient and Ambulatory Surgery Consumer Assessment of Healthcare Providers and Systems (OAS CAHPS) survey sample population and content; and the risk of low response rates—particularly in small or rural facilities, which could impact scoring.
Although the measure was not pilot tested in the ASC setting with facilities citing resource constraints, we believe that the instrument and methodology reasonably apply to the ASC setting as the measure concept was designed for use in both HOPDs and ASCs and many of the same surgical procedures are performed in both settings.[409] Measure harmonization across the Hospital OQR and ASCQR Programs enables meaningful comparisons of care for patients to assess quality between settings that offer similar services.
Regarding the PRMR Hospital Recommendation Group's concern about potential overlap between the Information Transfer PRO-PM and OAS CAHPS content and target population, the OAS CAHPS survey addresses overall quality of healthcare facility communication but does not assess patient understanding of discharge information related to medication, activity, and applicability/personalization. We believe that both surveys provide valuable insights into different aspects of a patient's experience related to discharge instructions. Additionally, to minimize duplication of patient sampling, resources are available to help facilities align administration of OAS CAHPS with other surveys.[410] In consideration of potential population overlap, we selected a timeframe of 2 to 7 days post-procedure for administration of the Information Transfer PRO-PM's survey to strike a balance between patient recovery and mitigate overlap with the initial administration of OAS CAHPS.
Regarding concerns about patient survey fatigue and risk of low response rates, the 9-item survey is concise, presenting a low burden for completion. Further, ASCs would not be penalized for patients' decisions to not complete the survey. Payment implications under the ASCQR Program are tied to the successful and timely reporting of required quality measure data, and an ASC that submits data to CMS in the form, manner, and timing specified, regardless of the number of surveys completed by the ASC's patient population, would be considered compliant with the measure requirements.
To review the Hospital Recommendation Group's voting summary, recommendations, and conditions for the Information Transfer PRO-PM please visit https://p4qm.org/PRMR/Resources.
e. Measure Endorsement
Under section 1833(i)(7)(B) of the Act, requirements for the development of outpatient measures for the Hospital OQR Program at section 1833(t)(17)(C) of the Act apply to the ASCQR Program, except as the Secretary may otherwise provide. Section 1833(t)(17)(C)(i) of the Act requires measures developed to reflect consensus among affected parties and, to the extent feasible and practicable, shall include measures set forth by one or more national consensus-based entities (not necessarily the contracted CBE). As we have noted in previous rulemaking, consensus among affected parties can be reflected in ways other than CBE endorsement, including through the measure development process, through broad acceptance and use of the measure(s), and through public comment (76 FR 74494). We have also noted that section 1833(t)(17) of the Act does not require that each measure we adopt for the ASCQR Program be CBE-endorsed (76 FR 74494).
A Technical Expert Panel consisting of interested parties, experts, and consumer advocates contributed to the development of the Information Transfer PRO-PM measure's survey design, measure cohort, and survey implementation, demonstrating a consensus-based approach to the measure's development.[411]
In the CY 2026 OPPS/ASC proposed rule, we explained that while we recognize the value of measures undergoing CBE endorsement review and prefer to use endorsed measures, at that time, we found no other CBE-endorsed measures for the ASC setting that address the topic of patients' understanding of clinical information related to their recovery for an outpatient procedure or surgery (90 FR 33779). We noted that we submitted the Information Transfer PRO-PM to the CBE for endorsement review in the Fall 2023 cycle (CBE #4210) for the Hospital OQR Program, and the CBE endorsed the measure on January 29, 2024.[412] The ASC-specific version of the Information Transfer PRO-PM is designed to use the same specifications as the Hospital OQR Program CBE-endorsed measure. We previously stated that we plan to pursue CBE endorsement for the measure's implementation in the ASC setting in a future measure endorsement cycle and we would continue to monitor implementation of the measure as part of the standard measure maintenance process.
f. Data Collection, Submission, and Reporting
In the CY 2026 OPPS/ASC proposed rule, we proposed that the Information Transfer PRO-PM would be calculated based on PRO data collected by ASCs directly or through their authorized third-party vendors through the Information Transfer PRO-PM survey instrument [413] distributed to patients or ( printed page 53959) their caregivers by electronic mail or text. We note that the Information Transfer PRO-PM survey is nonproprietary and free to use. We proposed that the survey be distributed within 2 to 7 days post-procedure or surgery. This timeframe minimizes the influence of variables related to the surgery or procedure, such as medications that could affect comprehension, fatigue, or acute pain, while ensuring timely reporting of patient experience related to recovery information.
In the pilot testing conducted by the measure developer using a third-party vendor, patients were sent a reminder to complete the survey 7 days after receipt. The survey remained open until pilot testing was completed; the mean length of time between the procedure date to the survey response date was 65 days. Based on these findings, we proposed a 65-day window for patient response to the survey.
We proposed to adopt the Information Transfer PRO-PM as a voluntary measure for the CY 2027 and CY 2028 reporting periods followed by mandatory reporting beginning with the CY 2029 reporting period/CY 2031 payment determination. We stated we would utilize the voluntary period to monitor the implementation and operationalization of the measure. We refer readers to section XVII.C. of this final rule with comment period for a discussion of the Information Transfer PRO-PM form, manner, and timing of data submission and reporting requirements.
We invited public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: Many commenters generally supported the Information Transfer PRO-PM, stating that it is important that patients have a clear understanding of their discharge instructions to enhance recovery. Some commenters noted that this measure addresses an important measurement area, and that improving patient understanding of discharge instructions can help reduce post-procedural readmissions and mortality, which reduces waste in the healthcare system. A few commenters also agreed that PRO-PMs like this one provide valuable insights into the effectiveness of communication with patients and patient satisfaction and can help identify opportunities for improvement in perioperative care. Another commenter asserted that assessing patient experience beyond the OAS CAHPS survey can drive improvements in care transitions, reduce readmissions, and enhance outcomes. Other commenters supported CMS's decision to introduce the measure through voluntary reporting.
Response: We thank commenters for their support, including their support of a voluntary reporting period. We agree that it is important for patients to have a clear understanding of their discharge instructions to enhance recovery and reduce adverse outcomes, particularly as procedures of higher complexity move to this setting. We agree that this measure addresses an important measurement area, that this measure can provide valuable feedback to clinicians about their communication practices, and it can highlight areas of needed improvement. However, after consideration of public comments, including comments raising concerns regarding low response rates and survey fatigue, we are not finalizing the proposal to adopt the Information Transfer PRO-PM for the ASCQR Program at this time.
Comment: Some commenters supported the proposal to adopt this measure stating that adoption would be a positive step towards aligning with the Hospital OQR Program. A commenter supported CMS' proposal to utilize this measure as specified with anonymous administration, supplying potential data collection options to address the anonymity requirement while emphasizing that the measure is evidence-based, has been tested to show moderate reliability, and has been endorsed by a CBE for the hospital outpatient department setting and has been adopted for the Hospital OQR Program.
Response: We thank the commenters for their support of the measure and for their support of the survey being administered anonymously.
Comment: A commenter supported the Information Transfer PRO-PM stating there is significant variation in the transmission of information between facilities and that patients are the only source of reliable information on the effectiveness of this communication. Another commenter expressed support for including the option to allow family caregivers to provide feedback for the Information Transfer PRO-PM because of the vital role family caregivers play in patient care.
Response: We thank the commenters for their support and for noting the importance of engaging patients and caregivers for information on the effectiveness of communication of information related to their care. We agree that family caregivers play an important role in patient care.
Comment: A commenter supported the use of web-based data collection for survey administration.
Response: We thank this commenter for the support for this mode of data collection.
Comment: A commenter recommended adding questions to the Information Transfer PRO-PM that are specialty specific. For example, this commenter noted that the survey can ask patients about their experience with anesthesia or pain medications, including questions on numbness or increased pain, to provide actionable feedback and support follow-up care after outpatient procedures.
Response: We thank the commenter for their suggested updates to the Information Transfer PRO-PM. While we are not finalizing the Information Transfer PRO-PM for the ASCQR Program at this time, we note that alignment across the quality reporting programs is important to ensure that clear and effective communication of information related to recovery is addressed in every healthcare delivery setting. Accordingly, we proposed to adopt this measure in the ASCQR Program consistent with its use in the Hospital OQR Program. While there are likely to be unique aspects related to patient recovery from an ASC procedure, adopting this measure in consistent form across quality reporting programs would enable cross-setting comparisons and evaluation utilizing a national, standardized survey instrument.
Comment: A commenter did not support the Information Transfer PRO-PM and stated that since a significant portion of the proposed information to be collected is included in the discharge summary the measure information is redundant.
Response: We disagree that this measure is redundant as the purpose of the discharge summary is to meaningfully convey post-surgery instructions to patients. The Information-Transfer PRO-PM was developed to assess the patient's experience during the discharge process and their understanding of these instructions.
Comment: Some commenters recommended engaging ASCs to determine the most effective means to collect data on patient understanding of discharge instructions. One commenter stated that this measure evaluates patients' comprehension rather than the materials and care provided by facilities. Another commenter stated that the Information Transfer PRO-PM ( printed page 53960) assesses service delivery, not patient outcomes, and will not be useful in improving clinical decision-making.
Response: The Information Transfer PRO-PM is designed to evaluate facilities based on their patients' perspective of their understanding of discharge instructions and experience based on the materials supplied by facilities. We believe that this measure can provide valuable feedback to facilities on the usefulness of their materials and their method in supplying these materials. We note that a patient's lack of understanding of clinical care instructions provided after a procedure and other aspects of health literacy have been linked to poor adherence to treatment, decreased patient safety, increased return to the emergency department, and lower levels of patient satisfaction; disproportionately increased rates of such adverse effects occur to patients with limited English proficiency and patients over age 65.[414 415] The Information Transfer PRO-PM addresses this important area of care coordination and offers an effective means of collecting data from patients themselves. Through this data collection, this measure can provide valuable feedback to clinicians about their communication practices and highlight areas of needed improvement in care delivery. However, after consideration of the concerns raised in public comments, we are not finalizing the proposal to adopt the Information Transfer PRO-PM for the ASCQR Program at this time.
Comment: Many commenters did not support adoption of the Information Transfer PRO-PM due to concerns about survey fatigue and low response rates. Several of these commenters recommended integrating the Information Transfer PRO-PM into the OAS CAHPS survey to reduce the potential for survey fatigue. A commenter noted that response rates to Federal surveys is declining as evidenced by reductions in response rates to Federal surveys in general and specifically the OAS CAHPS survey. Several commenters noted that administering two distinct surveys to the same patient cohort would require duplicative processes for identifying eligible patients, coordinating survey vendors, and managing survey distribution within overlapping timeframes. A few of these commenters stated that the OAS CAHPS survey is experiencing declining response rates over time and expressed concern that response rates for both surveys may be affected as patients may experience survey fatigue if they receive both surveys from ASCs. Another commenter requested clarification on whether multiple outreach attempts and reminders would be permitted in order to improve survey response rates.
Response: We thank the commenters for providing their concerns regarding survey fatigue and low response rates, and the suggestion to integrate the Information Transfer PRO-PM with the OAS CAHPS survey to reduce the number of surveys a patient could receive regarding their episode of care. We considered integrating questions from the Information Transfer PRO-PM survey into the OAS CAHPS survey to reduce the number of surveys a patient could receive regarding their episode of care. At that time we decided against doing so because it would necessitate changes to the specifications for administering OAS CAHPS. However, we acknowledge commenters' concerns and may consider the suggestion to combine the information transfer PRO-PM with OAS CAHPS in the future.
Research indicates that COVID-19 pandemic restrictions to in-person research greatly increased survey fatigue as researchers moved to using on-line methods [416 417] and while this affects all groups, differential effects are seen across industries [418] and demographics.[419] While we continue to believe that standardized patient-reported outcome measures, utilized across different payors including Federal agencies, are important to enable comparison,[420] we also recognize that survey fatigue manifests in declining quality and quantity of survey responses and has become a significant issue in information collection. We therefore understand commenters' concern that having two patient surveys for ASCs would likely result in the potential for low response rates and survey fatigue, even if ASCs provided multiple outreach attempts. Survey fatigue may also present greater challenges for ASCs because of their smaller administrative teams and fewer dedicated resources to manage patient outreach and survey processes.
We also acknowledge that patients receiving care from ASCs may experience more survey fatigue and lower response rates than patients in other settings due to the types of care and services provided by ASCs. For example, ASCs typically handle less complex and lower-risk, same-day procedures, so patients may be less motivated to complete surveys than they would for more serious conditions. Patients at ASCs may also be less willing to complete patient surveys for facilities with which they have less ongoing engagement after discharge, as is likely with ASCs. Patients at ASCs also may undergo multiple elective procedures, so they could receive duplicative patient surveys.
In consideration of the comments received related to concerns about low response rates and survey fatigue, we are not adopting the Information Transfer PRO-PM at this time. We will provide clarification regarding multiple outreach attempts to improve survey response rates if we decide to propose the Information Transfer PRO-PM in the future.
Comment: Many commenters expressed concern that the Information Transfer PRO-PM may increase administrative burden on facilities. Some of these commenters noted the challenge of integrating surveys into clinical workflows. A few commenters stated concerns that the burden of implementing this measure is greater than its potential benefits. Several commenters expressed significant concerns with the operational and logistical challenges related to implementing an additional, separately administered patient survey because ( printed page 53961) ASCs already administer OAS CAHPS. These commenters noted that the measure's administrative tasks, including data collection and tracking, could create significant burden for facilities with existing staff shortages which would divert resources from direct patient care. A commenter stated that some ASCs are beginning to adopt the OAS CAHPS survey in preparation for mandatory reporting so the addition of another survey to the workflow would be costly and administratively burdensome at this time.
Response: We acknowledge that collecting PRO-PM data may involve more burden and initial implementation effort than some other types of quality measures. While PRO-PMs typically require facilities to integrate data collection into clinical workflows, this integration could provide an opportunity for patient-reported outcomes to inform clinical decision-making. To provide more flexibility and reduce burden, we did not propose to require facilities to collect data in a standardized way for the Information Transfer PRO-PM. However, we understand that ASCs may have less experience integrating surveys into clinical workflows, so it may still be difficult for ASCs to implement this measure. Due to this concern, and concerns that the recent increase in the number of patient surveys may contribute to survey fatigue for patients, we are not finalizing adoption of the Information Transfer PRO-PM at this time.
Comment: A few commenters stated that the Methodology Report cites evidence from inpatient and outpatient hospitals in explaining the need for this measure. These commenters stated that there is no research indicating that ASCs have similar quality concerns. A commenter explained that all ASCs provide written discharge instructions in accordance with the Conditions for Coverage, and that there is no evidence that these instructions are insufficient in the ASC setting.
Response: We understand the commenters' concern that the Methodology Report did not cite studies related to discharge instructions in the ASC setting. We note that collecting data on measures aligned across the Hospital OQR Program and the ASCQR Program allows patients to compare performance across these two settings which provide similar services. Additionally, this measure would allow patients to compare performance between ASCs on discharge instructions, an area which was determined to be important by our Patient and Family Engagement (PFE) Work Group (89 FR 94407). However, as previously stated, we are not finalizing the proposal to adopt the Information Transfer PRO-PM for the ASCQR Program at this time.
Comment: A few commenters recommended administering the Information Transfer PRO-PM in the same nine languages as the OAS CAHPS survey to ensure consistency across surveys. Several commenters recommended requiring that facilities offer the survey in the patient's preferred language, if that language is one of these nine, to ensure inclusivity.
Response: We wish to clarify that the Information Transfer PRO-PM can be completed using a translator, proxy, or caregiver, and that ASCs would have been permitted to use interpretation and translation services in order to ensure that ASCs were administering the survey in the patient's preferred language. Although we are not finalizing adoption of the Information Transfer PRO-PM, we appreciate the commenters' recommendations and will consider available language options for the Information Transfer PRO-PM if we decide to propose this measure in the future.
Comment: Several commenters expressed doubts about the applicability of the measure as it has not been tested in the ASC setting. Some of these commenters recommended site-specific testing in the ASC setting before inclusion in the ASCQR Program. A commenter stated that the measure developer acknowledged the potential need to modify the measure for use among ASCs and recommended evaluating it for needed modifications before adoption. For example, a commenter noted that the survey introduction uses the word “hospital” twice and expressed concern that, even if the name of the facility is correct, this could cause patients to question the legitimacy or applicability of the survey. This commenter stated that measures should be fully tested in the setting for which they are being proposed before being adopted (even as voluntary measures) into quality reporting programs. A commenter recommended piloting the measure in an upcoming CMS Innovation Center (CMMI) Model as a means of testing the measure's feasibility for widespread implementation. A commenter requested clarification regarding whether survey response rates had been tested when both the OAS CAHPS survey and the Information Transfer PRO-PM survey are administered to the same patient population.
Response: We believe that the measure is applicable to the ASC setting because improving patient understanding of discharge information is an important part of providing quality care across all care settings. Historically discharge information is provided in written form after surgery because the effects of anesthesia and pain medication may impair the patient's ability to retain information. However, we understand commenters' concerns related to feasibility of implementing this measure without tailoring it to the ASC setting due to lack of testing in the ASC setting. We note that we are not finalizing this measure at this time, and that we will review the measure specifications for applicability to ASCs and will update language as necessary if we decide to propose this measure in the future. We will consider additional testing for the Information Transfer PRO-PM if we decide to propose this measure in the future. If we do conduct additional testing in the future, results of testing would reflect administration of the Information-Transfer PRO-PM and OAS CAHPS because OAS CAHPS is now mandatory for ASCs.
Comment: A few commenters expressed concern regarding potential conflicts between the requirements for the Information Transfer PRO-PM survey and the OAS CAHPS guidelines. A few commenters specifically mentioned that the OAS CAHPS guidelines recommend selecting the OAS CAHPS sample prior to any other survey and not including the same patients in more than one survey to ensure the highest response rates possible. A few commenters stated that the OAS CAHPS guidelines indicate that ASCs must coordinate with the OAS CAHPS Survey Coordination Team if they are conducting more than one Federally sponsored survey and that introduction of the Information Transfer PRO-PM would require all ASCs to coordinate with this team. These commenters expressed concern that the requirements for this survey contradict the goal of ensuring the highest response rate possible for the OAS CAHPS survey since the Information Transfer PRO-PM survey would be distributed before the OAS CAHPS survey, as most patients do not receive the OAS CAHPS survey until five to six weeks after the procedure. A commenter expressed concern that patients may not appreciate the distinction between the two surveys, leading them to discard the second survey. Some commenters recommended developing a coordinated administration plan that ensures patients do not receive more than one survey for the same encounter.
Response: We thank commenters for raising concerns regarding possible ( printed page 53962) conflicts with the requirements for the Information Transfer PRO-PM survey and the OAS CAHPS guidelines.
Regarding the concern that ASCs must coordinate with the OAS CAHPS Survey Coordination team, we wish to clarify that the OAS CAHPS FAQ document describes this as a recommendation and not a requirement, and note that it only would apply if more than one survey vendor was used or if the surveys were being self-administered.[421] We acknowledge that the timing and order of administering those surveys may impact the likelihood of patients responding to each survey. Additionally, we understand that administering the Information Transfer PRO-PM to every patient, not to a subset or sample of patients, would necessarily lead to some patients receiving multiple surveys. However, we believe that both surveys provide valuable insights into different aspects of a patient's experience. While we are not finalizing the Information Transfer PRO-PM in this final rule, we will take these comments into consideration if we propose the measure in the future.
Comment: A few commenters noted that the denominator cohort for the Information Transfer PRO-PM is different from the denominator cohort for OAS CAHPS, and that as a result, ASCs would need to separately determine patient eligibility for each survey, which could result in onerous processes related to survey administration. A commenter stated that the OAS CAHPS cohort includes codes and exclusions that are not replicated in the Information Transfer PRO-PM. This commenter recommended aligning the denominator cohorts and identifying a mechanism to ensure future standardization of the two surveys to reduce the burden of maintaining two different processes. Another commenter requested clarification regarding which survey, the OAS CAHPS survey or the Information Transfer PRO-PM survey, should be administered to patients first.
Response: We appreciate commenters' recommendation to align the Information Transfer PRO-PM and OAS CAHPS cohorts and to minimize the burden of administering the two surveys. During development of the Information Transfer PRO-PM, we considered incorporating the survey instrument into the OAS CAHPS survey to reduce the number of surveys a patient could receive regarding their episode of care. However, we decided against doing so at that time as the specifications for administering the OAS CAHPS (such as the survey timing, allowing multiple submission modes and requiring a CMS-approved vendor) do not align with the Information Transfer PRO-PM. The PRMR Hospital Recommendation Group recommended the Information Transfer PRO-PM with conditions for the Hospital OQR Program; the condition was to administer the survey at the time of the surgery or procedure so that there is no conflict with other measured pain and function outcomes to improve response rates. We considered this condition and determined that a survey administration window of 2 to 7 days post-procedure provides the necessary balance between allowing patients sufficient time for recovery and comprehension of the Information Transfer PRO-PM survey questions, while also mitigating potential overlap with the OAS CAHPS survey, which begins on the first day post-procedure and includes a reminder at 14 days. Therefore, to the commenter's question on which survey should be administered first under our proposal to adopt the Information Transfer PRO-PM, based on the survey timing guidelines mentioned above, the OAS CAHPS survey should be administered first (that is, first day post-procedure), followed by the Information Transfer PRO-PM survey at any point from 2 to 7 days post-procedure. Currently, there has been no testing measuring survey response rates when both surveys are administered to the same patient population. While we are not finalizing adoption of the Information Transfer PRO-PM in this final rule, we will take these comments into consideration if we propose the measure in the future.
Comment: A commenter requested clarification on when data submission specifications would be provided to vendors and whether there would be a point of contact for questions regarding those specifications. A commenter recommended that CMS provide materials such as technical guidance and standardized tools to assist with reporting.
Response: Data submission specifications for the reporting of this measure are available in the Hospital OQR Program Specifications Manual and would have applied to the ASCQR Program if the measure had been adopted as proposed.[422] Additionally, vendors may submit inquiries for the Information Transfer PRO-PM through the ServiceNow website. However, as previously stated, we are not finalizing the proposal to adopt the Information Transfer PRO-PM for the ASCQR Program at this time.
Comment: A commenter requested clarification on how responses of “Does Not Apply” would be treated in scoring and whether partially completed surveys would contribute to measure results. Another commenter requested clarification regarding whether facilities would receive domain-level feedback or only a composite score.
Response: Individual scores are calculated for each respondent by taking the sum of items for which the respondent gave the most positive response (either, “Yes” or “Very Clear”) and dividing by the number of items the respondent deemed applicable to their procedure or surgery. Applicable items are calculated by subtracting the sum of items for which the respondent selected “Does not apply” from the total number of survey items (nine). Only fully completed surveys would be included in the measure calculation.[423] If a facility administers the Information Transfer PRO-PM survey, they would be able to calculate domain level or composite results. If a facility uses a third-party to administer the survey, the granularity of data available to the facility would depend on the contract with their selected vendor. However, as previously explained, we are not finalizing the proposal to adopt the Information Transfer PRO-PM for the ASCQR Program at this time.
Comment: Some commenters had concerns with the measure specifications. These commenters recommended that measure scores be adjusted to account for patient factors beyond the control of the facility such as age, education, surgery type, time between surgery and survey response, overall physical and mental health, health literacy, and language barriers. Some of these commenters recommended that other survey factors such as non-response rates, a minimum response-rate threshold, and annual review to identify any potential bias be considered. A commenter stated that combining the Information Transfer PRO-PM survey with the OAS CAHPS survey would allow data from both surveys to be adjusted in the same manner.
Response: We understand commenters' concerns about measure ( printed page 53963) specifications with regard to patient factors beyond the control of the facility. We note the measure developer conducted testing to examine the need for risk adjustment to the measure for a set of factors such as patients self-reported health status, history of procedures, age, and education, and found there was not a significant relationship between the adjusted and unadjusted measure scores. Therefore, given the lack of a significant relationship between adjusted and unadjusted scores, there was empirical evidence to support an unadjusted measure and that providers and facilities should be able to provide clear, personalized instruction to all patients about their recovery from a procedure or surgery.[424] Additional details regarding measure testing and measure methodology, are available on the QualityNet website: https://qualitynet.cms.gov/outpatient/measures/PRO-PM/methodology. However, as previously stated, we are not finalizing the proposal to adopt the Information Transfer PRO-PM for the ASCQR Program at this time.
Comment: A few commenters expressed concern about the operational feasibility of the survey timing. Specifically, some commenters expressed concern about issuing survey invitations in the period 2 to 7 days post-procedure. These commenters stated that because of coding practices it may not be feasible to assign CPT-4 or HCPCS Level II codes within this timeframe as well as submit patient files to vendors with sufficient time for the vendor to process files, confirm patient eligibility, and issue invitations. A commenter recommended updating the eligibility definition to rely on data elements available in near real time (for example, use of anesthesia or sedation) or extending the timeline for survey administration. A commenter expressed concern about the 65-day timeline for patients to respond to the survey. The commenter referenced CMS' statement that in the pilot the mean length of time between the procedure date and the survey response date was 65 days and stated that the 65-day period was actually the amount of time that it took to send the survey invitation.
Response: We appreciate commenters' feedback regarding the feasibility of the proposed survey administration timeline, including concerns about the period for issuing invitations (2 to 7 days post-procedure) and the overall survey response window. We acknowledge commenters' operational concerns related to the availability of CPT-4 and HCPCS Level II codes and the ability of facilities and their vendors to identify eligible patients and submit files within the proposed timeframe. The proposed 2- to 7-day invitation window was selected to promote timely contact with patients following the clinical encounter and to minimize recall bias by focusing responses on the recent episode of care. These operational timing parameters were informed by the pilot testing conducted by the measure developer.
With respect to the 65-day observation highlighted by commenters, we clarify that during pilot testing, the survey remained open until sufficient response was attained with the mean interval between the procedure date and the survey response date being 65 days, or approximately two months. We identified this finding as the basis for proposing a 65-day response window to allow sufficient time for patients who are sent an invitation and for non-responders to receive reminders and to submit completed surveys.
At this time, we believe the timing parameters for invitation issuance and the proposed 65-day response window are appropriate to support valid, comparable measurement across facilities while balancing operational feasibility. However, we are not finalizing the Information Transfer PRO-PM for the ASCQR Program at this time.
Comment: A commenter expressed concern that limiting survey administration to email and text communications may affect representativeness and data quality because patients who have limited internet access may not be included in the survey. A commenter expressed concern about limiting access to email and text communications because people are becoming more reluctant to complete surveys due to concerns about data privacy, specifically associated with emails and text communications from unknown or unverified entities. Another commenter stated that the burden is more pronounced for ASCs specializing in ophthalmology, as their ophthalmology patients cannot easily check their emails or text messages due to limited vision.
Response: We appreciate the commenters' concerns about representativeness and data quality as well as the potential impact of this measure to ASCs specializing in ophthalmology. The measure developer did not identify systemic challenges in reaching patients through email or text message. Additionally, as distribution of the survey occurs 2 to 7 days after the procedure, the patient has up to 65 days to respond, and the survey can be completed using a translator, proxy, or caregiver, the influence of post-surgical variables can be minimized.
However, we note that we are not finalizing adoption of this measure at this time and may re-evaluate available survey modalities if we propose the measure in the future.
Comment: A commenter expressed concern that the measure fails to exclude certain patients, potentially violating the Telephone Consumer Protection Act (TCPA) and the Health Insurance Portability and Accountability Act (HIPAA). This commenter proposed excluding: (1) patients without email or text messaging access, (2) patients who have not given (or have withdrawn) prior consent for text message survey invitations (to comply with the TCPA), and (3) patients who requested confidential admission (to adhere to HIPAA). A commenter expressed concern that some ASCs' legal teams interpret a patient opting-out of text survey invitations as opting-out of all text-based communications. This commenter recommended that prior to adopting the Information Transfer PRO-PM, CMS provide guidance on how to allow patients to opt-out of the survey without opting out of important patient communications, such as appointment reminders.
Response: We thank the commenter for expressing these concerns regarding the TPCA and HIPAA as they relate to survey administration. The TPCA establishes rules for how surveys can be administered via phone. If this measure were adopted, facilities and their vendors would need to comply with TCPA regulations to avoid penalties and maintain patient privacy. These requirements are in regard to prior consent, limitations on the use of automatic dialers, and restrictions on artificial or prerecorded messages when contacting patients by phone for the Information Transfer PRO-PM. ASCs would need to work with their legal teams to determine opt-in or opt-out language that clearly indicates the patient's communication preferences. However, we reiterate that we are not finalizing the proposal to adopt the Information Transfer PRO-PM for the ASCQR Program at this time.
Comment: A few commenters stated that the requirement for anonymity limits their ability to use the survey data to investigate any issues identified and ( printed page 53964) to develop targeted action plans and quality improvement efforts, which defeats the purpose of the measure. A commenter stated that the measure has not been tested with a self-administration option and recommended testing self-administration and resolving barriers prior to adopting this measure. A few commenters requested guidance on how facilities could administer the survey anonymously without working with an outside vendor. A commenter expressed concern about the survey's current framing around anonymity as the instrument introduction informs patients that their responses are “completely anonymous,” yet facilities must necessarily use protected health information to identify and contact eligible patients, creating at least a temporary link between patient identity and survey responses. The commenter stated that this promise of total anonymity could increase distrust for patients, whereas stating that the responses would be confidential both aligns with patients' understanding of how healthcare organizations handle their most sensitive data and also aligns with other CMS patient surveys.
Response: We thank the commenters for their input. We agree that anonymity places constraints on use of the measure data which must be balanced with confidentiality concerns. We acknowledge that there are both advantages and disadvantages to maintaining anonymity in patient surveys. Anonymity may encourage patients to provide more candid and reliable feedback about their experiences, which can enhance the validity of the information collected. However, as commenters noted, anonymity can limit the ability of facilities to conduct targeted follow-up or quality improvement activities and may, in some cases, contribute to patient distrust if the anonymity promise is not clearly explained. We appreciate the commenters' insights on these challenges and we agree that the balance between protecting patient privacy and enabling meaningful quality improvement is important.
The survey instrument notes that patient responses are anonymous and that neither the patient's name nor any other identifying information will be shared with their clinician or facility. We recognize that vendors and facilities must use protected health information (such as patient names, contact information, and encounter dates) to invite and remind patients to complete the electronic survey during the administration process. Consistent with prior CMS surveys, such as OAS CAHPS, the intent of this Information Transfer PRO-PM's anonymity language is to indicate that individual patient responses will not be reported or publicly displayed with patient identifiers and will be handled in a manner that protects patient privacy.
When a facility administers the survey directly (without a vendor), it may leverage existing survey tools that include features that protect patient anonymity. Facilities administering the survey themselves can follow-up with all eligible patients throughout the 65-day response window based on the procedure date, rather than the survey administration date. Where vendors are used, we expect both vendors and facilities to comply with all applicable privacy protections and business-associate agreements under HIPAA. They must also meet any minimum business and quality requirements outlined in CMS guidance for vendor administered surveys. If we propose to adopt the Information Transfer PRO-PM in the future, we would provide guidance to support facilities that wish to administer surveys internally or without vendor involvement.
We acknowledge that the phrase “completely anonymous” may be interpreted to mean that no possible link to the identity of the respondent ever exists, which is not operationally feasible where contact information must be used to invite and remind patients to complete the survey. To reduce possible confusion and patient distrust, and to align with language used in other CMS patient surveys, we will consider revising the instrument introduction and associated text to clarify that: (i) contact information is used only to invite and remind eligible patients, (ii) individual survey responses will be protected and will not be reported to CMS or published with personal identifiers, and (iii) aggregate or de-identified data will be used for measure calculation and reporting. Although we are not finalizing adoption of this measure at this time we note that this clarification would be reflected in our operational guidance for implementation if this measure is considered for the ASCQR Program in the future. We will also consider including this clarification in our operational guidance for implementation in the Hospital OQR Program.
We note that the proposed voluntary reporting period was also intended to allow for refinement of mode-of-administration recommendations and to identify barriers to self-administration through operational experience. We will consider including additional guidance or flexibilities in this area in future iterations of the measure. Additionally, we recognize that challenges with recruiting ASCs to participate in testing during initial measure development limited opportunities to fully evaluate self-administration approaches, and similar challenges could resurface if we pursue additional testing in this area in the future.
Comment: A commenter stated that facilities already work with Patient and Family Advisory Councils to improve patient communications and that these engagements are more likely to provide meaningful improvements than an anonymous survey which does not allow for follow-up.
Response: We agree that Patient and Family Advisory Councils are a valuable resource, and encourage facilities to engage with these councils to improve their discharge planning processes and overall patient communication.
Comment: A few commenters expressed concern regarding the implementation timeline of the Information Transfer PRO-PM and recommended that CMS delay implementation and extend the use of voluntary reporting periods. A commenter recommended that CMS monitor ASC feedback during the voluntary reporting period to ensure ASCs do not face widespread technical or operational challenges which would require postponing mandatory implementation.
Response: We understand commenters' concern regarding the timeline for the Information Transfer PRO-PM, and we note that we are not finalizing adoption of this measure in the ASCQR Program at this time.
Comment: A commenter stated that when the measure underwent CBE review for the hospital outpatient setting the committee found that the measure did not meet requirements for scientific acceptability, specifically that the sample was too small to report reliability by decile and that the results for validity testing were non-significant.
Response: We acknowledge the commenter's concern regarding the measure's scientific acceptability, but note that the measure developer conducted pilot testing in 26 HOPDs across five states and demonstrated that the measure is both reliable and
( printed page 53965)meaningful.[425] Reliability of the measure was assessed with the Cronbach alpha score [426] to determine whether the survey questions reliably measure the same underlying characteristic, that is, patients' assessment of the clarity and applicability of recovery instruction. The Cronbach alpha score indicated that the survey items are reliable.[427] The measure developer also found that facility performance scores in the pilot study demonstrated moderate reliability based on a signal-to-noise ratio. This analysis estimated variance across facilities and measured facility-specific standard errors to determine the extent to which variance in facility scores can be attributed to variance in actual performance.[428] More information about the testing, feasibility, scientific acceptability, meaningfulness, and validity of the Information Transfer PRO-PM for the HOPD setting is available at https://p4qm.org/measures/4210.
During measure testing, the developer conducted extensive outreach for testing partners, however, ASCs were unavailable at the time of the testing period due to competing demands of implementing other PRO-PMs. The measure developer completed two pilot studies in the HOPD setting. The average annual case volume of an ASC is 4,714, indicating it would take a minimum of three months to meet the minimum requirement of respondents. Given the similar patient or caregiver interactions and goals in ASCs and HOPDs, we expect testing results to have similar reliability.
Comment: A commenter noted that this measure has not met statutory requirements for inclusion in the ASCQR Program due to not being endorsed by a Consensus-Based Entity (CBE) for use in the ASC setting.
Response: We thank the commenter for their observation that the measure has not been endorsed by the CBE for use in the ASC setting. We recognize the value of measures undergoing review for potential CBE endorsement and of measure endorsement specific to care setting. We note that the Information Transfer PRO-PM went through the rigorous measure development lifecycle outlined at the CMS Measures Management System website [429] which includes measure testing and reliability analysis. Furthermore, for the Hospital OQR and ASCQR Programs, we note that section 1833(t)(17) of the Act does not require that each measure we adopt be CBE-endorsed (76 FR 74494), but states that the Hospital OQR and ASCQR Programs, to the extent feasible and practicable, shall include measures set forth by one or more national consensus building entities. We reviewed measures endorsed by consensus organizations and were unable to identify any other measures on this topic endorsed by a consensus organization in the ASC setting, so the inclusion of an endorsed measure is not feasible or practicable. Section 1833(t)(17)(C)(i) of the Act also requires measures included in the Hospital OQR and ASCQR Programs to reflect consensus among affected parties. As we have stated in previous rulemaking (75 FR 72064 and 76 FR 74494), consensus among affected parties can be reflected in ways other than CBE endorsement, including through the measure development process, through broad acceptance and use of the measure(s), and through public comment. However, in light of the significant number of negative public comments received during the comment period opposing the adoption of this measure for the ASCQR Program, we have concerns that there may be a lack of consensus among interested parties. These concerns reflect an additional reason we are not finalizing this measure at this time.
Comment: A commenter stated that the requirement for consensus has not been met through broad acceptance of the measure by ASCs. The commenter stated that the ASC representative on the TEP did not support the use of the measure in the ASC setting and that ASC interested parties have not supported the measure in public comments.
Response: Section 1833(t)(17)(C)(i) of the Act requires measures included in the Hospital OQR and ASCQR Programs to reflect consensus among affected parties. As we have stated in previous rulemaking, consensus among affected parties can be reflected in various ways, including through the measure development process, through broad acceptance and use of the measure(s), and through public comment.
As previously stated, we have concerns regarding a potential lack of consensus among interested parties, given the significant number of negative public comments received during the comment period opposing the adoption of this measure for the ASCQR Program. This concern represents an additional reason we are not finalizing this measure at this time.
After consideration of public comments, we are not finalizing adoption of the Information Transfer PRO-PM for the ASCQR Program at this time.
2. Summary of Previously Finalized and Newly Finalized ASCQR Program Measure Set for CY 2026 to CY 2031 Payment Determinations
Table 140 summarizes the previously finalized and newly finalized ASCQR Program measure set for the CY 2026 to CY 2031 payment determinations. Table 140 reflects the removal of the FCHE, Screening for SDOH, Screen Positive Rate for SDOH, and the COVID-19 Vaccination Coverage Among HCP measures as discussed in section XIV.C., we note that we are not finalizing the addition of the Information Transfer PRO-PM as discussed in section XVII.B.1. of this final rule with comment period.
( printed page 53966) ( printed page 53967)We refer readers to the QualityNet website at https://qualitynet.cms.gov/asc/ascqr for additional information on the reporting periods and submission deadlines for each measure finalized and proposed in the ASCQR Program.
3. ASCQR Program Measures and Topics for Future Consideration
We refer readers to section XIV.B. of this final rule with comment period for information regarding our cross-program ( printed page 53968) Request for Information on measure concepts regarding well-being and nutrition for consideration in the ASCQR Program.
C. Updates to the Form, Manner, and Timing of ASCQR Program Data Submission
In the CY 2026 OPPS/ASC proposed rule, we proposed to establish data submission and reporting requirements for Patient-Reported Outcome-Based Performance Measures (PRO-PMs) for the ASCQR Program, including for the proposed Information Transfer PRO-PM (90 FR 33782).
1. Data Submission and Reporting Requirements for PRO-PMs
a. Data Submission Requirement for PRO-PMs
In the CY 2024 OPPS/ASC final rule with comment period (88 FR 82041), we finalized that for the Total Hip Arthroplasty and/or Total Knee Arthroplasty PRO-PM, ASCs must use the Hospital Quality Reporting (HQR) system for data submission as specified for a PRO-PM. In the CY 2026 OPPS/ASC proposed rule, we proposed to apply this submission method to PRO-PMs generally, including the Information Transfer PRO-PM (90 FR 33782). Specifically, we proposed that ASCs must use the HQR system for data submission for any PRO-PM that we adopt for the ASCQR Program measure set. ASCs may choose to: (1) directly submit their PRO-PM data to CMS using the HQR system; or (2) utilize a third-party entity, such as a vendor or registry, to submit their data using the HQR system. The HQR system allows for data submission using multiple file formats (such as .CSV and .XML) or a manual data entry option, allowing ASCs additional flexibility in data submission.
b. Data Submission and Reporting Requirements for the Information Transfer PRO-PM
In the CY 2026 OPPS/ASC proposed rule, we discussed the proposed adoption of the Information Transfer PRO-PM beginning with voluntary reporting for the CY 2027 and CY 2028 reporting periods followed by mandatory reporting beginning with the CY 2029 reporting period/CY 2031 payment determination (90 FR 33776 through 33779). We proposed that the reporting period for this measure would include data collection for procedures performed from January 1 through and including December 31 of the year that is two years prior to the applicable payment determination year. Therefore, ASCs would attribute patient survey responses to the CY reporting period during which the patient's procedure was completed. For example, if a patient undergoes a procedure on December 20, 2027, and their survey response is received on January 4, 2028, that response would be attributed to the CY 2027 reporting period. In the CY 2026 OPPS/ASC proposed rule we proposed a 65-day response window for collecting patient survey responses (90 FR 33776 through 33779). Under this 65-day response window policy, ASCs may collect survey responses for a reporting period as late as March of the year preceding the applicable payment determination year.
We proposed to require ASCs to submit their Information Transfer PRO-PM data in aggregate numerators and denominators by May 15 of the year prior to the applicable payment determination year in the HQR system. As codified at 42 CFR 416.310(f), all deadlines occurring on a Saturday, Sunday, or legal holiday, or on any other day, all or part of which is declared to be a non-workday for Federal employees by statute or Executive Order would be extended to the first business day thereafter. For example, for the first voluntary reporting period, data collected for the Information Transfer PRO-PM from surgical procedures performed January 1, 2027, through December 31, 2027, would be submitted to CMS's HQR system by May 15, 2028. For the first mandatory reporting period, data collected for the Information Transfer PRO-PM from surgical procedures performed January 1, 2029, through December 31, 2029, would be submitted to CMS's HQR system by May 15, 2030, for the CY 2031 payment determination.
We proposed to require ASCs to offer all patients meeting the measure's denominator specifications the opportunity to complete the survey and to report on all completed surveys received. For ASCs that anticipate receiving more than 200 completed surveys, these facilities would have the option to either: (1) survey and report data on their entire eligible Information Transfer PRO-PM patient population, or (2) randomly sample their eligible Information Transfer PRO-PM patient population to collect and report data from 200 completed surveys. Thus, to reduce burden, facilities with large patient populations would have the choice to randomly sample a sufficient number of patients to yield at least 200 completed surveys in a reporting period. ASCs that are unable to collect 200 completed surveys would not be able to perform random sampling and would instead be required to submit data on survey responses from all completed surveys received.
A minimum random sample size of 200 completed surveys would ensure the reliability of the measure, consistent with what is required for the OAS CAHPS measure for ASCs (86 FR 63908 through 63909). We note that under the Hospital OQR Program, a minimum sample size of 300 is required for the Information Transfer PRO-PM as this is a recommended minimum sample size for a population of 1,500 to provide a 95 percent confidence interval for a population of over 10,000; this is also generally accepted as a minimum sample size for stable population estimates.[430 431] However, as ASCs are expected to have less varied populations, we believe the sample size of 200 completed surveys, as determined to be sufficient for the OAS CAHPS survey, is appropriate. The 200 surveys would provide the appropriate balance of ensuring sufficient confidence in the results of the Information Transfer PRO-PM survey, while reducing the overall burden of the survey for facilities with large patient populations.
We invited public comments on this proposal.
Comment: A commenter expressed concern regarding the measure's feasibility as CMS requires a minimum random sample size of 200 completed surveys and asked for clarification regarding the proposal for the minimum random sample size. Another commenter expressed concern that lower-volume facilities would be unable to meet the minimum of 300 survey responses. A commenter noted that extrapolation from the results of the 8-month pilot test in the hospital outpatient department shows that only 38 percent of facilities would have reached the threshold of 200 completed surveys over the course of a year. The commenter stated that this indicates that the threshold of 200 completed surveys is not feasible.
Response: Under the Hospital OQR Program, a minimum random sample size of 300 completed surveys is recommended for a population of 1,500 to provide a 95 percent confidence interval and a 90 percent confidence interval for a population of over 10,000 ( printed page 53969) by reducing the standard error (89 FR 94420 through 954421). As ASCs are expected to have less varied populations, we determined the sample size of 200 completed surveys, which aligns with the sample size for the OAS CAHPS survey, is appropriate. The 200 surveys would provide the appropriate balance of ensuring sufficient confidence in the results of the Information Transfer PRO-PM, while reducing the overall burden of the survey for facilities with large patient populations. We note that we would require data to be submitted on all completed surveys for ASCs unable to achieve 200 completed surveys. However, as discussed in section XVII.B.1. of this final rule with comment period, we are not finalizing our proposal to adopt the Information Transfer PRO-PM into the ASCQR Program measure set at this time and therefore we are also not finalizing our proposed data submission and reporting requirements to be applied to PRO-PMs generally.
Comment: A commenter requested clarification on the apparent discrepancy between permitting sampling when sending the Information Transfer PRO-PM survey and the requirement to offer all patients meeting the measure's denominator specifications the opportunity to complete the survey.
Response: If the Information Transfer PRO-PM had been adopted as proposed, we would have required that all patients that meet the measure's denominator specifications have the opportunity to complete the survey. For facilities that received more than 200 completed surveys, the facility would have the option to randomly sample from these surveys for reporting the measure results. The 200 surveys would provide the appropriate balance of ensuring sufficient confidence in the results of the Information Transfer PRO-PM survey, while reducing the overall burden of the survey for facilities with large patient populations.[432] We note that all questions on the survey must be completed to meet the 200-survey minimum. Partially completed surveys should not be counted towards the 200-survey minimum.
After consideration of public comments, we are not finalizing adoption of the Information Transfer PRO-PM for the ASCQR Program at this time, or our proposed data submission and reporting requirements to be applied to PRO-PMs generally.
2. ASCQR Program Extraordinary Circumstances Exception (ECE) Policy
We refer readers to section XIV.D. of this final rule with comment period for our cross-program policy to codify updates to the ECE policy for the ASCQR Program.
D. Payment Reduction for ASCs That Fail To Meet the ASCQR Program Requirements
1. Statutory Background
We refer readers to the CY 2012 OPPS/ASC final rule with comment period (76 FR 74492 through 74493) for a detailed discussion of the statutory background regarding payment reductions for ASCs that fail to meet the ASCQR Program requirements.
2. Policy Regarding Reduction to the ASC Payment Rates for ASCs That Fail To Meet the ASCQR Program Requirements for a Payment Determination Year
The national unadjusted payment rates for many services paid under the ASC payment system are equal to the product of the ASC conversion factor and the scaled relative payment weight for the APC to which the service is assigned. For CY 2026, the ASC conversion factor is equal to the conversion factor calculated for the previous year updated by the productivity-adjusted hospital market basket update factor. The productivity adjustment is set forth in section 1833(i)(2)(D)(v) of the Act. The productivity-adjusted hospital market basket update was the annual update for the ASC payment system for a 5-year period (CY 2019 through CY 2023), which was extended an additional 2 years (through CY 2025) in the CY 2024 OPPS/ASC final rule with comment period (88 FR 81960). As discussed in section XIII. of this final rule with comment period, we are finalizing our proposal to continue using the productivity-adjusted hospital market basket update as the update factor for the ASC payment system for CY 2026. Under the ASCQR Program, in accordance with section 1833(i)(7)(A) of the Act and as discussed in the CY 2013 OPPS/ASC final rule with comment period (77 FR 68499), any annual increase in certain payment rates under the ASC payment system shall be reduced by 2.0 percentage points for ASCs that fail to meet the reporting requirements of the ASCQR Program. This reduction applied beginning with the CY 2014 payment rates (77 FR 68500). For a complete discussion of the calculation of the ASC conversion factor and our finalized proposal to update the ASC payment rates using the inpatient hospital market basket update for CYs 2019 through 2023, we refer readers to the CY 2019 OPPS/ASC final rule with comment period (83 FR 59073 through 59080).
In the CY 2013 OPPS/ASC final rule with comment period (77 FR 68499 through 68500), in order to implement the requirement to reduce the annual update for ASCs that fail to meet the ASCQR Program requirements, we finalized the following policies: (1) to calculate a full update conversion factor and an ASCQR Program reduced update conversion factor; (2) to calculate reduced national unadjusted payment rates using the ASCQR Program reduced update conversion factor that would apply to ASCs that fail to meet their quality reporting requirements for that calendar year payment determination; and (3) that application of the 2.0 percentage point reduction to the annual update may result in the update to the ASC payment system being less than zero prior to the application of the productivity adjustment. The ASC conversion factor is used to calculate the ASC payment rate for services with the following payment indicators (listed in Addenda AA and BB to this final rule with comment period, which are available via the internet on the CMS website): “A2,” “D2,” “G2,” “P2,” “R2”, and “Z2,” as well as the service portion of device-intensive procedures identified by “J8” (77 FR 68500). We finalized our proposal that payment for all services assigned the payment indicators listed would be subject to the reduction of the national unadjusted payment rates for applicable ASCs using the ASCQR Program reduced update conversion factor (77 FR 68500).
The conversion factor is not used to calculate the ASC payment rates for separately payable services that are assigned status indicators other than payment indicators “A2”, “D2”, “G2,” “J8”, “P2”, “R2”, and “Z2.” These services include separately payable drugs and biologicals, pass-through devices that are contractor-priced, brachytherapy sources that are paid based on the OPPS payment rates, and certain office-based procedures, radiology services, and diagnostic tests where payment is based on the PFS nonfacility PE RVU-based amount, and a few other specific services that receive cost-based payment (77 FR 68500). As a result, we also finalized our proposal that the ASC payment rates for these ( printed page 53970) services would not be reduced for failure to meet the ASCQR Program requirements because the payment rates for these services are not calculated using the ASC conversion factor and, therefore, are not affected by reductions to the annual update (77 FR 68500).
Office-based surgical procedures (generally those performed more than 50 percent of the time in physicians' offices) and separately paid radiology services (excluding covered ancillary radiology services involving certain nuclear medicine procedures or involving the use of contrast agents) are paid at the lesser of the PFS nonfacility PE RVU-based amounts or the amount calculated under the standard ASC ratesetting methodology. Similarly, in the CY 2015 OPPS/ASC final rule with comment period (79 FR 66933 through 66934), we finalized our proposal that payment for certain diagnostic test codes within the medical range of CPT codes for which separate payment is allowed under the OPPS will be at the lower of the PFS nonfacility PE RVU-based (or technical component) amount or the rate calculated according to the standard ASC ratesetting methodology when provided integral to covered ASC surgical procedures. In the CY 2013 OPPS/ASC final rule with comment period (77 FR 68500), we finalized our proposal that the standard ASC ratesetting methodology for this type of comparison would use the ASC conversion factor that has been calculated using the full ASC update adjusted for productivity. This is necessary so that the resulting ASC payment indicator, based on the comparison, assigned to these procedures or services is consistent for each HCPCS code, regardless of whether payment is based on the full update conversion factor or the reduced update conversion factor.
For ASCs that receive the reduced ASC payment for failure to meet the ASCQR Program requirements, we have noted our belief that it is both equitable and appropriate that a reduction in the payment for a service should result in proportionately reduced coinsurance liability for beneficiaries (77 FR 68500). Therefore, in the CY 2013 OPPS/ASC final rule with comment period (77 FR 68500), we finalized our proposal that the Medicare beneficiary's national unadjusted coinsurance for a service to which a reduced national unadjusted payment rate applies will be based on the reduced national unadjusted payment rate.
In the CY 2013 OPPS/ASC final rule with comment period, we finalized our proposal that all other applicable adjustments to the ASC national unadjusted payment rates would apply in those cases when the annual update is reduced for ASCs that fail to meet the requirements of the ASCQR Program (77 FR 68500). For example, the following standard adjustments would apply to the reduced national unadjusted payment rates: the wage index adjustment; the multiple procedure adjustment; the interrupted procedure adjustment; and the adjustment for devices furnished with full or partial credit or without cost (77 FR 68500). We believe that these adjustments continue to be equally applicable to payment for ASCs that do not meet the ASCQR Program requirements (77 FR 68500).
In the CY 2015 through CY 2025 OPPS/ASC final rules with comment period, we did not make any other changes to these policies. We proposed to continue applying these policies for the CY 2026 reporting period/CY 2028 payment determination and for subsequent years.
We did not receive public comments on these policies, and therefore, we are finalizing as proposed the continuation of these policies for the CY 2026 reporting period/CY 2028 payment determination and for subsequent years.
XVIII. Overall Hospital Quality Star Rating Modification To Emphasize the Safety of Care Measure Group
A. Summary
In the CY 2025 OPPS/ASC final rule with comment period (89 FR 94514 through 94521), we summarized broad public input received on a Request for Information (RFI) discussing potential methodologic modifications to the Safety of Care measure group within the Overall Hospital Quality Star Rating that is published on the provider comparison tool on Medicare.gov ( https://www.medicare.gov/care-compare/). The potential modifications discussed in that RFI aimed to emphasize the contribution of the Safety of Care measure group to the Overall Hospital Quality Star Rating. In that RFI, we also noted our intention to potentially issue additional RFIs or undertake rulemaking on this topic in the future.
Patient safety constitutes a fundamental component of the CMS National Quality Strategy, representing a sustained commitment to fostering optimal health outcomes and ensuring the safest possible care for all patients.[433] As we noted in the CY 2025 OPPS/ASC final rule with comment period (89 FR 94514 through 94521), we believe that increasing the influence of the Safety of Care measure group is a necessary and appropriate methodological change. Patient safety is cornerstone to healthcare delivery and the foundational principle of professional oaths is to “do no harm.” Prioritizing safety for both patients and healthcare workers aligns with this fundamental commitment. Considering the public input received and further internal analyses conducted, we proposed to make the following modifications to the Overall Hospital Quality Star Rating methodology: (1) implement a 4-star cap for hospitals in the lowest-performing quartile of the Safety of Care measure group for the 2026 Overall Hospital Quality Star Rating, and (2) implement a blanket 1-star reduction for hospitals in the lowest-performing quartile of the Safety of Care measure group for the 2027 Overall Hospital Quality Star Rating and thereafter. Both the 4-star cap and the blanket 1-star reduction apply to hospitals with at least three measures in the Safety Measure Group.
B. Background
The Overall Hospital Quality Star Rating provides a summary of certain existing hospital quality information on Medicare.gov [434] based on publicly available quality measure results reported through CMS' hospital quality measurement programs, by assigning hospitals between 1 and 5 stars, a way that is simple and easy for patients to understand (85 FR 86193). The Overall Hospital Quality Star Rating methodology was developed and is maintained according to the guiding principles of scientific validity, maximizing inclusion of hospitals and measure information, accounting for heterogeneity of available measures and hospital reporting, accommodating changes in the underlying measures, aligning with CMS hospital quality measure programs to the extent feasible, transparency of the methodology, and responsiveness to input from interested parties. The Overall Hospital Quality Star Rating was first introduced and reported on our Hospital Compare website in July 2016 (now reported on Care Compare on Medicare.gov) and has been published multiple times. In this rule, for the Overall Hospital Quality Star Rating, the term “publish” refers to the public posting of the Overall Hospital Quality Star Rating and “refresh” refers to the public posting quality measure and program data via ( printed page 53971) Care Compare on Medicare.gov or its successor website.
In the CY 2021 OPPS/ASC final rule with comment period (85 FR 86193), we codified the Overall Hospital Quality Star Rating methodology, including several methodology refinements, intended to improve the simplicity and predictability of measure emphasis within the methodology over time, and comparability of ratings among hospitals. We also finalized the inclusion of Veterans Health Administration (VHA) hospitals and Critical Access Hospitals (CAHs) in the Overall Hospital Quality Star Rating. In the CY 2023 OPPS/ASC final rule with comment period (87 FR 72233), we provided additional information on the previously finalized policy to incorporate VHA hospitals and finalized a proposal to amend 42 CFR 412.190 to revise how we would publish the Overall Hospital Quality Star Rating annually. In the CY 2025 OPPS/ASC final rule with comment period (89 FR 94514 through 94521) we summarized public input received on the following potential methodological updates to greater emphasize patient safety in the Overall Hospital Quality Star Rating: (1) Reweighting the Safety of Care Measure Group, (2) Policy-based 1-Star Reduction for Poor Performance on Safety of Care, and (3) Reweighting the Safety of Care measure group combined with a Policy-based Star Rating Cap. We refer readers to section XXIV. (Overall Hospital Quality Star Rating Modification to Emphasize the Safety of Care Measure Group: RFI) of the CY 2025 OPPS/ASC final rule with comment period (89 FR 94514 through 94521) for additional information.
C. Current Overall Hospital Quality Star Rating Methodology (§ 412.190)
Measures reported on the provider comparison tool on Medicare.gov [435] that meet the criteria for inclusion in the Overall Hospital Quality Star Rating are organized into five conceptually coherent measure groups: Safety of Care, Mortality, Readmission, Patient Experience (all of which include outcome measures), and Timely and Effective Care (which includes a selection of process measures).
The current Overall Hospital Quality Star Rating methodology includes eight general steps. First, measures are selected from those publicly reported on Care Compare on Medicare.gov through certain CMS hospital inpatient and outpatient quality programs. Second, the direction of all included measures that indicate better performance with a lower score are reversed to uniformly reflect that a higher score indicates better performance for all the measures, and all measure scores are standardized to a single, common scale to account for differences in measure score units. Third, measures are arranged into measure groups. Each measure group contains several publicly reported measures to produce a robust measure group score, which is reflective of differences in hospital quality. Fourth, the measure group scores are calculated as a simple average of the measure scores. Measure group scores are then standardized to a common scale, making varying scores comparable. Fifth, the hospital summary score is calculated as a weighted average of the standardized measure group scores. Specifically, each measure group score is multiplied by the assigned weight for that measure group. The weighted measure group scores are then summed up to generate the hospital summary score. If a hospital has no measure scores in a measure group (for example, by not achieving sufficient sample size in any of the measures), the weight is redistributed proportionally across the remaining measure groups. Sixth, minimum reporting thresholds are applied. To receive an Overall Hospital Quality Star Rating, hospitals must report at least three measures in each of at least three measure groups, one of which must be either the Mortality or Safety of Care measure groups. Seventh, peer grouping is applied. Hospitals are grouped into one of three peer groups based on the number of measure groups for which they report at least three measures: a three-measure group peer group, a four-measure group peer group, and a five-measure group peer group. Eighth, a clustering algorithm is applied within each peer group to assign hospital summary scores to Overall Hospital Quality Star Ratings so that 1 star is the lowest and 5 stars is the highest.
For additional details regarding the current methodology, we refer readers to § 412.190(d) and the Overall Hospital Quality Star Rating Methodology Reports, available at https://qualitynet.cms.gov/inpatient/public-reporting/overall-ratings/resources.
D. Modification to the Overall Hospital Quality Star Rating Methodology
In the CY 2025 OPPS/ASC final rule with comment period (89 FR 94514 through 94521), we presented three options and analyses (utilizing data from the July 2023 publication of the Overall Hospital Quality Star Rating) for potential methodological updates to emphasize Safety of Care in the Overall Hospital Quality Star Rating and summarized the public comments received. The majority of commenters did not support updating the methodology at that time. While some commenters expressed support for potential changes, there was no consensus on a preferred option (reweighting, the policy-based 1-star reduction, or reweighting combined with the 4-star cap). We refer readers to the CY 2025 OPPS/ASC RFI (89 FR 94514 through 94521), where we detailed the importance of prioritizing Safety of Care within the Overall Hospital Quality Star Rating.
Following the publication of that final rule, we conducted further internal analyses utilizing updated data from the July 2024 publication of the Overall Hospital Quality Star Rating (the most recent publicly released results as of the writing of the CY 2026 OPPS/ASC proposed rule) to reassess the correlation between the Safety of Care measure group and performance in the Overall Hospital Quality Star Rating.
To receive an Overall Hospital Quality Star Rating, hospitals must have at least three measures in each of at least three measure groups, one of which must be Mortality or Safety of Care. However, because the application of minimum reporting thresholds and peer grouping assignment occur strictly after the calculation of measure group scores and overall summary scores, any hospital with at least one measure in any group will have a measure group score for that group—that is, once a hospital meets the Overall Hospital Quality Star Rating reporting threshold, all measure groups for which it has any measure scores are included in its rating. In other words, a hospital with one or two Safety of Care measures can still receive an Overall Hospital Quality Star Rating if it still has at least three measures in Mortality and in two of the other measure groups; in this case, the hospital would still receive a Safety of Care measure group score based on the one or two measures it does have. Only a hospital qualifying for an Overall Hospital Quality Star Rating with zero Safety of Care measures would not have a Safety of Care measure group score.
There were 2,847 hospitals that met the criteria to receive an Overall Hospital Quality Star Rating in 2024. Among the 2,847 rated hospitals, 2,803 (99 percent) had at least one Safety of Care measure and therefore received a Safety of Care measure group score, while 2,475 (87 percent) had at least three Safety of Care measures. Our analysis showed that hospitals in the lowest-performing quartile of the Safety of Care measure group tended to receive lower Overall Hospital Quality Star ( printed page 53972) Ratings (being more likely to receive 1 or 2 stars and less likely to receive 4 or 5 stars than other hospitals) (Table 141). However, some hospitals performed in the lowest quartile (lowest-performing 25 percent, indicating poor Safety of Care performance relative to other hospitals) of the Safety of Care measure group and still received a 5-star rating. Of the 2,847 hospitals that received an Overall Hospital Quality Star Rating, 695 hospitals scored in the lowest quartile of the Safety of Care measure group, of which 595 hospitals had at least three Safety of Care measures. Of these 595 hospitals, 14 received a 5-star rating, representing 0.5 percent of all rated hospitals (Table 141). These 14 hospitals attained a 5-star rating despite having the lowest quartile Safety of Care measure group performance by achieving high scores across the other measure groups.
As we noted in the CY 2025 OPPS/ASC final rule with comment period (89 FR 94514 through 94521), we believe that a methodological change to increase the importance of the Safety of Care measure group is appropriate. This change is informed by landmark reports on healthcare quality,[436 437] along with the COVID-19 public health emergency, which revealed persistent patient and workforce safety risks and system vulnerabilities.[438] In response, Federal efforts—such as the National Action Alliance to Advance Patient and Workforce Safety and recommendations from the President's Council of Advisors on Science and Technology—are reinforcing patient safety as a national priority, aligned with CMS' initiatives like the National Quality Strategy and the Universal Foundation.[439 440 441] In particular, addressing the issue of hospitals receiving a high Overall Hospital Quality Star Rating despite performing in the lowest quartile of the Safety of Care measure group is critical to achieving CMS' vision of emphasizing and aligning the importance of patient safety across CMS programs. We therefore proposed to make the following two-stage methodologic updates to § 412.190(a)(2) and adding a new paragraph (a)(3)); the first stage would be a narrow but focused transitional step to promptly address the most pressing concern that hospitals in the lowest-performing quartile of the Safety of Care measure group achieve the highest possible Overall Hospital Quality Star Rating while allowing hospitals and interested parties more time to prepare for the second stage, which will increase the impact of the Safety of Care measure group across all hospitals more broadly.
We also proposed changes to paragraphs (b)(1), (e) and (f) to reflect updates to the regulation text uses of Overall Hospital Quality Star Rating and Care Compare on Medicare.gov language. In addition, we proposed removing the reference to “as defined in § 400.200 of this chapter.”
For the methodologic updates:
Stage 1: Implement a 4-Star Cap for Hospitals in the Lowest Quartile of the Safety of Care Measure Group Performance Beginning in 2026 (§ 412.190(d)(9)(i))
We proposed to limit hospitals in the lowest quartile of Safety of Care (based on at least three measure scores) to a maximum of 4 stars out of 5. The Overall Hospital Quality Star Rating methodology would be unchanged through step eight with the exception of redesignating paragraph (d)(5) as (6), and paragraph (d)(6) as (5) (assignment of star ratings using K-means clustering as described previously in this section), with the cap being applied as a new “step nine”: Any hospital that is assigned 5 stars in step eight but has a lowest quartile Safety of Care score (based on at least three Safety of Care measures) would be reassigned to 4 stars.
Using 2024 Overall Hospital Quality Star Rating data, implementing a cap of 4 stars in the lowest quartile of Safety of Care with at least three safety measures would result in 14 hospitals, out of 2,847 hospitals, receiving a lower Overall Hospital Quality Star Rating. This update provides a targeted, direct, and timely solution to the acute concern of hospitals receiving the highest possible 5-star rating despite performing in the lowest quartile of the Safety of Care measure group. Further, the implementation timeline reflects a deliberate and proactive effort to act swiftly and strategically, reinforcing patient safety as a national priority. ( printed page 53973)
We acknowledge in the CY 2025 OPPS/ASC final rule with comment period that only applying a 4-star maximum to hospitals in the lowest quartile of Safety of Care with at least three safety measures would have less impact than other options discussed in that rule. However, to promptly address the most pressing concern, the proposed 4-star maximum functions as an interim step, allowing hospitals and interested parties additional time to prepare for Stage 2:
Stage 2: Implement a Blanket 1-Star Reduction for Hospitals in the Lowest Quartile of Safety of Care Measure Group Performance for the 2027 Overall Hospital Quality Star Ratings and Later Years (§ 412.190(d)(9)(ii))
We proposed to reduce the Overall Hospital Quality Star Rating of any hospital in the lowest quartile of Safety of Care (based on at least three measure scores) by 1 star, to a minimum 1-star rating. The Overall Hospital Quality Star Rating methodology would be unchanged through step eight (assignment of star ratings using K-means clustering), with the blanket reduction replacing the 4-star cap in the new step nine: any hospital assigned a 2, 3, 4, or 5-star rating in step eight, but that has a lowest quartile Safety of Care score (based on at least three Safety of Care measures) would be reduced to 1, 2, 3, or 4 stars, respectively.
Using 2024 Overall Hospital Quality Star Rating data, applying a 1-star reduction for all hospitals in the lowest quartile of Safety of Care with at least three safety measures would result in 459 hospitals, out of 2,847 hospitals, receiving a lower Overall Hospital Quality Star Rating. This update would emphasize safety by applying a higher standard for patient safety to hospitals across a broad range of overall performance, rather than limiting it to the few 5-star hospitals in the lowest quartile of Safety of Care (with at least three Safety of Care measures). Since the minimum possible Overall Hospital Star Rating will remain 1 star, hospitals already getting one star would not get a further star reduction and therefore would effectively be exempt from this adjustment consistent with established assignment of ratings between 1-5 whole stars (85 FR 86193). This approach also aligns with CMS' overarching objective of advancing patient safety and reinforcing our commitment to continuous improvement across the healthcare system.
When determining the quartiles of Safety of Care measure group scores, we will use the distribution from all hospitals with at least 1 Safety of Care measure whether they qualify for an Overall Hospital Quality Star Rating, in alignment with the guiding principle of the Overall Hospital Quality Star Rating of inclusiveness of hospital and measure information.
Using the data for the July 2024 Overall Hospital Quality Star Rating, we evaluated the proportion of hospitals that would be impacted by the proposed methodological changes, stratified by various hospital characteristics (Table 142). As previously noted, a larger proportion of hospitals would be impacted by the blanket 1-star reduction (Stage 2 proposed methodological change) compared to the targeted 4-star cap (Stage 1 proposed methodological change). Our simulation revealed that teaching hospitals, non-safety-net hospitals, VHA hospitals, non-CAHs, large hospitals (100+ beds), urban hospitals, and non-specialty hospitals could be more likely to observe a change in Overall Hospital Quality Star Rating by both Stage 1 and Stage 2 proposed methodological changes than their counterparts. We recognize that with only 14 hospitals experiencing a change in Overall Hospital Quality Star Rating by the Stage 1 proposed methodological change, the general ability of this observation is limited. In part, this is because these hospitals are more likely to receive an Overall Hospital Quality Star Rating and have three or more Safety of Care measures than their non-teaching, safety-net, non-VHA, CAH, small, rural, and specialty counterparts (Table 142). However, these differences in hospital characteristics are not strongly determinative of a hospital's overall rating, with hospitals of any characteristic being capable of receiving either high or low ratings.
( printed page 53974)We received public comments on these proposals. The following is a summary of the comments we received and our responses. ( printed page 53975)
Comment: Many commenters support our proposal to emphasize the Safety of Care measure group within the Overall Hospital Quality Star Rating methodology utilizing a two-stage approach, which would implement a 4-star cap in CY 2026 for hospitals in the lowest quartile of Safety of Care measure group performance (and with at least three Safety of Care measures), followed by a uniform 1-star reduction for hospitals in the lowest quartile of Safety of Care measure group performance (and with at least three Safety of Care measures) beginning in CY 2027. Commenters agreed that patient safety is foundational to assessing hospital quality and should be heavily emphasized in the Overall Hospital Quality Star Rating. Commenters stated increasing the weight of Safety of Care aligns the Overall Hospital Quality Star Rating with patient priorities and would encourage hospitals to invest in safety improvements, thereby increasing transparency and accountability across healthcare systems, and incentivizing improvements in patient safety. One commenter anticipates the proposed updates will support the goals of Pay for Performance programs (like Hospital-Acquired Condition Reduction Program (HACRP)), drive improvements across other Overall Hospital Quality Star Rating measure groups and encourage policies that improve patient satisfaction and promote timely care, while maintaining a strong focus on quality metrics and transparency.
Response: We thank commenters for their support of the proposed modification to the Overall Hospital Quality Star Rating methodology which aligns with our vision to emphasize the importance of patient safety across CMS programs.
Comment: In addition to general support, commenters offered several specific suggestions and considerations. Commenters supported CMS's efforts to strengthen the role of patient safety in the Overall Hospital Quality Star Rating and urged the agency to proceed thoughtfully, with continued interested party engagement and a focus on equitable, transparent implementation. A few commenters recommended that CMS conduct additional testing or simulation of the proposed methodology before full implementation, to allow for refinement and avoid confusion or disincentivizing reporting. A few commenters requested more information and early notification on hospital performance on the Safety of Care measure group to support course correction ahead of public reporting. One commenter highlighted the potential for disproportionate impact on small, rural, and safety-net hospitals and encouraged CMS to monitor these effects during implementation. One commenter expressed concern that hospitals could avoid submitting Safety of Care measures to maintain a higher Overall Hospital Quality Star Rating and recommended awarding a 4- or 5-star rating only to hospitals that submit at least three measures from the Safety of Care group.
A number of commenters suggested alterations to the proposed policy. A few commenters recommended capping hospitals in the lowest quartile of Safety of Care at no more than 2 stars or prohibiting 4- or 5-star ratings altogether, for hospitals with poor patient safety performance. These commenters emphasized that facilities with a pattern of avoidable harm should not be classified as “average” or “above average” in overall quality. Additionally, one commenter advocated for CMS to ensure that the Overall Hospital Quality Star Rating is reported at the individual facility (CCN) level, rather than at the health system level, to ensure that patients receive accurate and meaningful information about the specific hospitals where they receive care.
A few commenters encouraged CMS to support data infrastructure improvements, such as automating quality reporting, aligning state reporting standards, and exploring use of the Trusted Exchange Framework and Common Agreement (TEFCA) for data sharing. One commenter noted the role of occupational therapy in reducing patient harm and improving Safety of Care outcomes and encouraged CMS to consider how multidisciplinary interventions contribute to improved patient safety performance.
Response: We thank commenters for supporting our proposal. We appreciate commenters' suggestions to conduct additional testing before implementation and to provide additional Safety of Care measure group information; however, we disagree that additional testing is needed. We refer commenters to the testing results in this rule, as well as the results calculated using the July 2023 Overall Star Rating as reported in the CY 2025 OPPS/ASC final rule with comment period (89 FR 94514 through 94521) which produced similar findings. We also appreciate the concern expressed regarding small, rural, and safety-net hospitals. We note that in our July 2024 Overall Hospital Quality Star Rating simulated results, hospitals with these characteristics were less likely to observe a change in their rating through Stage 1 and Stage 2 compared to their counterparts (90 FR 33787). We intend to monitor how different types of hospitals, such as small, rural, and safety-net hospitals, may be affected by this methodology update, as we have throughout the ongoing maintenance of the Overall Hospital Quality Star Rating.
We appreciate the commenter's suggestion to report the Overall Hospital Quality Star Rating at the CCN (facility) level rather than the health system level; while this change is not feasible at this time, CMS will consider future reevaluation work related to this suggestion. As noted in the CY 2021 OPPS/ASC final rule with comment period (85 FR 85866), “hospitals sharing the same CCN must combine data collection and submission across their multiple campuses for all clinical measures for public reporting purposes. Under our current policy, we publish quality data by the corresponding hospital CCN and indicate instances where data from two or more hospitals are combined to form the publicly reported measures on the Hospital Compare website and the successor Care Compare website. In the CY 2014 OPPS/ASC proposed rule ( 78 FR 43645 ), we noted that in a situation in which a larger hospital has taken over ownership of a smaller hospital, the smaller hospital's CCN is replaced by the larger hospital's CCN (the principal CCN). For data display purposes, we only display data received under the principal CCN. If both hospitals submit data, those data are not distinguishable in the warehouse [107] and are calculated together as one hospital” (85 FR 86182).
We appreciate the commenter's concern that hospitals may want to avoid reporting unfavorable Safety of Care results to avoid receiving a lower Overall Hospital Quality Star Rating. The Overall Hospital Quality Star Rating explicitly includes only measures for which all hospitals are required to collect and submit data by CMS's hospital quality reporting & payment programs. Each underlying measure has established thresholds (such as minimum case count) for a hospital's score to be publicly reported to ensure reliable measurement; if a hospital does not meet the threshold for a measure, its score is not publicly reported. In this case its score is considered “missing” and its performance does not factor into its Overall Hospital Quality Star Rating. We note this determination is made exclusively by CMS after all data are submitted; in general, hospitals cannot pick and choose which measures of the Overall Hospital Quality Star Rating they submit, and cannot choose to have ( printed page 53976) any measure withheld from public reporting.
We thank commenters for their suggestion to cap hospitals in the lowest quartile of Safety of Care at no more than 2 stars or prohibiting 4- or 5-star ratings altogether to prevent these hospitals from being categorized as “average” or “above average” in overall quality. We emphasize that while this update will increase the emphasis on Safety of Care, the Overall Hospital Quality Star Rating is still intended to be a summary of overall quality, and other measure groups will still contribute meaningfully to a hospital's rating.
We appreciate the comment regarding TEFCA. It is not addressed here, as it is out-of-scope for this methodology update. We also appreciate the commenter's suggestion to consider the role of occupational therapy in reducing patient harm and how other clinical interventions can improve Safety of Care. This proposed Overall Hospital Quality Star Rating methodology update is meant to complement and support other efforts within CMS to drive improvements in patient safety, most notably the HACRP. We believe this update will further encourage hospitals to explore such means to improve safety.
Comment: A few commenters discussed specific concerns with the potential disproportionate impact of the proposed approach on small and rural hospitals. One commenter explained that these hospitals often have limited Safety of Care measures available for reporting, making the Safety of Care measure group heavily weighted in their Overall Hospital Quality Star Rating. A few commenters stated that emphasis on this measure group would skew overall ratings and diminish the value of the system for small and rural hospitals. A few commenters noted that rural facilities also face resource constraints and challenges related to staffing and infrastructure, which may force them to divert resources away from other critical measure groups to prioritize Safety of Care. One commenter noted that rural hospitals already contend with physician shortages, maternity care deserts, and ongoing obstetric unit closures, and cautioned that capping or reducing these hospitals' Overall Hospital Quality Star Rating could exacerbate rural health disparities, particularly in maternal health outcomes. One commenter noted that applying a uniform star rating reduction across all hospitals could misrepresent performance and deepen existing disparities in how rural hospital quality is perceived. A few commenters recommended exempting hospitals that report only one or two Safety of Care measures from the proposed 1-star reduction and instead adopt a stratification or rural-specific risk adjustment to avoid disadvantaging these hospitals.
Response: We thank commenters for their concerns regarding the impact of the proposed update to the Overall Hospital Quality Star Rating methodology for small and rural hospitals. As demonstrated in Table 142, there are high, average, and low performers of all hospital types both in Safety of Care and in the Overall Hospital Quality Star Rating more generally. As demonstrated in Table 142, using July 2024 simulation data, 1 percent of CAHs were rated and reported at least three Safety of Care measures compared to 75 percent of non-CAH's. Similarly, 35 percent of rural hospitals were rated and reported at least three Safety of Care Measures compared to 75 percent of urban hospitals. Furthermore, comparing the “N Rated & 3+ Safety measures” and “N Rated & 3+ measures & Q1 Safety” columns in Table 142, shows that among those meeting the criteria outlined in those columns, there were close to 25 percent of hospitals performing in the bottom quartile of Safety of Care regardless of hospital type. Therefore, there is not a strong association between hospital type and actual Safety of Care performance.
Comment: A few commenters suggested that hospitals with greater patient volumes scored on more Safety of Care measures (particularly larger facilities such as teaching hospitals or VHA hospitals) may have higher potential for score reductions, not because of poor quality, but because they are more exposed to scoring variation; or, conversely, that hospitals without enough data to calculate performance on Safety of Care measures may avoid Overall Hospital Quality Star Rating reductions altogether.
Response: We thank commenters for their concerns regarding the potential impact of the proposed update to the Overall Hospital Quality Star Rating methodology for larger hospitals. As demonstrated in Table 142, there are high, average, and low performers of all hospital types both in Safety of Care and in the Overall Hospital Quality Star Rating more generally. While structural differences may exist for larger, higher-acuity hospitals (such as VHA hospitals and academic medical centers) that could make improved Safety of Care performance more difficult to achieve, we note that these hospitals do tend to both have more Safety of Care measures available and larger volumes for the measures they do have, both factors that increase measure reliability and reduce the possibility of score reductions based on chance variation.
Comment: A few commenters stated that hospitals caring for a significant proportion of high-acuity and complex patients (such as teaching hospitals and VHA hospitals) could be disproportionately affected by higher numbers of adverse events captured in the Safety of Care measures, even if overall quality is high. One commenter suggested that Safety of Care measures may not sufficiently account for clinical complexity, particularly for large tertiary hospitals and those performing high volumes of complex cases, which could obscure meaningful quality differences and discourage hospitals from treating high-risk patients. The commenter recommended implementation of additional safeguards such as more detailed risk adjustment, procedure-specific stratification, clear definitions of complications, and a peer review or appeals process before automatically reducing a hospital's rating. One commenter suggested that a blanket 1-star reduction might disproportionately affect institutions caring for vulnerable populations such as: hospitals serving older adults, patients with chronic conditions, or communities with limited access to post-acute care. The commenter was concerned this could suppress patient volume, exacerbate disparities, and create challenges in recruiting clinical talent, ultimately undermining patient safety outcomes.
Response: We thank commenters for their concerns regarding the impact of the proposed update to the Overall Hospital Quality Star Rating methodology for hospitals with higher-acuity or more clinically complex patients. We note that all individual measures comprising the Safety of Care group are already risk adjusted for clinical factors at the measure level prior to reporting, and thus such differences are already implicitly factored into the Safety of Care measure group score. For example, “calculations for the Hospital-Associated Infection (HAI) measures adjust for differences in the characteristics of hospitals and patients using a Standardized Infection Ratio.” [442] This increased emphasis on Safety of Care supports other ongoing efforts within CMS to promote patient safety, chiefly including the HACRP, ( printed page 53977) and aligns with CMS's intent to incentivize improvements in patient safety for all patients. Finally, regarding a commenter's suggestion to implement a peer review or appeals process before applying the 4-star cap or 1-star reduction, we do not believe a peer review or an appeals process is necessary since hospitals can use preview periods to review the methodology and their results, as well as reach out with questions. Since the Overall Hospital Quality Star Rating is calculated using individual measures publicly reported via Care Compare on Medicare.gov, hospitals can use established processes under each program to review and correct individual measure scores. Given that the Overall Hospital Quality Star Rating is published annually, there are regular opportunities for hospitals to improve their performance.
Comment: A few commenters suggested that reputational penalties for facilities could exacerbate financial consequences, particularly as the Overall Hospital Quality Star Rating is increasingly tied to payer contracts and value-based payment programs.
Response: We appreciate commenters' concerns regarding reputational and financial impacts of the Overall Hospital Quality Star Rating. We want to emphasize that the Overall Hospital Quality Star Rating is a summary of quality measurements that are already publicly reported as part of CMS hospital quality reporting & payment programs, as a simple, easily interpreted score to assist consumers and the public in assessing a hospital's overall quality. While the individual measures comprising the Overall Hospital Quality Star Rating factor into payment determination of those quality programs, the Overall Hospital Quality Star Rating itself does not have any direct impact on CMS payment.
Comment: Several commenters opposed adopting a quartile-based methodology for categorizing performance in the Safety of Care measure group. Commenters stated that the quartile approach is statistically flawed, arbitrary, and not aligned with the purpose of assessing hospital safety. A few commenters noted that quartiles reflect only relative ranking, not performance against an absolute or meaningful standard. A few commenters noted under this method, hospitals may be labeled as “low performers” even if they demonstrate strong safety outcomes, while hospitals ranked in the top quartile may not necessarily meet high safety benchmarks. A few commenters expressed that this creates volatility, as a hospital's placement could shift between quartiles based on small changes in peer performance rather than meaningful changes in its own outcomes.
A few commenters proposed replacing the quartile-based approach with a system based on fixed, evidence-based benchmarks that reflect meaningful differences in performance, rather than rankings relative to peers. A few commenters recommended using thresholds based on standard deviations below the mean, which would limit 1-star reductions to a smaller proportion of hospitals and more precisely target those with significantly lower Safety of Care performance. A few commenters highlighted that the impact of the proposed quartile-based 1-star reduction would be disproportionately broad. They stated that reducing ratings for nearly 20 percent of hospitals is excessive relative to the identified issue and supported alternative statistical methods to more precisely identify poor performers or outliers.
A few commenters also identified a concern with the calculation of the lowest quartile as described in the CY 2026 OPPS/ASC proposed rule. One commenter noted that the calculation included hospitals with fewer than three Safety of Care measures in the distribution and suggested that only hospitals with at least three measures be considered when calculating the quartiles to produce a more accurate assessment of performance, reduce volatility, and increase comparability in future public reporting.
Response: We thank commenters for expressing their concerns regarding the quartile-based methodology for categorizing performance in the Safety of Care measure group. We acknowledge that using a quartile-based approach for the 1-star reduction could sometimes result in hospitals receiving a lower Overall Hospital Quality Star Rating even if overall performance improved. We reiterate the foundational principle of “do no harm” in our rationale for making this methodology update to the Safety of Care measure group. The quartile approach to define poor performance emphasizes patient safety globally; all hospitals should be working to produce safer outcomes and minimize adverse events in their facilities. Additionally, we retire “topped out” measures on which most hospitals have come to perform highly (in which case such measures would no longer be eligible for the Overall Hospital Quality Star Rating). Currently no Safety of Care measures approach topped-out status, indicating continued room for improvement.
We appreciate commenters' suggestions to replace the quartile-based approach with a fixed benchmark or statistical outlier approach, as well as suggestions that the quartile-based approach would impact too many hospitals. We believe the quartile-based approach in conjunction with the requirement of at least three Safety of Care measures provides a suitable balance between emphasizing Safety of Care broadly across all hospitals (not just at a few extremely low performers) while also ensuring that only hospitals with demonstrably below-average performance would be subject to Overall Hospital Quality Star Rating reduction.
We acknowledge commenters' concern that, in the context of the 1-star reduction methodology update, the quartiles are inclusive of all hospitals that report at least one Safety of Care measure (even though only hospitals that report at least three Safety of Care measures will be subject to the 1-star reduction). We base the quartile calculation on the full Safety of Care measure group score distribution in order to be inclusive of the national Safety of Care landscape (including hospitals with Safety of Care data that do not receive an Overall Hospital Quality Star Rating). This is in alignment with the Overall Hospital Quality Star Rating's guiding principles of inclusiveness of quality information. We also note that this accommodates potential for collective improvement—if hospitals with at least three Safety of Care measures improve in performance relative to those with one or two, then fewer than 25 percent of those with at least three measures would find themselves in that lowest overall quartile. Conversely, if using only rated hospitals with at least three Safety of Care measures for the distribution, by definition 25 percent of all such hospitals would be subject to reduction.
Comment: Several commenters raised concerns that this proposed update to the Overall Hospital Quality Star Rating overvalues the safety relative to other aspects of hospital quality that are also important to patients. Additionally, commenters stated that singling out the Safety of Care measure group for additional emphasis over the other measure groups does not accurately reflect overall hospital quality.
A few commenters also stated concern that the Overall Hospital Quality Star Rating already struggles to summarize diverse domains of care into a single composite score and the proposed updates may exacerbate these limitations. Commenters stated that further increasing the influence of the Safety of Care measure group would ( printed page 53978) undermine the balance among measure groups, potentially downgrading hospitals that perform well in other areas of quality. A few commenters stated that this could misrepresent overall quality, unfairly penalize hospitals addressing isolated safety issues, and erode public trust in the ratings.
A few commenters expressed concern that this approach could distort results, limit the value of the ratings for patients, and fail to drive meaningful performance improvement, particularly given that many hospitals already receive two- or three-star ratings that signal a need for improvement in multiple respects.
Response: We appreciate commenters' concern that the proposed methodology updates could exacerbate perceived limitations of the Overall Hospital Quality Star Rating, and acknowledge concerns related to the general concept of the Overall Hospital Quality Star Rating serving as a quality summary based on a diverse set of measures.
We also appreciate commenters' concerns regarding the proposed methodology updates and how emphasizing the Safety of Care measure group through the Star Cap and 1-star reduction in turn deemphasizes the influence of the other four measure groups. We note that even hospitals in the lowest quartile of Safety of Care can still achieve ratings as high as 4 stars in this proposal based on strong performance in the other measure groups; while this proposed update increases the emphasis on Safety of Care relative to other measure groups, it does not drastically alter the overall approach.
We reiterate that “. . . Federal efforts—such as the National Action Alliance to Advance Patient and Workforce Safety and recommendations from the President's Council of Advisors on Science and Technology—are reinforcing patient safety as a national priority, aligned with CMS' initiatives like the National Quality Strategy and the Universal Foundation.[265 266 267].” The proposed methodology updates better align the Overall Hospital Quality Star Rating with other CMS efforts.
Patients are advised through Care Compare on Medicare.gov to consider a variety of factors beyond the Overall Hospital Quality Star Rating when choosing a hospital. The Overall Hospital Quality Star Rating is intended to serve as a complementary tool to existing efforts (such as the Hospital Consumer Assessment of Healthcare Providers and Systems (HCAHPS) star ratings (implemented in April 2015)) and will not replace the reporting of any individual quality measures.
Comment: A few commenters raised concerns that the Safety of Care measure group is based on a limited set of measures, which could reduce comparability across hospitals and may not fully capture the complexity of patient safety performance. A few commenters noted that the current measure set is narrow in scope and predominantly inpatient-focused, making the Safety of Care measure group less applicable to many hospitals; particularly low-volume rural and small hospitals may be unable to report on many of the included measures. One commenter encouraged CMS to identify patient safety measures that are relevant to a broader set of hospitals, including outpatient and observation settings. Another commenter recommended adding additional measures that capture broader dimensions of safety, such as patient safety indicators (PSIs), perinatal and maternal health measures, and Hospital Harm measures (contingent upon validation and interested party vetting). The commenter also recommended breaking down the PSI-90 composite into its individual component measures and weighing them appropriately to provide a more nuanced view of patient safety. Another commenter noted that the composition of the Safety of Care measure group is subject to change as CMS adds or removes measures from the various hospital quality reporting & payment programs.
Response: We thank commenters for their feedback on the composition of the Safety of Care measure group. As noted, the Overall Hospital Quality Star Rating summarizes existing measures that are already publicly reported via Care Compare on Medicare.gov as required by CMS. Accordingly, new measures are only added to the Overall Hospital Quality Star Rating after being first added to Care Compare on Medicare.gov (the requirements and timing of which are established by the relevant CMS program) and meeting established inclusion criteria. The Overall Hospital Quality Star Rating methodology is designed and intended to be flexible and accommodate changes to the measures publicly reported through Care Compare on Medicare.gov as the universe of measures required and reported via these CMS programs evolves.
Comment: A few commenters also emphasized that hospitals may be evaluated on different subsets of measures, which they believed undermined comparability and fairness in Overall Hospital Quality Star Rating. One commenter raised specific concerns regarding the comparability of ratings between large hospitals (with more Safety of Care measures) and small hospitals (with fewer).
Several commenters urged CMS to refine its approach by incorporating improved risk adjustment and stratification and ensuring that modifications to the Overall Hospital Quality Star Rating methodology do not unfairly affect hospitals based on facility type, patient acuity, or mission. Several commenters criticized the lack of adequate risk adjustment for social and demographic factors, noting that hospitals serving higher-risk, underserved, or rural populations could be unfairly affected despite delivering high-quality care. Commenters stated that this could lead to unintended consequences, such as exacerbating disparities or discouraging care for vulnerable populations.
Response: We thank commenters for their concerns regarding comparability of the Overall Hospital Quality Star Rating due to variation in hospital size and other contextual factors. We would like to note that “peer grouping” [Step 7 in the methodology] hospitals based on the number of measure groups for which they report at least three measures is intended to improve comparability of Overall Hospital Quality star ratings by accounting for differences in measure information. Peer grouping is applied independent of the measure and measure group reporting threshold and would therefore not result in any reduction in the number or type of hospitals receiving star ratings or the number or type of measures or measure groups contributing to hospital scores” (85 FR 86232). Therefore, peer grouping ensures hospitals are compared to other hospitals with similar quality measure information on an “apples-to-apples” basis. Furthermore, we acknowledge concerns that not all hospitals are scored on the same set of Safety of Care measures but note that the current methodology is designed to accommodate such differences in alignment with our guiding principle of inclusiveness of hospitals and measure information. In other words, the intention of the Overall Hospital Quality Star Rating is to summarize quality information that each hospital does in fact have publicly available, with each measure in a given group reflecting a common underlying construct.
We also thank commenters for their suggestions related to risk adjustment for social and demographic factors. As noted in the 2021 OPPS/ASC final rule with comment period, “In the past, we have not stratified or adjusted any of the ( printed page 53979) measures, measure groups, summary scores, or star ratings by social risk factor variables within the Overall Hospital Quality Star Rating methodology, primarily based on the original guiding principles of the Overall Hospital Quality Star Rating. The Overall Hospital Quality Star Rating is meant to summarize the existing quality measure information that is publicly reported through CMS programs, including Hospital IQR Program, Hospital OQR Program, HRRP, HAC Reduction Program, and Hospital VBP Program, on Hospital Compare or its successor websites. Individual measures undergo rigorous development and reevaluation processes under each program that include extensive analytic testing and interested parties engagement. As such, individual measure methodologies as specified under each program, including approaches to risk adjustment, are included within the Overall Star Rating. As measure data and methodologies are updated under each of the programs, they are subsequently reflected within the Overall Star Rating methodology” (85 FR 86218).
Comment: Several commenters suggested the proposal is methodologically flawed and not proportional to the issue it seeks to address. One commenter emphasized that the changes are being driven by a narrow concern affecting only 14 hospitals (0.5 percent of rated facilities) and do not justify a sweeping modification to the entire Overall Hospital Quality Star Rating framework. The commenter stated that the Overall Hospital Quality Star Rating system already oversimplifies hospital quality and that further emphasizing a single domain, particularly through penalties, risks distorting the meaning of the rating and diminishing its usefulness to patients.
A few commenters stated that the proposal would not lead to meaningful safety improvements and may instead confuse patients by implying that a lower Overall Hospital Quality Star Rating indicates poor safety, when the methodology may not support that conclusion. One commenter urged CMS to reconsider the proposed approach in favor of one that is more equitable, transparent, and methodologically sound.
Response: We thank commenters for their concern regarding whether the proposed Overall Hospital Quality Star Rating methodology updates adequately address the overall CMS goal of prioritizing patient safety across programs. We would like to highlight that, while eliminating the possibility of hospitals achieving a 5-star rating despite being in the lowest quartile of Safety of Care is part of the intent of this update, it is not the sole purpose; through the Stage 2 methodology update (1-star reduction), we are recognizing the broader Federal effort to reinforce patient safety as a national priority across hospitals more globally.
We appreciate a commenter's concern that the Overall Hospital Quality Star Rating oversimplifies hospital quality and that emphasizing Safety of Care distorts the intention of the Overall Hospital Quality Star Rating and diminishes its usefulness. However, we disagree and note that the Overall Hospital Quality Star Rating has always been intended to be an accessible, easily interpreted summary of available hospital quality information. Established measure groups and respective weightings were developed and maintained with interested parties, expert and public input upholding an Overall Hospital Quality Star Rating guiding principle of transparency.
We appreciate commenters' concern that some consumers may equate a lower Overall Hospital Quality Star Rating with poor Safety of Care performance. While it is true there will be cases in which a hospital receives a low Overall Hospital Quality Star Rating despite very good Safety of Care performance, this can be achieved only by having consistently poor performance across the other measure groups. We intend the Overall Hospital Quality Star Rating to remain a summary of all of a hospital's quality information; while this update will increase the emphasis on Safety of Care, the other measure groups will remain integral in determining a hospital's ultimate rating.
Comment: A few commenters encouraged CMS to adopt a more incremental and transparent approach to implementing the proposed methodologic updates to the Overall Hospital Quality Star Rating. Commenters expressed concerns about the timing and phased approach to implementing the proposed changes, recommending that CMS delay the proposal to allow hospitals sufficient time to respond and improve. A few commenters urged CMS to adopt a forward-looking approach to implementation, recommending that the proposed changes, particularly the star cap in 2026 and 1-star reduction in 2027, be delayed until at least 2028 to allow hospitals sufficient time to respond to the new methodology and improve safety performance. The commenters emphasized that the use of lagged data does not reflect current hospital safety performance, urging CMS to incorporate more current or near-real-time data sources such as electronic clinical quality measures (eCQMs) for public reporting. One commenter expressed concern that outdated or poorly contextualized data on Care Compare on Medicare.gov may mislead patients and harm hospitals' reputations, particularly when the timeframes for individual measures are inconsistent or poorly labeled. Another commenter stated if the proposal is finalized, hospitals should be provided with timely preview reports clearly indicating whether a hospital's Overall Hospital Quality Star Rating was downgraded due to Safety of Care performance.
Response: We acknowledge commenter's concern about outdated data and collection periods that do not always align across measures. However, the data collection period and refreshed timelines are established on a measure-by-measure basis so that results are released as soon as possible, while also enabling sufficient data collection for measure reliability and accounting for the time necessary to collect, process, and validate data and results.
We acknowledge commenters' concern with the implementation timing of the proposed Overall Hospital Quality Star Rating methodology updates. We believe the proposed approach is already an incremental process to first apply the 4-star cap in CY 2026 followed by the 1-star reduction in CY 2027. Furthermore, the Safety of Care RFI included in the CY 2025 OPPS/ASC Proposed Rule (89 FR 94514 through 94521) provided the public with information about potential methodology updates that were being considered by CMS. While we acknowledge concerns that a hospital may not have the opportunity to respond to performance results between the implementation of the star cap and the 1-star reduction, we note that the Safety of Care measure group and its component measures have been a prominent part of the Overall Hospital Quality Star Rating since its inception, with hospitals' Safety of Care performance already contributing substantially to their Overall Hospital Quality Star Rating. Furthermore, hospitals should always be striving to uphold patient safety in their care settings given the foundational principle of “Do no harm.”
We also acknowledge commenters' concern with the use of lagged data in the Overall Hospital Quality Star Rating; We note that the challenge of the lag between the data collection period, the public reporting of individual measures, ( printed page 53980) and the publication of the Overall Hospital Quality Star Rating is well known. The main driver of the gap is the data collection periods for the individual measures which is outside the purview of the Overall Hospital Quality Star Rating methodology, and often necessary as longer periods are needed for smaller volume hospitals. It takes longer periods of time to get sufficient samples for statistical stability for the risk-adjusted measures. Therefore, as noted in prior rules, “. . . the data collection period for each measure varies depending on measure specifications that set minimum case requirements to ensure individual measure reliability and meet the requirements of CMS quality programs, as detailed in each program's respective rules as well as on Hospital Compare or its successor website” (85 FR 86202). Regarding eCQMs specifically, as these measures are required for reporting through Care Compare on Medicare.gov, they will be assessed for measure inclusion criteria in the Overall Hospital Quality Star Rating. However, we want to highlight that data collection periods for eCQMs (as for all measures) are not dictated by the Overall Hospital Quality Star Rating but rather by their respective CMS quality program; the Overall Hospital Quality Star Rating remains a summary of quality data that has already been publicly reported.
We appreciate commenters' recommendations regarding hospitals being provided with preview reports that indicate if a 1-star reduction was applied. As in prior publications of the Overall Hospital Quality Star Rating, CMS will continue to provide Hospital-Specific Reports and supporting resources during the preview period to help hospitals understand and interpret their results.
Comment: A few commenters raised concerns that the proposed policy's complexity and punitive nature may confuse patients and providers and erode trust in the Overall Hospital Quality Star Rating system as a reliable public reporting tool. One commenter specifically expressed concern that the Stage 2 approach could cause sudden declines in ratings that patients and consumers may not understand. The commenter stated that CMS already uses other programs, such HACRP, to incentivize safety improvement and that layering additional reputational penalties may duplicate or conflict with existing incentives. The commenter added that focusing on modifying the Overall Hospital Quality Star Rating methodology itself does little to improve how safety is actually evaluated or advanced.
A few commenters called for increased transparency around the methodology including clearer documentation on how hospitals are grouped into peer comparisons, how quartiles are calculated, and how measure groups contribute to the Overall Hospital Quality Star Rating, to help hospitals anticipate and manage performance. A few commenters provided similar suggestions related to public documentation describing how patient safety measures are selected for each hospital and how performance distributions affect the Overall Hospital Quality Star Rating year after year. Commenters also recommended enhancing public education efforts to highlight what the Overall Hospital Quality Star Rating represents, preventing misinterpretation by patients and others.
Response: We acknowledge commenters' concerns about the staged process for the Overall Hospital Quality Star Ratings reductions, however more than 50 percent of hospitals already receive different ratings year-to-year just as a result of the evolution of the underlying data. The additional marginal effect of the 1-star reduction is fairly minimal in the scope of ordinary year-on-year changes and is likely less than the one-time impact of other significant methodological changes in past years (most significantly the switch from latent variable modeling to explicit average measure group scoring and the introduction of peer grouping) (85 FR 86193). Furthermore, this update reflects our belief (informed by interested parties' input) that patient safety should have greater emphasis than it does under the current methodology, and that this is a necessary and appropriate step to prevent hospitals from receiving unjustifiably high ratings despite poor Safety of Care outcomes.
We appreciate a commenter's position that there are existing CMS programs that drive patient safety improvement and that additional efforts may be conflicting, but we respectfully disagree and believe that further emphasis of patient safety in the Overall Hospital Quality Star Rating aligns with other efforts to improve patient safety including through HACRP.
We thank commenters for their suggestions related to transparency about peer groups, quartile calculation and measure group contributions. We would like to refer readers to resources related to these topics already publicly available. While hospitals' peer groups are not publicly reported (as a result of prior interested parties' feedback that suggested this may cause confusion for patients), a hospital's own peer group can be found in their Hospital-Specific Report. Additionally, detailed information about the peer grouping process is described in v4.1 of the Overall Hospital Quality Star Rating Methodology, available at https://qualitynet.cms.gov/inpatient/publicreporting/overall-ratings/resources. Peer group distribution as well as summary score ranges for each peer group can be found in the Quarterly Updates and Specifications Report that accompanies each Overall Hospital Quality Star Rating release.
In regard to how Safety of Care measures are selected for each hospital, in the process of assigning an Overall Hospital Quality Star Rating, all reported Safety of Care measures are incorporated. Hospitals participating in CMS programs are required to collect data for measures reported via Care Compare on Medicare.gov. Each underlying measure has established thresholds (such as minimum case count) for a hospital's score to be publicly reported to ensure reliable measurement. If a hospital does not meet threshold for a measure, a measure score for the hospital is not publicly reported, and therefore a score for that given measure does not factor into the Overall Hospital Quality Star Rating. However, in general, hospitals do not have a choice in measures for which they submit data, and for which their performance is scored and reported.
Comment: One commenter pointed to the significant methodological changes to individual measures finalized in the Fiscal Year (FY) 2026 Inpatient Prospective Payment System (IPPS) final rule (90 FR 36997 through 37027), such as the use of Medicare Advantage (MA) data, changes in risk adjustment models, removal of COVID-19 vaccination measures, and re-baselining of HAI measures, and urged CMS to delay implementation of Stage 2 for a year after updated data is publicly available and after conducting a comprehensive analysis of the combined effects before finalizing further penalties. The commenter noted implementing multiple methodological changes simultaneously may obscure the impact of individual updates, compound penalties that do not reflect actual quality performance, and undermine transparency by limiting public understanding and visibility.
Response: We acknowledge the concern expressed that methodologic changes in some underlying measures may subsequently affect the Overall Hospital Quality Star Rating. The Overall Hospital Quality Star Rating methodology was designed to ( printed page 53981) accommodate the ongoing evolution of publicly reported quality measures, including instances when existing measures make methodologic updates. CMS vets these updates extensively in advance with interested parties, experts, and the public based on established measure evaluation criteria to ensure the updated specifications are suitable for public reporting. The purpose of the Overall Hospital Quality Star Rating remains to simply summarize these publicly reported data, which must be independently validated as providing a meaningful quality signal prior to public reporting of any results.
https://qualitynet.cms.gov/inpatient/publicreporting/overall-ratings/resources
Comment: Commenters offered a variety of alternative approaches to address concerns with the proposed modifications to the Safety of Care measure group and the Overall Hospital Quality Star Rating.
A few commenters suggested that changes to the Overall Hospital Quality Star Rating, such as a star reduction, only be made if a hospital remains in the lowest quartile for multiple consecutive years, which would help account for normal performance variation and support long-term improvement efforts.
One commenter further proposed revising the minimum requirements for obtaining an Overall Hospital Quality Star Rating. The commenter suggested elimination of the Mortality domain as a minimum requirement and that hospitals be required to report on at least three Safety of Care measures to qualify for an Overall Hospital Quality Star Rating; thus, ensuring the measure group's performance is adequately represented and meaningful within the rating and reducing the need for blanket point reductions. A few commenters suggested alternatives to blanket penalties in public reporting, proposing the use of performance flags or annotations to highlight poor Safety of Care scores without reducing hospitals' Overall Hospital Quality Star Rating, thereby providing meaningful safety information while preserving rating comparability.
A few commenters encouraged CMS to increase the weight of the Safety of Care domain within the existing methodology (for example, from 22 percent to 30 percent) while reducing the weight of other domains proportionally, allowing safety to have a greater influence on the Overall Hospital Quality Star Rating without introducing categorical penalties. One commenter proposed that a more flexible, customizable Overall Hospital Quality Star Rating system that would allow users to assign their own weights to domains such as safety, mortality, or patient experience would reflect individual preferences and better supporting patient decision-making.
One commenter recommended CMS add additional rural-relevant patient safety measures or revise the methodology to include only measure groups with at least three reported measures to ensure composite scores more accurately reflect performance and do not discourage voluntary reporting of safety data, such as National Healthcare Safety Network (NHSN) HAI metrics. One commenter urged CMS to separate HAI measures from the broader Safety of Care measure group, allowing them to stand alone and improving the interpretability and actionability of these metrics for hospitals and the public alike. One commenter recommended exploring the use of more real-time structural patient safety measures, such as the Inpatient Hospital Patient Safety structural measure, to provide a more current and equitable assessment of safety performance. A few commenters specifically noted that many CAHs are excluded from the Overall Hospital Quality Star Rating due to measure reporting thresholds, and even when included, often report only one or two Safety of Care measures, which can disproportionately impact their Overall Hospital Quality Star Rating.
A few commenters called for improvements in data transparency, including clearer and more timely preview reports, enhanced technical assistance, and access to simulation tools to help hospitals prepare for the 2027 methodology change. A few commenters also called for CMS to provide plain-language explanations of methodological changes for patients, families, and advocates. One commenter emphasized the importance of engaging frontline clinicians and staff in future refinements and cautioned that changes to the Overall Hospital Quality Star Rating, such as a star reduction, could discourage transparency and self-reporting of safety events. Commenters urged CMS to implement these changes in a way that promotes fairness, supports continuous improvement, and maintains the credibility and usefulness of the Overall Hospital Quality Star Rating system.
Response: We appreciate commenters' alternative suggestions and recommendations to emphasize Safety of Care in the Overall Hospital Quality Star Rating methodology.
We acknowledge a commenter's recommendation to only apply the 1-star reduction to hospitals that are in the lowest quartile of the Safety of Care measure group for consecutive years. However, we want to reiterate that the goal of this methodology update is to emphasize patient safety across all hospitals, not just consistently low performers in the Safety of Care measure group.
We appreciate a commenter's suggestion to change the Overall Hospital Quality Star Rating eligibility criteria to require hospitals to report at least three measures in the Safety of Care measure group rather than the existing criteria of a hospital needing to report at least three measures in the Safety of Care measure group or the Mortality measure group. The current approach of requiring hospitals to report at least three measures in either Safety of Care or Mortality was vetted and partially informed by interested parties' feedback emphasizing the relative importance of Mortality and Safety of Care while still maintaining the Overall Hospital Quality Star Rating principle of optimizing inclusivity of measure information. This is meant to ensure that there is sufficient measure information available to allow for fair comparisons that reflect multiple dimensions of quality (particularly in the key domains of patient safety and preventing mortality), while still retaining flexibility to accommodate hospitals with varying measure reporting profiles.
We acknowledge commenters' suggestion to implement a performance flag or annotation to indicate poor Safety of Care performance rather than a 4-star cap or 1-star reduction. Currently, we are proceeding with the proposed methodology update to be consistent with the objective of the Overall Hospital Quality Star Rating to provide a single summary metric.
We appreciate commenters' recommendations to reconsider the weightings of the measure groups that contribute to the Overall Hospital Quality Star Rating, implement a customized weighting scheme, or separate out the HAI measures from the Safety of Care measure group. However, as noted in our v4.1 Methodology Report, the measure groups and respective weighting scheme were established during measure development with substantial interested parties, expert, and public input. The weighting of the Mortality, Safety of Care, Readmission, and Patient Experience measure groups will remain 22 percent and the weighting of the Timely & Effective Care measure group 12 percent. ( printed page 53982)
While we acknowledge the suggestion to include the Inpatient Hospital Patient Safety structural measure, structural measures are currently excluded from the Overall Hospital Quality Star Rating as they cannot be easily combined with other measures that are captured on a continuous scale with more granular data.
We thank commenters for their suggestions related to transparency as it is a key guiding principle for maintaining the Overall Hospital Quality Star Rating. Interested parties' input was gathered at two Provider Leadership Workgroup meetings, two Person and Family Engagement Workgroup meetings and one Technical Expert Panel meeting between 2023 and 2024 in addition to the public comment period correlated with the Safety of Care Request for Information included in the CY 2025 OPPS Proposed Rule (89 FR 94514 through 94521). We will consider how to increase transparency by providing plain-language explanations of methodological changes for patients, families and advocates. Accompanying Overall Hospital Quality Star Rating methodology updates, we strive to continue upholding the principle of transparency by publicly posting the R pack and input file used for the Overall Hospital Quality Star Rating calculations. We encourage questions about the methodology to be submitted via the QualityNet Question and Answer Tool.
We received no comments on our proposed adjustments to paragraphs (b)(1), (e) and (f). We are finalizing our proposal without modification to reflect updates to the regulation text uses of Overall Hospital Quality Star Rating and Care Compare on Medicare.gov language.
We received no comment on our proposed removal of the reference to “as defined in § 400.200 of this chapter.” We are finalizing as proposed.
After consideration of public comments, we are finalizing our proposal without modification to make the following two-stage methodologic updates to the Overall Hospital Quality Star Rating to emphasize the Safety of Care measure group (applicable to hospitals with at least three measures in the Safety of Care Measure Group): Stage 1: Implement a 4-star cap for hospitals in the lowest quartile of the Safety of Care measure group performance beginning in 2026 followed by Stage 2: Implement a blanket 1-star reduction for hospitals in the lowest quartile of Safety of Care measure group performance for the 2027 Overall Hospital Quality Star Ratings and later years.
XIX. Updates to Requirements for Hospitals to Make Public a List of Their Standard Charges
A. Introduction and Overview
1. Statutory Basis and Background
Section 1001 of the Patient Protection and Affordable Care Act (Pub. L. 111-148), as amended by section 10101 of the Health Care and Education Reconciliation Act of 2010 (Pub. L. 111-152), amended Title XXVII of the Public Health Service Act (the PHS Act), in part, by adding a new section 2718(e). Section 2718 of the PHS Act, entitled “Bringing Down the Cost of Health Care Coverage,” requires each hospital operating within the United States for each year to establish and update, and make public a list of the hospital's standard charges for items and services provided by the hospital, including for diagnosis-related groups established under section 1886(d)(4) of the Act. Section 2718(b)(3) of the PHS Act requires the Secretary of the Department of Health and Human Services (“Secretary” or “HHS”) to issue regulations to enforce the provisions of section 2718 of the PHS Act, and, in so doing, the Secretary may provide for appropriate penalties.
In the final rule that appeared in the November 27, 2019 Federal Register (84 FR 65524) titled “Medicare and Medicaid Programs: CY 2020 Hospital Outpatient PPS Policy Changes and Payment Rates and Ambulatory Surgical Center Payment System Policy Changes and Payment Rates: Price Transparency Requirements for Hospitals to Make Standard Charges Public” (hereafter referred to as the CY 2020 HPT final rule), we adopted requirements for hospitals to make public their standard charges in two ways: (1) as a comprehensive machine-readable file (MRF); and (2) in a consumer-friendly format. We codified these requirements at 45 CFR part 180. We also explained our belief that these two different methods of making hospital standard charges public are necessary to ensure that such data are available to consumers through data aggregation methods (for example, via integration into price transparency tools, electronic health records, and consumer apps), and direct availability to consumers searching for hospital-specific charge information. We stated our belief that innovators could use this information to create more useful data products for healthcare consumers to effectively compare prices. Moreover, we believe that employers (that offer or sponsor employee health plans), researchers, policy officials, and similar members of the public could utilize this data to promote competition and choice, ultimately helping to improve healthcare value.
Subsequently, in the CY 2022 OPPS/ASC final rule with comment period (86 FR 63941), we strengthened the hospital price transparency (HPT) enforcement process to improve compliance rates and made other updates to the requirements. Specifically, we: (1) increased the penalty amount for noncompliance through the use of a scaling factor based on hospital bed count; (2) deemed state forensic hospitals that meet certain requirements to be in compliance with the requirements of 45 CFR part 180; and (3) prohibited certain actions that we concluded were barriers to accessing the standard charge information, including prohibiting hospitals from designing their MRFs so as to make them inaccessible to automated searches and direct downloads.
In the CY 2024 OPPS/ASC final rule with comment period (88 FR 82079), we revised several HPT requirements to improve access to, and the usability of, hospital standard charge information; standardize the way hospital charges are presented; align, where feasible, certain HPT requirements and processes with requirements in the Transparency in Coverage (TiC) initiative; and strengthen and streamline our monitoring and enforcement capabilities. Specifically, we finalized: (1) a requirement that hospitals make a good faith effort to ensure standard charge information is true, accurate, and complete, and include a statement affirming this in the MRF; (2) new data elements that hospitals must include in the MRF, as well as a requirement that hospitals encode standard charge information in a CMS template layout; (3) a requirement that hospitals include a .txt file in the root folder that includes a direct link to the MRF and a link in the footer on its website that links directly to the publicly available web page that hosts the link to the MRF; and (4) improvements to our enforcement process by updating our methods to assess hospital compliance, requiring hospitals to acknowledge receipt of warning notices, and publicizing more information about CMS enforcement activities related to individual hospital compliance.
In these final rules, we stated that our policies requiring public release of hospital standard charge information are a necessary and important first step in ensuring transparency in prices of healthcare services for consumers. We also recognized that the release of hospital standard charge information is ( printed page 53983) not sufficient to achieve our ultimate price transparency goals. We noted that the regulations are, therefore, designed to address some of the barriers that limit price transparency, with a goal of requiring hospitals to make meaningful price information available to patients and employers to support a more competitive, innovative, affordable, and higher quality healthcare system.
On February 25, 2025, the White House issued Executive Order 14221, “Making America Healthy Again by Empowering Patients with Clear, Accurate, and Actionable Healthcare Pricing Information,” to empower patients with clear, accurate, and actionable healthcare pricing information.[443] The Executive Order states, in part, that the Departments of the Treasury, Labor, and HHS (the Departments) shall take action to:
- Require disclosure of actual prices of items and services, not estimates;
- Ensure pricing information is standardized and easily comparable across hospitals and health plans; and
- Update their enforcement policies designed to ensure compliance with transparent reporting of complete, accurate, and meaningful data.
Executive Order 14221 directs HHS to take actions to continue to implement and enforce existing statutory requirements for hospitals to make public a list of standard charges in accordance with guidelines developed by the Secretary. Consistent with the Executive Order and to better attain the goals we have articulated in previous HPT rulemaking—requiring hospitals to make meaningful price information available to consumers, employers, policymakers, and others to support a more competitive, innovative, affordable, and higher quality healthcare system—in the CY 2026 OPPS/ASC proposed rule, CMS proposed several updates to the regulations at 45 CFR part 180.
In the CY 2020 HPT final rule at § 180.20, we established a definition of “standard charge” as the regular rate established by the hospital for an item or service provided to a specific group of paying patients. In the CY 2026 OPPS/ASC proposed rule (90 FR 33790), we proposed subsequent updates to required MRF data elements that contextualize the standard charges, intended to improve the comparison of standard charge information and enable more meaningful disclosures to the public.
In the CY 2024 OPPS/ASC final rule with comment period (88 FR 82079), we established the requirement for each hospital, beginning April 1, 2024, to affirm in its MRF that the hospital has, to the best of its knowledge and belief, included all applicable standard charge information in accordance with the requirements of 45 CFR part 180 and that the information displayed is true, accurate, and complete as of the date indicated in the file. As described in the CY 2026 OPPS/ASC proposed rule, we proposed to strengthen this requirement, beginning January 1, 2026, by replacing it with an attestation in the MRF, and that attestation would also contain new specifications (compared to existing affirmation requirements). We stated in the CY 2026 OPPS/ASC proposed rule that these specifications would include that the hospital has: (1) included all applicable payer-specific negotiated charges in dollars that can be expressed as a dollar amount and for payer-specific negotiated charges that are not knowable in advance or cannot be expressed as a dollar amount, the hospital has provided in the MRF all necessary information available to the hospital for the public to be able to derive the dollar amount, including, but not limited to the specific fee schedule or components referenced in such percentage, algorithm, or formula, and (2) included the name of the hospital's chief executive officer, president, or senior official designated to oversee the encoding of true, accurate, and complete data.
In addition, pursuant to the authority provided to the Secretary under section 2718(b)(3) of the PHS Act to promulgate regulations to enforce section 2718 of the PHS Act, we previously have established regulations for enforcing the provisions of section 2718(e) of the PHS Act, including appropriate penalties. In the CY 2026 OPPS/ASC proposed rule, we proposed an additional change to encourage faster resolution of HPT civil monetary penalties (CMPs) and to reduce the amount of a CMP, under certain conditions, when the hospital waives its right to an administrative law judge (ALJ) hearing.
2. Summary of Final Policies
In this final rule with comment period, we are finalizing our proposals, with modifications, to revise the HPT regulations to enhance clarity and standardization in hospital disclosure of standard charges. Specifically, we are finalizing with modification our proposals to add to § 180.20 definitions for “tenth (10th) percentile allowed amount,” “median allowed amount,” and “ninetieth (90th) percentile allowed amount,” which are values hospitals will encode when a payer-specific negotiated charge is based on a percentage or algorithm, to more accurately reflect the distribution of actual amounts that hospitals have received for an item or service. In tandem with that, we are finalizing revisions to § 180.50 to remove the requirement for hospitals to disclose the estimated allowed amount, and, instead, require hospitals to disclose the 10th percentile, median, and 90th percentile allowed amounts, as well as the count of allowed amounts, in MRFs when payer-specific negotiated charges are based on percentages or algorithms. We are also finalizing, with modification, our proposal to require that hospitals use electronic data interchange (EDI) 835 electronic remittance advice (ERA) transaction data. Hospitals will be required to use EDI 835 ERA transaction data or an alternative, equivalent source of remittance data that includes the same information as EDI 835 ERA transaction data would include, to calculate and encode the allowed amounts. In addition, we are finalizing our proposals, with modifications, to require that hospitals comply with specific instructions regarding the methodology, including a lookback period, that must be used to calculate those amounts. We are finalizing that these policies are effective as of January 1, 2026, but we will delay enforcement of the requirements until April 1, 2026.
Additionally, we are finalizing, with modifications, our proposed amendments to § 180.50 to require hospitals to attest that in the MRF, to the best of the hospital's knowledge and belief, the hospital has included all applicable standard charge information in accordance with the requirements of this section and the information encoded is true, accurate, and complete as of the date in the file. We also are finalizing our proposal that hospitals attest in the MRF that the hospital has included all applicable payer-specific negotiated charges as dollars that can be expressed as a dollar amount, and for payer-specific negotiated charges that are not knowable in advance or cannot be expressed as a dollar amount, the hospital has provided in the MRF all necessary information available to the hospital for the public to be able to derive a dollar amount, including, but not limited to, the specific fee schedule or components referenced in such percentage, algorithm, or formula. Furthermore, we are finalizing our proposal that hospitals encode in the MRF the name of the hospital chief executive officer, president, or senior ( printed page 53984) official designated to oversee the encoding of true, accurate, and complete data. In addition, to advance the comparability of HPT data with other healthcare data, we are finalizing our proposal to require that hospitals encode their organizational, or Type 2, National Provider Identifier(s) (NPIs) in the MRFs. We are finalizing an effective date of January 1, 2026, for the revisions at § 180.50, including removal of the estimated allowed amount, disclosure of the 10th percentile, median, 90th percentile allowed amounts and the count of allowed amounts, the attestation requirements, and inclusion of NPIs. However, as discussed in more detail below, we will delay enforcement of these finalized revisions until April 1, 2026.
Finally, to encourage faster resolution and payment of CMPs, and in exchange for a hospital's admission of having violated HPT requirements, we are finalizing our proposal to update § 180.90 to, under certain circumstances, reduce the amount of a CMP by 35 percent when a hospital waives its right to an ALJ hearing. We are finalizing at new § 180.90(c)(4) that, effective beginning January 1, 2026, the amount of a CMP would be reduced by 35 percent should a hospital submit to CMS a written notice requesting to waive its right to a hearing under § 180.100 within 30 calendar days of the date of the notice of imposition of the CMP.
Collectively, these changes aim to improve transparency in hospital pricing, facilitate efficient enforcement of the HPT requirements, and empower consumers with actionable pricing information.
B. Modifications to the Requirements for Making Public Hospital Standard Charges at 45 CFR 180.50
1. Background
a. CY 2024 OPPS/ASC Final Rule With Comment Period
This section of the background recites relevant history from the CY 2024 OPPS/ASC final rule with comment period, and all references in this section pertain to it.
In the CY 2024 OPPS/ASC final rule with comment period (88 FR 82083, 82097, 82184), we indicated we understand that hospitals establish payer-specific negotiated charges in many ways, ranging from basic fee schedules (in which dollar amounts for specific items and services are known) to grouper methodologies (in which a base rate in dollars has been established but may then be modified depending on other factors like transfers or outliers), to “percent of billed charges” schemes (in which the dollar amount varies from person to person and is not known until the services are performed). We demonstrated in Figure A in the CY 2024 OPPS/ASC final rule with comment period (88 FR 82098) the components of an MS-DRG algorithm. An example of how Figure A may translate into an algorithm encoded in the MRF may be: base rate multiplied by the MS-DRG weight; outlier payment of $4,303 per diem when length of stay is greater than 2 times the average length of stay. Based on our experiences reviewing MRFs since finalizing the CY 2024 OPPS/ASC final rule with comment period, we have observed factors included in algorithms such as, but not limited to: weights based on resources required, mix of services provided within an episode of care, and thresholds or caps on the overall price of services billed within an episode of care. Therefore, we reiterate our stance outlined in the CY 2024 OPPS/ASC final rule with comment period that not all hospitals can produce a payer-specific negotiated charge in dollars that meets the definition of a “standard charge.”
As indicated in the CY 2024 OPPS/ASC final rule with comment period, we finalized a requirement for hospitals to display an estimated allowed amount which would provide needed context, in dollars, for instances where the hospital's payer-specific negotiated charge is based on a percentage or algorithm. We defined a new data element, the “estimated allowed amount,” at § 180.20, as the average dollar amount that the hospital has historically received from a third party payer for an item or service.
We noted that we heard from interested parties that, when a hospital has negotiated a payer-specific negotiated charge that is based on an algorithm, an estimate displayed in dollars within the MRF is useful, particularly for making comparisons across hospitals (88 FR 82099). We stated, for example, that an estimate displayed in dollars would permit users to make price comparisons across hospitals when, regarding the same procedure and payer/plan, one hospital has established a payer-specific negotiated charge as an algorithm and a second has established a payer-specific negotiated charge as a dollar amount. After considering what additional data could be required in the MRF to provide further needed context for a payer-specific negotiated charge that is expressed as an algorithm or a percentage, we finalized the estimated allowed amount as a new data element at § 180.20. We also required at § 180.50(b)(2)(ii)(C) that hospitals calculate and encode an estimated allowed amount, in dollars, when hospitals have established a payer-specific negotiated charge that is based on a percentage or an algorithm. We stated that the estimated allowed amount is the average reimbursement in dollars that the hospital has received from the payer in the past. We further stated that the estimated allowed amount is therefore not prospective and is also not based on the hospital's chargemaster, which, as we understand it, contains only gross charges for itemized items and services, or claims submitted to the payer. As we explained (88 FR 82099 through 82100), because the estimated allowed amount data element is meant to provide an estimate of what the algorithm produces in dollars, across the universe covered by a particular payer's plan, such an amount should reflect the amount the hospital expects to be reimbursed for the item or service (or service package), on average. We stated that we believed this information provides context to the public that is necessary to compare payer-specific negotiated charges across hospitals and is a valuable benchmark that innovators can use to develop price estimator tools to estimate an individual's personalized out-of-pocket costs. We stated that we believed this information, when paired with the algorithm encoded in the MRF, would promote greater transparency of hospital standard charges that can be useful to MRF users.
b. Background Subsequent to the CY 2024 OPPS/ASC Final Rule With Comment Period
Since the CY2024 OPPS/ASC final rule with comment period was finalized, we have continued to gain experience with the implementation of the estimated allowed amount data element and received public feedback and questions requesting that we further clarify its calculation. Based on our observations through comprehensive audits and feedback from users of the data, and consistent with Executive Order 14221, we proposed to revise the HPT regulations to recast the estimated allowed amount data element to better require, through new data elements, disclosure of dollar amounts for items and services in hospital MRFs, which we believe would enhance transparency and comparability of payer-specific negotiated charges across hospitals. Specifically, and as further discussed later in this section, we proposed to require hospitals to report four new data elements when a payer-specific ( printed page 53985) negotiated charge is based on a percentage or algorithm—the median allowed amount (which would replace the estimated allowed amount data element), the 10th percentile and 90th percentile allowed amounts, and the count of allowed amounts used to calculate the median, 10th, and 90th percentile allowed amounts.
2. General Comments
Comment: Many commenters, including, for example, hospitals and hospital associations, information technology developers, researchers, employers, payers, healthcare consumers, and consumer advocates expressed support for the Administration's goals of making more meaningful price information available to patients to support a more competitive, innovative, affordable, and higher quality healthcare system. Many commenters also supported our continued commitment to improving price transparency and the clarity of healthcare pricing. Many commenters agreed that price transparency supports consumers' ability to plan and make informed decisions about their care and to select high value providers. Several commenters indicated how complete, accurate, and timely price transparency can be a powerful tool allowing employers, innovators, researchers, policy makers, and public and private payers to have a truer picture of where cost pressures may lie and how they may be mitigated.
Response: We appreciate the overwhelming support for CMS price transparency policies, which include HPT, TiC, and the implementation of the No Surprises Act (NSA). We agree with commenters who believe that price transparency can stimulate provider competition, empower healthcare consumers, and result in lower healthcare costs. We agree that transparency in healthcare pricing is integral to supporting a transition to value-based care. We further agree that transparency in healthcare pricing is a societal benefit that can facilitate competition and comparison shopping to lower healthcare costs, and that policies that promote providing accurate and complete data ensure users of the MRF (such as employers and researchers) have the information they need to draw comparisons for contracting or research purposes, or support innovators as they develop products to enable patients' well-informed healthcare decisions.
Comment: Several commenters were generally supportive of the proposed regulatory requirements. Several commenters agreed that while some proposals may be useful, CMS should focus on policies that directly help patients understand the costs of their care rather than increasing administrative requirements on hospitals. A few commenters appreciated the agency revisiting the HPT policies to ensure that the information hospitals disclose in their MRFs is accurate and complete, so the information is maximally actionable for healthcare purchasers and consumers. A few commenters supported our efforts to simplify and streamline requirements, including MRF standardization, as such standardization facilitates actionable comparisons, and reduces the administrative burden of complying with these rules. One commenter indicated that ensuring that MRFs are available, timely, and accurate is the foundation upon which innovators can build tools that actually help patients navigate the cost of healthcare, but the commenter expressed that the data in the MRFs are insufficient on their own to fully support a patient's understanding of their individual price, and offered the view that final rules governing MRF creation must work in tandem with the NSA policy.
Response: We thank commenters for their support of the proposals. As we stated in the CY 2020 HPT final rule (84 FR 65571), we believe there is a direct connection between hospital standard charge information transparency and more affordable healthcare and lower healthcare costs. We believe healthcare markets can work more efficiently and provide consumers with higher value healthcare if we promote policies that encourage choice and competition. As we have stated on numerous occasions (for example, 84 FR 65526), we believe that transparency in healthcare pricing is critical to enabling patients to become active consumers so they can lead the drive towards value. As we stated in the CY 2020 HPT final rule (84 FR 65571), we continue to encourage hospitals to provide consumers with cost information in a consumer-friendly manner. We agree with commenters that standardization facilitates more actionable comparisons of the MRF data, and that over time standardization will reduce burden to hospitals and payers. We continue to affirm the premise we articulated in the CY 2024 OPPS/ASC final rule with comment period (88 FR 82080), that HPT regulations requiring hospitals to make public standard charges are a necessary and important first step for driving competition and in ensuring transparency in healthcare prices for the public. But, while foundational, we believe the release of hospital standard charge information alone is insufficient to achieve our ultimate goals for price transparency to help drive marketplace competition and consumer shopping. Instead, healthcare consumers' more complete understanding of real prices would come in tandem with other price transparency policies, including TiC and NSA.
Comment: Many commenters who opposed the proposals highlighted the operational and data complexities for hospitals—particularly for small, rural, and/or safety-net hospitals—in implementing the proposed changes. Several commenters provided examples of the operational burden, noting recent revenue losses, constrained resources, necessity of hiring vendor support to meet the requirements, and concern about the amount of time required for implementation. Several commenters noted that the steady stream of new HPT requirements has forced hospitals into a far more frequent update cycle, and asked that we stop adding new requirements. Several commenters recommended that CMS maintain the HPT rules in their current form and seek ways to reduce hospital burden. A few commenters cited Executive Order 14192 (Unleashing Prosperity Through Deregulation, issued January 31, 2025) and stated that we should be actively seeking ways to reduce hospital burden. A few of these commenters urged us to ensure HPT requirements are implemented with flexibility, clear technical guidance, and a supportive compliance framework that acknowledges the complexity of hospital pricing arrangements.
Response: We believe the benefits of these proposals to the public outweigh the burden on hospitals. The American public overwhelmingly supports healthcare price transparency. A September 2024 poll found that 92 percent of Americans want requirements for hospitals to provide upfront prices.[444] Additionally, these proposals align with the President's Executive Order 14221 directing that we require the disclosure of actual prices and ensure that pricing information is easily comparable across hospitals. We believe that continued use of a standardized format to encode and display this information will improve the public's understanding of the standard charges hospitals have established as well as reduce hospitals' burden over time. We will provide ( printed page 53986) technical guidance and examples of how to encode the new data elements we are finalizing in this rule on the CMS Hospital Price Transparency—Data Dictionary GitHub Repository, as well as in guidance on the HPT resources page on the CMS website. After consideration of comments, we are finalizing increased burden estimates found in section XXIII., Collection of Information Requirements”, of this final rule with comment period. Further, we are delaying enforcement (discussed in more detail in section XIX.B.7. of this final rule with comment period) of the revisions to § 180.50 to April 1, 2026, to provide hospitals with more time to comply.
Comment: Many commenters stated that we should prioritize efforts to improve the price comparison tools that are accessible to consumers who want to know their out-of-pocket costs, instead of layering more hospital regulatory requirements with respect to tools like MRFs that are not consumer-friendly. A few commenters asked CMS not to finalize these proposals, stating that the burden to implement them would outweigh the utility of the information for patients, and that the vast amounts of data presented in the MRF format, including hospital “standard charges,” are not consumer-friendly and do not provide patients with the personalized, prospective information they need to make informed decisions.
Response: We appreciate commenters' suggestions that we should focus on improving policies that directly help patients understand costs, and we may revisit the consumer-friendly display requirements in future rulemaking. While we understand the comment that MRFs may not be primarily intended for direct patient use, we disagree that they have little utility for patients and the public. The MRF format is designed to be parsed and analyzed by entities including innovators, employers, researchers, and journalists who transform that structured data into products, tools, research, and reports to support patients' ability to understand and compare hospital prices, which ultimately drives competition and directly benefits patients.
Since the HPT requirements went into effect on January 1, 2021, we have seen numerous examples of researchers and industry experts using MRFs to uncover potential savings by analyzing variation in payer-specific negotiated charges and discounted cash prices for the same items and services within and across hospitals; employers using the data to negotiate more competitive rates; and innovators identifying and aggregating the hospital price data for consumers to make more meaningful comparisons.[445 446 447 448] The impacts are beginning to be reflected in hospital pricing data. For example, a research report by Turquoise Health found that negotiated rates began converging between December 2021 and June 2024, and researchers from the University of Miami found that in Florida, “patients seeking elective, self-paid care responded [to hospital price transparency] by choosing hospitals that were transparent about their prices, and hospitals, in turn, responded by simplifying their pricing and reducing the intensity of services.” [449 450]
Comment: Many commenters pointed out the need for more policy coordination across the various price transparency initiatives (HPT, TiC, NSA and state level efforts), and the importance of not addressing HPT policies in isolation. These commenters stated that price transparency efforts would benefit from a comprehensive review of the numerous and sometimes conflicting requirements at both the State and Federal levels and urged us to focus future efforts to reform price transparency on streamlining policies to reduce the risk of conflicting information while improving accuracy, as well as alleviating costly administrative burden for both providers and insurers. Several commenters explained that with the recent Federal requirements for insurers and health plans to publicly share detailed pricing data, hospitals are no longer the most suitable or effective entities for this task. One commenter strongly felt that these proposals are shifting the responsibility of the plan's benefit design and education from the payers themselves to providers.
Response: We believe the HPT requirements we proposed to modify are complementary to other price transparency efforts such as the NSA. We note that in the CY 2024 OPPS/ASC final rule with comment period (88 FR 82079), we sought comment on alignment between HPT policies related to the consumer-friendly display requirements at § 180.60 which we may consider in future rulemaking. We acknowledge some states may have their own additional or more stringent requirements to promote price transparency. We also acknowledge that for patients with employer-based insurance, their health plans may have additional data to help the patient understand their out-of-pocket obligation, but, as stated above, the MRFs are a key foundational data source to support driving efficiency in health care. We continue to evaluate opportunities to improve coordination and further alignment across related Federal price transparency initiatives and policies.
Comment: Many commenters provided other suggestions on how to improve the HPT requirements and resources. In particular, commenters made recommendations on how to enhance the CMS template for the MRF. Several commenters recommended we add or remove data elements/attributes to or from the CMS template, particularly related to the hospital license number, hospital points of contact, the de-identified minimum negotiated charge, the de-identified maximum negotiated charge, the standard charge methodology, payer/plan name, billing class, stop loss, outliers, carve-outs, general contract provisions, a count of services, and plain language item/service descriptions. A few commenters suggested we further align data element/attribute names in the HPT MRFs with those in the TiC MRFs. A few commenters recommended limiting the display of items and services in the MRF. A few commenters recommended that we require hospitals to combine standard charge information with quality, health outcomes data and/or clinical data. A few commenters recommended that we require hospitals to group standard charge information by episodes of care. One commenter suggested eliminating the JSON format of the CMS template.
Several commenters provided recommendations for potential updates to the technical specifications in the CMS Hospital Price Transparency-Data Dictionary GitHub repository or to the Hospital Price Transparency validator tool. A few commenters requested we ( printed page 53987) increase the number of errors displayed in the online validator tool. A few commenters recommended that we clarify in the Hospital Price Transparency-Data Dictionary GitHub repository technical specifications that hospitals are required to make public their discounted cash prices for items and services, if the hospital has established a discounted cash price for an item or service. Several commenters requested that we provide enhanced technical assistance, guidance, or support to specific kinds of hospitals or in specific scenarios and increase engagement with hospitals. A few commenters suggested we provide financial support to hospitals to help them implement any new requirements finalized. One commenter recommended we implement the HHS Office of Inspector General's (OIG) recommendation to create a training and compliance program tailored for small and rural hospitals. One commenter requested we establish a multi-stakeholder technical advisory group, made up of hospitals, physicians, information technology firms, accounting firms, insurers, and patients to develop future HPT requirements and guidance.
A few commenters suggested we make updates to the requirements for displaying shoppable services in a consumer-friendly manner at § 180.60, with some of these commenters indicating we should eliminate these requirements as they feel they are no longer necessary as a result of the TiC and NSA regulations. Other commenters recommended that we: update, expand, or enhance the requirements for displaying shoppable services in a consumer-friendly manner; require hospitals to combine standard charge information with quality, health outcomes, and/or clinical data; and require hospitals to group standard charge information by episodes of care.
A few commenters requested we modify or vary the scope of the HPT requirements. A few commenters suggested tailoring the HPT requirements based on hospitals' specialty/size/patient volume. One commenter requested we expand the HPT requirements to include the disclosure of professional fees for providers not directly employed by hospitals.
Other commenters offered other HPT-related suggestions, with one recommending we consider strategies states are using to improve and enforce state-level HPT requirements, and another requesting that we further engage with patients in developing future HPT requirements.
Several commenters provided suggestions related to improving the TiC and NSA regulations, the 340B program, transparency of Medicare and Medicare Advantage rates, and extending the HPT requirements to other providers.
Response: While these comments are out of scope for this final rule because they do not relate to the specific proposals, we thank commenters for their suggestions on how to improve the HPT requirements and resources and may consider these comments with respect to future rulemaking, guidance, resources, and/or HPT enforcement process improvement.
3. Definitions
At § 180.20, we proposed to add definitions for three new data elements, the “median allowed amount,” the “tenth (10th) percentile allowed amount,” and the “ninetieth (90th) percentile allowed amount.” We stated in the proposed rule that these data elements would be defined as follows:
- “Median allowed amount” is defined as the median of the total allowed amounts the hospital has historically received from a third party payer for an item or service for a time period no longer than the 12 months prior to posting the machine-readable file. Should the calculated median fall between two observed allowed amounts, the median allowed amount is the next highest observed value.
- “Tenth (10th) percentile allowed amount” is defined as the 10th percentile of the total allowed amounts the hospital has historically received from a third party payer for an item or service for a time period no longer than the 12 months prior to posting the machine-readable file. Should the calculated percentile fall between two observed allowed amounts, the 10th percentile allowed amount is the next highest observed value.
- “Ninetieth (90th) percentile allowed amount” is defined as the 90th percentile of total allowed amounts the hospital has historically received from a third party payer for an item or service for a time period no longer than the 12 months prior to posting the machine-readable file. Should the calculated percentile fall between two observed allowed amounts, the 90th percentile allowed amount is the next highest observed value.
We discuss these definitions in more detail in sections below.
Comment: A few commenters supported our proposals to add the definitions for these three data elements. The commenters noted that more precise definitions will ensure that hospitals are interpreting the specifications correctly and patients will have more accurate data to allow them to make more informed decisions.
Response: We thank commenters for their support.
Comment: We received many comments on the proposed 12-month lookback period referenced in these definitions, as well as on the proposed definitions of the allowed amounts and how to calculate them when the percentiles fall between two observed allowed amounts.
Response: We address these comments in later sections on the lookback period and allowed amounts. As discussed in those sections, in light of these comments, we are finalizing these proposals with modification. Specifically, we are finalizing our proposed definitions for three new data elements, the “median allowed amount,” the “tenth (10th) percentile allowed amount,” and the “ninetieth (90th) percentile allowed amount” with modification to revise the phrase, “no longer than the 12 months” to state, “no less than 12 months and no longer than 15 months.”
Final Action: After consideration of public comments, we are finalizing the proposed definitions of “median allowed amount,” “tenth (10th) percentile allowed amount,” and “ninetieth (90th) percentile allowed amount” with modification to reflect a lookback period of no less than 12 months and no longer than 15 months prior to posting the MRF. These data elements will be defined as follows:
- “Median allowed amount” is defined as the median of the total allowed amounts the hospital has historically received from a third party payer for an item or service for a time period no less than 12 months and no longer than 15 months prior to posting the machine-readable file. Should the calculated median fall between two observed allowed amounts, the median allowed amount is the next highest observed value.
- “Tenth (10th) percentile allowed amount” is defined as the 10th percentile of the total allowed amounts the hospital has historically received from a third-party payer for a time period no less than 12 months and no longer than 15 months prior to posting the machine-readable file. Should the calculated percentile fall between two observed allowed amounts, the 10th percentile allowed amount is the next highest observed value.
- “Ninetieth (90th) percentile allowed amount” is defined as the 90th percentile of total allowed amounts the hospital has historically received from a third-party payer for a time period no ( printed page 53988) less than 12 months and no longer than 15 months prior to posting the machine-readable file. Should the calculated percentile fall between two observed allowed amounts, the 90th percentile allowed amount is the next highest observed value.
4. Replacing the Estimated Allowed Amount With the Allowed Amounts Data Elements and the Count of Allowed Amounts Data Element
a. Background on Encoding Payer-Specific Negotiated Charges as Dollar Amounts
As noted in the CY 2024 OPPS/ASC final rule with comment period (88 FR 82099), we have learned that most commercial contracting methods allow a hospital to identify and display as a dollar figure the payer-specific negotiated charges they have established with third party payers. Accordingly, we stated that we expect that, for most contracting scenarios, a hospital's payer-specific negotiated charges can also be expressed as a dollar amount.
As discussed in the CY 2026 OPPS/ASC proposed rule (90 FR 33476), hospitals and MRF users have indicated in inquiries to CMS that they are confused about our current requirements for encoding payer-specific negotiated charges, so we clarified our current policy in the proposed rule. We stated that if a dollar amount can be derived from a hospital's payer-specific negotiated charge, it must be encoded as a dollar value in the MRF. For items and services encoded in the MRF with a “standard charge methodology” of “case rate,” “per diem,” or a known “fee schedule,” we stated that we expect hospitals will be able to encode a “payer-specific negotiated charge: dollar amount.” We recognized that there may be situations where the payer-specific negotiated charge is a percentage of a fee schedule that is not available to the hospital. In such instances, under our existing policies, we stated the hospital must encode a “payer-specific negotiated charge: percentage” and an estimated allowed amount (which we stated would be replaced with the median allowed amount should our proposal be finalized) and may indicate in the additional notes data element the type of fee schedule. We noted that hospitals encoding a case rate or per diem as the standard charge methodology must encode the dollar amount for the service package base rate, which may be coupled with a “payer-specific negotiated charge: algorithm” and an estimated allowed amount (which we stated would be replaced with the median allowed amount should our proposal be finalized). We encouraged readers to review the scenarios and examples on the CMS Hospital Price Transparency—Data Dictionary GitHub Repository website for examples of how to encode standard charge information,[451] and additional guidance on CMS' HPT website.[452]
Comment: A few commenters commented on our clarification that if a dollar amount can be derived from a hospital's payer-specific negotiated charge, it must be encoded as a dollar value in the MRF. One commenter indicated they appreciated the May 22, 2025 guidance we provided at the CMS Hospital Price Transparency website's “Resources” page on this topic, titled “Updated Hospital Price Transparency Guidance Implementing the President's Executive Order (E.O.) `Making America Healthy Again by Empowering Patients with Clear, Accurate, and Actionable Healthcare Pricing Information,' ” (“May 22, 2025 guidance”) indicating they believe it is an important step towards ensuring the accuracy and completeness of the MRF data. Another commenter indicated they had not encountered any scenarios where a known fee schedule, case rate, or per diem exists outside of a more complicated and detailed algorithm and provided examples of factors that may further modify a fee schedule, case rate, or per diem, including carveouts, “lesser of” methodology, stop loss provisions, exclusions, and grouper logic. Therefore, this commenter recommended CMS reconsider the emphasis on the “payer-specific negotiated charge: dollar amount” data element in the MRF. A few commenters indicated they do not currently encode the “estimated allowed amount” if they can calculate the “payer-specific negotiated charge: dollar amount” of a known fee schedule, case rate, or per diem.
Response: We thank the commenters for their support of our guidance that if a dollar amount can be derived from a hospital's payer-specific negotiated charge, it must be encoded as a dollar value in the MRF. We continue to believe that for many items and services encoded in hospitals' MRFs with a “standard charge methodology” of “case rate,” “per diem,” or a known “fee schedule,” hospitals will be able to encode a “payer-specific negotiated charge: dollar amount,” as evidenced by a few commenters that indicated they are currently doing so. We recognize that there may be situations where the payer-specific negotiated charge for a particular item or service is further modified by a percentage or an algorithm, as suggested by one commenter. We clarify that if there are percentages or algorithms that modify the negotiated rate for an individual item or service, hospitals must encode the base rate for that item or service in the “payer-specific negotiated charge: dollar amount,” which must be coupled with a payer-specific negotiated charge percentage or algorithm, and hospitals must also encode the allowed amounts and count of allowed amounts we are finalizing in this rule (discussed in more detail below). For payer contract provisions that are negotiated at an aggregate level across multiple items and services (for example, claim level or episode of care), in the CMS Hospital Price Transparency—Data Dictionary GitHub Repository we provide guidance on the use of an optional data element, “general contract provisions,” that hospitals may, but are not required to use to convey this information. We point readers to the Hospital Price Transparency—Data Dictionary GitHub Repository available at https://github.com/CMSgov/hospital-price-transparency for additional details about this optional data element.
b. Replacing the Estimated Allowed Amount With the Median Allowed Amount and Adding the 10th and the 90th Percentile Allowed Amounts
In the CY 2026 OPPS/ASC proposed rule (90 FR 33476), we proposed to revise § 180.50(b)(2)(ii)(C) to require, at new § 180.50(b)(2)(ii)(C)( 2), that, beginning January 1, 2026, if a payer-specific negotiated charge is based on a percentage or algorithm, the hospital must calculate and encode the median allowed amount in dollars for that item or service. As noted above, we proposed to define “median allowed amount” in § 180.20 as the median of the total allowed amounts the hospital has historically received from a third party payer for an item or service for a time period no longer than the 12 months prior to posting the MRF. We stated in the proposed rule that should the calculated median fall between two observed allowed amounts (in other words, where the total count, n, is an even number), we proposed that the median allowed amount would be the next highest observed value. As we stated in the proposed rule, we believe requiring hospitals to encode the ( printed page 53989) median allowed amount in the circumstances described in § 180.50(b)(2)(ii)(C), rather than the estimated allowed amount, defined as the average dollar amount that the hospital has historically received from a third party payer for an item or service, would improve the public's ability to better understand, and, therefore, more meaningfully use, payer-specific negotiated charges, and would make such charges more comparable across hospitals.
We also proposed to revise § 180.50(b)(2)(ii)(C) to require at new § 180.50(b)(2)(ii)(C)( 2) that, beginning January 1, 2026, if a payer-specific negotiated charge is based on a percentage or algorithm, the hospital must calculate and encode the 10th and 90th percentile allowed amounts in dollars for that item or service. We proposed to define “tenth (10th) percentile allowed amount” in § 180.20 as the 10th percentile of the total allowed amounts the hospital has historically received from a third party payer for an item or service for a time period no longer than the 12 months prior to posting the MRF. We also proposed that if the calculated percentile falls between two observed allowed amounts, the 10th percentile allowed amount is the next highest observed value. We proposed to define “ninetieth (90th) percentile allowed amount” in § 180.20 as the 90th percentile of the total allowed amounts the hospital has historically received from a third party payer for an item or service for a time period no longer than the 12 months prior to posting the MRF. We also proposed that if the calculated percentile falls between two observed allowed amounts, the 90th percentile allowed amount is the next highest observed value.
Comment: We received many comments in support of our proposals to replace the estimated allowed amount with the median allowed amount and to add the 10th and 90th percentile allowed amounts. Several commenters indicated they believe the median, 10th percentile, and 90th percentile allowed amounts would be more useful than the estimated allowed amount, better reflecting what hospitals have been reimbursed for items and services. One commenter indicated they believe the median allowed amount would provide a more “robust and nuanced picture” of negotiated charges within the MRF. One commenter indicated they believe the median, 10th percentile, and 90th percentile allowed amounts would enhance “data utility for researchers, innovators, purchasers, and consumers seeking to understand price variation across hospitals and payers in a market.”
Response: We thank commenters for their support of the proposals. As we discussed in the CY 2026 OPPS/ASC proposed rule (90 FR 33792 through 33793), and in agreement with commenters, we continue to believe that requiring the median, 10th percentile, and 90th percentile allowed amounts will provide greater context and clarity with respect to the payer-specific negotiated charge than the estimated allowed amount. The median, 10th percentile, and 90th percentile allowed amounts will further improve the public's ability to understand the actual price of care, particularly when making comparisons across hospitals. As we stated in the CY 2026 OPPS/ASC proposed rule, allowed amount data elements will help MRF users to develop patient-level solutions to aid in patient financial planning and decision-making. The availability of a range of reference points, including lower (10th percentile), median (50th percentile), and upper (90th percentile) allowed amounts, will better enable healthcare consumers to compare cost information across hospitals, empowering them to better manage budgets, avoid unexpected financial burdens, and make more fully informed and value-conscious health care choices.
Likewise, researchers, innovators, policy officials, employers, and others MRF users will be able to use the information to improve data analysis and more precisely model healthcare costs and cost estimation algorithms, provide insights into healthcare pricing dynamics, and gain a deeper understanding of price dispersion across contracts that might provide a basis for negotiation and advocacy to more effectively bargain with healthcare providers and payers to yield more competitive pricing. Furthermore, the 90th percentile allowed amount will be helpful for assessing financial risk and identifying cases where costs exceed typical ranges. This information could assist researchers and innovators in refining cost predictions and contribute to better risk management strategies. As stated in the CY 2026 OPPS/ASC proposed rule (90 FR 33794), we continue to believe that for consumers with insurance plans that include coinsurance and deductibles, the 10th and 90th percentile allowed amounts would provide critical potential lower and upper reference points for estimating out-of-pocket expenses.
Comment: We also received comments opposing our proposals to replace the estimated allowed amount with the median allowed amount and to add the 10th and 90th percentile allowed amounts. Many commenters questioned the value of replacing the estimated allowed amount with the median allowed amount and adding the 10th and 90th percentile allowed amounts, indicating that they saw no additional benefit in these proposals for patients. Several commenters indicated they believe that replacing the estimated allowed amount with the median allowed amount and adding the 10th and 90th percentile allowed amounts would “overwhelm” patients with information that is difficult to interpret. A few commenters maintained that patients are more interested in knowing what their out-of-pocket costs may be for an upcoming hospital visit than negotiated rates between hospitals and insurers, or what hospitals have been reimbursed for items and services. A few commenters indicated that requiring the median, 10th percentile, and 90th percentile allowed amounts in the MRFs may be misleading for patients who would look at this information as a representation of what they would be expected to pay should they receive that item or service at the hospital. Therefore, several commenters recommended that we focus on advancing tools and requirements that provide patients with individualized and accurate pre-service estimates that factor in cost-sharing amounts/coinsurance, patient progress toward meeting their deductible, co-payments, and other pertinent information.
Response: We disagree with commenters who indicated there is no value for patients in replacing the estimated allowed amount with the median allowed amount and adding the 10th and 90th percentile allowed amounts. As we stated in the CY 2024 OPPS/ASC final rule with comment period (88 FR 82079), while the MRF format is designed to be used by innovators, employers and researchers and to be ingested into machines for further processing of the data, the information contained in a MRF is critical for driving competition and directly beneficial for patients. We believe, as discussed in the CY 2026 OPPS/ASC proposed rule (90 FR 33792 through 33793), that replacing the estimated allowed amount with the median allowed amount and adding the 10th and the 90th percentile allowed amounts will better enable price estimator tools to develop and estimate an individual's personalized out-of-pocket cost, as suggested by some commenters, ultimately providing patients with the information some ( printed page 53990) commenters stressed patients are most interested in and need.
Comment: Many commenters indicated that they oppose the proposals to replace the estimated allowed amount with the median allowed amount and add the 10th and 90th percentile allowed amounts due to the burden they place on hospitals. In particular, commenters expressed concerns about the specific instruction, in the definition of the median allowed amount and the 10th and 90th percentile allowed amounts, stating that should the calculated percentile “fall between two observed allowed amounts, the percentile allowed amount is the next highest observed value.” These commenters indicated this deviation from the standard way of calculating a percentile when it falls between two observed values would require hospitals to implement custom formulas for these calculations rather than the “out-of-the-box” formulas, increasing hospital burden. Several commenters requested that CMS not finalize these proposals due to resource constraints, particularly for small and rural hospitals, and a few commenters indicated the proposed effective date of January 1, 2026, would make complying with the requirements, if finalized as proposed, especially challenging. A few commenters requested we provide technical support and training for hospitals to assist them with implementing the requirements, should we finalize them as proposed. One commenter suggested we convene a group of stakeholders to evaluate how to display allowed amounts within the MRF and allow flexibility for hospitals to display their negotiated charges in the MRF as they deem appropriate until such time as the group could meet.
Response: We appreciate commenters' concerns that the proposed methodology for arriving at a value when the median, 10th percentile, or 90th percentile allowed amounts fall between two different values is not standard and may require additional programming or calculations by hospitals and this creates additional burden. However, as we stated in the CY 2026 OPPS/ASC proposed rule (90 FR 33792 through 33793), we believe the need to identify the actual dollar value a hospital has received is consistent with our statutory authority and aligns with the Administration's goals of ensuring pricing information is standardized and easily comparable across hospitals when a hospital's payer-specific negotiated charge is based on a percentage or algorithm.
As with previous HPT rulemaking, we will provide technical guidance and examples of how to encode the new data elements we are finalizing in this rule on the CMS Hospital Price Transparency—Data Dictionary GitHub Repository ( https://github.com/CMSgov/hospital-price-transparency), as well as guidance on the HPT resources page on the CMS website ( https://www.cms.gov/priorities/key-initiatives/hospital-price-transparency/resources). We thank the commenter for their suggestion to convene a stakeholder group to evaluate the use and display of allowed amounts within the MRF and to continue to allow flexibility until we do, but decline the commenter's suggestion at this time as we believe we have received sufficient feedback on encoding allowed amounts within the MRF via the public comment period on the proposed rule as well as in response to the “CMS Hospital Price Transparency Accuracy and Completeness Request for Information” (the public comment period for which closed on July 21, 2025), in which commenters recommended hospital MRFs should list a precise dollar amount for items and services whenever possible.
Comment: Several commenters expressed concern about replacing the estimated allowed amount with the median allowed amount and adding the 10th and 90th percentile allowed amounts so soon after the January 1, 2025 effective date of the estimated allowed amount requirement. These commenters questioned why CMS believes the estimated allowed amount is insufficient to provide context to the payer-specific negotiated charge when it is based on a percentage or algorithm and why the median allowed amount is more useful to consumers of the MRF. One commenter questioned why our proposals in the proposed rule deviate from our May 22, 2025 guidance.
Response: As discussed in the CY 2026 OPPS/ASC proposed rule, on February 25, 2025, the President issued Executive Order 14221 directing that HHS act to require disclosure of actual prices and ensure pricing information is easily comparable across hospitals. Consistent with the Executive Order and the feedback we have received from interested parties, we have considered ways to improve the requirement for hospitals to make public actual dollar amounts in the MRF to further transparency and comparability of hospital pricing information. Specifically, we considered the usefulness of the estimated allowed amount, as defined at § 180.20, in providing necessary context for the payer-specific negotiated charge and in facilitating comparisons across hospitals. As we stated in the proposed rule, we believe that the payer-specific negotiated charge should be better contextualized and more precisely encoded to improve the MRF users' ability to understand and use hospital standard charges. While the estimated allowed amount provides useful additional context and enhances transparency and comparability of hospital standard charges, the median, 10th percentile, and 90th percentile allowed amounts more accurately reflect what the hospital is reimbursed.
Our May 22, 2025 guidance regarding the use of the estimated allowed amount was an important first step towards requiring the disclosure of the actual prices of items and services, not estimates, and ensuring pricing information is standardized and easily comparable across hospitals and health plans. We realized, however, that additional requirements would be needed to achieve that aim. Toward that end, we proposed in the CY 2026 OPPS/ASC proposed rule to replace the estimated allowed amount with the median allowed amount and to add the 10th and 90th percentile allowed amounts, requiring hospitals to encode actual dollar values they have received for an item or service when the payer-specific negotiated charge is based on an algorithm or percentage. Consistent with the President's Executive Order and our goal of advancing the disclosure of the actual prices of items and services, we are finalizing our proposals to replace the estimated allowed amount with the median allowed amount and to add the 10th and 90th percentile allowed amounts.
Comment: Several commenters requested, as an alternative to our proposal to replace the estimated allowed amount with the median allowed amount and to add the 10th and 90th percentile allowed amounts, that CMS require hospitals, without exception, to make public all their payer-specific negotiated charges “in dollars and cents,” thus eliminating the need for the currently required estimated allowed amount or the proposed median, 10th percentile, and 90th percentile allowed amounts. These commenters indicated they believe this is the only way to be able to make “apples-to-apples” comparisons across hospitals and for patients to understand what they might be expected to pay for an item or service. One of these commenters suggested we prohibit hospitals from charging patients more than a payer-specific negotiated charge dollar amount and presume that any MRF that has payer-specific negotiated charges represented as percentages and algorithms is automatically non-compliant with our requirements. ( printed page 53991) Conversely, one commenter requested we indicate in the final rule that the allowed amounts would not represent a guaranteed price because they do not necessarily represent the amount an individual would expect to pay for the item or service.
Response: We point these commenters to the discussion in the CY 2024 OPPS/ASC final rule with comment period (88 FR 82079) where we acknowledged and agreed with commenters that, although critical for determining an individual's out-of-pocket obligation, hospital standard charges do not represent either an individual's out-of-pocket obligation or a “real, guaranteed price.” However, as we noted in the CY 2024 OPPS/ASC final rule with comment period, individualized estimates in dollars may be obtained directly, in many circumstances, from providers and payers through other Federal price transparency efforts such as those implementing the NSA and TiC requirements. As such, we continue to strongly encourage individual consumers to avail themselves of hospital and payer price estimator and comparison tools, and to seek out “good faith estimates” from hospitals to comply with separate requirements implementing the NSA, which may provide up-front pricing that can be used to dispute final charges that are substantially in excess of the up-front amounts. Additionally, we reiterate, as we stated in the CY 2020 HPT final rule and the CY 2024 OPPS/ASC final rule with comment period, that we continue to encourage hospitals to provide consumers with cost information in a consumer-friendly manner. Furthermore, as we noted in the CY 2024 OPPS/ASC final rule with comment period, we understand the desire for individual patients to access hospital prices in dollars and cents. However, as discussed in that rule, we continue to disagree that all hospitals can produce a payer-specific negotiated charge in dollars that meets the definition of a “standard charge” because hospitals establish payer-specific negotiated charges in many ways, ranging from basic fee schedules (in which dollar amounts for specific items and services are known), to grouper methodologies (in which a base rate in dollars has been established but may then be modified depending on other factors like transfers or outliers), to “percent of billed charges” schemes (in which the dollar amount varies from person to person). Finally, as discussed in the CY 2024 OPPS/ASC final rule with comment period, we believe that section 2718(e) of the PHS Act directs the Secretary to tell hospitals how to display their standard charges, not how to establish them or that they must establish them.
We continue to believe that the policies we are finalizing in this final rule with comment period will greatly improve the transparency of payer-specific negotiated charges and will make such charges more comparable across hospitals.
Comment: A few commenters recommended CMS keep the requirement to encode the estimated allowed amount in the MRF instead of replacing it with the median allowed amount and adding the 10th and 90th percentile allowed amounts, indicating they believe that given the volume and complexity of the MRFs, the average would provide a more accurate estimate of what a hospital has historically received for that item or service. One commenter indicated that “the law of large numbers ensures that the sample mean will converge towards the true population mean as the sample size grows. This assures that with enough data, such as that contained in MRFs, the average provides a more accurate estimate of the overall characteristic of the population being studied.”
Response: We recognize that the extent to which a sample statistic provides a reasonable representation of the population statistic depends, at least in part, on the sample size. Therefore, we proposed hospitals encode the count of allowed amounts data element (described in a later section) so that consumers of the MRFs can make informed decisions about the data. The commenter did not provide additional reasons for why the mean is a preferred statistic to the median, 10th percentile, and 90th percentile allowed amounts. We provided our reasons for proposing hospitals encode the median, 10th percentile, and 90th percentile allowed amounts in the proposed rule. Specifically, we stated we believe these allowed amounts would be a more accurate reflection of the amount a hospital would be reimbursed for an item or service. Thus, we proposed to revise § 180.50(b)(2)(ii)(C) to require hospitals to encode, beginning January 1, 2026, the median allowed amount, rather than the estimated allowed amount, if a payer-specific negotiated charge is based on an algorithm or percentage. We explained why the median allowed amount would be a more accurate and useful indicator of the allowed amount instead of a mean or average allowed amount with an outlier example in the proposed rule, that we reiterate here.
( printed page 53992)In the scenario detailed in Table 143, the mean of the claim remittance amounts is $35,000, which exceeds all the other values except for the $200,000 outlier and would not reasonably reflect the allowed amount for that item or service. By contrast, the $20,000 median would be a more accurate reflection of the allowed amount for that item and service and the amount a hospital typically would be reimbursed for that item or service. As we stated in the proposed rule, requiring the median rather than the average is consistent with generally accepted statistical principles for assessing the central point of a distribution when there are outliers.[453]
Comment: A few commenters requested clarification on, or seemed to misinterpret, our proposal to replace the estimated allowed amount with the median allowed amount. One commenter suggested regular changes in the gross charge will impact the calculation of the allowed amount. One commenter requested we clarify what a hospital should encode when they have no remittance data from which to calculate the median allowed amount. One commenter asked if CMS finalizes the proposals, would a hospital be required to encode the median, 10th percentile, and 90th percentile allowed amounts if they have encoded a payer-specific negotiated charge as a dollar amount.
Response: We thank commenters for the questions. We clarify that we proposed to define the median allowed amount as the median of the total allowed amounts the hospital has historically received from a third party payer. Since the median allowed amount would be based on the remittances received from a third party payer, we do not see how changes in the gross charge, which is defined in regulation as the charge for an individual item or service that is reflected on a hospital's chargemaster, absent any discounts, would impact the calculation of the allowed amounts. We also clarify that should a hospital have no remittance data for a particular item or service within the 12-to-15 month lookback period prior to posting the MRF (discussed in a later section), a hospital would encode “0” for the count of allowed amounts for that item and service and may leave the median, 10th percentile, and 90th percentile allowed amounts in the MRF blank. Further, we clarify that if a hospital has encoded a payer-specific negotiated charge in the MRF as a dollar amount, and that dollar amount represents the full payer-specific negotiated charge that is not further modified by a percentage or algorithm, the hospital would not be required to encode the allowed amounts. For example, if the hospital's payer-specific negotiated charge is 70 percent of the Medicare payment rate, then the hospital would calculate and encode the payer-specific negotiated charge as a dollar amount, and would not need to further describe the percentage or algorithm or provide the allowed amounts. If a payer-specific negotiated charge is a dollar amount and can be calculated, in part, but does not fully account for the payer-specific negotiated charge, however, then the hospital must calculate and encode the payer-specific negotiated charge dollar amount and must also describe the algorithm or percentage and encode the allowed amounts. For example, if the service has a base rate of $3,495, and is further modified by an algorithm, then the hospital would encode the base rate of $3,495 in the standard charge dollar data element, and then describe the algorithm that further modifies that base rate in the standard charge algorithm data element, and encode the allowed amounts. Figure 3 is an example of the information required for the algorithm description.
Comment: One commenter requested CMS reconsider in future rulemaking requiring hospitals to encode a “maximum allowed amount” that would reflect the maximum total allowed amount a hospital has historically received for a third party payer for an item and service.
Response: In the CY 2024 OPPS/ASC final rule with comment period (88 FR 82100), we stated that we agreed with commenters that the display of a maximum allowed amount could provide some clarity about the highest dollar amount a consumer might be obligated to pay (once the consumer calculates their own potential out-of-pocket obligation based on the displayed maximum allowed amount). For example, suppose, with respect to a particular payer and plan, the maximum allowed amount for an item or service was displayed as $1,500, the plan featured a 20 percent coinsurance requirement, and the individual had already met any applicable annual deductible. In such a scenario, the individual would likely not be required to pay more than $300 (20 percent of $1,500) for the indicated item or service.[454]
At that time, however, we elected not to adopt commenters' suggestions to require hospitals to encode the maximum allowed amount because, as we stated, a maximum dollar value derived from past remittances or other data sources could include outliers, thereby potentially misrepresenting an individual's required payment for an item or service. As we indicated in the CY 2026 OPPS/ASC proposed rule (88 FR 82101), the display of the maximum allowed amount could be skewed to the point where it would not present useful information to consumers or the public. As opposed to the maximum allowed amount, however, the 90th percentile of the total allowed amounts for an item or service would be more representative of the dollar amount the individual might be responsible for paying, less subject to extreme outliers, and would provide an additional data point to contextualize the dollar value when the payer-specific negotiated charge for an item or service is based on a percentage or algorithm. Similarly, we stated we believe that setting a threshold based at the 10th percentile would exclude outliers on the low end, and, when combined with the other data elements, provide MRF users with a better understanding of the realistic range of payer-specific negotiated charges. We will continue to evaluate the utility of the maximum allowed amount, and may revisit this alternative in future rulemaking.
Final Action: After consideration of public comments, we are finalizing our proposals to replace the estimated allowed amount with the median allowed amount and to add the 10th and 90th percentile allowed amounts, including our proposed methodology for calculating the allowed amounts should the calculated percentile fall between two observed allowed amounts. As described earlier, we are revising § 180.50(b)(2)(ii)(C) to require, at new § 180.50(b)(2)(ii)(C)( 2), that, beginning January 1, 2026, if a payer-specific negotiated charge is based on a percentage or algorithm, the hospital must calculate and encode the 10th percentile, median, and 90th percentile allowed amounts in dollars for that item or service, but delaying enforcement until April 1, 2026.
As noted previously, we also are finalizing, with modification, our proposed definition of “median allowed amount” in § 180.20, such that the ( printed page 53993) “median allowed amount” will be defined as the total allowed amounts the hospital has historically received from a third party payer for an item or service for a time period no less than 12 months and no longer than 15 months prior to posting the machine-readable file. We are finalizing, with modification, our proposed definition of “tenth (10th) percentile allowed amount” in § 180.20 as the 10th percentile of the total allowed amounts the hospital has historically received from a third party payer for an item or service for a time period no less than 12 months and no longer than 15 months prior to posting the machine-readable file. We are finalizing, with modification, our proposed definition of “ninetieth (90th) percentile allowed amount” in § 180.20 as the 90th percentile of the total allowed amounts the hospital has historically received from a third party payer for an item or service for a time period no less than 12 months and no longer than 15 months prior to posting the machine-readable file. We are also finalizing at § 180.20 that should the calculated percentile fall between two observed allowed amounts (in other words, where the total count, n, is an even number), the allowed amount will be the next highest observed value.
c. Calculation of Allowed Amounts
(1) Determining the “Total Allowed Amount”
In the CY 2026 OPPS/ASC proposed rule (90 FR 33795), we noted that, under the proposed definitions of “median allowed amount,” “10th percentile allowed amount,” and “90th percentile allowed amount” at § 180.20, hospitals would calculate the allowed amount considering the “total allowed amount” for an item or service. As with the estimated allowed amount in the current rule, and as we explained in the CY 2024 OPPS/ASC final rule with comment period (88 FR 82101), the amount should reflect the total amount the hospital was reimbursed for the item or service (or service package). We stated in the CY 2026 OPPS/ASC proposed rule (90 FR 33795) that the “total allowed amount” dollar figure would be derived from the gross charge minus contractual adjustments and consist of the portion billed to a payer for a particular plan and the portion, if any, billed to the patient. As we indicated in the CY 2026 OPPS/ASC proposed rule, we believe defining the “total allowed amount” this way would help to enhance consistency in how hospitals calculate this contextual data element, increasing comparability across hospitals.
We received no comments about this provision and are finalizing as proposed.
(2) Data Source for Calculating the Allowed Amounts
In the CY 2024 OPPS/ASC final rule with comment period (88 FR 82101), we stated our belief at the time that hospitals should retain flexibility, in the interest of reducing burden, to determine the best data source(s) for calculating the estimated allowed amount data element, though we agreed with commenters that using information from EDI 835 ERA transaction data would appear to meet our requirements. To enhance the consistency of hospital standard charge information and the comparability of the median allowed amount, and the 10th percentile and 90th percentile allowed amounts, and in accord with what commenters had earlier suggested in the CY 2024 OPPS/ASC final rule with comment period, in the CY 2026 OPPS/ASC proposed rule (90 FR 33476), we proposed to require that hospitals only use EDI 835 ERA transaction data to calculate and encode the allowed amounts. As we had indicated in prior rulemaking (88 FR 82100 through 82101), EDI 835 ERA transaction data, the electronic transaction data that provides claim payment information that hospitals use to track and analyze their claims and reimbursement patterns, including any adjustments made to the claim such as denials, reductions or increases to the amount charged, and expected patient co-payments, co-insurance, or secondary coverage, would meet the requirement to calculate an allowed amount. We sought comment on the proposal to require that hospitals only use EDI 835 ERA transaction data to calculate and encode the allowed amounts. We also sought comment on whether there are instances where a hospital would not have access to EDI 835 ERA transaction data and whether there are alternative data sources we should consider requiring hospitals to use to calculate the allowed amounts and count of allowed amounts.
Comment: Several commenters supported our proposal to require that hospitals only use EDI 835 ERA transaction data to calculate and encode the allowed amounts, indicating that the proposal would enhance comparability across hospitals and improve the utility of MRFs. One commenter indicated that the requirement would improve consumers' ability to “comparison shop” between hospitals as they will have confidence that the allowed amounts were calculated using common sources of data. Another commenter indicated that EDI 835 ERA transaction data is the “superior option” for calculating the allowed amounts and suggested that other data sources would be less reliable. One commenter maintained that EDI 835 ERA transaction data is an industry standard and the most comprehensive source of transaction data from which to calculate the allowed amounts and urged us not to deem any other data sources as a compliant alternative to EDI 835 ERA transaction data.
Response: We thank commenters for the support of our proposal to require that hospitals only use EDI 835 ERA transaction data to calculate and encode the allowed amounts. We agree with commenters that suggested EDI 835 ERA transaction data is sent by many payers and thus requiring its use to calculate and encode the allowed amounts would enhance the consistency of hospital standard charge information and the comparability of the median allowed amount, and the 10th percentile and the 90th percentile allowed amounts. However, as discussed in a later comment summary, several commenters indicated that there are scenarios in which a hospital does not have access to EDI 835 ERA transaction data. In order to ensure that all hospitals subject to the HPT requirements are able to calculate and encode the allowed amounts, we are requiring hospitals to use EDI 835 ERA transaction data or an alternative, equivalent source of remittance data that includes the same information as EDI 835 ERA transaction data would include (as discussed in more detail below).
Comment: Many commenters opposed our proposal to require that hospitals only use EDI 835 ERA transaction data to calculate and encode the allowed amounts due to the perceived burden of the proposal. Several commenters indicated that requiring hospitals to only use EDI 835 transaction data would require significant resource investment, coordination within the hospital, and, potentially, coordination with their EHR vendors. Several commenters maintained that it would be difficult to only use EDI 835 ERA transaction data to calculate and encode the allowed amounts because the data is not standardized, the data may not capture item or service level detail, payers use claim adjustment reason codes (CARC) and remittance advice reason codes (RARC) inconsistently, and payers frequently deviate from Health Insurance Portability and Accountability Act (HIPAA) standard transaction requirements. A few commenters suggested that using EDI 835 ERA transaction data to calculate ( printed page 53994) and encode the allowed amounts may be particularly difficult for small and rural hospitals with limited technical capacity.
Several commenters opposed the proposal to require hospitals to only use EDI 835 ERA transaction data to calculate and encode the allowed amounts because they indicated there are scenarios where a hospital does not have EDI 835 ERA transaction data. Several commenters noted that some payers still send paper remittances and a few commenters indicated that they have electronic remittance data from some payers, but it is not EDI 835 ERA transaction data. A few commenters indicated hospitals may lack direct access to EDI 835 ERA transaction data. A few commenters indicated that hospitals may receive reimbursement in the form of per month supplements, value-based payment distributions, or other payment arrangements that may not be reflected in EDI 835 ERA transaction data, and in such cases, one commenter suggested hospitals be allowed to reconcile EDI 835 ERA transaction data with other data sources to ensure the allowed amounts reflect the full reimbursement for a particular item or service. One commenter noted that CMS' proposal does not account for hospitals with integrated healthcare delivery systems which rely on internal allowed amount fee schedules as an alternative, equivalent data source and would produce the same allowed amount outputs as the EDI 835 ERA transaction data, but without significant administrative inefficiencies. Another commenter suggested that if a hospital does not have access to EDI 835 ERA transaction data, they support allowing the use of an alternative data source.
Response: We believe the requirements we are finalizing in this final rule with comment period will strengthen HPT through further standardization and comparability of the allowed amounts across hospitals, and that the benefits to the public outweigh the burden on hospitals. We appreciate commenters who suggested that there are reimbursements that may not be reflected in the EDI 835 ERA transaction data. Acknowledging the merits of these comments, we are finalizing that hospitals be required to use EDI 835 ERA transaction data or an alternative, equivalent source of remittance data that includes the same information as EDI 835 ERA transaction data would include. We clarify that if a hospital uses an alternative, equivalent source of remittance data to calculate and encode the allowed amounts, such data source must allow the hospital to calculate the “total allowed amount,” meaning the gross charge minus contractual adjustments, consisting of the portion billed to a payer for a particular plan and the portion, if any, billed to the patient. Further, as discussed in more detail in another section of this final rule with comment period, although the new requirements at § 180.50 will be effective January 1, 2026, we are delaying enforcement of these requirements to April 1, 2026, to provide hospitals with more time to comply. We believe our finalized policy to give hospitals the option to use an alternative, equivalent source of remittance data that includes the same information as EDI 835 ERA transaction data is responsive to commenters' concerns about the lack of availability of EDI 835 ERA transaction data for certain hospitals and mitigates concerns of burden associated with accessing EDI 835 ERA transaction data for such hospitals. At the same time, acknowledging commenters' concerns about burden we have increased burden estimates in section XXIII. (Collection of Information Requirements) of this final rule with comment period.
Comment: Several commenters opposed our proposal to only use EDI 835 ERA transaction data to calculate and encode the allowed amounts because they disagree with the EDI data they have been provided by payers, or believe there are errors in the data they have received. A few commenters indicated that the allowed amounts encoded in hospitals' MRFs may be much lower than the payer-specific negotiated charge for those items and services if CMS finalizes the requirement to use only EDI 835 ERA transaction data, as hospitals may have ongoing payment disputes with payers at the time the MRF is updated. One commenter indicated it would be “unreasonable” to require hospitals to use EDI 835 ERA transaction data because hospitals lack control over the accuracy of the data.
Response: We disagree with the commenter that indicated it would be unreasonable for us to require hospitals to use EDI 835 ERA transaction data to calculate and encode the allowed amounts. As indicated by commenters that supported our proposal, many payers send EDI 835 ERA transaction data to hospitals and thus it would be a consistent and reliable source of data for hospitals to use to calculate and encode the allowed amounts. However, as indicated elsewhere in this section, we are finalizing requiring hospitals to use EDI 835 ERA transaction data or an alternative, equivalent source of remittance data that includes the same information as EDI 835 ERA transaction data would include. Additionally, we recognize that some remittance data initially provided to a hospital may be later adjusted for a variety of reasons that may modify the total allowed amount for an item or service. However, as discussed in the CY 2026 OPPS/ASC proposed rule (90 FR 33796), timely filing limits for claims vary by state and payer, with a typical range of 30 days to 12 months from the date of service.[455] [456] [457] [458] In addition, research suggests that many healthcare claims are adjudicated within 30 days.[459] Therefore, we believe much of the remittance data hospitals will use to encode the allowed amounts will be finalized within 13 months, and that our finalized lookback period (discussed in a later section) that includes no less than 12 months and no longer than 15 months of data prior to posting the MRF should provide sufficient time for hospitals to receive corrected remittance data for most of the data that is used. We acknowledge that there may be some instances where the remittance data hospitals use to calculate and encode the allowed amounts may change after the MRF is posted, due to additional adjustments. However, we believe the need for hospitals to use more recent remittance data to enhance comparability outweighs the need to capture the final disposition on a claim.
Comment: A few commenters provided alternative suggestions to or requested clarification of our proposal for hospitals to calculate and encode the allowed amounts using only EDI 835 ERA transaction data. One commenter questioned whether we are proposing to require hospitals to calculate and encode a payer-specific negotiated charge that is not based on a percentage or algorithm using EDI 835 ERA transaction data. One commenter recommended we publish a standardized guide for calculating the allowed amounts at an item or service ( printed page 53995) level. One commenter suggested we require hospitals to encode the date of the most recent chargemaster update and the weighted average percentage increase applied to gross charges since the date of last update.
Response: As indicated in the CY 2020 HPT final rule (84 FR 65551), the payer-specific negotiated charge is defined as the charge that a hospital has negotiated with a third party payer for an item or service. We clarified in that rule (84 FR 65534) that the payer-specific negotiated charge can be found in other parts of the hospital billing and accounting systems than the chargemaster, or in rate tables or the rate sheets found in hospital in-network contracts with third party payers indicating the agreed upon rates for the provision of various hospital services. Since EDI 835 ERA transaction data is the electronic transaction data from payers that provides claim payment information rather than the payer-specific negotiated charge for an item or service, we clarify that we would not expect, nor should hospitals use, EDI 835 ERA transaction data to encode their payer-specific negotiated charges. We thank the commenter for the suggestion to publish a standardized guide for calculating the allowed amounts at an item or service level, but given the variation in how hospitals contract for items and services, we believe we would be unable to develop a standardized guide for calculating the allowed amounts that would apply to all items and services offered by all hospitals. We thank the commenter for the suggestion to require hospitals to encode the date of the most recent chargemaster update and the weighted average percentage increase applied to gross charges since the date of last update. We may consider these suggestions for future rulemaking.
Final Action: After consideration of public comments, we are finalizing our proposal, with modification. We are finalizing the requirement that hospitals must use EDI 835 ERA transaction data or an alternative equivalent source of remittance data that includes the same information as EDI 835 ERA transaction data would include, to calculate and encode the allowed amounts for items and services based on a percentage or algorithm in the MRF.
(3) Lookback Period for Calculating the Allowed Amounts
In the CY 2024 OPPS/ASC final rule with comment period, we declined to specify a lookback period for hospitals when calculating the estimated allowed amount. This flexibility was intended to reflect the variations in frequency and timing with which hospitals negotiate contracts with payers. The estimated allowed amount was intended to reflect the average reimbursement in dollars a hospital received. Our expectation was that hospitals would calculate the historical amount they received from a payer for an item or service based on the most recent reimbursement under that negotiated algorithm or percentage.
However, as we stated in the proposed rule, we have come to understand that if hospitals use substantially different lookback periods, particularly across multiple years, it could distort the allowed amounts, for example, because of pricing changes over time such as inflation, efficiencies, or the introduction of new products or services. Additionally, hospitals using varied lookback periods reduces comparability across MRFs.
As such, to help ensure that all hospitals calculate the allowed amount data elements consistently and calculate them based on the most recent reimbursements, we proposed to require that hospitals base the median allowed amount, the 10th and 90th percentile allowed amounts, and the count of allowed amounts (discussed in a later section) on EDI 835 ERA transaction data from no longer than 12 months prior to posting the MRF. We proposed that if the negotiated percentage or algorithm associated with the allowed amounts was only used for a portion of the 12-month time period prior to posting the MRF, the hospital would encode the median allowed amount, 10th and 90th percentile allowed amounts, and count of allowed amounts from the EDI 835 ERA transaction data for the portion of time that the percentage or algorithm was used. We proposed that if the negotiated percentage or algorithm associated with the allowed amounts was used for the entire 12-month time period prior to posting the MRF, the hospital would encode the median allowed amount, 10th and 90th percentile allowed amounts, and count of allowed amounts from the EDI 835 ERA transaction data for the entire 12-month time period prior to posting the MRF. We stated that a hospital may therefore need to use different lookback periods to calculate the allowed amounts for each payer, depending on when a contract was negotiated. We acknowledged that there may be situations where the EDI 835 ERA transaction data is not yet final or may change after the allowed amounts are encoded in the MRF due to additional adjustments being applied to a claim(s), so we clarified that the allowed amounts should be based on the EDI 835 ERA transaction data available at the time the MRF is updated.
We considered the efficacy of various lookback periods to calculate and encode the allowed amount data elements, and looked to research to help us gauge potential lookback periods for generating price data based on historic claims remittances.[460] [461] As we discuss below, we considered a 3- or 6-month lookback period, and requiring hospitals to use a rolling 12-month period prior to when the MRF posted.
We stated in the proposed rule that while a shorter lookback period (3- or 6-month) could be useful for accounting for recent healthcare trends and identifying quick changes in allowed amounts, with respect to less frequently provided items and services, we acknowledged that hospitals may not have any remittance data from such a short time period from which to derive the allowed amount data elements, which would result in numerous blanks in the MRF. Additionally, we noted that timely filing limits for claims vary by state and payer, with a typical range of 30 days to 12 months from the date of service, with some longer claim periods.[462] [463] [464] [465] [466] [467] Therefore, we stated that using a 3-month or 6- ( printed page 53996) month lookback period to derive the median, 10th percentile, and 90th percentile allowed amounts, and count of allowed amounts, could result in numerous blanks in the MRF for some items and services and would not accomplish our goal of providing MRF users with meaningful and comparable price data.
After considering these alternative options, we proposed that the lookback period for the median allowed amount, 10th and 90th percentile allowed amounts, and count of allowed amounts (discussed in a later section) be based on EDI 835 ERA transaction data from no longer than 12 months prior to posting the MRF. As discussed previously, we proposed that if the negotiated percentage or algorithm associated with the allowed amounts was only used for a portion of the 12-month time period prior to posting the file, the hospital would encode the median allowed amount, 10th and 90th percentile allowed amounts, and count of allowed amounts from the EDI 835 ERA transaction data for the portion of time that the percentage or algorithm was used. We stated that limiting the lookback period to no more than 12 months prior to posting the MRF would be consistent with section 2718(e) of the Public Health Service Act that refers to “for each year,” and our regulations that require the MRF to be updated at least annually (§§ 180.50(e) and 180.60(e)). Additionally, we stated that for most items and services, it would allow hospitals the ability to amass sufficient claims remittance data at the payer and plan level to encode a price. We stated that where a hospital's payer contracts were initiated or renegotiated in a lesser period than the previous 12 months, with respect to those contracts, we proposed that a hospital would apply whatever period of applicability existed. As noted above, under the proposal, hospitals may need to employ different lookback periods for payers and plans, depending on when a contract was negotiated. We indicated our belief that this approach would help achieve our goal of ensuring pricing information is standardized and easily comparable across hospitals when a hospital's payer-specific negotiated charge is based on a percentage or algorithm, as well as the purpose of Executive Order 14221 to promote disclosure of a real price. We solicited comment on our proposal to require that the lookback period for the median allowed amount, the 10th and 90th percentile allowed amounts, and count of allowed amounts be based on EDI 835 ERA transaction data from no longer than 12 months prior to posting the MRF.
Comment: A few commenters supported our proposal to require that the lookback period for the median allowed amount, the 10th and 90th percentile allowed amounts, and count of allowed amounts be no longer than 12 months prior to posting the MRF. A few commenters indicated that a shorter lookback period would result in numerous blanks in the MRFs. One commenter stated that our proposal would provide a reasonable amount of time without skewing the data by including long lookback periods which would not consider changes such as inflation over that time period.
Response: We thank commenters for their support of our proposal.
Comment: Many commenters expressed concern about the proposal. While most of these commenters supported the standardization of the lookback period, many commenters expressed concern with the 12-month timeframe. Several commenters noted that 12 months is not sufficient as hospitals typically pull the data several months before posting and have claims adjudication lags effectively meaning that hospitals will only be able to use 6 to 8 months of data. One commenter suggested allowing hospitals to select any contiguous 12-month period within the 24 months prior to posting the MRF to allow for delays in payer adjudication. One commenter suggested that CMS allow time for hospitals to incorporate payment data from only the final month of the 12-month lookback period into the MRF before publication. Several commenters recommended a lookback period of at least 18 months. One commenter recommended an 18-24 month lookback period.
Response: We thank commenters for their comments on our proposal. We believe it is important to standardize the lookback period to improve comparability across MRFs by eliminating variances due to different lookback periods, without distorting the data by including data from several years prior that do not account for inflation, efficiencies, or the introduction of new products or services. However, we understand commenters' concerns that requiring a lookback period of 12 months from the date the MRF is posted may not result in data that truly reflects 12 months. As several commenters stated, hospitals may pull the data to encode the allowed amounts several months prior to posting the MRF, thus eliminating those months from the data calculations. We agree with commenters that this would result in several months not being included in the data calculations, effectively shortening the 12-month lookback period. In light of these comments, we are finalizing, with modifications, our proposed lookback period.
We are modifying the proposed definitions of “median allowed amount,” “10th percentile allowed amount,” and “90th percentile allowed amount” at § 180.20 to replace the phrase, “no longer than 12 months” with “no less than 12 months and no longer than 15 months,” thus requiring hospitals to use a lookback period of no less than 12 months and no longer than 15 months prior to posting the MRF. This modification solidifies our intent in the proposed rule to ensure hospitals calculate the allowed amount data elements over a more consistent time period and calculate them based on the most recent reimbursements to enhance comparability. As noted earlier, we agree with commenters that a 12-month lookback period from the date of posting of the MRF may eliminate several months of data if hospitals pull the data a few months prior to posting. We believe the modifications we are finalizing to the lookback period help to address this concern by allowing a longer lookback period in situations where hospitals need time to pull and prepare data prior to posting the MRF. We expect hospitals to use at least the most recent 12 months of data that are available to them. However, in the case where hospitals need additional time to pull or prepare the data before posting the MRF, hospitals may use data from up to 15 months prior to the date the file is posted to help ensure they have adequate data to encode the allowed amounts. We agree with commenters that using a shorter, static 12-month period to derive the data could result in less information. We believe these modifications will result in hospitals encoding 12 months of data for all allowed amounts, striking a balance of fewer blanks in the files with meaningful data. We note that nothing precludes a hospital from using 15 months of data to encode the “median allowed amount,” “10th percentile allowed amount,” and “90th percentile allowed amount.”
Comment: Several commenters stated that a 12 month-lookback period may not be enough time for a meaningful count when the hospital has recently negotiated or renegotiated a contract or an item or service is rarely performed or newly introduced. A few commenters pointed out that, as proposed, there will be varying amounts of data available as hospitals negotiate contracts at different times. A few commenters noted the complexity of a segmented lookback ( printed page 53997) period if a contract changes midyear or if there are multiple allowed amounts that vary as a result of a lookback period that includes a contract change. One commenter requested clarification on whether hospitals should limit the claims utilized in cases where a contract has become effective within the 12-month window. Another commenter requested clarity on what defines the threshold of a change in a contracted amount.
Response: We agree with commenters that a 12-month lookback period for data to encode the “median allowed amount,” “10th percentile allowed amount,” and “90th percentile allowed amount” may not be enough for a meaningful amount of data when the hospital has recently negotiated or renegotiated a contract, or an item or service is rarely performed or newly introduced. We believe the modifications we are finalizing to the lookback period help to address this concern by providing a longer period to amass adequate data to encode a value. As we discussed earlier, we also believe a more standardized lookback period will improve comparability across MRFs. A lookback period of no less than 12 months and no longer than 15 months prior to posting the MRF, as described in more detail earlier in this section, provides for a more consistent lookback period across hospitals than a static 12-month lookback period, which we believe helps to alleviate commenters' concerns about having varying amounts of data from which to encode the allowed amounts for an item or service and the complexity of a segmented lookback period if a contract changes midyear. We agree with commenters that the proposed requirement may have allowed instances where hospitals could have very short lookback periods and a lack of data depending on when a contract was negotiated or renegotiated. We believe that our finalized requirement will address this issue. We agree with commenters that it is important for hospitals to have a lookback period of at least 12 months prior to posting the file to provide MRF users with meaningful and comparable data, and that this may require additional flexibility. As such, and in response to these comments, we clarify that hospitals should report data from both prior to and after a contract negotiation date to capture a full 12-month period regardless of when the contract negotiation takes place. In the event that hospitals renegotiate a contract during the lookback period, hospitals should use data from both prior to and after the negotiation date and encode one figure for each of the allowed amount data elements. Regardless of how many times a contract was modified or the amount changed, the hospital should use all data available over a 12-month lookback period. We are making this change to address commenters' concerns that only including data from the most recent contract negotiation date would limit the amount of data used to encode allowed amounts.
Comment: A few commenters opposed our proposal. A few commenters requested that CMS retain the current requirements which allow hospitals the flexibility to use a lookback period of their choosing from which to encode the allowed amounts. One commenter expressed concern that the proposal will significantly increase the administrative burden on hospitals as the commenter believes it is a complex approach. A few commenters expressed concern that this requirement could require heavy manual analytics to view which rate codes changed during the course of the year at the contract level and extract those specific values.
Response: We appreciate commenters' concerns; however, as we stated in the proposed rule, the current flexibility has resulted in distorted allowed amounts because of pricing changes over time such as inflation, efficiencies, or the introduction of new products or services and reduced comparability across MRFs. By standardizing the lookback period, we believe we are simplifying the process and decreasing the amount of manual work needed to encode the allowed amounts. In particular, if a hospital renegotiates a contract during the lookback period, the hospital will include data from both before and after the renegotiated contract. We believe that this mitigates the additional burden mentioned by commenters about requiring analytics to view which rate codes changed during the course of the year and extract those specific values. We also believe the benefit of the modification we are finalizing to require hospitals to use a lookback period of no less than 12 months and no longer than 15 months prior to posting the MRF outweighs any burden that hospitals may experience, as users of the MRF will be able to better understand and use the data.
Comment: A few commenters offered additional suggestions with respect to this proposal. One commenter suggested that because it will be difficult for MRF users to know how many months of EDI 835 ERA transaction data were used in the allowed amount calculations, hospitals should also be required to encode the effective date of the contract in the MRF. Another commenter recommended an option by which a hospital could leave the field blank if it had less than 6 months of data available. One commenter suggested requiring hospitals to update their files every 6 months to ensure the most recent and complete data is provided and to minimize the number of blanks when hospitals have no historical claim remittance data.
Response: We thank commenters for providing additional suggestions with respect to this proposal, and, as we have explained, we are finalizing modifications to the proposal that we believe address several commenters' concerns. Our intent is to standardize the lookback period to improve comparability without distorting the data with a longer lookback period or creating unnecessary blanks in the MRF with a shorter lookback period. We believe our finalized lookback period of no less than 12 months and no longer than 15 months prior to posting the MRF strikes the right balance. We note that nothing precludes a hospital from adding additional information in the “Additional Notes” field, such as the effective date of the contract.
Final action: After consideration of public comments, we are finalizing our proposal with modification. We are finalizing that the lookback period for the median allowed amount, the 10th and 90th percentile allowed amounts, and count of allowed amounts is a time period of no less than 12 months and no longer than 15 months prior to posting the MRF. As such, we are revising the definitions of “median allowed amount”, “10th percentile allowed amount”, and “90th percentile allowed amount” at § 180.20 to reflect a lookback period of no less than 12 months and no longer than 15 months prior to posting the MRF. We expect hospitals to use at least the most recent 12 months of data that is available to them. However, in the case where hospitals need additional time to pull or prepare the data before posting the MRF, hospitals may use data from up to 15 months prior to the date the MRF is posted to help ensure they have an adequate number of data points to encode the allowed amounts. The 12 to 15 months of data must be contiguous. In the event that a hospital renegotiates a contract during the 12- to 15-month span, the hospital must include data from both prior to and after the negotiation date.
d. Hospitals To Encode the Count of Allowed Amounts
As part of our proposal to require hospitals to encode the median, 10th ( printed page 53998) percentile, and 90th percentile allowed amounts if a hospital's payer-specific negotiated charge is based on an algorithm or percentage, we also proposed to require hospitals to encode the count of allowed amounts from the EDI 835 ERA transaction data used to calculate the median, 10th percentile, and 90th percentile allowed amounts. We proposed that the same count of allowed amounts would be used to calculate the median, 10th percentile, and 90th percentile allowed amounts (90 FR 33797). We solicited comment on our proposed revision to § 180.50(b)(2)(ii)(C) to require hospitals to calculate and encode the count of allowed amounts used to calculate the median, 10th percentile, and 90th percentile allowed amounts, as well as on our proposal that hospitals encode this data element with the actual number of allowed amounts used within the EDI 835 ERA transaction data. We also solicited comment on the alternative we considered of encoding the count of allowed amounts using a standardized range of the number of allowed amounts used within the EDI 835 ERA transaction data, rather than the actual number of allowed amounts, and sought comment on standardized range values of counts of allowed amounts that would be useful. In our proposal, we indicated our belief that this data element would also help drive understanding of the accuracy and completeness of the file, where applicable, and we also proposed that hospitals would be required to disclose why they were unable to calculate the median, 10th percentile and 90th percentile allowed amounts based on a lack of EDI 835 ERA transaction data.
Comment: Several commenters expressed their support for hospitals to encode the count of allowed amounts that were used to calculate the median, 10th percentile, and 90th percentile allowed amounts when the payer-specific negotiated charge is based on a percentage or algorithm. A few commenters indicated that the median, 10th percentile, and 90th percentile allowed amounts need to be interpreted in the context of the number of cases (or quantities) for which each procedure is performed and that disclosing the count of allowed amounts would help MRF users establish the credibility of this data, while further noting the policy aims to improve the accuracy and usability of the pricing data that hospitals must report.
Response: We thank commenters for their support of this proposal. We believe this additional context regarding the number of values used to calculate the median, 10th percentile, and 90th percentile allowed amounts—because more price volatility might reasonably be anticipated with respect to a less frequently performed service—would help MRF users determine whether allowed amounts are reasonably good approximations of what typically would be generated by the payer-specific negotiated charge percentage or algorithm. Knowing the number of allowed amounts used to derive the allowed amounts will better enable MRF users to assess how representative the median, 10th percentile, and 90th percentile allowed amounts are of the overall price distribution for the item or service.
Comment: A few commenters did not support the proposal to add the count of allowed amounts, citing the additional burden to encode this data from EDI 835 ERA transaction data.
Response: We acknowledge comments that cited the additional burden to calculate the count of allowed amounts used when calculating the median, 10th percentile, and 90th percentile allowed amounts, derived from EDI 835 ERA transaction data. In response to these comments, we have increased burden estimates, and as addressed above, have allowed additional flexibility to use an alternative, equivalent source of remittance data to address those concerns. As we stated in the CY 2026 OPPS/ASC proposed rule, we believe that requiring hospitals to encode the count of allowed amounts provides valuable information to allow MRF users to assess how representative the median, 10th percentile, and 90th percentile allowed amounts are of the overall price distribution for the item or service, with the reassurance of their statistical validity increasing with the increasing number of allowed amounts used to make the calculation. We believe that the benefit to the public to be able to understand the amount of data or number of remittances used to determine the allowed amounts outweighs the additional burden incurred by hospitals in encoding the count of allowed amounts. Additionally, as described in more detail later in this final rule with comment period, we are delaying enforcement of these new requirements to April 1, 2026 to allow hospitals additional time to implement the requirements.
Comment: Many commenters expressed concerns that, for low-volume services— especially in rural settings— the requirement to encode the count of allowed amounts could mean publishing very small claim counts that risk disclosure of protected health information and conflict with long-standing CMS and Federal data suppression standards. Commenters stated that publishing such small counts would not only raise HIPAA concerns but could also allow MRF users to back into sensitive information. A few commenters provided alternative approaches to address privacy concerns, including masking counts under either 10 or 11 by encoding an indicator such as “<10” or “<11” as the count of allowed amounts value, stating this indicator would alleviate privacy concerns. One commenter also suggested that CMS only require the 10th and 90th percentile allowed amount values when the count of allowed amounts is greater than 10, explaining doing so would better statistically capture true 10th and 90th percentile values and would, in outlier situations, address privacy concerns. This commenter also recommended that median values could be statistically calculated with two claims or greater.
Response: Commenters' persuasive recommendations with respect to encoding the count of allowed amounts for low-volume services lead us to finalize modifications to our proposal. We believe that masking counts of remittances greater than zero but less than 11 would align with the CMS Cell Suppression Policy [468] and alleviate commenters' concerns regarding any potential patient privacy risks. Should the actual number of allowed amounts within the EDI 835 ERA transaction data, or an alternative, equivalent source of remittance data used to calculate the allowed amount data elements, range from 1-10, hospitals will be required to encode a valid value “1 through 10” to indicate that there were from 1 through 10 allowed amounts used to calculate the median, 10th percentile, and 90th percentile allowed amounts. We will provide further instruction on how to encode the valid value in the CMS Hospital Price Transparency—Data Dictionary GitHub Repository.
We also acknowledge that, in certain situations (for example, in the case of a new hospital, or a hospital contracting with a new payer organization), a hospital may have no historical claim remittance history from which to derive a median, 10th percentile, or 90th percentile allowed amount for a payer and plan. We discussed this in the proposed rule (90 FR 33797), albeit with respect to a 12-month, as opposed to a 12- to 15-month, lookback period. Should a hospital have a “0” count of allowed amounts from the 12- to 15-month lookback period prior to posting ( printed page 53999) the MRF from which to derive the allowed amounts for a particular item or service, we are finalizing that it: (1) must encode “0” as the value for the count of allowed amounts for a specific payer and plan; (2) leave blank the associated MRF median, 10th percentile, and 90th percentile allowed amounts given there is no applicable data to encode; and (3) must encode information to explain the hospital's insufficient claim remittance history in the additional notes data element. In particular, should a hospital have no claims for the payer, we are requiring that a hospital encode why there is a “0” count of allowed amounts in the additional notes data element. We also note that nothing would preclude a hospital from updating its MRF when it has one or more remittances for an item or service. We also require that hospitals exclude zero-dollar claims from the count of allowed amounts because, as we noted in the proposed rule (90 FR 33797), they would result in a misleading and skewed calculation of the median, 10th percentile, and 90th percentile allowed amounts. Zero-dollar claims are health care claims submitted by the hospital to a payer organization where the payment amount is zero. Hospitals submit claims that result in no payment for some items and services because they provide information for the payer to calculate and process payment for the mix of services furnished, not because it results in a separate payment for that item or service. We appreciate the alternative suggested by one commenter that CMS only require the 10th and 90th percentile allowed amount values when counts are 11 or greater, but disagree as we would err on the side of the completeness of the MRF and the data encoded within.
Comment: One commenter stated, in response to the alternative we considered to encode the count of allowed amounts as ranges rather than a precise count, that requiring a range would present unnecessary burden to hospitals; thus, this commenter supported finalizing a precise count in the final rule.
Response: We agree with the commenter that our proposal offers greater precision and information, and reduces the burden on the hospital to encode the count of allowed amounts data element as a range.
Final action: After consideration of the comments received, we are finalizing at new § 180.50(b)(2)(ii)(C)( 2) that if the payer-specific negotiated charge is based on a percentage or algorithm, the hospital must calculate and encode the total number of allowed amount remittances from the EDI 835 ERA transaction data or an alternative, equivalent source of remittance data used to calculate the median, 10th percentile, and 90th percentile allowed amounts. Should the total count of allowed amount remittances be greater than 0 but less than 11, or 1 through 10, a hospital must indicate that at least 1 but no more than 10 allowed amount remittances were used in the calculation, using the valid value described in the CMS Hospital Price Transparency—Data Dictionary GitHub Repository website. We refer readers to section XIX.B.4.c. (2) of this final rule with comment period for discussion of the remittance data we are requiring hospitals to use to determine the count of allowed amounts. Should a hospital have a “0” count of allowed amounts within their remittance data from the 12- to 15-month lookback period from which to derive the allowed amounts for a particular item or service, we are finalizing that it: (1) must encode “0” as the value for the count of allowed amounts for a specific payer and plan; (2) leave the median, 10th percentile, and 90th percentile allowed amounts in the MRF blank as there is no data to encode; and (3) must encode information to explain the hospital's insufficient claim remittance history in the additional notes data element using the examples provided in the Hospital Price Transparency—Data Dictionary GitHub Repository. We also require that hospitals exclude zero-dollar claims from the count of allowed amounts.
5. Modification to the MRF Affirmation Statement
We proposed to supplant the existing affirmation requirement by, instead, specifying at new § 180.50(a)(3)(iii) that, beginning January 1, 2026, hospitals will be required to attest in their MRFs to the following statement: “The hospital has included all applicable standard charge information in accordance with the requirements of § 180.50, and the information encoded is true, accurate, and complete as of the date in the file. The hospital has included all payer-specific negotiated charges in dollars that can be expressed as a dollar amount. For payer-specific negotiated charges that cannot be expressed as a dollar amount in the machine-readable file or not knowable in advance, the hospital attests that the payer-specific negotiated charge is based on a contractual algorithm, percentage or formula that precludes the provision of a dollar amount and has provided all necessary information available to the hospital for the public to be able to derive the dollar amount, including, but not limited to, the specific fee schedule or components referenced in such percentage, algorithm or formula.” We also proposed at new § 180.50(a)(3)(iv) that, beginning January 1, 2026, the hospital must encode within the MRF the name of the hospital chief executive officer, president, or senior official designated to oversee the encoding of true, accurate and complete data as directed in § 180.50(a)(3)(iii).
We stated in the proposed rule (90 FR 33798) that since the proposed new attestation requirements at § 180.50(a)(3)(iii)-(iv) would supplant, with significantly stronger provisions, certain existing requirements, we believe those particular existing requirements would become superfluous and proposed to remove them. Provisions that we proposed to remove, effective December 31, 2025, include the affirmation requirement now at § 180.50(a)(3)(ii) and the requirement at § 180.50(a)(3)(i), which states that, beginning January 1, 2024, each hospital must make a good faith effort to ensure that the standard charge information encoded in the MRF is true, accurate, and complete as of the date indicated in the MRF. Specifically, we stated that we believe the attestation requirement we proposed would not only incorporate those concepts, but, in fact, would mandate significantly heightened hospital recognition of their responsibilities than what we presently require. We stated that we believe our proposed attestation requirements, if finalized as proposed, would reduce public confusion related to whether all standard charges for hospital items and services, where possible, are included within the MRF as dollar amounts. Additionally, we stated that, should our proposal be finalized as proposed, it would establish that the hospital has provided all available information to enable the public to derive a dollar amount, including, but not limited to, the specific fee schedule or components referenced in a percentage, algorithm or formula. We explained that we believe this would provide the necessary reassurance that hospitals have provided in their MRFs meaningful, accurate information to MRF users about their standard charges for health care items and services in order for those users to fully realize the intended use of the MRFs as expressed in the CY 2020 HPT final rule—that is, for enhancing the public's ability to use the data in, for example, innovator developed consumer tools and in EHRs at the point of care for value-based ( printed page 54000) referrals, or to aggregate and use the data to increase competition.
We stated in the proposed rule (90 FR 33799) that requiring an individual's name be specified would also, we believe, expedite our ability to quickly identify an individual at the hospital to obtain, where necessary, further clarity regarding the MRF data. In connection with this proposed requirement, we proposed to add a new general data element, attester name, and stated that, should the proposal be finalized as proposed, we would provide, on the CMS Hospital Price Transparency—Data Dictionary GitHub Repository website, instruction on how to encode this data element.
We also sought comment on an alternative approach, that is, whether CMS should require hospitals to post on their publicly available websites that host the hospital MRF, a standalone attestation document that would be signed by a hospital senior official. We received public comments on these proposals.
Comment: Several commenters supported the proposal to strengthen the current HPT requirements by replacing the “affirmation” standard with a more robust “attestation.” These commenters agreed that hospitals should attest that they have included all applicable payer-specific negotiated charges that can be expressed in dollars, and for charges not knowable in advance or that cannot be expressed as a dollar amount, hospitals should be required to include in the MRFs all necessary information available to enable the public to derive the dollar amount—such as the specific fee schedule or components referenced in any percentage, algorithm, or formula. A few commenters appreciated the proposal to rescind existing requirements that a hospital merely make a “good faith effort” to ensure their MRF data are true, accurate, and complete. One commenter expressed specific support for the removal of the phrase “to the best of their knowledge” from the proposed attestation statement. One commenter stated that without such attestation, hospital accountability for encoding accurate and complete data does not exist and the new requirement will demonstrate to the public that the Federal government and hospitals take seriously their obligation to provide accurate, reliable, actionable prices. One commenter underscored the requirement that hospitals must provide all necessary information for an average patient to determine the dollar amount without third-party assistance.
Response: We appreciate commenters' support for the proposed attestation statement. We proposed to adopt this attestation to make clear to hospitals and MRF users our expectations that the hospital will accurately and completely encode all available standard charge information. If the hospital established a payer-specific negotiated charge as a dollar amount, the hospital must display the payer-specific negotiated charge as a dollar amount. If the hospital is unable to display a payer-specific negotiated charge solely as a dollar amount, such as when the payer-specific negotiated charge is further modified by a percentage or algorithm, the hospital will be required to provide all necessary information available to the hospital to derive a dollar amount. We intend this public declaration to establish for MRF users and for CMS actionable certainty on the accuracy and completeness of the standard charge information displayed. We also intend that this public declaration will increase hospital accountability to MRF users that the data is complete as of the date indicated in the file. We believe demonstrating our strengthened expectations will result in the public display by hospitals of more meaningful data for MRF users. We agree with commenters that supported our removing the good faith effort provision, as this statement did not provide adequate assurances that all standard charge information was encoded in the MRF.
As discussed in more detail later in this section, we are finalizing the proposed attestation requirement, but with modification to retain the current phrase, “to the best of its knowledge and belief.” We appreciate the comment that patients will use the MRF to be able to understand the dollar amount for an item or service, but caution that the standard charge information encoded within the MRF reflects the standard charge, or the regular rate established by the hospital for an item or service provided to a specific group of paying patients, which is not necessarily reflective of an individualized charge for an episode of care. We encourage patients to use the HPT consumer-friendly display files and tools and the TiC tools that take into consideration the patient's plan design and avail themselves of the protections provided under the NSA, in order to obtain an individualized charge for their care.
Comment: Many commenters opposed the proposed attestation language, in particular, the phrase “has provided all necessary information available to the hospital for the public to be able to derive the dollar amount.” Many commenters stated that the proposed attestation fails to account for the reality of hospital billing, which they stated depends, in significant part, on insurer behavior and calculations which in turn depend on a host of factors that cannot be easily calculated by a third party, such as whether the service was provided in conjunction with other services, the applicability of any volume discounts or stop loss amounts, as well as other unique features of a patient's insurance plan. Commenters also noted that inpatient hospital reimbursement is typically based on an entire treatment case rather than a single service as well as on patient acuity, and stated that in many cases all the procedures that may be required cannot be known in advance. Several commenters stated that the phrase “has provided all necessary information available to the hospital for the public to be able to derive the dollar amount” incorrectly assumes that a member of the public is (1) capable of prospectively anticipating their every need during an episode of care; and (2) has the specialized knowledge and understanding of processes and nuances that underly hospital billing, coding, and reimbursement and can accurately use the algorithms displayed. Commenters stated that there will always be some additional piece of information that an individual could use to more accurately calculate a particular charge and noted that hospitals can never be entirely certain that they have provided “all necessary information . . . for the public to be able to derive the dollar amount.” In addition, commenters stated that publicizing negotiated rates and frameworks or even the full text of every single payer contract would not be sufficient to enable a patient to calculate their final bill, noting that because of insurers' penchant for proprietary systems and calculations, there may be nuances that are not readily available to or shareable by hospitals.
Several commenters further expressed concern that the proposed attestation language appears to require hospitals to guarantee the absolute accuracy of complex data and apply a strict liability standard and were concerned about the practicality of expecting flawless MRF data given the volume and complexity of hospital contracts and payment arrangements. Several commenters noted that the proposed median, and 10th and 90th percentile, allowed amounts are based exclusively on EDI 835 ERA transaction data which are developed by the payer and that hospitals do not have control over the accuracy and completeness of this data. Commenters stated that it would be unreasonable to hold hospitals accountable for validating and attesting ( printed page 54001) to the accuracy and completeness of data developed by a third party. One commenter noted that individual patient cost data requires detailed information not necessarily within hospitals' control (for example, payer algorithms, plan features, etc.). A few commenters also expressed concern that the proposed attestation that the hospital “has provided all necessary information available to the hospital for the public to be able to derive the dollar amount” risks ensnaring hospitals and health systems in a trap: since hospitals are unable to provide this detailed information, they cannot meet the new requirements of the attestation.
Given this, many commenters recommended retaining the existing “good faith effort” affirmation and the phrase “to the best of the hospital's knowledge and belief,” which they stated reflects what hospitals can realistically provide without raising the risk of liability for the failure to identify every single piece of information (“all”) that may exist. One commenter stated specifically that this is the most responsible option given the complexity of hospital price transparency reporting and the structural limits on the data hospitals receive from payers, and asserted that these qualifying phrases represent bona fide, reasonable commitments to integrity and transparency which are commonly used in legal certifications and affirmations. One commenter specifically asked that CMS include the language, “available information to best of our knowledge.” A few commenters stated that, at the very least, the agency must clarify the reasonable scope of “all necessary information.”
A few commenters stated that, because it would be impossible for hospitals to unilaterally provide sufficient information to enable patients to undertake these calculations on their own, the proposed attestation requirement would be arbitrary and capricious. Some of these commenters asserted that the imposition of an impossible requirement is, by definition, arbitrary and capricious and cited case law to support this belief. A few commenters further stated that at the very least, CMS' failure to consider the operability of this requirement would be arbitrary and capricious, with one commenter again citing case law to support their position.
Response: In the CY 2020 HPT final rule we asked hospitals to display their standard charges, and, subsequently, we encountered many MRFs strewn with blanks as hospitals indicated they could not display their charges as a dollar amount. In the CY 2024 OPPS/ASC final rule with comment period, based on hospital statements that many hospitals could not calculate a payer-specific negotiated charge as their payer-specific negotiated charge was based on a percentage of billed charges or based on a formula or algorithm that may be determined after the episode of care, we finalized new data elements, the payer-specific negotiated charge percentage and the payer-specific negotiated charge algorithm, to provide MRF users with an understanding of how the payer-specific negotiated charge was calculated, and a contextual data element, the estimated allowed amount, which is a dollar amount reflecting the average of the historical reimbursement (allowed amount) received from a third party payer for an item or service, required to be displayed when no payer-specific negotiated charge dollar can be encoded. We allowed hospitals the flexibility to describe their algorithms. Some hospitals met these additional requirements by providing additional data that gave MRF users valuable understanding on how the payer-specific negotiated charge is calculated and enough information that innovators, employers, and researchers could then draw meaningful comparisons. But other hospitals continued to provide opaque descriptions of algorithms with no anchor to help MRF users understand the calculation, sometimes using nine 9s in the estimated allowed amount field,[469] and with some indicating they did not have enough claims data to calculate any estimated allowed amounts for any items or services in their MRF. We are issuing this fourth final rule to reiterate, and further, our goal of ensuring that pricing information is standardized and easily comparable across hospitals. In this final rule with comment period, we reinforce our requirement that hospitals must calculate and encode a payer-specific negotiated charge dollar amount when possible and, when the payer-specific negotiated charge is based on an algorithm or percentage, must do more than merely offer a good faith effort to enter their payer-specific negotiated charge percentage or algorithms; instead, hospitals must provide MRF users with a detailed explanation, when the payer-specific negotiated charge is based on a percentage or algorithm, of how the payer-specific negotiated charge is derived. In this rule, we describe the data source to be used to calculate the allowed amount data elements, providing flexibility on the lookback period to calculate the data, and attempting, again, to provide MRF users more fulsome data.
We disagree with commenters who stated that it is impossible for hospitals to comply with our requirements. We understand that hospitals may not know plan design, payer-based algorithms, or patient acuity, that are determined after the episode of care is complete, and also understand that an MRF in JSON or CSV that captures standard charge information at the item or service level cannot possibly account for every patient scenario. But we remind commenters that an MRF represents a point in time—as of the date in the file—and require that hospitals attest that the information therein is accurate and complete. We require that hospitals calculate a payer-specific negotiated charge in a dollar amount, and, if the hospital cannot, that it provide all the information “available to the hospital,” which would include information in the hospital's systems and payer contracts, in a way that a reasonable MRF user can understand.
We understand that some data is provided to hospitals from payers, and that hospitals cannot vouch that the EDI 835 ERA transaction data provided by the payer is accurate and complete, but hospitals can attest that their calculations of the allowed amounts follow the requirements specified within this regulation completely and accurately. We likewise understand that hospitals cannot provide everything that everyone might need to know, because such information may not be available to them, but we expect them to provide sufficient and necessary information that is available to them for a reasonable objective person to derive the price for an item or service. Through this final rule with comment period, we are requiring hospitals to take the steps necessary to provide all information available to the hospital, including in their systems and contracts, to support the public to be able to derive the dollar amount. Provided here are some examples.
If the payer-specific negotiated charge is based on 115 percent of the Medicare allowed amount, the hospital is required to perform the necessary calculations ( printed page 54002) and encode the payer-specific negotiated charge dollar amount. If the payer-specific negotiated charge is based on 87 percent of total billed charges, when the payment reflects the total of all charges provided at the time of the service, the hospitals should encode 87 in the payer-specific negotiated charge percentage data element and then calculate the 10th percentile, median, and 90th percentile allowed amount data elements in dollars so that MRF users can use that data to draw comparisons.
The HPT regulations state that if the payer-specific negotiated charge is based on a percentage or algorithm, the MRF must also describe the percentage or algorithm that determines the dollar amount for the item or service, and, beginning January 1, 2025, calculate and encode an estimated allowed amount in dollars for that item or service. For payer-specific negotiated charges based on algorithms and payment formulas, we believe that all necessary information means all factors that comprise an algorithm that determines how the payer-specific negotiated charge is derived should be encoded in the MRF. Examples include, but are not limited to, patient demographics, hospital demographics, clinical characteristics, length of stay, resource utilization, and adjustments based on mix of services performed.
Figure 1 is an example of where a hospital provides all the contractual data on how the price is calculated, but in a way that a reasonable, objective MRF user would not be able to interpret it and use it to draw comparisons; therefore, Figure 1 illustrates an approach that would not meet the regulatory requirements:
Figure 1: Example of an Unacceptable Payer-Specific Negotiated Charge Algorithm
Figure 2 provides an example where a hospital provides an algorithm that provides adequate context so that an MRF user may be able to replicate the formula to derive a price.
Figure 2: Example of an Acceptable Payer-Specific Negotiated Charge Algorithm
Figure 3 provides an example where the case rate is encoded as a payer-specific negotiated dollar amount, and then further modified.
( printed page 54003)Figure 3: Example of an Acceptable Payer-Specific Negotiated Charge Algorithm
We will provide additional examples in the CMS Hospital Price Transparency Data Dictionary GitHub Repository.
We have also observed that some hospitals provide their contractual language in the payer-specific negotiated charge dollar data element and provide additional useful descriptions of what the contractual language includes using one of the additional notes data elements provided. Nothing would preclude a hospital from also providing a plain language description in the additional notes data elements in the MRF.
We disagree with commenters that our proposal is arbitrary and capricious. We are asking hospitals to provide within the MRF, all standard charge information that they have in their systems and contracts to support the public to be able to derive the dollar amount. We are revising the attestation statement to retain the phase “to the best of its knowledge and belief” to acknowledge that there may be varied interpretations of contract calculations that could lead to variation in price, that the hospital may not know at the time of MRF development every factor that may impact the price or the payer may not have disclosed all factors to the hospital, and that for many hospital services the price is determined after services are performed at the episode of care level, not at the line item level, which would hinder an MRF user from being able to derive a price prior to services being performed.
Comment: A few commenters questioned that the attestation in its current form risks exposing hospitals to undue risk under the False Claims Act (FCA) and stated that CMS' existing assurances that this requirement falls outside the scope of the FCA are insufficient to allay hospital and health system concerns. Although commenters appreciated CMS' perspective, as set forth in the proposed rule, that noncompliance with this new requirement would not implicate the FCA, commenters expressed concern that there is no evidence that the Department of Justice, regulators, or judges would agree. These commenters stated that, to ensure that these new attestation requirements do not generate FCA challenges, CMS must articulate why the rule would not give rise to FCA liability. Commenters suggested that the Department of Justice could co-issue a policy stating that it would move to dismiss all FCA cases based on this attestation requirement, and stated that, short of that, no signatory could be confident that an attestation would not be used against them, or their institution.
Response: We appreciate commenters' concerns but forcefully reiterate, as stated in the CY 2024 OPPS/ASC final rule with comment period (88 FR 82086, 82116), that this final rule with comment period falls outside the scope of the FCA.
In tandem with that, and as discussed in response to a comment summary earlier in this section, we have elected not to finalize the removal of the statement, “to the best of the hospital's knowledge and belief” from the attestation statement, to reflect that we do not view the attestation as an absolute guarantee of perfection, but instead, as a reassurance to CMS and MRF users that to the best of the hospital's knowledge and belief, it has included all applicable standard charge information in accordance with the requirements of § 180.50, and the information encoded is true, accurate, and complete as of the date in the file. We believe that the inclusion of this statement should help allay commenters' concerns. We further note that while we have strengthened the attestation statement to require the hospital to attest that it has provided “all necessary information available to the hospital for the public to be able to derive the dollar amount” in cases where the payer-specific negotiated charges cannot be expressed as a dollar amount in the MRF or are not knowable in advance, we emphasize that this attestation requires the hospital to provide information that is “available” ( printed page 54004) to the hospital, which does not require hospitals to provide any information to which they do not have access.
Comment: Several commenters expressed concern that the requirement to include “all necessary information” for the public to derive the dollar amount for cells that use algorithms or formulas could expose hospitals to significant legal challenges because many pricing algorithms rely on proprietary groupers or logic developed and owned by payers or third-party vendors. Commenters stated that such methodologies or reimbursement structures are often protected under intellectual property agreements or confidentiality clauses in hospital contracts, and requiring hospitals to encode such logic in the MRF could expose them to contractual breaches or legal liability. Commenters further noted that encoding such information may not even be technically possible because some components are completely and wholly managed by the payer and/or third-party vendor. A few commenters generally expressed concern that under the proposed attestation language, hospitals would be required to include privately negotiated contract terms in their MRFs or contract data that is contractually protected. A few commenters stated that the original price transparency rule struck an appropriate balance by requiring disclosure of prices without mandating hospitals to publish the full text of their contracts or all the underlying formula mechanics. Commenters asserted that the proposed attestation requirement goes well beyond the intent of helping patients understand prices, veering instead into revealing trade secrets that do not meaningfully help a patient estimate their out-of-pocket cost. Commenters believed that the attestation's purpose should simply be to ensure hospitals are making a good-faith, comprehensive effort to post the required standard charge information
Response: Consistent with the Executive Order 14221, Making America Healthy Again by Empowering Patients with Clear, Accurate, and Actionable Healthcare Pricing Information, the requirements we are finalizing advance our longstanding goals of equipping health care consumers with more meaningful and actionable price information to make well-informed healthcare decisions and supporting a more competitive, innovative, affordable, and higher quality healthcare system. This extends to ensuring that the MRF contains all accurate and complete information that the MRF users may need in order to be able to derive a payer-specific negotiated charge, including those formulas and algorithms used to derive the payer-specific negotiated charge.
Hospitals have had 4 years to demonstrate their good faith effort to make public the required standard charge information, but in that time we have assessed hospitals 2,531 warning notices, required 1,419 plans of corrective action, and imposed 27 CMPs (totaling 3,977 enforcement actions, and in the proposed rule we noted that only 7,416 hospitals meet our regulatory hospital definition (90 FR 33835)). Meanwhile, many MRF users are still confused by the dearth of data in the MRF files and whether the hospitals have indeed disclosed all the items and services they offer and the corresponding required standard charges for those items and services.[470] As we stated earlier in this rule and in prior rules, we believe MRF users (for example innovators, researchers, employers) will use this data to analyze and draw more meaningful comparisons of hospital standard charge information, identify outliers and encourage market competition, support more effective contract negotiations, and develop downstream tools to support patient price comparisons.
We do not discount commenters assertions that various confidentiality provisions may generally protect the terms of various contracts or products that intersect with price transparency, but reiterate the point we articulated in the CY 2020 HPT Final Rule (84 FR 65544) that such contracts typically include exceptions where Federal law requires such a disclosure. Our view, which we explained in the CY 2020 HPT Final Rule (84 FR 65544), is that the Defend Trade Secrets Act of 2016 (18 U.S.C. 1905), which we do not believe is applicable here, applies only to trade secrets that are “misappropriated,” which is defined by reference to, among other things, “improper means,” where there was a “duty to maintain the secrecy,” or “accident or mistake.” We do not believe any of the meanings of the term “misappropriation” under the Defend Trade Secrets Act apply to a circumstance where an agency rule requires disclosure of certain information. Making these charges public enables, among others, health care consumers to be better informed about the cost of care, enabling them to compare options and make informed decisions. In turn, this transparency fosters competition among providers, advancing our broader goal of lowering overall healthcare costs.
We also disagree with commenters asserting there are many pricing algorithms that rely on proprietary groupers or logic developed and owned by payers or third-party vendors for which they cannot provide information used to determine what the hospital will be reimbursed. While there may be components of such negotiated fee schedules that may depend on proprietary data or algorithms (such as proprietary groupers), this would not preclude a hospital from being able to provide all the information available to it to reflect the negotiated amount for that item or service, either as a payer-specific negotiated charge dollar amount or the formula or algorithm and the historically received allowed amounts. To support hospital implementation efforts, we will provide examples of how to encode specific scenarios in the CMS Hospital Price Transparency—Data Dictionary.
Comment: Several commenters were concerned that implementing the proposed attestation statement would overwhelm the file with conditional logic and supporting documentation that cannot conform to the file schema and would undermine the goal of machine-readability and comparability.
Response: We thank commenters for these comments. In addition to the examples we have provided, we will provide additional examples on how to encode data and information required in the attestation statement on the CMS Hospital Price Transparency—Data Dictionary GitHub Repository website.
Comment: A few commenters supported our proposal to encode within the MRF the name of the hospital chief executive officer, president, or other senior official designated to oversee the encoding of true accurate and complete data. These commenters stated their belief that this requirement appropriately enhances executive accountability and reinforces the importance of leadership engagement in ensuring transparent and reliable pricing information for the public.
Response: We thank commenters for their support of this proposal. As discussed in the proposed rule (90 FR 33799), we believe the inclusion of the name of a hospital senior official will provide CMS actionable and enforceable certainty, and MRF users additional assurance, including in situations where the hospital's payer-specific negotiated charge is based on a contractual algorithm, percentage, or formula by which a hospital genuinely cannot specify a dollar amount, that the data were reviewed and verified by the ( printed page 54005) hospital's leadership, and would expedite our ability to quickly identify an individual at the hospital to obtain, where necessary, further clarity regarding the MRF data. For the reasons stated previously, we believe inclusion of the name of the CEO, president, or senior hospital official designated to oversee the encoding of true, accurate, and complete data in the MRF is reasonable and appropriate to achieve our stated goals.
Comment: Many commenters disagreed with the proposal to require hospitals to encode the name of the CEO, president, or senior hospital official designated to oversee the encoding of true, accurate, and complete data in the MRF because it would be unnecessarily burdensome to the hospital executive, hinder their ability to fulfill their primary role of focusing on strategic vision and overall governance, or introduce delays without improving accuracy. Several commenters stated that it would be unreasonable to expect the executive to personally validate potentially hundreds of thousands or millions of data points, noting that such executives are likely to rely on staff review and confirmation of the data. Instead, commenters stated that CMS should trust the good faith of others within the hospital who are far closer to the information and can verify its accuracy far more directly and easily than someone higher on the organizational chart with broader responsibility. Commenters also noted that compiling and interpreting standard charge information is extraordinarily complex, requiring interpretation of numerous assumptions and formulas, changing regulatory requirements, and intricate contract terms, and expressed concern that there is no recognition that given the vastness of the data required, an error could be overlooked despite all efforts to prevent it. One commenter further noted that there are entire software solutions designed to understand payment rates. Several commenters stated that the attestation requirement places a hospital executive in the untenable position of having to certify information beyond their practical control or granular knowledge, potentially exposing them to legal risks, with one commenter specifically recommending that the attestation be provided by the compliance officer or revenue cycle leader directly involved in the MRF development.
A few commenters expressed concern that CMS offered no support for requiring hospitals to encode the name of the CEO, president, or senior official and stated that “conclusory reasoning” cannot withstand scrutiny. Commenters noted that the agency did not consider the significant administrative burdens on the named attester, nor did it provide any support for why other employees within a hospital would not provide “meaningful, accurate information.” These commenters stated that absent actual support for this proposal, the agency should not add to the burdens of hospital leaders.
A few commenters generally stated that the current affirmation is sufficient and that it is unnecessary to require hospitals to make an attestation and encode the name of the hospital CEO, president, or senior official. One commenter asked if the hospital executive name encoded in the proposed attestation needed to match who the hospital designates as the CMS authorized and delegated official when under compliance review.
Response: We disagree with commenters and believe the reassurance to MRF users that the MRF contains data that is accurate and complete as of the date encoded in the MRF far outweighs any burden to the hospital executives whose names are encoded in the MRF. We believe the proposed requirement that hospitals encode in the MRF the name of a senior hospital official as the attester appropriately strengthens the attestation and the reassurances it provides to MRF users and CMS by providing accountability at a senior management level for the accuracy and completeness of the data. While we recognize that the senior hospital executive may not be directly involved in MRF development, we expect the named attester, as part of their role in the overall governance of the hospital, would, as necessary, be able to consult with the appropriate personnel more directly involved in MRF development, such as a compliance officer or revenue cycle leader as suggested by a commenter, in order to obtain the necessary information to make the required attestation. As such, we do not believe the proposed attestation requirement will hinder the executive's primary role within the hospital, create delays in the MRF development process, or place the attester in a position of having to attest to information of which they have no knowledge. Other CMS programs require a senior hospital executive to attest to complex financial information; for example, CMS requires a Chief Financial Officer or hospital administrator to read, prepare, and sign CMS-2552-10 (Hospital and Hospital Health Care Complex Cost Report) after the cost report has been completed.[471]
We have provided some flexibility on the title of the senior hospital executive who would attest to the file's accuracy and completeness, and indicated in the proposed rule (90 FR 33799) that we would expect the senior hospital official who is named as the attester in the file would be the same individual who would submit to CMS, if requested as part of CMS' monitoring oversight, a certification of the accuracy and completeness of the MRF data under § 180.70(a)(2)(iv). We recognize that we did not include in section XXIII. (Collection of Information Requirements) the applicable burden level for CEO review of the MRF and have added an additional 2 hours to both the one time and annual burden estimates.
Comment: A few commenters stated that requiring the proposed attester name data element would raise concern that any unintentional errors in the MRFs may be punitively attached to the name of a single person, including exposing hospital executives to excessive liability or public criticism over unintentional, technical variances. The commenters stated that since the MRFs are published on the public-facing websites of hospitals, they urged CMS to not finalize the proposed “attester name” data element. One commenter believed that this would pose unnecessary privacy and security concerns and would not, in any way, advance the utility of price transparency regulations. One commenter further stated that the proposal raised potential safety and security concerns for the hospital executive named on the MRF, noting that requiring a named individual to publicly affirm the accuracy and completeness of complex pricing data could inadvertently expose that person to unintended harm. Given this, commenters urged CMS to retain the existing language requiring a good faith affirmation that the data is true, accurate, and complete.
Response: We acknowledge commenters' apprehension that the addition of the hospital senior official name could create privacy or security concerns; however, senior officials' names are already disclosed in multiple other public documents, including on the hospital's own website and in press releases, on state hospital licensure websites and, for Medicare-certified hospitals, information on hospital administration, including senior ( printed page 54006) officials' names, is publicly disclosed on the CMS website in the “Hospital All Owners” dataset and well as in the National Plan & Provider Enumeration System (NPPES) NPI Registry. Further, we believe that, by virtue of our modification that includes retaining the statement “to the best of its knowledge and belief,” hospitals and senior hospital officials would be less likely to receive public criticism over unintentional, technical variances.
Comment: A few commenters noted that requiring attestation by a CEO or other senior official would be duplicative of other existing hospital price transparency regulatory requirements. One commenter noted that hospitals are currently required to submit a certification of MRF data accuracy and completeness from an authorized hospital official on CMS' request; and to include a point of contact in their .txt files. This commenter believed these existing processes are fully appropriate and adequate, particularly given the complex nature of this data, noting that having a centralized hospital liaison who can either directly answer an MRF data question and/or refer to the appropriate individual, as applicable, would ensure efficiency and clarity. This commenter further noted that CMS already has the authority to request a certification. One commenter urged CMS to omit the proposed change to add another, more senior contact person in the MRF and keep the process as is. Another commenter stated that if CMS requires encoding the CEO's name within the MRF, it recommended direct communication with the individual named in the .txt file first to address any concerns, and if no response is provided, communication be directed to the CEO and the individual, recommending that CMS prioritize the use of email communication over postal mail to prevent delays. Another commenter stated that seeking a certification from a hospital official would be duplicative of the initial attestation without offering additional clarity on whether the file is truly compliant in the eyes of the regulators.
Response: We disagree with commenters that this information is duplicative as the MRF .txt file contact name is a person who can answer technical questions from entities such as innovators and researchers about the file structure and data encoded within it, but not attest to accuracy and completeness of the data. We further disagree that the proposed requirements are duplicative of the certification requirement at § 180.70(a)(2)(iv) as our stated goal for adding the attester name was to provide further reassurance to the public of the accuracy and completeness of the MRF, while the certification requirement is provided directly and only to CMS under the compliance process.
Comment: We received one comment in response to our alternative that the attestation reside in a separate document on the hospital's website. That commenter responded that the attestation statement and attestor name should reside within the MRF.
Response: We appreciate the comment and agree that the attestation confirmation and attestor name should reside in the MRF.
Comment: We received comments on a few additional alternatives for this proposal. One commenter suggested the addition of a signature within the file, and another commenter suggested we require the senior hospital official to specifically have one of the following titles: the hospital's Chief Executive Officer, Chief Financial Officer, Chief Revenue Officer, or their equivalents. One commenter suggested an alternative, that in order to provide all necessary information for the public to derive a dollar amount, the hospital should make public a separate formula sheet providing the complete underlying formula by which actual reimbursement would be determined, because algorithms, formulas, and percentages make the file no longer machine-readable.
Response: We decline to require a signature within the MRF file, given that the MRF required formats, JSON and CSV, do not permit the inclusion of electronic signature solutions. If a hospital is found noncompliant, CMS already has the authority under § 180.70(a)(2)(iv)-(v) to require the authorized hospital official to submit to us a signed certification as to the accuracy and completeness of the standard charge information encoded within the MRF. We decline at this time to be more prescriptive regarding the title of the senior hospital official, as we stated in the proposed rule that we want this name to align to the authorized hospital official who would submit to us the aforementioned signed certification, but we may revisit this suggestion in future rulemaking. We also decline the suggestion to require hospitals to provide additional files outside of the MRF, and require that hospitals encode all relevant data within the MRF.
Final Action: After consideration of public comments, we are finalizing, effective January 1, 2026, our proposal to supplant the existing affirmation requirement with the proposed attestation statement at new § 180.50(a)(3)(iii), with a modification to add the following phrase to the beginning of the attestation: “To the best of its knowledge and belief,”. In addition, we are finalizing, as proposed, new § 180.50(a)(3)(iv) to require that, beginning January 1, 2026, the hospital must encode within the MRF the name of the hospital chief executive officer, president, or senior official designated to oversee the encoding of true, accurate and complete data as directed in § 180.50(a)(3)(iii). We are also modifying proposed § 180.50(a)(3)(i) and (ii) to clarify that these provisions apply beginning January 1, 2024 through December 31, 2025, such that these provisions will no longer be effective as of January 1, 2026. Finally, as discussed in section XIX.B.7. (Effective Dates), although these revisions to the attestation requirements will be effective January 1, 2026, we will delay enforcement of these revisions until April 1, 2026.
6. Requirement To Report Hospital National Provider Identifier (NPI) Information in the Machine Readable File
We proposed to revise § 180.50(b)(2)(i)(A) to require hospitals, beginning January 1, 2026, to report a unique identifier, specifically their NPI(s), in their MRFs. As we explained in the proposed rule, we believe that having hospitals add their NPI(s) to the MRF would improve the comparability of HPT and other healthcare data, including health plan transparency data from the TiC MRFs. Below, we explain the details of this proposal, including how we proposed that hospitals would encode their NPI(s) in their MRFs.
An NPI is a unique 10-digit number used to identify healthcare providers and organizations, including hospitals.[472] All healthcare providers that are HIPAA-covered entities must obtain an NPI.[473] Health care providers who are individuals are assigned a Type 1 NPI and healthcare providers that are organizations are assigned a Type 2 NPI (69 FR 3440). Type 2 NPIs are also known as organizational NPIs. “Subparts” of organizations—which are components of the same organization that may be separately licensed or identified [474] —may also obtain a Type 2 NPI (69 FR 3441) if they conduct HIPAA ( printed page 54007) standard transactions separately [475] from the main organization (45 CFR 162.410(a)(1)). Entities and individuals maintain NPIs unless they are deactivated upon request, death, or dissolution (45 CFR 162.408(c)), and NPIs do not change if provider name, EIN, or state licensure changes (69 FR 3441). There are several internet-based NPI lookup tools available online, including CMS' NPPES NPI registry.[476] NPIs are commonly used in other CMS systems for financial transactions, and for other healthcare data sets, including claims, utilization, and quality data sets.
Under the current HPT regulations at § 180.50, hospitals must provide identifying information, including hospital name, address, license number, and the Employer Identification Number (EIN) either in the MRF file name or the file itself. We stated in the proposed rule that while these elements help to identify the hospital, interested parties have told us that they are inadequate to facilitate comparing hospital MRF data with other datasets that include hospital-related information and that a standard identifier would bolster these efforts. In particular, commenters stated that the lack of a standard identifier in the MRF hinders efforts to compare standard charge information across files and limits opportunities to automate the comparison and analysis of HPT and TiC MRF data. Innovators, researchers, and other MRF users have stressed to us the importance of including standard identifiers to streamline data and reduce the complexity of analyzing numerous and different disclosures. The currently required hospital license number is useful to help crosswalk the name of the hospital with the state license number, but because it differs from the identifier required in the TiC files, innovators and researchers have noted that they find it difficult to compare across files. We also note that EINs, while required as part of the naming convention for the hospital MRF and included in the TiC MRF, are generally not included in CMS datasets or other public financial and claims datasets. Therefore, we stated in the proposed rule that we believe it is important to require hospitals to report a standard identifier, specifically the NPI, which is used in the TiC MRFs and in CMS systems such as the Provider Enrollment, Chain, and Ownership System (PECOS), to maximize comparability across data and files.
Moreover, Executive Order 14221 directs HHS to ensure that pricing information is standardized and easily comparable across hospitals and health plans.[477] We stated in the proposed rule that, to this end, we believe it is important to align the HPT MRF and the TiC identifier data element. Under the TiC final rule (85 FR 72158), and as described in the TiC GitHub schemas for the “In-Network File,” “Out-Of-Network Allowed Amount File,” and the optional “Provider Reference File,” most group health plans and health insurance issuers must post pricing information, and such pricing information must be associated with a provider's NPI to ensure that consumers have reliable data and can make informed healthcare purchasing decisions.[478] We explained in the proposed rule that we believe aligning the NPI across the HPT and TiC MRFs would support improved cross-comparison among hospital data and health plan data, providing users of both MRFs further context about hospital standard charges.
We specifically proposed to require, beginning January 1, 2026, that hospitals report, in a newly created general data element in the MRF, any Type 2 NPI(s) that has a primary taxonomy code starting with `28' (indicating hospital) or `27' (indicating hospital unit) and that is active as of the date of the most recent update to the standard charge information. We proposed to limit the Type 2 NPI(s) that hospitals would report to only those that meet this taxonomy criteria, because while hospitals may have more NPIs beyond these criteria for other departments or units, these taxonomy codes limit the number of NPIs to only those indicating hospital or hospital unit. In the case that hospitals have more than one NPI that meet the proposed criteria above, we proposed that hospitals would be required to report in the general data element all active Type 2 NPIs meeting the criteria. We stated that we would include additional technical instructions in the CMS data dictionary and CSV template and JSON schema in the Hospital Price Transparency—Data Dictionary GitHub Repository available at https://github.com/CMSgov/hospital-price-transparency. We sought comment on our proposal and any additional taxonomy codes that would be necessary or helpful to consider.
We considered in the proposed rule, as an alternative, that should a hospital have multiple Type 2 NPIs, it would be required to report only one NPI. We stated that with this alternative, MRF users could crosswalk the NPI to identify additional NPIs. A review of the publicly available January 2025 data from PECOS, the online Medicare enrollment system, found that only approximately 10 percent of hospital enrollment applications reported multiple NPIs.[479] This data was cross-walked with NPPES data to find the provider taxonomy code (a 10-digit code that designates classification or specialization), whether the NPI was still active in the system, and whether an NPI was classified as an organization subpart. The majority of reported NPIs for applications with multiple NPIs were active. Some applications reported as many as 27 NPIs with a hospital or hospital unit taxonomy. However, the median number of NPIs with a hospital or hospital unit taxonomy was two and the average number of NPIs with a hospital or hospital unit taxonomy was 1.9. For this reason, we stated in the proposed rule that we believe requiring hospitals to include NPIs that meet our proposed criteria would not pose a significant burden or, for most hospitals, significantly increase the amount of data stored in the MRFs.
We also considered requiring that hospitals include other identifiers in their MRF, such as the CMS Certification Number (CCN). CCNs are assigned by CMS and used to identify healthcare facilities participating in the Medicare Part A and Medicaid programs.[480 481] Hospitals primarily have assigned CCNs as entities, but CCNs can also be used to identify specific hospital locations or units, especially when those units operate under the same organizational umbrella but at different sites. CCNs do not change when hospital ownership changes, but hospital mergers, acquisitions, and consolidations can result in CCN changes. We elected not to propose to require that hospitals encode CCNs because CCNs are limited to Medicare- or Medicaid-participating hospitals, while the HPT regulations apply to all hospitals in the United States (with exceptions listed at § 180.30(b)), and, also, the inclusion of ( printed page 54008) CCNs would not align with the TiC provider identifier requirements.
We sought comment on our proposal, as well as any additional, or alternative, taxonomy codes that commenters believe would be necessary or helpful to consider. We also sought comment on other standard identifiers that may be useful in providing needed context for and streamlining the alignment of price transparency data.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: Many commenters supported our proposal to revise § 180.50(b)(2)(i)(A) to require hospitals to report a unique identifier, specifically their Type 2 NPI(s), in their MRFs. Supportive commenters stated that the Type 2 NPI is a valuable identifier for linking to claims data and provider directories, and the inclusion of this information in the MRF would enhance interoperability across datasets, including TiC files. Further, commenters indicated that adding NPI information to the MRFs would allow payers, employers, and researchers to match hospital pricing information more reliably with utilization and outcomes data to identify variation in negotiated rates, analyze payment trends, and develop tools that empower consumers to make informed healthcare purchasing decisions.
Response: We thank commenters for their support of this proposal.
Comment: Several commenters did not support the proposal to include the NPI in the MRF. A few commenters believed it would increase burden to hospitals. One commenter questioned our analysis that only 10 percent of hospitals nationwide would report multiple NPIs. One commenter opposed the inclusion of the NPI as it could add technical and operational complexity when combined with other CMS requirements. This commenter argued that including the NPI with detailed pricing data could expose specific contracting arrangements.
Response: We thank commenters for their comments on this subject. We disagree with commenters that this proposal would add significant burden to hospitals or that it would add complexity to the files. We reiterate that the Type 2 NPI would be listed as a general data element (in CSV) or data attribute (in JSON) in the MRFs that would only need to be included once in the file, not repeated for each item and service. This would be similar to the hospital name, location, and address data that innovators and researchers use to correctly attribute files to locations. Since the requirement is confined to one data element/attribute where the hospital is being asked to list all their NPIs only once, we do not anticipate a significant increase in file size. As we indicated in the proposed rule, our review of the publicly available January 2025 data from PECOS, the online Medicare enrollment system, found that only approximately 10 percent of hospital enrollment applications reported multiple NPIs registered as hospital or hospital units.[482] This is validated by an Office of the Assistant Secretary for Planning and Evaluation (ASPE) report that found that the “vast majority” of hospitals only have one NPI and a “very small” number of hospitals have multiple NPIs.[483] We anticipate that MRF users have the necessary sophistication and tools to navigate multiple NPIs and would appreciate the additional information. Furthermore, because the TiC files already include this information, we do not believe it exposes sensitive contracting information, but, rather, allows alignment and cross-comparison with other data sets.
Comment: A few commenters opposed the proposal because, they stated, insurers are not required to report NPIs in their TiC files, creating an uneven playing field. Another commenter expressed that the requirement to include Type 2 NPIs in the MRF would not make it possible to compare the information with TiC files, stating that TiC requires attributes that specifically define the payer network, plan, and item/service billing class.
Response: While insurers do not have NPIs, we disagree with commenters that insurers are not required to report NPIs in their TiC files, as they do report pricing information that must be associated with a provider's NPI. As we stated in the proposed rule, we proposed the NPI requirement to align more closely with the TiC requirements. Under the TiC final rule (85 FR 72158), and as described in the TiC GitHub schemas for the “In-Network File,” “Out-Of-Network Allowed Amount File,” and the optional “Provider Reference File,” most group health plans and health insurance issuers must post pricing information, and such pricing information must be associated with a provider's NPI to ensure that consumers have reliable data and can make informed healthcare purchasing decisions.[484] We also disagree that the inclusion of the NPI will not improve comparability with the TiC files. While most group health plans and health insurance issuers do include additional data such as billing class, plan market type, and payer network, we believe the inclusion of the NPI will allow for a direct crosswalk between the hospital MRFs and the TiC MRFs. Prior to the inclusion of this data element, innovators and researchers would have to match files by hospital name, EIN, or hospital address. Because hospital name and address are not standardized, there may be variances between hospital and TiC MRFs. EINs may be used across health systems and may not be distinguishable between hospitals within the same health system. The NPI is a standard, unique, 10-digit identifier that hospitals have readily available for inclusion in their MRFs. Furthermore, we believe that some commenters may not have understood our proposal to require hospitals to enter their organizational taxonomy code(s), which begin with `28' or '27.' We are clarifying that our proposal was for hospitals to encode the NPI associated with a taxonomy code reflecting either a hospital or a hospital unit, not that the hospitals should enter the actual taxonomy code.
Comment: Several commenters suggested alternatives to our proposal for consideration. One commenter recommended that CMS require both the NPI and the CCN in the file to strengthen the link between hospital pricing data and other CMS datasets. One commenter recommended that CMS consider replacing the EIN in the file naming convention with the CCN. A few commenters recommended that CMS limit the requirement to a single Type 2 NPI that is associated with a primary taxonomy code starting with `28' but not require Type 2 NPIs beginning with a primary taxonomy code `27,' which represent hospital units, to reduce burden and confusion. One commenter suggested that hospitals include the TIN or EIN that is applicable for each negotiated rate. One commenter suggested that we require all NPIs associated with the hospital, beyond the NPIs associated with the taxonomy codes starting with `28' or `27.'
Response: We thank commenters for their suggestions and alternatives. We decline to include the CCNs in the files as CCNs are limited to Medicare- or ( printed page 54009) Medicaid-participating hospitals, while the HPT regulations apply to all hospitals in the United States (with exceptions listed at § 180.30(b)). Additionally, we note that the inclusion of CCNs would not align with the TiC provider identifier requirements.
As we stated, the vast majority of hospitals have only one Type 2 NPI, and therefore we do not believe it would be overly burdensome to require hospitals to encode all Type 2 NPIs that have a primary taxonomy code starting with `28' or `27.' Furthermore, a review of the publicly available January 2025 data from PECOS, indicates that not all hospitals, such as psychiatric hospitals, have a Type 2 NPI with a primary taxonomy code starting with `28.' [485] Therefore, requiring both strikes a balance ensuring that all hospitals subject to the HPT requirements will have an NPI listed in their MRF.
We do not believe it is necessary for hospitals to include all of their NPIs or include the NPI with each item and service. Innovators and researchers should have the ability to crosswalk to additional NPIs, if necessary, once they have a Type 2 NPI that is associated with primary taxonomy code starting with `28' or `27.' We also decline to include the EIN/TIN for each item and service. We do not believe this is necessary, as it will continue to be required in the file name and as it is applicable to the hospital, it would be applicable to all items and services.
We clarify that this requirement will require hospitals to report their Type 2 NPIs that are associated with a primary taxonomy code starting with `28' or `27.' As we described above, NPIs are unique 10-digit numbers used to identify healthcare providers and organizations, including hospitals. They are associated with taxonomy codes that identify a healthcare provider's specialty and type. Primary taxonomy codes that begin `28' represent hospitals. Primary taxonomy codes that begin with `27' represent hospital units. When applying for a NPI from the NPPES, a healthcare provider must select the Healthcare Provider Taxonomy Code or code description that the health care provider determines most closely describes the healthcare provider's type/classification/specialization, and report that code or code description in the NPI application.[486] Readers can reference the Medicare Provider and Supplier Taxonomy Crosswalk to familiarize themselves with the descriptions for each provider taxonomy code.[487] Hospitals are only required to report their Type 2 NPIs that meet the criteria specified at § 180.50(b)(2)(i)(A), not the taxonomy codes.
Final action: After consideration of public comments, we are finalizing, as proposed, the requirement that, beginning January 1, 2026, hospitals must report, in a newly created general data element in the MRF, any Type 2 NPI(s) that are associated with a primary taxonomy code starting with `28' (indicating hospital) or `27' (indicating hospital unit) and that is active as of the date of the most recent update to the standard charge information. As noted in the “Effective Dates” section below, while this requirement will become effective January 1, 2026, we will delay enforcement to April 1, 2026.
7. Effective Dates
In the CY 2026 OPPS/ASC proposed rule, we proposed several revisions to § 180.50, that would be effective January 1, 2026, including removing the requirement for hospitals to disclose the estimated allowed amount, and, instead, disclose the median, and 10th and 90th percentile, allowed amounts, and the count of allowed amounts; requiring an attestation that the hospital has included all applicable standard charge information in the MRF and that the information encoded is true, accurate, and complete as of the date in the file; requiring the name of the hospital chief executive officer, president, or senior official overseeing the encoding of the data; and a requirement that hospitals must encode their Type 2 (organizational) NPIs. We also proposed at § 180.90, beginning January 1, 2026, to reduce the amount of a CMP by 35 percent, under certain conditions, when a hospital waives its right to an ALJ hearing.
Comment: We received many comments related to the effective date of the proposed requirements at § 180.50, with nearly all such commenters asserting that our proposed timeline was aggressive and that it would be unreasonable to require hospitals to update the data elements and implement the changes by January 1, 2026. Many commenters recommended alternative effective dates, ranging from 60 days to 2 years beyond our proposed effective date of January 1, 2026, that they considered more reasonable. Many commenters indicated that hospitals would need additional time to review new guidance, develop plans for updating the files, coordinate with vendors, encode the data, validate for accuracy, and refresh and post the files. A few commenters expressed concern that a January 1, 2026 implementation date would disrupt hospitals that are then updating their files in anticipation of annual updates. Several commenters expressed concern that hospitals only recently implemented the required CMS standardized templates and other requirements as of January 1, 2025, and would like more time before incorporating additional changes. A few commenters requested that we delay enforcement of the proposed requirements, rather than delay the implementation date.
Response: We believe that hospitals should adopt the new and updated data elements as soon as possible to improve public use of hospital standard charge information. However, in light of the comments, we understand that hospitals may require additional time to develop and encode the additional required information, particularly the new data requirements associated with the median, 10th percentile, and 90th percentile allowed amounts, and the count of allowed amounts. Therefore, while we are finalizing revisions to § 180.50 effective beginning January 1, 2026, we will delay enforcement of those specific new requirements at § 180.50 until April 1, 2026.
Final action: After consideration of public comments, we are finalizing that the effective date of the revisions at § 180.50, including removal of the estimated allowed amount, disclosure of the median, and 10th and 90th percentile, allowed amounts and the count of allowed amounts, the attestation requirement, and the NPIs, will be January 1, 2026. However, we will delay enforcement of these finalized revisions until April 1, 2026. This 3-month enforcement delay will apply solely to enforcement actions based on the new requirements at revised § 180.50 and in no way will affect already-initiated compliance actions or actions for noncompliance with other requirements under part 180 as they are currently being implemented. We believe this 3-month enforcement delay will provide hospitals with sufficient time to update their systems, and review, validate, and post their files.
C. Improved and Enhanced Enforcement
1. Background
In the CY 2020 HPT final rule (84 FR 65524), we established actions that ( printed page 54010) would address hospital noncompliance with the requirements under §§ 180.50 and 180.60, which may include issuing a written warning notice, requesting a corrective action plan (CAP), and imposing CMPs on noncompliant hospitals and publicizing these penalties on a CMS website. In the CY 2022 OPPS/ASC final rule with comment period (86 FR 63941), we increased the amount of CMPs to which a hospital could be subject to a minimum total penalty of $300/day that applies to smaller hospitals with a bed count of 30 or fewer, and a penalty of $10/bed/day for hospitals with a bed count greater than 30, not to exceed a maximum daily dollar amount of $5,500. In the CY 2024 OPPS/ASC final rule with comment period (88 FR 82113), we finalized several enhancements to our enforcement process by updating our methods to assess hospital compliance, requiring hospitals to acknowledge receipt of warning notices, working with health system officials to address noncompliance issues in one or more hospitals that are part of a health system, and publicizing more information about CMS enforcement activities related to individual hospital compliance. We also finalized revisions to § 180.70(a)(2) to add activities that CMS may use to monitor and assess for compliance. Specifically, we revised § 180.70(a)(2)(iii) to indicate that we may conduct an audit and comprehensive compliance review of a hospital's standard charge information posted on a publicly available website. We received comments on our general enforcement activities.
Comment: Several commenters supported our previous regulatory changes to address noncompliance, including the increase in the CMPs for noncompliance, and stated their appreciation of the enforcement efforts taken to address compliance violations to date. Several commenters indicated they desired an enhanced focus on enforcement by CMS to ensure widespread compliance with the HPT rule in an expeditious and efficient manner. Several commenters suggested we increase the CMPs for noncompliant hospitals, while a few commenters opposed the issuance of CMPs or the publicization of compliance actions against hospitals, particularly for those that demonstrate they are making a good faith effort to comply with the requirements immediately following the effective date of new requirements.
Response: We thank commenters for their support of our enforcement activities to date. In a fact sheet, “Hospital Price Transparency Enforcement Updates,” that we posted on April 26, 2023,[488] we provided updates on improvements we implemented regarding our enforcement process, including streamlining the process to no longer issue a warning notice to a hospital that has not posted an MRF or shoppable services list/price estimator tool. This was intended to encourage hospitals to more quickly comply with our HPT requirements, and especially the fundamental HPT requirements to make public an MRF and a consumer-friendly display of shoppable services.
Consistent with Executive Order 14221, we continue to believe that it is critically important that all hospitals comply with applicable HPT regulations. As CMS identifies hospitals without an MRF and/or shoppable services file, we will continue to prioritize those cases for immediate compliance by expediting the compliance process and sending hospitals a CAP request letter.
Comment: Several commenters provided suggestions on how to improve the HPT enforcement process for hospitals and for individuals reporting potential noncompliance with the HPT requirements. A few commenters noted the recent CMPs imposed on smaller hospitals and encouraged us to continue providing hospitals with robust technical assistance and sufficient opportunities to correct deficiencies prior to issuance of a CMP. A few commenters recommended increased clarity in warning notices provided to noncompliant hospitals. A few commenters suggested we formally notify hospitals that are deemed to be in compliance with the Hospital Price Transparency requirements following monitoring and assessment. Other commenters offered other ideas, ( printed page 54011) including: recommending that we establish a hotline for individuals to report potential HPT noncompliance and following up on those reports within 60 days, recommending that we conduct random audits of hospital websites to assess compliance with the requirements, and suggesting that we require hospitals to publicize on their websites the status of their CMS HPT requirements compliance.
Response: We thank commenters for their suggestions on how to improve the HPT requirements and the monitoring and assessment process. We may consider these comments for future rulemaking, guidance, resources, and as part of our HPT monitoring process.
2. Civil Money Penalties: Waiver of Hearing, Automatic Reduction of Penalty Amount
In prior HPT rulemaking,[489] we issued regulations that established processes to enforce the HPT requirements, including issuance of CMPs when a noncompliant hospital fails to respond to our request to submit a CAP or comply with the requirements of the CAP (§ 180.90(a)). The HPT regulations set forth the criteria we use to determine the CMP amount (§ 180.90(c)) and permit hospitals to appeal a CMP imposed by us within 30 days of issuance of the notice of imposition of a CMP (§§ 180.100 and 180.110). As of September 2025, we have issued CMP notices to 27 hospitals, 22 of which have exercised their right to appeal the CMP to an ALJ.[490] Hospitals may elect to appeal for many reasons, including disagreeing with our assessment of the law or facts underlying our determination, seeking to protect their reputation and/or avoid other civil or state regulatory actions, or other reasons.
We are aware that in some other CMS enforcement programs, entities subject to CMPs receive a 35 percent discount to the CMP amount if they waive their appeal rights.[491] For example, in the FY 2024 Skilled Nursing Facility Prospective Payment System final rule (88 FR 53200, 53326), we discussed our experience over the years with the CMP reduction pertaining to LTC facilities. We noted there how, between CYs 2016 and 2022 (but for CY 2017 that was not referenced), around 80 percent of LTC facilities submitted waivers, with the figure rising to 91 percent in CY 2021 but retreating to 81 percent in CY 2022, while also a considerable percentage of the remaining facilities did not submit a waiver but also not did not contest the penalty and its basis. Throughout the period, between 2 to 6 percent of facilities availed themselves of the full hearing process.
Given respondents' widespread invocation of the LTC facility enforcement appeal waiver provision, we considered whether offering hospitals the opportunity to receive a reduced penalty—in some circumstances, and in exchange for their acknowledging their HPT noncompliance—could expedite timely payment of CMPs. Among our considerations, we believed that hospitals that elect this waiver opportunity pursuant to such a proposal would be demonstrating their acceptance of responsibility for HPT noncompliance, and consequently also their commitment to timely achieving future compliance, which would be key to helping us achieve our overarching HPT goal of ensuring this information, in compliant form, is accessible to healthcare consumers.
We therefore proposed at new § 180.90(c)(4), and subject to the exceptions discussed below, that the amount of a CMP would be reduced by 35 percent should a hospital submit to CMS a written notice waiving its right to a hearing under § 180.100 within 30 calendar days of the date of the notice of imposition of the CMP. We also proposed that if a hospital waives its right to appeal a CMP and receives a 35 percent reduction in accordance with § 180.90(c)(4), the hospital: (1) would not be eligible to receive a 35 percent reduction under § 180.90(c)(4) on any subsequent CMPs issued under § 180.90(f) that result from the same instance(s) of noncompliance (that is, continuing violations); and (2) would waive its right to appeal the subsequent CMPs for any such continuing violations. As discussed above, in waiving its right to appeal and receiving a 35 percent reduction with respect to the initial CMP, we believe a hospital would be demonstrating acceptance of responsibility for HPT noncompliance and a commitment to achieving future compliance without further intervention; further appeal rights or CMP reductions in the face of continuing violations would be not consistent with that underlying rationale for the CMP discount.
At § 180.90(c)(4), we proposed that, in certain situations, CMS would decline to make available to hospitals the opportunity to have a CMP amount reduced. First, we proposed that, should a hospital not affirmatively waive its right to a hearing in accordance with the procedures specified at proposed § 180.90(c)(4), the CMP amount would not be reduced. We explained that the proposed timeframe (within 30 calendar days of the date of notice of imposition of the CMP) would provide a hospital ample opportunity to elect whether to exercise its option to waive a hearing. Second, we proposed that, should CMS impose upon a hospital a CMP for HPT noncompliance going to the core of the HPT requirements—specified as failing to make public either: (1) an MRF as required in § 180.40(a), or (2) any shoppable services in a consumer-friendly format (either in the form of a shoppable services file or an internet price estimator tool) as required in § 180.40(b)—the hospital would be ineligible to avail itself of such an opportunity. As reflected in Table 144, through the compliance review process, CMS has encountered instances where hospitals have not made public an MRF and/or a consumer-friendly list of shoppable services (either a shoppable services file or internet price estimator tool). We stated in the proposed rule that a hospital that fails to abide by such core HPT requirements—entirely depriving the public access to these important tools—would forfeit the opportunity to avail itself of a penalty reduction and would be required to pay in full a CMP. For example, should CMS impose upon a hospital a CMP for failing to make public an MRF as required by § 180.40(a), even if it did have a shoppable services file or internet price estimator tool as required by § 180.40(b), such hospital would not be eligible for a reduction to its CMP by waiving its appeal rights (and the same would pertain were a hospital to have an MRF as required by § 180.40(a), but not a shoppable services file or internet price estimator tool as required by § 180.40(b)). We explained that this exception would be appropriate because we finalized, and codified at 42 CFR part 180, the requirement that hospitals make public their standard charges in two ways (as an MRF and in a consumer-friendly format), effective beginning January 1, 2021; in other words, hospitals have been subject to this requirement for more than 4 years and failing even to try to comply with the requirement renders any effort to ( printed page 54012) accept responsibility or commit to future compliance less credible than a hospital whose efforts to comply have simply fallen short.
We noted that our proposal would not preclude a hospital, so long as it did not seek a waiver, from requesting a hearing, nor would waiving the right to a hearing remove from the hospital's record the fact of its HPT noncompliance. Rather we proposed that should a hospital choose to waive its right to a hearing, it would accept CMS' determination that it was noncompliant. Significantly, whether or not a hospital would elect to waive the right to a hearing, it would still be required to achieve compliance to avoid the potential imposition of additional CMPs pursuant to § 180.90(f). We also expected that this proposal would benefit both CMS and the hospital by reducing or eliminating the time, resources, expenses, and other potential burden otherwise attributable to prosecuting or defending the administrative appeals processes.
Finally, we also proposed to make conforming revisions to § 180.90(d)(1) and to add a new § 180.90(d)(2) to take into account the proposed provisions at § 180.90(c)(4), which would allow for a reduction to the CMP amount were certain criteria to be met, as discussed above. We proposed to redesignate current § 180.90(d)(2) and (3) as § 180.90(d)(3) and (4), respectively.
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: A few commenters supported our proposal at new § 180.90(c)(4), that the amount of a CMP be reduced by 35 percent should a hospital submit to CMS a written notice requesting to waive its right to a hearing under § 180.100 within 30 calendar days of the date of the notice of imposition of the CMP. One commenter noted that this proposal strikes a fair balance between accountability and administrative efficiency, encouraging timely resolution of violations while reinforcing the importance of transparency. The commenter indicated their support of the proposal as a constructive incentive to help ensure that patients and stakeholders have access to accurate and actionable pricing information.
Response: We appreciate the commenters' support for our proposal.
Comment: A few commenters indicated that CMS' proposal to reduce penalties by 35 percent puts undue pressure on hospitals to waive their rights to appeal and due process. One commenter requested that CMS consider hospitals' good faith efforts to implement corrective actions addressing ongoing noncompliance to avoid further imposition of CMPs and sought clarification on their understanding that the regulations currently allow for reduced penalties. One commenter stated that the current regulations at § 180.90 already permit CMS to reduce CMPs.
Response: We disagree with the commenters that the 35 percent reduction in CMPs described in our proposed rule puts undue pressure on hospitals to waive their rights. By accepting the 35 percent reduction in their CMP amount, a hospital acknowledges and accepts CMS' noncompliance determination and demonstrates its commitment to correct its behavior going forward. Commenters point to no case law finding that crediting acceptance of responsibility for legal violations violates due process, and if a hospital disagrees with the legal or factual basis of our determination, it may exercise its right to appeal as outlined in § 180.100. We thank the commenter who indicated that § 180.90 already permits CMS to reduce CMPs, but reinforce that this proposal would benefit both CMS and the hospital by reducing or eliminating the time, resources, expenses, and other potential burden otherwise attributable the administrative appeals processes.
Comment: One commenter requested we withdraw the 35 percent reduction in CMPs described in our proposed rule because it will incentivize noncompliant hospitals to pay reduced CMPs and remain noncompliant as a course of business, rather than come into compliance with the HPT rules. Another commenter called for increased penalties and more enforcement.
Response: We do not agree with commenters that the 35 percent reduction in CMPs will incentivize hospitals to remain noncompliant with our requirements. Hospitals can continue to receive additional CMPs for the same violations if they remain out of compliance, but they will not be eligible to receive a 35 percent reduction in CMPs for the same instance(s) (that is, continuing violations) of noncompliance. Therefore, hospitals cannot remain noncompliant as a course of business and continue to receive a 35 percent CMP reduction. We acknowledge that there is a risk a hospital might take advantage of the 35 percent reduction to the CMP to avoid affirmative efforts to comply with the HPT requirements until CMS conducts an audit and identifies the violations, however, we believe this risk to be minimal and mitigated by the fact that hospitals are not eligible for the 35 percent reduction in CMPs if they receive a subsequent CMP for the same violation. We also note that those hospitals found to not have an MRF or consumer-friendly display of shoppable services would not be eligible for this CMP reduction in the first instance. We remain committed to ensuring that all hospitals comply with the HPT rules, and will continue to assess CMPs for violations, as appropriate. We believe that structuring the 35 percent CMP reduction in this way maximizes the likelihood that the reduction will be used mainly by hospitals that are accepting responsibility for past failures and are committed to coming into compliance. Should that belief prove false, we may revisit the 35 percent CMP reduction. For now, however, we decline to withdraw the proposal.
Comment: One commenter called for stronger enforcement actions and increased penalties for noncompliant hospitals.
Response: We thank the commenters for their suggestion. Currently, we do not see the need to increase penalties as we are seeing increasing compliance rates among hospitals subject to the HPT regulations. As we note above, CMS remains committed to ensuring that all hospitals comply with the HPT rules and will continue to assess CMPs for violations, as appropriate.
Comment: Several commenters noted that the current requirements have achieved high compliance rates through increased enforcement efforts and collaboration. A few commenters noted that this waiver is unnecessary due to the existing high levels of compliance.
Response: We thank commenters and agree with their assessment that the HPT program has achieved increasing levels of compliance since enforcement began in January 1, 2021. However, we do not agree with the commenters' assessment that this proposal is unnecessary. We believe that both CMS and the hospital would benefit from this proposal by reducing or eliminating the time, resource, and expense burden that may be incurred with a protracted appeals process. Further, we believe this will continue to increase hospital compliance as we believe, by virtue of its terms that we describe above, that only hospitals committed to achieving timely compliance will avail themselves of it.
Final Action: After consideration of the public comments received, we are finalizing our proposal at new § 180.90(c)(4) with clarifying edits, with an effective date of January 1, 2026. Specifically, we are adding the word ( printed page 54013) “subsequent” before the word “civil monetary penalties” in the second sentence so that this sentence now reads, “A hospital that receives a 35 percent reduction in a civil monetary penalty under this paragraph is not eligible to receive a 35 percent reduction for any subsequent civil monetary penalties imposed pursuant to continuing violations according to § 180.90(f) and also waives its right to appeal under § 180.100 any subsequent civil monetary penalties imposed for such continuing violations.” In addition, we are revising the introductory language of the third sentence to clarify that the reduction in “the amount of a civil monetary penalty” referred to this sentence means the “35 percent” reduction in accordance with § 180.90(c)(4).
In addition, we are finalizing, as proposed, conforming revisions to § 180.90(d)(1) and new § 180.90(d)(2) to take into account the final provisions at § 180.90(c)(4). Finally, as proposed, we are redesignating current § 180.90(d)(2) and (3) as § 180.90(d)(3) and (4), respectively.
XX. Market-Based Medicare Severity-Diagnosis Related Groups (MS-DRG) Relative Weight Data Collection and Change in Methodology for Calculating MS-DRG Relative Weights Under the Inpatient Prospective Payment System
A. Overview
In the FY 2021 IPPS/LTCH PPS final rule (85 FR 58873 through 58892), we discussed the need for Medicare to reduce its reliance on the hospital chargemaster and develop market-based approaches to payment under the Medicare FFS system. We continue to believe this is the case.
In that rulemaking (85 FR 58891), we adopted a policy that required hospitals to report on the Medicare cost report the median payer-specific negotiated charge that the hospital had negotiated with all of its Medicare Advantage Organizations (MAOs), by MS-DRG, effective for cost reporting periods ending on or after January 1, 2021. In the same final rule, we adopted the use of the median payer-specific negotiated charge by MS-DRG for MAOs in the market-based MS-DRG relative weight methodology finalized for relative weight calculations beginning in FY 2024. In the FY 2022 IPPS/LTCH PPS final rule (86 FR 45319), we repealed both the collection of market-based rate information on the Medicare cost report and the market-based MS-DRG relative weight methodology and stated that we would continue to evaluate and consider the usefulness and appropriateness of market-based data for ratesetting purposes. After further consideration, as discussed in section XX.C. of this final rule with comment period, we once again proposed, with modifications (as discussed in section XX.C.2. of this final rule with comment period), to require that hospitals report on the Medicare cost report, beginning January 1, 2026, the median [492] of the payer-specific negotiated charges (hereinafter referred to as the “median payer-specific negotiated charge”) that the hospital has negotiated with all of its MAOs, by MS-DRG, for use in a market-based MS-DRG relative weight methodology, effective for the relative weights calculated for FY 2029.
In the FY 2021 IPPS/LTCH PPS final rule (85 FR 58885), we discussed our authority for adopting a market-based MS-DRG relative weight data collection and MS-DRG relative weight methodology. Sections 1815(a) and 1833(e) of the Act provide authority to collect data for purposes of determining the amount of payments due to a provider under the Medicare program. Specifically, sections 1815(a) and 1833(e) of the Act state that no Medicare payments will be made to a provider unless it has furnished information requested by the Secretary to determine payment amounts due under the Medicare program and pertain to CMS' authority to collect information on the Medicare cost report. We also discussed CMS' authority under section 1886(d)(4) of the Act to assign and update MS-DRG weighting factors to reflect relative resource use. In particular, section 1886(d)(4)(B) of the Act requires that for each diagnosis-related group the Secretary shall assign an appropriate weighting factor which reflects the relative hospital resources used with respect to discharges classified within that group compared to discharges classified within other groups, and section 1886(d)(4)(C)(i) of the Act requires that the weighting factors be adjusted at least annually to reflect changes in treatment patterns, technology, and other factors which may change the relative use of hospital resources.
In the CY 2026 OPPS/ASC proposed rule, we proposed for cost reporting periods ending on or after January 1, 2026, to collect on the Medicare cost report the median payer-specific negotiated charge that the hospital has negotiated with all of its MAOs, by MS-DRG. We proposed to utilize this data within a proposed methodology for calculating the IPPS MS-DRG relative weights to reflect relative market-based pricing, effective in FY 2029. This proposal reflected certain modifications to the policy as finalized in the FY 2021 IPPS/LTCH PPS final rule (85 FR 58873 through 58892), as discussed further in section XX.C. of this final rule with comment period. As stated previously, and in the CY 2026 OPPS/ASC proposed rule, we continue to believe there is a need for Medicare to reduce its reliance on the hospital chargemaster and develop market-based approaches to payment under the Medicare FFS system. We discuss in further detail in this section our evaluation and reconsideration of the usefulness and appropriateness of market-based data for ratesetting purposes since the FY 2022 IPPS/LTCH PPS final rule. As discussed in greater detail in section XX.C.2. of this final rule with comment period, the CY 2026 OPPS/ASC proposed rule also provided instruction on how hospitals would calculate the median of the payer-specific negotiated charges for an MS-DRG using data from the machine-readable file (MRF) that hospitals are required to disclose under the hospital price transparency regulations at 45 CFR part 180. The CY 2026 OPPS/ASC proposed rule also addressed circumstances when hospitals use something other than MS-DRGs as a basis for reporting under those hospital price transparency requirements.
As described further in section XX.C.2. of this final rule with comment period, we specifically proposed that for the purposes of reporting the data on the cost report, hospitals would report the median of the payer-specific negotiated charges for an MS-DRG that the hospital has disclosed for all of its MAOs on the most recent version of the MRF that the hospital is required to disclose under 45 CFR 180.40(a). If the hospital disclosed the payer-specific negotiated charge for an MS-DRG as a dollar amount, the hospital would use the dollar amount disclosed on its MRF under 45 CFR 180.50(b)(2)(ii)(C) in determining the median of the payer-specific negotiated charges to be reported on its Medicare cost report, as discussed further in section XX.C.2 of this final rule with comment period. If the hospital disclosed the payer-specific negotiated charge as a percentage or algorithm on the MRF, we proposed that the hospital would instead use the proposed “median allowed amount” (as discussed in section XIX. of this final rule with comment period) to calculate the ( printed page 54014) median of the payer-specific negotiated charges.[493] The hospital would then report the median payer-specific negotiated charge on its Medicare cost report, as also discussed further in section XX.C.2. of this final rule with comment period. In the CY 2026 OPPS/ASC proposed rule, we stated that we believed this approach of utilizing data required for disclosure on the MRF under 45 CFR 180.50(b)(2)(ii)(C) in determining the median of the payer-specific negotiated charges would help streamline requirements for hospitals and result in less administrative burden overall because hospitals would already be required to calculate and disclose these data in compliance with the hospital price transparency requirements. For additional details on hospital price transparency requirements, including MRF requirements and the modifications to the hospital price transparency requirements, we refer readers to section XIX. of this final rule with comment period and https://www.cms.gov/priorities/key-initiatives/hospital-price-transparency/hospitals.
As described in greater detail in section XX.C. of this final rule with comment period, we proposed that the median payer-specific negotiated charges as reported on the Medicare cost report would be used in a proposed market-based methodology to calculate IPPS MS-DRG relative weights beginning in FY 2029 to reflect the relative hospital resources used to provide inpatient services to patients. The use of the median payer-specific negotiated charges would replace the current use of gross charges that are reflected on a hospital's chargemaster and cost information from Medicare cost reports for the development of the IPPS MS-DRG relative weights.
B. Factors Considered
As discussed in the FY 2021 IPPS/LTCH PPS final rule (85 FR 58873 through 58892), to reduce the Medicare program's reliance on the hospital chargemaster and to support the development of a market-based approach to payment under the Medicare FFS system, we finalized our proposal to require that hospitals report certain market-based payment rate information on their Medicare cost report for cost reporting periods ending on or after January 1, 2021. In that same rulemaking, we also adopted a market-based MS-DRG relative weight methodology using that information. In the FY 2022 IPPS/LTCH PPS final rule (86 FR 45319), we repealed both the collection of market-based rate information on the Medicare cost report and the market-based MS-DRG relative weight methodology and stated that we would continue to evaluate and consider the usefulness and appropriateness of market-based data for ratesetting purposes.
As noted in the FY 2022 IPPS/LTCH PPS rulemaking, we have continued to consider the use of market-based rate information for purposes of the IPPS relative weight methodology, including for the reasons discussed in the FY 2021 IPPS/LTCH PPS final rule (85 FR 58874 through 58875) regarding reducing the Medicare program's reliance on the hospital chargemaster and supporting the development of a market-based approach to payment under the Medicare FFS system, as well as additional factors since the repeal of the prior policies.
For example, in the FY 2021 IPPS/LTCH PPS proposed rule we described research that chargemasters are usually highly inflated and that these inflated charges have been used to secure higher payments from Medicare and private payers (85 FR 32790). We indicated that some hospitals' charges do not reflect market rates. Hospital bills that are generated off these chargemaster rates can be inherently unreasonable when judged against prevailing market rates. We stated that recognizing that chargemaster (gross) rates rarely reflect true market costs, we believed that by reducing our reliance on the hospital chargemaster we could adjust Medicare payment rates so that they reflect the relative market value for inpatient items and services. As part of our efforts since the FY 2022 repeal, we have examined more recent research on hospital chargemasters, which is generally consistent with the discussion in the FY 2021 rulemaking regarding whether hospital chargemasters reflect true market costs. Recent research by Linde and Egede [494] concluded that higher chargemaster markups are associated with higher hospital profitability. They delineated four potential causal pathways that may connect chargemaster markups to hospital profitability. First, chargemaster prices are commonly billed to uninsured patients and therefore may increase profits via higher payments (or payment settlements) with uninsured patients. Second, higher chargemasters may yield higher payments from insured individuals that seek care out-of-network, or who receive care at in-network facilities but are cared for by out-of-network providers. Third, chargemaster prices do in many cases serve as reference prices for the contractual payments between private insurers and hospitals. As such, higher chargemaster prices may yield increased profits by increasing payments from private payors. Fourth, higher chargemaster prices may allow hospitals to increase the cost-saving value of liabilities that end up being written off as bad debt, and therefore increased hospital profits.
We have also continued to consider the available research comparing Medicare, MAO, and commercial payment rates since the repeal. As discussed in the FY 2021 IPPS/LTCH PPS final rule (85 FR 58874 through 58877), we reviewed available literature to compare Medicare FFS and MAO payment rates and how those MAO rates may reflect the relative hospital resources used within an MS-DRG differently than our current cost-based methodology.
As discussed in the FY 2021 rulemaking, Berenson et al.[495] surveyed senior hospital and health plan executives and found that MA plans nominally pay only 100 to 105 percent of traditional Medicare rates and, in real economic terms, possibly less. Respondents broadly identified three primary reasons for near payment equivalence:
- Statutory and regulatory provisions that limit out-of-network payments to traditional Medicare rates,
- De facto budget constraints that MA plans face because of the need to compete with traditional Medicare and other MA plans, and
- A market equilibrium that permits relatively lower MA rates as long as commercial rates remain well above the traditional Medicare rates.
As also discussed in the FY 2021 rulemaking, Baker et al.[496] used data from Medicare and the Health Care Cost Institute (HCCI) to identify the prices paid for hospital services by FFS Medicare, MA plans, and commercial insurers in 2009 and 2012. They ( printed page 54015) calculated the average price per admission, and its trend over time, in each of the three types of insurance for fixed baskets of hospital admissions across metropolitan areas. After accounting for differences in hospital networks, geographic areas, and case-mix between MA and FFS Medicare, they found that MA plans paid 5.6 percent less for hospital services compared to FFS Medicare. For the time period studied, the authors suggest that at least one channel through which MA plans paid lower prices was by obtaining greater discounts on types of FFS Medicare admissions that were known to have very short lengths-of-stay. They also found that the rates paid by commercial plans were much higher than those of either MA or FFS Medicare, and that this differential was growing. At least some of this difference they indicated came from the much higher prices that commercial plans paid for certain service lines.
Maeda and Nelson [497] also analyzed data from the HCCI in their research. They compared the hospital prices paid by MA organizations and commercial plans with Medicare FFS prices using 2013 claims from the HCCI. The HCCI claims were used to calculate hospital prices for private insurers, and Medicare's payment rules were used to estimate Medicare FFS prices. The authors focused on stays at acute care hospitals in metropolitan statistical areas (MSAs). They found MA prices to be roughly equal to Medicare FFS prices, on average, but commercial prices were 89 percent higher than FFS prices. In addition, commercial prices varied greatly across and within MSAs, but MA prices varied much less. Although they noted that they used slightly different methods to calculate Medicare FFS prices, the authors considered their results generally consistent with the Baker et al. study findings in that hospital payments by MA plans were much more similar to Medicare FFS levels than they were to commercial payment levels.
In their study, Maeda and Nelson also examined whether the ratio of MA prices to FFS prices varied across DRGs to assess whether there were certain DRGs for which MA plans tended to pay more or less than FFS. They ranked the ratio of MA prices to FFS prices and adjusted for outlier payments. The authors found that “there were some DRGs where the average MA price was much higher than FFS and there were some DRGs where the average MA price was a bit lower than FFS.” For example, for the time period in question, on average, MA plans paid 129 percent more than FFS for rehabilitation stays (DRG 945), 33 percent more for depressive neuroses (DRG 881), and 27 percent more for stays related to psychoses (DRG 885). But MA plans paid an average of 9 percent less than FFS for stays related to pathological fractures (DRG 542) and wound debridement and skin graft (DRG 464) (see Online Appendix Table 5 from their study). The authors state these results suggest that there may be certain services where MA plans pay more than FFS possibly because the FFS rates for those services are too low, but that there may be other services where MA plans pay less than FFS possibly because the FFS rates for those DRGs are too high (Maeda, Nelson, 2018 p. 5).
In addition to this research discussed in the FY 2021 rulemaking, we have also considered more recent research comparing Medicare FFS rates, MAO rates, and rates of other commercial payers, some of which used data that was made public under the provisions of the Hospital Price Transparency regulations. Meiselbach et al.[498] used 2022 price information disclosed by hospitals to examine the ratio of commercial-to-MA prices negotiated by the same insurer and found that median prices were two to three times higher for commercial plans than MA plans in the same hospital for the same service. They attributed the relatively lower MA prices to the same reasons outlined by Berenson et al. Based on price transparency data from 22 dyads of large hospitals and insurers, Randall and Duffy [499] found that, for a market basket of inpatient services, prices for health insurance exchange plans were 143.3 percent of those for MA organizations and about 89 percent of those for commercial group insurance plans.
This more recent research does not directly address the relationship between payer-specific charges negotiated between hospitals and MAOs and Medicare IPPS payment rates, but it is generally consistent in other respects to the earlier research we cited in the FY 2021 IPPS/LTCH PPS final rule (85 FR 58876 through 58877) indicating that hospital payments by MAOs are much more similar to Medicare FFS levels than they are to commercial payment levels. We continue to believe that payer-specific charges negotiated between hospitals and MAOs and Medicare IPPS payment rates are generally well-correlated. In the FY 2022 IPPS/LTCH PPS final rule we indicated that we agreed with commenters that we needed to further consider the questions raised by commenters regarding the ability of the payer-specific charges negotiated between hospitals and MAOs to represent market-based pricing given the relationship between Medicare FFS and MAO rates. After considering this issue further since the FY 2022 rulemaking, we do not believe that the current general correlation between the two precludes the ability of this data over time to reflect market-based pricing for at least some services. As discussed in the FY 2021 IPPS/LTCH PPS final rule (85 FR 58883), MA rates to MA contracted inpatient hospitals are not required to be the same as (or based on) Medicare FFS rates; the Medicare statute only requires MAOs to pay FFS rates to a health care provider for services furnished to an MA enrollee when the MAO does not have a contract with the health care provider. We believe that to the extent hospitals and MAOs over time negotiate different relative relationships for some services than the relationships that exist under the IPPS, this information adds value to the IPPS and should be incorporated. For example, in the FY 2021 IPPS final rule we stated that we believe the rates that hospitals negotiate with MAOs capture the relative resource use to provide services to patients in order to maximize profits (or, in the case of not-for-profit hospitals, net income), subject to market constraints and conditions (supply and demand, community benefit requirements, etc.). Therefore, we stated we believed that payer-specific negotiated charges provide greater insight into the resource use of a hospital (85 FR 58886). After further consideration, recognizing that there is currently general correlation between the Medicare FFS and MAO rates, we believe that the ability of the payer specific negotiated charges to provide these insights over time still holds true.
Another factor that we considered in our current proposal is the experience hospitals have gained through the process of disclosing the payer-specific negotiated charge information for the purpose of the hospital price transparency requirements. In calculating the median payer-specific ( printed page 54016) negotiated charges to be reported on the Medicare cost report for use in the proposed market-based relative weight methodology, hospitals would use the same payer-specific negotiated charge information that hospitals are required to disclose under the requirements (45 CFR 180.40(a)) that we initially finalized in the Hospital Price Transparency final rule (84 FR 65524), beginning January 1, 2021. Over the last four years, hospitals have become increasingly familiar with the hospital price transparency requirements and procedures necessary to disclose payer-specific negotiated charges. CMS has also taken enforcement actions against hospitals that have failed to comply with the price transparency requirements.[500] We believe that this increased familiarity, experience, and enforcement has improved the data integrity of this information, simplified the initial administrative burden in disclosing this data, and means that this data is now more robust for Medicare ratesetting purposes than it was when we repealed the prior market-based policies.
An additional factor we considered was the ending of the COVID-19 public health emergency (PHE). To the extent commenters previously raised concerns regarding the need for additional flexibilities as hospitals continue to recover from the COVID-19 PHE, as summarized in the FY 2022 IPPS/LTCH final rule (86 FR 45319), the COVID-19 PHE expired on May 11, 2023.
Considering these factors, we proposed to require that hospitals report on the Medicare cost report the median payer-specific negotiated charge that the hospital has negotiated with all of its MAO payers, by MS-DRG, effective for cost reporting periods ending on or after January 1, 2026, and to use this data in a new market-based MS-DRG relative weight methodology, beginning in FY 2029.
We stated in the CY 2026 OPPS/ASC proposed rule that, if the policy were finalized, we intend to make our analysis of this market-based data available for public review prior to the proposed effective date of this market-based relative weight methodology in FY 2029, including the estimated potential payment impact on the MS-DRG relative weights. As under the current methodology, the impact of any MS-DRG relative weight changes on an individual hospital would depend on the mix of services provided by that particular hospital.
C. Market-Based MS-DRG Relative Weight Estimation
1. Overview
Section 1886(d)(4)(A) of the Act states that the Secretary shall establish a classification of inpatient hospital discharges by diagnosis-related groups and a methodology for classifying specific hospital discharges within these groups. Section 1886(d)(4)(B) of the Act states that for each such diagnosis-related group the Secretary shall assign an appropriate weighting factor which reflects the relative hospital resources used with respect to discharges classified within that group compared to discharges classified within other groups. For the reasons previously discussed, we stated in the CY 2026 OPPS/ASC proposed rule that we believe the use of median payer-specific negotiated charge data for a hospital's MAOs, to be collected on the Medicare cost report, may support the development of an appropriate market-based approach to payment under the Medicare FFS system by incorporating such data into the estimation of the relative hospital resources used with respect to discharges classified within a single MS-DRG compared to discharges classified within other MS-DRGs, as required by statute.
As discussed, since the FY 2022 IPPS/LTCH PPS final rule, we have continued to evaluate and consider the usefulness and appropriateness of market-based data for ratesetting purposes. Based on this review, in the CY 2026 OPPS/ASC proposed rule we stated that we believed it would be appropriate to propose the use of hospitals' median payer-specific negotiated charges for MAOs, to be collected on the Medicare cost report as described previously, within a proposed new methodology for calculating the MS-DRG relative weights to reflect a more market-based approach, using our authority under sections 1886(d)(4)(A), 1886(d)(4)(B), and 1886(d)(4)(C) of the Act.
2. Market-Based Data Collection
In order to support the development of a relative market-based payment methodology under the IPPS, we proposed to collect market-based payment rate data on the Medicare cost report for cost reporting periods ending on or after January 1, 2026. This proposed data collection was similar to the market-based data collection as finalized in the FY 2021 IPPS/LTCH PPS final rule (85 FR 558873 through 58892), with additional modifications to use the payer-specific negotiated charges from the hospital's most recent MRF published prior to the submission of its cost report, to reflect proposed revisions to the hospital price transparency regulations at 45 CFR 180, and to better address when the payer-specific negotiated charge is based on a percentage or algorithm, in response to previous concerns (85 FR 58884).
Specifically, we proposed that hospitals would report on their cost report the median of the payer-specific negotiated charges that the hospital negotiated with its MAOs, by MS-DRG, beginning with cost reporting periods ending on or after January 1, 2026. Sections 1815(a) and 1833(e) of the Act provide that no Medicare payments will be made to a provider unless it has furnished the information, as may be requested by the Secretary, to determine the amount of payments due to the provider under the Medicare program. We require that providers follow reasonable cost principles under section 1861(v)(1)(A) of the Act when completing the Medicare cost report. Under the regulations at 42 CFR 413.20 and 413.24, we define adequate cost data and require cost reports from providers on an annual basis. As previously discussed, the collection of this market-based data on the Medicare cost report would allow for the adoption of a market-based strategy to determine the appropriate weighting factors to reflect the relative hospital resources used with respect to hospital discharges, as required under sections 1886(d)(4)(B) and 1886(d)(4)(C) of the Act.
As discussed in the FY 2021 IPPS/LTCH PPS final rule (85 FR 58877), Medicare certified providers, such as Medicare certified hospitals, are required to submit an annual cost report to their Medicare Administrative Contractor (MAC). The Medicare cost report contains provider information such as facility characteristics, cost and charges by cost center, in total and for Medicare, Medicare settlement data, and financial statement data. The cost report must be submitted in a standard (ASCII) electronic cost report (ECR) format. CMS maintains the cost report data in the Healthcare Cost Report Information System (HCRIS) data set. The HCRIS data supports our payment policymaking, congressional studies, legislative health care reimbursement initiatives, Medicare profit margin analysis, and relative weight updates. As such, data from hospital cost reports beginning on or after May 1, 2010 is reflected on the HCRIS dataset, and available for public access and use.
In the CY 2026 OPPS/ASC proposed rule, we stated that if we were to finalize the proposal to collect the proposed market-based information (specifically, the median payer-specific ( printed page 54017) negotiated charges negotiated between a hospital and all its MAOs, by MS-DRG) on the cost report, the data would become publicly accessible on the HCRIS dataset in a de-identified manner and would be usable for analysis by third parties. The data would, by definition, be de-identified since we proposed that the hospital calculate the median rate (that is, the specific rate that is negotiated between a hospital and a specific MAO for an MS-DRG would not be reported and need to be de-identified). For more information or to obtain HCRIS data we refer readers to https://www.cms.gov/data-research/statistics-trends-and-reports/cost-reports/cost-reports-fiscal-year.
We proposed that the hospital would determine the weighted median of the payer-specific negotiated charges that the hospital negotiated with its MAOs, by MS-DRG, as follows:
Step 1. Using the hospital's most recent MRF as of the hospital's cost report filing date identify the following information: (a) each MAO payer-specific negotiated charge under 45 CFR 180.50(b)(2)(ii) that the hospital has negotiated with its MAOs for inpatient items or services (for example, discharges), and (b) the code under 45 CFR 180.50(b)(2)(iv)(A) for each payer-specific negotiated charge. If the payer-specific negotiated charge is based on a percentage or algorithm, the hospital would identify and substitute the dollar amount in the MRF required under 45 CFR 180.50(b)(2)(ii)(C) for the percentage or algorithm. Exclude any payer-specific negotiated charges that represent capitated payment.
Step 2. For the cost reporting period, sum the number of inpatient discharges for each MAO for each MS-DRG. Exclude inpatient discharges where payment was made on a capitated basis.
Step 3. For each MS-DRG, list each MAO payer-specific negotiated charge (from Step 1) the number of times as there were inpatient discharges that occurred during the cost reporting period for that MAO (from Step 2).
Step 4. For each MS-DRG, compute the median [501] of the MAO payer-specific negotiated charge in the list from Step 3. To compute the median, using the list in Step 3, order the list in Step 3 from the lowest MAO payer-specific negotiated charge to the highest; if the list contains an odd number of charges the median is the middle value in the list, or if the list contains an even number of charges the median is the mean of the two middle values. For each MS-DRG, this median is the weighted median MAO payer-specific negotiated charge for that MS-DRG.
As we discussed in the FY 2021 rulemaking, we recognize that the payer-specific negotiated charges negotiated between MAOs and hospitals may in some cases be based on a system other than MS-DRGs. If there are codes identified in (b) of Step 1 that are not MS-DRG codes, or discharges in Step 2 that are not classified to MS-DRGs, the hospital would crosswalk those codes or classify those discharges to MS-DRGs. Hospitals can utilize the CMS GROUPER and associated definitions manual for this purpose. Hospitals have access to the publicly available version of the CMS Grouper used to group ICD-10 diagnosis and procedure codes to MS-DRGs.[502] This software and associated definitions manual can be used to crosswalk the code(s) in the MRF or classify the discharge to an MS-DRG code.
We note that, in section XIX. of the CY 2026 OPPS/ASC proposed rule, we proposed to amend the regulations at 45 CFR 180 as they relate to a standard charge that is based on a percentage or algorithm. Specifically, we proposed in section XIX. of the CY 2026 OPPS/ASC proposed rule, that, beginning January 1, 2026, hospitals would be required to report a new data element, the “median allowed amount,” instead of the “estimated allowed amount” reported at present, and that the median allowed amount would be defined as the median of the total allowed amount that the hospital has historically received from a third-party payer (including MAOs) for an item or service. We also proposed in section XIX. of the CY 2026 OPPS/ASC proposed rule that if a payer-specific negotiated charge is based on a percentage or algorithm, the hospital's MRF would have to describe the percentage or algorithm that determines the dollar amount for the item or service and the hospital would have to calculate and encode the median allowed amount in dollars for that item or service. We proposed in section XIX. of the CY 2026 OPPS/ASC proposed rule that, to calculate the `median allowed amount,' hospitals would be required to use electronic remittance advice transaction data, and that the dollar amount would reflect no longer than a 12-month time period prior to the posting of the most recent MRF. Accordingly, we proposed that the dollar amount in the MRF required under 45 CFR 180.50(b)(2)(ii)(C) for the percentage or algorithm in Step 1 would be the “median allowed amount” if the proposed amendment was finalized. We also proposed that if CMS did not finalize changes to 45 CFR 180.50(b)(2)(ii)(C), the dollar amount would be the “estimated allowed amount” under the current regulations. We note that in section XIX. of this final rule with comment period, CMS is finalizing, with modification, that beginning January 1, 2026, if a hospital's payer specific negotiated amount is based on an algorithm or percentage, the hospital would be required to describe the percentage or algorithm and report a new data element, the “median allowed amount”, instead of the “estimated allowed amount”, and that the “median allowed amount” is defined as the median of the total allowed amounts the hospital has historically received from a third-party payer for an item or service for a time period no less than 12 months and no longer than 15 months prior to the date the MRF is posted. Should the calculated median fall between two observed allowed amounts, the median allowed amount is the next highest observed value. In section XIX. of this final rule with comment period, CMS is finalizing as proposed that if a payer-specific negotiated charge is based on a percentage or algorithm, the hospital must describe the algorithm or percentage and calculate and encode the median allowed amount in dollars for that item or service. Finally, in section XIX. of this final rule with comment period, CMS is finalizing, with modification, that hospitals must use EDI 835 ERA transaction data or an alternative equivalent source of remittance data that includes the same information as EDI 835 ERA transaction data would include, to calculate and encode the allowed amounts for items and services based on a percentage or algorithm in the MRF. We refer readers to section XIX. of this final rule with comment period for more information regarding the specific final policy.
A simplified example for the purpose of illustrating this process is as follows:
For its cost reporting period ending on September 30, 2026, a hospital had MAO payer-specific negotiated charges for MS-DRG 123 for five MAOs: MA1, MA2, MA3, MA4, and MA5.
The hospital filed its cost report on February 28, 2027.
The hospital made available to the public its MRF on January 1, 2027. This MRF did not contain MAO payer-specific negotiated charges for MA5 because the hospital stopped contracting with MA5 and began contracting with a new MAO, MA6. ( printed page 54018)
Step 1. The hospital identified the following MAO payer-specific negotiated charge information for MS-DRG 123 from its January 1, 2027 MRF:
- MA1: $7,400
- MA2: $7,200
- MA3: $7,500
- MA4: $7,300 (algorithm-based)
- MA6: $7,400
Note, as the payer-specific negotiated charge for MA4 was based on an algorithm, the hospital substituted the dollar amount in the MRF required under 45 CFR 180.50(b)(2)(ii)(C) for the algorithm.
Step 2. The hospital summed the number of inpatient discharges that occurred during the cost report period ending September 30, 2026, for each MAO for MS-DRG 123.
- MA1: 2 discharges
- MA2: 1 discharge
- MA3: 1 discharge
- MA4: 3 discharges
- MA5: 2 discharges
Step 3. The hospital listed each MAO payer-specific negotiated charge (from Step 1) the number of times as there were inpatient discharges that occurred during the cost reporting period for that MAO (from Step 2).
- MA1: $7,400, $7,400
- MA2: $7,200
- MA3: $7,500
- MA4: $7,300, $7,300, $7,300
For example, the $7,400 MA1 charge from Step 1 was listed two times because there were two discharges for MS-DRG 123 that occurred during the cost report period ending September 30, 2026, for MA1; the MRF charge of $7,200 for MA2 was listed once because there was one discharge; the MRF charge of $7,500 for MA3 was listed once because there was one discharge; the MRF charge of $7,300 for MA4 was listed three times because there were three discharges, there is no MRF charge for MA5 as the hospital no longer contracted with that MAO, and the MRF charge of $7,400 for MA6 was not listed as there were no discharges during the cost reporting period for that MAO.
Step 4. The median charge for MS-DRG 123 is $7,300 because that is the median of the charges in the list from Step 3.[503] (Note that if the list had contained an even number of charges, the median would have been the mean of the two middle numbers).[504]
- $7,200-MA2
- $7,300-MA4
- $7,300-MA4
- $7,300-MA4
- $7,400-MA1
- $7,400-MA1
- $7,500-MA3
For purposes of this calculation, we proposed to define the term “payer-specific negotiated charge” as the charge that a hospital has negotiated with a MAO for an item or service. We proposed to use this definition of payer-specific negotiated charge because it would capture the charges that are negotiated between hospitals and MAOs and be able to provide the data needed to support the use of market-based information for payment purposes within the MS-DRG relative weight calculation. For consistency, the definition of “payer-specific negotiated charge” that we proposed is the same as the definition at 45 CFR 180.20 for purposes of our requirements for hospitals to make their standard charges available to the public. We also proposed to define “items and services” as all items and services, including individual items and services and service packages, that could be provided by a hospital to a patient in connection with an inpatient admission for which the hospital has established a standard charge.[505] (With respect to service packages, we note that an MS-DRG, as established by CMS under the MS-DRG classification system, is a type of service package consisting of items and services based on patient diagnosis and other characteristics.) We proposed this definition of “items and services” because we believe it captures the types of items and services, including service packages, that a hospital would use to calculate and report the median payer-specific negotiated charge for each MS-DRG to support the use of market-based rate information by MS-DRG within the MS-DRG relative weight calculation. For purposes of this calculation, an MAO is defined as in 42 CFR 422.2 and means a public or private entity organized and licensed by a State as a risk-bearing entity (with the exception of provider-sponsored organizations receiving waivers) that is certified by CMS as meeting the MA contract requirements. We note that these definitions are the same as those finalized in the FY 2021 IPPS/LTCH PPS final rule.
As finalized in the FY 2021 IPPS/LTCH PPS final rule (85 FR 58888), we proposed that subsection (d) hospitals in the 50 states and DC, as defined at section 1886(d)(1)(B) of the Act, and subsection (d) Puerto Rico hospitals, as defined under section 1886(d)(9)(A) of the Act, would be required to report the median payer-specific negotiated charge information. In the CY 2026 OPPS/ASC proposed rule, we noted that hospitals that do not negotiate payment rates and only receive non-negotiated payments for service would be exempted from this proposed data collection. Examples of subsection (d) hospitals that only receive non-negotiated payment rates include hospitals operated by an Indian Health Program as defined in section 4(12) of the Indian Health Care Improvement Act or Federally owned and operated facilities. In the CY 2026 OPPS/ASC proposed rule, we noted that the proposed data collection requirement would apply to a smaller subset of hospitals as compared to the public reporting requirements under the hospital price transparency regulations. We recognized that Critical Access Hospitals (CAHs) may, in some instances, negotiate payment rates; however, because CAHs are not subsection (d) hospitals and are not paid on the basis of MS-DRGs, CAHs would not be subject to the proposed data collection requirement. We also noted that rural emergency hospitals would not be subject to the proposed data collection requirement given that they do not provide inpatient services.
On March 12, 2025, CMS announced the intention to end the Maryland Total Cost of Care Model.[506] We proposed that hospitals in Maryland, which are currently paid under the Maryland Total Cost of Care Model, would be exempted from this data collection requirement during the performance period of that Model. Following the end of the performance period of the Maryland Total Cost of Care Model, Maryland hospitals would no longer be exempt from this data collection requirement.
In the CY 2026 OPPS/ASC proposed rule, we stated that further instructions for the reporting of the proposed market-based data collection requirement on the Medicare cost report would be discussed in a forthcoming new Information Collection Request, which we stated was currently under development.
In the CY 2026 OPPS/ASC proposed rule, we stated that we believed that the administrative burden for the proposal was reduced by utilizing data that hospitals would disclose under existing and proposed hospital price transparency requirements relative to if hospitals did not already have this data compiled. We referred readers to section XXII.E. of the CY 2026 OPPS/ASC proposed rule for discussion of the ( printed page 54019) estimated burden for hospitals as a result of the proposed policy.
We also proposed to amend 42 CFR 413.20(d)(3) to reflect this proposed requirement. Specifically, we proposed to amend § 413.20(d)(3) to require hospitals to report the median payer-specific negotiated charge by MS-DRG for MAOs on the Medicare cost report. We proposed to capture this proposed data collection requirement in regulation at § 413.20(d)(3)(i)(B). We proposed that this requirement would be effective for cost reporting periods ending on or after January 1, 2026.
3. Market Based MS-DRG Relative Weight Methodology
As previously discussed, we proposed a new market-based methodology for estimating the MS-DRG relative weights, beginning in FY 2029. We noted that the proposed market-based MS-DRG relative weight methodology would be the same market-based MS-DRG relative weight methodology that was initially adopted in the FY 2021 IPPS/LTCH PPS final rule (85 FR 58879 through 58881). Specifically, we proposed to implement a methodology for calculating the MS-DRG relative weights using the median payer-specific negotiated charge for MAOs for each MS-DRG, as described in this section and reported on the cost report. For the reasons discussed in section XX.B. of the CY 2026 OPPS/ASC proposed rule and this final rule with comment period, based on our further review, in the CY 2026 OPPS/ASC proposed rule we stated we believed that using the median payer-specific negotiated charge for MAOs within the MS-DRG relative weight calculation would allow for a more market-based approach to determining Medicare FFS reimbursement.
Below is a description of the steps for the proposed MS-DRG relative weight methodology change using the payer-specific negotiated charge data. We refer readers to the FY 2021 IPPS/LTCH PPS final rule (85 FR 58880 through 58881) for additional discussion of the finalized methodology which we reproposed.
- Step One: Standardize the Median Payer-Specific Negotiated Charges: In order to make the median payer-specific negotiated charges from the cost reports more comparable among hospitals, we would standardize the median payer-specific negotiated charges reported on the cost report by removing the effects of differences in area wage levels, and cost-of living adjustments for hospital claims from Alaska and Hawaii, in the same manner as under the current MS-DRG relative weight calculation for those effects.
- Step Two: Create a Single Weighted Average Standardized Median MAO Payer-Specific Negotiated Charge by MS-DRG Across Hospitals: For each MS-DRG, we would create a single weighted average across hospitals of the standardized median payer-specific negotiated charges. We would weight the standardized payer-specific negotiated charge for each MS-DRG for each hospital using that hospital's Medicare transfer-adjusted case count for that MS-DRG, with transfer adjusted case counts calculated the same way as under the current MS-DRG relative weight methodology. We note that, as discussed in the FY 2025 IPPS/LTCH PPS final rule (89 FR 69109), the current MS-DRG relative weight methodology does not include MA cases as discharges for Medicare beneficiaries enrolled in a MA managed care plan are excluded from the relative weight methodology. We believe that using the Medicare transfer-adjusted case counts would be a reasonable approach to combining the data across hospitals because it would reflect relative volume and transfer activity (that is, larger hospitals responsible for more discharges would be weighted more heavily in the calculation, hospitals that transfer more often would be weighted less heavily).
- Step Three: Create a Single National Weighted Average Standardized MAO Payer-Specific Negotiated Charge Across all MS-DRGs: We would create a single national weighted average across MS-DRGs of the results of Step Two, where the weights are the national Medicare transfer adjusted case counts by MS-DRG.
- Step Four: Calculate the Market-based Relative Weights: For each MS-DRG, the market-based relative weight would be calculated as the ratio of the single weighted average standardized median MAO payer-specific negotiated charge for that MS-DRG across hospitals from Step Two to the single national weighted average standardized median MAO payer-specific negotiated charge across all MS-DRGs from Step Three.
- Step Five: Normalize the Market-based Relative Weights: We noted that as under the current cost-based MS-DRG relative weight methodology, the market-based relative weights would be normalized by an adjustment factor so that the average case weight after recalibration would be equal to the average case weight before recalibration. We stated that as under the current cost-based relative weight estimation methodology, the normalization adjustment is intended to help ensure that recalibration by itself neither increases nor decreases total payments under the IPPS, as required by section 1886(d)(4)(C)(iii) of the Act.
In the CY 2026 OPPS/ASC proposed rule, we stated that we believed initially there would be minimal impacts to the relative weights calculated under the proposed market based MS-DRG relative weight methodology (which would utilize the median payer-specific negotiated charge data negotiated between hospitals and their MAOs) beginning in FY 2029, given the relationship between the MAO rates and Medicare FFS rates (as evidenced by feedback from commenters as discussed in the FY 2021 IPPS/LTCH PPS final rule and the results of our literature review). In the CY 2026 OPPS/ASC proposed rule we stated that, if the proposed policy were finalized, we would expect, for some period of time following implementation of the proposed market-based MS-DRG relative weight methodology, to continue to estimate and publicly provide, for informational purposes, the MS-DRG relative weights as calculated using our current cost-based estimation methodology.
In addition, similar to our discussion in the FY 2021 IPPS/LTCH PPS final rule (85 FR 58886 through 58887), in the CY 2026 OPPS/ASC proposed rule we stated that, if the proposed policy were finalized, we would intend to provide additional opportunity for the public to review the MAO median payer-specific negotiated charge data received prior to the utilization of this data in the market-based MS-DRG relative weight methodology beginning in FY 2029. We continue to believe this would allow for additional discussions, public review, and conversation about utilizing this market-based data in the MS-DRG relative weight methodology.
We sought comment on all elements of the proposed market-based data collection for cost reporting periods ending on or after January 1, 2026, and market-based methodology for estimating the MS-DRG relative weights beginning in FY 2029. We also sought comments on potential unintended consequences of the proposal, if any, including special considerations if needed to mitigate those potential consequences for certain hospitals. We also sought comment on how these or other market-based strategies could be utilized in additional Medicare FFS payment systems and the benefits of these market-based approaches.
In this section, we summarize and respond to the public comments received on these proposals. Commenters included individuals, consumer and patient advocacy organizations, hospitals and health ( printed page 54020) systems, hospital and State hospital associations, medical associations, health benefits consultants, health information technology (IT) organizations, and academic institutions, among others.
Comment: The vast majority of commenters were opposed to the proposals that hospitals report median payer specific negotiated charges for MAOs on the Medicare cost report and that CMS use that data to recalibrate the IPPS MS-DRG relative weights. Commenters asserted that the proposals are not authorized by and conflict with CMS's statutory authority, are arbitrary and capricious, and require new unsupported expansive interpretations of the statute. Commenters indicated that the statute requires the MS-DRG weights reflect “the relative hospital resources used”—that is a resource-based system—not market rates negotiated with MA plans. Commenters stated that the cost reporting provisions CMS invoked authorize collection only of information necessary to determine appropriate payment amounts due to a provider—not the collection of information that they asserted cannot lawfully be used to set payments.
More specifically, many commenters stated that the text of section 1886(d)(4)(B) of the Act compels a resource-based, not a market-based methodology. Commenters discussed the statutory requirements that CMS assign to each MS-DRG “an appropriate weighting factor which reflects the relative hospital resources used.” They stated that the negotiated rates are influenced by many factors, including bargaining leverage, patient populations, network needs, utilization management practices, local market concentration, out-of-network Medicare payment requirements, quality and value initiatives, episodes of care, and other contract and pricing dynamics rather than relative hospital resource use at the MS-DRG level.
With respect to the data collection proposal, commenters stated that sections 1815(a) and 1833(e) of the Act permit CMS to require providers to furnish information necessary to determine amounts due (for example, to process claims and compute legally authorized payments) but establish no broader authority. Commenters asserted that these statutory authorities do not authorize and cannot be interpreted to authorize a policy to collect third party negotiated rates because CMS cannot lawfully use that data to set IPPS payments.
In contrast to the proposed approach, many commenters indicated that the current MS-DRG cost-based methodology established in the FY 2007 IPPS rulemaking is a longstanding and better framework for improving the MS-DRG relative weights consistent with the statute. Commenters indicated that although CMS is focused on the charges in the hospital chargemaster as being inflated and not reflecting resource use, the current methodology is not based on charges, but rather on charges converted to cost.
Some commenters who opposed finalizing our proposal agreed with CMS that conceptually a methodology that relies more on market-based concepts would be preferable to the current system of administered pricing, but disagreed that our current proposal was an appropriate approach because they asserted that it risked circular and destabilizing effects across both FFS and MA and could create uncertainty within the MA program, FFS program, and value-based payment models that depend on a stable FFS baseline. They indicated CMS should engage with interested parties through multiple avenues to identify effective, stable alternatives. Examples of suggested alternatives included more sophisticated cost accounting systems, improved cost reporting, and CMS demonstrations.
A few commenters expressed support for our proposal in terms of improving the efficiency of care, ensuring payments are set appropriately, better reflecting hospital resources for inpatient items and services and improving the accuracy of MS-DRGs. One commenter who supported our proposal stated it would result in greater alignment of the MS-DRG relative weights with the statutory requirements. The commenter stated that MA rates are more likely to reflect the relativity of hospitals' true resources because those rates represent the ending point of the negotiations between hospitals and plans, as opposed to the chargemasters which may reflect the starting point of the negotiations. The commenter also indicated that although Medicare Advantage is not a market-based system, the proposal would create an opportunity for negotiations to reflect market dynamics even if the immediate impact of the proposal is minimal due to the current extremely close correlation between the MS-DRG relative weights and the MA rates.
Response: We agree with commenters that CMS is required to assign and update MS-DRG weighting factors to reflect relative resource use. Section 1886(d)(4)(A) of the Act states that the Secretary shall establish a classification of inpatient hospital discharges by diagnosis-related groups and a methodology for classifying specific hospital discharges within these groups. Section 1886(d)(4)(B) of the Act states that for each such diagnosis-related group the Secretary shall assign an appropriate weighting factor which reflects the relative hospital resources used with respect to discharges classified within that group compared to discharges classified within other groups. Section 1886(d)(4)(C)(i) of the Act states that the Secretary shall adjust the weighting factors at least annually to reflect changes in treatment patterns, technology, and other factors which may change the relative use of hospital resources. As indicated by commenters with respect to the current methodology, relative resources are accounted for when hospitals establish the costs of services. We continue to believe that the costs of services are considered when hospitals and payers negotiate rates. Commenters noted that the negotiated rates are influenced by many factors, including bargaining leverage, patient populations, network needs, utilization management practices, local market concentration, out-of-network Medicare payment requirements, quality and value initiatives, episodes of care, and other contract and pricing dynamics. While the negotiated rates may reflect a variety of factors, it does not follow that the resources necessary to perform the services based on these negotiated rates would not be considered in the negotiations. As an extreme example for purposes of illustration, in contract negotiations hospitals would not generally negotiate payments of $1,500 for heart transplant cases and MA organizations would not generally negotiate payments of $150,000 for simple pneumonia cases because those rates would not reflect the relative resources required to provide those services. Furthermore, as network needs ( e.g. network adequacy) are considerations in the negotiations between hospitals and payers and network needs involve anticipated patient utilization, we do not believe that hospitals and payers would consider anticipated patient utilization when negotiating contracts without considering the resources necessary (that is, costs) to provide those items and services for that level of patient utilization anticipated. We continue to believe that payer-specific negotiated charges that hospitals negotiate with MA organizations capture the relative resources used to provide services to patients in order to maximize profits (or, in the case of not-for-profit hospitals, net income), subject to market ( printed page 54021) constraints and conditions (supply and demand, community benefit requirements, etc.).
Because we believe the payer-specific negotiated charges reflect relative resources used ( i.e. costs) for the reasons described earlier, our proposal is entirely consistent with and based on longstanding interpretations of the statute with respect to the relationship between resources and costs. We disagree with commenters that our proposed policy is in any way arbitrary or capricious or relies on new expansive unsupported interpretations of the statute. For the same reasons, we also disagree with commenters who stated we lack authority and did not articulate a sufficient policy basis for our data collection policy. As discussed in the CY 2026 OPPS/ASC proposed rule, sections 1815(a) and 1833(e) of the Act provide us with the authority to collect data for purposes of determining the amount of payments due to the provider under the Medicare program. We proposed to collect this negotiated charge data so that it may be used in determining relative weights for purposes of payment under the IPPS. We also note that we referenced the requirement that providers follow reasonable cost principles under section 1861(v)(1)(A) of the Act when completing Medicare cost reports and reiterate that section 1861(v)(1)(A) of the Act requires reporting of data elements beyond just cost, including non-cost items and items used to determine the cost of services.
In response to comments that the current approach for establishing the MS-DRG relative weights involves estimating cost from charges, we note that it still relies on data derived from hospital chargemasters as input and it still relies on an estimation methodology to convert those charges to cost. As we indicated in the FY 2007 IPPS rulemaking (71 FR 47894-95) when initially adopting a charges-to-cost methodology, no payment methodology can be perfect because DRG-specific costs cannot be determined. We indicated that we believed that the cost-based methodology represented an improvement over the prior charge-based methodology. Similarly, we believe that while a market-based methodology, a different type of cost-based methodology, may also not be perfect, it would be an improvement over the current methodology. By using payer specific negotiated charges, we can reduce our reliance on the hospital chargemaster and utilize this data in Medicare payment methodologies so that payments more closely reflect the market cost and therefore the relative market value and resource utilization for inpatient items and services.
In response to comments that our proposed approach risks circular and destabilizing effects across both FFS and MA and could create uncertainty within the MA program, FFS program, and value-based payment models that depend on a stable FFS baseline, we disagree. We continue to believe that if market-based data (median payer-specific negotiated charges for MA organizations) are incorporated into the calculation of the MS-DRG relative weights, initially there will be limited impact on the relative weights given the current similarity between MA organization rates and Medicare FFS rates. As discussed in the CY 2026 OPPS/ASC proposed rule, we intend to provide an opportunity for the public to review the data we collect prior to use of these data in a market-based relative weight methodology. To the extent the data shows that there would be more than a limited impact on the relative weights initially, which we do not believe will be the case, CMS will be able to further consider the impact and appropriate approach to utilizing this market-based data in the MS-DRG relative weight methodology. We will continue to provide impact analyses of changes in the MS-DRG relative weights in the annual IPPS rulemaking. Also, as discussed in the CY 2026 OPPS/ASC proposed rule we expect for some period of time following implementation of this market-based MS-DRG relative weight methodology to continue to estimate and publicly provide the MS-DRG relative weights calculated using the current methodology for informational purposes. We also note that our adoption of a market-based methodology does not preclude continued engagement with interested parties to identify further improvements to our payment systems.
Comment: Commenters stated that the research CMS cited in the CY 2026 OPPS/ASC proposed rule does not analyze relative hospital resource utilization at the MS-DRG level and that CMS cannot use this research to support that the MA negotiated payment rates capture relative resource use. Furthermore, some commenters stated that the MA non-interference clause at section 1854(a)(6)(B)(iii) of the Act bars CMS from requiring particular price structures in MA provider contracts and therefore prohibits CMS from ever ensuring any correlation between MA rates and resource use.
Response: As previously discussed in this final rule with comment period, we believe the payer-specific negotiated charges would better reflect relative resources used because the rates that hospitals negotiate with MAOs capture the relative resource use to provide services to patients in order to maximize profits (or, in the case of not-for-profit hospitals, net income), subject to market constraints and conditions (supply and demand, community benefit requirements, etc.). Therefore, for the reasons previously discussed, we believe that payer-specific negotiated charges provide greater insight into the resource use of a hospital. The primary focus of the literature discussion in this context is the correlation between the payer specific charges negotiated between hospitals and MA organizations and Medicare IPPS payment rates and the implications of that correlation to analyze the impacts of the use of these data in a market-based relative weight methodology, as discussed in section XXV.C.7 of this final rule with comment period, both initially and over time, for the relative weights to the extent that the MA rates and the FFS rates differ for some services. In the CY 2026 OPPS/ASC proposed rule, we described research that chargemasters are usually highly inflated and that inflated charges have been used to secure higher payments; we discussed available literature to compare Medicare FFS and MAO payment rates and how those MAO payment rates may (emphasis added) reflect the relative hospital resources used within an MS-DRG differently than our current cost-based system, and we indicated that we considered more recent research comparing Medicare FFS rates, MAO rates, and rates of other commercial payers, some of which used data that was made public under the provisions of the Hospital Price Transparency regulations (90 FR 33805-33807). In the FY 2021 IPPS final rule we stated that taken as a whole, we continued to believe that the body of research discussed in that rulemaking suggests that payer specific charges negotiated between hospitals and MA organizations are generally well-correlated with Medicare IPPS payment rates, and payer-specific charges negotiated between hospitals and other commercial payers are generally not as well-correlated with Medicare IPPS payment rates. With respect to either type of payer-specific negotiated charges, there may be instances where those negotiated charges may (emphasis added) reflect the relative hospital resources used within an MS-DRG differently than our current cost-based methodology (85 FR 58877). As ( printed page 54022) discussed in the FY 2021 IPPS/LTCH PPS final rule (85 FR 58883) and the CY 2026 OPPS/ASC proposed rule (90 FR 33807), the MA rates to MA contracted inpatient hospitals are not required to be the same as (or based on) Medicare FFS rates; the Medicare statute only requires MAOs to pay FFS rates to a health care provider for services furnished to an MA enrollee when the MAO does not have a contract with the health care provider. Recognizing that there is currently a general correlation as shown by the research we cited, we believe that to the extent hospitals and MAOs over time negotiate different relative relationships for some services than the well correlated relationships that currently exist under the IPPS, this information adds value to the IPPS regarding resource use and should be incorporated (90 FR 33807).
We agree with commenters that the MA non-interference clause at section 1854(a)(6)(B)(iii) of the Act generally bars CMS from requiring particular price structures in MA provider contracts. However, our proposal does not create any requirements for particular price structures in MA provider contracts.
Comment: Commenters stated that the proposals would create distortions in the MS-DRG relative weights and that CMS has not modelled the impacts of or otherwise accounted for these distortions in the proposed policy. Commenters asserted these distortions are evidence as to why the proposed approach would be unlawful if adopted (that is, not resource-based), as well as evidence as to why the proposed approach is inappropriate even if CMS had the authority to adopt it. Commenters discussed the statutory requirement at section 1886(d)(4)(C)(i) of the Act to update the relative weights at least annually to reflect “changes in treatment patterns, technology . . . and other factors which may change the relative use of hospital resources” and asserted that the proposal failed to do so. Commenters stated that because MA rates are often based on Medicare FFS rates the proposal would over time lock in outdated weights in a closed loop that adds no independent information on these factors. According to the commenters, this locked in “circularity” would contradict the statutory requirement to update weights to reflect changes in resource use. Some commenters stated that over time as the MS-DRG weights become outdated MA plans and hospitals would rely more on non-MS-DRG based approaches. Commenters further stated that even absent circularity, the MA negotiated rates cannot assist with determining when changes to the MS-DRG classification system itself are necessary (for example splitting MS-DRGs) because unlike the current case-level methodology the MA negotiated rates do not provide information on resource use variations within an MS-DRG. Commenters also stated that the MS-DRG relative weights could be distorted by a lag between when a hospital updates its price transparency data and when it submits its cost report.
Commenters stated that in cases where MA plans pay hospitals for inpatient services based on charges, MS-DRG relative weights would be distorted by different cost-to-charge ratios across departments. Commenters stated the proposed process would yield higher relative weights for MS-DRGs that had higher mark-ups of charges relative to costs to the extent some MA plans still pay hospitals a rate based on discounts off charges.
Commenters stated that the combined data across hospitals would not be nationally representative for a variety of reasons. Commenters stated that in cases where MA plans pay hospitals for inpatient services as a percentage of FFS Medicare payments, MS-DRG relative weights would be distorted by existing FFS Medicare policy-based payments that do not correspond to the relative cost of providing the service. Commenters cited the example of FFS Medicare payments to hospitals including uncompensated care payments to help support hospitals' costs of treating the uninsured as well as wage index policies reflected in the current payment rates. Commenters stated the proposal would yield higher relative weights for MS-DRGs for procedures disproportionately performed at hospitals that receive these additional policy-based payments. Commenters indicated that the proposed methodology would use no data from subsection (d) providers that exclusively contract with MAOs on a capitated basis and/or have no MA network participation agreements. They also stated payment elements that are excluded from the payer-specific negotiated rates for MAOs may skew the data. For example, they stated that the payer-specific negotiated rate would exclude components of the total payment amount that are based on risk-sharing or value-based payment methodologies, and the use of those payment methodologies is not uniformly distributed across hospitals. Commenters stated that using Medicare data on fee-for-service case counts to develop a single weighted average standardized median MAO payer-specific negotiated rate by MS-DRG across hospitals means that data from hospitals with less MA penetration, which have less at stake in MA negotiations, is weighted more heavily than data from hospitals with high MA penetration that have more at stake. Commenters also stated that the impact of stoploss provisions and prior authorizations are similarly not uniformly distributed and would skew the relative weights. Commenters also asserted that the proposed approach is not actually market-based due to a lack of competing hospitals and/or a lack of competing MAOs in many areas.
Commenters also stated that CMS has not and cannot analyze the impacts of its proposed policy because it has not yet collected the data and contrasted the impact analysis of this proposed policy with the impact analysis performed when CMS adopted the current cost-based MS-DRG methodology. Commenters stated that the policy could reduce access to care, particularly for high-cost new technologies such as innovative cell and gene therapies, and in rural communities.
Response: With respect to the comments that our proposals would create distortions in the MS-DRG relative weights, we note that there is some similarity between these comments and the comments that we received during the FY 2021 rulemaking (85 FR 58883). We appreciate the additional feedback from commenters regarding differences in payment methodologies and other factors that influence the contracts between MA organizations and hospitals. We thank commenters for their concerns regarding the comparability of payer-specific negotiated charges by MS-DRG due to these and other factors and the potential impact on the MS-DRG relative weights. We believe, as discussed earlier, based on the literature review we conducted and feedback from commenters, that MA rates and Medicare FFS rates are often similar and/or are highly reliant on one another. However, as previously discussed, MA rates to MA contracted inpatient hospitals are not required to be the same as (or based on) Medicare FFS rates. We continue to believe that if market-based data (median payer-specific negotiated charges for MA organizations) are incorporated into the calculation of the MS-DRG relative weights, initially there will be limited impact on the relative weights given the current similarity between MA organization rates and Medicare FFS rates, but that over time the proposal would create an opportunity for negotiations to reflect market dynamics, as noted by a commenter who supported our proposed policy. As discussed ( printed page 54023) earlier, to the extent the data shows that there would be more than a limited impact on the relative weights initially, which we do not believe will be the case, we intend to provide an opportunity for additional public input and will continue to provide impact analyses of changes in the MS-DRG relative weights in the annual IPPS rulemaking. We remain open to adjusting our finalized policy for the MS-DRG relative weights through future rulemaking prior to the effective date if appropriate.
We note that we did not propose modifications to the process for making adjustments to the MS-DRG classification system itself, nor did we propose changes to the new technology or outlier payment policies at this time. We may revisit these issues in future rulemaking as part of our larger goal of reducing reliance on the hospital chargemaster.
Comment: Commenters indicated that the proposed data collection on Worksheet S-12 would be administratively burdensome and that it was often unclear how the hospital should perform the required calculations. For example, commenters indicated that it was not clear from the proposed instructions how hospitals should (1) crosswalk the MAO's payment methodology to an MS-DRG for payers that do not use the MS-DRG as a basis of payment and (2) calculate the cases for the weighted median.
With respect to the crosswalking issue, commenters indicated that hospitals will not readily be able to crosswalk an MAO's rate for an item or service (or bundle of items and services) to a particular MS-DRG when the MAO does not use an MS-DRG-based rate. For example, commenters indicated that the proposed instructions do not provide information about how a provider would determine an MS-DRG rate for an MAO that pays for an inpatient stay based on a per-diem rate where the length of stay for cases that map to a particular MS-DRG will vary between patients.
Commenters also indicated that an MAO's payment methodology might bundle different cases together such that some cases within an MS-DRG are paid at one rate and others that map to that same MS-DRG are paid at a different rate or such that a single rate is applied to a range of discharges that map to multiple MS-DRGs. Commenters stated the proposed instructions assume that it would be a simple matter of crosswalking one rate to a single discharge and provide no guidance on how the MAO's rate should be determined for a particular MS-DRG when this is not the case. Commenters indicated that to the extent that a hospital would be required to identify each patient discharge to which a particular rate applied, then determine the MS-DRG that would have applied to that case, and then calculate or determine an MS-DRG rate for that payer based on that claims history, CMS severely understates the hospital burden in completing Worksheet S-12 and that this burden and approach is wholly unsupported.
Commenters indicated the calculation of the weighted median for each MS-DRG requires summing the number of inpatient discharges for each MAO for each MS-DRG, but a hospital might get a different result depending on whether inpatient discharges are counted as (1) all inpatient discharges for the MAO's members, (2) those inpatient discharges where the MAO made payment (whether inpatient or otherwise), or (3) those inpatient discharges where the MAO paid for the care at the inpatient rate.
Response: In response to comments that it was not clear from the proposed instructions how hospitals should crosswalk the MAO's payment methodology to an MS-DRG for payers that do not use the MS-DRG as a basis of payment, as discussed in the CY 2026 OPPS/ASC proposed rule (90 FR 33808) if there are codes identified that are not MS-DRG codes, or discharges that are not classified to MS-DRGs, the hospital would crosswalk those codes or classify those discharges to MS-DRGs. Hospitals can utilize the CMS GROUPER and associated definitions manual for this purpose. Hospitals have access to the publicly available version of the CMS Grouper used to group ICD-10 diagnosis and procedure codes to MS-DRGs.[507] This software and associated definitions manual can be used to crosswalk the code(s) in the MRF or classify the discharge to an MS-DRG code.
In response to comments that a hospital would be required to (1) identify each patient discharge to which a particular rate applied, then (2) determine the MS-DRG that would have applied to that case, and then (3) calculate or determine an MS-DRG rate for that payer based on that claims history, we agree with the first two statements, but is it unclear what the commenters mean by the third statement with respect to the claims history. The rates associated with the discharge would come from the MRF and would not be recalculated from the claims history for this purpose. For example, if the rate associated with an MS-DRG discharge was strictly based on a per diem, the hospital would multiply the per diem amount from the MRF by the number of days of the hospital stay. More specifically, as discussed in the CY 2026 OPPS/ASC proposed rule (90 FR 33808) and applied to this situation, in Step 1 the hospital would use its most recent MRF as of the hospital's cost report filing date to identify the MAO payer-specific negotiated charge under 45 CFR 180.50(b)(2)(ii) that the hospital has negotiated with its MAOs for the discharge. Because the payer-specific negotiated charge is based on an algorithm, the hospital would identify and substitute the dollar amount in the MRF required under 45 CFR 180.50(b)(2)(ii)(C) for the algorithm. In this example, that means that the hospital would identify the per diem amount in the MRF and substitute the per diem amount times the length of stay for that discharge for the algorithm.
In response to the comments indicating that the calculation of the weighted median for each MS-DRG requires summing the number of inpatient discharges for each MAO for each MS-DRG, and stating that a hospital might get a different result depending on whether inpatient discharges are counted as (1) all inpatient discharges for the MAO's members, (2) those inpatient discharges where the MAO made payment (whether inpatient or otherwise), or (3) those inpatient discharges where the MAO paid for the care at the inpatient rate, the hospital should use (3) those inpatient discharges where the MAO paid for the care at the inpatient rate as the sole purpose of the discharges in the calculation is to weight the rates contained in the MRF.
We continue to believe that hospitals have the capacity, based on the instructions provided within this final rule with comment period, and the forthcoming revision of the Information Collection Request currently approved under OMB control number 0938-0050 to report this data on the Medicare cost report for cost reporting periods ending on or after January 1, 2026. We may provide additional guidance as appropriate or as determined necessary. Absent additional guidance, we believe that hospitals have the capability to report this market-based data for cost reporting periods ending on or after January 1, 2026.
The burden associated with our proposal is discussed in section XXII of this final rule with comment period. ( printed page 54024)
Comment: Commenters stated that proposing a methodology for one payment system within the CY 2026 OPPS/ASC proposed rule of a different payment system does not reflect the intent of the annual rulemaking cycles as it will not result in the amount or kind of interested party feedback necessary to properly evaluate the proposal.
Response: We may, in certain circumstances, propose policies in a rule associated with a different payment system, including when those policies have cross-cutting implications or when statutory or operational considerations necessitate timely implementation. We believe that the established notice-and-comment rulemaking process for the OPPS provides interested parties with the opportunity to review the proposal and affords all interested parties the ability to submit feedback. We also note the substantial overlap between subsection (d) hospitals and hospitals subject to the OPPS.
After consideration of the comments received, and for the reasons previously discussed, we are finalizing our proposed market-based data collection requirement as proposed. Specifically, we are finalizing that hospitals would report on the Medicare cost report the median payer-specific negotiated charge that the hospital has negotiated with all of its MA organization payers, by MS-DRG, for cost reporting periods ending on or after January 1, 2026. We are also finalizing our proposal that if the hospital disclosed the payer-specific negotiated charge as a percentage or algorithm on the MRF, the hospital would use the “median allowed amount” (as finalized in section XIX. of this final rule with comment period) to calculate the median of the payer-specific negotiated charges. To determine the median payer-specific negotiated charge for MA organizations for a given MS-DRG, a hospital would follow the process as outlined in section XX.C.2. of this final rule with comment period. We are finalizing our definitions of “payer-specific negotiated charge,” “MA organization” and “items and services,” as proposed. For the purposes of calculating and reporting the median payer-specific negotiated charge the hospital has negotiated with all of its MA organization payers, by MS-DRG, we define an MA organization the same way as proposed, and defined in 42 CFR 422.2; namely, an MA organization means a public entity or private entity organized and licensed by a State as a risk-bearing entity (with the exception of provider-sponsored organizations receiving waivers) that is certified by CMS as meeting the MA contract requirements. We are finalizing as proposed that subsection (d) hospitals in the 50 States and DC, as defined at section 1886(d)(1)(B) of the Act, and subsection (d) Puerto Rico hospitals, as defined under section 1886(d)(9)(A) of the Act, would be required to report this median payer-specific negotiated charge information. As discussed in the CY 2026 OPPS/ASC proposed rule, hospitals that do not negotiate payment rates and only receive non-negotiated payments for service would be exempted from this data collection.
We are finalizing our proposed amendment to the regulations to reflect this data collection requirement at 42 CFR 413.20(d)(3), without modification. Specifically, we are finalizing our proposal to amend § 413.20(d)(3) to require hospitals to report the median payer-specific negotiated charge by MS-DRG for MAOs on the Medicare cost report. This data collection requirement is effective for cost reporting periods ending on or after January 1, 2026. As stated in the CY 2026 OPPS/ASC proposed rule, further instructions for the reporting of this market-based data collection requirement on the Medicare cost report will be discussed in a forthcoming new Information Collection Request. This new information collection request will be submitted to OMB for review under control number 0938-1486 (CMS-10935). The OMB control number will not be valid until formally approved by OMB. Please see section XXII.E. of this final rule with comment period for further details. We may provide additional guidance regarding this data collection policy as determined appropriate or necessary. However, absent additional guidance, we believe that hospitals have the capability to report this market-based data, as required, for cost reporting periods ending on or after January 1, 2026.
We are also finalizing the adoption of a market-based MS-DRG relative weight methodology effective for FY 2029. We are finalizing the market-based MS-DRG relative weight methodology, as described within the CY 2026 OPPS/ASC proposed rule, without modification. Specifically, we will begin using the median payer-specific negotiated charge by MS-DRG for MA organizations in the market-based MS-DRG relative weight methodology beginning with the relative weights calculated for FY 2029. We also remain open to making modifications and refinements to this market-based methodology, through rulemaking prior to the FY 2029 effective date. We are not finalizing, at this time, a transition period to this market-based MS-DRG relative weight methodology. We may, however, consider this in future rulemaking prior to FY 2029. We expect, for some period of time, following implementation of this market-based MS-DRG relative weight methodology, as discussed in the CY 2026 OPPS/ASC proposed rule, to continue to estimate and publicly provide the MS-DRG relative weights calculated using the cost-based estimation methodology for informational purposes.
We will continue to consider ways to reduce the role of hospital chargemasters in Medicare IPPS payments, as we described in the CY 2026 OPPS/ASC proposed rule, to further reflect market-based approaches in Medicare FFS payments, to the extent permitted by law.
XXI. Graduate Medical Education Accreditation
A. Executive Order 14279
Executive Order 14279 (April 23, 2025), entitled “Reforming Accreditation to Strengthen Higher Education,” directs the Attorney General, in consultation with the Secretary of Health and Human Services, to “investigate and take appropriate action to terminate unlawful discrimination by American medical schools or graduate medical education entities that is advanced by the Liaison Committee on Medical Education or the Accreditation Council for Graduate Medical Education or other accreditors of graduate medical education, including unlawful `diversity, equity, and inclusion' requirements under the guise of accreditation standards.” [508] The Executive Order further directs that standards for training doctors should focus solely on providing the highest quality care, and should not require or encourage educational institutions to discriminate unlawfully on the basis of race.
The Accreditation Council for Graduate Medical Education (`ACGME') is the primary organization in the United States that currently conducts accreditation for Graduate Medical Education (`GME') Programs. While ACGME accreditation is a voluntary process, programs that are not accredited by the ACGME generally do not receive Medicare funding from CMS for Direct Graduate Medical Education (DGME) and Indirect Medical Education (IME). Additionally, if the ACGME withdraws accreditation, residents generally must receive assistance to ( printed page 54025) continue their education from other ACGME-accredited programs.[509]
Historically, the ACGME identified `diversity, equity, and inclusion' as a primary value of the organization and a central component of its vision for graduate medical education.[510] The ACGME's Common Program Requirements required that institutions “must engage in practices that focus on mission-driven, ongoing, systematic recruitment and retention of a diverse and inclusive workforce of residents, fellows (if present), faculty members, senior administrative staff members,” and that organizations' “programs implement policies and procedures related to recruitment and retention of individuals underrepresented in medicine and medical leadership.” [511] In practice, many such diversity, equity, and inclusion programs unlawfully discriminate against Americans on the basis of race. In Students for Fair Admissions v. President and Fellows of Harvard College (2023), the U.S. Supreme Court held that race-based admissions policies, even when focused on the goal of diversity, violate the Equal Protection Clause of the Fourteenth Amendment unless they satisfy strict scrutiny.[512] While the ruling applies specifically to admissions decisions at institutions of higher education, its broader reasoning—especially the requirement that any use of race be narrowly tailored to a compelling interest—strongly suggests that race-conscious elements in Diversity, Equity, and Inclusion (DEI) initiatives in Federally funded education programs are generally impermissible. These programs raise particular concerns in the medical context, where patients and the larger society have a compelling need for medical education to be focused primarily on excellence and delivering the best possible care to patients.
B. Definition of “Approved Medical Residency Programs”
Section 1886(h) of the Act, as added by section 9202 of the Consolidated Omnibus Budget Reconciliation Act (COBRA) of 1985 (Pub. L. 99-272), and as currently implemented in CMS regulations at 42 CFR 413.75 through 413.83, establishes a methodology for determining payments to hospitals for the direct costs of approved graduate medical education (GME) programs. Section 1886(h)(2) of the Act sets forth a methodology for the determination of a hospital-specific base-period per resident amount (PRA) that is calculated by dividing a hospital's allowable direct costs of GME in a base period by its number of full-time equivalent (FTE) residents in the base period. In general, Medicare direct GME payments are calculated by multiplying the hospital's updated PRA by the weighted number of FTE residents working in all areas of the hospital complex (and at non-provider sites, when applicable), and the hospital's Medicare share of total inpatient days.
Section 1886(d)(5)(B) of the Act provides for a payment adjustment known as the indirect medical education (IME) adjustment under the IPPS for hospitals that have residents in an approved GME program, to account for the higher indirect patient care costs of teaching hospitals relative to nonteaching hospitals. The regulations regarding the calculation of this additional payment are located at 42 CFR 412.105. The hospital's IME adjustment applied to the DRG payments is calculated based on the ratio of the hospital's number of FTE residents training in either the inpatient or outpatient departments of the IPPS hospital (and, for discharges occurring on or after October 1, 1997, at non-provider sites, when applicable) to the number of inpatient hospital beds.
Hospitals may receive direct GME and IME payments for residents in “approved medical residency training programs.” Section 1886(h)(5)(A) of the Act defines an “approved medical residency training program” as “a residency or other postgraduate medical training program participation in which may be counted toward certification in a specialty or subspecialty and includes formal postgraduate training programs in geriatric medicine approved by the Secretary.”
The regulations at § 413.75(b) define an “approved medical residency program” for purposes of direct GME payment as a program that meets one of four criteria: (1) is approved by one of the national organizations specified in the regulations at § 415.152; (2) may count towards certification of the participant in a specialty or subspecialty listed in the current edition of certain publications specified in the regulations; (3) is approved by the ACGME as a fellowship program in geriatric medicine; or (4) is a program that would be accredited except for the accrediting agency's reliance upon an accreditation standard that involves induced abortions, regardless of whether the standard provides exceptions or exemptions. The regulations at § 412.105(f)(1)(i) define an “approved teaching program” similarly for purposes of IME payment.
The regulations at § 415.152 define an “approved graduate medical education program” as a residency program approved by one of the following national organizations (or their predecessors): The Accreditation Council for Graduate Medical Education (ACGME), the American Osteopathic Association (AOA), the Commission on Dental Accreditation (CODA) of the American Dental Association, and the Council on Podiatric Medical Education (CPME) of the American Podiatric Medical Association. Thus, in general, under §§ 413.75(b) and 412.105(f)(1)(i), an “approved” program can be a program that is accredited by one of these national organizations, or one that leads toward board certification by the American Board of Medical Specialties (ABMS).
The statute gives CMS authority to specify additional criteria for approved GME programs. Therefore, to ensure that accreditation for approved medical residency programs is in compliance with applicable laws related to race-based admission policies and to improve the accreditation process, in the CY 2026 OPPS/ASC proposed rule (90 FR 33811 through 33812), we proposed that accreditors may not require as part of accreditation, or otherwise encourage institutions to put in place, diversity, equity, and inclusion programs that encourage unlawful discrimination on the basis of race or other violations of Federal law. The effective date of this proposed policy would be January 1, 2026. Additionally, we noted that the Secretary may recognize other organizations that meet or exceed Medicare's requirements as accreditors to increase the potential for competition in the accreditation space and improve the quality of the accreditation process.
Our intent in issuing this proposal was to ensure that accreditors of academic medical institutions are focused on the mission of ensuring excellence in graduate medical education, of improving the potential for competition in the accreditation space, and of eliminating unlawful and discriminatory DEI programs. We invited commenters' feedback on this proposal. The following is a summary of ( printed page 54026) the comments we received and our responses.
Comment: CMS received numerous comments in support of our proposal to modify the definition of “approved medical residency program” so that accreditors may not require as part of accreditation, or otherwise encourage institutions to put in place, diversity, equity, and inclusion programs that encourage unlawful discrimination on the basis of race or other violations of Federal law. A majority of these commenters also expressed support for the Secretary's authority to recognize other organizations as accreditors of graduate medical education programs.
Many commenters that supported the proposal also recommended that CMS expand the policy to explicitly prohibit the use of accreditation standards that violate Federal healthcare conscience laws, including the Church Amendments (42 U.S.C. 300a-7), the Coats-Snowe Amendment (42 U.S.C. 238n), certain provisions of the Affordable Care Act (Pub. L. 111-148), and the Weldon Amendment. These statutory provisions generally prohibit discrimination against recipients of certain Federal funding who refuse to perform abortions or provide other services in violation of their moral or religious convictions. Specifically, a commenter recommended defining an approved program at § 412.105(f)(1)(i)(A) as one that “[i]s approved by one of the national organizations listed in § 415.152 of this chapter, provided that the national organization does not use accreditation criteria that promote or emphasize diversity, equity, inclusion, or awareness based on race, color, sex, sexual orientation or identity, national origin, or any other characteristic which serves as a proxy to achieve the same ends, or that would cause entities or individuals to act contrary to objections protected by Federal conscience and nondiscrimination statutes, including 42 U.S.C. 300a-7, 42 U.S.C. 238n, 42 U.S.C. 18113, or the Weldon Amendment, for example, Consolidated Appropriations Act, 2023, Public Law 117-328, div. H, title V General Provisions, section 507(d)(1) (Dec. 29, 2022)) [ sic].” The commenter recommended similar language for other sections of the regulations subject to this proposal. Other commenters stated that the existing regulations at §§ 413.75(b) and 412.105(f)(1)(i)(D), which recognize approved programs that would be accredited except for the accrediting agency's reliance upon an accreditation standard that requires an entity to perform an induced abortion, should be expanded to include other services, such as in vitro fertilization, surrogacy, certain forms of family planning, sterilization, sex-rejecting procedures, assisted suicide, euthanasia, medical aid in dying, voluntary stopping of eating and drinking, and inducing death for organ harvesting. A commenter also urged CMS to require that abortion training be offered under an opt-in only model, as opposed to the ACGME's current opt-out requirement.
Commenters stated that these additional provisions are necessary to protect individuals and other healthcare entities from discrimination on the basis of their moral and religious beliefs. Some commenters argued that, as a result of current requirements, individual physicians and faith-based institutions are effectively forced to act in violation of their conscientious objections in order to complete their training or secure Medicare GME funding; several respondents described instances in which they or others experienced allegedly discriminatory treatment as a result of their objections to certain training requirements.
A commenter that supported the proposal also expressed concern that Medicare GME funding may be used to pay for abortions, in violation of the Hyde Amendment, which generally prohibits the use of Federal funding for abortion except under limited circumstances.
Another commenter that supported the proposal encouraged CMS to add language to the regulations that would prevent accreditors from engaging in word play as a means of circumventing the proposed policy. Specifically, the commenter recommended defining an approved program at § 412.105(f)(1)(i)(A) as one that “[i]s approved by one of the national organizations listed in § 415.152 of this chapter, provided that the national organization does not use accreditation criteria that either promote or emphasize diversity, equity, inclusion, or awareness based on race, color, sex, sexual orientation or gender identity, national origin, or any other characteristic which serves as a proxy to achieve the same ends or would cause the hospitals to violate, or reasonably cause the hospitals to believe that they would violate, Federal civil rights laws if adopted by the hospitals.” The commenter recommended similar language for other sections of the regulations subject to this proposal.
Response: We thank the commenters for their support of our proposal.
In response to comments recommending further expansion of the proposed policy, we may take these comments into consideration for future rulemaking. We emphasize that regardless of the inclusion of explicit language in the GME regulations, no entity or individual may be forced to act contrary to objections protected by Federal conscience and nondiscrimination statutes. We also note with regard to the Hyde Amendment that both direct GME and IME payments are made only with respect to services otherwise payable under Medicare, and that abortion services are not payable under Medicare except under the limited circumstances specified in the Hyde Amendment.
In response to a commenter's concerns regarding potential circumvention of the proposed regulations, we believe the language we proposed is sufficiently clear to prevent gaming. However, as we also note in response to a subsequent comment, we agree that the regulations should more explicitly specify the types of practices that will be prohibited under our finalized policy. Therefore, we are finalizing, with modification, our proposed change to the definition of “approved medical residency program” and equivalent terms in the regulations at 42 CFR 412.105(f)(1)(i), 413.75(b) and 415.152, to state that accrediting organizations may not use accreditation criteria that promote or encourage discrimination on the basis of race, color, national origin, sex, age, disability, or religion, including the use of those characteristics or intentional proxies for those characteristics as a selection criterion for employment, program participation, resource allocation, or similar activities, opportunities, or benefits.
In connection with this policy, we emphasize the independent obligation of all recipients (and subrecipients) of Federal financial assistance to comply with all applicable Federal civil rights laws and, thus, not to discriminate on the bases prohibited by such laws. Thus, we further note that prohibited practices include all other conduct in violation of Federal antidiscrimination laws, including any “unlawful practices” under the Attorney General's Guidance for Recipients of Federal Funding Regarding Unlawful Discrimination (July 29, 2025). Any person or entity that believes that they have been subjected to discrimination in violation of Federal civil rights/antidiscrimination laws can file a complaint with HHS' Office for Civil Rights; information can be located at https://www.hhs.gov/ocr/complaints/index.html.
As we gain more experience with this policy, we may revisit this issue in future rulemaking if we determine that ( printed page 54027) it is necessary to further refine the regulations to prevent gaming.
Comment: Several commenters opposed our proposed change to the definition of “approved medical residency programs”. Commenters expressed concern that the proposal would create uncertainty for hospitals, disrupt residency training, and potentially exacerbate physician shortages in various specialties. A few commenters emphasized the importance of a diverse physician workforce in achieving positive health outcomes, addressing upstream drivers of health (such as housing and food insecurity), and reducing health disparities, with a commenter adding that the shift away from training informed by diverse American experiences risks eroding trust in healthcare institutions, especially among underserved groups. Another commenter cited studies that they believe demonstrate the continuing adverse effects of implicit bias and systemic racism on public health, as well as the positive impacts that increased diversity and inclusion have on innovation, research, and patient outcomes.
A commenter stated that the proposed policy is unnecessary in light of developments within the GME community that have taken place since the publication of the CY 2026 OPPS/ASC proposed rule. The commenter reported that on September 5, 2025, the ACGME announced that, in response to Federal directives related to diversity, equity and inclusion, it was removing its DEI-related accreditation requirements and closing its Department of DEI. The commenter added that they are not aware of any other GME accrediting organization that includes DEI-related standards within its requirements.
Another commenter requested that, if the proposed policy were to be finalized, CMS should explicitly specify what sort of activities remain permissible for accreditors under the regulations, and whether these include activities such as holistic applicant review, targeted mentorship and coaching, curricula on language access, cultural humility, upstream drivers of health, and outreach that does not rely on impermissible classifications.
Response: We respectfully disagree with the commenters' objections. As we stated in the CY 2026 OPPS/ASC proposed rule, we believe that race-conscious elements of diversity, equity and inclusion policies such as those historically required by the ACGME are generally impermissible under Federal law, as strongly suggested by the Supreme Court's ruling in Students for Fair Admissions v. President and Fellows of Harvard College. In addition, we are unpersuaded by commenters' arguments that diversity, equity and inclusion programs are necessary for achieving positive health outcomes or maintaining trust in healthcare institutions: rather, as we stated in the CY 2026 OPPS/ASC proposed rule, we believe that patients and the larger society have a compelling need for medical education to be focused primarily on excellence and delivering the best possible care to patients.
While our understanding is that the ACGME has removed its DEI-related accreditation standards, we do not believe this renders the proposed policy unnecessary, since the ACGME or another accrediting body might seek to reinstate such requirements in the future. Regarding what activities would remain permissible for accreditors under the proposal, the regulatory text that we are finalizing in this final rule with comment period states that an accrediting organization must not use accreditation criteria that promote or encourage discrimination on the basis of race, color, national origin, sex, age, disability, or religion, including the use of those characteristics or intentional proxies for those characteristics as a selection criterion for employment, program participation, resource allocation, or similar activities, opportunities, or benefits. We emphasize that accrediting bodies should review their requirements to ensure that any activities such as those mentioned by the commenter do not serve as a proxy for unlawful discrimination based on race or other characteristics.
Comment: Several commenters objected to our statement in the CY 2026 OPPS/ASC proposed rule that the Secretary may recognize other organizations that meet or exceed Medicare's requirements as accreditors to increase the potential for competition in the accreditation space and improve the quality of the accreditation process. Commenters expressed concern that the recognition of new accrediting bodies with divergent standards would create uncertainty for hospitals, undermine the rigor of the accreditation process, and jeopardize patient care by diminishing the quality of residency training. A commenter stated that if CMS recognizes additional accreditors, such organizations should be required to: (1) Publish transparent standards and conflict-of-interest safeguards; (2) Demonstrate parity or improvement in patient-safety and competency expectations compared to existing accrediting bodies; and (3) Ensure grandfathering protections so that residents currently in training are not disrupted.
Response: We disagree that the recognition of additional accreditors for graduate medical education programs would lead to the circumstances described by commenters. For instance, we have not observed that the existence of multiple accreditors for various specialties of nursing and allied health education programs has undermined the quality of training or patient care in those specialties. Any new GME accreditors recognized by the Secretary would be subject to the same requirements as current accreditors, including the requirements we are adopting in this final rule with comment period.
After consideration of public comments, we are finalizing, with modification, our proposed change to the definition of “approved medical residency program” and equivalent terms at §§ 412.105(f)(1)(i), 413.75(b), and 415.152, to specify that accrediting organizations must not use accreditation criteria that promote or encourage discrimination on the basis of race, color, national origin, sex, age, disability, or religion, including the use of those characteristics or intentional proxies for those characteristics as a selection criterion for employment, program participation, resource allocation, or similar activities, opportunities, or benefits.
We also note that, in line with the Administration's commitment to preventing and reducing chronic disease through improved diet and public health measures, we are considering for future rulemaking how best to encourage accrediting bodies to incorporate nutrition education requirements into the accreditation standards for graduate medical education programs. These efforts would build on the Secretary's recent request to U.S. medical education organizations to submit written plans detailing the scope, timeline, standards alignment, measurable milestones, and accountability measures of their nutrition education commitments.[513]
( printed page 54028)XXII. Payment for Indirect and Direct Graduate Medical Education Costs (§§ 412.105 and 413.75 Through 413.83)
Notice of Closure of Teaching Hospital and Opportunity To Apply for Available Slots
A. Background
Section 5506 of the Patient Protection and Affordable Care Act (Pub. L. 111-148), as amended by the Health Care and Education Reconciliation Act of 2010 (Pub. L. 111-152) (collectively, “Affordable Care Act”), authorizes the Secretary to redistribute residency slots after a hospital that trained residents in an approved medical residency program closes. Section 5506 of the Affordable Care Act instructs the Secretary to establish a process by regulation that redistributes slots from teaching hospitals that close to hospitals that meet certain criteria, with priority given to certain hospitals including those located in the same Core Based Statistical Area (CBSA), in a contiguous CBSA or in the same state as the closed hospital.
Specifically, section 5506 of the Affordable Care Act amended the Act by adding subsection (vi) to section 1886(h)(4)(H) of the Act and modifying language at section 1886(d)(5)(B)(v) of the Act, to instruct the Secretary to establish a process to increase the full-time equivalent (FTE) resident caps for other hospitals based upon the FTE resident caps in teaching hospitals that closed on or after a date that is 2 years before the date of enactment (that is, March 23, 2008). In the CY 2011 Outpatient Prospective Payment System (OPPS) final rule with comment period (75 FR 72264), we established regulations at 42 CFR 413.79(o) and an application process for qualifying hospitals to apply to CMS to receive direct GME and IME FTE resident cap slots from the hospital that closed. We made certain additional modifications to § 413.79 in the FY 2013 IPPS/LTCH PPS final rule (77 FR 53434), and we made changes to the section 5506 application process in the FY 2015 IPPS/LTCH PPS final rule (79 FR 50122 through 50134). The procedures we established apply both to teaching hospitals that closed on or after March 23, 2008, and on or before August 3, 2010, and to teaching hospitals that close after August 3, 2010 (75 FR 72215).
B. Notice of Closure of Pontiac General Hospital Located in Pontiac, MI, and the Application Process—Round 26
We have learned of the closure of Pontiac General Hospital, located in Pontiac, MI (CCN 230013). Accordingly, this notice serves to notify the public of the closure of this teaching hospital and initiate another round (“Round 26”) of the application and selection process. This round will be the 26th round (“Round 26”) of the application and selection process. Table 145 contains the identifying information and IME and direct GME FTE resident caps for the closed teaching hospital, which are part of the Round 26 application process under section 5506 of the Affordable Care Act.
C. Application Process for Available Resident Slots
The application period for hospitals to apply for slots under section 5506 of the Affordable Care Act is 90 days following notice to the public of a hospital closure (77 FR 53436). Therefore, hospitals that wish to apply for and receive slots from the previously noted hospital's FTE resident caps must submit applications using the electronic application intake system, Medicare Electronic Application Request Information System (MEARISTM ), with application submissions for Round 26 due no later than February 19, 2026. The section 5506 application can be accessed at https://mearis.cms.gov/public/home.
CMS will only accept Round 26 applications submitted via MEARISTM . Applications submitted through any other method will not be considered. Within MEARISTM , we have built in several resources to support applicants:
- Please refer to the “Resources” section for guidance regarding the application submission process athttps://mearis.cms.gov/public/resources.
- Technical support is available under “Useful Links” at the bottom of the MEARISTM web page.
- Application related questions can be submitted to CMS using the form available under “Contact” athttps://mearis.cms.gov/public/resources.
Application submission through MEARISTM will not only help CMS track applications and streamline the review process, but it will also create efficiencies for applicants when ( printed page 54029) compared to a paper submission process.
We have not established a deadline by when CMS will issue the final determinations to hospitals that receive slots under section 5506 of the Affordable Care Act. However, we review all applications received by the application deadline and notify applicants of our determinations as soon as possible.
We refer readers to the CMS Direct Graduate Medical Education (DGME) website at https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/direct-graduate-medical-education-dgme. Hospitals should access this website for a list of additional section 5506 guidelines for the policy and procedures for applying for slots, and the redistribution of the slots under sections 1886(h)(4)(H)(vi) and 1886(d)(5)(B)(v) of the Act.
XXIII. Collection of Information Requirements
Under the Paperwork Reduction Act of 1995 (PRA), 44 U.S.C. 3501-3520, we are required to provide notice in the Federal Register and solicit public comment before a collection of information requirement is submitted to the Office of Management and Budget (OMB) for review and approval. To fairly evaluate whether an information collection should be approved by OMB, 44 U.S.C. 3506(c)(2)(A) requires that we solicit comment on the following issues:
- The need for the information collection and its usefulness in carrying out the proper functions of our agency.
- The accuracy of our estimate of the information collection burden.
- The quality, utility, and clarity of the information to be collected.
- Recommendations to minimize the information collection burden on the affected public, including automated collection techniques.
We solicited public comment on each of these issues for the following sections of this document that contain information collection requirements (ICRs):
A. ICRs for the Hospital Outpatient Quality Reporting (OQR) Program
1. Background
In sections XIV. and XV. of this final rule with comment period, we discuss the finalized requirements for the Hospital OQR Program. The Hospital OQR Program is generally aligned with the CMS quality reporting program for hospital inpatient services known as the Hospital Inpatient Quality Reporting (IQR) Program. We refer readers to the CY 2025 OPPS/ASC final rule with comment period (89 FR 94522 through 94530) for detailed discussions of the previously finalized Hospital OQR Program ICRs which are currently approved under OMB control number 0938-1109 (expiration date January 31, 2026).
We are: (1) removing the COVID-19 Vaccination Coverage Among Healthcare Personnel (HCP) measure beginning with the CY 2024 reporting period/CY 2026 payment determination; (2) removing the Hospital Commitment to Health Equity (HCHE) measure beginning with the CY 2025 reporting period/CY 2027 payment determination; (3) removing the Screening for Social Drivers of Health (SDOH) measure beginning with the CY 2025 reporting period; (4) removing the Screen Positive Rate for SDOH measure beginning with the CY 2025 reporting period; (5) modifying the Excessive Radiation Dose or Inadequate Image Quality for Diagnostic Computed Tomography (CT) in Adults (Hospital Level—Outpatient) eCQM (Excessive Radiation eCQM) from mandatory reporting beginning with the CY 2027 reporting period to continue voluntary reporting in the CY 2027 reporting period and subsequent years; (6) adopting the Emergency Care Access & Timeliness electronic clinical quality measure (eCQM) with voluntary reporting for the CY 2027 reporting period, followed by mandatory reporting beginning with the CY 2028 reporting period/CY 2030 payment determination; (7) removing the Median Time from Emergency Department (ED) Arrival to ED Departure for Discharged ED Patients measure beginning with the CY 2028 reporting period/CY 2030 payment determination; and (8) removing the Left Without Being Seen (LWBS) measure beginning with the CY 2028 reporting period/CY 2030 payment determination.
In section XIV.D. of this final rule with comment period, we are also updating our Extraordinary Circumstances Exception (ECE) Policy for the Hospital OQR Program. This update will explicitly include extensions as a type of extraordinary circumstances relief option, in addition to exceptions. Because the process for requesting or granting an ECE will remain the same as the current ECE process, these updates will not affect burden associated with the submission of the ECE form.
In the CY 2025 OPPS/ASC final rule with comment period, we calculated reporting burden estimates for the Hospital OQR Program by utilizing the Bureau of Labor Statistics (BLS) mean hourly wage rate for Medical Records Specialists (89 FR 94522 through 94523). Specifically, we used the industry-specific wage for Medical Records Specialists working in “general medical and surgical hospitals”, as this categorization aligns the closest with the Hospital OQR Program care setting. The most recent data from BLS' May 2024 National Occupational Employment and Wage Estimates reflects a median hourly wage of $27.53 per hour for Medical Records Specialists working in “general medical and surgical hospitals” (SOC 29-2072).[514] We calculated the cost of overhead, including fringe benefits, at 100 percent of the median hourly wage, consistent with previous years. This is a rough adjustment, both because fringe benefits and overhead costs vary significantly by employer and methods of estimating these costs vary widely in the literature. Nonetheless, we believe that doubling the hourly wage rate ($27.53 × 2 = $55.06) to estimate total cost burden is reasonably accurate. Accordingly, unless otherwise specified, we calculate cost burden to hospitals using a wage plus benefits estimate of $55.06 per hour throughout the discussion in this section of this final rule with comment period for the Hospital OQR Program.
In the CY 2025 OPPS/ASC final rule with comment period, our burden estimates assumed that approximately 3,200 hospital outpatient departments (HOPDs) will report data to the Hospital OQR Program (89 FR 94523). For this final rule with comment period, based on the most recent available data from the CY 2024 Hospital OQR Program payment determination, we estimate that 3,200 HOPDs will report data to the Hospital OQR Program for the CY 2026 reporting period/CY 2028 payment determination and future years.
2. Information Collection Burden Estimate for the Removal of the COVID-19 Vaccination Coverage Among HCP Measure Beginning With CY 2024 Reporting Period/CY 2026 Payment Determination
As discussed in section XIV.C.1. of this final rule with comment period, we are removing the COVID-19 Vaccination Coverage Among HCP measure beginning with the CY 2024 reporting period/CY 2026 payment determination. The information collection burden associated with this measure is currently approved under OMB control number 0920-1317. To report this measure, HOPDs have the option to ( printed page 54030) manually enter data directly into the Centers for Disease Control and Prevention (CDC) National Healthcare Safety Network (NHSN) web-based application or by uploading a CSV file. CDC estimates that each HOPD requires between 40 minutes (0.67 hours) to upload a CSV file and 45 minutes (0.75 hours) monthly to enter the data manually. CDC assumes that manual data entry will be completed by a Microbiologist with a wage rate of $58.60/hour and uploading of a CSV file will be completed by an Information Technologist with a wage rate of $56.50/hour. Therefore, we estimate that this policy will result in a decrease in burden of between 25,600 hours (0.67 hours × 12 months × 3,200 HOPDs) at a savings of $1,446,400 (25,600 hours × $56.50/hour) and 28,800 hours (0.75 hours × 12 months × 3,200 HOPDs) at a savings of $1,687,680 (28,800 hours × $58.60/hour) annually across all 3,200 HOPDs under OMB control number 0920-1317.
We did not receive public comments on this burden estimate.
3. Information Collection Burden Estimate for the Removal of the HCHE Measure Beginning With the CY 2025 Reporting Period/CY 2027 Payment Determination
As discussed in section XIV.C.2. of this final rule with comment period, we are removing the HCHE measure beginning with the CY 2025 reporting period/CY 2027 payment determination. The information collection burden associated with this measure is currently approved under OMB control number 0938-1109. The currently approved information collection burden estimate for this measure assumes HOPDs spend approximately 10 minutes (0.167 hours) annually to report measure data. Therefore, for all participating HOPDs, we estimate removal of this measure will decrease burden by approximately 533 hours (0.167 hours × 3,200 HOPDs) at a savings of $29,347 (533 hours × $55.06/hour).
We did not receive public comments on this burden estimate.
4. Information Collection Burden Estimate for the Removal of the Screening for SDOH Measure Beginning With the CY 2025 Reporting Period
In section XIV.C.3. of this final rule with comment period, we are removing the Screening for SDOH measure beginning with the CY 2025 reporting period. There are two components to this measure: patient screening for five health related social needs domains and hospital submission of aggregated hospital-level measure data. We have previously estimated each patient requires 2 minutes (0.033 hours) to complete the screening and each hospital requires 10 minutes (0.167 hours) annually to report this measure.
We determine the cost for patients (or their representative) to complete the screening using a post-tax wage of $25.63/hour based on assumptions from the report “Valuing Time in U.S. Department of Health and Human Services Regulatory Impact Analyses: Conceptual Framework and Best Practices”, which identifies an approach for valuing time when individuals undertake administrative and other tasks on their own time.[515] To derive the costs for patients (or their representatives), a measurement of the usual weekly earnings of wage and salary workers of $1,192 is divided by 40 hours to calculate an hourly pre-tax wage rate of $29.80/hour.[516] This rate is adjusted downwards by an estimate of the effective tax rate for median income households of about 14 percent calculated by comparing pre- and post-tax income,[517] resulting in the post-tax hourly wage rate of $25.63/hour. Unlike our State and private sector wage adjustments, we are not adjusting beneficiary wages for fringe benefits and other indirect costs because the individuals' activities, if any, will occur outside the scope of their employment.
Under OMB control number 0938-1109, we estimated 206,325,645 HOPD visits annually that would have resulted in screening once the measure became mandatory. Therefore, for all participating HOPDs, we estimate removal of this measure will decrease burden for voluntary reporting for the CY 2025 reporting period by approximately 1,719,380 hours for 51,581,411 patients (0.033 hours × 206,325,645 patients × 50 percent response rate × 50 percent of HOPDs) at a savings of $44,067,709 (1,719,380 hours × $25.63/hour). For mandatory reporting beginning with the CY 2026 reporting period, we estimate a decrease in burden of 6,877,522 hours (206,325,645 patients × 0.033 hours per patient) at a savings of $176,270,889 (6,877,522 hours × $25.63/hour). With regard to measure reporting, we estimate a decrease in burden of 267 hours (3,200 HOPDs × 50 percent of HOPDs × 0.167 hours per HOPD) at a savings of $14,701 (267 hours × $55.06/hour) for voluntary reporting for the CY 2025 reporting period and 533 hours annually (0.167 hours × 3,200 HOPDs) at a savings of $29,347 (533 hours × $55.06/hour) for mandatory reporting beginning with the CY 2026 reporting period.
We did not receive public comments on this burden estimate.
5. Information Collection Burden Estimate for the Removal of the Screen Positive Rate for SDOH Measure Beginning With the CY 2025 Reporting Period
In section XIV.C.3. of this final rule with comment period, we are removing the Screen Positive Rate for SDOH measure beginning with the CY 2025 reporting period. For this measure, HOPDs are required to report on an annual basis the number of patients who screen positive for one or more of the five SDOH domains divided by the total number of patients screened (reported as five separate rates). We previously estimated each HOPD requires 10 minutes (0.167 hours) annually to report this measure. Therefore, we estimate removal of this measure will decrease burden by 267 hours (3,200 HOPDs × 50 percent of HOPDs × 0.167 hours per HOPD) at a savings of $14,701 (267 hours × $55.06/hour) for voluntary reporting for the CY 2025 reporting period and 533 hours annually (0.167 hours × 3,200 HOPDs) at a savings of $29,347 (533 hours × $55.06/hour) for mandatory reporting beginning with the CY 2026 reporting period.
We did not receive public comments on this burden estimate.
6. Information Collection Burden Estimate for the Adoption of the Emergency Care Access & Timeliness eCQM With Voluntary Reporting for the CY 2027 Reporting Period, Followed by Mandatory Reporting Beginning With the CY 2028 Reporting Period/CY 2030 Payment Determination
As discussed in section XV.B.1. of this final rule with comment period, we are adopting the Emergency Care Access & Timeliness eCQM beginning with voluntary reporting for the CY 2027 reporting period, followed by mandatory reporting beginning with the CY 2028 reporting period/CY 2030 payment determination. Similar to the information collection burden for the Appropriate Treatment for ST-Segment ( printed page 54031) Elevation Myocardial Infarction (STEMI) and Excessive Radiation Dose or Inadequate Image Quality for Diagnostic CT in Adults eCQMs currently approved under OMB control number 0938-1109, we assume a Medical Records Specialist will require 10 minutes (0.167 hours) to submit the data required per quarter for each HOPD or 40 minutes (0.67 hours; 10 minutes × 4 quarters) annually. For voluntary reporting for the CY 2027 reporting period, HOPDs will be able to voluntarily submit at least one quarter and up to four quarters of data. For estimation purposes, similar to the assumptions previously used for the STEMI and Excessive Radiation Dose or Inadequate Image Quality for Diagnostic CT in Adults eCQMs, we estimate 20 percent of HOPDs will voluntarily report one quarter of data for the measure in the CY 2027 reporting period, with 100 percent of HOPDs reporting the measure as required in subsequent years (86 FR 63962 and 63963, and 88 FR 82134). For voluntary reporting for the CY 2027 reporting period, we estimate an annual burden for voluntarily participating HOPDs of 107 hours (3,200 HOPDs × 20 percent × 0.167 hours × 1 quarter) at a cost of $5,891 (107 hours × $55.06/hour). Beginning with the CY 2028 reporting period, we estimate the annual burden for all participating HOPDs to be 2,133 hours (0.67 hours × 3,200 HOPDs) at a cost of $117,443 (2,133 hours × $55.06/hour). With respect to any costs/burdens unrelated to data submission, we refer readers to the Regulatory Impact Analysis in section XXVI. of this final rule with comment period.
We received public comments on these burden estimates. The following is a summary of the comments we received and our responses.
Comment: A commenter asserted that the burden estimate associated with the Emergency Care Access & Timeliness eCQM, and eCQMs in general, includes only the burden associated with the submission of data and does not include the cost or effort associated with other activities such as: education to providers and staff on the impact of the measure on their workflow; modifications to documentation as appropriate; evaluation of measure report details against EHR documentation, especially on fallouts and exclusions to identify opportunities for improved discrete documentation; time needed for review of ONC Project Tracking for ongoing issues identified with the eCQM measures; development of trending and benchmark reports for awareness of reported outcomes prior to submission of data; reprocessing of data when errors have been identified and corrected; and meetings with vendors and IT, quality, leadership, and other staff to assure an understanding of what is reported on Medicare.gov/Care Compare, its impact on public reporting, star reporting, and use by other reporting agencies.
Response: Because we assume the collection of data for eCQMs is already being collected in each HOPD's electronic health record (EHR) system as part of the HOPD's patient workflow, the burden estimates provided in this final rule with comment period include only the time associated with submission of data to CMS. However, as noted in the Regulatory Impact Analysis in section XXVI.C.3.b. of this final rule with comment period, we agree with the commenter that there are additional recurring and non-recurring activities associated with the adoption of new eCQM measures.
After consideration of public comments, we are declining to update our burden estimate.
7. Information Collection Burden Estimate for the Removal of the Median Time From ED Arrival to ED Departure for Discharged ED Patients Measure Beginning With the CY 2028 Reporting Period/CY 2030 Payment Determination
As discussed in section XV.B.2. of this final rule with comment period, we are removing the Median Time from ED Arrival to ED Departure for Discharged ED Patients measure beginning with the CY 2028 reporting period/CY 2030 payment determination, when reporting for the Emergency Care Access & Timeliness eCQM becomes mandatory. The information collection burden associated with this measure is currently approved under OMB control number 0938-1109. The currently approved information collection burden estimate for this measure assumes an average of 289 cases are reported annually per HOPD, and HOPDs require approximately 2.9 minutes (0.049 hours) per case to perform the necessary chart abstraction and report measure data. Therefore, we estimate removal of this measure will decrease burden by approximately 14.2 hours (0.049 hours × 289 cases) at a savings of $782 per HOPD (14.2 hours × $55.06/hour). Therefore, for all participating HOPDs, we estimate a decrease in annual burden of 45,440 hours (14.2 hours per HOPD × 3,200 HOPDs) at a savings of $2,501,926 (45,440 hours × $55.06/hour).
We did not receive public comments on this burden estimate.
8. Information Collection Burden Estimate for the Removal of the Left Without Being Seen Measure Beginning With the CY 2028 Reporting Period/CY 2030 Payment Determination
As discussed in section XV.B.2. of this final rule with comment period, we are removing the Left Without Being Seen measure beginning with the CY 2028 reporting period/CY 2030 payment determination, when reporting for the Emergency Care Access & Timeliness eCQM becomes mandatory. The information collection burden associated with this measure is currently approved under OMB control number 0938-1109. The currently approved information collection burden estimate for this measure assumes HOPDs spend approximately 10 minutes (0.167 hours) annually to report measure data. Therefore, for all participating HOPDs, we estimate removal of this measure will decrease burden by approximately 533 hours (0.167 hours × 3,200 HOPDs) at a savings of $29,347 (533 hours × $55.06/hour).
We did not receive public comments on this burden estimate.
9. Information Collection Burden Estimate To Modify the Excessive Radiation eCQM From Mandatory Reporting Beginning With the CY 2027 Reporting Period To Continue Voluntary Reporting in the CY 2027 Reporting Period and Subsequent Years
As discussed in section XV.B.3. of this final rule with comment period, we are modifying the reporting requirements for the Excessive Radiation eCQM by maintaining voluntary reporting instead of mandatory reporting of the measure, beginning with the CY 2027 reporting period. The information collection burden associated with this measure is currently approved under OMB control number 0938-1109 and estimates HOPDs spend approximately 10 minutes (0.167 hours) per quarter annually to report measure data. In the CY 2024 OPPS/ASC final rule with comment period, where we adopted the Excessive Radiation eCQM beginning with voluntary reporting in the CY 2026 reporting period, we estimated that 20 percent of hospitals will voluntarily report one quarter of data for the measure and 100 percent of hospitals would report data for the measure once mandatory reporting began with the CY 2027 reporting period/CY 2029 payment determination. We also finalized to gradually increase the number of quarters of data hospitals would be required to report on the measure ( printed page 54032) starting with two self-selected quarters for the CY 2027 reporting period/CY 2029 payment determination, and all four quarters for the CY 2028 reporting period/CY 2030 payment determination (88 FR 82134).
Because HOPDs will no longer be required to report this measure under the modification to extend voluntary reporting beginning with the CY 2027 reporting period, we estimate the revised data submission burden, reflecting 20 percent of hospitals voluntarily reporting, will be 107 hours (3,200 HOPDs × 20 percent × 0.167 hours × 1 quarter) at a cost of $5,891 (107 hours × $55.06/hour) annually. This updated burden estimate is a decrease of 960 hours [(3,200 HOPDs × 0.167 hours × 2 quarters)−(3,200 HOPDs × 20 percent × 0.167 hours × 1 quarter)] at a savings of $52,858 (960 hours × $55.06) for the CY 2027 reporting period, and a decrease of 2,027 hours [(3,200 HOPDs × 20 percent × 0.167 hours × 3 quarters) + (3,200 HOPDs × 80 percent × 0.167 hours × 4 quarters)] at a savings of $111,607 (2,027 hours × $55.06/hour) annually beginning with the CY 2028 reporting period.
In addition, for this measure as described under OMB control number 0938-1109, participating HOPDs must follow the process for running their chosen vendor's translation software prior to sending data to its EHR for measure calculation and reporting. We estimate participating HOPDs spend approximately 15 minutes (0.25 hours) annually to conduct these activities prior to data submission. Therefore, for all participating HOPDs, we estimate modification of this measure from mandatory to voluntary reporting will decrease annual burden associated with these activities by approximately 640 hours (0.25 hours × 80 percent × 3,200 HOPDs) at a savings of $35,238 (640 hours × $55.06/hour) beginning with the CY 2027 reporting period.
We received public comments on these burden estimates. The following is a summary of the comments we received and our responses.
Comment: A commenter stated their opinion that the burden estimate associated with the Excessive Radiation eCQM includes only the burden associated with the submission of data and does not include the cost or effort associated with other activities such as: contracted physicist time for scanner reconfiguration and validation; health information system, radiology information system, and EHR modifications to create separate low-, routine-, and high-dose orderable exams; radiologist and technologist labor for workflow redesign and training; IT integration across picture archiving and communication systems, dose monitoring systems, and reporting vendors; and scanner manufacturer support to ensure redesigned protocols remain consistent with each scanner's technology and capability. The commenter further noted that first-year costs associated with these activities range between $20,000 and $50,000 with additional annual expenses incurred thereafter.
Response: Reporting on this measure will continue to be voluntary, and as such HOPDs can determine their readiness to implement any changes necessary if they elect to report on this measure. Because we assume the collection of data for eCQMs is already being collected in each HOPD's EHR system as part of the HOPD's patient workflow, the burden estimates provided in this final rule with comment period include only the time associated with submission of data to CMS. However, as noted in the Regulatory Impact Analysis in section XXVI.C.3.b. of this final rule with comment period, we agree with the commenter that there are additional recurring and non-recurring activities associated with adoption of new eCQM measures.
After consideration of public comments, we are declining to update our burden estimate.
10. Summary of Information Collection Burden Estimates for the Hospital OQR Program
Tables 146 through 150 summarizes the information collection burden changes under OMB control number 0938-1109 (expiration date January 1, 2026). We estimate that removal of the COVID-19 Vaccination Coverage Among HCP measure in this final rule with comment period will result in a decrease in information collection burden of between 25,600 hours at a savings of $1,446,400 and 28,800 hours at a savings of $1,687,680 under OMB control number 0920-1317. We also estimate that the remaining measure removals, adoptions, and modifications in this final rule with comment period will result in a decrease in information collection burden of 6,924,988 hours at a savings of $178,884,367 annually for all 3,200 program-eligible HOPDs from the CY 2025 reporting period/CY 2027 payment determination through the CY 2028 reporting period/CY 2030 payment determination. We submitted the revised information collection estimates to OMB for approval under OMB control number 0938-1109. With respect to any costs/burdens unrelated to data submission, we refer readers to the Regulatory Impact Analysis in section XXVI.C.3 of this final rule with comment period.
We invited public comments on the information collection requirements and whether our estimated burden is a reasonable estimate but did not receive any comments other than those previously discussed.
( printed page 54033) ( printed page 54034) ( printed page 54035) ( printed page 54036) ( printed page 54037)B. ICRs for the Rural Emergency Hospital Quality Reporting (REHQR) Program
1. Background
In sections XIV. and XVI. of this final rule with comment period, we discuss the finalized changes to requirements for the REHQR Program. The REHQR Program is generally aligned with the CMS quality reporting program for HOPDs, known as the Hospital OQR Program. We refer readers to the CY 2025 OPPS/ASC final rule with comment period (89 FR 94530 through 94533) for detailed discussions of the previously finalized REHQR Program ICRs, which have been submitted for OMB approval under OMB control number 0938-1454 (expiration date April 30, 2027).
We are: (1) removing the HCHE measure beginning with the CY 2025 reporting period/CY 2027 program determination; (2) removing the Screening for SDOH measure beginning with the CY 2025 reporting period; (3) removing the Screen Positive Rate for SDOH measure beginning with the CY 2025 reporting period; and (4) adopting the Emergency Care Access & Timeliness eCQM beginning with the CY 2027 reporting period/CY 2029 program determination as an optional measure. In section XIV.D. of this final rule with comment period, we are also updating our Extraordinary Circumstances Exception (ECE) Policy for the REHQR Program. This update will explicitly include extensions as a type of extraordinary circumstances relief option, in addition to exceptions. Because the process for requesting or granting an ECE will remain the same as the current ECE process, these updates will not affect burden associated with the submission of the ECE form.
In the CY 2025 OPPS/ASC final rule with comment period, we calculated reporting burden estimates for the REHQR Program by utilizing the BLS mean hourly wage rate for Medical Records Specialists (89 FR 94530). Specifically, we used the industry-specific wage for Medical Records Specialists working in “general medical and surgical hospitals,” as this categorization aligns the closest with the REHQR Program care setting. The most recent data from BLS' May 2024 National Occupational Employment and Wage Estimates reflects a median hourly wage of $27.53 per hour for Medical Records Specialists working in “general medical and surgical hospitals” (SOC 29-2072).[518] We calculated the cost of overhead, including fringe benefits, at 100 percent of the median hourly wage, consistent with previous years. This is necessarily a rough adjustment, both because fringe benefits and overhead costs vary significantly by employer and methods of estimating these costs vary widely in the literature. Nonetheless, we believe that doubling the hourly wage rate ($27.53 × 2 = $55.06) to estimate total cost is a reasonably accurate estimation method. Accordingly, unless otherwise specified, we will calculate cost burden to REHs using a wage plus benefits estimate of $55.06 per hour throughout the discussion in this section of this rule for the REHQR Program.
In the CY 2025 OPPS/ASC final rule with comment period, our burden estimates were based on the 33 acute care and critical access hospital conversions to REH status as of September 27, 2024 (89 FR 94530). For this final rule with comment period, based on the actual number of acute care and critical access hospital conversions to REH status as of April 11, 2025, we estimate that 38 REHs will report data to the REHQR Program during the CY 2026 reporting period unless otherwise noted. While the exact number of REHs required to submit data may vary due to status changes to and from an REH, as reiterated in section XVI. of this final rule with comment period, REHs are required by statute to submit quality data. Therefore, for purposes of estimating burden, we assume that all 38 REHs will submit data under the REHQR Program for the CY 2026 reporting period and future years.
2. Information Collection Burden Estimate for the Removal of the HCHE Measure Beginning With the CY 2025 Reporting Period/CY 2027 Program Determination
As discussed in section XIV.C.2. of this final rule with comment period, we are removing the HCHE measure beginning with the CY 2025 reporting ( printed page 54038) period/CY 2027 program determination. The information collection burden associated with this measure is currently approved under OMB control number 0938-1454. The currently approved information collection burden estimate for this measure assumes REHs spend approximately 10 minutes (0.167 hours) annually to report measure data. Therefore, for all participating REHs, we estimate removal of this measure will decrease burden by approximately 6 hours (0.167 hours x 38 REHs) at a savings of $349 (6 hours x $55.06/hour).
We did not receive public comments on this burden estimate.
3. Information Collection Burden Estimate for the Removal of the Screening for SDOH Measure Beginning With the CY 2025 Reporting Period
In section XIV.C.3. of this final rule with comment period, we are removing the Screening for SDOH measure beginning with the CY 2025 reporting period. There are two components to this measure: patient screening for five health related social needs domains and hospital submission of aggregated hospital-level measure data. We have previously estimated each patient requires 2 minutes (0.033 hours) to complete the screening and each hospital requires 10 minutes (0.167 hours) annually to report this measure.
We determine the cost for patients (or their representative) to complete the screening using a post-tax wage of $25.63/hour based on assumptions from the report “Valuing Time in U.S. Department of Health and Human Services Regulatory Impact Analyses: Conceptual Framework and Best Practices,” which identifies an approach for valuing time when individuals undertake administrative and other tasks on their own time.[519] To derive the costs for patients (or their representatives), a measurement of the usual weekly earnings of wage and salary workers of $1,192 is divided by 40 hours to calculate an hourly pre-tax wage rate of $29.80/hour.[520] This rate is adjusted downwards by an estimate of the effective tax rate for median income households of about 14 percent calculated by comparing pre- and post-tax income,[521] resulting in the post-tax hourly wage rate of $25.63/hour. Unlike our State and private sector wage adjustments, we are not adjusting beneficiary wages for fringe benefits and other indirect costs because the individuals' activities, if any, will occur outside the scope of their employment.
Under OMB control number 0938-1454, we estimated 11,798 patients annually would have been screened per REH when reporting on the measure became mandatory. For voluntary reporting in the CY 2025 reporting period, we estimate that 50 percent of REHs will survey 50 percent of patients. Therefore, for all participating REHs with regard to patient screening, we estimate removal of this measure will decrease burden for voluntary reporting for the CY 2025 reporting period by 3,699 hours for 112,081 patients (0.033 hours × 11,798 patients × 50 percent response rate × 19 REHs) at a savings of $94,797 (3,699 hours × $25.63/hour). For mandatory reporting beginning with the CY 2026 reporting period, we estimate a decrease in burden of 14,795 hours (448,324 patients × 0.033 hours per patient) at a savings of $379,188 (14,795 hours × $25.63/hour). With regard to measure reporting, we estimate a decrease in burden of 3 hours (38 REHs × 50 percent of REHs × 0.167 hours per REH) at a savings of $175 (3 hours × $55.06/hour) for voluntary reporting for the CY 2025 reporting period and 6 hours annually (38 REHs × 0.167 hours) at a savings of $349 (6 hours × $55.06/hour) for mandatory reporting beginning with the CY 2026 reporting period.
We did not receive public comments on this burden estimate.
4. Information Collection Burden Estimate for the Removal of the Screen Positive Rate for SDOH Measure Beginning With the CY 2025 Reporting Period
In section XIV.C.3. of this final rule with comment period, we are removing the Screen Positive Rate for SDOH measure beginning with the CY 2025 reporting period. For this measure, REHs are required to report on an annual basis the number of patients who screen positive for one or more of the five SDOH domains divided by the total number of patients screened (reported as five separate rates). We previously estimated each REH requires 10 minutes (0.167 hours) annually to report this measure. Therefore, we estimate the removal of this measure will decrease burden by 3 hours (38 REHs × 50 percent of REHs × 0.167 hours) at a savings of $175 (3 hours × $55.06/hour) for voluntary reporting for the CY 2025reporting period and 6 hours (38 REHs × 0.167 hours) at a savings of $349 (6 hours × $55.06/hour) annually for mandatory reporting beginning with the CY 2026 reporting period.
We did not receive public comments on this burden estimate.
5. Information Collection Burden Estimate for the Adoption of the Emergency Care Access & Timeliness eCQM Beginning With the CY 2027 Reporting Period/CY 2029 Program Determination
As discussed in section XVI.B.1. of this final rule with comment period, we are adopting the Emergency Care Access & Timeliness eCQM beginning with the CY 2027 reporting period/CY 2029 program determination. We refer readers to the discussion of information collection burden associated with the proposal to adopt a similar measure for the Hospital OQR Program in section XXIII.A.6. of this final rule with comment period. Because this will be the first eCQM adopted in the REHQR Program, we are also finalizing that REHs will be provided with the option of reporting either the Median Time for Discharged ED Patients measure or the Emergency Care Access & Timeliness eCQM to meet program requirements. We assume a Medical Records Specialist will require 10 minutes (0.167 hours) to submit the data required per quarter for each REH, therefore, for each REH that elects to report the Emergency Care Access & Timeliness eCQM, we estimate an annual burden of 40 minutes (0.67 hours; 10 minutes × 4 quarters) annually at a cost of $36.92 (0.67 hours × $55.06/hour). Because we are currently unable to estimate the number of REHs that will elect to report the Emergency Care Access & Timeliness eCQM instead of the Median Time for Discharged ED Patients measure, we base our estimate of total burden for the REHQR Program solely on the time to report the Median Time for Discharged ED Patients measure. For reporting the Median Time for Discharged ED Patients measure, we have previously estimated that a Medical Records Specialist would require 12.2 hours per REH annually or 464 hours (12.2 hours × 38 REHs) at a cost of $25,526 (464 hours × $55.06/hour) across all REHs.
We received public comments on this burden estimate. The following is a summary of the comments we received and our responses. ( printed page 54039)
Comment: A commenter stated their opinion that the burden estimate associated with the Emergency Care Access & Timeliness eCQM and eCQMs in general includes only the burden associated with the submission of data and does not include the cost or effort associated with other activities such as: education to impacted providers and staff on the impact of the measure on their workflow; modifications to documentation as appropriate; evaluation of measure report details against EHR documentation, especially on fallouts and exclusions to identify opportunities for improved discrete documentation; time needed for review of ONC Project Tracking for ongoing issues identified with the eCQM measures; development of trending and benchmark reports for awareness of reported outcomes prior to submission of data; reprocessing of data when errors have been identified and corrected; and meetings with vendors and IT, quality, leadership, and other staff to assure an understanding of what is reported on Medicare.gov/Care Compare, its impact on public reporting, star reporting, and use by other reporting agencies.
Response: Because we assume the collection of data for eCQMs is already being collected in each REH's EHR system as part of the REH's patient workflow, the burden estimates provided in this final rule include only the time associated with submission of data to CMS. However, as noted in the Regulatory Impact Analysis in section XXVI.C.4.b. of this final rule with comment period, we agree with the commenter that there are additional recurring and non-recurring activities associated with adoption of new eCQM measures.
After consideration of public comments, we are declining to update our burden estimate.
6. Summary of Information Collection Burden Estimates for the REHQR Program
Tables 151 through 153 summarizes the information collection burden changes for the REHQR Program. We estimate that the measure adoptions and removals in this final rule with comment period will result in a decrease of 14,813 hours at a savings of $380,235 for 38 REHs annually from the CY 2025 reporting period through the CY 2027 reporting period. We submitted these information collection estimates to OMB for approval under OMB control number 0938-1454. With respect to any costs/burdens unrelated to data submission, we refer readers to the Regulatory Impact Analysis in section XXVI.C.4. of this final rule with comment period.
We invited public comments on the information collection requirements and whether our estimated burden is a reasonable estimate but did not receive any comments other than those previously discussed.
( printed page 54040) ( printed page 54041)C. ICRs for the Ambulatory Surgical Center Quality Reporting (ASCQR) Program
1. Background
In sections XIV. and XVII. of this final rule with comment period, we discuss the finalized requirements for the ASCQR Program. We refer readers to the CY 2025 OPPS/ASC final rule with comment period (89 FR 94533 through 94537) for detail regarding the previously finalized ASCQR Program ICRs which are currently approved under OMB control number 0938-1270 (expiration date July 31, 2027).
We are: (1) removing the COVID-19 Vaccination Coverage Among HCP measure beginning with the CY 2024 reporting period/CY 2026 payment determination; (2) removing the Facility Commitment to Health Equity (FCHE) measure beginning with the CY 2025 reporting period/CY 2027 payment determination; (3) removing the Screening for SDOH measure beginning with the CY 2025 reporting period; and (4) removing the Screen Positive Rate for SDOH measure beginning with the CY 2025 reporting period. We are not finalizing the Patient Understanding of Key Information Related to Recovery After a Facility-Based Outpatient Procedure or Surgery, Patient Reported Outcome-Based Performance measure (Information Transfer PRO-PM).
In section XIV.D. of this final rule with comment period, we are also updating our Extraordinary Circumstances Exception (ECE) Policy for the ASCQR Program. This update will explicitly include extensions as a type of extraordinary circumstances relief option, in addition to exceptions. Because the process for requesting or granting an ECE will remain the same as the current ECE process, these updates will not affect burden associated with the submission of the ECE form.
In the CY 2025 OPPS/ASC final rule with comment period, we calculated reporting burden estimates for the ASCQR Program by utilizing the BLS mean hourly wage rate for Medical Records Specialists (89 FR 94534). Specifically, we used the industry-specific wage for Medical Records Specialists working in the “general medical and surgical hospitals” ( printed page 54042) industry, as this categorization aligns the closest with the ASCQR Program care setting. The most recent data from BLS' May 2024 National Occupational Employment and Wage Estimates reflects a median hourly wage of $27.53 per hour for Medical Records Specialists working in “general medical and surgical hospitals” (SOC 29-2072).[522] We calculated the cost of overhead, including fringe benefits, at 100 percent of the median hourly wage, consistent with previous years. This is necessarily a rough adjustment, both because fringe benefits and overhead costs vary significantly by employer and methods of estimating these costs vary widely in the literature. Nonetheless, doubling the hourly wage rate ($27.53 × 2 = $55.06) to estimate total cost is a reasonably accurate estimation method. Accordingly, unless otherwise specified, we will calculate cost burden to ASCs using a wage plus benefits estimate of $55.06 per hour throughout the discussion in this section of this rule for the ASCQR Program.
Based on the most recent analysis of the CY 2025 payment determination data, we found that, of the 6,012 ASCs that were actively billing Medicare, 4,271 were required to participate in the ASCQR Program. Of the 1,741 ASCs not required to participate in the program, 319 ASCs did so and met full requirements. On this basis, we estimate that 4,590 ASCs (4,271 + 319) will submit data for the ASCQR Program for the CY 2026 reporting period/CY 2028 payment determination and future years.
We received public comments on these burden estimates. The following is a summary of the comments we received and our responses.
Comment: A commenter stated their opinion that CMS' estimate of the costs associated with implementing the Information Transfer PRO-PM are too low and does not believe the utility of the measure is outweighed by the burden.
Response: We thank the commenter for its comment and note that we are not finalizing the Information Transfer PRO-PM measure.
2. Information Collection Burden Estimate for the Removal of the COVID-19 Vaccination Coverage Among HCP Measure Beginning With CY 2024 Reporting Period/CY 2026 Payment Determination
As discussed in section XIV.C.1. of this final rule with comment period, we are removing the COVID-19 Vaccination Coverage Among HCP measure beginning with the CY 2024 reporting period/CY 2026 payment determination. The information collection burden associated with this measure is currently approved under OMB control number 0920-1317.
To report this measure, ASCs have the option to manually enter data directly into CDC's NHSN web-based application or to upload a CSV file. CDC estimates that each ASC requires between 40 minutes (0.67 hours) to upload a CSV file and 45 minutes (0.75 hours) monthly to enter the data manually. CDC assumes that manual data entry will be completed by a Microbiologist with a wage rate of $58.60/hour and uploading of a CSV file would be completed by an Information Technologist with a wage rate of $56.50/hour. Therefore, we estimate that this proposal will result in a decrease in burden of between 36,720 hours (0.67 hours × 12 months × 4,590 ASCs) at a savings of $2,074,680 (36,720 hours × $56.50/hour) and 41,310 hours (0.75 hours × 12 months × 4,590 ASCs) at a savings of $2,420,766 (41,310 hours × $58.60/hour) annually across all 4,590 ASCs under OMB control number 0920-1317.
We did not receive public comments on this burden estimate.
3. Information Collection Burden Estimate for the Removal of the FCHE Measure Beginning With the CY 2025 Reporting Period/CY 2027 Payment Determination
As discussed in section XIV.C.2. of this final rule with comment period, we are removing the FCHE measure beginning with the CY 2025 reporting period/CY 2027 payment determination. The information collection burden associated with this measure is currently approved under OMB control number 0938-1270.
The currently approved information collection burden estimate for this measure assumes ASCs spend approximately 10 minutes (0.167 hours) annually to report measure data. Therefore, for all participating ASCs, we estimate removal of this measure will decrease burden by approximately 765 hours (0.167 hours × 4,590 ASCs) at a savings of $42,121 (765 hours × $55.06/hour).
We did not receive public comments on this burden estimate.
4. Information Collection Burden Estimate for the Removal of the Screening for SDOH Measure Beginning With the CY 2025 Reporting Period
In section XIV.C.3. of this final rule with comment period, we are removing the Screening for SDOH measure beginning with the CY 2025 reporting period. There are two components to this measure's burden calculation: patient screening for five health related social needs domains and ASC submission of aggregated ASC-level measure data. We previously estimated each patient requires 2 minutes (0.033 hours) to complete the screening and each ASC requires 10 minutes (0.167 hours) annually to report this measure. We determine the cost for patients (or their representative) to complete the screening using a post-tax wage of $25.63/hour based on assumptions from the report “Valuing Time in U.S. Department of Health and Human Services Regulatory Impact Analyses: Conceptual Framework and Best Practices,” which identifies the approach for valuing time when individuals undertake administrative and other tasks on their own time.[523] To derive the costs for patients (or their representatives), a measurement of the usual weekly earnings of wage and salary workers of $1,192 is divided by 40 hours to calculate an hourly pre-tax wage rate of $29.80/hour.[524] This rate is adjusted downwards by an estimate of the effective tax rate for median income households of about 14 percent calculated by comparing pre- and post-tax income,[525] resulting in the post-tax hourly wage rate of $25.63/hour. Unlike our State and private sector wage adjustments, we are not adjusting beneficiary wages for fringe benefits and other indirect costs because the individuals' activities, if any, will occur outside the scope of their employment.
Under OMB control number 0938-1270, we estimated an average of 4,765 patients per ASC annually would have been screened once the measure became mandatory. Therefore, consistent with ( printed page 54043) the burden estimates for this measure under OMB control number 0938-1270, for all participating ASCs with regard to patient screening, we estimate removal of this measure will decrease burden for voluntary reporting for the CY 2025 reporting period by approximately 182,262 hours for 5,467,838 patients (0.033 hours × 4,765 patients × 50 percent response rate × 4,590 ASCs × 50 percent of ASCs) at a savings of $4,671,375 (182,262 hours × $25.63/hour). Beginning with the mandatory reporting for the CY 2026 reporting period, we estimate a decrease in burden of approximately 729,045 hours for 21,871,350 patients (4,765 patients × 4,590 ASCs × 0.033 hours per patient) at a savings of $18,685,423 (729,045 hours × $25.63/hour). With regard to measure reporting, we estimate the removal of this measure will decrease burden for voluntary reporting for the CY 2025 reporting period by 383 hours (4,590 ASCs × 50 percent of ASCs × 0.167 hours per ASC) at a savings of $21,088 (383 hours × $55.06/hour) and 765 hours annually (0.167 hours × 4,590 ASCs) at a savings of $42,121 (765 hours × $55.06/hour) for mandatory reporting beginning with the CY 2026 reporting period.
We did not receive public comments on this burden estimate.
5. Information Collection Burden Estimate for the Removal of the Screen Positive Rate for SDOH Measure Beginning With the CY 2025 Reporting Period
In section XIV.C.3. of this final rule with comment period, we are removing the Screen Positive Rate for SDOH measure beginning with the CY 2025 reporting period. For this measure, ASCs are required to report on an annual basis the number of patients who screen positive for one or more of the five SDOH domains divided by the total number of patients screened (reported as five separate rates). We previously estimated each ASC requires 10 minutes (0.167 hours) annually to report this measure. Therefore, consistent with the burden estimates for this measure under OMB control number 0938-1270, we estimate the removal of this measure will decrease burden for voluntary reporting for the CY 2025 reporting period by 383 hours (4,590 ASCs × 50 percent of ASCs × 0.167 hours per ASC) at a savings of $21,088 (383 hours × $55.06/hour) and 765 hours annually (0.167 hours × 4,590 ASCs) at a savings of $42,121 (765 hours × $55.06/hour) for mandatory reporting beginning with the CY 2026 reporting period.
We did not receive public comments on this burden estimate.
6. Summary of Information Collection Burden Estimates for the ASCQR Program
Tables 154 through 158 summarizes the information collection burden changes for OMB control number 0938-1270 (expiration date July 31, 2027). We estimate that the removal of the COVID-19 Vaccination Coverage Among HCP Measure in this final rule with comment period will result in a decrease in information collection burden of between 36,720 hours at a savings of $2,074,680 and 41,310 hours at a savings of $2,420,766 under OMB control number 0920-1317. We also estimate that the remaining measure removals and adoptions in this final rule with comment period will result in an overall decrease in information collection burden of 731,340 hours at a savings of $18,811,786 annually for all 4,590 program-eligible ASCs from the CY 2025 reporting period/CY 2027 payment determination through the CY 2029 reporting period/CY 2031 payment determination. We submitted the revised information collection estimates to OMB for approval under OMB control number 0938-1270. With respect to any costs/burdens unrelated to data submission, we refer readers to the Regulatory Impact Analysis in section XXVI.C.5. of this final rule with comment period.
We invited public comments on the information collection requirements and whether our estimated burden is a reasonable estimate but did not receive any comments other than those previously discussed.
( printed page 54044) ( printed page 54045) ( printed page 54046)D. Summary of Information Collection Burden Estimates for the Overall Hospital Quality Star Rating
The Overall Hospital Quality Star Rating uses measures that are publicly reported on Hospital Compare or its successor websites under the public reporting authority of each individual hospital program furnishing measure data. The burden associated with measures included in the Overall Hospital Quality Star Rating, including requesting withholding of measures from public reporting, is already captured in the respective hospital programs' ICRs and represents no increased information collection burden to hospitals.
Therefore, as the Overall Hospital Quality Star Rating utilizes output data from CMS hospital quality and payment programs, there is no additional information collection burden. The burden is accounted for under OMB control numbers 0938-1109, 0938-1022, 0938-1352, 0920-0666, 0938-0981, 0938-1240 and 0938-1197.
E. ICRs for Payer-Specific Negotiated Charges Data Collection
Section XX. of this final rule with comment period discusses the collection of market-based payment rate information by MS-DRG on the Medicare cost report for cost reporting periods ending on or after January 1, 2026. Hospitals would report the median payer-specific negotiated charge by MS-DRG for payers that are Medicare Advantage Organization (MAOs). We proposed to collect this market-based information on new worksheet Supplemental to Form CMS-2552-10, Weighted Median MAO Payer-Specific Negotiated Charge Data Worksheet. This new information collection request will be submitted to OMB for review under control number 0938-1486 (CMS-10935). The OMB control number will not be valid until formally approved by OMB.
As described further in section XX.C.3. of this final rule with comment period, for the purposes of reporting the data on the cost report, we proposed and are finalizing that hospitals would report the median of the payer-specific negotiated charges for an MS-DRG that the hospital has disclosed for all of its MAOs on the most recent version of the MRF that the hospital is required to disclose under the hospital price transparency regulations. We believe reporting this market-based information would result in less burden for hospitals given that hospitals are required to make public their payer-specific negotiated charges for the same service packages under the requirements we finalized in the Hospital Price Transparency final rule, which became effective January 1, 2021. We refer readers to the Hospital Price Transparency final rule for the full burden assessment analysis for the requirements set forth within that final rule (84 FR 65524). We also refer readers to section XIX. of this final rule with comment period, where we discuss our proposal to amend the hospital price transparency regulations at 45 CFR 180 to require that, beginning January 1, 2026, hospitals would report a new data element, the “median allowed amount”, instead of the “estimated allowed amount” reported at present, and that the median allowed amount would be defined as the median of the total allowed amounts that the hospital has historically received from a third-party payer (including MAOs) for an item or service. For purposes of the market-based rate information we proposed to collect on the Medicare cost report, in determining the median of the payer-specific negotiated charges to report on its cost report, we proposed that if the proposal to amend the regulations at 45 CFR 180 was finalized, the “median allowed amount” would be used for instances in which the payer-specific negotiated charge reported on the MRF is based on a percentage or algorithm. Otherwise, we proposed that the “estimated allowed amount” (as defined under current regulations) would be used in determining the median of the payer-specific negotiated charges for instances in which the payer-specific negotiated charge is based on a percentage or algorithm. We note that in section XIX. of this final rule with comment period, CMS is finalizing, that beginning January 1, 2026, if a hospital's payer specific negotiated amount is based on an algorithm or percentage, the hospital would be required to describe the percentage or algorithm and report a new data element, the “median allowed amount”, instead of the “estimated allowed amount”, and that the “median allowed amount” is defined as the median of the total allowed amounts the hospital has historically received from a third-party payer for an item or service for a time period no less than 12 months and no longer than 15 months prior to the date the MRF is posted. Should the calculated median fall between two observed allowed amounts, the median allowed amount is the next highest observed value. In section XIX. of this final rule with comment period, CMS is finalizing as proposed that if a payer-specific negotiated charge is based on a percentage or algorithm, the hospital must describe the algorithm or percentage and calculate and encode the median allowed amount in dollars for that item or service. Finally, in section XIX. of this final rule with comment period, CMS is finalizing, with modification, that hospitals must use EDI 835 ERA transaction data or an alternative equivalent source of remittance data that includes the same information as EDI 835 ERA transaction data would include, to calculate and encode the allowed amounts for items and services based on a percentage or algorithm in the MRF. We refer readers to section XIX. of this final rule with comment period for more information regarding the specific final policy. In the CY 2026 OPPS/ASC proposed rule we stated that we believed that because hospitals would already be required to publicly report the payer-specific negotiated charge information that they would use to calculate these medians, the additional calculation and reporting of the median payer-specific negotiated charge would result in less burden for hospitals than if hospitals did not already have this information compiled to disclose on the MRF under the hospital price transparency requirements. For additional details on hospital price transparency requirements, including MRF requirements and the final modifications to the hospital price transparency requirements, we refer readers to section XIX. of this final rule with comment period and https://www.cms.gov/priorities/key-initiatives/hospital-price-transparency/hospitals.
Burden hours estimate the time (number of hours) required for each IPPS hospital to complete ongoing data gathering and recordkeeping tasks, search existing data resources, review instructions, and complete the Supplemental to Form CMS-2552-10, Weighted Median MAO Payer-Specific Negotiated Charge Data Worksheet. In the CY 2026 OPPS/ASC proposed rule we stated that the most recent data from the System for Tracking Audit and Reimbursement, an internal CMS data system maintained by the Office of Financial Management (OFM), reported that 3,038 hospitals, the current number of Medicare certified IPPS hospitals, file Form CMS-2552-10 annually.
As discussed in section XX.C.2. of this final rule with comment period, we proposed that subsection (d) hospitals in the 50 states and DC, as defined at section 1886(d)(1)(B) of the Act, and subsection (d) Puerto Rico hospitals, as defined under section 1886(d)(9)(A) of the Act, would be required to report the median payer-specific negotiated charge ( printed page 54047) information. We proposed that hospitals that do not negotiate payment rates and only receive non-negotiated payments for service would be exempted from this definition. We noted that this proposed data collection requirement would apply to a smaller subset of hospitals as compared to the public reporting requirements under the hospital price transparency regulations. Under our proposal, hospitals that would be exempted from this policy included, Critical Access Hospitals (CAHs), hospitals in Maryland, which are currently paid under the Maryland Total Cost of Care Model, during the performance period of that Model, hospitals operated by an Indian Health Program as defined in section 4(12) of the Indian Health Care Improvement Act, and Federally owned and operated facilities, and non-subsection (d) hospitals. In the CY 2026 OPPS/ASC proposed rule, we also noted that rural emergency hospitals would not be subject to this data collection requirement given that they do not provide inpatient services. Based on this proposal, we estimated that 3,038 hospitals (which excludes hospitals described earlier as being exempted from this proposal) would be required to comply with this market-based data collection requirement.
Based on our understanding of the resources necessary to report this information, in the CY 2026 OPPS/ASC proposed rule we estimated an average annual burden per hospital of 20 hours (5 hours for recordkeeping and 15 hours for reporting) for the Supplemental to Form CMS-2552-10: Weighted Median MAO Payer-Specific Negotiated Charge Data Worksheet. This estimate included effort that would be necessary to crosswalk inpatient discharges to an MS-DRG, specifically if a hospital is not familiar with the MS-DRG classification system, for use in calculating the median payer-specific negotiated charges. In the CY 2026 OPPS/ASC proposed rule we stated that the burden was minimized because the median payer-specific negotiated charge data that we proposed to collect on the Supplemental to Form CMS-2552-10: Weighted Median MAO Payer-Specific Negotiated Charge Data Worksheet is based on payer-specific data that would already be maintained by the hospital, the data from the MRF that hospitals are required to disclose under the hospital price transparency regulations at 45 CFR part 180. In the CY 2026 OPPS/ASC proposed rule we stated we believed that since hospitals assign the underlying ICD-10-CM principal diagnosis, and any other secondary diagnosis codes and ICD-10-PCS procedure codes, which determine how patients are assigned to an MS-DRG, hospitals are able to associate those items and services to MS-DRGs for each discharge. Additionally, hospitals that are not as familiar with MS-DRGs have access to the most current publicly available version of the CMS Grouper used to group ICD-10 codes to MS-DRGs, and are able to use this software to uniformly group inpatient items and services to MS-DRGs, either initially by proactively using the same Grouper version used by CMS, or retrospectively after an inpatient hospital stay, but prior to submitting this information on the hospital cost report.
In the CY 2026 OPPS/ASC proposed rule, we estimated the total annual burden hours as follows: 3,038 hospitals times 20 hours per hospital equals 60,760 annual burden hours.
The 5 hours for recordkeeping include hours for bookkeeping, accounting and auditing clerks; the 15 hours for reporting include accounting and audit professionals' activities. In the CY 2026 OPPS/ASC proposed rule we stated that we believed the basic median calculation would be captured within the recordkeeping portion of this assessment.
Based on the most recent Bureau of Labor Statistics (BLS) in its 2024 Occupation Outlook Handbook, the mean hourly wage for Category 43-3031 (bookkeeping, accounting and auditing clerks) is $25.01 ( https://www.bls.gov/oes/current/oes433031.htm). We added 100 percent of the mean hourly wage to account for fringe and overhead benefits, which calculates to $50.02 ($25.01 + $25.01) and multiplied it by 5 hours, to determine the annual recordkeeping costs per hospital to be $250.10 ($50.02 × 5 hours).
The mean hourly wage for Category 13-2011 (accounting and audit professionals) is $44.96 ( www.bls.gov/oes/current/oes132011.htm). We added 100 percent of the mean hourly wage to account for fringe and overhead benefits, which calculates to $89.92 ($44.96 + $44.96) and multiplied it by 15 hours, to determine the annual reporting costs per hospital to be $1,348.80 ($89.92 × 15 hours). We calculated the total annual cost per hospital of $1,598.90 by adding the recordkeeping costs of $250.10 plus the reporting costs of $1,348.80 (Table 157). We estimated the total annual cost to be $4,857,458.20 ($1,598.90 × 3,038 IPPS hospitals) (Table 158).
( printed page 54048)Comment: Some commenters expressed concern that CMS underestimated the amount of time and burden it will take hospitals to collect, organize, properly format, calculate, update and report the median payer-specific negotiated charges by MS-DRG. According to commenters, the proposal's newly added burden is at odds with the Administration's goal of reducing administrative burden in healthcare. Commenters stated that hospitals cannot complete the task of implementing the reporting requirements themselves, nor have they been able to find vendors capable of accomplishing the task. Commenters noted that a health system operating in numerous States will have multiple contracts for each individual hospital, within each State, and with each payer. Commenters stated that this could result in the system needing to arrange the payer-specific negotiated charges for hundreds of discharges for a given MS-DRG across hundreds of different payer contracts in order to determine the median. Additionally, commenters stated that some MA organizations do not pay based on MS-DRGs and as a result, hospitals will need to calculate an MS-DRG based on the same or similar package of services. Commenters noted that this process becomes even more complicated if MAOs do not pay the hospital based on FFS rates.
According to commenters, the task of tabulating the data required for complying with this proposed policy could encompass reviewing data from thousands of patients from a single hospital taking well over the estimated 20 hours. According to a commenter, even if CMS moved to market-based weights, it would still need cost data for other purposes (outlier payments, new technology add-on payments, etc.) and thus, hospitals would shoulder additional reporting duties rather than any offsetting reduction. According to the commenter, this would create a dual system: cost reports still collecting traditional cost/charge info (for outliers and transparency enforcement) plus this new, additive median price reporting. The commenter stated that this duplication further undermines any claim of burden reduction or simplification.
According to a commenter, each hospital would be required to ensure its chargemaster team, contracting department, and finance staff coordinate to correctly identify all negotiated rates for each MS-DRG across potentially dozens of contracts, then calculate medians (with volume-weighting for each payer's discharges). For hospitals with complex contracts (per diems, etc.), the commenter stated that this means analyzing internal claims data to derive the “allowed amounts” per case and then the median, which it described as an extremely labor-intensive task. According to the commenter, this is an example of new regulation requirements that will divert critically needed resources to administrative tasks that do not benefit patients. The commenter stated that every hour staff spend on this reporting is an hour not spent on patient care improvement, revenue cycle for actual claims, or compliance with existing requirements. The commenter stated that this burden is especially onerous for smaller and rural hospitals that have fewer staff and already face compliance challenges and limited resources and it is precisely those hospitals that often have less sophisticated contracting (or more capitated arrangements) and will struggle to compute medians from messy data.
One commenter stated that they simply do not have the manpower to perform this collection and would need to contract this out to a third-party vendor leading to a cost of $32,000 a year. The commenter also stated that this would cause an increase in employee time for coordinating data capture and ensuring quality data and that the cost would continue to increase as well because of the complexity of the requirements and the resources to create the data.
Some commenters recommended CMS collect MA negotiated data from hospital or payor websites via MRFs or gather it directly since CMS already contracts with MAO plans, rather than requiring hospitals to extract and re-report the same information on the cost report, to avoid unnecessary administrative burden.
Response: We continue to note as discussed in the CY 2026 OPPS/ASC proposed rule that under the Hospital Price Transparency requirements hospitals are already required to publicly report the payer-specific negotiated charge information that they will use to calculate median payer-specific negotiated charges by MS-DRG for payers that are MA organizations. We therefore continue to believe that the additional calculation and reporting requirements in this final rule with comment period for this policy would result in less burden overall for hospitals absent those price transparency requirements since hospitals will already have the initial data compiled and the incremental additional burden of reporting medians by MS-DRG would be limited.
In the CY 2026 OPPS/ASC proposed rule (90 FR 33831), we estimated a total annual burden to hospitals of 20 hours per hospital: 5 hours for recordkeeping, including hours for bookkeeping, accounting and auditing clerks; and 15 hours for reporting, including accounting and audit professionals' activities. We acknowledge that different hospitals may face more or less ( printed page 54049) burden and do not assert that the total annual burden for every hospital will be precisely 20 hours, but we continue to believe that the 20 hour estimate per hospital for this data collection is reasonable given that hospitals will already be required to disclose the data used to calculate the median payer-specific negotiated charges under the Hospital Price Transparency requirements. We estimated an initial annual burden of 60,760 annual burden hours for 3,038 hospitals, at cost of $1,598.90 per hospital, or $4,857,458.20 across all hospitals. Although commenters did not agree with our proposed burden estimate for cost and hours, they did not provide additional information, to include a breakdown of tasks and hours estimates across hospitals, to support a burden estimate revision. In addition, we note that in response to comments in the FY 2021 IPPS/LTCH PPS final rule (85 FR 59015 through 59016), we increased the burden estimate from 15 hours to 20 hours after consideration of comments stating that additional effort would be necessary to crosswalk discharges to an MS-DRG, specifically if a hospital is not familiar with the MS-DRG classification system, for use in calculating the median payer-specific negotiated charges. In light of this prior burden estimate revision to account for the additional effort that commenters stated would be necessary to crosswalk discharges to an MS-DRG, and since hospitals are already required to publicly report the payer-specific negotiated charge information which they will use to calculate these medians, in accordance with the Hospital Price Transparency requirements in effect at the time that this data collection requirement goes into effect, we continue to believe that the burden estimate is reasonable. As noted earlier, we continue to believe that the additional calculation and reporting of the median payer-specific negotiated charge will result in less administrative burden overall for hospitals than in the absence of the Hospital Price Transparency requirements since hospitals are already required to have this information compiled and the burden associated with that compilation is already assumed.
In response to the comments recommending CMS collect MA negotiated data from hospital or payor websites via MRFs or gather it directly, we believe hospitals are more familiar with their own data and are best positioned to calculate the medians across payers.
In summary, for the reasons discussed we are maintaining our estimate for the hours associated with recordkeeping at 5 and our estimate of hours associated with reporting at 15, which equals 20 hours of annual burden per hospital and 60,760 hours of estimated annual burden across all 3,038 hospitals. This equals a cost of $1,598.90 per hospital, or $4,857,458.20 across all hospitals.
F. ICRs for Medicare OPPS Drug Acquisition Cost Survey
a. Background
In section V.C. of this final rule with comment period, we discuss our intent to conduct a survey of hospitals' drug acquisition costs. Section 1833(t)(14)(A)(iii) of the Act required the Secretary to set payment rates for specified covered outpatient drugs (SCODs) [526] beginning in 2006 at the amount the Secretary determined to be the average acquisition cost for the drug for that year, at least when certain hospital acquisition cost survey data is available. To collect the cost survey data for the Secretary to use for 2006 payment rates, section 1833(t)(14)(D)(i)(I) of the Act required the Comptroller General of the U.S. to conduct a survey in each of 2004 and 2005 to determine the hospital acquisition cost for each SCOD. To inform payment rates in later years, section 1833(t)(14)(D)(ii) of the Act requires the Secretary periodically to conduct surveys of hospital acquisition costs for each SCOD.
The GAO conducted the required surveys in 2004 and 2005, and, in reporting the results in 2006, recommended that the Secretary thereafter validate, “on an occasional basis—possibly every 5 or 10 years—average sales price (ASP) data that manufacturers report to CMS for developing SCOD payment rates.” [527] CMS has not, however, conducted its own survey of the acquisition costs for each SCOD for all hospitals paid under the OPPS. Accordingly, under section 1833(t)(14)(D)(ii) of the Act, we intend to conduct a survey, with the survey submission window opening by early CY 2026, of the acquisition costs for each separately payable drug acquired by all hospitals paid under the OPPS. We intend for the survey to be completed in time for the survey results to be used to inform policy making beginning with the CY 2027 OPPS/ASC proposed rule.
Additionally, on April 18, 2025, President Trump signed Executive Order (E.O.) 14273, “Lowering Drug Prices by Once Again Putting Americans First.” [528] Section 5 of the E.O., “Appropriately Accounting for Acquisition Costs of Drugs in Medicare,” directs the Secretary of HHS to publish in the Federal Register a plan to conduct a survey under section 1833(t)(14)(D)(ii) of the Act so he can determine the hospital acquisition cost for covered outpatient drugs at hospital outpatient departments.
b. OPPS Drug Acquisition Cost Survey Description and Burden Calculation
From January 1, 2026, through March 31, 2026, we intend to survey hospitals paid under the OPPS for their drug acquisition costs, including for SCODs, and drugs and biologicals CMS historically treats as SCODs. The survey is designed to impose the least amount of burden on hospitals as possible while ensuring we capture the required data to inform payment rates as required by statute. As part of this data collection, we will survey hospitals only about drugs that are separately paid under the OPPS and will ask hospitals to report the total acquisition cost, net of all rebates and discounts, of each drug by National Drug Code (NDC) purchased during the 1-year timeframe of July 1, 2024 through June 30, 2025. We are asking hospitals to incorporate all rebates and discounts in their acquisition cost for each NDC, including discounts directly applicable to an individual NDC, but also those discounts that are not necessarily linked to a single NDC, but could be a discount linked to a certain invoice, or discounts linked to purchases made over a certain time period, such as prompt pay discounts, wholesaler discounts, or other discounts. We understand that certain discounts may depend on whether an eligible patient receives the drug. That is true, for example, for drugs acquired through the 340B program. We are therefore asking for hospitals to separately list their acquisition costs for drug NDCs acquired through the 340B program and those drug NDCs acquired outside of the 340B program to ensure that all discounts are accurately captured and represent the hospital's acquisition costs. In the CY 2026 OPPS/ASC proposed rule, we welcomed comments on whether other common drug discount programs have a similar structure or should otherwise also be separately noted.
There are approximately 700 drug HCPCS codes that will be subject to the survey, with most HCPCS codes having multiple NDCs per HCPCS code. During ( printed page 54050) the CY 2026 OPPS/ASC proposed rule stage, we published a draft list of the NDCs that would be included in the survey, if finalized, so hospitals would have ample opportunity to review and prepare to report their acquisition costs for those NDCs. We noted there may be slight adjustments to this NDC list, but we expected the final list would be similar to the draft list. We recognized that hospitals may not have acquired all drugs on this list, and hospitals are not expected to provide data for NDCs for which they do not have acquisition cost data. We are collecting acquisition cost data by NDC as we understand most hospitals acquire drugs from wholesalers and manufacturers based on NDCs rather than other identifiers, such as HCPCS billing codes. We expect this method will likely reduce hospital burden, as hospitals can simply report the cost at which they acquired the drug without significant calculations. Additionally, we have designed the survey so that only the total cost and the total units of the drug acquired need to be reported. This means that only two fields of information are required per NDC: total net acquisition cost—non-340B and total units purchased—non-340B. If the same drug NDC is purchased multiple times throughout the given timeframe, only the total cost of all of the drug acquired during the given timeframe plus the total number of units purchased is needed. As we previously discussed, for each NDC, we are asking hospitals to report the total acquisition cost, net of all rebates and discounts, which includes all discounts attributable to each specific NDC as well as those discounts attributable to multiple NDCs. For those discounts received for drugs acquired through the 340B Program, since those discounts may be dependent on whether a 340B eligible patient receives the drug, we are asking for hospitals to separately list their acquisition costs for those drug NDCs acquired through the 340B program and those drug NDCs acquired outside of the 340B program. This means for 340B covered entity hospitals that acquire NDCs through the 340B program, they are to submit up to four fields of information for each NDC depending on their acquisition patterns: total net acquisition cost—non-340B, total units purchased—non-340B, total net acquisition cost 340B, total units purchased 340B.
We will assume the burden of performing any additional calculations. We believe this collection of information is based on common information that the hospital already has in its records from its drug purchase history.
This survey will apply to all hospitals paid under the OPPS, which for purposes of our burden calculations we estimated to be 3,500 hospitals in the CY 2026 OPPS/ASC proposed rule. Based on our understanding of hospital practices, in the CY 2026 OPPS/ASC proposed rule we estimated the total time for each hospital to respond to the survey to be 73.5 hours, which includes time required to review instructions, gather data (including potentially from hospital wholesalers), perform basic addition calculations, and enter data. As previously mentioned, we will take every practical step to streamline the data collection for each hospital.
We estimated 73.5 hours to complete the survey by aggregating time from the four roles that are most likely to be responsible. These roles are described below:
- A Top Executive (11-1000) will likely review the survey request and designate a Submitter prior to survey distribution.
- A Lawyer (23-1011) will likely review the survey request, the survey, and requirements for compliance.
- A Pharmacy Technician (29-1051) will likely register for the module and apply to fill out the survey. Once the survey is distributed, the Pharmacy Technician will review the survey. Then, the Pharmacy Technician will request data from suppliers, and/or pull data from internal systems, ensure data are in the appropriate format, manually enter data OR upload data into system, review data, make corrections as needed, and certify data.
- A Pharmacist (29-2052) will likely review the survey request and data that are pulled by the Pharmacy Technician.
As described in Table 159, we estimated the total burden hours as follows: 3,500 hospitals times 73.5 hours per hospital equals 257,250 hours. We used data from the Occupational Employment and Wage Statistics (Hospital-Specific Wages) [529] for all ( printed page 54051) salary estimates. In this regard, the previous table presents the mean hourly wage, the cost of fringe benefits, and the adjusted hourly wage for providers that are responsible for completing the survey. We added 100 percent of the mean hourly wage to account for fringe and overhead benefits.
We received public comments on these proposals. We refer readers to section V.C. of this final rule with comment period for the summary of comments and our responses to them. We reiterate below one comment especially relevant to the calculation of burden discussed in this section.
Comment: Many commenters alleged that, given the complexity and scale of the required data collection and analysis, conducting the survey will impose a significant burden on hospitals and that CMS's estimate of that burden grossly underestimates the cost, time and resources that will be necessary to complete the survey. Commenters also stated that CMS is wrong to assume that the reporting will be done by pharmacy technicians and that the survey will actually be completed by pharmacists, which will cost more. One commenter opined that completing the survey would demand the concerted effort of multi-disciplinary teams, including pharmacy, supply chain, finance, legal, and reimbursement professionals, far beyond what pharmacy technicians alone could provide. The commenter stated that CMS's own Information Collection Review document “acknowledges the breadth of the undertaking, but fails to account for the extensive coordination needed with vendors and suppliers, from whom much of the requested data must be sourced. The time and resources required to collect, clean, analyze, and accurately report thousands of distinct drug prices would divert critical personnel from patient care and other essential hospital functions”. Many other commenters also emphasized the point that the cost of completing the survey would come at the expense of the hospital's ability to provide essential care to patients and quoted the GAO's 2006 report to the Congress in which the GAO concluded that the surveys it conducted “created a considerable burden for hospitals” and that to submit the required price data hospitals “had to divert staff from their normal duties, thereby incurring additional costs.” Many commenters stated that the financial burden of completing the survey would be exacerbated by upcoming Medicaid and Medicare reductions under the Inflation Reduction Act.
Response: We have taken the burdens on hospitals of completing the survey and the GAO's conclusions relating to that burden very seriously in the design and implementation of the survey. We have created a survey instrument and survey process that we think will minimize, to the greatest extent possible, the staffing and financial burden on hospitals of collecting and reporting the necessary information to CMS. The survey instrument consists of a streamlined online portal, where hospitals can either directly enter acquisition costs or download and reupload an excel template of acquisition costs. There will be technical assistance available for those with any issues that arise during the submission process. We have taken the feedback from commenters into careful consideration when finalizing the survey tool. We believe future surveys will be periodic in nature, as to limit the burden that hospitals face on a reoccurring basis.
We were persuaded that the role of the pharmacist in the data collection effort may be greater than 1 hour. Therefore, based on comments, we are now estimating 1 working day of 8 hours, for a pharmacist to assist in completing the survey. This is reflected in Table 160. Additionally, reflected in Table 160 is an updated number of total hospitals expected to respond to the survey. This number was determined by assessing which CCNs were paid under the OPPS during the survey period for a drug or biological under the OPPS. Additionally, in the rare event that a hospital is paid under the OPPS, but does not have any acquisition costs for the entire year period that is being surveyed, we'd still expect a submission by the hospital. During the CY 2026 OPPS/ASC proposed rule stage, we published a draft list of the NDCs that would be included in the survey, if finalized, so hospitals would have ample opportunity to review and prepare to report their acquisition costs for those NDCs. We noted there may be slight adjustments to this NDC list, but we expected the final list would be similar to the draft list. We have refined this list by removing certain NDCs that were not separately payable under the OPPS to ensure that we are only surveying necessary drugs.
( printed page 54052)After consideration of public comments as discussed in section V.C. of this final rule with comment period, we are finalizing our intent to conduct a survey of OPPS drug acquisition costs, subject to the clarifications and modifications discussed in this final rule with comment period and outlined in the final package approved by OMB. Based on the updates to the burden calculations in response to commenters, we now estimate the cost per response to be $5,047.15 and the total estimated cost across all survey respondents to be $22,712,175.
This new information collection request will be submitted to OMB for review under control number 0938-1487 (CMS-10931). The OMB control number will not be valid until formally approved by OMB.
G. ICRs for Hospital Price Transparency
In a final rule published in November 2019 (84 FR 65524) (herein referred to as the CY 2020 HPT final rule), we adopted requirements for hospitals to make public their standard charges in two ways: (1) as a comprehensive machine-readable file (MRF); and (2) in a consumer-friendly format. We codified these requirements at 45 CFR 180.50 and 180.60, respectively.
The proposed changes to the information collection request were submitted to OMB for review under control number 0938-1369 (CMS-10707). The previously approved requirements and burden associated with 0938-1369 lapsed due to administrative oversight. Specifically, CMS failed to submit the revisions to 0938-1369 that pertained to the 2024 hospital price transparency requirements in the CY 2024 OPPS/ASC final rule with comment period (88 FR 81540). Therefore, we included the finalized burden mentioned in the CY 2024 OPPS/ASC final rule with comment period and the new burden associated with the CY 2026 OPPS proposed rule in the request for reinstatement.
In the CY 2020 HPT final rule, we originally estimated the number of hospitals subject to the HPT requirements to be 6,002. We finalized an initial one-time burden of 150 hours and cost of $11,898.60 per hospital, resulting in a total national burden of 900,300 hours (150 hours × 6,002 hospitals) and $71,415,397 ($11,898.60 × 6,002 hospitals) for hospitals to build processes and make required system updates to make their standard charge information publicly available: (1) as a comprehensive MRF and (2) in a consumer-friendly format. Additionally, we estimated an ongoing annual burden of 46 hours per hospital with a cost of $3,610.88 per hospital, resulting in a total national burden of 276,092 hours (46 hours × 6,002 hospitals) and total cost of $21,672,502 ($3,610.88 × 6,002 hospitals), to make required annual updates to the hospitals' standard charge information. For a detailed discussion of the cost estimates for the requirements related to hospitals making their standard charge information publicly available, we refer readers to our discussion in the collection of information section in the CY 2020 HPT final rule (84 FR 65591 through 65596).
In the CY 2024 OPPS/ASC final rule with comment period (88 FR 82080 through 82114), we finalized revisions to the regulations at 45 CFR 180.50 related to making public hospital standard charges in an MRF. First, we finalized adding data elements to be included in the hospital's MRF and to require hospitals to conform to a CMS template layout. Second, to enhance automated access to the MRF, we finalized that hospitals include a .txt file in the root folder of the public website it selects to host its MRF in the form and manner specified by CMS that includes a standardized set of fields, and a link in the footer on its website that is labeled “Hospital Price Transparency” and links directly to the publicly available web page that hosts the link to the MRF.
( printed page 54053)As explained in the CY 2024 OPPS/ASC final rule with comment period, we increased the number of hospitals that we believed to be subject to these requirements from 6,002 to 7,098, which, in turn, increased the estimated national burden. The reason for this increase was because in the CY 2020 HPT final rule (84 FR 65591), we relied on data from the American Hospital Association (AHA).[530] For the collection of information estimates in the CY 2024 OPPS/ASC final rule with comment period we used updated hospital numbers based on the publicly available dataset from the Homeland Infrastructure Foundation-Level Data (HIFLD) hospital dataset.[531] The HIFLD dataset compiles a directory of hospital facilities based on data acquired directly from State hospital licensure information and Federal sources and validates this data annually. Thus, we stated our belief that the HIFLD dataset is more comprehensive than the AHA Directory. To estimate the number of hospitals subject to these requirements in the CY 2024 OPPS/ASC proposed rule with comment period, we leveraged the HIFLD hospital dataset to identify 8,013 total hospitals. We then subtracted 379 hospitals HIFLD identified as “closed” as well as hospitals that are deemed under the regulation to have met requirements (see 45 CFR 180.30) which included 339 Federally owned non-military and military hospitals, and 197 State, local, and district run forensic hospitals. We therefore estimated that the CY 2024 OPPS/ASC final rule with comment period would apply to 7,098 hospitals operating within the U.S that meet the HPT regulation's definition of “hospital” at 45 CFR 180.20.
In the CY 2024 OPPS/ASC final rule with comment period (88 FR 82151), we estimated the total initial one-time burden to implement the CMS standard template and conform to the data dictionary to be 120 hours (5 hours for a Lawyer + 5 hours for a General and Operations Manager + 80 hours for a Business Operations Specialist + 30 hours for a Network and Computer System Administrator) per hospital with a cost of $10,587.10 ($787.40 for a Lawyer + $590.70 for a General and Operations Manager + $6,406.40 for a Business Operations Specialist + $2,802.60 for a Network and Computer System Administrator) per hospital. The initial one-time national burden was calculated to be $75,147,235.80 dollars ($10,587.10 per hospital × 7,098 hospitals). As we indicated in the CY 2026 OPPS/ASC proposed rule, we still believe this estimate to be an accurate estimate of the one-time burden for a new hospital to implement the CMS standard template and conform to the data dictionary. However, CMS is not presently aware of any new hospitals that are beginning operations. We find it challenging to determine the number of new hospitals that are opened each year because distinguishing brand-new hospitals from expansions, new locations, or mergers is inherently arduous. Many hospitals open satellite facilities or rebrand existing ones under similar names, creating ambiguity in identifying independent entities. Additionally, there is no standardized or centralized database that categorizes hospitals based on their origin, and regulatory processes often overlap for new openings, expansions, and mergers, making it difficult to rely on licensing data alone. Complex ownership structures within healthcare systems further blur the lines between new hospitals and extensions of existing networks. Marketing strategies and naming conventions can also mislead public perception, as hospitals often promote new locations as “new” regardless of their operational independence. Finally, data inconsistencies and delays in reporting further complicate efforts to verify whether a hospital is truly new. Because we find it difficult to determine a new hospital, we will still account for the original one-time burden to implement the CMS standard template that we calculated in the CY 2024 OPPS/ASC final rule with comment period, but we will no longer account for this one-time burden moving forward.
Additionally, we finalized in the CY 2024 OPPS/ASC final rule with comment period an estimated ongoing annual national burden of 383,292 hours (54 hours × 7,098 hospitals) and an annual national cost of $32,370,571 dollars ($4,560.52 per respondent × 7,098 hospitals), which represented a $10,698,069 ($32,370,571−$21,672,502) increase over our previous estimated ongoing national annual burden for subsequent years for hospitals to update their standard charge information in the CMS standard template and conform to the data dictionary.
As we discuss in more detail below, in addition to providing updated one-time estimates for hospitals to implement new MRF data elements, updating wage rates for the ongoing annual estimates, and adding an additional labor category for Chief Executives, we are also updating the number of hospitals estimated to be subject to the HPT requirements, in this final rule with comment period.
For this final rule with comment period, we updated the number of hospitals estimated to be subject to the HPT requirements using the same methodology as we did in the CY 2024 OPPS/ASC final rule with comment period. There were 8,340 hospitals most recently identified in the HIFLD hospital dataset.[532] We subtracted 374 hospitals HIFLD identified as “closed” as well as hospitals that are deemed under the regulation to have met requirements which included 352 Federally owned non-military and military hospitals, and 198 State, local, and district run forensic hospitals. We therefore estimate that, for this final rule with comment period, 7,416 hospitals would meet the HPT regulation's definition of “hospital” at 45 CFR 180.20.
We estimated the hourly cost for each labor category used in this analysis by referencing the Bureau of Labor Statistics report on Occupational Employment and Wages (May 2024).[533] We included labor categories for General and Operations Managers, Business Operations Specialists, and Network and Computer Systems Administrators for this final rule with comment period as we still believe these labor categories are associated with the one-time and annual burden related to the implementation of HPT requirements. As discussed below, we have also added a labor category for Chief Executives in response to comments on the CY 2026 OPPS/ASC proposed rule (that are addressed in section XIX. of this final rule with comment period), that suggested we failed to account for the burden for the hospital chief executive officer, president, or senior official designated to oversee the encoding of true, accurate, and complete data in the MRF ( printed page 54054) to review and attest to the information (See Table 161.)
As discussed in section XIX. of this final rule with comment period, while we believe the “estimated allowed amount” (defined at § 180.20 as the average dollar amount that the hospital has historically received from a third party payer for an item or service) provides useful additional context and enhances transparency and comparability of hospital standard charges, we acknowledge that these average dollar amounts do not necessarily apply to any particular individual, nor do they necessarily represent the actual dollar amount an individual would pay for an item or service. Therefore, we proposed to require hospitals to report four new data elements when the payer-specific negotiated charge is based on a percentage or algorithm—the median allowed amount (which would replace the estimated allowed amount data element), the 10th percentile allowed amount, the 90th percentile allowed amount, and the count of allowed amounts. We also proposed to require that hospitals abide by specific instructions regarding the data source and methodology, including the lookback period, that should be used to calculate the median, 10th and 90th percentile allowed amounts. As we indicated in the CY 2026 OPPS/ASC proposed rule, we believe that the median, 10th and 90th percentile allowed amounts would provide greater context and clarity with respect to the payer-specific negotiated charge, would be a better consumer benchmark than the estimated allowed amount, and better enable price estimator tools to develop and estimate an individual's personalized out-of-pocket cost, enabling MRF users to more easily compare such standard charges across hospitals. After consideration of public comments, discussed in an earlier section of this final rule with comment period, we are finalizing with modification our proposals to replace the estimated allowed amount with the median allowed amount and to add the 10th and 90th percentile allowed amounts, including our proposed methodology for calculating the allowed amounts should the calculated percentile fall between two observed allowed amounts, effective January 1, 2026. We are finalizing with modification our proposal that a hospital must calculate and encode the total number of allowed amount remittances that were used to calculate the median, 10th and 90th percentile allowed amounts, using the valid values and instructions that will be detailed in the CMS Hospital Price Transparency—Data Dictionary GitHub Repository website. We are also finalizing with modification our proposals to require that hospitals abide by specific instructions regarding the data source and methodology, including the lookback period, that should be used to calculate the median, 10th and 90th percentile allowed amounts effective January 1, 2026.
We also proposed that beginning January 1, 2026, hospitals must attest in their MRF that they have included all applicable standard charge information in accordance with the requirements of 45 CFR 180.50, and the information encoded is true, accurate, and complete as of the date in the file, and the hospital has included all payer-specific negotiated charges in dollars that can be expressed as a dollar amount. For payer-specific negotiated charges that cannot be expressed as a dollar amount in the MRF, or are not knowable in advance, the hospital would attest that the payer-specific negotiated charge is based on a contractual algorithm, percentage or formula that precludes the provision of a dollar amount and has provided all necessary information available to the hospital for the public to be able to derive the dollar amount, including, but not limited to, the specific fee schedule or components referenced in such percentage, algorithm or formula. Additionally, we proposed that, beginning January 1, 2026, the hospital must encode within the MRF the name of the chief executive officer, president, or senior official designated to oversee the encoding of true, accurate and complete data in the MRF. As we indicated in the CY 2026 OPPS/ASC proposed rule, we believe these proposed requirements would provide the necessary reassurance that hospitals have provided in their MRFs meaningful, accurate information to users of the MRF about their standard charges for health care items and services. However, after consideration of public comments, discussed in an earlier section of this final rule with comment period, we are finalizing, effective January 1, 2026, our proposal to supplant the existing affirmation requirement with the proposed attestation statement at new ( printed page 54055) § 180.50(3)(iii), with a modification to add to the following phrase to the beginning of the attestation: “To the best of its knowledge and belief,”. In addition, we are finalizing, as proposed, new § 180.50(a)(3)(iv) to require that, beginning January 1, 2026, the hospital must encode within the MRF the name of the hospital chief executive officer, president, or senior official designated to oversee the encoding of true, accurate and complete data as directed in § 180.50(a)(3)(iii).
We also proposed in the CY 2026 OPPS/ASC proposed rule adding a standard identifier, specifically the hospital's National Provider Identifiers (NPIs) to the MRFs. As we indicated in the CY 2026 OPPS/ASC proposed rule, we believe that adding a standard identifier to the file would advance the comparability of the HPT data with other healthcare data, including health plan transparency data from the Transparency in Coverage (TiC) MRFs. After consideration of public comments, discussed in an earlier section of this final rule with comment period, we are finalizing, as proposed, the requirement that, beginning January 1, 2026, hospitals must report, in a newly created general data element in the MRF, any Type 2 NPI(s) that are associated with a primary taxonomy code starting with `28' (indicating hospital) or `27' (indicating hospital unit) and that is active as of the date of the most recent update to the standard charge information.
As we indicated in the CY 2026 OPPS/ASC proposed rule, we still believe that, by now, hospitals have largely developed standardized processes and procedures for encoding the existing estimated allowed amount and general data elements, like hospital license number, in the MRF and that modifying their existing processes to include the four new data elements related to the proposed allowed amounts and hospital NPI would not entail a significant amount of additional work for hospitals. Furthermore, hospitals are required to encode the affirmation statement in the MRF currently, therefore we still believe the additional burden related to the proposed attestation statement is the requirement for hospitals to encode the name of the senior official making the attestation. However, as discussed in more detail below, after consideration of public comments, we are increasing both the one-time and annual burden hours estimates associated with the information collections in this final rule with comment period.
We indicated in the CY 2026 OPPS/ASC proposed rule, we believed hospitals would incur an initial one-time cost to update their processes and systems to (1) identify and collect the newly proposed data elements, and (2) encode the standard charge information for the newly proposed data elements in the CMS standard template. To implement the proposed requirements, we estimated that it would take a Business Operations Specialist (BLS 13-1000), on average, 4 hours (at a cost of $87.52 per hour) to develop and update the necessary processes and procedures and develop the requirements to implement the proposed data elements and a General and Operations Managers (BLS 11-1021), on average, 1 hours (at a cost of $128.00 per hour) to review the updates.
Therefore, we believed the one-time burden estimate to be 37,080 hours for all hospitals (5 hours × 7,416 hospitals) at a cost of $3,545,441.28 (7,416 hospitals × [($87.52 × 4 hours) + ($128.00 × 1 hours)]); see Table 162. As we indicated in the CY 2026 OPPS/ASC proposed rule, we believe the benefits to users of the MRF of having this additional information would justify the initial one-time burden to hospitals to update their processes and systems to identify and collect the newly proposed data elements and encode the standard charge information for the newly proposed data elements in the CMS standard template.
For the annual burden estimate we relied on our previous assumptions related to labor categories and number of hours as we did in the CY 2024 OPPS/ASC final rule with comment period (88 FR 82153). We estimated it would take a General and Operations Manager 2 hours per hospital, to review and determine updates in compliance with requirements. We estimated the ongoing time for a Business Operations Specialist to be 40 hours per hospital, to identify and gather the required data elements on an annual basis. We estimated that it would take a Computer System Administrator 12 hours to maintain and post the MRF in a manner that conforms to the CMS standard template, which brings the total burden per hospital to 54 hours. Therefore, we estimated a total annual burden of 400,464 hours for all hospitals (7,416 hospitals × 54 hours) at a cost of $36,519,350.40 (7,416 hospitals × [($128/hour × 2 hours) + ($87.52/hour × 40 hours) + ($97.30/hour × 12 hours)]); see Table 163.
( printed page 54056)We received public comments on these information collections. The following is a summary of the comments we received and our responses.
Comment: While many commenters indicated that our proposed changes to the HPT requirements outlined in the CY 2026 OPPS/ASC proposed rule and/or the HPT requirements in general are burdensome, as discussed in prior sections of this final rule with comment period, only several commenters commented on our specific burden estimates for these information collections. Several commenters stated that CMS's projected annual burden estimate was underestimated, noting an undefined, but “significantly higher” burden and cost to implement the proposed policies. A few commenters stated that hospitals that contract with outside vendors or consultants to update their MRFs on an annual basis, do so at a cost of $10,000 to $30,000 per hospital and those without vendor support report needing 5 to 30 full-time-equivalents (FTEs) to update their MRFs on an annual basis. One commenter indicated that they recently updated their MRF to meet the annual update requirement and it required 5 times the amount of hours CMS estimated for the annual burden estimates. One commenter indicated that hospitals incur annual vendor fees of up to $250,000 to update their MRFs and that even with vendor support, hospitals may have three FTEs spend an entire month developing and validating their MRFs.
Similarly, several commenters indicated that CMS' one-time burden estimate to encode the new data elements in the MRF as proposed in the CY 2026 OPPS/ASC proposed rule was underestimated. A few commenters maintained that they expect to spend $20,000-$30,000 to elicit vendor support to encode the new data elements in the MRF by the January 1, 2026, effective date proposed in the CY 2026 OPPS/ASC proposed rule. One commenter indicated they believe they will need to spend thousands of dollars per hospital to hire a third-party vendor or devote an internal “project management team” to support the encoding of the new data elements. One commenter incorrectly stated that we estimated a one-time burden estimate of 20 hours to implement the new HPT requirements, at a cost of $1,598.90, and that this estimate significantly understated the real cost of implementing the proposals. One commenter indicated they had already spent more than the cost of the entire one-time burden estimate in just reviewing the proposed rule.
Response: We appreciate commenters' concerns and the varying range of estimates provided by commenters suggests that hospitals have different operational and administrative processes and systems that impact the projected burden of encoding the new data elements and meeting the requirement to update the MRF annually. To address this variability, we allow hospitals to choose which CMS MRF template format they use, providing hospitals some flexibility to select the least burdensome format and layout to develop and update their MRF. We expect that, as indicated in the CY 2026 OPPS/ASC proposed rule, more than a year after the implementation of the CMS MRF standard template, some hospitals have well developed automated processes in place that they leverage to minimize the burden associated with making hospital standard charge information public in their current MRFs. Additionally, as discussed in more detail in prior sections of this final rule with comment period and as with previous HPT rulemaking, we will provide technical guidance and examples of how to encode the new data elements we are finalizing in this rule on the CMS Hospital Price Transparency—Data Dictionary GitHub Repository, as well as guidance on the HPT resources page on the CMS website to further minimize the burden to hospitals.
Moreover, in order to further reduce burden, as discussed in more detail in prior sections of this final rule with comment period, we are delaying enforcement of our requirements to encode the new data elements in the MRF. Specially, we are finalizing at § 180.50, the removal of the estimated allowed amount, disclosure of the 10th percentile, median, 90th percentile allowed amounts and the count of allowed amounts, the attestation requirements, and the requirement to encode hospital NPIs effective January 1, 2026. However, we will delay enforcement of these finalized revisions until April 1, 2026. We believe this 3-month enforcement delay will provide hospitals with sufficient additional time to encode the new data elements and review their MRFs prior to making them public online.
We continue to believe that increased standardization and comparability of the MRFs benefit consumers of the MRF, and that this benefit outweighs the burden imposed by these requirements. However, we are swayed by commenters that suggested we underestimated the one-time burden of encoding the new data elements in the MRF. Therefore, we have increased the one-time burden estimate for the General and Operations Manager and Business Operations Specialist labor categories by doubling those estimates in this final rule with comment period. Additionally, we have also added one-time and annual burden estimates for Chief Executives in response to comments on the “Modification to the MRF Affirmation Statement” section of the CY 2026 OPPS/ASC proposed rule, addressed in an earlier section of this final rule with comment period, that suggested we failed to account for the burden for the hospital chief executive officer, president, or senior official designated to oversee the encoding of true, accurate, and complete data in the MRF to review and attest to the information. However, we have retained our existing annual burden estimates for the General and Operations Manager and Business Operations Specialist labor categories as we believe it is reasonable to assume that the burden to ( printed page 54057) hospitals for encoding the new data elements finalized in this rule will lessen with subsequent annual updates to the MRF once hospitals have developed standardized processes and procedures for doing so such that increasing the annual burden estimates for these labor categories is unnecessary.
Final Action: After consideration of public comments, we are increasing both our one-time and annual burden estimates. To implement the encoding of the new data elements we are finalizing in this final rule with comment period, we now estimate that it will take a Business Operations Specialist (BLS 13-1000), on average, 8 hours (at a cost of $87.52 per hour) to develop and update the necessary processes and procedures and develop the requirements to implement the new data elements and a General and Operations Managers (BLS 11-1021), on average, 2 hours (at a cost of $128.00 per hour) to review the updates, and a Chief Executive (BLS 11-1011) 2 hours (at a cost of $252.82) to review and attest to the accuracy and completeness of the data in the MRF. Therefore, we believe the one-time burden estimate to be 88,992 hours for all hospitals (12 hours × 7,416 hospitals) at a cost of $10,840,708.80 (7,416 hospitals × [($87.52 × 8 hours) + ($128.00 × 2 hours) + ($252.82 × 2 hours]); see Table 164.
Additionally, we still estimate it will take a General and Operations Manager (BLS 11-1021), 2 hours (at a cost of $128.00 per hour) per hospital to review and determine updates in compliance with the annual update requirement. We still estimate the ongoing time for a Business Operations Specialist (BLS 13-1000), to be 40 hours (at a cost of $87.52 per hour) per hospital, to identify and gather the required data elements on an annual basis. We still estimate that it will take a Computer System Administrator (BLS 15-1244) 12 hours (at a cost of $97.30 per hour) to maintain and post the MRF in a manner that conforms to the CMS standard template. However, we now estimate it will take a Chief Executive (BLS 11-1011) 2 hours (at a cost of $252.82) to review and attest to the accuracy and completeness of the data in the MRF each year prior to posting the MRF online, which now brings the total ongoing annual burden per hospital to 56 hours. Therefore, we estimate a total ongoing annual burden of 415,296 hours for all hospitals (7,416 hospitals × 56 hours) at a cost of $40,269,176.60 (7,416 hospitals × [($128/hour × 2 hours) + ($87.52/hour × 40 hours) + ($97.30/hour × 12 hours) + ($252.82/hour × 2 hours]); see Table 165. The annual burden is increased by 2 hours from 54 hours to 56 hours per hospital as compared to the CY 2024 OPPS/ASC final rule with comment period, which reflects the additional burden annually associated with the new requirements finalized in this final rule with comment period.
If you comment on these information collection, that is, reporting, recordkeeping or third-party disclosure requirements, please submit your comments to the Office of Information and Regulatory Affairs, Office of Management and Budget,
Attention: CMS Desk Officer, CMS-1834-FC
Fax: (202) 395-6974; or
Email: OIRA_submission@omb.eop.gov
XXIV. Files Available to the Public via the Internet
The Addenda to the OPPS/ASC proposed rules and final rules with comment period are published and available via the internet on the CMS website. In the CY 2019 OPPS/ASC final rule with comment period (83 FR 59154), for CY 2019, we changed the format of the OPPS Addenda A, B, and C by adding a column titled “Copayment Capped at the Inpatient Deductible of $1,364.00” where we flag, through use of an asterisk, those items and services with a copayment that is equal to or greater than the inpatient hospital deductible amount for any given year (the copayment amount for a ( printed page 54058) procedure performed in a year cannot exceed the amount of the inpatient hospital deductible established under section 1813(b) of the Act for that year). In the CY 2022 OPPS/ASC final rule with comment period (85 FR 86266), we updated the format of the OPPS Addenda A, B, and C by adding a column titled “Drug Pass-Through Expiration during Calendar Year” where we flagged, through the use of an asterisk, each drug for which pass-through payment was expiring during the calendar year on a date other than December 31. For CY 2026 and subsequent years, we proposed to retain these columns that are updated to reflect the drug codes for which pass-through payment is expiring in the applicable year.
In the CY 2023 OPPS/ASC final rule with comment period (87 FR 72250) for CY 2023, we changed the format of the OPPS Addenda A, B, and C by adding a column titled “Drug Pass-Through Expiration during Calendar Year” to include devices, so that the column reads: “Drug and Device Pass-Through Expiration during Calendar Year” where we flagged, through the use of an asterisk, each drug and device for which pass-through payment was expiring during the calendar year on a date other than December 31.
For CY 2024, we deleted the column titled “Copayment Capped at the Inpatient Deductible” and instead added a new column for “Adjusted Beneficiary Copayment” to identify any copayment adjustment due to either the inpatient deductible amount copayment cap or the inflation-adjusted copayment of a Part B rebatable drug per section 1833(t)(8)(F) and section 1833(i)(9) of the Act, as added by section 11101 of the Inflation Reduction Act (IRA). We also added another column for notes. The “Note” column contains multiple messages including, but not limited to, inflation-adjusted copayment of a Part B rebatable drug, the copayment for a code capped at the inpatient deductible, or 8 percent of the reference product add-on applied for a biosimilar.
In addition, for CY 2024, we updated the format of the OPPS Addenda A, B, and C by adding another column for “IRA Coinsurance Percentage” to identify the percentage for the inflation-adjusted copayment of a Part B rebatable drug per section 1833(t)(8)(F) and section 1833(i)(9) of the Act, as added by section 11101 of the Inflation Reduction Act (IRA).
For CY 2026 and subsequent years, we proposed to keep the same format for the addenda A, B, and C, and we did not propose any additional changes for CY 2026.
We did not receive any public comments related to the format of the OPPS Addenda A, B, and C and are adopting the addenda format as proposed.
To view the Addenda to this final rule with comment period pertaining to CY 2026 payments under the OPPS, we refer readers to the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices; select “CMS-1834-FC” from the list of regulations. All OPPS Addenda to this final rule with comment period are contained in the zipped folder titled “2026 NFRM OPPS Addenda” in the related links section at the bottom of the page. To view the Addenda to this final rule with comment period pertaining to CY 2026 payments under the ASC payment system, we refer readers to the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/ambulatory-surgical-center-asc/asc-regulations-and-notices; select “CMS-1834-FC” from the list of regulations. The ASC Addenda to this final rule with comment period are contained in a zipped folder titled “2026 NFRM Addendum AA, BB, DD1, DD2, EE, and FF” in the related links section at the bottom of the page.
XXV. Response to Comments
Because of the large number of public comments, we normally receive on Federal Register documents, we are not able to acknowledge or respond to them individually. We will consider all comments we receive by the date and time specified in the DATES section of this preamble; and, when we proceed with a subsequent document, we will respond to the comments in the preamble to that document.
XXVI. Economic Analyses
A. Statement of Need
This final rule with comment period is necessary to make updates to the Medicare hospital OPPS rates. It is also necessary to make changes to the payment policies and rates for outpatient services furnished by hospitals and CMHCs in CY 2026. We are required under section 1833(t)(3)(C)(ii) of the Act to update annually the OPPS conversion factor used to determine the payment rates for APCs. We also are required under section 1833(t)(9)(A) of the Act to review, not less often than annually, and revise the groups, the relative payment weights, and the wage and other adjustments described in section 1833(t)(2) of the Act. We must review the clinical integrity of payment groups and relative payment weights at least annually. We are revising the APC relative payment weights using claims data for services furnished on and after January 1, 2024, through and including December 31, 2024, and processed through June 30, 2025, and updated HCRIS cost report information.
This final rule with comment period is also necessary to make updates to the ASC payment rates for CY 2026, enabling CMS to make changes to payment policies and payment rates for covered surgical procedures and covered ancillary services that are performed in ASCs in CY 2026. Because ASC payment rates are based on the OPPS relative payment weights for most of the procedures performed in ASCs, the ASC payment rates are updated annually to reflect annual changes to the OPPS relative payment weights. In addition, we are required under section 1833(i)(1) of the Act to review and update the list of surgical procedures that can be performed in an ASC, not less frequently than every 2 years.
In the CY 2019 OPPS/ASC final rule with comment period (83 FR 59075 through 59079), we finalized a policy to update the ASC payment system rates using the hospital market basket update instead of the CPI-U for CY 2019 through 2023. In the CY 2024 OPPS/ASC final rule with comment period, we finalized a policy to extend the 5-year interim period by an additional 2 years, through CY 2024 and CY 2025, to enable us to more accurately analyze whether the application of the hospital market basket update to the ASC payment system resulted in a migration of services from the hospital setting to the ASC setting (88 FR 81960). As discussed in section XIII. of this final rule with comment period, we are extending our utilization of the hospital market basket update as the update factor for the ASC payment system for one additional year (through CY 2026). The ASC impacts discussed below reflect our application of the hospital market basket update for CY 2026.
In addition, this final rule with comment period is necessary to make policy changes for facilities reporting data under the Hospital OQR, REHQR, and ASCQR Programs. The primary objective of these quality reporting programs is to promote higher quality, more efficient health care for Medicare beneficiaries by collecting and reporting on quality-of-care metrics. This information is made available to consumers, both to empower Medicare beneficiaries and inform decision making, as well as to incentivize healthcare facilities to make continued improvements. This rule is also ( printed page 54059) necessary to modify the methodology for the Overall Hospital Quality Star Ratings to emphasize and align the importance of patient safety across CMS programs. The Overall Hospital Quality Star Ratings information is publicly available.
Also, this final rule with comment period is necessary to enhance clarity and standardization in hospital disclosure of standard charges. The Hospital Price Transparency regulations requiring public release of hospital standard charge information are a necessary and important first step in ensuring transparency in prices of healthcare services for consumers.
B. Overall Impact of Provisions of This Final Rule With Comment Period
We have examined the impacts of this rule as required by Executive Order 12866, “Regulatory Planning and Review”; Executive Order 13132, “Federalism”; Executive Order 13563, “Improving Regulation and Regulatory Review”; Executive Order 14192, “Unleashing Prosperity Through Deregulation”; the Regulatory Flexibility Act (RFA) (Pub. L. 96-354); section 1102(b) of the Social Security Act; and section 202 of the Unfunded Mandates Reform Act of 1995 (Pub. L. 104-4); and the Congressional Review Act (5 U.S.C. 804(2)).
Executive Orders 12866 and 13563 direct agencies to assess all costs and benefits of available regulatory alternatives and, if regulation is necessary, to select those regulatory approaches that maximize net benefits (including potential economic, environmental, public health and safety, and other advantages; distributive impacts; and equity). Section 3(f) of Executive Order 12866 defines a “significant regulatory action” as any regulatory action that is likely to result in a rule that may: (1) have an annual effect on the economy of $100 million or more or adversely affect in a material way the economy, a sector of the economy, productivity, competition, jobs, the environment, public health or safety, or State, local, or tribal governments or communities; (2) create a serious inconsistency or otherwise interfere with an action taken or planned by another agency; (3) materially alter the budgetary impact of entitlements, grants, user fees, or loan programs or the rights and obligations of recipients thereof; or (4) raise novel legal or policy issues arising out of legal mandates, or the President's priorities.
A regulatory impact analysis (RIA) must be prepared for a regulatory action that is significant under section 3(f)(1) of E.O. 12866. Based on our estimates, the Office of Management and Budget's (OMB) Office of Information and Regulatory Affairs (OIRA) has determined this rulemaking is significant per section 3(f)(1). Accordingly, we have prepared a Regulatory Impact Analysis that to the best of our ability presents the costs and benefits of the rulemaking. Therefore, OMB has reviewed this final rule with comment period, and the Departments have provided the following assessment of their impact. In accordance with subtitle E of the Small Business Regulatory Enforcement Fairness Act of 1996 (also known as the Congressional Review Act), OIRA has also determined that this final rule meets the criteria set forth in 5 U.S.C. 804(2).
We estimate that the total increase in Federal Government expenditures under the OPPS for CY 2026, compared to CY 2025, due to the changes to the OPPS in this final rule with comment period, will be approximately $1.77 billion. Taking into account our estimated changes in enrollment, utilization, and case-mix for CY 2026 we estimate that the OPPS expenditures, including beneficiary cost-sharing, for CY 2026 will be approximately $101.0 billion, which is approximately $8.0 billion higher than estimated OPPS expenditures in CY 2025. Table 167 of this final rule with comment period displays the distributional impact of the CY 2026 changes in OPPS payment to various groups of hospitals and for CMHCs.
We note that under our proposed CY 2026 policy, drugs and biologicals are generally paid at ASP plus 6 percent, WAC plus 6 percent, or 95 percent of AWP, as applicable.
We estimate that the final update to the conversion factor will increase total OPPS payments by 2.6 percent in CY 2026. The final changes to the APC relative payment weights, the final changes to the wage indexes, the continuation of a payment adjustment for rural SCHs, including EACHs, and the final payment adjustment for cancer hospitals would not increase total OPPS payments because these changes to the OPPS are budget neutral. However, these updates would change the distribution of payments within the budget neutral system. We estimate that the total change in payments between CY 2025 and CY 2026, considering all budget-neutral payment adjustments, changes in estimated total outlier payments, the application of the frontier State wage adjustment, the payment adjustment for drug administration services furnished at excepted off campus PBDs, in addition to the application of the OPD fee schedule increase factor after all adjustments required by sections 1833(t)(3)(F), 1833(t)(3)(G), and 1833(t)(17) of the Act will increase total estimated OPPS payments by 2.4 percent. We note that, as previously discussed in section V.B.7 of this final rule with comment period, we reduce payments for non-drug items and services for hospitals for whom the annual reduction to payment amounts under § 419.32(b)(1)(iv)(B)(12) applies by 0.5 percentage points in CY 2026. We estimate that this reduction would reduce OPPS spending by $275 million in CY 2026.
We estimate the total increase (from changes to the ASC provisions in this final rule with comment period, as well as from enrollment, utilization, and case-mix changes) in Medicare expenditures (not including beneficiary cost-sharing) under the ASC payment system for CY 2026 compared to CY 2025, to be approximately $450 million. Tables 168 and 169 of this final rule with comment period display the redistributive impact of the CY 2026 changes regarding ASC payments, grouped by specialty area and then grouped by procedures with the greatest ASC expenditures, respectively.
C. Detailed Economic Analyses
1. Estimated Effects of OPPS Changes in This Final Rule With Comment Period
a. Limitations of Our Analysis
The distributional impacts presented here are the projected effects of the proposed CY 2026 policy changes on various hospital groups. We post our hospital-specific estimated payments for CY 2026 on the CMS website with the other supporting documentation for this final rule with comment period. To view the hospital-specific estimates, we refer readers to the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient. On the website, select “Regulations and Notices” from the left side of the page and then select “CMS-1834-FC” from the list of regulations and notices. The hospital-specific file layout and the hospital-specific file are listed with the other supporting documentation for this final rule. We show hospital-specific data only for hospitals whose claims were used for modeling the impacts shown in Table 167 of this final rule with comment period. We do not show hospital-specific impacts for hospitals whose claims we were unable to use. We refer readers to section II.A. of this final rule with comment period for a discussion of the hospitals whose claims we do not use for ratesetting or impact purposes. ( printed page 54060)
We estimate the effects of the individual policy changes by estimating payments per service, while holding all other payment policies constant. We use the best data available but do not attempt to predict behavioral responses to our policy changes in order to isolate the effects associated with specific policies or updates, but any policy that changes payment could have a behavioral response. In addition, we have not made any adjustments for future changes in variables, such as service volume, service-mix, or number of encounters.
b. Estimated Effects To Control Unnecessary Increases in the Volume of Outpatient Services Furnished in Excepted Off-Campus Provider Based Departments (PBDs)
In section X.A. of this final rule with comment period, we discuss our CY 2026 policy to control for unnecessary increases in the volume of outpatient services by paying for drug administration services furnished at an off-campus PBD at an amount equal to the site-specific PFS payment rate for nonexcepted items and services furnished by a nonexcepted off-campus PBD (the PFS payment rate). Specifically, we pay for HCPCS codes billed with modifier “PO” and assigned to and paid through drug administration APCs 5691 through 5694 at an amount equal to the site-specific PFS payment rate for nonexcepted items and services furnished by a nonexcepted off-campus PBD (the PFS payment rate). For a discussion of the PFS relativity adjuster that is used to pay for all drug administration services provided at all off-campus PBDs, we refer readers to the CY 2018 PFS final rule with comment period discussion (82 FR 53023 through 53024), as well as the CY 2019 PFS proposed rule.
To develop an estimated impact of this policy, we began with CY 2024 outpatient claims data used, for claim lines with HCPCS codes assigned for payment through drug administration APCs 5691 through 5694 that contained modifier “PO” because the presence of this modifier indicates that such claims were billed for services furnished by an off-campus department of a hospital paid under the OPPS. We then simulated payment for the remaining claim lines as if they were paid at the PFS-equivalent rate, removing a portion of the payment associated with rural Sole Community Hospitals based on our finalized exception for those hospitals. An estimate of the final policy that includes the effects of estimated changes in enrollment, utilization, and case-mix based on the FY 2026 Mid-Session review budget approximates the estimated decrease in total payments at $290 million, with Medicare OPPS payments decreasing by $220 million and beneficiary copayments decreasing by $70 million in CY 2026.
This estimate is utilized for the accounting statement displayed in Table 170 of this final rule with comment period because the impact of this final CY 2026 policy, which is not budget neutral, is combined with the impact of the OPD update, which is also not budget neutral, to estimate changes in Medicare spending under the OPPS as a result of the changes in this rule.
We note our estimates may differ from the actual effect of the proposed policy due to offsetting factors, such as changes in provider behavior. We note that by removing this payment differential that may influence site-of-service decision-making, we anticipate an associated decrease in the volume of drug administration services provided in the excepted off-campus PBD setting.
Comment: A commenter requested additional clarification regarding the figures in Table 111 of the CY 2026 OPPS/ASC proposed rule. They noted that while their estimates remained relatively similar to the Table in CY 2026, for the latter years of the table their estimates differed significantly.
Response: Table 166 of this final rule with comment period updates the estimated effect of changes related to the excepted off-campus drug administration payment policy. In the table displaying the effects of the final policy, the CY 2026 estimates are primarily isolated to effects on OPPS payments and are also what are included in Table 170 of this final rule with comment period. However, in CY 2027 and later years of Table 166 of this final rule with comment period, the savings estimates also include the impact of this policy on reducing Medicare Advantage payments starting in CY 2027, and this is why the estimates in Table 111 of the CY 2026 OPPS/ASC proposed rule were greater than the commenter's estimates beginning in CY 2027.
c. Estimated Effects of OPPS Changes on Hospitals
Table 167 shows the estimated impact of the final rule with comment period on hospitals. Historically, the first line of the impact table, which estimates the change in payments to all facilities, has always included cancer and children's hospitals, which are held harmless to their pre-Balanced Budget Act (BBA) amount. We also include CMHCs in the first line that includes all providers. We include a second line for all hospitals, excluding permanently held harmless hospitals and CMHCs. ( printed page 54061)
We present separate impacts for CMHCs in Table 167, and we discuss them separately below, because CMHCs are paid only for partial hospitalization and intensive outpatient program services under the OPPS and are a different provider type from hospitals. In the CY 2025 OPPS/ASC final rule with comment period (89 FR 94269 through 94270), we finalized paying CMHCs for partial hospitalization services and intensive outpatient services under APCs 5851 through 5854. For CY 2026, we are maintaining the same APC structure and revising our methodology for calculating APC payment rates. Specifically, we are finalizing our proposal to apply the 40 percent Medicare Physician Fee Schedule (MPFS) Relativity Adjuster to calculate PHP and IOP payment rates for CMHCs.
The estimated increase in the total payments made under the OPPS is determined largely by the increase to the conversion factor under the statutory methodology. The distributional impacts presented do not include assumptions about changes in volume and service-mix. The conversion factor is updated annually by the OPD fee schedule increase factor, as discussed in detail in section II.B. of this final rule with comment period.
Section 1833(t)(3)(C)(iv) of the Act provides that the OPD fee schedule increase factor is equal to the market basket percentage increase applicable under section 1886(b)(3)(B)(iii) of the Act, which we refer to as the IPPS market basket percentage increase. The final IPPS market basket percentage increase applicable to the OPD fee schedule for CY 2026 is 3.3 percent. Section 1833(t)(3)(F)(i) of the Act reduces that 3.3 percent by the productivity adjustment described in section 1886(b)(3)(B)(xi)(II) of the Act, which is a 0.7 percentage point for CY 2026 (which is also the productivity adjustment for FY 2026 in the FY 2026 IPPS/LTCH PPS proposed rule (90 FR 18257)) resulting in the proposed CY 2026 OPD fee schedule increase factor of 2.6 percent. We are using the OPD fee schedule increase factor of 2.6 percent in the calculation of the final CY 2026 OPPS conversion factor. Section 10324 of the Affordable Care Act, as amended by HCERA, further authorized additional expenditures outside budget neutrality for hospitals in certain frontier States that have a wage index less than 1.0000. The amounts attributable to this frontier State wage index adjustment are incorporated in the estimates in Table 167 of this final rule with comment period.
To illustrate the impact of the CY 2026 changes, our analysis begins with a baseline simulation model that uses the CY 2025 relative payment weights, the CY 2025 final OPPS wage indexes that include reclassifications, and the final CY 2025 conversion factor. Table 167 shows the estimated redistribution of the increase or decrease in payments for CY 2026 over CY 2025 payments to hospitals and CMHCs as a result of the following factors: the impact of the APC reconfiguration and recalibration changes between CY 2025 and CY 2026 (Column 2); the wage indexes and the provider adjustments (Column 3); the combined impact of all of the changes described in the preceding columns plus the 2.6 percent OPD fee schedule increase factor update to the conversion factor (Column 4); the additional estimated impact for the payment adjustment for drug administration furnished at excepted off campus PBDs (Column 5); the estimated impact taking into account all payments for CY 2026 relative to all payments for CY 2025, including the impact of changes in estimated outlier payments and changes to the pass-through payment estimate (Column 6).
We did not model an explicit budget neutrality adjustment for the rural adjustment for SCHs because we are maintaining the current adjustment percentage for CY 2026. Because the final updates to the conversion factor (including the update of the OPD fee schedule increase factor), the estimated cost of the rural adjustment, and the estimated cost of projected pass-through payment for CY 2026 are applied uniformly across services, observed redistributions of payments in the impact table for hospitals largely depend on the mix of services furnished by a hospital (for example, how the APCs for the hospital's most frequently furnished services would change), and the impact of the wage index changes on the hospital. However, total payments made under this system and the extent to which this final rule with comment period would redistribute money during implementation also will depend on changes in volume, practice patterns, and the mix of services billed between CY 2025 and CY 2026 by various groups of hospitals, which CMS cannot forecast.
Overall, we estimate that the final rates for CY 2026 will increase Medicare OPPS payments by an estimated 2.4 percent. Removing payments to cancer and children's hospitals because their payments are held harmless to the pre-OPPS ratio between payment and cost and removing payments to CMHCs results in an estimated 2.5 percent increase in Medicare payments to all other hospitals. These estimated payments will not significantly impact other providers. We note that providers not considered “new providers” for purposes of the 340b remedy offset would receive an adjustment to their OPPS payment rates.
Column 1: Total Number of Hospitals
The first line in Column 1 in Table 167 shows the total number of facilities (3,543), including designated cancer and children's hospitals and CMHCs, for which we were able to use CY 2024 hospital outpatient and CMHC claims data to model CY 2025 and CY 2026 payments, by classes of hospitals, for CMHCs and for dedicated cancer hospitals. We excluded all hospitals and CMHCs for which we could not plausibly estimate CY 2025 or CY 2026 payment and entities that are not paid under the OPPS. The latter entities include CAHs, IHS and tribal hospitals, and hospitals located in Guam, the U.S. Virgin Islands, Northern Mariana Islands, American Samoa, and the State of Maryland. This process is discussed in greater detail in section II.A. of this final rule with comment period. At this time, we are unable to calculate a DSH variable for hospitals that are not also paid under the IPPS because DSH payments are only made to hospitals paid under the IPPS. Hospitals for which we do not have a DSH variable are grouped separately and generally include freestanding psychiatric hospitals, rehabilitation hospitals, and long-term care hospitals. We show the total number of OPPS hospitals (3,439), excluding the hold harmless cancer and children's hospitals and CMHCs, on the second line of the table. We excluded cancer and children's hospitals because section 1833(t)(7)(D) of the Act permanently holds harmless cancer hospitals and children's hospitals to their “pre-BBA amount” as specified under the terms of the statute, and therefore, we removed them from our impact analyses. We show the isolated impact on the 34 CMHCs at the bottom of the impact table (Table 167) and discuss that impact separately below.
Column 2: APC Recalibration—All Changes
Column 2 shows the estimated effect of APC recalibration. Column 2 also reflects any changes in multiple procedure discount patterns or conditional packaging that occur as a result of the changes in the relative magnitude of payment weights. As a result of APC recalibration, we estimate that urban hospitals will experience a 0.1 increase, with the impact ranging ( printed page 54062) from no change to an increase of 0.2, depending on the number of beds. Rural hospitals will experience a decrease of 0.4 percent overall. Major teaching hospitals will experience no change.
Column 3: Wage Indexes and the Effect of the Provider Adjustments
Column 3 demonstrates the combined budget neutral impact of the APC recalibration, the updates for the wage indexes with the FY 2026 IPPS post-reclassification wage indexes, the rural adjustment, the frontier adjustment, and the cancer hospital payment adjustment. We modeled the independent effect of the budget neutrality adjustments and the OPD fee schedule increase factor by using the relative payment weights and wage indexes for each year and using a CY 2025 conversion factor that included the OPD fee schedule increase and a budget neutrality adjustment for differences in wage indexes.
Column 3 reflects the independent effects of the updated wage indexes, including the application of budget neutrality for the rural floor policy on a nationwide basis, as well as the final CY 2026 changes in wage index policy, discussed in section II.C. of this final rule with comment period. We did not model a budget neutrality adjustment for the rural adjustment for SCHs because we are continuing the rural payment adjustment of 7.1 percent to rural SCHs for CY 2026, as described in section II.E. of this final rule with comment period. We modeled a budget neutrality adjustment for the final cancer hospital payment adjustment because the final payment-to-cost ratio target for the cancer hospital payment adjustment in CY 2026 is 0.87, which is the same PCR target adopted in the CY 2025 OPPS/ASC final rule with comment period (89 FR 93979). We note that, in accordance with section 16002 of the 21st Century Cures Act, we apply a budget neutrality factor calculated as if the cancer hospital adjustment target payment-to-cost ratio was 0.88, not the 0.87 target payment-to-cost ratio we discuss in section II.F. of this final rule with comment period.
We modeled the independent effect of updating the wage indexes by varying only the wage indexes, holding APC relative payment weights, service-mix, and the rural adjustment constant and using the CY 2026 scaled weights and a CY 2025 conversion factor that included a budget neutrality adjustment for the effect of the changes to the wage indexes between CY 2025 and CY 2026.
Column 4: All Budget Neutrality Changes Combined With the Market Basket Update
Column 4 demonstrates the combined impact of all the final changes previously described and the update to the conversion factor of 2.6 percent. Overall, these changes would increase payments to urban hospitals by 2.8 percent and to rural hospitals by 2.4 percent. Rural sole community hospitals would receive an estimated increase of 2.8 percent while other rural hospitals would receive an estimated increase of 1.8 percent.
Column 5—Final Off-Campus PBD Drug Administration Payment Policy
Column 5 displays the estimated effect of our final CY 2026 policy to pay for drug administration services assigned to APCs 5691 through 5694 when billed with modifier “PO” at a PFS-equivalent rate. We note that the numbers provided in this column isolate the estimated effect of this final policy adjustment relative to the numerator of Column 4. Therefore, the numbers reported in Column 5 show how much of the difference between the estimates in Column 4 and the estimates in Column 6 are a result of the off-campus PBD drug administration policy.
Column 6: All Changes With Outlier—Final CY 2026 Update
Column 6 depicts the full impact of the final CY 2026 policies on each hospital group by including the effect of all changes for CY 2026 and comparing them to all estimated payments in CY 2025. Column 6 shows the combined budget neutral effects of Columns 2 and 3; the effect of the off-campus provider-based department drug administration policy; the OPD fee schedule increase; the impact of estimated OPPS outlier payments, as discussed in section II.G. of this final rule with comment period; the Hospital OQR Program payment reduction for the small number of hospitals in our impact model that failed to meet the reporting requirements (discussed in section XV. of this final rule with comment period); and other rule adjustments to the CY 2026 OPPS payments.
Of those hospitals that failed to meet the Hospital OQR Program reporting requirements for the full CY 2025 update (and assumed, for modeling purposes, to be the same number for CY 2026), we included 75 hospitals in our model because they had both CY 2024 claims data and recent cost report data. We estimate that the cumulative effect of all changes for CY 2026 would increase payments to all facilities by 2.4 percent for CY 2026. We modeled the independent effect of all changes in Column 6 using the final relative payment weights for CY 2025 and the proposed relative payment weights for CY 2026. We used the final conversion factor for CY 2025 of $89.169 and a CY 2026 conversion factor of $91.415 discussed in section II.B. of this final rule with comment period.
Column 6 contains simulated outlier payments for each year. We used the 1-year charge inflation factor used in the FY 2026 IPPS/LTCH PPS final rule (90 FR 37227) of 5.5 percent (1.05505) to increase charges on the CY 2024 claims, and we used the overall CCR in the July 2025 Outpatient Provider-Specific File (OPSF) to estimate outlier payments for CY 2025. Using the CY 2024 claims and a 5.5 percent charge inflation factor, we currently estimate that outlier payments for CY 2025, using a multiple threshold of 1.75 and a fixed-dollar threshold of $7,175, would be approximately 0.95 percent of total payments. The estimated current outlier payments of 0.95 percent are incorporated in the comparison in Column 5. We used the same set of claims and a charge inflation factor of 11.3 percent (1.11313) and the CCRs in the July 2025 OPSF, with an adjustment of 0.956081 (90 FR 37227), to reflect relative changes in cost and charge inflation between CY 2025 and CY 2026, to model the final CY 2026 outliers at 1.0 percent of estimated total payments using a multiple threshold of 1.75 and a fixed dollar threshold of $6,225. The charge inflation and CCR inflation factors are discussed in detail in the FY 2026 IPPS/LTCH PPS final rule (90 FR 37224 through 37228).
Overall, we estimate that facilities will experience an increase of 2.4 percent under this final rule in CY 2026 relative to total spending in CY 2025. This projected increase (shown in Column 6) of Table 167 of this final rule with comment period reflects the final 2.6 percent OPD fee schedule increase factor, adding the 0.05 difference in estimated outlier payments between CY 2025 (0.95 percent) and CY 2026 (1.0 percent), including the -0.3 percent decrease due to the payment adjusted for drug administration at off campus PBDs, plus 0.07 percent for the change in the pass-through payment estimate between CY 2025 and CY 2026. We estimate that the combined effect of all changes for CY 2026 will increase payments to urban hospitals by 2.6 percent. Overall, we estimate that rural hospitals will experience a 2.3 percent increase as a result of the combined effects of all the changes for CY 2026. ( printed page 54063)
Among hospitals, by teaching status, we estimate that the impacts resulting from the combined effects of all changes include an increase of 2.4 percent for major teaching hospitals and an increase of 2.6 percent for nonteaching hospitals. Minor teaching hospitals will experience an estimated increase of 2.7 percent.
In our analysis, we also have categorized hospitals by type of ownership. Based on this analysis, we estimate that voluntary hospitals will experience an increase of 2.5 percent, proprietary hospitals will experience an increase of 3.3 percent, and governmental hospitals will experience an increase of 2.1 percent.
Reduction for Providers Subject to the 340B Remedy Offset
In column 7 we have included additional information to account for estimated changes in the CY 2026 OPPS for providers subject to the 340B Remedy Offset.
( printed page 54064) ( printed page 54065) ( printed page 54066) ( printed page 54067)d. Estimated Effects of OPPS Changes on CMHCs
The last line of Table 167 demonstrates the isolated impact on CMHCs, which furnished only partial hospitalization and intensive outpatient program services under the OPPS during CY 2024. As discussed in section VIII.C. of this final rule with comment period, we are finalizing for CY 2026 to continue paying CMHCs using APCs 5851 through 5854. We modeled the impact of this APC policy, assuming CMHCs will continue to provide the same PHP and IOP care as seen in the CY 2024 claims used for ratesetting in the final rule with comment period. We did not exclude days with one or two services from our modeling for CY 2026, because our final rule policy would pay the per diem rate for APC 5853 for such days in CY 2026. As a result of the final PHP APC changes for CMHCs, we estimate that CMHCs will experience a 0.7 percent decrease in CY 2026 payments relative to their CY 2025 payments (shown in Column 2). For a detailed discussion of our final PHP and IOP policies, please see section VIII. of this final rule with comment period.
Column 3 shows the estimated impact of adopting the final FY 2026 wage index values, which result in an estimated increase of 0.2 percent to CMHCs.
Column 4 shows that combining the OPD fee schedule increase factor, along with the final changes in APC policy for CY 2026 and the final FY 2026 wage index updates, will result in an estimated increase of 2.1 percent.
e. Estimated Effect of OPPS Changes on Beneficiaries
For services for which the beneficiary pays a copayment of 20 percent of the payment rate, the beneficiary's payment would increase for services for which the OPPS payments would rise and decrease for services for which the OPPS payments would fall. For further discussion of the calculation of the national unadjusted copayments and minimum unadjusted copayments, we refer readers to section II.H. of this final rule with comment period. In all cases, section 1833(t)(8)(C)(i) of the Act limits beneficiary liability for copayment for a procedure performed in a year to the hospital inpatient deductible for the applicable year.
We estimate that the aggregate beneficiary coinsurance percentage would be approximately 18 percent for all services paid under the OPPS in CY 2026. The estimated aggregate beneficiary coinsurance reflects general system adjustments. We note that the individual payments, and therefore copayments, associated with services may differ based on the setting in which they are furnished. However, at the aggregate system level, we do not currently observe significant impact on beneficiary coinsurance as a result of those policies.
f. Estimated Effects of OPPS Changes on Other Providers
The relative payment weights and payment amounts established under the OPPS affect the payments made to ASCs, as discussed in section XIII. of this final rule with comment period. Hospitals, CMHCs, and ASCs will be affected by the changes in this final rule. Additionally, the payment policies we established for IOP services affect RHCs and FQHCs. These providers of IOP are not paid under the OPPS and are not included in the impact analysis shown in Table 167. However, the final payment amount for OPPS APC 5861 will affect payments to RHCs and FQHCs since under sections 1834(o)(5)(A) and 1834(y)(3)(A) of the Act payment for IOP services in these settings is required to be equal to the payment determined for IOP services in the hospital outpatient department.
g. Estimated Effects of OPPS Changes on the Medicare and Medicaid Programs
The effect of the update on the Medicare program is expected to be an increase of $1.77 billion in program payments for OPPS services furnished in CY 2026. The effect on the Medicaid program is expected to be limited to copayments that Medicaid may make on behalf of Medicaid recipients who are also Medicare beneficiaries. We estimate that the changes in this final rule with comment period will increase these Medicaid beneficiary payments by approximately $145 million in CY 2026. Currently, there are approximately 11.5 million dual-eligible beneficiaries, which represent approximately 40 percent of Medicare Part B fee-for-service beneficiaries. The impact on Medicaid was determined by taking 40 percent of the beneficiary cost-sharing impact. The national average split of Medicaid payments is 58 percent Federal payments and 42 percent State payments. Therefore, for the estimated $145 million Medicaid increase, approximately $85 million will be from the Federal Government and $60 million will be from State governments.
h. Alternative OPPS Policies Considered
Alternatives to the OPPS changes we proposed and the reasons for our selected alternatives are discussed throughout this final rule with comment period.
Alternatives Considered for the Final Payment Policy for Skin Substitute Products
We considered several alternatives to our policy to group skin substitute products based on FDA regulatory category. For example, we considered grouping skin substitute products based on their composition (for example, whether they are non-synthetic or synthetic) or by graft type ( e.g., allograft or xenograft). We also considered grouping all products together to set a single payment rate or creating new categories reflecting product cost, similar to our current payment policy. All of these alternatives considered would be implemented in a budget neutral manner and would involve unpackaging the current costs of skin substitute products from the application procedures, resulting in separate payments for skin substitute products under the OPPS and ASC. For a more detailed discussion of our CY 2026 payment policy for skin substitutes, please see section V. of this final rule with comment period.
2. Estimated Effects of CY 2026 ASC Payment System Changes
Most ASC payment rates are calculated by multiplying the ASC conversion factor by the ASC relative payment weight. As discussed fully in section XIII. of this final rule with comment period, we are setting the CY 2026 ASC relative payment weights by scaling the final CY 2026 OPPS relative payment weights by the final CY 2026 ASC scalar of 0.872. The estimated effects of the updated relative payment weights on payment rates are varied and are reflected in the estimated payments displayed in Tables 168 and 169.
Beginning in CY 2011, section 3401 of the Affordable Care Act requires that the annual update to the ASC payment system after application of any quality reporting reduction be reduced by a productivity adjustment. In CY 2019, we adopted a policy for the annual update to the ASC payment system to be the hospital market basket update for CY 2019 through CY 2023. In the CY 2024 OPPS/ASC final rule with comment period, we extended this 5-year interim period an additional 2 years through CYs 2024 and 2025. As discussed in further detail in section XIII. of this final rule with comment period, we are finalizing an extension of our utilization of the hospital market basket update as the update factor to the ASC payment system for 1 additional year (through CY 2026). Section 1886(b)(3)(B)(xi)(II) of the ( printed page 54068) Act defines the productivity adjustment to be equal to the 10-year moving average of changes in annual economy-wide private nonfarm business multifactor productivity (as projected by the Secretary for the 10-year period, ending with the applicable fiscal year, year, cost reporting period, or other annual period). For ASCs that fail to meet their quality reporting requirements, the CY 2026 payment determinations would be based on the application of a 2.0 percentage point reduction to the hospital market basket update for CY 2026. We calculated the final CY 2026 ASC conversion factor by adjusting the CY 2025 ASC conversion factor ($54.895) by 1.0000 to account for changes in the pre-floor and pre-reclassified hospital wage indexes between CY 2025 and CY 2026, which includes our policy to limit wage index declines of greater than 5 percent, and by applying the CY 2026 hospital market basket update factor of 2.6 percent (which is equal to the final inpatient hospital market basket percentage increase of 3.3 percent reduced by a productivity adjustment of 0.7 percentage point). The final CY 2026 ASC conversion factor is $56.322 for ASCs that successfully meet the quality reporting requirements.
a. Limitations of Our Analysis
Presented here are the projected effects of the final changes for CY 2026 on Medicare payment to ASCs. A key limitation of our analysis is our inability to predict changes in ASC service-mix between CY 2024 and CY 2026 with precision. We believe the net effect on Medicare expenditures resulting from the final CY 2026 changes would be small in the aggregate for all ASCs. However, such changes may have differential effects across surgical specialty groups, as ASCs continue to adjust to the payment rates based on the policies of the revised ASC payment system. We are unable to accurately project such changes at a disaggregated level. Clearly, individual ASCs would experience changes in payment that differ from the aggregated estimated impacts presented below.
b. Estimated Effects of ASC Payment System Policies on ASCs
Some ASCs are multispecialty facilities that perform a wide range of surgical procedures from excision of lesions to hernia repair to cataract extraction; others focus on a single specialty and perform only a limited range of surgical procedures, such as ophthalmology, digestive system, or orthopedic procedures. The combined effect of the final update to the payments on an individual ASC would depend on a number of factors, including, but not limited to, the mix of services the ASC provides, the volume of specific services provided by the ASC, the percentage of its patients who are Medicare beneficiaries, and the extent to which an ASC provides different services in the coming year. The following discussion includes tables that display estimates of the impact of the final CY 2026 updates to the ASC payment system on Medicare payments to ASCs, assuming the same mix of services, as reflected in our CY 2024 claims data. Table 168 depicts the estimated aggregate percent change in payment by surgical specialty or ancillary items and services group by comparing estimated CY 2025 payments to estimated CY 2026 payments, and Table 169 shows a comparison of estimated CY 2025 payments to estimated CY 2026 payments for items and procedures that we estimate would receive the most Medicare payment in CY 2025.
In Table 168, we have aggregated the surgical HCPCS codes by specialty group, grouped all HCPCS codes for covered ancillary items and services into a single group, and then estimated the effect on aggregated payment for surgical specialty and ancillary items and services groups. The groups are sorted for display in descending order by estimated Medicare program payment to ASCs. The following is an explanation of the information presented in Table 168.
- Column 1—Surgical Specialty or Ancillary Items and Services Group indicates the surgical specialty into which ASC procedures are grouped and the ancillary items and services group, which includes all HCPCS codes for covered ancillary items and services. To group surgical procedures by surgical specialty, we used the CPT code range definitions and Level II HCPCS codes and Category III CPT codes, as appropriate, to account for all surgical procedures to which the Medicare program payments are attributed.
- Column 2—Estimated CY 2025 ASC Payments were calculated using CY 2024 ASC utilization data (the most recent full year of ASC utilization) and CY 2025 ASC payment rates. The surgical specialty groups are displayed in descending order based on estimated CY 2025 ASC payments.
- Column 3—Estimated CY 2026 Percent Change is the aggregate percentage increase or decrease in Medicare program payment to ASCs for each surgical specialty or ancillary items and services group that is attributable to final updates to ASC payment rates for CY 2026 compared to CY 2025.
As shown in Table 168, for the six specialty groups that account for the most ASC utilization and spending, we estimate that the final update to ASC payment rates for CY 2026 would result in a 4 percent increase in aggregate payment amounts for eye and ocular adnexa procedures, a 2 percent increase in aggregate payment amounts for musculoskeletal system procedures, a 2 percent increase in aggregate payment amounts for nervous system procedures, a 3 percent increase in aggregate payment amounts for digestive system procedures, a 5 percent increase in aggregate payment amounts for cardiovascular system procedures, and a 12 percent increase in aggregate payment amounts for genitourinary system procedures. We note that these changes can be a result of different factors, including updated data, payment weight changes, and changes in policy. In general, spending in each of these categories of services is increasing due to the 2.6 percent payment rate update. After the payment rate update is accounted for, aggregate payment increases or decreases for a category of services can be higher or lower than a 2.6 percent increase, depending on if payment weights in the OPPS APCs that correspond to the applicable services increased or decreased or if the most recent data show an increase or a decrease in the volume of services performed in an ASC for a category. For example, we estimate a 4 percent increase in eye surgical procedure payments. The increase in expenditures for eye surgical procedures is largely a result of the 2.6 percent hospital market basket update. The large increases in genitourinary procedure expenditures are a result of higher APC level assignment in the OPPS of newer prostate biopsy procedure codes compared to prior prostate biopsy procedure codes. For estimated changes for selected procedures, we refer readers to Table 168.
( printed page 54069)Table 169 shows the estimated impact of the updates to the revised ASC payment system on aggregate ASC payments for selected surgical procedures during CY 2026. The table displays 30 of the procedures receiving the greatest estimated CY 2025 aggregate Medicare payments to ASCs. The HCPCS codes are sorted in descending order by estimated CY 2025 program payment.
- Column 1-CPT/HCPCS code.
- Column 2-Short Descriptor of the HCPCS code.
- Column 3-Estimated CY 2025 ASC Payments were calculated using CY 2024 ASC utilization (the most recent full year of ASC utilization) and the CY 2025 ASC payment rates. The estimated CY 2025 payments are expressed in millions of dollars.
- Column 4-Estimated CY 2026 Percent Change reflects the percent differences between the estimated ASC payment for CY 2025 and the estimated payment for CY 2026 based on the final update.
c. Estimated Effects of ASC Payment System Policies on Beneficiaries
We estimate that the CY 2026 update to the ASC payment system will be generally positive (that is, result in lower cost-sharing) for beneficiaries with respect to the procedures we are finalizing to add to the ASC CPL for CY 2026. First, other than certain preventive services where coinsurance and the Part B deductible are waived to comply with sections 1833(a)(1) and (b) of the Act, the ASC coinsurance rate for all procedures is 20 percent. This contrasts with procedures performed in HOPDs under the OPPS, where the beneficiary is responsible for copayments that range from 20 percent to 40 percent of the procedure payment (other than for certain preventive services), although the majority of HOPD procedures have a 20-percent copayment. Second, in almost all cases, the ASC payment rates under the ASC payment system are lower than payment rates for the same procedures under the OPPS. Therefore, the beneficiary coinsurance amount under the ASC payment system will usually be less than the OPPS copayment amount for the same services. (The only exceptions will be if the ASC coinsurance amount exceeds the hospital inpatient deductible since the statute requires that OPPS copayment amounts not exceed the hospital inpatient deductible. Therefore, in limited circumstances, the ASC coinsurance amount may exceed the hospital inpatient deductible and, therefore, the OPPS copayment amount for similar services.) Beneficiary coinsurance for services migrating from physicians' offices to ASCs may decrease or increase under the ASC payment system, depending on the particular service and the relative payment amounts under the MPFS compared to the ASC. While the ASC payment system bases most of its payment rates on hospital cost data used to set OPPS relative payment weights, services that are performed a majority of the time in a physician office are generally paid the lesser of the ASC amount according to the standard ASC ratesetting methodology or at the nonfacility practice expense-based amount payable under the PFS. For those additional procedures that we finalized to designate as office-based in CY 2026, the beneficiary coinsurance amount under the ASC payment system generally will be no greater than the ( printed page 54071) beneficiary coinsurance under the PFS because the coinsurance under both payment systems generally is 20 percent (except for certain preventive services where the coinsurance is waived under both payment systems).
Accounting Statements and Tables for OPPS and ASC Payment System
As required by OMB Circular A-4 (available on the Office of Management and Budget website at https://trumpwhitehouse.archives.gov/sites/whitehouse.gov/files/omb/circulars/A4/a-4.pdf), we have prepared accounting statements to illustrate the impacts of the OPPS and ASC changes in this final rule with comment period. The first accounting statement, Table 170, illustrates the classification of expenditures for the CY 2026 estimated hospital OPPS incurred benefit impacts associated with the final CY 2026 OPD fee schedule increase and the final policy for drug administration services furnished at excepted off-campus PBDs. The second accounting statement, Table 171, illustrates the classification of expenditures associated with the 2.6 percent CY 2026 update to the ASC payment system, based on the provisions of the final rule with comment period and the baseline spending estimates for ASCs. Both tables classify most estimated impacts as transfers. The third accounting statement, Table 172, outlines the one-time burden associated with the HPT requirements we are finalizing in this rule.
We did not receive public comments on this provision, and therefore, we are finalizing as proposed.
3. Effects of Changes in Requirements for the Hospital Outpatient Quality Reporting (OQR) Program
a. Background
We refer readers to the CY 2025 OPPS/ASC final rule with comment period (89 FR 94561 and 94562) for the previously estimated effects of changes to the Hospital OQR Program for the CY 2025 reporting period and subsequent years. Of the 3,014 hospital outpatient departments (HOPDs) that met eligibility requirements for the CY 2025 payment determination for the Hospital OQR Program, we determined that 42 HOPDs did not meet the program requirements to receive the full annual Outpatient Department (OPD) fee schedule increase factor while an additional 54 HOPDs elected not to participate.
b. Impact of CY 2026 OPPS/ASC Final Rule Policies
We are finalizing: (1) removal of the COVID-19 Vaccination Coverage Among Healthcare Personnel (HCP) measure beginning with the CY 2024 reporting period/CY 2026 payment determination; (2) removal of the Hospital Commitment to Health Equity (HCHE) measure beginning with the CY 2025 reporting period/CY 2027 payment determination; (3) removal of the Screening for Social Drivers of Health (SDOH) measure beginning with the CY 2025 reporting period; (4) removal of the Screen Positive Rate for SDOH measure beginning with the CY 2025 reporting period; (5) adoption of the Emergency Care Access & Timeliness electronic clinical quality measure (eCQM) with voluntary reporting for the CY 2027 reporting period followed by mandatory reporting beginning with the CY 2028 reporting period/CY 2030 payment determination; (6) removal of the Median Time from Emergency Department (ED) Arrival to ED Departure for Discharged ED Patients (Median Time for Discharged ED Patients) measure beginning with the CY 2028 reporting period/CY 2030 payment determination; (7) removal of ( printed page 54072) the Left Without Being Seen (LWBS) measure beginning with the CY 2028 reporting period/CY 2030 payment determination; and (8) modification of the Excessive Radiation Dose or Inadequate Image Quality for Diagnostic Computed Tomography (CT) in Adults (Hospital Level—Outpatient) eCQM (Excessive Radiation eCQM) from mandatory reporting beginning with the CY 2027 reporting period to continue voluntary reporting in the CY 2027 reporting period and subsequent years.
In section XV.D. of this final rule with comment period, we updated our Extraordinary Circumstances Exception (ECE) policy for the Hospital OQR Program. This update will explicitly include extensions as a type of extraordinary circumstances relief option, in addition to exceptions. Because the process for requesting or granting an ECE will remain the same as the current ECE process, these updates will not affect burden associated with the submission of the ECE form.
We refer readers to section “XXIII.A. Collection of Information” of this final rule with comment period for a detailed discussion of the calculations estimating the changes to the information collection and reporting burden for finalized data requirements under the Hospital OQR Program for the estimated 3,200 program-eligible HOPDs. As shown in summary tables in section XXIII.A.10, we estimate a total information collection and reporting burden decrease of 6,924,988 hours at a savings of $178,884,367 annually associated with our proposals for the CY 2028 reporting period/CY 2030 payment determination and subsequent years compared to our currently approved information collection burden estimates under OMB control number 0938-1109 (expiration date January 31, 2026). We also estimate a decrease of between 25,600 hours at a savings of $1,446,400 and 28,800 hours at a savings of $1,687,680 in information collection burden associated with the removal of the COVID-19 Vaccination Coverage Among HCP measure compared to the currently approved information collection burden estimates and under OMB control number 0920-1317 (expiration date January 31, 2028).
In section XV.B.1. of this final rule with comment period, we adopted the Emergency Care Access & Timeliness eCQM. Similar to the effects associated with the ST-Segment Elevation Myocardial Infarction (STEMI) eCQM finalized in the CY 2022 OPPS/ASC final rule with comment period (86 FR 63984 and 63985), we believe that costs associated with adoption of eCQMs are multifaceted and include not only the burden associated with reporting but also the costs associated with implementing and maintaining program requirements, such as maintaining measure specifications in hospitals' electronic health record (EHR) systems for the eCQMs used in the Hospital OQR Program.
In section XV.B.2. of this final rule with comment period, we finalized the removal of the Median Time from ED Arrival to ED Departure for Discharged ED Patients and LWBS measures in coordination with the adoption of the Emergency Care Access & Timeliness eCQM. Because these measures will be replaced by the Emergency Care Access & Timeliness eCQM, we believe HOPDs will be positively impacted by the decreased effort required to report one eCQM rather than one chart-abstracted measure and one web-based measure.
In section XV.B.3, of this final rule with comment period, we modified the reporting requirements for the Excessive Radiation eCQM by maintaining voluntary reporting instead of mandatory reporting of the measure, beginning with the CY 2027 reporting period. In the CY 2024 OPPS/ASC final rule with comment period, we stated that for the Excessive Radiation eCQM, HOPDs may incur costs associated with implementing and maintaining program requirements, such as maintaining measure specifications in hospitals' electronic health record (EHR) systems (88 FR 82167). HOPDs would be required to link their EHR and PACS data to their chosen vendor's translation software to calculate the measure, which we estimate would require no more than 1 hour to complete. In section XXIII.A.9., we estimate that 20 percent of HOPDs will report this measure annually. Because some HOPDs may only elect to report this eCQM for some reporting periods, we are unable to assume that 80 percent of HOPDs will not report this measure in any reporting period. However, for HOPDs who elect not to report this eCQM in any reporting period, the modification will result in a savings of no more than 1 hour and $55 (1 hour × $55.06) as well as any costs that would be associated with implementing and maintaining program requirements specific to this eCQM.
Regarding the remaining policies, we do not believe these policies will result in any additional economic impact beyond those discussed in section “XXIII.A. Collection of Information” of this final rule with comment period.
We did not receive public comments on the financial impact of our proposals.
4. Effects of Changes in Requirements for the Rural Emergency Hospital Quality Reporting (REHQR) Program
a. Background
We refer readers to the CY 2025 OPPS/ASC final rule with comment period (89 FR 94562 and 94563) for the previously estimated effects of changes to the REHQR Program for the CY 2025 reporting period and subsequent years. For the CY 2026 reporting period, we have estimated there will be 38 REHs required to report under the REHQR Program based on hospital conversions as of April 11, 2025. We use this number of REHs for our impact analyses knowing that more jurisdictions will pass or amend necessary legislation enabling transitions, acknowledging that the number of conversions could be less than or significantly greater than this estimate with time.
b. Impact of CY 2026 OPPS/ASC Final Rule Policies
We are finalizing: (1) removal of the HCHE measure beginning with the CY 2025 reporting period/CY 2027 program determination; (2) removal of the Screening for SDOH measure beginning with the CY 2025 reporting period/CY 2027 program determination; (3) removal of the Screen Positive Rate for SDOH measure beginning with the CY 2025 reporting period/CY 2027 program determination; and (4) adoption of the Emergency Care Access & Timeliness eCQM beginning with the CY 2027 reporting period/CY 2029 program determination as optional in lieu of reporting the chart-abstracted Median Time for Discharged ED Patients. In section XVI.D. of this final rule with comment period, we also updated our ECE policy for the REHQR Program. This update will explicitly include extensions as a type of extraordinary circumstances relief option, in addition to exceptions. Because the process for requesting or granting an ECE will remain the same as the current ECE process, these updates will not affect burden associated with the submission of the ECE form.
We refer readers to section “XXIII.B. Collection of Information” of this final rule with comment period for a detailed discussion of the calculations estimating the changes to the information collection and reporting burden for finalized data requirements under the REHQR Program for the estimated 38 REHs. As shown in summary tables in section XXIII.B.6. of this final rule with comment period, we estimate a total information collection and reporting burden decrease of 14,813 hours at a savings of $380,235 annually associated ( printed page 54073) with our policies for the CY 2027 reporting period/CY 2029 program determination and subsequent years compared to our currently approved information collection burden estimates under OMB control number 0938-1454 (expiration date April 30, 2027).
In section XVI.B.1. of this final rule with comment period, we adopted the Emergency Care Access & Timeliness eCQM. Similar to the effects associated with the STEMI eCQM finalized for the Hospital OQR Program in the CY 2022 OPPS/ASC final rule with comment period (86 FR 63984 and 63985), we believe that costs associated with adoption of eCQMs are multifaceted and include not only the burden associated with reporting but also the costs associated with implementing and maintaining program requirements, such as maintaining measure specifications in REHs' EHR systems for the eCQMs used in the REHQR Program. Because REHs will have the option to report the Emergency Care Access & Timeliness eCQM or the more burdensome Median Time for Discharged ED Patients measure, we believe REHs will be positively impacted by the decreased effort required to report the Emergency Care Access & Timeliness eCQM in the long-term following initial implementation in the EHR.
Regarding the remaining policies, we do not believe these policies will result in any additional economic impact beyond those discussed in section “XXIII.B. Collection of Information” of this final rule with comment period.
We did not receive public comments on the financial impact of our proposals.
5. Effects of Changes in Requirements for the Ambulatory Surgical Center Quality Reporting (ASCQR) Program
a. Background
We refer readers to the CY 2025 OPPS/ASC final rule with comment period (89 FR 94563) for the previously estimated effects of changes to the ASCQR Program for the CY 2025 reporting period and subsequent years. Based on the most recent analysis of the CY 2025 payment determination data, we found that, of the 6,012 ambulatory surgical centers (ASCs) that were actively billing Medicare, 4,271 were required to participate in the ASCQR Program. Of the 1,741 ASCs not required to participate in the program, 319 ASCs did so and met full requirements. On this basis, we estimate that 4,590 ASCs (4,271 + 319) will submit data for the ASCQR Program for the CY 2026 reporting period and subsequent years unless otherwise noted. We note that this estimate is an increase of 115 ASCs from our estimate of 4,475 provided in the CY 2025 OPPS/ASC final rule with comment period (89 FR 94563) due to more recent data analysis regarding numbers of eligible ASCs.
b. Impact of CY 2025 OPPS/ASC Final Rule Policies
We are finalizing: (1) removal of the COVID-19 Vaccination Coverage Among HCP Measure beginning with the CY 2024 reporting period/CY 2026 payment determination; (2) removal of the Facility Commitment to Health Equity (FCHE) measure beginning with the CY 2025 reporting period/CY 2027 payment determination; (3) removal of the Screening for SDOH measure beginning with the CY 2025 reporting period/CY 2027 payment determination; and (4) removal of the Screen Positive Rate for SDOH measure beginning with the CY 2025 reporting period/CY 2027 payment determination.
In section XVII.D. of this final rule with comment period, we also updated our ECE policy for the ASCQR Program. This update will explicitly include extensions as a type of extraordinary circumstances relief option, in addition to exceptions. Because the process for requesting or granting an ECE will remain the same as the current ECE process, these updates will not affect burden associated with the submission of the ECE form.
We refer readers to section “XXIII.C. Collection of Information” of this final rule with comment period for a detailed discussion of the calculations estimating the changes to the information collection and reporting burden for proposed data requirements under the ASCQR Program for the estimated 4,590 program-eligible ASCs. As shown in summary tables in section XXIII.C.7. of this final rule with comment period, we estimate a total information collection and reporting burden decrease of 731,340 hours at a cost of $18,811,786 annually associated with our policies for the CY 2029 reporting period/CY 2031 payment determination and subsequent years compared to our currently approved information collection burden estimates under OMB control number 0938-1270 (expiration date July 31, 2027). We also estimate a decrease of between 36,720 hours at a savings of $2,074,680 and 41,310 hours at a savings of $2,420,766 in information collection burden associated with the removal of the COVID-19 Vaccination Among HCP measure compared to the currently approved information collection burden estimates and under OMB control number 0920-1317 (expiration date January 31, 2028).
We do not believe these policies will result in any additional economic impact beyond those discussed in section “XXIII.C. Collection of Information” of this final rule with comment period.
We did not receive public comments on the financial impact of our proposals.
6. Effects of Requirements for the Overall Hospital Quality Star Rating
a. Background
In section XVIII. Overall Hospital Quality Star Rating Modification to Emphasize the Safety of Care Measure Group of this final rule with comment period, we discussed our finalized policies as they relate to the Overall Hospital Quality Star Rating methodology. The Overall Hospital Quality Star Rating uses measures that are publicly reported on the provider comparison tool on Medicare.gov ( https://www.medicare.gov/care-compare/) under the public reporting authority of each individual hospital program furnishing measure data. The burden associated with measures included in the Overall Hospital Quality Star Rating, including forms used to request withholding of publicly reported measure data and the Overall Hospital Quality Star Rating (for CAHs), is already captured in respective hospital programs' burden estimates and represents no increased information collection burden to hospitals.
b. Impact of CY 2026 OPPS/ASC Proposed Rule Policies
In this CY 2026 OPPS/ASC proposed rule, we are using the most recent data from the Bureau of Labor Statistics, which reflects a median hourly wage of $24.16 per hour for a Medical Records and Health Information Technician professional.[534] We calculate the cost of overhead, including fringe benefits, at 100 percent of the hourly wage estimate, consistent with the previous year. This is necessarily a rough adjustment, both because fringe benefits and overhead costs vary significantly from employer- to-employer and because methods of estimating these costs vary widely from study-to-study. Therefore, we believe that doubling the hourly wage rate ($24.16 × 2 = $48.32) to estimate total cost is a reasonably accurate estimation method. Accordingly, we calculate the cost burden to hospitals using a wage ( printed page 54074) plus benefits estimate of $48.32 per hour. We estimate that the non-information collection burden associated with all non-Veterans Health Administration (VHA) hospitals reviewing their Overall Hospital Quality Star Rating preview report prior to public reporting to be 2 hours per hospital, which includes time to review the report and ask any questions about the calculation necessary to increase comprehension. Estimating that approximately 4,600 hospitals would receive an Overall Hospital Quality Star Rating hospital specific report (HSR), regardless of if they meet the reporting thresholds to be assigned a star rating, we estimate the overall non- information collection burden to be $444,544 annually ($48.32 × 2 hours per preview report × once per year × 4,600 hospitals). For CAHs specifically, which are included in the estimate above, we estimate that 1,300 CAHs would be eligible for an Overall Hospital Quality Star Rating, which represents a burden of $125,632 annually (1,300 CAHs × 2 hours per preview report × once per year × $48.32).
Within this rule, for CY 2026 Overall Hospital Quality Star Rating and subsequent years, we finalized our policy to make the following two-stage methodological updates to emphasize the importance of the Safety of Care measure group to the Overall Hospital Quality Star Rating methodology: (1) implement a 4-star cap for poor performance in the Safety of Care measure group (lowest performing quartile) for the 2026 Star Rating, and (2) implement a blanket 1-star reduction for poor performance in the Safety of Care measure group (lowest performing quartile) for the 2027 Star Rating and thereafter.
To simulate the impact of the proposed Overall Hospital Quality Star Rating methodology, we used the July 2024 refresh of the Overall Hospital Quality Star Rating (the most recent publicly released results as of the writing of the proposal) to describe the overall distribution and reclassification of the Overall Hospital Quality Star Rating across different types of hospitals. The update to the Overall Hospital Quality Star Rating methodology in CY 2026 that will limit hospitals in the lowest quartile of Safety of Care (based on at least three measure scores) to a maximum of 4 out of 5 stars (Stage 1 methodological change) would have resulted in 14 (0.30 percent) hospitals receiving a lower Overall Hospital Quality Star Rating in the July 2024 simulation. The update to the Overall Hospital Quality Star Rating methodology beginning in CY 2027 that will reduce the Overall Hospital Quality Star Rating of any hospital in the lowest quartile of Safety of Care (based on at least three measure scores) by 1 star, to a minimum 1-star rating (Stage 2 methodological change) would have resulted in 459 (9.90 percent) hospitals receiving a lower Overall Hospital Quality Star Rating (in the simulation).
Utilizing the Stage 1 methodology update, fewer hospitals would have received a different Overall Hospital Quality Star Rating and changes in the Overall Hospital Quality Star Rating are less easily attributed to specific hospital characteristics, as very few hospitals of any type would be affected. In contrast, the Stage 2 methodological update resulted in teaching hospitals, non-safety-net hospitals, VHA hospitals, non-CAHs, large hospitals (100+ beds), and non-specialty hospitals being more likely to receive a lower Overall Hospital Quality Star Rating in the July 2024 simulation. (Table 173).
( printed page 54075) ( printed page 54076)7. Effects of a Final Market-Based MS-DRG Relative Weight Methodology
In section XX. of this final rule with comment period we are finalizing the adoption of a market-based methodology for determining the MS-DRG relative weights beginning in FY 2029 utilizing the median payer-specific negotiated charge information we will be collecting on the cost report. More specifically, the Medicare cost report will collect the median of the payer-specific negotiated charge that the hospital has negotiated with all of its MAOs, by MS-DRG, effective for cost reporting periods ending on or after January 1, 2026.
We note that the estimated total annual burden hours for this final policy are as follows: 3,038 hospitals times 20 hours per hospital equals 60,760 annual burden hours and $4,857,458.20. We refer readers to section XXIII.E. of this final rule with comment period for further analysis of this assessment.
Under the finalized methodology, we will apply a budget neutrality factor to ensure that the overall payment impact of any MS-DRG relative weight changes is budget neutral, as required by section 1886(d)(4)(C)(iii) of the Act and consistent with our current practice.
As discussed in the CY 2026 OPPS/ASC proposed rule, similar to our discussion in the economic analysis section of the FY 2021 IPPS/LTCH PPS final rule (85 FR 59089 through 59090) regarding the market-based MS-DRG relative weight methodology, once we have access to the weighted payer-specific negotiated charge information at the MS-DRG level from the cost reports we would be able to more precisely estimate the payment impact of adopting this market-based MS-DRG relative weight methodology for payments beginning in FY 2029. We also stated that we intended to provide these more precise estimates prior to the FY 2029 effective date. However, to explore the potential impacts more generally, in the CY 2026 OPPS/ASC proposed rule we explained that we conducted a literature search to compare the payment rates of Medicare FFS, MA organizations, and other commercial payers, which is discussed in section XX.B. of this final rule with comment period. As discussed in that section, the payer-specific charges negotiated between hospitals and MAOs are generally well-correlated with Medicare IPPS payment rates, although in the future this may change over time for some services. As discussed in response to comments on the impacts in section XX.C.3. of this final rule with comment period, we continue to believe that if market-based data (median payer-specific negotiated charges for MA organizations) are incorporated into the calculation of the MS-DRG relative weights, initially there will be limited impact on the relative weights given the current similarity between MA organization rates and Medicare FFS rates. To the extent the data shows that there would be more than a limited impact on the relative weights initially, which we do not believe will be the case, we intend to provide an opportunity for the public to review the data we collect prior to implementation. This will allow for additional discussions, public review, and feedback on utilizing this market-based data in the MS-DRG relative weight methodology. We will continue to provide impact analyses of changes in the MS-DRG relative weights in the annual IPPS rulemaking. Also, as discussed in the CY 2026 OPPS/ASC proposed rule we expect for some period of time following implementation of this market-based MS-DRG relative weight methodology to continue to estimate and publicly provide the MS-DRG relative weights calculated using the current methodology for informational purposes.
As previously noted, once we have access to the payer-specific negotiated charge information at the MS-DRG level, we can more precisely estimate the potential payment impact, which we intend to do in future rulemaking, prior to the FY 2029 effective date of the market-based MS-DRG relative weight methodology. As under the current methodology, the impact of any MS- DRG relative weight changes on an individual hospital would depend on the mix of services provided by that particular hospital.
8. Graduate Medical Education Accreditation
In section XXI. of this final rule with comment period, we are finalizing, with modification, our proposal that accreditors may not require as part of accreditation or otherwise encourage institutions to put in place diversity, equity, and inclusion programs that encourage unlawful discrimination on the basis of race or other violations of Federal law. Specifically, we are changing the definition of “approved medical residency program” and equivalent terms in the regulations to state that accrediting organizations may not use accreditation criteria that promote or encourage discrimination on the basis of race, color, national origin, sex, age, disability, or religion, including the use of those characteristics or intentional proxies for those characteristics as a selection criterion for employment, program participation, resource allocation, or similar activities, opportunities, or benefits. The effective date of this policy is January 1, 2026. We believe there is no financial impact associated with this policy because, as of January 1, 2026, we do not expect that current or future accrediting organizations would continue to or newly require or otherwise encourage institutions to put in place diversity, equity, and inclusion programs that encourage unlawful discrimination on the basis of race or other violations of Federal law.
9. Effects Relating to Hospital Price Transparency
a. Background
Since the January 1, 2021, effective date of the CY 2020 Hospital Price Transparency (HPT) final rule, hospitals have been required to make their standard charges available to the public. Consistent with Executive Order 14221, and to further advance the goals articulated in previous HPT rulemaking of requiring hospitals to make meaningful price information available to consumers, employers, policymakers, and others to support a more competitive, innovative, and affordable healthcare system, we proposed several updates to the HPT regulations in the CY 2026 OPPS/ASC proposed rule to enhance the clarity and standardization of hospital disclosure of standard charges. We are finalizing with modifications revisions to § 180.20 to add definitions for “tenth (10th) percentile allowed amount”, “median allowed amount”, and “ninetieth (90th) percentile allowed amount,” which are values a hospital will encode when a payer-specific negotiated charge is based on a percentage or algorithm, to more accurately reflect the distribution of actual amounts that a hospital has received for an item or service. In tandem with that, we are finalizing revisions to § 180.50 to remove the requirement for hospitals to disclose the estimated allowed amount, and, instead, require hospitals to disclose the 10th percentile, median, and 90th percentile allowed amounts, as well as the count of allowed amounts, in MRFs when payer-specific negotiated charges are based on percentages or algorithms. We are also finalizing, with modification, our proposal to require that hospitals use electronic data interchange (EDI) 835 electronic remittance advice (ERA) transaction data or an alternative, equivalent source of remittance data to ( printed page 54077) calculate and encode the allowed amounts. We are finalizing our proposals, with some modifications, to require that hospitals comply with specific instructions regarding the methodology, including a lookback period, that must be used to calculate those amounts.
Additionally, we are finalizing with modifications our proposed revisions to § 180.50 to require hospitals to attest that in the MRF, to the best of the hospital's knowledge and belief, the hospital has included all applicable standard charge information in accordance with the requirements of this section and the information encoded is true, accurate, and complete as of the date in the file. We are finalizing our proposal that hospitals attest in the MRF that the hospital has included all applicable payer-specific negotiated charges as dollars that can be expressed as a dollar amount, and for payer-specific negotiated charges that are not knowable in advance or cannot be expressed as a dollar amount, the hospital has provided in the MRF all necessary information available to the hospital for the public to be able to derive a dollar amount, including, but not limited to, the specific fee schedule or components referenced in such percentage, algorithm, or formula. Furthermore, we are finalizing our proposal that hospitals encode in the MRF the name of the hospital chief executive officer, president, or senior official designated to oversee the encoding of true, accurate, and complete data. In addition, to advance the comparability of HPT data with other healthcare data, we are finalizing our proposal to require that hospitals encode their organizational, or Type 2, National Provider Identifier(s) (NPIs) in the MRFs.
Finally, to encourage faster resolution and payment of CMPs, and in exchange for a hospital's admission of having violated HPT requirements, we are finalizing our proposal with clarifying edits to update § 180.90 to reduce the amount of a CMP by 35 percent, under certain conditions, when a hospital waives its right to an ALJ hearing. These changes aim to improve transparency in hospital pricing, facilitate efficient enforcement of the HPT requirements, and empower consumers with actionable pricing information.
We are finalizing an effective date of January 1, 2026, for the revisions at § 180.50, including removal of the estimated allowed amount, disclosure of the 10th percentile, median, 90th percentile allowed amounts and the count of allowed amounts, the attestation requirements, and inclusion of NPIs. However, we will delay enforcement of these finalized policies until April 1, 2026. We are finalizing at new § 180.90(c)(4) that, effective beginning January 1, 2026, the amount of a CMP would be reduced by 35 percent should a hospital submit to CMS a written notice requesting to waive its right to a hearing under § 180.100 within 30 calendar days of the date of the notice of imposition of the CMP.
b. Overall Estimated Burden on Hospitals Due to HPT Requirements
To analyze the costs of the requirements, we used an updated baseline that assumes the existing requirements (those adopted in the CY 2020 HPT final rule, the CY 2022 OPPS/ASC final rule with comment period and the CY 2024 OPPS/ASC final rule with comment period and still codified at 45 CFR part 180) remain in place over the time horizon of this RIA.
In the CY 2024 OPPS/ASC final rule with comment period, we estimated an ongoing annual national burden of 383,292 hours (54 hours × 7,098 hospitals) and an annual national cost of $32,370,571 dollars ($4,560.52 per respondent × 7,098 hospitals), which represented a $10,698,069 ($32,370,571−$21,672,502) increase over our previous estimated ongoing national annual burden for subsequent years for hospitals to update their standard charge information in the CMS standard template and conform to the data dictionary.
We indicated in the CY 2026 OPPS/ASC proposed rule, we believe hospitals would incur an initial one-time cost to update their processes and systems to (1) identify and collect the newly proposed data elements, and (2) encode the standard charge information for the newly proposed data elements in the CMS standard template. To implement the proposed requirements, we estimated that it would take a Business Operations Specialist (BLS 13-1000), on average, 4 hours (at a cost of $87.52 per hour) to develop and update the necessary processes and procedures and develop the requirements to implement the proposed data elements and a General and Operations Managers (BLS 11-1021), on average, 1 hours (at a cost of $128.00 per hour) to review the updates.
Therefore, we stated in the CY 2026 OPPS/ASC proposed rule that we believed the one-time burden estimate to be 37,080 hours for all hospitals (5 hours × 7,416 hospitals) at a cost of $3,545,441.28 (7,416 hospitals × [($87.52 × 4 hours) + ($128.00 × 1 hours)]); see Table 163. As we indicated in the CY 2026 OPPS/ASC proposed rule, we believe the benefits to users of the MRF of having this additional information would justify the initial one-time burden to hospitals to update their processes and systems to identify and collect the newly proposed data elements and encode the standard charge information for the newly proposed data elements in the CMS standard template.
For this final rule with comment period, we updated the number of hospitals estimated to be subject to the HPT requirements using the same methodology as we did in the CY 2024 OPPS/ASC final rule with comment period. There were 8,340 hospitals most recently identified in the HIFLD hospital dataset.[535] We subtracted 374 hospitals HIFLD identified as “closed” as well as hospitals that are deemed under the regulation to have met requirements which included 352 Federally owned non-military and military hospitals, and 198 State, local, and district run forensic hospitals. We therefore estimate that, for this final rule with comment period, 7,416 hospitals would meet the HPT regulation's definition of “hospital” at 45 CFR 180.20.
After consideration of public comments, we are increasing both our one-time and ongoing annual burden estimates as demonstrated in section “XXIII. Collection of Information”. To establish processes and requirements related to the new data elements we are finalizing in this final rule with comment period, we now estimate that it will take a Business Operations Specialist (BLS 13-1000), on average, 8 hours (at a cost of $87.52 per hour) to develop and update the necessary processes and procedures and develop the requirements to implement the proposed data elements, a General and Operations Managers (BLS 11-1021), on average, 2 hours (at a cost of $128.00 per hour) to review the updates, and a Chief Executive (BLS 11-1011) 2 hours (at a cost of $252.82) to review and attest to the accuracy and completeness of the data in the MRF. We added the labor category for Chief Executives in response to comments on the CY 2026 OPPS/ASC proposed rule, addressed in an earlier section of this final rule with comment period, that suggested we failed to account for the burden for the Chief Executive Officer, president, or senior official designated to oversee the encoding of true, accurate, and complete data in the MRF to review and attest to ( printed page 54078) the information. Therefore, we believe the one-time burden estimate to implement the new requirements finalized in this final rule with comment period to be 88,992 hours for all hospitals (12 hours × 7,416 hospitals) at a cost of $10,840,708.80 (7,416 hospitals × [($87.52 × 8 hours) + ($128.00 × 2 hours) + ($252.82 × 2 hours]). For the annual burden, we added the labor category for Chief Executives in response to comments on the CY 2026 OPPS/ASC proposed rule, and we now estimate a total of 415,296 hours for all hospitals (7,416 hospitals × 56 hours) at a cost of $40,269,176.60 (7,416 hospitals × [($128/hour × 2 hours) + ($87.52/hour × 40 hours) + ($97.30/hour × 12 hours) + ($252.82/hour × 2 hours]), to include the burden for the senior hospital official. The annual burden is increased by 2 hours from 54 hours to 56 hours per hospital as compared to the CY 2024 OPPS/ASC final rule with comment period, which reflects the additional burden annually associated with the new requirements finalized in this final rule with comment period.
c. Benefit of Policies
As indicated in the CY 2026 OPPS/ASC proposed rule (90 FR 33856), although we cannot quantify the benefits of including additional data elements and encoding such data in the CMS required MRF template, we believe any opportunity to provide further context about standard charges, including through the addition of contextual information when the payer-specific negotiated charge is based on a percentage or algorithm, will be helpful to all consumers of the MRF as they analyze the data to identify cost savings and ways to stimulate market competition. We believe the attestation statement we are finalizing and the requirement to add the name of a senior official to publicly attest to the accuracy and completeness of the data encoded within the file will reduce public confusion related to whether all standard charges for hospital items and services are included within the MRF as dollar amounts, if possible, and the hospital has provided all necessary information available to the hospital for the public to be able to derive the dollar amount, including, but not limited to, the specific fee schedule or components referenced in such percentage, algorithm or formula. We also believe the addition of the NPI data element will catalyze more fulsome HPT and Transparency in Coverage (TiC) analysis.
(1) Benefits to Hospitals
As indicated in the CY 2026 OPPS/ASC proposed rule (90 FR 33856), hospitals, either directly or through management or actuarial consultants, are consuming the data released in the MRFs for their own operational purposes. Hospitals consume and analyze MRF data to improve negotiation strategies with employers and third party payers, improve contracting strategies, and demonstrate value in the negotiation process.[536] [537] Hospitals also use the MRF data to determine pricing strategies and to identify and hone their competitive advantage,[538] as well as to improve their revenue cycle efficiency.[539] Further, we continue to believe, as indicated in the CY 2026 OPPS/ASC proposed rule that, for those hospitals that are assessed a CMP, the reduction of the CMP we are finalizing if the hospital elects not to appeal CMS' findings, would offset some of the hospital financial burden, including legal fees and costs associated with challenging the imposition—including requesting and defending a hearing before an Administrative Law Judge (ALJ) and any subsequent appeals.
(2) Benefits to Other Interested Parties
As discussed in the CY 2020 HPT final rule (84 FR 65538) and the CY 2026 OPPS/ASC proposed rule (90 FR 33856), we believe public access to hospital standard charge information can be useful to the public, including patients who need to obtain services from a hospital, consumers of healthcare who wish to view hospital standard charge information prior to selecting a hospital, employers and State governments searching for lower cost options for health care coverage, and other users of the MRF who may develop consumer-friendly price transparency tools or perform price analyses to uncover disparities or drive value-based policy development. Since the effective date of the HPT regulations, innovators have been compiling HPT data sets and making them available for employers, researchers, and journalists to perform cost comparison studies and publish findings.
As discussed in the CY 2026 OPPS/ASC proposed rule (90 FR 33856 through 33857), feedback from interested parties, specifically innovators and researchers, has illuminated the need to detangle complex hospital contracting methods through the provision of data elements that help define the algorithm or percentage set forth in hospital MRFs. The allowed amount data elements we are finalizing in this final rule with comment period will support a better understanding of the range of payer-specific negotiated charge dispersion, which would further assist employers and consumers to understand a hospital's value as compared to other hospitals, stimulating competition and potentially resulting in price convergence to drive more predictable and consistent health care costs.[540]
With regard to the proposals we are finalizing in this final rule with comment period to strengthen the affirmation statement requirement, beginning January 1, 2026, by replacing it with an attestation in the MRF that would contain new specifications (relative to existing affirmation requirements) and to require hospitals to encode the name of the chief executive officer, president or senior official designated to oversee the encoding of true, accurate and complete data in the MRF, we continue to believe they will provide the necessary reassurance that hospitals have provided in their MRFs meaningful, accurate information to users of the MRF about their standard charges for health care items and services.
With regard to the NPI data element, as indicated in the CY 2026 OPPS/ASC proposed rule (90 FR 33857), including identifiers used for financial transactions in the MRF will make it easier for key participants in price negotiations (for example, employers and payers) to programmatically identify hospitals in their internal financial databases, such as claims data, ( printed page 54079) to conduct more in-depth payment and volume analyses to support contract negotiations and potentially reduce healthcare costs for consumers. We believe requiring a standard identifier will also help CMS, researchers, and innovators reduce dependence on manual processes for identifying the hospital and the hospital locations and increase opportunities for automated processes.
d. Limitations of Our Analysis
As stated in the CY 2024 OPPS/ASC final rule with comment period (88 FR 82174) and the CY 2026 OPPS/ASC proposed rule (90 FR 33857), it would be difficult for us to conduct a detailed quantitative analysis of the impact of requiring hospitals to make HPT information publicly available, given the lack of studies at the national level on the impact of the HPT regulations. Thus, in assessing the impact of our proposals, we rely on qualitative evidence of, and experiences with, the use of the public HPT data and feedback from consumers of the MRFs, as well as our own experiences reviewing the MRFs. Specifically, as discussed in the CY 2026 OPPS/ASC proposed rule, we have noted through our own reviews that many hospital MRFs lack dollar values when the standard charge for an item or service is based on an algorithm or percentage and have heard from interested parties the limitations of drawing meaningful comparisons without necessary context to help understand the standard charge in dollars for such items or services. We also have received comments from MRF users since the effective date of the 2024 OPPS/ASC final rule with comment period indicating that requiring hospitals to make a good faith effort did not go far enough to convey CMS' intent that all standard charge information available must be encoded in the MRF, with commenters suggesting our requirement to allow a good faith estimate may actually deter hospitals from providing fully complete and accurate standard charge data in their MRF. As we indicated in the proposed rule, we also heard users of the MRFs who questioned a hospital's inability to encode dollar amounts for the payer-specific negotiated charge data elements, and whether the complex contracting methodologies used by hospitals and payer organizations could only be expressed through an algorithm or formula, and not a dollar amount. We noted that we understand that users of the MRF may find the addition of algorithms make price comparisons among hospitals challenging when only an algorithm is available because the algorithms may not be consumer friendly. We continue to believe that our proposal to require an attestation, as opposed to merely requiring an affirmation statement, may enhance users' confidence that the data encoded is accurate and complete.
In addition, as indicated in the CY 2026 OPPS/ASC proposed rule (90 FR 33857), in discussions with innovators and researchers, we have heard that the addition of an NPI as a hospital unique identifier would allow more effective data crosswalking between hospital HPT MRFs and TiC MRFs, as well as to other CMS datasets that contain hospital quality data. We continue to believe this regulation would provide the additional context needed for consumers of the MRFs to create meaningful dollar comparisons that would ultimately benefit consumers through development of cost comparison tools, increased competition, or improved price negotiations with employers.
e. Alternatives Considered
The revisions to the HPT regulations we are finalizing in this final rule with comment period are designed to further address some of the barriers identified that limit price transparency, with a goal of increasing competition among healthcare providers to bring down costs. Specifically, this final rule with comment period aims to make meaningful price information via hospital standard charges more readily available to the public by providing additional contextual information, displayed as a dollar value, in those instances where standard charges are based on a percentage or an algorithm, as well as provide needed hospital identifier values to enable innovators and researchers to combine the HPT data with other claims and quality data to further empower consumer decision-making. We considered several alternative approaches, including other methodologies and lookback periods for calculating the allowed amount data elements, and whether to display specific counts of allowed amounts or ranges. We discuss these alternatives in section XIX. of this final rule with comment period. Specifically, we considered EDI 835 ERA transaction data lookback period alternatives of 3 months, 6 months, as well as requiring hospitals to use a rolling 12-month period prior to when the MRF posted. Based on comments received, we are ultimately finalizing a lookback period of no less than 12 months and no longer than 15 months. We sought comment on data sources other than the EDI 835 ERA transaction data to use to derive the allowed amount data elements. We received comments that not all hospitals have access to the EDI 835 ERA data for every item and service and that some payers provide paper or alternative electronic sources of remittance data. Based on comments, we are finalizing that hospitals use EDI 835 ERA transaction data or an alternative, equivalent source of remittance data to calculate and encode the allowed amounts. We also considered whether hospitals could encode a range of allowed amounts, as opposed to the exact count of allowed amounts, to achieve our objective of providing needed context to the allowed amount data element values. We received one comment indicating that adopting a range would be more burdensome than encoding the exact amount and are thus finalizing that hospitals must encode the count of allowed amounts.
We considered several alternative options to updating the required affirmation within the MRF, as discussed in section XIX. of this final rule with comment period. We considered asking the official to submit their MRF attestation directly to CMS, using a CMS developed template that would provide evidence of the accuracy and completeness of the MRF, and we also considered requiring the hospitals to post a more detailed attestation document that is signed by a senior official on the publicly available website that hosts the MRF. However, after consideration of public comments, we continue to believe these alternatives to be less useful than our proposals as they would either not meet the stated need to alert the public to the hospital's declaration of the accuracy and completeness of the data encoded within the MRF, or the attestation would not “travel” inside the MRF like the affirmation statement.
In addition, we considered different types of hospital identifiers, specifically the Employer Identification Number and the CMS Certification Number. Ultimately, however, we continue to believe that the alternatives would either limit the usefulness of hospital standard charge information or increase burden for hospitals without any additional benefit for users of MRF standard charge information.
Comment: Many commenters expressed concern about the financial and administrative burden for hospitals to comply with the proposed hospital price transparency requirements. Several commenters commented on the alternatives to our proposals outlined earlier in this section and section XIX. (Updates to Requirements for Hospitals to Make Public a List of Their Standard Charges section) that we considered. ( printed page 54080)
Response: We refer readers to the more substantial discussion of these comments and our responses to these comments in section XIX. (Updates to Requirements for Hospitals to Make Public a List of Their Standard Charges section) and section “XXIII. Collection of Information”. After consideration of the public comments, we are finalizing with modifications our proposals and our burden estimates.
D. Regulatory Review Cost Estimation
Due to the uncertainty involved with accurately quantifying the number of entities that will review the rule, we assume that the total number of unique commenters on this year's proposed rule will be the number of reviewers of this final rule with comment period. We acknowledge that this assumption may understate or overstate the costs of reviewing this rule. It is possible that not all commenters reviewed this year's rule in detail, and it is also possible that some reviewers chose not to comment on the proposed rule. For these reasons we believe that the number of past commenters would be a fair estimate of the number of reviewers of this rule. We welcomed any public comments on the approach in estimating the number of entities that would review the proposed rule. We did not receive any public comments specific to our solicitation.
We also recognize that different types of entities are in many cases affected by mutually exclusive sections of this final rule with comment period, and therefore for the purposes of our estimate we assume that each reviewer reads approximately 50 percent of the rule. We sought comments on this assumption. We did not receive any public comments specific to our solicitation.
Using the wage information from the Bureau of Labor Statistics (BLS) for medical and health service managers (Code 11-9111), we estimate that the cost of reviewing this rule is $113.42 per hour, including overhead and fringe benefits ( https://www.bls.gov/oes/current/oes_nat.htm). Assuming an average reading speed, we estimate that it would take approximately 8 hours for the staff to review half of this final rule with comment period. For each entity that reviews the rule, the estimated cost is $907.36 (8 hours × $113.42). Therefore, we estimate that the total cost of reviewing this regulation is $2,760,189 ($907.36 × 3,042).
E. Regulatory Flexibility Act (RFA) Analysis
The RFA requires agencies to analyze options for regulatory relief of small entities, if a rule has a significant impact on a substantial number of small entities. For purposes of the RFA, we estimate that many hospitals and CAHs are considered small businesses either by the Small Business Administration's size standards with total revenues of $41.5 million or less in any single year or by the hospital's not-for-profit status. Most ASCs and most CMHCs are considered small businesses with total revenues of $16.5 million or less in any single year. While we note the limited availability of certain information for OPPS providers, we estimate that approximately 3,000 OPPS providers included in the impact analysis would be considered small entities. As the small entity category would represent the majority of the providers in the table, the individual impact table categories would provide more specific estimated hospital impacts. While the estimated impacts of this final rule with comment period vary by OPPS provider category, many of those categories will be within the range of 1.5 to 2.5 percent. For details, we refer readers to the Small Business Administration's “Table of Size Standards” at http://www.sba.gov/content/small-business-size-standards.
Individuals and States are not included in the definition of a small entity. As its measure of significant economic impact on a substantial number of small entities, HHS uses a change in revenue of more than 3 to 5 percent. We believe that this threshold will not be reached by the requirements in this final rule with comment period, since as noted earlier in this section, most estimated changes will be below that range. Therefore, the Secretary has certified that this final rule with comment period will not have a significant economic impact on a substantial number of small entities.
In addition, section 1102(b) of the Act requires us to prepare a regulatory impact analysis if a rule may have a significant impact on the operations of a substantial number of small rural hospitals. This analysis must conform to the provisions of section 604 of the RFA. For purposes of section 1102(b) of the Act, we define a small rural hospital as a hospital that is located outside of a metropolitan statistical area and has 100 or fewer beds. We estimate that this final rule with comment period will increase payments to small rural hospitals by approximately 2.4 percent; therefore, it should have a negligible impact on approximately 526 small rural hospitals. We note that the estimated payment impact for any category of small entity will depend on both the services that they provide as well as the payment policies and/or payment systems that may apply to them. Therefore, the most applicable estimated impact may be based on the specialty, provider type, or payment system.
The analysis above, together with the remainder of this preamble, provides a regulatory flexibility analysis and a regulatory impact analysis.
F. Unfunded Mandates Reform Act (UMRA)
Section 202 of the Unfunded Mandates Reform Act of 1995 (UMRA) also requires that agencies assess anticipated costs and benefits before issuing any rule whose mandates require spending in any 1 year of $100 million in 1995 dollars, updated annually for inflation. In 2025, that threshold is approximately $187 million. This final rule with comment period will not impose a mandate that will result in the expenditure by State, local, and Tribal Governments, in the aggregate, or by the private sector, of more than $187 million in any 1 year.”
G. Federalism
Executive Order 13132 establishes certain requirements that an agency must meet when it promulgates a proposed rule (and subsequent final rule) that imposes substantial direct requirement costs on State and local governments, preempts State law, or otherwise has Federalism implications. We have examined the OPPS and ASC provisions included in this final rule with comment period in accordance with Executive Order 13132, Federalism, and have determined that they will not have a substantial direct effect on State, local, or tribal governments, preempt State law, or otherwise have a federalism implication. As reflected in Table 166 of this final rule with comment period, we estimate that OPPS payments to governmental hospitals (including State and local governmental hospitals) will increase by 2.1 percent under this final rule with comment period. While we do not know the number of ASCs or CMHCs with government ownership, we anticipate that it is small. The analyses we have provided in this section of this final rule with comment period, in conjunction with the remainder of this document, demonstrate that this rule is consistent with the regulatory philosophy and principles identified in Executive Order 12866, the RFA, and section 1102(b) of the Act.
This final rule with comment period will affect payments to a substantial number of small rural hospitals and a small number of rural ASCs, as well as other classes of hospitals, CMHCs, and ASCs, and some effects may be ( printed page 54081) significant. However, as noted in section XXVI. of this final rule with comment period, this rule should not have a significant effect on small rural hospitals.
H. E.O. 14192, “Unleashing Prosperity Through Deregulation”
Executive Order 14192, entitled “Unleashing Prosperity Through Deregulation” was issued on January 31, 2025, and requires that “any new incremental costs associated with new regulations shall, to the extent permitted by law, be offset by the elimination of existing costs associated with at least 10 prior regulations”. This final rule with comment period, is neither an E.O. 14192 deregulatory action nor an E.O. 14192 deregulatory action, due to generating no more than de minimis costs.
This final regulation is subject to the Congressional Review Act provisions of the Small Business Regulatory Enforcement Fairness Act of 1996 (5 U.S.C. 801 et seq.) and has been transmitted to the Congress and the Comptroller General for review.
XXVII. Waiver of 60-Day Delay of Effective Date
In the absence of an appropriation for FY 2026 or a Continuing Resolution, the Federal government funding for HHS lapsed on October 1, 2025. During this funding lapse, only excepted or exempt operations have continued, which significantly delayed work on this final rule with comment period. CMS identified funding that allowed the agency to restore additional day-to-day operations on a temporary basis beginning on October 27, 2025. However, most of the work on this final rule with comment period was not completed in accordance with our usual schedule for final CY OPPS/ASC payment rules, which aims for an issuance date of November 1, followed by an effective date of January 1, to ensure that the policies are effective at the start of the calendar year to which they apply.
We ordinarily provide a 60-day delay in the effective date of final rules after the date they are issued. The 60-day delay in effective date required by the Congressional Review Act, 5 U.S.C. 801(a)(3), can be waived, however, if the agency finds, for good cause, that notice and public procedure thereon are impracticable, unnecessary, or contrary to the public interest, and the agency incorporates the finding and a brief statement of reasons in the rule issued, 5 U.S.C. 808(2). We believe it would be impracticable and contrary to the public interest to delay the effective date of the OPPS and ASC final rule with comment period for the following reasons.
If the effective date of this final rule with comment period mentioned above in this document is delayed by 60 days, the finalized OPPS and ASC payment system updated policies (including the Hospital Outpatient Quality Report (OQR) Program, Rural Emergency Hospital Quality Reporting (REHQR) Program, the Ambulatory Surgical Center Quality Reporting (ASCQR) Program, Overall Hospital Quality Star Rating, the Hospital Price Transparency, Medicare Severity Diagnostis Related Groups (MS-DRGs), and Graduate Medical Education (GME) policies will not be effective as of the beginning of the payment year, and CMS will be compelled to continue to use obsolete reimbursement rates which will disadvantage health care providers and suppliers. We note that our waiver of the delayed effective date only applies to the OPPS and ASC payment system policies that are adopted in this final rule with comment period.
In accordance with sections 1833(t)(9) and 1833(i)(2)(D) of the Act, the OPPS and the ASC payment systems are calendar year payment systems. We typically issue the OPPS/ASC final rule with comment period by November 1 of each year to both comply with the statutory requirement under section 1833(t)(9)(A) of the Act to annually review and update these payment systems on a calendar year basis and ensure 60-days' notice so that the payment policies for these systems are effective on January 1, the first day of the calendar year to which the policies are intended to apply. The Hospital OQR Program, REHQR Program, and the ASCQR Program are intended to align with the OPPS and the ASC payment systems, respectively.
In this final rule with comment period, we review and revise payment rates for specific services and adopt or revise other policies that relate to the OPPS/ASC payment system for CY 2026. Section 1833(t)(9)(A) of the Act requires that no less often than annually, the Secretary shall review and revise the groups, the relative payment weights, and the wage and other adjustments for outpatient services to take into account changes in medical practice, changes in technology, the addition of new services, new cost data, and other relevant information and factors. In addition, section 1833(i)(2)(A) of the Act states that the payment amount made for facility services furnished in connection with a surgical procedure furnished to an individual in an ambulatory surgical center should be reviewed and updated annually to take account of varying conditions in different areas.
We find that, in order to comply with the statutory requirements to annually review and update these payment systems and ensure that the payment policies for these systems are effective on an annual basis, we must ensure that the rule is effective no later than 1 full year after the effective date of the previous year's update (that is, January 1 of each year). Moreover, it is in the public interest to waive the 60-day delay of the effective date, so that providers, and suppliers are adequately reimbursed starting at the beginning of the calendar year.
Therefore, we find good cause exists to waive the 60-day delay in the effective date for this final rule with comment period.
Mehmet Oz, Administrator of the Centers for Medicare & Medicaid Services, approved this document on November 20, 2025.
List of Subjects
42 CFR Part 410
- Diseases
- Health facilities
- Health professions
- Laboratories
- Medicare
- Reporting and recordkeeping requirements
- Rural areas
- X-rays
42 CFR Part 412
- Administrative practice and procedure
- Health facilities
- Medicare
- Puerto Rico
- Reporting and recordkeeping requirements
42 CFR Part 413
- Diseases
- Health facilities
- Medicare
- Puerto Rico
- Reporting and recordkeeping requirements
42 CFR Part 415
- Health facilities
- Health professions
- Medicare
- Reporting and recordkeeping requirements
42 CFR Part 416
- Health facilities
- Health professions
- Medicare
- Reporting and recordkeeping requirements
42 CFR Part 419
- Hospitals
- Medicare
- Reporting and recordkeeping requirements
45 CFR Part 180
- Hospital Price Transparency
For the reasons set forth in the preamble, the Centers for Medicare & Medicaid Services amends 42 CFR chapter IV as follows:
PART 410—SUPPLEMENTARY MEDICAL INSURANCE (SMI) BENEFITS
1. The authority citation for part 410 continues to read as follows:
2. Section 410.27 is amended by revising paragraph (a)(1)(iv)(B)( 1) to read as follows:
(a) * * *
(1) * * *
(iv) * * *
(B) * * *
( 1) For purposes of this section, direct supervision means that the physician or nonphysician practitioner must be immediately available to furnish assistance and direction throughout the performance of the procedure. It does not mean that the physician or nonphysician practitioner must be present in the room when the procedure is performed. For pulmonary rehabilitation, cardiac rehabilitation, and intensive cardiac rehabilitation services, direct supervision must be furnished as specified in §§ 410.47 and 410.49, respectively. The presence of the physician or nonphysician practitioner for the purpose of the supervision of pulmonary rehabilitation, cardiac rehabilitation, and intensive cardiac rehabilitation services includes virtual presence through audio/video real-time communications technology (excluding audio-only); and
3. Section 410.28 is amended by revising paragraph (e)(2)(iii) to read as follows:
(e) * * *
(2) * * *
(iii) The presence of the physician or nonphysician practitioner under paragraphs (e)(2)(i) and (ii) of this section includes virtual presence through audio/video real-time communications technology (excluding audio-only) for services without a 010 or 090 global surgery indicator.
PART 412—PROSPECTIVE PAYMENT SYSTEMS FOR INPATIENT HOSPITAL SERVICES
4. The authority citation for part 412 continues to read as follows:
5. Section 412.3 is amended by revising paragraph (d)(2) introductory text and removing paragraphs (d)(2)(i) and (ii) to read as follows:
(d) * * *
(2) An inpatient admission for a surgical procedure specified by Medicare as inpatient only under § 419.22(n) of this chapter is generally appropriate for payment under Medicare Part A regardless of the expected duration of care. Procedures no longer specified as inpatient only under § 419.22(n) of this chapter are appropriate for payment under Medicare Part A in accordance with paragraph (d)(1) or (3) of this section. Claims for services and procedures removed from the inpatient only list under § 419.22 of this chapter on or after January 1, 2021 are exempt from certain medical review activities until the Secretary determines that the service or procedure is more commonly performed in the outpatient setting than the inpatient setting.
6. Section 412.105 is amended by—
a. Revising paragraphs (f)(1)(i)(A) through (C); and
b. Adding paragraph (f)(1)(i)(E).
The revisions and addition read as follows:
(f) * * *
(1) * * *
(i) * * *
(A) Is approved by one of the national organizations listed in § 415.152 of this chapter, provided that the national organization does not use accreditation criteria that promote or encourage discrimination on the basis of race, color, national origin, sex, age, disability, or religion, including the use of those characteristics or intentional proxies for those characteristics as a selection criterion for employment, program participation, resource allocation, or similar activities, opportunities, or benefits.
(B) May count towards certification of the participant in a specialty or subspecialty listed in the current edition of either of the following publications, provided that listing in either of those publications, or in successor information sources, does not require the program to promote or encourage discrimination on the basis of race, color, national origin, sex, age, disability, or religion, including the use of those characteristics or intentional proxies for those characteristics as a selection criterion for employment, program participation, resource allocation, or similar activities, opportunities, or benefits:
( 1) The Directory of Graduate Medical Education Programs published by the American Medical Association.
( 2) The Annual Report and Reference Handbook published by the American Board of Medical Specialties.
(C) Is approved by the Accreditation Council for Graduate Medical Education (ACGME), or other organization designated by the Secretary, as a fellowship program in geriatric medicine, provided that the Council or other organization does not use accreditation criteria that promote or encourage discrimination on the basis of race, color, national origin, sex, age, disability, or religion, including the use of those characteristics or intentional proxies for those characteristics as a selection criterion for employment, program participation, resource allocation, or similar activities, opportunities, or benefits.
(E) Is a program that would be accredited except for the accrediting agency's reliance upon an accreditation standard that requires an entity to promote or encourage discrimination on the basis of race, color, national origin, sex, age, disability, or religion, including the use of those characteristics or intentional proxies for those characteristics as a selection criterion for employment, program participation, resource allocation, or similar activities, opportunities, or benefits.
7. Section 412.190 is amended by—
a. Revising paragraph (a)(2);
b. Adding paragraph (a)(3);
c. Revising paragraph (b)(1) introductory text;
d. Redesignating paragraph (d)(5) as (d)(6), and paragraph (d)(6) as (d)(5);
e. Adding paragraph (d)(9); and
f. Revising paragraphs (e) and (f).
The revisions and additions read as follows
(a) * * * ( printed page 54083)
(2) To update the methodology that will be used to calculate the Overall Hospital Quality Star Ratings to emphasize the contribution of the Safety of Care measure group to the Overall Hospital Quality Star Rating. This change aims to address the issue of hospitals receiving a high Star Rating despite performance in the lowest quartile of the Safety of Care measure group.
(3) The guiding principles of the Overall Hospital Quality Star Rating are as follows. In developing and maintaining the Overall Hospital Quality Star Ratings, we strive to:
(i) Use scientifically valid methods that are inclusive of hospitals and measure information and able to accommodate underlying measure changes;
(ii) Align with Care Compare on Medicare.gov and CMS programs;
(iii) Provide transparency of the methods for calculating the Overall Hospital Quality Star Rating; and
(iv) Be responsive to stakeholder input.
(b) * * *
(1) Sources of Data. Measures are selected from those publicly reported on Care Compare on Medicare.gov through certain CMS hospital inpatient and outpatient quality programs:
(d) * * *
(9) Emphasize Safety of Care. (i) Apply a 4-star cap for hospitals in the lowest quartile of the Safety of Care measure group performance in Calendar Year 2026. Any hospital that is assigned 5 stars in step eight but has a lowest quartile Safety of Care score (based on at least three Safety of Care measures) would be reassigned to 4 stars.
(ii) Apply a blanket 1-Star reduction for hospitals in the lowest quartile of Safety of Care measure group performance beginning in Calendar Year 2027 and later years. Any hospital assigned a 2, 3, 4, or 5-star rating in step eight, but with a lowest quartile Safety of Care score (based on at least three Safety of Care measures) would be reduced to 1, 2, 3, or 4 stars, respectively.
(e) Preview period prior to publication. CMS provides hospitals the opportunity to preview their Overall Hospital Quality Star Rating prior to publication. Hospitals have at least 30 days to preview their results, and if necessary, can reach out to CMS with questions.
(f) Suppression of Overall Hospital Quality Star Rating —(1) Subsection (d) hospitals. CMS may consider suppressing Overall Hospital Quality Star Rating for subsection (d) hospitals only under extenuating circumstances that affect numerous hospitals (as in, not an individualized or localized issue) as determined by CMS, or when CMS is at fault, including but not limited to when:
(i) There is an Overall Hospital Quality Star Rating calculation error by CMS;
(ii) There is a systemic error at the CMS quality program level that substantively affects the Overall Hospital Quality Star Rating calculation; or;
(iii) If a Public Health Emergency substantially affects the underlying measure data.
(2) CAHs. (i) CAHs may request to withhold their Overall Hospital Quality Star Rating from publication on Care Compare on Medicare.gov so long as the request for withholding is made, at the latest, during the Overall Hospital Quality Star Rating preview period.
(ii) CAHs may request to have their Overall Hospital Quality Star Rating withheld from publication on Care Compare on Medicare.gov, as well as their data from the public input file, so long as the request is made during the CMS quality program-level 30-day confidential preview period for the Care Compare refresh data used to calculate the Overall Hospital Quality Star Ratings.
PART 413—PRINCIPLES OF REASONABLE COST REIMBURSEMENT; PAYMENT FOR END-STAGE RENAL DISEASE SERVICES; PROSPECTIVELY DETERMINED PAYMENT RATES FOR SKILLED NURSING FACILITIES; PAYMENT FOR ACUTE KIDNEY INJURY DIALYSIS
8. The authority citation for part 413 continues to read as follows:
9. Section 413.20 is amended by revising paragraph (d)(3) to read as follows:
(d) * * *
(3)(i) The provider must furnish the contractor—
(A) Upon request, copies of patient service charge schedules and changes thereto as they are put into effect; and
(B) Its median payer-specific negotiated charge by MS-DRG for payers that are Medicare Advantage (MA) organizations, as applicable, and changes thereto as they are put into effect.
(ii) The contractor evaluates the charge schedules as specified in paragraph (d)(3)(i) of this section to determine the extent to which they may be used for determining program payment.
10. Section 413.75 is amended in paragraph (b) by revising the definitions of “Approved geriatric program” and “Approved medical residency program” to read as follows:
(b) * * *
Approved geriatric program means a fellowship program of one or more years in length that is approved by one of the national organizations listed in § 415.152 of this chapter under that respective organization's criteria for geriatric fellowship programs, provided that the national organization does not use accreditation criteria that promote or encourage discrimination on the basis of race, color, national origin, sex, age, disability, or religion, including the use of those characteristics or intentional proxies for those characteristics as a selection criterion for employment, program participation, resource allocation, or similar activities, opportunities, or benefits.
Approved medical residency program means a program that meets one of the following criteria:
(i) Is approved by one of the national organizations listed in § 415.152 of this chapter, provided that the national organization does not use accreditation criteria that promote or encourage discrimination on the basis of race, color, national origin, sex, age, disability, or religion, including the use of those characteristics or intentional proxies for those characteristics as a selection criterion for employment, program participation, resource allocation, or similar activities, opportunities, or benefits.
(ii) May count towards certification of the participant in a specialty or subspecialty listed in the current edition of either of the following publications, provided that listing in either of those publications, or in successor information sources, does not require the program to promote or encourage discrimination on the basis of race, color, national origin, sex, age, disability, or religion, including the use of those characteristics or intentional proxies for those characteristics as a ( printed page 54084) selection criterion for employment, program participation, resource allocation, or similar activities, opportunities, or benefits:
(A) The Directory of Graduate Medical Education Programs published by the American Medical Association, and available from American Medical Association, Department of Directories and Publications, 515 North State Street, Chicago, Illinois 60610; or
(B) The Annual Report and Reference Handbook published by the American Board of Medical Specialties, and available from American Board of Medical Specialties, One Rotary Center, Suite 805, Evanston, Illinois 60201.
(iii) Is approved by the Accreditation Council for Graduate Medical Education (ACGME), or other organization designated by the Secretary, as a fellowship program in geriatric medicine, or other organization designated by the Secretary, provided that the Council or other organization does not use accreditation criteria that promote or encourage discrimination on the basis of race, color, national origin, sex, age, disability, or religion, including the use of those characteristics or intentional proxies for those characteristics as a selection criterion for employment, program participation, resource allocation, or similar activities, opportunities, or benefits.
(iv) Is a program that would be accredited except for the accrediting agency's reliance upon an accreditation standard that requires an entity to perform an induced abortion or require, provide, or refer for training in the performance of induced abortions, or make arrangements for such training, regardless of whether the standard provides exceptions or exemptions.
(v) Is a program that would be accredited except for the accrediting agency's reliance upon an accreditation standard that requires an entity to promote or encourage discrimination on the basis of race, color, national origin, sex, age, disability, or religion, including the use of those characteristics or intentional proxies for those characteristics as a selection criterion for employment, program participation, resource allocation, or similar activities, opportunities, or benefits.
PART 415—SERVICES FURNISHED BY PHYSICIANS IN PROVIDERS, SUPERVISING PHYSICIANS IN TEACHING SETTINGS, AND RESIDENTS IN CERTAIN SETTINGS
11. The authority citation for part 415 continues to read as follows:
12. Section 415.152 is amended by revising the definition of “Approved graduate medical education (GME) program” to read as follows:
Approved graduate medical education (GME) program means one of the following:
(1) A residency program approved by the Accreditation Council for Graduate Medical Education, by the American Osteopathic Association, by the Commission on Dental Accreditation of the American Dental Association, or by the Council on Podiatric Medical Education of the American Podiatric Medical Association, or other organization determined by the Secretary, provided that the applicable organization does not use accreditation criteria that promote or encourage discrimination on the basis of race, color, national origin, sex, age, disability, or religion, including the use of those characteristics or intentional proxies for those characteristics as a selection criterion for employment, program participation, resource allocation, or similar activities, opportunities, or benefits.
(2) A program otherwise recognized as an “approved medical residency program” under § 413.75(b) of this chapter.
PART 416—AMBULATORY SURGICAL SERVICES
13. The authority citation for part 416 is revised to read as follows:
14. Section 416.164 is amended by—
a. Revising paragraphs (a)(5), (b)(5) and (6); and
b. Adding paragraph (b)(7).
The revisions and addition read as follows:
(a) * * *
(5) Medical and surgical supplies not on pass-through status under subpart G of part 419 of this subchapter and not covered ancillary skin substitute supplies under paragraph (b) of this section;
(b) * * *
(5) Certain radiology services and certain diagnostic tests for which separate payment is allowed under the OPPS;
(6) Non-opioid pain management drugs, biologicals, and medical devices as determined by CMS under § 416.174; and
(7) Groups of skin substitute supply products.
15. Section 416.166 is revised to read as follows:
(a) Covered surgical procedures. (1) Effective for services furnished on or after January 1, 2008 through December 31, 2025, covered surgical procedures are those procedures that meet the general standards described in paragraph (b)(1) of this section (whether commonly furnished in an ASC or a physician's office) and are not excluded under paragraph (c) of this section; and
(2) Effective for services furnished on or after January 1, 2026, covered surgical procedures are those procedures that meet the requirements described in paragraph (b)(2) of this section (whether commonly furnished in an ASC or a physician's office).
(b) Requirements for covered surgical procedures —(1) General Standards. Effective for services furnished on or after January 1, 2008 through December 21, 2025, subject to the exclusions in paragraph (c) of this section, covered surgical procedures are surgical procedures specified by the Secretary and published in the Federal Register and/or via the internet on the CMS website that are separately paid under the OPPS, that would not be expected to pose a significant safety risk to a Medicare beneficiary when performed in an ASC, and for which standard medical practice dictates that the beneficiary would not typically be expected to require active medical monitoring and care at midnight following the procedure.
(2) Effective for services furnished on or after January 1, 2026, covered surgical procedures are surgical procedures specified by the Secretary that are published in the Federal Register and/or via the internet on the CMS website and that:
(i) Are separately paid under the OPPS; and
(ii) Are not:
(A) Currently designated as requiring inpatient care under § 419.22(n) of this subchapter; ( printed page 54085)
(B) Only able to be reported using a CPT unlisted surgical procedure code; or
(C) Otherwise excluded under § 411.15 of this chapter.
(c) General exclusions effective January 1, 2008, through December 31, 2025. Notwithstanding paragraph (b)(1) of this section, covered surgical procedures do not include those surgical procedures that:
(1) Generally result in extensive blood loss;
(2) Require major or prolonged invasion of body cavities;
(3) Directly involve major blood vessels;
(4) Are generally emergent or life-threatening in nature;
(5) Commonly require systemic thrombolytic therapy;
(6) Are designated as requiring inpatient care under § 419.22(n) of this subchapter;
(7) Can only be reported using a CPT unlisted surgical procedure code; or
(8) Are otherwise excluded under § 411.15 of this chapter.
(d) Physician considerations beginning January 1, 2026. Physicians should consider the following safety factors as to a specific beneficiary when determining whether to perform a covered surgical procedure. The covered procedure:
(1) Is not expected to pose a significant safety risk when performed in an ASC;
(2) Is one of which standard medical practice dictates the beneficiary would not typically be expected to require active medical monitoring and care at midnight following the procedure;
(3) Generally results in extensive blood loss;
(4) Requires major or prolonged invasion of body cavities;
(5) Directly involves major blood vessels;
(6) Is generally emergent or life-threatening in nature; and
(7) commonly requires systemic thrombolytic therapy.
(e) Additions to the list of ASC covered surgical procedures beginning January 1, 2026. On or after January 1, 2026, CMS adds surgical procedures to the list of ASC covered procedures as follows:
(1) CMS identifies a surgical procedure that meets the requirements at paragraph (b)(2) of this section.
(2) CMS is notified of a surgical procedure that could meet the requirements at paragraph (b)(2) of this section and CMS confirms that such surgical procedure meets those requirements.
16. Section 416.171 is amended by revising paragraphs (a)(2)(iii) through (viii) to read as follows:
(a) * * *
(2) * * *
(iii) For CY 2019 through CY 2026, the update is the hospital inpatient market basket percentage increase applicable under section 1886(b)(3)(B)(iii) of the Act.
(iv) For CY 2027 and subsequent years, the update is the Consumer Price Index for All Urban Consumers (U.S. city average) as estimated by the Secretary for the 12-month period ending with the midpoint of the year involved.
(v) For CY 2014 through CY 2018, the Consumer Price Index for All Urban Consumers update determined under paragraph (a)(2)(ii) of this section was reduced by 2.0 percentage points for ASCs that failed to meet the standards for reporting of ASC quality measures as established by the Secretary for the corresponding calendar year.
(vi) For CY 2019 through CY 2026, the hospital inpatient market basket percentage increase determined under paragraph (a)(2)(iii) of this section is reduced by 2.0 percentage points for an ASC that fails to meet the standards for reporting of ASC quality measures as established by the Secretary for the corresponding calendar year.
(vii) For CY 2027 and subsequent years, the Consumer Price Index for All Urban Consumers update determined under paragraph (a)(2)(iv) of this section is reduced by 2.0 percentage points for an ASC that fails to meet the standards for reporting of ASC quality measures as established by the Secretary for the corresponding calendar year.
(viii)(A) For CY 2011 through CY 2018, the Consumer Price Index for All Urban Consumers determined under paragraph (a)(2)(ii) of this section, after application of any reduction under paragraph (a)(2)(iv) of this section, was reduced by the productivity adjustment described in section 1886(b)(3)(B)(xi)(II) of the Act.
(B) For CY 2019 through CY 2026, the hospital inpatient market basket percentage increase determined under paragraph (a)(2)(iii) of this section, after application of any reduction under paragraph (a)(2)(v) of this section, is reduced by the productivity adjustment described in section 1886(b)(3)(B)(xi)(II) of the Act.
(C) For CY 2027 and subsequent years, the Consumer Price Index for All Urban Consumers determined under paragraph (a)(2)(iv) of this section, after application of any reduction under paragraph (a)(2)(vii) of this section, is reduced by the productivity adjustment described in section 1886(b)(3)(B)(xi)(II) of the Act.
17. Section 416.174 is amended by revising paragraphs (a) introductory text, (b) introductory text, and (c)(1) and (2) to read as follows:
(a) Eligibility for separate payment for non-opioid pain management drugs and biologicals. From January 1, 2025, through December 31, 2027, a non-opioid drug or biological is eligible for separate payment if CMS determines it meets the following requirements:
(b) Eligibility for separate payment for non-opioid medical devices. From January 1, 2025, through December 31, 2027, a medical device is eligible for separate payment if CMS determines it meets all of the following requirements:
(c) * * *
(1) For a qualifying drug or biological as defined in paragraph (a) of this section, the amount of payment is the amount determined under section 1847A of the Act for the drug or biological that exceeds the portion of the otherwise applicable Medicare OPD fee schedule amount, subject to paragraph (c)(3) of this section.
(2) For a qualifying medical device as defined in paragraph (b) of this section, the amount of payment is the amount of the hospital's charges for the device, adjusted to cost, that exceeds the portion of the otherwise applicable Medicare OPD fee schedule amount, subject to paragraph (c)(3) of this section.
18. Section 416.310 is amended by revising paragraph (d) to read as follows:
(d) Extraordinary circumstance exception (ECE) —(1) General rule. CMS may grant an ECE with respect to the reporting requirements under this section in the event of extraordinary circumstances beyond the control of the ASC. For purposes of this paragraph (d), an extraordinary circumstance is an event beyond the control of an ASC (for example, a natural or man-made disaster such as a hurricane, tornado, earthquake, terrorist attack, or bombing) that affected the ability of the ASC to comply with one or more applicable reporting requirements with respect to a calendar year. ( printed page 54086)
(2) Process for requesting an ECE. (i) An ASC may request an ECE within 60 calendar days of the date that the extraordinary circumstance occurred by submitting the information specified by CMS at QualityNet or a successor website.
(ii) CMS notifies the ASC of its decision on the request, in writing, via email. In the event that CMS grants an ECE to the ASC, the written decision specifies whether the ASC is exempted from one or more reporting requirements or whether CMS has granted the ASC an extension of time to comply with one or more reporting requirements.
(3) Authority to Grant an ECE. CMS may grant an ECE to one or more ASCs that have not requested an ECE if CMS determines that—
(i) A systemic problem with a CMS data collection system directly impacted the ability of the ASC to comply with a quality data reporting requirement; or
(ii) An extraordinary circumstance has affected an entire region or locale. Any ECE granted under this paragraph (d)(3) specifies whether the affected ASCs are exempted from one or more reporting requirements or whether CMS has granted the ASCs an extension of time to comply with one or more reporting requirements.
PART 419—PROSPECTIVE PAYMENT SYSTEM FOR HOSPITAL OUTPATIENT DEPARTMENT SERVICES
19. The authority citation for part 419 continues to read as follows:
20. Section 419.2 is amended by revising paragraphs (b)(16) and (18) to read as follows:
(b) * * *
(16) Drugs and biologicals that function as supplies when used in a surgical procedure including, but not limited to products, excluding skin substitutes, that aid wound healing;
(18) Certain services described by add-on codes (excluding skin substitute product add-on codes that are assigned to status indicator “S1”).
21. Section 419.22 is amended by revising paragraph (n) to read as follows:
(n) Services and procedures that the Secretary designates as requiring inpatient care. Effective beginning on January 1, 2026, the Secretary shall eliminate the list of services and procedures designated as requiring inpatient care through a 3-year transition, with the list eliminated in its entirety by January 1, 2029.
22. Section 419.23 is removed.
23. Section 419.43 is amended by revising paragraphs (k)(1) introductory text and (k)(2) introductory text to read as follows:
(k) * * *
(1) Eligibility for separate payment for non-opioid pain management drugs and biologicals. From January 1, 2025, through December 31, 2027, a drug or biological is eligible for separate payment if CMS determines it meets the following requirements:
(2) Eligibility for separate payment for non-opioid medical devices. From January 1, 2025, through December 31, 2027, a medical device is eligible for separate payment if CMS determines it meets the following requirements:
24. Section 419.46 is amended by revising paragraph (e) to read as follows:
(e) Extraordinary circumstance exception (ECE). (1) General rule. CMS may grant an ECE with respect to the reporting requirements under this section in the event of extraordinary circumstances beyond the control of the hospital. For purposes of this paragraph (e), an extraordinary circumstance is an event beyond the control of a hospital (for example, a natural or man-made disaster such as a hurricane, tornado, earthquake, terrorist attack, or bombing) that affected the ability of the hospital to comply with one or more applicable reporting requirements with respect to a calendar year.
(2) Process for requesting an ECE. (i) A hospital may request an ECE within 60 calendar days of the date that the extraordinary circumstance occurred by submitting the information specified by CMS at QualityNet or a successor website.
(ii) CMS notifies the hospital of its decision on the request, in writing, via email. In the event that CMS grants an ECE to the hospital, the written decision specifies whether the hospital is exempted from one or more reporting requirements or whether CMS has granted the hospital an extension of time to comply with one or more reporting requirements.
(3) Authority to Grant an ECE. CMS may grant an ECE to one or more hospitals that have not requested an ECE if CMS determines that—
(i) A systemic problem with a CMS data collection system directly impacted the ability of the hospital to comply with a quality data reporting requirement; or
(ii) An extraordinary circumstance has affected an entire region or locale. Any ECE granted under this paragraph (e)(3) specifies whether the affected hospitals are exempted from one or more reporting requirements or whether CMS has granted the hospitals an extension of time to comply with one or more reporting requirements.
25. Section 419.47 is amended by revising the section heading and paragraph (a) and to read as follows:
(a) Creation of a new HCPCS code for Category B IDE studies that have a treatment arm and a placebo control arm. CMS will create a new HCPCS code, or revise an existing HCPCS code, to describe a Category B IDE study, which will include both the treatment and placebo control arms, related device(s) of the study, as well as routine care items and services, as specified under § 405.201 of this chapter, when CMS determines that:
(1) The Medicare coverage IDE study criteria in § 405.212 of this chapter are met; and
(2) A new or revised code is necessary to preserve the scientific validity of such a study, such as by preventing the unblinding of the study.
26. Section 419.49 is added to read as follows:
(a) General rule. CMS provides for an additional payment beyond the standard payment to a hospital for a dose of Tc- ( printed page 54087) 99m derived from Mo-99, if at least 50 percent of the Mo-99 in the Tc-99m generator that produced the dose was both irradiated and processed in the United States.
(1) Domestically produced Mo-99 refers to Mo-99 that was both irradiated and processed in the United States.
(2) Irradiated refers to the process of bombarding a uranium or molybdenum target with radiation in order to produce Mo-99. Irradiation is typically performed with a nuclear reactor or particle accelerator.
(3) Processed refers to the purification of Mo-99 from irradiated material.
(b) Exclusions. A dose of Tc-99m does not qualify for the add-on payment if more than 50 percent of the Mo-99 in the Tc-99m generator was irradiated or processed outside the United States, even if the Mo-99 has been loaded into a Tc-99m generator in the United States or if the Tc-99m has been eluted at a radiopharmacy in the United States.
(1) Eluted refers to the process by which Tc-99m is chemically separated from Mo-99 within the generator and collected in an elution vial.
(2) [Reserved]
27. Section 419.64 is amended by removing paragraph (a)(4)(iv).
28. Section 419.95 is amended by revising paragraph (g) and adding paragraph (h) to read as follows:
(g) Extraordinary circumstance exception (ECE). (1) General rule. CMS may grant an ECE with respect to the reporting requirements under this section in the event of extraordinary circumstances beyond the control of the REH. For purposes of this paragraph (g), an extraordinary circumstance is an event beyond the control of an REH (for example, a natural or man-made disaster such as a hurricane, tornado, earthquake, terrorist attack, or bombing) that affected the ability of the REH to comply with one or more applicable reporting requirements with respect to a calendar year.
(2) Process for requesting an ECE. (i) An REH may request an ECE within 60 calendar days of the date that the extraordinary circumstance occurred by submitting the information specified by CMS at QualityNet or a successor website.
(ii) CMS notifies the REH of its decision on the request, in writing, via email. In the event that CMS grants an ECE to the REH, the written decision specifies whether the REH is exempted from one or more reporting requirements or whether CMS has granted the REH an extension of time to comply with one or more reporting requirements.
(3 ) Authority to Grant an ECE. CMS may grant an ECE to one or more REHs that have not requested an ECE if CMS determines that—
(i) A systemic problem with a CMS data collection system directly impacted the ability of the REH to comply with a quality data reporting requirement; or
(ii) An extraordinary circumstance has affected an entire region or locale. Any ECE granted under this paragraph (g)(3) specifies whether the affected REHs are exempted from one or more reporting requirements or whether CMS has granted the REHs an extension of time to comply with one or more reporting requirements.
(h) Requirements for submission of electronic clinical quality measures (eCQMs) under the REHQR Program. When reporting eCQMs under the REHQR Program, REHs must adhere to the following requirements:
(1) REHs must utilize technology certified to the Office of the National Coordinator for Health Information Technology's (ONC's) health information technology (IT) certification criteria, as adopted and updated in 45 CFR 170.315, for reporting eCQMs under the REHQR Program.
(2) REHs must use health IT certified to all eCQMs that are available to report under the REHQR Program.
(3) REHs must use the most recent version of the eCQM electronic measure specifications for the applicable reporting period available on the Electronic Clinical Quality Improvement Resource Center website at https://ecqi.healthit.gov/, or another website as designated by CMS.
(4) The requirements set forth in paragraphs (h)(1) through (3) of this section apply only where an REH opts to report an eCQM.
For the reasons set forth in the preamble, the Department of Health and Human Services amends 45 CFR part 180 as set forth below:
PART 180—HOSPITAL PRICE TRANSPARENCY
29. The authority citation for part 180 continues to read as follows:
30. Section 180.20 is amended by adding definitions of “Median allowed amount”, “Ninetieth (90th) percentile allowed amount”, and “Tenth (10th) percentile allowed amount” in alphabetical order to read as follows.
Median allowed amount means the median of the total allowed amounts the hospital has historically received from a third party payer for an item or service for a time period no less than 12 months and no longer than 15 months prior to posting the machine-readable file. Should the calculated median fall between two observed allowed amounts, the median allowed amount is the next highest observed value.
Ninetieth (90th) percentile allowed amount means the 90th percentile of the total allowed amounts the hospital has historically received from a third party payer for an item or service for a time period no less than 12 months and no longer than 15 months prior to posting the machine-readable file. Should the calculated percentile fall between two observed allowed amounts, the 90th percentile allowed amount is the next highest observed value.
Tenth (10th) percentile allowed amount means the 10th percentile of the total allowed amounts the hospital has historically received from a third party payer for an item or service for a time period no less than 12 months and no longer than 15 months prior to posting the machine-readable file. Should the calculated percentile fall between two observed allowed amounts, the 10th percentile allowed amount is the next highest observed value.
31. Section 180.50 is amended by revising paragraphs (a)(3), (b)(2)(i)(A), and (b)(2)(ii)(C) to read as follows:
(a) * * *
(3) Each hospital must:
(i) Beginning January 1, 2024 through December 31, 2025, make a good faith effort to ensure that the standard charge information encoded in the machine-readable file is true, accurate, and complete as of the date indicated in the machine-readable file.
(ii) Beginning January 1, 2024 through December 31, 2025, affirm in its machine-readable file that, to the best of its knowledge and belief, the hospital has included all applicable standard charge information in accordance with the requirements of this section, and that the information encoded is true, accurate, and complete as of the date indicated in the machine-readable file. ( printed page 54088)
(iii) Beginning January 1, 2026, attest in its machine-readable file the following: To the best of its knowledge and belief, this hospital has included all applicable standard charge information in accordance with the requirements of 45 CFR 180.50, and the information encoded is true, accurate, and complete as of the date in the file. This hospital has included all payer-specific negotiated charges in dollars that can be expressed as a dollar amount. For payer-specific negotiated charges that cannot be expressed as a dollar amount in the machine-readable file or not knowable in advance, the hospital attests that the payer-specific negotiated charge is based on a contractual algorithm, percentage or formula that precludes the provision of a dollar amount and has provided all necessary information available to the hospital for the public to be able to derive the dollar amount, including, but not limited to, the specific fee schedule or components referenced in such percentage, algorithm or formula.
(iv) Beginning January 1, 2026, encode the name of the hospital chief executive officer, president, or senior official designated to oversee the encoding of true, accurate, and complete data as directed in in paragraph (a)(3)(iii) of this section.
(b) * * *
(2) * * *
(i) * * *
(A) Hospital name, license number, location name(s) and address(es) under the single hospital license to which the list of standard charges applies, and beginning January 1, 2026, Type 2 (organizational) National Provider Identifier(s) (NPI). Location name(s) and address(es) must include, at minimum, all inpatient facilities and stand-alone emergency departments; and
(ii) * * *
(C) Whether the standard charge indicated should be interpreted by the user as a dollar amount, or if the standard charge is based on a percentage or algorithm. If the standard charge is based on a percentage or algorithm, the machine-readable file (MRF) must also describe the percentage or algorithm that determines the dollar amount for the item or service, and
(1) Beginning January 1, 2025 through December 31, 2025, calculate and encode an estimated allowed amount in dollars for that item or service; and
(2) Beginning January 1, 2026, calculate and encode the tenth (10th) percentile allowed amount, the median allowed amount, and the ninetieth (90th) percentile allowed amount in dollars for that item or service. Hospitals must also calculate and encode the total number of allowed amount remittances that were used to calculate the 10th percentile allowed amount, median allowed amount, and 90th percentile allowed amount.
32. Section 180.90 is amended by—
a. Adding paragraph (c)(4);
b. Revising paragraph (d)(1);
c. Redesignating paragraphs (d)(2) and (3) as paragraphs (d)(3) and (4), respectively; and
d. Adding new paragraph (d)(2).
The additions and revision read as follows:
(c) * * *
(4) Except as provided in this paragraph, the amount of a civil monetary penalty is reduced by 35 percent if the hospital submits a written notice to CMS requesting to waive its right to a hearing under § 180.100 within 30-calendar days of the date of the notice of imposition of the civil monetary penalty. A hospital that receives a 35 percent reduction in a civil monetary penalty under this paragraph is not eligible to receive a 35 percent reduction for any subsequent civil monetary penalties imposed pursuant to continuing violations according to § 180.90(f) and also waives its right to appeal under § 180.100 any subsequent civil monetary penalties imposed for such continuing violations. A hospital is not eligible to request that CMS reduce the amount of a civil monetary penalty by 35 percent in accordance with this paragraph if—
(i) The hospital does not request to waive its right to a hearing in accordance with this paragraph; or
(ii) CMS imposed the CMP because the hospital failed to make public an MRF as required at § 180.40(a) or failed to make public a consumer-friendly list of standard charges as required at § 180.40(b).
(d) * * *
(1) A hospital that does not meet the criteria to receive a reduction to the civil monetary penalty that had been imposed upon it as set forth in paragraph (c)(4) of this section must pay the civil monetary penalty in full within 60 calendar days after the date of the notice of imposition of a civil monetary penalty from CMS under paragraph (b) of this section.
(2) A hospital that meets the criteria to receive a reduction to the civil monetary penalty that had been imposed upon it as set forth in paragraph (c)(4) of this section must pay the civil monetary penalty, as reduced in accordance with paragraph (c)(4) of this section, within 60 calendar days after the date of the notice of imposition of a civil monetary penalty from CMS under paragraph (b) of this section.
Robert F. Kennedy, Jr.,
Secretary, Department of Health and Human Services.
Footnotes
1. Status indicator “J1” denotes Hospital Part B Services Paid Through a Comprehensive APC. Further information can be found in CY 2026 Addendum D1.
Back to Citation2. Status indicator “J2” denotes Hospital Part B Services That May Be Paid Through a Comprehensive APC. Further information can be found in CY 2026 Addendum D1.
Back to Citation3. Status Indicator “T” is defined as a “Procedure or Service, Multiple Procedure Reduction Applies” the OPPS payment status is “Paid under OPPS; separate APC payment.” Definitions to all OPPS payment status indicators are available in Addenda D1 to this final rule with comment period.
Back to Citation4. https://www.cms.gov/regulations-and-guidance/guidance/transmittals/downloads/r3523cp.pdf.
Back to Citation5. https://www.cms.gov/Regulations-and-Guidance/Guidance/Manuals/Downloads/bp102c15.pdf.
Back to Citation6. Status Indicator “T” is defined as a “Procedure or Service, Multiple Procedure Reduction Applies” the OPPS payment status is “Paid under OPPS; separate APC payment.” Definitions to all OPPS payment status indicators are available in Addenda D1 to this final rule with comment period.
Back to Citation7. Status Indicator “Q” is defined as a “STV-Packaged Codes” the OPPS payment status is “Paid under OPPS; Addendum B displays APC assignments when services are separately payable. (1) Packaged APC payment if billed on the same claim as a HCPCS code assigned status indicator “S,” “T,” or “V.” (2) Composite APC payment if billed with specific combinations of services based on OPPS composite-specific payment criteria. Payment is packaged into a single payment for specific combinations of services. (3) In other circumstances, payment is made through a separate APC payment.” Definitions to all OPPS payment status indicators are available in Addenda D1 to this final rule with comment period.
Back to Citation9. https://www.cms.gov/files/document/medicare-part-b-inflation-rebate-program-initial-guidance.pdf.
Back to Citation10. https://www.cms.gov/files/document/medicare-part-b-inflation-rebate-program-revised-guidance.pdf.
Back to Citation11. In addition, beginning with the April 2023 ASP Drug Pricing file, the file includes the coinsurance percentage for each drug and specifies “inflation-adjusted coinsurance” in the “Notes” column if the coinsurance for a drug is less than 20 percent of the Medicare Part B payment amount. Drug pricing files are available at https://www.cms.gov/medicare/medicare-fee-for-service-part-b-drugs/mcrpartbdrugavgsalesprice.
Back to Citation15. Currently approved under OMB control number 0938-0860; expires October 31, 2027.
Back to Citation16. Luxturna. FDA Package Insert. Available: https://www.fda.gov/media/109906/download.
Back to Citation17. LUXTURNA REIMBURSEMENT GUIDE FOR TREATMENT CENTERS. https://mysparkgeneration.com/uploads/2022/09/LUXTURNA-Reimbursement-Guide-for-Treatment-Centers-ISI-Update-April-2022-P-RPE65-US-320025.pdf.
Back to Citation18. ClinicalTrials.gov. “Randomized Controlled Pivotal Trial of Autologous Bone Marrow Cells Using the CardiAMP Cell Therapy System in Patients With Refractory Angina Pectoris and Chronic Myocardial Ischemia.” Accessed May 10, 2022. https://clinicaltrials.gov/ct2/show/NCT03455725?term=NCT03455725&rank=1.
Back to Citation19. ClinicalTrials.gov. “Randomized Controlled Pivotal Trial of Autologous Bone Marrow Mononuclear Cells Using the CardiAMP Cell Therapy System in Patients With Post Myocardial Infarction Heart Failure.” Accessed May 10, 2022. https://clinicaltrials.gov/ct2/show/NCT02438306.
Back to Citation20. Jelymyto Package Insert, Revised: 01/2021. https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/211728s002lbl.pdf.
Back to Citation21. ClinicalTrials.gov. “The HistoSonics System for Treatment of Primary and Metastatic Liver Tumors Using Histotripsy (#HOPE4LIVER) (#HOPE4LIVER).” Accessed May 10, 2022. https://clinicaltrials.gov/ct2/show/study/NCT04573881.
Back to Citation22. Subsequently, the FDA approved a prior approval supplemental new drug application (sNDA) providing for the following labeling modification: expansion of the indication to include monotherapy of SpravatoTM. (esketamine) for treatment resistant depression (TRD). See https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2025/211243Orig1s016ltr.pdf.
Back to Citation23. The REMS document is available at https://www.fda.gov/drugs/drug-safety-and-availability/risk-evaluation-and-mitigation-strategies-rems, and labeling can be found at https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm.
Back to Citation24. See “The HistoSonics System for Treatment of Primary Solid Renal Tumors Using Histotripsy (#HOPE4KIDNEY) at https://clinicaltrials.gov/study/NCT05820087.
Back to Citation25. See FDA Package Insert. https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/208742s007lbl.pdf.
Back to Citation28. https://www.cms.gov/medicare/payment/fee-schedules/physician/federal-regulation-notices.
Back to Citation29. To apply for OPPS transitional device pass-through status, applicants complete an application that is subject to the Paperwork Reduction Act of 1995 (PRA) (44 U.S.C. 3501 et seq.). This information collection (CMS-10052) is currently approved under OMB control number 0938-0857 and has an expiration date of November 30, 2025. The information collection is in the resubmission process. The 60-day FR notice published on August 21, 2025 (90 FR 40831). The 30-day notice will publish at a future date in the Federal Register before the information collection is formally submitted to OMB for reapproval.
Back to Citation30. The Medicare Coverage Database defines DDD as degenerative disc disease. In addition, we believe that DDD is commonly referred to as degenerative disk disease in the healthcare industry.
Back to Citation31. For more information on the aprevo® Cervical ACDF System's indications, we refer readers to the November 15, 2024, FDA 510(k) clearance letter (K242260) https://www.accessdata.fda.gov/cdrh_docs/pdf24/K242260.pdf.
Back to Citation32. The aprevo® Intervertebral Fusion Device (IFD) received quarterly approval under the alternative pathway effective October 1, 2021, and final approval in the CY 2023 OPPS/ASC final rule (87 FR 71891 through 71895). CMS established device category code C1831 based on the approval of the aprevo® IFD. Device pass-through payment status eligibility for C1831 expired effective September 30, 2024.
Back to Citation33. Centers for Medicare & Medicaid Services (2017). Pub 100-04 Medicare Claims Processing, Transmittal 3941, Change Request 10417, dated December 22, 2017. Accessed at https://www.cms.gov/regulations-and-guidance/guidance/transmittals/2017downloads/r3941cp.pdf.
Back to Citation34. Centers for Medicare & Medicaid Services (2022). Pub 100-04 Medicare Claims Processing, Transmittal 11305, Change Request 12666, dated March 24, 2022. Accessed at https://www.cms.gov/files/document/r11305cp.pdf.
Back to Citation35. Centers for Medicare & Medicaid Services (2024). Pub 100-04 Medicare Claims Processing, Transmittal 12816, Change Request 13784, dated August 29, 2024. Accessed at https://www.cms.gov/files/document/r12816cp.pdf.
36. Centers for Medicare & Medicaid Services (2024). Hospital Outpatient Prospective System Quarterly Addenda Updates October 2024, Addendum B. Accessed at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/addendum-a-b-updates/october-2024-updated-10/02/2024-0.
Back to Citation37. As stated in the CY 2023 OPPS/ASC final rule with comment period (87 FR 71892), the aprevo® (IFD) is indicated for use as an adjunct to fusion at one or more levels of the lumbar spine in patients having an Oswestry Disability Index (ODI) >40 and diagnosed with severe symptomatic adult spinal deformity (ASD) conditions. These patients should have had 6 months of non-operative treatment. The devices are intended to be used with autologous and/or allogenic bone graft comprised of cancellous and/or cortico-cancellous bone graft. These implants may be implanted via a variety of open or minimally invasive approaches. These approaches may include anterior lumbar interbody fusion or lateral lumbar interbody fusion.
Back to Citation38. For examples, please see our decisions regarding CavaClear Inferior Vena Cava Filter Removal Laser Sheath (88 FR 81718 through 81720), which differed from devices described in C2629 and C1773 by mechanism of action, clinical use, impacted anatomy, and FDA clearance pathway; CERAMENT® G (88 FR 81723 through 81725), which differed from the device described in C1734 by composition, mechanisms of action, indication for use, intended patient population, associated treatment cases and procedures, and FDA designation and classification; and AGENTTM. Paclitaxel-Coated Balloon Catheter (89 FR 94136 through 94137), which differed from devices described in C2623 by indication and type of procedure.
Back to Citation39. Centers for Medicare & Medicaid Services (2023). Pub 100-04 Medicare Claims Processing,Transmittal 11801, Change Request 13031, dated January 20, 2023. Accessed at https://www.cms.gov/files/document/r11801cp.pdf.
Back to Citation40. The SCOUT MD Reflectors are implanted percutaneously in soft tissue and may remain in place for 30 days or longer.
Back to Citation41. Effective July 1, 2013, CMS deleted C1879 (Tissue marker, implantable) because it is described by A4648 (Tissue marker, implantable, any type). Centers for Medicare & Medicaid Services (2013). Pub 100-04 Medicare Claims Processing (Transmittal 2718) in CMS Manual System. Accessed at https://www.cms.gov/regulations-and-guidance/guidance/transmittals/2013-transmittals-items/r2718cp.
Back to Citation42. We noted the applicant selected APC 5072 and an APC payment rate of $1,620.24 for the three tests of the cost criteria. However, for our calculation, we selected APC 5071, which we believe had the lowest applicable APC payment rate of $703.59 found in the CY 2025 OPPS/ASC final rule with comment period, among the APCs related to the HCPCS/CPT codes provided by the applicant. We selected the HCPCS/CPT code level device offset amount of $240.56 related to HCPCS 19287 in APC 5071. We stated that, based on our initial assessment in the CY 2026 OPPS/ASC proposed rule, using the APC payment rate of $703.59 and the device offset amount of $240.56 would result in the SCOUT MDTM Delivery System meeting the cost significance requirement.
Back to Citation43. Center of Medicare & Medicaid Services (2025). Pub 100-04 Medicare Claims Processing, Transmittal 13425, Change Request 14223, dated September 22, 2025. Accessed at https://www.cms.gov/files/document/r13425cp.pdf.
Back to Citation44. Nerve Protection Evaluation: Revision Cubital Tunnel Syndrome Decompression (COVERED); (NCT06117501).
Back to Citation45. Bhojani, N, Morris, K, White, J, Rojanasarot, S Tran, E.D, Monga, M. (2025). Post-operative infection with a single-use ureteroscope with real-time intrarenal pressure monitoring vs. all other single-use ureteroscopes. Expert Review of Medical Devices, 1-9. https://doi.org/10.1080/17434440.2025.2557403.
Back to Citation46. As discussed in section IV.A.2 (New Device Pass-Through Applications for CY 2023) of the CY 2023 OPPS/ASC final rule with comment period, we approved C1747 (Endoscope, single-use ( i.e., disposable), urinary tract, imaging/illumination device (insertable)), as a new device category for pass-through status under the OPPS, with an effective date of January 1, 2023. For the full discussion on the criteria used to evaluate device pass-through applications, refer to the CY 2023 OPPS/ASC final rule with comment period, which was published in the Federal Register on November 23, 2022 (87 FR 71929 through 71934). We note that C1747 was established for a ureteroscope that can only be used for a single procedure and cannot be reprocessed. As such, C1747 only describes devices that cannot be reprocessed.
Back to Citation47. U.S. Food and Drug Administration. (2020, April 2). Decision Summary for K193095 [Ambu® aScopeTM. 4 Cysto]. U.S. Department of Health and Human Services. https://www.accessdata.fda.gov/cdrh_docs/pdf19/K193095.pdf.
Back to Citation48. Geldmaker, L.E., Baird, B.A., Lyon, T.D., Regele, E.J., Wajswol, E.J., Pathak, R.A., Petrou, S.P., Haehn, D.A., Gajarawala, N.M., Ball, C.T., Broderick, G.A., & Thiel, D.D. (2023). Conversion to disposable cystoscopes decreased post-procedure encounters and infections compared to reusable cystoscopes. Urology Practice, 12 (1), 58-64. https://doi.org/10.1097/UPJ.0000000000000410.
Back to Citation49. Anderson, S., Patterson, K., Skolarikos, A., Somani, B., Bolton, D.M., & Davis, N.F. (2024). Perspectives on technology: To use or to reuse, that is the endoscopic question—a systematic review of single-use endoscopes. BJU International, 133 (1), 14-24. https://doi.org/10.1111/bju.16206.
50. Johnson, B.A., Raman, J.D., Best, S.L., & Lotan, Y. (2023). Prospective randomized trial of single-use vs reusable cystoscope for ureteral stent removal. Journal of Endourology, 37 (10). https://doi.org/10.1089/end.2023.0134.
Back to Citation51. Anderson, 2024, op. cit.
Back to Citation52. Johnson, 2023, op. cit.
Back to Citation53. Lee, J., Kaplan-Marans, E., Jivanji, D., Tennenbaum, D., & Schulman, A. (2022). Post-cystoscopy infections and device malfunctions in reprocessed flexible cystoscopes in a national database. Canadian Journal of Urology, 29 (6), 11361-11365. https://pubmed.ncbi.nlm.nih.gov/36495577.
54. Muscarella, L.F. (2022, January 28). Contamination of flexible endoscopes and associated infections: A comprehensive review and analysis of FDA adverse event reports. Discussions in Infection Control. https://lfm-hcs.com/2022/01/contamination-of-flexible-endoscopes-and-associated-infections/.
55. U.S. Food and Drug Administration. (2021, April 1). FDA is investigating reports of infections associated with reprocessed urological endoscopes: Agency is taking action to remind health care providers about the proper way to clean certain devices for reuse. [FDA News Release]. https://www.fda.gov/news-events/press-announcements/fda-investigating-reports-infections-associated-reprocessed-urological-endoscopes.
56. U.S. Food and Drug Administration. (2025, January 31). Update on alert: Endoscope accessories forceps/irrigation plug issue from Olympus. https://www.fda.gov/medical-devices/medical-device-recalls/update-alert-endoscope-accessories-forcepsirrigation-plug-issue-olympus.
Back to Citation57. U.S. Food and Drug Administration. (2024, June 6). About the Manufacturer and User Facility Device Experience (MAUDE) database. U.S. Department of Health and Human Services. https://www.fda.gov/medical-devices/mandatory-reporting-requirements-manufacturers-importers-and-device-user-facilities/about-manufacturer-and-user-facility-device-experience-maude-database.
Back to Citation58. U.S. Food and Drug Administration, 2024, op. cit.
Back to Citation59. U.S. Food and Drug Administration, 2024, op. cit.
Back to Citation60. U.S. Food and Drug Administration, 2021, op. cit.
Back to Citation61. U.S. Food and Drug Administration, 2025, op. cit.
Back to Citation62. In the CY 2023 OPPS/ASC final rule with comment period CMS approved Uretero1TM. as a new device category for transitional pass-through payment status and established C1747 as a new device category beginning in January 2023 (87 FR 7129 through 71934) effective January 1, 2023.
Back to Citation63. Chew, B.H., Miller, L.E., Morris, K.C., Shin, Y.E., Tsacogianis, T., White, J., Rojanasarot, S., & Forbes, C.M. (2025) Healthcare Utilization and Complications Associated With Single-Use Versus Reusable Flexible Cystoscopy in Hospital Outpatient Settings. Cureus, 17 (9): e91879. https://doi.org/10.7759/cureus.91879.
Back to Citation64. Device Pass-through Requirements: https://mearis.cms.gov/public/resources.
Back to Citation65. https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient.
Back to Citation66. To apply for OPPS transitional Pass-Through Payment Status and New Technology Ambulatory Payment Classification (APC), applicants complete an application that is subject to the Paperwork Reduction Act of 1995 (PRA) (44 U.S.C. 3501 et seq.). This information collection (CMS-10008) is currently approved under OMB control number of 0938-0802 and has an expiration date of July 31, 2027.
Back to Citation67. In the CY 2025 OPPS/ASC final rule with comment period (89 FR 93948), we finalized the diagnostic radiopharmaceuticals policy to separately pay those products when the per-day costs are greater than a threshold. Please refer to section II.A.3.c. of the CY 2025 OPPS/ASC final rule for more information regarding this policy.
Back to Citation68. https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient.
Back to Citation69. In the CY 2025 OPPS/ASC final rule with comment period (89 FR 93948), we finalized the diagnostic radiopharmaceuticals policy to separately pay those products when the per-day costs are greater than a threshold. Please refer to section II.A.3.c. of the CY 2025 OPPS/ASC final rule for more information regarding this policy.
Back to Citation70. https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient.
Back to Citation71. In the CY 2025 OPPS/ASC final rule with comment period (89 FR 93948), we finalized a diagnostic radiopharmaceuticals policy to separately pay those products when the per-day costs are greater than a threshold. Please refer to section II.A.3.c. of the CY 2025 OPPS/ASC final rule for more information regarding this policy.
Back to Citation72. https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient.
Back to Citation73. Medicare Payment Advisory Committee. June 2005 Report to the Congress. Chapter 6: Payment for pharmacy handling costs in hospital outpatient departments. Available at https://www.medpac.gov/wp-content/uploads/import_data/scrape_files/docs/default-source/reports/June05_ch6.pdf.
Back to Citation74. https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient.
Back to Citation75. https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient.
Back to Citation76. https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient.
Back to Citation78. Government Accountability Office. “Medicare Part B Drugs: “Action Needed to Reduce Financial Incentives to Prescribe 340B Drugs at Participating Hospitals.” June 2015. Available at https://www.gao.gov/assets/gao-15-442.pdf.
Back to Citation79. Office of Inspector General. “Part B Payment for 340B Purchased Drugs. OEI-12-14-00030”. November 2015. Available at: https://oig.hhs.gov/oei/reports/oei-12-14-00030.pdf.
Back to Citation80. Medicare Payment Advisory Commission. March 2016 Report to the Congress: Medicare Payment Policy. March 2016. Available at Medicare Payment Advisory Commission. March 2016 Report to the Congress: Medicare Payment Policy. March 2016. Available at https://www.medpac.gov/document/http-www-medpac-gov-docs-default-source-reports-may-2015-report-to-the-congress-overview-of-the-340b-drug-pricing-program-pdf/.
Back to Citation81. The 69.46 percent of AWP was calculated by first reducing the original 95 percent of AWP price by 6 percent to generate a value that is similar to ASP or WAC with no percentage markup. Then we applied the 22.5 percent reduction to ASP/WAC- similar AWP value to obtain the 69.46 percent of AWP, which was similar to either ASP minus 22.5 percent or WAC minus 22.5 percent.
Back to Citation85. Available at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices/cms-1834-p.
Back to Citation86. See 88 FR 77169 through 77181; see also Id. at 77156 through 77161; 90 FR 33633 through 33634.
Back to Citation88. Prior Advance Notice and Rate Announcement documents are available at https://www.cms.gov/medicare/payment/medicare-advantage-rates-statistics/announcements-and-documents.
Back to Citation90. 89 FR 101607 (December 16, 2024); https://www.federalregister.gov/documents/2024/12/16/2024-29505/reimbursement-rates-for-calendar-year-2025.
Back to Citation91. Sections 321(a) and 322(b) of the Public Health Service Act (42 U.S.C. 248(a) and 249(b)), Public Law 83-568 (42 U.S.C. 2001(a)), and the Indian Health Care Improvement Act (25 U.S.C. 1601 et seq.).
Back to Citation92. https://oig.hhs.gov/reports-and-publications/workplan/summary/wp-summary-0000894.asp.
Back to Citation93. The term “FDA regulatory categories” is used in this final rule with comment period when referring to the basis for CMS' payment policies but is not intended to reflect or imply that the products discussed within this final rule with comment period are characterized as such or grouped together by FDA.
Back to Citation94. CMS Skin Substitutes Town Hall, which was held virtually on January 18, 2023. More information regarding the CMS Skin Substitutes Town Hall such as links to recording and transcripts is available at https://www.cms.gov/medicare/payment/fee-schedules/physician/skin-substitutes#:~:text=The%20CMS%20Skin%20Substitutes%20Town,Physician%20Fee%20Schedule%20(PFS).
Back to Citation95. We note that establishments may seek feedback from FDA regarding their self-determination analysis and conclusion that a particular product is a 361 HCT/P. See, for example, https://www.fda.gov/vaccines-blood-biologics/tissue-tissue-products/tissue-reference-group.
Back to Citation96. No premarket authorization is required for 361 HCT/Ps.
97. https://www.fda.gov/industry/fda-user-fee-programs/medical-device-user-fee-amendments-mdufa.
98. These numbers include either a review within 180 days for decisions without advisory committee input or a review within 320 days for decisions with advisory committee input, respectively.
99. PDUFA performance goals call for FDA to review and act on 90 percent of original BLA submissions within 10 months of the 60-day filing date. Other regulatory pathways may have different timelines. See https://www.fda.gov/patients/learn-about-drug-and-device-approvals/fast-track-breakthrough-therapy-accelerated-approval-priority-review; https://www.fda.gov/drugs/development-approval-process-drugs.
100. https://www.fda.gov/industry/fda-user-fee-programs/prescription-drug-user-fee-amendments.
Back to Citation103. Tettelbach W, Armstrong DG, Driver V, et al. Safeguarding access, fiscal responsibility and innovation: a comprehensive reimbursement framework for CAMPs to preserve the Medicare Trust Fund. J Wound Care. 2025;34(10):Ahead of Print. doi:10.12968/jowc.2025.0396.
Back to Citation104. See Regulatory Considerations for HCT/Ps: Minimal Manipulation and Homologous Use, July 2020 (pg. 19).
Back to Citation105. FDA Executive Summary Prepared for the October 26 & 27, 2022 Meeting of the General and Plastic Surgery Devices Panel of the Medical Devices Advisory Panel Classification of Wound Dressings with Animal-derived Materials (Section 3). Available at https://www.fda.gov/media/162539/download.
Back to Citation106. A 510(k) is a premarket submission made to the FDA generally by the manufacturer of a new device to demonstrate that the device to be marketed is substantially equivalent to a legally marketed device that is not subject to premarket approval (sections 510(k) and 513(i) of the FD&C Act).
Back to Citation107. Office of Inspector General, U.S. Department of Health and Human Services. Medicare Part B Payment Trends for Skin Substitutes Raise Major Concerns About Fraud, Waste, and Abuse. September 10, 2025.
Back to Citation108. 89.6 percent of AWP was calculated by first reducing the usual 95 percent of AWP price by 6 percent to generate a value that is similar to WAC with no percentage markup.
Back to Citation111. “Eluted” refers to the process by which Tc-99m is chemically separated from Mo-99 within the generator and collected in an elution vial.
Back to Citation112. For the definition of a SCOD, see section 1833(t)(14)(B) of the Act at https://www.ssa.gov/OP_Home/ssact/title18/1833.htm.
Back to Citation115. Government Accountability Office. “Medicare Hospital Pharmaceuticals: “Survey Shows Price Variation and Highlights Data Collection Lessons and Outpatient Rate-Setting Challenges for CMS” April 2006. Available at https://www.gao.gov/assets/gao-06-372.pdf.
Back to Citation118. https://www.reginfo.gov/public/do/PRAViewDocument?ref_nbr=202507-0938-017.
Back to Citation120. https://www.gao.gov/assets/gao-06-372.pdf. Page 7.
Back to Citation121. FSS pricing from the Veteran Affairs' (VA's) pharmaceutical pricing database is publicly available at the NDC level and published at https://www.va.gov/opal/nac/fss/pharmPrices.asp.
Back to Citation122. section 340B(a)(1) of the Public Health Service Act. https://www.hrsa.gov/about/faqs/what-difference-between-340b-ceiling-price-package-adjusted-price-which-are-both-published-340b.
Back to Citation123. In the CY 2025 OPPS/ASC final rule with comment period, we finalized the high-cost diagnostic radiopharmaceuticals policy to separately pay those products when the per-day costs are greater than a threshold. Please refer to section II.A.3.c. of this final rule with comment period for more information regarding this policy.
Back to Citation124. Estimated costs are updated to reflect the Medicare hospital outpatient claims data for each HCPCS code as such data becomes available. Prior to the availability of Medicare hospital outpatient claims data, estimated costs are based on the device cost and projected CY OPPS utilization provided by the device manufacturer in the device pass-through application, As such, the final estimated pass-through costs for HCPCS codes C1600, C1601, C1602, C1603 and C1604 are updated based on CY 2024 Medicare hospital outpatient claims data available for this final rule with comment period. The final estimated pass-through costs for HCPCS codes C1605, C1606, C8000, C1735, C1736, C1737, C1738, C1739, and C9610 are based on information provided by the device manufacturers in the respective device pass-through applications regarding the device cost and the projected CY 2026 OPPS utilization.
Back to Citation125. As discussed in section IV.A.2 of this final rule with comment period, the VasQ pass-through application was preliminarily approved for transitional pass-through payment under the alternative pathway effective October 1, 2024, and the SCOUT MDTM Surgical Guidance System pass-through application was preliminarily approved for transitional pass-through payment under the alternative pathway effective January 1, 2025. We are finalizing the approvals for device pass-through payment status for VasQ and the SCOUT MDTM Surgical Guidance System in this final rule with comment period. Due to the timing of the preliminary approvals, the CY 2026 spending estimate for VasQ and the SCOUT MDTM Surgical Guidance System is included in the CY 2026 spending estimate for the first group of devices.
Back to Citation126. Centers for Medicare & Medicaid Services (2025). Pub 100-04 Medicare Claims Processing, Transmittal 13425, Change Request 14223, dated September 22, 2025. Accessed at https://www.cms.gov/files/document/r13425cp.pdf.
127. Per the CY 2016 OPPS/ASC final rule with comment period, device pass-through applications are still submitted to CMS through the quarterly process, but the applications are subject to notice and comment rulemaking in the next applicable OPPS annual rulemaking cycle. Under this process, all applications that are preliminarily approved upon quarterly review will automatically be included in the next applicable OPPS annual rulemaking cycle (80 FR 70417 through 70418). Applications received after the March 3, 2025, deadline for the remaining 2025 quarters (the quarters beginning June 1, September 1, and December 1 of 2025), will be discussed in the CY 2027 OPPS/ASC proposed rule. As such, we expect to include and discuss the device applications associated with HCPCS codes C1740, C1741 and C1742 in the CY 2027 OPPS/ASC proposed and final rules with comment period.
Back to Citation128. As we discussed in the CY 2023 OPPS/ASC final rule with comment period, our longstanding ratesetting methodology defaults any CMHC CCR that is not available or any CMHC CCR greater than one to the Statewide hospital CCR associated with the provider's urban/rural designation and their State location.
Back to Citation129. Click on the link labeled “CY 2026 OPPS/ASC Notice of Final Rulemaking”, which can be found under the heading “Hospital Outpatient Prospective Payment System Rulemaking” and open the claims accounting document link at the bottom of the page, which is labeled “2026 NFRM OPPS Claims Accounting (PDF)”.
Back to Citation130. https://www.medpac.gov/wp-content/uploads/2023/03/Mar23_MedPAC_Report_To_Congress_SEC.pdf.
Back to Citation131. In this rulemaking, we proposed to eliminate, the IPO list, beginning in CY 2026, with all services being removed from the list over the course of a three-year transition period. The CY 2026 IPO List can be found here: Hospital Outpatient PPS, https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/HospitalOutpatientPPS/index.
Back to Citation132. https://www.healthaffairs.org/doi/10.1377/hlthaff.2016.0830.
133. https://onlinelibrary.wiley.com/doi/10.1111/1475-6773.13613.
Back to Citation134. https://jamanetwork.com/journals/jamainternalmedicine/fullarticle/2463591.
135. https://www.healthaffairs.org/doi/10.1377/hlthaff.2020.01183.
136. https://onlinelibrary.wiley.com/doi/10.1111/1475-6773.14172.
Back to Citation137. https://www.siteneutral.org/wp-content/uploads/2016/06/14_USON-Moran-Report-08272013.pdf.
Back to Citation142. Based on our analysis of claims data and Medicare FFS enrollment.
Back to Citation143. Based on our analysis of claims data.
Back to Citation144. HCPCS included in the chemotherapy administration category are: 96423, 96549, 96401, 96402, 96405, 96411, 96415, 96417, 96406, 96409, 96422, 96542, 96413, 96416, 96420, 96425, 96440, 96446, 96450, G0498.
Back to Citation153. Available in Table IV.B6. at https://www.cms.gov/Research-Statistics-Data-and-Systems/Statistics-Trends-and-Reports/ReportsTrustFunds/Downloads/TR2019.pdf.
Back to Citation154. Available in Tables IV.B3. and B6 at https://www.cms.gov/oact/tr/2024.
Back to Citation155. Available in Table IV.B3. at https://www.cms.gov/oact/tr/2024.
Back to Citation158. In comparison—in CY 2020 when the clinic visit volume control method was fully phased-in the estimated savings was approximately $800 million, with approximately $640 million of the savings accruing to Medicare, and approximately $160 million saved by Medicare beneficiaries in the form of reduced copayments.
Back to Citation159. Dranove D, Ody C. Employed for Higher Pay? how Medicare Payment Rules Affect Hospital Employment of Physicians. American economic journal. Economic policy. 2019;11(4):249-271. https://www.aeaweb.org/articles?id=10.1257/pol.
160. Neprash HT, Chernew ME, Hicks AL, Gibson T, McWilliams JM. Association of Financial Integration between Physicians and Hospitals with Commercial Health Care Prices. JAMA internal medicine. 2015;175(12):1932-1939. https://jamanetwork.com/journals/jamainternalmedicine/fullarticle/2463591.
161. Post B, Norton EC, Hollenbeck B, Buchmueller T, Ryan AM. Hospital-physician Integration and Medicare's Site-based Outpatient Payments. Health services research. 2021;56(1):7-15. https://pmc.ncbi.nlm.nih.gov/articles/PMC7839648/.
Back to Citation165. Based on our analysis of claims data and Medicare FFS enrollment.
Back to Citation166. Based on our analysis of claims data.
Back to Citation167. HCPCS included in the chemotherapy administration category are: 96423, 96549, 96401, 96402, 96405, 96411, 96415, 96417, 96406, 96409, 96422, 96542, 96413, 96416, 96420, 96425, 96440, 96446, 96450, G0498.
Back to Citation168. Available in Table IV.B6 at https://www.cms.gov/Research-Statistics-Data-and-Systems/Statistics-Trends-and-Reports/ReportsTrustFunds/Downloads/TR2019.pdf.
Back to Citation171. Kane, Carol K., Recent Changes in Physician Practice Arrangements: Private Practice Dropped to Less Than 50 Percent of Physicians in 2020, Chicago, Ill.: American Medical Association, 2021.
Back to Citation173. https://www.healthaffairs.org/doi/full/10.1377/hlthaff.2018.0472.
174. https://doi.org/10.1016/j.jhealeco.2005.04.009.
175. https://www.healthaffairs.org/doi/10.1377/hlthaff.2013.1279.
176. https://doi.org/10.1016/j.jhealeco.2018.04.001.
177. https://onlinelibrary.wiley.com/doi/full/10.1002/hec.3502.
178. https://www.healthaffairs.org/doi/full/10.1377/hlthaff.2021.01007.
Back to Citation179. https://doi.org/10.1016/j.jhealeco.2018.04.001.
180. https://www.healthaffairs.org/doi/full/10.1377/hlthaff.2020.01006.
Back to Citation181. https://doi.org/10.1016/j.jhealeco.2016.08.006.
182. https://doi.org/10.1002/hec.3502.
Back to Citation184. https://pmc.ncbi.nlm.nih.gov/articles/PMC1361007/.
185. https://jamanetwork.com/journals/jama/fullarticle/1917439.
186. https://jamanetwork.com/journals/jamainternalmedicine/fullarticle/2463591.
187. https://pmc.ncbi.nlm.nih.gov/articles/PMC7080686/.
188. https://onlinelibrary.wiley.com/doi/10.1111/1475-6773.13613.
189. https://doi.org/10.1016/j.jhealeco.2016.12.007.
190. https://www.healthaffairs.org/doi/full/10.1377/hlthaff.2020.01006.
191. https://doi.org/10.1016/j.jhealeco.2016.08.006.
192. https://pmc.ncbi.nlm.nih.gov/articles/PMC8810743/.
Back to Citation198. 42 CFR 485.601 through 485.647.
Back to Citation206. Mills CA, Yeager VA, Unroe KT, Holmes A, Blackburn J. The impact of rural general hospital closures on communities—A systematic review of the literature. J Rural Health. 2024;40:238-248. https://doi.org/10.1111/jrh.12810.
Back to Citation207. In the CY 2023 OPPS/ASC final rule with comment period, we removed § 410.27(a)(1)(iv)(D) in its entirety and added its language regarding pulmonary rehabilitation, cardiac rehabilitation, and intensive cardiac rehabilitation services and the virtual presence of a physician through audio/video real-time communications technology during the PHE to the newly designated § 410.27(a)(1)(iv)(B)( 1) (87 FR 72024).
Back to Citation208. Ibid.
Back to Citation210. On May 22, 2025, CMS announced that responsibility for short-stay reviews will be transitioned from the BFCC-QIOs to the MACs, as of September 1, 2025. https://www.cms.gov/data-research/monitoring-programs/medicare-fee-service-compliance-programs/medical-review-and-education/hospital-patient-status-reviews. BFCC-QIOs will continue to review post-payment inpatient hospital claims for higher weighted Diagnosis Related Groups, hospital discharge and service termination appeals, and quality of care concerns. While the BFCC-QIO will not specifically select short stay claims for the purpose of assessing compliance with the two-midnight rule, if the BFCC-QIO encounters a hospital short inpatient stay claim when reviewing for other reasons, the medical record will also be reviewed for compliance with the two-midnight rule.
Back to Citation212. Medicare Payment Advisory Committee. March 2025 Report to the Congress. Chapter 3: Hospital inpatient and outpatient services, pp. 61-94. Available at https://www.medpac.gov.
Back to Citation214. FDA approval letter, April 10, 2025, (K250371) https://www.accessdata.fda.gov/cdrh_docs/pdf25/K250371.pdf.
Back to Citation215. O'Connor LA, Dua A, Orhurhu V, Hoepp LM, Quinn CC. Opioid Requirements After Intercostal Cryoanalgesia in Thoracic Surgery. J Surg Res. 2022; 274:232-241.
216. Maxwell CM, Weksler B, Houda J, Fernando HC. Intercostal Cryoablation During Video-Assisted Lung Resection Can Decrease Postoperative Opioid Use. Innovations 2023 18(4):352-356.
217. Jaroszewski DE, Bostoros P, Farina JM, Botros MM, Aly MR, Peterson M, Lackey J, Pulivarthi KV, Smith B, Craner R, Stearns JD. Evolution of Pain Control for Adult Pectus Excavatum Repair. Ann Thorac Surg. 2024;117(4):829-837.
218. Graves CE, Moyer J, Zobel MJ, Mora R, Smith D, O'Day M, Padilla BE. Intraoperative intercostal nerve cryoablation during the Nuss procedure reduces length of stay and opioid requirement: A randomized clinical trial. J Pediatric Surg. 2019 Nov;54(11):2250-2256.
219. Miller DL, Hutchins J, Ferguson MA, Barhoush Y, Achter E, Kuckelman JP. Intercostal Nerve Cryoablation During Lobectomy for Postsurgical Pain: A Safe and Cost-Effective Intervention. Pain Ther. 2025 Feb; 14(1):317-328. doi: 10.1007/s40122-024-00694-3.
220. Kleiboeker HL, Hall DJ, Lowery EM, Hayney MS, Maloney JD, DeCamp MM, McCarthy DP. Intercostal nerve cryoablation as part of an opioid-sparing protocol reduces opioid and epidural use after lung transplant. JHLT Open. 2024;4:100084.
221. Notrica DM, McMahon LE, Hahn A, Ndikintum N, Dua M, Jaroszewski DE. Intercostal Nerve Cryoablation During Pectus Excavatum Surgery for Postoperative Pain Management: A Systematic Review and Meta-Analysis. Ann Surg. 2025; doi: 10.1097/SLA.0000000000006855. Epub ahead of print.
Back to Citation222. FDA Approval Letter, November 7, 2020, http://www.accessdata.fda.gov/cdrh_docs/pdf19/K192860.pdf.
Back to Citation223. FDA Approval Letter, November 7, 2020, http://www.accessdata.fda.gov/cdrh_docs/pdf19/K192860.pdf.
Back to Citation224. Liang, T.-W., Shen, C.-H., Wu, Y.-S., Chang, Y.-T. (2024) Erector spinae plane block reduces opioid consumption and improves incentive spirometry volume after cardiac surgery: A 4 | Page Eitan Medical North America Inc. 65 Enterprise—STE 485, Aliso Viejo, CA 92656 www.eitanmedical.com Tel. 877-541-9944 | Fax. 949-288-5482 retrospective cohort study. Journal of the Chinese Medical Association, 87(5): 550-557. DOI: ad10.1097/JCMA.0000000000001086.
Back to Citation225. FDA Approval Letter, March 10, 2023, https://www.accessdata.fda.gov/cdrh_docs/pdf21/K213744.pdf.
Back to Citation226. FDA Approval Letter (SonoPlex), June 27, 2025, https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfpmn/pmn.cfm?ID=K243682.
227. FDA Approval Letter (SonoBlock), November 11, 2024, https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfpmn/pmn.cfm?ID=K241954.
228. FDA Approval Letter (SonoTap), March 24, 2025, https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfpmn/pmn.cfm?ID=K243525.
Back to Citation229. Sahap et al. (2023), Ultrasound-guided vs Laparoscopic-assisted Transversus Abdominis Plane Block for Laparoscopic Cholecystectomy: A Randomized Prospective Study.
230. Cosarcan (2024), The effect of ultrasound-guided rectus sheath block on postoperative analgesia in robot assisted prostatectomy: A randomized controlled trial.
231. Kukreja et al. (2023), Quality of recovery after pericapsular nerve group (PENG) block for primary total hip arthroplasty under spinal anaesthesia: a randomised controlled observer-blinded trial.
Back to Citation232. FDA approval letter, June 20, 2008, https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfpmn/pmn.cfm?ID=K080675.
Back to Citation233. Narayan et al (2021), Continuous Wound Infusion as an Alternative to Continuous Epidural Infusion for Postoperative Analgesia in Renal Transplant Surgery: A Prospective Randomized Controlled Trial.
Back to Citation234. FDA approval letter, May 4, 1992, https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfpmn/pmn.cfm?id=K911202.
235. FDA approval letter, March 14, 2025, https://www.accessdata.fda.gov/cdrh_docs/pdf25/K250774.pdf.
Back to Citation236. FDA approval letter, May 4, 1992, https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfpmn/pmn.cfm?id=K911202.
Back to Citation237. FDA approval letter (E-Cath), July 13, 2016, https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfpmn/pmn.cfm?ID=K152952.
238. FDA approval letter (SonoLong), March 1, 2012, https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfpmn/pmn.cfm?ID=K113188.
Back to Citation239. Al Ja'bari et al. (2019), A randomised controlled trial of the pectoral nerves-2 (PECS-2) block for radical mastectomy.
Back to Citation240. Id.
Back to Citation241. Kim et al. (2022), Initiation Timing of Continuous Interscalene Brachial Plexus Blocks in Patients Undergoing Shoulder Arthroplasty: A Retrospective Before and-After Study.
Back to Citation242. FDA approval letter, August 24 2017, https://www.accessdata.fda.gov/cdrh_docs/pdf17/K170982.pdf.
Back to Citation243. Abbasian, et al (2022). Multimodal continuous ambulatory erector spinae catheter pain protocol for early recovery following Nuss procedure: a retrospective cohort study; American Society of Regional Anesthesia & Pain Medicine.
Back to Citation244. FDA Approval letter, June 30, 2023, https://www.accessdata.fda.gov/cdrh_docs/pdf23/P230020A.pdf.
Back to Citation245. Kapural L, Melton J, Kim B, Mehta P, Sigdel A, Bautista A, Petersen EA, Slavin KV, Eidt J, Wu J, Elshihabi S, Schwalb JM, Garrett HE Jr, Veizi E, Barolat G, Rajani RR, Rhee PC, Guirguis M, Mekhail N. Primary 3-Month Outcomes of a Double-Blind Randomized Prospective Study (The QUEST Study) Assessing Effectiveness and Safety of Novel High-Frequency Electric Nerve Block System for Treatment of Post- Amputation Pain. J Pain Res. 2024;17:2001-2014. https://doi.org/10.2147/JPR.S463727.
Back to Citation246. Id.
Back to Citation247. FDA Approval Letter, June 16, 2020. https://www.accessdata.fda.gov/cdrh_docs/pdf19/P190021A.pdf.
Back to Citation248. Gilligan, et al. 2024. Five-Year Longitudinal Follow-Up of Restorative Neurostimulation Shows Durability of Effectiveness in Patients With Refractory Chronic Low Back Pain Associated With Multifidus Muscle Dysfunction. Neuromodulation. 2024 Jul;27(5):930-943. doi: 10.1016/j.neurom.2024.01.006. Epub 2024 Mar 12. PMID: 38483366.
Back to Citation249. FDA 510(k) clearance K192114, 2019, https://www.accessdata.fda.gov/cdrh_docs/pdf19/K192114.pdf.
Back to Citation250. Khan, et al., Cryo-Pneumatic Compression Results in a Significant Decrease in Opioid Consumption After Shoulder Surgery: A Multicenter Randomized Controlled Trial. Am J Sports Med. 2024 Sep;52(11):2860-2865.
Back to Citation252. Centers for Disease Control and Prevention. (May 2024). About Emotional Well-Being. Available at https://www.cdc.gov/emotional-well-being/about/#cdc_behavioral_basics_types-health-benefits. Accessed: April 30, 2025.
Back to Citation253. Health Measures. (March 2023). PROMIS. Available at https://www.healthmeasures.net/explore-measurement-systems/promis.
254. World Health Organization (October 2024). The World Health Organization-Five Well-Being Index (WHO-5). Available at https://www.who.int/publications/m/item/WHO-UCN-MSD-MHE-2024.01.
255. American Psychological Association. Patient Health Questionnanire-9 (PHQ-9). Available at https://www.apa.org/depression-guideline/patient-health-questionnaire.pdf.
256. COPM. (2025). The COPM is an individualized, client-centered outcome measure. Available at https://www.thecopm.ca.
257. Shirely Ryan Ability Lab. (April 2018). Occupational Self-Assessment. Available at https://www.sralab.org/rehabilitation-measures/occupational-self-assessment.
258. Mapi Research Trust. (2012). Self-Management Ability Scale-Short Form (SMAS-S). Available at https://eprovide.mapi-trust.org/instruments/self-management-ability-scale-short-form.
Back to Citation259. American Speech-Language-Hearing Association. (2020). Functional Communication Measures (FCMs). Available at https://www.asha.org/siteassets/noms/slp-noms-functional-communication-measures.pdf?srsltid=AfmBOoqymclX2j2Mt7aSFrul0T_PdYL1oNzRZAuzzYO1SBp9mHNe5r0i.
Back to Citation260. Office of Disease Prevention and Health Promotion. United States Core Data for Interoperability. Available at https://odphp.health.gov/foodismedicine/federal-resource-hub/united-states-core-data-interoperability.
261. eCQI Resource Center. (June, 2025). FHIR—Fast Healthcare Interoperability Resources. Available at https://ecqi.healthit.gov/fhir/about.
Back to Citation262. Centers for Disease Control and Prevention. (2025). Weekly COVID-19 Vaccination Module for Healthcare Personnel. Available at https://www.cdc.gov/nhsn/pdfs/hps/covidvax/2025-hcp-combined-protocol-508.pdf. Accessed: April 30, 2025.
Back to Citation263. U.S. Department of Health and Human Services. (2023). COVID-19 Public Health Emergency. Available at https://www.hhs.gov/coronavirus/covid-19-public-health-emergency/index.html. Accessed: April 30, 2025.
Back to Citation264. Centers for Disease Control and Prevention. (2025). COVID Data Tracker. Available at https://covid.cdc.gov/covid-data-tracker/#trends_totaldeaths_select_00. Accessed: April 30, 2025.
Back to Citation265. Centers for Disease Control and Prevention. (2025). COVID Data Tracker. Available at https://covid.cdc.gov/covid-data-tracker/#trends_weeklydeaths_select_00. Accessed: April 30, 2025.
Back to Citation266. Centers for Disease Control and Prevention. (2025). COVID Data Tracker. Available at https://covid.cdc.gov/covid-data-tracker/#trends_weeklydeaths_select_00. Accessed: April 30, 2025.
Back to Citation267. Centers for Medicare & Medicaid Services (2024, December). Blueprint Measure Lifecycle. Measures Management System. Available at https://mmshub.cms.gov/blueprint-measure-lifecycle-overview.
Back to Citation268. Agency for Healthcare Research and Quality. (Oct 2025) Types of Health Care Quality Measures. Available at https://www.ahrq.gov/talkingquality/measures/types.html.
Back to Citation269. Centers for Medicare & Medicaid Services (2024, December). Blueprint Measure Lifecycle. Measures Management System. Available at https://mmshub.cms.gov/blueprint-measure-lifecycle-overview.
Back to Citation270. Centers for Medicare & Medicaid Services. (May 2024). Quality Program Extraordinary Circumstances Exceptions (ECE) Request Form. QualityNet. Available at https://qualitynet.cms.gov/files/677e843f50ed8df7419f60e1?filename=HQR_ECE_Req_Form_CY_2025.pdf. Accessed: April 30, 2025.
Back to Citation271. Centers for Medicare & Medicaid Services. Hospital OQR Program Extraordinary Circumstances Exceptions (ECE) Policy: https://qualitynet.cms.gov/outpatient/oqr/participation%23tab2#tab2; REHQR Program Extraordinary Circumstances Exceptions (ECE) Policy: https://qualitynet.cms.gov/reh/rehqr/participation#tab2; and ASCQR Program Extraordinary Circumstances Exceptions (ECE) Policy: https://qualitynet.cms.gov/asc/ascqr/participation%23tab3#tab2. Accessed: April 30, 2025.
Back to Citation272. Centers for Medicare & Medicaid Services. Hospital OQR Program Extraordinary Circumstances Exceptions (ECE) Policy https://qualitynet.cms.gov/outpatient/oqr/participation%23tab2#tab2; REHQR Program Extraordinary Circumstances Exceptions (ECE) Policy: https://qualitynet.cms.gov/reh/rehqr/participation#tab2; and ASCQR Program Extraordinary Circumstances Exceptions (ECE) Policy: https://qualitynet.cms.gov/asc/ascqr/participation%23tab3#tab2. Accessed: April 30, 2025.
Back to Citation273. Moore, C. & Heckmann R. (2025). Hospital Boarding In The ED: Federal, State, And Other Approaches. Health Affairs Forefront. Available at https://www.healthaffairs.org/content/forefront/hospital-boarding-ed-federal-state-and-other-approaches. Accessed: April 30, 2025.
Back to Citation274. Moore, C. & Heckmann R. (2025). Hospital Boarding In The ED: Federal, State, And Other Approaches. Health Affairs Forefront. Available at https://www.healthaffairs.org/content/forefront/hospital-boarding-ed-federal-state-and-other-approaches. Accessed: April 30, 2025.
Back to Citation275. Agency for Healthcare Research and Quality. (2025). Technical Report: AHRQ Summit To Address Emergency Department Boarding. Available at https://www.ahrq.gov/sites/default/files/wysiwyg/topics/ed-boarding-summit-report.pdf. Accessed: April 30, 2025.
Back to Citation276. Gaieski, D.F., Agarwal, A.K., Mikkelsen, M.E., Drumheller, B., Cham Sante, S., Shofer, F.S., Goyal, M., & Pines, J.M. (2017). The impact of ED crowding on early interventions and mortality in patients with severe sepsis. The American Journal of Emergency Medicine, 35 (7), 953-960. Available at https://doi.org/10.1016/j.ajem.2017.01.061. Accessed: April 30, 2025.
277. Laam L.A., Wary A.A., Strony R.S., Fitzpatrick M.H., & Kraus C.K. (2021). Quantifying the impact of patient boarding on emergency department length of stay: All admitted patients are negatively affected by boarding. Journal of American College Emergency Physicians, 2(2):e12401. Available at https://doi.org/10.1002/emp2.12401. Accessed: April 30, 2025.
Back to Citation278. Janke, A.T., Melnick, E.R., & Venkatesh, A.K. (2022). Monthly Rates of Patients Who Left Before Accessing Care in US Emergency Departments, 2017-2021. JAMA, 5(9), e2233708. Available at https://doi.org/10.1001/jamanetworkopen.2022.33708. Accessed: April 30, 2025.
Back to Citation279. Laam L.A., Wary A.A., Strony R.S., Fitzpatrick M.H., & Kraus C.K. (2021). Quantifying the impact of patient boarding on emergency department length of stay: All admitted patients are negatively affected by boarding. Journal of American College Emergency Physicians, 2(2):e12401. Available at https://doi.org/10.1002/emp2.12401. Accessed: April 30, 2025.
Back to Citation280. Hsuan, C., Segel, J.E., Hsia, R.Y., Wang, Y., & Rogowski, J. (2023). Association of emergency department crowding with inpatient outcomes. Health Services Research, 58(4), 828-843. Available at https://doi.org/10.1111/1475-6773.14076. Accessed: April 30, 2025.
Back to Citation281. Gaieski, D.F., Agarwal, A.K., Mikkelsen, M.E., Drumheller, B., Cham Sante, S., Shofer, F.S., Goyal, M., & Pines, J.M. (2017). The impact of ED crowding on early interventions and mortality in patients with severe sepsis. The American Journal of Emergency Medicine, 35(7), 953-960. Available at https://doi.org/10.1016/j.ajem.2017.01.061. Accessed: April 30, 2025.
Back to Citation282. Reznek, M.A., Larkin, C.M., Scheulen, J.J., Harbertson, C.A., & Michael, S.S. (2021). Operational factors associated with emergency department patient satisfaction: Analysis of the Academy of Administrators of Emergency Medicine/Association of Academic Chairs of Emergency Medicine national survey. Academic Emergency Medicine: Official Journal of the Society for Academic Emergency Medicine, 28(7), 753-760. Available at https://doi.org/10.1111/acem.14278. Accessed: April 30, 2025.
283. Loke, D.E., Green, K.A., Wessling, E.G., Stulpin, E.T., & Fant, A.L. (2023). Clinicians' Insights on Emergency Department Boarding: An Explanatory Mixed Methods Study Evaluating Patient Care and Clinician Well-Being. Joint Commission Journal on Quality and Patient Safety, 49(12), 663-670. Available at https://doi.org/10.1016/j.jcjq.2023.06.017. Accessed: April 30, 2025.
Back to Citation284. Gaieski, D.F., Agarwal, A.K., Mikkelsen, M.E., Drumheller, B., Cham Sante, S., Shofer, F.S., Goyal, M., & Pines, J.M. (2017). The impact of ED crowding on early interventions and mortality in patients with severe sepsis. The American Journal of Emergency Medicine, 35(7), 953-960. Available at https://doi.org/10.1016/j.ajem.2017.01.061. Accessed: April 30, 2025.
Back to Citation285. The Emergency Care Access and Timeliness eCQM was previously named the Emergency Care Capacity and Quality (ECCQ) eCQM. The name of the measure has been updated to better reflect the purpose of the measure based on feedback from the Pre-Rulemaking Measure Review (PRMR) Hospital Recommendation Group Meeting on January 16, 2025. Available at https://p4qm.org/sites/default/files/2025-02/PRMR-Hospital-Recommendation-Group-Meeting-Summary.pdf. Accessed: April 30, 2025.
Back to Citation286. Partnership for Quality Measurement. Emergency Care Capacity and Quality. Available at https://p4qm.org/measures/4625e. Accessed: April 30, 2025.
Back to Citation287. For proposed measure specifications, we refer readers to the eCQI Resource Center at https://ecqi.healthit.gov/ecqm/hosp-outpt/2027/cms1244v1, or the CMS QualityNet Hospital OQR Program website at https://qualitynet.cms.gov/outpatient.
Back to Citation288. For proposed measure specifications, we refer readers to the eCQI Resource Center at https://ecqi.healthit.gov/ecqm/hosp-outpt/2027/cms1244v1, or the CMS QualityNet Hospital OQR Program website at https://qualitynet.cms.gov/outpatient.
Back to Citation289. ED observations stays are defined as an observation encounter where the patient remains physically in an area under control of the ED and under the care of an ED clinician inclusive of observation in a hospital bed. Partnership for Quality Measurement. Emergency Care Capacity and Quality. Available at https://p4qm.org/measures/4625e. Accessed: April 30, 2025.
Back to Citation290. Specific codes required to calculate the numerator are outlined in the value set data dictionary and eCQM package (Quality Data Model—QDM output). Please refer to the “Measure Calculation” Section for information: https://p4qm.org/measures/4625e. Accessed: April 30, 2025.
Back to Citation291. Partnership for Quality Measurement. Emergency Care Capacity and Quality. Available at https://p4qm.org/measures/4625e. Accessed: April 30, 2025.
Back to Citation292. Partnership for Quality Measurement. Emergency Care Capacity and Quality. Available at https://p4qm.org/measures/4625e. Accessed: April 30, 2025.
Back to Citation293. The Joint Commission. (2012). Patient Flow through the Emergency Department. Available at https://www.jointcommission.org/en-us/standards/r3-report/r3-report-4. Accessed: April 30, 2025.
294. American College of Emergency Physicians. (2024). Emergency Department Boarding and Crowding. Available at https://www.acep.org/administration/crowding--boarding. Accessed: April 30, 2025.
Back to Citation295. Partnership for Quality Measurement. Emergency Care Capacity and Quality. Available at https://p4qm.org/measures/4625e. Accessed: April 30, 2025.
Back to Citation296. Partnership for Quality Measurement. Emergency Care Capacity and Quality. Available at https://p4qm.org/measures/4625e. Accessed: April 30, 2025.
Back to Citation297. For proposed measure specifications, we refer readers to the CMS QualityNet Hospital OQR Program website at https://qualitynet.cms.gov/outpatient.
Back to Citation298. The principal diagnosis (first listed diagnosis at ED discharge) will be used to define strata inclusion. For this measure's purpose, mental health diagnoses do not include substance use disorder diagnoses. Mental health refers to mental health diagnoses, life stressors and crises, and stress-related physical symptoms.
Back to Citation299. Partnership for Quality Measurement. (2025). 2024-2025 Pre-Rulemaking Measure Review (PRMR) Recommendation Group Final Meeting Summary: Hospital Committee. Available at https://p4qm.org/sites/default/files/2025-02/PRMR-Hospital-Recommendation-Group-Meeting-Summary.pdf. Accessed: April 30, 2025.
Back to Citation300. Partnership for Quality Measurement. (2025). 2024-2025 Pre-Rulemaking Measure Review (PRMR) Recommendation Group Final Meeting Summary: Hospital Committee. Available at https://p4qm.org/sites/default/files/2025-02/PRMR-Hospital-Recommendation-Group-Meeting-Summary.pdf. Accessed: April 30, 2025.
Back to Citation301. Partnership for Quality Measurement. (2025). 2024-2025 Pre-Rulemaking Measure Review (PRMR) Recommendation Group Final Meeting Summary: Hospital Committee. Available at https://p4qm.org/sites/default/files/2025-02/PRMR-Hospital-Recommendation-Group-Meeting-Summary.pdf. Accessed: April 30, 2025.
Back to Citation302. Dyas, S.R., Greenfield, E., Messimer, S., Thotakura, S., Gholston, S., Doughty, T., Hays, M., Ivey, R., Spalding, J., & Phillips, R. (2015). Process-Improvement Cost Model for the Emergency Department. Journal of Healthcare Management, 60(6): 442-57. Available at https://doi.org/10.1097/00115514-201511000-00011. Accessed: April 30, 2025.
Back to Citation303. Partnership for Quality Measurement. (2024). 2024 Pre-Rulemaking Measure Review Preliminary Assessment. Available at https://p4qm.org/sites/default/files/2024-12/PRMR-PA-MUC2024-075.pdf. Accessed: April 30, 2025.
Back to Citation304. Partnership for Quality Measurement. (2025). 2024-2025 Pre-Rulemaking Measure Review (PRMR) Recommendation Group Final Meeting Summary: Hospital Committee. Available at https://p4qm.org/sites/default/files/2025-02/PRMR-Hospital-Recommendation-Group-Meeting-Summary.pdf. Accessed: April 30, 2025.
Back to Citation305. Partnership for Quality Measurement. (2025). 2024-2025 Pre-Rulemaking Measure Review (PRMR) Recommendation Group Final Meeting Summary: Hospital Committee. Available at https://p4qm.org/sites/default/files/2025-02/PRMR-Hospital-Recommendation-Group-Meeting-Summary.pdf. Accessed: April 30, 2025.
Back to Citation306. Yale New Haven Health Services Corporation. (September 2024). Technical Expert Panel (TEP) Evaluation of Measure Emergency Care Capacity and Quality Electronic Clinical Quality Measure (eCQM). Available at https://mmshub.cms.gov/sites/default/files/ECCQ-TEP-Summary-Report-081624.pdf. Accessed: April 30, 2025.
Back to Citation307. Centers for Medicare & Medicaid Services. (2023). Electronic Clinical Quality Measures (eCQMs) Specification, Testing, Standards, Tools, and Community. Available at https://mmshub.cms.gov/sites/default/files/eCQM-Specifications-Testing-Standards-Tools-Community.pdf. Accessed: April 30, 2025.
Back to Citation308. For measure specifications, we refer readers to the eCQI Resource Center at https://ecqi.healthit.gov/ecqm/hosp-outpt/2027/cms1244v1, or the CMS QualityNet Hospital OQR Program website at https://qualitynet.cms.gov/outpatient.
Back to Citation309. The principal diagnosis (first listed diagnosis at ED discharge) will be used to define strata inclusion. For this measure's purpose, mental health diagnoses do not include substance use disorder diagnoses. Mental health refers to mental health diagnoses, life stressors and crises, and stress-related physical symptoms.
Back to Citation310. The Joint Commission. (2012). Patient Flow through the Emergency Department. Available at https://www.jointcommission.org/en-us/standards/r3-report/r3-report-4. Accessed: April 30, 2025.
311. American College of Emergency Physicians. (2024). Emergency Department Boarding and Crowding. Available at https://www.acep.org/administration/crowding-boarding. Accessed: April 30, 2025.
Back to Citation312. For more information about the risk factors considered by the measure developer, we refer readers to the Risk Factor Conceptual Model available at https://p4qm.org/sites/default/files/2025-10/4625e-ECAT-Risk-Factor-Conceptual-Model.pdf.
Back to Citation313. Yale New Haven Health Services Corporation. (April 2024). Public Comment Summary. Available at https://mmshub.cms.gov/sites/default/files/ECCQFinalSpreadsheetofPublicComments04152024.xlsx. Accessed: October 28, 2025.
Back to Citation314. Yale New Haven Health Services Corporation. (April 2024). Public Comment Summary. Available at https://mmshub.cms.gov/sites/default/files/ECCQFinalSpreadsheetofPublicComments04152024.xlsx. Accessed: October 28, 2025.
Back to Citation315. Median Time from ED Arrival to ED Departure for Discharged ED Patients. Available at https://ecqi.healthit.gov/sites/default/files/ecqm/measures/CMS32v8.html.
Back to Citation316. American College of Emergency Physicians. Augustine, J. (2022). Data Registries in Emergency Care. Clinical Emergency Data Registry (CEDR). Available at https://www.acep.org/cedr/newsroom/spring-2022/data_registries_in_emergency_care/. Accessed: October 28, 2025.
Back to Citation317. Welch, S.J., Augustine, J.J., Dong, L., Savitz, L.A., Snow, G., James, B.C. (2012). Volume-Related Differences in Emergency Department Performance. The Joint Commission Journal on Quality and Patient Safety 38 (9): 395-402. Available at https://doi.org/10.1016/s1553-7250(12)38050-1.
Back to Citation318. Partnership for Quality Measurement. Emergency Care Access and Timeliness. Available at https://p4qm.org/measures/4625e. Accessed: October 28, 2025.
319. Venkatesh, A., Ravi, S., Rothenberg, C., et al. (2021). Fair Play: Application of Normalized Scoring to Emergency Department Throughput Quality Measures in a National Registry. Annals of emergency medicine, 77 (5), 501-510. Available at https://doi.org/10.1016/j.annemergmed.2020.10.021. Accessed: November 12, 2025.
Back to Citation320. eCQI Resource Center. Emergency Care Access & Timeliness (HOQR). Available at https://ecqi.healthit.gov/ecqm/hosp-outpt/2027/cms1244v1. Accessed: November 13, 2025.
Back to Citation321. To access the supplemental attachment, select attachments under the downloads option on the https://p4qm.org/measures/4625e website and select section 7.1 in the zip file.
Back to Citation322. Partnership for Quality Measurement. Emergency Care Access and Timeliness. Available at https://p4qm.org/measures/4625e. Accessed: October 15, 2025. Accessed: November 13, 2025.
Back to Citation323. Partnership for Quality Measurement. Emergency Care Access and Timeliness. Available at https://p4qm.org/measures/4625e. Accessed: October 15, 2025. Accessed: November 13, 2025.
Back to Citation324. Partnership for Quality Measurement. Emergency Care Capacity and Quality. Available at https://p4qm.org/measures/4625e. Accessed: April 30, 2025.
Back to Citation325. eCQI Resource Center. Excessive Radiation Dose or Inadequate Image Quality for Diagnostic Computed Tomography in Adults eCQM—Measure Clarification. Available at https://ecqi.healthit.gov/excessive-radiation-dose-or-inadequate-image-quality-diagnostic-computed-tomography-adults-ecqm-measure-clarification. Accessed June 5, 2025.
Back to Citation326. In that notice, we also clarified that while CMS is not requiring vendors to demonstrate their software's capabilities to CMS, hospitals and clinicians that choose to do so may request information from a vendor about a specific software's ability to generate and transform the radiology data into the necessary format.
Back to Citation327. Smith-Bindman R, Yu S, Wang Y, et al. (2022). An Image Quality-informed Framework for CT Characterization. Radiology, 302(2), 380-389. Available at https://doi.org/10.1148/radiol.2021210591. Accessed: November 13, 2025.
Back to Citation328. Smith-Bindman R, Yu S, Wang Y, et al. (2022). An Image Quality-informed Framework for CT Characterization. Radiology, 302(2), 380-389. Available at https://doi.org/10.1148/radiol.2021210591. Accessed: November 13, 2025.
Back to Citation329. American College of Radiology (2015). Development and Revision Handbook. Available at https://www.acr.org/-/media/ACR/Files/Practice-Parameters/DevelopmentHandbook.pdf. Accessed: November 13, 2025.
Back to Citation330. Stecker MS, Balter S, Towbin RB, et al. (2009). Guidelines for Patient Radiation Dose Management. Journal of Vascular and Interventional Radiology. 20(7): S263-S273. Available at https://doi.org/10.1016/j.jvir.2009.04.037. Accessed: November 13, 2025.
Back to Citation331. Halliburton SS, Abbara S, Chen MY, et al. (2011). Society of Cardiovascular Computed Tomography. SCCT guidelines on radiation dose and dose-optimization strategies in cardiovascular CT. J Cardiovasc Comput Tomogr. 5(4): 198-224. Available at https://doi.org/10.1016/j.jcct.2011.06.001. Accessed: November 13, 2025.
Back to Citation332. Hirshfeld JW, Ferrari VA, Bengel FM, et al. (2018). 2018 ACC/HRS/NASCI/SCAI/SCCT Expert Consensus Document on Optimal Use of Ionizing Radiation in Cardiovascular Imaging: Best Practices for Safety and Effectiveness. Catheter Cardiovasc Interv. 92: E35-E97. Available at https://doi.org/10.1002/ccd.27659. Accessed: November 13, 2025.
Back to Citation333. Image Wisely 2020. Available at https://www.imagewisely.org/. Accessed: November 13, 2025.
Back to Citation334. FDA (2019). Computed Tomography (CT). Available at https://www.fda.gov/radiation-emitting-products/medical-x-ray-imaging/computed-tomography-ct#6. Accessed: November 13, 2025.
Back to Citation335. We note that at the time of the measure's endorsement, NQF served as the CBE responsible for measure endorsement activities. The CBE role has since transitioned to Battelle, which now administers the PQM and oversees both the pre-rulemaking and measure endorsement processes.
Back to Citation336. Smith-Bindman, R., Chu, P. W., Azman Firdaus, H., Stewart, C., Malekhedayat, M., Alber, S., Bolch, W. E., Mahendra, M., Berrington de González, A., & Miglioretti, D. L. (2025b). Projected Lifetime Cancer Risks From Current Computed Tomography Imaging. JAMA Internal Medicine, 185 (6). Available at https://doi.org/10.1001/jamainternmed.2025.0505.Accessed: November 13, 2025.
Back to Citation337. Greffier J, Hamard A, Pereira F, et al. (2020). Image quality and dose reduction opportunity of deep learning image reconstruction algorithm for CT: a phantom study. Eur Radiol, 30(7), 3951-3959. Available at https://doi.org/10.1007/s00330-020-06724-w.Accessed: November 13, 2025.
338. Gottumukkala RV, Kalra MK, Tabari A, Otrakji A, Gee MS (2019). Advanced CT Techniques for Decreasing Radiation Dose, Reducing Sedation Requirements, and Optimizing Image Quality in Children. Radiographics, 39(3), 709-726. Available at https://doi.org/10.1148/rg.2019180082.Accessed: November 13, 2025.
339. Den Harder AM, Willemink MJ, van Doormaal PJ, et al. (2018). Radiation dose reduction for CT assessment of urolithiasis using iterative reconstruction: A prospective intra-individual study. Eur Radiol, 28(1), 143-150. Available at https://doi.org/10.1007/s00330-017-4929-2. Accessed: November 13, 2025.
340. Rob S, Bryant T, Wilson I, Somani BK (2017). Ultra-low-dose, low-dose, and standard-dose CT of the kidney, ureters, and bladder: is there a difference? Results from a systematic review of the literature. Clin Radiol, 72(1), 11-15. Available at https://doi.org/10.1016/j.crad.2016.10.005. Accessed: November 13, 2025.
341. Konda SR, Goch AM, Leucht P, et al. (2016). The use of ultra-low-dose CT scans for the evaluation of limb fractures: is the reduced effective dose using CT in orthopaedic injury (REDUCTION) protocol effective? Bone Joint J, 98-B(12), 1668-1673. Available at https://doi.org/10.1302/0301-620X.98B12.BJJ-2016-0336.R1. Accessed: November 13, 2025.
Back to Citation342. Smith-Bindman, R., Alber, S. A., Kwan, M. L., Pequeno, P., Bolch, W. E., Bowles, E. J. A., Greenlee, R. T., Stout, N. K., Weinmann, S., Moy, L. M., Stewart, C., Francisco, M., Kofler, C., Duncan, J. R., Ducore, J., Mahendra, M., Pole, J. D., & Miglioretti, D. L. (2025). Medical Imaging and Pediatric and Adolescent Hematologic Cancer Risk. New England Journal of Medicine. Available at https://doi.org/10.1056/nejmoa2502098.Accessed: November 13, 2025.
Back to Citation343. For additional information on REHs, we refer readers to a CMS Fact Sheet on REHs (Sept. 2024). Available at https://www.cms.gov/files/document/rural-emergency-hospitals-factsheet-september-2024.pdf.
Back to Citation344. Moore, C. & Heckmann R. (2025). Hospital Boarding In The ED: Federal, State, And Other Approaches. Health Affairs Forefront. Available at https://www.healthaffairs.org/content/forefront/hospital-boarding-ed-federal-state-and-other-approaches. Accessed: April 30, 2025.
Back to Citation345. Moore, C. & Heckmann R. (2025). Hospital Boarding In The ED: Federal, State, And Other Approaches. Health Affairs Forefront. Available at https://www.healthaffairs.org/content/forefront/hospital-boarding-ed-federal-state-and-other-approaches. Accessed: April 30, 2025.
Back to Citation346. Agency for Healthcare Research and Quality. (2025). Technical Report: AHRQ Summit To Address Emergency Department Boarding. Available at https://www.ahrq.gov/sites/default/files/wysiwyg/topics/ed-boarding-summit-report.pdf. Accessed: April 30, 2025.
Back to Citation347. Gaieski, D.F., Agarwal, A.K., Mikkelsen, M.E., Drumheller, B., Cham Sante, S., Shofer, F.S., Goyal, M., & Pines, J.M. (2017). The Impact of ED Crowding on Early Interventions and Mortality in Patients with Severe Sepsis. The American Journal of Emergency Medicine, 35 (7), 953-960. Available at https://doi.org/10.1016/j.ajem.2017.01.061. Accessed: April 30, 2025.
348. Laam L.A., Wary A.A., Strony R.S., Fitzpatrick M.H., & Kraus C.K. (2021). Quantifying the Impact of Patient Boarding on Emergency Department Length of Stay: All Admitted Patients are Negatively Affected by Boarding. Journal of American College Emergency Physicians, 2 (2), e12401. Available at https://doi.org/10.1002/emp2.12401. Accessed: April 30, 2025.
Back to Citation349. Janke, A.T., Melnick, E.R., & Venkatesh, A.K. (2022). Monthly Rates of Patients Who Left Before Accessing Care in US Emergency Departments, 2017-2021. Journal of the American Medical Association, 5 (9), e2233708. Available at https://doi.org/10.1001/jamanetworkopen.2022.33708. Accessed: April 30, 2025.
Back to Citation350. Gaieski, D.F., Agarwal, A.K., Mikkelsen, M.E., Drumheller, B., Cham Sante, S., Shofer, F.S., Goyal, M., & Pines, J.M. (2017). The Impact of ED Crowding on Early Interventions and Mortality in Patients with Severe Sepsis. The American Journal of Emergency Medicine, 35 (7), 953-960. Available at https://doi.org/10.1016/j.ajem.2017.01.061. Accessed: April 30, 2025.
Back to Citation351. Laam L.A., Wary A.A., Strony R.S., Fitzpatrick M.H., & Kraus C.K. (2021). Quantifying the Impact of Patient Boarding on Emergency Department Length of Stay: All Admitted Patients are Negatively Affected by Boarding. Journal of American College Emergency Physicians, 2 (2), e12401. Available at https://doi.org/10.1002/emp2.12401. Accessed: April 30, 2025.
Back to Citation352. Mohr, N.M., Wu, C., Ward, M.J., McNaughton, C.D., Faine, B., Pomeranz, K., Richardson, K., & Kaboli, P.J. (2022). Transfer Boarding Delays Care More in Low-volume Rural Emergency Departments: A Cohort Study.The Journal of Rural, 38 (1), 282-292. Available at https://doi.org/10.1111/jrh.12559. Accessed: April 30, 2025.
353. Usher, M., Sahni, N., Herrigel, D., Simon, G., Melton, G.B., Joseph, A., & Olson, A. (2018). Diagnostic Discordance, Health Information Exchange, and Inter-Hospital Transfer Outcomes: A Population Study. Journal of General Internal Medicine, 33 (9), 1447-53. Available at https://doi.org/10.1007/s11606-018-4491-x. Accessed: April 30, 2025.
Back to Citation354. McNaughton, C.D., Bonnet, K., Schlundt, D., Mohr, N.M., Chung, S., Kaboli, P.J., & Ward, M.J. (2020). Rural Interfacility Emergency Department Transfers: Framework and Qualitative Analysis. The Western Journal of Emergency Medicine, 21 (4), 858-865. Available at https://doi.org/10.5811/westjem.2020.3.46059. Accessed: April 30, 2025.
Back to Citation355. The Emergency Care Access & Timeliness eCQM was previously named the Emergency Care Capacity and Quality (ECCQ) eCQM. The name of the measure has been updated to better reflect the purpose of the measure based on feedback from the Pre-Rulemaking Measure Review (PRMR) Hospital Recommendation Group Meeting on January 16, 2025. Available at https://p4qm.org/sites/default/files/2025-02/PRMR-Hospital-Recommendation-Group-Meeting-Summary.pdf.
Back to Citation356. The Median Time for Discharged ED Patients was adopted in the REHQR Program in the CY 2024 OPPS/ASC final rule with comment period (88 FR 49832).
Back to Citation357. Partnership for Quality Measurement. (n.d.). Emergency Care Capacity and Quality. Available at https://p4qm.org/measures/4625e. Accessed: April 30, 2025.
Back to Citation358. For proposed measure specifications, we refer readers to the CMS QualityNet REHQR Program website at https://qualitynet.cms.gov/reh/rehqr.
Back to Citation359. This measure component is calculated at the encounter level by subtracting “Decision to Transfer” order time from “ED Departure Time” for visits with the ED disposition of “Transferred” (to an acute care hospital) and then flagging as a numerator event if >240 minutes.
Back to Citation360. ED observations stays are defined as an observation encounter where the patient remains physically in an area under control of the ED and under the care of an ED clinician inclusive of observation in a hospital bed. Partnership for Quality Measurement. Emergency Care Capacity and Quality. Available at https://p4qm.org/measures/4625e. Accessed: April 30, 2025.
Back to Citation361. Partnership for Quality Measurement. (n.d.). Emergency Care Capacity and Quality. Available at https://p4qm.org/measures/4625e. Accessed: April 30, 2025.
Back to Citation362. Partnership for Quality Measurement. (n.d.). Emergency Care Capacity and Quality. Available at https://p4qm.org/measures/4625e. Accessed: April 30, 2025.
Back to Citation363. Partnership for Quality Measurement. (n.d.). Emergency Care Capacity and Quality. Available at https://p4qm.org/measures/4625e. Accessed: April 30, 2025.
Back to Citation364. Yale New Haven Health Services Corporation. (2024). Technical Expert Panel (TEP) Evaluation of Measure Emergency Care Capacity and Quality Electronic Clinical Quality Measure (eCQM). Available at https://mmshub.cms.gov/sites/default/files/ECCQ-TEP-Summary-Report-081624.pdf. Accessed: April 30, 2025.
Back to Citation365. Partnership for Quality Measurement. (n.d.). Emergency Care Capacity and Quality. Available at https://p4qm.org/measures/4625e. Accessed: April 30, 2025.
Back to Citation366. Partnership for Quality Measurement. (n.d.). Emergency Care Capacity and Quality. Available at https://p4qm.org/measures/4625e. Accessed: April 30, 2025.
Back to Citation367. Partnership for Quality Measurement. (n.d.). Section 7.1 Supplemental Attachment. Available at https://p4qm.org/sites/default/files/2024-10/4625e- section-7.1-supplemental-attachment.pdf. Accessed: April 30, 2025.
Back to Citation368. Because REHs are typically a low volume setting, volume standardization is not applied to calculate the Emergency Care Access & Timeliness eCQM, unlike the version of the Emergency Care Access & Timeliness eCQM finalized for adoption into the Hospital OQR Program in section XV.B.1. of this final rule with comment period.
Back to Citation369. The principal diagnosis (first listed diagnosis at ED discharge) will be used to define strata inclusion. For this measure's purpose, mental health diagnoses do not include substance use disorder diagnoses. Mental health refers to mental health diagnoses, life stressors and crises, and stress-related physical symptoms.
Back to Citation370. Partnership for Quality Measurement. (2025). 2024-2025 Pre-Rulemaking Measure Review (PRMR) Recommendation Group Final Meeting Summary: Hospital Committee. Available at https://p4qm.org/sites/default/files/2025-02/PRMR-Hospital-Recommendation-Group-Meeting-Summary.pdf. Accessed: April 30, 2025.
Back to Citation371. Partnership for Quality Measurement. (2025). 2024-2025 Pre-Rulemaking Measure Review (PRMR) Recommendation Group Final Meeting Summary: Hospital Committee. Available at https://p4qm.org/sites/default/files/2025-02/PRMR-Hospital-Recommendation-Group-Meeting-Summary.pdf. Accessed: April 30, 2025.
Back to Citation372. Partnership for Quality Measurement. (2025). 2024-2025 Pre-Rulemaking Measure Review (PRMR) Recommendation Group Final Meeting Summary: Hospital Committee. Available at https://p4qm.org/sites/default/files/2025-02/PRMR-Hospital-Recommendation-Group-Meeting-Summary.pdf. Accessed: April 30, 2025.
Back to Citation373. Dyas, S.R., Greenfield, E., Messimer, S., Thotakura, S., Gholston, S., Doughty, T., Hays, M., Ivey, R., Spalding, J., & Phillips, R. (2015). Process-Improvement Cost Model for the Emergency Department. Journal of Healthcare Management, 60 (6), 442-57. Available at https://doi.org/10.1097/00115514-201511000-00011. Accessed: April 30, 2025.
Back to Citation374. Partnership for Quality Measurement. (2024). 2024 Pre-Rulemaking Measure Review Preliminary Assessment. Available at https://p4qm.org/sites/default/files/2024-12/PRMR-PA-MUC2024-075.pdf. Accessed: April 30, 2025.
375. Partnership for Quality Measurement. (2025). Fall 2024 Cycle Endorsement and Maintenance (E&M) Technical Report. Available at https://p4qm.org/sites/default/files/Initial%20Recognition%20and%20Management/material/EM-Fall-2024-Initial-Recognition-Final-Project-Report.pdf. Accessed: April 30, 2025.
Back to Citation376. Partnership for Quality Measurement. (2025). 2024-2025 Pre-Rulemaking Measure Review (PRMR) Recommendation Group Final Meeting Summary: Hospital Committee. Available at https://p4qm.org/sites/default/files/2025-02/PRMR-Hospital-Recommendation-Group-Meeting-Summary.pdf. Accessed: April 30, 2025.
Back to Citation377. Partnership for Quality Measurement. (2025). 2024-2025 Pre-Rulemaking Measure Review (PRMR) Recommendation Group Final Meeting Summary: Hospital Committee. Available at https://p4qm.org/sites/default/files/2025-02/PRMR-Hospital-Recommendation-Group-Meeting-Summary.pdf. Accessed: April 30, 2025.
Back to Citation378. Partnership for Quality Measurement (n.d.). Emergency Care Capacity and Quality. Available at https://p4qm.org/measures/4625e. Accessed: April 30, 2025.
379. Yale New Haven Health Services Corporation. (April 2024). Technical Expert Panel (TEP) Evaluation of Measure Equity of Emergency Care Capacity and Quality Electronic Clinical Quality Measure (eCQM). Available at https://mmshub.cms.gov/sites/default/files/ECCQ-TEP-2-Summary-Report.pdf. Accessed: April 30, 2025.
380. Yale New Haven Health Services Corporation. (September 2024). Technical Expert Panel (TEP) Evaluation of Measure Emergency Care Capacity and Quality Electronic Clinical Quality Measure (eCQM). Available at https://mmshub.cms.gov/sites/default/files/ECCQ-TEP-Summary-Report-081624.pdf. Accessed: April 30, 2025.
Back to Citation381. Partnership for Quality Measurement. (2025). Fall 2024 Cycle Endorsement and Maintenance (E&M) Technical Report. Available at https://p4qm.org/sites/default/files/Initial%20Recognition%20and%20Management/material/EM-Fall-2024-Initial-Recognition-Final-Project-Report.pdf. Accessed: April 30, 2025.
Back to Citation382. Centers for Medicare & Medicaid Services. (2023). Electronic Clinical Quality Measures (eCQMs) Specification, Testing, Standards, Tools, and Community. Available at https://mmshub.cms.gov/sites/default/files/eCQM-Specifications-Testing-Standards-Tools-Community.pdf. Accessed: April 30, 2025.
Back to Citation383. The Median Time for Discharged ED Patients was adopted in the REHQR Program in the CY 2024 OPPS/ASC final rule with comment period (88 FR 49832).
Back to Citation384. Partnership for Quality Measurement (n.d.). Emergency Care Capacity and Quality. Available at https://p4qm.org/measures/4625e. Accessed: April 30, 2025.
Back to Citation385. Partnership for Quality Measurement. Median Time from ED Arrival to ED Departure for Discharged ED Patients. Available at https://p4qm.org/measures/0496. Accessed: April 30, 2025.
Back to Citation386. Centers for Medicare & Medicaid Services. (December 2024). HCAHPS Fact Sheet. Available at https://hcahpsonline.org/globalassets/hcahps/facts/hcahps_fact_sheet_december_2024.pdf.
Back to Citation387. Partnership for Quality Measurement. Section 7.1 Supplemental Attachment. Available at https://p4qm.org/sites/default/files/2024-10/4625e-section-7.1-supplemental-attachment.pdf.
Back to Citation388. QRDA I is an individual patient-level quality report that contains quality data for one patient for one or more eCQMs. QRDA creates a standard method to report quality measure results in a structured, consistent format and can be used to exchange eCQM data between systems. For further detail on QRDA I, the most recently available QRDA I specifications and Implementation Guides (IGs) can be found at https://ecqi.healthit.gov/qrda/versions.
Back to Citation389. The Hospital Quality Reporting (HQR) System (formerly referred to as the QualityNet Secure Portal) is the only CMS-approved website for secure communications and healthcare quality data exchange to and within various CMS quality reporting programs. For more information regarding the HQR System, we refer readers to the CMS eCQI Resource Center available at https://ecqi.healthit.gov/tool/hospital-quality-reporting-hqr-system.
Back to Citation390. For additional information on the Median Time for Discharged ED Patients measure, we refer readers to the specifications manuals for the REHQR Program, available at https://qualitynet.cms.gov/reh/specifications-manuals.
Back to Citation391. DelSole, E.M., Makanji, H.S., & Kurd, M.F. (2019). Current trends in ambulatory spine surgery: a systematic review. J Spine Surg. 5(Suppl 2):S124-S132. Available at https://doi.org/10.21037/jss.2019.04.12. Accessed: April 29, 2025.
392. Kondamuri, N.S., Miller, A.L., Rathi, V.K., et al. (2020). Trends in Ambulatory Surgery Center Utilization for Otolaryngologic Procedures among Medicare Beneficiaries, 2010-2017. Otolaryngol Head Neck Surg. 162(6):873-880. Available at https://doi.org/10.1177/0194599820914298. Accessed: April 29, 2025.
393. Shariq, O.A., Bews, K.A., Etzioni, D.A., et al. (2023). Performance of General Surgical Procedures in Outpatient Settings Before and After Onset of the COVID-19 Pandemic. JAMA Netw Open.; 6(3):e231198. Available at https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2801919. Accessed: April 29, 2025.
Back to Citation394. DeSai, C., Janowiak, K., Secheli, B., et al. (2021). Empowering patients: simplifying discharge instructions. BMJ Open Quality; 10(3)001419. Available at http://doi.org/10.1136/bmjoq-2021-001419. Accessed: April 8, 2025.
395. Malevanchik, L., Wheeler, M., Gagliardi, K., Karliner L., & Shah, S.J. (2021). Disparities After Discharge: The Association of Limited English Proficiency and Postdischarge Patient-Reported Issues. Available at The Joint Commission Journal on Quality and Patient Safety, 47(12):775-782. https://doi.org/10.1016/j.jcjq.2021.08.013. Accessed: April 8, 2025.
Back to Citation396. Choudhry, A.J., Younis, M., Ray-Zack, M.D., et al. (2019). Enhanced readability of discharge summaries decreases provider telephone calls and patient readmissions in the posthospital setting. Surgery. 165(4):789-794. Available at https://doi.org/10.1016/j.surg.2018.10.014. Accessed: April 8, 2025.
397. Becker, C., Zumbrunn, S., Beck, K., et al. (2021). Interventions to Improve Communication at Hospital Discharge and Rates of Readmission: A Systematic Review and Meta-analysis. JAMA Netw Open. 4(8):e2119346. Available at https://doi.org/10.1001/jamanetworkopen.2021.19346. Accessed: April 8, 2025.
Back to Citation398. Downey, E., & Olds, D.M. (2021). Comparison of Documentation on Inpatient Discharge and Ambulatory End-of-Visit Summaries. Journal of Healthcare Quality. 43(3):e43-e52. Available at https://doi.org/10.1097/JHQ.0000000000000269. Accessed: April 8, 2025.
Back to Citation399. A copy of the survey instrument is available at https://www.cms.gov/files/document/patient-understanding-key-information-related-recovery-after-facility-based-outpatient-procedure-or.pdf.
Back to Citation400. Murdoch, M., et al. (2014). Impact of different privacy conditions and incentives on survey response rate, participant representativeness, and disclosure of sensitive information: a randomized controlled trial. BMC Medical Research Methodology. July 16:14-90. Available at https://bmcmedresmethodol.biomedcentral.com/articles/10.1186/1471-2288-14-90. Accessed: October 29, 2025.
Back to Citation401. Centers for Medicare & Medicaid Services. (April 2024). Patient Understanding of Key Information Related to Recovery After a Facility-Based Outpatient Procedure or Surgery, Patient Reported Outcome-Based Performance Measure. Available at https://www.cms.gov/files/document/patient-understanding-key-information-related-recovery-after-facility-based-outpatient-procedure-or.pdf. Accessed: April 29, 2025.
Back to Citation402. For more information on what the Cronbach alpha score determines and how it is used, we refer readers to: Tavakol, M., & Dennick, R. (2011). Making sense of Cronbach's alpha. Int J Med Educ. 27;2: 53- 55. https://www.ijme.net/archive/2/cronbachs-alpha.pdf. Accessed: April 30, 2025.
Back to Citation403. Centers for Medicare & Medicaid Services. (April 2024). Patient Understanding of Key Information Related to Recovery After a Facility-Based Outpatient Procedure or Surgery, Patient Reported Outcome-Based Performance Measure. Available at https://www.cms.gov/files/document/patient-understanding-key-information-related-recovery-after-facility-based-outpatient-procedure-or.pdf. Accessed: April 29, 2025.
Back to Citation404. Centers for Medicare & Medicaid Services. (April 2024). Patient Understanding of Key Information Related to Recovery After a Facility-Based Outpatient Procedure or Surgery, Patient Reported Outcome-Based Performance Measure. Available at https://www.cms.gov/files/document/patient-understanding-key-information-related-recovery-after-facility-based-outpatient-procedure-or.pdf. Accessed: April 29, 2025.
Back to Citation405. Partnership for Quality Measurement. Submission Tool and Repository Measure Database. Available at https://p4qm.org/measures/4210. Accessed: April 8, 2025.
Back to Citation406. The proposed ASCQR Program measure specifications can be found at https://qualitynet.cms.gov/asc.
Back to Citation407. The Information Transfer PRO-PM is identified on the MUC List as MUC2024-073.
408. Partnership for Quality Measurement. (2025). 2024-2025 Pre-Rulemaking Measure Review (PRMR) Recommendation Group Final Meeting Summary: Hospital Committee. Available at https://p4qm.org/sites/default/files/2025-02/PRMR-Hospital-Recommendation-Group-Meeting-Summary.pdf. Accessed: April 29, 2025.
Back to Citation409. Centers for Medicare & Medicaid Services. (April 2024). Patient Understanding of Key Information Related to Recovery After a Facility-Based Outpatient Procedure or Surgery, Patient Reported Outcome-Based Performance Measure. Available at https://www.cms.gov/files/document/patient-understanding-key-information-related-recovery-after-facility-based-outpatient-procedure-or.pdf. Accessed: April 29, 2025.
Back to Citation410. OAS CAHPS. (2024). 2024 Introduction to the OAS CAHPS Survey, Self-Paced Training. Available at https://oascahps.org/Training/Training-Materials. Accessed: April 29, 2025.
Back to Citation411. Centers for Medicare & Medicaid Services. (March 22). Methodology Report For Public Comment: Patient Understanding of Key Information Related to Recovery From an Outpatient Surgery or Procedure. Available at https://www.cms.gov/files/document/methodology-report-public-comment.pdf. Accessed: April 29, 2025.
Back to Citation412. Partnership for Quality Measurement. (2024). Fall 2023 Management of Acute and Chronic Events Meeting Summary. Available at https://p4qm.org/sites/default/files/Management%20of%20Acute%20Events%2C%20Chronic%20Disease%2C%20Surgery%2C%20and%20Behavioral%20Health/material/EM-Acute-Chronic-Events-Fall2023-Endorsement-Meeting-Summary.pdf. Accessed: April 8, 2025.
Back to Citation413. A copy of the survey instrument is available at https://www.cms.gov/files/document/patient- understanding-key-information-related-recovery-after-facility-based-outpatient-procedure-or.pdf.
Back to Citation414. DeSai, C., Janowiak, K., Secheli, B., et al. (2021). Empowering patients: simplifying discharge instructions. BMJ Open Quality; 10(3)001419. Available at http://doi.org/10.1136/bmjoq-2021-001419. Accessed: April 8, 2025.
415. Malevanchik, L., Wheeler, M., Gagliardi, K., Karliner L., & Shah, S.J. (2021). Disparities After Discharge: The Association of Limited English Proficiency and Postdischarge Patient-Reported Issues, The Joint Commission Journal on Quality and Patient Safety, 47(12):775-782. Available at https://doi.org/10.1016/j.jcjq.2021.08.013. Accessed: April 8, 2025.
Back to Citation416. Assistant Secretary for Planning and Evaluation. (2017). Final Report Volume I: Background Paper, Declining Response Rates in Federal Surveys: Trends and Implications. Available at https://aspe.hhs.gov/reports/final-report-volume-i-background-paper-declining-response-rates-federal-surveys-trends-implications#:~:text=Over%20the%20last%20decade%2C%20survey,of%20Data%2C%20Surveys%2C%20&%20Indicators. Accessed: October 2, 2025.
417. de Koning R, Egiz A, Kotecha J, et al. (2021). Survey Fatigue During the COVID-19 Pandemic: An Analysis of Neurosurgery Survey Response Rates. Frontiers in Surgery. Available at https://doi.org/10.3389/fsurg.2021.690680. Accessed: October 29, 2025. Accessed: October 29, 2025.
Back to Citation418. Brown RF, St John A, Hu, Y, Sandhu G (2024). Differential Electronic Survey Response: Does Survey Fatigue affect Everyone Equally? Journal of Surgical Research. Available at https://journalofsurgicalresearch.com/retrieve/pii/S0022480423005127. Accessed: October 29, 2025.
Back to Citation419. Porter SR, Whitcomb ME, Weitzer WH. (2004). Multiple Surveys of Students and Survey Fatigue. Chapter 5, Overcoming Survey Research Problems. Available at https://oia.unm.edu/surveys/survey-fatigue.pdf. Accessed: October 29, 2025.
Back to Citation420. What Are Patient-Reported Measures? Content last reviewed February 2025. Agency for Healthcare Research and Quality, Rockville, MD. Available at https://www.ahrq.gov/cahps/about-cahps/patient-experience/prems-proms/index.html. Accessed: October 29, 2025.
Back to Citation421. OAS CAHPS. Frequently Asked Questions for Hospitals and ASCs OAS CAHPS®. OAS CAHPS. Available at https://oascahps.org/OAS_Facility_FAQs.docx. Accessed: October 14, 2025.
Back to Citation422. Centers for Medicare & Medicaid Services. QualityNet. Available at https://qualitynet.cms.gov/files/6830945005ca4cea73650e74?filename=OQR_v19.0_SpecsManual_2026.pdf. Accessed: October 6, 2025.
Back to Citation423. Partnership for Quality Measurement. Submission Tool and Repository Measure Database. Available at https://p4qm.org/measures/4210. Accessed: April 8, 2025.
Back to Citation424. Centers for Medicare & Medicaid Services. (April 2024). Patient Understanding of Key Information Related to Recovery After a Facility-Based Outpatient Procedure or Surgery, Patient Reported Outcome-Based Performance Measure (PRO-PM). Available at https://qualitynet.cms.gov/outpatient/measures/PRO-PM/methodology. Accessed: November 12, 2025.
Back to Citation425. Centers for Medicare & Medicaid Services. (April 2024). Patient Understanding of Key Information Related to Recovery After a Facility-Based Outpatient Procedure or Surgery, Patient Reported Outcome-Based Performance Measure. Available at https://www.cms.gov/files/document/patient-understanding-key-information-related-recovery-after-facility-based-outpatient-procedure-or.pdf. Accessed: April 29, 2025.
Back to Citation426. For more information on what the Cronbach alpha score determines and how it is used, we refer readers to: Tavakol, M., & Dennick, R. (2011). Making sense of Cronbach's alpha. Int J Med Educ. 27;2: 53-55. Available at https://www.ijme.net/archive/2/cronbachs-alpha.pdf. Accessed: April 30, 2025.
Back to Citation427. Centers for Medicare & Medicaid Services. (April 2024). Patient Understanding of Key Information Related to Recovery After a Facility-Based Outpatient Procedure or Surgery, Patient Reported Outcome-Based Performance Measure. Available at https://www.cms.gov/files/document/patient-understanding-key-information-related-recovery-after-facility-based-outpatient-procedure-or.pdf. Accessed: April 29, 2025.
Back to Citation428. Centers for Medicare & Medicaid Services. (April 2024). Patient Understanding of Key Information Related to Recovery After a Facility-Based Outpatient Procedure or Surgery, Patient Reported Outcome-Based Performance Measure. Available at https://www.cms.gov/files/document/patient-understanding-key-information-related-recovery-after-facility-based-outpatient-procedure-or.pdf. Accessed: April 29, 2025.
Back to Citation429. CMS. Measure Management Lifecycle Overview. Available at https://mmshub.cms.gov/blueprint-measure-lifecycle-overview. Accessed: October 5, 2025.
Back to Citation430. Ahmad, H., & Halim, H. (2017). Determining Sample Size for Research Activities. Selangor Business Review, 2(1), 20-34. Available at https://sbr.journals.unisel.edu.my/index.php/sbr/article/view/12. Accessed: April 8, 2025.
431. Voorhis, C., & Morgan, B. (2007). Understanding Power and Rules of Thumb for Determining Sample Size. Tutorials in Quantitative Methods for Psychology. 3(2), 43-50. Available at www.doi.org/10.20982/tqmp.03.2.p043. Accessed: April 8, 2025.
Back to Citation432. Malevanchik L., Wheeler M., Gagliardi K., Karliner L., Shah S.J. (2021). Disparities After Discharge: The Association of Limited English Proficiency and Postdischarge Patient-Reported Issues, The Issues, The Joint Commission Journal on Quality and Patient Safety, 47(12):775-782. Available at https://doi.org/10.1016/j.jcjq.2021.08.013. Accessed: October 29, 2025.
Back to Citation433. https://www.cms.gov/files/document/cms-national-quality-strategy-handout.pdf.
Back to Citation434. https://www.medicare.gov/care-compare/resources/hospital/overall-star-rating.
Back to Citation436. Institute of Medicine (US) Committee on Quality of Health Care in America, Kohn, L.T., Corrigan, J.M., & Donaldson, M.S. (Eds.). (2000). To Err is Human: Building a Safer Health System. National Academies Press (US).
437. Quality of Health Care in America. (2001). Crossing the Quality Chasm: A New Health System for the 21st Century. National Academies Press (US).
Back to Citation438. Agency for Healthcare Research and Quality. (February 2021). National Healthcare Quality and Disparities Report chartbook on patient safety. Rockville, MD. Available at https://www.ahrq.gov/sites/default/files/wysiwyg/research/findings/nhqrdr/chartbooks/patientsafety/2019qdr-patientsafety-chartbook.pdf.
Back to Citation439. AHRQ. (2023). National Action Alliance To Advance Patient and Workforce Safety. https://www.ahrq.gov/cpi/about/otherwebsites/actionalliance.html.
441. Fleisher, L.A., Schreiber, M., Cardo, D., Srinivasan, A. (2022). Health Care Safety during the Pandemic and Beyond—Building a System That Ensures Resilience. The New England Journal of Medicine, 386(7): 609-611. DOI: 10.1056/NEJMp2118285.
Back to Citation442. Centers for Medicare & Medicaid Services. (n.d.). Complications & deaths. Provider Data Catalog. https://data.cms.gov/provider-data/topics/hospitals/complications-deaths#complications.
Back to Citation443. Exec. Order No 14,221 (2025). https://www.govinfo.gov/content/pkg/FR-2025-02-28/pdf/2025-03440.pdf.
Back to Citation444. PatientRightsAdvocate.org. (2024). Transparency Survey 2024. https://www.patientrightsadvocate.org/patient-rights-advocate-bipartisan-pollster-memo-2024.
Back to Citation445. Mouslim. https://doi.org/10.1377/forefront.20211103.716124.
446. Smith, C., Singleton, A., Lewis, D., & Allen, B. (2022, May 3). Hospital price transparency data: Case studies for how to use it. Milliman. https://us.milliman.com/en/insight/hospital-price-transparency-data-case-studies-for-how-to-use-it.
447. Minemyer, P. (2022, September 8). New playbook aims to help employers, plan sponsors negotiate hospital prices. Fierce Healthcare: Payer. https://www.fiercehealthcare.com/payers/new-playbook-aims-help-employers-plan-sponsors-negotiate-hospital-prices.
448. https://turquoise.health/patients.
Back to Citation449. Xiao, F. (2024, October 23). Examining the healthcare market response to transparency regulations: Healthcare prices have begun to converge. Here's a detailed look into our analysis. Turquoise Health. https://blog.turquoise.health/examining-rate-trends-by-annualized-rate-change-over-time/.
450. Yaraghi, N., & Pan, X. (2025, September 9). The hospital price transparency rule is working, but patients still need help using it. Brookings Institute. https://www.brookings.edu/articles/the-hospital-price-transparency-rule-is-working-but-patients-still-need-help-using-it.
Back to Citation451. CMS, (2024, June), Hospital-Price-Transparency Examples, Hospital Price Transparency, GitHub. https://github.com/CMSgov/hospital-price-transparency/tree/master/examples.
Back to Citation452. CMS, (2025, May), Resources, Hospital Price Transparency website at https://www.cms.gov/priorities/key-initiatives/hospital-price-transparency/resources.
Back to Citation453. Cooksey, R.W. (2020). Descriptive Statistics for Summarising Data. In: Illustrating Statistical Procedures: Finding Meaning in Quantitative Data. https://doi.org/10.1007/978-981-15-2537-7_5. https://doi.org/10.1007/978-981-15-2537-7_5.
Back to Citation454. We note that this scenario is slightly different than we had portrayed at 88 FR 81540, 82101 which, in retrospect, we realized was erroneous with respect to the deductible.
Back to Citation455. Washington State Office of the Insurance Commissioner. What medical providers need to know about health insurance. https://www.insurance.wa.gov/what-medical-providers-need-know-about-health-insurance.
456. Texas Department of Insurance. (Last Updated on March 25, 2025) Prompt Pay FAQ. https://www.tdi.texas.gov/hprovider/ppsb418faq.html.
457. Centers for Medicare & Medicaid Services (CMS). (2011) Transmittal 2140: Changes to the Time Limits for Filing Medicare Fee-For-Service Claims. https://www.cms.gov/regulations-and-guidance/guidance/transmittals/downloads/r2140cp.pdf.
458. 42 CFR 447.45(d)(1) https://www.ecfr.gov/current/title-42/part-447/section-447.45#p-447.45(d)(1).
Back to Citation459. Orszag, Peter and Rekhi, Rahul. Real-Time Adjudication for Health Insurance Claims. https://onepercentsteps.com/wp-content/uploads/brief-rta-210208-1700.pdf.
Back to Citation460. National Academy for State Health Policy Palliative Care in Medicaid Costing Out the Benefit: Actuarial Analysis of Medicaid Experience, December 17, 2022 https://nashp.org/palliative-care-in-medicaid-costing-out-the-benefit-actuarial-analysis-of-medicaid-experience/.
461. Xie, Q.Y., Schreier, G., Hoy, M., Liu, Y., Neubauer, S., Chang, D.C.W., Redmond, S.J., & Lovell, N.H. (2016). Analyzing health insurance claims on different timescales to predict days in hospital. Journal of Biomedical Informatics, 60, 187-196. https://www.sciencedirect.com/science/article/pii/S1532046416000034.
Back to Citation462. National Academy for State Health Policy. Palliative Care in Medicaid Costing Out the Benefit: Actuarial Analysis of Medicaid Experience, December 17, 2022 https://nashp.org/palliative-care-in-medicaid-costing-out-the-benefit-actuarial-analysis-of-medicaid-experience/.
463. Xie, Q.Y., Schreier, G., Hoy, M., Liu, Y., Neubauer, S., Chang, D.C.W., Redmond, S.J., & Lovell, N.H. (2016). Analyzing health insurance claims on different timescales to predict days in hospital. Journal of Biomedical Informatics, 60, 187-196. https://www.sciencedirect.com/science/article/pii/S1532046416000034.
464. Washington State Office of the Insurance Commissioner. What medical providers need to know about health insurance. https://www.insurance.wa.gov/what-medical-providers-need-know-about-health-insurance.
465. Texas Department of Insurance. (Last Updated on March 25, 2025) Prompt Pay FAQ. https://www.tdi.texas.gov/hprovider/ppsb418faq.html.
466. Centers for Medicare & Medicaid Services (CMS). (2011) Transmittal 2140: Changes to the Time Limits for Filing Medicare Fee-For-Service Claims. https://www.cms.gov/regulations-and-guidance/guidance/transmittals/downloads/r2140cp.pdf.
467. 42 CFR 447.45(d)(1). https://www.ecfr.gov/current/title-42/part-447/section-447.45#p-447.45(d)(1).
Back to Citation468. CMS Cell Size Suppression Policy, Current Version January 26, 2024: https://resdac.org/articles/cms-cell-size-suppression-policy.
Back to Citation469. We acknowledge that, in past guidance, we established the use of the “nine 9s” policy. See https://www.cms.gov/files/document/cms-hpt-webinar-10-21-2024.pdf. (“CMS recommends that the hospital encode 999999999 (nine 9s) in the data element value to indicate that there is not sufficient historic claims history to derive the estimated allowed amount, and then update the file when sufficient history is available.”). Later recognizing that was a suboptimal approach, however, in subsequent guidance dated May 22, 2025, we said that “[h]ospitals should discontinue encoding 999999999 (nine 9s) in the estimated allowed amount data element within the MRF and should instead encode an actual dollar amount”. https://www.cms.gov/files/document/updated-hpt-guidance-encoding-allowed-amounts.pdf.
Back to Citation470. Data is inclusive of compliance actions from January 1, 2021, through August 31, 2025.
Back to Citation472. https://www.cms.gov/regulations-and-guidance/administrative-simplification/nationalprovidentstand.
Back to Citation473. Ibid.
Back to Citation474. Guidance on NPI Enumeration; 45 CFR 162.412(b). https://www.cms.gov/files/document/guidance-national-provider-identifier-npi-enumeration-pdf.pdf.
Back to Citation476. CMS's NPPES registry is available online at the following website address: https://npiregistry.cms.hhs.gov/.
Back to Citation479. CMS Hospital Enrollments and Hospital Additional NPIs datasets https://data.cms.gov/provider-characteristics/hospitals-and-other-facilities/hospital-enrollments.
Back to Citation480. https://www.cms.gov/files/document/provider-enrollment-certification-roadmap.pdf.
481. https://www.cms.gov/Regulations-and-Guidance/Guidance/Manuals/Downloads/pim83c10.pdf.
Back to Citation482. CMS Hospital Enrollments and Hospital Additional NPIs datasets https://data.cms.gov/provider-characteristics/hospitals-and-other-facilities/hospital-enrollments.
Back to Citation483. Welch WP, Xu L, De Lew N, et al. Ownership of Hospitals: An Analysis of Newly-Released Federal Data & A Method for Assessing Common Owners: Data Point [internet]. Washington (DC): Office of the Assistant Secretary for Planning and Evaluation (ASPE); 2023 Aug. Available from: https://www.ncbi.nlm.nih.gov/books/NBK616123/.
Back to Citation485. CMS Hospital Enrollments and Hospital Additional NPIs datasets https://data.cms.gov/provider-characteristics/hospitals-and-other-facilities/hospital-enrollments.
Back to Citation486. https://data.cms.gov/resources/medicare-provider-and-supplier-taxonomy-crosswalk-methodology.
Back to Citation488. https://www.cms.gov/newsroom/fact-sheets/hospital-price-transparency-enforcement-updates.
Back to Citation489. The CY 2020 HPT final rule ( https://www.federalregister.gov/d/2019-24931/p-683), CY 2022 OPPS/ASC final rule ( https://www.federalregister.gov/d/2021-24011/p-4135), and CY 2024 OPPS/ASC final rule with comment period ( https://www.federalregister.gov/d/2023-24293/p-5090).
Back to Citation490. CMS (2025, June) Enforcement Actions. https://www.cms.gov/priorities/key-initiatives/hospital-price-transparency/enforcement-actions.
Back to Citation491. See, for example,42 CFR 488.1245(c)(2)(ii) (Hospice); 42 CFR 488.845(c)(2)(ii) (Home Health Agency); 42 CFR 488.436(b) (Long-Term Care (LTC) Facility).
Back to Citation492. More precisely as discussed later in this section, the weighted median MAO payer-specific negotiated charges where the MAO payer-specific negotiated charges are weighted by the number of inpatient discharges for each of those payers that occurred during the cost reporting period. We simply refer to the median for ease of discussion.
Back to Citation493. As discussed further in section XX.C.2. of this final rule with comment period, we proposed that if CMS did not finalize the proposal to amend 45 CFR 180.50(b)(2)(ii)(C), hospitals would use the “estimated allowed amount” as required under the hospital price transparency regulations for purposes of calculating the median payer-specific negotiated charge that is reported on the cost report.
Back to Citation494. Linde S., Egede L.E. Do Chargemaster Prices Matter?: An Examination of Acute Care Hospital Profitability. Med Care. 2022 Aug 1;60(8):623-630.
Back to Citation495. Berenson R.A., Sunshine J.H., Helms D., Lawton E. Why Medicare Advantage plans pay hospitals traditional Medicare prices. Health Aff (Millwood). 2015;34(8):1289-1295.
Back to Citation496. Baker L.C., Bundorf M.K., Devlin A.M., Kessler D.P. Medicare Advantage plans pay less than traditional Medicare pays. Health Aff. (Millwood). 2016;35(8):1444-1451.
Back to Citation497. Maeda J.L.K., Nelson L. How Do the Hospital Prices Paid by Medicare Advantage Plans and Commercial Plans Compare with Medicare Fee-for-Service Prices? The Journal of Health Care Organization, Provision, and Financing. 2018;55(1-8).
Back to Citation498. Meiselbach M.K., Wang Y., Xu Jianhui, Bai G., Anderson G.F. Hospital Prices for Commercial Plans Are Twice Those For Medicare Advantage Plans When Negotiated By The Same Insurer. Health Aff. 2023;42(8):1110-1118.
Back to Citation499. Randall S., Duffy E.L. Insurers Negotiate Lower Hospital Prices for HIX Than for Commercial Groups. The American Journal of Managed Care. 2022;28(9): e347-e350.
Back to Citation500. For example, see https://www.cms.gov/priorities/key-initiatives/hospital-price-transparency/enforcement-actions.
Back to Citation501. The middle number; found by ordering all data points and selecting the one in the middle (or if there are two middle numbers, taking the mean of those two numbers).
Back to Citation503. Ordering the payer-specific negotiated charges from Step 3 from lowest to highest as {$7,200, $7,300, $7,300, $7,300, $7,400, $7,400, $7,500} the median, or middle, charge in that list is the fourth charge of $7,300.
Back to Citation504. For example, if the list had been {$7,300, $7,300, $7,400, $7,500} the median would have been $7,350, the mean of $7,300 and $7,400 (the two middle values are the second and third charges of $7,300 and $7,400).
Back to Citation505. We noted in the CY 2026 OPPS/ASC proposed rule that our proposed definition here of “items and services” is the same as the definition at 45 CFR 180.20, but for the examples included there and omitting the reference to outpatient department visits, as here we would not require hospitals to calculate the median of their payer-specific negotiated charges for items and services provided in the hospital outpatient setting.
Back to Citation508. 90 FR 17529. https://www.federalregister.gov/documents/2025/04/28/2025-07376/reforming-accreditation-to-strengthen-higher-education.
Back to Citation510. ACGME, Policies and Procedures, February 2, 2025. https://www.acgme.org/globalassets/pdfs/ab_acgmepoliciesprocedures.pdf.
Back to Citation511. ACGME, Guide to the Common Program Requirements, March 2024, https://www.acgme.org/globalassets/pdfs/guide-to-the-common-program-requirements-residency.pdf?utm_source=chatgpt.com.
Back to Citation512. Students for Fair Admissions, Inc. v. President and Fellows of Harvard College, June 2023. https://www.supremecourt.gov/opinions/22pdf/20-1199_hgdj.pdf.
Back to Citation513. https://www.hhs.gov/press-room/hhs-education-nutrition-medical-training-reforms.html.
Back to Citation514. U.S. Bureau of Labor Statistics. (2025). Occupational Outlook Handbook, Medical Records Specialists. Available at: https://data.bls.gov/oes/#/industry/622100. Accessed: April 8, 2025.
Back to Citation515. Office of the Assistant Secretary for Planning and Evaluation. (2017). Valuing Time in U.S. Department of Health and Human Services Regulatory Impact Analyses: Conceptual Framework and Best Practices. Available at https://aspe.hhs.gov/reports/valuing-time-us-department-health-human-services-regulatory-impact-analyses-conceptual-framework. Accessed: June 24, 2025.
Back to Citation516. Bureau of Labor and Statistics. (2025). Usual Weekly Earnings of Wage and Salary Workers, Fourth Quarter 2025. Available at https://www.bls.gov/news.release/pdf/wkyeng.pdf. Accessed: March 3, 2025.
Back to Citation517. Guzman, G. & Kollatr, M. (2024). Income in the United States: 2023. Available at https://www2.census.gov/library/publications/2024/demo/p60-282.pdf. Accessed: June 24, 2025.
Back to Citation518. U.S. Bureau of Labor Statistics. (2025). Occupational Outlook Handbook, Medical Records Specialists. Available at https://data.bls.gov/oes/#/industry/622100. Accessed: June 24, 2025.
Back to Citation519. Office of the Assistant Secretary for Planning and Evaluation. (2017). Valuing Time in U.S. Department of Health and Human Services Regulatory Impact Analyses: Conceptual Framework and Best Practices. Available at https://aspe.hhs.gov/reports/valuing-time-us-department-health-human-services-regulatory-impact-analyses-conceptual-framework. Accessed: June 24, 2025.
Back to Citation520. Bureau of Labor and Statistics. (2025). Usual Weekly Earnings of Wage and Salary Workers, Fourth Quarter 2025. Available at https://www.bls.gov/news.release/pdf/wkyeng.pdf. Accessed: March 3, 2025.
Back to Citation521. Guzman, G. & Kollatr, M. (2024). Income in the United States: 2023. Available at https://www2.census.gov/library/publications/2024/demo/p60-282.pdf. Accessed: June 24, 2025.
Back to Citation522. U.S. Bureau of Labor Statistics. (2025). Occupational Outlook Handbook, Medical Records Specialists. Available at: https://data.bls.gov/oes/#/industry/622100. Accessed: April 8, 2025.
Back to Citation523. Office of the Assistant Secretary for Planning and Evaluation. (2017). Valuing Time in U.S. Department of Health and Human Services Regulatory Impact Analyses: Conceptual Framework and Best Practices. Available at https://aspe.hhs.gov/reports/valuing-time-us-department-health-human-services-regulatory-impact-analyses-conceptual-framework. Accessed: June 24, 2025.
Back to Citation524. Bureau of Labor and Statistics. (2025). Usual Weekly Earnings of Wage and Salary Workers, Fourth Quarter 2025. Available at https://www.bls.gov/news.release/pdf/wkyeng.pdf. Accessed: March 3, 2025.
Back to Citation525. Guzman, G. & Kollatr, M. (2024). Income in the United States: 2023. Available at https://www2.census.gov/library/publications/2024/demo/p60-282.pdf. Accessed: June 24, 2025.
Back to Citation526. For the definition of a SCOD, see section 1833(t)(14)(B) of the Act at https://www.ssa.gov/OP_Home/ssact/title18/1833.htm.
Back to Citation530. American Hospital Association. Fast Facts on U.S. Hospitals, 2019. Available at https://www.aha.org/statistics/fast-facts-us-hospitals.
Back to Citation531. Homeland Infrastructure Foundation-Level Data (HIFLD) hospital dataset accessed on May 3, 2023, located at https://hifld-geoplatform.hub.arcgis.com/maps/9e318142490c4884bf74932af437c6c2/about. Note: All HIFLD open datasets, including the hospital dataset, have been discontinued as of September 30, 2025, and are no longer publicly available online.
Back to Citation532. Homeland Infrastructure Foundation-Level Data (HIFLD) hospital dataset accessed on April 2, 2025, located at https://hifld-geoplatform.hub.arcgis.com/maps/9e318142490c4884bf74932af437c6c2/about. Note: All HIFLD open datasets, including the hospital dataset, have been discontinued as of September 30, 2025, and are no longer publicly available online.
Back to Citation533. U.S. Bureau of Labor Statistics, May 2024 National Occupational Employment and Wage Estimates United States, Occupational Employment and Wage Statistics. Accessed at https://www.bls.gov/oes/tables.htm.
Back to Citation534. Bureau of Labor Statistics, U.S. Department of Labor, Occupational Outlook Handbook, Medical Records Specialists, at https://www.bls.gov/ooh/healthcare/medical-records-and-health-information-technicians.htm.
Back to Citation535. HIFLD Open was discontinued on August 26, 2025. This information is now accessible via the Geospatial Information Infrastructure (GII).
Back to Citation536. Clarify Insights Center. (2023, August 15). How hospitals can use price transparency data to negotiate better contracts with payers. Retrieved from https://clarifyhealth.com/insights/blog/how-hospitals-can-use-price-transparency-data-to-negotiate-better-contracts-with-payers/.
537. Gomes, C. (2023, July 31). Why healthcare providers should harness price transparency data. Medlyze—Price Transparency Data and Analysis for the Healthcare Industry Insights. Retrieved from https://www.medlyze.com/blog/why-healthcare-providers-should-harness-the-power-of-price-transparency-data.
Back to Citation538. Xiao, F. (2024, October 25). Is price transparency helping? Here are three ways to tell: Let's explore our biggest indicators of change: competition, pricing, and power. Turquoise Health Blog. Retrieved from https://blog.turquoise.health/is-price-transparency-helping-heres-three-ways-to-tell/.
Back to Citation539. Healthcare Financial Management Association. (2023, August 28). Leverage healthcare price transparency data to promote financial sustainability. Retrieved from https://www.hfma.org/price-transparency/leverage-healthcare-price-transparency-data-to-promote-financial-sustainability/.
Back to Citation540. Xiao, F. (2024, October 25). Is price transparency helping? Here are three ways to tell. Turquoise Health. Retrieved from https://blog.turquoise.health/is-price-transparency-helping-heres-three-ways-to-tell/.
Back to CitationBILLING CODE 4120-01-P
BILLING CODE 4120-01-C
BILLING CODE 4120-0-P
BILLING CODE 4120-0-C
BILLING CODE 4120-01-P
[FR Doc. 2025-20907 Filed 11-21-25; 4:15 pm]
BILLING CODE 4120-01-P




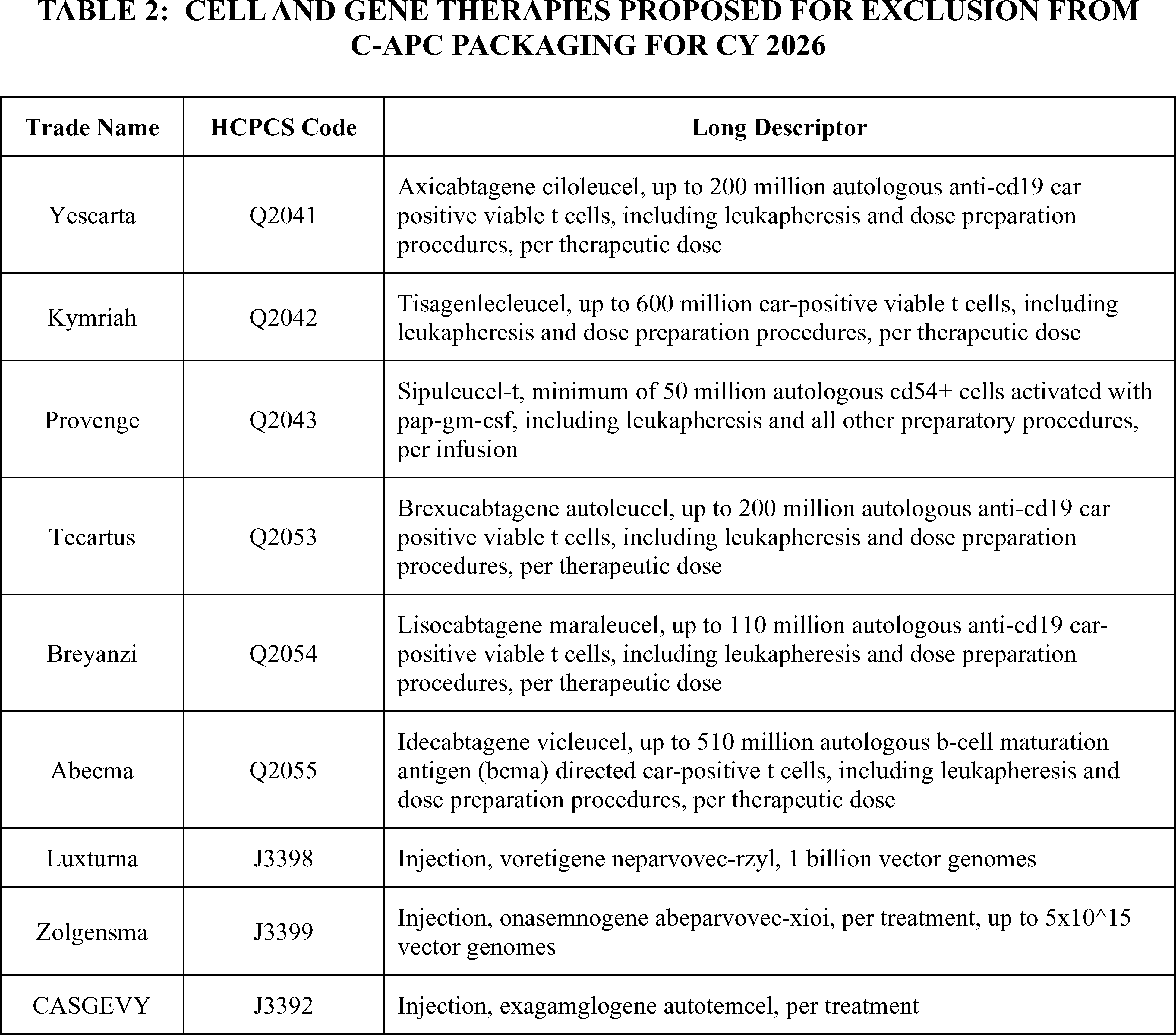

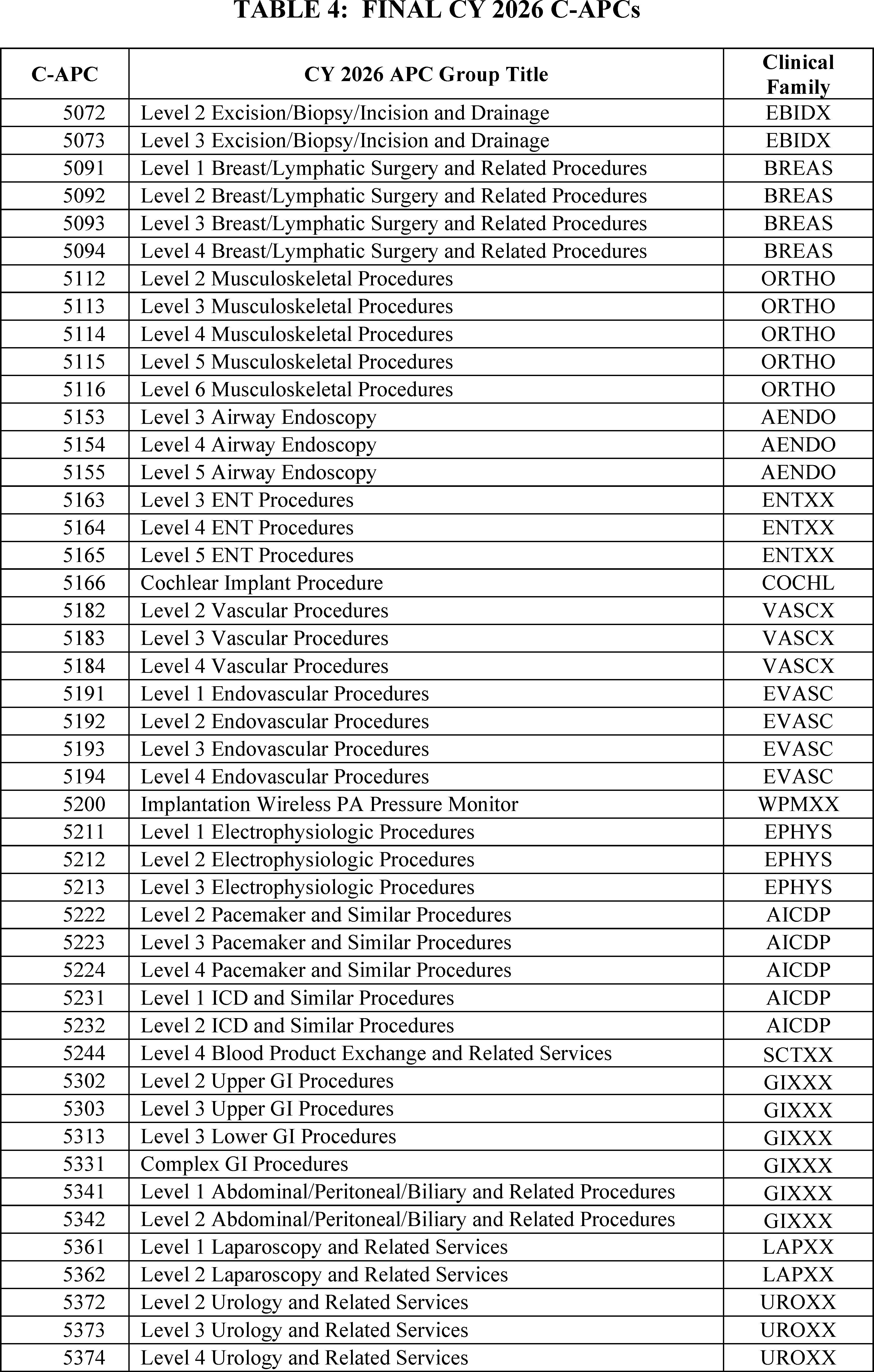
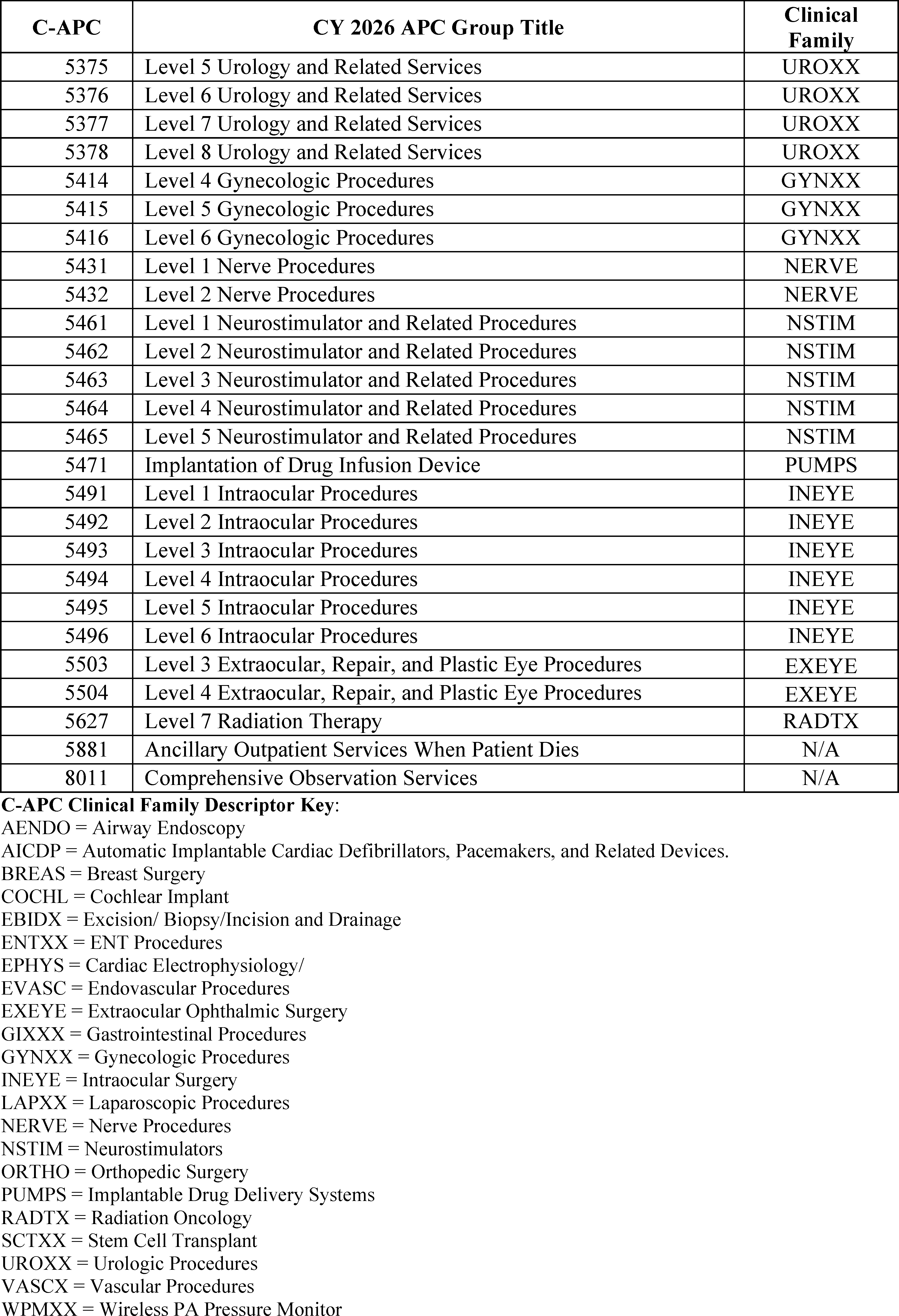
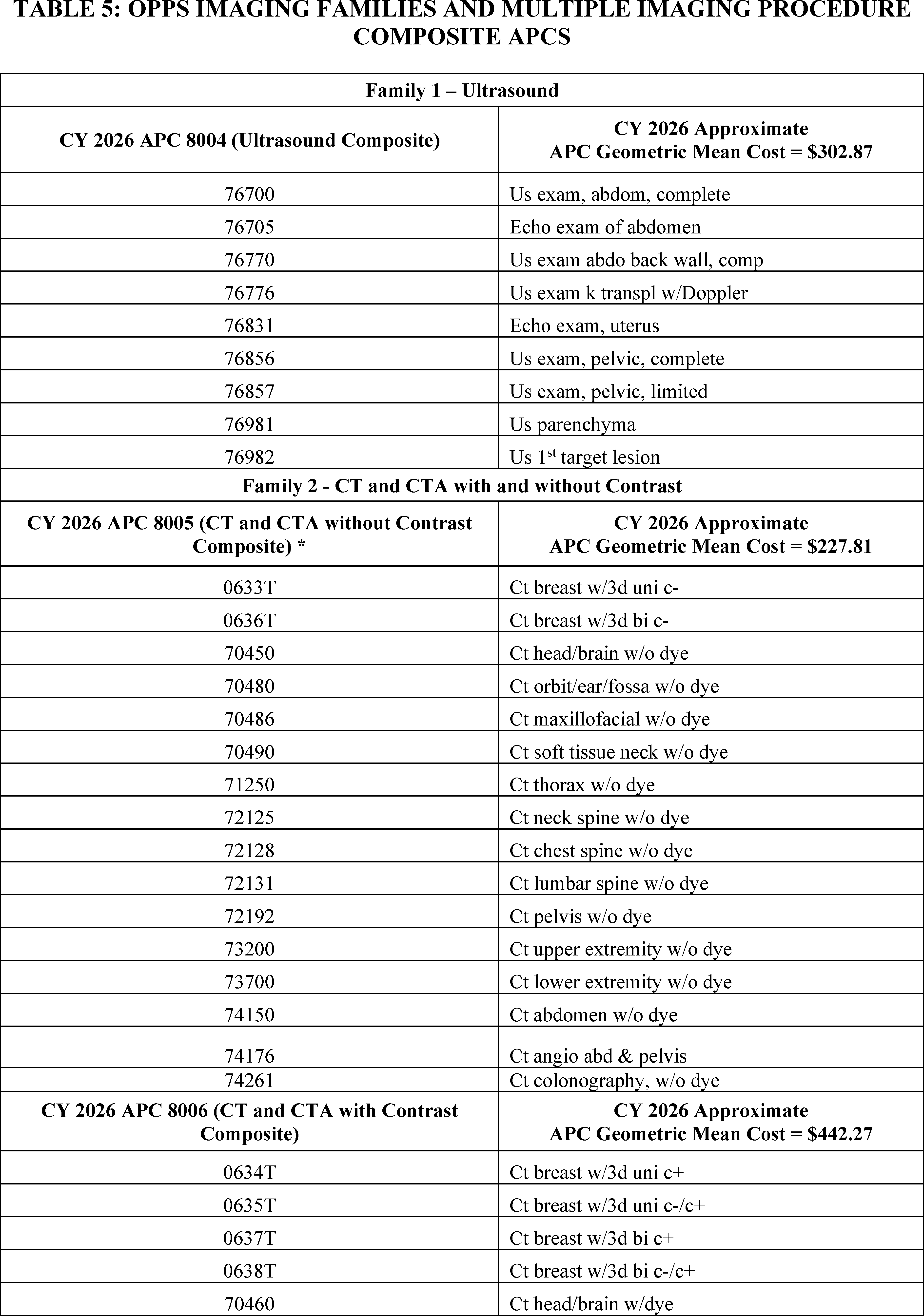


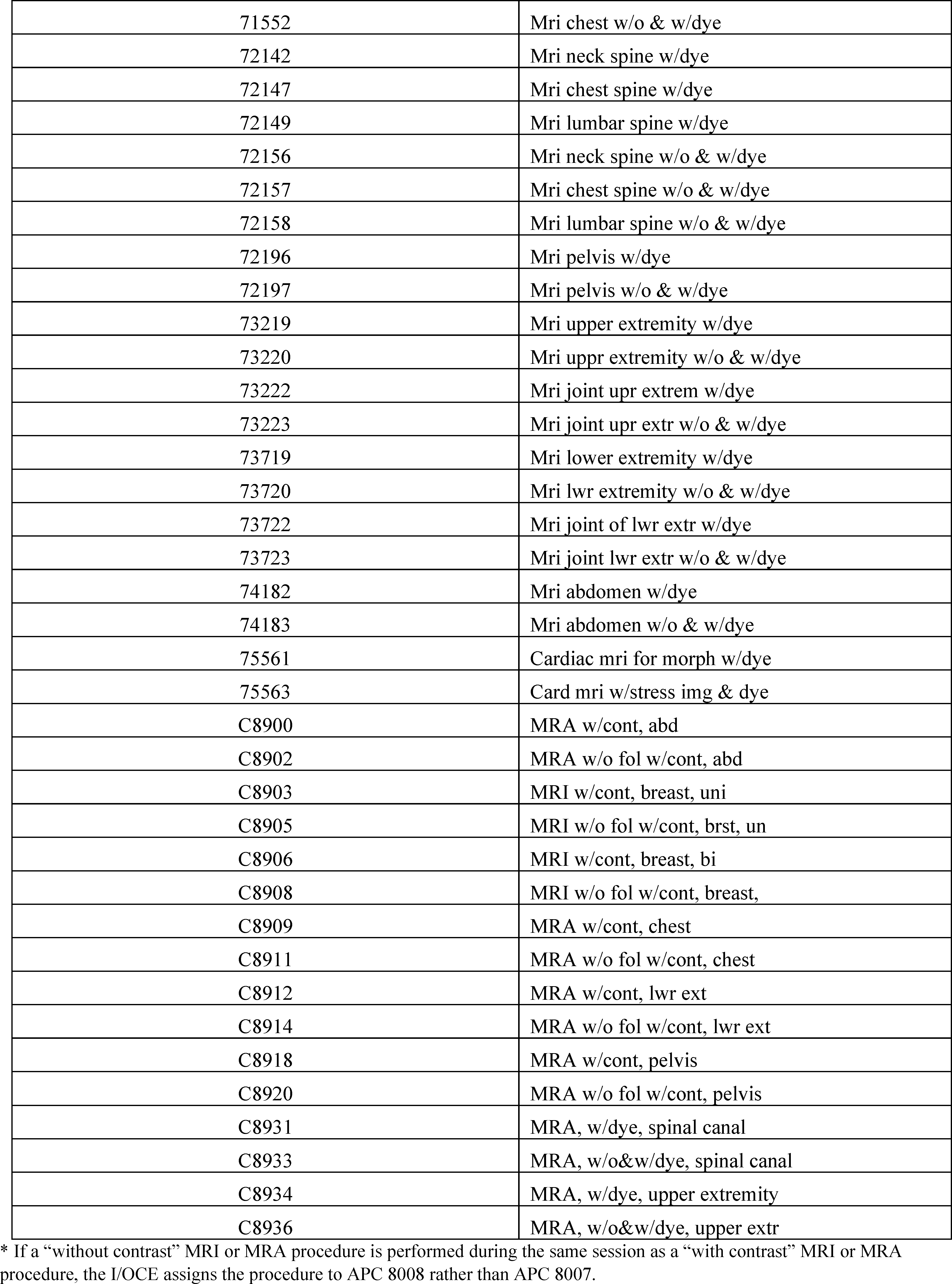




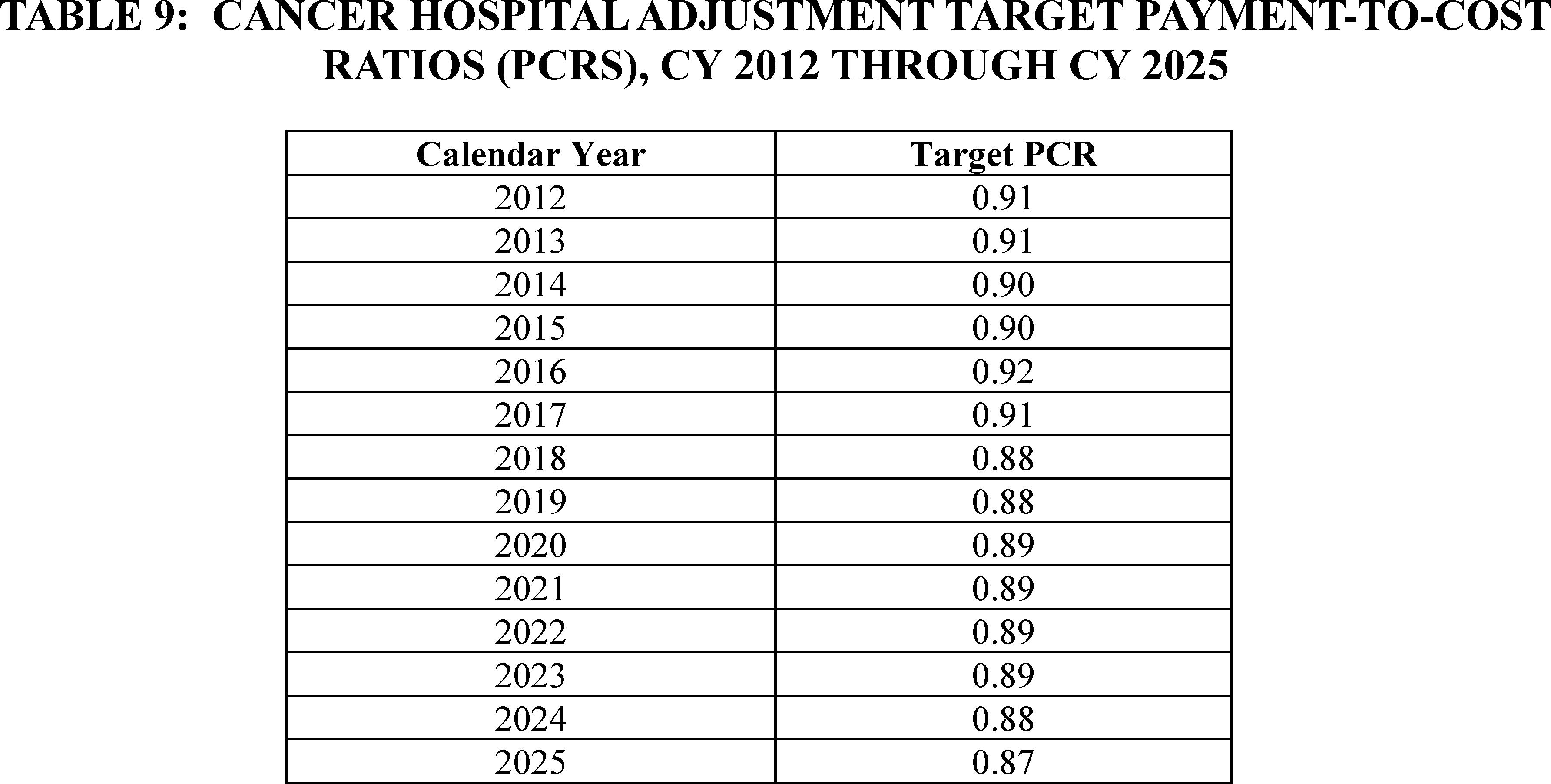
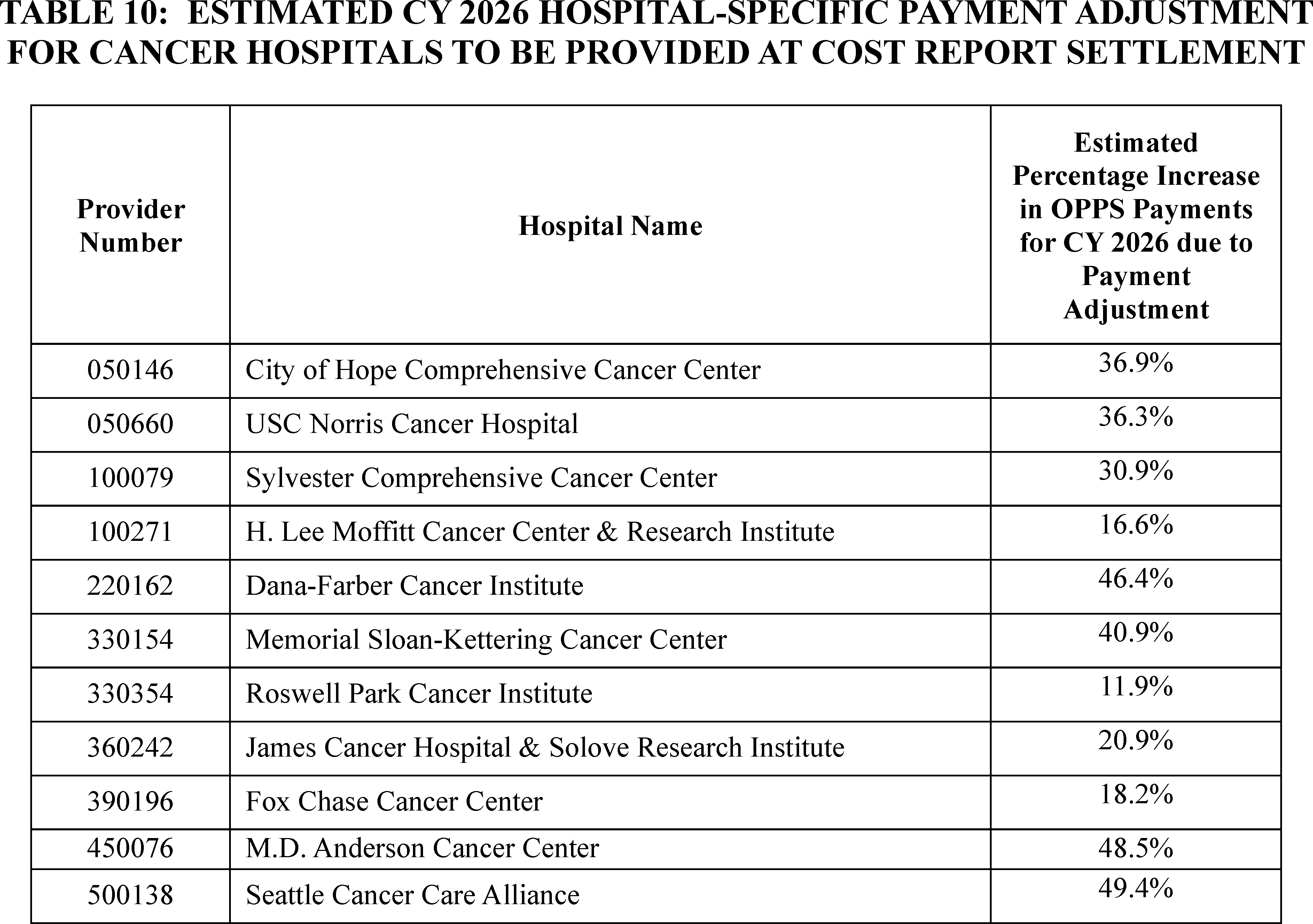

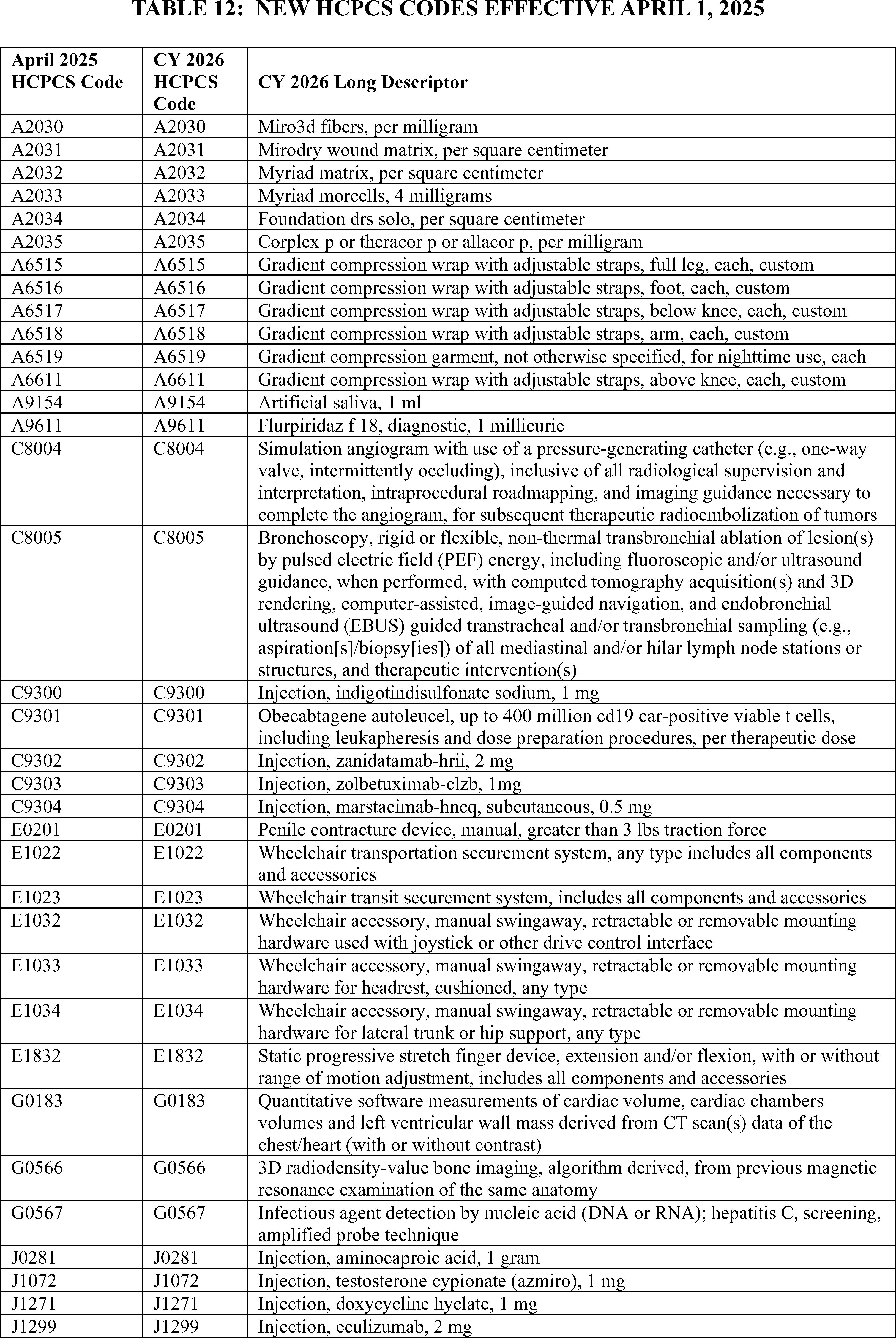
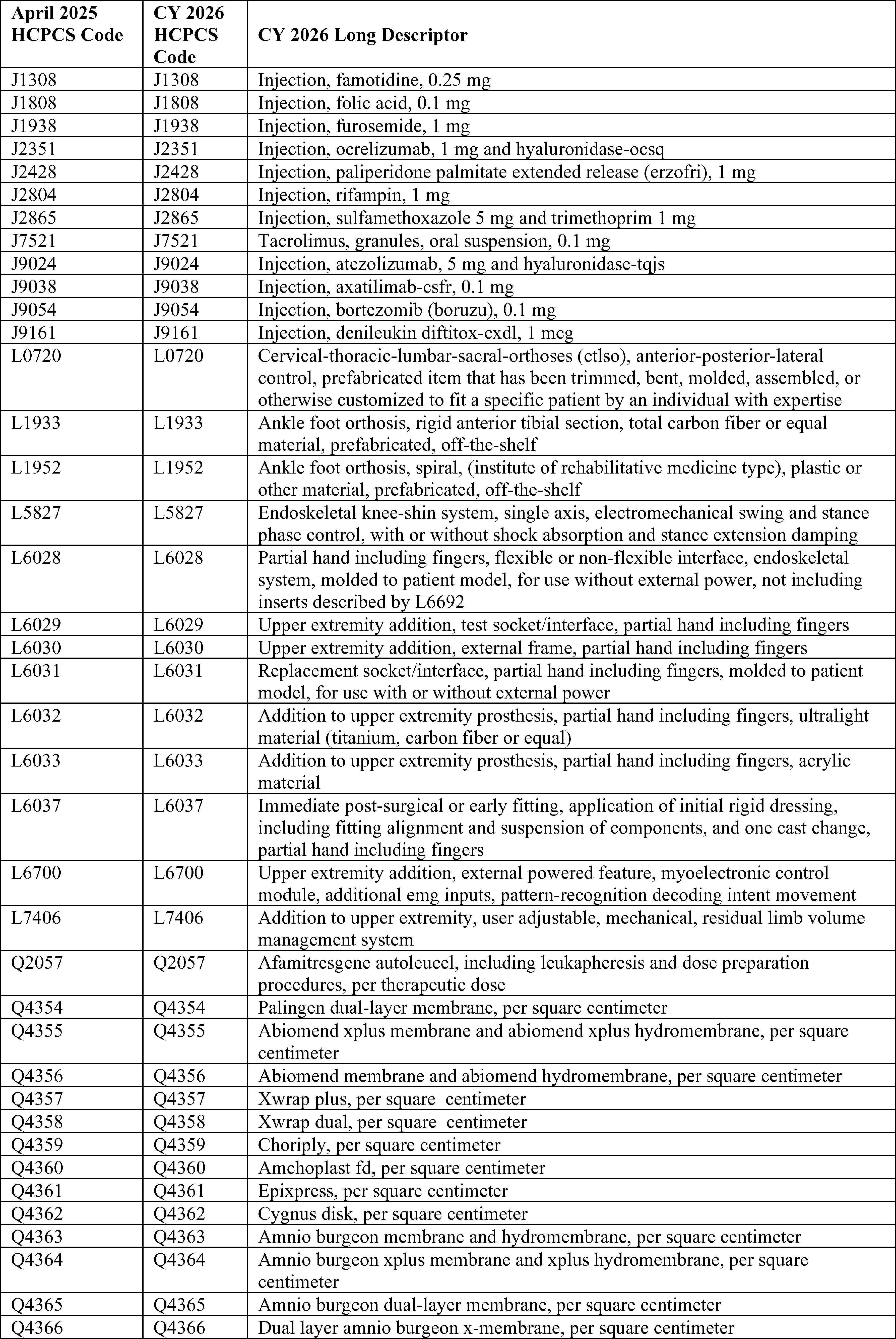
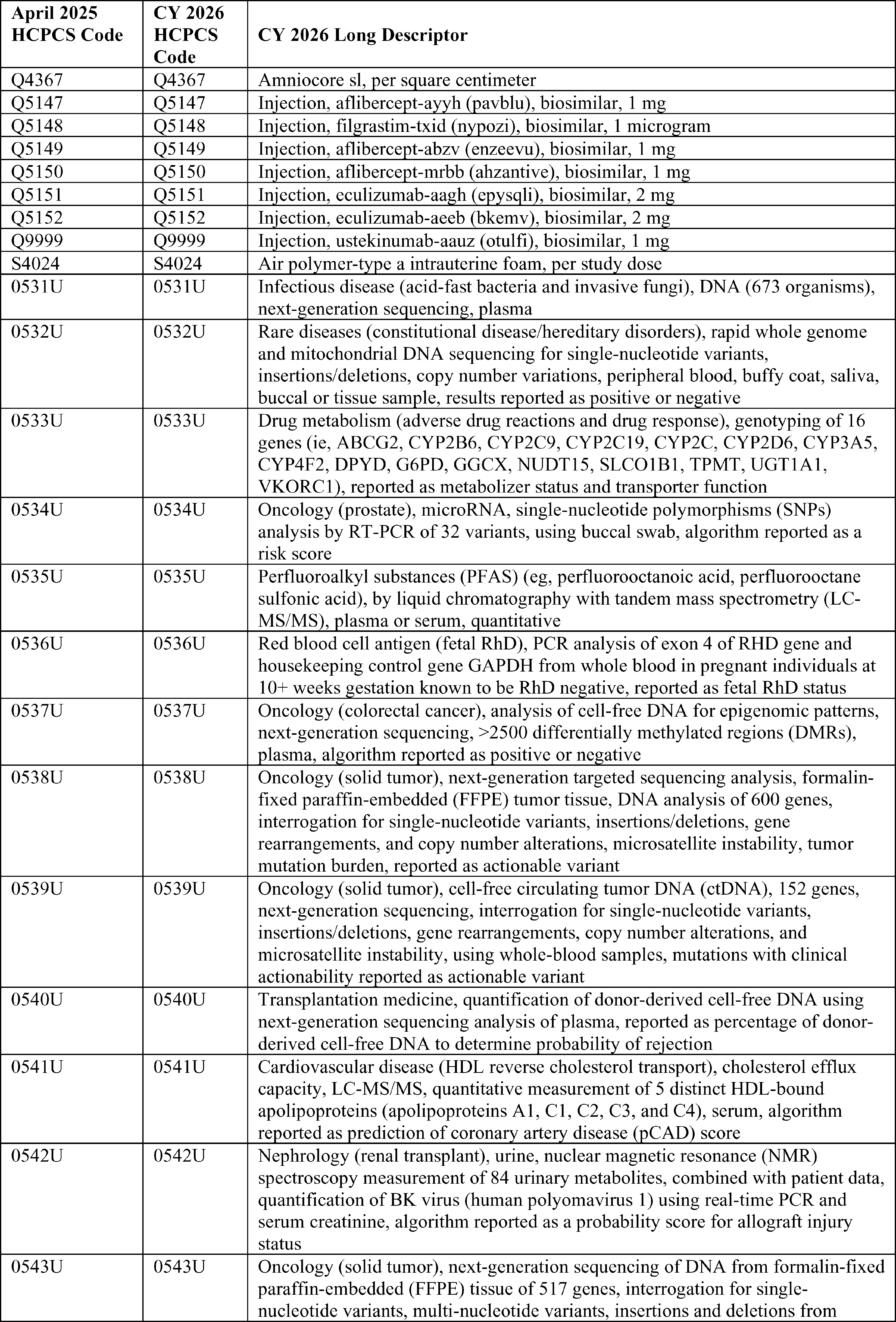
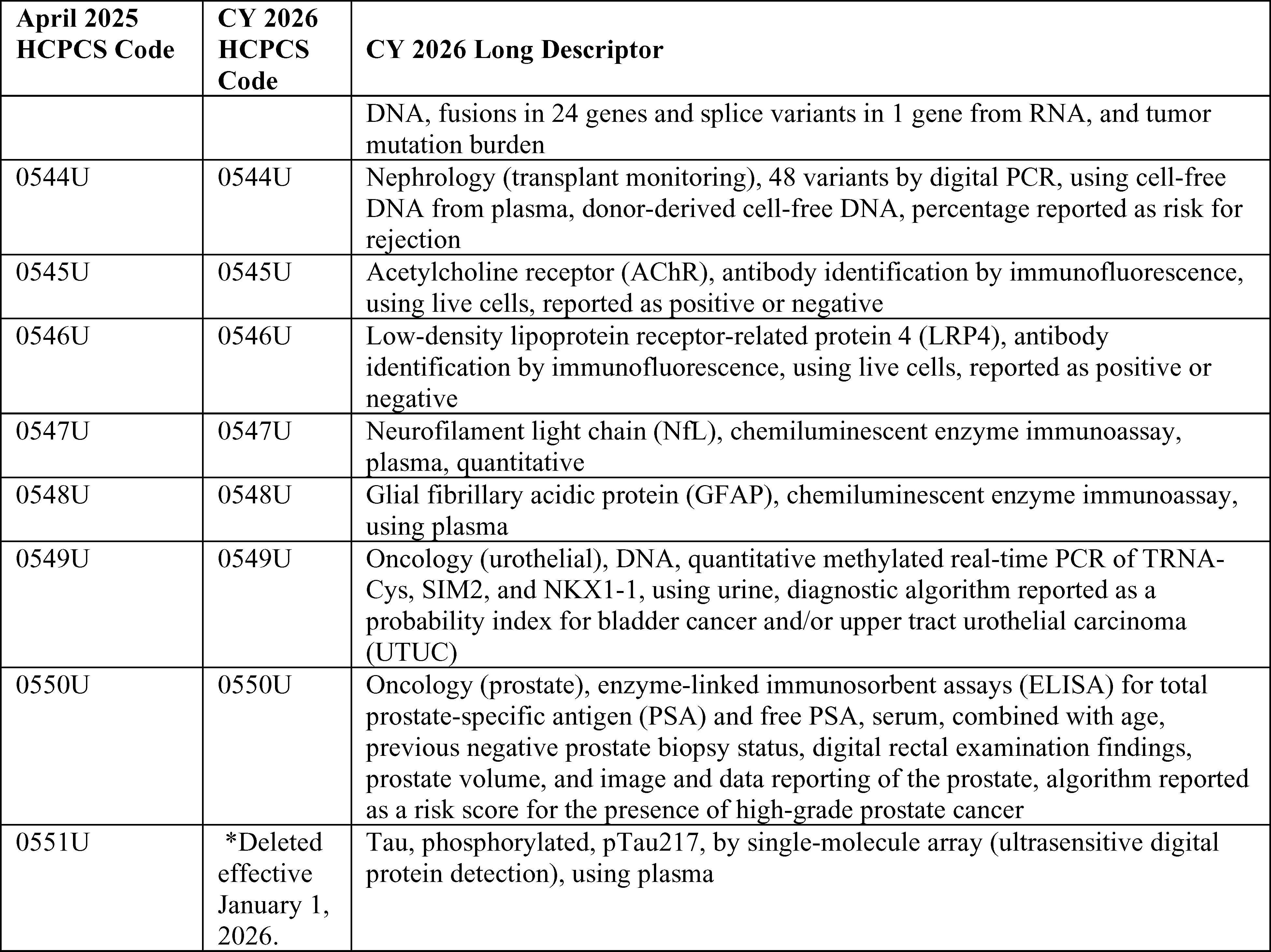
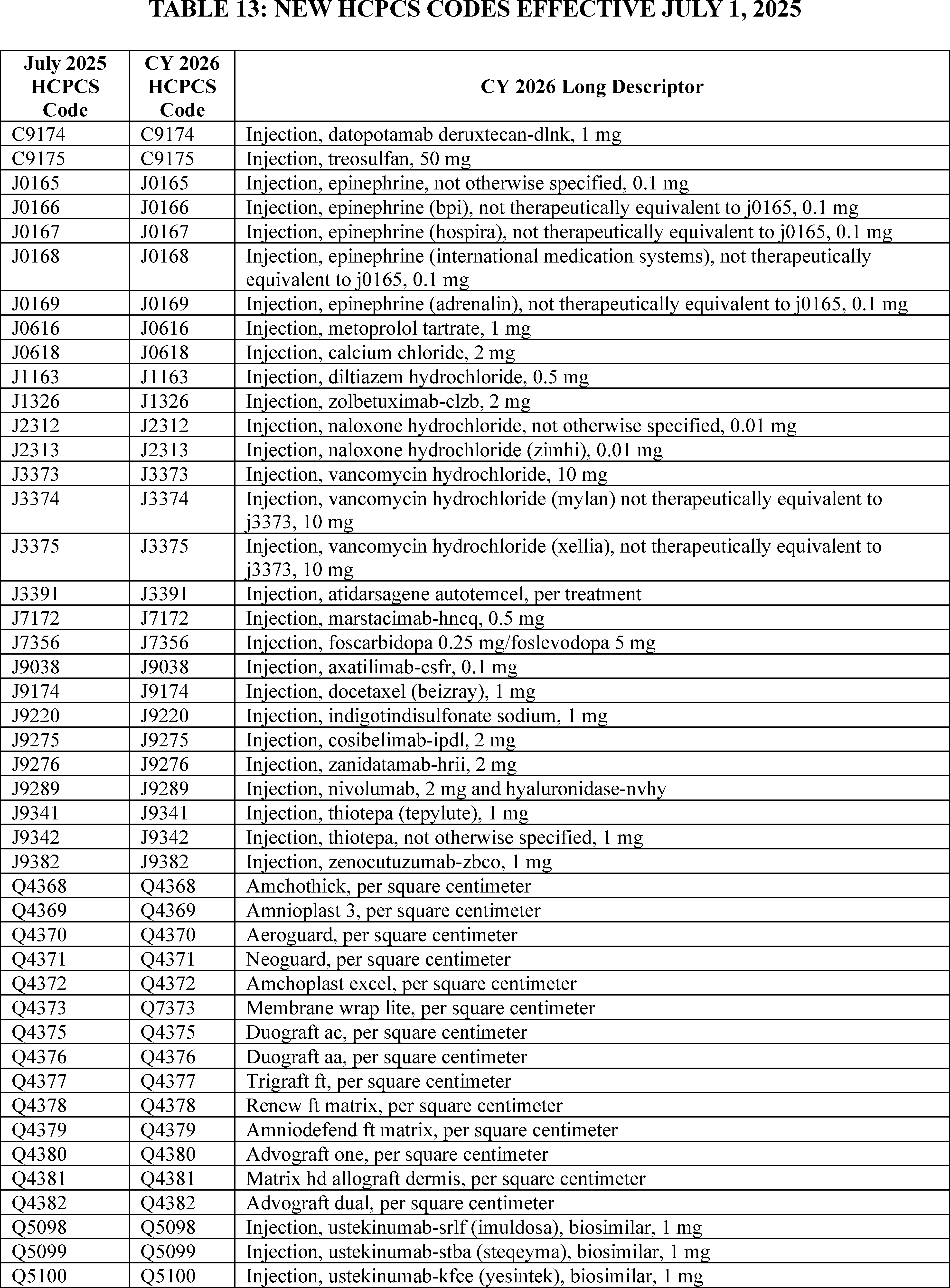
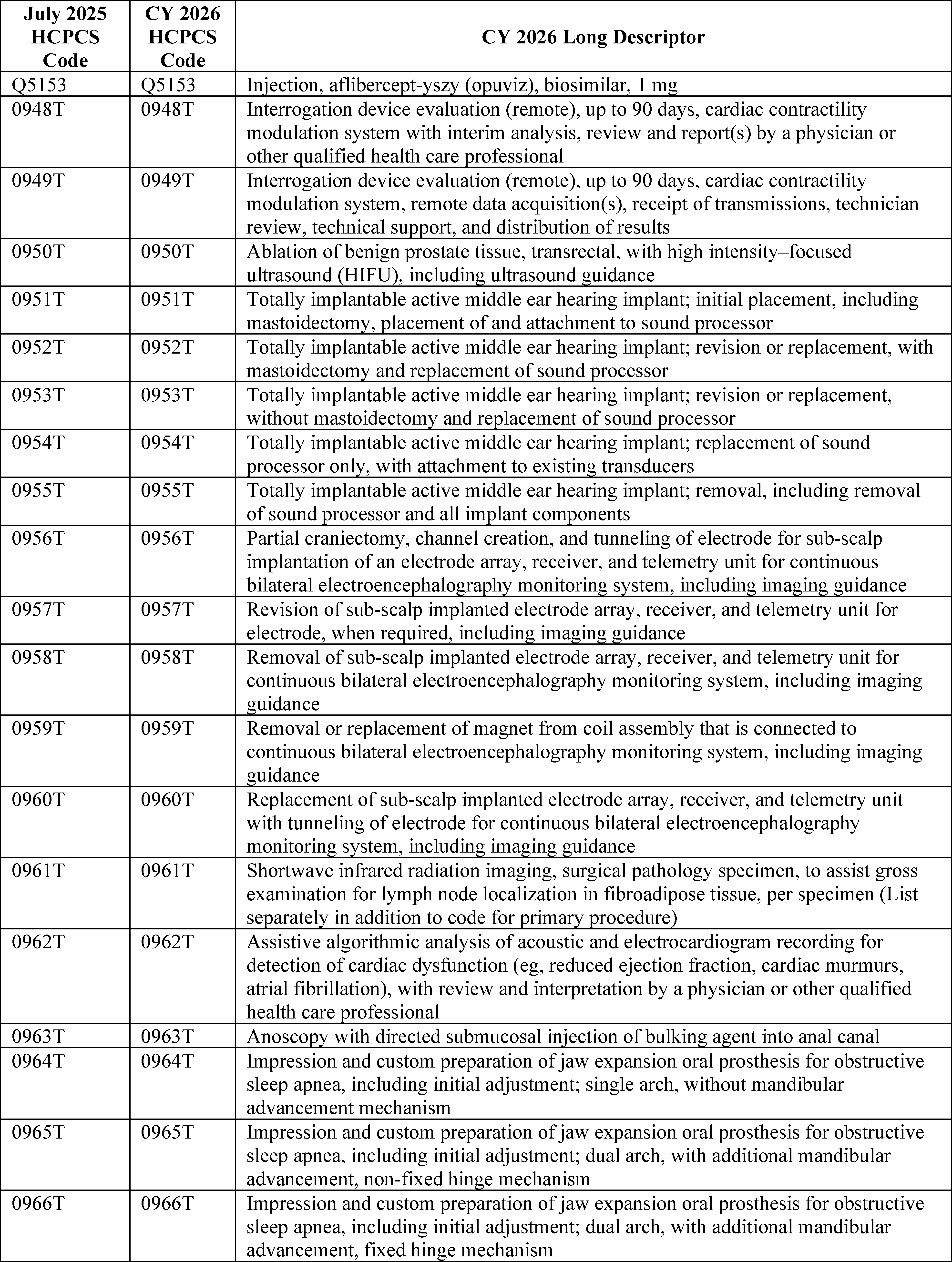
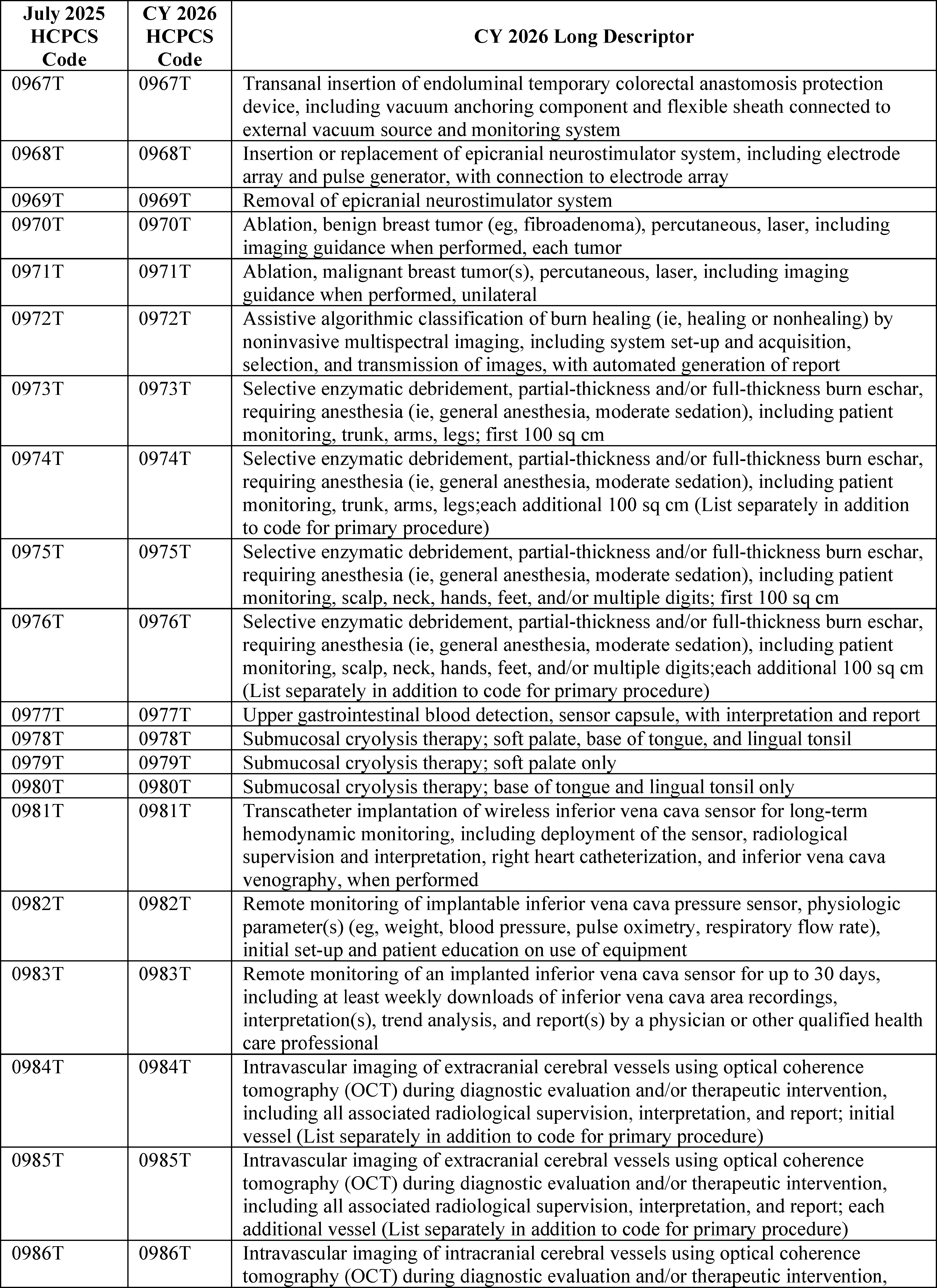
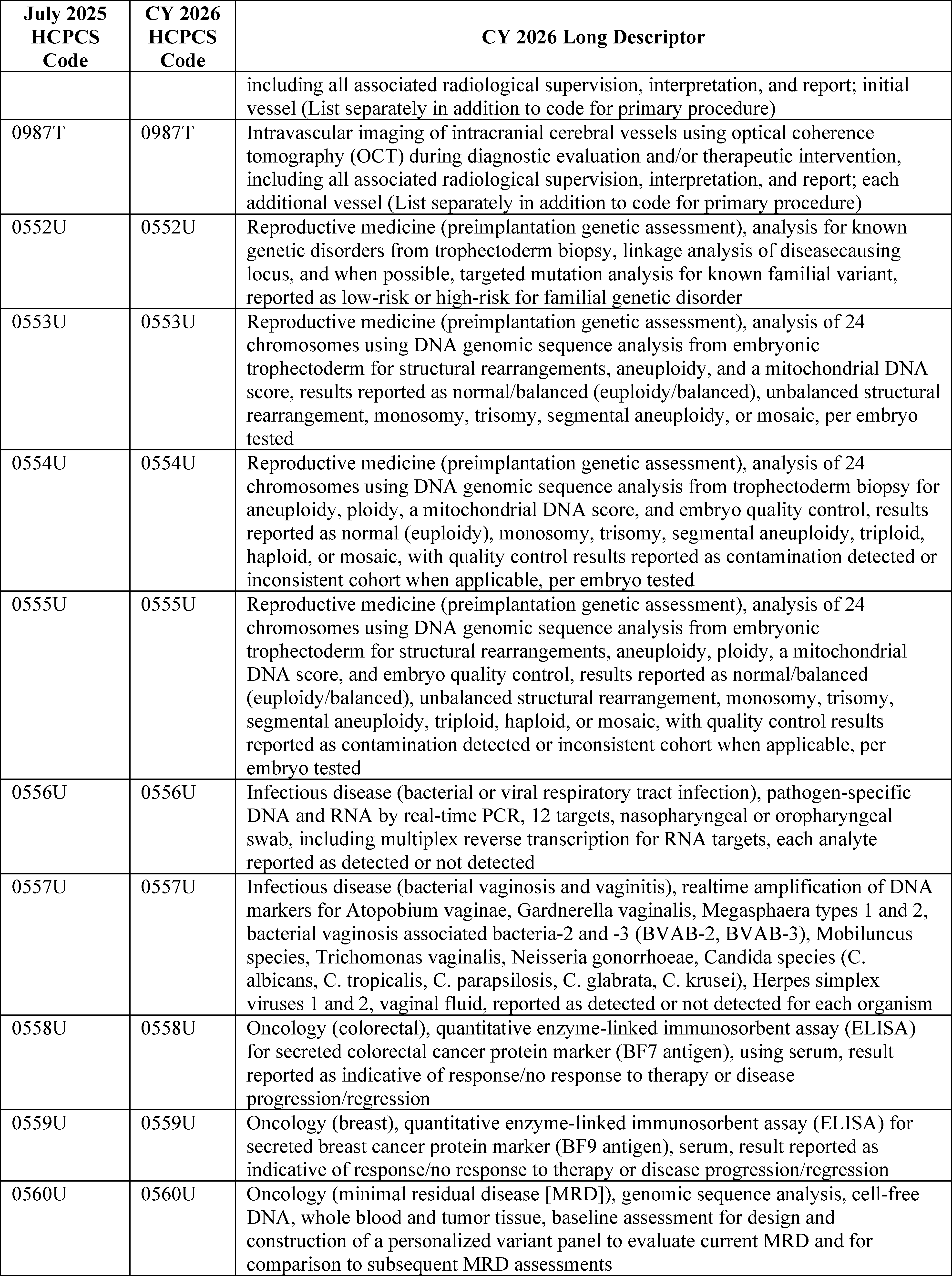
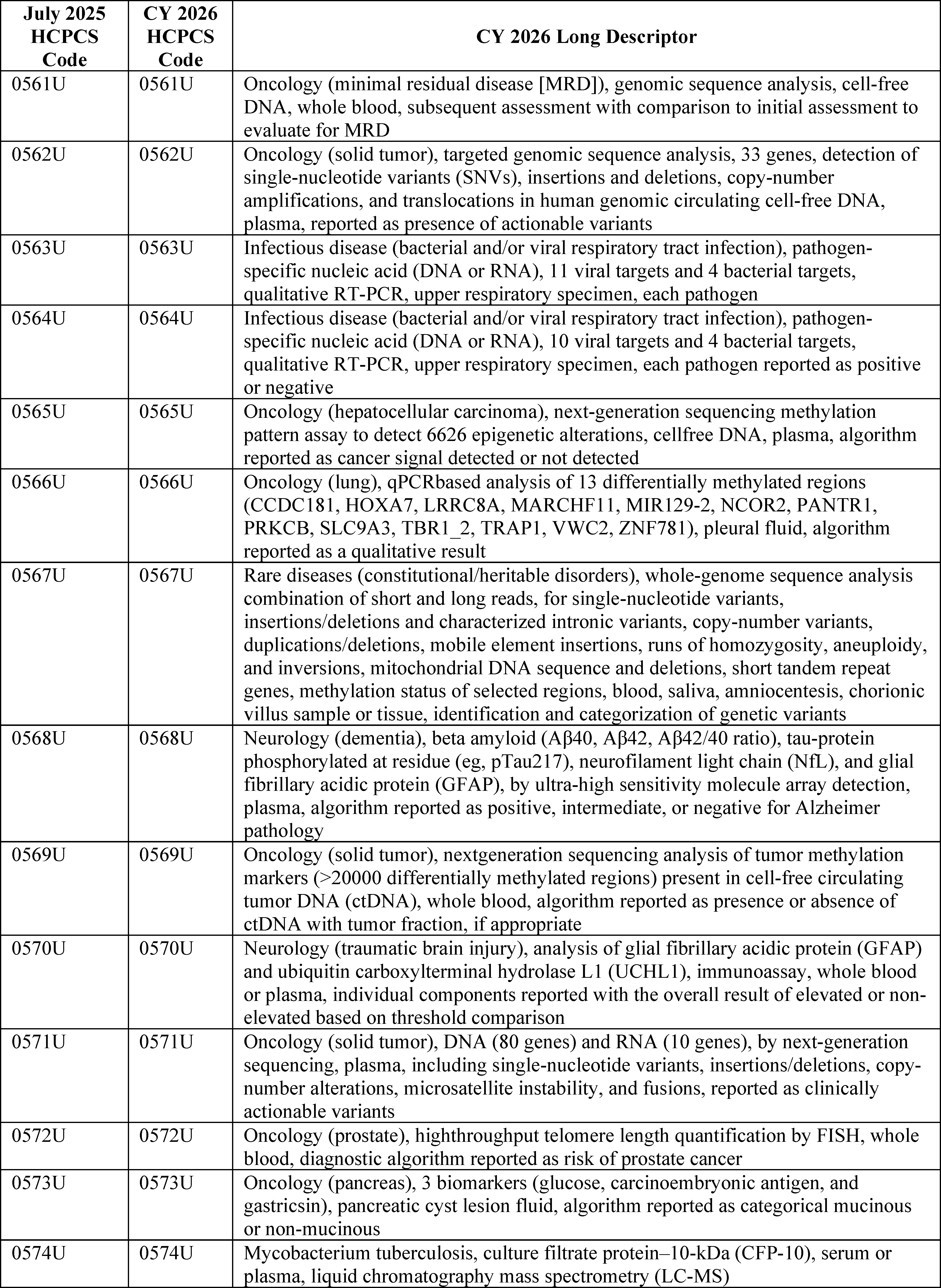
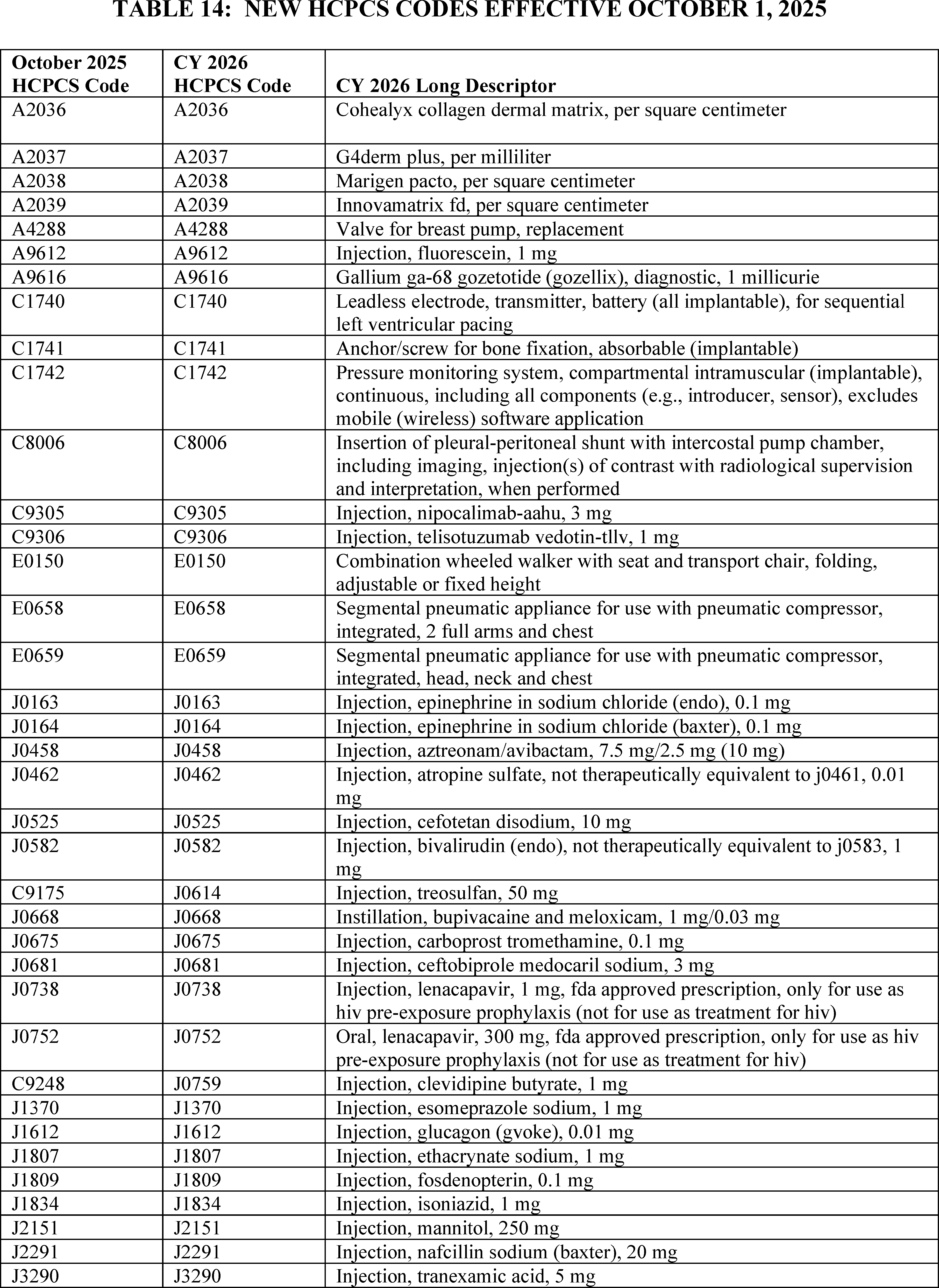
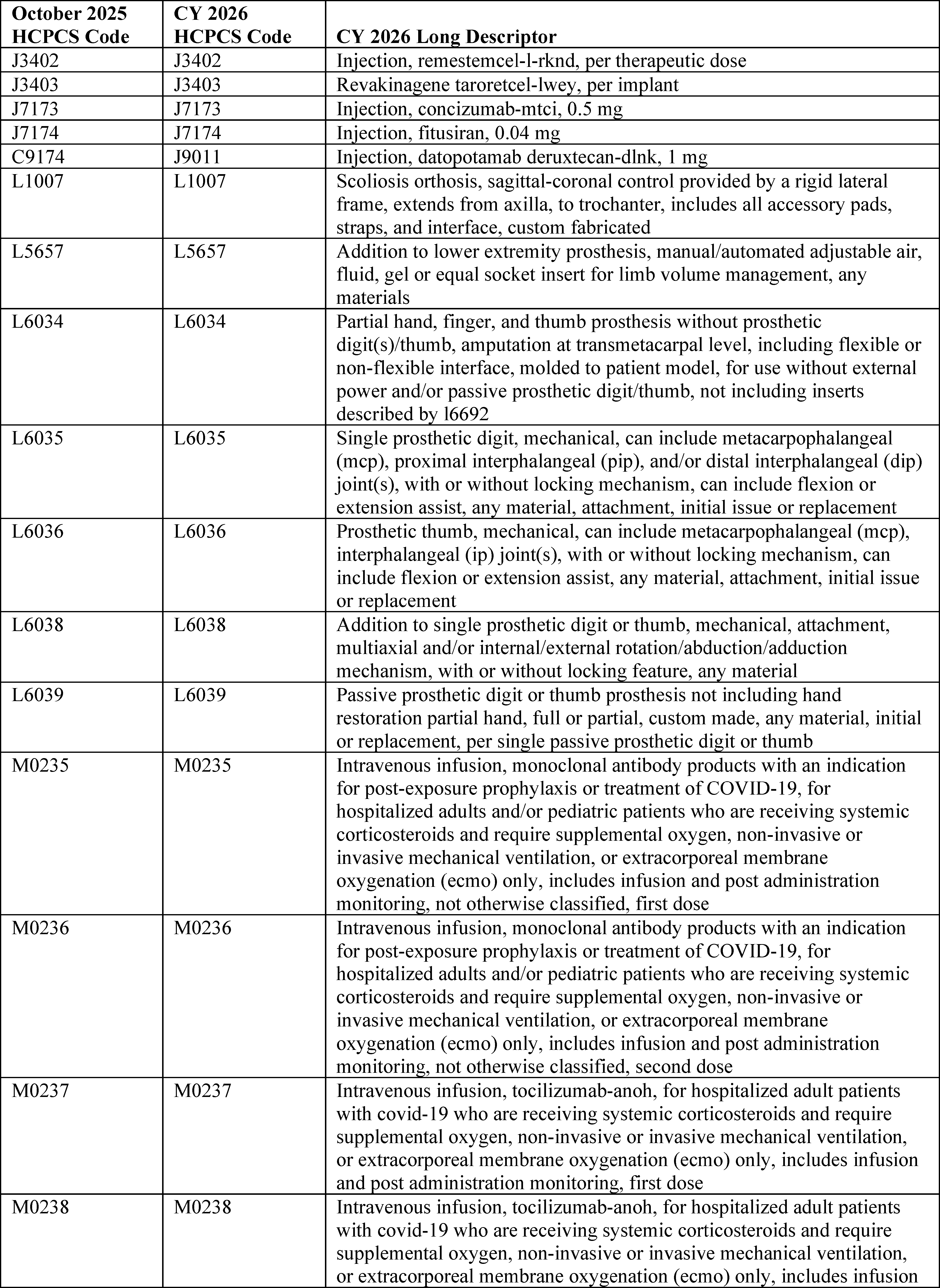
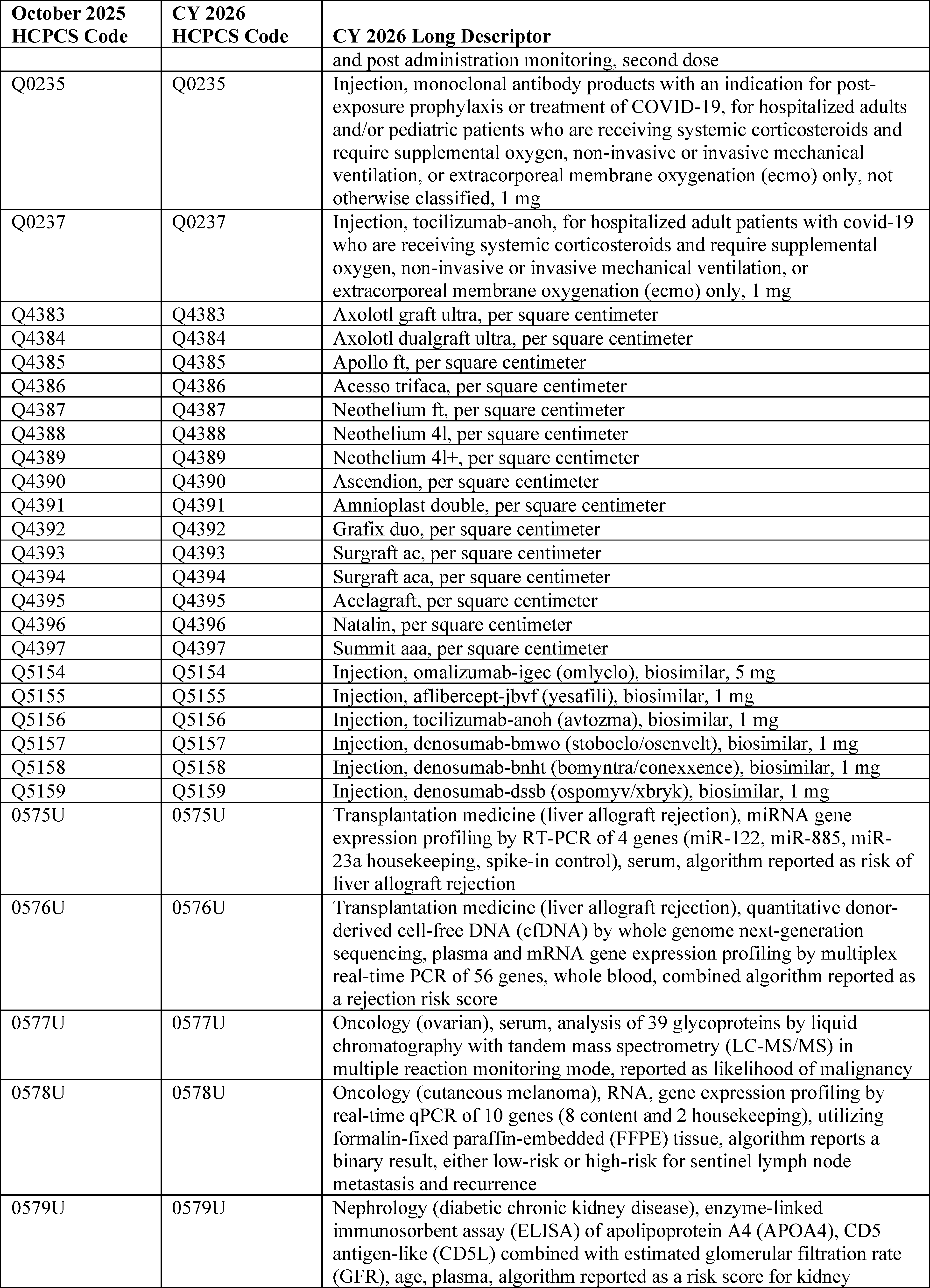
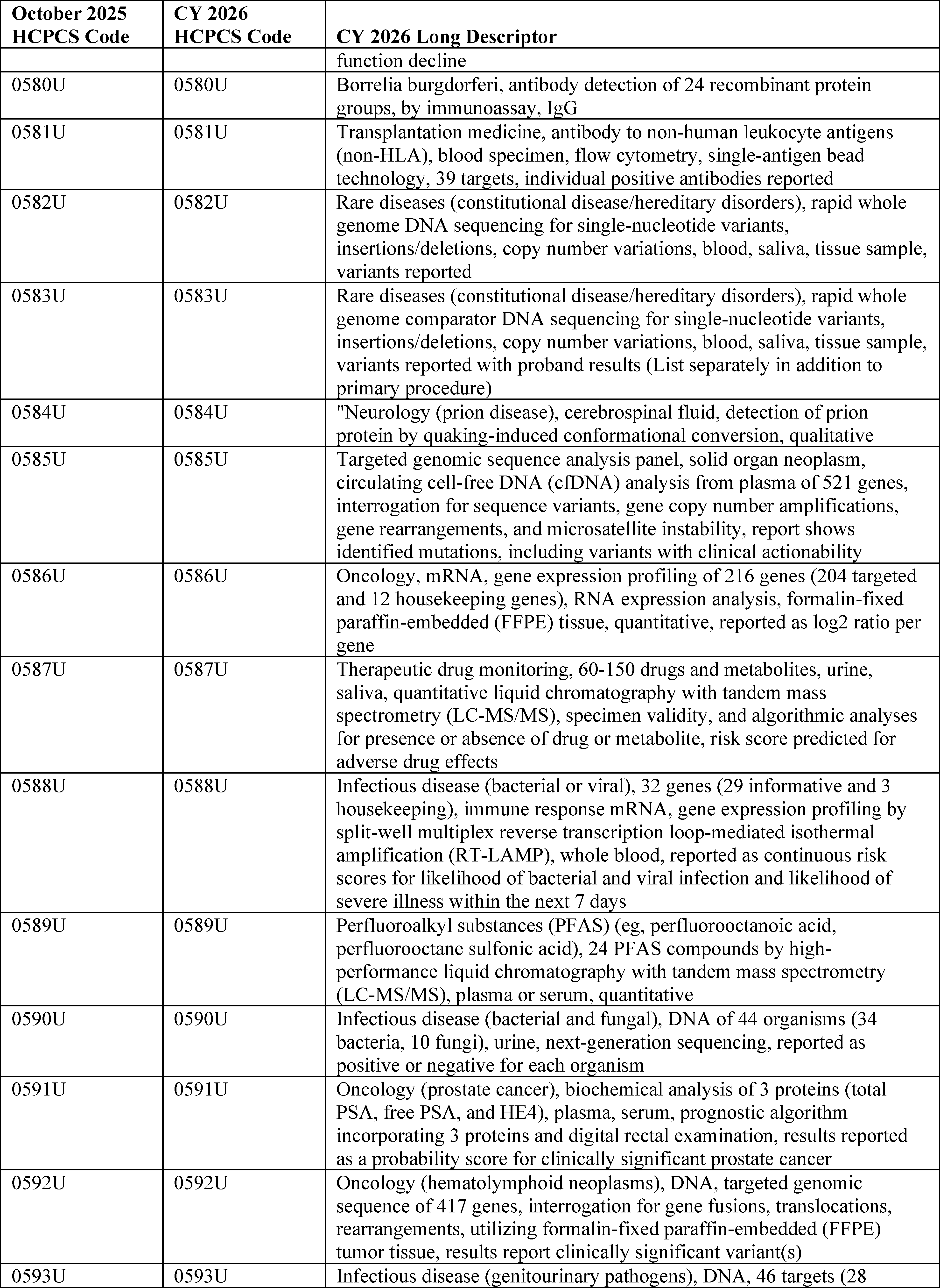

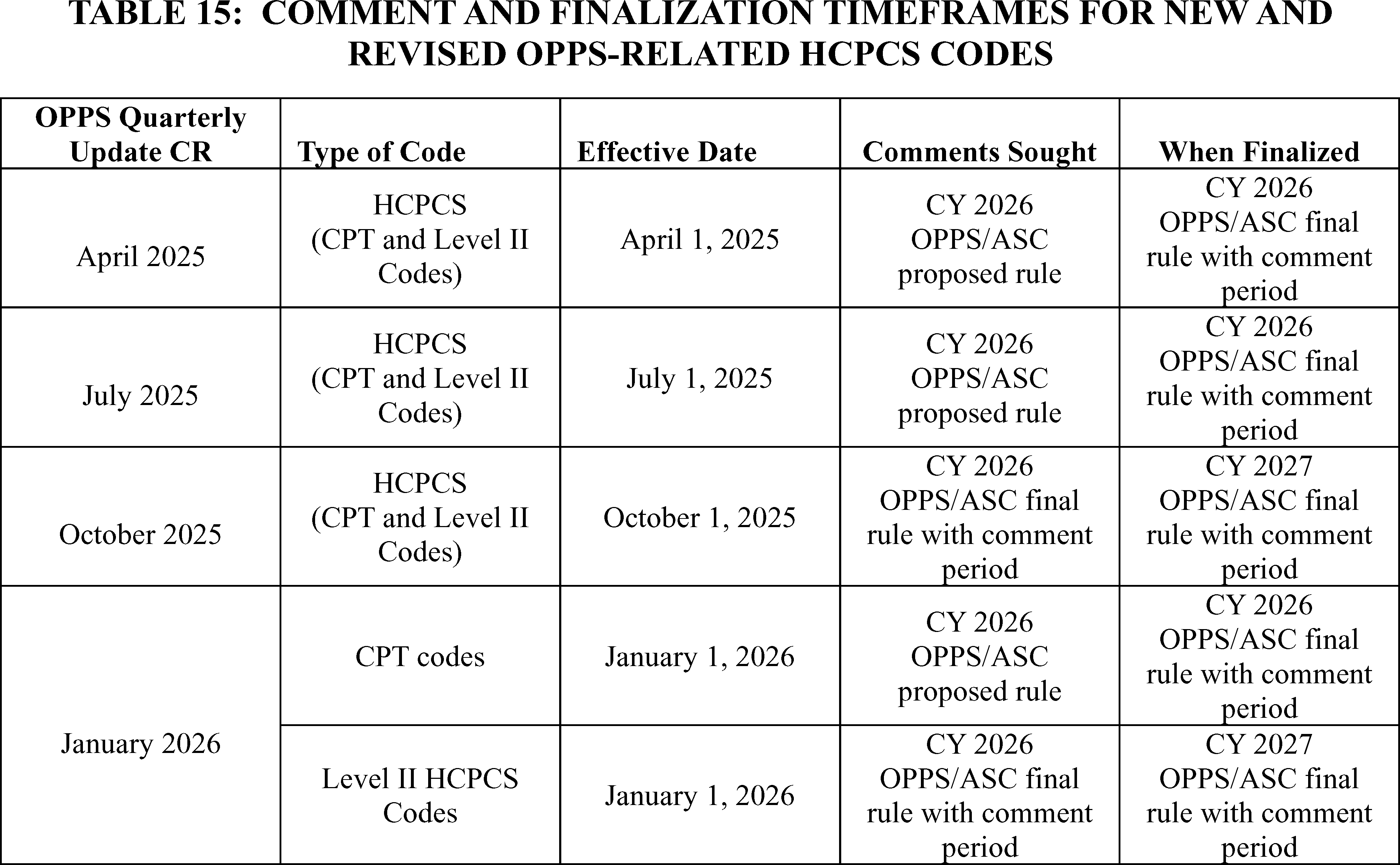
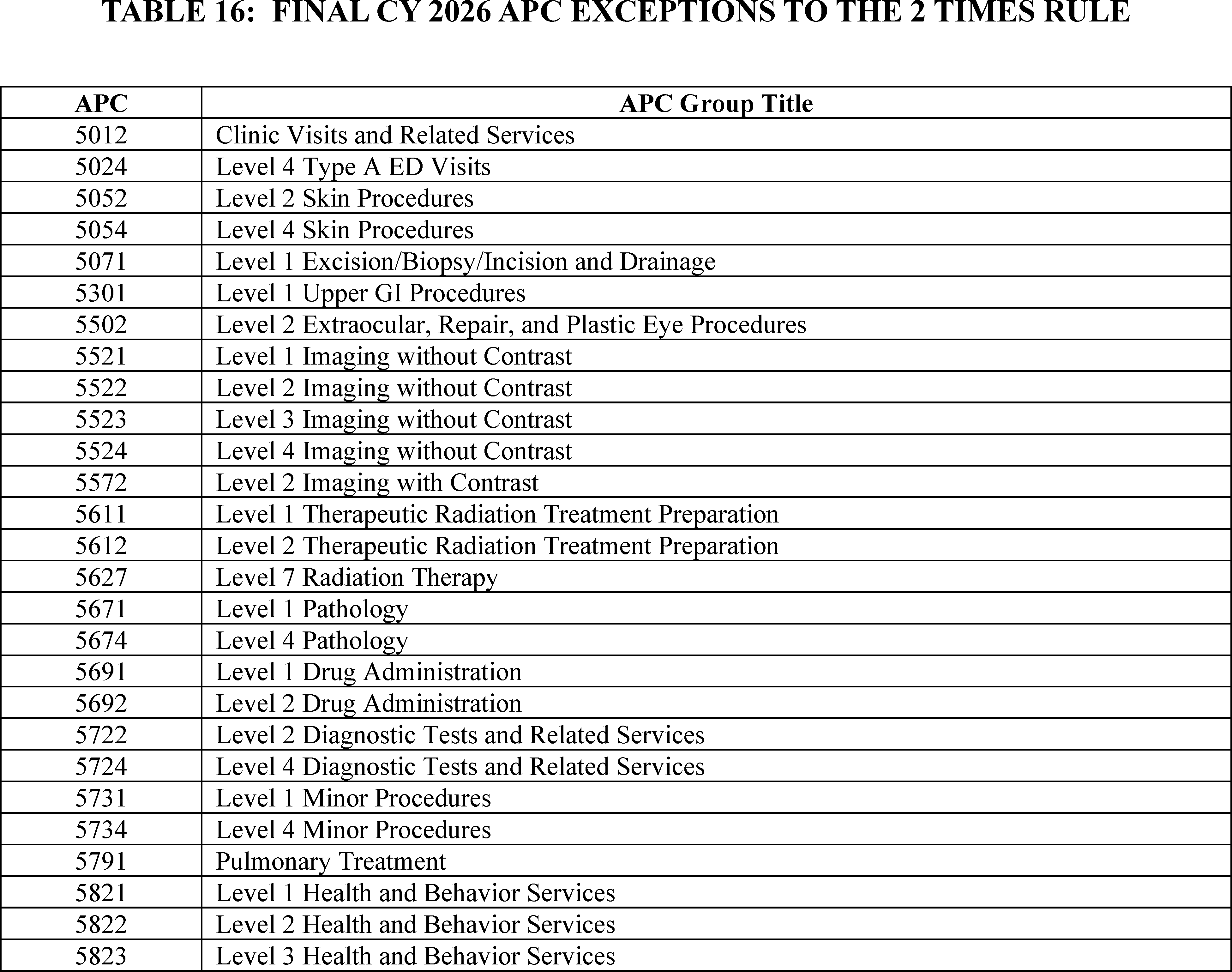










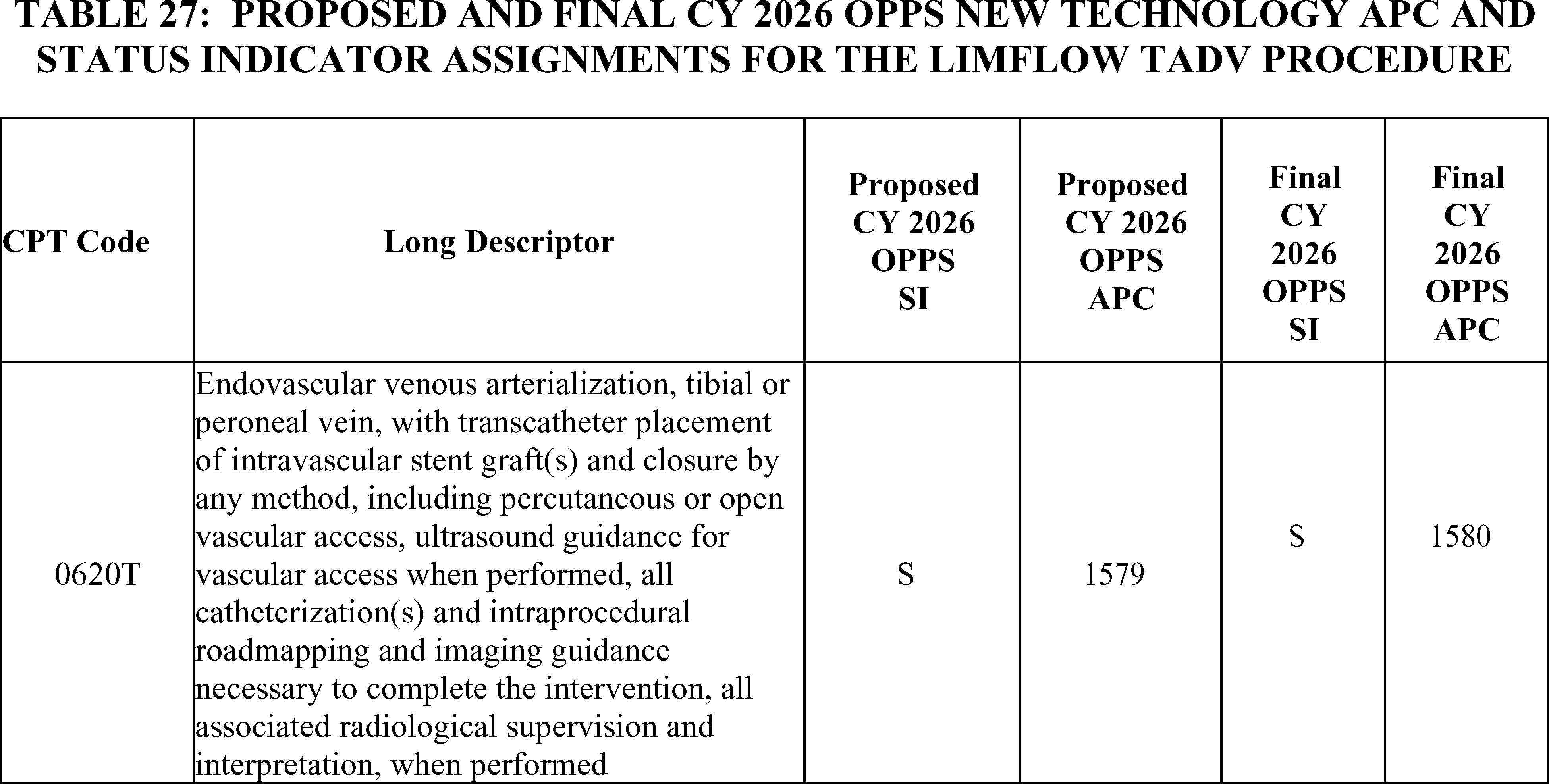


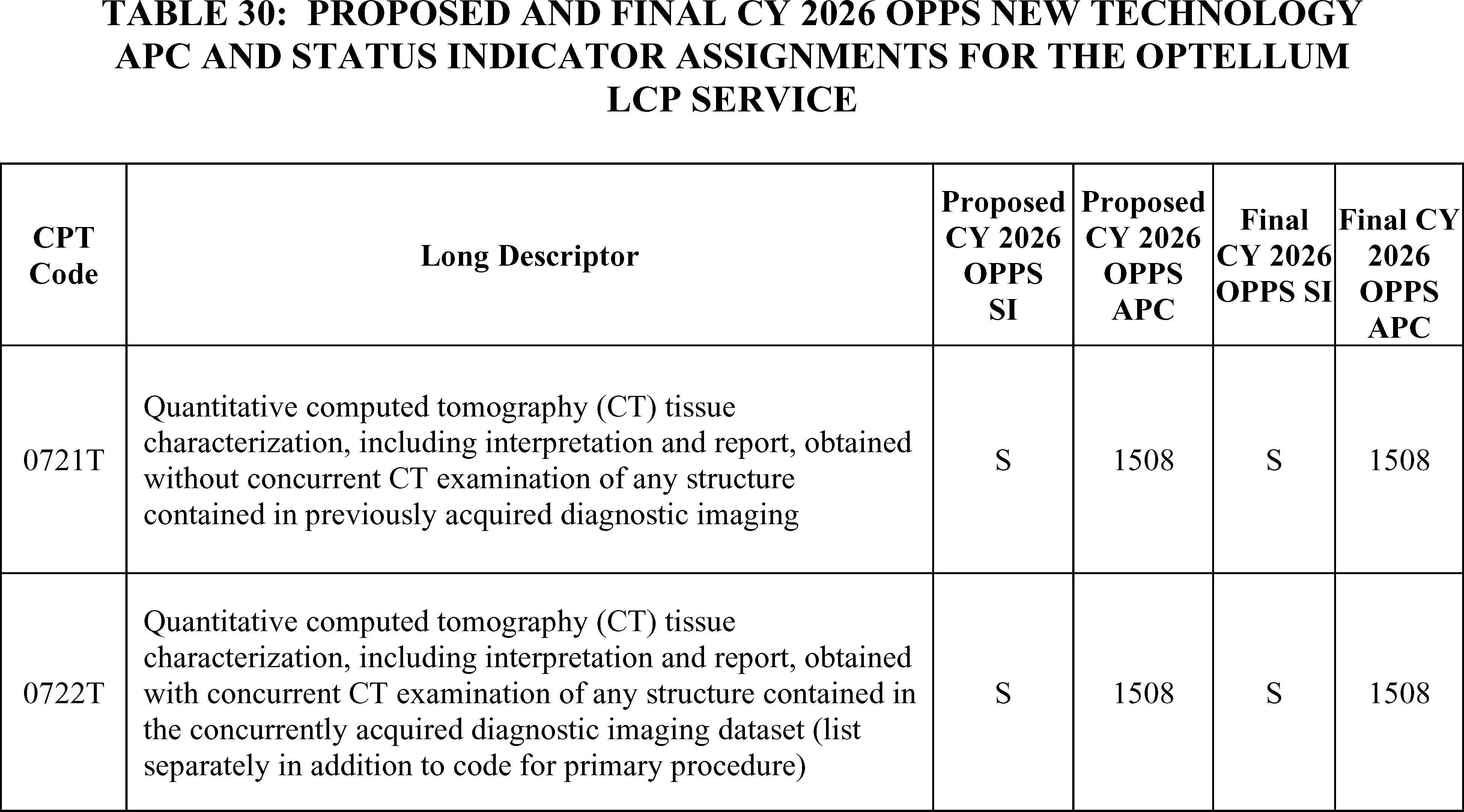
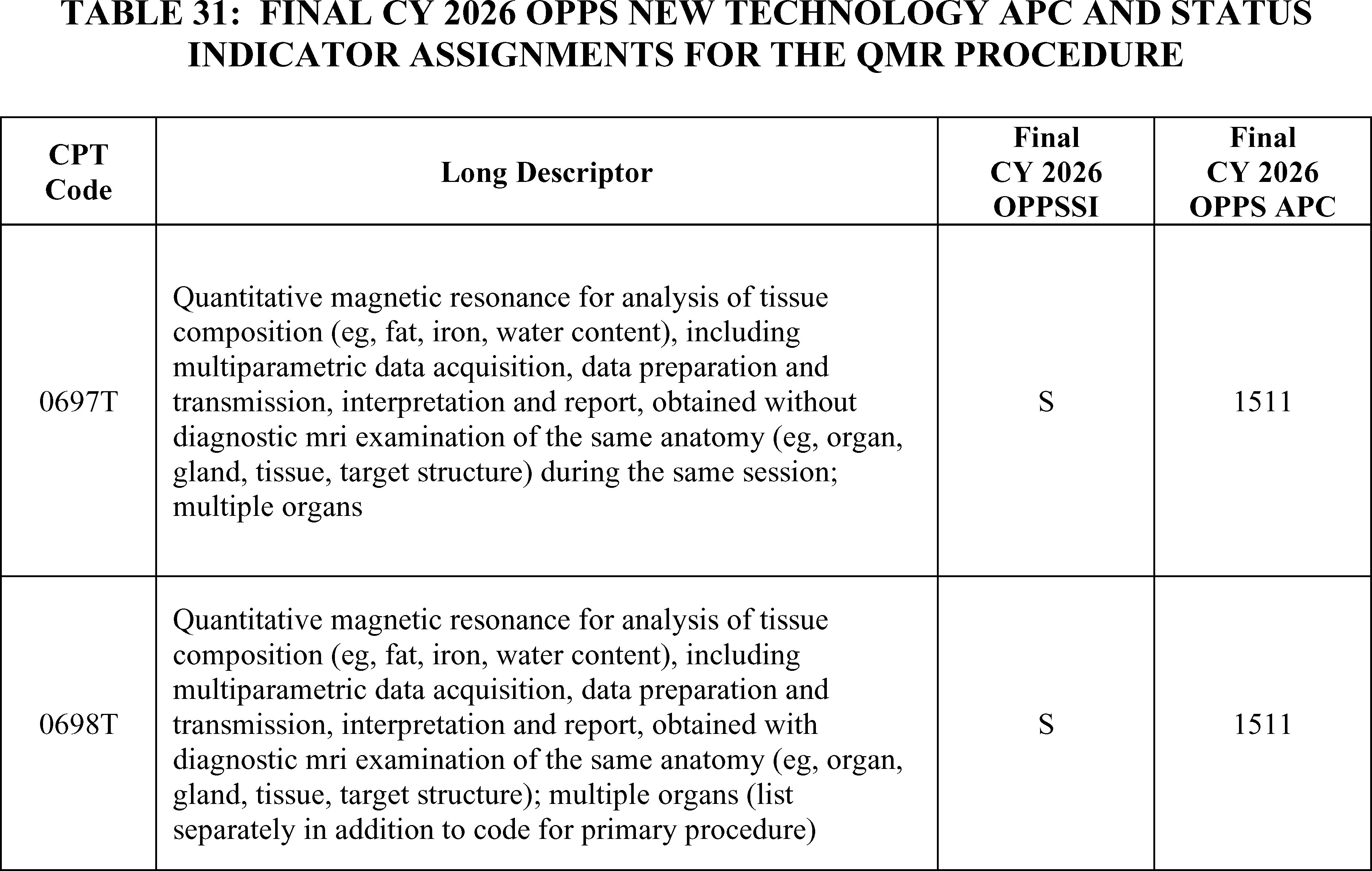

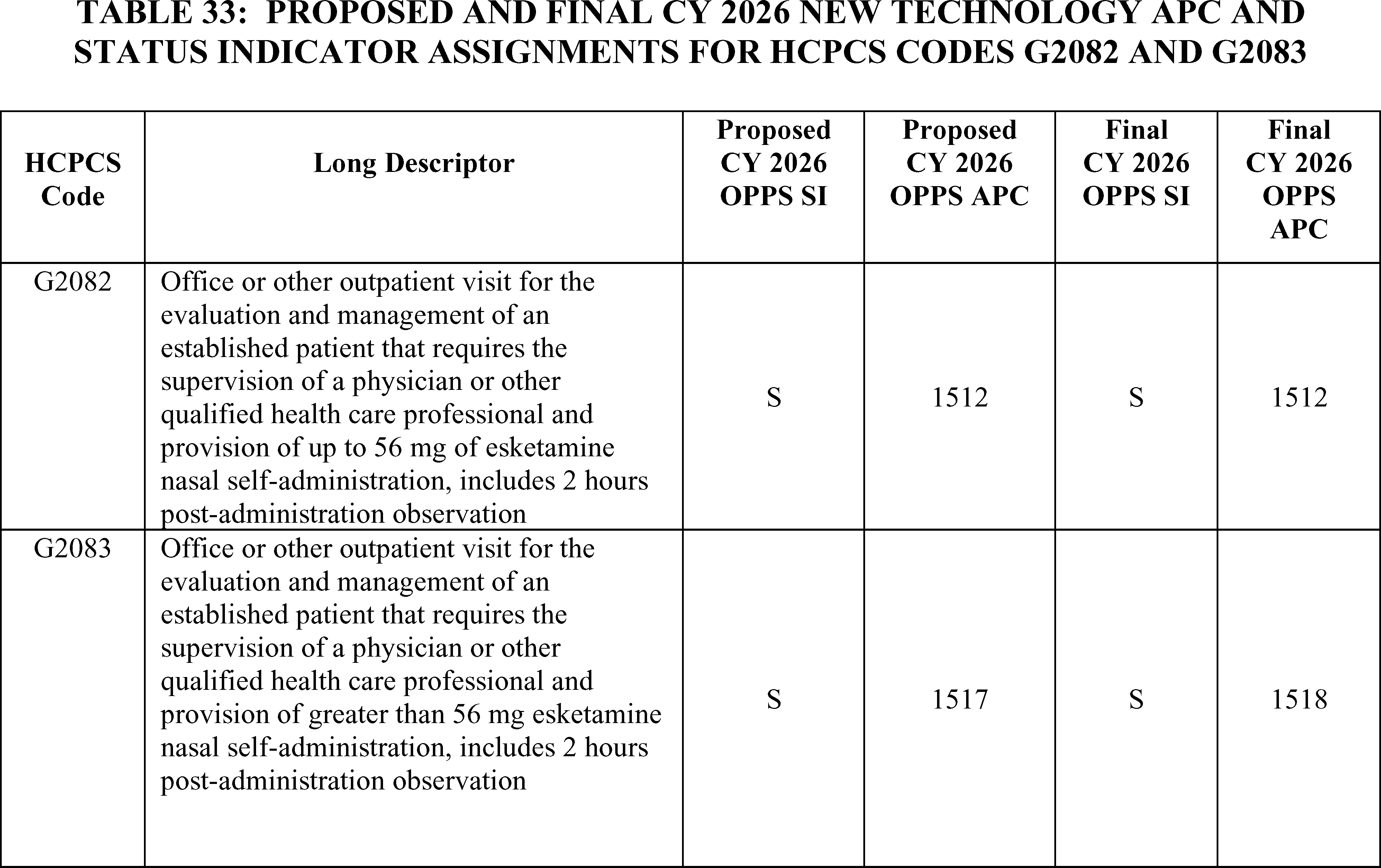




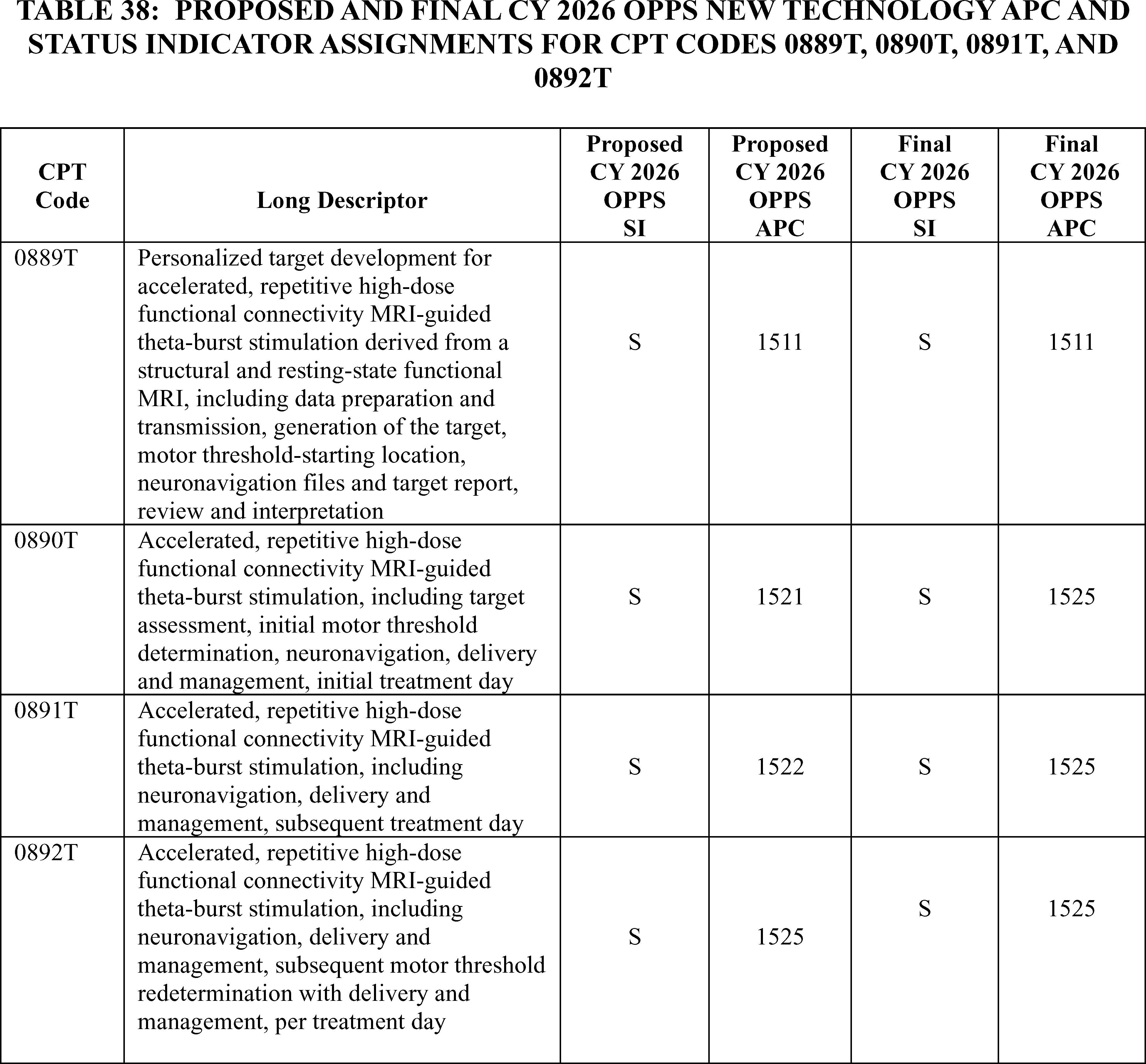




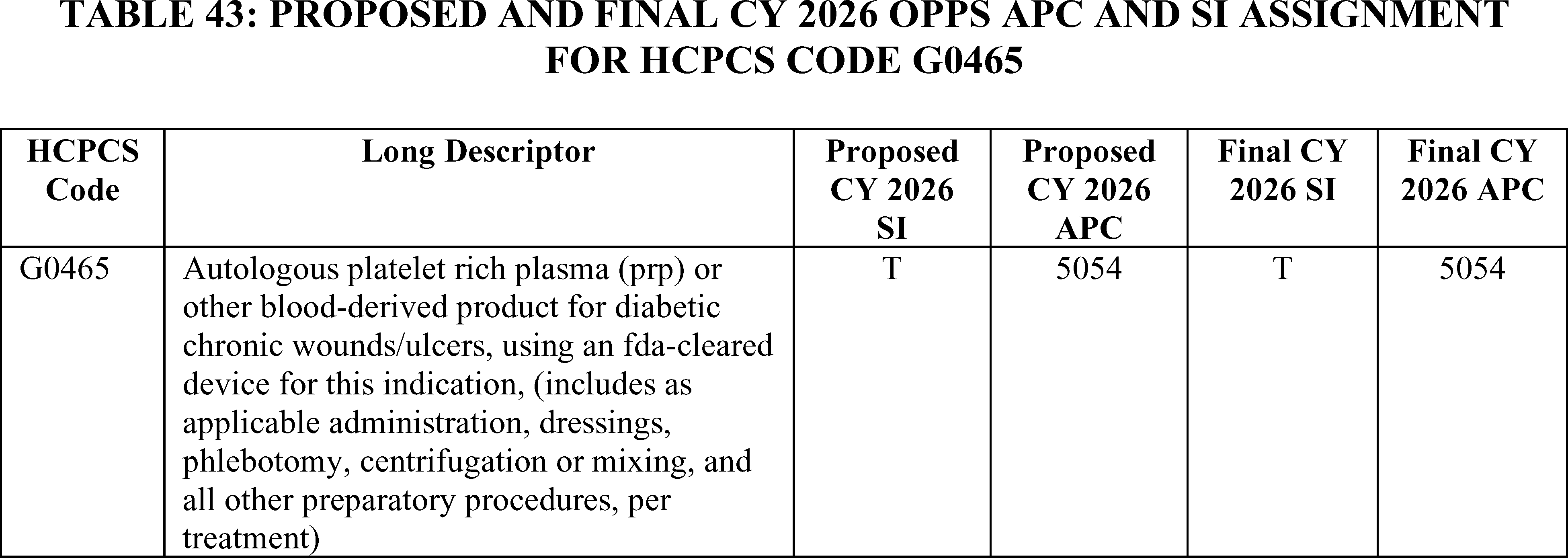



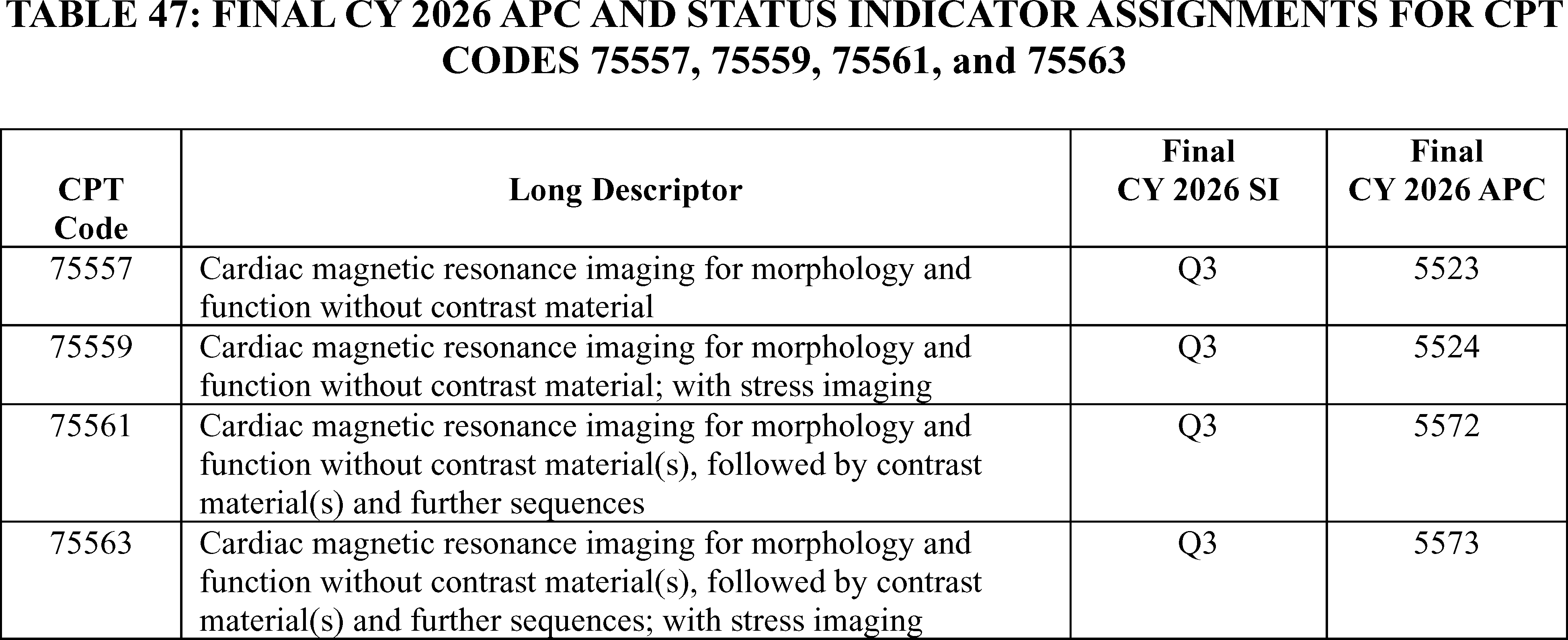









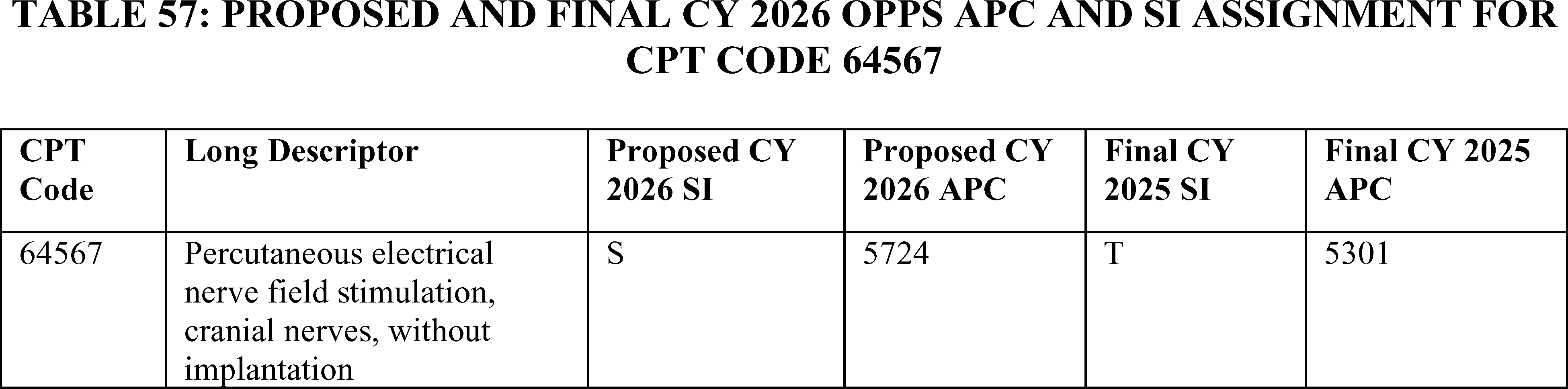



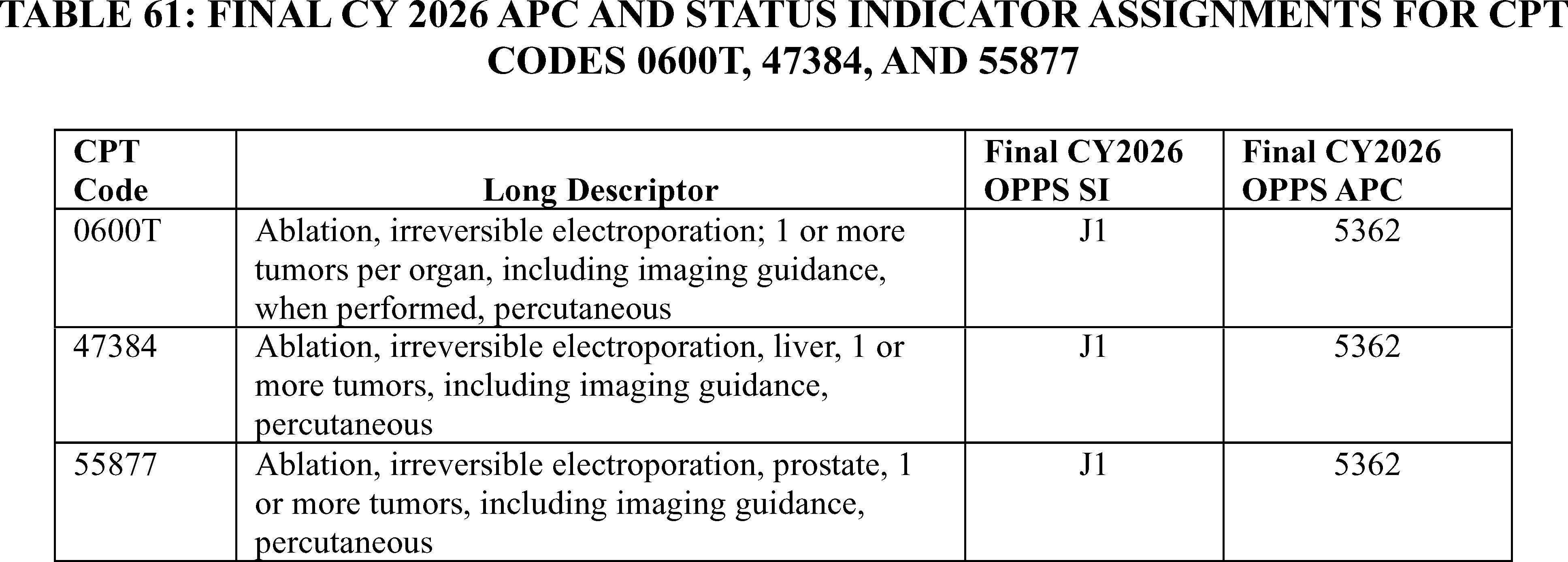





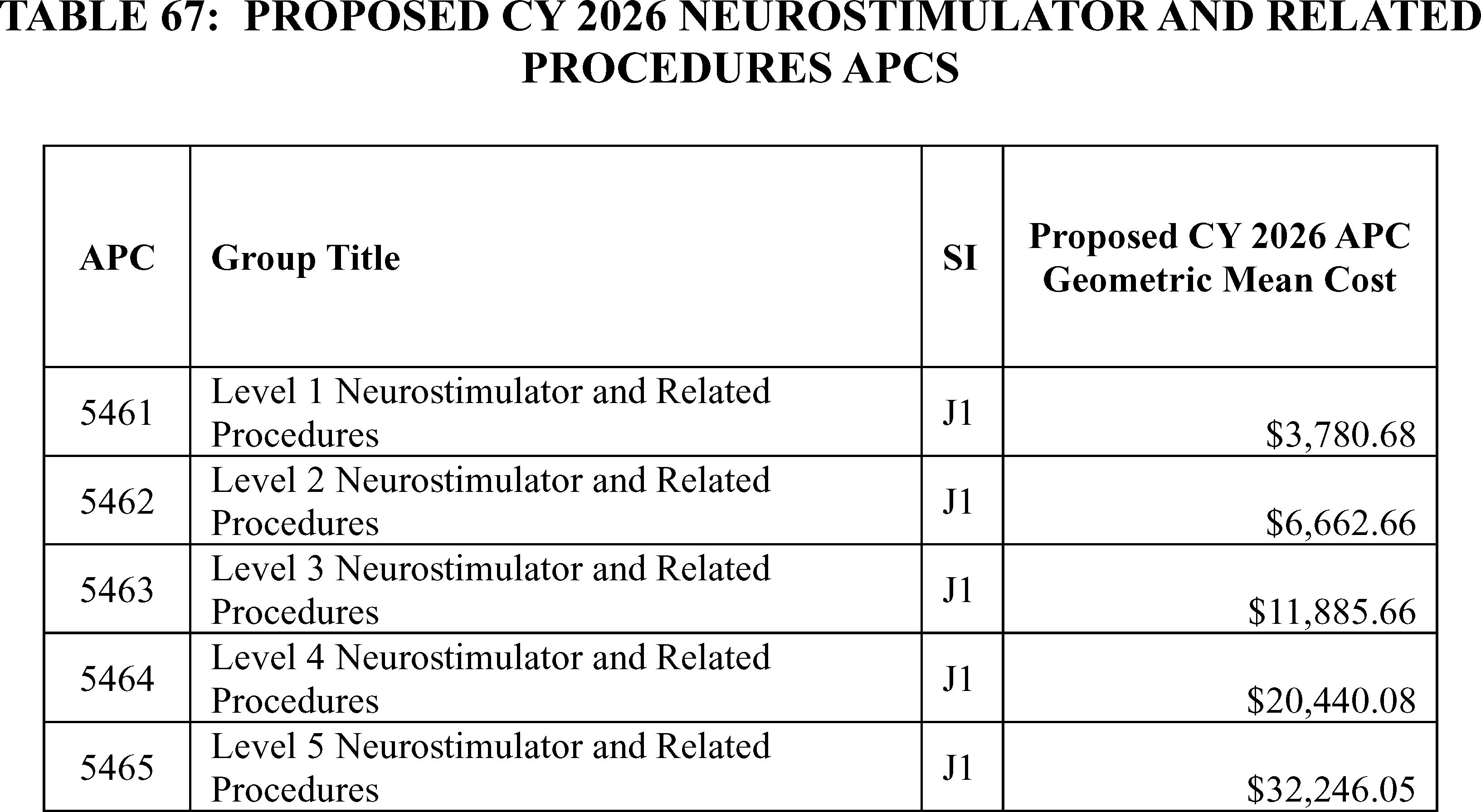
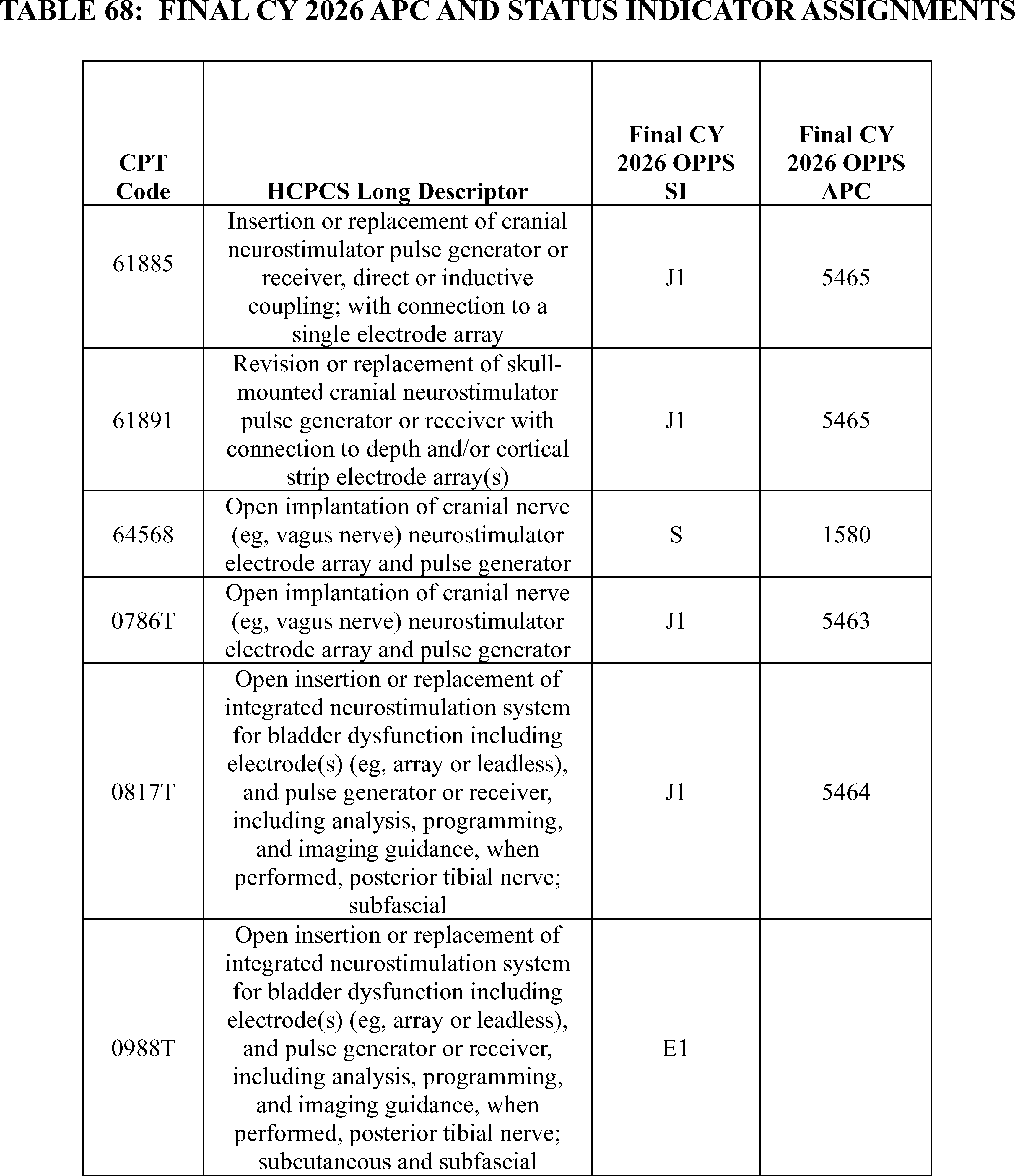
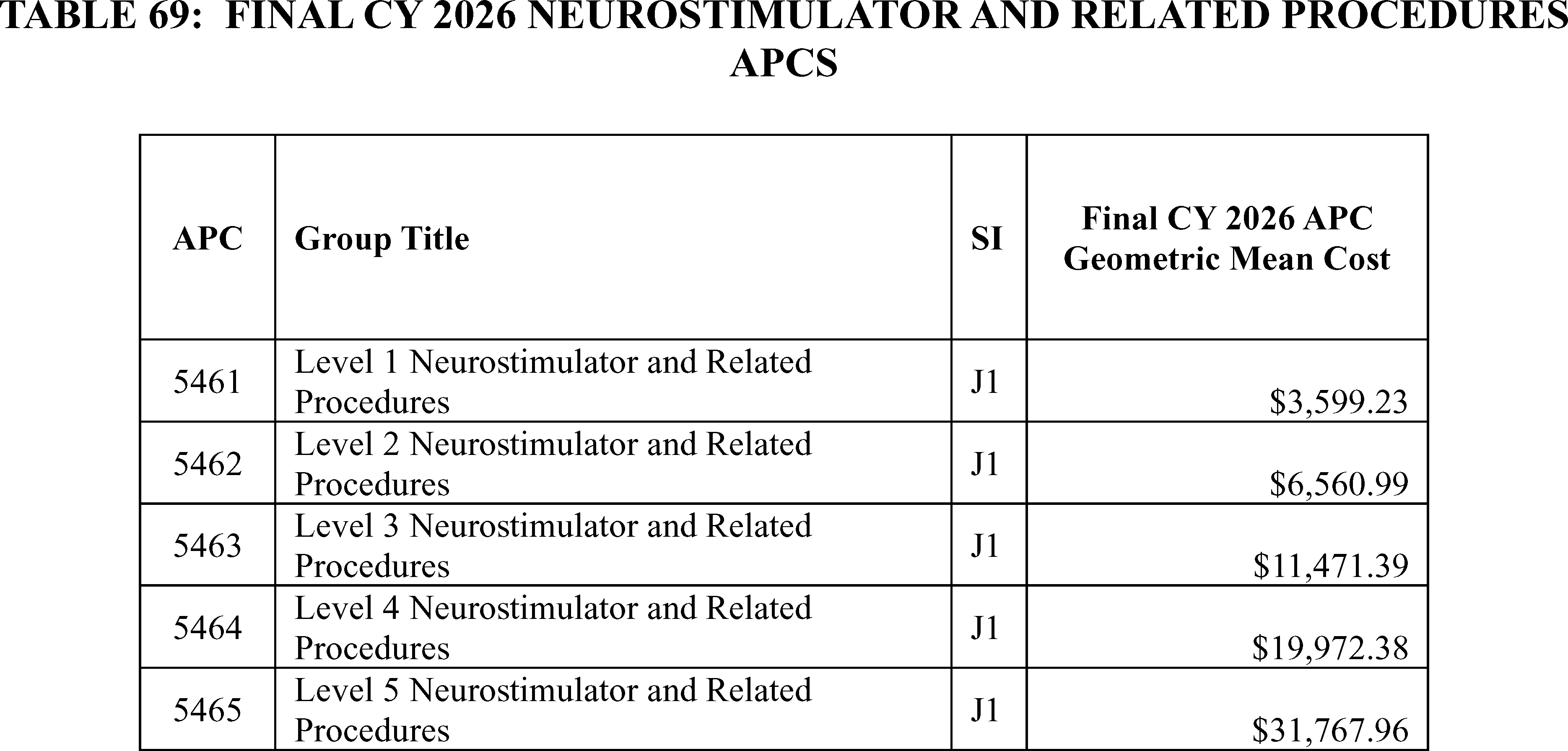






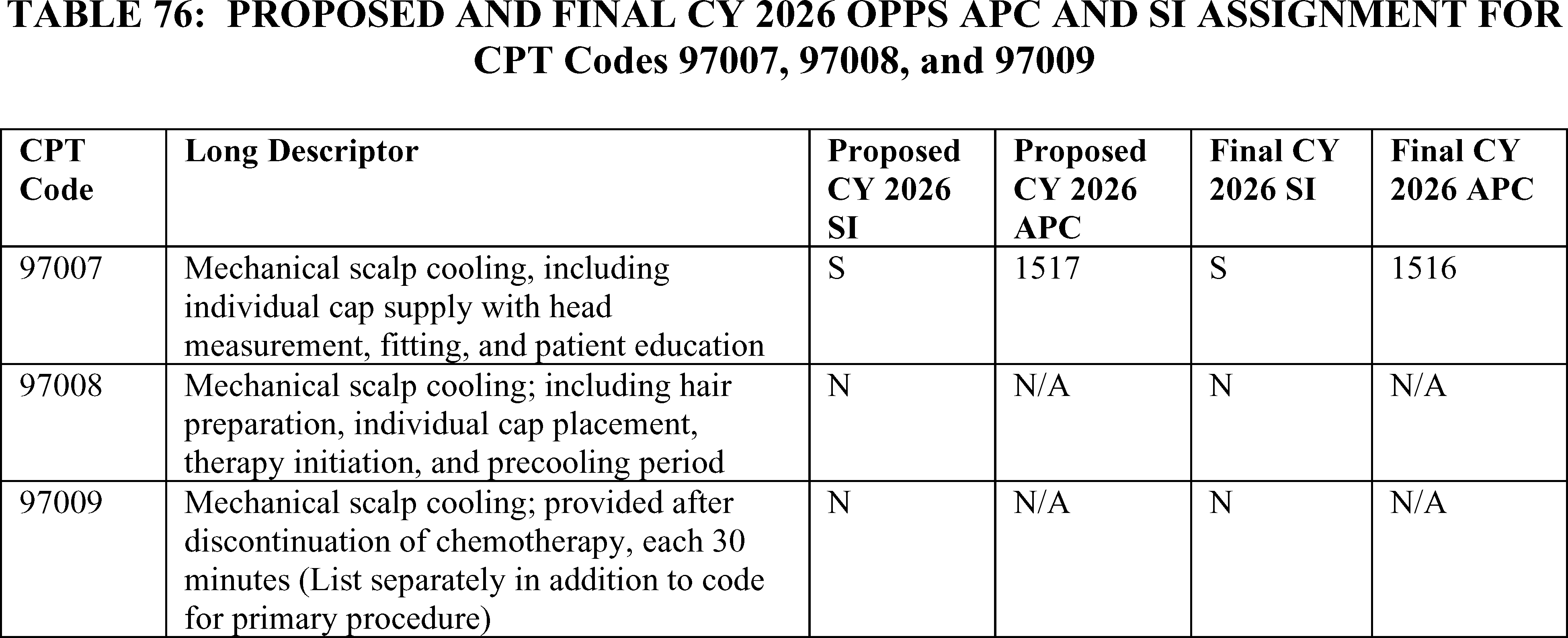















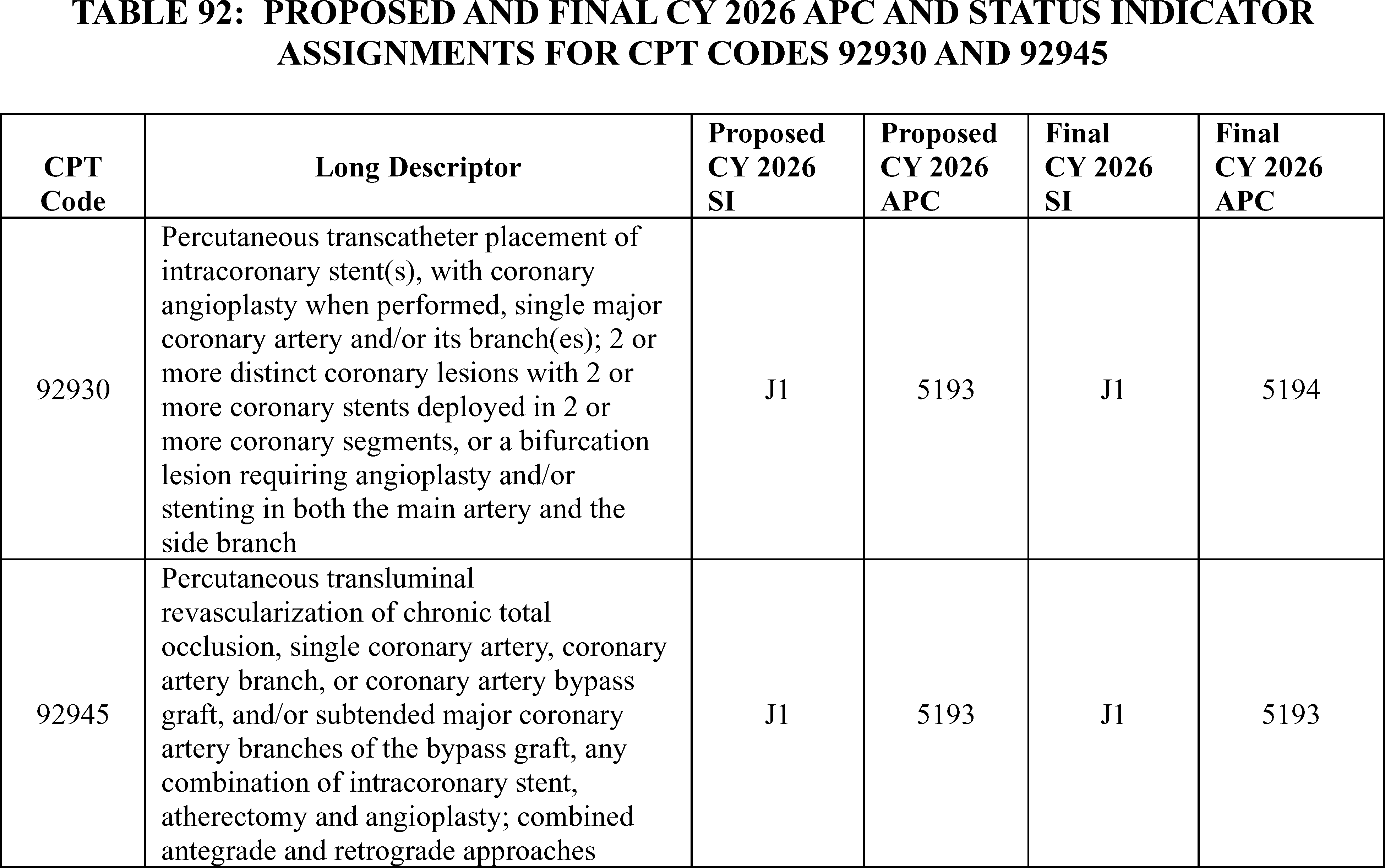
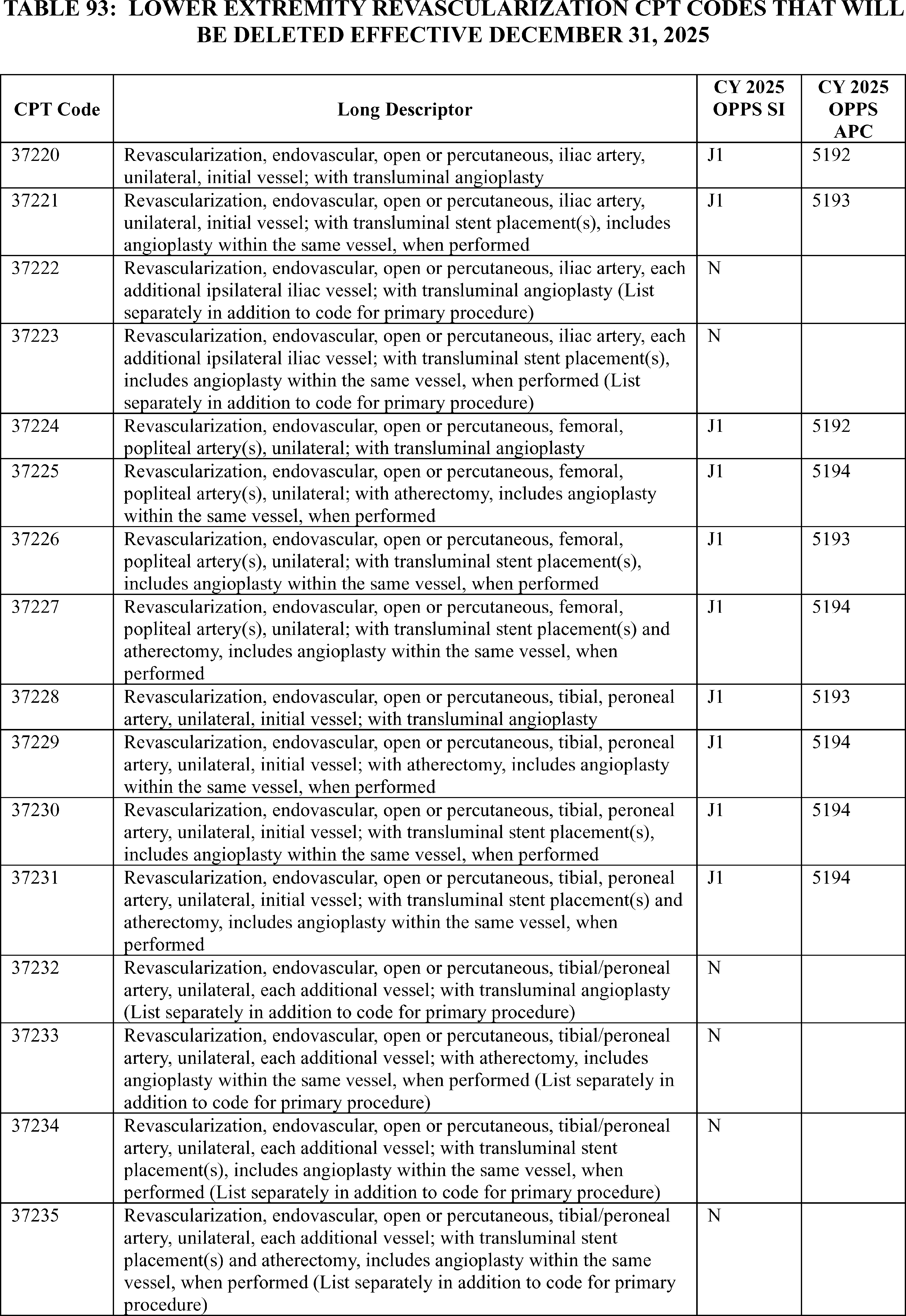

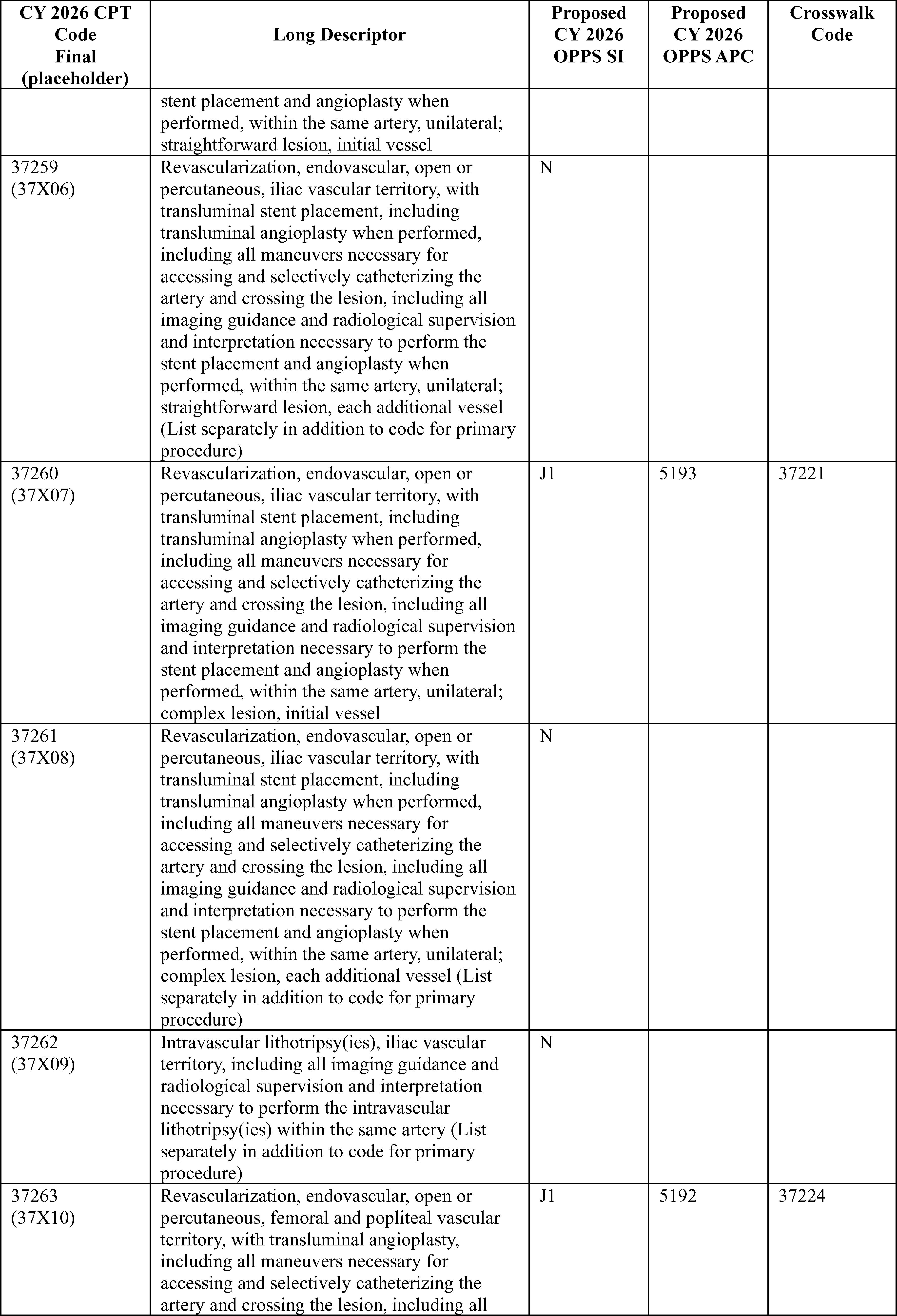



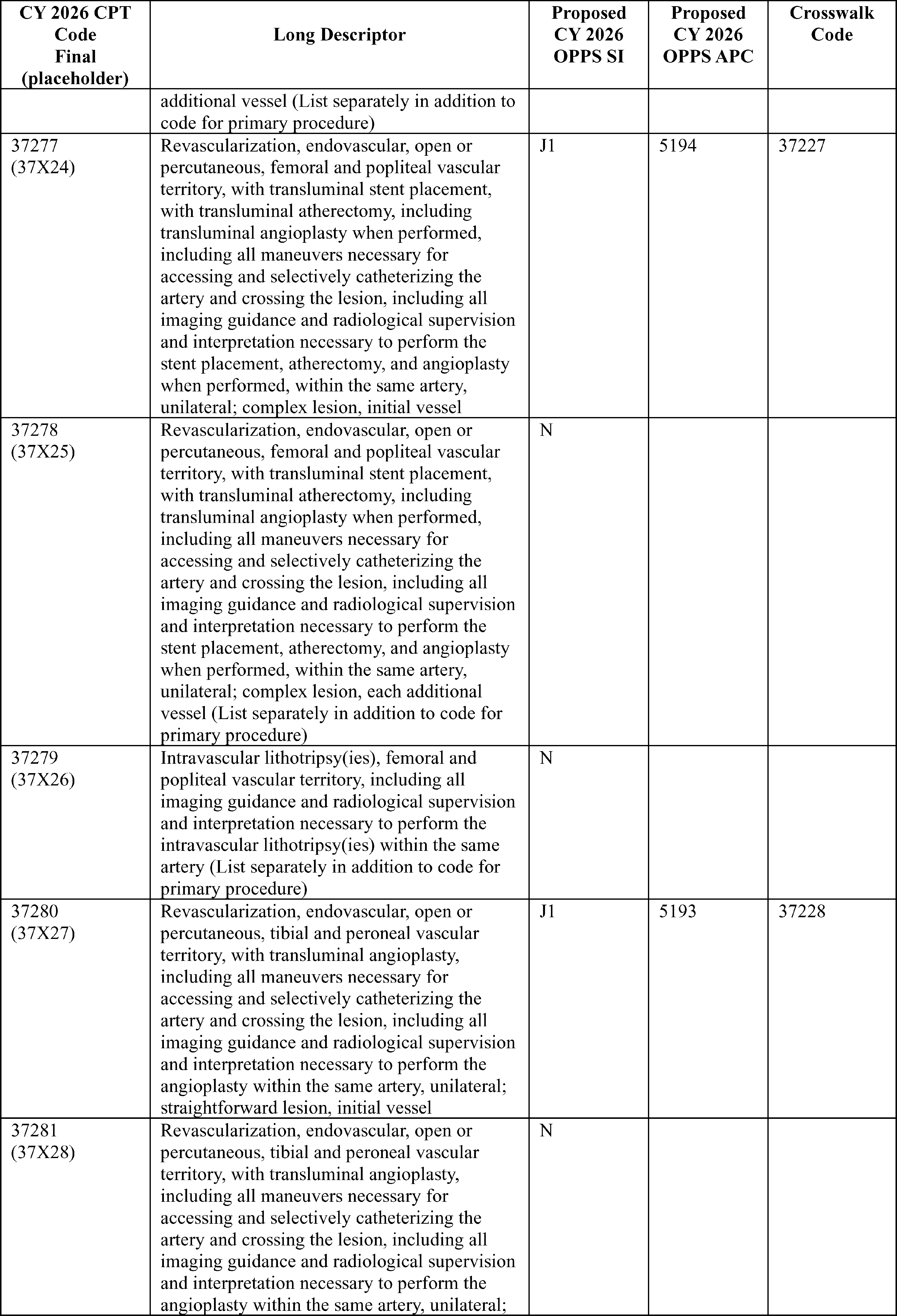






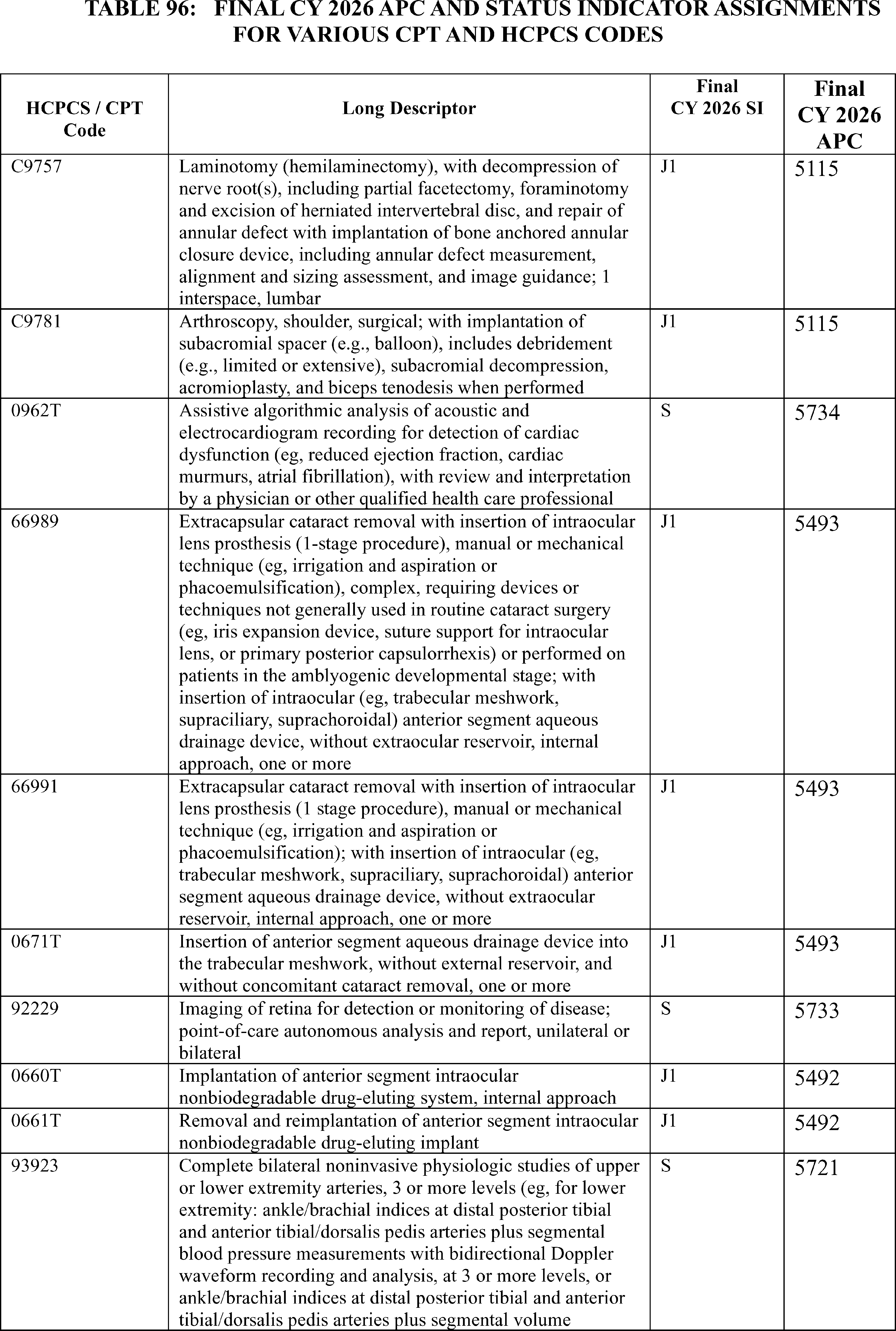

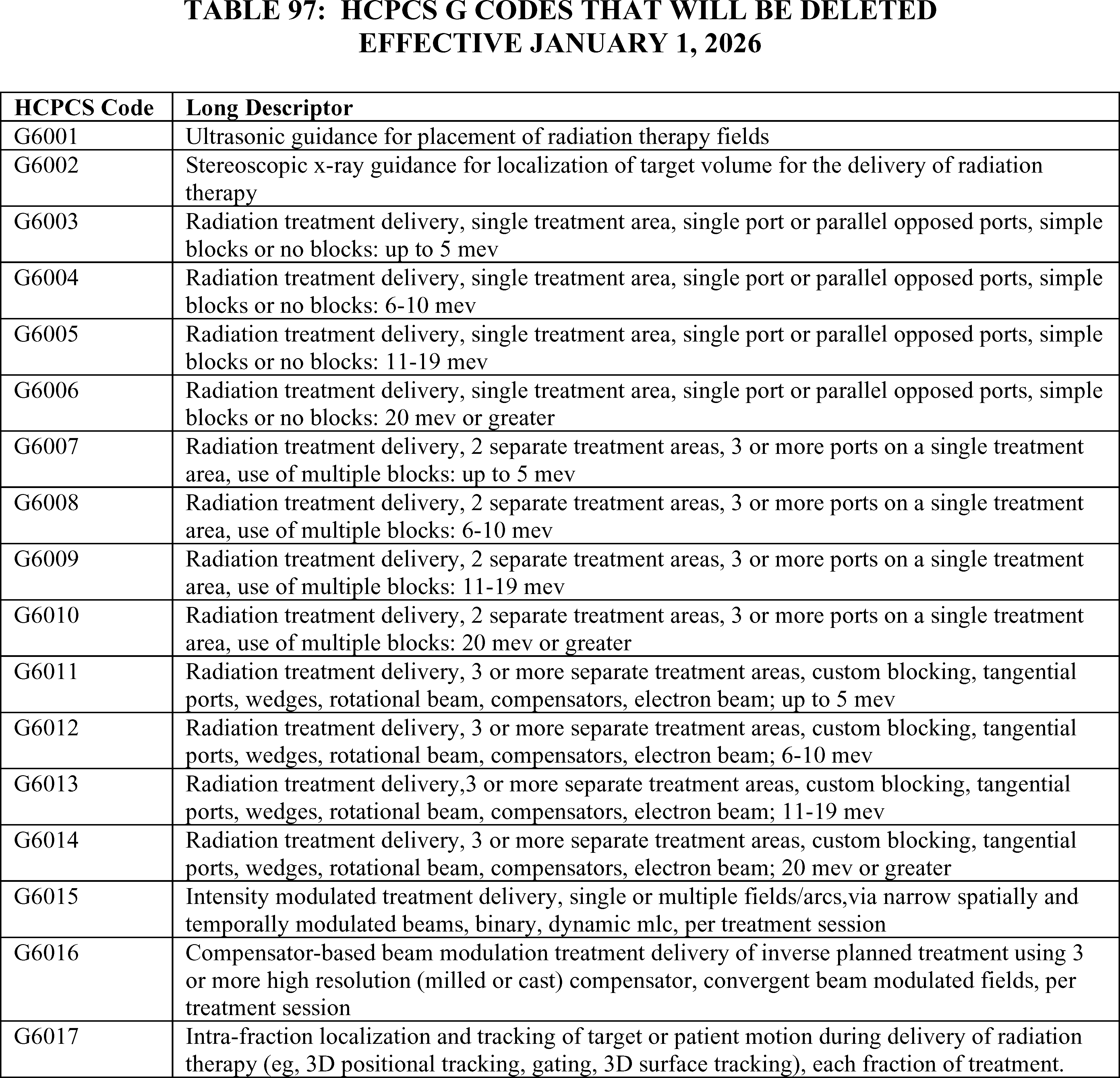

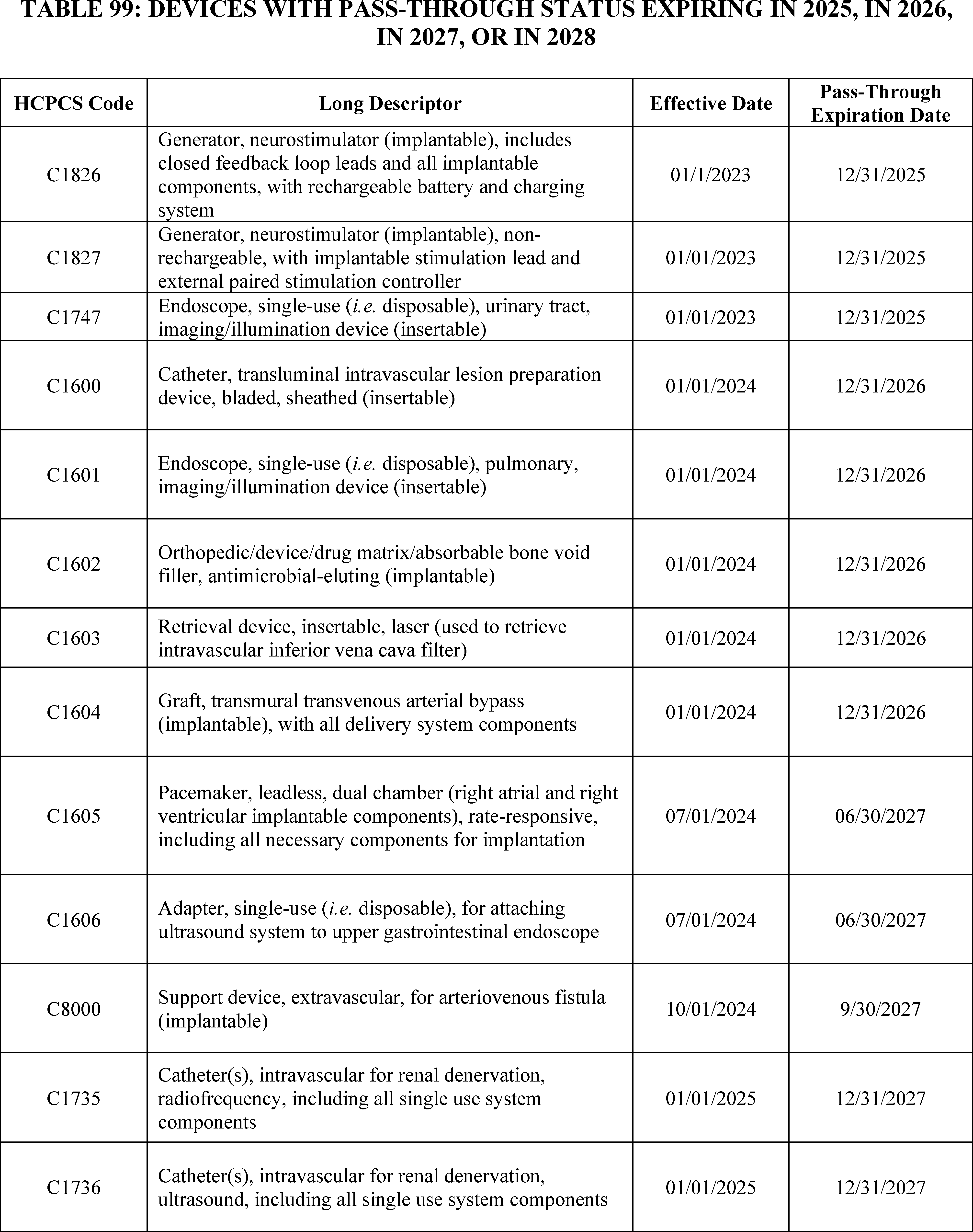

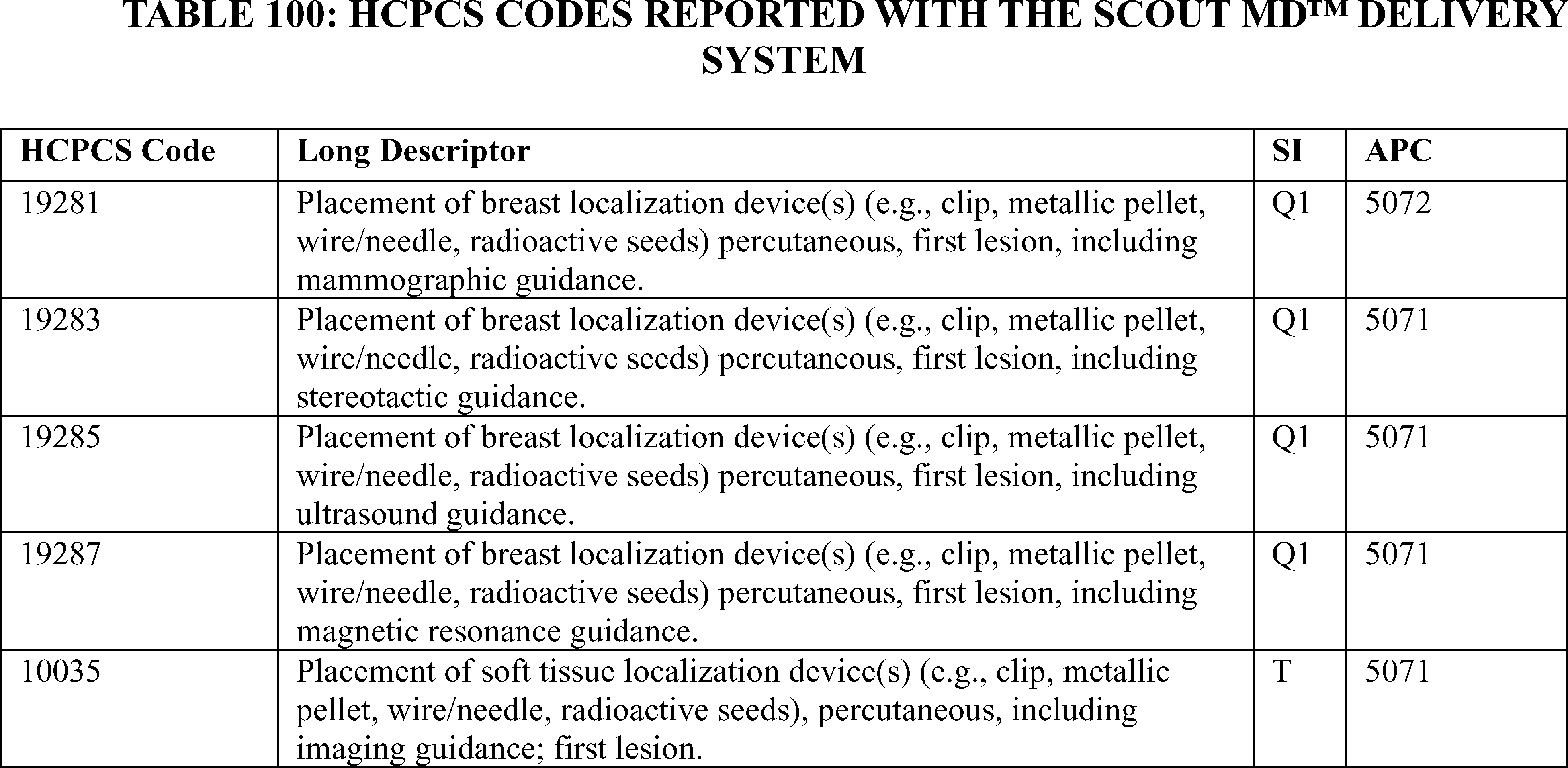






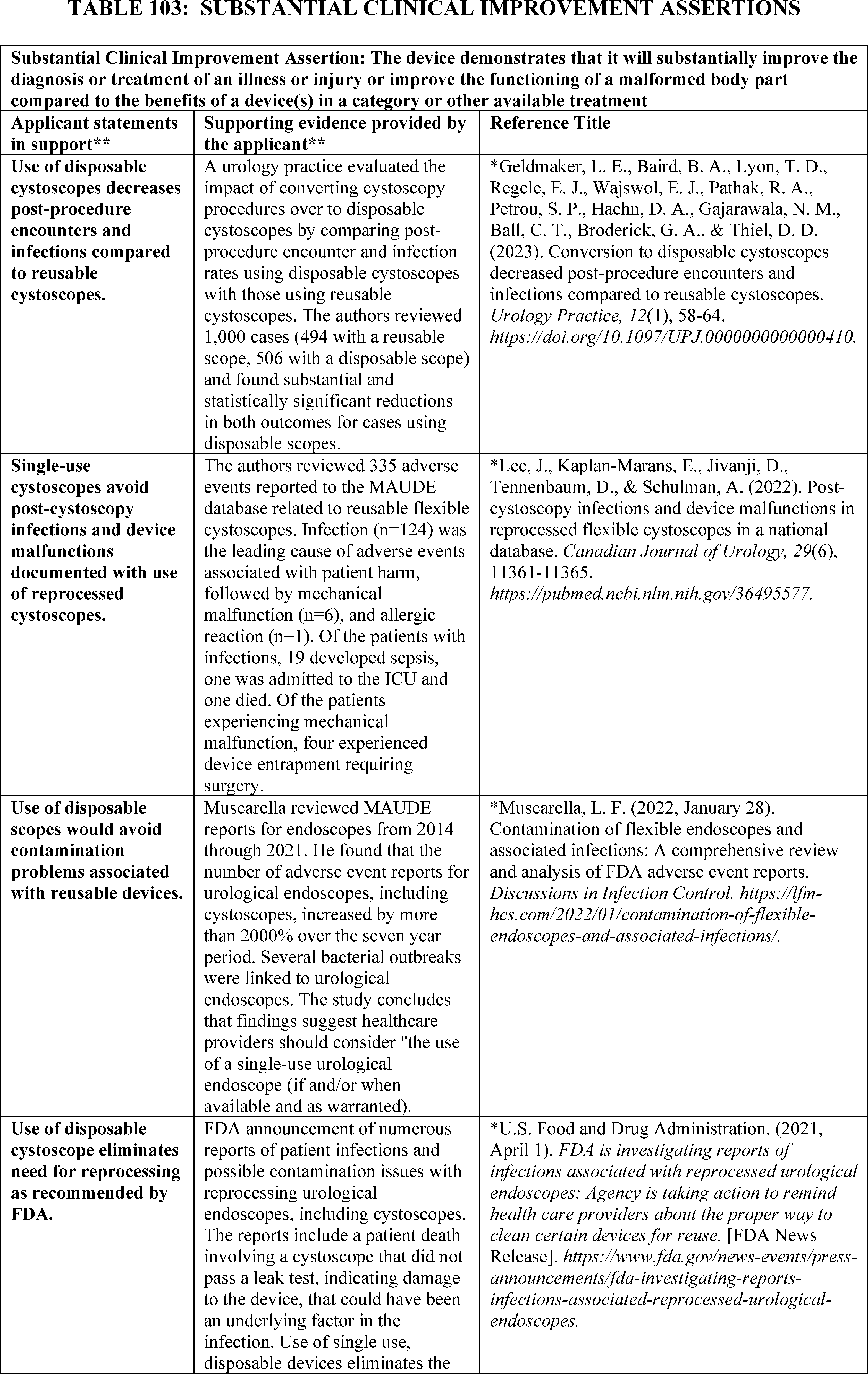

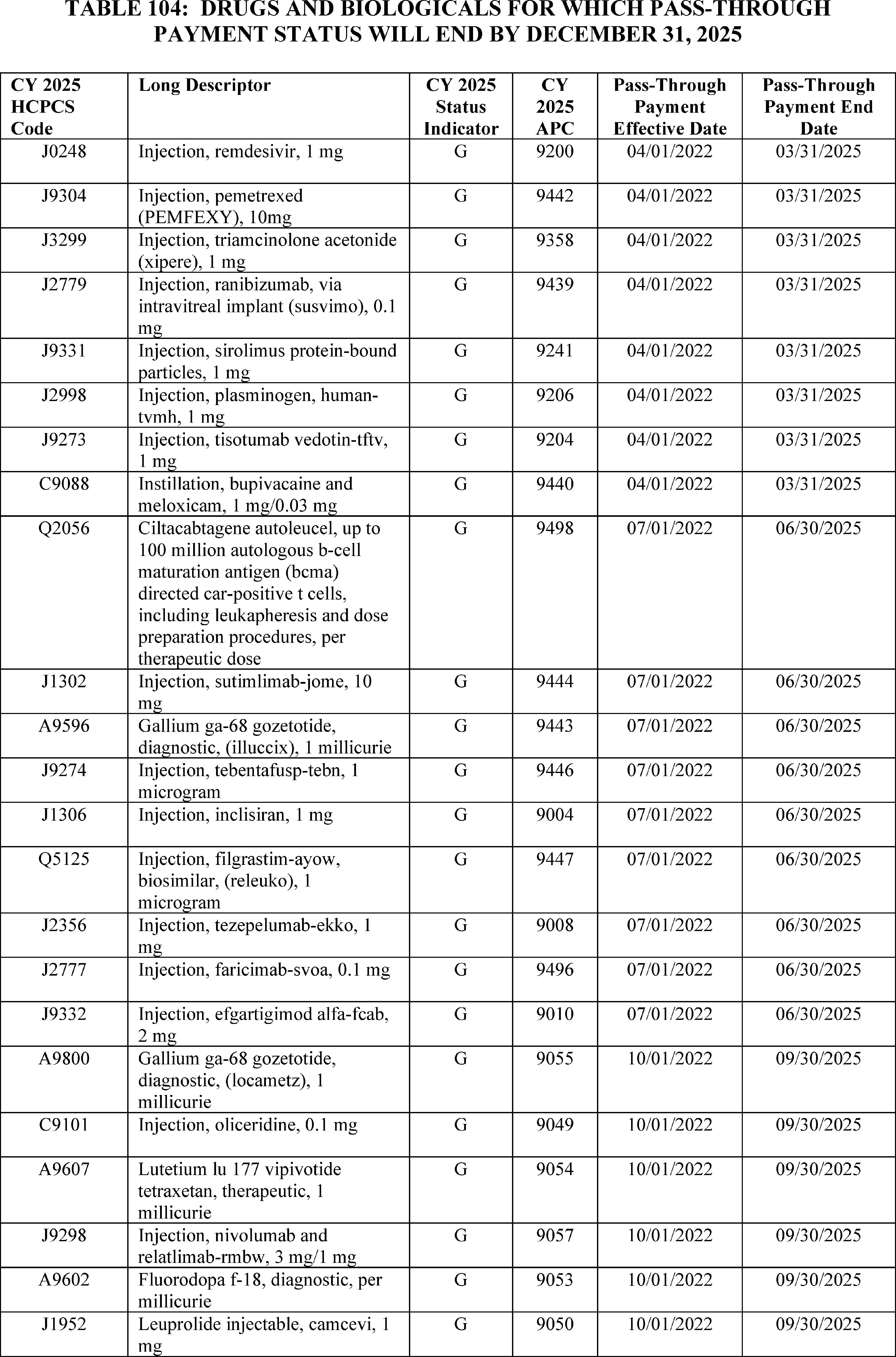
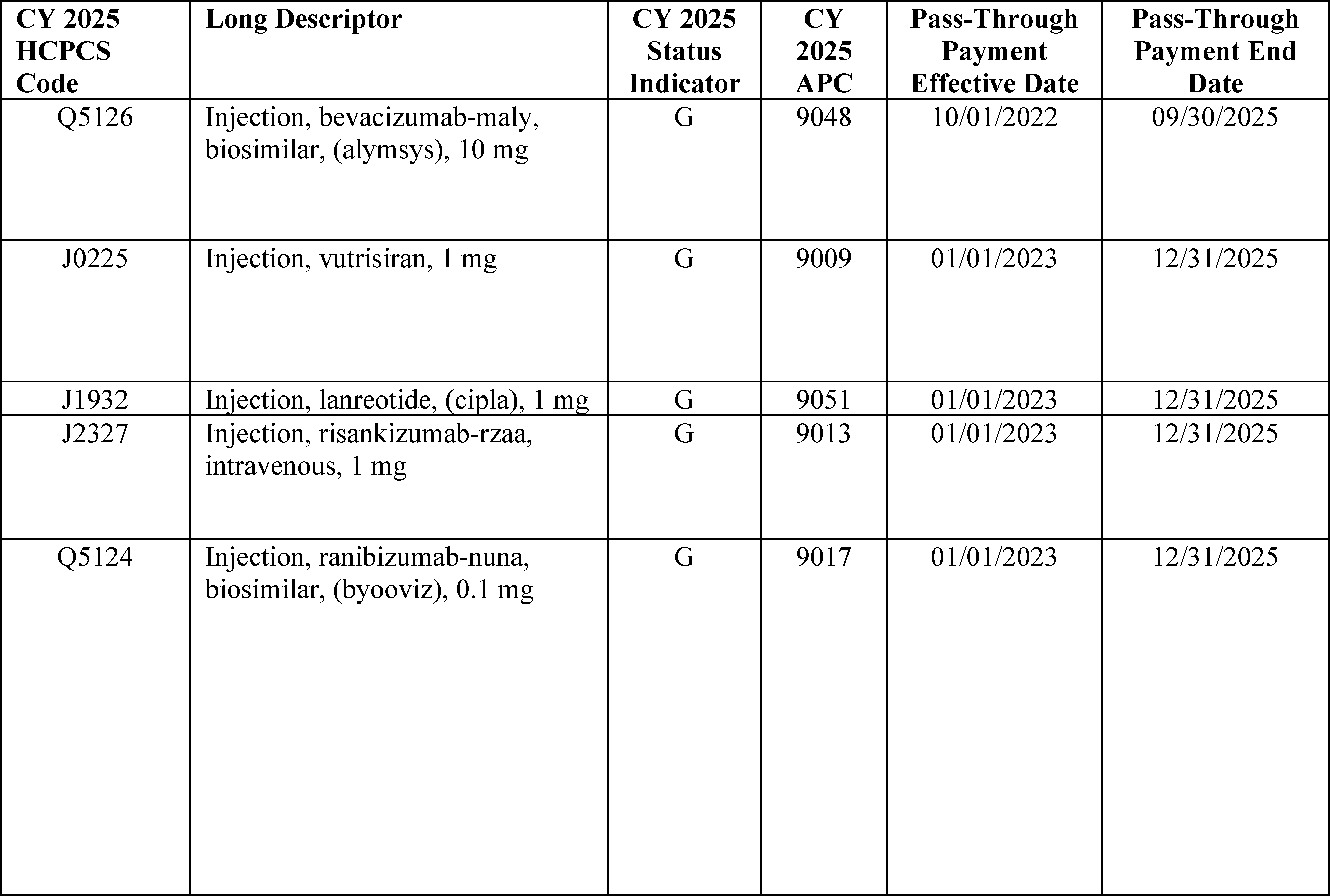
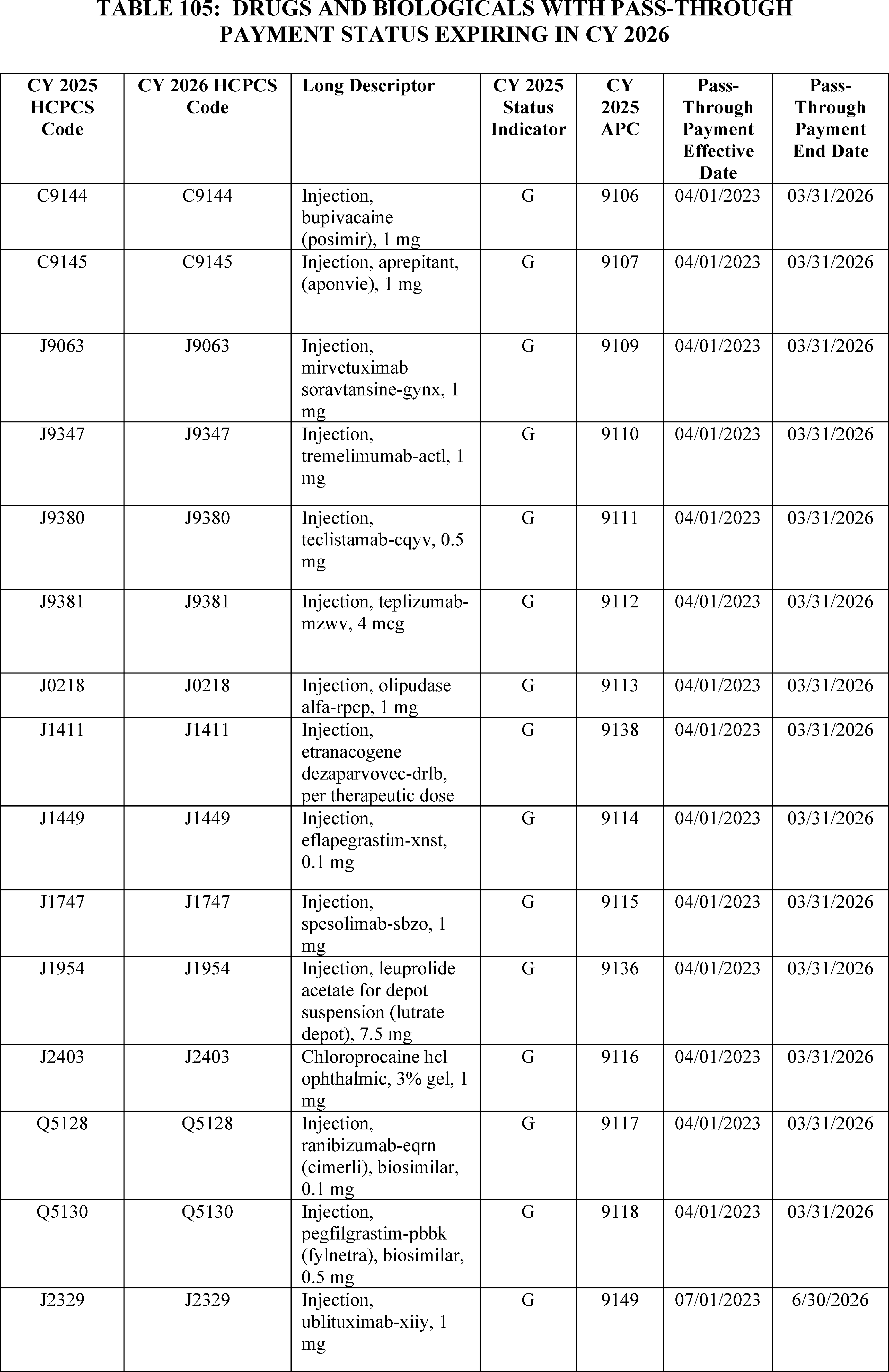
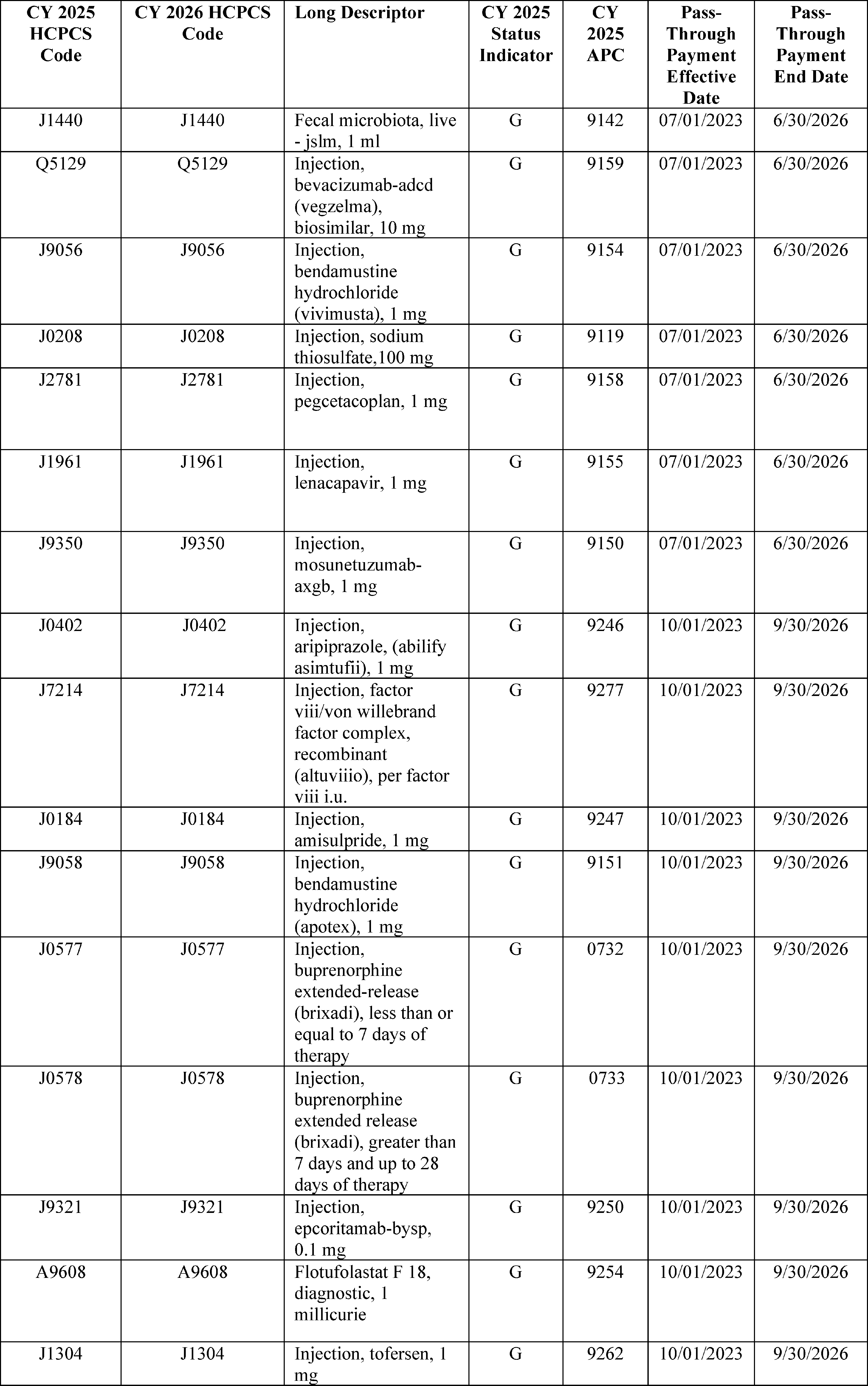
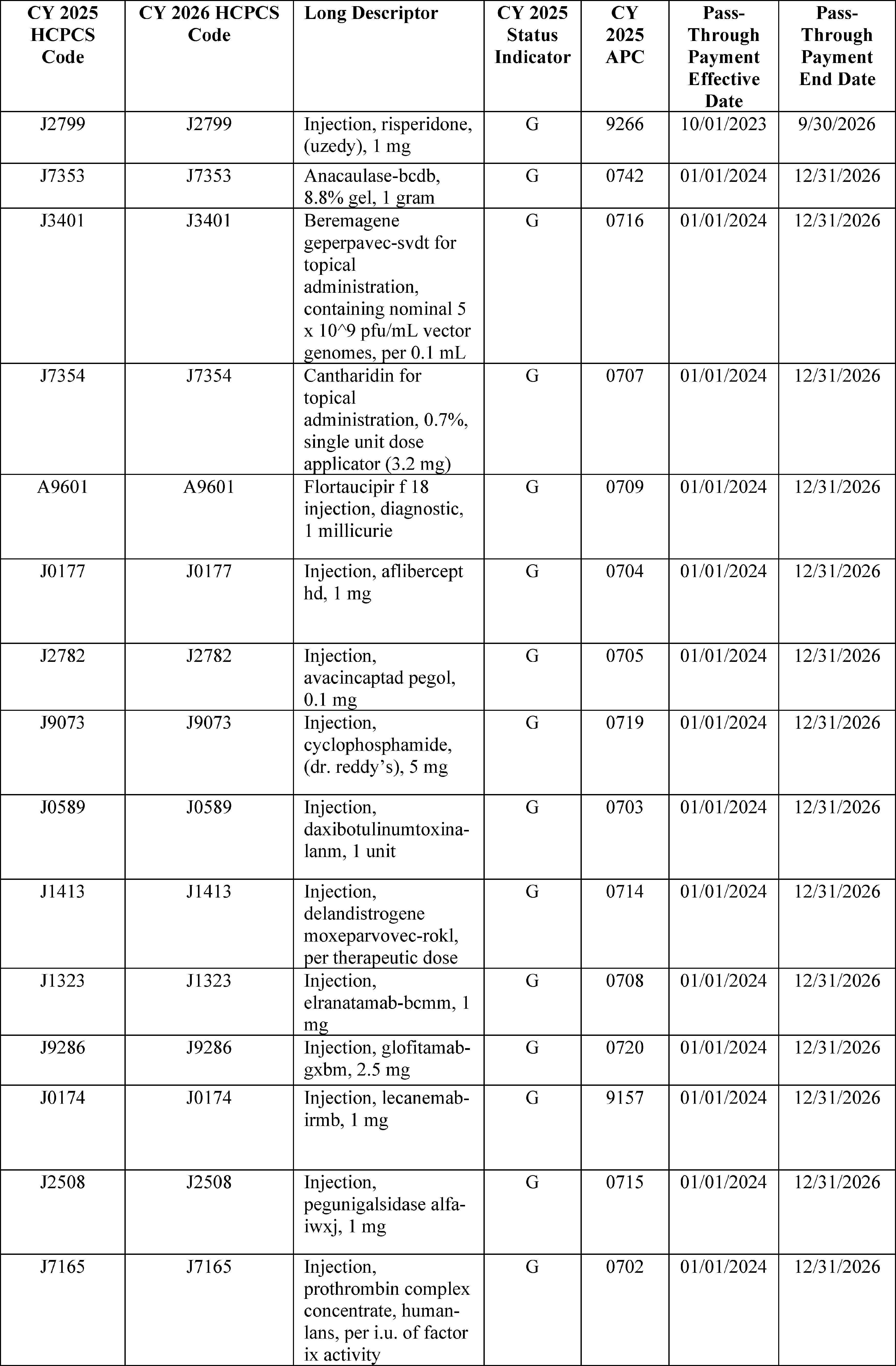
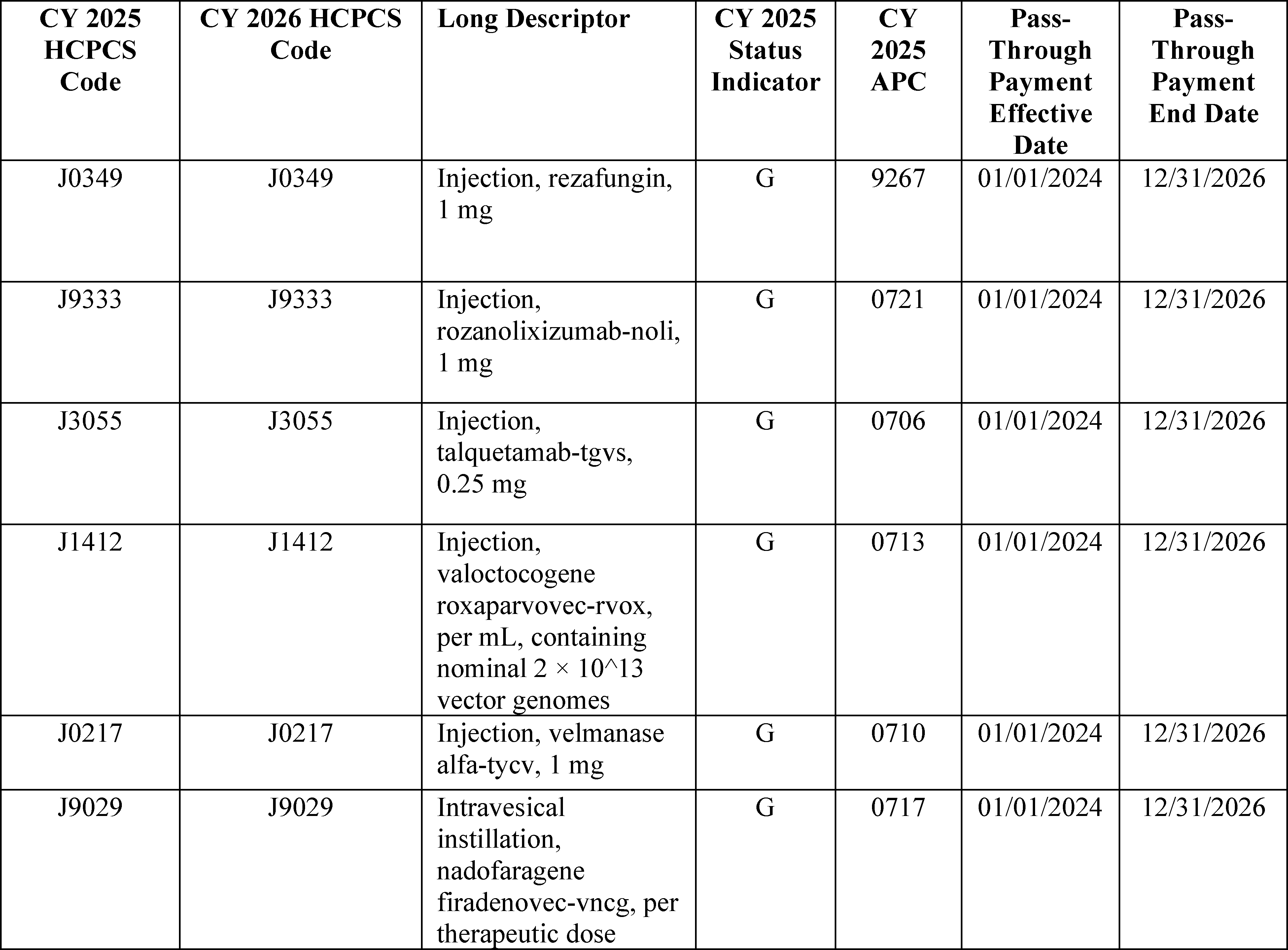
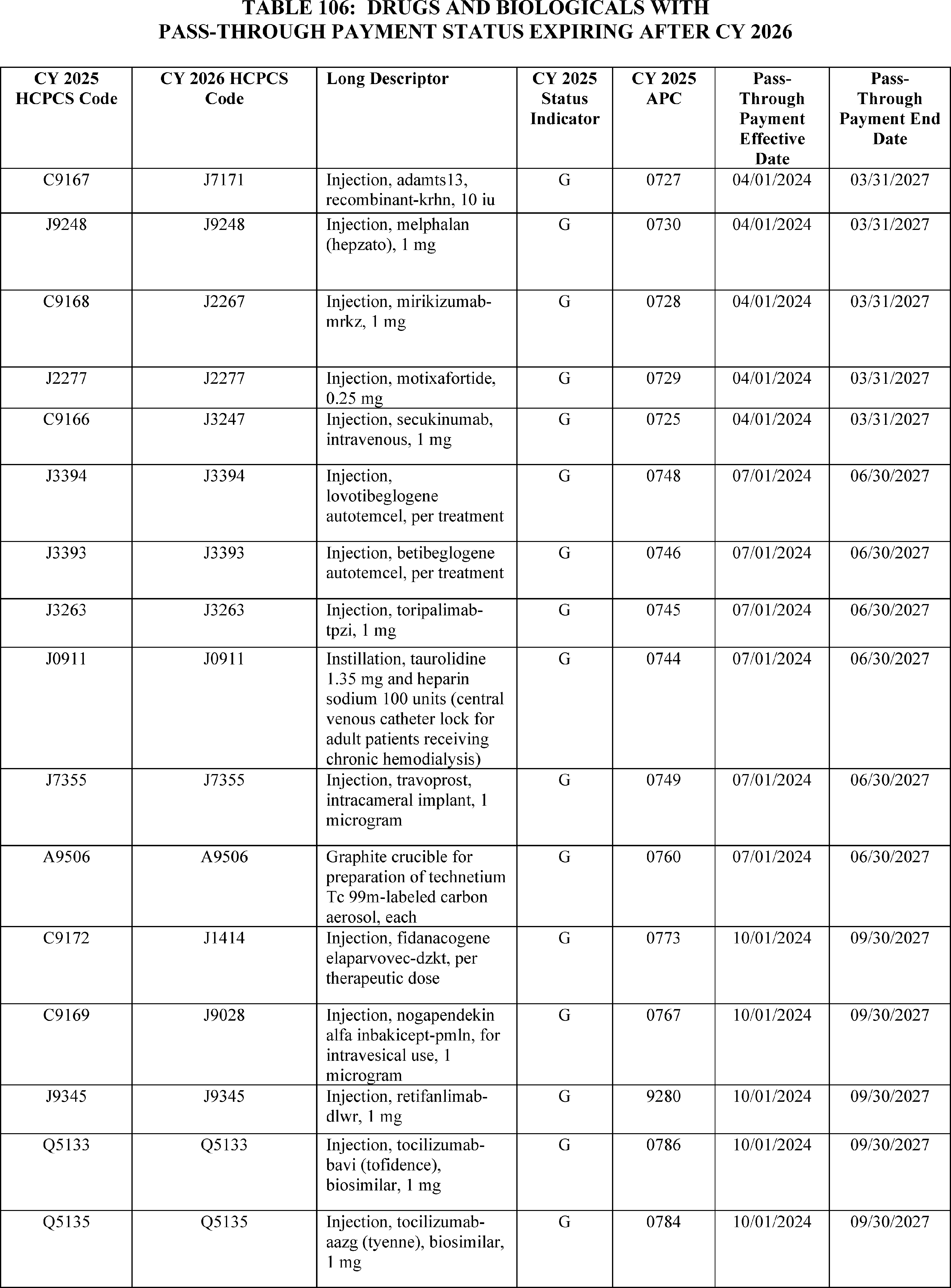
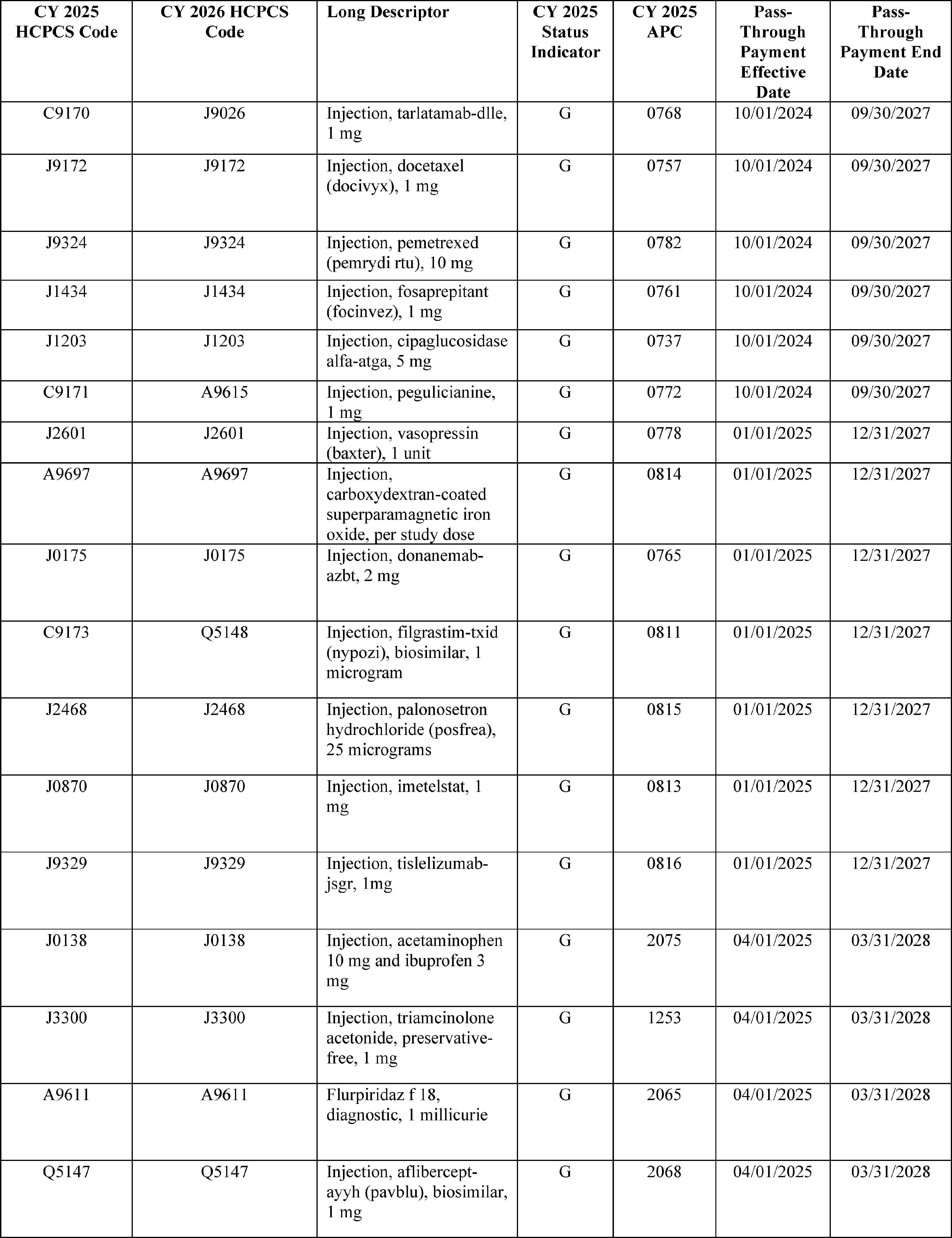
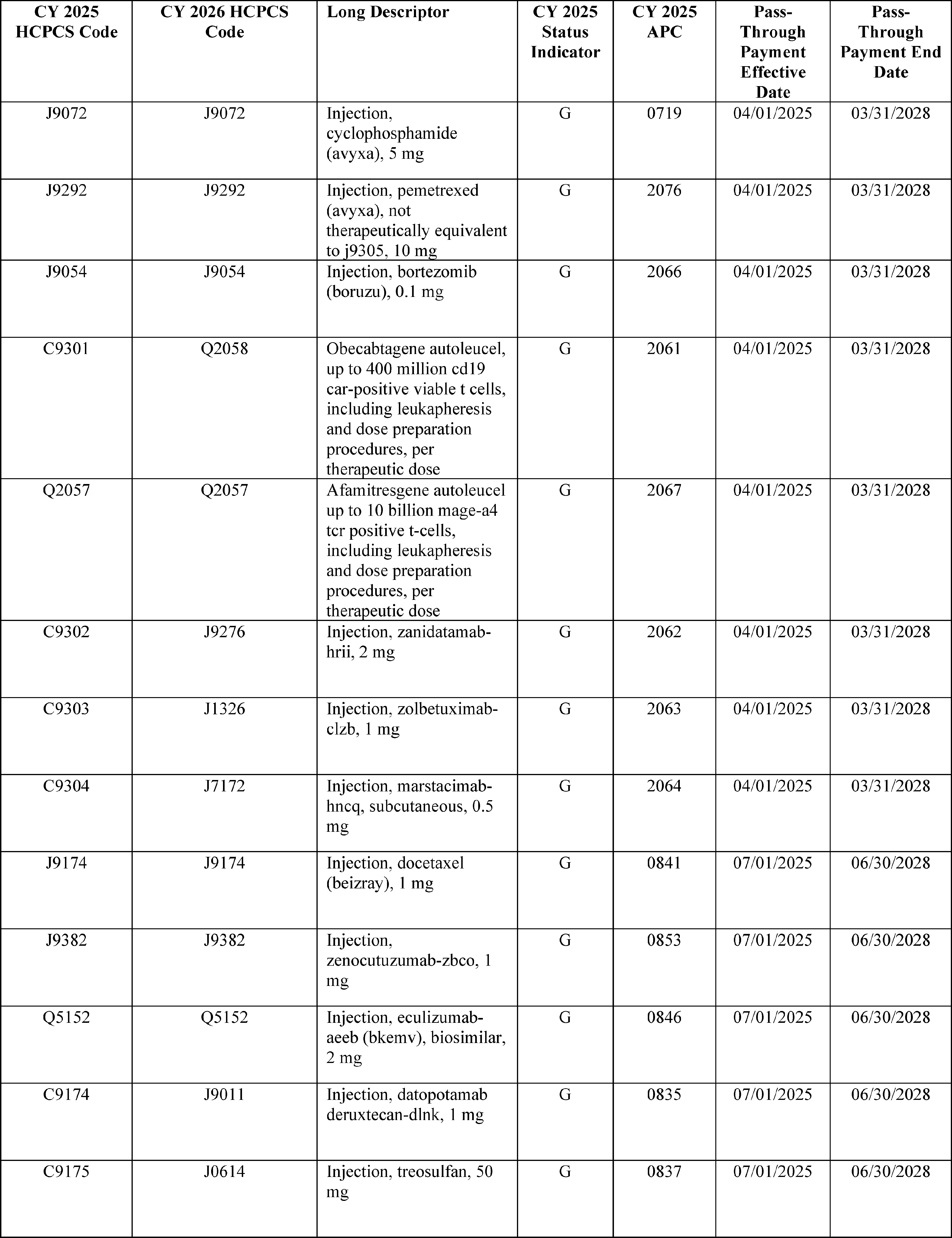
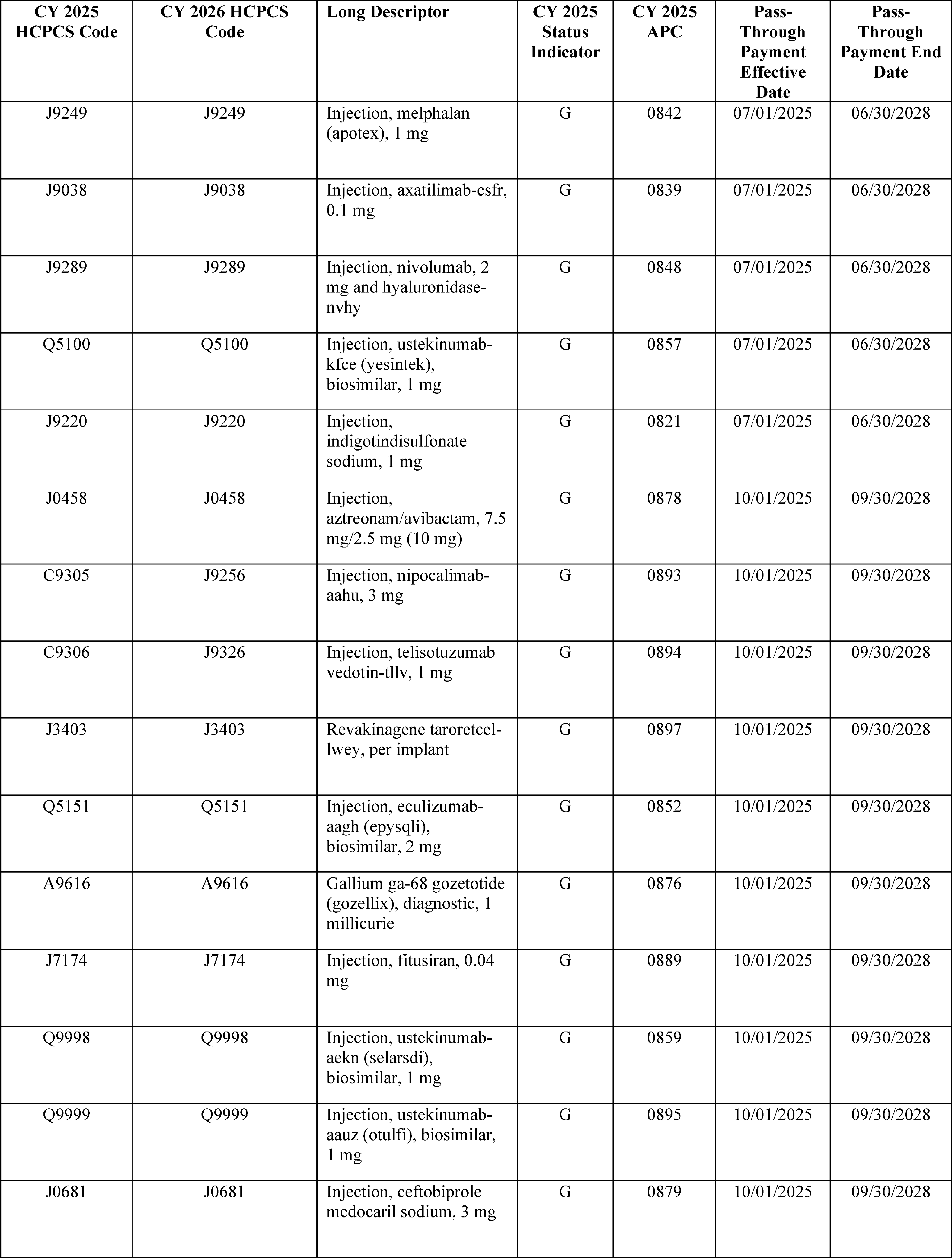


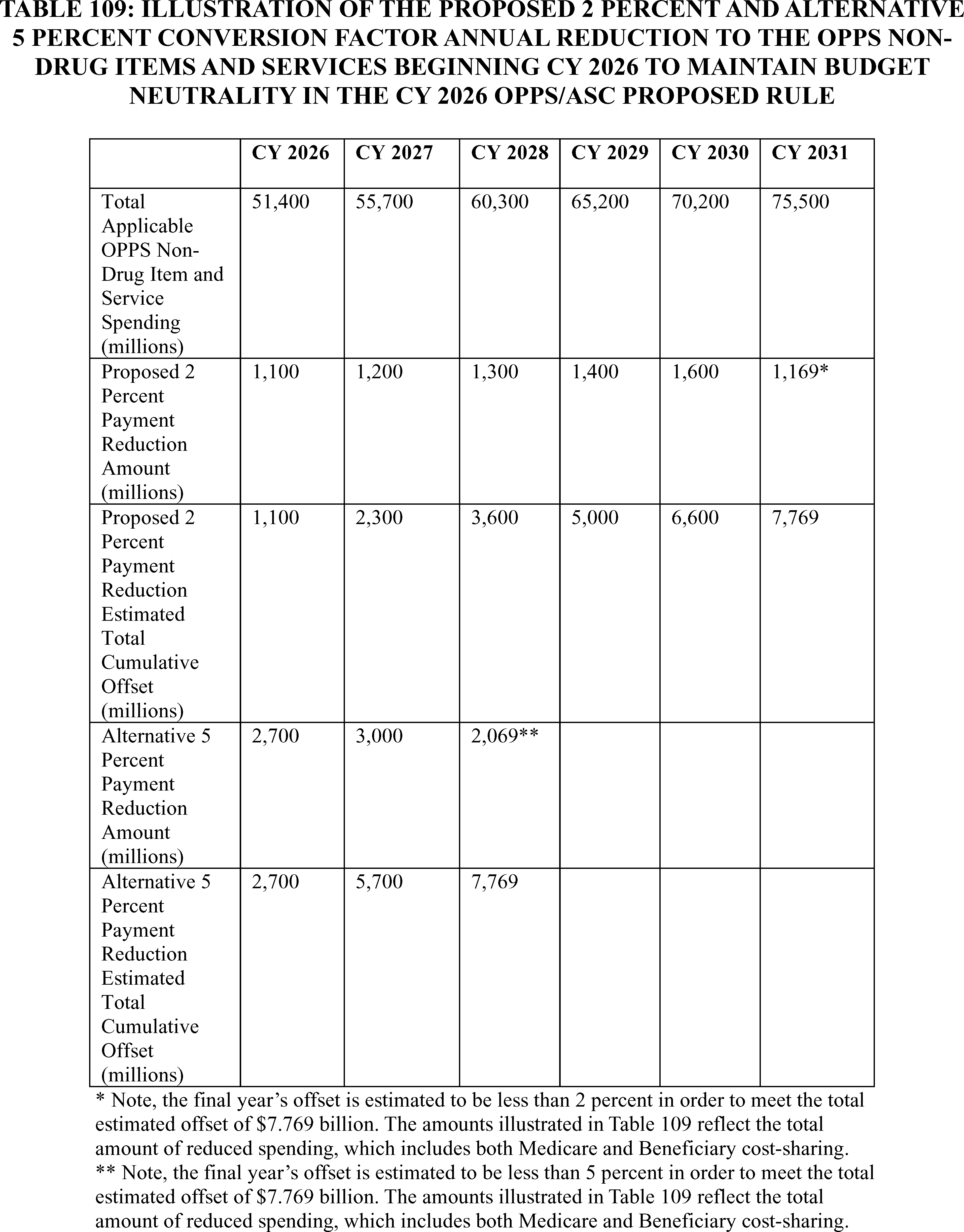



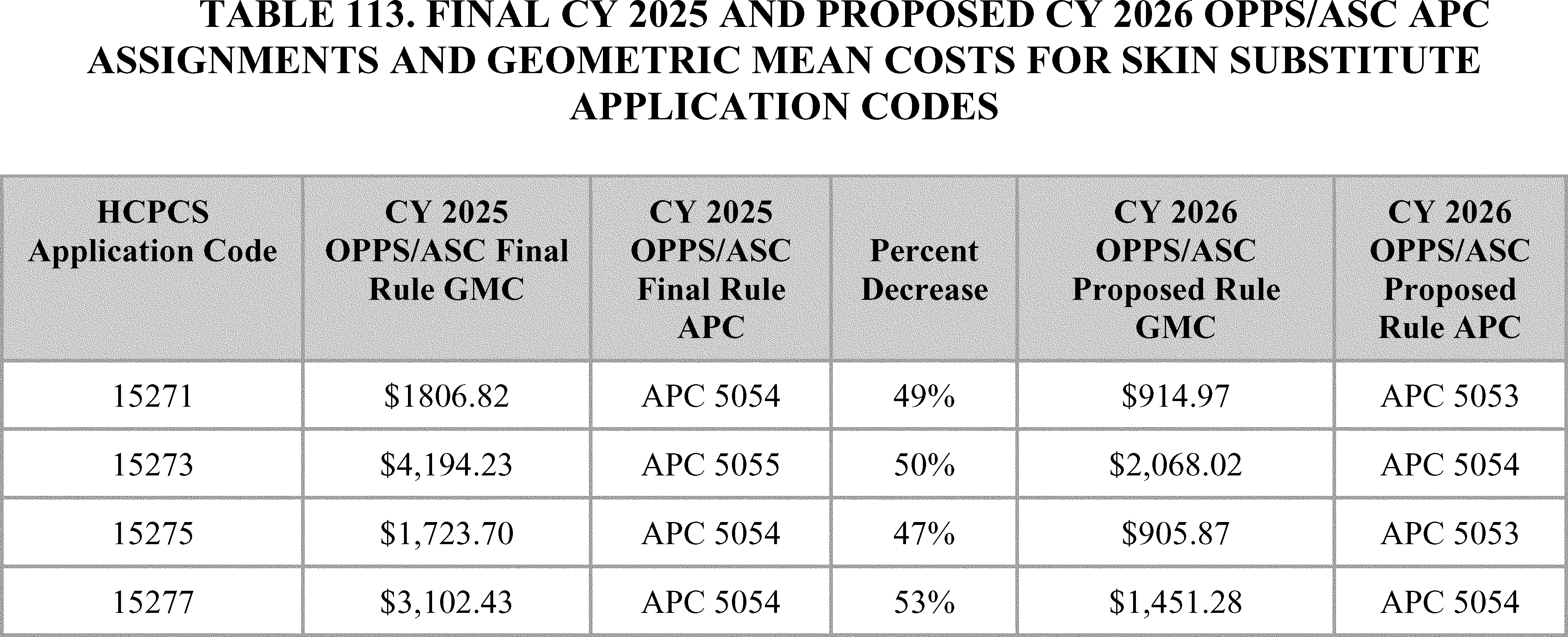


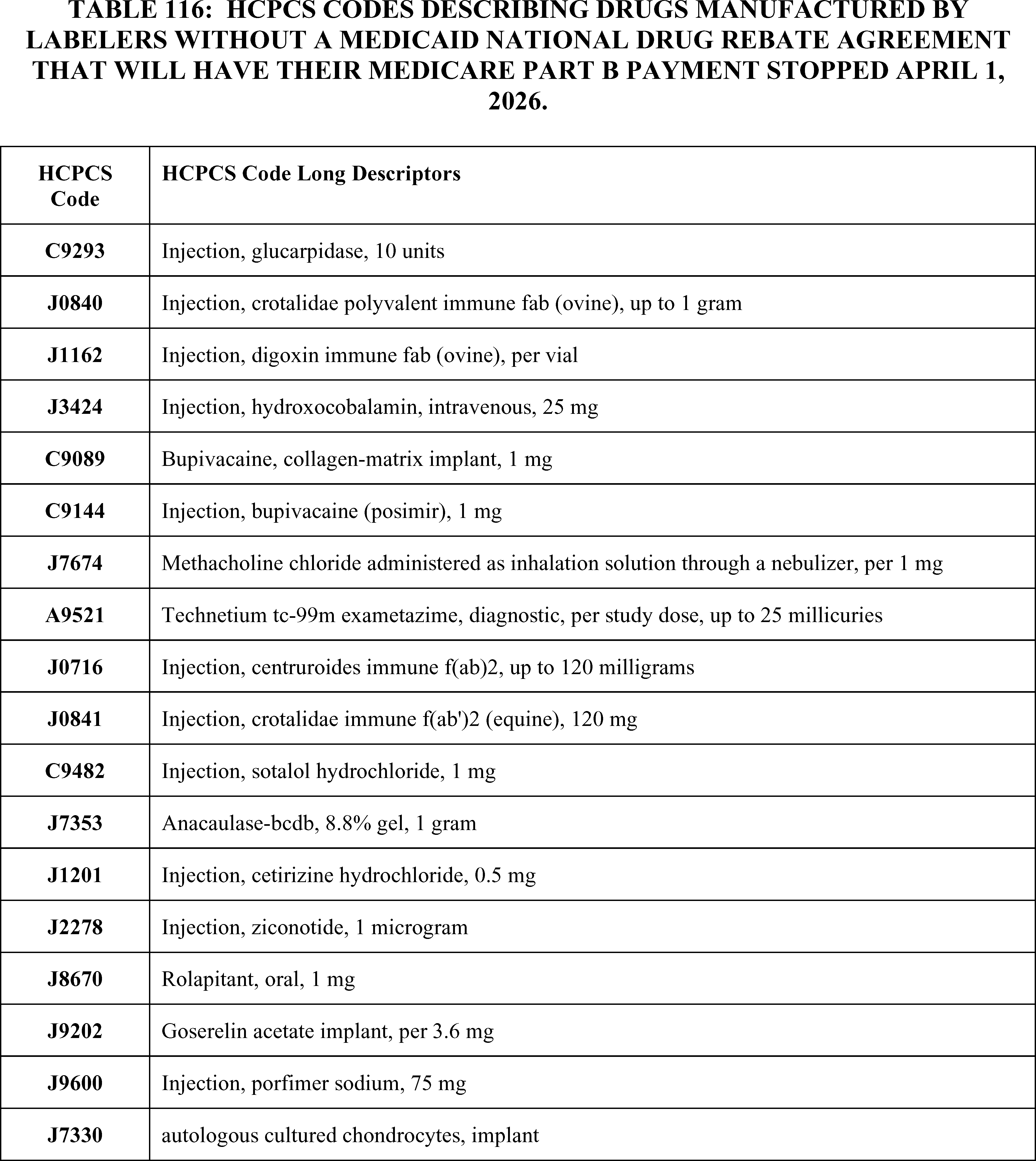

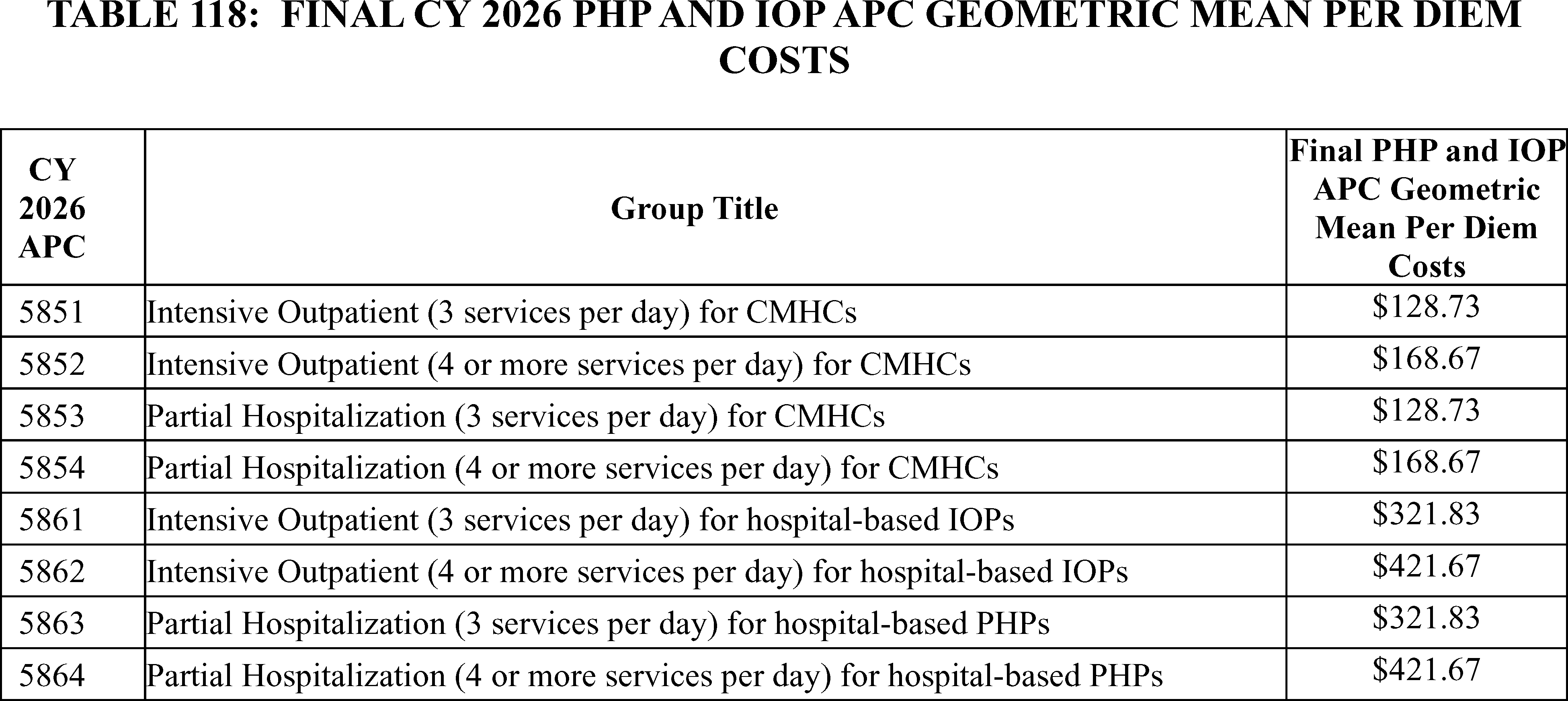
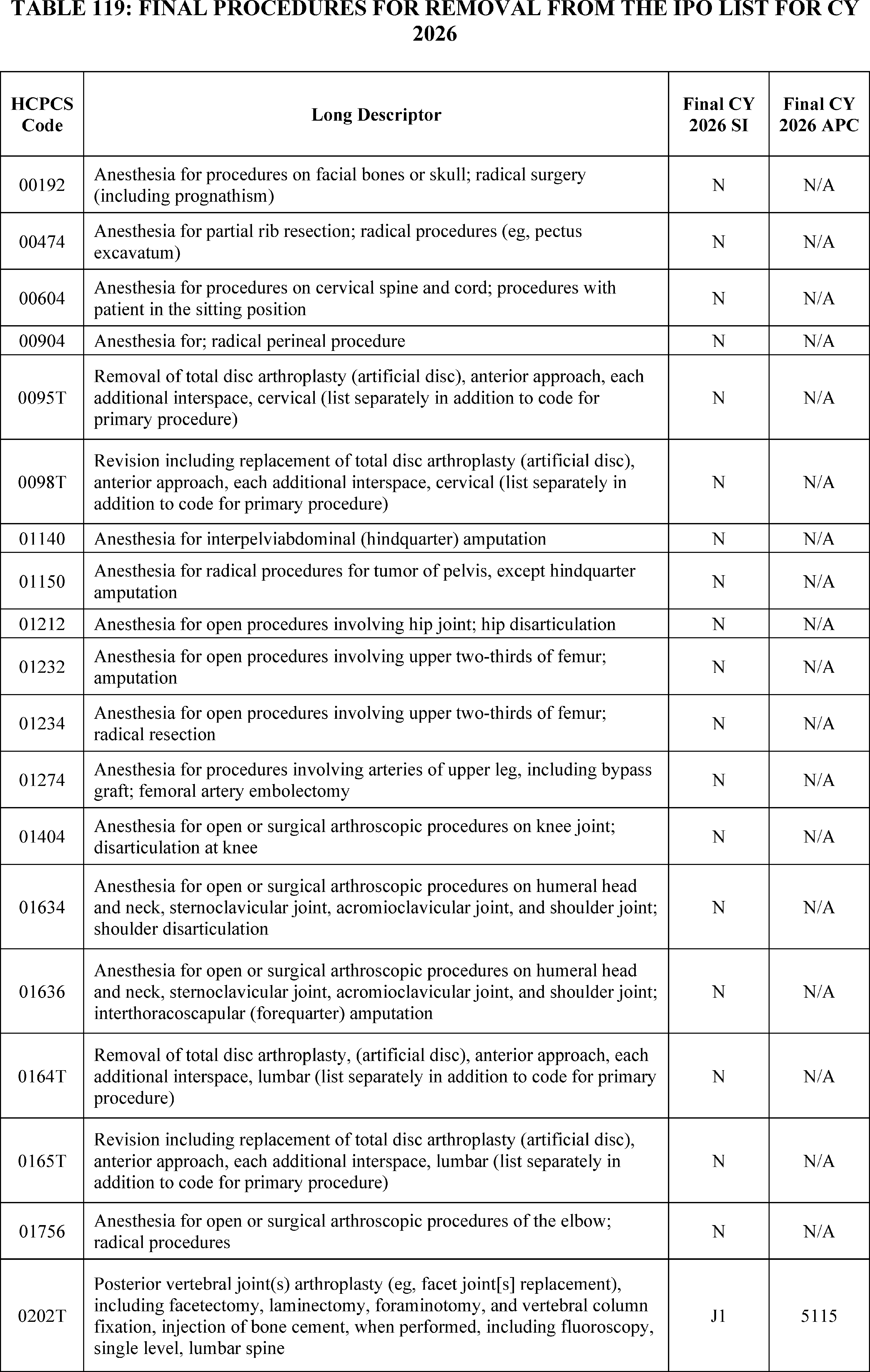
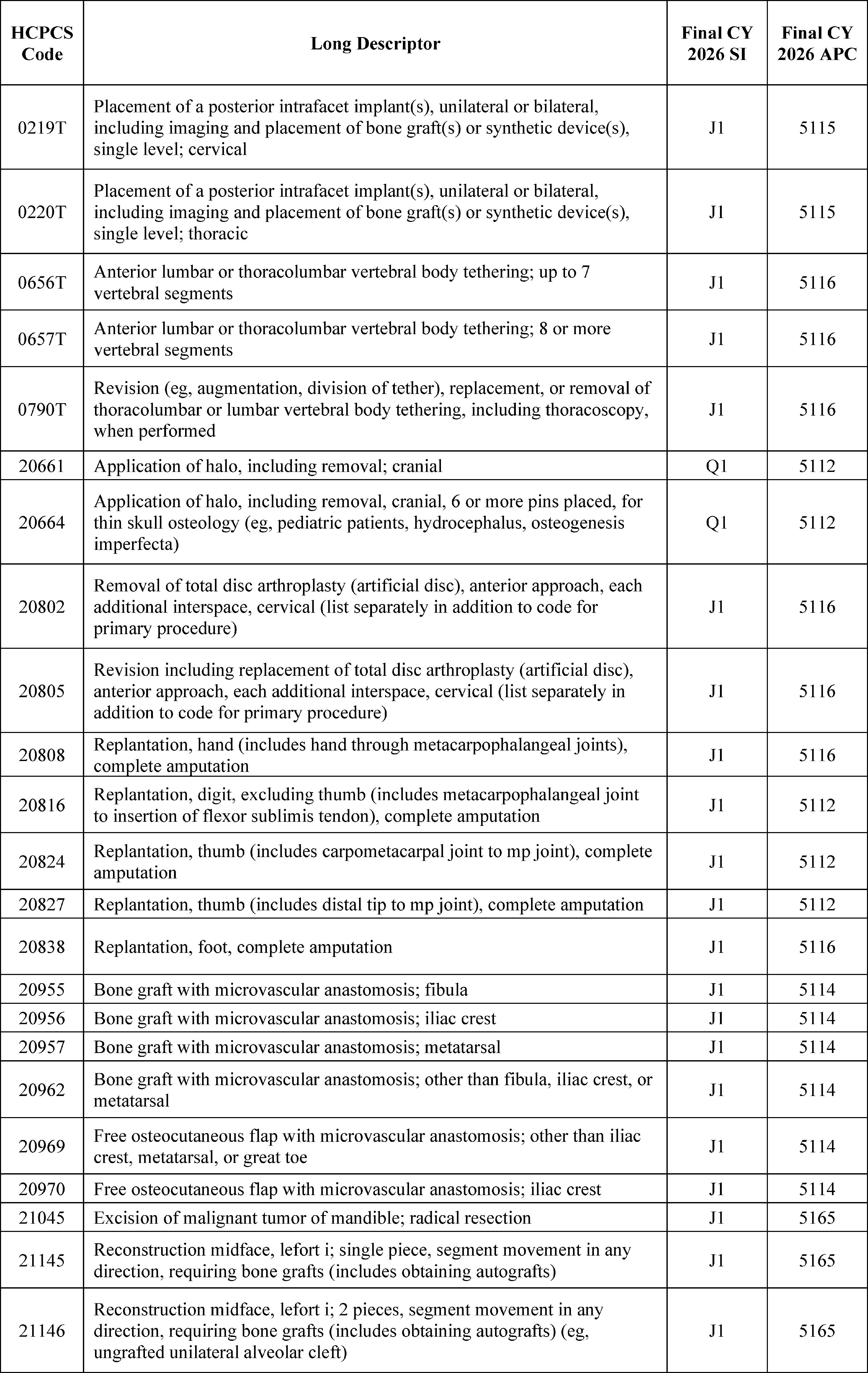
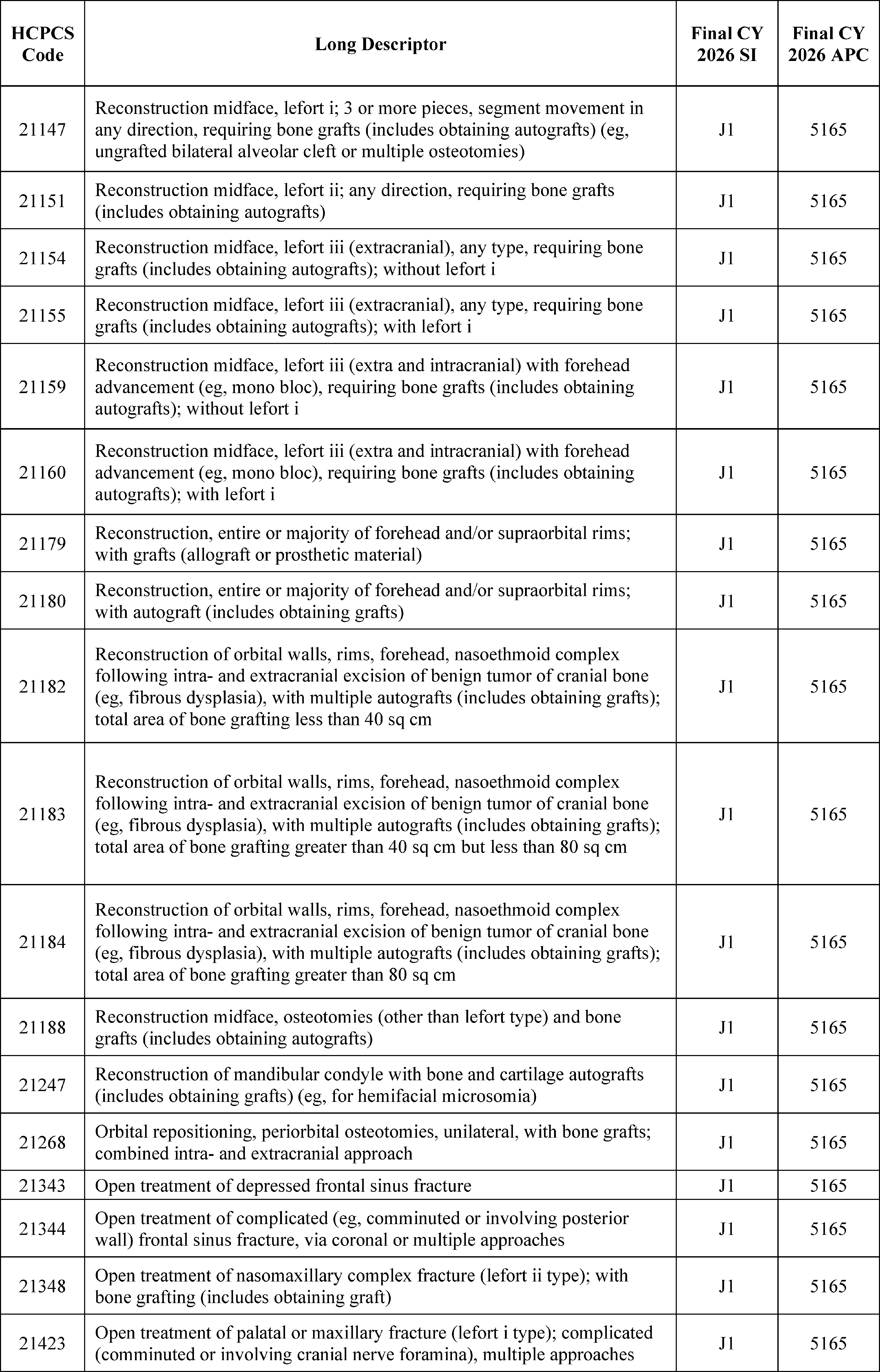
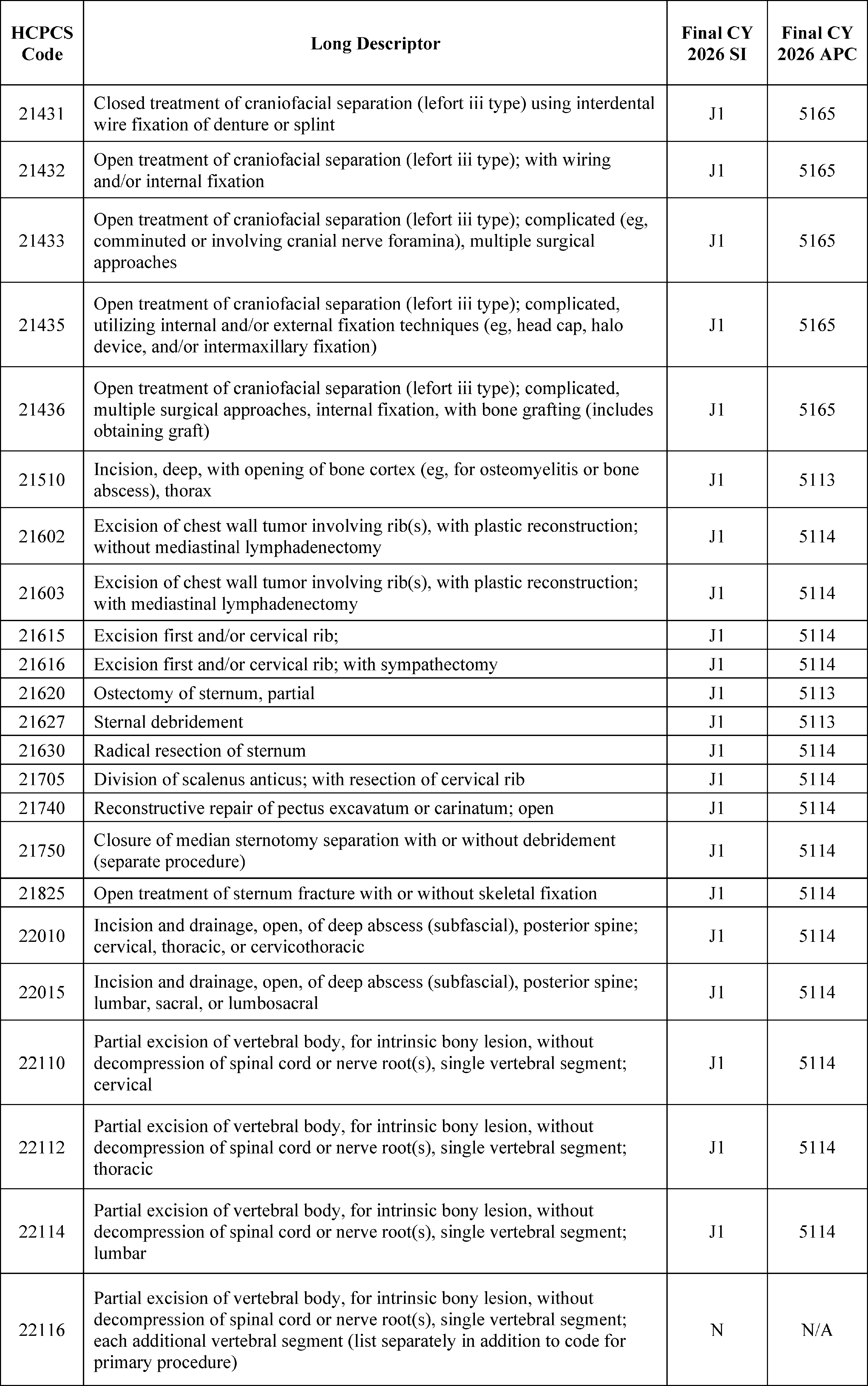
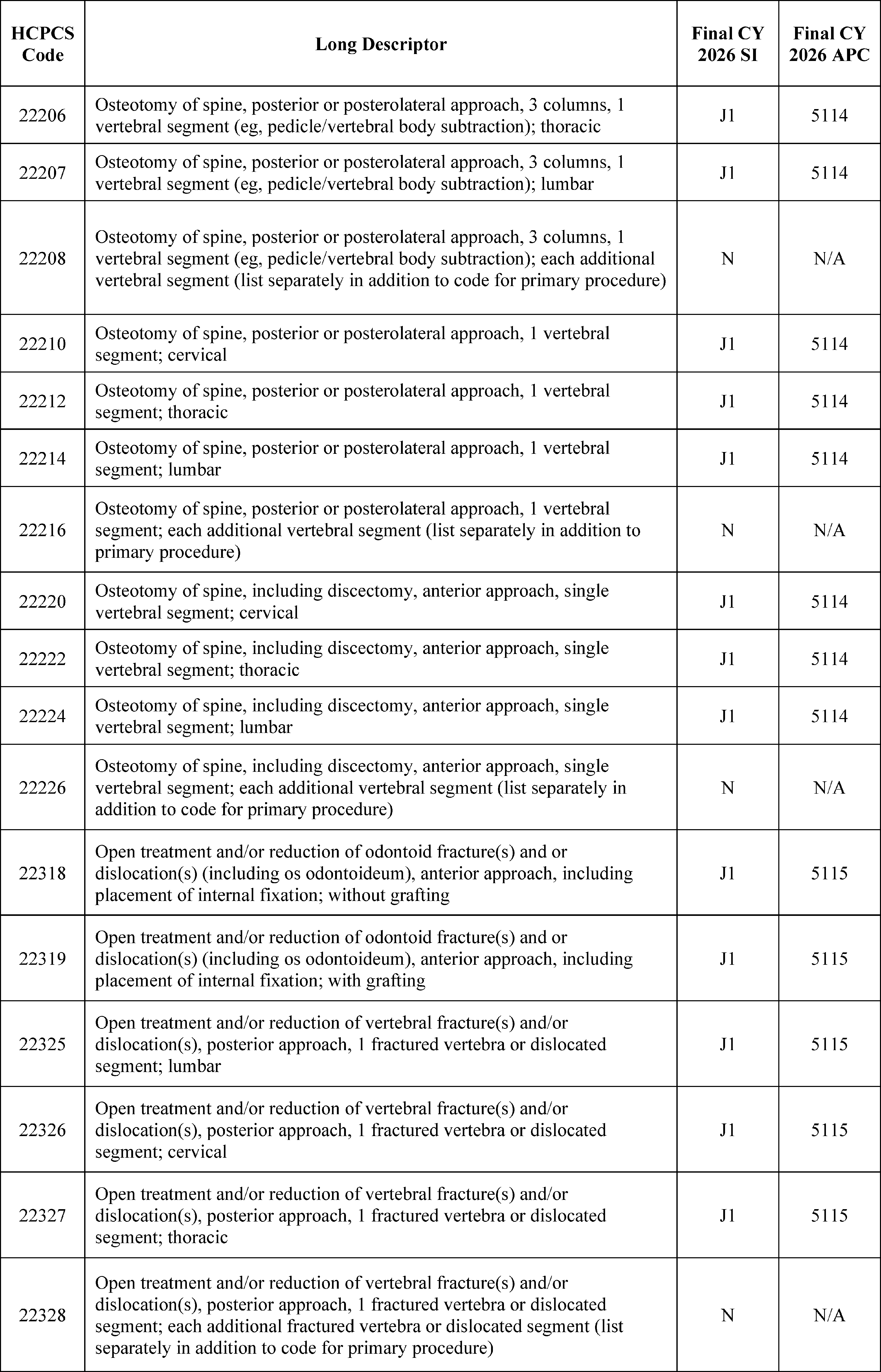
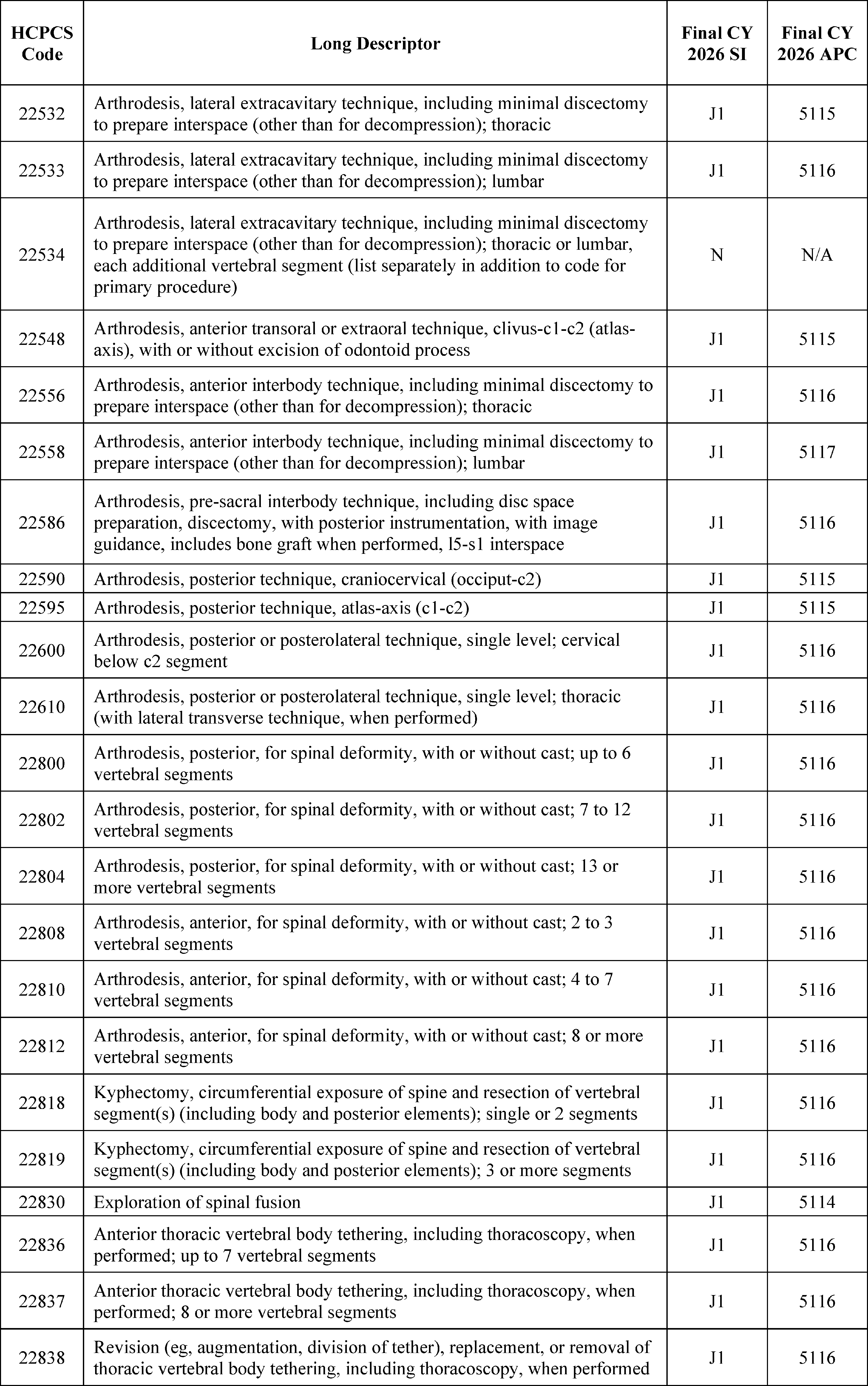
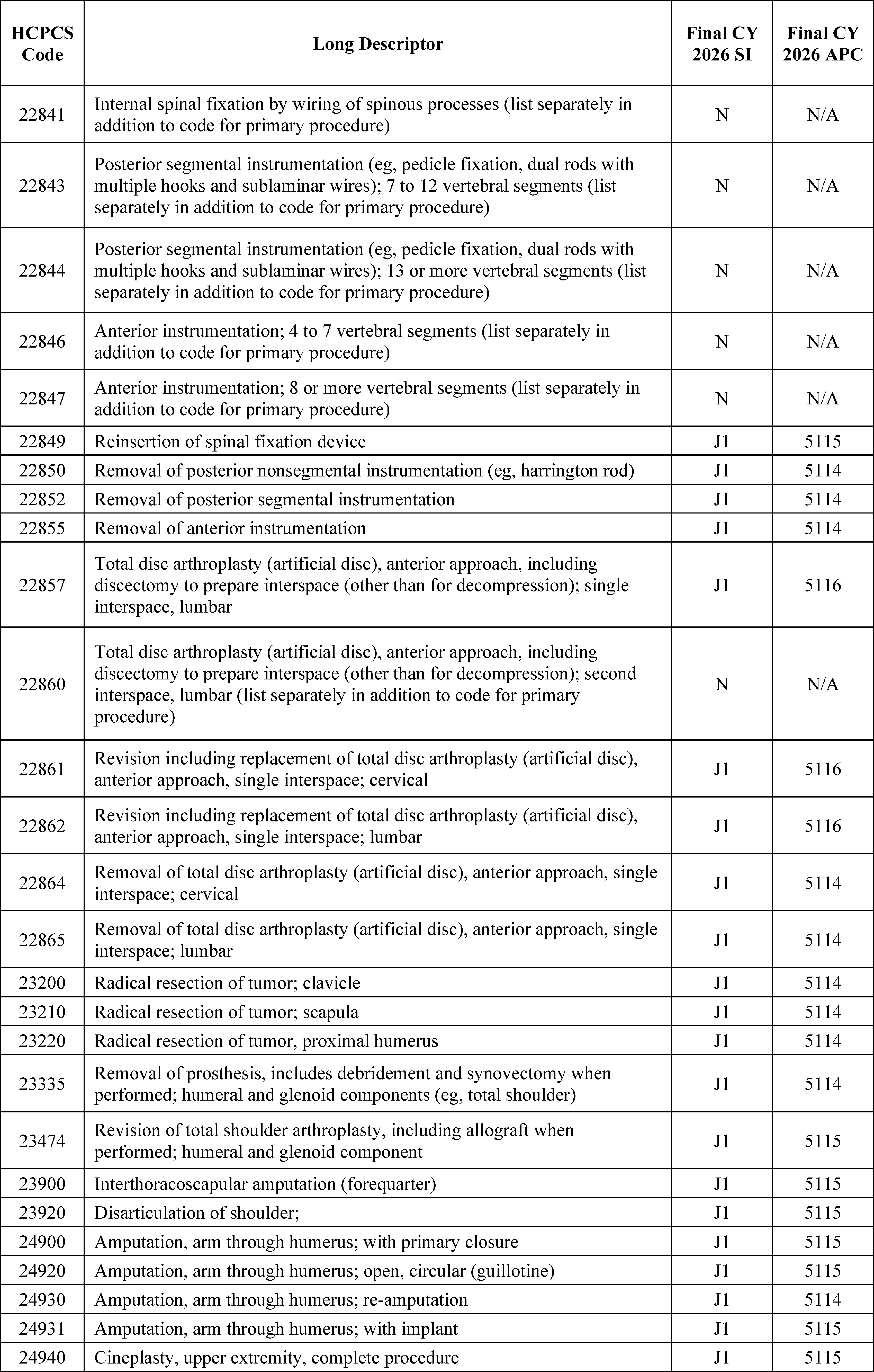
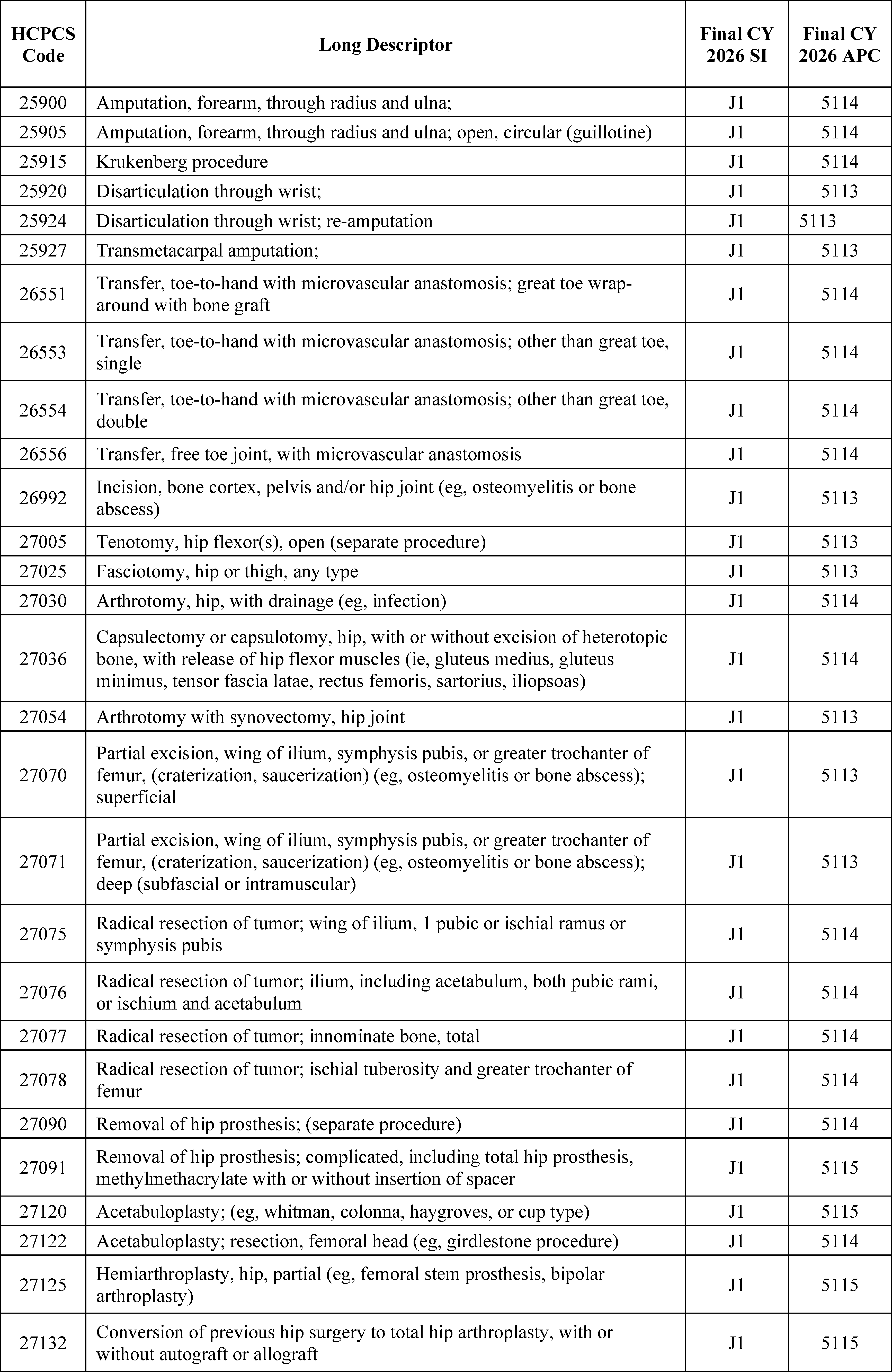
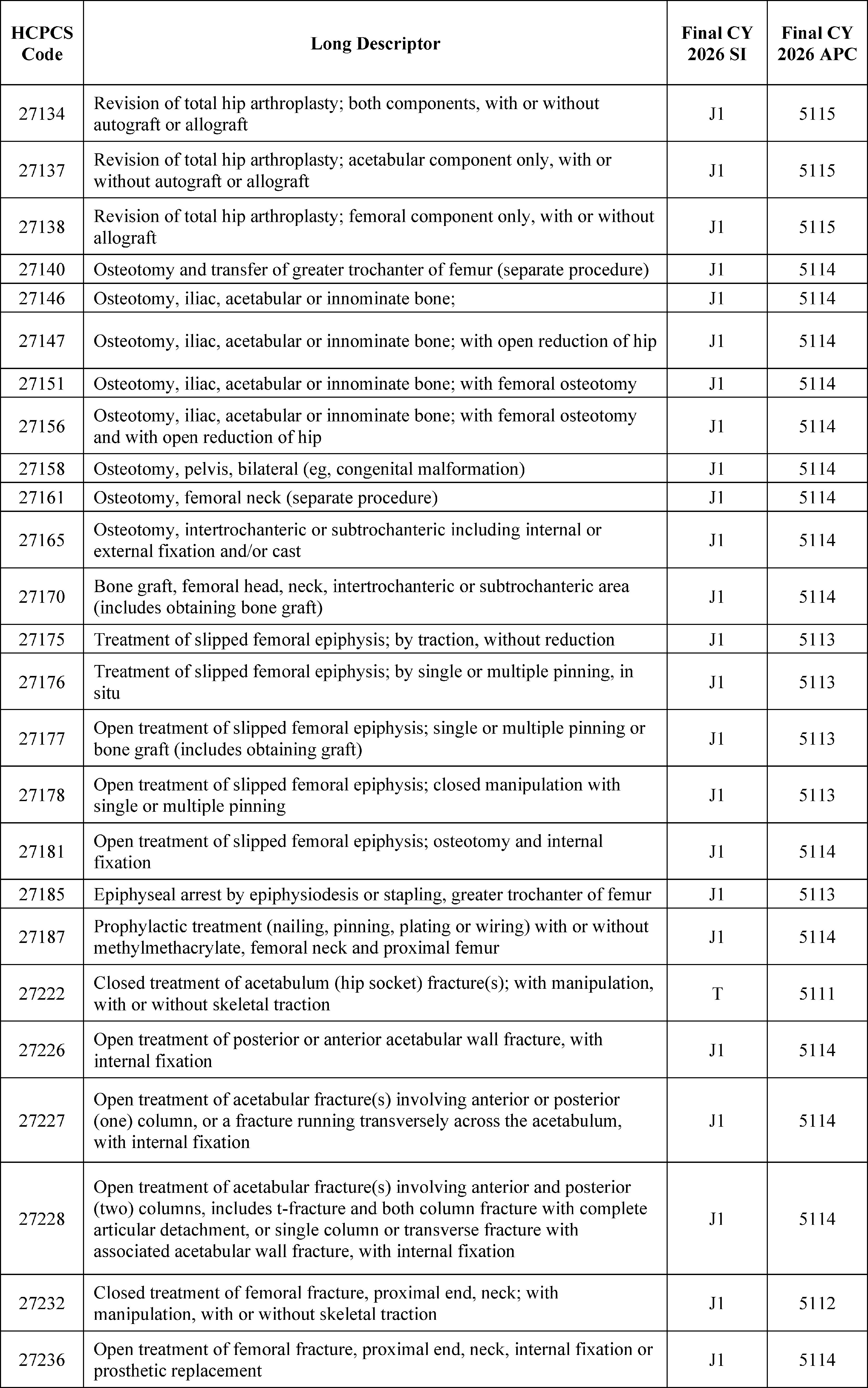
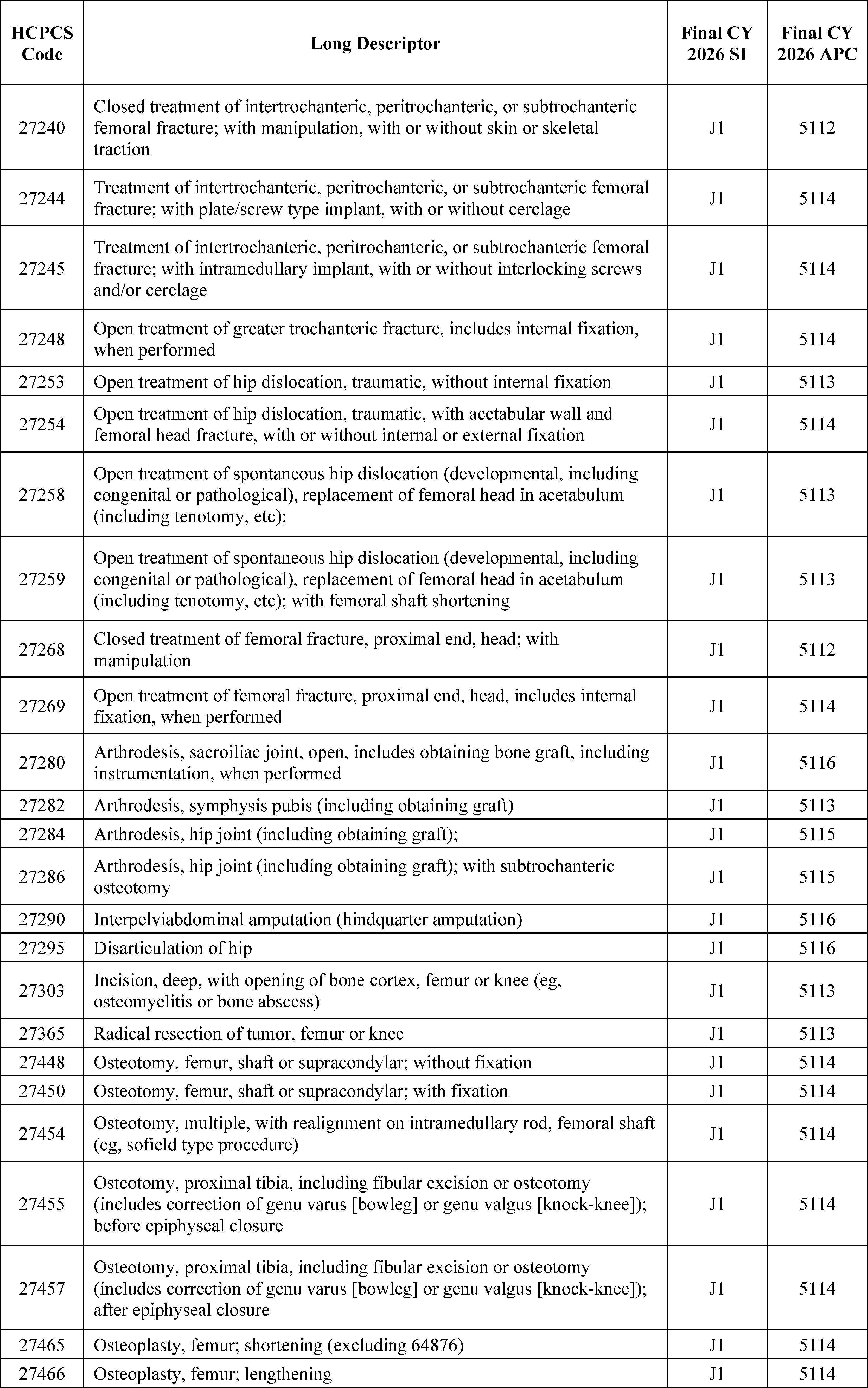
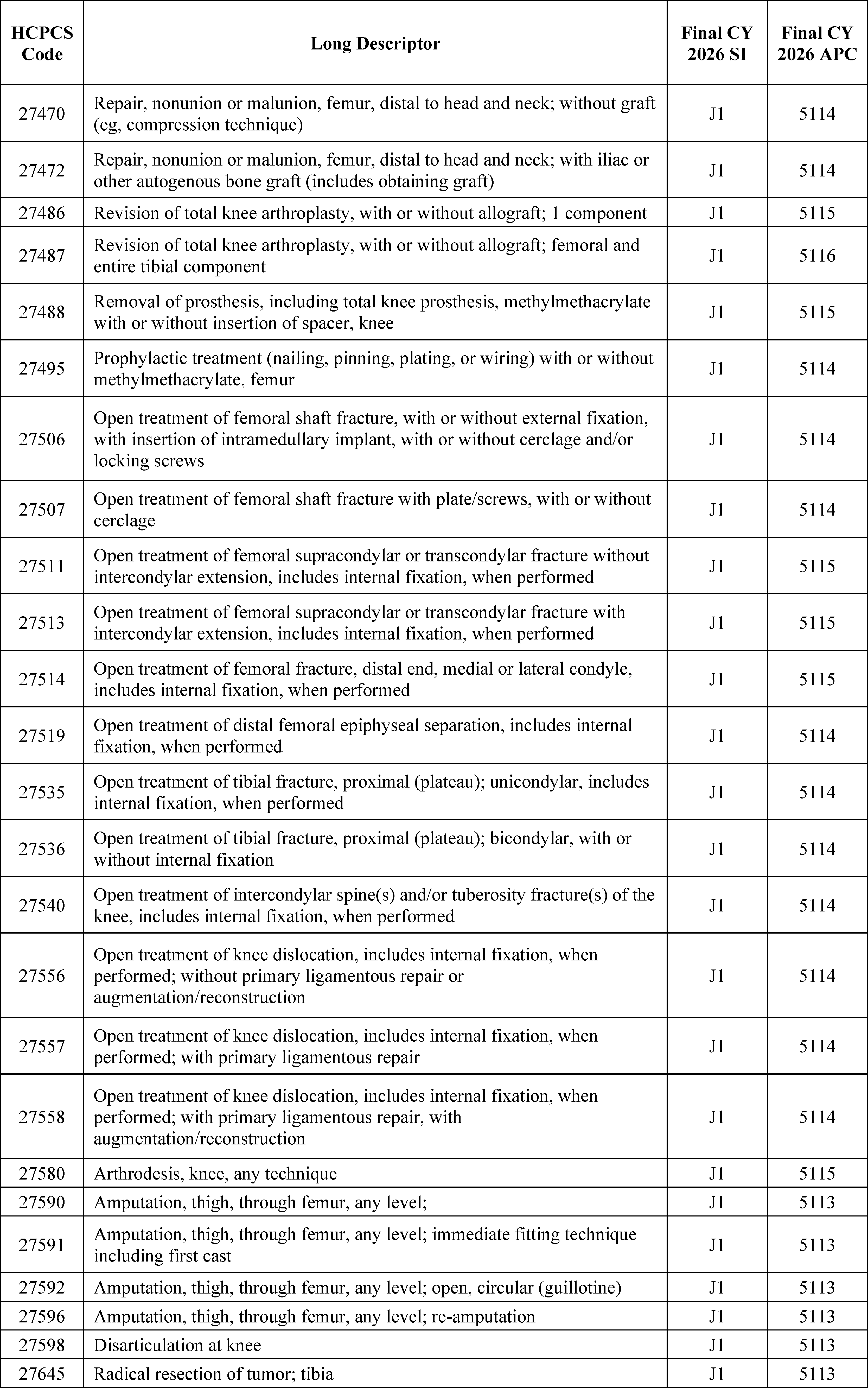
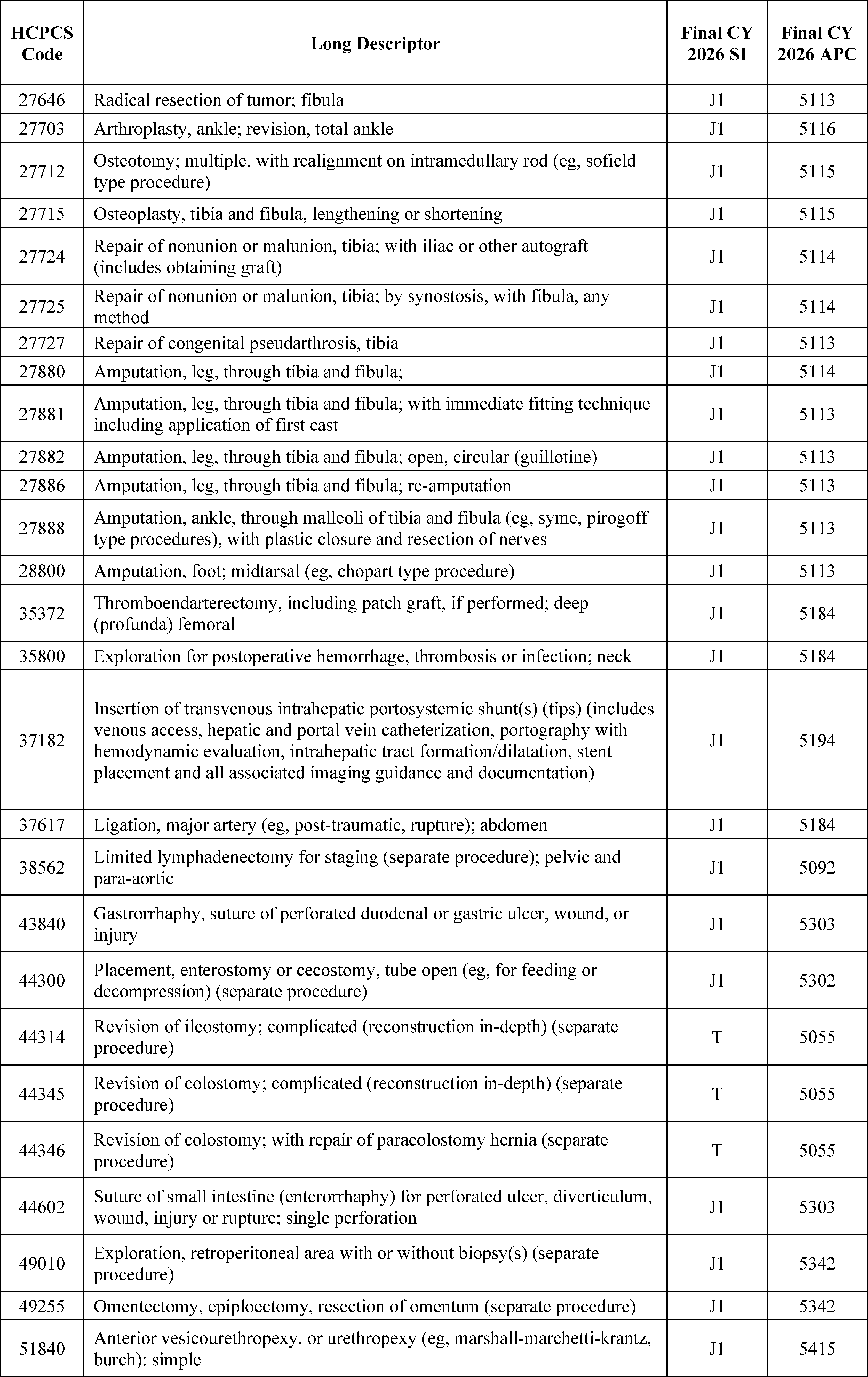
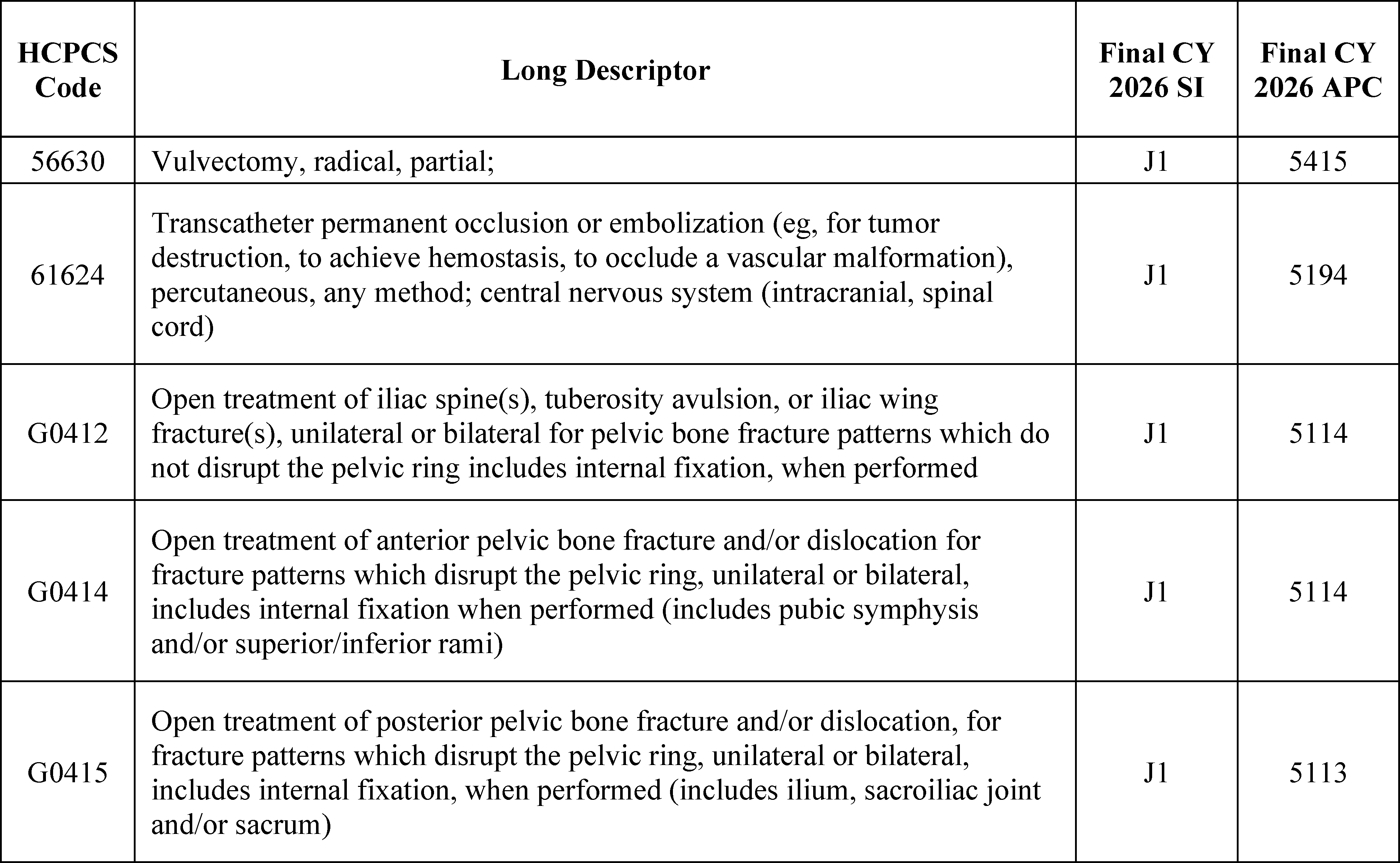



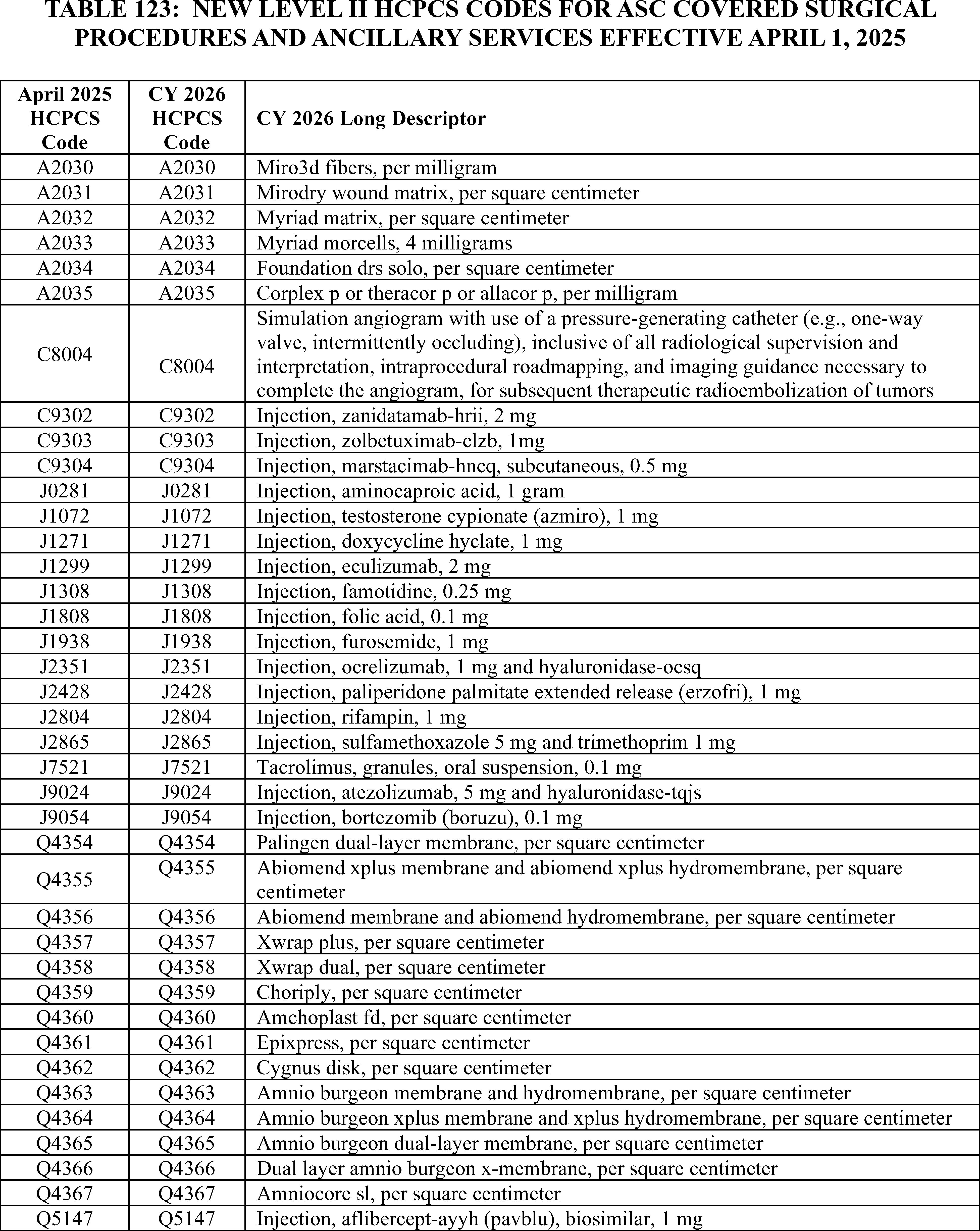
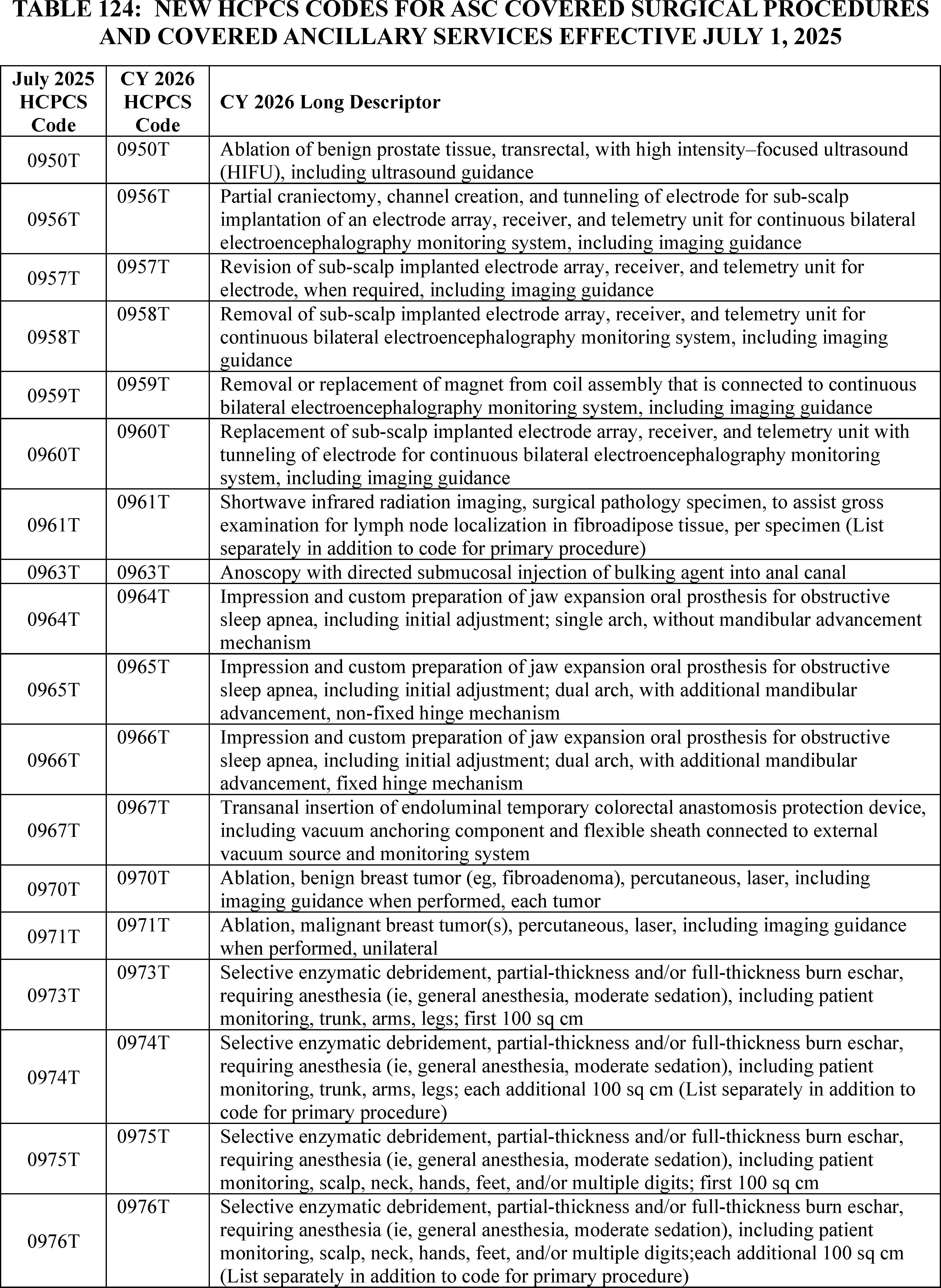
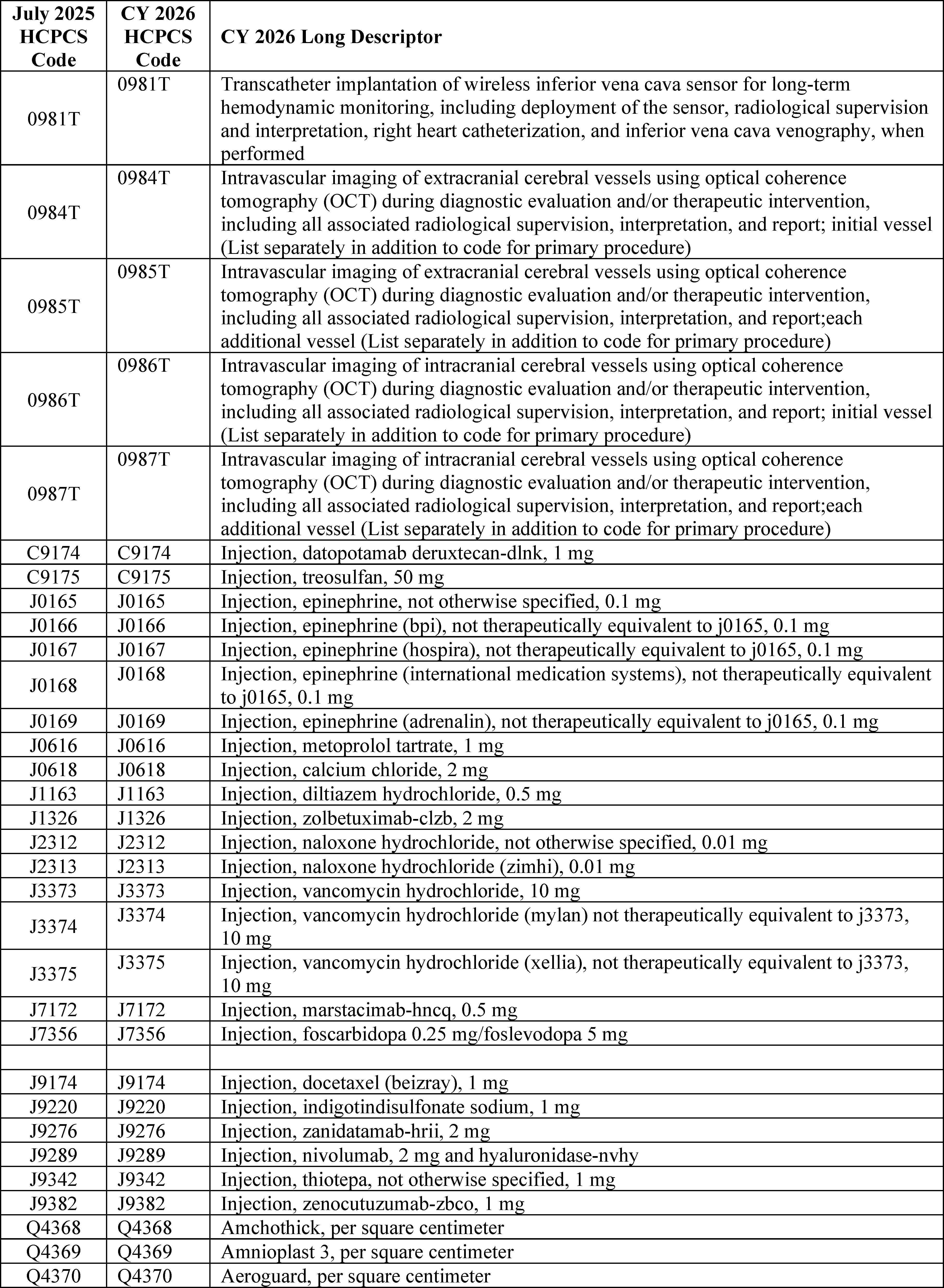
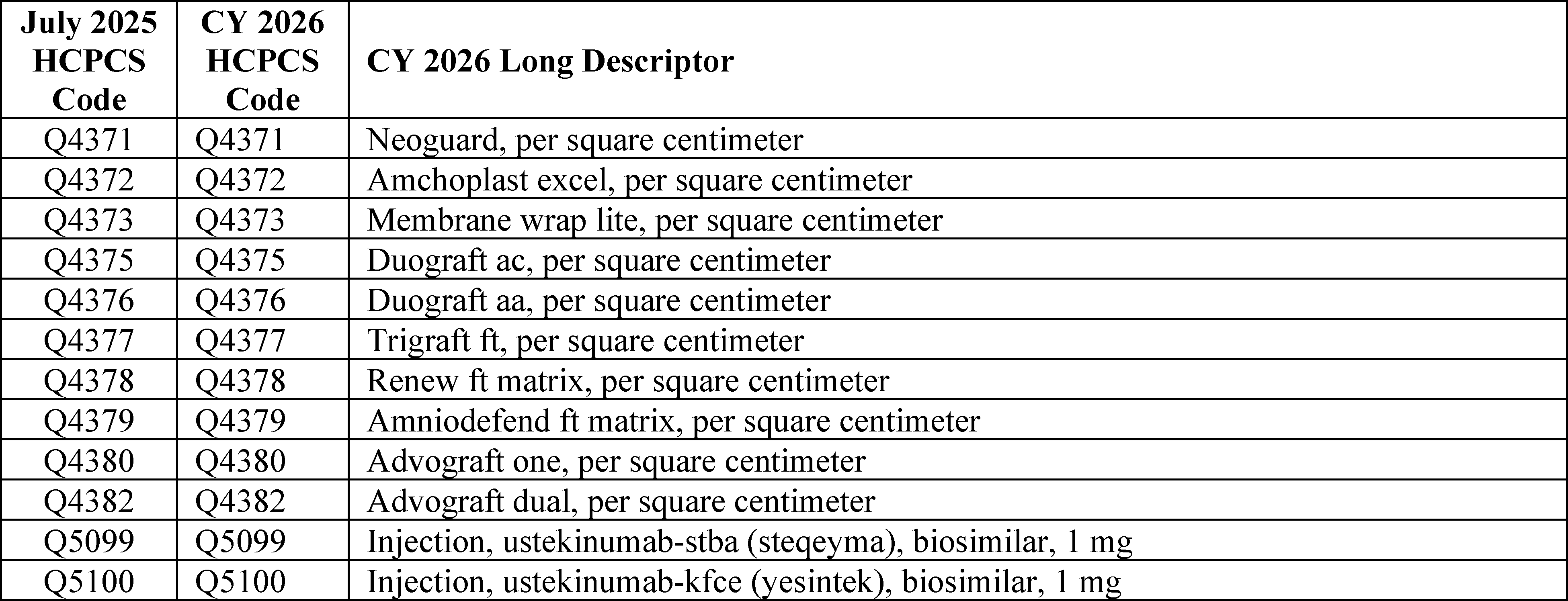
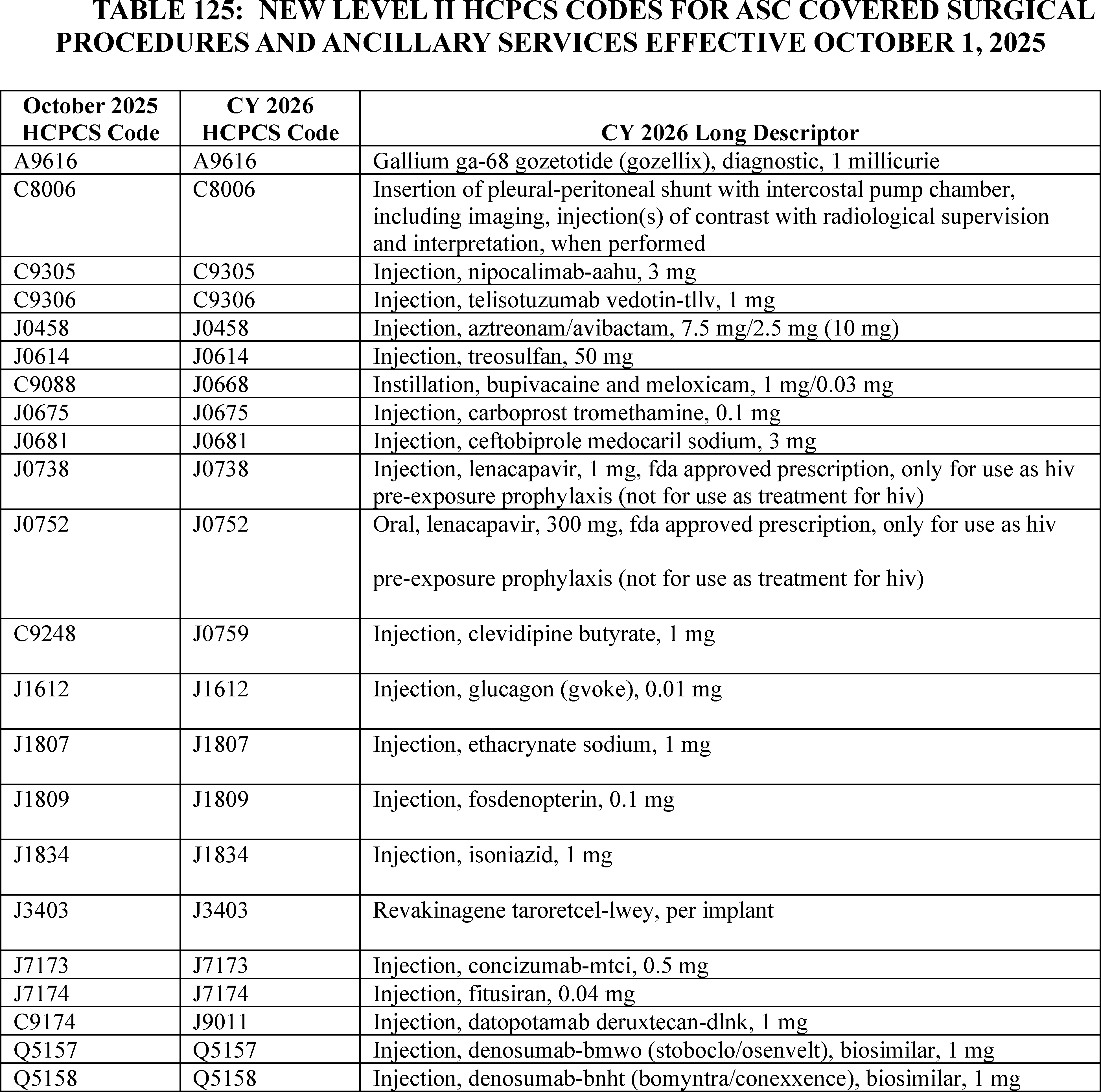
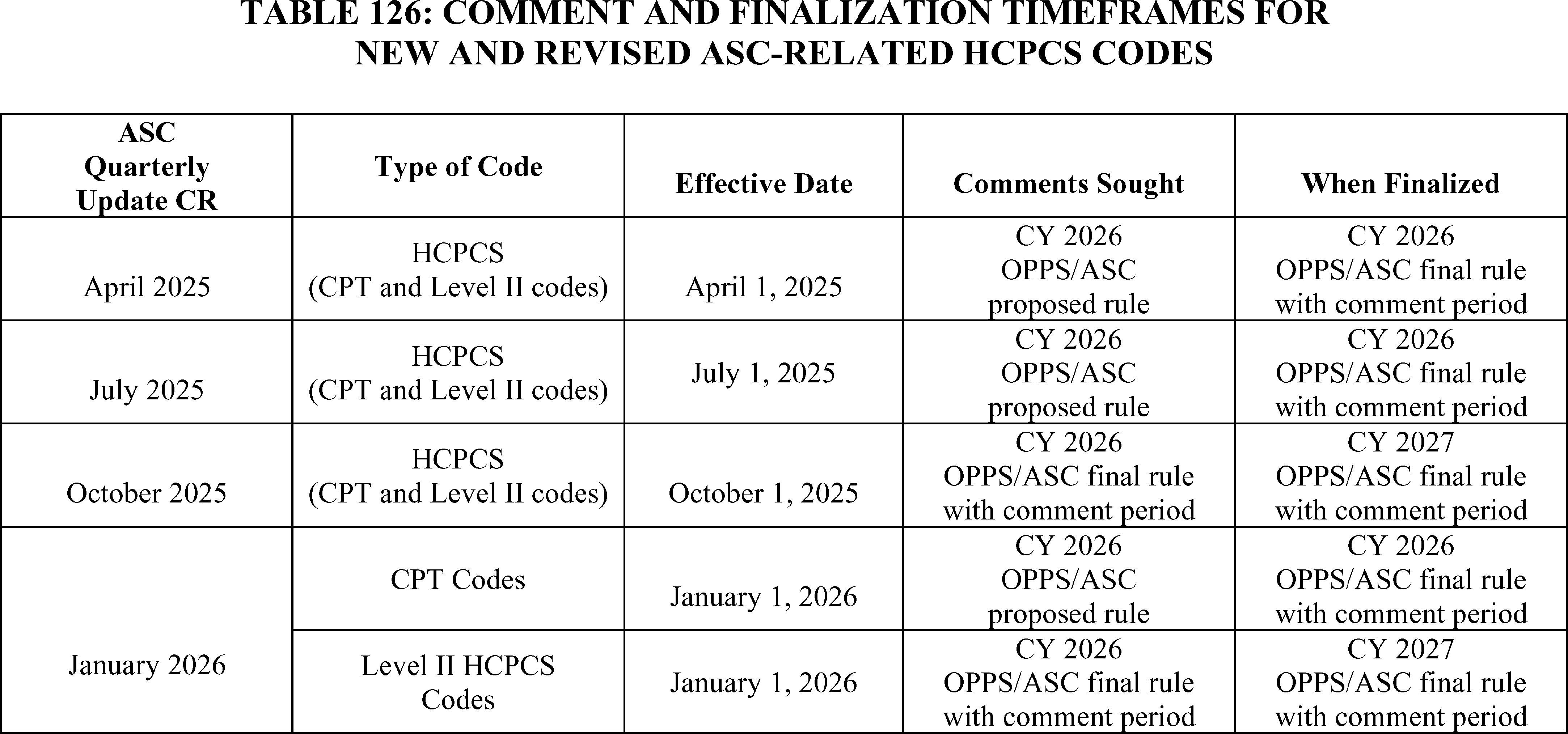



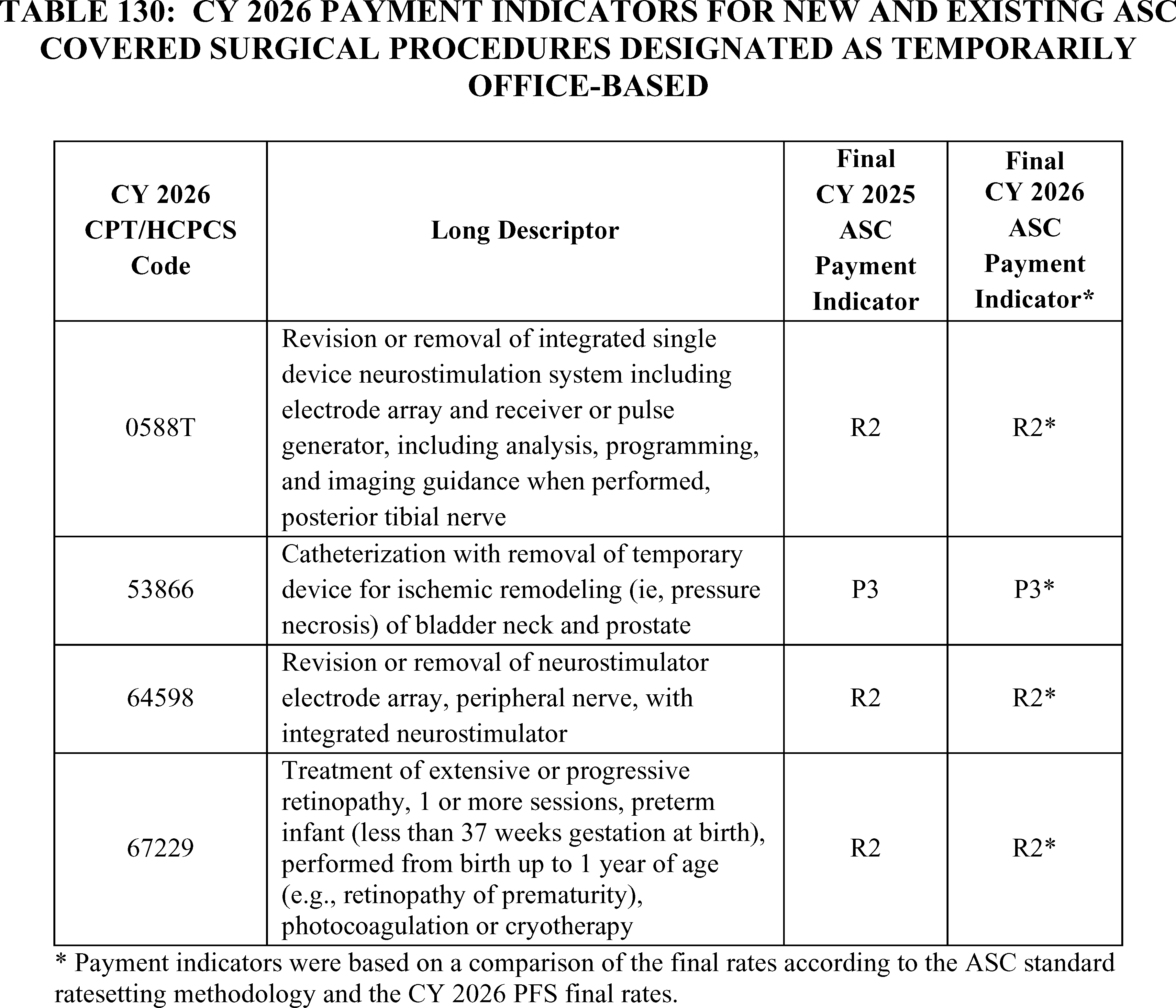
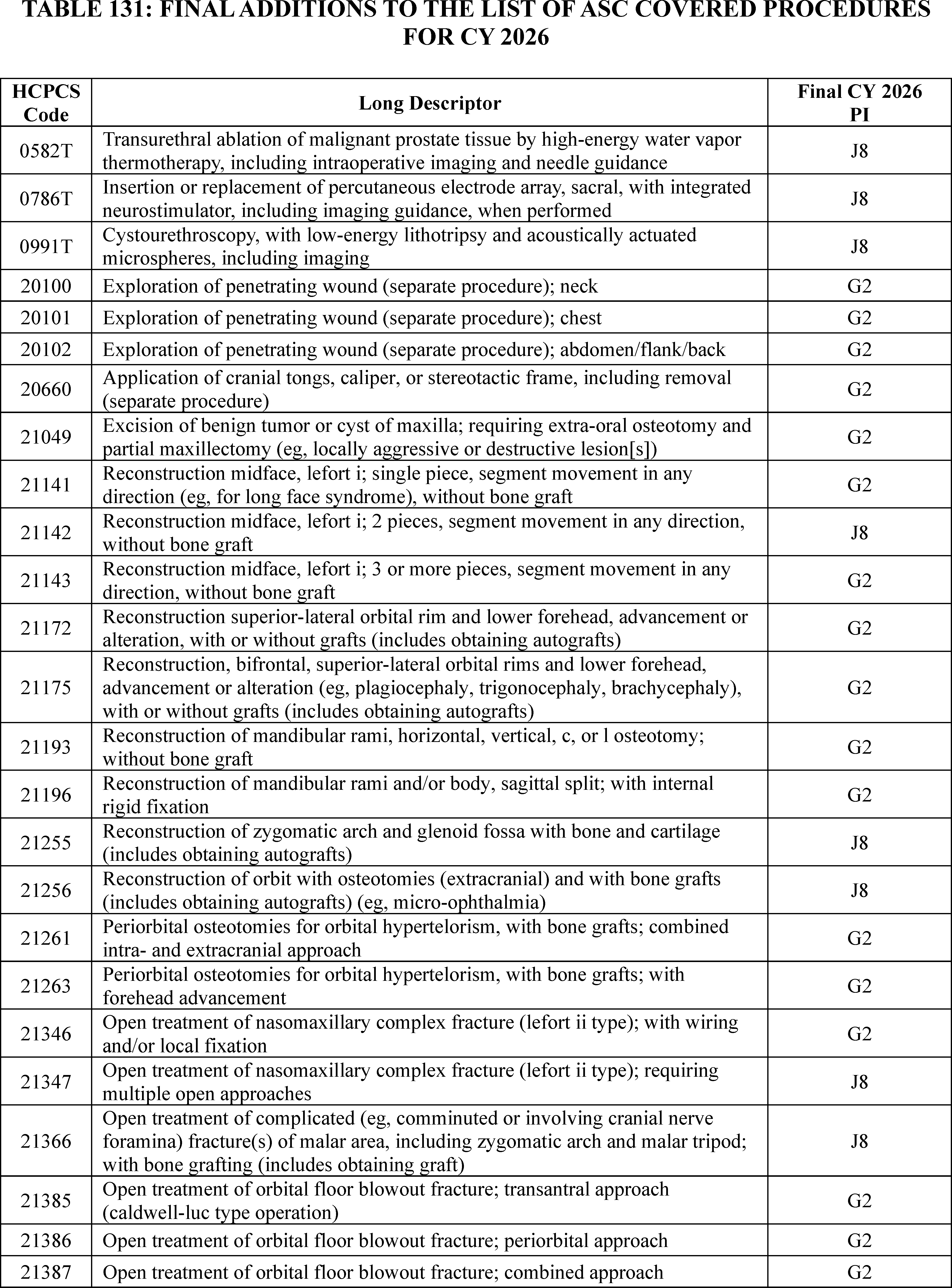
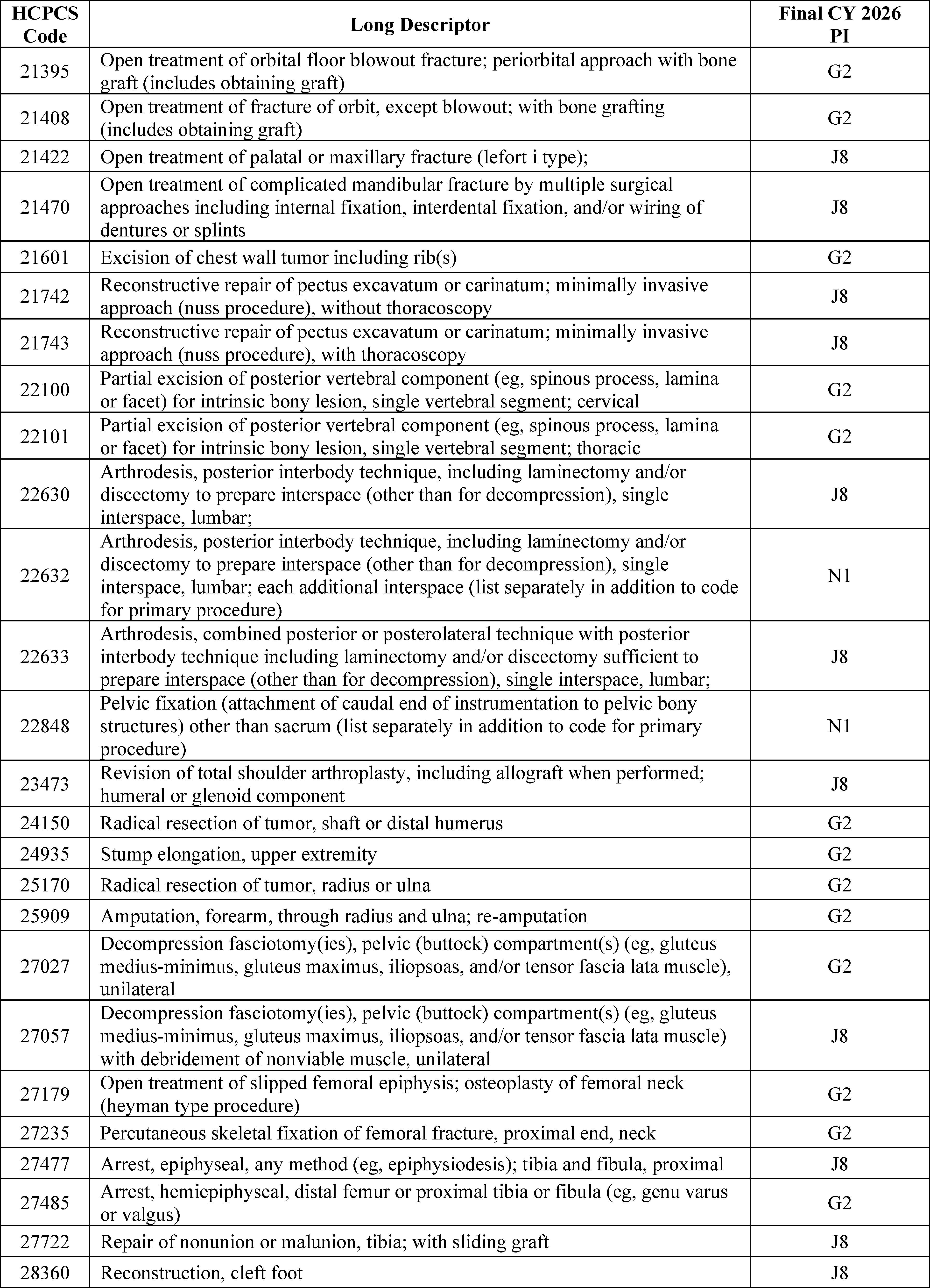
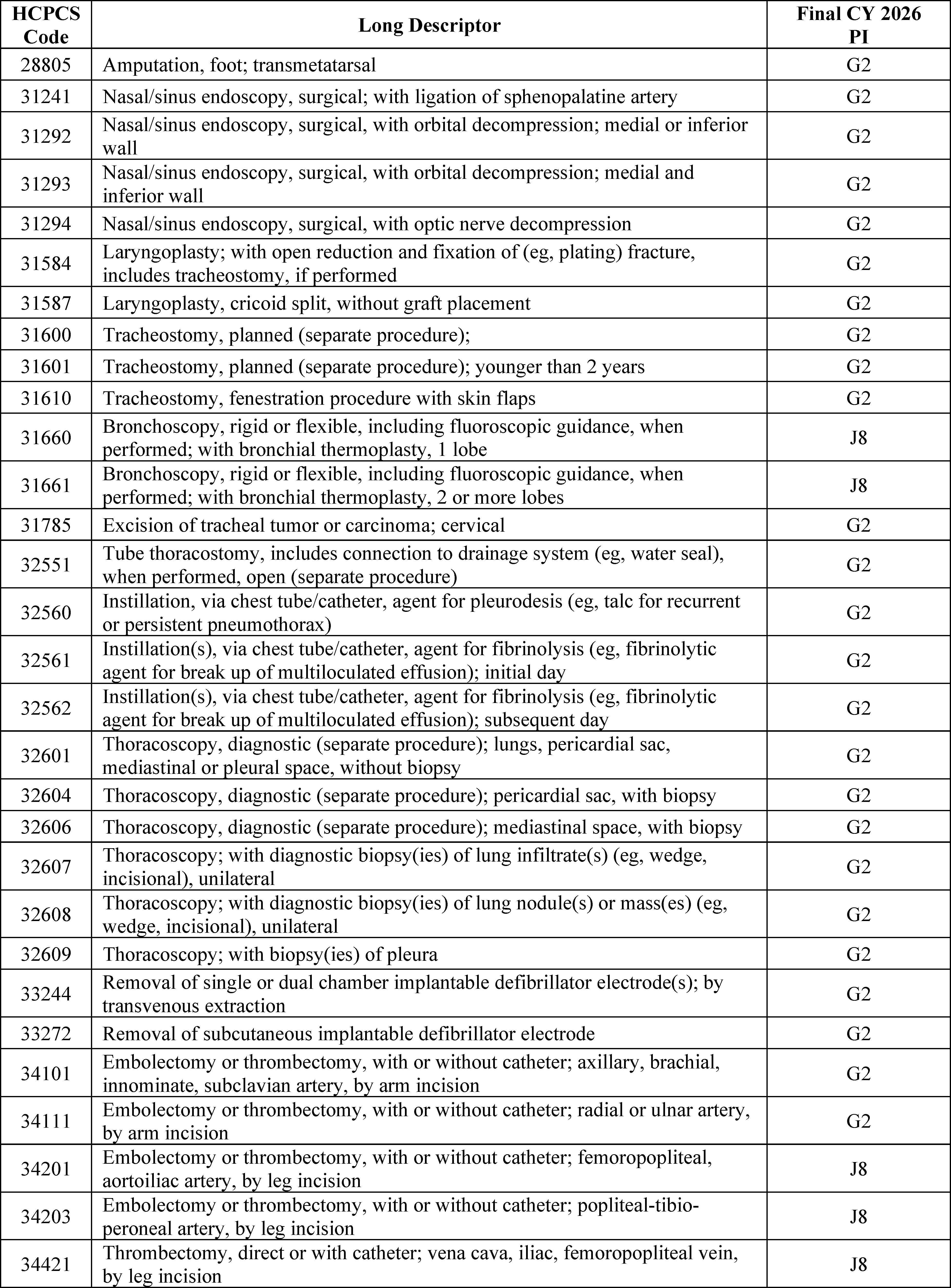
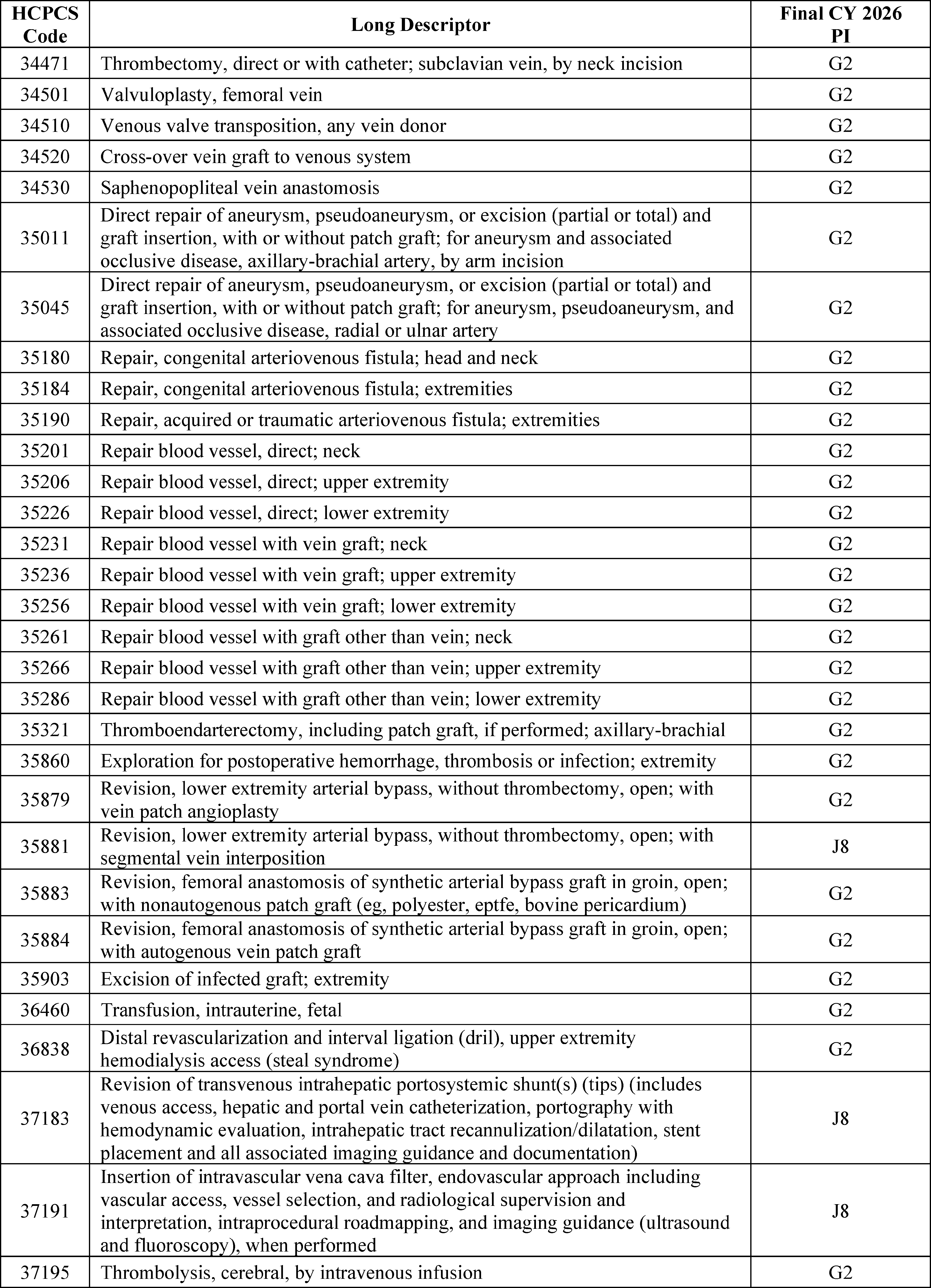
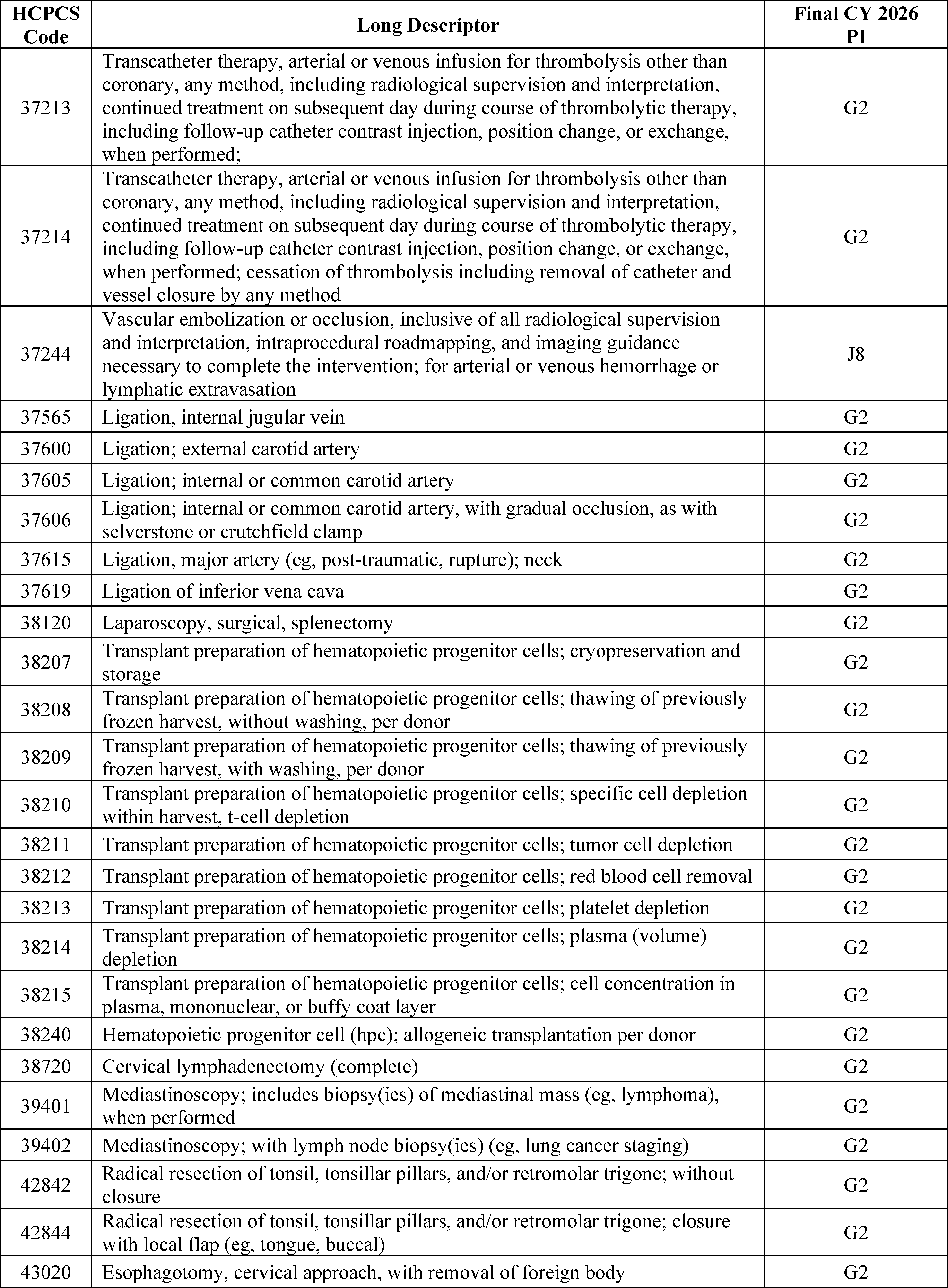
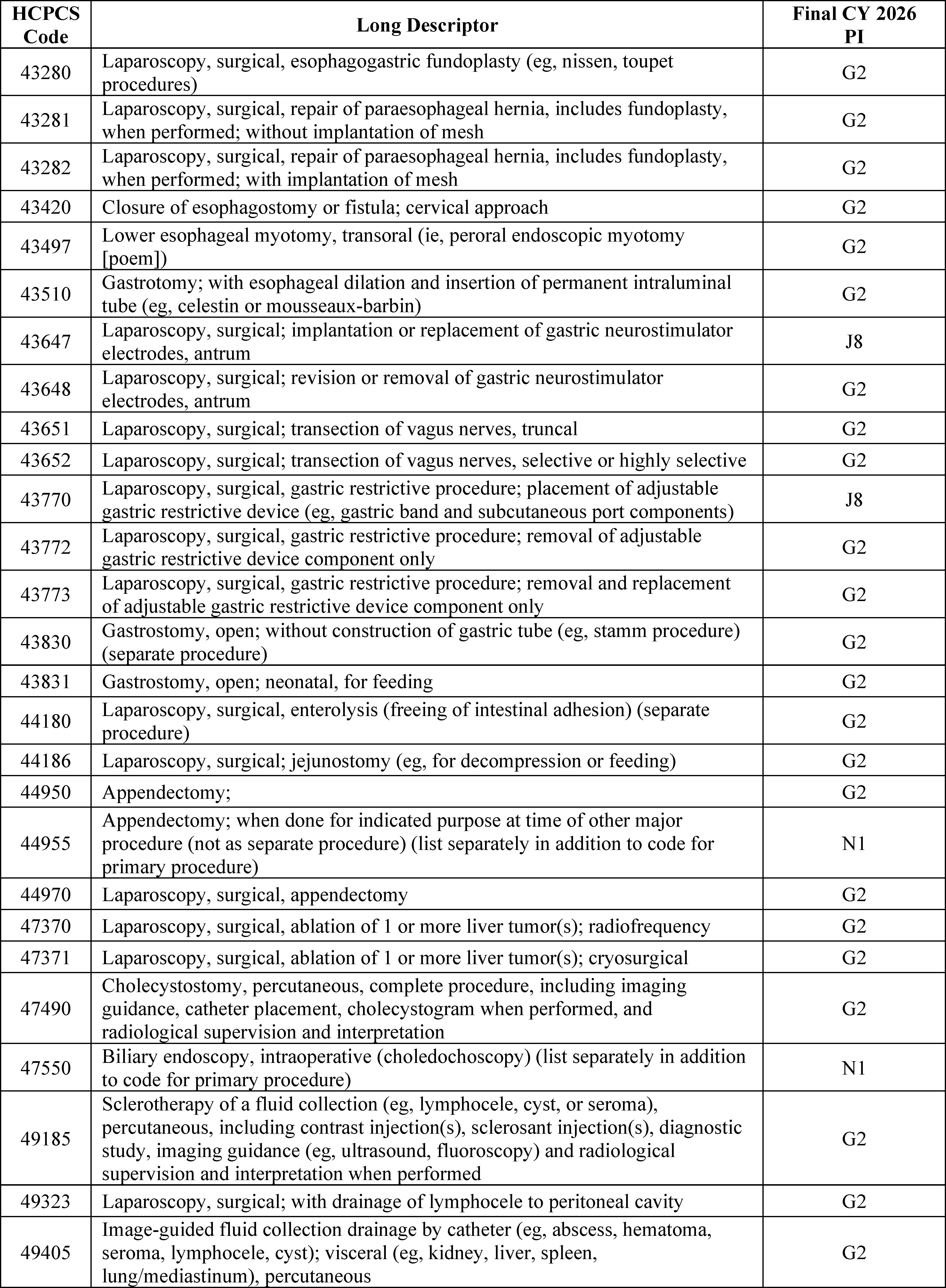
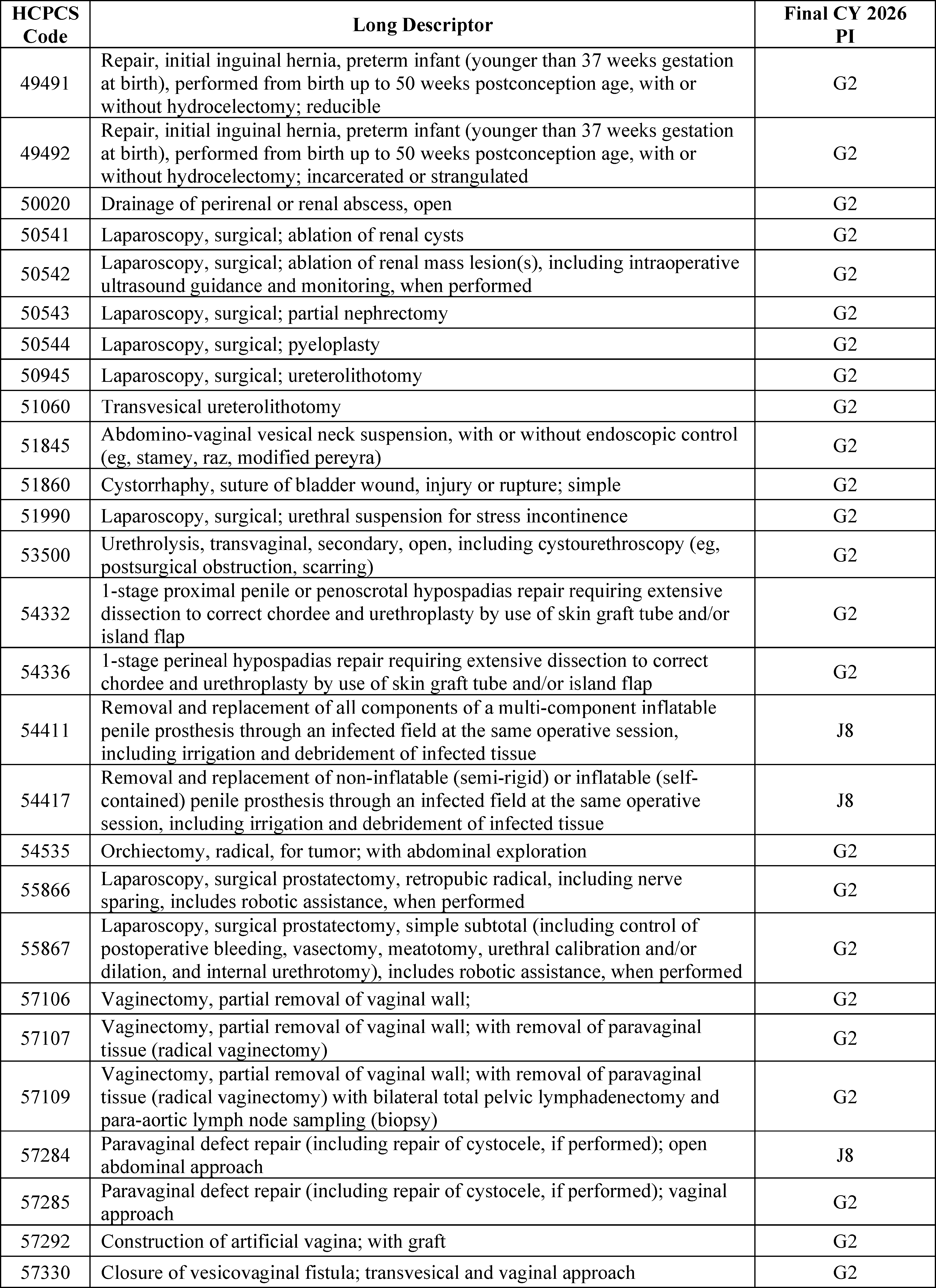
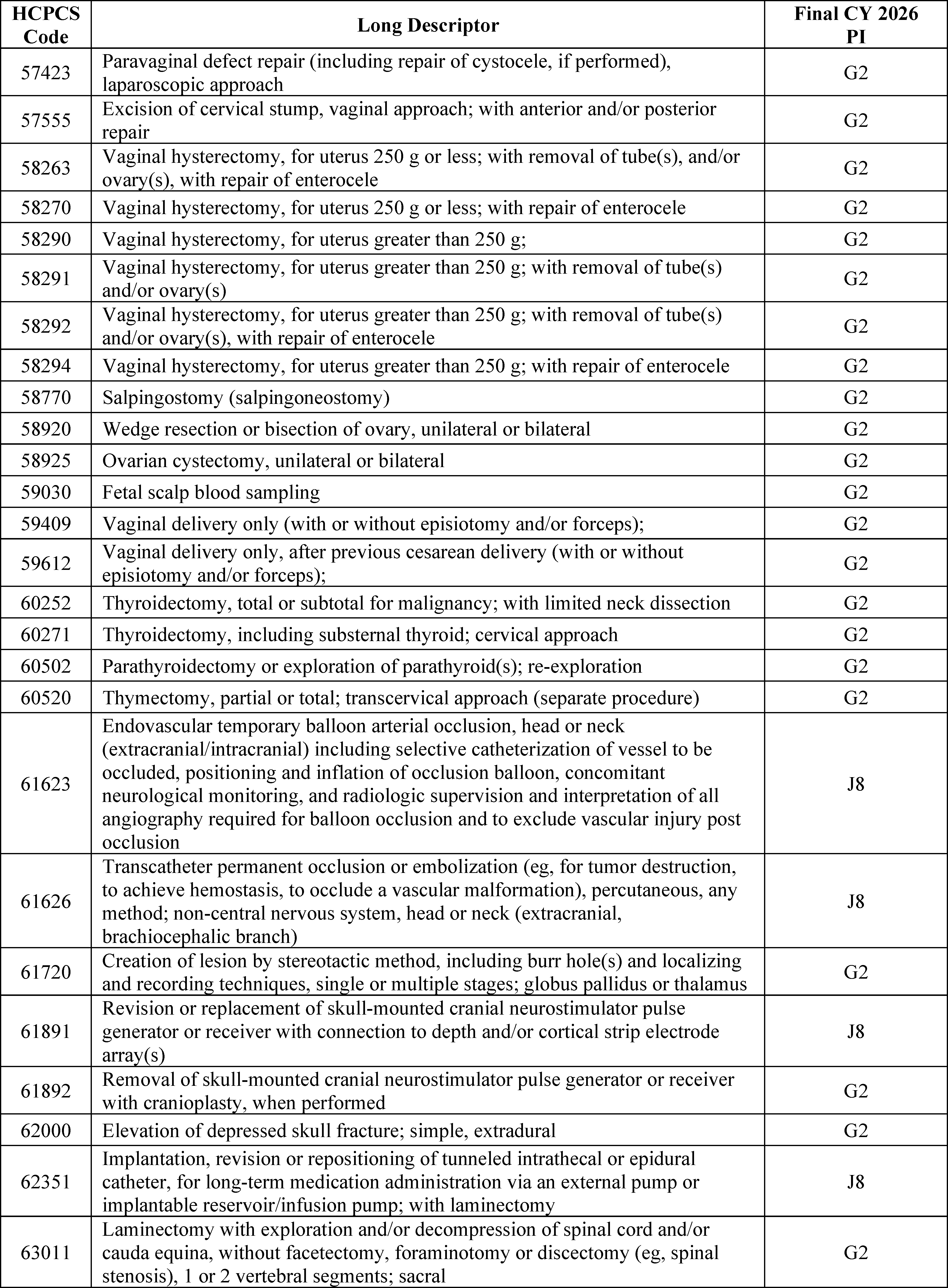
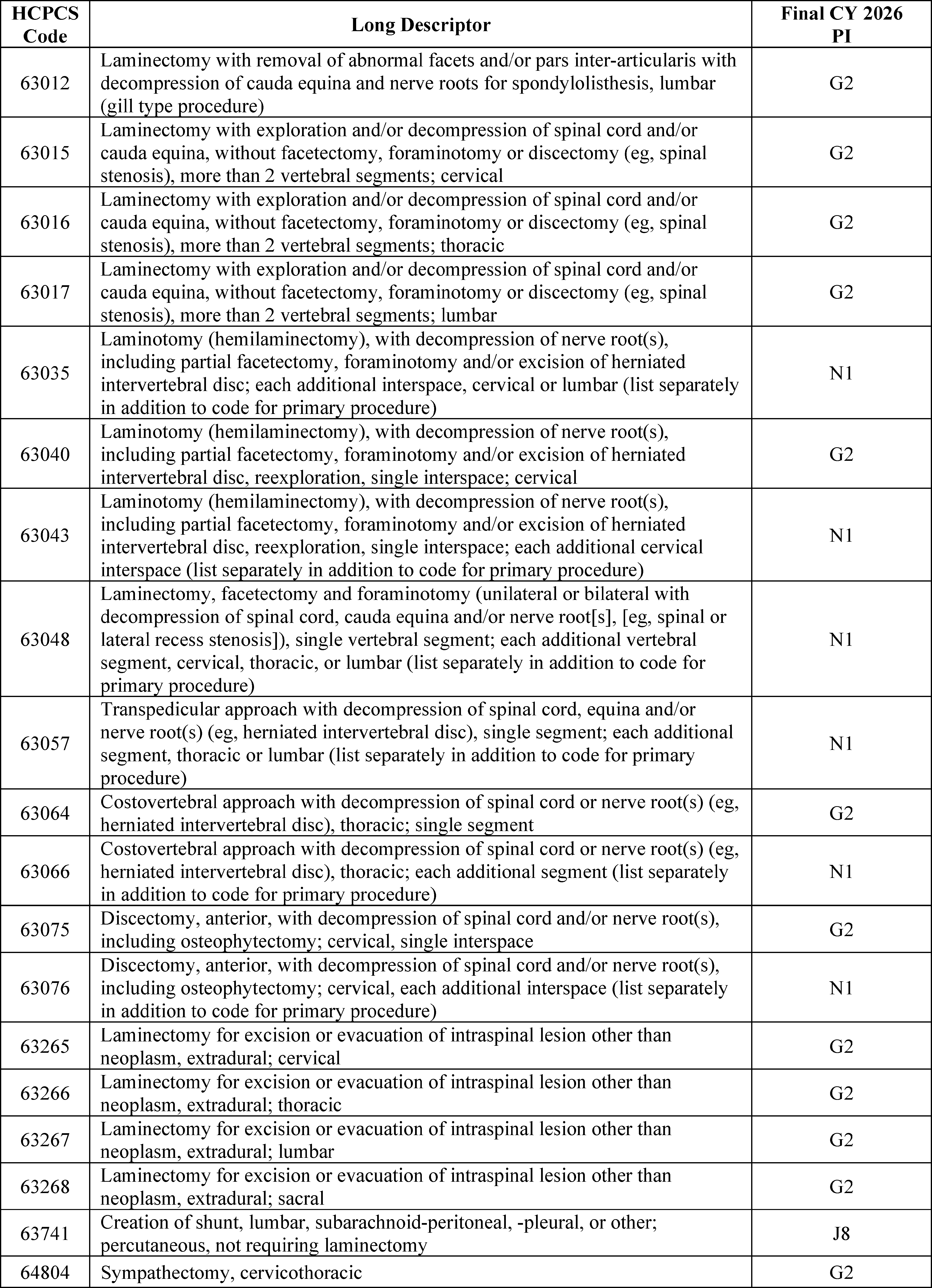
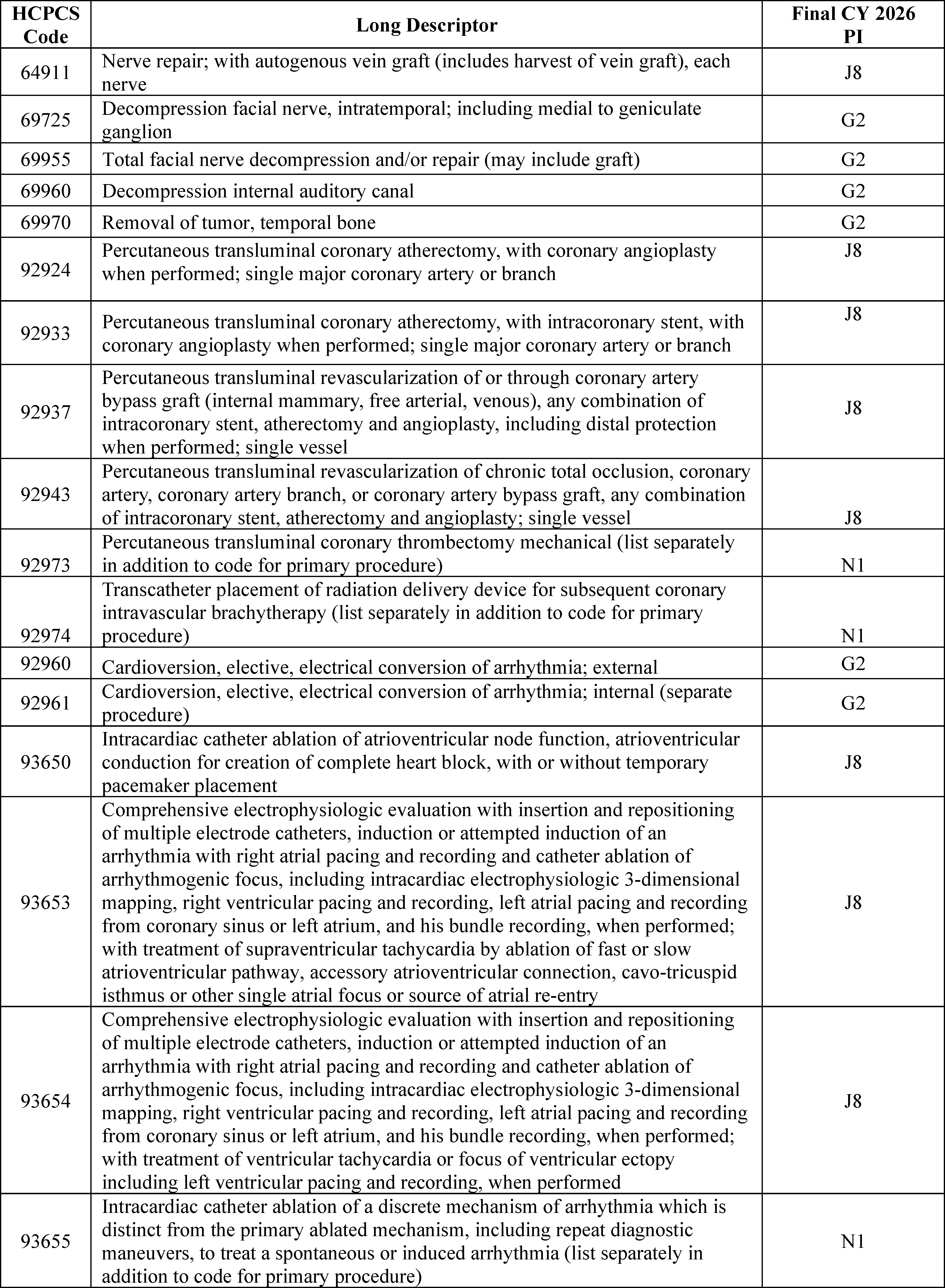
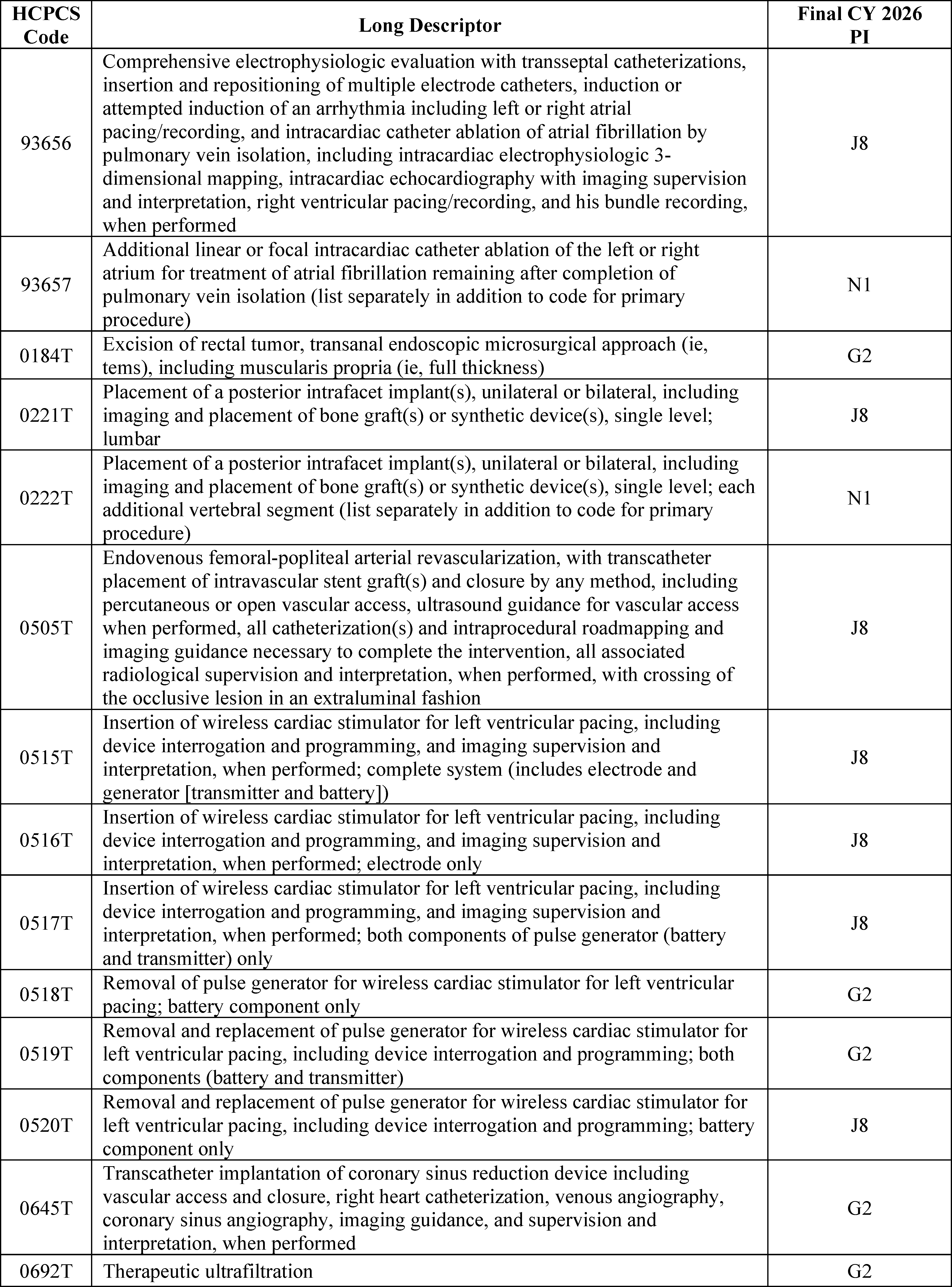
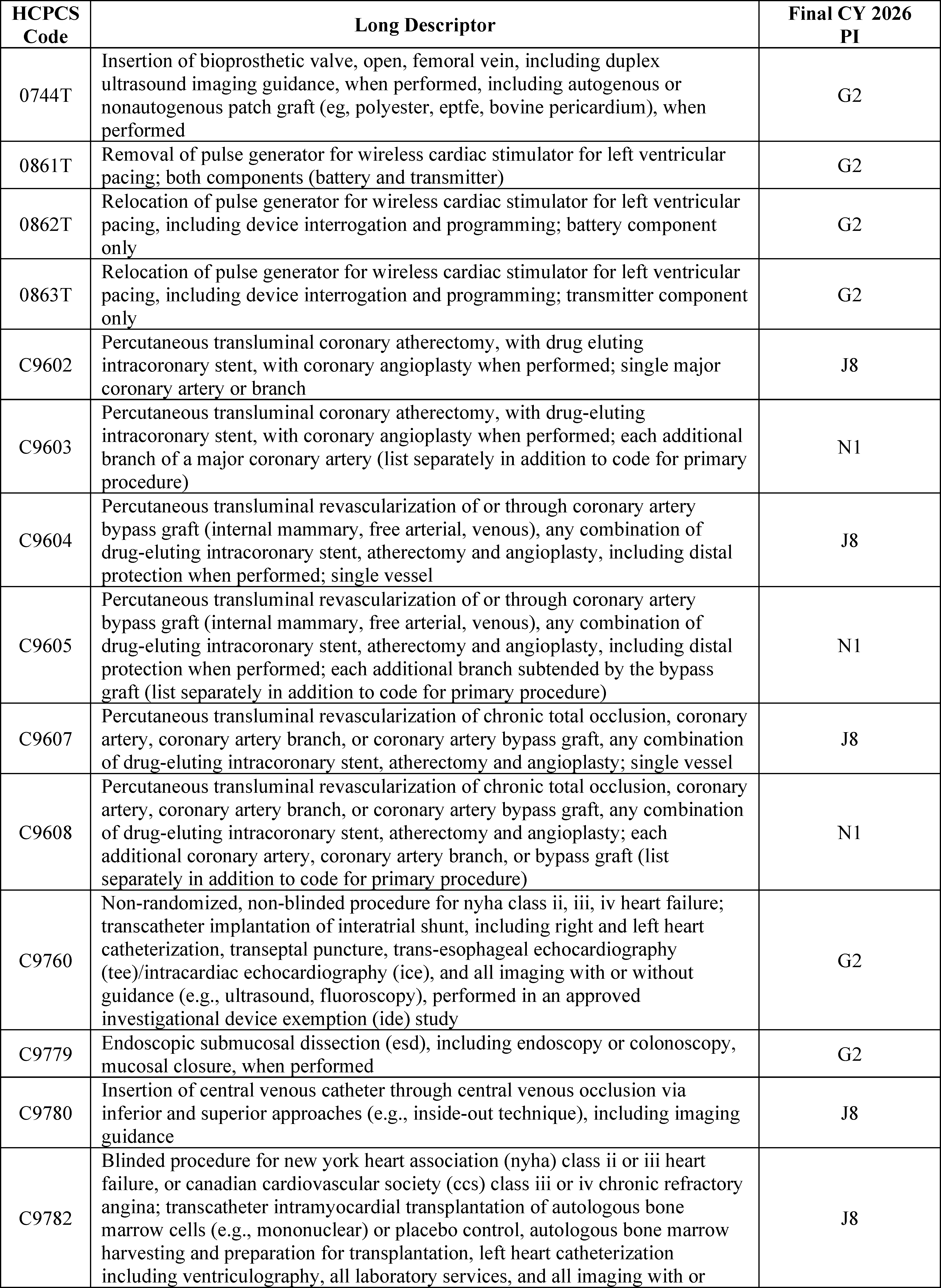
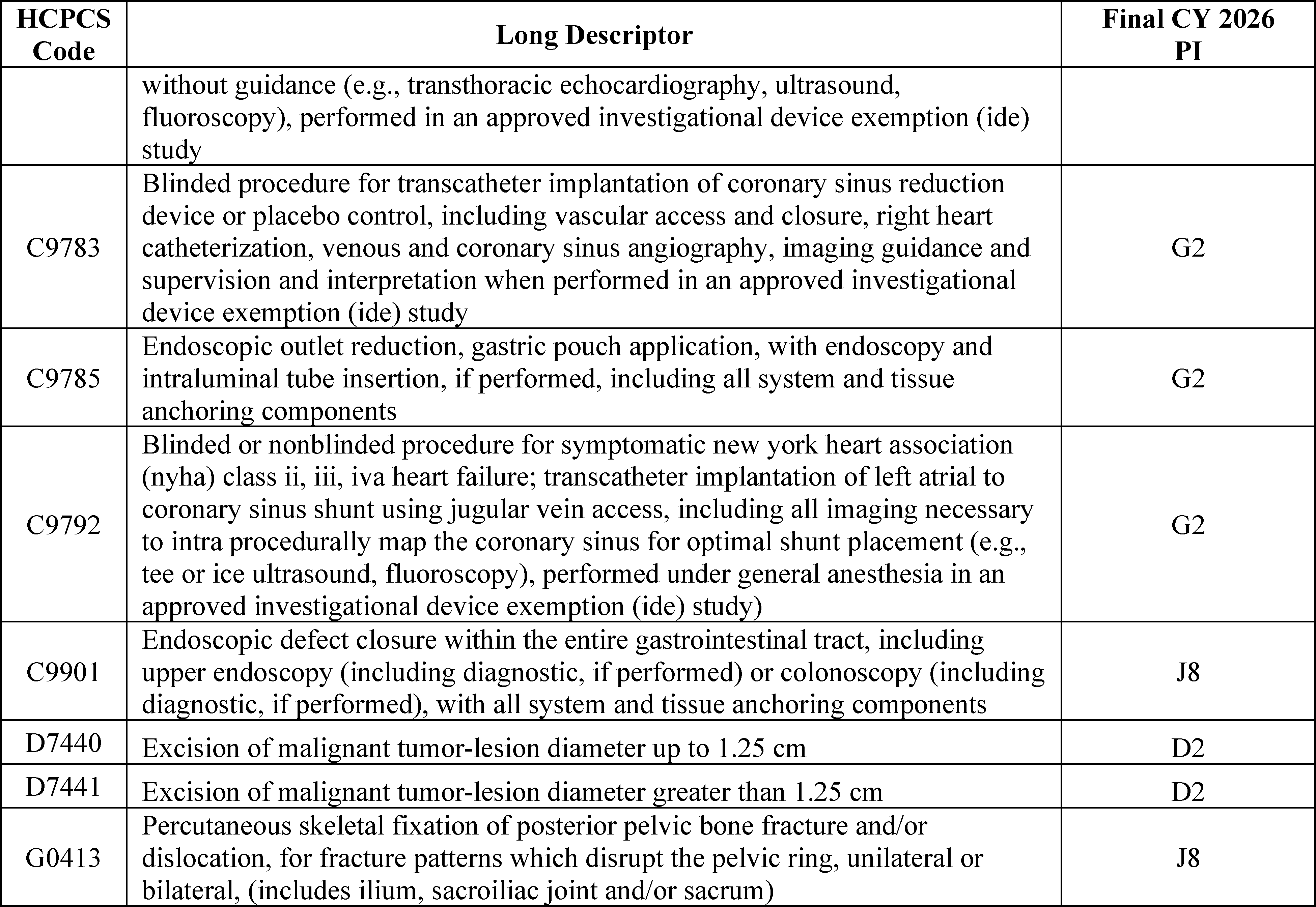

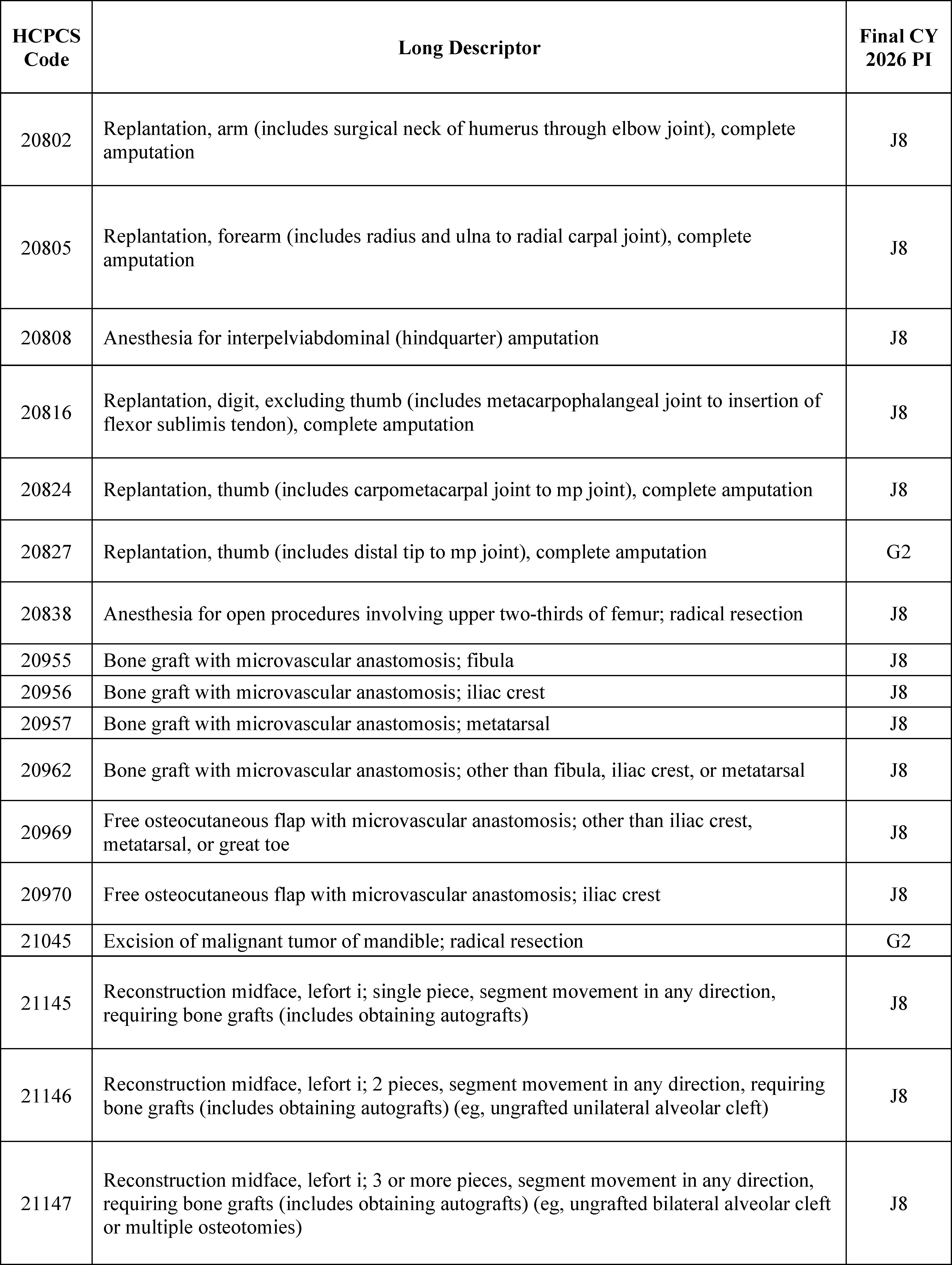

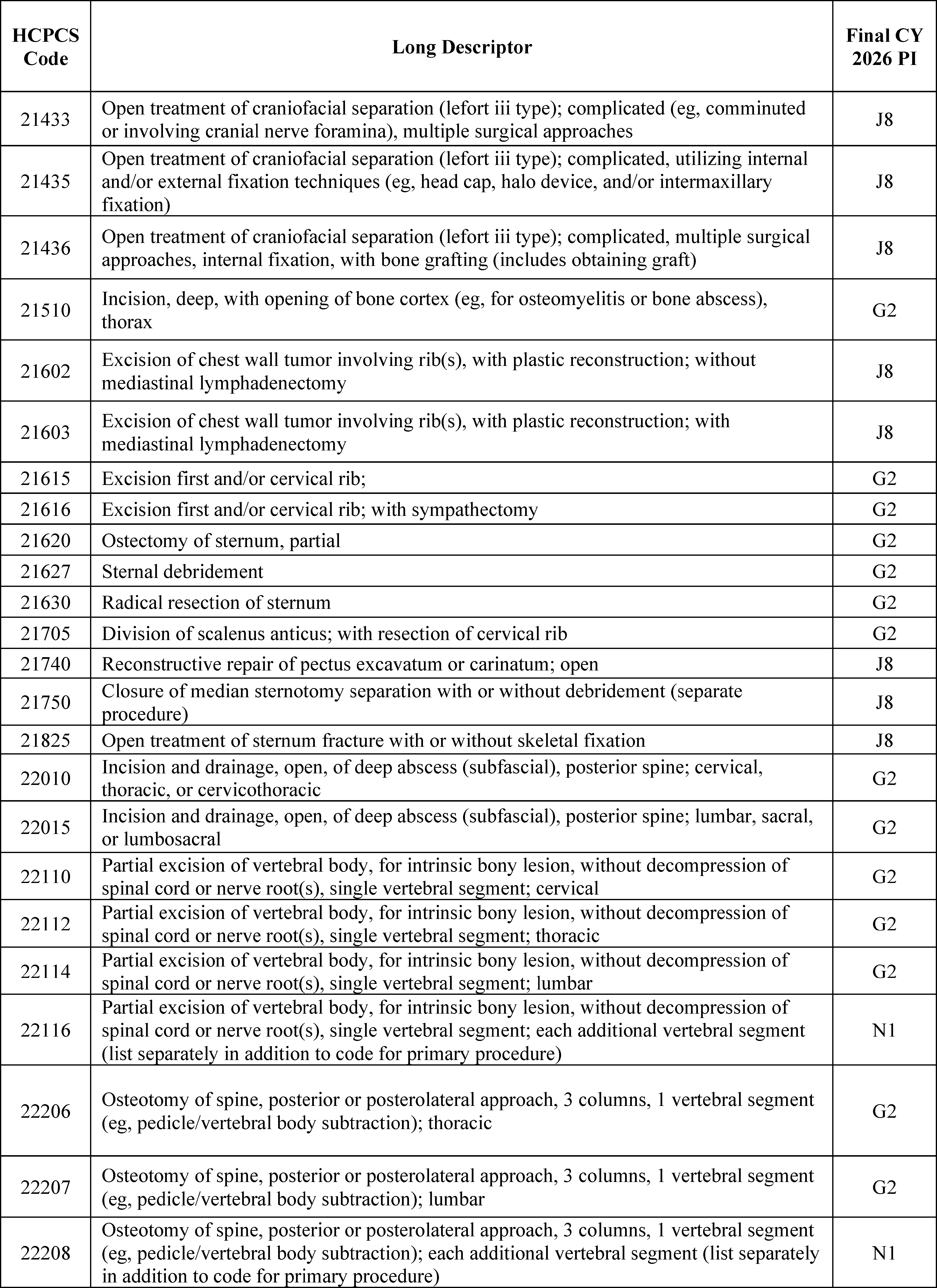
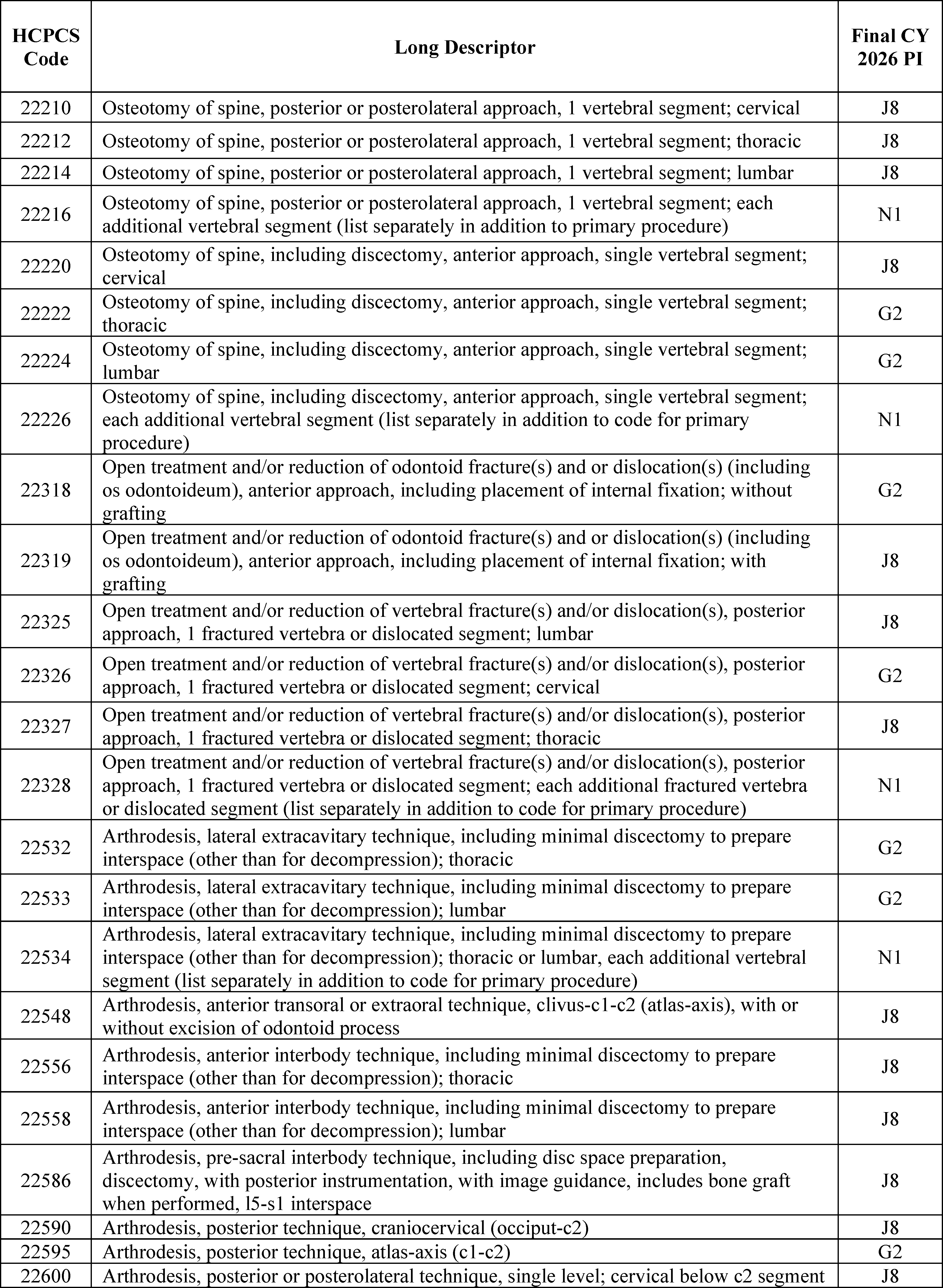
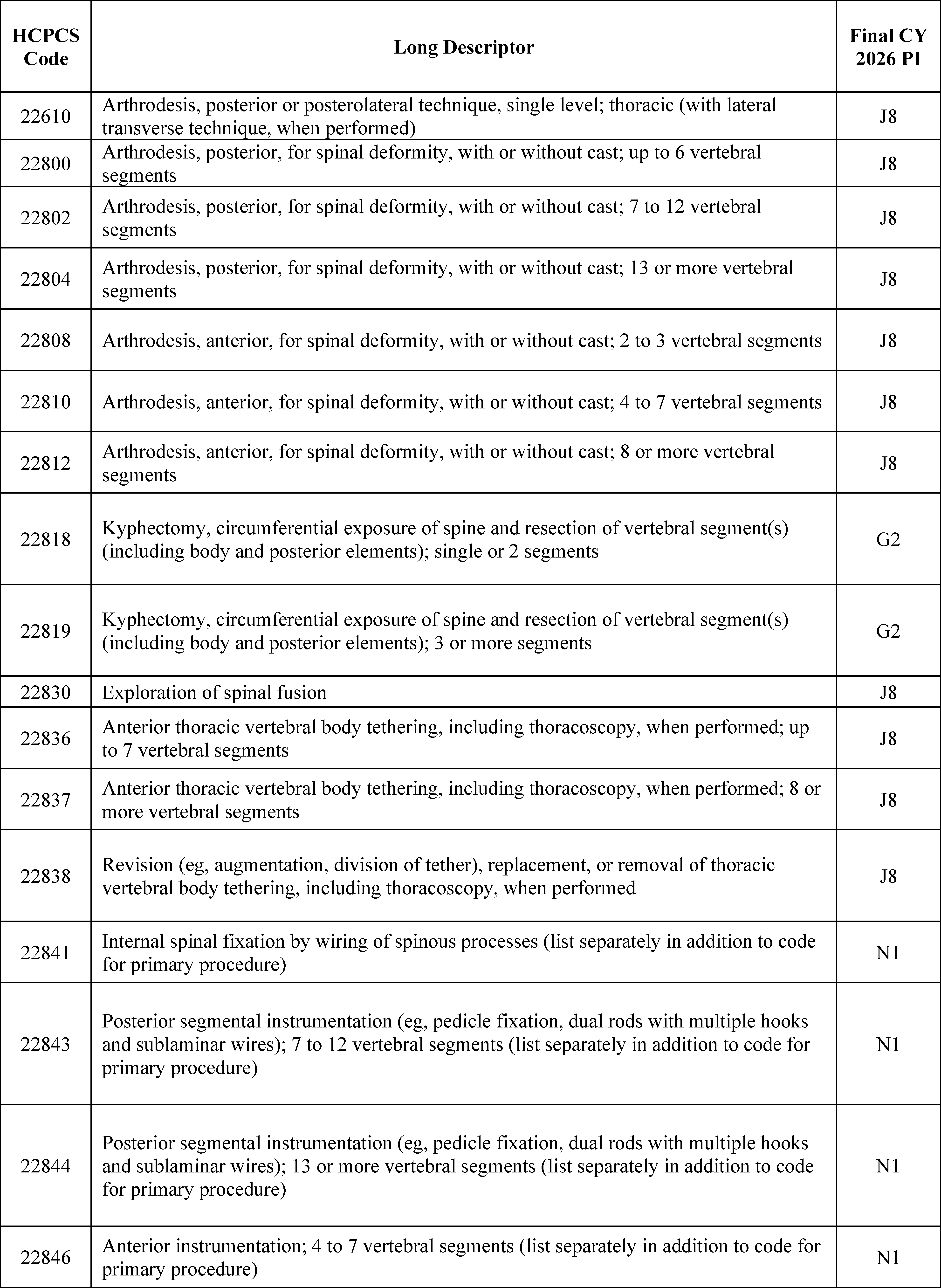
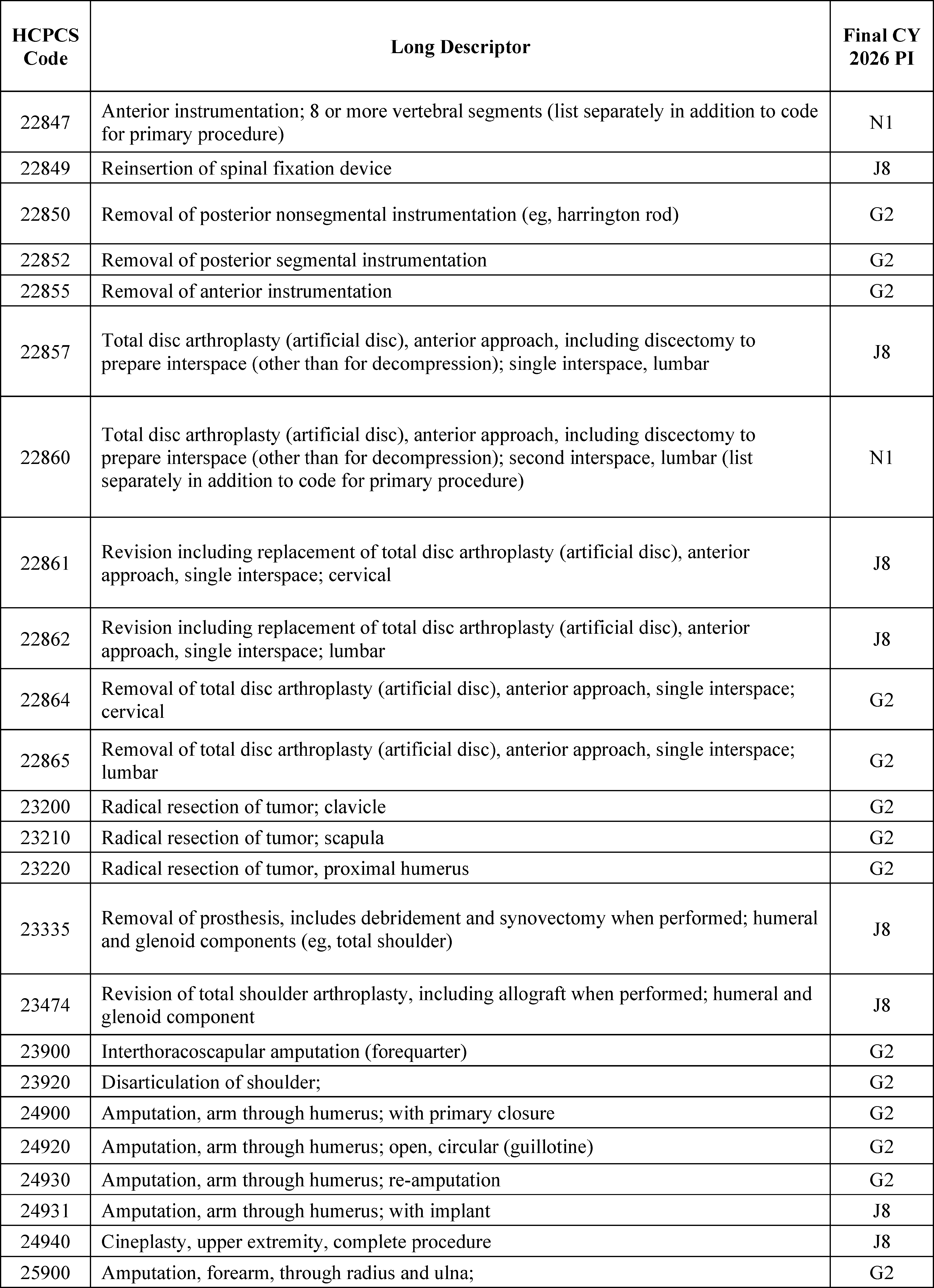
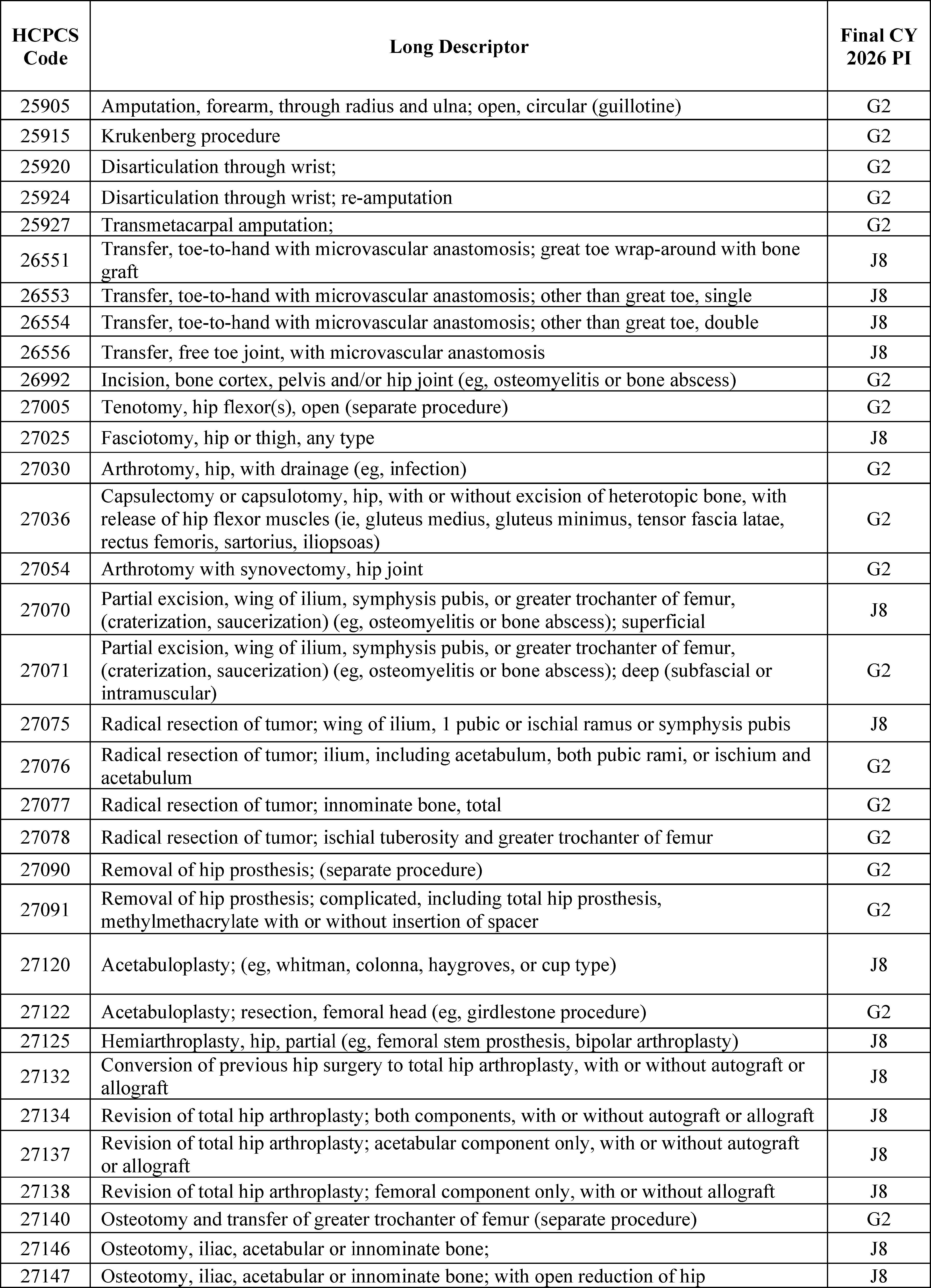
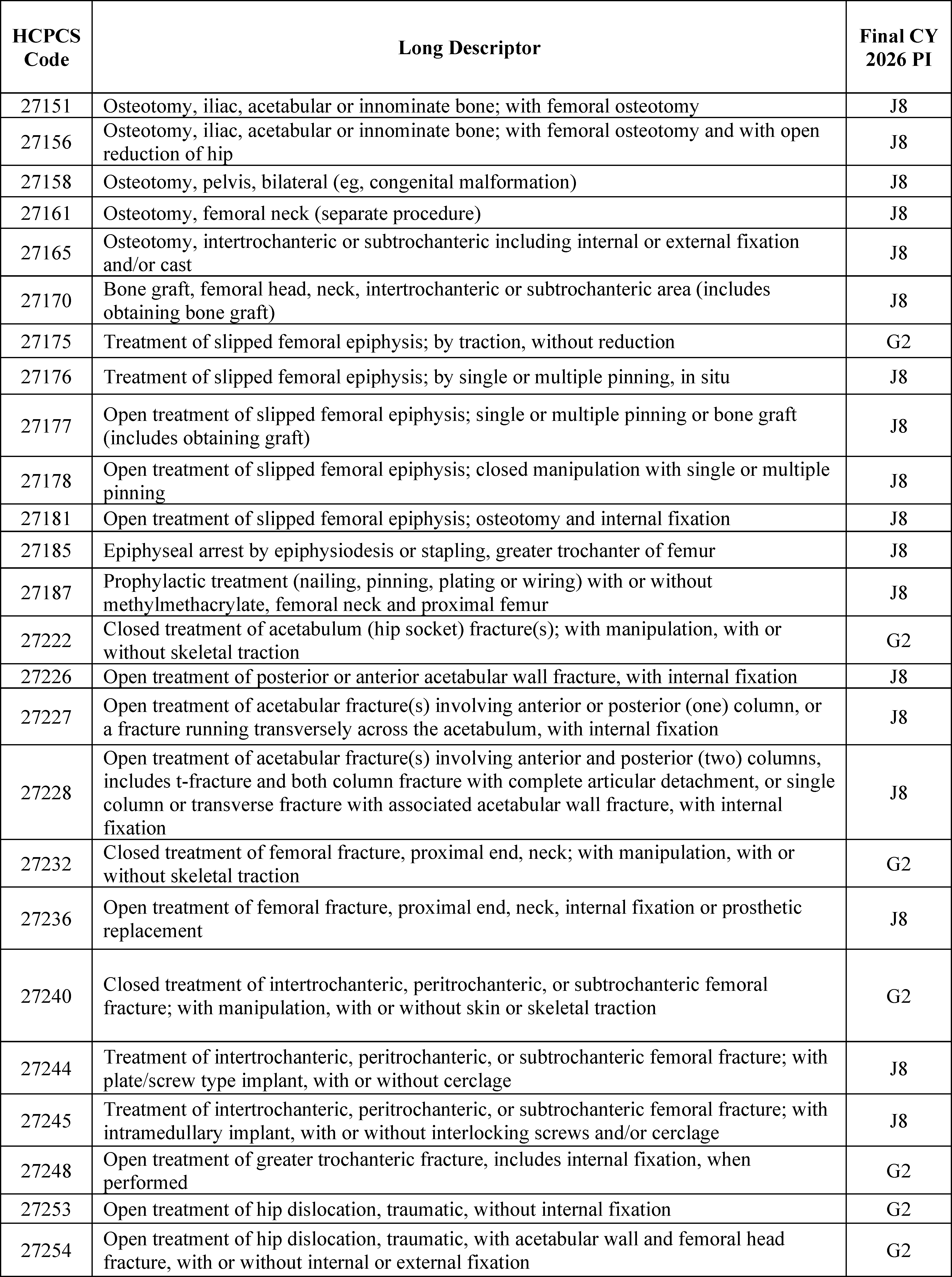
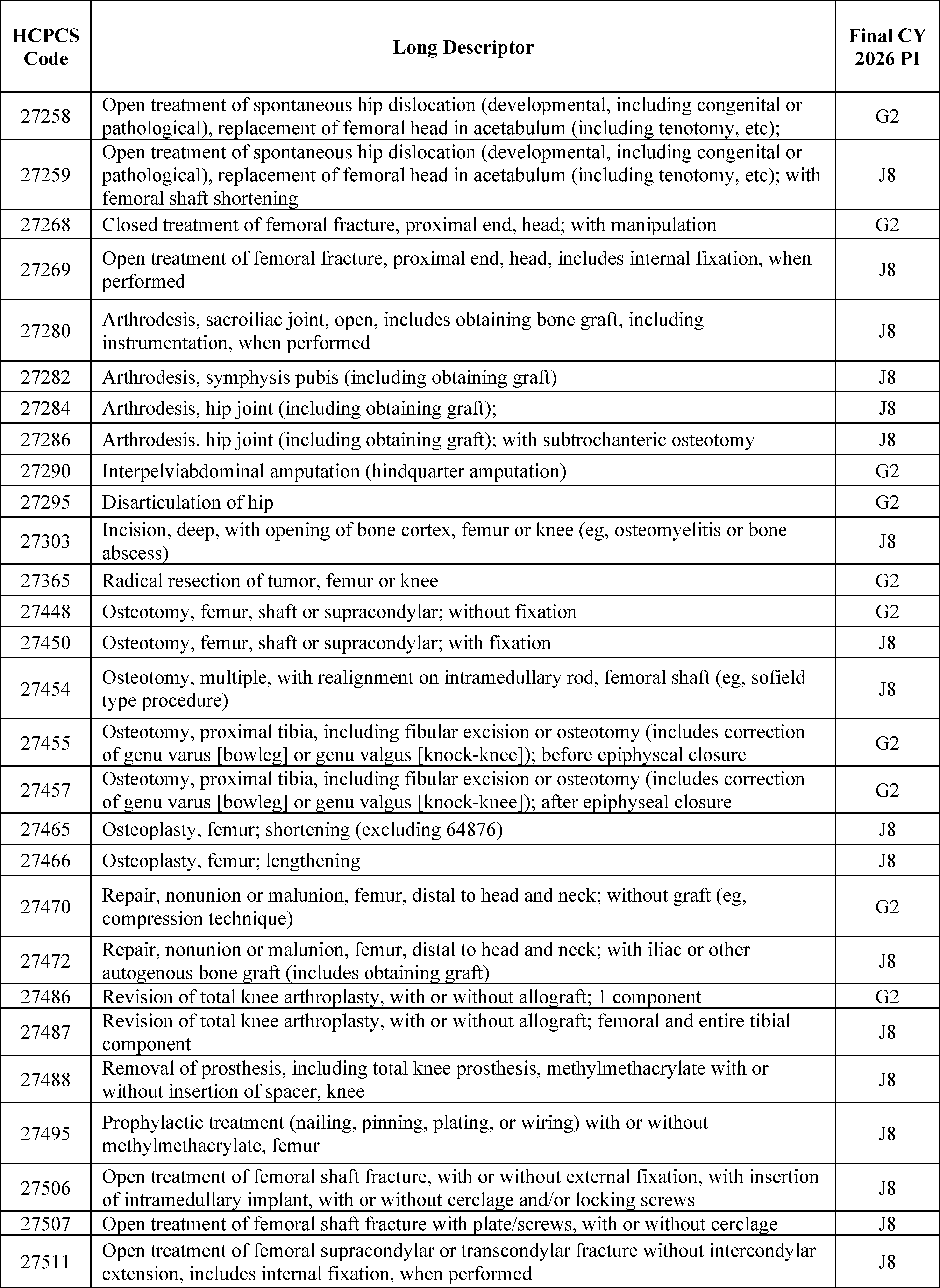
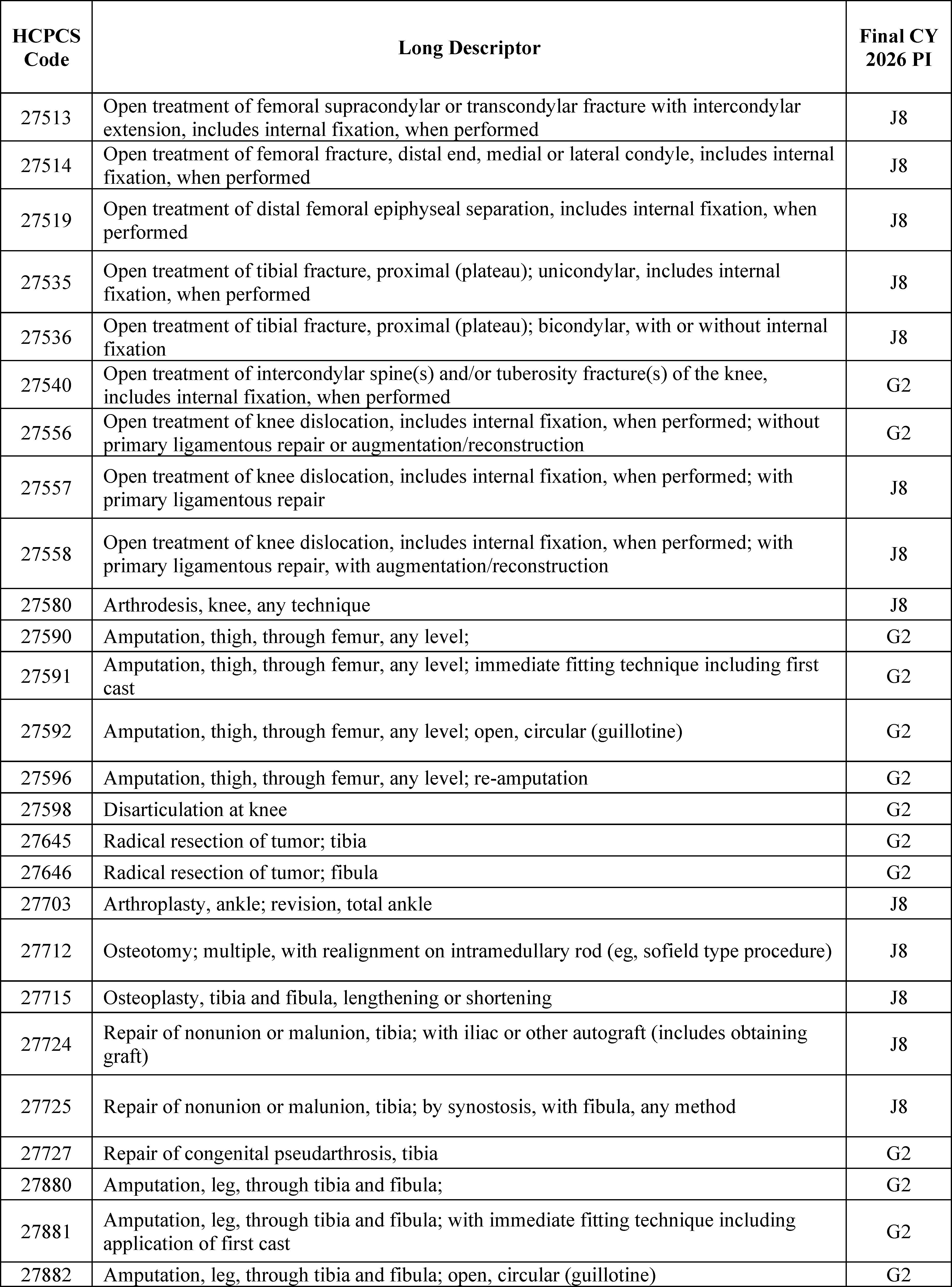
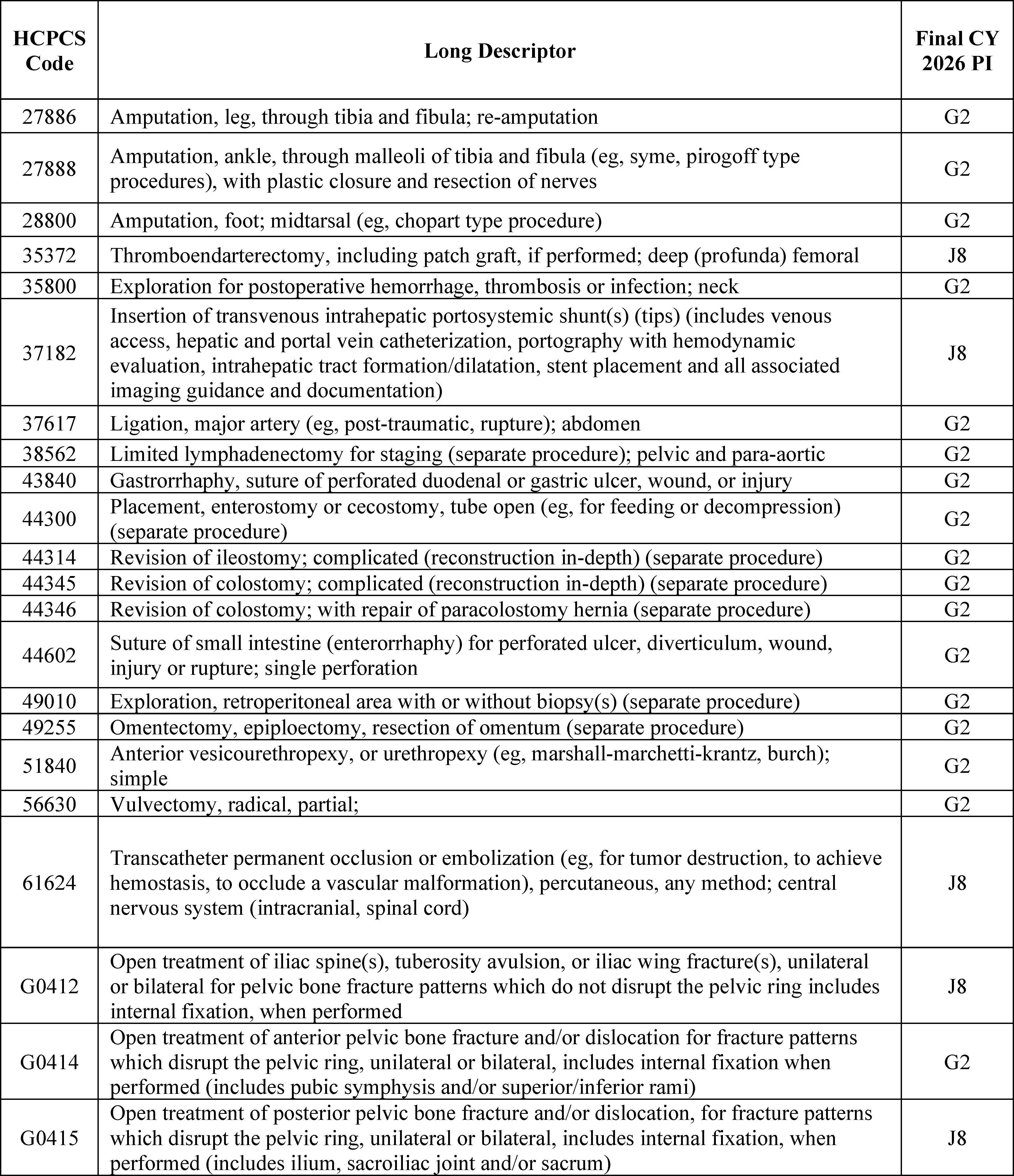

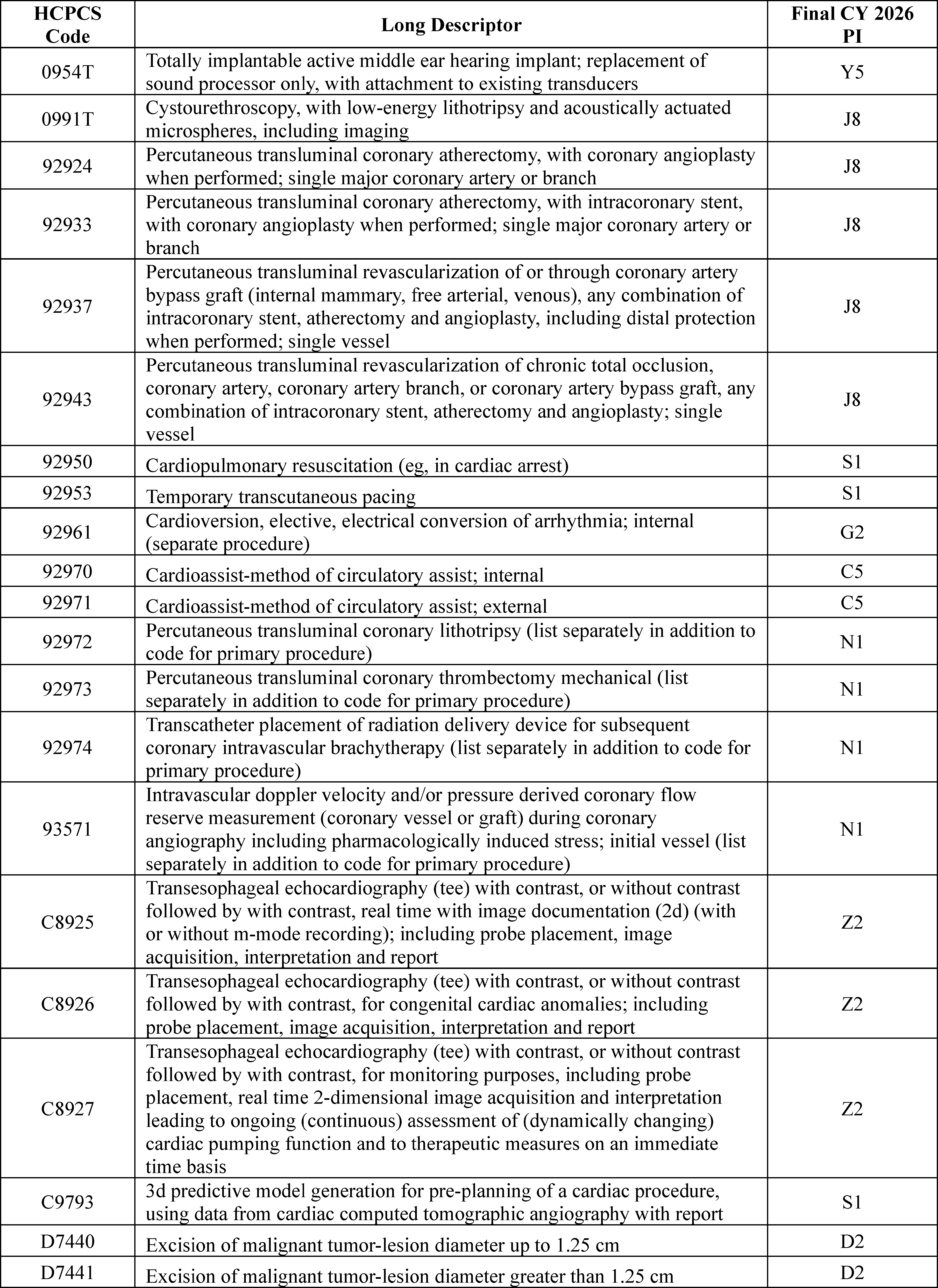
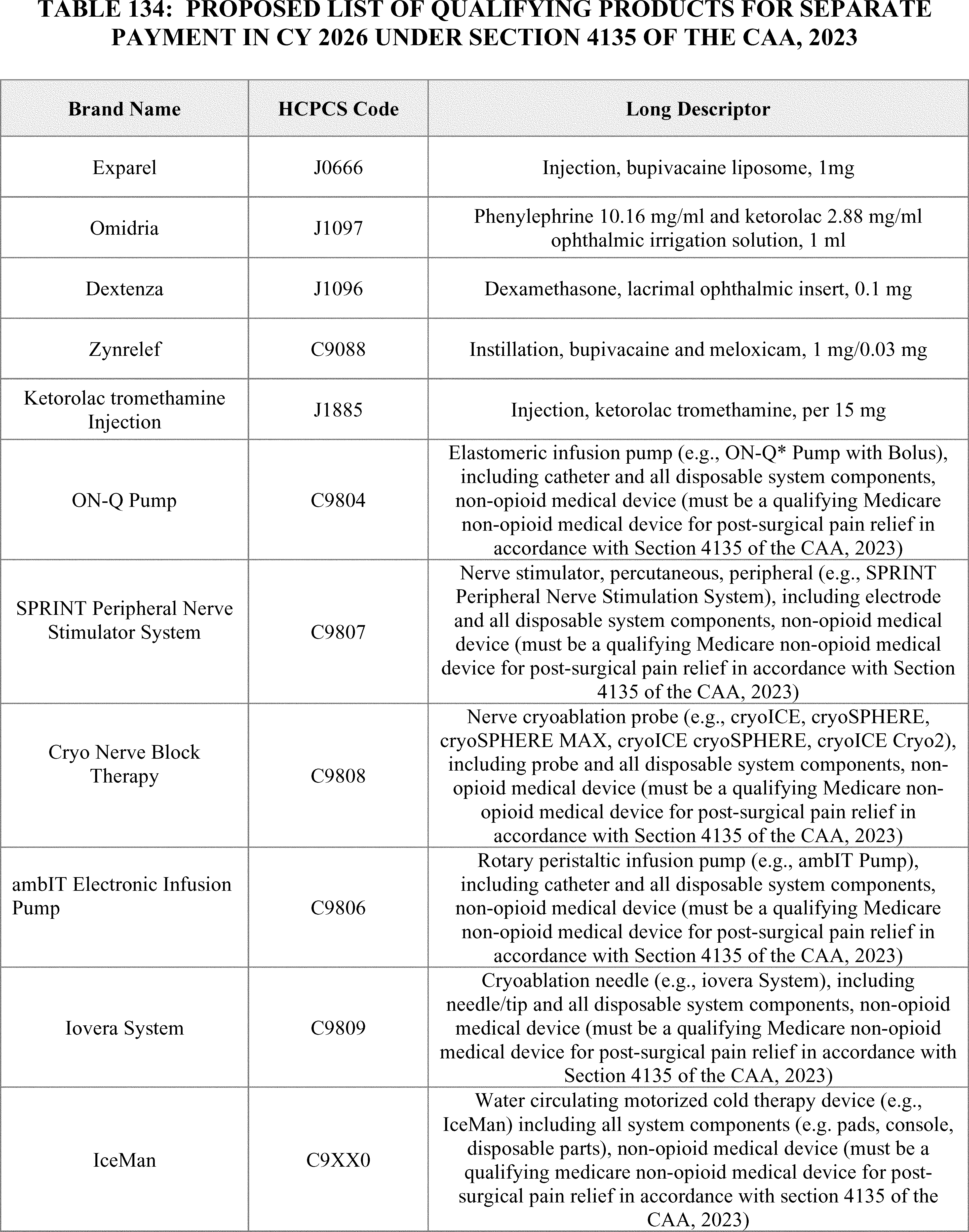
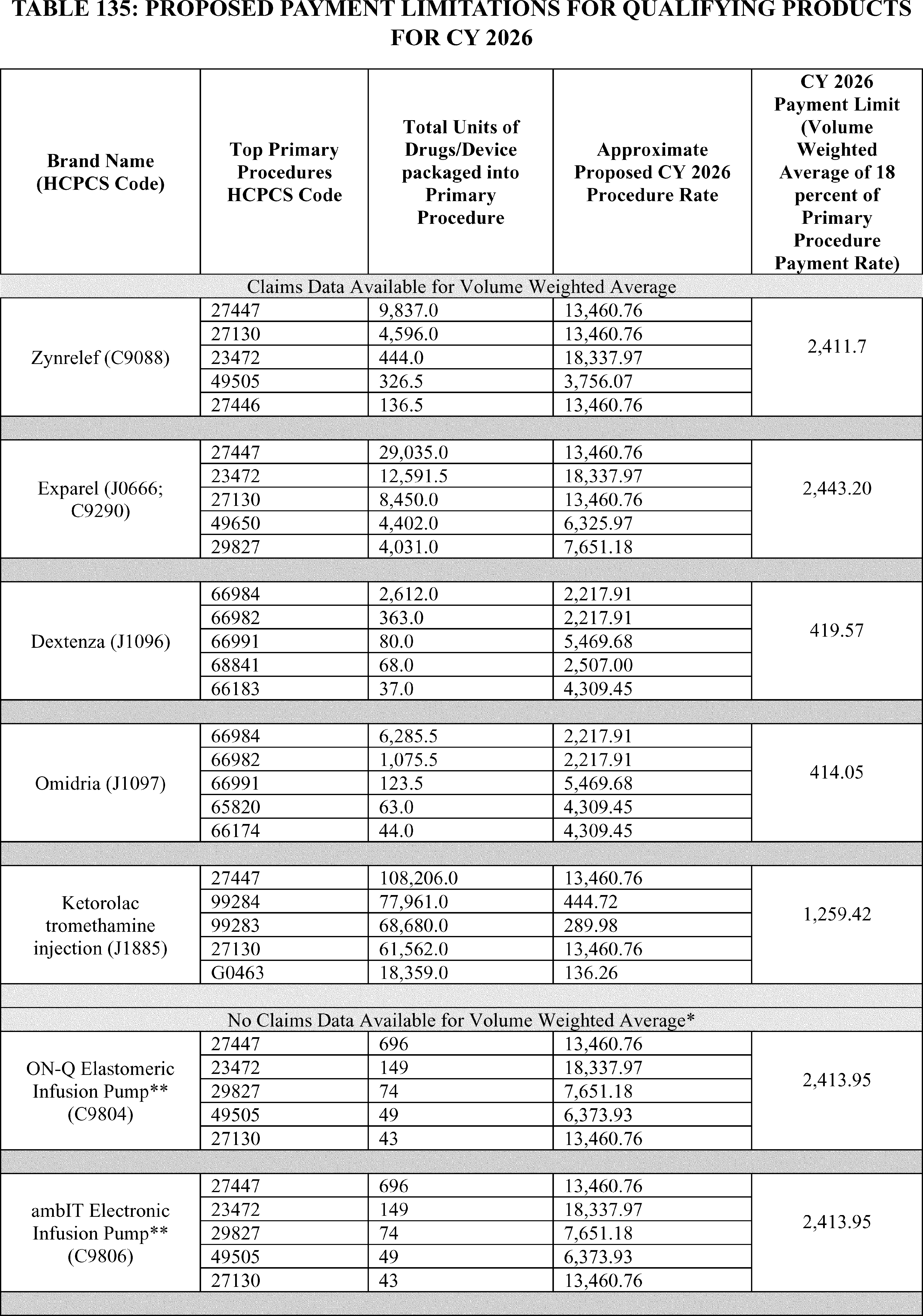
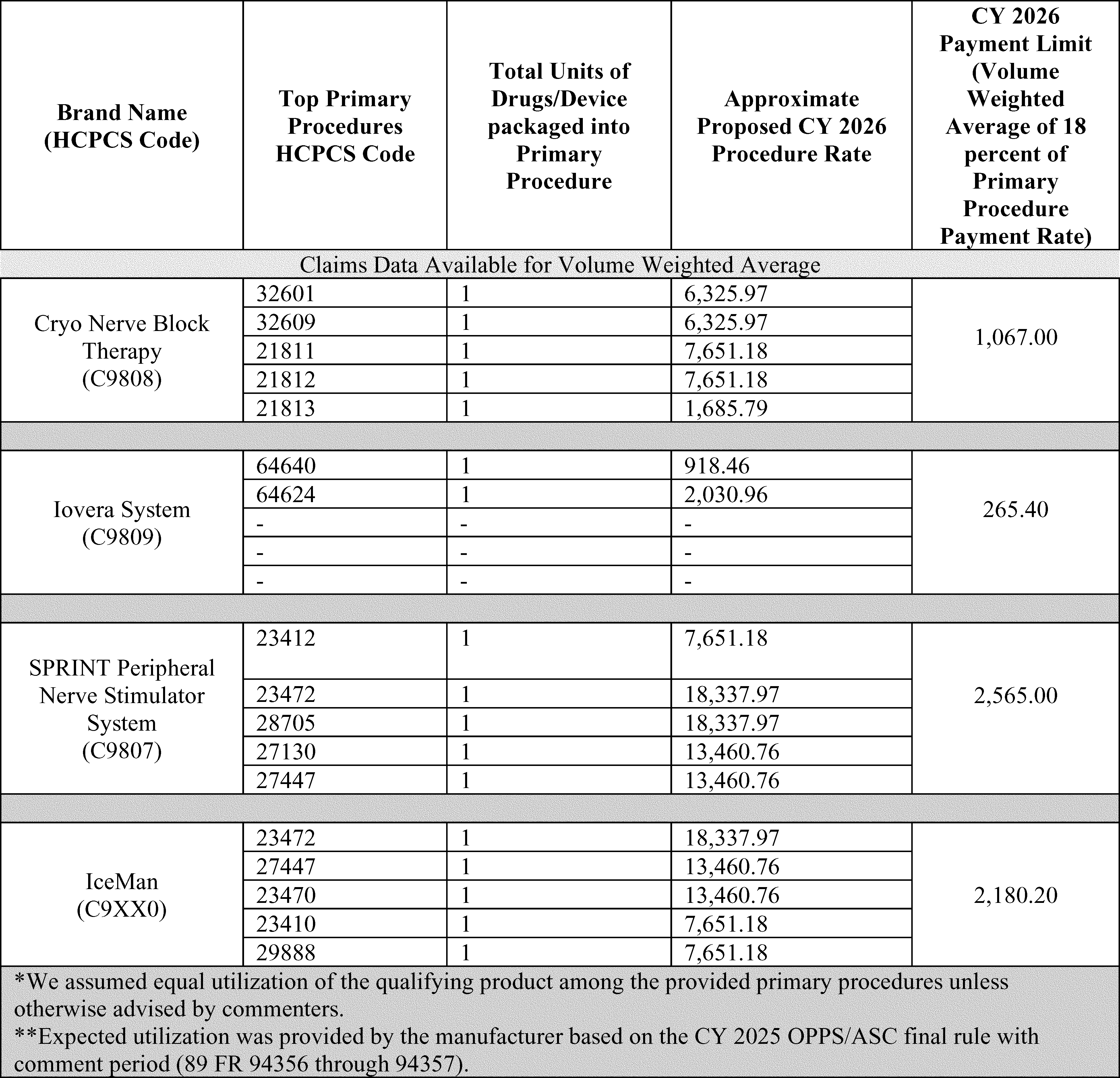
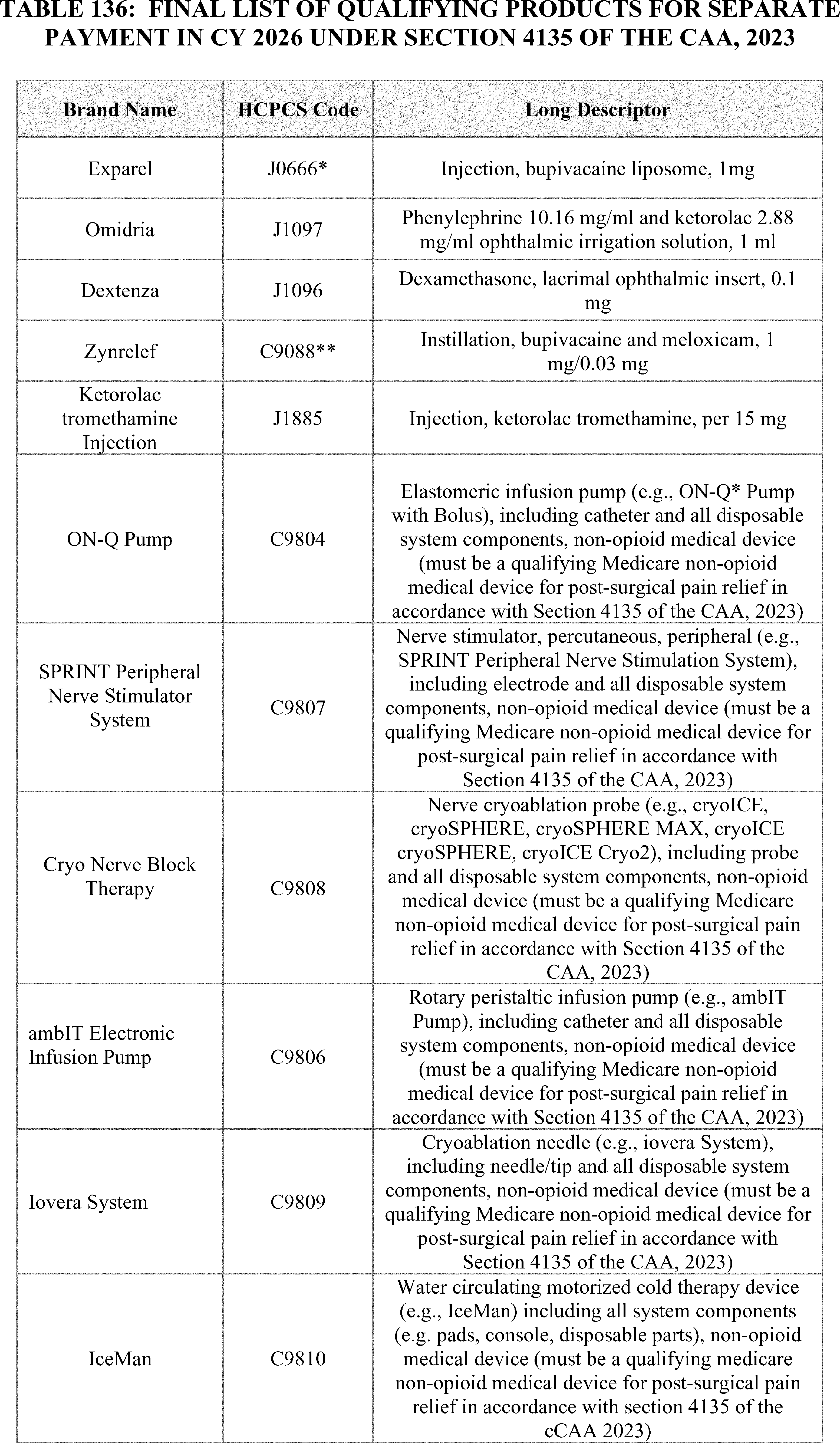
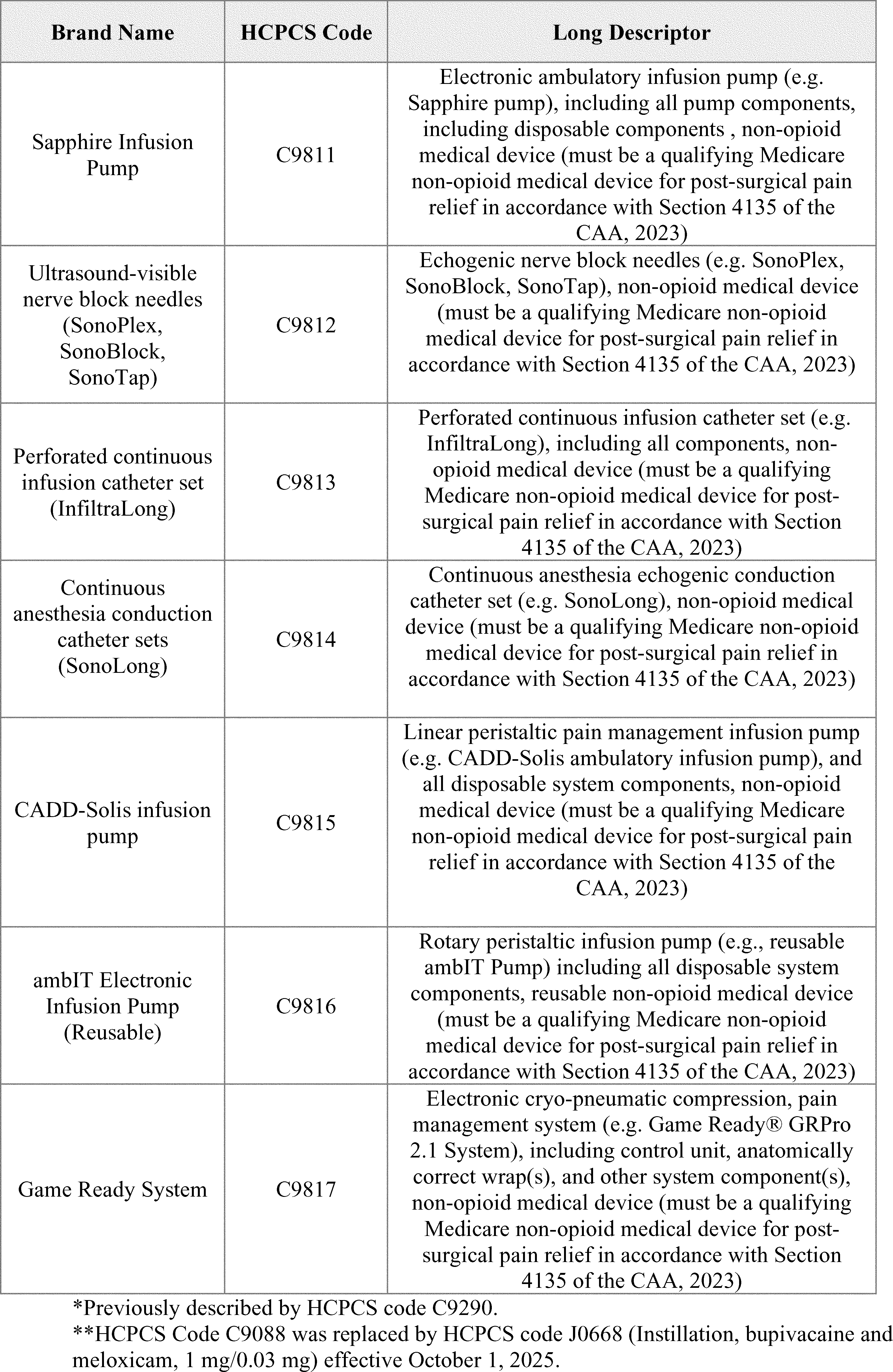
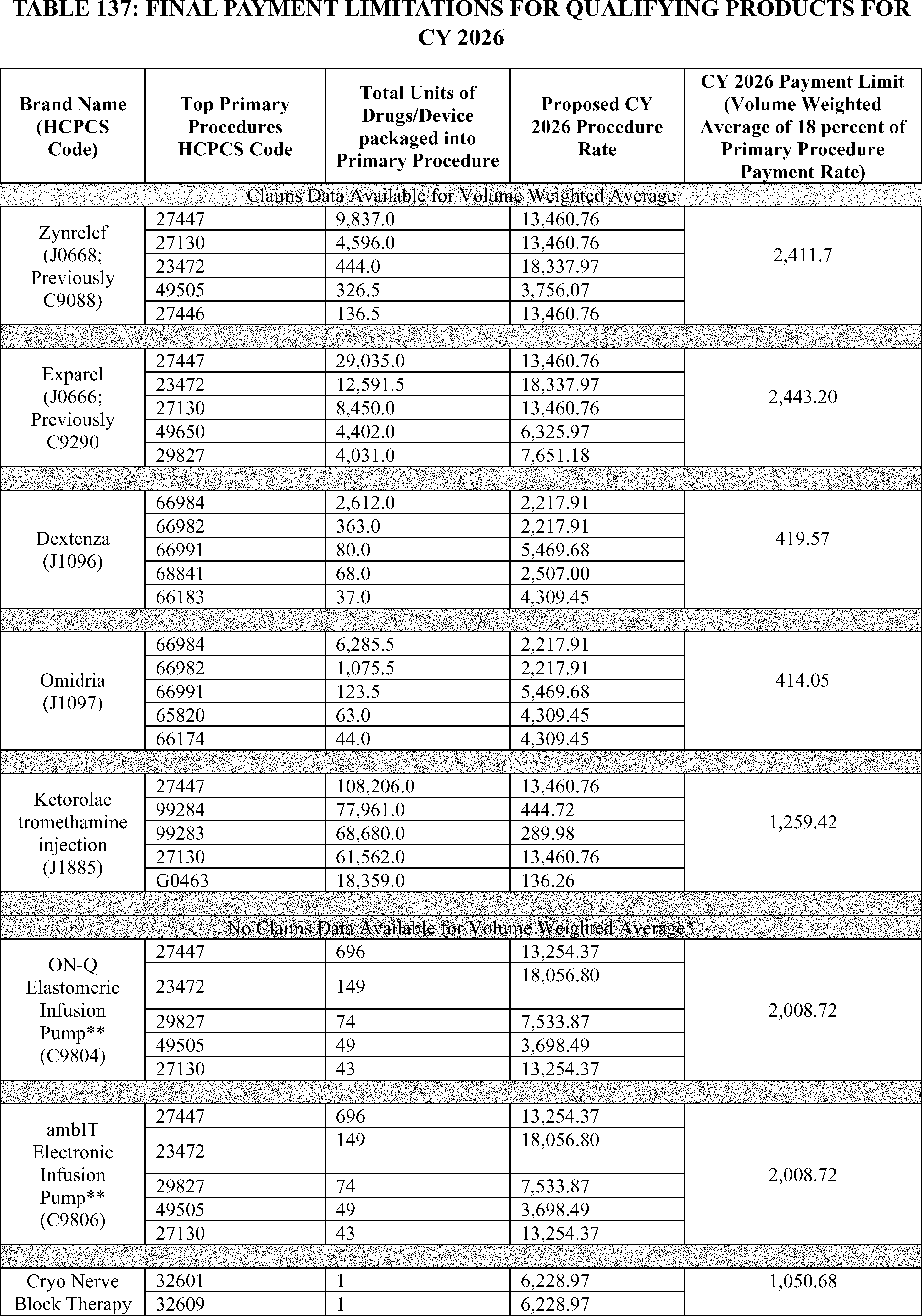
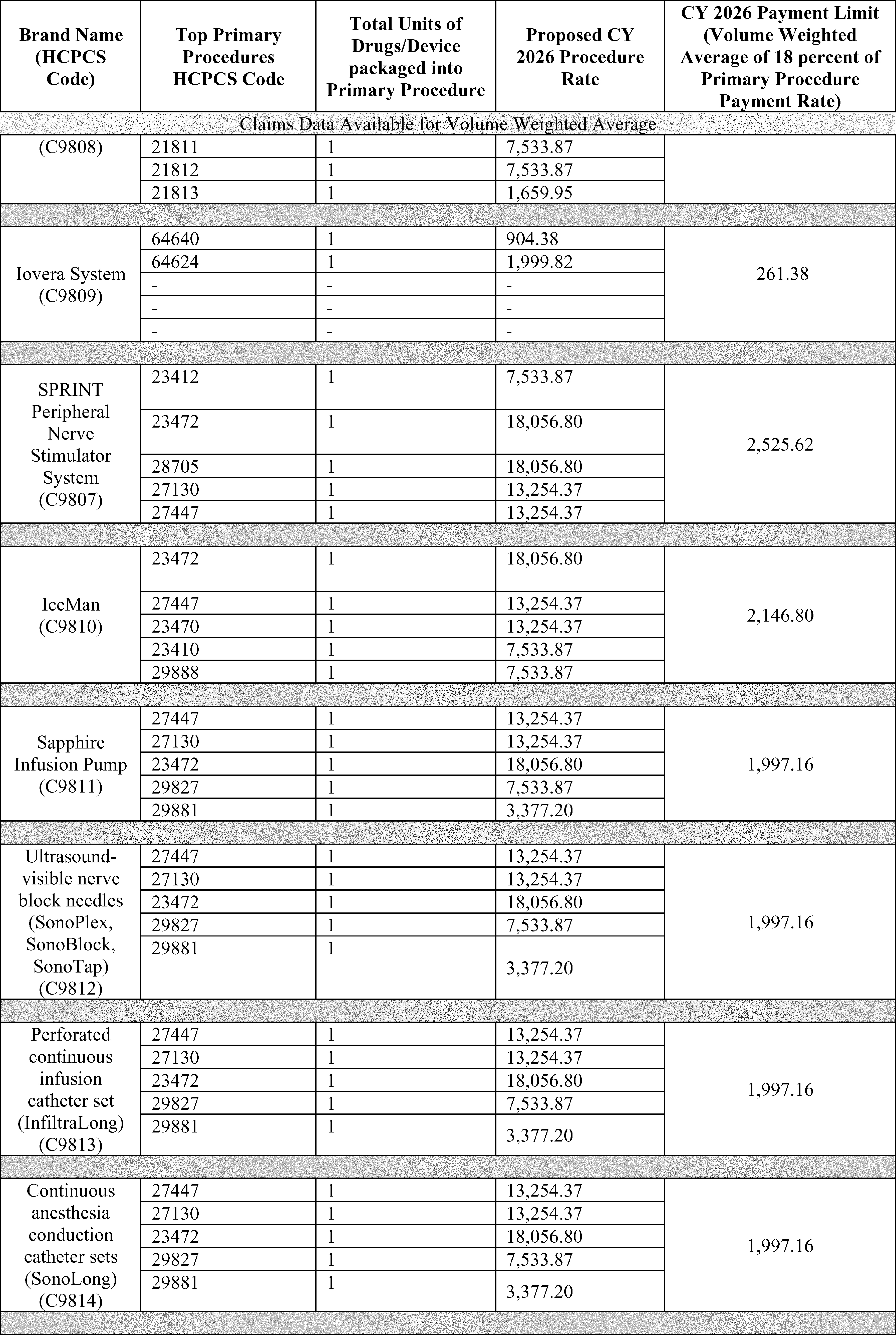
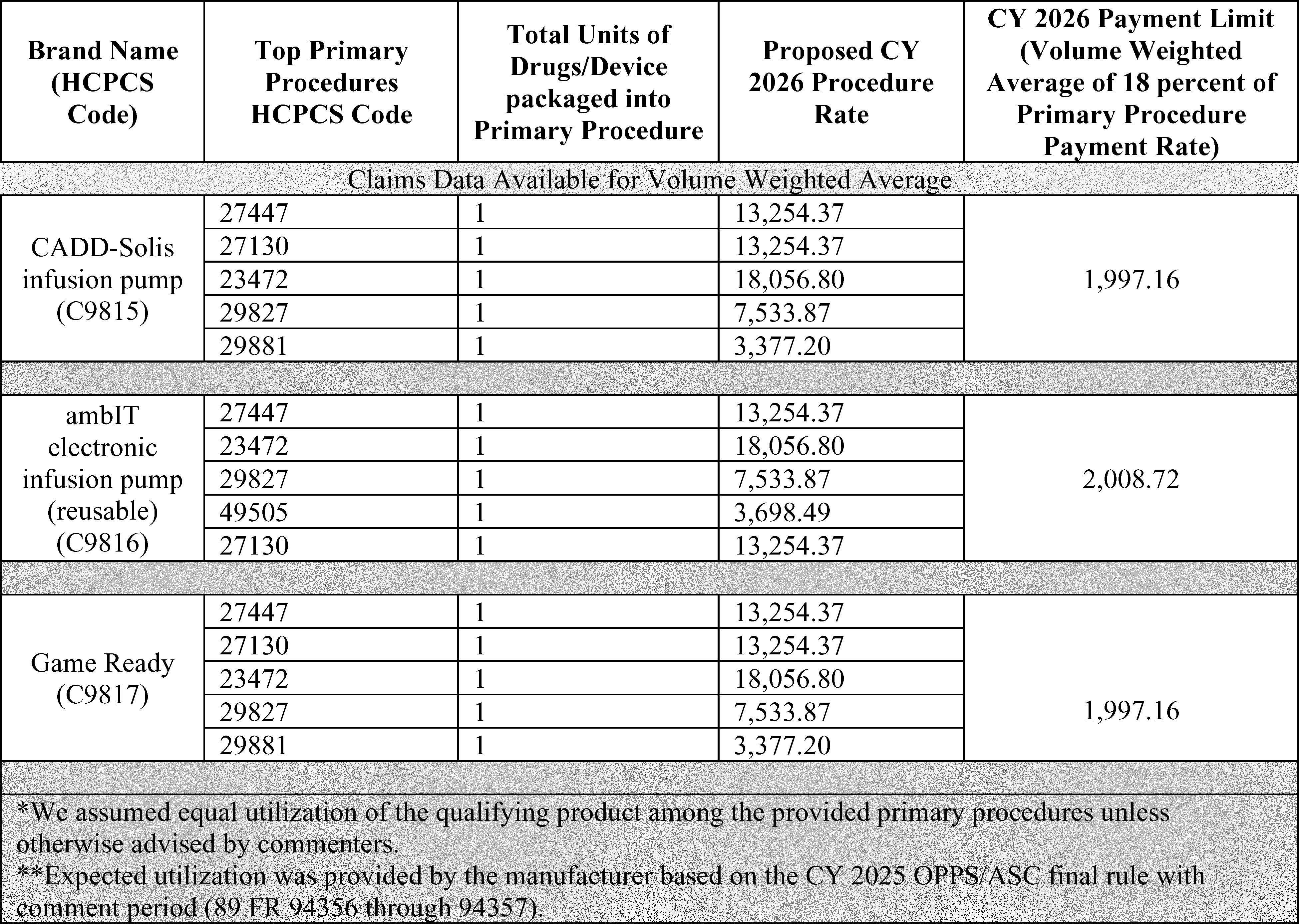
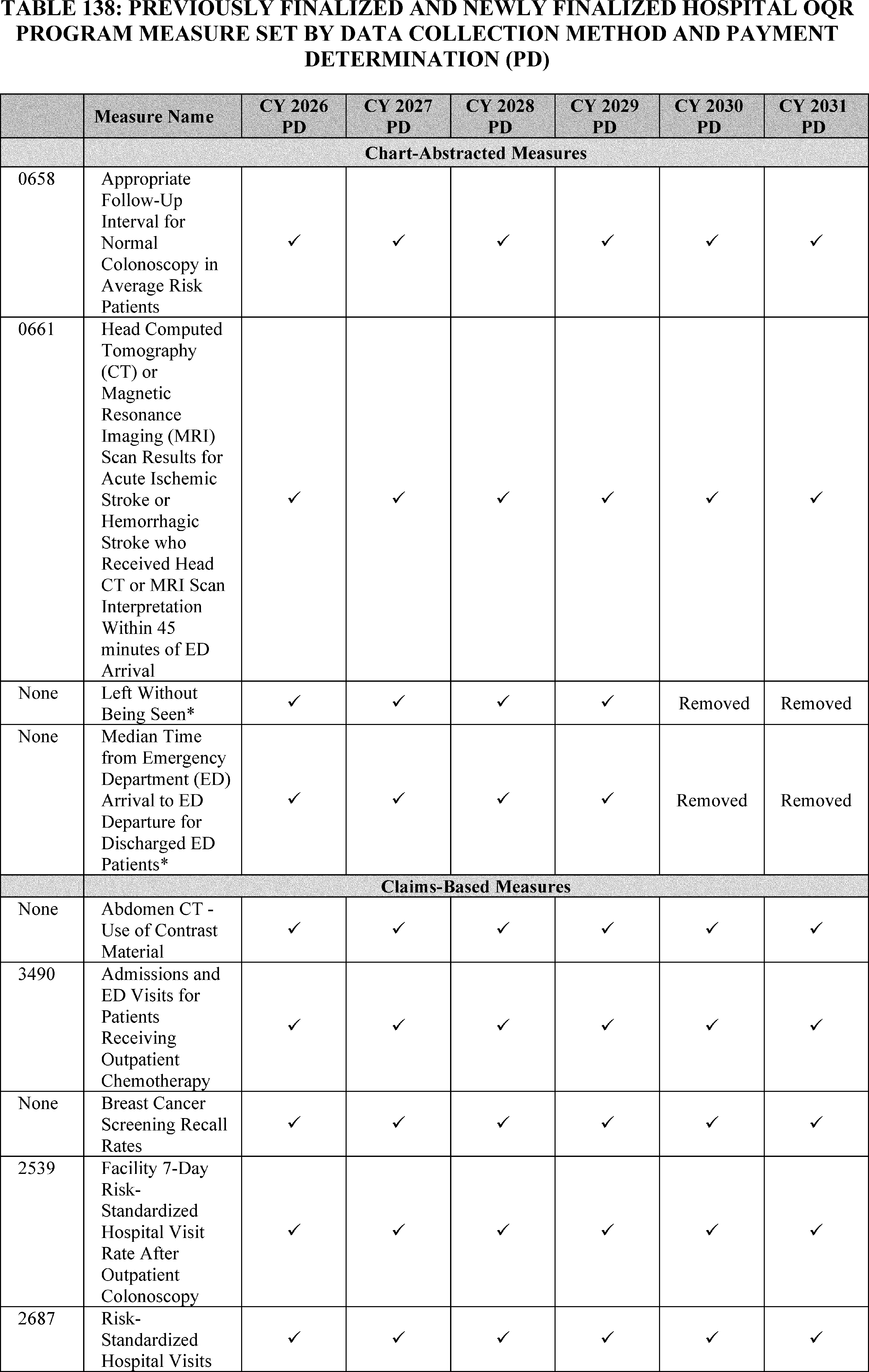

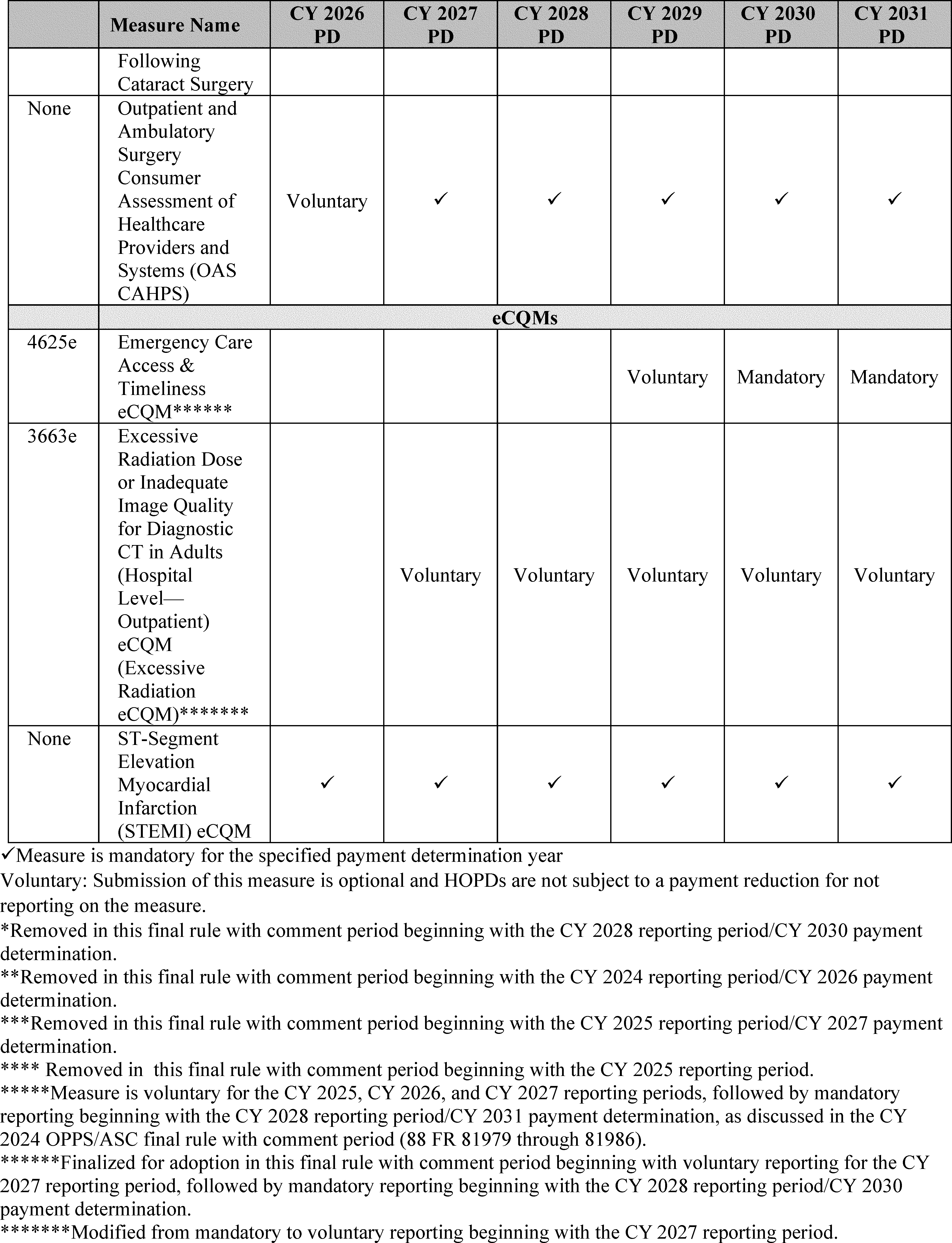
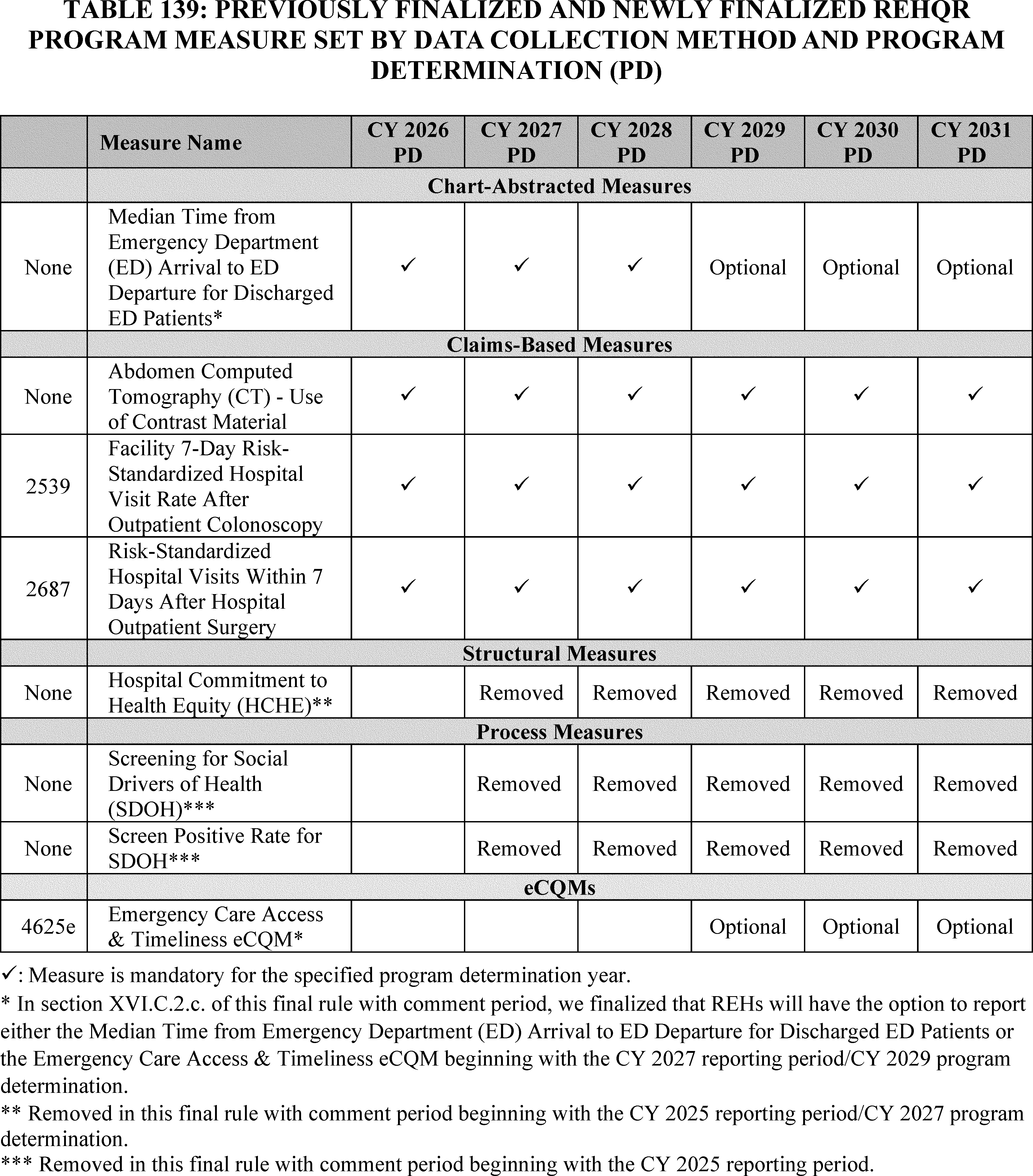
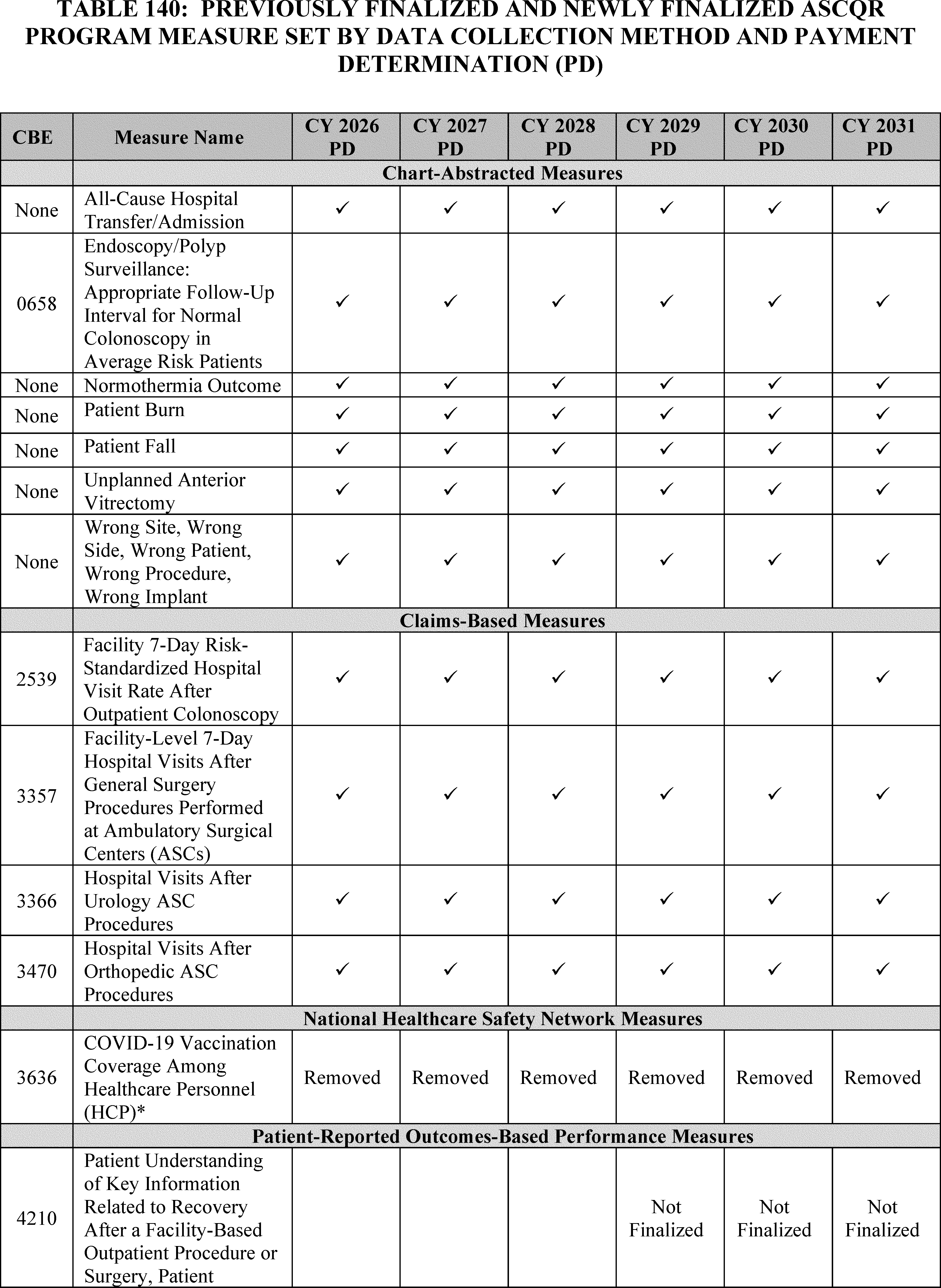
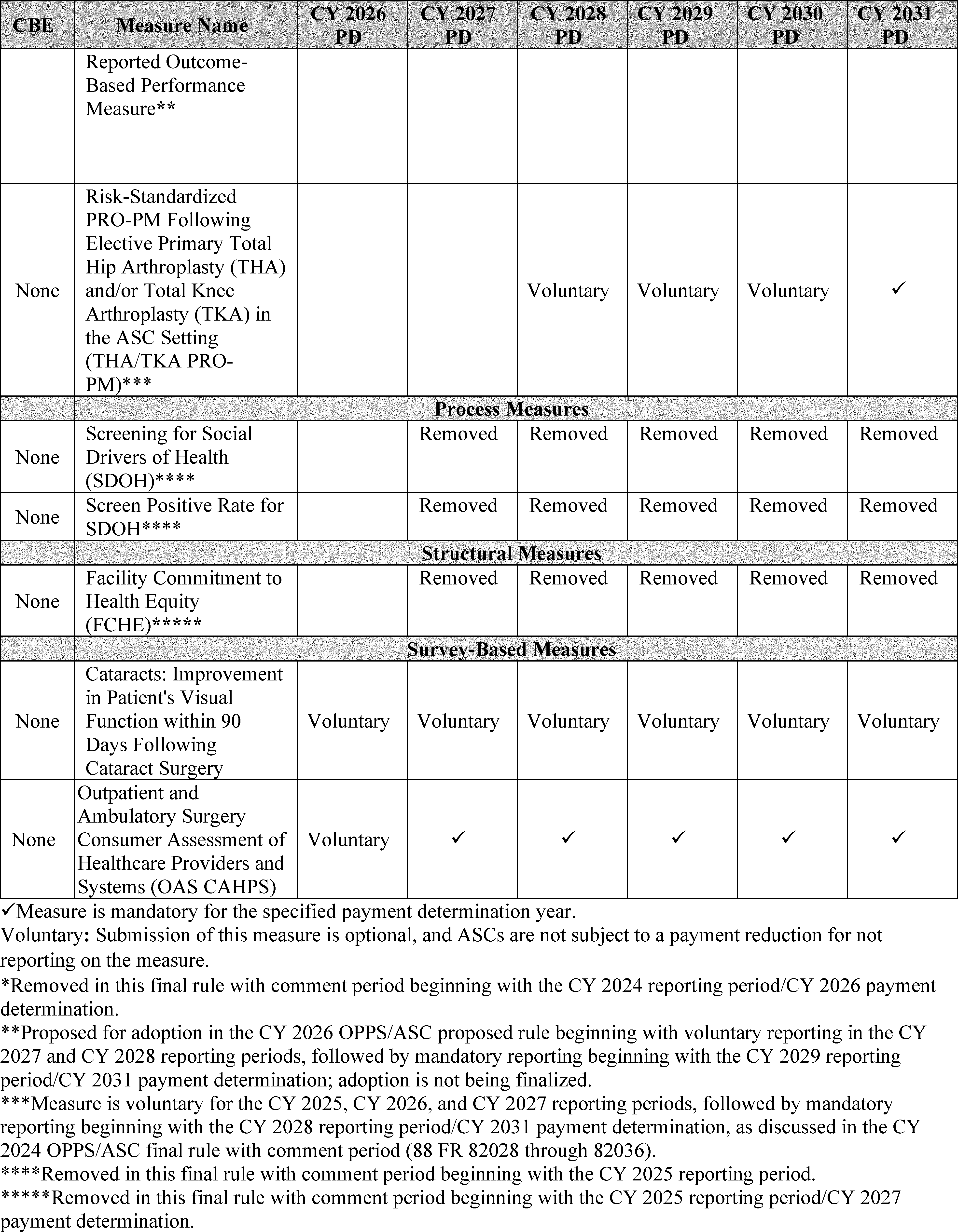
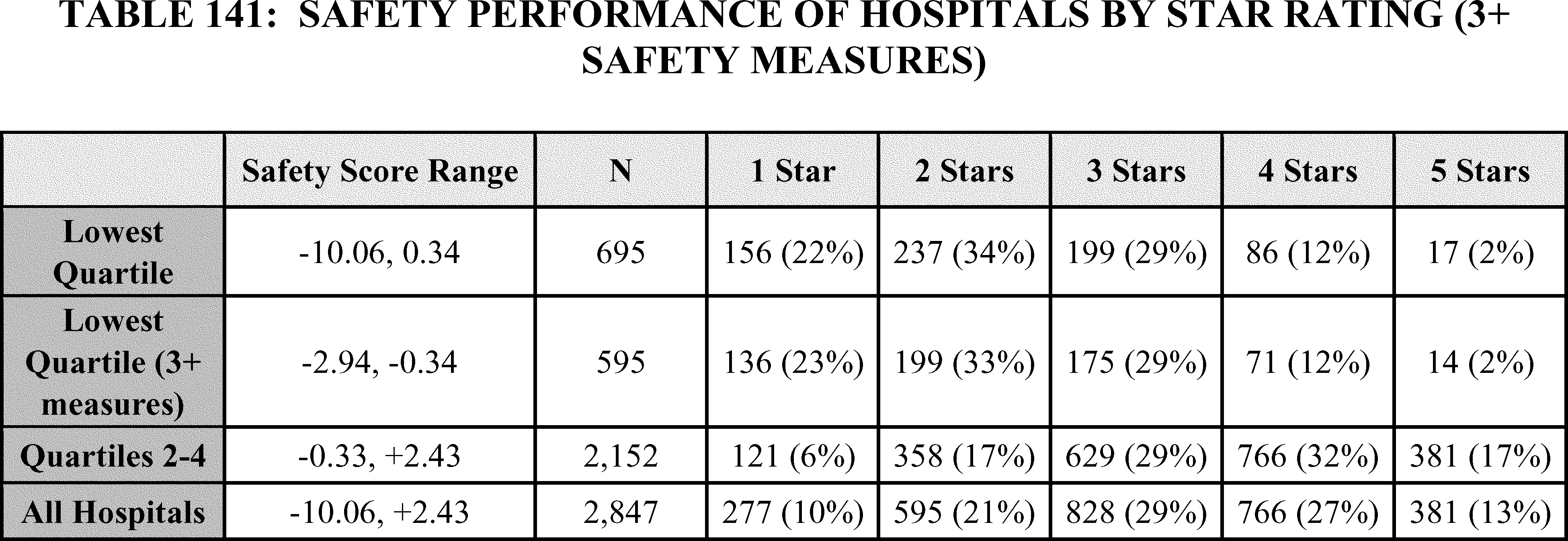
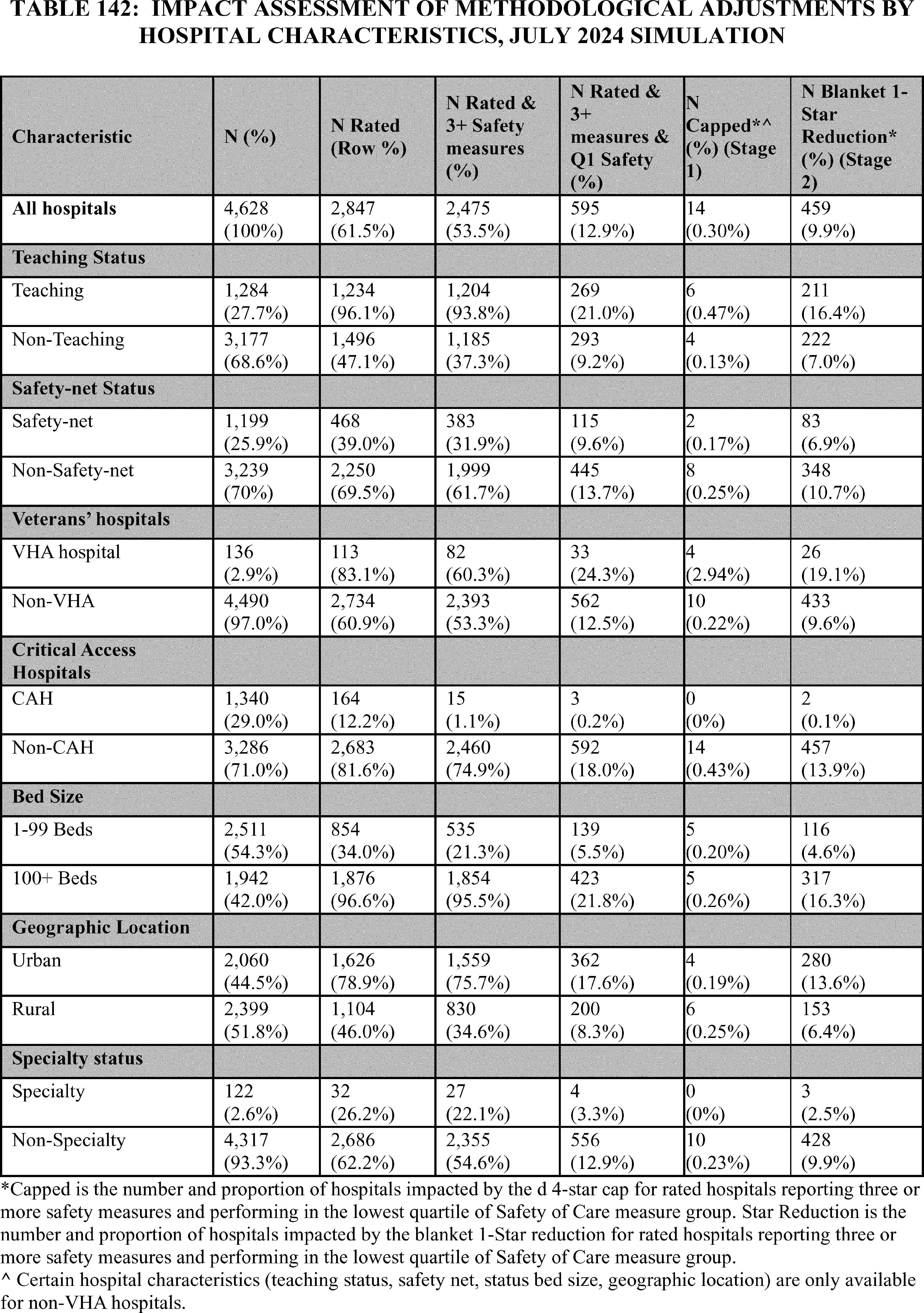

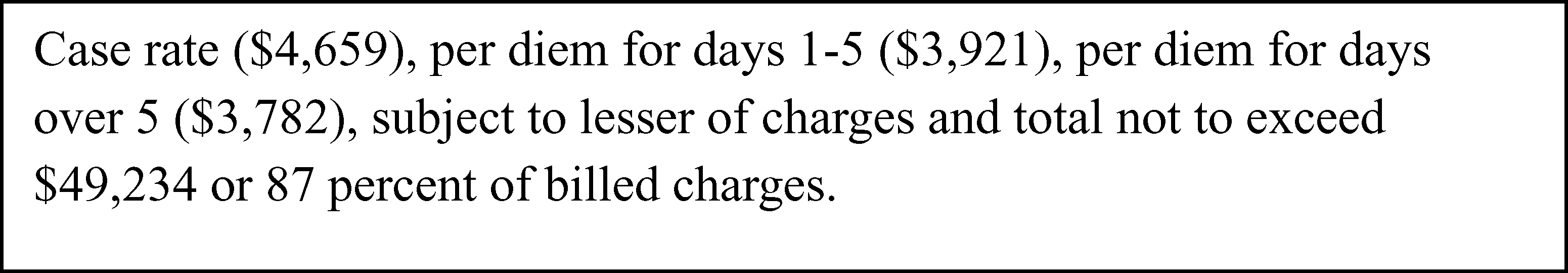





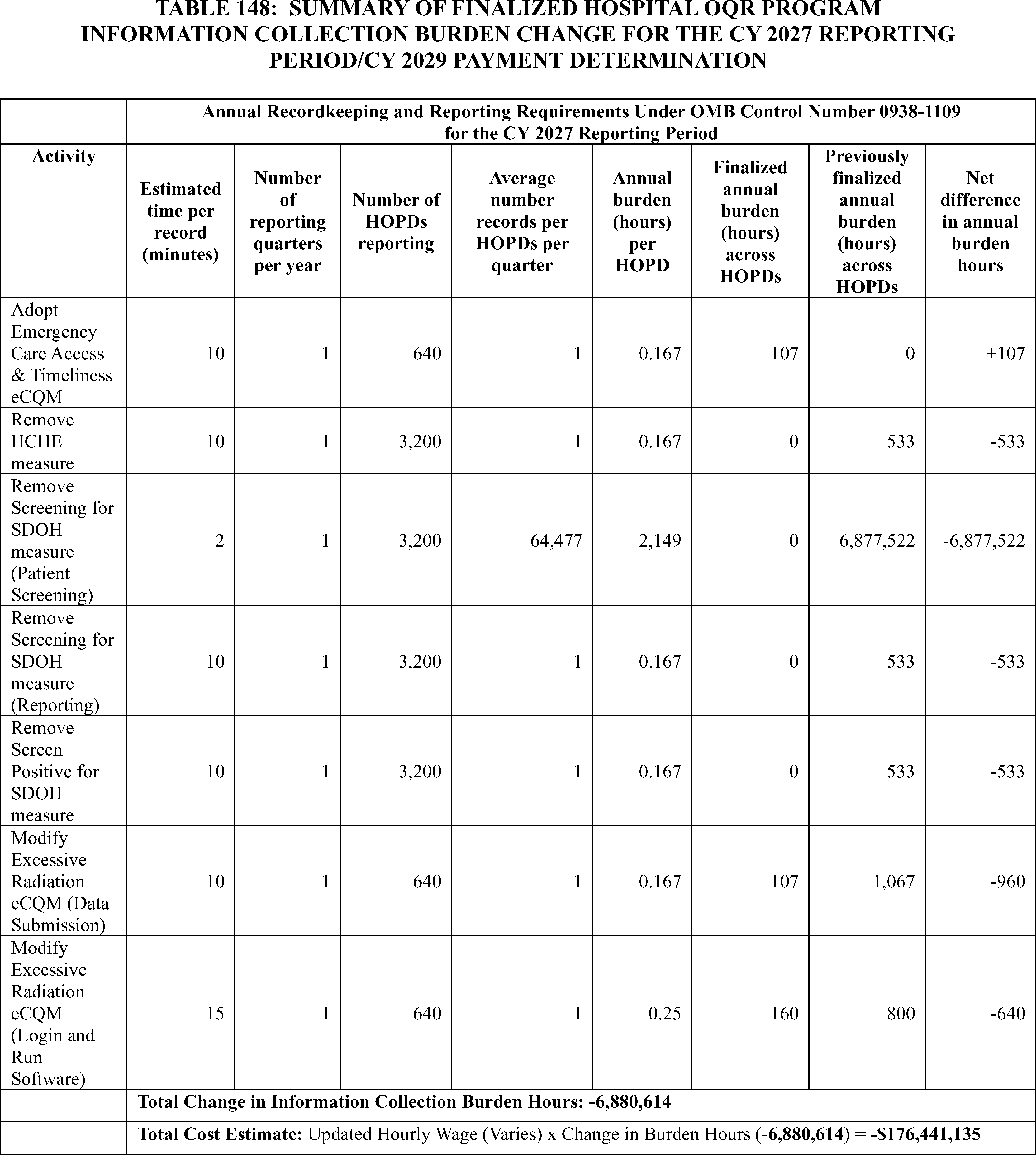
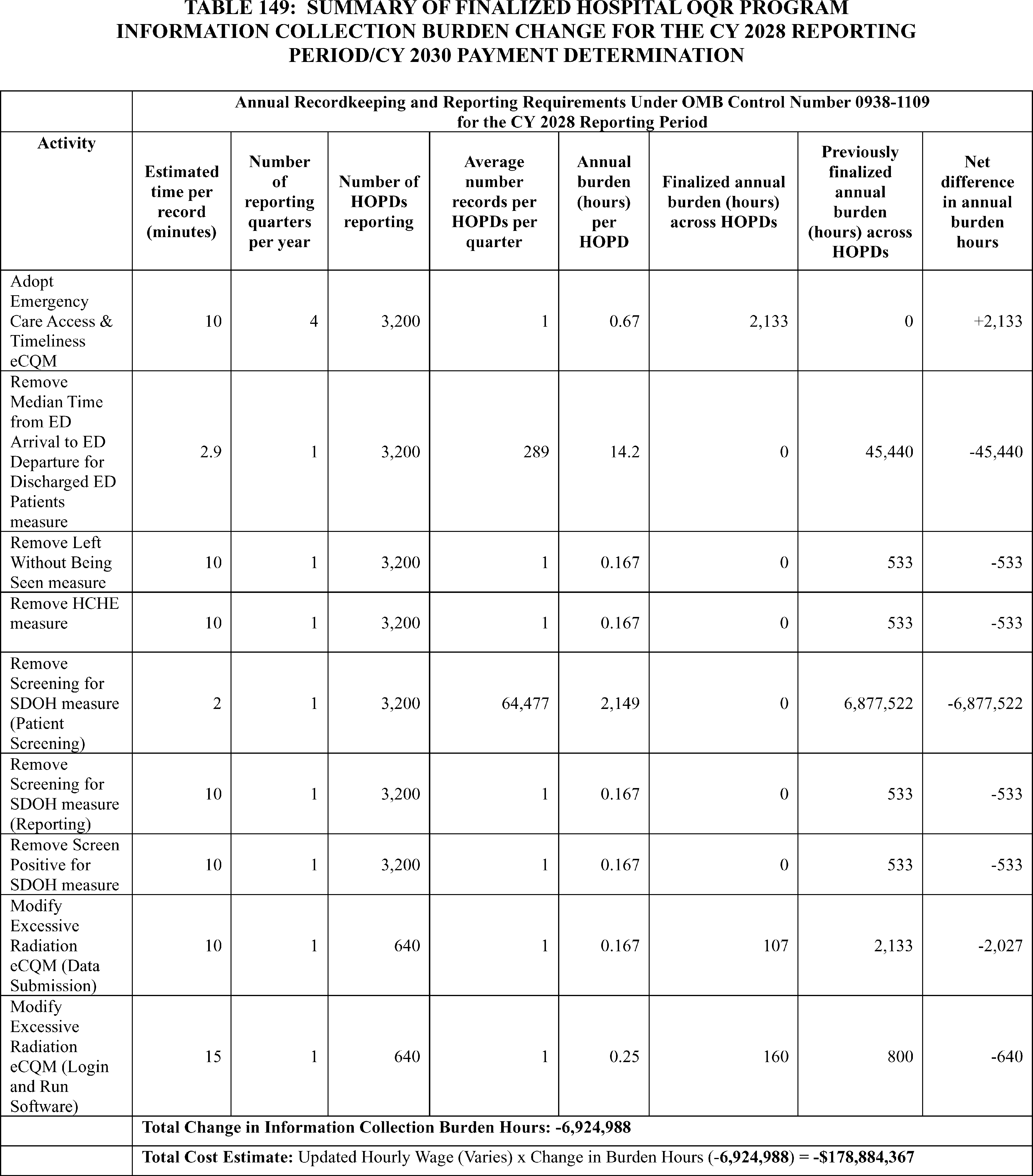
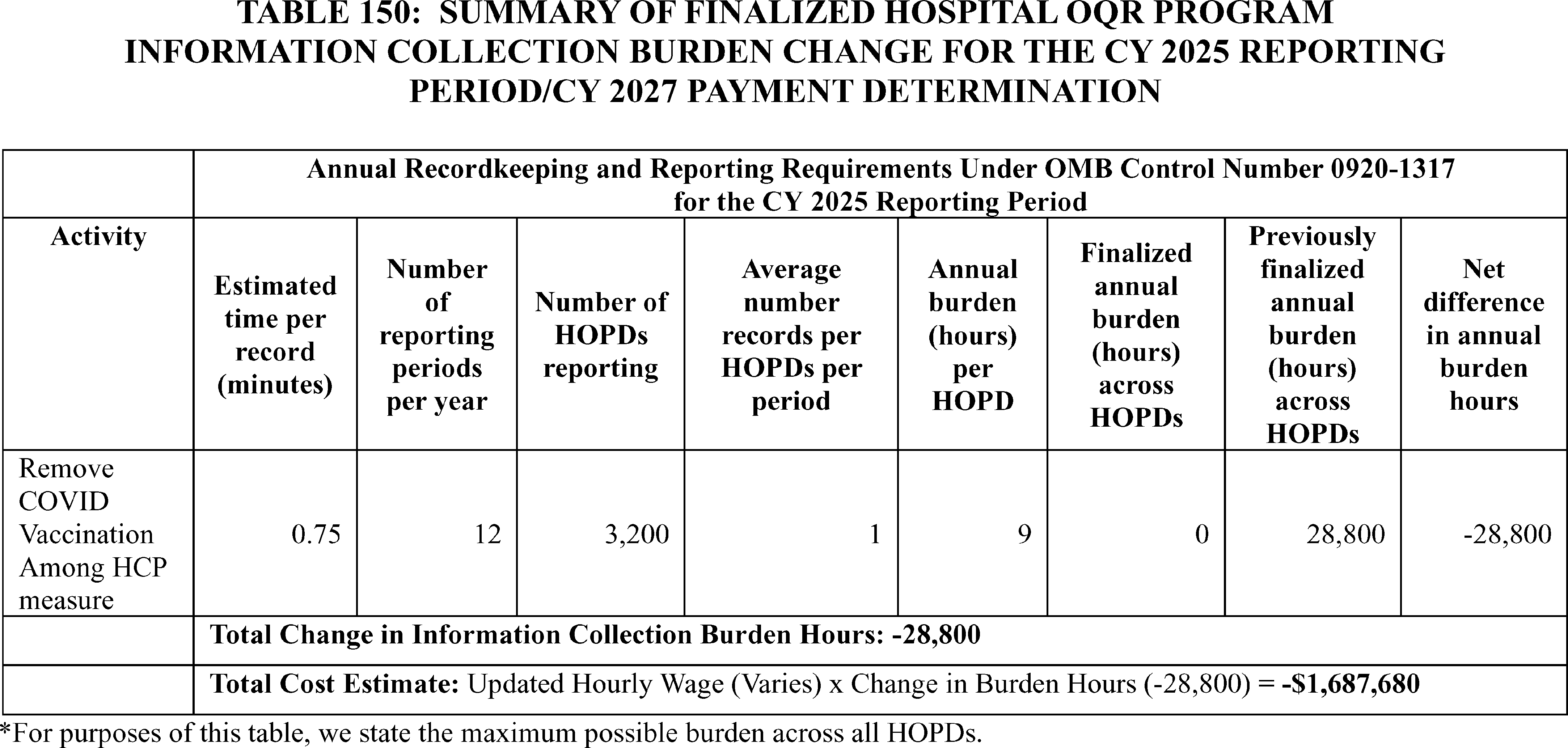

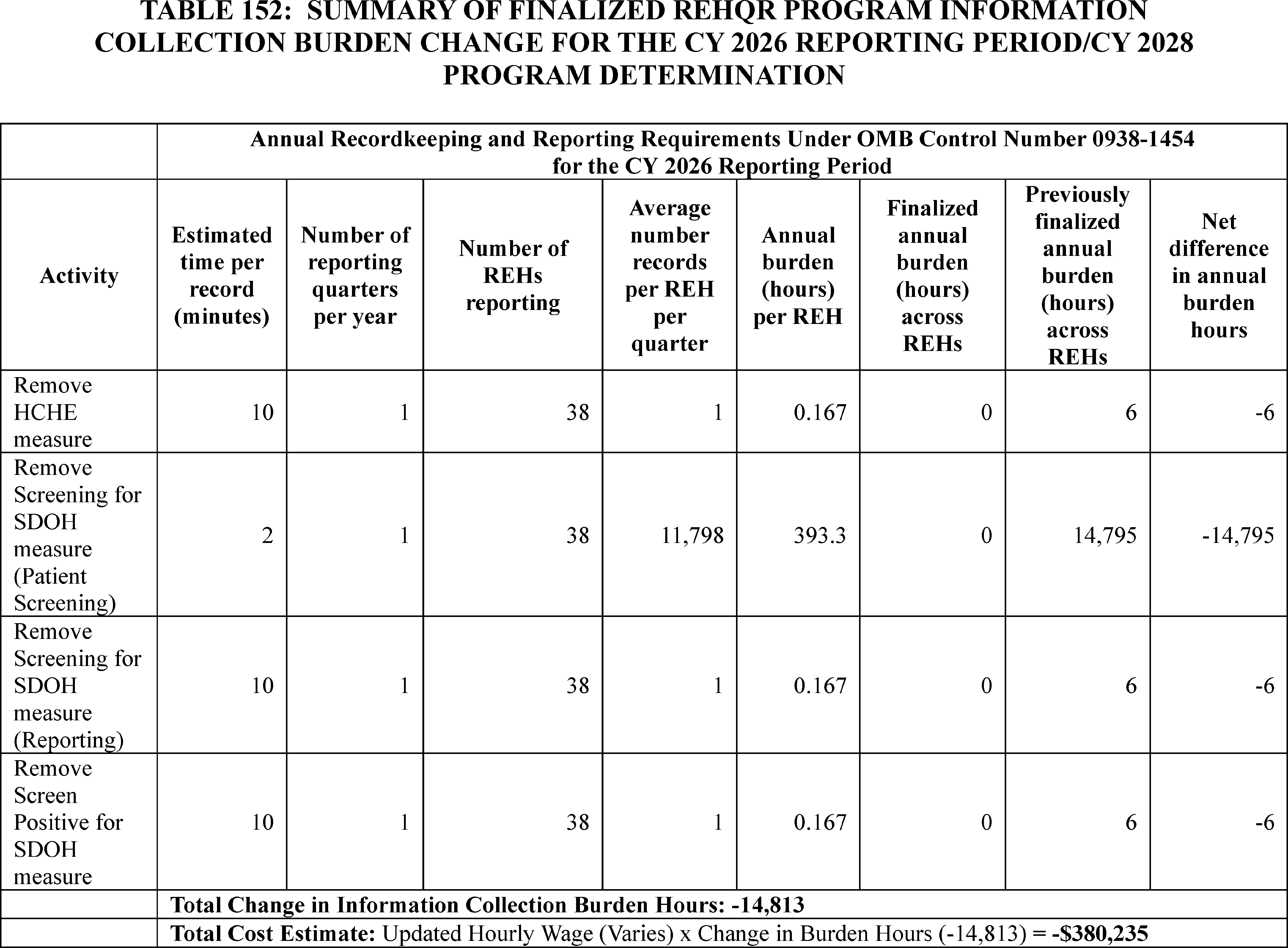
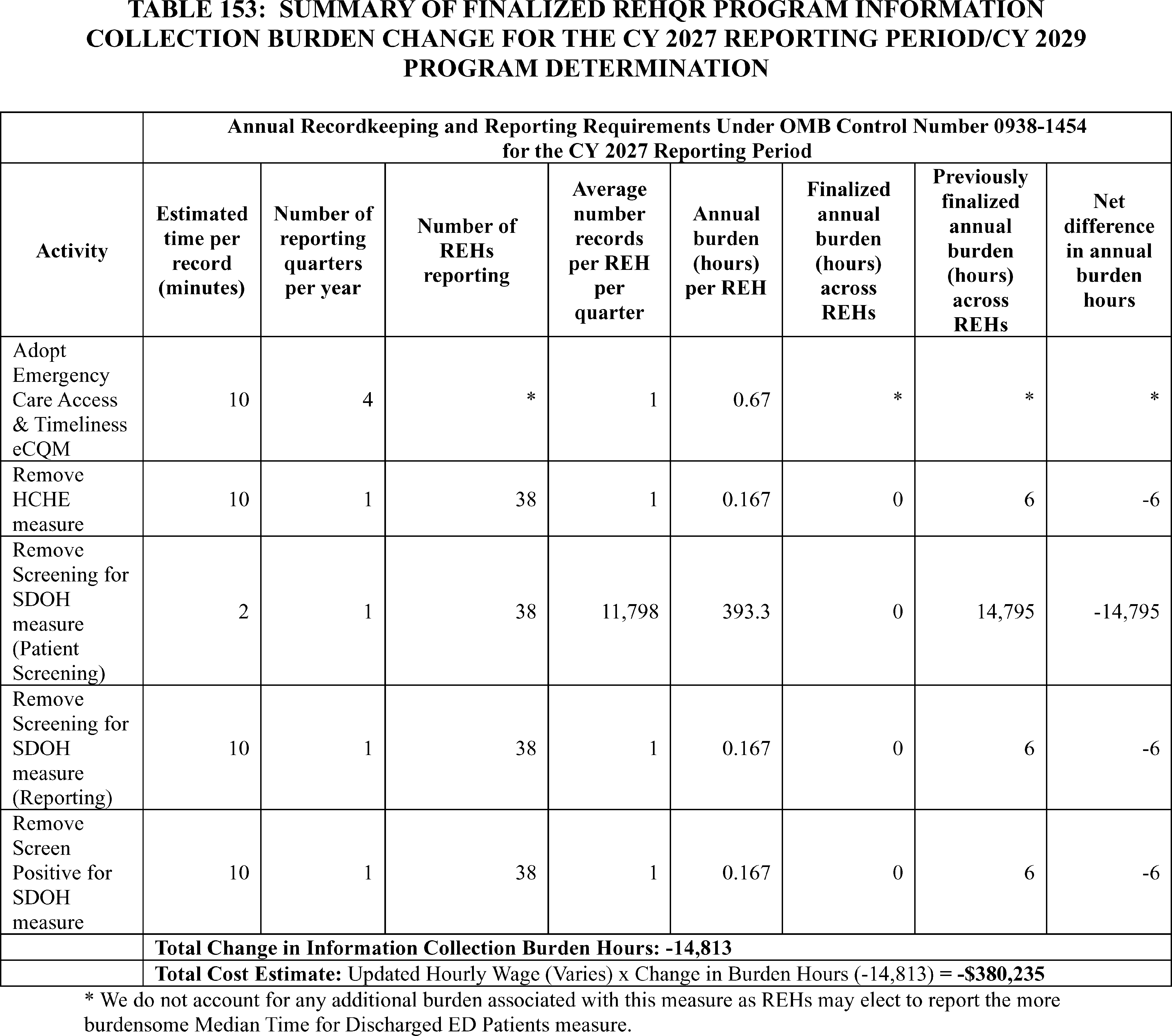



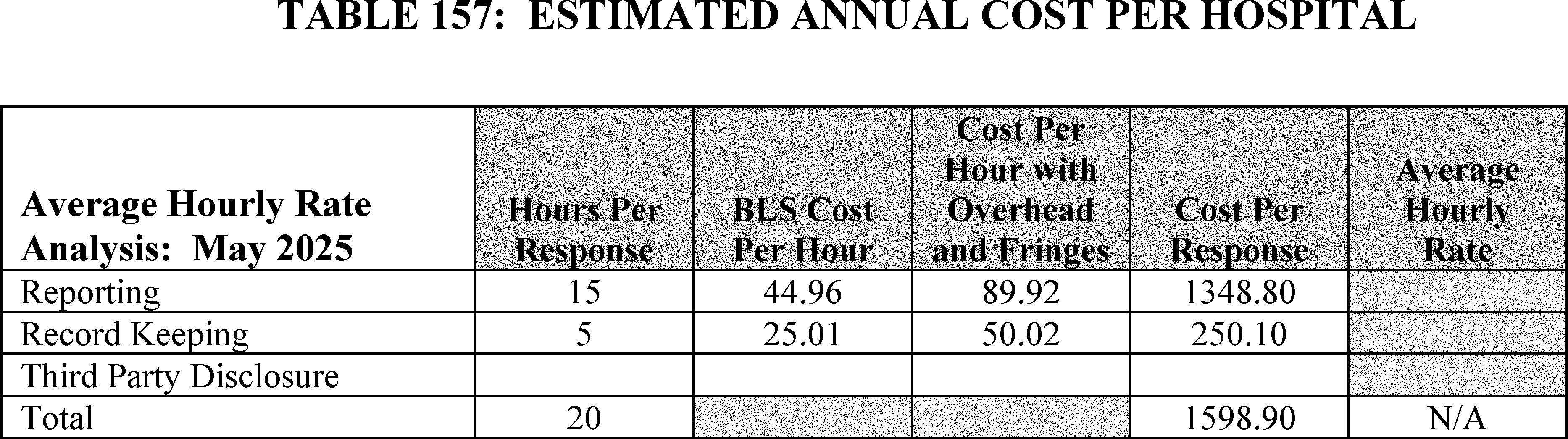


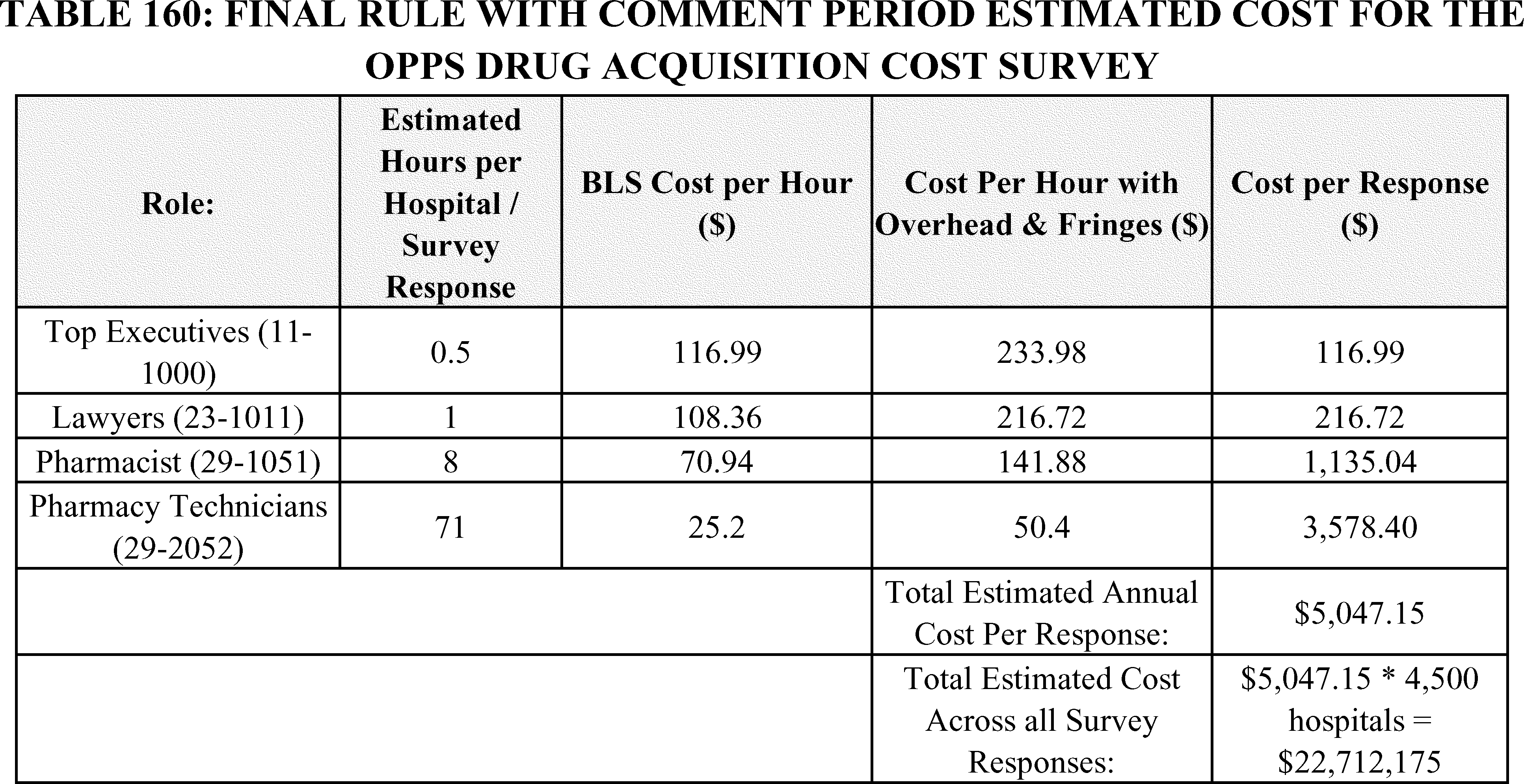
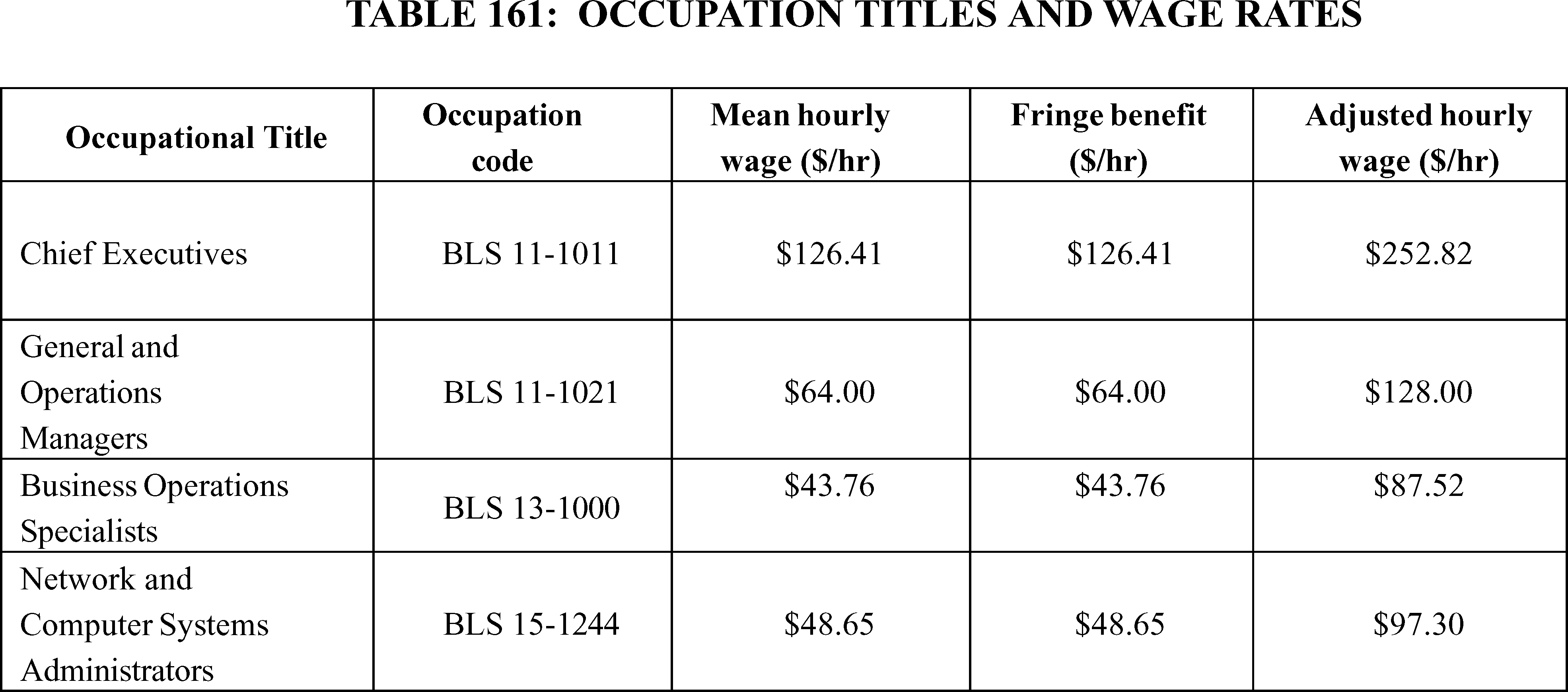




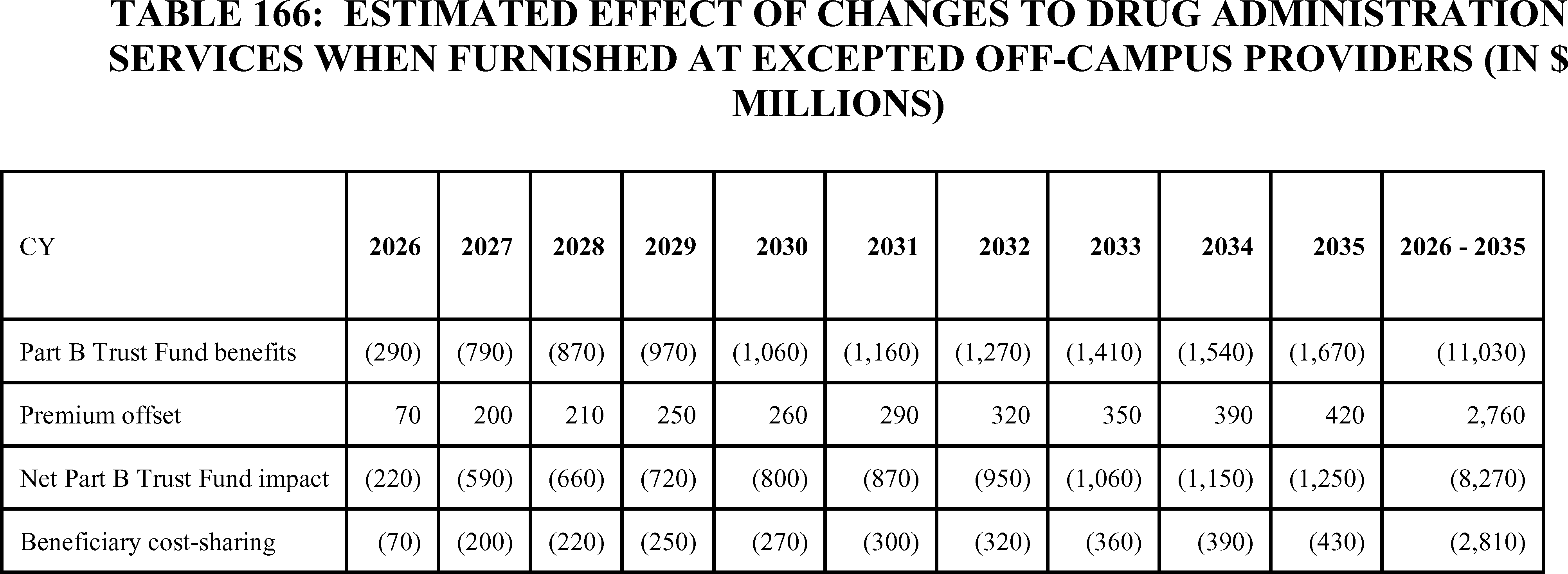
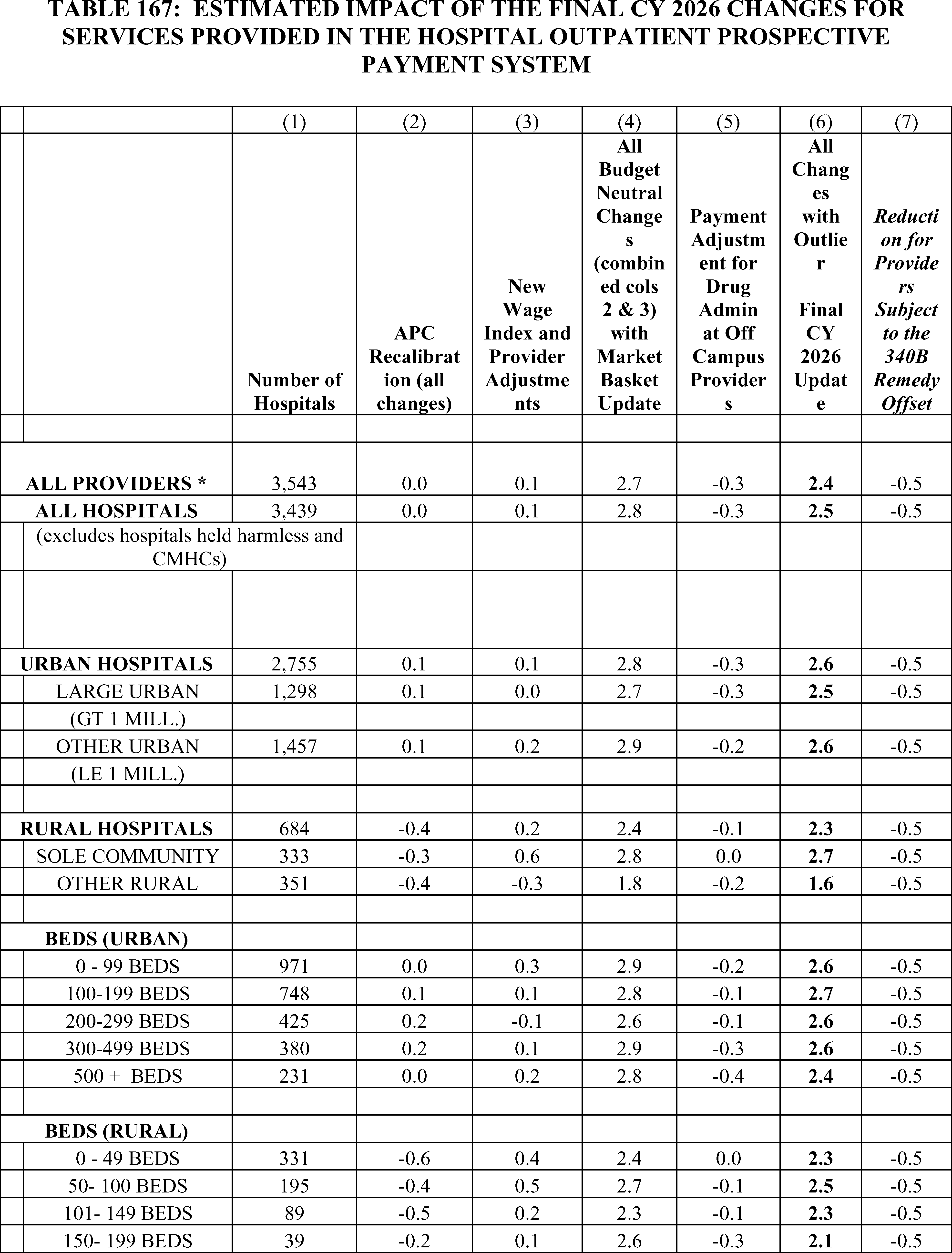
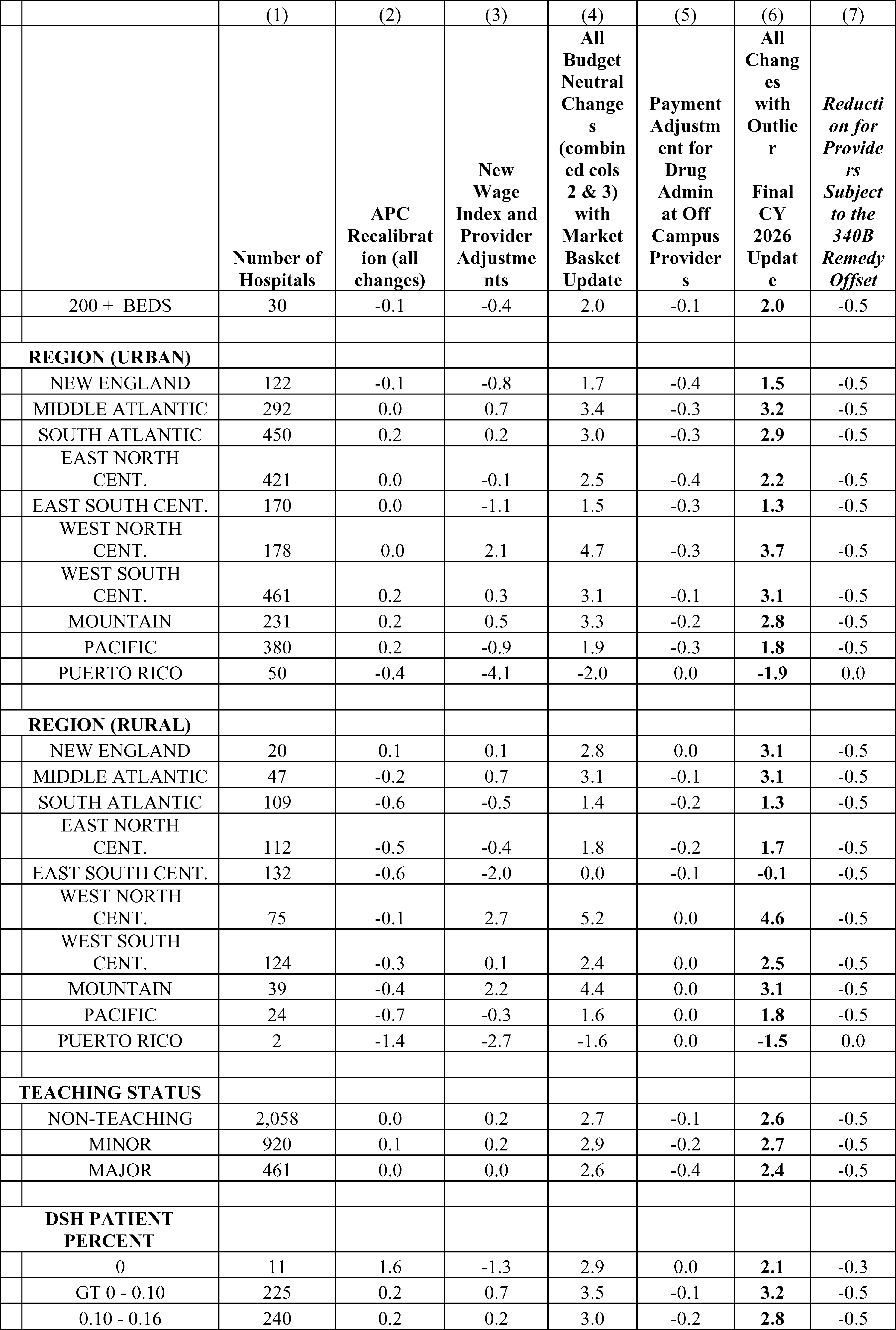
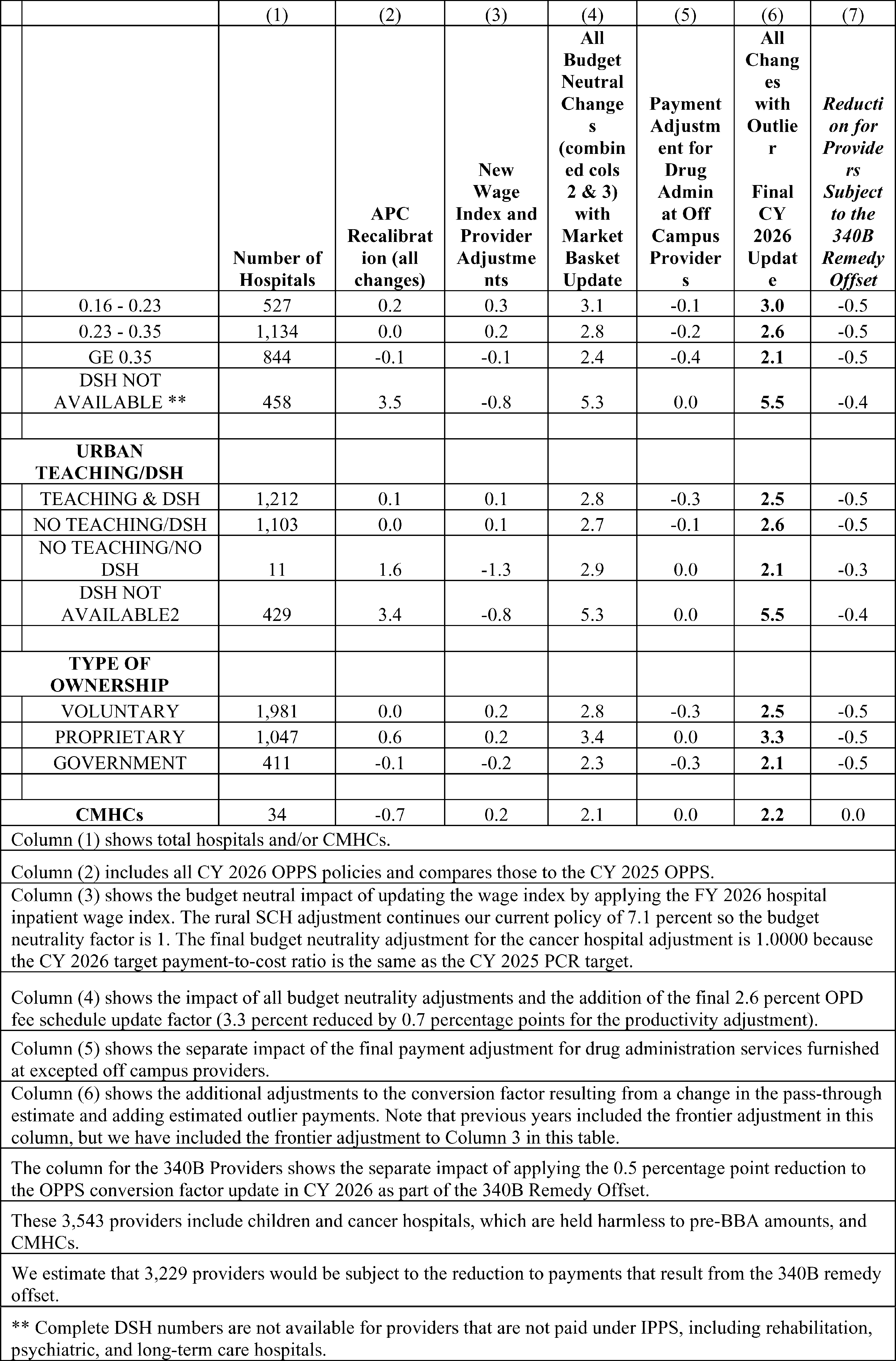
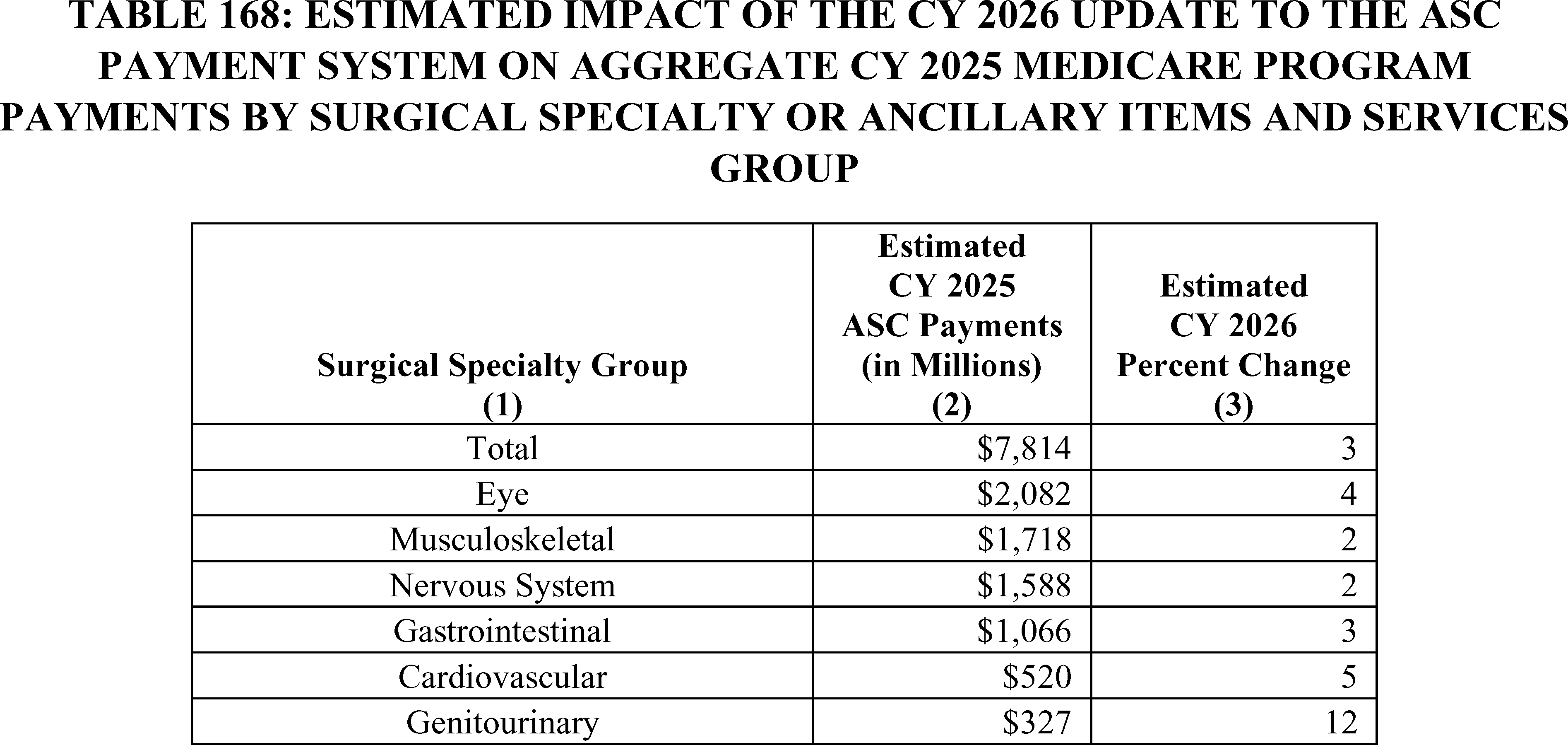
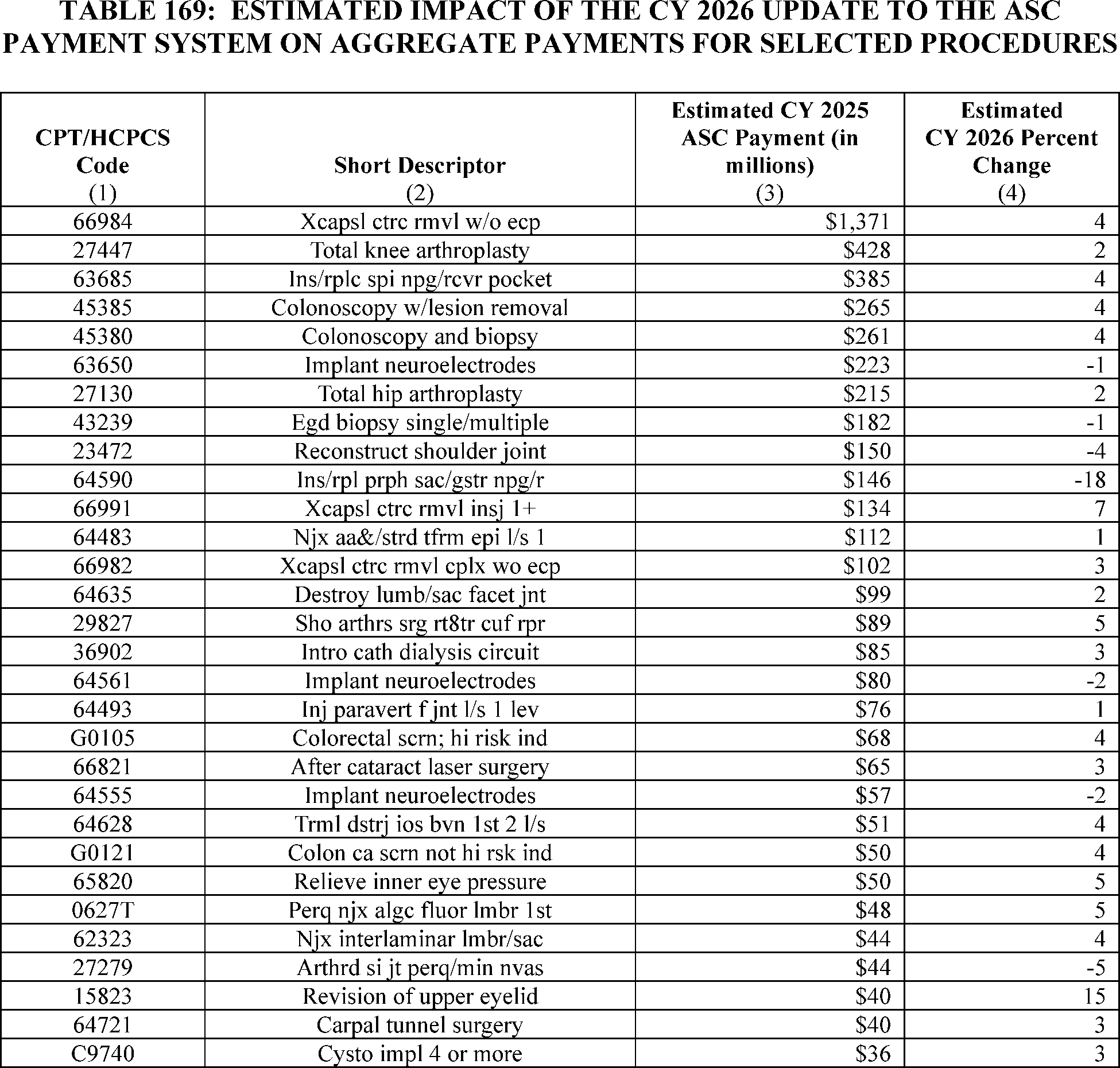



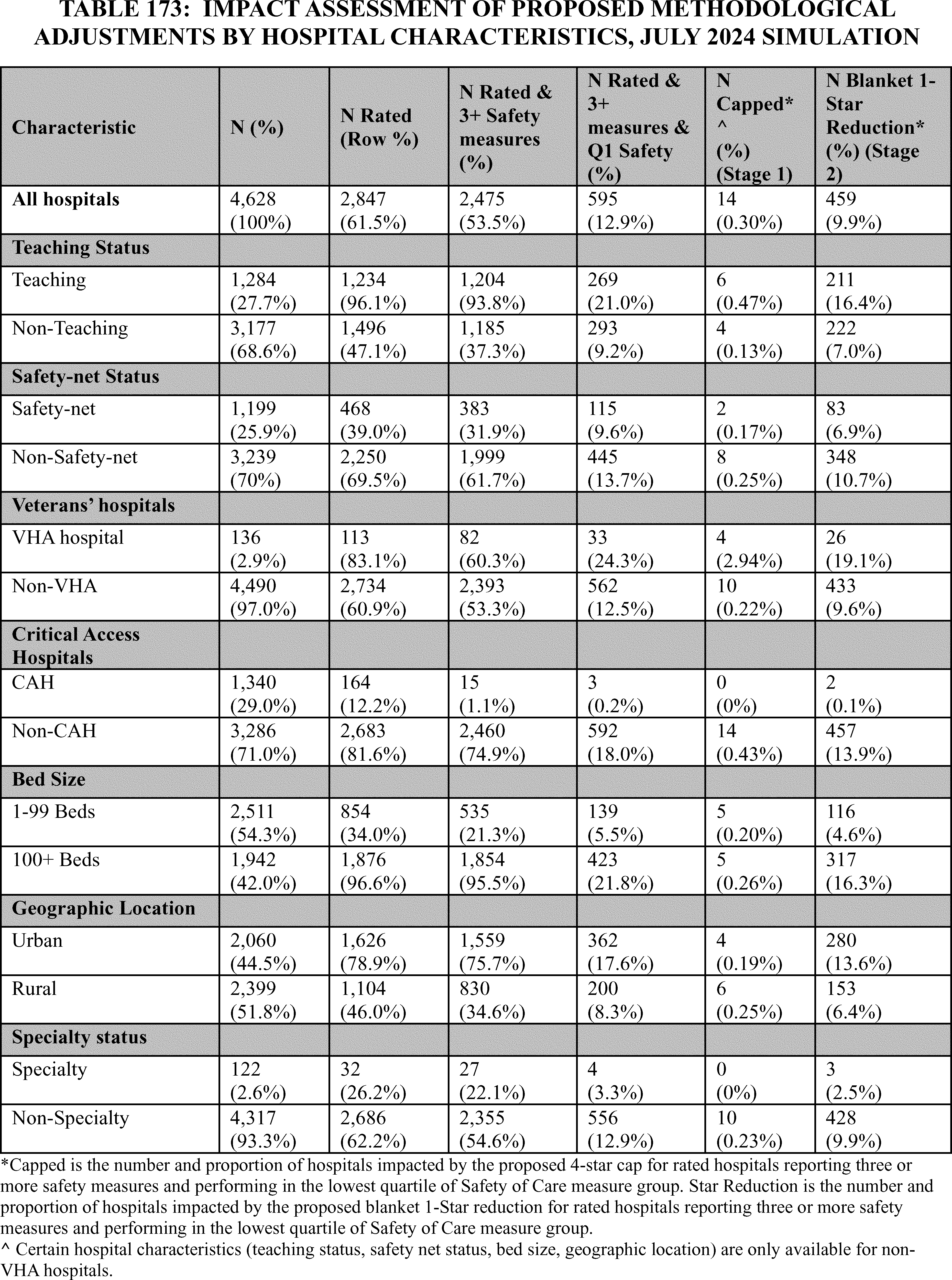
49 comments have been received at Regulations.gov.
Agencies review all submissions and may choose to redact, or withhold, certain submissions (or portions thereof). Submitted comments may not be available to be read until the agency has approved them.